Abstract
Background
Developmental coordination disorder (DCD) is a motor disorder of unknown aetiology that may have long‐term consequences on daily activities, and psychological and physical health. Studies investigating risk factors for DCD have so far provided inconsistent results.
Objectives
To assess, using a parent‐report screening tool, risk of DCD in school‐age very preterm children born in Italy, and investigate the associated early biomedical and sociodemographic factors.
Methods
A prospective area‐based cohort (804 children, response rate 73.4%) was assessed at 8–11 years of age in three Italian regions. Perinatal data were abstracted from medical records. DCD risk was measured using the Italian‐validated version of the Developmental Coordination Disorder Questionnaire (DCDQ‐IT). For this study, children with cognitive deficit (i.e. intelligence quotient <70), cerebral palsy, severe vision and hearing disabilities, and other impairments affecting movement were excluded. A total of 629 children were analysed. We used inverse probability weighting to account for loss to follow‐up, and multilevel, multivariable modified Poisson models to obtain adjusted risk ratio (aRR) and 95% confidence interval (CI). Missing values in the covariates were imputed.
Results
195 children (weighted proportion 31.8%, 95% CI 28.2, 35.6) scored positive on the DCDQ‐IT, corresponding to the 15th centile of the reference Movement‐ABC test. Factors associated with overall DCD risk were male sex (aRR 1.35, 95% CI 1.05, 1.73), intrauterine growth restriction (aRR 1.45, 95% CI 1.14, 1.85), retinopathy of prematurity (aRR 1.62, 95% CI 1.07, 2.45), and older maternal age at delivery (aRR 1.39, 95% CI 1.09, 1.77). Complete maternal milk feeding at discharge from the neonatal unit and higher parental socio‐economic status were associated with decreased risk.
Conclusions
Both biomedical and sociodemographic factors increase DCD risk. These findings can contribute to elucidating the origins of this disorder, and assist in the identification of children at risk for early referral and intervention.
Keywords: developmental coordination disorder, early diagnosis, risk factors, very preterm birth
Synopsis.
1. Study question
What factors are associated with risk of developmental coordination disorder in very preterm children?
2. What's already known
Developmental coordination disorder is a condition of unknown aetiology, more frequent in males and preterm children. Inadequate motor skills lead to disadvantages in school education, home tasks, and leisure activities, with impact on mental and physical health. Previous studies investigating risk factors have provided inconsistent results.
3. What this study adds
In children born very preterm, both biomedical (intrauterine growth restriction, antepartum haemorrhage, and retinopathy of prematurity) and sociodemographic (older maternal age and smoking in pregnancy) variables are associated with increased risk of developmental coordination disorder. Full maternal milk feeding at discharge and higher socio‐economic status are associated with lower risk.
1. BACKGROUND
Developmental coordination disorder (DCD) is a condition of unknown aetiology 1 characterised by a level of motor performance substantially lower than expected for chronological age and motor skill learning opportunities, which significantly interferes with daily activities and is not explained by intellectual disability or other known medical conditions or diseases such as visual, neurological or physical impairments. 2 The onset of symptoms occurs during the developmental period. 2 The reported prevalence in the general paediatric population is about 5%–6%, 1 but can range from 12% to over 50% in children born preterm. 3 , 4 , 5 , 6 , 7 , 8 Males are 2–7 times more likely to be affected compared with females. 1 , 3 , 4 , 5 , 6 , 7
Although the diagnosis is rarely made before 5–6 years of age, the onset can generally be traced back to early childhood, with difficulties in the acquisition of motor skills such as walking, use of common tools, catching an object, riding a bicycle, drawing and handwriting, leading in time to disadvantage in home tasks, recreational activities, and school curriculum. 1 DCD children have a mean intelligence quotient in the average range, yet they are more likely to have school outcomes poorer than peers. 9 They tend to engage in fewer physical and group activities, 10 with negative consequences on their self‐esteem, mental 11 and physical 12 health, social relations, and overall quality of life. 13 The difficulties encountered by these children may be increased by the presence of co‐morbidities such as attention deficit and hyperactivity disorder, learning disabilities, and specific language impairment. 14 The few available longitudinal studies suggest that, although remission is possible, in a substantial proportion of cases the problems in motor coordination continue to adolescence 8 and even adulthood, 15 and may substantially affect academic, professional, and emotional life.
Previous studies have investigated risk factors for DCD in the ante‐, peri‐, and neonatal periods, but results have been inconsistent. While males are generally found at higher risk, 1 , 4 , 5 , 6 , 7 a recent review states that this applies to the general paediatric population only. 16 Associations were reported between DCD and pregnancy exposures, such as active 4 , 17 , 18 , 19 and passive 20 , 21 maternal smoking, and with medical conditions including threatened abortion before 20 weeks, 22 infection/inflammation, 23 , 24 fetal distress during labour 22 , 25 and intrauterine growth restriction, 4 , 26 although this latter was not always confirmed 14 or explored 22 in subsequent studies.
Neonatal morbidities, including seizures, 22 prolonged jaundice, 22 disruption of brain white matter, 27 retinopathy of prematurity, 23 and bronchopulmonary dysplasia, 22 or treatments such as postnatal steroids 3 , 25 were reported to increase the risk of DCD. The impact of sociodemographic factors such as younger 28 and older 22 maternal age, low 29 and high 6 parental education, maternal unemployment, 28 and high professional status 4 was also reported and attributed to related perinatal conditions, or to the potential to modify environmental variables directly affecting child motor development. 30
Neuroimaging research suggests that DCD is related to brain pathology, and recently, Dewey et al 27 documented reduced volumes in white matter and total brain tissues, particularly in areas associated with motor functions such as the cerebellum, corpus callosum, thalamus, and basal ganglia, together with alterations in the microstructural organisation of brain white matter, possibly reflecting reduced myelination and axonal size.
Overall, however, despite the large number of studies carried out, most often with very preterm or low‐birthweight children, research findings are not yet conclusive and, as noted by a recent review, “surprisingly little is known about the aetiology of DCD.” 16
This study aimed to investigate the biological, medical, and sociodemographic factors associated with DCD risk assessed at school age. We focussed on very preterm (VP, i.e. <32 weeks of gestational age) children because of the higher frequency of DCD in this population and the availability of data from a prospective area‐based VP cohort followed up to school age in Italy.
2. METHODS
2.1. Cohort selection
The ACTION (Accesso alle Cure e Terapie Intensive Ostetrico‐Neonatali—access to intensive obstetrical and neonatal care) follow‐up project is an area‐based prospective cohort study including all very preterm infants born in 2003–2005 in five Italian regions, with follow‐up at 2 years of corrected age. 31 In three of the regions (Friuli‐Venezia Giulia, Toscana, and Lazio), a second wave of follow‐up was carried out at school age (mean 9.2 years, standard deviation 0.7) to assess general health, cognitive and neuropsychological development, and motor functions including the risk of DCD.
Out of 1350 newborns admitted to neonatal intensive care units (NICUs) in these regions, 1103 were discharged alive (Figure 1). There were eight documented deaths before school age, leaving 1095 children eligible for follow‐up. At school age, 158 children could not be traced, and for 133, parents refused participation. Thus, we collected information on 804 children (response rate 73.4%). In accordance with the DCD diagnostic criteria 2 for this study, we excluded children with cerebral palsy and other physical conditions limiting movement such as shorter limb; severe vision and hearing disabilities; and cognitive deficit, defined as full‐scale intelligence quotient (FSIQ) <70. In 65 cases, the parent‐report questionnaire used to assess DCD risk was missing or incomplete in some of the domains, precluding computation of the total score. We were able to impute 21 cases using the average of the completed questions within the same domain. Thus, 629 children were available for the analysis.
FIGURE 1.
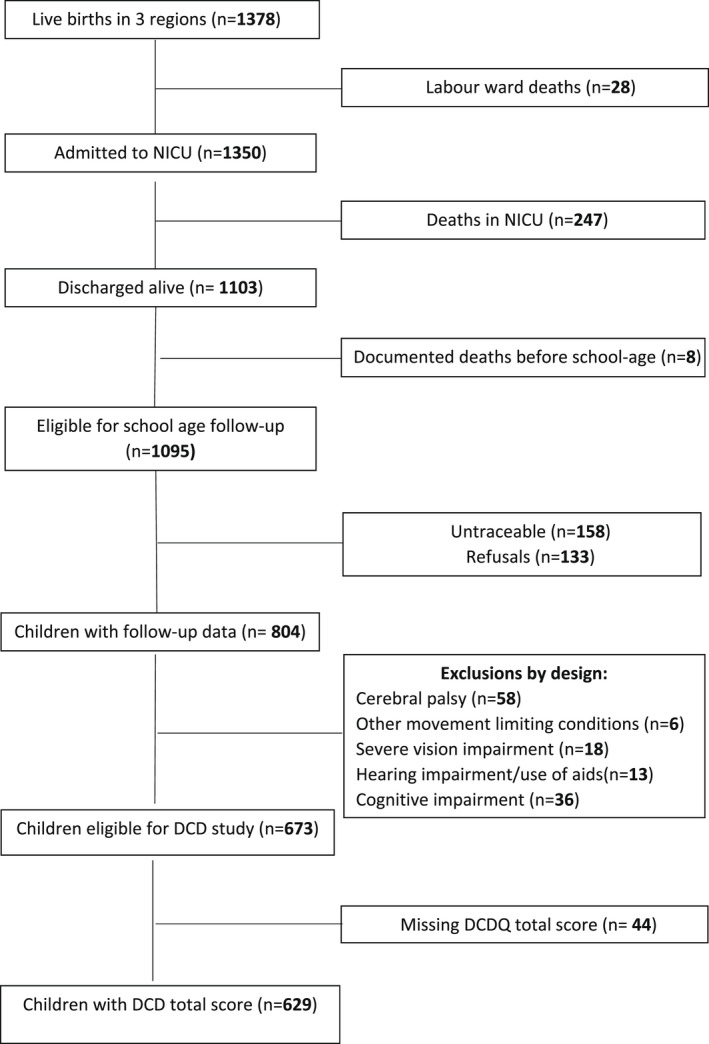
Flow chart of the study cohort
2.2. Measurements and data collection
Maternal and neonatal data were abstracted from clinical records using a data collection form with agreed definitions. GA was recorded as the best obstetrical estimate using information on the last menstrual period and ultrasound measurements. Small‐for‐gestational‐age (SGA) status was defined as birthweight <10th percentile using European intrauterine references. 32 Recorded morbidities in NICU included intraventricular haemorrhage (IVH) grades 3–4, periventricular leucomalacia (PVL), retinopathy of prematurity (ROP) stages 3–4, and bronchopulmonary dysplasia (BPD), defined as oxygen supplementation at 36 weeks’ postmenstrual age.
School‐age follow‐up was carried out in 4 hospital sites (2 in Friuli‐Venezia Giulia, 1 in Tuscany, and 1 in Lazio). Information on the child's health, including the presence and severity of cerebral palsy, data on vision and hearing, and use of aids were collected according to agreed definitions. We used the Italian‐validated version of the Kaufman Assessment Battery for Children, second edition (KABC‐II), to measure cognitive development (Mental Processing Index, MPI, equivalent to FSIQ). Values below the minimum were used to impute cases with missing assessment but documented severe cognitive deficit (n = 5).
The risk of DCD was assessed using the Developmental Coordination Disorder Questionnaire, 33 Italian‐validated version (DCDQ‐IT). 34 This 15‐item parent‐report tool describes child motor abilities in three separate areas (Control during Movement, Fine Motor/Handwriting, and General Coordination) using a set of positive affirmations (e.g. “Your child throws a ball in a controlled and accurate fashion”). Parents are asked to provide their assessment according to a 5‐point Likert scale (from 1, “not at all similar to your child,” to 5 “completely similar to your child”), in comparison with same‐age peers. The individual item scores are added to compute three area subscores and a total score (range 15–75), with higher values indicating no suspect/risk of DCD. Based on the total score, each child can be classified as “indicated, or suspected, DCD” versus “probably not DCD,” according to the age‐specific (5–7, 8–9, and 10–15 years) cut‐offs.
The validation of the Italian version (DCDQ‐IT) was carried out using the Movement Assessment Battery for Children (MABC, 1st edition) as reference standard. In a first study carried out on a clinical sample, 34 the DCDQ‐IT showed a sensitivity of 88% (95% CI 69, 97%) and a specificity of 96% (95% CI 86, 99%), while a second one based on a community sample led to lower values, that is 73% and 70%, respectively, for the age 8–10 years. 35
For this study, the questionnaire was completed by the parents in paper form at the time of the hospital visit.
2.3. Outcomes
The main outcome was the proportion of children at risk for DCD in the cohort, based on the DCDQ‐IT results. Secondary outcomes were the proportion of children at risk in the three DCD domains of Control during Movement, Fine motor/Handwriting, and General Coordination.
2.4. Statistical analysis
We used the Italian cut‐offs on DCDQ‐IT corresponding to the 15th percentile of the MABC reference standard in the validation study 35 to identify the children at risk for DCD in our very preterm population. We carried out uni‐ and multivariable analyses to explore the relation between potential predictors and indicated DCD status. Variables considered for inclusion in the analyses were region, GA, SGA status, multiple births, pregnancy complications, child sex, Apgar score at 5’, severe morbidities in NICU (intraventricular haemorrhage—IVH—stages 3–4; periventricular leucomalacia—PVL; any sepsis and/or necrotising enterocolitis—NEC; retinopathy of prematurity—ROP—stages 3–4; and bronchopulmonary dysplasia—BPD), feeding at discharge from NICU, and child age at DCDQ assessment. Sociodemographic variables were maternal country of birth (Italy or otherwise), age at delivery, and any maternal smoking in pregnancy as reported by the mother at the 2‐year follow‐up. The education and professional level of both parents were used to compute Hollingshead's composite index as a measure of household socio‐economic status (SES). 36
We used multilevel modified Poisson regression analysis 37 to obtain unadjusted and adjusted risk ratios (RRs) and 95% CIs of scoring positive at the 15th percentile cut‐off. Maternal identification code was included as random effect to take into account correlation within multiple births. To account for the missing cases due to loss to follow‐up, all analyses used inverse probability weights 38 to attribute a higher weight to subjects with characteristics of non‐responders (Table S1). Variables associated with the main or any of the secondary outcomes were retained in the final models.
As sensitivity analysis, we present all tables without IPW (Tables S2–S5).
Data analysis was carried out with STATA 17.0 SE (Stata Corporation, College Station, Texas).
2.4.1. Missing data
Missing values in covariates were all <5%, with the exception of any sepsis or NEC (16%) and maternal antenatal smoking (17%). We carried out multiple imputations with chained equations (MICE). 39 Data were assumed to be “missing at random.” Fifty data sets were imputed, using all variables included in the model as predictor or outcome.
2.5. Ethics approval
The school‐age follow‐up study (Prot. RF‐2009‐1511846) was approved by the Ethics Committee at the coordinating institute, Paediatric Hospital Bambino Gesù, IRCCS, on 9 May 2012 (Prot. N. 282 LB). Ethics Committees in Toscana and Friuli‐Venezia Giulia confirmed the approval. Written parental consent was obtained at the recruitment of the cohort and at follow‐up.
3. RESULTS
Table 1 shows the characteristics of the study population. The proportions are weighted to account for loss to follow‐up. Overall, more than half of the children were males, and almost one third was born from multiple births; 27.9% were SGA, defined as birthweight by gestational age <10th centile. Over 80% had a mother born in Italy, and in 37.1% of the children, maternal age was ≥35 years at delivery. A story of maternal smoking in pregnancy was reported for about 11% of the children. Hollingshead's composite index allowed to group children as belonging to low (36.8%), intermediate (35.0%), and high (28.2%) SES, respectively.
TABLE 1.
Characteristics of the study population by developmental coordination disorder risk (weighted proportions, column)
| Total (N = 629) | Not at risk for DCD (n = 434) | At risk for DCD (n = 195) | ||||
|---|---|---|---|---|---|---|
| N | % | n | % | n | % | |
| Child variables: | ||||||
| Child sex | ||||||
| Female | 278 | 43.6 | 211 | 47.5 | 67 | 35.3 |
| Male | 351 | 56.4 | 223 | 52.5 | 128 | 64.7 |
| Gestational age, weeks | ||||||
| <28 | 131 | 19.3 | 92 | 19.6 | 39 | 18.6 |
| ≥28 | 498 | 80.7 | 342 | 80.4 | 156 | 81.4 |
| Birthweight by GA | ||||||
| ≥10th percentile | 454 | 72.1 | 327 | 75.4 | 127 | 65.1 |
| <10th percentile | 175 | 27.9 | 107 | 24.6 | 68 | 34.9 |
| Multiple births | ||||||
| No | 442 | 72.6 | 307 | 73.2 | 135 | 71.3 |
| Yes | 187 | 27.4 | 127 | 26.8 | 60 | 28.7 |
| Apgar at 5 min | ||||||
| 0–6 | 110 | 17.3 | 76 | 17.0 | 34 | 17.9 |
| 7–10 | 517 | 82.7 | 357 | 83.0 | 160 | 82.1 |
| Any sepsis and/or NEC | ||||||
| No | 434 | 80.9 | 304 | 80.0 | 130 | 82.9 |
| Yes | 96 | 19.1 | 71 | 20.0 | 25 | 17.1 |
| Severe brain damage a | ||||||
| No | 595 | 95.1 | 417 | 95.9 | 178 | 93.3 |
| Yes | 28 | 4.9 | 16 | 4.1 | 12 | 6.7 |
| Retinopathy of prematurity stages 3–4 | ||||||
| No | 602 | 96.4 | 420 | 97.7 | 182 | 93.6 |
| Yes | 22 | 3.6 | 10 | 2.3 | 12 | 6.4 |
| Bronchopulmonary dysplasia | ||||||
| No | 590 | 93.8 | 411 | 94.9 | 179 | 91.3 |
| Yes | 39 | 6.2 | 23 | 5.1 | 16 | 8.7 |
| Feeding at discharge | ||||||
| Mixed/full formula | 469 | 76.7 | 307 | 73.0 | 162 | 84.8 |
| Maternal milk only | 158 | 23.3 | 125 | 27.0 | 33 | 15.2 |
| Child age at DCD assessment, y | ||||||
| 8–9 | 413 | 65.8 | 289 | 66.8 | 124 | 63.5 |
| 10–11 | 216 | 34.2 | 145 | 33.2 | 71 | 36.5 |
| Parental variables | ||||||
| Maternal age | ||||||
| <35 years | 384 | 62.9 | 279 | 66.0 | 105 | 56.2 |
| ≥35 years | 241 | 37.1 | 152 | 34.0 | 89 | 43.8 |
| Maternal country of birth | ||||||
| Italy | 516 | 83.7 | 353 | 83.6 | 163 | 84.0 |
| Other | 113 | 16.3 | 81 | 16.4 | 32 | 16.0 |
| Pregnancy hypertensive disorders | ||||||
| No | 493 | 80.0 | 343 | 80.1 | 150 | 79.8 |
| Yes | 128 | 20.0 | 88 | 19.9 | 40 | 20.2 |
| Antepartum haemorrhage | ||||||
| No | 543 | 86.8 | 380 | 87.8 | 163 | 84.7 |
| Yes | 78 | 13.2 | 51 | 12.2 | 27 | 15.3 |
| PROM | ||||||
| No | 447 | 71.6 | 314 | 72.3 | 133 | 70.2 |
| Yes | 177 | 28.4 | 119 | 27.7 | 58 | 29.8 |
| Any smoking in pregnancy | ||||||
| No | 469 | 88.9 | 337 | 91.4 | 132 | 83.5 |
| Yes | 53 | 11.1 | 30 | 8.6 | 23 | 16.5 |
| Maternal education | ||||||
| Upper secondary or more | 458 | 70.6 | 332 | 74.9 | 126 | 61.5 |
| Lower secondary or less | 166 | 29.4 | 99 | 25.1 | 67 | 38.5 |
| Paternal education | ||||||
| Upper secondary or more | 380 | 60.3 | 270 | 62.7 | 110 | 55.0 |
| Lower secondary or less | 230 | 39.7 | 150 | 37.3 | 80 | 45.0 |
| Maternal occupation | ||||||
| Manager/professional | 89 | 13.3 | 63 | 13.5 | 26 | 12.8 |
| White‐collar worker | 242 | 38.5 | 186 | 43.5 | 56 | 27.6 |
| Manual worker | 98 | 16.3 | 58 | 13.6 | 40 | 22.1 |
| Unemployed/housewife | 192 | 31.9 | 122 | 29.4 | 70 | 37.5 |
| Paternal occupation | ||||||
| Manager/professional | 166 | 26.4 | 120 | 27.6 | 46 | 24.1 |
| White‐collar worker | 292 | 48.3 | 197 | 48.2 | 95 | 48.5 |
| Manual worker | 108 | 19.3 | 71 | 18.4 | 37 | 21.2 |
| Unemployed | 32 | 6.0 | 22 | 5.8 | 10 | 6.2 |
| Hollingshead's SES index | ||||||
| Low | 211 | 36.8 | 133 | 33.7 | 78 | 43.5 |
| Medium | 219 | 35.0 | 153 | 35.6 | 66 | 33.7 |
| High | 185 | 28.2 | 139 | 30.7 | 46 | 22.8 |
| Region | ||||||
| FVG | 98 | 14.8 | 71 | 15.5 | 27 | 13.1 |
| Lazio | 325 | 53.7 | 212 | 50.9 | 113 | 59.8 |
| Tuscany | 206 | 31.5 | 151 | 33.6 | 55 | 27.1 |
Intraventricular haemorrhage grades III–IV and/or periventricular leucomalacia/porencephaly.
Overall, 195 children (weighted proportion 31.8%, 95% CI 28.2, 35.6) scored <15th centile reference cut‐off, indicating risk of DCD. Figures stratified by sex and by DCDQ domain are shown in Table 2. In all domains, risks were higher for males.
TABLE 2.
Risk for developmental coordination disorder by child sex (weighted proportions and 95% CIs)
| Total | At risk for DCD | ||
|---|---|---|---|
| N | n | % (95% CI) | |
| Total DCD | |||
| Female | 278 | 67 | 25.7 (20.7, 31.2) |
| Male | 351 | 128 | 36.5 (31.6, 41.8) |
| Control during movement | |||
| Female | 278 | 85 | 31.4 (26.0, 37.1) |
| Male | 351 | 124 | 35.7 (30.8, 40.9) |
| Fine motor/handwriting | |||
| Female | 278 | 59 | 22.3 (17.9, 28.0) |
| Male | 351 | 153 | 44.2 (39.0, 49.6) |
| General coordination | |||
| Female | 278 | 59 | 23.5 (18.6, 28.8) |
| Male | 351 | 117 | 33.2 (28.5, 38.5) |
Table 3 shows the weighted distribution of DCD risk, overall and by domain, by the characteristics of the study population, together with univariable RRs and 95% confidence intervals.
TABLE 3.
Distribution of developmental coordination disorder risk, overall and by domain, by the characteristics of the study population (weighted proportions and risk ratios) a
| Total (N = 629) | Total DCD | Control during movement | Fine motor/handwriting | General coordination | |||||
|---|---|---|---|---|---|---|---|---|---|
| At risk (n = 195) | Risk ratio | At risk (n = 209) | Risk ratio | At risk (n = 212) | Risk ratio | At risk (n = 176) | Risk ratio | ||
| n | n (%) | (95% CI) | n (%) | (95% CI) | n (%) | (95% CI) | n (%) | (95% CI) | |
| Child variables | |||||||||
| Child sex | |||||||||
| Female | 278 | 67 (25.7) | 1.00 (Reference) | 85 (31.4) | 1.00 (Reference) | 59 (22.3) | 1.00 (Reference) | 59 (23.5) | 1.00 (Reference) |
| Male | 351 | 128 (36.5) | 1.42 (1.10, 1.83) | 124 (35.7) | 1.14 (0.90, 1.43) | 153 (44.2) | 1.98 (1.52, 2.58) | 117 (33.2) | 1.41 (1.07, 1.87) |
| Gestational age, weeks | |||||||||
| <28 | 131 | 39 (30.7) | 1.00 (Reference) | 45 (36.2) | 1.00 (Reference) | 42 (32.7) | 1.00 (Reference) | 44 (35.6) | 1.00 (Reference) |
| ≥28 | 498 | 156 (32.1) | 1.04 (0.77, 1.41) | 164 (33.2) | 0.92 (0.69, 1.21) | 170 (35.1) | 1.07 (0.80, 1.44) | 132 (27.4) | 0.77 (0.57, 1.03) |
| Birthweight by GA | |||||||||
| ≥10th percentile | 454 | 127 (28.7) | 1.00 (Reference) | 141 (31.8) | 1.00 (Reference) | 144 (32.7) | 1.00 (Reference) | 118 (27.2) | 1.00 (Reference) |
| <10th percentile | 175 | 68 (39.8) | 1.39 (1.09, 1.76) | 68 (38.8) | 1.22 (0.96, 1.55) | 68 (39.6) | 1.21 (0.96, 1.52) | 58 (33.3) | 1.22 (0.93, 1.60) |
| Multiple births | |||||||||
| No | 442 | 135 (31.3) | 1.00 (Reference) | 152 (34.4) | 1.00 (Reference) | 145 (33.7) | 1.00 (Reference) | 131 (30.6) | 1.00 (Reference) |
| Yes | 187 | 60 (33.3) | 1.06 (0.81, 1.40) | 57 (32.0) | 0.93 (0.70, 1.24) | 67 (37.3) | 1.11 (0.84, 1.46) | 45 (24.5) | 0.80 (0.56, 1.13) |
| Apgar at 5 min | |||||||||
| 0–6 | 110 | 34 (32.9) | 1.00 (Reference) | 39 (37.8) | 1.00 (Reference) | 39 (37.7) | 1.00 (Reference) | 35 (32.4) | 1.00 (Reference) |
| 7–10 | 517 | 160 (31.6) | 0.96 (0.71, 1.30) | 169 (33.0) | 0.87 (0.66, 1.15) | 172 (34.0) | 0.90 (0.68, 1.20) | 141 (28.2) | 0.87 (0.64, 1.20) |
| Any sepsis and/or NEC | |||||||||
| No | 434 | 130 (30.5) | 1.00 (Reference) | 137 (32.1) | 1.00 (Reference) | 135 (31.7) | 1.00 (Reference) | 117 (27.3) | 1.00 (Reference) |
| Yes | 96 | 25 (26.6) | 0.87 (0.60, 1.27) | 30 (31.1) | 0.97 (0.69, 1.36) | 32 (34.7) | 1.09 (0.80, 1.50) | 29 (31.4) | 1.15 (0.81, 1.63) |
| Severe brain damage | |||||||||
| No | 595 | 178 (30.7) | 1.00 (Reference) | 193 (33.0) | 1.00 (Reference) | 199 (34.6) | 1.00 (Reference) | 159 (27.7) | 1.00 (Reference) |
| Yes | 28 | 12 (42.3) | 1.38 (0.86, 2.20) | 11 (39.6) | 1.20 (0.73, 1.97) | 9 (30.1) | 0.87 (0.48, 1.56) | 12 (41.6) | 1.50 (0.93, 2.42) |
| Retinopathy of prematurity stage 3–4 | |||||||||
| No | 602 | 182 (31.1) | 1.00 (Reference) | 195 (33.1) | 1.00 (Reference) | 198 (34.1) | 1.00 (Reference) | 162 (27.9) | 1.00 (Reference) |
| Yes | 22 | 12 (56.3) | 1.81 (1.22, 2.68) | 13 (57.5) | 1.73 (1.18, 2.55) | 13 (57.2) | 1.68 (1.14, 2.48) | 13 (60.9) | 2.18 (1.52, 3.14) |
| Bronchopulmonary dysplasia | |||||||||
| No | 590 | 179 (31.0) | 1.00 (Reference) | 192 (33.0) | 1.00 (Reference) | 199 (34.7) | 1.00 (Reference) | 161 (28.3) | 1.00 (Reference) |
| Yes | 39 | 16 (44.6) | 1.44 (0.99, 2.10) | 17 (45.1) | 1.37 (0.93, 2.01) | 13 (34.1) | 0.98 (0.61, 1.57) | 15 (38.4) | 1.36 (0.87, 2.11) |
| Feeding at discharge | |||||||||
| Mixed/full formula | 469 | 162 (35.2) | 1.00 (Reference) | 166 (35.7) | 1.00 (Reference) | 178 (38.8) | 1.00 (Reference) | 143 (31.3) | 1.00 (Reference) |
| Maternal milk only | 158 | 33 (20.7) | 0.59 (0.42, 0.83) | 43 (27.3) | 0.76 (0.57, 1.03) | 34 (21.0) | 0.54 (0.39, 0.76) | 33 (21.2) | 0.68 (0.48, 0.95) |
| Child age at follow‐up, y | |||||||||
| 8–9 | 413 | 124 (30.7) | 1.00 (Reference) | 154 (37.6) | 1.00 (Reference) | 132 (32.9) | 1.00 (Reference) | 108 (26.8) | 1.00 (Reference) |
| 10–11 | 216 | 71 (33.9) | 1.10 (0.86, 1.42) | 55 (26.5) | 0.70 (0.53, 0.93) | 80 (38.1) | 1.16 (0.90, 1.49) | 68 (33.1) | 1.24 (0.94, 1.63) |
| Parental variables | |||||||||
| Maternal age | |||||||||
| <35 years | 384 | 105 (28.4) | 1.00 (Reference) | 113 (30.1) | 1.00 (Reference) | 127 (34.1) | 1.00 (Reference) | 102 (27.9) | 1.00 (Reference) |
| ≥35 years | 241 | 89 (37.5) | 1.32 (1.04, 1.68) | 95 (40.0) | 1.33 (1.05, 1.68) | 84 (35.6) | 1.04 (0.82, 1.33) | 74 (30.8) | 1.10 (0.84, 1.45) |
| Maternal country of birth | |||||||||
| Italy | 516 | 163 (31.9) | 1.00 (Reference) | 177 (34.2) | 1.00 (Reference) | 174 (34.5) | 1.00 (Reference) | 145 (28.4) | 1.00 (Reference) |
| Other | 113 | 32 (31.3) | 0.98 (0.70, 1.38) | 32 (31.6) | 0.92 (0.66, 1.29) | 38 (35.3) | 1.02 (0.75, 1.39) | 31 (31.5) | 1.11 (0.78, 1.57) |
| Pregnancy Hypertensive Disorders | |||||||||
| No | 493 | 150 (31.5) | 1.00 (Reference) | 166 (34.8) | 1.00 (Reference) | 167 (35.3) | 1.00 (Reference) | 135 (28.5) | 1.00 (Reference) |
| Yes | 128 | 40 (31.9) | 1.01 (0.75, 1.36) | 40 (30.5) | 0.88 (0.65, 1.19) | 42 (32.9) | 0.93 (0.69, 1.25) | 37 (29.7) | 1.04 (0.75, 1.46) |
| Antepartum haemorrhage | |||||||||
| No | 543 | 163 (30.8) | 1.00 (Reference) | 178 (33.3) | 1.00 (Reference) | 185 (35.2) | 1.00 (Reference) | 140 (26.5) | 1.00 (Reference) |
| Yes | 78 | 27 (36.6) | 1.19 (0.85, 1.65) | 28 (37.7) | 1.13 (0.82, 1.57) | 24 (32.1) | 0.91 (0.64, 1.30) | 32 (43.2) | 1.63 (1.20, 2.22) |
| PROM | |||||||||
| No | 447 | 133 (31.0) | 1.00 (Reference) | 147 (33.5) | 1.00 (Reference) | 146 (34.0) | 1.00 (Reference) | 121 (28.0) | 1.00 (Reference) |
| Yes | 177 | 58 (33.1) | 1.07 (0.82, 1.39) | 59 (34.3) | 1.02 (0.79, 1.32) | 64 (36.8) | 1.08 (0.84, 1.39) | 52 (30.5) | 1.09 (0.82, 1.46) |
| Any smoking in pregnancy | |||||||||
| No | 469 | 132 (28.7) | 1.00 (Reference) | 145 (31.3) | 1.00 (Reference) | 146 (31.7) | 1.00 (Reference) | 126 (27.6) | 1.00 (Reference) |
| Yes | 53 | 23 (45.7) | 1.59 (1.14, 2.23) | 25 (49.4) | 1.58 (1.15, 2.17) | 23 (43.2) | 1.36 (0.96, 1.92) | 18 (35.2) | 1.28 (0.86, 1.90) |
| Hollingshead SES index | |||||||||
| Low | 211 | 78 (37.4) | 1.00 (Reference) | 79 (38.1) | 1.00 (Reference) | 79 (38.7) | 1.00 (Reference) | 71 (34.6) | 1.00 (Reference) |
| Medium | 219 | 66 (30.5) | 0.81 (0.61, 1.08) | 69 (31.0) | 0.81 (0.61, 1.08) | 78 (36.3) | 0.94 (0.71, 1.23) | 53 (24.8) | 0.71 (0.52, 0.99) |
| High | 185 | 46 (25.6) | 0.68 (0.50, 0.94) | 55 (30.4) | 0.80 (0.59, 1.08) | 48 (26.6) | 0.69 (0.50, 0.95) | 48 (26.5) | 0.76 (0.55, 1.06) |
| Region | |||||||||
| FVG | 98 | 27 (28.2) | 1.00 (Reference) | 30 (31.2) | 1.00 (Reference) | 30 (30.7) | 1.00 (Reference) | 25 (25.8) | 1.00 (Reference) |
| Lazio | 325 | 113 (35.4) | 1.25 (0.87, 1.80) | 127 (39.2) | 1.25 (0.89, 1.77) | 119 (37.5) | 1.22 (0.86, 1.73) | 95 (30.3) | 1.17 (0.79, 1.76) |
| Tuscany | 206 | 55 (27.4) | 0.97 (0.64, 1.45) | 52 (25.8) | 0.83 (0.55, 1.24) | 63 (31.7) | 1.03 (0.70, 1.52) | 56 (28.1) | 1.09 (0.70, 1.68) |
Risk ratios were computed using modified Poisson univariable models. Random effect: maternal identification code.
The results of multivariable analysis are presented in Table 4. Male sex was confirmed to be a risk factor, particularly in the Fine motor/Handwriting domain (aRR 1.87, 95% CI 1.43, 2.43), but showed no association with Control during Movement. Also, previous ROP (aRR 1.62, 95% CI 1.07, 2.45), SGA (aRR 1.45, 95% CI 1.14, 1.85), and maternal age at delivery ≥35 years (aRR 1.39, 95% CI 1.09, 1.77) were associated with overall DCD risk. Smoking in pregnancy and antepartum haemorrhage increased DCD risk in the Control during Movement (1.39, 95% CI 0.98, 1.96) and General Coordination (aRR 1.73, 95% CI 1.27, 2.36) domains, respectively. Exclusive maternal milk feeding at discharge from NICU, high SES index, and, for Control during Movement only, older child age were associated with lower DCD risk.
TABLE 4.
Factors associated with developmental coordination disorder risk: results of multivariable analysis (weighted data) a
| Total DCD | Control during movement | Fine motor/handwriting | General coordination | |
|---|---|---|---|---|
| aRR (95% CI) | aRR (95% CI) | aRR (95% CI) | aRR (95% CI) | |
| Child sex | ||||
| Female | 1.00 (Reference) | 1.00 (Reference) | 1.00 (Reference) | 1.00 (Reference) |
| Male | 1.35 (1.05, 1.73) | 1.11 (0.88, 1.39) | 1.87 (1.43, 2.43) | 1.35 (1.03, 1.77) |
| Birthweight by GA | ||||
| ≥10 percentile | 1.00 (Reference) | 1.00 (Reference) | 1.00 (Reference) | 1.00 (Reference) |
| <10 percentile | 1.45 (1.14, 1.85) | 1.24 (0.97, 1.58) | 1.22 (0.97, 1.54) | 1.35 (1.03, 1.77) |
| Antepartum haemorrhage | ||||
| No | 1.00 (Reference) | 1.00 (Reference) | 1.00 (Reference) | 1.00 (Reference) |
| Yes | 1.27 (0.92, 1.76) | 1.19 (0.86, 1.65) | 0.93 (0.66, 1.32) | 1.73 (1.27, 2.36) |
| ROP (grade 3–4) | ||||
| No | 1.00 (Reference) | 1.00 (Reference) | 1.00 (Reference) | 1.00 (Reference) |
| Yes | 1.62 (1.07, 2.45) | 1.70 (1.13, 2.55) | 1.33 (0.87, 2.05) | 2.02 (1.39, 2.93) |
| Feeding on discharge | ||||
| Artificial milk partial/complete | 1.00 (Reference) | 1.00 (Reference) | 1.00 (Reference) | 1.00 (Reference) |
| Maternal milk only | 0.66 (0.47, 0.93) | 0.82 (0.61, 1.11) | 0.61 (0.43, 0.85) | 0.74 (0.52, 1.05) |
| Maternal age at delivery | ||||
| <35 years | 1.00 (Reference) | 1.00 (Reference) | 1.00 (Reference) | 1.00 (Reference) |
| ≥35 years | 1.39 (1.09, 1.77) | 1.36 (1.07, 1.72) | 1.10 (0.87, 1.39) | 1.19 (0.91, 1.56) |
| Smoking in pregnancy | ||||
| No | 1.00 (Reference) | 1.00 (Reference) | 1.00 (Reference) | 1.00 (Reference) |
| Yes | 1.33 (0.92, 1.91) | 1.39 (0.98, 1.96) | 1.17 (0.83, 1.65) | 1.11 (0.74, 1.68) |
| Hollingshead's SES index | ||||
| Low | 1.00 (Reference) | 1.00 (Reference) | 1.00 (Reference) | 1.00 (Reference) |
| Medium | 0.82 (0.62, 1.08) | 0.82 (0.62, 1.08) | 0.97 (0.74, 1.26) | 0.74 (0.54, 1.01) |
| High | 0.72 (0.52, 0.99) | 0.82 (0.60, 1.11) | 0.75 (0.54, 1.03) | 0.84 (0.60, 1.18) |
| Age at DCDQ assessment, y | ||||
| 8–9 | 1.00 (Reference) | 1.00 (Reference) | 1.00 (Reference) | 1.00 (Reference) |
| 10–11 | 1.08 (0.84, 1.38) | 0.70 (0.53, 0.92) | 1.12 (0.88, 1.41) | 1.20 (0.92, 1.57) |
Multivariable multilevel modified Poisson regression analysis based on imputed data (50 imputations). Random effect: maternal identification code. aRR is adjusted risk ratio.
4. COMMENT
4.1. Principal findings
In this area‐based study of school‐age VP children, we found an overall proportion of DCD risk of 31.8%, consistent with the results of the literature. 7 , 8 Both antenatal factors (older maternal age, antepartum haemorrhage, and smoking in pregnancy) and child variables (male sex, SGA, and ROP stages 3–4) were associated with increased DCD risk, overall and/or in specific domains. Full maternal milk feeding at discharge from the neonatal unit and high family SES were associated with decreased risk. Gestational age was not related to DCD risk in our data, most likely because of our study restriction to VP births only. 5
4.2. Strengths of the study
The strengths of this study are the relatively large size and the area‐based prospective recruitment of the cohort. The response rate was satisfactory for this type of study, and availability of baseline data allowed us to use IPW to adjust for loss to follow‐up. The proportion of missing values in covariates was below 5% except for antenatal smoking, and multiple imputations were used to retain all cases in multivariable analyses.
4.3. Limitations of the data
The main limitation of this study is the use of a parental questionnaire for the assessment of DCD. However, after standardised motor tests, DCDQ is considered one of the best tools to determine whether a child may have the disorder or is unlikely to be affected. 40 It was translated into several languages, and studies using it are included in systematic reviews. 16 A second limitation is the use of maternal reported smoking in pregnancy, without biological marker measurement to verify the exposure. Although previous studies have validated self‐reported smoking by pregnant women, 41 others have not. 42 Misclassification of smokers into non‐smokers cannot be excluded, but would likely lead to underestimation of the association with the outcome.
4.4. Interpretation
Consistent with most previous research, we found that males had higher DCD risk compared with females. Male sex has since long been linked to worse perinatal outcomes, including neurological and developmental disabilities. 43 The underlying mechanisms are poorly understood, and the male disadvantage has been attributed to a general biological or genetic vulnerability, partly linked to the single X chromosome. More recently, sex‐related genetic polymorphisms in the genes encoding proinflammatory cytokines have been reported, 44 as well as differences in the DNA methylation placental profile, suggesting that male fetuses might be more vulnerable to epigenetic processes affecting neurodevelopment. 45
Our results regarding the role of SGA are consistent with those first described in the Danish National Birth cohort, 4 as well as with the review by Murray et al. 26 who reported a higher risk of motor impairment among SGA children born preterm. Differences in the definition of SGA may in part explain the inconsistencies with other studies. 14 Similar to the Danish cohort study, 4 we used an intrauterine reference, 32 which provides a better estimate of fetal growth restriction, particularly for very preterm children. 46
Despite the exclusion of children with severe visual impairments, we found that previous ROP was the strongest predictor of DCD risk in our population. Previous studies have linked ROP to non‐visual developmental disabilities, especially in the cognitive and motor areas, 47 even in case of favourable vision outcomes and independently of acquired major brain injury. 48 , 49 Involvement of the macula area of the retina has been especially linked to subsequent non‐visual disability. 47 Neuroimaging data have shown associations between ROP, developmental impairment, and brain abnormalities such as reduced volumes 49 and maturational delay of the brain white matter, particularly in the posterior regions, optic radiations, and posterior limbs of the internal and external capsule, where the primary visual and motor pathways are housed. 48 These findings suggest that a common cause may lead to impaired neurovascular and neural development in the retina and in the brain. One postulated mechanism involves the anabolic hormone insulin‐like growth factor 1 (IGF‐1), whose low levels after preterm birth may impair brain growth and maturation, as well as normal angiogenesis, leading in the retina to the pathologic revascularisation, which is the hallmark of ROP. 48 , 50 Postnatal systemic inflammation with activation of proinflammatory cytokines was also proposed as common cause, 51 either by itself or in combination with IGF‐1 deficiency. 52 Inflammation can exert an influence also before birth. 53 Antenatal haemorrhage, which is considered a marker of inflammation, was associated in our study with increased risk of General Coordination problems.
As with other developmental outcomes, 54 we found that socio‐economic variables such as maternal age, smoking in pregnancy, parental education, and occupational profession may be important in shaping the neuropsychological profile of a child, and in our study were associated with risk of motor problems even when controlling for biomedical factors. The root of these relations may be both socio‐economic and educational, linked to the level of home affordances, 30 opportunities for motor activities, training, and outdoor leisure time, as well as to more active parenting practices and stimulating environment. 55
While the effect of maternal smoking on intrauterine growth is well known, its role in developmental disorders, and specifically DCD, has been less researched. We found that both SGA and smoking were independently associated with increased DCD risk, although in different domains, with only smoking affecting Control during Movement. These findings are consistent with previous research showing a relation between antenatal smoking and motor difficulties, 17 even when cotinine markers were used 18 , 20 and when socio‐economic factors and alcohol intake were taken into account. 19 Cigarette smoke contains several harmful substances that can cross the placenta and affect the child prenatally. 56 Nicotine binds to receptors of several organs, including the brain. The cerebellum, which is particularly rich in nicotine receptors, is responsible for general coordination of movements and body balance, and has been implicated in the pathogenesis of DCD. 27
The benefits of breast feeding on the child health and development, including cognition, have been largely discussed in the literature, providing justification for including this variable in our analyses. Belfort et al. 57 found that, in infants below 30 weeks of GA, breastmilk intake in the first 28 days of life was associated with greater brain deep nuclear grey matter at term equivalent age, and better motor and cognitive performance at 7 years of age. However, these findings were not replicated by subsequent studies, 55 , 58 which did not identify any feeding associated differences in the achievement of motor milestones in very preterm infants. Our study found that, adjusting for maternal age, smoking in pregnancy, and SES index, maternal milk feeding at discharge from the neonatal unit was associated with lower DCD risk, particularly overall and in the Fine Motor/Handwriting domains. Proposed mechanisms linking breast feeding with brain development are the nutritional properties of breastmilk, 57 and possibly the quality of mother–infant relation that may be enhanced by breast feeding. 55 , 57 Recently, the report by Kar et al.y 59 of an association between breast‐feeding exclusivity and duration and brain global and regional white matter microstructure has provided additional support to the link between breast feeding and infant motor development.
5. CONCLUSIONS
Our study confirms the high risk of DCD in children born very preterm and highlights the association with both biomedical and socio‐economic variables. The pattern of associations differs according to the specific motor domains. General coordination appears to be influenced mainly by biomedical factors (sex, SGA, antepartum haemorrhage, and a history of severe ROP), while Control during Movement is affected also by maternal age and smoking in pregnancy. The identification of these factors and their differential distribution across motor domains, together with the accumulating findings of neuroimaging studies, can contribute to elucidating the aetiopathogenetic mechanisms of DCD, as well as to the discussion about possible different DCD subtypes. Additionally, they may alert parents, teachers, and physicians to suspect DCD when an association between these risk factors and motor problems is present, referring the child for specialist assessment and early intervention, rather than dismissing the issue as mere clumsiness that will be overgrown with time.
Author contributions
Stefania Zoia and Marina Biancotto drafted the manuscript. Alessandra Valletti and Laura Montelisciani carried out statistical analysis; Ileana Croci was in charge of the database management. Fabio Voller and Franca Rusconi in Tuscany, Marco Carrozzi and Valeria Chiandotto in Friuli Venezia Giulia, and Domenico di Lallo, Barbara Caravale and Stefano Vicari in Lazio contributed to the preparation of the study instruments and to the coordination of data collection. Marina Cuttini initiated the study, provided overall project coordination, supervised statistical analysis and finalized the manuscript. All Authors critically reviewed the manuscript for important aspects and approved the final version.
Supporting information
Table S1
Table S2
Table S3
Table S4
Table S5
ACKNOWLEDGEMENTS
Data collection and neurodevelopmental assessments were carried out by Gina Riccio, Chiara Giorno, Grazia Giana, and Caterina Rosa in Lazio; Valentina Svelto in Toscana; and Emanuela Pessina, Nicoletta Bressan, Dora Cosentini, and Cristiana Melon in Friuli‐Venezia Giulia. The study central coordination staff in Roma included Maria Lacchei and Angela Pignatelli. We thank the IRTEF Institute for data coding and development of the database. We are very grateful to the families and children who participated in our study.
Zoia S, Biancotto M, Caravale B, et al. Early factors associated with risk of developmental coordination disorder in very preterm children: A prospective area‐based cohort study in Italy. Paediatr Perinat Epidemiol. 2022;36:683–695. doi: 10.1111/ppe.12878
Funding information
The ACTION follow‐up study was funded by the Italian Ministry of Health (RF‐2009‐1511846) and by two unrestricted grants from Chiesi Farmaceutici S.p.A. Additional resources for this work came from the European Union's Horizon 2020 Program (GA No. 733280)
REFERENCES
- 1. Blank R, Barnett AL, Cairney J, et al. International clinical practice recommendations on the definition, diagnosis, assessment, intervention, and psychosocial aspects of developmental coordination disorder. Dev Med Child Neurol. 2019;61:242‐285. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. American Psychiatric Association . Diagnostic and Statistical Manual of Mental Disorders (5th edition). American Psychiatric Association; 2013. [Google Scholar]
- 3. Zwicker JG, Yoon SW, Mackay M, Petrie‐Thomas J, Rogers M, Synnes AR. Perinatal and neonatal predictors of developmental coordination disorder in very low birthweight children. Arch Dis Child. 2013;98:118‐122. [DOI] [PubMed] [Google Scholar]
- 4. Faebo Larsen R, Hvas Mortensen L, Martinussen T, Nybo Andersen AM. Determinants of developmental coordination disorder in 7‐year‐old children: a study of children in the Danish National Birth Cohort. Dev Med Child Neurol. 2013;55:1016‐1022. [DOI] [PubMed] [Google Scholar]
- 5. Caravale B, Herich L, Zoia S, et al. Risk of Developmental Coordination Disorder in Italian very preterm children at school age compared to general population controls. Eur J Paediatr Neurol. 2019;23:296‐303. [DOI] [PubMed] [Google Scholar]
- 6. Du W, Ke L, Wang Y, Hua J, Duan W, Barnett AL. The prenatal, postnatal, neonatal, and family environmental risk factors for Developmental Coordination Disorder: A study with a national representative sample. Res Dev Disabil. 2020;104:103699. [DOI] [PubMed] [Google Scholar]
- 7. Evensen KAI, Ustad T, Tikanmäki M, Haaramo P, Kajantie E. Long‐term motor outcomes of very preterm and/or very low birth weight individuals without cerebral palsy: A review of the current evidence. Semin Fetal Neonatal Med. 2020;25:101116. [DOI] [PubMed] [Google Scholar]
- 8. Spittle AJ, Dewey D, Nguyen TN, et al. Rates of developmental coordination disorder in children born very preterm. J Pediatr. 2021;231:61‐67. [DOI] [PubMed] [Google Scholar]
- 9. Harrowell I, Hollén L, Lingam R, Emond A. The impact of developmental coordination disorder on educational achievement in secondary school. Res Dev Disabil. 2018;72:13‐22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Izadi‐Najafabadi S, Ryan N, Ghafooripoor G, Gill K, Zwicker JG. Participation of children with developmental coordination disorder. Res Dev Disabil. 2019;84:75‐84. [DOI] [PubMed] [Google Scholar]
- 11. Lingam R, Jongmans MJ, Ellis M, Hunt LP, Golding J, Emond A. Mental health difficulties in children with developmental coordination disorder. Pediatrics. 2012;129:e882‐e891. [DOI] [PubMed] [Google Scholar]
- 12. Hendrix CG, Prins MR, Dekkers H. Developmental coordination disorder and overweight and obesity in children: a systematic review. Obes Rev. 2014;15:408‐423. [DOI] [PubMed] [Google Scholar]
- 13. Karras HC, Morin DN, Gill K, Izadi‐Najafabadi S, Zwicker JG. Health‐related quality of life of children with Developmental Coordination Disorder. Res Dev Disabil. 2019;84:85‐95. [DOI] [PubMed] [Google Scholar]
- 14. Bolk J, Farooqi A, Hafström M, Åden U, Serenius F. Developmental Coordination Disorder and its association with developmental comorbidities at 6.5 Years in apparently healthy children born extremely preterm. JAMA Pediatr. 2018;172:765‐774. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Tal‐Saban M, Zarka S, Grotto I, Ornoy A, Parush S. The functional profile of young adults with suspected Developmental Coordination Disorder (DCD). Res Dev Disabil. 2012;33:2193‐2202. [DOI] [PubMed] [Google Scholar]
- 16. van Hoorn JF, Schoemaker MM, Stuive I, et al. Risk factors in early life for developmental coordination disorder: a scoping review. Dev Med Child Neurol. 2021;63:511‐519. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Trasti N, Vik T, Jacobsen G, Bakketeig LS. Smoking in pregnancy and children's mental and motor development at age 1 and 5 years. Early Hum Dev. 1999;55:137‐147. [DOI] [PubMed] [Google Scholar]
- 18. Christensen LH, Høyer BB, Pedersen HS, et al. Prenatal smoking exposure, measured as maternal serum cotinine, and children's motor developmental milestones and motor function: A follow‐up study. Neurotoxicology. 2016;53:236‐245. [DOI] [PubMed] [Google Scholar]
- 19. Suyama S, Yagyu K, Araki A, et al. Risk factors for motor coordination problems in preschool‐aged children. Pediatrics Int. 2020;62:1177‐1183. [DOI] [PubMed] [Google Scholar]
- 20. Evlampidou I, Bagkeris M, Vardavas C, et al. Prenatal second‐hand smoke exposure measured with urine cotinine may reduce gross motor development at 18 Months of Age. J Pediatr. 2015;167:246‐252. [DOI] [PubMed] [Google Scholar]
- 21. Mahlberg N, James ME, Bulten R, Rodriguez C, Kwan M, Cairney J. Investigating the association between exposure to second hand smoke in utero and developmental coordination disorder. Front Pediatr. 2019;7:1‐7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Hua J, Gu G, Jiang P, Zhang L, Zhu L, Meng W. The prenatal, perinatal and neonatal risk factors for children's developmental coordination disorder: a population study in mainland China. Res Dev Disabil. 2014;35:619‐625. [DOI] [PubMed] [Google Scholar]
- 23. Goyen TA, Lui K. Developmental coordination disorder in "apparently normal" schoolchildren born extremely preterm. Arch Dis Child. 2009;94:298‐302. [DOI] [PubMed] [Google Scholar]
- 24. Holst C, Jørgensen SE, Wohlfahrt J, Nybo Andersen AM, Melbye M. Fever during pregnancy and motor development in children: a study within the Danish National Birth Cohort. Dev Med Child Neurol. 2015;57:725‐732. [DOI] [PubMed] [Google Scholar]
- 25. Arnaud C, Daubisse‐Marliac L, White‐Koning M, et al. Prevalence and associated factors of minor neuromotor dysfunctions at age 5 years in prematurely born children. Arch Pediatr Adolesc Med. 2007;161:1053‐1061. [DOI] [PubMed] [Google Scholar]
- 26. Murray E, Fernandes M, el Faz M, Kennedy SH, Villar J, Stein A. Differential effect of intrauterine growth restriction on childhood neurodevelopment: a systematic review. BJOG. 2015;122:1062‐1072. [DOI] [PubMed] [Google Scholar]
- 27. Dewey D, Thompson DK, Kelly CE, et al. Very preterm children at risk for developmental coordination disorder have brain alterations in motor areas. Acta Paediatr. 2019;108:1649‐1660. [DOI] [PubMed] [Google Scholar]
- 28. Moreira RS, Magalhães LC, Dourado JS, Lemos SM, Alves CR. Factors influencing the motor development of prematurely born school‐aged children in Brazil. Res Dev Disabil. 2014;35:1941‐1951. [DOI] [PubMed] [Google Scholar]
- 29. Strooband KFB, de Rosnay M, Okely AD. Prevalence and risk factors of pre‐schoolers’ fine motor delay within vulnerable Australian communities. J Paediatr Child Health. 2021;57:114‐120. [DOI] [PubMed] [Google Scholar]
- 30. Freitas TC, Gabbard C, Caçola P, Montebelo MI, Santos DC. Family socioeconomic status and the provision of motor affordances in the home. Braz J Phys Ther. 2013;17:319‐327. [DOI] [PubMed] [Google Scholar]
- 31. Cuttini M, Caravale B, Carnielli V, et al. A two‐year follow‐up study of very preterm infants in Italy: aims and study design. Paediatr Child Health. 2009;19:S145‐S151. [Google Scholar]
- 32. Zeitlin J, Bonamy AE, Piedvache A, et al. Variation in term birth weight across European countries affects the prevalence of small for gestational age among very preterm infants. Acta Paediatr. 2017;106:1447‐1455. [DOI] [PubMed] [Google Scholar]
- 33. Wilson BN, Crawford SG, Green D, Roberts G, Aylott A, Kaplan BJ. Psychometric properties of the revised Developmental Coordination Disorder Questionnaire. Phys Occup Ther Pediatr. 2009;29:182‐202. [DOI] [PubMed] [Google Scholar]
- 34. Caravale B, Baldi S, Gasparini C, Wilson BN. Cross‐cultural adaptation, reliability and predictive validity of the Italian version of Developmental Coordination Disorder Questionnaire (DCDQ). Eur J Paediatr Neurol. 2014;18:267‐272. [DOI] [PubMed] [Google Scholar]
- 35. Caravale B, Baldi S, Capone L, Presaghi F, Balottin U, Zoppello M. Psychometric properties of the Italian version of the Developmental Coordination Disorder Questionnaire (DCDQ‐Italian). Res Dev Disabil. 2015;36:543‐550. [DOI] [PubMed] [Google Scholar]
- 36. Hollingshead AA. Four‐factor index of social status. Yale University; 1975. Unpublished manuscript. [Google Scholar]
- 37. Zou GY, Donner A. Extension of the modified Poisson regression model to prospective studies with correlated binary data. Stat Methods Med Res. 2013;22:661‐670. [DOI] [PubMed] [Google Scholar]
- 38. Piedvache A, van Buuren S, Barros H, Ribeiro AI, Draper E, Zeitlin J. EPICE Research group. Strategies for assessing the impact of loss to follow‐up on estimates of neurodevelopmental impairment in a very preterm cohort at 2 years of age. BMC Med Res Methodol. 2021;21:118‐126. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39. White IR, Royston P, Wood AM. Multiple imputation using chained equations: issues and guidance for practice. Stat Med. 2011;30:377‐399. [DOI] [PubMed] [Google Scholar]
- 40. Harris SR, Mickelson ECR, Zwicker G. Diagnosis and management of developmental coordination disorder. CMAJ. 2015;187:659‐665. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41. Klebanoff MA, Levine RJ, Morris CD, et al. Accuracy of self‐reported cigarette smoking among pregnant women in the 1990s. Paediatr Perinat Epidemiol. 2001;15:140‐143. [DOI] [PubMed] [Google Scholar]
- 42. England LJ, Grauman A, Qian C, et al. Misclassification of maternal smoking status and its effects on an epidemiologic study of pregnancy outcomes. Nicotine Tob Res. 2007;9:1005‐1013. [DOI] [PubMed] [Google Scholar]
- 43. Marlow N, Wolke D, Bracewell MA, Samara M. EPICure Study Group. Neurologic and developmental disability at six years of age after extremely preterm birth. NEJM. 2005;352:9‐19. [DOI] [PubMed] [Google Scholar]
- 44. Bi D, Chen M, Zhang X, et al. The association between sex‐related interleukin‐6 gene polymorphisms and the risk for cerebral palsy. J Neuroinflammation. 2014;11:100‐111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45. Lesseur C, Armstrong DA, Murphy MA, et al. Sex‐specific associations between placental leptin promoter DNA methylation and infant neurobehavior. Psychoneuroendocrinology. 2014;40:1‐9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46. Hutcheon JA, Platt RW. The missing data problem in birth weight percentiles and thresholds for "small‐for‐gestational‐age". Am J Epidemiol. 2008;167:786‐792. [DOI] [PubMed] [Google Scholar]
- 47. Beligere N, Perumalswamy V, Tandon M, et al. Retinopathy of prematurity and neurodevelopmental disabilities in premature infants. Semin Fetal Neonatal Med. 2015;20:346‐353. [DOI] [PubMed] [Google Scholar]
- 48. Glass TJA, Chau V, Gardiner J, et al. Severe retinopathy of prematurity predicts delayed white matter maturation and poorer neurodevelopment. Arch Dis Child Fetal Neonatal Ed. 2017;102:F532‐F537. [DOI] [PubMed] [Google Scholar]
- 49. Sveinsdóttir K, Ley D, Hövel H, et al. Relation of retinopathy of prematurity to brain volumes at term equivalent age and developmental outcome at 2 years of corrected age in very preterm infants. Neonatology. 2018;114:46‐52. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50. Hellström A, Engström E, Hård AL, et al. Postnatal serum insulin‐like growth factor I deficiency is associated with retinopathy of prematurity and other complications of premature birth. Pediatrics. 2003;112:1016‐1020. [DOI] [PubMed] [Google Scholar]
- 51. Morken TS, Dammann O, Skranes J, Austeng D. Retinopathy of prematurity, visual and neurodevelopmental outcome, and imaging of the central nervous system. Semin Perinatol. 2019;43:381‐389. [DOI] [PubMed] [Google Scholar]
- 52. Hellgren G, Löfqvist C, Hansen‐Pupp I, et al. Increased postnatal concentrations of pro‐inflammatory cytokines are associated with reduced IGF‐I levels and retinopathy of prematurity. Growth Hormon IGF Res. 2018;39:19‐24. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53. Gagliardi L, Rusconi F, Da Frè M, et al. Pregnancy disorders leading to very preterm birth influence neonatal outcomes: results of the population‐based ACTION cohort study. Pediatr Res. 2013;73(6):794‐801. [DOI] [PubMed] [Google Scholar]
- 54. Arrhenius B, Gyllenberg D, Chudal R, et al. Social risk factors for speech, scholastic and coordination disorders: a nationwide register‐based study. BMC Public Health. 2018;18:739‐748. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55. Valentini NC, de Borba LS, Panceri C, Smith BA, Procianoy RS, Silveira RC. Early detection of cognitive, language, and motor delays for low‐income preterm infants: a Brazilian cohort longitudinal study on infant neurodevelopment and maternal practice. Front Psych. 2021;12:753551. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56. Rogers JM. Tobacco and pregnancy. Reprod Toxicol. 2009;28:152‐160. [DOI] [PubMed] [Google Scholar]
- 57. Belfort MB, Anderson PJ, Nowak VA, et al. Breast milk feeding, brain development, and neurocognitive outcomes: a 7‐year longitudinal study in infants born at less than 30 weeks’ gestation. J Pediatr. 2016;177:133‐139 e131. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58. Michels KA, Ghassabian A, Mumford SL, et al. Breastfeeding and motor development in term and preterm infants in a longitudinal US cohort. Am J Clin Nutr. 2017;106:1456‐1462. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59. Kar P, Reynolds JE, Grohs MN, et al. Association between breastfeeding during infancy and white matter microstructure in early childhood. NeuroImage 2021;236:118084. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Table S1
Table S2
Table S3
Table S4
Table S5


