Abstract
DNA sequence and Southern blot analyses were used to determine the genetic defect of a Haemophilus ducreyi pyocin-resistant lipooligosaccharide (LOS) mutant, HD35000R. The region of the HD35000R chromosome containing the suspected mutation was amplified, and sequence analysis detected a 3,189-bp deletion. This deletion resulted in the loss of the entire waaQ gene, another open reading frame that encodes a putative homolog to a hypothetical protein (HI0461) of H. influenzae, the gene encoding an argininosuccinate synthase homolog, and a change in the 3′ sequence of the lgtF gene. Southern blot analysis confirmed that no genomic rearrangements had occurred. Isogenic LOS mutants and the respective complemented mutants were evaluated for susceptibility to pyocin C. The mutants expressing truncated LOS were resistant to lysis by pyocin C, and complementation restored sensitivity to the pyocin. We conclude that HD35000R is defective in both glycosyltransferase genes and that pyocin resistance is due to truncation of the full-length LOS molecule.
Haemophilus ducreyi is a gram-negative organism which causes chancroid. This genital ulcerative disease is endemic in many developing countries, with infection rates being highest in Africa and Asia, where prostitution is an important risk factor (40). Occasionally, sporadic epidemics occur in the United States; however, most of these outbreaks are associated with drug use and the sale of sex for drugs or money (8, 9). Although it is difficult to obtain reliable data, the World Health Organization estimated that approximately 7 million new cases of chancroid infection occurred in 1995 worldwide (43). In addition, chancroid is one of a number of genital ulcer diseases that serve as cofactors for human immunodeficiency virus transmission (7). Because the actual skin lesions are the likely site of human immunodeficiency virus entry, identification and characterization of virulence factors that contribute to ulcer formation have become an important goal of current H. ducreyi research efforts.
One potential virulence factor of H. ducreyi is the lipooligosaccharides (LOS). The LOS of H. ducreyi structurally resembles LOS from other gram-negative mucosal pathogens, such as Haemophilus influenzae, Neisseria meningitidis, and Neisseria gonorrhoeae (5, 22–24). The LOS molecules from these three human pathogens are important virulence factors involved in adherence to host epithelial cells, serum resistance, and evasion of the host immune system (16, 25, 31, 32, 39, 42). While these data suggest similar functions for H. ducreyi LOS, the actual role of LOS in chancroid is currently undefined. The inability to identify or construct LOS mutants in H. ducreyi was initially a major obstacle; however, this was overcome using pyocin lysis (4, 13) and transposon-based mutagenesis (15, 38) to identify mutants defective in expression of LOS biosynthesis from H. ducreyi strain 35000.
Pyocins, bacteriocins produced by Pseudomonas aeruginosa, have structures similar to contractile bacteriophage tails and are thought to use the lipopolysaccharide or LOS molecule as a receptor (18). The mechanism of action of these particles is through formation of a pore in the membrane and subsequent disruption of membrane potential resulting in cell death (41). Pyocins also arrest protein and nucleic acid synthesis of the bacteria (17, 19, 27, 29). Although the mechanism(s) allowing bacteria to survive pyocin lysis is unknown, pyocin-resistant strains of N. gonorrhoeae and H. ducreyi have truncated LOS molecules (4, 11, 13, 26). These pyocin mutants have been used to identify genes involved in LOS biosynthesis (13, 21, 34), and several pyocin-resistant gonococci have been characterized at the DNA level (35).
More recently, we described an H. ducreyi pyocin C survivor, designated HD35000R (13). This pyocin mutant, derived from H. ducreyi strain 35000, was used to clone genes involved in LOS biosynthesis using complementation. These data suggested that HD35000R had disruptions in both the waaQ and lgtF glycosyltransferase genes. The aim of this study was to investigate the genetic defect of HD35000R using Southern blot and DNA sequence analyses and to begin to define the interaction of pyocin C with LOS.
Bacterial strains and culture conditions.
H. ducreyi strain 35000 is a wild-type strain isolated in Winnipeg, Canada. HD35000R was derived from 35000 as a pyocin-resistant mutant (13). The isogenic LOS mutants, 35000glu-, 35000hep-, and 35000hepglu- were constructed in our laboratory and have been described previously (13). pLS88 is an H. ducreyi shuttle vector (10). 35000glu-(pGLU), 35000hep-(pHEP), and 35000hepglu-(pLS88HG.13) are complemented strains derived from the electroporation of plasmids containing the respective wild-type gene(s) into the respective mutant (13). 35000hepglu-(pGLU) was derived from the electroporation of the plasmid containing the lgtF gene. H. ducreyi 35000HP is a human passaged variant of 35000 and has been described previously (1). All H. ducreyi strains were grown at 35°C in 5% CO2 on chocolate agar plates (6). Escherichia coli strains were grown at 37°C on Luria-Bertani agar plates or in Luria-Bertani broth containing 5-bromo-4-chloro-3-indolyl-β-d-galactopyranoside (X-Gal) (40 μg/ml) and/or isopropyl-β-d-thiogalactopyranoside (IPTG) (25 μg/ml) and kanamycin (50 μg/ml) as needed. P. aeruginosa strain C was grown in Pseudomonas broth (11).
Chemicals, reagents, and enzymes.
Plasmid DNA was isolated using Qiagen purification kits. Restriction enzymes were purchased from New England Biolabs, Inc. Standard methods were used for restriction endonuclease analysis, ligations, and transformation of plasmid DNA (33).
Identification of the genetic defect in HD35000R.
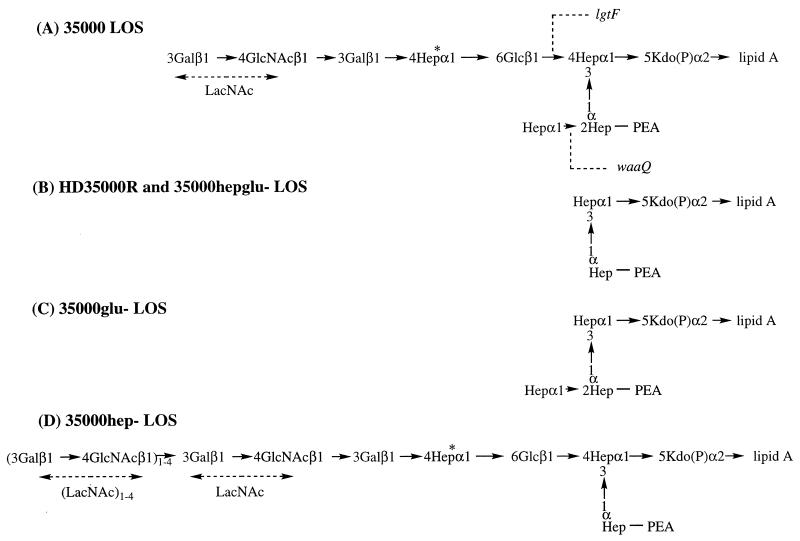
Previously, we reported the identification and characterization of a pyocin-resistant LOS mutant designated HD35000R (13). Matrix-assisted laser desorption ionization mass spectrometry analysis of O-deacylated LOS from HD35000R indicated that the LOS terminated with a core consisting of only two of the three heptose residues with no additional branch structures (Fig. 1B), compared to the wild-type strain 35000 (Fig. 1A). This mutant was complemented with a plasmid containing the waaQ and lgtF genes (13). Further, an isogenic mutant disrupted in both genes produced the same LOS phenotype as HD35000R (Fig. 1B); therefore, we hypothesized that HD35000R had mutations in both glycosyltransferase genes. Repeated attempts to amplify these genes using primers developed to portions of the waaQ and lgtF genes were unsuccessful for reasons which will be explained below.
FIG. 1.
Biochemical structures of H. ducreyi 35000 LOS and LOS structures expressed by genetically defined 35000 mutants, as determined by Filiatrault et al. (13). (A) The major LOS structure from H. ducreyi 35000 consists of lipid A, keto-deoxyoctulosonic acid, a triheptose core, and one main oligosaccharide branch. This structure can also be substituted for by sialic acid. (B) HD35000R synthesizes a LOS molecule which lacks the main oligosaccharide branch and the third heptose residue of the core. LOS synthesized by HD35000R and 35000hepglu- were identical. (C) The LOS produced by the lgtF mutant, 35000glu-, consists of lipid A, keto-deoxyoctulosonic, and the triheptose core. (D) The waaQ mutant, 35000hep-, produces an LOS molecule similar to the wild-type strain 35000; however, the structure lacks the third heptose of the core and additional lactosamine repeats are added to the terminal portion of the molecule. In addition, no sialic acid is present in the waaQ LOS structure. The locations of where the gene products of the β1,4 glucosyltransferase (LgtF) and the heptosyltransferase III (WaaQ) act are indicated by dashed lines. All core heptoses are l-glycero-d-manno-heptose with the exception of the branch heptose (asterisk), which is d-glycero-d-manno-heptose. Abbreviations are as follows: LacNAc, N-acetyllactosamine; NeuAC, sialic acid; Gal, galactose; Glu, glucose; GlcNAc, N-acetylglucosamine; Hep, heptose; KDO, keto-deoxyoctulosonic acid; PEA, phosphoethanolamine.
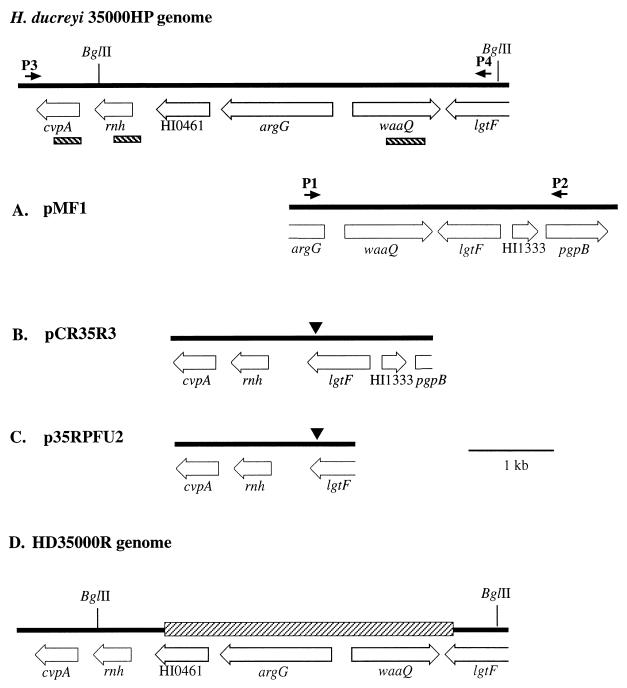
The wild-type plasmid library constructed in pLS99 was used a second time to complement HD35000R, and additional sequence 5′ to the waaQ gene was obtained (13). This complementing clone was designated pMF1 (Fig. 2A). Using this sequence, primers were designed for the argininosuccinate synthase homolog (P1), which is 5′ to waaQ, and the pgpB homolog (P2), which is upstream of lgtF (Fig. 2A). PCR was performed with H. ducreyi 35000 and HD35000R genomic DNA. The expected 2.8-kb product was amplified from the wild-type strain. A slightly larger amplicon was amplified from HD35000R chromosomal DNA, using the same primer pair. These amplicons were purified using the GeneClean II kit (Bio 101), cloned into the pCR2.1 vector (Invitrogen), and the nucleotide sequences of both strands were determined. The nucleotide sequence of the plasmid clone containing the PCR product from the wild type was identical with the nucleotide sequence of H. ducreyi 35000HP (http://www.ncbi.nlm.nih.gov /Microb_blast/unfinishedgenome.html; data not shown). Sequence analysis of the DNA fragment from HD35000R, designated pCR35R3 (Fig. 2B), identified an open reading frame (ORF) which encodes a polypeptide that shares 59% identity and 72% similarity to the colicin V production proteins of H. influenzae (14) and E. coli (3) and therefore has been designated cvpA. Colicin V is a proteinaceous bacterial toxin produced by many strains of E. coli and other members of the Enterobacteriaceae and is associated with the pathogenicity of these organisms (2). Interestingly, the genetic organization of the S-type pyocins of P. aeruginosa is similar to that of colicins (12, 36). While production of a colicin may allow survival in a stressful environment and provide H. ducreyi with an advantage over other organisms, more studies are needed to determine if this organism possesses a similar bacteriocin system and if production of this toxic factor contributes to the pathogenesis of H. ducreyi.
FIG. 2.
Partial ORF map of the H. ducreyi genome and the plasmids characterized in this study. The ORFs are indicated by open arrows which designate the direction of transcription. The solid arrows represent the oligonucleotide primers used in PCR. The hatched bars indicate the probes used for Southern blot analysis. The point where the deletion occurred in HD35000R is indicated (▾). (A) Plasmid pMF1 is a plasmid which complements HD35000R. (B) Plasmid pCR35R3 contains a 3.1-kb PCR product from HD35000R chromosomal DNA using primers P1 and P2. (C) Plasmid p35RPFU#2 is pCRBlunt vector with a 2.16-kb PCR product from HD35000R chromosomal DNA using primers P3 and P4. (D) Partial ORF map of the HD35000R genome. The deleted region is designated by the hatched bar.
The derived amino acid sequence of the next ORF has significant homology with numerous RNases, including the RNase H from H. influenzae (76% identity and 85% similarity) (14). The next ORFs were the lgtF, HI1333, and a portion of pgpB (Fig. 2B). An unexpected observation was the lack of the ORF which has a derived amino acid sequence similar to the argininosuccinate synthase of H. influenzae (ArgG) (14), the region where primer P1 was developed. Using dot plot matrix analysis, we found that this particular segment of DNA in the H. ducreyi genome contained a sequence at the 5′ end which was the reverse complement of the sequence at the 3′ end. PCR using a single primer, P2, confirmed that this DNA fragment could be amplified (data not shown).
In order to analyze the DNA region surrounding the LOS genes and identify the position of the colicin V gene in relation to the LOS biosynthesis genes, the H. ducreyi 35000HP genomic sequence was obtained from the NCBI unfinished microbial genome BLAST database (http://www.ncbi.nlm.nih.gov/icrob_blast /unfinishedgenome.html) and the High Throughput Sequencing Center at the University of Washington, Seattle. A homology search of the putative colicin V gene with the H. ducreyi 35000HP genome revealed that this gene is ∼3 kb upstream of the glycosyltransferase genes (Fig. 2 [top]). In this gene cluster cvpA is preceded by the gene encoding the RNase H-like protein and an ORF which has a derived amino acid sequence with 42% identity and 66% similarity to a conserved hypothetical membrane protein of H. influenzae (HI0461) (14) and N. meningitidis (30) (Fig. 2 [top]). These results suggest that the chromosomal DNA of HD35000R contains a large deletion.
Based on the unexpected results obtained initially with primers P1 and P2 (above), a second PCR of this region was performed using another primer set to further verify the suspected deletion in strain HD35000R. The entire region spanning the colicin V gene to the lgtF from H. ducreyi 35000 and HD35000R chromosomal DNA was amplified with oligonucleotide primers P3 and P4 (Fig. 2 [top]). A 5.35-kb fragment was amplified from wild-type 35000 and a 2.16-kb fragment was obtained from HD35000R as predicted. The smaller size of the PCR fragment obtained from HD35000R was consistent with a 3.18-kb deletion. This 2.16-kb product was cloned into the pCRBlunt vector (Invitrogen) to form p35RPFU2 (Fig. 2C), and the sequence was determined and analyzed. DNA sequences of p35RPFU2 were aligned to 35000HP. Sequence from p35RPFU2 was identical to 35000HP except for a 3,189-bp deletion. In Fig. 2D the location of the deletion is depicted by a cross-hatched bar. Sequence analysis did not reveal any unique characteristics of the region where the deletion occurred. These results are consistent with the structural and complementation data of HD35000R LOS (13) (Fig. 1).
In analyzing the DNA sequence obtained from HD35000R, we observed that a portion of the 5′ end of the HI0461 ORF had been fused to the 3′ end of the lgtF gene in frame, resulting in the replacement of the last 37 bp of lgtF. The derived amino acid sequence of the HD35000R protein differs from the sequence of the 35000HP protein by 10 amino acids (data not shown). This suggests that a fusion protein would be generated between LgtF and a portion of the HI0461 ORF in HD35000R, although the LOS analysis suggests that this is a nonfunctional protein. LgtF is assigned to family 2 of the glycosyltransferase enzymes (http://afmb.cnrs-mrs.fr/∼pedro /CAZY/db.html). These enzymes are inverting glycosyltransferases that possess a nucleotide binding domain (domain A) within the N-terminal region (37). However, glycosyltransferases from the same family demonstrate little to no sequence conservation throughout their C-terminal region (20). Therefore, residues which may be involved in substrate recognition and catalytic activity do not appear to be conserved. By creation of an in-frame fusion between the two gene portions of lgtF and HI0461, critical residues in the C-terminal region of the LgtF may have been eliminated, resulting in loss of substrate binding. The LOS structure of HD35000R suggests that the mutation, which occurred in HD35000R, disrupts properties of this enzyme, and that one or more of the C-terminal 13 amino acids of LgtF are required for proper function. It is, however, possible that the fusion protein may not be produced or is unstable. Further studies are required to determine the specific residues that are important in LgtF transferase activity.
Southern blot analysis.
To determine whether a chromosomal rearrangement or deletion of this region had occurred, Southern blot analysis was performed as previously described (13), using the NEBlot Phototope labeling kit and Phototope-Star detection kit (New England Biolabs), with the exception that hybridizations were performed at 60°C in 100 ng of denatured biotinylated probe per ml of hybridization fluid. Chromosomal DNA was isolated from H. ducreyi strains, as previously described (13), and digested to completion with BglII, electrophoresed on a 0.7% agarose gel, and transferred to Immobilon-Ny+ membrane (Millipore) by capillary blotting overnight. Probes to the cvpA, rnh, and waaQ genes were generated by PCR, utilizing the primers listed in Table 1 and the plasmids pCR35R3 and pMF1 as templates, and were used to probe BglII-digested chromosomal DNA from H. ducreyi strain 35000 and HD35000R. The cvpA probe hybridized to a single fragment of ∼2.2 kb from H. ducreyi strain 35000 and HD35000R (data not shown). When chromosomal DNA from H. ducreyi strain 35000 was probed with a portion of the waaQ gene, a band of ∼4.6 kb was observed (data not shown). This probe did not hybridize with chromosomal DNA from HD35000R, demonstrating that HD35000R does not contain this portion of the waaQ gene (data not shown). The rnh probe hybridized to a fragment of ∼4.6 kb from H. ducreyi strain 35000 and an ∼1.4-kb fragment from HD35000R chromosomal DNA (data not shown). These results are consistent with a 3.18-kb deletion in HD35000R, demonstrating that no chromosomal DNA rearrangements of these genes occurred.
TABLE 1.
Primers used in this study
| Primer | Oligonucleotide sequence (5′-3′) | Description |
|---|---|---|
| P1 | CCAACCTTTTGACCTAATGGGAG | |
| P2 | AAATCCAAGAATAATGCCGAC | |
| P3 | TAATGCCGACAATGCCGCAC | |
| P4 | GCAAGGTTTTGGTCCACAGAGAC | |
| P5 | TGTCGCAGATAAACCTGTGGTG | Forward primer used with P6 to amplify a 522-bp product to probe for cvpA |
| P6 | CGATGAACTGGCTAAACTCGGC | Reverse primer used with P5 to amplify a 522-bp product to probe for cvpA |
| P7 | GATGCCTGTTGAGCCTCAGATTC | Forward primer used with P8 to amplify a 505-bp product to probe for waaQ |
| P8 | TTGTTTACCGCTAGGGGGACAG | Reverse primer used with P7 to amplify a 502-bp product to probe for waaQ |
| P9 | TCCTTGACTCACTTTTTTCTGATGC | Forward primer used with P10 to amplify a 437-bp product to probe for rnh |
| P10 | GCGTTCGTCCAATGTTTATGTTG | Reverse primer used with P9 to amplify a 437-bp product to probe for rnh |
Pyocin sensitivity of the LOS mutants and complemented mutants.
HD35000R was initially selected based on resistance to pyocin C. In order to confirm that pyocin resistance was a result of the truncation or loss of LOS genes, LOS mutants were assayed for sensitivity to pyocin C. Isogenic LOS mutants of H. ducreyi strain 35000 lacking expression of the heptosyltransferase III (WaaQ), the β1,4 glucosyltransferase (LgtF), and both glycosyltransferases were constructed and characterized previously (13). The heptosyltransferase mutant produces an LOS molecule which is similar to the wild type but lacks the third heptose of the triheptose core and contains additional lactosamine repeats (Fig. 1D) (13). The glucosyltransferase mutant produces a truncated LOS that terminates after the triheptose core and lacks the main oligosaccharide branch (Fig. 1C) (13). The LOS glycoform expressed by the double mutant contains two of the heptose sugars of the core (Fig. 1B) (13). Pyocin was isolated from cultures of P. aeruginosa strain C by the method described by Morse et al. (28), and the pyocin lysis assay was performed as described previously (4, 11).
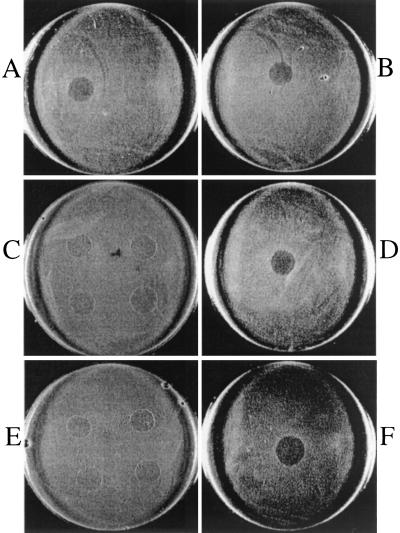
Both the glucosyltransferase mutant (35000glu-) and the double mutant (35000hepglu-) were resistant to pyocin lysis (Fig. 3B and D). However, the heptosyltransferase mutant (35000hep-) was sensitive to lysis by pyocin C, suggesting that pyocin interacts with the full-length chain (Fig. 3C). To test whether sensitivity could be restored to 35000glu- and 35000hepglu-, the complemented mutants were analyzed. Complementing the glucosyltransferase with the wild-type lgtF and the double mutant with lgtF and waaQ restored sensitivity to pyocin C (Fig. 3E and F). The complemented waaQ mutant remained sensitive, as expected (data not shown). In addition, the double mutant was complemented with only the lgtF gene. This strain, which displays the same LOS phenotype as the waaQ mutant, was also sensitive to the pyocin (data not shown). These results suggest that the main oligosaccharide branch is involved in susceptibility to this pyocin, since the heptosyltransferase mutant and the wild type, which possess a common oligosaccharide chain, are sensitive. These studies demonstrate that the core is not involved in pyocin attachment, since the LOS synthesized by the heptosyltransferase mutant lacks the third heptose of the core and since this mutant remains sensitive.
FIG. 3.
Pyocin C sensitivity of H. ducreyi 35000, isogenic LOS mutants, and complemented LOS mutants. Wild-type strain 35000 and isogenic LOS mutants were electroporated with the pLS88 shuttle vector or pLS88 containing the respective wild-type gene(s) as previously described (13). All strains were then tested for pyocin sensitivity and observed for a zone of lysis, as described in Materials and Methods. (A) 35000(pLS88); (B) 35000hep-(pLS88); (C) 35000glu-(pLS88); (D) 35000glu-(pGLU); (E) 35000hepglu-(pLS88); (F) 35000hepglu-(pLS88HG.13).
Conclusions.
Genetic evaluation of several pyocin-resistant gonococci revealed a 12-bp deletion and a point mutation in the phosphoglucomutase gene and a nonsense mutation in the rfaF gene (35). The authors hypothesized that because there were no large deletions, it was unlikely that there was an interaction of pyocin DNA with the bacterial DNA. However, these studies did not investigate whether there were other deletions or rearrangements in the remainder of the chromosome. Therefore, the possibility that there were large chromosomal deletions in these mutants cannot be excluded.
Because all pyocin mutants described to date synthesize truncated LOS molecules compared to the parent strain, the lipopolysaccharide and LOS molecules have been implicated as the receptor for R-type pyocins in P. aeruginosa (28) and N. gonorrhoeae (11, 18, 44), respectively. Although many N. gonorrhoeae prototype and pyocin mutant strains have been tested for pyocin sensitivity (11), several drawbacks of this method include the instability of pyocin mutants, the possibility of other mutations in pyocin-resistant strains, and the ability of N. gonorrhoeae to phase vary its LOS molecule. The novelty of this study is the evaluation of stable isogenic H. ducreyi LOS mutants and their respective complemented mutants for pyocin sensitivity, demonstrating that sensitivity or resistance to pyocin is due solely to the LOS molecule.
In this study, we evaluated isogenic mutants previously constructed in our laboratory. As expected, the mutants, which produced truncated LOS molecules (35000glu- and 35000hepglu-), were resistant to pyocin lysis by pyocin C. Complementation of the H. ducreyi lgtF and waaQ lgtF double mutant with the wild-type genes in trans restored sensitivity to pyocin C. In addition, complementation of the H. ducreyi waaQ lgtF mutant with the lgtF gene alone restored sensitivity to pyocin. This finding suggests that the main oligosaccharide branch of the LOS molecule is involved in susceptibility to this pyocin and that the third heptose of the core is not required. In addition, novel genes present in the H. ducreyi genome were identified, suggesting that this organism may contain a bacteriocin-like system similar to those described for other pathogens.
Acknowledgments
This work was supported by the National Institutes of Health grant R01 AI30006 (to A.A.C.). M.J.F. was partially supported by training grant AI07614-01 from the National Institutes of Health.
REFERENCES
- 1.Al-Tawfiq J A, Thornton A C, Katz B P, Fortney K R, Todd K D, Hood A F, Spinola S M. Standardization of the experimental model of Haemophilus ducreyi infection in human subjects. J Infect Dis. 1998;178:1684–1687. doi: 10.1086/314483. [DOI] [PubMed] [Google Scholar]
- 2.Blanco J E, Blanco M, Mora A, Blanco J. Production of toxins (enterotoxins, verotoxins, and necrotoxins) and colicins by Escherichia coli strains isolated from septicemic and healthy chickens: relationship with in vivo pathogenicity. J Clin Microbiol. 1997;35:2953–2957. doi: 10.1128/jcm.35.11.2953-2957.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Blattner F R, Plunkett III G, Bloch C A, Perna N T, Burland V, Riley M, Collado-Vides J, Glasner J D, Rode C K, Mayhew G F, Gregor J, Davis N W, Kirkpatrick H A, Goeden M A, Rose D J, Mau B, Shao Y. The complete genome sequence of Escherichia coli K-12. Science. 1997;277:1453–1474. doi: 10.1126/science.277.5331.1453. [DOI] [PubMed] [Google Scholar]
- 4.Campagnari A A, Karalus R, Apicella M, Melaugh W, Lesse A J, Gibson B W. Use of pyocin to select a Haemophilus ducreyi variant defective in lipooligosaccharide biosynthesis. Infect Immun. 1994;62:2379–2386. doi: 10.1128/iai.62.6.2379-2386.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Campagnari A A, Spinola S M, Lesse A J, Kwaik Y A, Mandrell R E, Apicella M A. Lipooligosaccharide epitopes shared among gram-negative non-enteric mucosal pathogens. Microb Pathog. 1990;8:353–362. doi: 10.1016/0882-4010(90)90094-7. [DOI] [PubMed] [Google Scholar]
- 6.Campagnari A A, Wild L M, Griffiths G E, Karalus R J, Wirth M A, Spinola S M. Role of lipooligosaccharides in experimental dermal lesions caused by Haemophilus ducreyi. Infect Immun. 1991;59:2601–2608. doi: 10.1128/iai.59.8.2601-2608.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Centers for Disease Control and Prevention. HIV prevention through early detection and treatment of other sexually transmitted diseases—United States. Recommendations of the Advisory Committee for HIV and STD prevention. Atlanta, Ga: Centers for Disease Control and Prevention; 1998. [PubMed] [Google Scholar]
- 8.Chirgwin K, DeHovitz J A, Dillon S, McCormack W M. HIV infection, genital ulcer disease, and crack cocaine use among patients attending a clinic for sexually transmitted diseases. Am J Public Health. 1991;81:1576–1579. doi: 10.2105/ajph.81.12.1576. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.DiCarlo R P, Armentor B S, Martin D H. Chancroid epidemiology in New Orleans men. J Infect Dis. 1995;172:446–452. doi: 10.1093/infdis/172.2.446. [DOI] [PubMed] [Google Scholar]
- 10.Dixon L G, Albritton W L, Willson P J. An analysis of the complete nucleotide sequence of the Haemophilus ducreyi broad-host-range plasmid pLS88. Plasmid. 1994;32:228–232. doi: 10.1006/plas.1994.1060. [DOI] [PubMed] [Google Scholar]
- 11.Dudas K C, Apicella M A. Selection and immunochemical analysis of lipooligosaccharide mutants of Neisseria gonorrhoeae. Infect Immun. 1988;56:499–504. doi: 10.1128/iai.56.2.499-504.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Duport C, Baysse C, Michel-Briand Y. Molecular characterization of pyocin S3: a novel S-type pyocin from Pseudomonas aeruginosa. J Biol Chem. 1995;270:8920–8927. doi: 10.1074/jbc.270.15.8920. [DOI] [PubMed] [Google Scholar]
- 13.Filiatrault M J, Gibson B W, Schilling B, Sun S, Munson R S, Jr, Campagnari A A. Construction and characterization of Haemophilus ducreyi lipooligosaccharide (LOS) mutants defective in expression of heptosyltransferase III and β1,4-glucosyltransferase: identification of LOS glycoforms containing lactosamine repeats. Infect Immun. 2000;68:3352–3361. doi: 10.1128/iai.68.6.3352-3361.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Fleischmann R D, Adams M D, White O, Clayton R A, Kirkness E F, Kerlavage A R, Bult C J, Tomb J F, Dougherty B A, Merrick J M, et al. Whole-genome random sequencing and assembly of Haemophilus influenzae Rd. Science. 1995;269:496–512. doi: 10.1126/science.7542800. [DOI] [PubMed] [Google Scholar]
- 15.Gibson B W, Campagnari A A, Melaugh W, Phillips N J, Apicella M A, Grass S, Wang J, Palmer K L, Munson R S., Jr Characterization of a transposon Tn916-generated mutant of Haemophilus ducreyi 35000 defective in lipooligosaccharide biosynthesis. J Bacteriol. 1997;179:5062–5071. doi: 10.1128/jb.179.16.5062-5071.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Harvey H A, Porat N, Campbell C A, Jennings M, Gibson B W, Phillips N J, Apicella M A, Blake M S. Gonococcal lipooligosaccharide is a ligand for the asialoglycoprotein receptor on human sperm. Mol Microbiol. 2000;36:1059–1070. doi: 10.1046/j.1365-2958.2000.01938.x. [DOI] [PubMed] [Google Scholar]
- 17.Iijima M. Mode of action of pyocin R1. J Biochem (Tokyo) 1978;83:395–402. doi: 10.1093/oxfordjournals.jbchem.a131926. [DOI] [PubMed] [Google Scholar]
- 18.Ikeda K, Egami F. Receptor substance for pyocin R. I. Partial purification and chemical properties. J Biochem (Tokyo) 1969;65:603–609. doi: 10.1093/oxfordjournals.jbchem.a129053. [DOI] [PubMed] [Google Scholar]
- 19.Kaziro Y, Tanaka M. Studies on the mode of action of pyocin. I. Inhibition of macromolecular synthesis in sensitive cells. J Biochem (Tokyo) 1965;57:689–695. [PubMed] [Google Scholar]
- 20.Keenleyside W J, Clarke A J, Whitfield C. Identification of residues involved in catalytic activity of the inverting glycosyl transferase WbbE from Salmonella enterica serovar Borreze. J Bacteriol. 2001;183:77–85. doi: 10.1128/JB.183.1.77-85.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Levin J C, Stein D C. Cloning, complementation, and characterization of an rfaE homolog from Neisseria gonorrhoeae. J Bacteriol. 1996;178:4571–4575. doi: 10.1128/jb.178.15.4571-4575.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Mandrell R E, Griffiss J M, Smith H, Cole J A. Distribution of a lipooligosaccharide-specific sialyltransferase in pathogenic and non-pathogenic Neisseria. Microb Pathog. 1993;14:315–327. doi: 10.1006/mpat.1993.1031. [DOI] [PubMed] [Google Scholar]
- 23.Melaugh W, Campagnari A A, Gibson B W. The lipooligosaccharides of Haemophilus ducreyi are highly sialylated. J Bacteriol. 1996;178:564–570. doi: 10.1128/jb.178.2.564-570.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Melaugh W, Phillips N J, Campagnari A A, Tullius M V, Gibson B W. Structure of the major oligosaccharide from the lipooligosaccharide of Haemophilus ducreyi strain 35000 and evidence for additional glycoforms. Biochemistry. 1994;33:13070–13078. doi: 10.1021/bi00248a016. [DOI] [PubMed] [Google Scholar]
- 25.Moran A P, Prendergast M M, Appelmelk B J. Molecular mimicry of host structures by bacterial lipopolysaccharides and its contribution to disease. FEMS Immunol Med Microbiol. 1996;16:105–115. doi: 10.1111/j.1574-695X.1996.tb00127.x. [DOI] [PubMed] [Google Scholar]
- 26.Morse S A, Apicella M A. Isolation of a lipopolysaccharide mutant of Neisseria gonorrhoeae: an analysis of the antigenic and biologic difference. J Infect Dis. 1982;145:206–216. doi: 10.1093/infdis/145.2.206. [DOI] [PubMed] [Google Scholar]
- 27.Morse S A, Jones B V, Lysko P G. Pyocin inhibition of Neisseria gonorrhoeae: mechanism of action. Antimicrob Agents Chemother. 1980;18:416–423. doi: 10.1128/aac.18.3.416. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Morse S A, Vaughan P, Johnson D, Iglewski B H. Inhibition of Neisseria gonorrhoeae by a bacteriocin from Pseudomonas aeruginosa. Antimicrob Agents Chemother. 1976;10:354–362. doi: 10.1128/aac.10.2.354. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Ohsumi M, Shinomiya T, Kageyama M. Comparative study on R-type pyocins of Pseudomonas aeruginosa. J Biochem (Tokyo) 1980;87:1119–1125. [PubMed] [Google Scholar]
- 30.Parkhill J, Achtman M, James K D, Bentley S D, Churcher C, Klee S R, Morelli G, Basham D, Brown D, Chillingworth T, Davies R M, Davis P, Devlin K, Feltwell T, Hamlin N, Holroyd S, Jagels K, Leather S, Moule S, Mungall K, Quail M A, Rajandream M A, Rutherford K M, Simmonds M, Skelton J, Whitehead S, Spratt B G, Barrell B G. Complete DNA sequence of a serogroup A strain of Neisseria meningitidis Z2491. Nature. 2000;404:502–506. doi: 10.1038/35006655. [DOI] [PubMed] [Google Scholar]
- 31.Preston A, Mandrell R E, Gibson B W, Apicella M A. The lipooligosaccharides of pathogenic gram-negative bacteria. Crit Rev Microbiol. 1996;22:139–180. doi: 10.3109/10408419609106458. [DOI] [PubMed] [Google Scholar]
- 32.Ram S, Sharma A K, Simpson S D, Gulati S, McQuillen D P, Pangburn M K, Rice P A. A novel sialic acid binding site on factor H mediates serum resistance of sialylated Neisseria gonorrhoeae. J Exp Med. 1998;187:743–752. doi: 10.1084/jem.187.5.743. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Sambrook J, Fritsch E F, Maniatis T. Molecular cloning: a laboratory manual. 2nd ed. Cold Spring Harbor, N.Y: Cold Spring Harbor Laboratory Press; 1989. [Google Scholar]
- 34.Sandlin R C, Apicella M A, Stein D C. Cloning of a gonococcal DNA sequence that complements the lipooligosaccharide defects of Neisseria gonorrhoeae 1291d and 1291e. Infect Immun. 1993;61:3360–3368. doi: 10.1128/iai.61.8.3360-3368.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Sandlin R C, Danaher R J, Stein D C. Genetic basis of pyocin resistance in Neisseria gonorrhoeae. J Bacteriol. 1994;176:6869–6876. doi: 10.1128/jb.176.22.6869-6876.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Sano Y, Kobayashi M, Kageyama M. Functional domains of S-type pyocins deduced from chimeric molecules. J Bacteriol. 1993;175:6179–6185. doi: 10.1128/jb.175.19.6179-6185.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Saxena I M, Brown R M, Jr, Fevre M, Geremia R A, Henrissat B. Multidomain architecture of β-glycosyl transferases: implications for mechanism of action. J Bacteriol. 1995;177:1419–1424. doi: 10.1128/jb.177.6.1419-1424.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Stevens M K, Klesney-Tait J, Lumbley S, Walters K A, Joffe A M, Radolf J D, Hansen E J. Identification of tandem genes involved in lipooligosaccharide expression by Haemophilus ducreyi. Infect Immun. 1997;65:651–660. doi: 10.1128/iai.65.2.651-660.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Swords W E, Buscher B A, Ver Steeg K, II, Preston A, Nichols W A, Weiser J N, Gibson B W, Apicella M A. Non-typeable Haemophilus influenzae adhere to and invade human bronchial epithelial cells via an interaction of lipooligosaccharide with the PAF receptor. Mol Microbiol. 2000;37:13–27. doi: 10.1046/j.1365-2958.2000.01952.x. [DOI] [PubMed] [Google Scholar]
- 40.Trees D L, Morse S A. Chancroid and Haemophilus ducreyi: an update. Clin Microbiol Rev. 1995;8:357–375. doi: 10.1128/cmr.8.3.357. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Uratani Y, Hoshino T. Pyocin R1 inhibits active transport in Pseudomonas aeruginosa and depolarizes membrane potential. J Bacteriol. 1984;157:632–636. doi: 10.1128/jb.157.2.632-636.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Vogel U, Frosch M. Mechanisms of neisserial serum resistance. Mol Microbiol. 1999;32:1133–1139. doi: 10.1046/j.1365-2958.1999.01469.x. [DOI] [PubMed] [Google Scholar]
- 43.World Health Organization. Sexually transmitted diseases: three hundred and thirty-three million new, curable cases in 1995. World Health Organization press release 64. Geneva, Switzerland: World Health Organization; 1995. [Google Scholar]
- 44.Winstanley F P, Blackwell C C, Tan E L, Patel P V, Parsons N J, Martin P M, Smith H. Alteration of pyocin-sensitivity pattern of Neisseria gonorrhoeae is associated with induced resistance to killing by human serum. J Gen Microbiol. 1984;130:1303–1306. doi: 10.1099/00221287-130-5-1303. [DOI] [PubMed] [Google Scholar]