Abstract
Background
Sodium-glucose co-transporter 2 inhibitors (SGLT2-i) have shown significant cardiovascular benefits in patients with and without type 2 diabetes mellitus (T2DM). They have also gained interest for their potential anti-arrhythmic role and their ability to reduce the occurrence of atrial fibrillation (AF) and ventricular arrhythmias (VAs) in T2DM and heart failure patients.
Objectives
To investigate in-hospital new-onset cardiac arrhythmias in a cohort of T2DM patients presenting with acute myocardial infarction (AMI) treated with SGLT2-i vs. other oral anti-diabetic agents (non-SGLT2-i users).
Methods
Patients from the SGLT2-I AMI PROTECT registry (NCT05261867) were stratified according to the use of SGLT2-i before admission for AMI, divided into SGLT2-i users vs. non-SGLT2-i users. In-hospital outcomes included the occurrence of in-hospital new-onset cardiac arrhythmias (NOCAs), defined as a composite of new-onset AF and sustained new-onset ventricular tachycardia (VT) and/or ventricular fibrillation (VF) during hospitalization.
Results
The study population comprised 646 AMI patients categorized into SGLT2-i users (111 patients) and non-SGLT2-i users (535 patients). SGLT2-i users had a lower rate of NOCAs compared with non-SGLT2-i users (6.3 vs. 15.7%, p = 0.010). Moreover, SGLT2-i was associated with a lower rate of AF and VT/VF considered individually (p = 0.032). In the multivariate logistic regression model, after adjusting for all confounding factors, the use of SGLT2-i was identified as an independent predictor of the lower occurrence of NOCAs (OR = 0.35; 95%CI 0.14–0.86; p = 0.022). At multinomial logistic regression, after adjusting for potential confounders, SGLT2-i therapy remained an independent predictor of VT/VF occurrence (OR = 0.20; 95%CI 0.04–0.97; p = 0.046) but not of AF occurrence.
Conclusions
In T2DM patients, the use of SGLT2-i was associated with a lower risk of new-onset arrhythmic events during hospitalization for AMI. In particular, the primary effect was expressed in the reduction of VAs. These findings emphasize the cardioprotective effects of SGLT2-i in the setting of AMI beyond glycemic control.
Trial registration
Data are part of the observational international registry: SGLT2-I AMI PROTECT. ClinicalTrials.gov, identifier: NCT05261867.
Keywords: sodium-glucose cotransporter 2 inhibitors (SGLT2-i), acute myocardial infarction, atrial fibrillation, ventricular arrhythmias, ventricular tachycardia, hyperglycemia
Introduction
Cardiac arrhythmias, specifically atrial fibrillation (AF) and ventricular arrhythmias (VAs), are common in the early phase of acute myocardial infarction (AMI), in both ST-segment elevation (STEMI) and non-ST-segment elevation myocardial infarction (NSTEMI) (1–3). Ischemia in myocardial infarction induces heterogeneity in excitability, refractoriness, and conduction, thus creating the substrate for the onset of potentially life-threatening arrhythmias (4–6). AF as new-onset arrhythmia occurs in about 5% of cases after STEMI, and the risk of new-onset AF in AMI patients increases by 60–77% (1, 2, 7). VAs, including ventricular tachycardia (VT) and/or ventricular fibrillation (VF), may occur at any time during AMI, especially in the inpatient phase, and represent a leading complication in this setting (1, 8). Although the incidence of VAs has declined in the hospital phase, mainly due to prompt revascularization with primary percutaneous coronary intervention (pPCI) and the early administration of medical therapy, the risk of arrhythmias, cardiac arrest, and sudden cardiac death remains high (1, 9–11). Moreover, the occurrence of VAs in early phase of AMI has been associated with an increased death rate during in-hospital period and 6-month follow-up (5, 12).
Sodium-glucose cotransporter 2 inhibitors (SGLT2-i) are a new class of drugs that act by inhibiting the reabsorption of sodium and glucose in the proximal tubules of the kidney. They were introduced as oral anti-diabetic drugs in patients with type 2 diabetes mellitus (T2DM), and later showed strong evidence of cardiovascular benefits in diabetic and non-diabetic patients with atherosclerotic cardiovascular disease and heart failure (HF), irrespective of the ejection fraction (13–16). In T2DM patients, the SGLT2-i have been shown to improve cardiac function (17), reduce hospitalizations, and positively impact prognosis in patients with stable ischemic heart disease undergoing surgical revascularization (18). In addition, they have an ameliorative effect on sympathetic tone, potentially reducing arrhythmic burden (19). Patients with AMI, even those with T2DM, are prone to developing new-onset arrhythmias due to autonomic, electrical, and structural remodeling, and glycemic fluctuations (20, 21). Although there is evidence for a reduction in new-onset arrhythmic events in patients treated with SGLT2-i, previous studies yielded inconclusive results and a variable association between SGLT2-i treatment and cardiac arrhythmias (22–24). Furthermore, the efficacy of SGLT2-i in preventing arrhythmic events in specific settings of diabetic patients with AMI remains unclear. To address this uncertainty, we evaluated the effect of SGLT2-i on the occurrence of new arrhythmic events during hospitalization in diabetic patients presenting with AMI.
Methods
Study design and population
The Cardioprotective Effect of Sodium-Glucose Cotransporter-2 Inhibitors in Diabetic Patients With Acute Myocardial Infarction registry (SGLT2-I AMI PROTECT, ClinicalTrials.gov, identifier: NCT05261867) is a multicenter international observational registry that collects data on consecutive diabetic patients admitted with AMI, both NSTEMI and STEMI, undergoing percutaneous coronary intervention (PCI), between January 2018 and November 2021. The study design and main results have been previously reported (25). Based on admission drug therapy, patients were divided into two groups: SGLT2-i users, if they were admitted to chronic SGLT2-i therapy (defined as ongoing treatment for at least 3 months before hospitalization) and non-SGLT2-i users, if they received other oral anti-diabetic (OAD) treatment. Patients on insulin therapy before hospitalization or with incomplete information on medical therapy at admission were excluded. Patients who started SGLT2-i therapy after the acute index event, or patients with type I diabetes mellitus, treated with coronary artery bypass grafting (CABG) as a revascularization strategy, with severe valvular heart disease, severe anemia, a history of bleeding, pulmonary embolism, glomerular filtration rate < 30 ml/min/1.73 m2 and malignancies were excluded. Patients with more than 20% missing values in the collected data were also excluded due to potential bias. All patients were treated with optimal medical therapy, in accordance with current guidelines. The current study was performed in accordance with the guidelines of the Declaration of Helsinki, and all patients provided informed consent.
Study endpoints
The primary endpoint was the occurrence of in-hospital new-onset cardiac arrhythmias (NOCAs), defined as a composite of new-onset AF and new-onset sustained VT/VF during hospitalization. New-onset of AF or sustained VT/VF was considered any detection of AF or VT/VF, as defined by international guidelines, using telemetry recording or electrocardiogram throughout hospitalization from admission to discharge in patients with no previous history of AF or established tachyarrhythmia disorders. An analysis of the individual components (AF and VT/VF) of the composite endpoint was performed.
Statistical analysis
Continuous variables are presented as mean and standard deviation (SD) or median and interquartile range, and categorical variables as numbers and percentages. The normal distribution was first assessed using the Kolmogorov–Smirnov goodness-of-fit test. Categorical data were compared using either the Pearson chi-square test or the Fisher exact test when appropriate, and continuous variables were compared using the non-parametric Mann–Whitney U-test, Student's t-test, or a one-way ANOVA followed by Tukey-Kramer post hoc correction, as appropriate. Univariate analysis was performed to identify variables associated with arrhythmic events. All variables found to have a P-value ≤0.1 in the univariate evaluation were considered to be candidates for a subsequent multivariate logistic regression analysis. We performed multivariate hierarchical binary logistic regression and multinomial logistic regression models to assess the predictability of variables on the occurrence of composite endpoints, and each of the components is presented as odds ratios (ORs) and 95% confidence intervals (CIs). Statistical significance was set as P < 0.05. All analyses were performed with the Statistical Package for the Social Sciences software version 25 (SPSS, IBM®, Armonk, New York) and R software (CRAN® 3.3.4).
Results
Study population and baseline characteristics
In the SGLT2-i AMI PROTECT registry, 1,118 diabetic patients with AMI were screened. After excluding non-eligible patients 646 patients were considered for the present analysis. According to the use of SGLT2-i before admission for AMI, the study cohort was divided into two groups: SGLT2-i users (n = 111) and non-SGLT2-i users (n = 535). Baseline characteristics are shown in Table 1. The median age of the overall population was 70 years (range 61–79), while the SGLT2-i group was younger, with a median of 66 years (range 59–73). Over three-quarters of the patients enrolled were male, and more than 26% had a history of AMI. There was no difference in cardiovascular risk factors, history of established cardiovascular disease, or drug therapy between groups, yet the use of sulfonylureas was lower in the SGLT2-i user group. With respect to clinical data at admission and procedural data, more than half of patients experienced NSTEMI (equally distributed between the two groups), and there were no differences in terms of Killip class at presentation, history of AF, VT or cardiac arrest, radial vascular access, multivessel disease, or complete revascularization between groups. A summary of the clinical and procedural data is provided in Table 2. No differences were found between SGLT2-i users and non-SGLT2-i users in the occurrence of electrolyte imbalances; no differences in potassium, calcium, and magnesium concentrations that could potentially impact the onset of arrhythmias. Admission heart rate was significantly lower in the group of patients receiving SGLT2-i (Table 2). Although there were no differences in Hb1Ac levels on admission, median admission blood glucose levels were significantly higher in the non-SGLT2-i user group (185 vs. 158 mg/dL, p = 0.007). Furthermore, subcutaneous (s.c.) and intravenous (i.v.) insulin therapy was less frequently used during hospitalization in the SGLT2-i users (51.4 vs. 73.6%, p < 0.01; 15.3 vs. 26.9%, p = 0.010, respectively). Albeit there were marked differences in glycemic control and insulin use, no difference in terms of glycemic variability between the two groups was found. In addition, although there was a trend toward lower mean SD and coefficient of variation (CV) in the group of SGLT2-i users, these differences did not reach significance (SD 44.7 vs. 51.0, p = 0.20; CV 0.25 vs. 0.27, p = 0.23).
Table 1.
Baseline characteristics of study population according to groups eligibility criteria.
| Total | SGLT2-i users | Non-SGLT2-i users | P -value | |
|---|---|---|---|---|
| (N = 646) | (N = 111) | (N = 535) | ||
| Age, median (IQR) | 70 (61–79) | 66 (59–73) | 72 (62–80) | <0.001 |
| Male Sex, n (%) | 498 (77.1) | 90 (81.1) | 405 (75.7) | 0.222 |
| BMI | 27.7 (25–31.3) | 27.1 (24.6–30) | 27.7 (25–31.4) | 0.245 |
| Smoking, n (%) | 370 (57.3) | 67 (60.4) | 303 (56.6) | 0.470 |
| Hypertension, n (%) | 541 (83.7) | 98 (88.3) | 443 (82.8) | 0.154 |
| Dyslipidemia, n (%) | 508 (78.6) | 90 (81.1) | 418 (78.1) | 0.490 |
| PAD, n (%) | 82 (12.7) | 16 (14.4) | 66 (12.3) | 0.550 |
| COPD, n (%) | 90 (13.9) | 15 (13.5) | 75 (14) | 0.889 |
| CKD, n (%) | 58 (9) | 10 (9) | 47 (8.8) | 0.886 |
| Previous TIA/CVA, n (%) | 52 (8) | 10 (9) | 42 (7.9) | 0.683 |
| Previous AMI, n (%) | 169 (26.2) | 30 (27) | 136 (25.4) | 0.724 |
| Previous PCI, n (%) | 183 (28.3) | 35 (31.5) | 144 (26.9) | 0.322 |
| Antiplatelets, n (%) | 321 (49.7) | 60 (54.1) | 261 (48.8) | 0.312 |
| Anticoagulation, n (%) | 55 (8.5) | 6 (5.4) | 49 (9.2) | 0.197 |
| RAAS, n (%) | 378 (58.5) | 69 (62.2) | 309 (57.8) | 0.391 |
| Diuretics, n (%) | 196 (30.3) | 31 (27.9) | 165 (30.8) | 0.543 |
| B-blockers, n (%) | 296 (45.8) | 55 (49.5) | 241 (45) | 0.386 |
| CCB, n (%) | 197 (30.5) | 35 (31.5) | 162 (30.3) | 0.794 |
| Statins, n (%) | 329 (50.9) | 61 (55) | 268 (50.1) | 0.351 |
| Low/moderate intensity | 238 (72.3) | 39 (63.9) | 199 (74.3) | 0.104 |
| High intensity | 91 (27.7) | 22 (36.1) | 69 (25.7) | |
| Ezetimibe, n (%) | 78 (12.1) | 15 (13.5) | 63 (11.8) | 0.609 |
| Metformin, n (%) | 467 (72.3) | 80 (72.1) | 387 (72.3) | 0.955 |
| Sulfonylureas, n (%) | 166 (25.7) | 13 (11.7) | 153 (28.6) | 0.001 |
| DPP-4 Inhibitors, n (%) | 54 (8.4) | 8 (7.2) | 46 (8.6) | 0.630 |
| GLP-1 Agonist, n (%) | 19 (2.9) | 5 (4.5) | 14 (2.6) | 0.284 |
Continuous variables are presented as mean ± SD or as median (IQR); categorical variables as number (%). AMI, acute myocardial infarction; BB, B-blockers; BMI, Body Mass Index; CCB, Calcium Channel Blockers; CKD, chronic kidney disease with 30 < GFR < 60 ml/min; COPD, chronic obstructive pulmonary disease; CVA, cerebrovascular accident; DPP-4, Dipeptidyl peptidase 4; GLP-1, Glucagon-like peptide-1; PAD, peripheral artery disease; PCI, Percutaneous Coronary Intervention; RAAS: renin-angiotensin-aldosterone system; TIA, transient ischaemic attack.
Table 2.
Clinical admission, procedural and angiographic characteristics.
| Total | SGLT2-i users | Non-SGLT2-i users | P -value | |
|---|---|---|---|---|
| (N = 646) | (N = 111) | (N = 535) | ||
| STEMI, n (%) | 309 (47.8) | 52 (46.8) | 257 (48) | 0.819 |
| ECG– balloon time (STEMI) | 3 (2–5) | 3 (2–6) | 3 (2–5) | 0.648 |
| SBP | 140 (125–160) | 140 (125–155) | 140 (125–160) | 0.639 |
| DBP | 80 (70–90) | 83 (70–90) | 80 (70–90) | 0.551 |
| HR | 81 (70–94) | 75 (68–86) | 83 (72–95) | <0.001 |
| NYHA > 2, n (%) | 113 (17.5) | 16 (14.4) | 101 (18.9) | 0.266 |
| Admission LVEF | 47 ± 11 | 48 ± 10 | 47 ± 11 | 0.183 |
| Killip Class ≥ 2, n (%) | 135 (20.9) | 18 (16.2) | 117 (21.9) | 0.183 |
| VT/VF, n (%) | 21 (3.3) | 2 (1.8) | 19 (3.6) | 0.344 |
| AF, n (%) | 58 (9) | 9 (8.1) | 49 (9.2) | 0.725 |
| Admission blood glucose | 180 (143–239) | 158 (139–205) | 185 (146–246) | 0.007 |
| K+, mmol/L (mean ± SD) | 4 ± 0.3 | 4.1 ± 0.5 | 3.9 ± 0.2 | 0.342 |
| Ca2+, mg/dl (mean ± SD) | 9.6 ± 2.3 | 9.9 ± 2 | 9.4 ± 2.4 | 0.087 |
| Mg, mg/dl (mean ± SD) | 2 ± 0.3 | 2.1 ± 0.4 | 2 ± 0.2 | 0.854 |
| Radial access, n (%) | 542 (83.9) | 92 (82.9) | 450 (84.1) | 0.748 |
| LM lesion, n (%) | 34 (5.3) | 5 (4.5) | 29 (5.4) | 0.694 |
| LAD lesion, n (%) | 361 (55.9) | 64 (57.7) | 297 (55.5) | 0.679 |
| CX lesion, n (%) | 168 (26) | 33 (29.7) | 135 (25.2) | 0.326 |
| RCA lesion, n (%) | 203 (31.4) | 35 (31.5) | 168 (31.4) | 0.979 |
| 1 Vessel lesion, n (%) | 271 (42) | 52 (46.8) | 219 (40.9) | 0.251 |
| 2 Vessels lesion, n (%) | 231 (35.8) | 33 (29.7) | 198 (37) | 0.145 |
| 3 Vessels lesion, n (%) | 140 (21.7) | 24 (21.6) | 116 (21.7) | 0.989 |
| TIMI Flow pre, (mean ± SD) | 1.2 ± 1.1 | 1.1 ± 1 | 1.2 ± 1.1 | 0.948 |
| TIMI Flow post, (mean ± SD) | 3 ± 0.3 | 3 ± 0.3 | 3 ± 0.3 | 0.678 |
| Complete revascularization, n (%) | 406 (62.8) | 86 (77.5) | 426 (79.6) | 0.611 |
Continuous variables are presented as mean±SD or as median (IQR); categorical variables as number (%). AF, Atrial Fibrillation; CX, circumflex artery; DBP, diastolic blood pressure; ECG, electrocardiogram; HR, heart rate; NYHA, New York Heart Association; LAD, left anterior descending artery; LM, left main; LVEF, Left ventricular ejection fraction; PCI, Primary Percutaneous Coronary Intervention; RCA, right coronary artery; SBP, systolic blood pressure; STEMI, ST-elevation myocardial infarction; TIMI, Thrombolysis in Myocardial Infarction; VF, Ventricular Fibrillation; VT, Ventricular Tachycardia.
In-hospital clinical outcomes
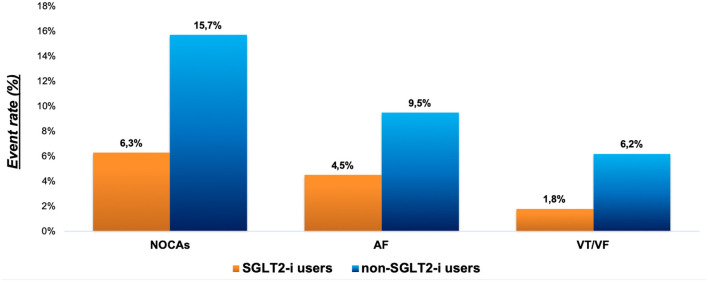
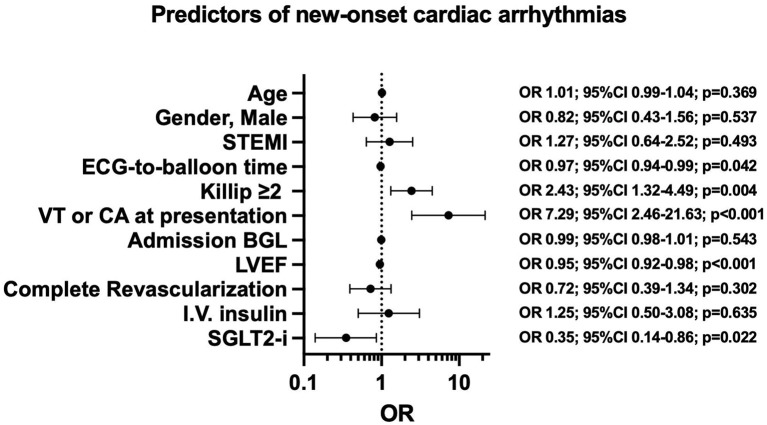
The median length of stay was 5 days (range 4–8), with no difference between SGLT2-i users and non-users. A total of 91 (14.1%) NOCAs, 56 (8.7%) new-onset AF, and 35 (5.4%) new-onset VT/VF events occurred during hospitalization. SGLT2-i users had a lower rate of primary endpoint compared with non-SGLT2-i users (6.3 vs. 15.7%, p = 0.010) (Figure 1). Furthermore, a lower rate of AF and VT/VF was observed in the SGLT2-i users group compared with the non-users at Pearson Chi-Square test (4.5 vs. 9.5% and 1.8 vs. 6.2%, respectively; p = 0.032) (Figure 2). There were no significant differences in terms of recurrent AMI (p = 0.84) or the need for circulatory support (p = 0.98) between groups, whereas SGLT2-i users had less frequently post-procedure renal function impairment (p = 0.022) (Table 3). The hierarchical univariate analysis revealed an unadjusted reduction of 63% (OR = 0.37; 95%CI 0.17–0.83; p = 0.015) in the risk of the primary endpoint for SGLT2-i users compared with non-SGLT2-i users. In the multivariate logistic regression model, after adjusting for all known confounders, the use of SGLT2-i was independently associated with the lower occurrence of the composite endpoint of NOCAs (OR = 0.35; 95%CI 0.14–0.86; p = 0.022), together with no-VAs at presentation, reduced ECG-to-balloon time, higher LVEF, and Killip class <2 at admission (Figure 3). When each component of the composite endpoint was separately appraised by multinomial logistic regression, after adjusting for potential confounders, SGLT2-i therapy remained an independent predictor of lower VT/VF occurrence (OR = 0.20; 95%CI 0.04–0.97; p = 0.046) but not for AF occurrence (Table 4), which showed a reduction, without reaching statistical significance (OR = 0.40; 95% CI 0.14–1.14; p = 0.086).
Figure 1.
Study design and occurrence of new-onset cardiac arrhythmias. AF, atrial fibrillation; AMI, acute myocardial infarction; NOCAs, new-onset cardiac arrhythmias; T2DM, type 2 diabetes mellitus; SGLT2-i, Sodium-glucose co-transporter 2 inhibitors; VF, Ventricular Fibrillation; VT, Ventricular Tachycardia.
Figure 2.
Primary endpoint and its individual components in SGLT2-i and non-SGLT2-i users. AF, atrial fibrillation; NOCAs, new-onset cardiac arrhythmias; SGLT2-i, Sodium-glucose co-transporter 2 inhibitors; VF, Ventricular Fibrillation; VT, Ventricular Tachycardia.
Table 3.
In-hospital outcomes of SGLT2-i users vs. non-SGLT2-i users.
| Total | SGLT2-i users | Non-SGLT2-i users | P -value | |
|---|---|---|---|---|
| (N = 646) | (N = 111) | (N = 535) | ||
| Hospital stay, days | 5 (4–8) | 5 (4–8) | 5 (4–8) | 0.526 |
| hs-TnI max | 2,368 (625–9,224) | 903 (278–2,438) | 3,155 (731–9,223) | <0.001 |
| HbA1c | 51 (45–59) | 52 (48–57) | 50 (44–60) | 0.137 |
| SD | 49.8 ±46.5 | 44.7 ±50.2 | 51 ±45.6 | 0.206 |
| CV | 0.27 ±0.19 | 0.25 ±0.20 | 0.27 ±0.19 | 0.234 |
| In-hospital glucose-lowering strategy | ||||
| Insulin s.c., n (%) | 430 (66.6) | 57 (51.4) | 394 (73.6) | <0.001 |
| Insulin i.v., n (%) | 65 (10.1) | 17 (15.3) | 144 (26.9) | 0.010 |
| In-hospital outcomes | ||||
| Arrhythmia, n (%) | 91 (14.1) | 7 (6.3) | 84 (15.7) | 0.010 |
| New-onset AF, n (%) | 56 (8.7) | 5 (4.5) | 51 (9.5) | |
| VT/VF, n (%) | 35 (5.4) | 2 (1.8) | 33 (6.2) | |
| Re-AMI, n (%) | 7 (1.1) | 1 (0.9) | 6 (1.1) | 0.838 |
| Re-PCI, n (%) | 13 (2.0) | 4 (3.6) | 9 (1.7) | 0.190 |
| IABP, n (%) | 23 (3.6) | 4 (3.6) | 19 (3.6) | 0.978 |
| CI-AKI, n (%) | 68 (10.5) | 6 (5.4) | 70 (13.1) | 0.022 |
Continuous variables are presented as mean±SD or as median (IQR); categorical variables as number (%). AF, Atrial Fibrillation; AMI, Acute Myocardial Infarction, CI-AKI, Contrast-Induced Acute Kidney Injury; Hs-TnI, high sensitivity Troponin I; IABP, Intra-Aortic Balloon Pump; i.v., intravenous; MACE, major adverse cardiovascular events; PCI, Primary Percutaneous Coronary Intervention; s.c., subcutaneous; VF, Ventricular Fibrillation; VT, Ventricular Tachycardia.
Figure 3.
Effects of predictors of new-onset cardiac arrhythmias. BGL, blood glucose level; CA, cardiac arrest; ECG, electrocardiogram; LVEF, Left ventricular ejection fraction; NOCAs, new-onset cardiac arrhythmias; SGLT2-i, Sodium-glucose cotransporter 2 inhibitors; STEMI, ST-elevation myocardial infarction; VT, ventricular tachycardia.
Table 4.
Multivariable analysis: Predictors of NOCAs, AF, VT/VF.
| NOCAs | AF | VT/VF | ||||||
|---|---|---|---|---|---|---|---|---|
| Variables | OR (CI, 95%) | p -value | Variables | OR (CI, 95%) | p -value | Variables | OR (CI, 95%) | p -value |
| Age | 1.01 (0.99–1.04) | 0.369 | Age | 1.02 (0.98–1.05) | 0.267 | Age | 0.99 (0.96–1.04) | 0.972 |
| Gender, male | 0.82 (0.43–1.56) | 0.537 | RWMA | 0.82 (0.24–1.90) | 0.455 | RWMA | 0.76 (0.18–3.13) | 0.702 |
| STEMI | 1.27 (0.64–2.52) | 0.493 | STEMI | 1.83 (0.84–3.98) | 0.124 | STEMI | 0.71 (0.23–2.27) | 0.570 |
| ECG-to-balloon time | 0.97 (0.94–0.99) | 0.042 | ECG-to-balloon time | 0.97 (0.93–1.00) | 0.062 | ECG-to-balloon time | 0.96 (0.90–1.02) | 0.194 |
| Killip≥2 | 2.43 (1.32–4.49) | 0.004 | Killip≥2 | 2.34 (1.15–4.77) | 0.019 | Killip≥2 | 2.54 (0.95–6.85) | 0.064 |
| VT or CA at presentation | 7.29 (2.46–21.63) | <0.001 | VT or CA at presentation | 2.77 (0.67–11.42) | 0.160 | VT or CA at presentation | 17.3 (4.99–60.0) | <0.001 |
| Admission BGL | 0.99 (0.98–1.01) | 0.543 | Peak Hs-TnI, ng/L | 1.00 (1.00–1.01) | 0.776 | Peak Hs-TnI, ng/L | 1.00 (1.00–1.01) | 0.267 |
| LVEF | 0.95 (0.92–0.98) | <0.001 | LVEF | 0.94 (0.90–0.97) | <0.001 | LVEF | 0.95 (0.91–0.99) | 0.043 |
| Complete revascularization | 0.72 (0.39–1.34) | 0.302 | Complete revascularization | 0.75 (0.38–1.48) | 0.401 | Complete revascularization | 0.62 (0.24–1.57) | 0.310 |
| I.V. insulin | 1.25 (0.50–3.08) | 0.635 | I.V. insulin | 1.12 (0.56–2.21) | 0.756 | I.V. insulin | 0.75 (0.29–1.91) | 0.547 |
| SGLT2-i | 0.35 (0.14–0.86) | 0.022 | SGLT2-i | 0.40 (0.14–1.14) | 0.086 | SGLT2-i | 0.20 (0.04–0.97) | 0.046 |
BGL, blood glucose level; CA, cardiac arrest; ECG, electrocardiogram; Hs-TnI, high sensitivity Troponin I; LVEF, Left ventricular ejection fraction; SGLT2-i, Sodium-glucose cotransporter 2 inhibitors; RWMA, regional wall motion abnormalities; STEMI, ST-elevation myocardial infarction; VT, ventricular tachycardia.
Discussion
The main findings of our study can be summarized as follows: (i) in the context of diabetic patients with AMI, the new-onset of in-hospital major arrhythmic events, as the composite endpoint, was significantly lower in SGLT2-i users compared to non-SGLT2-i users; (ii) when considered separately, AF and VT/VF events occurred less frequently in the group of patients on SGLT2-i therapy compared with non-SGLT2-i users; (iii) SGLT2-i was a significant predictor of lower incidence of NOCAs, independently of age, sex, STEMI presentation, complete revascularization, admission stress hyperglycemia, and use of i.v. insulin therapy. Conversely, longer ECG-balloon time, Killip class >2, VT or cardiac arrest as presentation and lower LVEF turn out to be independent predictors of higher arrhythmic burden; (iv) after adjusting for all confounding factors, the use of SGLT2-i was identified as an independent predictor of reduced VT/VF occurrence. Our study is the first to report the impact of SGLT2-i therapy on arrhythmic burden in a specific setting of AMI diabetic patients.
The relationship between SGLT2-i therapy and cardiac arrhythmias has been extensively studied in T2DM patients with HF, but reported results have been inconsistent (22, 24). A systematic review and meta-analysis of randomized controlled trials on SGLT2i in T2DM patients highlighted that no significant difference was noted in the occurrence of AF in SGLT2-i patients (26). A later and larger meta-analysis (27) confirmed no benefit in protecting from new-onset AF in diabetic patients treated with SGLT2-i compared to other glucose-lowering drugs. These results were confirmed by two large real-world observational studies (28, 29), which found no significant differences in the occurrence of AF when SGTL2-i were compared with dipeptidylpeptidase-4 (DPP-4) inhibitors (30). By contrast, a larger body of evidence supports a protective effect in terms of new-onset AF (31–37). A post-hoc study from the Declare-TIMI 58 trial showed that dapagliflozin was associated with a lower incidence of new-onset AF in high-risk diabetic patients, with a relative risk reduction of 19% (31). Ling et al. (32) demonstrated a lower incidence of AF when SGLT2-i were compared to DPP-4-inhibitors (32). Several meta-analyses have confirmed that SGLT2-i could significantly decrease the incidence of AF both in diabetic and non-diabetic patients, with a reduction rate ranging from 19 to 25 % (33–36). These results appear to be consistent in recent observational studies (32, 37). Moreover, there seem to be no differences in protection from cardiac arrhythmias among different SGLT2-i, assuming a class effect (38). Less robust data regarding ventricular arrhythmias have been reported, possibly due to the large heterogeneity of the populations studied (22, 24). Overall, there are encouraging data on reducing VAs in patients treated with SGLT2-i (35, 39). In a post hoc analysis of DAPA-HF, dapagliflozin was shown to reduce the risk of any serious VAs or sudden cardiac death when added to standard therapy in patients with HF with reduced ejection fraction (HFrEF) (39). In this analysis, the relative risk reduction of VAs (as a composite of VT and VF) occurrence was 21%, and the effect was consistent across each component of the composite outcome. Other meta-analytic data highlighted a protective effect of SGLT2-i on the occurrence of VT, showing no differences across comorbidities or baseline conditions (35). There are also conflicting results on the incidence of VAs in patients receiving SGLT2-i; in fact, a recent meta-analysis found no association between SGLT2i therapy and lower VAs in patients with T2DM and/or HF and/or chronic kidney disease (34).
Although numerous—yet conflicting—the aforementioned data concern patients with or without T2DM, most with HF and HFrEF, in a chronic setting. Data on the acute effects of dapagliflozin in T2DM patients with HFrEF were provided by Ilyas et al. (40), who observed a reduced ventricular ectopic burden, suggesting an early anti-arrhythmic benefit after 2 weeks of treatment (40). Nevertheless, none of these relate specifically to patients with AMI. Our study is distinguishable from these previous reports inasmuch as we investigated the impact that chronic SGLT2-i therapy may have on patients experiencing hospitalization for AMI. In our cohort, in-hospital NOCAs were significantly reduced by 64%; in particular, a more effective reduction in VAs drove this finding. In fact, SGLT2-i seemed to reduce the risk of developing VAs by 80%, with a trend in reducing the new-onset of AF that did not reach significance. It is pertinent to emphasize how this finding is clinically relevant since VAs are frequently due to ischemia and the resulting molecular mechanisms during AMI, as they are closely related to sudden cardiac deaths in the early stages of AMI (5, 12). This observation is also significant considering that patients who develop any cardiac arrhythmia, especially VAs, during AMI have increased risks of subsequent arrhythmic events after discharge and a worse long-term prognosis (41). In terms of reduction in arrhythmic events, our results are in line with a nationwide population-based longitudinal Taiwanese cohort study that showed a reduction in total new-onset arrhythmic events, but not of all of its components, among SLGT2-i users (42). This study found no differences regarding AF, supraventricular arrhythmias, and VAs when considered separately (42).
We can relate this benefit in terms of NOCAs reduction to several mechanisms and pathways involved in the pathogenesis of cardiac arrhythmias on which SGLT2-i may impact. The EMBODY trial (43) investigated the effects of SGLT2-i on cardiac sympathetic and parasympathetic activities in patients with AMI and T2DM. Using several surrogate indices, the authors demonstrated that the empagliflozin group experienced significant improvement in both cardiac sympathetic and parasympathetic nerve activities, with a focus on sympathetic activity that might promote the development of arrhythmias (43). The sympathetic activity might be involved in another mechanism of control by SGLT2-i (44). In our cohort, the improved modulation of sympathetic tone could be reflected by a lower heart rate in patients treated with SGLT2-i. Indeed, previous reports have shown a more balanced autonomic system activity in patients treated with SGLT2-i when compared with non-SGLT2-i users, also in patients without structural heart disease (19). Another beneficial effect of SGLT2-i is protection against hyperglycemia-induced sympathetic overstimulation (45, 46). Different, non-mutually exclusive, mechanisms might mediate the relationship between insulin therapy and cardiac arrhythmias. First, hyperinsulinemia occurring during insulin therapy might induce sympathetic nervous system stimulation and consequently increase heart rate and QTc, which are well-known risk factors for ventricular arrhythmias (47–49). Additionally, patients treated with insulin are much more exposed to perceived and unperceived hypoglycemic crises (50). It should be pointed out that the presence of hypoglycemia is known to increase circulating levels of several counter-regulatory hormones (e.g., catecholamines), which increase the release of glucose from the liver and allow recovery from hypoglycemia. However, the same catecholamines also target the heart, resulting in increased heart rate and QTc, with a higher outcome consisting of a greater prevalence and incidence of cardiac arrhythmias (50). Accordingly, our patients treated with SGLT2-i exhibited lower admission blood glucose levels than patients treated with other OAD agents. Moreover, the lower number of hypoglycemic episodes associated with reduced insulin therapy (both subcutaneous and intravenous) resulting from minor stress admission hyperglycemia further corroborated the reduced in-hospital occurrence of arrhythmias in SGLT2-i users. Nevertheless, blood glucose levels at admission and the use of i.v. insulin did not appear to be predictor of arrhythmic events in our analysis. Glycemic control might be involved in the anti-arrhythmic action of SGLT2-i, but the hypothesis that these drugs also prevent arrhythmias via glucose-independent pathways is an added advantage since these drugs are also administered to non-diabetic patients. SGLT2-i ability to affect some of the ionic currents in cardiomyocytes could also explain some of their anti-arrhythmic effects. In particular, SGLT2-i could attenuate the increase in INa − late in diabetic patients with and without HF, thus preventing ventricular repolarization prolongation and early afterdepolarization (51). In ventricular cardiomyocytes isolated from rats, dapagliflozin attenuated the decreased IK responsible for the potential prolongation and was thereby able to reduce the risk of cardiac arrhythmias in these preclinical models (52). SGLT2-i have also been shown to interact with Ca2+/calmodulin-dependent protein kinase II (CaMKII) (53), decreasing RyR2 phosphorylation thus reducing spontaneous diastolic Ca2+ release and sodium influx, both known to trigger delayed afterdepolarizations (54).
Finally, focusing on arrhythmias related to ischemia and reperfusion, it is relevant to highlight the results of an animal model that might reproduce our proposed acute ischemia setting (55). Hu et al. (55) aimed to investigate the impact of empagliflozin on myocardial ischemia/reperfusion-provoked arrhythmias in vivo. They found that pretreatment with empagliflozin protected from VAs induced by ischemia and reperfusion injury, and this benefit seems to be related to the activation of the ERK1/2-dependent signaling pathway (involved in cell survival processes) in a glucose-independent manner. Similarly, in ischemia-reperfusion rabbit models, empagliflozin was shown to reduce VAs, improving calcium cycling and mitochondrial fitness (56).
Several other molecular effects, as well as sodium hydrogen antiporter 1 (NHE1) receptor interaction, impact on oxidative stress modulation, myocardial extracellular matrix remodeling, and systemic inflammation, have been hypothesized to have a cardioprotective effect. In fact, we have actually demonstrated that AMI patients treated with SGLT2-i display a significantly lower inflammatory response and smaller infarct size than those receiving other OAD agents, independent of glucose control (25).
To date, real-world data on the cardioprotective effects and arrhythmia prevention of SGLT2i are mainly about diabetic patients, given that SGLT2-i prescription was only recently extended to non-diabetic patients. Therefore, further studies are needed to evaluate the effects of this class of drugs in the early stages of AMI, even in non-diabetic patients.
Study limitations
The results of our study should be interpreted in light of some limitations. First, our conclusions are limited by the study's observational design, so our study results should be considered hypothesis-generating. Second, the sample size was powered to evaluate only a “class effect” but not the “doses effect”. However, the sample size estimation was made by considering long-term all-cause death as the main outcome. The present study is a sub-analysis evaluating arrhythmic events, so the sample size may be underpowered. Further investigation will be necessary to determine the finding that should be considered as hypothesis-generating. Third, categorization into SGLT2-i users and non-SGLT2-i users was based on the patients' reported therapy at admission and medical records. However, we cannot determine the effective adherence to treatment with SGLT2i or other OAD drugs. Moreover, the risk of potential concealed conditions must be taken into account. These may not have been included in the multivariable model. Finally, a unified protocol for cardiac arrhythmia detection was not observed in the medical records. Therefore, the possibility of undetected arrhythmia cannot be ruled out in this study.
Conclusions
In a real-world clinical scenario of diabetic patients with AMI, the use of SGLT2-i was associated with a lower risk of new-onset cardiac arrhythmias during the in-hospital phase. In particular, the major effect seemed to be exerted on the reduction of VAs. Our findings are hypothesis-generating, and they turn on the spotlight on the pleiotropic and additional benefits of this class of drugs in patients with atherosclerotic cardiovascular disease, beyond their underlying role in controlling glycemic metabolism.
Data availability statement
The raw data supporting the conclusions of this article will be made available by the authors, without undue reservation.
Ethics statement
The studies involving human participants were reviewed and approved by Institutional Review Board of the IRCCS Azienda Ospedaliero-Universitaria of Bologna, Registration Number 600/2018/Oss/AOUBo. The patients/participants provided their written informed consent to participate in this study.
Author contributions
AC and FG contributed conception and design of the study. PP, LB, EG, NMi, NMo, FG, MA, AS, SA, GE, and AI organized the database and collected data. AC and LB performed the statistical analysis. AC, FG, and PC wrote the first draft of the manuscript. PP and LB wrote sections of the manuscript. GS, CS, AF, GC, CM, EB, NMi, NMo, JO, DV, CP, PC, and RM revised the article and approved the final version of the manuscript. All authors contributed to manuscript revision, read and approved the submitted version.
Funding
PP and GE report receiving a research grant from the CardioPaTh Ph.D. Program.
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Publisher's note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
References
- 1.Kalarus Z, Svendsen JH, Capodanno D, Dan G-A, De Maria E, Gorenek B, et al. Cardiac arrhythmias in the emergency settings of acute coronary syndrome and revascularization: an European Heart Rhythm Association (EHRA) consensus document, endorsed by the European Association of Percutaneous Cardiovascular Interventions (EAPCI), and European Acute Cardiovascular Care Association (ACCA). EP Eur. (2019) 21:1603–4. 10.1093/europace/euz163 [DOI] [PubMed] [Google Scholar]
- 2.Hindricks G, Potpara T, Dagres N, Arbelo E, Bax JJ, Blomström-Lundqvist C, et al. 2020 ESC Guidelines for the diagnosis and management of atrial fibrillation developed in collaboration with the European Association for Cardio-Thoracic Surgery (EACTS). Eur Heart J. (2020) 42:373–498. 10.1093/eurheartj/ehaa612 [DOI] [PubMed] [Google Scholar]
- 3.Al-Khatib SM, Stevenson WG, Ackerman MJ, Bryant WJ, Callans DJ, Curtis AB, et al. 2017 AHA/ACC/HRS Guideline for management of patients with ventricular arrhythmias and the prevention of sudden cardiac death. Circulation. (2018) 138: e272–e391. 10.1161/CIR.0000000000000549 [DOI] [PubMed] [Google Scholar]
- 4.Di Diego JM, Antzelevitch C. Ischemic ventricular arrhythmias: Experimental models and their clinical relevance. Hear Rhythm. (2011) 8:1963–8. 10.1016/j.hrthm.2011.06.036 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Sattler SM, Skibsbye L, Linz D, Lubberding AF, Tfelt-Hansen J, Jespersen T. Ventricular arrhythmias in first acute myocardial infarction: epidemiology, mechanisms, and interventions in large animal models. Front Cardiovasc Med. (2019) 6:158. 10.3389/fcvm.2019.00158 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Liang C, Li Q, Wang K, Du Y, Wang W, Zhang H. Mechanisms of ventricular arrhythmias elicited by coexistence of multiple electrophysiological remodeling in ischemia: a simulation study. PLoS Comput Biol. (2022) 18:e1009388. 10.1371/journal.pcbi.1009388 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Jabre P, Jouven X, Adnet F, Thabut G, Bielinski SJ, Weston SA, et al. Atrial fibrillation and death after myocardial infarction. Circulation. (2011) 123:2094–100. 10.1161/CIRCULATIONAHA.110.990192 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Henkel DM, Witt BJ, Gersh BJ, Jacobsen SJ, Weston SA, Meverden RA, et al. Ventricular arrhythmias after acute myocardial infarction: a 20-year community study. Am Heart J. (2006) 151:806–12. 10.1016/j.ahj.2005.05.015 [DOI] [PubMed] [Google Scholar]
- 9.Ibanez B, James S, Agewall S, Antunes MJ, Bucciarelli-Ducci C, Bueno H, et al. 2017 ESC Guidelines for the management of acute myocardial infarction in patients presenting with ST-segment elevation. Eur Heart J. (2018) 39:119–77. 10.5603/KP.2018.0041 [DOI] [PubMed] [Google Scholar]
- 10.Mehta RH Yu J, Piccini JP, Tcheng JE, Farkouh ME, Reiffel J, et al. Prognostic significance of postprocedural sustained ventricular tachycardia or fibrillation in patients undergoing primary percutaneous coronary intervention (from the HORIZONS-AMI Trial). Am J Cardiol. (2012) 109:805–12. 10.1016/j.amjcard.2011.10.043 [DOI] [PubMed] [Google Scholar]
- 11.Tran H V, Ash AS, Gore JM, Darling CE, Kiefe CI, Goldberg RJ. Twenty-five year trends (1986-2011) in hospital incidence and case-fatality rates of ventricular tachycardia and ventricular fibrillation complicating acute myocardial infarction. Am Heart J. (2019) 208:1–10. 10.1016/j.ahj.2018.10.007 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Deo R, Albert CM. Epidemiology and genetics of sudden cardiac death. Circulation. (2012) 125:620–37. 10.1161/CIRCULATIONAHA.111.023838 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Udell JA, Jones WS, Petrie MC, Harrington J, Anker SD, Bhatt DL, et al. Sodium glucose cotransporter-2 inhibition for acute myocardial infarction. J Am Coll Cardiol. (2022) 79:2058–68. 10.1016/j.jacc.2022.03.353 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.McMurray JJV, Solomon SD, Inzucchi SE, Køber L, Kosiborod MN, Martinez FA, et al. Dapagliflozin in patients with heart failure and reduced ejection fraction. N Engl J Med. (2019) 381:1995–2008. [DOI] [PubMed] [Google Scholar]
- 15.Zinman B, Wanner C, Lachin JM, Fitchett D, Bluhmki E, Hantel S, et al. Empagliflozin, cardiovascular outcomes, and mortality in type 2 diabetes. N Engl J Med. (2015) 373:2117–28. 10.1056/NEJMoa1504720 [DOI] [PubMed] [Google Scholar]
- 16.Anker SD, Butler J, Filippatos G, Ferreira JP, Bocchi E, Böhm M, et al. Empagliflozin in heart failure with a preserved ejection fraction. N Engl J Med. (2021) 385:1451–61. [DOI] [PubMed] [Google Scholar]
- 17.Marfella R, D'Onofrio N, Trotta MC, Sardu C, Scisciola L, Amarelli C, et al. Sodium/glucose cotransporter 2 (SGLT2) inhibitors improve cardiac function by reducing JunD expression in human diabetic hearts. Metabolism. (2022) 127:154936. 10.1016/j.metabol.2021.154936 [DOI] [PubMed] [Google Scholar]
- 18.Sardu C, Massetti M, Testa N, Martino L Di, Castellano G, Turriziani F, et al. Effects of sodium-glucose transporter 2 inhibitors (SGLT2-I) in patients with Ischemic Heart Disease (IHD) treated by coronary artery bypass grafting via MiECC: inflammatory burden, and clinical outcomes at 5 years of follow-up. Front Pharmacol. (2021) 12:777083. 10.3389/fphar.2021.777083 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Sardu C, Massimo Massetti M, Rambaldi P, Gatta G, Cappabianca S, Sasso FC, et al. SGLT2-inhibitors reduce the cardiac autonomic neuropathy dysfunction and vaso-vagal syncope recurrence in patients with type 2 diabetes mellitus: the SCAN study. Metabolism. (2022) 155243. 10.1016/j.metabol.2022.155243 [DOI] [PubMed] [Google Scholar]
- 20.Bell DSH, Goncalves E. Atrial fibrillation and type 2 diabetes: Prevalence, etiology, pathophysiology and effect of anti-diabetic therapies. Diabetes Obes Metab. (2019) 21:210–7. 10.1111/dom.13512 [DOI] [PubMed] [Google Scholar]
- 21.Bhar-Amato J, Davies W, Agarwal S. Ventricular arrhythmia after acute myocardial infarction: ‘The Perfect Storm'. Arrhythmia Electrophysiol Rev. (2017) 6:134. 10.15420/aer.2017.24.1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Kolesnik E, Scherr D, Rohrer U, Benedikt M, Manninger M, Sourij H, et al. SGLT2 inhibitors and their antiarrhythmic properties. Int J Mol Sci. (2022) 23:1678. 10.3390/ijms23031678 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Yin Z, Zheng H, Guo Z. Effect of Sodium-Glucose Co-transporter protein 2 inhibitors on arrhythmia in heart failure patients with or without type 2 diabetes: a meta-analysis of randomized controlled trials. Front Cardiovasc Med. (2022) 9:902923. 10.3389/fcvm.2022.902923 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Manolis AA, Manolis TA, Melita H, Manolis AS. Sodium-glucose cotransporter type 2 inhibitors and cardiac arrhythmias. Trends Cardiovasc Med. (2022) S1050-1738(22)00062-7. 10.1016/j.tcm.2022.04.003 [DOI] [PubMed] [Google Scholar]
- 25.Paolisso P, Bergamaschi L, Santulli G, Gallinoro E, Cesaro A, Gragnano F, et al. Infarct size, inflammatory burden, and admission hyperglycemia in diabetic patients with acute myocardial infarction treated with SGLT2-inhibitors: a multicenter international registry. Cardiovasc Diabetol. (2022) 21:77. 10.1186/s12933-022-01506-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Usman MS, Siddiqi TJ, Memon MM, Khan MS, Rawasia WF, Talha Ayub M, et al. Sodium-glucose co-transporter 2 inhibitors and cardiovascular outcomes: A systematic review and meta-analysis. Eur J Prev Cardiol. (2018) 25:495–502. 10.1177/2047487318755531 [DOI] [PubMed] [Google Scholar]
- 27.Li C, Liang S, Gao L, Liu H. Cardiovascular outcomes associated with SGLT-2 inhibitors versus other glucose-lowering drugs in patients with type 2 diabetes: a real-world systematic review and meta-analysis. PLoS ONE. (2021) 16:e0244689. 10.1371/journal.pone.0244689 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Norhammar A, Bodegård J, Nyström T, Thuresson M, Nathanson D, Eriksson JW. Dapagliflozin and cardiovascular mortality and disease outcomes in a population with type 2 diabetes similar to that of the DECLARE-TIMI 58 trial: a nationwide observational study. Diabetes Obes Metab. (2019) 21:1136–45. 10.1111/dom.13627 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Birkeland KI, Jørgensen ME, Carstensen B, Persson F, Gulseth HL, Thuresson M, et al. Cardiovascular mortality and morbidity in patients with type 2 diabetes following initiation of sodium-glucose co-transporter-2 inhibitors versus other glucose-lowering drugs (CVD-REAL Nordic): a multinational observational analysis. Lancet Diabetes Endocrinol. (2017) 5:709–17. 10.1016/S2213-8587(17)30258-9 [DOI] [PubMed] [Google Scholar]
- 30.Persson F, Nyström T, Jørgensen ME, Carstensen B, Gulseth HL, Thuresson M, et al. Dapagliflozin is associated with lower risk of cardiovascular events and all-cause mortality in people with type 2 diabetes (CVD-REAL Nordic) when compared with dipeptidyl peptidase-4 inhibitor therapy: A multinational observationa. Diabetes, Obes Metab. (2018) 20:344–51. 10.1111/dom.13077 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Zelniker TA, Bonaca MP, Furtado RHM, Mosenzon O, Kuder JF, Murphy SA, et al. Effect of dapagliflozin on atrial fibrillation in patients with type 2 diabetes mellitus. Circulation. (2020) 141:1227–34. 10.1161/CIRCULATIONAHA.119.044183 [DOI] [PubMed] [Google Scholar]
- 32.Ling AW-C, Chan C-C, Chen S-W, Kao Y-W, Huang C-Y, Chan Y-H, et al. The risk of new-onset atrial fibrillation in patients with type 2 diabetes mellitus treated with sodium glucose cotransporter 2 inhibitors versus dipeptidyl peptidase-4 inhibitors. Cardiovasc Diabetol. (2020) 19:188. 10.1186/s12933-020-01162-w [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Pandey AK, Okaj I, Kaur H, Belley-Cote EP, Wang J, Oraii A, et al. Sodium-Glucose Co-Transporter inhibitors and atrial fibrillation: a systematic review and meta-analysis of randomized controlled trials. J Am Heart Assoc. (2021) 10:e022222. 10.1161/JAHA.121.022222 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Fernandes GC, Fernandes A, Cardoso R, Penalver J, Knijnik L, Mitrani RD, et al. Association of SGLT2 inhibitors with arrhythmias and sudden cardiac death in patients with type 2 diabetes or heart failure: a meta-analysis of 34 randomized controlled trials. Hear Rhythm. (2021) 18:1098–105. 10.1016/j.hrthm.2021.03.028 [DOI] [PubMed] [Google Scholar]
- 35.Li H-L, Lip GYH, Feng Q, Fei Y, Tse Y-K, Wu M, et al. Sodium-glucose cotransporter 2 inhibitors (SGLT2i) and cardiac arrhythmias: a systematic review and meta-analysis. Cardiovasc Diabetol. (2021) 20:100. 10.1186/s12933-021-01293-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Zhou Z, Jardine MJ Li Q, Neuen BL, Cannon CP, de Zeeuw D, et al. Effect of SGLT2 inhibitors on stroke and atrial fibrillation in diabetic kidney disease. Stroke. (2021) 52:1545–56. 10.1161/STROKEAHA.120.031623 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Jhuo S-J, Lin T-H, Lin Y-H, Tsai W-C, Liu I-H, Wu B-N, et al. Clinical observation of SGLT2 inhibitor therapy for cardiac arrhythmia and related cardiovascular disease in diabetic patients with controlled hypertension. J Pers Med. (2022) 12:271. 10.3390/jpm12020271 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Suzuki Y, Kaneko H, Okada A, Itoh H, Matsuoka S, Fujiu K, et al. Comparison of cardiovascular outcomes between SGLT2 inhibitors in diabetes mellitus. Cardiovasc Diabetol. (2022) 21:67. 10.1186/s12933-022-01508-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Curtain JP, Docherty KF, Jhund PS, Petrie MC, Inzucchi SE, Køber L, et al. Effect of dapagliflozin on ventricular arrhythmias, resuscitated cardiac arrest, or sudden death in DAPA-HF. Eur Heart J. (2021) 42:3727–38. 10.1093/eurheartj/ehab560 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Ilyas F, Jones L, Tee SL, Horsfall M, Swan A, Wollaston F, et al. Acute pleiotropic effects of dapagliflozin in type 2 diabetic patients with heart failure with reduced ejection fraction: a crossover trial. ESC Hear Fail. (2021) 8:4346–52. 10.1002/ehf2.13553 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Sau A, Kaura A, Ahmed A, Patel KHK, Li X, Mulla A, et al. Prognostic significance of ventricular arrhythmias in 13 444 patients with acute coronary syndrome: a retrospective cohort study based on routine clinical data (NIHR Health Informatics Collaborative VA-ACS Study). J Am Heart Assoc. (2022) 11:e024260. 10.1161/JAHA.121.024260 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Chen H-Y, Huang J-Y, Siao W-Z, Jong G-P. The association between SGLT2 inhibitors and new-onset arrhythmias: a nationwide population-based longitudinal cohort study. Cardiovasc Diabetol. (2020) 19:73. 10.1186/s12933-020-01048-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Shimizu W, Kubota Y, Hoshika Y, Mozawa K, Tara S, Tokita Y, et al. Effects of empagliflozin versus placebo on cardiac sympathetic activity in acute myocardial infarction patients with type 2 diabetes mellitus: the EMBODY trial. Cardiovasc Diabetol. (2020) 19:148. 10.21203/rs.3.rs-35207/v2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Wan N, Rahman A, Hitomi H, Nishiyama A. The effects of Sodium-Glucose cotransporter 2 inhibitors on sympathetic nervous activity. Front Endocrinol. (2018) 9:421. 10.3389/fendo.2018.00421 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Verma S. Are the cardiorenal benefits of SGLT2 inhibitors due to inhibition of the sympathetic nervous system? JACC Basic to Transl Sci. (2020) 5:180–2. 10.1016/j.jacbts.2020.01.011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Dünser MW, Hasibeder WR. Sympathetic overstimulation during critical illness: adverse effects of adrenergic stress. J Intensive Care Med. (2009) 24:293–316. 10.1177/0885066609340519 [DOI] [PubMed] [Google Scholar]
- 47.Tran H V, Gore JM, Darling CE, Ash AS, Kiefe CI, Goldberg RJ. Hyperglycemia and risk of ventricular tachycardia among patients hospitalized with acute myocardial infarction. Cardiovasc Diabetol. (2018) 17:136. 10.1186/s12933-018-0779-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.de Lucia C, Piedepalumbo M, Paolisso G, Koch WJ. Sympathetic nervous system in age-related cardiovascular dysfunction: pathophysiology and therapeutic perspective. Int J Biochem Cell Biol. (2019) 108:29–33. 10.1016/j.biocel.2019.01.004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Marfella R, De Angelis L, Siniscalchi M, Rossi F, Giugliano D, Nappo F. The effect of acute hyperglycaemia on QTc duration in healthy man. Diabetologia. (2000) 43:571–5. 10.1007/s001250051345 [DOI] [PubMed] [Google Scholar]
- 50.Andersen A, Bagger JI, Baldassarre MPA, Christensen MB, Abelin KU, Faber J, et al. Acute hypoglycemia and risk of cardiac arrhythmias in insulin-treated type 2 diabetes and controls. Eur J Endocrinol. (2021) 185:343–53. 10.1530/EJE-21-0232 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Philippaert K, Kalyaanamoorthy S, Fatehi M, Long W, Soni S, Byrne NJ, et al. Cardiac late sodium channel current is a molecular target for the sodium/glucose cotransporter 2 inhibitor empagliflozin. Circulation. (2021) 143:2188–204. 10.1161/CIRCULATIONAHA.121.053350 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Durak A, Olgar Y, Degirmenci S, Akkus E, Tuncay E, Turan B, et al. SGLT2 inhibitor dapagliflozin suppresses prolonged ventricular-repolarization through augmentation of mitochondrial function in insulin-resistant metabolic syndrome rats. Cardiovasc Diabetol. (2018) 17:144. 10.1186/s12933-018-0790-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Mustroph J, Wagemann O, Lücht CM, Trum M, Hammer KP, Sag CM, et al. Empagliflozin reduces Ca/calmodulin-dependent kinase II activity in isolated ventricular cardiomyocytes. ESC Hear Fail. (2018) 5:642–8. 10.1002/ehf2.12336 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Trum M, Riechel J, Wagner S. Cardioprotection by SGLT2 inhibitors—does it all come down to Na+? Int J Mol Sci. (2021) 22:7976. 10.3390/ijms22157976 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Hu Z, Ju F, Du L, Abbott GW. Empagliflozin protects the heart against ischemia/reperfusion-induced sudden cardiac death. Cardiovasc Diabetol. (2021) 20:199. 10.1186/s12933-021-01392-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Azam MA, Chakraborty P, Si D, Du B, Massé S, Lai PFH, et al. Anti-arrhythmic and inotropic effects of empagliflozin following myocardial ischemia. Life Sci. (2021) 276:119440. 10.1016/j.lfs.2021.119440 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The raw data supporting the conclusions of this article will be made available by the authors, without undue reservation.