Summary
Background
There is limited information on the effects of statins on the outcomes of liver transplantation (LT), regarding either their use by LT recipients or donors.
Aim
To analyse the association between statin exposure and recipient and graft survival.
Methods
We included adult LT recipients with deceased donors in a nationwide prospective database study. Using a multistate modelling approach, we examined the effect of statins on the transition hazard between LT, biliary and vascular complications and death, allowing for recurring events. The observation time was 3 years.
Results
We included 998 (696 male, 70%, mean age 54.46 ± 11.14 years) LT recipients. 14% of donors and 19% of recipients were exposed to statins during the study period. During follow‐up, 141 patients died; there were 40 re‐LT and 363 complications, with 66 patients having two or more complications. Treatment with statins in the recipient was modelled as a concurrent covariate and associated with lower mortality after LT (HR = 0.35; 95% CI 0.12–0.98; p = 0.047), as well as a significant reduction of re‐LT (p = 0.004). However, it was not associated with lower incidence of complications (HR = 1.25; 95% CI = 0.85–1.83; p = 0.266). Moreover, in patients developing complications, statin use was significantly associated with decreased mortality (HR = 0.10; 95% CI = 0.01–0.81; p = 0.030), and reduced recurrence of complications (HR = 0.43; 95% CI = 0.20–0.93; p = 0.032).
Conclusions
Statin use by LT recipients may confer a survival advantage. Statin administration should be encouraged in LT recipients when clinically indicated.
Keywords: cardiovascular disease, dyslipidemia, solid organ transplantation, survival
Statin use after liver transplantation is associated with improved survival.

1. INTRODUCTION
Liver transplantation (LT) is considered the ultimate curative option for end‐stage liver disease and for non‐resectable hepatocellular carcinoma (HCC). Although survival rates after LT have progressively improved over the years, the first year post‐LT remains the critical period, summing 46% of the total deaths and 67% of re‐LT. 1 Initial outcomes are mainly determined by surgical or peri‐operative problems leading to primary non‐function or delayed graft function and ultimately to re‐LT. In contrast to this, long‐term outcomes are mainly affected by de novo or recurrent malignant tumours and cardiovascular disease. 2
Statins are inhibitors of 3‐hydroxy‐3‐methyl‐glutaryl‐coenzyme A reductase (HMG‐CoA reductase) widely used in the treatment of dyslipidemia and prophylaxis of cardiovascular events. 2 , 3 It has long been recognised that part of the benefits attributed to statins in cardiovascular disease are due to their pleiotropic effects that influence vascular remodelling and reverse endothelial dysfunction, among others. 4 These pleiotropic effects may likely explain the beneficial effects of statins in other conditions, such as sepsis 5 and cancer. 6 , 7 Recent studies reported beneficial effects of statins on chronic liver diseases, both in pre‐clinical models 8 , 9 , 10 and in clinical studies. 11 , 12 , 13 These range from improvement of hepatic sinusoidal endothelial function, leading to a reduction in intrahepatic vascular tone and portal pressure, to a decreased fibrogenesis that may translate into preventing disease progression and facilitating its regression. 14 Statins have also been shown to protect from lipopolysaccharide‐induced acute‐on‐chronic liver failure (ACLF) in cirrhotic rats 15 and to prevent liver function impairment after hypovolemic shock. 16 , 17 Interestingly, in preclinical models statins protect against ischemia/reperfusion injury in young and aged animals 18 and prolong liver graft preservation both in normal and liver grafts with steatosis considered at high‐risk of ischemia/reperfusion injury. 19 Furthermore, epidemiological studies in large cohorts of patients with chronic liver disease suggest a protective effect of statins reducing the rate of progression to cirrhosis, liver decompensation, development of HCC and death. 20 , 21 Classical indications for statins, including the treatment and prevention of cardiovascular diseases, may be relevant for the long‐term outcome after LT. 22 Moreover, the effects of statins protecting from ischemia/reperfusion injury could be beneficial in the early phase after LT, by reducing the incidence and severity of biliary and vascular complications. Therefore, we hypothesize that statin use in LT recipients may favourably influence the transition to adverse outcomes, including re‐LT, severe and recurrent biliary‐vascular complications, and death.
2. METHODS
2.1. Study design
We conducted a cohort study in a nationwide database, including all adult patients who underwent LT from May 2008 to December 2019 registered in the Swiss Transplant Cohort Study (STCS). The STCS is a prospective open cohort study including data from transplanted patients starting in 2008 from the three Swiss liver transplant centres: Bern, Zurich and Geneva. Data from the liver donors were obtained from the Swiss Organ Allocation System (SOAS) records, with permission from Swisstransplant.
The study was conducted in accordance with the Declaration of Helsinki and Good Clinical Practice. All the patients included in the present study had given their informed consent to be included in the STCS. In addition, the Bern Cantonal Ethics Committee (KEK‐ID 2020–02122) approved the present study, as well as the STCS (FUP 149).
2.2. Study population
We included all consecutive adult LT candidates who received a liver from a deceased donor and agreed to participate in the STCS. We collected data regarding liver disease aetiology, severity, comorbidities, use of statins and duration of exposure to these drugs. Regarding donor characteristics, we recorded demographic data, comorbidities, type of deceased donor, cause of death, biochemical characteristics and use of statins.
2.3. Study outcomes and definitions
We studied LT outcome according to statin exposure in the first 3 years after LT. The primary outcome was post‐LT mortality, while re‐LT and/or development of biliary‐vascular complications were secondary outcomes. Yet, we consider cancer and major cardiovascular events as secondary endpoint as well, arising in an ancillary analysis aimed primarily to better control for possible confounding factors. Patients who dropped out were considered alive until last follow‐up and then censored.
Re‐LT was defined as a new LT due to all types of graft loss. Biliary‐vascular complications included biliary leaks, stones and strictures and vascular thrombosis, vascular stenosis and ischemic cholangiopathy. 23 Because not all vascular thrombotic complications necessarily lead to a biliary event, the two types of complications were considered separately. Systemic cardiovascular complications included occurrence of a major cardiovascular event (documented myocardial infarction or stroke), thromboembolic event (pulmonary embolism) and peripheral arterial ischemic disease. Concerning cancers other than HCC, we considered all solid and haematological cancers excluding non‐melanoma skin cancers.
Statin exposure was assessed in both donors and recipients. For donors, statin use was assessed as a dichotomous variable at the time of donation, whereas for recipients, statin use was defined as concurrent use of statins for a given time t during follow‐up. In order to reduce the immortal time bias, exposure was considered as person‐time between prescription and end of follow‐up. 24 LT was considered time 0. The observation time after LT was 3 years.
2.4. Statistical analysis
For descriptive aims, we report the general characteristics of the study cohort across the study period. Quantitative and categorical variables were expressed as mean and standard deviation or median and interquartile range (25%–75%) and percentages, respectively. As supplementary analysis, we divided the recipients into statin non‐user and statin user groups through the study period and compared these using Welch's t‐test for continuous variables and Wilcoxon's rank‐sum test for rank data, respectively. Categorical variables were compared using Chi2, where appropriate.
Because only three patients had missing covariate data, we adopted a complete‐cases analysis for all models. We conducted the statistical analysis by adopting a multistate modelling approach in order to examine the effect of statin exposure (either as recipient currently under statin treatment or receiving a donor liver exposed to statins) on the transition hazards between transplant, biliary‐vascular complications and death, while allowing for recurring events such as re‐transplant and recurrent biliary‐vascular complications (Figure 1). Death was considered a terminal outcome, while re‐LT and biliary‐vascular complications were considered the main surrogate of graft loss and dysfunction, respectively. Therefore, the multistate modelling approach considered three main states of transition for the time‐dependent model: death, re‐LT and biliary‐vascular complications. Additional transitions are possible between each state, including recurrent events. The only exception is death, which represents a sinking state. Follow‐up for every patient started at the time of their first LT. The model was fitted using a Cox‐proportional hazards model (see more data in the Supplementary materials).
FIGURE 1.
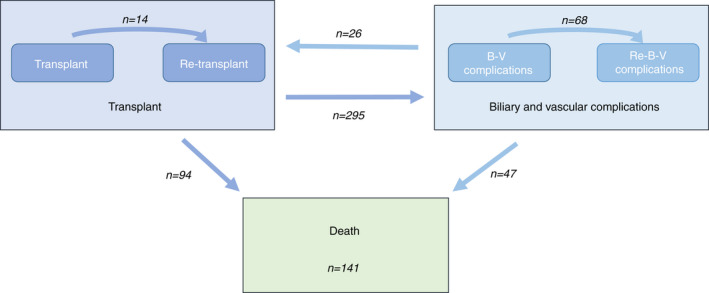
Graphical representation of the possible transitions. The numbers in the arrows indicate the observed transitions. The number of transitions from biliary‐vascular complications to re‐LT and death is given globally, including those coming from first or recurrent biliary‐vascular complications.
Administrative censoring was applied at 3 years because the primary interest of the study was the time immediately following LT. Effects might be different in the long‐term follow‐up since long‐term effects would progressively be more based on patients transplanted at an earlier date, which might therefore not generalise well to current standards.
Furthermore, we did not include HCC or other cancers in the model because of indications that statins could influence the risk of developing HCC 25 , 26 as well as a potential protective role of statins in other cancers. 27 , 28 Including these variables would therefore constitute post‐treatment conditioning and might bias the estimates. However, we additionally explored this possibility using a Cox regression model with HCC‐free survival and non‐HCC cancer‐free survival as outcomes.
All tests were two‐sided. Significance was accepted at p < 0.05. R version 4.1.0 (Development Core Team, 2021) was used for statistical calculations. The package survival (Therneau, T. M. [2020]. A Package for Survival Analysis in R. https://CRAN.R‐project.org/package=survival) was used to estimate the model.
3. RESULTS
3.1. General characteristics of the study population
Overall, 998 LT adult recipients were included in the study, with a total of 1038 grafts transplanted. Male was the prevalent gender (696 patients, 70%) with a mean age of 55 ± 11 years. Hepatocellular carcinoma (HCC) was present in 452 (45%) of the patients. The mean calculated MELD score at LT was 14 [IQR 8–24]. Demographic and clinical information regarding LT and comorbidities are summarised in Table 1. 72 (7%) of LT recipients were on statin therapy at time of LT. The total number of patients exposed to statins increased thereafter up to 19% and the time of statin use for 13.52/100 patient‐days. New cardiovascular complications onset in patients without previous cardiovascular events, new type of cardiovascular events, as well as newly diagnosed T2DM and arterial hypertension after LT occurred in 150 (15%), 257 (27%), 96 (10%) and 155 (16%), respectively, suggesting an underuse of statins in this setting as previously observed (22). Thirty‐five (3.5%) patients had recurrence of HCC, whereas de novo/recurrent extrahepatic cancers occurred in 111 (11.1%).
TABLE 1.
General baseline pre‐transplant characteristics of the LT recipients and recipients characteristics stratified into statin non‐user and statin user groups throughout the study period
| Overall (n = 998) | No exposure to statins (n = 808) | Exposure to statins (n = 190) | p | |
| Recipient sex, male, n (%)** | 696 (70%) | 537 (66%) | 159 (84%) | <0.001 |
| Age, years, mean ± sd** | 54.46 ± 11.14 | 53.32 ± 11.69 | 59.31 ± 6.53 | <0.001 |
| Indication to LT, n (%) | ||||
| Alcohol** | 343 (34%) | 259 (32%) | 84 (44%) | 0.002 |
| HCV | 259 (26%) | 220 (27%) | 39 (21%) | 0.07 |
| HBV | 111 (11%) | 86 (11%) | 25 (13%) | 0.391 |
| PSC/PBC/AIH* | 89 (9%) | 81 (10%) | 8 (4%) | 0.017 |
| NAFLD/NASH* | 99 (10%) | 72 (9%) | 27 (14%) | 0.04 |
| Other* | 133 (13%) | 117 (14%) | 16 (8%) | 0.036 |
| HCC, yes, n (%)** | 474 (48%) | 358 (44%) | 116 (61%) | <0.001 |
| MELD score at LT, median [Q2‐Q4]** | 14 [8–24] | 14 [8–26] | 12.5 [9–21] | 0.008 |
| MELD exception, yes, n (%) | 356 (36%) | 309 (38%) | 47 (25%) | <0.001 |
| BMI, kg/cm2, mean ± sd** | 26.53 ± 5.11 | 26.24 ± 5.17 | 27.78 ± 4.68 | <0.001 |
| BMI, classes, n (%)** | <0.001 | |||
| 18–24.9 | 441 (44%) | 383 (48%) | 58 (31%) | |
| 25–29.9 | 332 (33%) | 253 (31%) | 79 (42%) | |
| >30 | 222 (22%) | 169 (21%) | 53 (28%) | |
| CV disease, yes, n (%)** | 439 (44%) | 329 (41%) | 110 (58%) | <0.001 |
| CAD | 112 (11%) | 58 (7%) | 54 (28%) | <0.001 |
| DM type II, yes, n (%) | 315 (32%) | 252 (31%) | 63 (33%) | 0.668 |
| HTN, yes, n (%) | 411 (41%) | 343 (42%) | 109 (57%) | 0.765 |
| CKD, yes, n (%) | 286 (29%) | 235 (29%) | 51 (27%) | 0.592 |
| History of extrahepatic cancer, yes, n (%) | ||||
| Skin cancer, yes, n (%)* | 27 (3%) | 15 (2%) | 10 (5%) | 0.016 |
| Urogenital cancer, yes, n (%) | 19 (2%) | 18 (2%) | 1 (1%) | 0.149 |
| GI cancer, yes, n (%) | 0 | 0 | 0 | ‐ |
| Lung cancer, yes, n (%) | 2 (0%) | 2 (0%) | 0 | ‐ |
| Breast cancer, yes, n (%) | 10 (1%) | 10 (1%) | 0 | ‐ |
| Haematological cancer, yes, n (%) | 0 | 0 | 0 | ‐ |
| ORL cancer, yes, n (%) | 0 | 0 | 0 | ‐ |
| Neuroendocrine cancer, yes, n (%) | 5 (1%) | 4 (0%) | 1 (1%) | ‐ |
| Others, yes, n (%) | 38 (4%) | 36 (4%) | 6 (3%) | 0.548 |
| History of pre‐OLT Statin use, n (%)** | 65 (7%) | 7 (1%) | 58 (31%) | <0.001 |
| Transplant centre, n (%) | 0.819 | |||
| Centre 1 | 252 (25%) | 201 (25%) | 51 (27%) | |
| Centre 2 | 320 (32%) | 259 (32%) | 61 (32%) | |
| Centre 3 | 427 (43%) | 349 (43%) | 78 (41%) |
Note: Exposure and no exposure to statins relate to the whole follow‐up period of 3 years. Significances determined by chi‐squared or Fisher's exact test for categorical variables, Welch's T‐Test for variables summarised by mean ± sd and the Wilcoxon test where median [IQR] is reported.
Abbreviations: BMI, body mass index; CAD, coronary artery disease; CKD, chronic kidney disease; GI gastrointestinal; HBV, hepatitis B virus; HCC, hepatocellular carcinoma; HCV, hepatitis C virus; HTN, arterial hypertension; LT, liver transplantation; MELD, Model End‐Stage Liver Disease; NAFLD, non‐alcoholic fatty liver disease; NASH, non‐alcoholic steatohepatitis; PBC, primary biliary cholangitis; PSC, primary sclerosing cholangitis; T2DM, diabetes mellitus type 2.
p ≤ 0.05
p ≤ 0.01.
Recipients on statins were older (59 ± 7 vs. 53 ± 12‐year‐old), more frequently male (84% vs. 66%) had higher BMI (BMI 27.8 ± 4.7 vs. 26.2 ± 5.1) and more frequently cardiovascular disease (58% vs. 41%) (all p ≤ 0.001). Additionally, statin recipients were more frequently transplanted for alcohol‐related liver disease (44% vs. 32% in non‐exposed to statins, p = 0.002), non‐alcoholic steatohepatitis (NASH) (14% vs. 9%, p = 0.04) and HCC (61% vs. 44%) (Table 1).
The main characteristics of the study population 3‐year after LT are reported in Table 2.
TABLE 2.
Events observed in LT recipients within 3 years after transplant
| Data | |
|---|---|
| HCC, yes, n (%) | 35 (3.5%) |
| Extrahepatic cancer, yes, n (%) | |
| Skin cancer, yes, n (%) | 54 (5%) |
| Urogenital cancer, yes, n (%) | 4 (<1%) |
| GI cancer, yes, n (%) | 2 (0%) |
| Lung cancer, yes, n (%) | 8 (1%) |
| Breast cancer, yes, n (%) | 2 (0%) |
| Haematological cancer, yes, n (%) | 10 (1%) |
| Oropharyngeal cancer, yes, n (%) | 0 |
| Neuroendocrine cancer, yes, n (%) | 2 (0%) |
| Others, yes, n (%) | 32 (3%) |
| Statin users, n (%) | 190 (19%) |
| Statin use, 100 patient‐days | 13.52 |
| Number of biliary‐vascular complications per patient, n (%) | |
| 0 | 712 (71%) |
| 1 | 221 (22%) |
| 2 | 56 (6%) |
| 3 | 10 (1%) |
| Death, n (%) | 141 (14%) |
| Number of transplants per patient, n (%) | |
| 1 | 961 (96%) |
| 2 | 36 (4%) |
| 3 | 2 (<1%) |
Abbreviations: HCC, hepatocellular carcinoma; GI, gastrointestinal; LT, liver transplantation.
3.2. Donors' general characteristics
A total of 1038 deceased donors were included in the study. Male was the prevalent sex (608, 57%), mean age was 56.18 ± 16.86 years. DCD accounted for 122 (12%) livers, alcohol use was reported in almost half of the donors (482, 46%) and 127 (12%) were obese (Table 3). One hundred forty‐three donors (14%) were on statins at the time of donation. Donors receiving statins were older (68 vs. 54 years, p < 0.001) and had higher BMI (26.9 vs. 25.3, p < 0.001). Donors with T2DM were had 3.39 times more likely to receive statins (p < 0.001).
TABLE 3.
General characteristics of the donor population
| Data (n = 1038) | |
|---|---|
| Donor sex, male, n (%) | 593 (57%) |
| DCD, n (%) | 121 (12%) |
| Age, years, mean (SD) | 56.21 ± 16.87 |
| Causes of death, n (%) | |
| Anoxia | 248 (24) |
| Cerebral disease | 74 (7) |
| Cerebral hemorrhage | 473 (46) |
| Cerebral/spinal trauma | 206 (20%) |
| Other | 18 (2%) |
| Suicide | 18 (2%) |
| BMI, classes, n (%) | |
| 18–24.9 | 554 (53%) |
| 25–29.9 | 370 (35%) |
| >30 | 127 (12%) |
| BMI, kg/m2, mean (±SD) | 25.51 ± 4.20 |
| T2DM, yes, n (%) | 89 (8%) |
| Alcohol use, yes, n (%) | 472 (45%) |
| Cold ischemia time, min, mean (±SD) | 426 ± 117 |
| HBc antibody positive donor, yes, n (%) | 58 (6%) |
| Statin users, n (%) | 142 (14%) |
Note: Donor statin use was only recorded in 592 cases.
Abbreviations: BMI, body mass index; DCD, donor after circulatory death; LT, liver transplantation; T2DM, diabetes mellitus type 2.
3.3. Mortality, re‐transplant and vascular and biliary complication
During the follow‐up, 141 (14%) patients died. Median time to death from first LT was 241 days (IQR, 66–548 days). Causes of death included infections (n = 42, 30%), liver failure (n = 34, 24%), terminal cancer (n = 27, 19%), haemorrhage (n = 15, 11%) and cardiovascular disease (n = 10, 7%). Among patients who died 16 (11%) were exposed to statins. No statistically significant differences were shown for causes of death and statin exposure, however, none of recipients on statins died from cardiovascular event compared to 10 deaths from cardiovascular events in patients who never used statins (Figure 3). There were 40 re‐LT in 38 patients (4%) and 363 biliary‐vascular complications in 287 patients (29%).
FIGURE 3.

Different causes of death among recipients expose or not to statins.
3.4. Multistate model approach
In the multistate model (Table 4), any patient can migrate from one state to another with the only terminal state being death; migrations observed in the present study are summarised in Figure 1. Recipient statin use was associated with lower mortality after LT (HR = 0.35; 95% CI = 0.12–0.99; p = 0.047) (Figure 2). Statin use was also associated with lower hazard of re‐LT (p = 0.004), where the HR could not be reliably estimated because no statin users had re‐LT. Recipient statins' use was not associated with occurrence of biliary‐vascular complications (HR = 1.25; 95% CI = 0.85–1.83; p = 0.266). Regarding the transition from B‐V complications to other states, statin use was significantly associated with reduced likelihood of death (HR = 0.10; 95% CI = 0.01–0.81; p = 0.031), and of recurrent biliary‐vascular complications (HR = 0.43; 95% CI = 0.20–0.93; p = 0.033) (Figure 2). There was no significant effect of statin use on the likelihood of re‐LT after biliary‐vascular complications (HR = 1.12; 95% CI = 0.32–3.90, p = 0.858).
TABLE 4.
Multistate model considering survival in the different transition according to Cox regression analysis
| LT to re‐LT | B‐V complication to re‐LT | |||||
|---|---|---|---|---|---|---|
| HR | 95% CI (HR) | p‐value | HR | 95% CI (HR) | p‐value | |
| Statin use recipients, yes | 0.004 | 1.12 | 0.32–3.9 | 0.858 | ||
| Statins use donors, yes | 2.42 | 0.66–8.96 | 0.185 | 0.87 | 0.31–2.47 | 0.794 |
| Age recipient, 10 years | 1.21 | 0.73–2.01 | 0.460 | 0.80 | 0.58–1.09 | 0.151 |
| Age donor, 10 years | 1.00 | 0.92–1.09 | 0.933 | 0.81 | 0.64–1.02 | 0.072 |
| Donor type, DCD | 2.44 | 0.56–10.7 | 0.236 | 1.19 | 0.31–4.57 | 0.798 |
| T2DM recipient, yes | 1.39 | 0.5–3.87 | 0.527 | 1.12 | 0.43–2.95 | 0.813 |
| BMI donor, (kg/m2), 5 points | 1.24 | 0.41–3.7 | 0.706 | 1.59 | 0.67–3.76 | 0.292 |
| BMI recipient, (kg/m2), 5 points | 0.99 | 0.97–1 | 0.004 | 0.99 | 0.99–1 | 0.001 |
| Cardiovascular event | 1.39 | 0.5–3.87 | 0.527 | 1.12 | 0.43–2.95 | 0.813 |
| Date of LT, years since first patient included (linear) | 0.042 | <0.001 | ||||
| Date of LT, years since first patient included (nonlinear) | 0.022 | 0.028 | ||||
| Centre (gaussian frailty) | 0.879 | 0.003 | ||||
| LT to B‐V complication | B‐V complication to Re‐B‐V complication | |||||
| HR | 95% CI (HR) | p‐value | HR | 95% CI (HR) | p‐value | |
| Statin use recipients, yes | 1.25 | 0.85–1.83 | 0.266 | 0.43 | 0.2–0.93 | 0.032 |
| Statins use donors, yes | 0.95 | 0.68–1.34 | 0.778 | 0.47 | 0.22–1.02 | 0.056 |
| Age recipient, 10 years | 0.95 | 0.85–1.05 | 0.318 | 1.06 | 0.88–1.28 | 0.536 |
| Age donor, 10 years | 1.08 | 1–1.17 | 0.038 | 1.18 | 0.99–1.41 | 0.061 |
| Donor type, DCD | 1.06 | 0.72–1.55 | 0.776 | 1.23 | 0.69–2.21 | 0.484 |
| T2DM recipient, yes | 1.43 | 1.1–1.84 | 0.007 | 0.81 | 0.38–1.7 | 0.572 |
| BMI donor (kg/m2), 5 points | 0.80 | 0.61–1.03 | 0.083 | 1.34 | 0.83–2.18 | 0.234 |
| BMI recipient (kg/m2), 5 points | 1.00 | 1–1 | 0.594 | 1.00 | 1–1.01 | 0.526 |
| Cardiovascular event | 1.43 | 1.1–1.84 | 0.007 | 0.81 | 0.38–1.7 | 0.572 |
| Date of LT, years since first patient included (linear) | 0.172 | 0.456 | ||||
| Date of LT, years since first patient included (nonlinear) | <0.001 | 0.013 | ||||
| Centre (gaussian frailty) | 0.027 | 0.998 | ||||
| LT to death | B‐V complication to death | |||||
| HR | 95% CI (HR) | p‐value | HR | 95% CI (HR) | p‐value | |
| Statin use recipients, yes | 0.35 | 0.12–0.98 | 0.047 | 0.10 | 0.01–0.81 | 0.030 |
| Statins use donors, yes | 1.27 | 0.73–2.21 | 0.393 | 0.95 | 0.42–2.15 | 0.898 |
| Age recipient, 10 years | 1.33 | 1.06–1.67 | 0.016 | 1.60 | 1.11–2.3 | 0.012 |
| Age donor, 10 years | 1.00 | 0.97–1.02 | 0.830 | 1.06 | 0.88–1.28 | 0.519 |
| Donor type, DCD | 1.20 | 0.64–2.25 | 0.574 | 1.32 | 0.48–3.65 | 0.591 |
| T2DM recipient, yes | 1.73 | 1.04–2.88 | 0.035 | 3.23 | 1.74–5.97 | <0.001 |
| BMI donor, (kg/m2), 5 points | 0.99 | 0.64–1.54 | 0.979 | 1.14 | 0.57–2.27 | 0.711 |
| BMI recipient, (kg/m2), 5 points | 1.00 | 0.99–1 | 0.283 | 0.99 | 0.99–1 | 0.001 |
| Cardiovascular event | 1.73 | 1.04–2.88 | 0.035 | 3.23 | 1.74–5.97 | <0.001 |
| Date of LT, years since first patient included (linear) | 0.484 | <0.001 | ||||
| Date of LT, years since first patient included (nonlinear) | 0.014 | 0.005 | ||||
| Centre (gaussian frailty) | 0.029 | 0.433 | ||||
Abbreviations: BMI, body mass index; B‐V, biliary‐vascular; DCD, donor after circulatory death; LT, liver transplantation; T2DM, diabetes mellitus type 2.
FIGURE 2.

Cumulative transition hazards with significant effects of recipient statin use: Transplant to transplant; transplant to death; biliary‐vascular complication to biliary‐vascular complication; and biliary‐vascular complication to death.
Considering donor statin use, there was no effect on any of the six transitions. Donors' statin use showed a trend for a similar association to occurrence of biliary‐vascular complications as recipient statin use: HR = 0.47; 95% CI = 0.22–1.02, but it did not reach statistical significance (p = 0.056).
3.5. HCC recurrence and de novo/recurrence of cancer other than HCC
In order to better understand the impact of statins on survival, we built Cox models considering HCC recurrence and de novo/recurrence of non‐liver cancer, with death as a competing risk. While there was no association between HCC recurrence and statin use (Table S1), statin use was associated with a significant reduction of the risk of developing cancers other than HCC (HR = 0.48; 95% CI = 0.29–0.80; p = 0.005; Table S2).
4. DISCUSSION
The present study aimed at evaluating the effect of statin exposure of both recipients and donors on LT outcomes. Specifically, we wanted to investigate whether statin exposure could influence adverse outcomes, including death, re‐LT, severe biliary‐vascular complications, and recurrent biliary‐vascular complications.
The most significant result of our study is that statin use by LT recipients is associated with improved survival, both in patients with and without biliary‐vascular complications.
The study design was set up bearing in mind the main limitations affecting epidemiological studies, particularly in the LT setting. Therefore, we constructed a multistate model for the main analysis of the study, taking into account that each subject can transition to other state(s) in the course of the observation period and that this can happen several times (Figure 1). This approach ensures greater internal consistency than conventional approaches 22 and weights the accumulated risk for each transition. Statin use in the recipients was associated with a lower risk of transition from LT to death, and from biliary‐vascular complications to death. Recipients with concurrent statin use were never subject to re‐LT without prior biliary‐vascular complication in our cohort, which equates a significantly lower log‐likelihood (p = 0.004). In addition, recipient exposure to statins was associated with lower hazards of recurrent biliary‐vascular complications. This finding is particularly relevant considering that patients experiencing a first biliary‐vascular complication are more likely to develop a second one.
To our knowledge, this study is the first specifically designed to examine the pleiotropic protective effects of statins in LT. Recently, a study from the NailNASH Consortium analysed the outcome of 938 patients receiving LT for NASH cirrhosis. They concluded that statin use after LT favourably impacted survival (HR = 0.38; 95% CI = 0.19–0.75; p = 0.005), which is in accordance with our study. However, the protective effect of statins was not the main aim of the study and the results relate to a population known for higher risk of cardiovascular disease and likelihood of statin use compared to other LT indications. 25 A previous study by Patel et al. 22 examined the impact of coronary artery disease (CAD) and dyslipidemia on clinical outcomes after LT, and reported that in this context, statin exposure was associated with improved survival. Our findings in the present cohort are in line with this previous data, since among patients who died after LT being exposed to statins, none died from cardiovascular diseases. Additionally, they showed a considerable underuse of statins in LT recipients, even when clinically indicated, without observing side effects that could justify this underuse. 22 Statin underuse is also observed in the general population: only around 25% of subjects with an indication for statins are actually on statin therapy. Globally, up to 12.6% of annual cardiovascular deaths could be avoided if all patients eligible were on statins. 29 This could be estimated also in our cohort, where approximately 19% of recipients were exposed to statins, while circa 30% would have an indication for statins based on comorbidities. Under‐prescription of statins might partly reflect that attending physicians feel uncomfortable taking therapeutic decisions in LT recipients. On the other hand, the known interaction between cyclosporine and statins with potential increase in statins bioavailability may have led to the use of low potency hydrophilic statins (not metabolised through the cytochrome P‐450 3A4) or under‐use and under‐dosing of statins. 30 However, most LT recipients at present are under immunosuppressive regimens based on tacrolimus, whose interaction with statins is almost negligible. 30 There is thus no contraindication to the use of statins post‐LT, although slowly increasing the dose and monitoring immunosuppression trough levels is advisable.
Even if post‐LT cancer outcomes were not the primary objective of our study, 26 , 27 our findings confirmed a strong association between statin exposure and reduced incidence of development/recurrence of extrahepatic tumours. However, in contrast with other recent studies, it failed to demonstrate a significant association with decreased HCC recurrence. 26 , 31 This might be partly due to the short post‐LT observation period (3 years) not being able to capture all HCC recurrences, 32 as well as the coding of statins use as a concurrent covariate, which might not be appropriate for long‐term outcomes.
On the other hand, even within a limited number of events, we observed a trend for a protective effect of donor statin exposure against post‐LT biliary‐vascular complications, analogous to that observed for recipient statin exposure. From a mechanistic point of view, this data is in line with preclinical data in experimental models of ischemia/reperfusion injury and cold liver preservation, 16 , 17 and with the preliminary results of a recent randomised controlled trial. 33 Interestingly, the biologic effect of statins on vascular function and liver grafts is quite similar to that exerted by pulsatile flow perfusion 17 , 34 which successfully minimises biliary‐vascular complications after LT. 35
This study has some limitations, the main one being the applicability of the results of cohort studies to clinical practice, which usually require confirmation in ad hoc prospective studies. To minimise this limitation, we adopted a statistical strategy that is more process‐oriented than conventional regression analysis and that provides higher generalizability to real‐life scenarios. The total number of patients exposed to statins increased from 7% at the time of LT to 19% thereafter. It appears possible that clinicians were more confident to prescribe statins after LT rather to stable patients than to patients suffering from surgical complications or relevant liver dysfunction. However, we minimised selection bias inherent to cohort studies by including all adult patients receiving LT in the period of this nationwide study. Another potential limitation is that the short number of events early after liver transplantation and of donor statin users may have precluded observing and association between statin exposure and biliary/vascular complications very early after liver transplantation that perhaps would have been shown in a larger study. Unfortunately, fully modelling the time‐varying nature of the different effects of donor and recipient statin exposure would have required a significantly larger number of events than were available in our study.
In summary, our results suggest that the use of statins in LT recipients confers a survival advantage. Our data also confirm that statins are underused in LT recipients qualifying for statins, and further add to the body of evidence in favour of using statins in this population when there is a clinical indication. Considering that statins are cheap, safe and widely used and that current strategies for reducing graft loss and improving survival after LT are costly and still limited, our data suggest that statins may represent a new effective approach with relevant clinical impact in the post‐LT setting.
AUTHOR CONTRIBUTIONS
Chiara Becchetti: Conceptualization (equal); data curation (equal); investigation (equal); methodology (equal); writing – original draft (lead); writing – review and editing (equal). Melisa Dirchwolf: Conceptualization (equal); data curation (equal); investigation (equal); methodology (equal); writing – original draft (equal); writing – review and editing (equal). Jonas Schropp: Data curation (equal); formal analysis (equal); validation (equal). Giulia Magini: Data curation (equal); writing – review and editing (equal). Beat Mullhaupt: Data curation (equal); writing – review and editing (equal). Immer Franz: Data curation (equal); writing – review and editing (equal). Jean‐Francois Dufour: Data curation (equal); writing – review and editing (equal). Vanessa Banz: Funding acquisition (equal); writing – review and editing (equal). Annalisa Berzigotti: Conceptualization (equal); supervision (equal); writing – review and editing (equal). Jaime Bosch: Conceptualization (equal); funding acquisition (equal); methodology (equal); project administration (equal); supervision (equal); writing – review and editing (equal).
FUNDING INFORMATION
C.B. and J.B. were supported by the Stiftung für Leberkrankheiten Bern. The study was supported by the Swiss Transplant Cohort Study (FUP 149).
CONFLICT OF INTEREST
None.
Supporting information
Data S1 Supporting Information.
ACKNOWLEDGEMENTS
The Swiss Transplant Cohort Study is supported by the Swiss National Science Foundation (http://www.snf.ch), Unimedsuisse (https://www.unimedsuisse.ch) and the Transplant Centres. We would like to acknowledge Simona Rossi and Susanne Stampf who helped in the process of retrieving data from STCS. Open access funding provided by Inselspital Universitatsspital Bern. Open access funding provided by Inselspital Universitatsspital Bern.
APPENDIX A.
The members of the Swiss Transplant Cohort Study are: Patrizia Amico, Andres Axel, John‐David Aubert, Vanessa Banz, Beckmann Sonja, Guido Beldi, Christoph Berger, Ekaterine Berishvili, Isabelle Binet, Pierre‐Yves Bochud, Sanda Branca, Heiner Bucher, Thierry Carrel, Emmanuelle Catana, Yves Chalandon, Sabina De Geest, Olivier De Rougemont, Sophie De Seigneux, Michael Dickenmann, Joëlle Lynn Dreifuss, Michel Duchosal, Thomas Fehr, Sylvie Ferrari‐Lacraz, Nicola Franscini, Christian Garzoni, Paola Gasche Soccal, Christophe Gaudet, Déla Golshayan, Nicolas Goossens, Karine Hadaya, Jörg Halter, Dominik Heim, Christoph Hess, Sven Hillinger, Hans Hirsch, Patricia Hirt, Günther Hofbauer, Uyen Huynh‐Do, Franz Immer, Michael Koller(Head of the data centre), Mirjam Laager, Bettina Laesser, Roger Lehmann, Alexander Leichtle, Christian Lovis, Oriol Manuel, Hans‐Peter Marti, Pierre Yves Martin, Michele Martinelli, Valérie McLin, Katell Mellac, Aurélia Merçay, Karin Mettler, Nicolas Mueller (Chairman Scientific Committee), Antonia Müller, Ulrike Müller‐Arndt, Beat Müllhaupt, Mirjam Nägeli, Graziano Oldani, Manuel Pascual (Executive office), Klara Posfay‐Barbe, Juliane Rick, Anne Rosselet, Simona Rossi, Silvia Rothlin, Frank Ruschitzka, Thomas Schachtner, Urs Schanz, Stefan Schaub, Aurelia Schnyder, Macé Schuurmans, Thierry Sengstag, Federico Simonetta, Susanne Stampf, Jürg Steiger (Head, Excecutive office), Guido Stirniman, Ueli Stürzinger, Christian Van Delden (Executive office), Jean‐Pierre Venetz, Jean Villard, Julien Vionnet, Madeleine Wick (STCS coordinator), Markus Wilhlem, Patrick Yerly
Becchetti C, Dirchwolf M, Schropp J, Magini G, Müllhaupt B & Immer F, et al. Use of statins after liver transplantation is associated with improved survival: results of a nationwide study. Aliment Pharmacol Ther. 2022;56:1194–1204. 10.1111/apt.17192
Chiara Becchetti and Melisa Dirchwolf have contributed equally.
The Handling Editor for this article was Dr Colin Howden, and it was accepted for publication after full peer‐review.
Contributor Information
Jaume Bosch, Email: jaime.bosch@dbmr.unibe.ch.
Swiss Transplant Cohort Study:
Patrizia Amico, Andres Axel, John‐David Aubert, Vanessa Banz, Beckmann Sonja, Guido Beldi, Christoph Berger, Ekaterine Berishvili, Isabelle Binet, Pierre‐Yves Bochud, Sanda Branca, Heiner Bucher, Thierry Carrel, Emmanuelle Catana, Yves Chalandon, Sabina De Geest, Olivier De Rougemont, Sophie De Seigneux, Michael Dickenmann, Joëlle Lynn Dreifuss, Michel Duchosal, Thomas Fehr, Sylvie Ferrari‐Lacraz, Nicola Franscini, Christian Garzoni, Paola Gasche Soccal, Christophe Gaudet, Déla Golshayan, Nicolas Goossens, Karine Hadaya, Jörg Halter, Dominik Heim, Christoph Hess, Sven Hillinger, Hans Hirsch, Patricia Hirt, Günther Hofbauer, Uyen Huynh‐Do, Franz Immer, Michael Koller, Mirjam Laager, Bettina Laesser, Roger Lehmann, Alexander Leichtle, Christian Lovis, Oriol Manuel, Hans‐Peter Marti, Pierre Yves Martin, Michele Martinelli, Valérie McLin, Katell Mellac, Aurélia Merçay, Karin Mettler, Nicolas Mueller, Antonia Müller, Ulrike Müller‐Arndt, Beat Müllhaupt, Mirjam Nägeli, Graziano Oldani, Manuel Pascual, Klara Posfay‐Barbe, Juliane Rick, Anne Rosselet, Simona Rossi, Silvia Rothlin, Frank Ruschitzka, Thomas Schachtner, Urs Schanz, Stefan Schaub, Aurelia Schnyder, Macé Schuurmans, Thierry Sengstag, Federico Simonetta, Susanne Stampf, Jürg Steiger, Guido Stirniman, Ueli Stürzinger, Christian Van Delden, Jean‐Pierre Venetz, Jean Villard, Julien Vionnet, Madeleine Wick, Markus Wilhlem, and Patrick Yerly
DATA AVAILABILITY STATEMENT
The data that support the findings of this study are available from the corresponding author upon reasonable request." cd_value_code="text
REFERENCES
- 1. Adam R, Karam V, Cailliez V, O Grady JG, Mirza D, Cherqui D, et al. 2018 annual report of the European liver transplant registry (ELTR) ‐ 50‐year evolution of liver transplantation. Transpl Int. 2018;31:1293–317. [DOI] [PubMed] [Google Scholar]
- 2. Watt KDS, Pedersen RA, Kremers WK, Heimbach JK, Charlton MR. Evolution of causes and risk factors for mortality post‐liver transplant: results of the NIDDK long‐term follow‐up study. Am J Transplant. 2010;10:1420–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Bhopal RS. Epidemic of cardiovascular disease and diabetes: explaining the phenomenon in south Asians worldwide. Oxford, UK: Oxford University Press; 2019. [Google Scholar]
- 4. Stancu C, Sima A. Statins: mechanism of action and effects. J Cell Mol Med. 2001;5:378–87. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Patel JM, Snaith C, Thickett DR, Linhartova L, Melody T, Hawkey P, et al. Randomized double‐blind placebo‐controlled trial of 40 mg/day of atorvastatin in reducing the severity of sepsis in ward patients (ASEPSIS trial). Crit Care. 2012;16:R231. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Hong JY, Nam EM, Lee J, Park JO, Lee SC, Song SY, et al. Randomized double‐blinded, placebo‐controlled phase II trial of simvastatin and gemcitabine in advanced pancreatic cancer patients. Cancer Chemother Pharmacol. 2014;73:125–30. [DOI] [PubMed] [Google Scholar]
- 7. Konings IRHM, van der Gaast A, van der Wijk LJ, de Jongh FE, Eskens FALM, Sleijfer S. The addition of pravastatin to chemotherapy in advanced gastric carcinoma: a randomised phase II trial. Eur J Cancer. 2010;46:3200–4. [DOI] [PubMed] [Google Scholar]
- 8. Abraldes JG, Rodríguez‐Vilarrupla A, Graupera M, Zafra C, García‐Calderó H, García‐Pagán JC, et al. Simvastatin treatment improves liver sinusoidal endothelial dysfunction in CCl4 cirrhotic rats. J Hepatol. 2007;46:1040–6. [DOI] [PubMed] [Google Scholar]
- 9. Marrone G, Maeso‐Díaz R, García‐Cardena G, Abraldes JG, García‐Pagán JC, Bosch J, et al. KLF2 exerts antifibrotic and vasoprotective effects in cirrhotic rat livers: behind the molecular mechanisms of statins. Gut. 2015;64:1434–43. [DOI] [PubMed] [Google Scholar]
- 10. Trebicka J, Hennenberg M, Odenthal M, Shir K, Klein S, Granzow M, et al. Atorvastatin attenuates hepatic fibrosis in rats after bile duct ligation via decreased turnover of hepatic stellate cells. J Hepatol. 2010;53:702–12. [DOI] [PubMed] [Google Scholar]
- 11. Abraldes JG, Albillos A, Bañares R, Turnes J, González R, García–Pagán JC, et al. Simvastatin lowers portal pressure in patients with cirrhosis and portal hypertension: a randomized controlled trial. Gastroenterology. 2009;136:1651–8. [DOI] [PubMed] [Google Scholar]
- 12. Abraldes JG, Villanueva C, Aracil C, Turnes J, Hernandez‐Guerra M, Genesca J, et al. Addition of simvastatin to standard therapy for the prevention of variceal rebleeding does not reduce rebleeding but increases survival in patients with cirrhosis. Gastroenterology. 2016;150:1160–1170.e3. [DOI] [PubMed] [Google Scholar]
- 13. Zafra C, Abraldes JG, Turnes J, Berzigotti A, Fernández M, García‐Pagán JC, et al. Simvastatin enhances hepatic nitric oxide production and decreases the hepatic vascular tone in patients with cirrhosis. Gastroenterology. 2004;126:749–55. [DOI] [PubMed] [Google Scholar]
- 14. Gracia‐Sancho J, Russo L, García‐Calderó H, et al. Endothelial expression of transcription factor Kruppel‐like factor 2 and its vasoprotective target genes in the normal and cirrhotic rat liver. Gut. 2011;60:517–24. [DOI] [PubMed] [Google Scholar]
- 15. Tripathi DM, Vilaseca M, Lafoz E, Garcia‐Calderó H, Viegas Haute G, Fernández‐Iglesias A, et al. Simvastatin prevents progression of acute on chronic liver failure in rats with cirrhosis and portal hypertension. Gastroenterology. 2018;155:1564–77. [DOI] [PubMed] [Google Scholar]
- 16. Meireles CZ, Pasarin M, Lozano JJ, García‐Calderó H, Gracia‐Sancho J, García‐Pagán JC, et al. Simvastatin attenuates liver injury in rodents with biliary cirrhosis submitted to hemorrhage/resuscitation. Shock. 2017;47:370–7. [DOI] [PubMed] [Google Scholar]
- 17. Russo L, Gracia‐Sancho J, García‐Calderó H, Marrone G, García‐Pagán JC, García‐Cardeña G, et al. Addition of simvastatin to cold storage solution prevents endothelial dysfunction in explanted rat livers. Hepatology. 2012;55:921–30. [DOI] [PubMed] [Google Scholar]
- 18. Hide D, Warren A, Fernández‐Iglesias A, et al. Ischemia/reperfusion injury in the aged liver: the importance of the sinusoidal endothelium in developing therapeutic strategies for the elderly. J Gerontol Series A Biol Sci Med Sci. 2020;75:268–277. [DOI] [PubMed] [Google Scholar]
- 19. Gracia‐Sancho J, García‐Calderó H, Hide D, Marrone G, Guixé‐Muntet S, Peralta C, et al. Simvastatin maintains function and viability of steatotic rat livers procured for transplantation. J Hepatol. 2013;58:1140–6. [DOI] [PubMed] [Google Scholar]
- 20. Kim RG, Loomba R, Prokop LJ, Singh S. Statin use and risk of cirrhosis and related complications in patients with chronic liver diseases: a systematic review and meta‐analysis. Clin Gastroenterol Hepatol. 2017;15:1521–1530.e8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Simon TG, Bonilla H, Yan P, Chung RT, Butt AA. Atorvastatin and fluvastatin are associated with dose‐dependent reductions in cirrhosis and hepatocellular carcinoma, among patients with hepatitis C virus: results from ERCHIVES. Hepatology. 2016;64:47–57. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Patel SS, Rodriguez VA, Siddiqui MB, et al. The impact of coronary artery disease and statins on survival after liver transplantation. Liver Transplant. 2019;25:1514–23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Vivalda S, Zhengbin H, Xiong Y, Liu Z, Wang Z, Ye Q. Vascular and biliary complications following deceased donor liver transplantation: a meta‐analysis. Transplant Proc. 2019;51:823–32. [DOI] [PubMed] [Google Scholar]
- 24. Levesque LE, Hanley JA, Kezouh A, Suissa S. Problem of immortal time bias in cohort studies: example using statins for preventing progression of diabetes. BMJ. 2010;340:b5087–7. [DOI] [PubMed] [Google Scholar]
- 25. Rinella ME, Satapathy SK, Brandman D, Smith C, Elwir S, Xia J, et al. Factors impacting survival in those transplanted for NASH cirrhosis: data from the NailNASH consortium. Clin Gastroenterol Hepatol. 2022. 10.1016/j.cgh.2022.02.028. Online ahead of print. [DOI] [PubMed] [Google Scholar]
- 26. Cho Y, Kim MS, Nam CM, Kang ES. Statin use is associated with decreased hepatocellular carcinoma recurrence in liver transplant patients. Sci Rep. 2019;9:1467. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Demierre M‐F, Higgins PDR, Gruber SB, Hawk E, Lippman SM. Statins and cancer prevention. Nat Rev Cancer. 2005;5:930–42. [DOI] [PubMed] [Google Scholar]
- 28. Ahmadi M, Amiri S, Pecic S, Machaj F, Rosik J, Łos MJ, et al. Pleiotropic effects of statins: a focus on cancer. Biochim Biophys Acta Mol Basis Dis. 2020;1866:165968. [DOI] [PubMed] [Google Scholar]
- 29. Yang Q, Zhong Y, Gillespie C, Merritt R, Bowman B, George MG, et al. Assessing potential population impact of statin treatment for primary prevention of atherosclerotic cardiovascular diseases in the USA: population‐based modelling study. BMJ Open. 2017;7:e011684. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Skalicka B, Kubanek M, Malek I, Vymetalova Y, Hoskova L, Podzimkova M, et al. Conversion to tacrolimus and atorvastatin in cyclosporine‐treated heart transplant recipients with dyslipidemia refractory to fluvastatin. J Heart Lung Transplant. 2009;28:598–604. [DOI] [PubMed] [Google Scholar]
- 31. Lee HL, Lee SW, Jang JW, Bae SH, Choi JY, Yoon SK, et al. Anticancer effect of statins in patients undergoing liver transplantation for hepatocellular carcinoma. Liver Transpl. 2022;28:397–406. [DOI] [PubMed] [Google Scholar]
- 32. Sapisochin G, Bruix J. Liver transplantation for hepatocellular carcinoma: outcomes and novel surgical approaches. Nat Rev Gastroenterol Hepatol. 2017;14:203–17. [DOI] [PubMed] [Google Scholar]
- 33. Pagano D, Bosch J, Tuzzolino F, Oliva E, Ekser B, Zito G, et al. Donor simvastatin treatment is safe and might improve outcomes after liver transplantation: a randomized clinical trial. Transplantation. 2022. 10.1097/TP.0000000000004220. Online ahead of print. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Tullius SG, García‐Cardeña G. Organ procurement and perfusion before transplantation. N Engl J Med. 2009;360:78–80. [DOI] [PubMed] [Google Scholar]
- 35. van Rijn R, Schurink IJ, de Vries Y, van den Berg AP, Cortes Cerisuelo M, Darwish Murad S, et al. A randomized controlled trial of dual hypothermic oxygenated machine perfusion in donation after circulatory death liver transplantation. HPB. 2021;23:S673. [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data S1 Supporting Information.
Data Availability Statement
The data that support the findings of this study are available from the corresponding author upon reasonable request." cd_value_code="text


