Abstract
目的
探讨非小细胞肺癌(NSCLC)细胞外泌体源性FZD10在血管生成中的作用及其机制。
方法
采用超速离心法分离外泌体,并利用Western blot和RT-qPCR技术分析NSCLC细胞(95D和H1299)、正常人支气管上皮细胞(BEAS-2B)及其外泌体中FZD10的表达;通过转染FZD10-siRNA敲低FZD10表达,用FZD10未敲低和敲低的NSCLC细胞外泌体分别处理HUVEC细胞,利用体外血管生成实验观察其成管能力,采用ELISA和RT-qPCR技术分析血管生成相关因子VEGFA和Ang-1的表达;进一步利用Western blot分析外泌体源性FZD10对信号通路PI3K、Erk1/2和YAP/TAZ激活的影响。
结果
同BEAS-2B细胞及其外泌体相比较,FZD10在95D和H1299细胞及其外泌体中高表达(P < 0.01);95D和H1299细胞来源的外泌体可促进HUVEC的微管形成及VEGFA、Ang-1的蛋白分泌、mRNA的表达(P < 0.01),但在95D和H1299细胞敲低FZD10后这些效果受到抑制。FZD10的敲低可抑制PI3K、Erk1/2信号通路的激活,但对YAP/TAZ信号通路的影响不显著。
结论
NSCLC细胞外泌体源性FZD10可促进体外血管生成,其机制可能与PI3K、Erk1/2信号通路的激活有关。
Keywords: 外泌体, FZD10, 血管生成, 非小细胞肺癌
Abstract
Objective
To investigate the effect of exosomal FZD10 derived from non-small cell lung cancer (NSCLC) cells on angiogenesis of human umbilical venous endothelial cells (HUVECs) and explore the possible mechanism.
Methods
We analyzed the expression of FZD10 in two NSCLC cell lines (95D and H1299 cells), normal human bronchial epithelial cells (BEAS-2B cells) and their exosomes isolated by ultracentrifugation. Cultured HUVECs were treated with the exosomes derived from NSCLC cells or NSCLC cells transfected with FZD10-siRNA, and the changes in tube formation ability of the cells were analyzed using an in vitro angiogenesis assay. ELISA was performed to determine the concentration of VEGFA and Ang-1 in the conditioned media of HUVECs, and RT-qPCR was used to analyze the mRNA levels of VEGFA and Ang-1 in the HUVECs. The effects of exosomal FZD10 on the activation of PI3K, Erk1/2 and YAP/TAZ signaling pathways were evaluated using Western blotting.
Results
Compared with BEAS-2B cells and their exosomes, 95D and H1299 cells and their exosomes all expressed high levels of FZD10 (P < 0.01). The exosomes derived from 95D and H1299 cells significantly enhanced tube formation ability and increased the expressions of VEGFA and Ang-1 protein and mRNA in HUVECs (P < 0.01), but FZD10 knockdown in 95D and H1299 cells obviously inhibited these effects of the exosomes. Exosomal FZD10 knockdown suppressed the activation of PI3K and Erk1/2 signaling pathways, but had no obvious effect on the activation of YAP/TAZ signaling pathway.
Conclusion
Exosomal FZD10 derived from NSCLC cells promotes HUVEC angiogenesis in vitro, the mechanism of which may involve the activation of PI3K and Erk1/2 signaling pathways.
Keywords: exosomes, FZD10, angiogenesis, non-small cell lung cancer
肺癌是全球死亡率最高的恶性肿瘤[1, 2],其中非小细胞肺癌(NSCLC)约占所有原发肺癌的85%[3]。尽管新的NSCLC治疗方案(如酪氨酸激酶抑制剂的靶向治疗、免疫疗法等)的临床应用已取得了一定的进展[4-6],但耐药等因素使其疗效还是没有重大突破[7],NSCLC患者的死亡率仍然较高[3, 8]。侵袭、转移是导致NSCLC患者死亡的关键因素[9, 10],而血管生成在NSCLC侵袭、转移中具有重要作用,而且近年来发现外泌体可作为细胞间信息交流的媒介参与NSCLC侵袭[11]、转移[12-14]及血管生成[14],从而调控NSCLC的发展[11]。
Frizzled-10(FZD10)是G蛋白偶联受体中的一种亚型,在包括肺鳞癌等多种肿瘤组织和细胞中高表达[15-17]。研究发现FZD10可以通过外泌体为介质促进肿瘤细胞增殖[18],而且FZD10还被证实在中枢神经系统中与血管生成有关[19]。但FZD10在NSCLC血管生成中的作用尚不清楚,外泌体源性FZD10对肿瘤血管生成的影响还未见报道。因此,本文拟探讨NSCLC细胞来源外泌体中的FZD10(NSCLC细胞外泌体源性FZD10)对血管生成的影响,以期为NSCLC的防治提供潜在靶点。
1. 材料和方法
1.1. 细胞株及细胞培养
NSCLC细胞株95D(中国科学院细胞库),NSCLC细胞株H1299和人脐静脉内皮细胞(HUVEC)(ATCC),正常人支气管上皮细胞(BEAS-2B)(赛百慷(上海)生物技术股份有限公司)。95D、H1299和BEAS-2B细胞使用RPMI 1640+10%胎牛血清(去外泌体血清)+ 1%青霉素-链霉素培养,HUVEC使用DMEM+10%胎牛血清(SBI去外泌体血清)+1%青霉素-链霉素培养,所有细胞都在37 ℃、5% CO2的条件下传代培养,将处于指数生长期的细胞用作后续实验。
1.2. 主要仪器
高速冷冻离心机(HITACHI);超高速冷冻离心机Optima XPN-100(BECKMAN);ABI7500型实时荧光定量PCR仪(Thermo);荧光倒置显微镜TE2000-Nikon公司);Jem-1400型透射电子显微镜(Jeol)。
1.3. 主要试剂
兔抗人PI3K抗体、兔抗人p-PI3K抗体、兔抗人GAPDH抗体、兔抗人YAP/TAZ抗体、兔抗人p-YAP抗体、兔抗人CD9抗体、兔抗人Grp94抗体(CST);FZD10-siRNA系列试剂及转染试剂DharmaFECTTM Transfection Reagents-SiRNA(Thermo);In vitro angiogenesis assay kit ECM 62(Chemicon Millipore);Angiopoietin-1 (Ang-1) human ELISA kit(RD);VEGFA Human ELISA Kit、小鼠抗人CD81抗体、兔抗人FZD10抗体、兔抗人TSG101(Abcam);PKH26红色荧光染料(Sigma);兔抗人Erk1/2抗体、兔抗人p-Erk1/2抗体、羊抗兔IgG、HRP标记抗体、DAPI染料、裂解液RIPA、Trizol(碧云天生物科技公司);Matrigel基质胶(BD);SBI去外泌体血清(艾博瑞生物科技公司);SYBR® Premix Ex TaqTM Ⅱ(Tli RNaseH Plus)和PrimeScriptTM RT reagent Kit with gDNA Eraser(Perfect Real Time)(TaKaRa)。
1.4. 细胞转染
分别将Opti-MEN基础培养基、5 μmol/L FZD10-siRNA和转染试剂按照表 1的体积混合,并按照转染试剂说明书将FZD10-siRNA瞬时转染入95D、H1299细胞,并设转染对照组。
表 1.
不同规格培养皿所需的转染试剂体积
Volume of transfection reagents in the different culture plates
| Plates (well/plate) | Area (cm2/well) | Tube1 (μL/well) | Tube2 (μL/well) | (μL/well) | |||||
| 5 μmol/L siRNA | Opti-MEN | DharmaTECT | Opti-MEN | Complete media | Total volume | ||||
| 12 | 4 | 5 | 95 | 0.5 | 99.5 | 800 | 1000 | ||
| 6 | 10 | 10 | 190 | 1 | 199 | 1600 | 2000 | ||
| 1 | 60 | 40 | 760 | 4 | 796 | 6400 | 8000 | ||
1.5. 外泌体提取
将收集的细胞上清液通过2000 g离心30 min,取上清液再用10 000 g离心45 min,再取其上清液用0.22 μm过滤器过滤;过滤后的上清液用超高速(130 000 g)离心两次,70 min/次,弃掉上清液,外泌体沉淀用PBS重悬(一般情况,100 mL细胞培养上清液提取的外泌体用100 μL的PBS缓冲液重悬)。收集的外泌体可直接用于后续实验,或置-80 ℃冰箱保存1月。
1.6. 外泌体形态观察
取10 μL外泌体悬液滴入铜网,室温静置3 min,用等体积的2%醋酸双氧铀染液染色2 min,晾干30 min,使用电压80 kV的JEM-1400型透射电子显微镜观察外泌体的形态,并用Gatan832 CCD相机拍照保存。
1.7. 外泌体内化
将95D、H1299、BEAS-2B细胞外泌体分别和PKH26染料混匀,室温下避光静置5 min,加入等体积的10% BSA终止反应;再重新提取已标记的外泌体并重悬,将标记的外泌体与贴壁的HUVEC细胞共培养6 h,用4%多聚甲醛固定;再用DAPI染料进行细胞核染色,37 ℃温箱中静置10 min,在避光条件下,用荧光显微镜观察外泌体被细胞内化的情况。
1.8. 体外成管实验
根据Chemicon(Millipore)的In vitro angiogenesis assay kit说明书操作。主要步骤:将基质胶放在4 ℃冰箱过夜融化并按照10×稀释液: 基质胶=1∶9的比例稀释,用预冷的移液器取50 μL的基质胶包被96孔板,静置1 h使其凝固;预先按照2×106/孔将HUVEC细胞接种在六孔板中,用50 μg不同细胞来源的外泌体(NSCLC细胞、FZD10敲低的NSCLC细胞来源的外泌体)分别处理HUVEC细胞48 h,消化、重悬后以细胞总数为8000~10 000且细胞悬液体积不超过150 μL的HUVEC细胞接种到已凝固的基质胶上,培养6~8 h,于倒置显微镜下观察成管情况,使用软件Scion Image分析结果并计算成管总长度。
1.9. Western blot
使用强RIPA分别裂解细胞及其外泌体,提取总蛋白,使用BCA法检测蛋白原液浓度,再加热变性(CD81无需加热变性),除Erk1/2、p-Erk1/2分析取12 μg外,其余取50 μg上样、电泳(60 V浓缩30 min,再100 V电泳约1.5 h)、转膜、电转(100 V电转1.5 h)、封闭。加入一抗4 ℃孵育过夜(除兔抗人Erk1/2抗体和兔抗人p-Erk1/2抗体按1∶2000稀释外,其他一抗均按1∶1000稀释),洗涤后,再加入二抗室温孵育1~2 h(二抗全部按1∶2000稀释)。用ECL化学发光检测信号,条带结果用Image J软件进行灰度值分析。将GAPDH作为内参对照。
1.10. RT-qPCR
用Trizol分别裂解95D、H1299、BEAS-2B细胞及不同细胞来源的外泌体预处理的HUVEC,提取总RNA,测定A260/A280,通过该比值判断RNA样品的纯度并计算所提取样品的RNA浓度,计算公式为:CRNA=A260×0.33×2000 μg/mL。将纯度符合要求(A260/A280为1.8~2.0)的样品,按照说明书进行去基因组DNA后,在ABI7500扩增仪上进行RT-qPCR反应。扩增引物序列见表 2,所有引物全部由生工生物工程(上海)股份有限公司合成。逆转录(RT)条件:37 ℃ 15 min、85 ℃ 5 s;qPCR反应条件:42 ℃ 5 min、95 ℃ 10 s, 紧接着40个循环(95 ℃ 5 s、60 ℃ 31 s)。结果通过2-△△Ct值对扩增产物进行相对定量分析,将GAPDH作为内参对照。
表 2.
引物序列
Sequences of the primers
| Genes | Sequences (5′→ 3′) | GenBank No. |
| FZD10 | F: AGCAGGTCTCTACCCCCATC | NM_007197.4 |
| R: TAATCGGGGAGCACTTGAGC | ||
| VEGFA | F: CTTTCTGCTGTCTTGGGT | NM_001025366.3 |
| R: ACTTCGTGATGATTCTGCC | ||
| Ang-1 | F: CCAGTACAACACAAACGCTCT | NM_001146.5 |
| R: TCTCCGACTTCATGTTTTCCAC | ||
| GAPDH | F: ATGAATGGGCAGCCGTTAGG | NM_001256799.2 |
| R: CCCAATACGACCAAATCAGAGAT |
1.11. ELISA实验
按照说明书,采用ELISA法检测NSCLC细胞、FZD10敲低的NSCLC细胞来源的外泌体分别处理后的HUVEC条件培养基中VEGFA和Ang-1的浓度,检测前需稀释样品到合适浓度,再在波长450 nm的分光光度计中读取吸光度值A450 nm,使用软件ELISA CALC分析数据并计算浓度值。
1.12. 统计学分析
使用GraphPad Prism 6.0软件进行统计学分析。所有实验都重复3次,数据均用均数±标准差表示,两组间比较采用独立样本t检验;多组间比较采用单因素方差分析,多组组间两两比较方差齐时采用LSD检验。P < 0.05表示差异具有统计学意义。
2. 结果
2.1. 外泌体的鉴定
通过电子显微镜观察发现,采用超速离心法所分离的物质为单个或聚集成团的膜性结构囊泡,呈现较典型的外泌体形态特征(图 1A);利用Western blot鉴定发现,外泌体相关标志物CD81、CD9和TSG101均可在所提取的物质中表达,而内质网分子伴侣相关蛋白Grp94(非外泌体相关标志物)却不存在于所提取的物质中(图 1B)。说明本研究利用超速离心法从细胞上清液中分离出的物质是外泌体。
图 1.
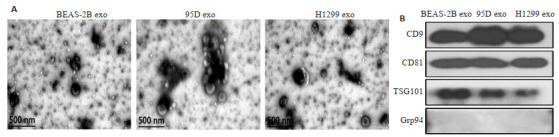
外泌体的鉴定
Identification of the isolated exosomes. A: Results of transmission electron microscopy. B: Results of Western blotting.
2.2. FZD10在NSCLC细胞及其外泌体中的表达
Western blot结果显示,与BEAS-2B细胞及其外泌体相比较,95D、H1299细胞中的FZD10蛋白水平升高(P < 0.01),且两株NSCLC细胞来源的外泌体中也均有FZD10蛋白的高表达(图 2A、B);RT-qPCR结果显示,两株NSCLC细胞中的FZD10 mRNA表达也高于对照细胞BEAS-2B(P < 0.01,图 2C)。
图 2.
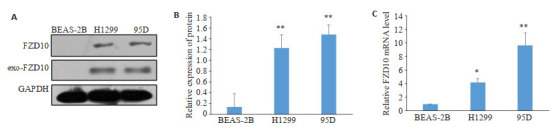
FZD10在NSCLC细胞及其外泌体中的表达
Expression of FZD10 in NSCLC cells and their exosomes. A, B: Western blotting for analyzing FZD10 protein expression. C: RT-qPCR for analyzing FZD10 mRNAlevels. *P < 0.05, **P < 0.01 vs BEAS-2B group.
2.3. NSCLC细胞来源的外泌体促进体外血管生成
外泌体内化结果显示,被标记的外泌体基本绕HUVEC细胞核单个或多个分布(图 3),HUVEC均可内化95D、H1299和BEAS-2B三种细胞来源的外泌体;体外成管实验结果显示:与BEAS-2B细胞来源的外泌体处理组相比较,95D、H1299两株NSCLC细胞来源的外泌体处理组的HUVEC总管长度明显增加(P < 0.01,图 4A、B)。ELISA检测和RT-qPCR分别从蛋白水平和mRNA转录水平证实,95D、H1299两株NSCLC细胞来源的外泌体明显促进了HUVEC中的血管生成相关标志物VEGFA和Ang-1的蛋白分泌和mRNA表达(P < 0.01,图 4C~E)。
图 3.
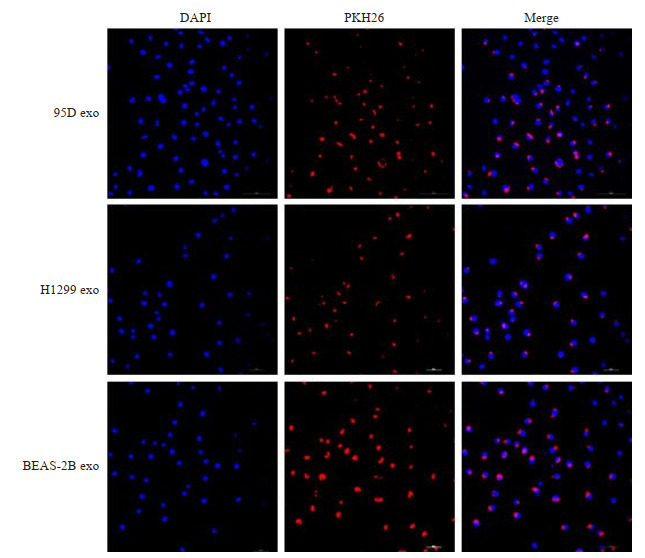
外泌体内化的结果
Results of exosome internalization (Original magnification×200).
图 4.
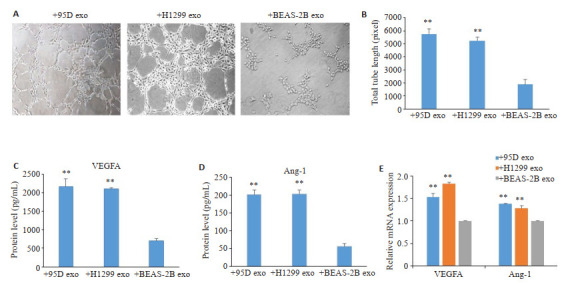
NSCLC细胞来源的外泌体对体外血管生成的影响
Effect of the exosomes derived from NSCLC cells on angiogenesis of HUVECs in vitro. A: Tube formation of the HUVECs was observed under a phase-contrast microscope (×200). B: Total tube length measured by Scion Image software. C, D: ELISA for analyzing VEGFA (C) and Ang-1 (D) concentrations in the conditioned media of HUVECs. E: RT-qPCR for analysis of the mRNA levels of VEGFA and Ang-1 in HUVECs. In+95D exo, +H1299 exo and +BEAS-2B exo groups, the HUVECs were treated with the exosomes derived from 95D, H1299 and BEAS-2B cells, respectively. **P < 0.01 vs +BEAS-2B exo group.
2.4. NSCLC细胞来源的外泌体通过FZD10促进体外血管生成
FZD10表达在95D、H1299细胞中被成功敲低(图 5A),FZD10敲低的NSCLC细胞来源的外泌体处理组(95D FZD10-KD exo、H1299 FZD10-KD exo)的HUVEC成管能力与FZD10未敲低的NSCLC细胞来源的外泌体处理组(95D exo、H1299 exo)相比明显降低,两组结果的总管长度差异具有统计学意义(P < 0.01,图 5B、C)。ELISA的检测结果显示:与95D exo处理组比较,95D FZD10-KD exo处理组的细胞条件培养液中血管生成相关标志物VEGFA和Ang-1浓度均明显降低(P < 0.01,图 5D、E)。RT-qPCR的结果也显示,95D FZD10-KD exo处理组细胞的VEGFA和Ang-1 mRNA表达水平也明显低于95D exo处理组(P < 0.01,图 5F)。
图 5.
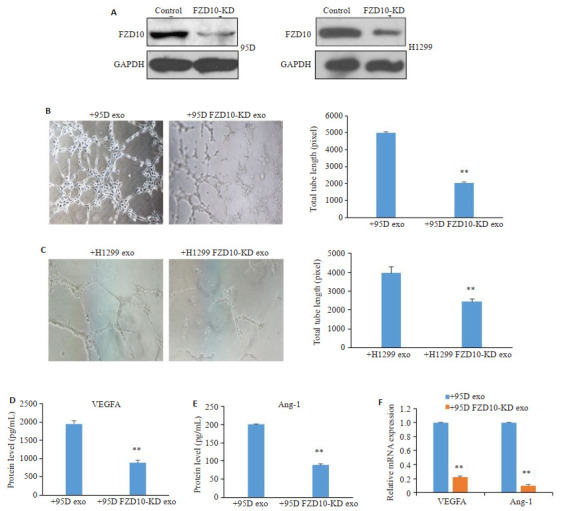
FZD10在NSCLC细胞来源的外泌体对体外血管生成影响中的作用
Role of FZD10 mediating the effect of the exosomes derived from NSCLC cells on angiogenesis of HUVECs in vitro. A: 95D and H1299 cells were transfected with FZD10-siRNA, and Western blotting was performed to identify the efficiency of knockdown. B, C: Tube formation of HUVECs treated with the exosomes derived from FZD10-siRNA-transfected 95D (B) and H1299 cells (C) was observed under a phase-contrast microscope (×200), and the total tube length was measured by Scion Image software. D, E: ELISA was performed to analyze VEGFA (D) and Ang-1 (E) concentration in the conditioned media of HUVECs. F: RT-qPCR was used to analyze the of VEGFA and Ang-1 mRNA levels in HUVECs. In + 95D exo and + 95D FZD10-KD exo groups, the HUVECs were treated with the exosomes derived from 95D and FZD10-siRNA-transfected 95D cells, respectively; the cells were treated likewise in +H1299 exo and +H1299 FZD10-KD exo groups. **P < 0.01 vs +95D exo or +H1299 exo group.
2.5. NSCLC细胞外泌体源性FZD10促进血管生成的分子机制
与95D exo对照组比较,95D FZD10-KD exo处理组p-PI3K和p-Erk1/2的表达明显下调(P < 0.05,图 6A);与H1299 exo对照组比较,H1299 FZD10-KD exo处理组p-PI3K和p-Erk1/2的表达降低(P < 0.05,图 6B)。但YAP/TAZ信号通路相关分子表达差异不显著(P < 0.05,图 6)。
图 6.
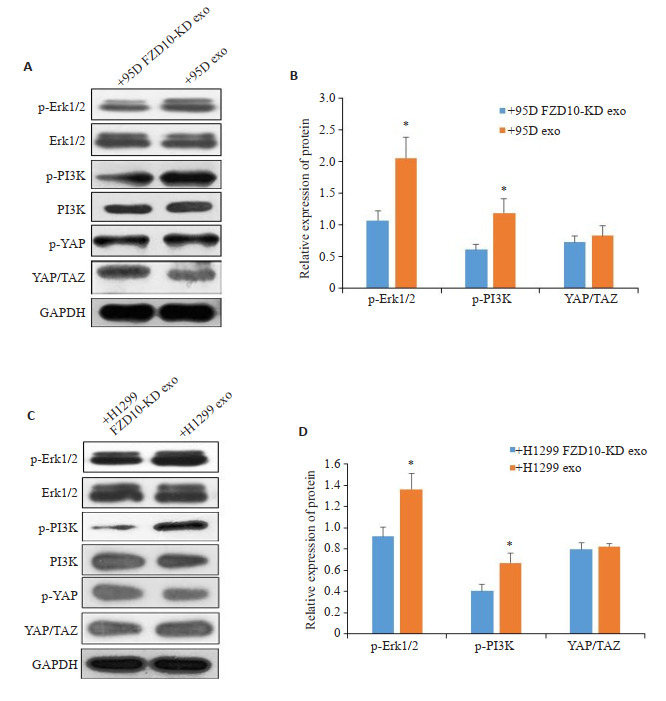
FZD10敲低对PI3K、Erk1/2、YAP/TAZ信号通路激活的影响
Effect of FZD10 knockdown on the activation of PI3K, Erk1/2 and YAP/TAZ signaling pathways. A, C: Western blotting for detecting protein expressions in 95D cells (A) and H1299 cells (C). B, D: Quantitative analysis of the protein expressions. *P < 0.05 vs +95D FZD10-KD exo or +H1299 FZD10-KD exo group.
3. 讨论
外泌体是由体内多种活细胞分泌的直径在30~150 nm的细胞外囊泡[30],其可作为细胞间通讯的重要桥梁,在NSCLC生长[12, 22]、侵袭[11]、转移[12, 13]、上皮-间质转化[21]、耐药[22]等中起重要作用,而且还发现外泌体可参与调控NSCLC血管生成[14]。FZD10是经典通路Wnt/β-catenin中的重要受体,其为影响肿瘤治疗效果的关键分子,可能为肿瘤防治的重要靶点[23-25]。近年发现,结肠癌细胞株来源的外泌体通过递送FZD10诱导了正常结肠上皮细胞株发生上皮-间质转化[26],而且FZD10外囊泡血浆浓度还与结直肠癌和胃癌发展有关[27]。这些报道说明FZD10可能可通过外泌体递送在肿瘤发展中发挥作用,但FZD10是否可通过外泌体递送在NSCLC发展中发挥作用还未见文献报道。
本研究发现FZD10不仅在NSCLC细胞(95D和H1299)中高表达,而且在其外泌体中也呈现高表达。与正常人支气管上皮细胞BEAS-2B来源的外泌体相比较,95D和H1299细胞来源的外泌体可明显促进HUVEC细胞管状样结构的形成及血管生成相关因子VEGF、Ang-1的蛋白分泌和mRNA表达(P < 0.01),说明NSCLC细胞(95D和H1299)来源的外泌体可促进血管生成;但FZD10敲低后,95D和H1299细胞来源的外泌体对HUVEC细胞管状样结构的形成及VEGF、Ang-1的蛋白分泌和mRNA表达的影响也受到了抑制(P < 0.01),说明FZD10可通过外泌体递送介导NSCLC血管生成,从而在NSCLC发展中起重要作用。因此,FZD10可能是NSCLC防治的潜在靶点。
为进一步探讨外泌体源性FZD10在NSCLC血管生成的作用机制,本研究对PI3K、Erk1/2、YAP/TAZ多条信号通路进行了初步分析。最近有研究报道在结肠癌和胃癌中外泌体源性FZD10通过Erk1/2信号通路增强了Ki-67的表达[28]。本研究结果也显示,FZD10敲低的95D和H1299细胞来源的外泌体可抑制PI3K、Erk1/2信号通路的激活,说明PI3K、Erk1/2信号通路可能参与了外泌体源性FZD10促进NSCLC血管生成的过程。有研究在中枢神经系统血管生成中发现FZD10以Gα 12/13依赖的方式诱导了YAP/TAZ的转录激活[19],但本研究在对FZD10敲低和未敲低95D和H1299细胞来源的外泌体YAP/TAZ及p-YAP表达的比较分析中,未发现YAP/TAZ信号通路参与外泌体源性FZD10促进NSCLC血管生成过程的调控,我们的结果与中枢神经系统模型结果的差异可能由于细胞种类的不同及肿瘤微环境的复杂性所致。
基于目前FZD10在NSCLC研究罕见,尤其是NSCLC细胞外泌体源性FZD10在血管生成中的研究在我们所能检索的资料范围之内未见报道,本研究不仅明确了FZD10在NSCLC细胞及其外泌体中高表达,而且还发现NSCLC细胞来源的FZD10通过外泌体递送介导体外血管生成。本研究的优势是将FZD10与血管生成之间的作用通过外泌体将两者联系起来,可进一步阐明FZD10在NSCLC发展中的作用机制,并可为NSCLC防治潜在靶点FZD10的发现提供实验依据。
Biography
吴小凤,硕士,技师,E-mail: 977830984@qq.com
Funding Statement
国家自然科学基金(81372511); 广东省基础与应用基础研究基金(2019A1515011081); 广东省“扬帆计划”培养高层次人才项目(201635011); 广东省普通高校特色创新类项目(自然科学) (2022KTSCX048)
Supported by National Natural Science Foundation of China (81372511)
Contributor Information
吴 小凤 (Xiaofeng WU), Email: 977830984@qq.com.
唐 旭东 (Xudong TANG), Email: tangxudong2599@126.com.
References
- 1.Yuan JM, Sun Y, Bu X, et al. Global, regional and national burden of lung cancer and its attributable risk factors in 204 countries and territories, 1990-2019. Eur J Cancer Prev. 2022;31(3):253–9. doi: 10.1097/CEJ.0000000000000687. [DOI] [PubMed] [Google Scholar]
- 2.Sung H, Ferlay J, Siegel RL, et al. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CACancer J Clin. 2021;71(3):209–49. doi: 10.3322/caac.21660. [DOI] [PubMed] [Google Scholar]
- 3.Ma ZJ, Chen GY, Chen YQ, et al. miR-937-3p promotes metastasis and angiogenesis and is activated by MYC in lung adenocarcinoma. Cancer Cell Int. 2022;22(1):31–8. doi: 10.1186/s12935-022-02453-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Chu TQ, Zhang W, Zhang B, et al. Efficacy and safety of first-line anlotinib-based combinations for advanced non-small cell lung cancer: a three-armed prospective study. Transl Lung Cancer Res. 2022;11(7):1394–404. doi: 10.21037/tlcr-22-438. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Zhao PF, Zhen HC, Zhao H, et al. Efficacy and safety of adjuvant EGFR-TKIs for resected non-small cell lung cancer: a systematic review and meta-analysis based on randomized control trials. BMC Cancer. 2022;22(1):328–37. doi: 10.1186/s12885-022-09444-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Song ZZ, Ren GY, Hu L, et al. Two case reports of non-small cell lung cancer patients harboring acquired EGFR T790M-Cis-C797S benefit from immune checkpoint inhibitor combined with platinum-based doublet chemotherapy. Ann Transl Med. 2022;10(12):719–28. doi: 10.21037/atm-22-2436. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Sun QY, Wei XZ, Wang ZL, et al. Primary and acquired resistance against immune check inhibitors in non-small cell lung cancer. Cancers. 2022;14(14):3294–305. doi: 10.3390/cancers14143294. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Muthusamy B, Pennell N. Chemoimmunotherapy for EGFR-mutant NSCLC: still no clear answer. J Thorac Oncol. 2022;17(2):179–81. doi: 10.1016/j.jtho.2021.11.012. [DOI] [PubMed] [Google Scholar]
- 9.Zhong YF, She YL, Deng JJ, et al. Deep learning for prediction of N2 metastasis and survival for clinical stage Ⅰ non-small cell lung cancer. Radiology. 2022;302(1):200–11. doi: 10.1148/radiol.2021210902. [DOI] [PubMed] [Google Scholar]
- 10.Zhou HL, Guan Q, Hou XY, et al. Epithelial-mesenchymal reprogramming by KLF4-regulated Rictor expression contributes to metastasis of non-small cell lung cancer cells. Int J Biol Sci. 2022;18(13):4869–83. doi: 10.7150/ijbs.73548. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.He YZ, Liu YH, Cha NE, et al. Exosomal circ_0048856 derived from non-small cell lung cancer contributes to aggressive cancer progression through downregulation of miR-1287-5p. Pathol Res Pract. 2022;232:153659–66. doi: 10.1016/j.prp.2021.153659. [DOI] [PubMed] [Google Scholar]
- 12.Wang R, Liu HL, Dong MQ, et al. Exosomal hsa_circ_0000519 modulates the NSCLC cell growth and metastasis via miR-1258/RHOV axis. Open Med (Wars) 2022;17(1):826–40. doi: 10.1515/med-2022-0428. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Rizwan MN, Ma YX, Nenkov M, et al. Tumor-derived exosomes: key players in non-small cell lung cancer metastasis and their implication for targeted therapy. Mol Carcinog. 2022;61(3):269–80. doi: 10.1002/mc.23378. [DOI] [PubMed] [Google Scholar]
- 14.Ma ZJ, Wei K, Yang FM, et al. Tumor-derived exosomal miR-3157-3p promotes angiogenesis, vascular permeability and metastasis by targeting TIMP/KLF2 in non-small cell lung cancer. Cell Death Dis. 2021;12(9):840–51. doi: 10.1038/s41419-021-04037-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Gugger M, White R, Song SS, et al. GPR87 is an overexpressed G-protein coupled receptor in squamous cell carcinoma of the lung. Dis Markers. 2008;24(1):41–50. doi: 10.1155/2008/857474. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Nagayama S, Yamada E, Kohno Y, et al. Inverse correlation of the up-regulation of FZD10 expression and the activation of beta-catenin in synchronous colorectal tumors. Cancer Sci. 2009;100(3):405–12. doi: 10.1111/j.1349-7006.2008.01052.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.陈 昌望, 王 璋, 周 玲, et al. FZD10在胃癌中的表达以及对胃癌细胞的影响. 海南医学院学报. 2019;25(11):853–7. [Google Scholar]
- 18.Scavo MP, Depalo N, Rizzi F, et al. FZD10 carried by exosomes sustains cancer cell proliferation. Cells. 2019;8(8):777–86. doi: 10.3390/cells8080777. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Hot B, Valnohova J, Arthofer E, et al. FZD10-Gα13 signalling axis points to a role of FZD10 in CNS angiogenesis. Cell Signal. 2017;32:93–103. doi: 10.1016/j.cellsig.2017.01.023. [DOI] [PubMed] [Google Scholar]
- 20.Liu JY, Ren LW, Li S, et al. The biology, function, and applications of exosomes in cancer. Acta Pharm Sin B. 2021;11(9):2783–97. doi: 10.1016/j.apsb.2021.01.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Jouida A, O'Callaghan M, Mc Carthy C, et al. Exosomes from EGFR-mutated adenocarcinoma induce a hybrid EMT and MMP9-dependant tumor invasion. Cancers. 2022;14(15):3776–85. doi: 10.3390/cancers14153776. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Shi L, Zhu WL, Huang YY, et al. Cancer-associated fibroblastderived exosomal microRNA-20a suppresses the PTEN/PI3K-AKT pathway to promote the progression and chemoresistance of nonsmall cell lung cancer. Clin Transl Med. 2022;12(7):e989–95. doi: 10.1002/ctm2.989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Chen JY, Zhang F, Ren XS, et al. Targeting fatty acid synthase sensitizes human nasopharyngeal carcinoma cells to radiation via downregulating frizzled class receptor 10. Cancer Biol Med. 2020;17(3):740–52. doi: 10.20892/j.issn.2095-3941.2020.0219. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Giraudet AL, Cassier PA, Iwao-Fukukawa C, et al. A first-in-human study investigating biodistribution, safety and recommended dose of a new radiolabeled MAb targeting FZD10 in metastatic synovial sarcoma patients. BMC Cancer. 2018;18(1):646. doi: 10.1186/s12885-018-4544-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Sudo H, Tsuji AB, Sugyo A, et al. FZD10-targeted α-radioimmunotherapy with 225 Ac-labeled OTSA101 achieves complete remission in a synovial sarcoma model. Cancer Sci. 2022;113(2):721–32. doi: 10.1111/cas.15235. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Scavo MP, Rizzi F, Depalo N, et al. A possible role of FZD10 delivering exosomes derived from colon cancers cell lines in inducing activation of epithelial-mesenchymal transition in normal colon epithelial cell line. Int J Mol Sci. 2020;21(18):6705–12. doi: 10.3390/ijms21186705. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Scavo MP, Cigliano A, Depalo N, et al. Frizzled-10 extracellular vesicles plasma concentration is associated with tumoral progression in patients with colorectal and gastric cancer. J Oncol. 2019;2019:2715968–77. doi: 10.1155/2019/2715968. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Scavo MP, Rizzi F, Depalo N, et al. Exosome released FZD10 increases ki-67 expression via phospho-ERK1/2 in colorectal and gastric cancer. Front Oncol. 2021;11:730093–102. doi: 10.3389/fonc.2021.730093. [DOI] [PMC free article] [PubMed] [Google Scholar]


