Abstract
We sought to determine whether racial and socioeconomic disparities in the utilization of deep brain stimulation (DBS) for Parkinson's disease (PD) have improved over time. We examined DBS utilization and analyzed factors associated with placement of DBS. The odds of DBS placement increased across the study period, whereas White patients with PD were 5 times more likely than Black patients to undergo DBS. Individuals, regardless of racial background, with 2 or more comorbidities were 14 times less likely to undergo DBS. Privately insured patients were 1.6 times more likely to undergo DBS. Despite increasing DBS utilization, significant disparities persist in access to DBS. ANN NEUROL 2022;92:246–254
Surgical treatment of Parkinson's disease (PD) with deep brain stimulation (DBS) has demonstrated efficacy that complements pharmacotherapies in managing the chronic motor symptoms of PD while improving the patients' quality of life and ability to perform daily activities. Multiple randomized clinical trials demonstrate superiority of DBS compared to medical management alone in selected patients with PD. 1 Therefore, evidence supports the efficacy and safety of DBS for PD.
Despite wide acceptance as an efficacious therapy for PD, significant disparities in DBS access have been identified, particularly for Black patients. 2 , 3 In the 10 years after US Food and Drug Administration (FDA) approval of DBS, Black patients with PD were 5 to 8 times less likely to receive DBS than White patients. 2 Since that time, much effort has been put into reducing health disparities. 4 In this study, we examined the National Inpatient Sample (NIS) from 2002 to 2018 to determine whether disparities in DBS access have improved in the last decade.
Methods
Sampling
We examined NIS data from the Healthcare Cost and Utilization Project (HCUP) and the Agency for Healthcare Research and Quality. 5 The database was queried from 2002 to 2018 using the International Disease Classification Ninth Revision (ICD‐9) and ICD Tenth Revision (ICD‐10) diagnostic and procedural codes (Fig 1A). We identified hospitalized patients with PD ≥18 years of age and the subset of patients admitted for DBS while excluding individuals with other neurodegenerative diagnoses.
FIGURE 1.
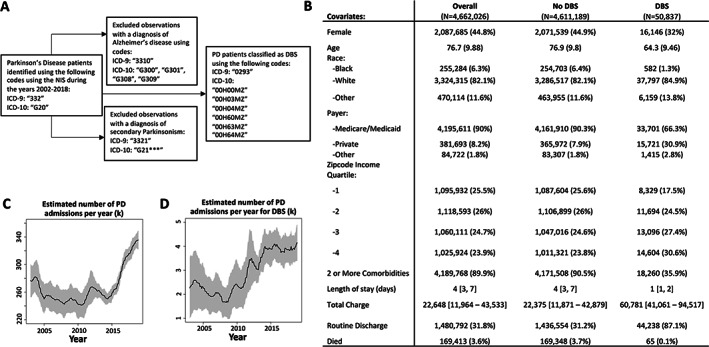
ICD codes used to define PD cohort and DBS cases (A). National estimates of patient characteristics summed or averaged across 2002 through 2018 (B). Patient characteristics are summarized by estimated mean (sample SD), median [IQR], or estimated N (estimated %) for continuous normal, continuous skewed, and categorical variables, respectively. Note that White patients predominated among both patients with PD without (82.1%) and with DBS (84.9%). Black patients composed 6.4% of PD admissions not undergoing DBS and 1.3% of those admitted for DBS placement. National estimates of PD admissions (all PD admission, including for DBS) over time (C) and national estimates of DBS procedures over time (D) in thousands. DBS = deep brain stimulation; ICD‐9 = International Disease Classification Ninth Revision; ICD‐10 = International Disease Classification Tenth Revision; IQR = interquartile range; PD = Parkinson's disease.
Patient Classification
The Elixhauser comorbidity software, supplied by HCUP, was used to identify comorbidities among our patient sample. 6 Patients were dichotomized based on the number of comorbidities using a cutoff of ≥2 based on an accepted threshold for defining multimorbidity, 7 re‐admission, and surgical complications. Patients were classified as follows: PD without DBS versus PD with DBS, and number of comorbidities (<2 vs ≥2 comorbidities). Patient demographics included sex, age, race, payer, and income quartile classification of patients' zip code derived from Claritas. Based on the NIS database structure, racial categories were defined as White, Black, and other.
Statistical Methods
All analyses were performed under the survey framework using sampling weights, clusters, and strata provided by the NIS database. Plots of procedures over time were constructed by estimating 12‐month running averages with a 95% confidence band. A multivariable survey logistic model assessed the odds of a PD admission undergoing DBS with and without an interaction term for year and race. A post hoc chi‐squared test examined the association between insurance type and year stratified by race. Analyses were performed using SAS software version 9.4 or R version 3.5.1 using the “survey” package. 6 Estimates of admission and surgical volume based on primary insurance type are displayed per 100,000 of the United States population with that respective insurance type, whereas those in the “other” category are shown per 100,000 uninsured individuals using data from the US census bureau. 8 , 9
Results
Characteristics of the Study Cohort and Time Trend Analysis of DBS Utilization
Figure 1B highlights characteristics of the overall cohort of patients with PD, individuals not undergoing DBS, and those hospitalized for DBS placement. Figure 1C and D illustrate the estimated number of hospitalizations for PD and patients undergoing DBS, respectively. Estimated DBS utilization increased by 82%, from approximately 2,200 DBS surgeries in 2002 to approximately 4,000 in 2018. Hospital admission of patients with PD increased from 285,000 in 2002 to 336,000 in 2018. To quantitatively assess the apparent increase in DBS after 2010, we divided the study period into 4 epochs. After adjusting for ≥2 comorbidities, age, sex, insurance, and income, stratified by year and race, the odds of patients with PD receiving DBS were comparable between the 2002 to 2005 and 2006 to 2009 period (odds ratio [OR] = 0.83, confidence interval [CI] = 0.59–1.18). However, patients with PD were more likely to receive DBS during the time periods 2010 to 2013 (OR = 1.45, CI = 1.07–1.97) and 2014 to 2018 (OR = 1.71, CI = 1.25–2.34) relative to 2002 to 2005.
Time Trends in DBS Placement as a Function of Insurer Type and Factors Associated with DBS
Figure 2A–C depict patients undergoing DBS between 2002 and 2018 as a function of primary insurer type. Although increased DBS utilization is observed for patients insured by Medicare/Medicaid (see Fig 2A), private insurer (see Fig 2B), and other insurers (see Fig 2C), growth in DBS is most evident in Medicare/Medicaid patients, as these patients account for the bulk of patients undergoing DBS.
FIGURE 2.
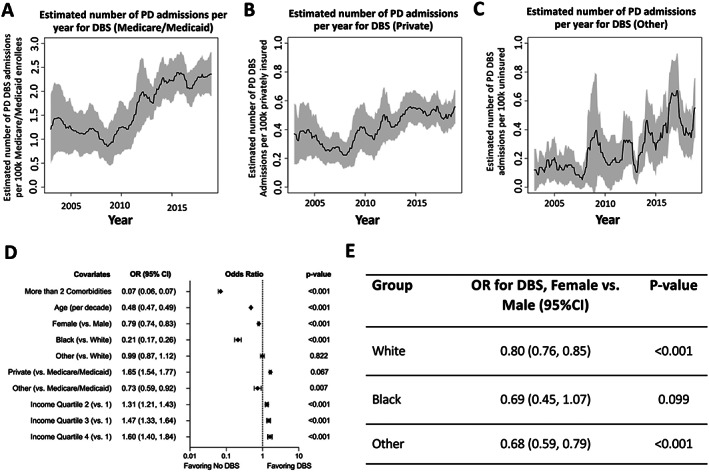
Changes in primary insurance for Medicare/Medicaid (A), private insurance (B), and other (C) over time in patients undergoing DBS. For A to C, data are displayed per 100,000 of the overall US population with that respective insurance type while those in the “other” category are shown per 100,000 uninsured individuals over the study period. Note that quantitatively, these time trends are characterized by slopes per year of 2.39, 0.08, and 0.77 for Medicare/Medicaid, private insurance, and other insurers, respectively. Results to a multi‐variable survey logistic regression model assessing the odds of DBS (D) adjusted for the year. Note that “other” types of insurance include patients that self‐pay; those that incur no hospital charges; or those that have their hospital admission paid via Worker's Compensation, Title V, or other government programs. Effect of sex on DBS by race after adjusting for year, 2 or more comorbidities, age, payer, and zip code income quartile (E). DBS = deep brain stimulation; CI = confidence interval; OR = odds ratio; PD = Parkinson's disease.
Private insurance was associated with increased odds of DBS compared with patients insured by Medicare/Medicaid (OR = 1.64, CI = 1.53–1.76). Patients primarily insured in the “other” category were less likely to undergo DBS (OR = 0.70, CI = 0.56–0.88). Black patients with PD were less likely to undergo DBS than White patients with PD (OR = 0.21, CI = 0.17–0.27). “Other” racial groups had similar odds of undergoing DBS compared with White patients (OR = 0.99, CI = 0.87–1.12). We found that women were significantly less likely to undergo DBS than men (OR = 0.79, CI = 0.75–0.83). Older age was associated with decreased likelihood of undergoing DBS (OR = 0.48 per decade, CI = 0.47–0.49). Finally, patients with ≥2 comorbidities were less likely to undergo DBS (OR = 0.07, CI = 0.06–0.07). Each subsequent income quartile was associated with increasing odds of undergoing DBS compared to the lowest quartile (all p < 0.001; Fig 2D).
To further evaluate the observed sex disparity in the likelihood of receiving DBS between female and male patients with PD, we performed a multivariate analysis across sex based on racial background. White female patients with PD were less likely than White male patients with PD to receive DBS (OR = 0.80, CI = 0.76–0.85). Similarly, female patients with PD from other racial backgrounds also demonstrated a reduced likelihood of undergoing DBS compared with male patients with PD from other racial backgrounds (OR = 0.68, CI = 0.59–0.79). We observed a similar trend for Black female patients with PD compared with Black male patients with PD (OR = 0.69, CI = 0.45–1.07), although this was not significant.
Given the large observed influence of comorbidities on the likelihood of receiving DBS, we evaluated the percentage of the overall cohort of patients with PD with ≥2 comorbidities, stratified by race (Fig 3A). We found that among patients with PD, 89.9% of White patients, 94.1% of Black patients, and 90.4% of other patients had ≥2 comorbidities. We then assessed the effect of racial background on the likelihood of receiving DBS based on comorbidities and found that among Black patients with ≥2 comorbidities, the odds of receiving DBS were 0.19 (CI = 0.14–0.25) compared with White patients with ≥2 comorbidities (Fig 3B). The odds of Black patients with <2 comorbidities receiving DBS was 0.23 (CI = 0.17–0.30) compared with White patients with <2 comorbidities. When comparing the odds of Black patients with PD receiving DBS compared with White patients with PD (regardless of the number of comorbidities among the racial groups) we found no significant difference in the interaction (p = 0.268), suggesting that comorbidities do not underlie the racial disparity between the likelihood of Black PD patients receiving DBS compared with White patients with PD. We also compared the odds of patients with PD from other racial backgrounds to White patients with PD and found no statistical differences between those with ≥2 comorbidities (OR = 0.96, CI = 0.82–1.11) and those with <2 comorbidities (OR = 1.01, CI = 0.88–1.16). Finally, among patients with PD from other racial groups (regardless of the number of comorbidities), we found no significant difference in the interaction (p = 0.495).
FIGURE 3.
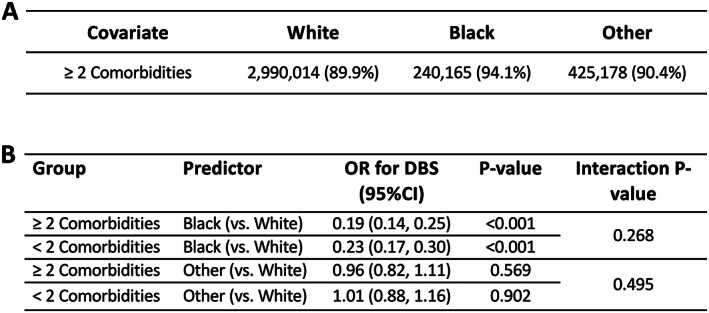
Descriptive statistics showing comorbidity by race, N (%), (A). Assessment of the effect of race on likelihood of DBS by comorbidities after adjusting for year, sex, age, payer, and zip code income quartile (B). DBS = deep brain stimulation; OR = odds ratio.
Disparity in DBS Utilization Rate over Time
Prior work has shown Black patients with PD in the United States are 5 to 8‐fold less likely to undergo DBS relative to White patients. 2 , 3 Correction of this disparity would require that the likelihood of DBS utilization in Black patients outpace that of White patients by 5 to 8 to 1. We tested whether this occurred by comparing the odds of DBS placement in Black and White patients pre‐ and post‐2010 (Fig 4A). The odds of undergoing DBS during the period 2010 to 2018 versus the period 2002 to 2009 for Black patients with PD was 2.24 (CI = 1.20–4.19). For the White patients, the corresponding OR was 1.76 (CI = 1.38–2.24). A direct comparison of Black and White patients undergoing DBS in 2002 to 2009 demonstrates that the odds of Black patients receiving DBS was 0.17 compared with White patients after adjusting for confounders (OR = 0.17, CI = 0.10–0.29, p < 0.001). In 2010 to 2018, the odds of Black patients receiving DBS was 0.21 compared with White patients (OR = 0.22, CI = 0.17–0.28, p < 0.001; Fig 4B). Therefore, more Black and White patients have received DBS since 2010, but the racial disparity in DBS has not meaningfully changed. There was no significant change in insurance type between racial groups (Fig 4C).
FIGURE 4.
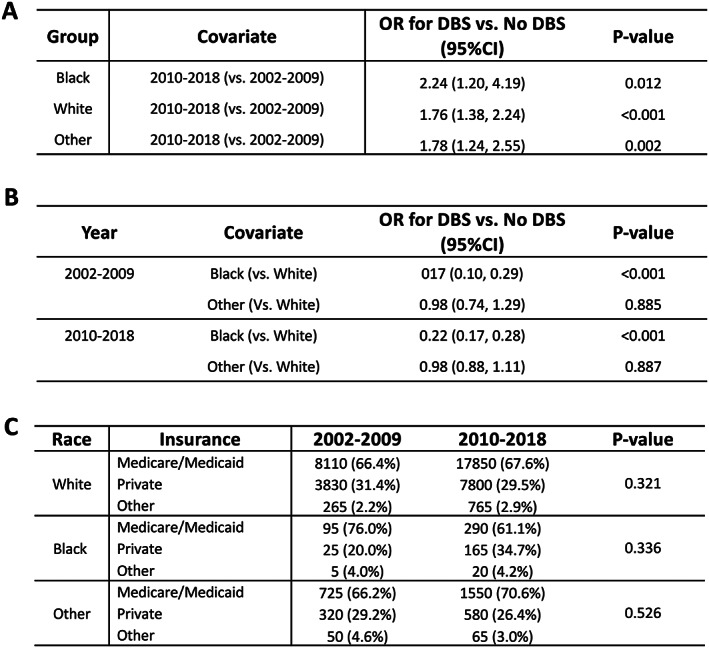
Results of a multivariable survey logistic regression model assess the association between race and DBS over time for Black patients, White patients, and other (A). Direct comparison of odds of undergoing DBS based on race across years (B), note that the model adjusted for 2 or more Elixhauser comorbidities, age, sex, insurance type, and zip code income quartile. Post hoc survey chi‐squared analysis to test if insurance type for those who received DBS differs across the study period by race (C). DBS = deep brain stimulation; CI = confidence interval; OR = odds ratio.
Discussion
As the US population ages, the prevalence of PD has been increasing, 10 as reflected in the overall increase in hospital admissions of these patients in the last 2 decades (see Fig 1C). Accordingly, and perhaps in concert with increased acceptance of DBS over time, there has been significant growth in DBS in patients with PD during the same period (see Fig 1D). Growth in DBS, however, has not meaningfully changed the racial disparity in DBS utilization over the last decade. After controlling for factors associated with DBS placement, including age, comorbidities, insurance type, and income quartile, Black patients were 5 times less likely to undergo DBS than White patients in the last decade, almost identical to the ratio seen in the prior decade (see Fig 4B). Analysis of the likelihood of receiving DBS across racial groups using a finer timescale (3‐year increments across the study period) did not demonstrate a significant change across the study period (data not shown), suggesting that the racial disparities have not narrowed with time. The findings demonstrate persistent disparities in the clinical practice of DBS that warrant consideration for improvement.
As an elective surgery aimed at improving the quality of life in patients with PD, surgeons and DBS teams must minimize the most common complications of DBS surgery (hemorrhage and infection) in order to justify its benefits. The selection process of DBS candidates is one major aspect of this risk management and it is therefore not surprising that patients with PD with ≥2 comorbidities are less likely to undergo surgery. We and others have previously demonstrated that selection criteria for DBS for patients appeared to be getting more liberal over time with surgical patients getting increasingly older and more likely to have comorbidities. 11 We considered that differences in the number of comorbidities may affect the likelihood of receiving DBS and that these differences may be different between Black and White patients with PD. We tested this hypothesis by comparing the likelihood of Black patients receiving DBS among those with <2 comorbidities and those with ≥2 comorbidities and found that Black patients from both groups were approximately 5 times less likely than White patients to receive DBS (Fig 3). Furthermore, there was no significant difference in the interaction among the comparison between these 2 groups, which suggests that the number of comorbidities does not contribute to the observed racial disparity. Although previous studies have demonstrated racial differences in the likelihood of comorbidities, such as obesity and diabetes mellitus, 12 this does not appear to be a significant factor in the racial disparity between Black and White patients with PD who receive DBS. One limitation of this analysis is that it only addresses the quantity of comorbidities and not necessarily the severity or quality of comorbidities. Nevertheless, the presence of 2 or more comorbidities remains the strongest predictor of not undergoing DBS and demonstrates that patients with PD still undergo a rigorous selection process.
Differences in surgical outcomes across racial groups may also contribute to the racial disparity in DBS utilization. For example, if White patients tend to have better outcomes than Black patients or patients from other racial groups, this may bias the recommendation for DBS therapy for the group with better outcomes. Unfortunately, there is limited literature to draw upon to address this question as it pertains to possible racial differences in postoperative outcomes following DBS surgery. One recent study found that White and non‐White patients with PD were similarly likely to be routinely discharged following DBS surgery. 13 However, there is evidence that postoperative outcomes (including length of stay and complications) are worse for Black patients compared with White patients following a range of major surgeries. 14 , 15 , 16 , 17 , 18 Black patients also have higher postoperative mortality rates. 19 , 20 , 21 Therefore, to what extent surgeon/institutional experience with racial disparities in outcome following DBS surgery may play a role in patient selection is unclear and should be explored in future analyses examining factors underlying the disparities in DBS for PD.
The sex disparity in the utilization of DBS for PD is another notable finding. We observed that female patients with PD are less likely to receive DBS than male patients with PD. Further analysis among genders across racial backgrounds demonstrates that whereas the odds of White female patients with PD receiving DBS is 0.8 compared with white male patients, women from other racial backgrounds are even less likely to receive DBS compared with men from other racial backgrounds. This trend was also observed for Black female patients compared with Black male patients, although the difference was not statistically significant, likely owing to inadequate power to detect a difference. The largest group of patients with PD in the United States is White men (gender prevalence estimated at 1.55 men and 1.00 women). 22 Despite the increased prevalence of PD in men, the number of patients referred for DBS is disproportional to the prevalence data. 23 , 24 A previous retrospective study at a single institution found that 75% of patients with PD referred for DBS were men. Furthermore, among patients that were referred and chose not to undergo DBS, patient preference was the dominant reason cited by female patients for not pursuing DBS, which was significantly more common than among male patients. 22 Therefore, sex referral bias as well as patient self‐selection may in part underlie the observed sex disparities, factors which may be amplified in non‐White patients with PD.
Increased DBS utilization has not yielded uniform benefit across racial groups. Epidemiological studies examining racial differences in incidence and prevalence of PD have demonstrated mixed findings. 25 Absent racial epidemiological differences, the persistent disparity that Black patients with PD are 5‐fold less likely to undergo DBS relative to White patients is notable. It should also be noted that the disparities in the utilization of DBS are unfortunately not unique to DBS surgery. Multiple studies have demonstrated that Black patients as well as patients from other racial minorities are less likely to receive surgery among a range of different procedures. 26 , 27 Furthermore, in the case of patients with epilepsy undergoing surgery over a similar time period as this study, the OR for Black patients receiving surgery was approximately 0.5 compared with White patients. 28 Our finding that the odds of Black patients receiving DBS is 0.2 compared with White patients is among the lowest for all elective surgery types. This highlights the importance of systemic factors inherent in the US medical system that contribute to the pervasive inequities of surgical care across racial groups. Several factors may underlie this disparity, including unconscious/implicit or conscious/explicit bias, which contributes to under‐representation of Black patients in specialty clinic referrals. 2 , 29 Interventions to change physician behavior generally focus on improving adherence to evidence‐based guidelines but these practices could be adapted to improving disparities in referrals or selection for DBS. Active methods, including continuing education, computerized decision support systems/reminders, and financial incentives, have been shown to effectively modify behavior. 30 Thus, whereas documenting disparities in health care increases awareness, reducing disparities depends upon changing physician behavior on a local, institutional level beginning with medical training. 31 For example, training physicians to utilize strategies, including individuation, 32 perspective taking, 33 and other strategies, 31 to combat implicit bias. Incorporating these strategies into undergraduate medical education would further spur systemic change necessary to truly reduce disparities in medical care.
Although changing individual behavior (either physician or patient) has been a main focus in disparities research, recent studies have highlighted that many additional factors, such as structural barriers, should be considered as contributing to racial disparities. For example, a potential barrier to access surgical care of PD is one of geography. Regional differences in the density of neurologists and neurosurgeons across urban and rural settings may influence access to care. Despite this hypothetical concern, a previous study examined this question and demonstrated that this does not appear to be a significant factor. 2 Whereas density of specialty care physicians and surgeons may not be a factor, access to specialty care via referral pattern biases or other mechanisms may underlie some of these disparities. Future multicenter registries that examine detailed patient demographic information (which is not available in a national dataset) would be helpful to better delineate the role geography may play in access to DBS therapy.
Socioeconomic status, and its complex association with many other factors, including racial background, access to transportation, social support, and time off of work for surgery and subsequent follow‐up visits (an important consideration given that frequent clinic visits are required for optimizing DBS therapy), represents another factor that may contribute to racial inequities in access to DBS. In agreement with a prior study, 3 we found that increasing income by zip code was a strong predictor of increased likelihood of receiving DBS compared to the lowest income quartile. In fact, at 1 year follow‐up of DBS placement, patients with PD with higher household incomes achieve better functional outcomes, 34 a factor which may bias referrals for DBS therapy toward higher income patients. Conversely, prior investigations suggest that Medicare coverage (a surrogate for lower socioeconomic status) among Black patients with PD may lead to a DBS surgery nonuse decision. 2 Significant differences in the medical management of PD have also been reported based on race and socioeconomic status. 35 Among surgical care more broadly, there is evidence to suggest that lower socioeconomic status is associated with reduced likelihood of receiving surgical care. 36 , 37 , 38 Socioeconomic status, therefore, is an important factor in the likelihood of receiving DBS therapy.
Marketing of DBS therapy may play a role in racial disparities in the utilization of DBS for PD. As one of the presumed drivers of DBS growth over the past 2 decades, marketing efforts may not be uniformly received and/or effective across racial backgrounds. The effects of device marketing for DBS systems have not been specifically investigated but the effects of direct to consumer marketing of pharmaceuticals has been studied. For example, Black patients are less likely to be exposed to direct to consumer marketing of pharmaceuticals compared with White patients, whereas Black patients are more likely to be influenced by such efforts. 39 Patient preference for innovative technologies, including implantable medical devices, may differ along racial lines which could play a role in how marketing efforts are received, although a previous study demonstrated that Black and White patients have similar preferences for implantable medical devices. 40
Patient‐specific factors may also contribute to racial disparity. Black patients with PD under‐report motor impairment compared with White patients and therefore present later in the disease process, delaying their diagnosis and treatment. 25 Studies examining this phenomenon have demonstrated that Black patients are more likely to view PD as a part of normal aging and therefore may be less likely to act on their symptoms to seek treatment. 41 A large single‐center study examining racial disparities in PD among 1,159 patients showed greater disability and disease severity in Black patients. 35 This finding is supported by Weuve et al who found that Black people may have higher rates of cognitive impairments, dementia, or other comorbidities among aged patients with PD, which may differentially limit their candidacy for DBS surgery, 42 although we found no differences in comorbidities among racial groups. Medical mistrust among Black patients due to repeated past mistreatment in studies, such as the Tuskegee Syphilis Study, 43 is also a factor contributing to delays in diagnosis and treatment. One recent study identified perceived discrimination due to income or insurance type as factors most strongly associated with medical mistrust among Black patients with perceived discrimination due to race/ethnic background being more weakly associated. 44 Highlighting patient factors that may influence referral or selection for DBS therapy is important. This is not because the patient's individual behavior fundamentally reduces the likelihood of receiving DBS but rather that the treating or referring physician must recognize patient‐specific factors that may unfairly bias the physician's treatment recommendations.
As discussed above, the exact causes of the persistent racial disparities in the use of DBS for PD are complex and multifactorial. Important open questions remain regarding DBS therapy. What is the rate that DBS is offered across racial groups? What is the rate at which DBS is offered but declined across racial groups? Although we cannot investigate these questions with our current data set, answering these questions may provide some degree of clarity regarding the root cause of racial disparities in DBS therapy for PD. For example, multicenter registries or single institution studies which track referrals and reasons for acceptance/decline would be able to provide some insight into the specific reasons for racial disparities and highlight potential avenues for improvement.
Increasing utilization of DBS for PD has not improved racial disparities in access to this important therapy. Although previous studies have identified racial disparities in DBS therapy, we believe that it is important to point out that nearly a decade after these findings were first reported, racial inequities in DBS therapy remain essentially unchanged. Highlighting persistent racial disparities in DBS is a preliminary effort to reform US health care delivery to ensure equal access to life altering treatments regardless of racial background.
Author Contributions
S.W.C., T.H.D., C.C.C., and R.A.M. contributed to the conception and design of the study. S.W.C., T.H.D., C.C.C., R.A.M., E.F.P., and J.D.H. were responsible for acquisition and analysis of data. S.W.C., T.H.D., E.F.P., A.N., A.L.R., S.G.N., J.T.H., A.N.P., M.A.H., J.D.H., C.C.C., and R.A.M. contributed to drafting the text and preparing the figures.
Potential Conflicts of Interest
The authors declare that they have no conflict of interest.
Acknowledgements
This study was supported in part by funding from MnDRIVE, a collaboration between the University of Minnesota and the State of Minnesota.
Contributor Information
Truong H. Do, Email: doxxx063@umn.edu.
Robert A. McGovern, Email: rmcgover@umn.edu.
References
- 1. Perestelo‐Pérez L, Rivero‐Santana A, Pérez‐Ramos J, et al. Deep brain stimulation in Parkinson's disease: meta‐analysis of randomized controlled trials. J Neurol 2014;261:2051–2060. [DOI] [PubMed] [Google Scholar]
- 2. Chan AK, McGovern RA, Brown LT, et al. Disparities in access to deep brain stimulation surgery for Parkinson disease: interaction between African American race and Medicaid use. JAMA Neurol 2014;71:291–299. [DOI] [PubMed] [Google Scholar]
- 3. Willis AW, Schootman M, Kung N, et al. Disparities in deep brain stimulation surgery among insured elders with Parkinson disease. Neurology 2014;82:163–171. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Fiscella K, Sanders MR. Racial and ethnic disparities in the quality of health care. Annu Rev Public Health 2016;37:375–394. [DOI] [PubMed] [Google Scholar]
- 5. Healthcare Cost and Utilization Project (HCUP) . HCUP National Inpatient Sample (NIS) [Internet] 2012; Available from: www.hcup-us.ahrq.gov/nisoverview.jsp
- 6. Johnson RA, Do TH, Palzer EF, et al. Pattern of technology diffusion in the adoption of stereotactic laser interstitial thermal therapy (LITT) in neuro‐oncology. J Neurooncol 2021;153:417–424. [DOI] [PubMed] [Google Scholar]
- 7. Johnston MC, Crilly M, Black C, et al. Defining and measuring multimorbidity: a systematic review of systematic reviews. Eur J Public Health 2019;29:182–189. [DOI] [PubMed] [Google Scholar]
- 8. Barnett JC, Berchick ER. Health insurance coverage in the United States: 2016. Washington, DC: US Government Printing Office, 2017. [Google Scholar]
- 9. National Intercensal Datasets : 2000. –2010; National population totals and components of change: 2010–2019 [internet]. United States Census Bureau 2016; [cited August 16, 2021]. Available from: https://www.census.gov/data/datasets/time-series/demo/popest/intercensal-2000-2010-national.html; https://www.census.gov/data/datasets/time-series/demo/popest/2010s-national-total.html
- 10. GBD 2016 Parkinson's Disease Collaborators . Global, regional, and national burden of Parkinson's disease, 1990–2016: a systematic analysis for the Global Burden of Disease Study 2016. Lancet Neurol 2018;17:939–953. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. McGovern RA, Sheehy JP, Zacharia BE, et al. Unchanged safety outcomes in deep brain stimulation surgery for Parkinson disease despite a decentralization of care. J Neurosurg 2013;119:1546–1555. [DOI] [PubMed] [Google Scholar]
- 12. Whitson HE, Hastings SN, Landerman LR, et al. Black‐white disparity in disability: the role of medical conditions. J Am Geriatr Soc 2011;59:844–850. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Fana M, Everett G, Fagan T, et al. Procedural outcomes of deep brain stimulation (DBS) surgery in rural and urban patient population settings. J Clin Neurosci 2020;72:310–315. [DOI] [PubMed] [Google Scholar]
- 14. Esnaola NF, Hall BL, Hosokawa PW, et al. Race and surgical outcomes: it is not all black and white. Ann Surg 2008;248:647–655. [DOI] [PubMed] [Google Scholar]
- 15. Thomas G, Almeida ND, Mast G, et al. Racial disparities affecting postoperative outcomes after brain tumor resection. World Neurosurg 2021;155:e665–e673. [DOI] [PubMed] [Google Scholar]
- 16. Sathianathen NJ, Jarosek SL, Fan Y, et al. Racial disparities in surgical outcomes among males following major urologic cancer surgery. Am J Prev Med 2018;55:S14–S21. [DOI] [PubMed] [Google Scholar]
- 17. Shahid H, Singh JA. Racial/ethnic disparity in rates and outcomes of Total joint arthroplasty. Curr Rheumatol Rep 2016;18:20. [DOI] [PubMed] [Google Scholar]
- 18. Amirian H, Torquati A, Omotosho P. Racial disparity in 30‐day outcomes of metabolic and bariatric surgery. Obes Surg 2020;30:1011–1020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Alosh H, Riley LH 3rd, Skolasky RL. Insurance status, geography, race, and ethnicity as predictors of anterior cervical spine surgery rates and in‐hospital mortality: an examination of United States trends from 1992 to 2005. Spine 2009;34:1956–1962. [DOI] [PubMed] [Google Scholar]
- 20. Curry WT Jr, Carter BS, Barker FG 2nd. Racial, ethnic, and socioeconomic disparities in patient outcomes after craniotomy for tumor in adult patients in the United States, 1988–2004. Neurosurgery 2010;66:427–437. discussion 437–8. [DOI] [PubMed] [Google Scholar]
- 21. Kim DH, Daskalakis C, Lee AN, et al. Racial disparity in the relationship between hospital volume and mortality among patients undergoing coronary artery bypass grafting. Ann Surg 2008;248:886–892. [DOI] [PubMed] [Google Scholar]
- 22. Shpiner DS, Di Luca DG, Cajigas I, et al. Gender disparities in deep brain stimulation for Parkinson's disease. Neuromodulation 2019;22:484–488. [DOI] [PubMed] [Google Scholar]
- 23. Katz M, Kilbane C, Rosengard J, et al. Referring patients for deep brain stimulation: an improving practice. Arch Neurol 2011;68:1027–1032. [DOI] [PubMed] [Google Scholar]
- 24. Willis AW, Evanoff BA, Lian M, et al. Geographic and ethnic variation in Parkinson disease: a population‐based study of US Medicare beneficiaries. Neuroepidemiology 2010;34:143–151. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Bailey M, Anderson S, Hall DA. Parkinson's disease in African Americans: a review of the current literature. J Parkinsons Dis 2020;10:831–841. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Haider AH, Scott VK, Rehman KA, et al. Racial disparities in surgical care and outcomes in the United States: a comprehensive review of patient, provider, and systemic factors. J Am Coll Surg 2013;216:482–92.e12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Best MJ, McFarland EG, Thakkar SC, Srikumaran U. Racial disparities in the use of surgical procedures in the US. JAMA Surg 2021;156:274–281. 10.1001/jamasurg.2020.6257. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Hamade YJ, Palzer EF, Helgeson ES, et al. Persistent racial and ethnic disparities as a potential source of epilepsy surgery underutilization: analysis of large national datasets from 2006–2016. Epilepsy Res 2021;176:106725. [DOI] [PubMed] [Google Scholar]
- 29. Wahid NA, Rosenblatt R, Brown RS Jr. A review of the current state of liver transplantation disparities. Liver Transpl 2021;27:434–443. [DOI] [PubMed] [Google Scholar]
- 30. Mostofian F, Ruban C, Simunovic N, Bhandari M. Changing physician behavior: what works? Am J Manag Care 2015;21:75–84. [PubMed] [Google Scholar]
- 31. Maina IW, Belton TD, Ginzberg S, et al. A decade of studying implicit racial/ethnic bias in healthcare providers using the implicit association test. Soc Sci Med 2018;199:219–229. [DOI] [PubMed] [Google Scholar]
- 32. Devine PG, Forscher PS, Austin AJ, Cox WTL. Long‐term reduction in implicit race bias: a prejudice habit‐breaking intervention. J Exp Soc Psychol 2012;48:1267–1278. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Burgess D, van Ryn M, Dovidio J, Saha S. Reducing racial bias among health care providers: lessons from social‐cognitive psychology. J Gen Intern Med 2007;22:882–887. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Genc G, Abboud H, Oravivattanakul S, et al. Socioeconomic status may impact functional outcome of deep brain stimulation surgery in Parkinson's disease. Neuromodulation 2016;19:25–30. [DOI] [PubMed] [Google Scholar]
- 35. Hemming JP, Gruber‐Baldini AL, Anderson KE, et al. Racial and socioeconomic disparities in parkinsonism. Arch Neurol 2011;68:498–503. [DOI] [PubMed] [Google Scholar]
- 36. Zak Y, Rhoads KF, Visser BC. Predictors of surgical intervention for hepatocellular carcinoma: race, socioeconomic status, and hospital type. Arch Surg 2011;146:778–784. [DOI] [PubMed] [Google Scholar]
- 37. Axelrod DA, Dzebisashvili N, Schnitzler MA, et al. The interplay of socioeconomic status, distance to center, and interdonor service area travel on kidney transplant access and outcomes. Clin J Am Soc Nephrol 2010;5:2276–2288. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. Cheung MC, Yang R, Byrne MM, et al. Are patients of low socioeconomic status receiving suboptimal management for pancreatic adenocarcinoma? Cancer 2010;116:723–733. [DOI] [PubMed] [Google Scholar]
- 39. Lee D, Begley CE. Racial and ethnic disparities in response to direct‐to‐consumer advertising. Am J Health Syst Pharm 2010;67:1185–1190. [DOI] [PubMed] [Google Scholar]
- 40. Groeneveld PW, Sonnad SS, Lee AK, et al. Racial differences in attitudes toward innovative medical technology. J Gen Intern Med 2006;21:559–563. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41. Pan S, Stutzbach J, Reichwein S, et al. Knowledge and attitudes about Parkinson's disease among a diverse group of older adults. J Cross Cult Gerontol 2014;29:339–352. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42. Weuve J, Barnes LL, Mendes de Leon CF, et al. Cognitive aging in Black and white Americans: cognition, cognitive decline, and incidence of Alzheimer disease dementia. Epidemiology 2018;29:151–159. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43. Gamble VN. Under the shadow of Tuskegee: African americans and health care. Am J Public Health 1997;87:1773–1778. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44. Bazargan M, Cobb S, Assari S. Discrimination and medical mistrust in a racially and ethnically diverse sample of California adults. Ann Fam Med 2021;19:4–15. [DOI] [PMC free article] [PubMed] [Google Scholar]


