Abstract
N6-methyladenosine (m6A) is a widely investigated RNA modification in studies on the “epigenetic regulation” of mRNAs that is ubiquitously present in eukaryotes. Abnormal changes in m6A levels are closely related to the regulation of RNA metabolism, heat shock stress, tumor occurrence, and development. m6A modifications are catalyzed by the m6A writer complex, which contains RNA methyltransferase-like 3 (METTL3), methyltransferase-like 14 (METTL14), Wilms tumor 1-associated protein (WTAP), and other proteins with methyltransferase (MTase) capability, such as RNA-binding motif protein 15 (RBM15), KIAA1429 and zinc finger CCCH-type containing 13 (ZC3H13). Although METTL3 is the main catalytic subunit, WTAP is a regulatory subunit whose function is to recruit the m6A methyltransferase complex to the target mRNA. Specifically, WTAP is required for the accumulation of METTL3 and METTL14 in nuclear speckles. In this paper, we briefly introduce the molecular mechanism of m6A modification. Then, we focus on WTAP, a component of the m6A methyltransferase complex, and introduce its structure, localization, and physiological functions. Finally, we describe its roles and mechanisms in cancer.
Subject terms: Oncogenes, Methylation
Facts
N6-methyladenosine RNA modification (m6A) is one of the most abundant modifications in eukaryotic mRNA, which plays an important role in cancer initiation and progression.
m6A methylation is catalyzed by a multicomponent methyltransferase complex including: METTL3, METTL14, WTAP, METTL16, KIAA1429, RBM15, RBM15B, ZC3H13. WTAP serves as an essential regulatory subunit in methyltransferase which recruits m6A methyltransferase complex to the target mRNA.
WTAP plays dual roles in cancer either as an oncogene or as a tumor suppressor. It might regulate cancer though m6A methylation or other signaling pathways.
Open questions
How does WTAP recruit methyltransferase complex to the target mRNA?
What determines WTAP localization and in what condition WTAP forms up complexes as WTAP-BCLAF1-THRAP3, WT1-WTAP, or METTL3-METTL14-WTAP?
Background
Epigenetics is a branch of genetics that investigates heritable changes in gene expression without changes in the nucleotide sequence [1, 2]. Epigenetic regulation has been observed in the context of DNA methylation [3], histone modifications [4], chromatin remodeling [5], transcriptional control [6], noncoding RNAs [7], and cancer immunotherapy [8]. Posttranscriptional modifications, including m1A [9], m5C [10], and m6A [11], are abundant and significant, especially m6A modifications, because they are considered the most abundant internal modification in eukaryotes [12], with approximately 25% of mRNAs carrying at least one m6A site [13, 14]. m6A modifications can be added not only to mRNAs but also to rRNAs, small nucleolar RNAs (snRNAs), and microRNAs [7, 15]. m6A modification affects RNA export, leads to spliced pre-mRNAs, and impacts RNA translation and stability [16]. Abnormal regulation of m6A has been observed in cancers, and its role as an oncogene or tumor suppressor depends on the cellular environment [17, 18].
The main methyltransferases are METTL3, METTL14, and WTAP, which form the m6A methyltransferase complex (MTC). The m6A level is largely dependent on the MTC. Numerous studies have revealed that the m6A level is of great concern in heart failure [19], testosterone synthesis [20], liver steatosis [21], and different cancers [22, 23]. The m6A modification plays a dual role in cancer biology and is important for the recognition of cancer progression and cancer therapy [24]. To provide a more comprehensive understanding of m6A methyltransferase, we focused on WTAP, a constituent of the m6A methyltransferase complex.
WTAP was first identified as a splicing factor and then confirmed to be the third component of methyltransferase [14, 25, 26]. In addition, WTAP fulfils several biological functions, including embryo development, cell cycle progression, cell differentiation, pre-mRNA splicing, and antiviral responses. In this review, we first describe the biological functions of WTAP in detail. Then, we focus on the role of WTAP in cancers either dependent or independent of METTL3-METTL14 methyltransferase and summarize the specific mechanisms of WTAP in tumorigenesis and development.
Molecular mechanism of m6A modification
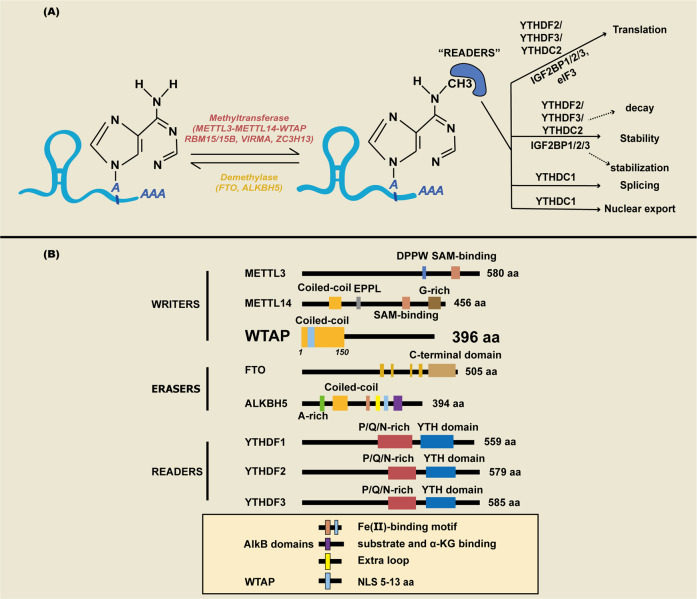
m6A is a widely investigated RNA modification in studies on “epigenetic regulation” [27, 28]. The m6A RNA modification accounts for 80% of all RNA modifications related to pre-mRNA splicing, miRNAs, lncRNAs, circRNA processing, translation efficiency, and mRNA stability [29]. m6A is a dynamic, reversible posttranscriptional modification. The residues of adenosine at the N6 position are localized in the 3ʹ untranslated region (UTR) of the mRNA or close to the termination codon [30, 31]. This modification can occur in different biological processes and is mediated by corresponding enzymes termed “writers,” “erasers,” and “readers” [32].
Methyltransferase-like protein 3 (METTL3) and S-adenosylmethionine (SAM)-binding protein [33] are the most significant components of the methyltransferase complex [34–38]. Methyltransferase-like protein 14 (METTL14) colocalizes with METTL3 in nuclear speckles at a 1:1 ratio [39–43], where it stabilizes the m6A methyltransferase complex (MTC) and recognizes specific RNA sequences (RRACH) [30, 44]. WTAP recruits METTL3 and METTL14 into nuclear speckles (associated with mRNA export) and is crucial for this unique localization [14, 25, 26]. Furthermore, RNA-binding motif protein 15 (RBM15) can bind to WTAP and recruit the MTC to specific RNA sites for m6A modification [45]. This process is important for the control of m6A-promoted X-chromosome inactivation in humans [46]. Zinc finger CCCH-type containing 13 (ZC3H13) interacts with WTAP to retain the MTC in nuclear speckles via its LC domain and thereby promotes its function [47, 48]. Other m6A writers have been revealed in recent years, including METTL16, METTL5, VIRMA, and ZCCHC4 [49–53].
After the “writers” mark the target mRNA, “reader” proteins, such as YT521-B homology (YTH) domain-containing protein [54–62], eukaryotic translation initiation factor 3 (eIF3) [63], the IGF2 mRNA binding protein (IGF2BP) family [64–67], and the heterogeneous nuclear ribonucleoprotein (HNRNP) protein family [68, 69], decode m6A methylation to generate signals for nuclear export, translation, RNA splicing, RNA stabilization, and decay [70].
Fat and obesity-related protein (FTO) [71–73] and alkB homolog 5 (ALKBH5) [74–76] are two essential enzymes for demethylation. “Erasers” are involved in building up the dynamic, reversible modification with “Writers” and “Readers” [77].
In general, m6A modification is an abundant and powerful epigenetic modification in eukaryotes. If one key enzyme is disordered, this dynamic modification is disrupted, which impacts human diseases (Table 1, Fig. 1).
Table 1.
Summary of m6A modification enzymes.
| Components | Enzymes | Intracellular localization | Biological functions | References |
|---|---|---|---|---|
| WRITERS | METTL3 | Cytoplasm, Nucleus, Nuclear speckles | m6A methyltransferase, DNA damage responses, DNA‒RNA hybrid, Cancer cell proliferation, Cell cycle progression and survival, Cancer cell resistance to radiotherapy and cisplatin | [35–38] |
| METTL14 | Nucleus | m6A methyltransferase, mRNA degradation or stabilization, LncRNA stabilization, pre-mRNA splicing, mRNA exportation, mRNA turnover in tumor proliferation, Metastasis, Self-renewal and tumor-initiating capacity | [41–44] | |
| WTAP | Cytoplasm, Nucleus, Nuclear speckles | m6A methyltransferase, Embryo development, Cell cycle progression and differentiation, Pre-mRNA splicing, Antiviral responses, Alternative splicing | [78, 82–84, 86, 87, 98, 100, 101] | |
| RBM15/ ZC3H13/ VIRMA | Nuclear speckles, Nucleus, Nuclear envelope, Nuclear membrane | m6A methyltransferase, Proliferation, invasion, migration, and apoptosis, Anchoring the m6A regulatory complex in the nucleus, Controls mouse embryonic stem cell self-renewal | [45, 48, 51, 70] | |
| ZCCHC4 | Nucleus, Cytoplasm | Methylates human 28 S rRNA, Interacts with a subset of mRNAs, Related to global translation, Cell proliferation | [52] | |
| METTL5 | Nucleus, Cell junction | m6A modification of 18 S rRNA, Promotes translation initiation, S6K activation, and cancer cell growth | [50, 53] | |
| METTL16 | Nucleus, Cytoplasm | m6A modification of U6 snRNA, lncRNAs, and introns of pre-mRNAs | [49] | |
| ERASERS | FTO | Cytoplasm, Nucleus, Nuclear speckles | Demethylation of m6A and m1A, Regulation of mRNA splicing and cell differentiation | [71–73] |
| ALKBH5 | Nuclear speckles | m6A demethylation, Participates in the regulation of mRNA nuclear export and mouse sperm development, Reduces tumoral proliferative, migration, and invasion activities | [74–76] | |
| READERS | YTHDF2/3 | Nucleus, Cytoplasm | mRNA stabilization/degradation, Regulates mRNA clearance, Regulates cancer cell proliferation, invasion and migration | [54, 58, 59, 83] |
| YTHDC1 | Nucleus, Nuclear speckles | Binds m6A-modified pre-mRNAs and mRNAs, and facilitates exon inclusion, splicing, mRNA nuclear-cytoplasmic export | [55, 60, 61] | |
| IGF2BP1–3 | Cytoplasm, Nucleus | Recognizes m6A through K homology domains and facilitates m6A-modified mRNA stabilization and protein translation | [64–67] | |
| YTHDC2 | Cytoplasm | Regulates mRNA translation or decay and mouse spermatogenesis | [56] | |
| YTHDF1 | Cytoplasm | Selectively recognizes m6A-modified mRNA, Promotes ribosome loading of m6A-modified mRNA, Interacts with initiation factors to facilitate translation initiation | [57, 62] | |
| hnRNPC/hnRNPG | Nucleus | Regulates mRNA structure and alternative splicing | [69] |
Fig. 1. Mechanism of m6A and fuctional domais in m6A methyltransferase.
A The dynamic molecular mechanism of m6A modification. m6A is installed by “writers” (METTL3/14, WTAP, RBM15/15B, VIRMA, and ZC3H13), removed by “erasers” (FTO, ALKBH5, and ALKBH3), and recognized by “readers” (YTHDC1/2, YTHDF1/2/3, IGF2BP1/2/3, HNRNP, and eIF3). B Functional domains in m6A writer, eraser, and reader proteins.
Overview of WTAP
Structure and cellular localization of WTAP
Wilms’ tumor 1-associating protein (WTAP) is encoded on human chromosomal region 6q25.3 [78]. WTAP is a 44 kDa protein that contains 396 amino acids and is encoded by the human homolog of FL (2)d [79]. WTAP localizes to both the nucleus and cytoplasm [25, 80]. WTAP is a key component in m6A modification, forming a complex with VIRMA, CBLL1, ZC3H13 (KIAA0853), RBM15/15B, and METTL3/14 [80]. WTAP contains an extended N-terminal coiled-coil region followed by an unstructured C-terminal part [81] (Fig. 1B). WTAP regulates the localization of the stable heterodimer core complex of METTL3/14 into nuclear speckles through amino acids 5–13 of the nuclear localization signal (NLS) (-PLPKKVRL- to -PLPGGVGL-) at its N-terminus [81]. Notably, the N-terminal coiled-coil region (1–150 amino acids) that contains the NLS is the binding surface of METTL3, which links to the helical structure at the N-terminus of METTL3, called the leader helix (LH) [81]. Although WT1 was found to interact with WTAP, it was confirmed that WT1 was dispensable for the regulation of m6A modification by WTAP [25] (Fig. 1B).
Biological functions of WTAP
Embryo development
In mice, WTAP plays an essential role in embryonic development. WTAP knockout embryos exhibit proliferative failure [82], and heterozygous mice die at embryonic day 10.5 [83]. In pigs, WTAP knockdown reduced the blastocyst rate and total m6A levels [84].
Cell cycle progression and differentiation
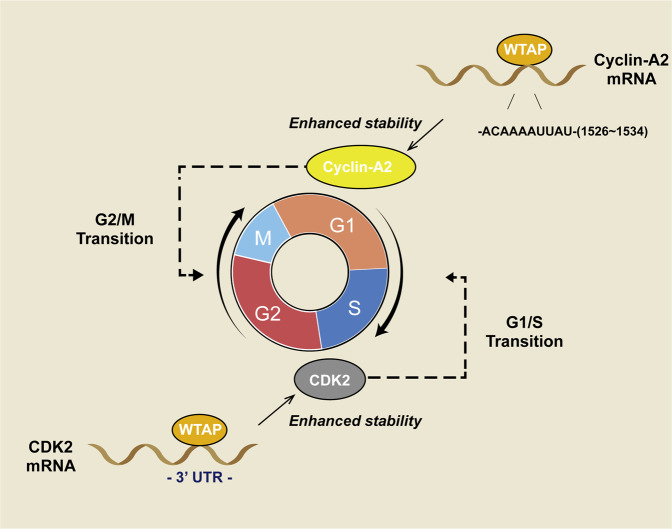
Cell proliferation and differentiation are the foundation of growth, development, reproduction, and heredity in organisms [85]. In human umbilical vein endothelial cells (HUVECs), decreased WTAP levels induced cell cycle arrest in the G2 phase. At the same time, the protein levels of cyclin-A2, B1, B2, and CDC20, which are related to the cell cycle [86, 87], were significantly decreased [82]. Mechanistically, WTAP stabilizes cyclin-A2 mRNA by binding to its AUUUA motif ACAAAUUAU, which corresponds to the 3ʹ UTR (1526–1534) [82]. These findings indicated that WTAP promotes the G2/M transition in HUVECs (Fig. 2) [82].
Fig. 2. The function of WTAP in cell cycle transition.
In keratinocytes and renal cell carcinoma cells, WTAP enhances the stability of the CDK2 mRNA by directly binding to its 3’-UTR. In human umbilical vein endothelial cells (HUVECs), WTAP stabilizes cyclin-A2 mRNA by binding to its AUUUA motif ACAAAUUAU, which corresponds to the 3ʹ UTR (1526–1534). These findings indicated that WTAP promotes the G1/S transition and the G2/M transition.
WTAP regulates CDK2 mRNA stability, which is related to the G1/S transition [88], in renal cell carcinoma (RCC) and keratinocytes [89]. During RCC cell proliferation, WTAP enhances the stability of the CDK2 mRNA by directly binding to its 3ʹ-UTR (Fig. 2) [89]. In psoriasis, WTAP not only stabilizes the CDK2 mRNA but also stabilizes the cyclin-A2 mRNA, which promotes the G2/M transition [90]. The binding motif of WTAP in the cyclin-A2 mRNA is ACAAAAUUAU (1526–1534) [82]. Smooth muscle cells (SMCs) proliferate during vascular restructuring and switch to a nonproliferative state when remodeling is complete [91]. The efficiency of WT1 binding to its target promoter is affected by WTAP in the nucleus. Amphiregulin belongs to the epidermal growth factor gene family, which serves as a strong mitogen in SMCs and is regulated by WT1 [92]. When WTAP levels decrease in SMCs, more WT1 bound to the promoter of amphiregulin, switching the cell to a proliferative state. Bcl-2, a protooncogenic apoptosis suppressor, is also activated by WT1 [93]. WTAP was upregulated when SMCs were in a nonproliferative state or the late stage of repair in the intima of injured arteries. Overexpression of WTAP prevents WT1 from binding to the Bcl-2 promoter, thereby downregulating Bcl-2 and activating apoptosis (Fig. 3A) [94].
Fig. 3.
A Model of the mechanism through which WTAP regulates SMC proliferation. The balance between WTAP and WT1 influences the state of SMCs. When the expression of WTAP is reduced, WT1-mediated transcriptional events proceed. Amphiregulin is a direct transcriptional target of WT1 that drives SMC proliferation by upregulating the EGF pathway. Thus, SMCs switch to a proliferative state. When the balance of WTAP and WT1 is reversed, WT1-mediated transcription may be blocked, and the transcription of Bcl-2, which is suppressed by WT1, is activated. SMC apoptosis is increased, and the cells switch to a nonproliferative state. B WTAP in the antiviral immune response. WTAP is degraded in virus-infected cells. After viral infection, degradation of WTAP leads to a decrease in the m6A level of IRF3 mRNA and IFNAR1 mRNA, which leads to IRF3 mRNA translation blockade and accelerated IFNAR1 mRNA degradation. This biological process restricts the antiviral immune response and maintains homeostasis.
pre-mRNA splicing
Alternative splicing of pre-mRNAs plays important roles in cell differentiation and development, and recent studies indicated that most human multiexon genes exhibit alternative splicing [8]. If this process is not highly regulated and accurate, it will lead to mis-splicing events, which may result in proteins with altered function [95].
WTAP interacts with the nuclear splicing factor WT1, forming a splicing complex [96]. Female-specific regulatory protein sex-lethal (SXL) affects sex-specific splicing by regulating the female-specific splicing of transformer (tra) pre-mRNA. Moreover, FL (2)D, the Drosophila homolog of WTAP, forms an RNA-independent complex with SXL [97]. When Fl(2)D was immunodepleted, alternative splicing of transformer pre-mRNA, the target of SXL regulation, was affected [98].
In Drosophila, FL(2)d is distributed throughout the entire eye-antennal imaginal disc and affects retinal development [96] by regulating the alternative splicing of the eye developmental gene Ultrabithorax (Ubx) [99]. In mammalian cells, WTAP and its complex (VIRMA, CBLL1, and ZC3H13) regulate alternative splicing and alternative polyadenylation via inhibitory mechanisms in GC-rich sequences [100].
Furthermore, WTAP was found in complexes related to splicing factors, including Snf, U170k, and the two U2AF subunits U2AF38 and U2AF50 [97]. In conclusion, WTAP is closely related to pre-mRNA splicing, but its specific role in this process remains unclear.
Antiviral responses
WTAP is degraded in virus-infected cells through the K48-linked ubiquitination-proteasome pathway upon activation of type I interferon (IFN-I) signaling. IFN-regulatory factor 3 (IRF3) and interferon-alpha/beta receptor subunit 1 (IFNAR1) are two key components involved in IFN-I signaling that are regulated by WTAP in an m6A-dependent manner. WTAP maintains the expression of IRF3 and IFNAR1 by enhancing IRF3 translation efficiency via m6A modification at its 5’UTR and improving IFNAR1 mRNA stability via m6A modification at its 3’UTR at the same time. Following viral infection, degradation of WTAP blocks IRF3 mRNA translation and accelerates IFNAR1 mRNA degradation, which restricts the antiviral immune response and maintains homeostasis (Fig. 3B) [101].
Expression of WTAP in cancers
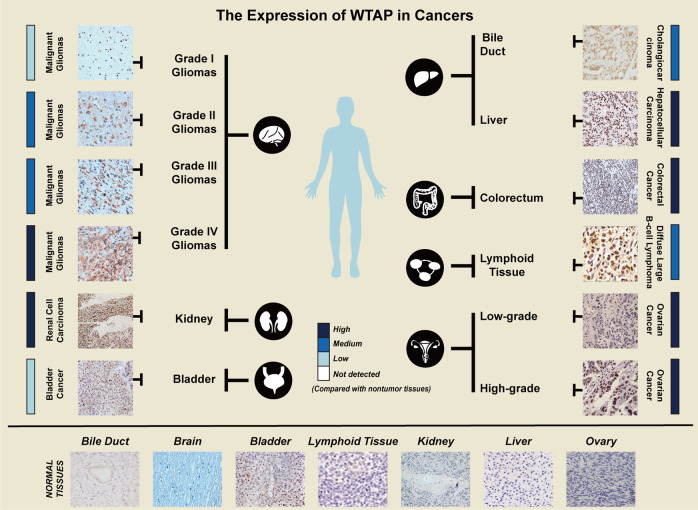
In patient tissue samples, immunohistochemistry results and western blot results have shown that WTAP is highly expressed in dozens of cancers (Fig. 4 Table 2).
Fig. 4. The function of WTAP in biological process.
Immunohistochemistry has been performed in many studies. Strong staining for WTAP was observed in grade IV gliomas, renal cell carcinoma, hepatocellular carcinoma, colorectal cancer, and high-grade ovarian carcinoma, with low staining in adjacent normal tissues.
Table 2.
WTAP expression in different cancers.
| Cancer | Expression | Role | References |
|---|---|---|---|
| Hepatocellular carcinoma | Upregulated | Oncogene | [105] |
| Osteosarcoma tumorigenesis | Upregulated | Oncogene | [106] |
| Gastric cancer | Upregulated | Oncogene | [107] |
| Acute myeloid leukemia | Upregulated | Oncogene | [114, 132] |
| Natural killer/T-cell lymphoma | Upregulated | Oncogene | [123] |
| Cholangiocarcinoma | Upregulated | Oncogene | [126] |
| Diffuse large B-cell lymphoma | Upregulated | Oncogene | [134] |
| Malignant glioma | Upregulated | Oncogene | [135] |
| Colorectal cancer | ? | Tumor Suppressor | [137] |
| Pancreatic ductal adenocarcinoma | Upregulated | Oncogene | [142] |
| Bladder cancer | Upregulated | Oncogene | [143] |
| Renal cell carcinoma | Upregulated | Oncogene | [89] |
| High-grade serous ovarian cancer | Upregulated | Oncogene | [145] |
| Non-small cell lung cancer | ? | Oncogene | [147] |
WTAP as an m6A methyltransferase in cancer
WTAP in hepatocellular carcinoma (HCC)
The overexpression of WTAP was found to be correlated with a poor prognosis in HCC, and WTAP expression promoted proliferation and metastasis in vitro and vivo [102]. ETS1 is a transcriptional activator that is typically regulated by the Ras/Raf/MEK/ERK pathway [103], and it serves as a tumor suppressor in HCC by downregulating the transcription of p21 and p27 [102]. The expression of ETS1 is regulated by HuR, an RNA-binding protein that binds to and stabilizes m6A-modified RNA [104], and WTAP. WTAP was confirmed to increase the m6A modification of ETS1 mRNA and interfere with the interaction between ETS1 mRNA and HuR. Thus, WTAP downregulates p21 and p27 expression to promote HCC proliferation (Fig. 5, Table 3) [102, 105].
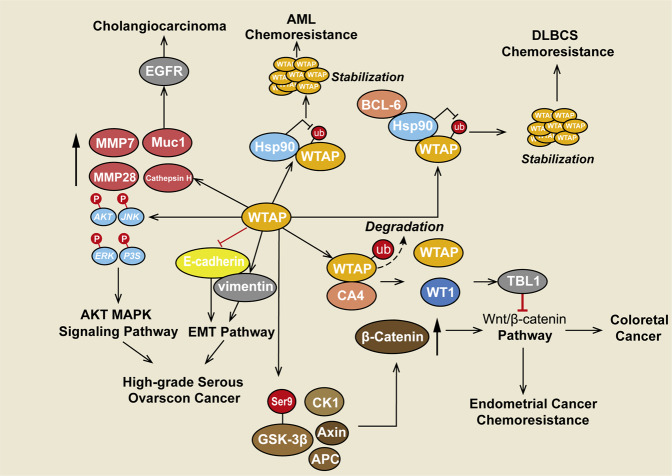
Fig. 5. WTAP serves as a methyltransferase in cancers.
WTAP plays a significant role in RNA methylation by recruiting METTL3/METTL14 to form a complex that binds to target RNAs. In this process, WTAP regulates the differential expression of oncogenes and tumor suppressor genes in an m6A-dependent manner. It enhances the stability of the HK2 and DUSP6 mRNAs, inducing drug resistance in hepatocellular carcinoma, gastric cancer, and NKTCL. Additionally, WTAP induces the degradation of the ETS1, HMBOX1, and c-Myc mRNAs in an m6A-dependent manner, enhancing HCC proliferation and suppressing the invasion and metastasis of osteosarcoma and acute myeloid leukemia.
Table 3.
WTAP as an m6A methyltransferase in cancer.
| Cancer | Biological function | Mechanism | Target | Regulator | References |
|---|---|---|---|---|---|
| Hepatocellular carcinoma | Enhance proliferation, migration | Downregulated the ETS1/p21, p27 axis in an m6A-mediated manner | ETS1/p21, p27 | / | [105] |
| Osteosarcoma tumorigenesis | Enhance proliferation, migration | Downregulated the HMBOX1/PI3K/AKT axis in an m6A-mediated manner | HMBOX1/PI3K/AKT | / | [106] |
| Gastric cancer | Enhance proliferation, migration | WTAP enhanced the stability of HK2 mRNA to regulate the gastric cancer Warburg effect | HK2 | / | [107] |
| Acute myeloid leukemia | Enhance proliferation | Performed m6A on c-Myc mRNA and enhanced its degradation | c-Myc | Cyclins and Hsp90 | [114, 132] |
| Natural killer/T-cell lymphoma | Promote resistance to cisplatin | Enhanced m6A on DUSP6 and stabilized its mRNA | DUSP6 | / | [123] |
WTAP in osteosarcoma
WTAP was found to be highly expressed in osteosarcoma, and it was a significant independent prognostic factor for overall survival [106]. Chen et al. found that upregulation of WTAP reduces the expression of HMBOX1, an oncogene that inhibits osteosarcoma proliferation and metastasis by downregulating the PI3K/AKT pathway. Specifically, WTAP regulated HMBOX1 in an m6A-dependent manner. The m6A modification sites in HMBOX1 are in the 3ʹUTR at 2767 and 3080 nucleotides. However, the reader of HMBOX1 m6A remains unclear (Fig. 5, Table 3) [106].
WTAP in gastric cancer
WTAP was found to be highly expressed in gastric cancer tissues, and its overexpression was correlated with poor prognosis [107]. HK2 plays significant roles in both the Warburg effect, a significant cause of relapse and pathogenesis in gastric cancer [108], and cancer cell immortalization [109]. WTAP promoted the proliferative ability of gastric cancer cells and increased their glycolytic capacity (glucose uptake, lactate production, and extracellular acidification rate) by stabilizing the hexokinase-2 (HK2) mRNA by binding to its 3ʹ-UTR m6A site (Fig. 5, Table 3) [107].
WTAP in hematological malignancies
WTAP was overexpressed in acute myeloid leukemia (AML) patients, and its expression was related to a poor survival rate. MYC is known as a master transcription factor that regulates genes essential for survival, cell proliferation, and metastasis [110, 111] and may act as a downstream regulator of the PI3K/AKT pathway [112, 113]. WTAP downregulates c-Myc expression by increasing the m6A modification of its mRNA [114]. Thus, high WTAP expression predicts poor prognosis in AML, and WTAP plays an epigenetic role in AML (Fig. 4, Table 2) [114].
It was also reported that PIWI-interacting RNAs (piRNAs) are related to diffuse large B-cell lymphoma (DLBCL) [115]. piRNA 30473 was highly expressed in DLBCLs, where it promoted proliferation and induced cell cycle arrest. Mechanistically, piRNA-30473 increased WTAP levels to upregulate the global m6A level. WTAP increased HK2 expression by enhancing its m6A level. The m6A reader IGF2BP2 was found to bind to the 5ʹUTR of HK2 mRNA, leading to its stabilization. HK2 is an essential kinase in glucose metabolism that is associated with tumor cell proliferation by enhancing aerobic glycolysis [116–119]. Overall, the piRNA-30473/WTAP/HK2 axis contributes to tumorigenesis by regulating m6A RNA methylation in DLBCL [115] (Fig. 5, Table 3).
Natural killer/T-cell lymphoma (NKTCL) exhibits high resistance to chemotherapy, which is related to the high expression of ATP binding cassette (ABC) transporter proteins as drug efflux pumps [120, 121]. Multidrug resistance-associated protein 1 (MRP1) and P-glycoprotein (P-gp) are two major proteins in the ABC transporter family that prevent the cellular accumulation of chemotherapy drugs [122]. WTAP was upregulated in NKTCL cell lines. Depletion of WTAP downregulated the expression of MRP1 and P-gp and blocked resistance to cisplatin [122, 123]. WTAP also upregulated the expression of dual-specificity phosphatase 6 (DUSP6) by stabilizing its mRNA by increasing the m6A modification of its transcript, which induced tumor progression and contributed to WTAP-induced drug resistance via the WTAP/m6A/DUSP6 axis (Fig. 5, Table 3) [123].
WTAP in endometrial carcinoma (EC)
WTAP was observed to be upregulated in endometrial cancer cell lines [124, 125]. WTAP activated the nuclear factor‐κB (NF‐κB) pathway by regulating the m6A modification of caveolin‐1 (CAV‐1) mRNA. Reduction of CAV-1 levels by WTAP could enhance the activity of the NF‐κB pathway, contributing to the pathogenesis of EC [124, 125].
Other functions of WTAP in cancer
WTAP in cholangiocarcinoma
WTAP shows a tendency toward overexpression in cholangiocarcinoma tissues. In addition, overexpression of WTAP induces the expression of MMP7, MMP28, cathepsin H, and Muc1 [126]. Notably, these enzymes are all involved in the degradation of the extracellular matrix, which can explain the increased invasion of cholangiocarcinoma cells and WTAP overexpression inside lymph nodes or vessels [127–130]. In addition, Muc1 was shown to regulate EGFR activity [131] to regulate the motility of cancer cells [126]. Therefore, the function of WTAP is an important in cholangiocarcinoma (Fig. 6, Table 4).
Fig. 6. Other functions of WTAP in cancers.
WTAP regulates the differential expression of oncogenes and tumor suppressor genes at the non-posttranscriptional level. WTAP induces the expression of Muc1, which regulates EGFR activity in cholangiocarcinoma. Hsp90 forms a complex with WTAP and stabilizes its protein level to promote chemoresistance in AML. In DLBCL, Hsp90 also stabilizes the WTAP protein, which forms a complex with BCL6. In colorectal cancer, CA4 interacts with WTAP and promotes its degradation in a polyubiquitination-dependent manner so that WT1 is released from the WT1-WTAP complex, resulting in the induction of transducin β-like protein 1 (TBL1) and the degradation of β-catenin, which blocks the Wnt pathway. WTAP was found to facilitate the nuclear translocation of β-catenin and enhance the phosphorylation of GSK3b at Ser9, which induced chemoresistance to cisplatin in endometrial carcinoma by activating the Wnt/β-catenin pathway. Additionally, WTAP was found to regulate the expression of the EMT-related proteins E-cadherin and vimentin. Furthermore, WTAP is involved in the activation of the AKT and MAPK pathways. Overall, WTAP contributes to cell proliferation, apoptosis, invasion, metastasis, and chemo- or radioresistance in different cancers.
Table 4.
Other functions of WTAP in cancer.
| Cancer | Biological function | Mechanism | Target | Regulator | References |
|---|---|---|---|---|---|
| Cholangiocarcinoma | Promote invasion, migration | / | MMP7, MMP28, Cathepsin H, Muc1 | / | [126] |
| Diffuse large B-cell lymphoma | Promote proliferation, counteract etopside-mediated apoptosis | / | / | Cyclins and Hsp90 | [134] |
| Colorectal cancer | / | WTAP supports CA4 in performing its tumor-suppressive function and releasing WT1 from the WTAP-WT1 complex | Carbonic anhydrase IV (CA4) | / | [137] |
| Renal cell carcinoma | Promote invasion proliferation and migration, accelerate cell cycle progression | Binds to the CKD2 transcript to enhance the function of its mRNA | / | / | [89] |
| High-grade serous ovarian cancer | Proliferation, migration and inhibition of apoptosis abilities | Regulates the epithelial-mesenchymal transition (EMT) pathway and AKT and MAPK signaling pathways | E-cadherin, Vimentin, AKT, JNK, ERK and p38 | / | [145] |
| Non-small cell lung cancer | Proliferation, migration and inhibition of apoptosis abilities | / | / | PCGEM1/miR-433–3p axis | [147] |
WTAP in hematological malignancies
In AML, the molecular chaperone Hsp90 interacted with and stabilized WTAP by decreasing its polyubiquitination, which promoted chemoresistance (Fig. 5, Table 3) [132]. This phenomenon was also observed in diffuse large B-cell lymphoma (DLBCL), a common type of non-Hodgkin lymphoma [133, 134] (Fig. 6, Table 4).
WTAP in malignant glioma
WTAP is overexpressed in glioma tissues compared to normal brain tissues. Furthermore, WTAP expression is associated with glioma grade and is an independent prognostic factor for shorter survival in patients with glioma. High expression of WTAP leads to a much lower overall survival rate than low WTAP expression in patients suffering from glioma. Therefore, WTAP may be a novel prognostic marker for glioma (Table 4) [135].
WTAP in endometrial carcinoma (EC)
WTAP also promoted chemoresistance of endometrial carcinoma (EC) cells to cisplatin by facilitating proliferation and repressing apoptosis. Mechanistically, WTAP enhanced the phosphorylation of GSK3β at Ser9, which facilitated the nuclear translocation of β-catenin [136]. Consequently, β-catenin activated the transcription of c-Myc, Survivin, and Bcl-xl to promote chemoresistance to cisplatin [136]. Overall, these results shed light on the strategies to modify the treatment response by altering chemoresistance to cisplatin (Fig. 6 Table 4) [124].
WTAP in colorectal cancer (CRC)
Carbonic anhydrase IV (CA4) is silenced in colorectal cancer (CRC) [137]. It was recently identified as a preferentially methylated gene that is expressed in normal colon tissues [138] and plays a tumor-suppressive function by inhibiting the Wnt/β-catenin signaling pathway [139, 140]. CA4 interacts with WTAP and promotes its polyubiquitination-dependent degradation [137]. WT1 is a negative regulator of the Wnt signaling pathway [141]. WT1 is released from the WT1-WTAP complex by CA4, resulting in the induction of transducing β-like protein 1 (TBL1) and the degradation of β-catenin. A lack of CA4 results in the activation of WNT/β-catenin signaling, which promotes CRC progression [137] (Fig. 6, Table 4).
WTAP in pancreatic ductal adenocarcinoma (PDAC)
The nuclear and cytoplasmic levels of WTAP were much higher in PDAC than in adjacent nontumor tissues [142]. High nuclear levels of WTAP were correlated with a more advanced tumor stage, while cytoplasmic WTAP levels were associated with histological trade and perineural invasion. In addition, high expression of WTAP in the nucleus and cytoplasm differed significantly by sex. Nuclear WTAP levels were identified as an independent prognostic indicator for PDAC and were associated with poor overall survival. Overall, WTAP may be a molecular biomarker in PDAC [142] (Table 4).
WTAP in bladder cancer
Immunohistochemical staining showed that WTAP expression in bladder cancer was significantly higher than that in normal tissues, and high expression of WTAP indicated a poor prognosis [143]. Moreover, both the mRNA and protein levels of WTAP were upregulated in bladder cancer, offering a potential novel approach for the diagnosis and treatment of bladder cancer (Table 4) [143].
WTAP in renal cell carcinoma (RCC)
In RCC, WTAP binds to the transcript of CDK2, a cell cycle-related protein [144], to enhance the stability of its mRNA, thus decreasing the percentage of cells in the G1 phase (Table 4) [89].
WTAP in high-grade serous ovarian cancer (HGSOC)
WTAP expression was correlated with a poor prognosis in high-grade serous ovarian cancer (HGSOC) [145]. Mechanistically, WTAP affected migration by regulating proteins related to the epithelial-mesenchymal transition (EMT) by decreasing E-cadherin expression and increasing vimentin expression. In addition, WTAP promoted the phosphorylation of AKT, JNK, ERK, and p38, indicating that WTAP might be involved in activation of the AKT and MAPK signaling pathways (Fig. 6, Table 4) [145].
It was also reported that family with sequence similarity 76-member A (FAM76A) and HBS1-like translational GTPase (HBS1L) are positively correlated with WTAP according to weighted gene coexpression network analysis (WGCNA), and both were correlated with a poor prognosis [146].
WTAP in non-small cell lung cancer (NSCLC)
High levels of the lncRNA PCGEM1, which is considered to promote cell growth, were detected in NSCLC. PCGEM1 was mostly distributed in the cytoplasm, indicating that it mostly performs its function at the posttranscriptional level. Furthermore, PCGEM1 was found to act as a sponge for miR-433–3p in NSCLC. WTAP is a downstream target of the PCGEM1/miR-433-3p axis. Overall, PCGEM1 plays an important role in NSCLC and can accelerate cancer progression via the miR-433-3p/WTAP axis (Table 4) [147].
WTAP in hepatoblastoma
Hepatoblastoma is a common primary malignant hepatic tumor of infancy and childhood that usually occurs in the first two years of life [148]. Hepatoblastoma susceptibility was correlated with WTAP gene variants. The genotype frequencies of three WTAP single nucleotide polymorphisms (SNPs: rs7766006 G > T, rs9457712 G > A, and rs1853259 A > G) were evaluated in Chinese children, including 313 hepatoblastoma patients and 1446 controls. However, only the rs7766006 GT/TT genotype exhibited a significant association with hepatoblastoma risk. Rs7766006 T was associated with a decrease in WTAP mRNA levels. Thus, WTAP SNPs potentially play a role in hepatoblastoma via genetic modification [149].
Future prospects
WTAP was first reported to be a splicing factor. In the following years, its biological functions have gradually been uncovered, including functions in m6A modification, embryo development, cell cycle progression and differentiation, pre-mRNA splicing, and antiviral responses. With the development of techniques for detecting m6A modification, WTAP was revealed to be a part of the MTC and to participate in m6A modification with both METTL3 and METTL14 and other methyltransferases. In human umbilical vein endothelial cells, WTAP promotes G2/M transition, while in smooth muscle cells, overexpression of WTAP prevents WT1 from binding to the Bcl-2 promoter, thereby downregulating Bcl-2 and activating apoptosis. In renal cell carcinoma, keratinocytes, and psoriasis, WTAP regulates the G1/S transition and G2/M transition by stabilizing specific mRNAs. Thus, WTAP may be a potential biomarker for changes in cell proliferation and differentiation. WTAP is also associated with chemoresistance in hematological malignancies and endometrial carcinoma by upregulating the expression of MRP1 and P-gp and enhancing the phosphorylation of GSK3β at Ser9. These results shed light on the potential of targeting WTAP for the prevention of chemoresistance to cisplatin. During metabolism, WTAP can stabilize the HK2 mRNA, which is associated with aerobic glycolysis and the Warburg effect in diffuse large B-cell lymphoma. The therapeutic schedule can be developed according to this metabolic phenomenon. High expression of WTAP was confirmed in malignant gliomas, renal cell carcinoma, hepatocellular carcinoma, colorectal cancer, and ovarian cancer, which is related to progression and poor prognosis (Fig. 6, Table 4), suggesting that WTAP might be a biomarker for the above cancers. In liver cancer, WTAP was observed to increase the m6A level of the ETS1 mRNA, thereby facilitating cancer progression. Similarly, WTAP was found to induce the proliferation and metastasis of osteosarcoma by regulating HMBOX1 m6A modification. In gastric cancer, WTAP enhanced HK2 mRNA stability through m6A modification. In natural killer/T-cell lymphoma, WTAP upregulated DUSP6 expression through m6A modification, inducing drug resistance. In acute myeloid leukemia, WTAP downregulated c-Myc expression by increasing the m6A modification of its mRNA, making cells resistant to chemotherapy drugs. These cases indicated that the role of WTAP as a methyltransferase is vital in cancer progression. Although no small-molecule inhibitors of RNA methyltransferases and WTAP have been discovered, FTO demethylation inhibitors have been identified. Rhein can bind the FTO catalytic domain to suppress m6A demethylation [150]. CHTB, N-CDPCB and meclofenamic acid 2 (MA2) have been revealed to be FTO inhibitors through structure-based virtual screening and biochemical analyses [151, 152]. R-2-hydroxyglutarate (R-2HG) inhibits FTO activity and increases global m6A modification, which has been tested in vitro and in mice [153]. These effects suggest that WTAP-targeted inhibitors may be developed in the future and that a deeper understanding of m6A modification is warranted.
Conclusion
At present, our understanding of WTAP is insufficient due to a lack of further experiments and additional samples. m6A has gradually become a significant focus of cancer research, but the role of WTAP in this process is still at an early stage. Furthermore, the localization of WTAP in nuclear speckles and the formation of a complex with METTL3 and METTL14 need to be further investigated, since this knowledge may be useful for understanding the role of m6A modification in cancer biology. In conclusion, many studies have revealed WTAP as a potential biomarker for predicting cancer progression, since it participates in alternative splicing, cell cycle regulation and methylation. Thus, efforts should be made to develop the potential of WTAP for therapies targeting tumorigenesis and tumor development.
Author contributions
QH and JM collected the related papers and drafted the manuscript. BZ and ZL participated in the design of the review. XC initiated the study and revised the manuscript. All authors read and approved the final manuscript.
Funding
This work was supported by The National Natural Science Foundation of China (No. 81874189 to BZ).
Data availability
The materials that support the conclusion of this review have been included within the article.
Competing interests
The authors declare no competing interests.
Footnotes
Edited by Ivano Amelio
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
These authors contributed equally: Qibo Huang, Jie Mo.
Contributor Information
Zhibin Liao, Email: zhibliao@hust.edu.cn.
Xiaoping Chen, Email: chenxpchenxp@163.com.
Bixiang Zhang, Email: bixiangzhang@hust.edu.cn.
References
- 1.Mann DA. Epigenetics in liver disease. Hepatology. 2014;60:1418–25. doi: 10.1002/hep.27131. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Sapienza C, Issa JP. Diet, nutrition, and cancer epigenetics. Annu Rev Nutr. 2016;36:665–81. doi: 10.1146/annurev-nutr-121415-112634. [DOI] [PubMed] [Google Scholar]
- 3.Cedar H, Bergman Y. Programming of DNA methylation patterns. Annu Rev Biochem. 2012;81:97–117. doi: 10.1146/annurev-biochem-052610-091920. [DOI] [PubMed] [Google Scholar]
- 4.Black JC, Van Rechem C, Whetstine JR. Histone lysine methylation dynamics: establishment, regulation, and biological impact. Mol Cell. 2012;48:491–507. doi: 10.1016/j.molcel.2012.11.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Clapier CR, Cairns BR. The biology of chromatin remodeling complexes. Annu Rev Biochem. 2009;78:273–304. doi: 10.1146/annurev.biochem.77.062706.153223. [DOI] [PubMed] [Google Scholar]
- 6.Kemper JK. Regulation of FXR transcriptional activity in health and disease: Emerging roles of FXR cofactors and post-translational modifications. Biochim Biophys Acta. 2011;1812:842–50. doi: 10.1016/j.bbadis.2010.11.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Mercer TR DM, Mattick JS. Long non-coding RNAs: insights into functions. Nat Rev Genet. 2009;10:155–9. doi: 10.1038/nrg2521. [DOI] [PubMed] [Google Scholar]
- 8.Villanueva L, Alvarez-Errico D, Esteller M. The contribution of epigenetics to cancer immunotherapy. Trends Immunol. 2020;41:676–91. doi: 10.1016/j.it.2020.06.002. [DOI] [PubMed] [Google Scholar]
- 9.Li X, Xiong X, Zhang M, Wang K, Chen Y, Zhou J, et al. Base-resolution mapping reveals distinct m(1)A methylome in nuclear- and mitochondrial-encoded transcripts. Mol Cell. 2017;68:993–1005.e1009. doi: 10.1016/j.molcel.2017.10.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Motorin Y, Lyko F, Helm M. 5-methylcytosine in RNA: detection, enzymatic formation and biological functions. Nucleic Acids Res. 2010;38:1415–30. doi: 10.1093/nar/gkp1117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Xia T, Wu X, Cao M, Zhang P, Shi G, Zhang J, et al. The RNA m6A methyltransferase METTL3 promotes pancreatic cancer cell proliferation and invasion. Pathol Res Pr. 2019;215:152666. doi: 10.1016/j.prp.2019.152666. [DOI] [PubMed] [Google Scholar]
- 12.Xie S, Chen W, Chen K, Chang Y, Yang F, Lin A, et al. Emerging roles of RNA methylation in gastrointestinal cancers. Cancer Cell Int. 2020;20:585. doi: 10.1186/s12935-020-01679-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Desrosiers R FK, Rottman F. Identifcation of methylated nucleo sides in messenger RNA from Novikof hepatoma cells. Proc Natl Acad Sci USA. 1974;71:3971–5. doi: 10.1073/pnas.71.10.3971. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Meyer KD, Jaffrey SR. Rethinking m(6)A Readers, writers, and erasers. Annu Rev Cell Dev Biol. 2017;33:319–42. doi: 10.1146/annurev-cellbio-100616-060758. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Lee M, Kim B, Kim VN. Emerging roles of RNA modification: m(6)A and U-tail. Cell. 2014;158:980–7. doi: 10.1016/j.cell.2014.08.005. [DOI] [PubMed] [Google Scholar]
- 16.He L, Li H, Wu A, Peng Y, Shu G, Yin G. Functions of N6-methyladenosine and its role in cancer. Mol Cancer. 2019;18:176. doi: 10.1186/s12943-019-1109-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Ma JZ, Yang F, Zhou CC, Liu F, Yuan JH, Wang F, et al. METTL14 suppresses the metastatic potential of hepatocellular carcinoma by modulating N(6) -methyladenosine-dependent primary MicroRNA processing. Hepatology. 2017;65:529–43. doi: 10.1002/hep.28885. [DOI] [PubMed] [Google Scholar]
- 18.Han J, Wang JZ, Yang X, Yu H, Zhou R, Lu HC, et al. METTL3 promote tumor proliferation of bladder cancer by accelerating pri-miR221/222 maturation in m6A-dependent manner. Mol Cancer. 2019;18:110. doi: 10.1186/s12943-019-1036-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Berulava T, Buchholz E, Elerdashvili V, Pena T, Islam MR, Lbik D, et al. Changes in m6A RNA methylation contribute to heart failure progression by modulating translation. Eur J Heart Fail. 2020;22:54–66. doi: 10.1002/ejhf.1672. [DOI] [PubMed] [Google Scholar]
- 20.Chen Y, Wang J, Xu D, Xiang Z, Ding J, Yang X, et al. m(6)A mRNA methylation regulates testosterone synthesis through modulating autophagy in Leydig cells. Autophagy. 2021;17:457–75. doi: 10.1080/15548627.2020.1720431. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Zhou B, Liu C, Xu L, Yuan Y, Zhao J, Zhao W, et al. N(6) -Methyladenosine Reader Protein YT521-B Homology Domain-Containing 2 Suppresses Liver Steatosis by Regulation of mRNA Stability of Lipogenic Genes. Hepatology. 2021;73:91–103. doi: 10.1002/hep.31220. [DOI] [PubMed] [Google Scholar]
- 22.Pan Y, Ma P, Liu Y, Li W, Shu Y. Multiple functions of m(6)A RNA methylation in cancer. J Hematol Oncol. 2018;11:48. doi: 10.1186/s13045-018-0590-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Cui Q, Shi H, Ye P, Li L, Qu Q, Sun G, et al. m(6)A RNA methylation regulates the self-renewal and tumorigenesis of glioblastoma stem cells. Cell Rep. 2017;18:2622–34. doi: 10.1016/j.celrep.2017.02.059. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Jiang X, Liu B, Nie Z, Duan L, Xiong Q, Jin Z, et al. The role of m6A modification in the biological functions and diseases. Signal Transduct Target Ther. 2021;6:74. doi: 10.1038/s41392-020-00450-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Ping XL, Sun BF, Wang L, Xiao W, Yang X, Wang WJ, et al. Mammalian WTAP is a regulatory subunit of the RNA N6-methyladenosine methyltransferase. Cell Res. 2014;24:177–89. doi: 10.1038/cr.2014.3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Liu J, Yue Y, Han D, Wang X, Fu Y, Zhang L, et al. A METTL3-METTL14 complex mediates mammalian nuclear RNA N6-adenosine methylation. Nat Chem Biol. 2014;10:93–95. doi: 10.1038/nchembio.1432. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Li S, Kuo HD, Yin R, Wu R, Liu X, Wang L, et al. Epigenetics/epigenomics of triterpenoids in cancer prevention and in health. Biochem Pharm. 2020;175:113890. doi: 10.1016/j.bcp.2020.113890. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Lin YT, Wu KJ. Epigenetic regulation of epithelial-mesenchymal transition: focusing on hypoxia and TGF-beta signaling. J Biomed Sci. 2020;27:39. doi: 10.1186/s12929-020-00632-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Maity A, Das B. N6-methyladenosine modification in mRNA: machinery, function and implications for health and diseases. FEBS J. 2016;283:1607–30. doi: 10.1111/febs.13614. [DOI] [PubMed] [Google Scholar]
- 30.Dominissini D, Moshitch-Moshkovitz S, Schwartz S, Salmon-Divon M, Ungar L, Osenberg S, et al. Topology of the human and mouse m6A RNA methylomes revealed by m6A-seq. Nature. 2012;485:201–6. doi: 10.1038/nature11112. [DOI] [PubMed] [Google Scholar]
- 31.Meyer KD, Saletore Y, Zumbo P, Elemento O, Mason CE, Jaffrey SR. Comprehensive analysis of mRNA methylation reveals enrichment in 3’ UTRs and near stop codons. Cell. 2012;149:1635–46. doi: 10.1016/j.cell.2012.05.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Dai D, Wang H, Zhu L, Jin H, Wang X. N6-methyladenosine links RNA metabolism to cancer progression. Cell Death Dis. 2018;9:124. doi: 10.1038/s41419-017-0129-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Wang X, Huang J, Zou T, Yin P. Human m(6)A writers: Two subunits, 2 roles. RNA Biol. 2017;14:300–4. doi: 10.1080/15476286.2017.1282025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Wu R, Jiang D, Wang Y, Wang X. N (6)-Methyladenosine (m(6)A) methylation in mRNA with a dynamic and reversible epigenetic modification. Mol Biotechnol. 2016;58:450–9. doi: 10.1007/s12033-016-9947-9. [DOI] [PubMed] [Google Scholar]
- 35.Xu K, Yang Y, Feng GH, Sun BF, Chen JQ, Li YF, et al. Mettl3-mediated m(6)A regulates spermatogonial differentiation and meiosis initiation. Cell Res. 2017;27:1100–14. doi: 10.1038/cr.2017.100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Zhang C, Chen L, Peng D, Jiang A, He Y, Zeng Y, et al. METTL3 and N6-methyladenosine promote homologous recombination-mediated repair of DSBs by modulating DNA-RNA hybrid accumulation. Mol Cell. 2020;79:425–42.e427. doi: 10.1016/j.molcel.2020.06.017. [DOI] [PubMed] [Google Scholar]
- 37.Xiang Y, Laurent B, Hsu C-H, Nachtergaele S, Lu Z, Sheng W, et al. RNA m6A methylation regulates the ultraviolet-induced DNA damage response. Nature. 2017;543:573–6. doi: 10.1038/nature21671. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Visvanathan A, Patil V, Arora A, Hegde AS, Arivazhagan A, Santosh V, et al. Essential role of METTL3-mediated m(6)A modification in glioma stem-like cells maintenance and radioresistance. Oncogene. 2018;37:522–33. doi: 10.1038/onc.2017.351. [DOI] [PubMed] [Google Scholar]
- 39.Wang X, Feng J, Xue Y, Guan Z, Zhang D, Liu Z, et al. Structural basis of N(6)-adenosine methylation by the METTL3-METTL14 complex. Nature. 2016;534:575–8. doi: 10.1038/nature18298. [DOI] [PubMed] [Google Scholar]
- 40.Zhou H, Yin K, Zhang Y, Tian J, Wang S. The RNA m6A writer METTL14 in cancers: Roles, structures, and applications. Biochim Biophys Acta Rev Cancer. 2021;1876:188609. doi: 10.1016/j.bbcan.2021.188609. [DOI] [PubMed] [Google Scholar]
- 41.Gu C, Wang Z, Zhou N, Li G, Kou Y, Luo Y, et al. Mettl14 inhibits bladder TIC self-renewal and bladder tumorigenesis through N(6)-methyladenosine of Notch1. Mol Cancer. 2019;18:168. doi: 10.1186/s12943-019-1084-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Yang X, Zhang S, He C, Xue P, Zhang L, He Z, et al. METTL14 suppresses proliferation and metastasis of colorectal cancer by down-regulating oncogenic long non-coding RNA XIST. Mol Cancer. 2020;19:46. doi: 10.1186/s12943-020-1146-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Du L, Li Y, Kang M, Feng M, Ren Y, Dai H, et al. USP48 is upregulated by Mettl14 to attenuate hepatocellular carcinoma via regulating SIRT6 stabilization. Cancer Res. 2021;81:3822–34. doi: 10.1158/0008-5472.CAN-20-4163. [DOI] [PubMed] [Google Scholar]
- 44.Chen X, Xu M, Xu X, Zeng K, Liu X, Pan B, et al. METTL14-mediated N6-methyladenosine modification of SOX4 mRNA inhibits tumor metastasis in colorectal cancer. Mol Cancer. 2020;19:106. doi: 10.1186/s12943-020-01220-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Wang X, Tian L, Li Y, Wang J, Yan B, Yang L, et al. RBM15 facilitates laryngeal squamous cell carcinoma progression by regulating TMBIM6 stability through IGF2BP3 dependent. J Exp Clin Cancer Res. 2021;40:80. doi: 10.1186/s13046-021-01871-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Patil DP, Chen CK, Pickering BF, Chow A, Jackson C, Guttman M, et al. m(6)A RNA methylation promotes XIST-mediated transcriptional repression. Nature. 2016;537:369–73. doi: 10.1038/nature19342. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Knuckles P, Lence T, Haussmann IU, Jacob D, Kreim N, Carl SH, et al. Zc3h13/Flacc is required for adenosine methylation by bridging the mRNA-binding factor Rbm15/Spenito to the m(6)A machinery component Wtap/Fl(2)d. Genes Dev. 2018;32:415–29. doi: 10.1101/gad.309146.117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Wen J, Lv R, Ma H, Shen H, He C, Wang J, et al. Zc3h13 regulates nuclear RNA m(6)A methylation and mouse embryonic stem cell self-renewal. Mol Cell. 2018;69:1028–38.e1026. doi: 10.1016/j.molcel.2018.02.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Pendleton KE, Chen B, Liu K, Hunter OV, Xie Y, Tu BP, et al. The U6 snRNA m(6)A methyltransferase METTL16 regulates SAM synthetase intron retention. Cell. 2017;169:824–35.e814. doi: 10.1016/j.cell.2017.05.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Rong B, Zhang Q, Wan J, Xing S, Dai R, Li Y, et al. Ribosome 18Sm(6)A methyltransferase METTL5 promotes translation initiation and breast cancer cell growth. Cell Rep. 2020;33:108544. doi: 10.1016/j.celrep.2020.108544. [DOI] [PubMed] [Google Scholar]
- 51.Yue Y, Liu J, Cui X, Cao J, Luo G, Zhang Z, et al. VIRMA mediates preferential m(6)A mRNA methylation in 3’UTR and near stop codon and associates with alternative polyadenylation. Cell Disco. 2018;4:10. doi: 10.1038/s41421-018-0019-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Ma H, Wang X, Cai J, Dai Q, Natchiar SK, Lv R, et al. N(6-)Methyladenosine methyltransferase ZCCHC4 mediates ribosomal RNA methylation. Nat Chem Biol. 2019;15:88–94. doi: 10.1038/s41589-018-0184-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Richard EM, Polla DL, Assir MZ, Contreras M, Shahzad M, Khan AA, et al. Bi-allelic variants in METTL5 cause autosomal-recessive intellectual disability and microcephaly. Am J Hum Genet. 2019;105:869–78. doi: 10.1016/j.ajhg.2019.09.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Liu, J, Gao, M, Xu, S, Chen, Y, Wu, K, Liu, H et al. YTHDF2/3 Are required for somatic reprogramming through different RNA deadenylation pathways. Cell Rep. 2020;32. 10.1016/j.celrep.2020.108120. [DOI] [PubMed]
- 55.Xiao W, Adhikari S, Dahal U, Chen YS, Hao YJ, Sun BF, et al. Nuclear m(6)A reader YTHDC1 regulates mRNA splicing. Mol Cell. 2016;61:507–19. doi: 10.1016/j.molcel.2016.01.012. [DOI] [PubMed] [Google Scholar]
- 56.Hsu PJ, Zhu Y, Ma H, Guo Y, Shi X, Liu Y, et al. Ythdc2 is an N(6)-methyladenosine binding protein that regulates mammalian spermatogenesis. Cell Res. 2017;27:1115–27. doi: 10.1038/cr.2017.99. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Liu T, Wei Q, Jin J, Luo Q, Liu Y, Yang Y, et al. The m6A reader YTHDF1 promotes ovarian cancer progression via augmenting EIF3C translation. Nucleic Acids Res. 2020;48:3816–31. doi: 10.1093/nar/gkaa048. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Sheng H, Li Z, Su S, Sun W, Zhang X, Li L, et al. YTH domain family 2 promotes lung cancer cell growth by facilitating 6-phosphogluconate dehydrogenase mRNA translation. Carcinogenesis. 2020;41:541–50. doi: 10.1093/carcin/bgz152. [DOI] [PubMed] [Google Scholar]
- 59.Chen X, Zhou X, Wang X. m(6)A binding protein YTHDF2 in cancer. Exp Hematol Oncol. 2022;11:21. doi: 10.1186/s40164-022-00269-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Yan, W, Kasowitz, SD, Ma, J, Anderson, SJ, Leu, NA, Xu, Y et al. Nuclear m6A reader YTHDC1 regulates alternative polyadenylation and splicing during mouse oocyte development. PLOS Genetics. 2018;14, 10.1371/journal.pgen.1007412. [DOI] [PMC free article] [PubMed]
- 61.Roundtree IA, Luo GZ, Zhang Z, Wang X, Zhou T, Cui Y, et al. YTHDC1 mediates nuclear export of N6-methyladenosine methylated mRNAs. Elife. 2017;6:e31311. doi: 10.7554/eLife.31311. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Wang X, Zhao BS, Roundtree IA, Lu Z, Han D, Ma H, et al. N(6)-methyladenosine modulates messenger RNA translation efficiency. Cell. 2015;161:1388–99. doi: 10.1016/j.cell.2015.05.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Meyer KD, Patil DP, Zhou J, Zinoviev A, Skabkin MA, Elemento O, et al. 5’ UTR m(6)A promotes cap-independent translation. Cell. 2015;163:999–1010. doi: 10.1016/j.cell.2015.10.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Huang H, Weng H, Sun W, Qin X, Shi H, Wu H, et al. Recognition of RNA N(6)-methyladenosine by IGF2BP proteins enhances mRNA stability and translation. Nat Cell Biol. 2018;20:285–95. doi: 10.1038/s41556-018-0045-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Muller S, Glass M, Singh AK, Haase J, Bley N, Fuchs T, et al. IGF2BP1 promotes SRF-dependent transcription in cancer in a m6A- and miRNA-dependent manner. Nucleic Acids Res. 2019;47:375–90. doi: 10.1093/nar/gky1012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Lan Q, Liu PY, Bell JL, Wang JY, Huttelmaier S, Zhang XD, et al. The emerging roles of RNA m(6)A methylation and demethylation as critical regulators of tumorigenesis, drug sensitivity, and resistance. Cancer Res. 2021;81:3431–40. doi: 10.1158/0008-5472.CAN-20-4107. [DOI] [PubMed] [Google Scholar]
- 67.Hu X, Peng WX, Zhou H, Jiang J, Zhou X, Huang D, et al. IGF2BP2 regulates DANCR by serving as an N6-methyladenosine reader. Cell Death Differ. 2020;27:1782–94. doi: 10.1038/s41418-019-0461-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Alarcon CR, Goodarzi H, Lee H, Liu X, Tavazoie S, Tavazoie SF. HNRNPA2B1 is a mediator of m(6)A-dependent nuclear RNA processing events. Cell. 2015;162:1299–308. doi: 10.1016/j.cell.2015.08.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Wang LC, Chen SH, Shen XL, Li DC, Liu HY, Ji YL, et al. M6A RNA methylation regulator HNRNPC contributes to tumorigenesis and predicts prognosis in glioblastoma multiforme. Front Oncol. 2020;10:536875. doi: 10.3389/fonc.2020.536875. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Wang T, Kong S, Tao M, Ju S. The potential role of RNA N6-methyladenosine in Cancer progression. Mol Cancer. 2020;19:88. doi: 10.1186/s12943-020-01204-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Mathiyalagan P, Adamiak M, Mayourian J, Sassi Y, Liang Y, Agarwal N, et al. FTO-dependent N(6)-methyladenosine regulates cardiac function during remodeling and repair. Circulation. 2019;139:518–32. doi: 10.1161/CIRCULATIONAHA.118.033794. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Su R, Dong L, Li C, Nachtergaele S, Wunderlich M, Qing Y, et al. R-2HG exhibits anti-tumor activity by targeting FTO/m(6)A/MYC/CEBPA signaling. Cell. 2018;172:90–105.e123. doi: 10.1016/j.cell.2017.11.031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Zhao X, Yang Y, Sun BF, Shi Y, Yang X, Xiao W, et al. FTO-dependent demethylation of N6-methyladenosine regulates mRNA splicing and is required for adipogenesis. Cell Res. 2014;24:1403–19. doi: 10.1038/cr.2014.151. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Zhang S, Zhao BS, Zhou A, Lin K, Zheng S, Lu Z, et al. m(6)A demethylase ALKBH5 maintains tumorigenicity of glioblastoma stem-like cells by sustaining FOXM1 expression and cell proliferation program. Cancer Cell. 2017;31:591–606 e596. doi: 10.1016/j.ccell.2017.02.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Zheng G, Dahl JA, Niu Y, Fedorcsak P, Huang CM, Li CJ, et al. ALKBH5 is a mammalian RNA demethylase that impacts RNA metabolism and mouse fertility. Mol Cell. 2013;49:18–29. doi: 10.1016/j.molcel.2012.10.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Guo X, Li K, Jiang W, Hu Y, Xiao W, Huang Y, et al. RNA demethylase ALKBH5 prevents pancreatic cancer progression by posttranscriptional activation of PER1 in an m6A-YTHDF2-dependent manner. Mol Cancer. 2020;19:91. doi: 10.1186/s12943-020-01158-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Zaccara S, Ries RJ, Jaffrey SR. Reading, writing and erasing mRNA methylation. Nat Rev Mol Cell Biol. 2019;20:608–24. doi: 10.1038/s41580-019-0168-5. [DOI] [PubMed] [Google Scholar]
- 78.Little NA HN, Davies RC. Identification of WTAP, a novel Wilms’ tumour 1-associating protein. Hum Mol Genet. 2000;9:2231–9. doi: 10.1093/oxfordjournals.hmg.a018914. [DOI] [PubMed] [Google Scholar]
- 79.Small TW, Penalva LO, Pickering JG. Vascular biology and the sex of flies: regulation of vascular smooth muscle cell proliferation by wilms’ tumor 1-associating protein. Trends Cardiovasc Med. 2007;17:230–4. doi: 10.1016/j.tcm.2007.08.002. [DOI] [PubMed] [Google Scholar]
- 80.Horiuchi K, Kawamura T, Iwanari H, Ohashi R, Naito M, Kodama T, et al. Identification of Wilms’ tumor 1-associating protein complex and its role in alternative splicing and the cell cycle. J Biol Chem. 2013;288:33292–302. doi: 10.1074/jbc.M113.500397. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Scholler E WF, Treiber T, Ringle S, Treiber N, Flatley A, et al. Interactions, localization, and phosphorylation of the m (6) a generating METTL3-METTL14-WTAP complex. RNA. 2018;24:499–512. doi: 10.1261/rna.was. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Horiuchi K, Umetani M, Minami T, Okayama H, Takada S, Yamamoto M, et al. Wilms’ tumor 1-associating protein regulates G2/M transition through stabilization of cyclin A2 mRNA. Proc Natl Acad Sci USA. 2006;103:17278–83. doi: 10.1073/pnas.0608357103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Fukusumi Y, Naruse C, Asano M. Wtap is required for differentiation of endoderm and mesoderm in the mouse embryo. Dev Dyn. 2008;237:618–29. doi: 10.1002/dvdy.21444. [DOI] [PubMed] [Google Scholar]
- 84.Hao, J, Huang, S, Wang, D, Jin, Y, Zhang, M, Zhang, J et al. Loss of WTAP impairs early parthenogenetic embryo development. Animals (Basel). 2021;11. 10.3390/ani11061675. [DOI] [PMC free article] [PubMed]
- 85.Liu L, Michowski W, Kolodziejczyk A, Sicinski P. The cell cycle in stem cell proliferation, pluripotency and differentiation. Nat Cell Biol. 2019;21:1060–7. doi: 10.1038/s41556-019-0384-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.O’Farrell CFLAPH. The roles of deosophila cyclins A and B in mitotic control. Cell. 1990;61:535–47. doi: 10.1016/0092-8674(90)90535-m. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Michele Pagano RP, Verde Fulvia, Ansorge Wilhelm, Draetta Giulio. Cyclin A is required at two points in the human cell cycle. EMBO J. 1992;11:961–71. doi: 10.1002/j.1460-2075.1992.tb05135.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Hume S, Dianov GL, Ramadan K. A unified model for the G1/S cell cycle transition. Nucleic Acids Res. 2020;48:12483–501. doi: 10.1093/nar/gkaa1002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Tang J, Wang F, Cheng G, Si S, Sun X, Han J, et al. Wilms’ tumor 1-associating protein promotes renal cell carcinoma proliferation by regulating CDK2 mRNA stability. J Exp Clin Cancer Res. 2018;37:40. doi: 10.1186/s13046-018-0706-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Kong Y, Wu R, Zhang S, Zhao M, Wu H, Lu Q, et al. Wilms’ tumor 1-associating protein contributes to psoriasis by promoting keratinocytes proliferation via regulating cyclinA2 and CDK2. Int Immunopharmacol. 2020;88:106918. doi: 10.1016/j.intimp.2020.106918. [DOI] [PubMed] [Google Scholar]
- 91.Owens GK, Brian R Wamhoff MSK. Molecular regulation of vascular smooth muscle cell differentiation in development and disease. Physiol Rev. 2004;84:767–801. doi: 10.1152/physrev.00041.2003. [DOI] [PubMed] [Google Scholar]
- 92.Shin HS, Lee HJ, Nishida M, Lee MS, Tamura R, Yamashita S, et al. Betacellulin and amphiregulin induce upregulation of cyclin D1 and DNA synthesis activity through differential signaling pathways in vascular smooth muscle cells. Circ Res. 2003;93:302–10. doi: 10.1161/01.RES.0000086803.64109.9E. [DOI] [PubMed] [Google Scholar]
- 93.Mayo MW, Wang CY, Drouin SS, Madrid LV, Marshall AF, Reed JC, Weissman BE, Baldwin AS. A WT1 modulates apoptosis by transcriptionally upregulating the bcl-2 proto-oncogene. The EMBO J. 1999;18:3990–4003. doi: 10.1093/emboj/18.14.3990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Small TW, Bolender Z, Bueno C, O’Neil C, Nong Z, Rushlow W, et al. Wilms’ tumor 1-associating protein regulates the proliferation of vascular smooth muscle cells. Circ Res. 2006;99:1338–46. doi: 10.1161/01.RES.0000252289.79841.d3. [DOI] [PubMed] [Google Scholar]
- 95.Montes M, Sanford BL, Comiskey DF, Chandler DS. RNA splicing and disease: animal models to therapies. Trends Genet. 2019;35:68–87. doi: 10.1016/j.tig.2018.10.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Anderson AM, Weasner BP, Weasner BM, Kumar JP. The Drosophila Wilms Tumor 1-Associating Protein (WTAP) homolog is required for eye development. Dev Biol. 2014;390:170–80. doi: 10.1016/j.ydbio.2014.03.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Penn JK, Graham P, Deshpande G, Calhoun G, Chaouki AS, Salz HK, et al. Functioning of the Drosophila Wilms’-tumor-1-associated protein homolog, Fl(2)d, in Sex-lethal-dependent alternative splicing. Genetics. 2008;178:737–48. doi: 10.1534/genetics.107.081679. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Ortega A, Niksic M, Bachi A, Wilm M, Sanchez L, Hastie N, et al. Biochemical function of female-lethal (2)D/Wilms’ tumor suppressor-1-associated proteins in alternative pre-mRNA splicing. J Biol Chem. 2003;278:3040–7. doi: 10.1074/jbc.M210737200. [DOI] [PubMed] [Google Scholar]
- 99.Burnette JM, Hatton AR, Lopez AJ. Trans-acting factors required for inclusion of regulated exons in the ultrabithorax mRNAs of Drosophila melanogaster. Genetics Society of America. 1999;151:1517–29. doi: 10.1093/genetics/151.4.1517. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Horiuchi K, Kawamura T, Hamakubo T. Wilms’ tumor 1-associating protein complex regulates alternative splicing and polyadenylation at potential G-quadruplex-forming splice site sequences. J Biol Chem. 2021;297:101248. doi: 10.1016/j.jbc.2021.101248. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Ge Y, Ling T, Wang Y, Jia X, Xie X, Chen R et al. Degradation of WTAP blocks antiviral responses by reducing the m(6) A levels of IRF3 and IFNAR1 mRNA. EMBO Rep. 2021:e52101, 10.15252/embr.202052101. [DOI] [PMC free article] [PubMed]
- 102.Coqueret O. New roles for p21 and p27 cell-cycle inhibitors: a function for each cell compartment? TRENDS Cell Biol. 2003;13:65–70. doi: 10.1016/s0962-8924(02)00043-0. [DOI] [PubMed] [Google Scholar]
- 103.Wasylyk C, Bradford AP, Gutierrez-Hartmann A, Wasylyk B. Conserved mechanisms of Ras regulation of evolutionary related transcription factors, Ets1 and Pointed P2. Oncogene. 1997;14:899–913. doi: 10.1038/sj.onc.1200914. [DOI] [PubMed] [Google Scholar]
- 104.Wang Y, Li Y, Toth JI, Petroski MD, Zhang Z, Zhao JC. N6-methyladenosine modification destabilizes developmental regulators in embryonic stem cells. Nat Cell Biol. 2014;16:191–8. doi: 10.1038/ncb2902. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Chen Y, Peng C, Chen J, Chen D, Yang B, He B, et al. WTAP facilitates progression of hepatocellular carcinoma via m6A-HuR-dependent epigenetic silencing of ETS1. Mol Cancer. 2019;18:127. doi: 10.1186/s12943-019-1053-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Chen S, Li Y, Zhi S, Ding Z, Wang W, Peng Y, et al. WTAP promotes osteosarcoma tumorigenesis by repressing HMBOX1 expression in an m(6)A-dependent manner. Cell Death Dis. 2020;11:659. doi: 10.1038/s41419-020-02847-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Yu H, Zhao K, Zeng H, Li Z, Chen K, Zhang Z, et al. N(6)-methyladenosine (m(6)A) methyltransferase WTAP accelerates the Warburg effect of gastric cancer through regulating HK2 stability. Biomed Pharmacother. 2021;133:111075. doi: 10.1016/j.biopha.2020.111075. [DOI] [PubMed] [Google Scholar]
- 108.Liberti MV, Locasale JW. The Warburg effect: how does it benefit cancer cells? Trends Biochem Sci. 2016;41:211–8. doi: 10.1016/j.tibs.2015.12.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Lis P, Dylag M, Niedzwiecka K, Ko YH, Pedersen PL, Goffeau A et al. The HK2 Dependent “Warburg Effect” and mitochondrial oxidative phosphorylation in cancer: targets for effective therapy with 3-bromopyruvate. Molecules. 2016;21 10.3390/molecules21121730 [DOI] [PMC free article] [PubMed]
- 110.Dang CV. MYC on the path to cancer. Cell. 2012;149:22–35. doi: 10.1016/j.cell.2012.03.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Meyer N,PL. Reflecting on 25 years with MYC. Nat Rev Cancer. 2008;8:976–90. doi: 10.1038/nrc2231. [DOI] [PubMed] [Google Scholar]
- 112.Zhang F, Li K, Yao X, Wang H, Li W, Wu J, et al. A miR-567-PIK3AP1-PI3K/AKT-c-Myc feedback loop regulates tumour growth and chemoresistance in gastric cancer. EBioMedicine. 2019;44:311–21. doi: 10.1016/j.ebiom.2019.05.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Wei C, Dong X, Lu H, Tong F, Chen L, Zhang R, et al. LPCAT1 promotes brain metastasis of lung adenocarcinoma by up-regulating PI3K/AKT/MYC pathway. J Exp Clin Cancer Res. 2019;38:95. doi: 10.1186/s13046-019-1092-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Naren D, Yan T, Gong Y, Huang J, Zhang D, Sang L, et al. High Wilms’ tumor 1 associating protein expression predicts poor prognosis in acute myeloid leukemia and regulates m(6)A methylation of MYC mRNA. J Cancer Res Clin Oncol. 2021;147:33–47. doi: 10.1007/s00432-020-03373-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Huiying Han GF, Song S, Jiang Y, Qian C, Zhang W, Qi Su XX, et al. piRNA-30473 contributes to tumorigenesis and poorprognosis by regulating m6A RNA methylation in DLBCL. blood. 2021 doi: 10.1182/blood.2019003764/1759249/blood.2019003764.pdf. [DOI] [PubMed] [Google Scholar]
- 116.Mathupala SP, Ko YH, Pedersen PL. Hexokinase II: cancer’s double-edged sword acting as both facilitator and gatekeeper of malignancy when bound to mitochondria. Oncogene. 2006;25:4777–86. doi: 10.1038/sj.onc.1209603. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Wilson JE. Isozymes of mammalian hexokinase: structure, subcellular localization and metabolic function. J Exp Biol. 2003;206:2049–57. doi: 10.1242/jeb.00241. [DOI] [PubMed] [Google Scholar]
- 118.Shi T, Ma Y, Cao L, Zhan S, Xu Y, Fu F, et al. B7-H3 promotes aerobic glycolysis and chemoresistance in colorectal cancer cells by regulating HK2. Cell Death Dis. 2019;10:308. doi: 10.1038/s41419-019-1549-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Fan K, Fan Z, Cheng H, Huang Q, Yang C, Jin K, et al. Hexokinase 2 dimerization and interaction with voltage-dependent anion channel promoted resistance to cell apoptosis induced by gemcitabine in pancreatic cancer. Cancer Med. 2019;8:5903–15. doi: 10.1002/cam4.2463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Xudong Zhanga, Lu Zhao b,1, Xin Li a, Xinhua Wanga, Ling Li a, Xiaorui Fua, & Zhenchang Suna, Z. L. a., Feifei Nana, Yu Changa, Mingzhi Zhanga,∗. ATP-binding cassette sub family C member 4 (ABCC4) is overexpressed in human NK/T-cell lymphoma and regulates chemotherapy sensitivity: Potential as a functional therapeutic target. Leukemia Research. 2015. 10.1016/j.leukres.2015.10.001. [DOI] [PubMed]
- 121.Zhu L, Zhang X, Fu X, Li Z, Sun Z, Wu J, et al. c-Myc mediated upregulation of long noncoding RNA SNHG12 regulates proliferation and drug sensitivity in natural killer/T-cell lymphoma. J Cell Biochem. 2019;120:12628–37. doi: 10.1002/jcb.28529. [DOI] [PubMed] [Google Scholar]
- 122.Roger Estrada-Tejedor GFE. Predicting drug resistance related to ABC transporters using unsupervised Consensus Self Organizing Maps. Sci Rep. 2017;8:6803. doi: 10.1038/s41598-018-25235-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Ma H, Shen L, Yang H, Gong H, Du X, Li J. m6A methyltransferase Wilms’ tumor 1-associated protein facilitates cell proliferation and cisplatin resistance in NK/T cell lymphoma by regulating dual-specificity phosphatases 6 expression via m6A RNA methylation. IUBMB Life. 2021;73:108–17. doi: 10.1002/iub.2410. [DOI] [PubMed] [Google Scholar]
- 124.Xie W, Liu N, Wang X, Wei L, Xie W, Sheng X. Wilms’ tumor 1-associated protein contributes to chemo-resistance to cisplatin through the Wnt/beta-catenin pathway in endometrial cancer. Front Oncol. 2021;11:598344. doi: 10.3389/fonc.2021.598344. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Li Q, Wang C, Dong W, Su Y, Ma Z. WTAP facilitates progression of endometrial cancer via CAV-1/NF-kappaB axis. Cell Biol Int. 2021;45:1269–77. doi: 10.1002/cbin.11570. [DOI] [PubMed] [Google Scholar]
- 126.Jo HJ, Shim HE, Han ME, Kim HJ, Kim KS, Baek S, et al. WTAP regulates migration and invasion of cholangiocarcinoma cells. J Gastroenterol. 2013;48:1271–82. doi: 10.1007/s00535-013-0748-7. [DOI] [PubMed] [Google Scholar]
- 127.Egeblad M, Werb Z. New functions for the matrix metalloproteinases in cancer progression. Nat Rev Cancer. 2002;2:161–74. doi: 10.1038/nrc745. [DOI] [PubMed] [Google Scholar]
- 128.Itatsu K, Sasaki M, Yamaguchi J, Ohira S, Ishikawa A, Ikeda H, et al. Cyclooxygenase-2 is involved in the up-regulation of matrix metalloproteinase-9 in cholangiocarcinoma induced by tumor necrosis factor-alpha. Am J Pathol. 2009;174:829–41. doi: 10.2353/ajpath.2009.080012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.von Bredow DC, Cress AE, Howard EW, Bowden GT, Nagle RB. Activation of gelatinase-tissue-inhibitors-of-metalloproteinase complexes by matrilysin. Biochem. 1998;J1:965–72, 10.1042/bj3310965. [DOI] [PMC free article] [PubMed]
- 130.Nakanishi H. Cathepsin regulation on microglial function. Biochim Biophys Acta Proteins Proteom. 2020;1868:140465. doi: 10.1016/j.bbapap.2020.140465. [DOI] [PubMed] [Google Scholar]
- 131.Hisatsune A, Nakayama H, Kawasaki M, Horie I, Miyata T, Isohama Y, et al. Anti-MUC1 antibody inhibits EGF receptor signaling in cancer cells. Biochem Biophys Res Commun. 2011;405:377–81. doi: 10.1016/j.bbrc.2011.01.029. [DOI] [PubMed] [Google Scholar]
- 132.Bansal H, Yihua Q, Iyer SP, Ganapathy S, Proia DA, Penalva LO, et al. WTAP is a novel oncogenic protein in acute myeloid leukemia. Leukemia. 2014;28:1171–4. doi: 10.1038/leu.2014.16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Li S, Young KH, Medeiros LJ. Diffuse large B-cell lymphoma. Pathology. 2018;50:74–87. doi: 10.1016/j.pathol.2017.09.006. [DOI] [PubMed] [Google Scholar]
- 134.Kuai Y, Gong X, Ding L, Li F, Lei L, Gong Y, et al. Wilms’ tumor 1-associating protein plays an aggressive role in diffuse large B-cell lymphoma and forms a complex with BCL6 via Hsp90. Cell Commun Signal. 2018;16:50. doi: 10.1186/s12964-018-0258-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Xi Z, Xue Y, Zheng J, Liu X, Ma J, Liu Y. WTAP expression predicts poor prognosis in malignant glioma patients. J Mol Neurosci. 2016;60:131–6. doi: 10.1007/s12031-016-0788-6. [DOI] [PubMed] [Google Scholar]
- 136.Clevers H, Nusse R. Wnt/beta-catenin signaling and disease. Cell. 2012;149:1192–205. doi: 10.1016/j.cell.2012.05.012. [DOI] [PubMed] [Google Scholar]
- 137.Zhang J, Tsoi H, Li X, Wang H, Gao J, Wang K, et al. Carbonic anhydrase IV inhibits colon cancer development by inhibiting the Wnt signalling pathway through targeting the WTAP-WT1-TBL1 axis. Gut. 2016;65:1482–93. doi: 10.1136/gutjnl-2014-308614. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.Fleming RE, Parkkila S, Parkkila AK, Rajaniemi H, Waheed A, Sly WS. Carbonic anhydrase IV expression in rat and human gastrointestinal tract regional, cellular, and subcellular localization. J Clin Invest. 1995;96:2907–13. doi: 10.1172/JCI118362. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Kim MK-H, McGarry TJ, Broin PO, Flatow JM, Golden AA-J, Licht JD, An integrated genome screen identifies the Wnt signaling pathway as a major target of WT1. Proc Natl Acad Sci USA. 2009;106:11154–9, 10.1073/pnas.0901591106. [DOI] [PMC free article] [PubMed]
- 140.S I Matsuzawa JCR. Siah-1, SIP, and Ebi collaborate in a novel pathway for b-catenin degradation linked to p53 Responses. Mol Cell. 2001;7:915–26. doi: 10.1016/s1097-2765(01)00242-8. [DOI] [PubMed] [Google Scholar]
- 141.Kim MS, Yoon SK, Bollig F, Kitagaki J, Hur W, Whye NJ, et al. A novel Wilms tumor 1 (WT1) target gene negatively regulates the WNT signaling pathway. J Biol Chem. 2010;285:14585–93. doi: 10.1074/jbc.M109.094334. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142.Li BQ, Huang S, Shao QQ, Sun J, Zhou L, You L, et al. WT1-associated protein is a novel prognostic factor in pancreatic ductal adenocarcinoma. Oncol Lett. 2017;13:2531–8. doi: 10.3892/ol.2017.5784. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.Chen L, Wang X. Relationship between the genetic expression of WTAP and bladder cancer and patient prognosis. Oncol Lett. 2018;16:6966–70. doi: 10.3892/ol.2018.9554. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144.Satyanarayana A, Kaldis P. A dual role of Cdk2 in DNA damage response. Cell Div. 2009;4:9. doi: 10.1186/1747-1028-4-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Yu HL, Ma XD, Tong JF, Li JQ, Guan XJ, Yang JH. WTAP is a prognostic marker of high-grade serous ovarian cancer and regulates the progression of ovarian cancer cells. Onco Targets Ther. 2019;12:6191–201. doi: 10.2147/OTT.S205730. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146.Wu LS, Qian JY, Wang M, Yang H. Identifying the role of Wilms tumor 1 associated protein in cancer prediction using integrative genomic analyses. Mol Med Rep. 2016;14:2823–31. doi: 10.3892/mmr.2016.5528. [DOI] [PubMed] [Google Scholar]
- 147.Weng L, Qiu K, Gao W, Shi C, Shu F. LncRNA PCGEM1 accelerates non-small cell lung cancer progression via sponging miR-433-3p to upregulate WTAP. BMC Pulm Med. 2020;20:213. doi: 10.1186/s12890-020-01240-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 148.Sharma D, Subbarao G, Saxena R. Hepatoblastoma. Semin Diagn Pathol. 2017;34:192–200. doi: 10.1053/j.semdp.2016.12.015. [DOI] [PubMed] [Google Scholar]
- 149.Zhuo ZJ, Hua RX, Chen Z, Zhu J, Wang M, Yang Z, et al. WTAP gene variants confer hepatoblastoma susceptibility: a seven-center case-control study. Mol Ther Oncolytics. 2020;18:118–25. doi: 10.1016/j.omto.2020.06.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 150.Chen B, Ye F, Yu L, Jia G, Huang X, Zhang X, et al. Development of cell-active N6-methyladenosine RNA demethylase FTO inhibitor. J Am Chem Soc. 2012;134:17963–71. doi: 10.1021/ja3064149. [DOI] [PubMed] [Google Scholar]
- 151.He W, Zhou B, Liu W, Zhang M, Shen Z, Han Z, et al. Identification of a novel small-molecule binding site of the fat mass and obesity associated protein (FTO) J Med Chem. 2015;58:7341–8. doi: 10.1021/acs.jmedchem.5b00702. [DOI] [PubMed] [Google Scholar]
- 152.Qiao Y, Zhou B, Zhang M, Liu W, Han Z, Song C, et al. A novel inhibitor of the obesity-related protein FTO. Biochemistry. 2016;55:1516–22. doi: 10.1021/acs.biochem.6b00023. [DOI] [PubMed] [Google Scholar]
- 153.Huang Y, Yan J, Li Q, Li J, Gong S, Zhou H, et al. Meclofenamic acid selectively inhibits FTO demethylation of m6A over ALKBH5. Nucleic Acids Res. 2015;43:373–84. doi: 10.1093/nar/gku1276. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The materials that support the conclusion of this review have been included within the article.