Baruj Benacerraf and Hugh O. McDevitt are sometimes called the discoverers of immune response genes because their seminal research showed that the immune response was under genetic control. The primary genes identified by this pair are located in the major histocompatibility complex (MHC), known as HLA genes in humans. In 1972, the two wrote a prescient commentary that appeared in Science.1 In essence, they argued that if genes control the immune response, and if some diseases are triggered by the immune response, then those same genes might influence who receives a diagnosis of diseases like rheumatoid arthritis, systemic lupus erythematosus, or Graves’ disease. Their prediction was validated approximately 1 year later2 and has been shown to hold true for more than 100 diseases.3 For most genetically complex immune-mediated diseases, the HLA genes affect the likelihood of disease developing more than any other gene identified in genome-wide association studies.
Although Benacerraf and McDevitt made an insightful deduction, their reasoning might have been incorrect. Fifty years ago, many immune-mediated diseases were labelled autoimmune. Rheumatoid arthritis, systemic lupus erythematosus, and Graves’ disease are characterized by autoantibodies, so it makes sense to believe that the immune system is attacking itself, and it is plausible that immune response genes of the major histocompatibility complex affect the magnitude of that response. But this logic has 2 flaws. First, many immune-mediated diseases like sarcoidosis, ankylosing spondylitis, or birdshot chorioretinopathy (BSCR) are not necessarily autoimmune diseases.4 Disease-causing autoantibodies are not characteristic of these diseases. Second and more importantly, even in diseases in which autoantibodies are detectable, rarely, if ever, has a study documented that the HLA genes affect the immune response to the putative autoantigen. In celiac disease, the HLA allele, DQ2.5, does impact the immune response to peptides derived from gluten,5 which is present in diet. An immune response to gluten is causally related to celiac disease, although gluten itself is not an autoantigen. Nevertheless, celiac disease is arguably the disease that best connects disease predisposition to a genetically controlled immune response.
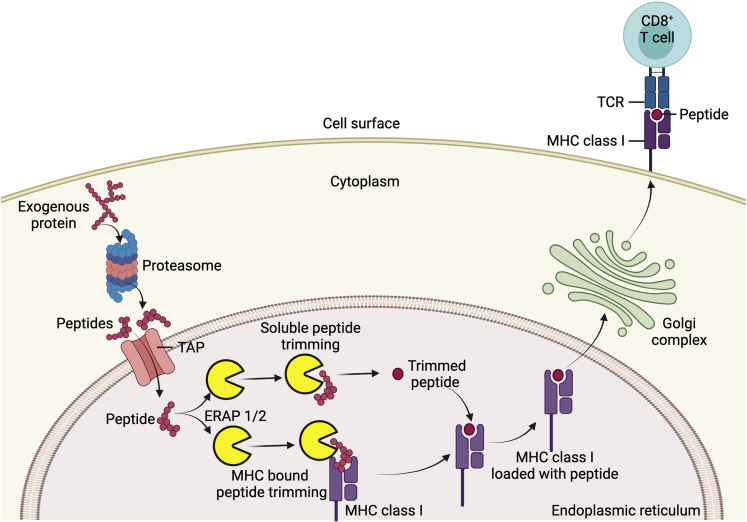
The HLA system is the most polymorphic germline genetic system known. The major histocompatibility complex includes class I genes encoded by HLA A, B, and C loci and class II genes encoded by DR, DP, and DQ loci. HLA class I molecules present antigen to CD8 T cells (Fig 1), which perform killer and suppressor functions. HLA class II molecules present antigen to CD4 T cells, which are often labeled helper T cells. For most loci, hundreds of different alleles exist. Considering that an allele is inherited from each parent and some loci have hundreds of allotypes, there are many millions of combinations of different gene products. A teleologic argument is that this genetic diversity helps to protect a species from extinction during a pandemic.
Figure 1.
Diagram showing the role of endoplasmic reticulum aminopeptidase (ERAP) in major histocompatibility complex (MHC) class I antigen processing and presentation. Exogenous proteins are digested into smaller peptides by the proteasome in the cytoplasm. These peptides are then translocated into the endoplasmic reticulum by transporter associated with antigen processing (TAP) proteins, where they undergo N-terminus trimming in the soluble or MHC bound form, by ERAP1, ERAP2, or both. The resulting peptide–MHC class I complex leaves the endoplasmic reticulum to reach cell surface via the Golgi complex. This peptide–MHC class I complex then binds to the T-cell receptor (TCR) and initiates antigen-specific response in CD8+ T cells.
Class II genes are most likely to influence susceptibility to a disease. The likelihood of many forms of uveitis developing is known to be affected by HLA alleles.6 Of all the HLA disease associations, the HLA allele that most affects the predisposition to a disease is HLA-A29, which is associated with BSCR, whose likelihood to develop increases roughly 150-fold by virtue of this HLA type.7
HLA molecules have a pocket that provides the contact with the antigenic peptide that is presented to the T cell. To be presented, the peptide must be a specific size and must include specific amino acids with specific spacing between these amino acids. For HLA class I, the peptide that is loaded into the pocket is trimmed and fitted inside the cell before the HLA complex is transported to the cell surface (Fig 1). Peptide trimming is performed by endoplasmic reticulum aminopeptidase (ERAP).8,9 Two associated genes, ERAP1 and ERAP2, can each be expressed as different isoforms. Furthermore, ERAP1 and ERAP2 have complementary and distinct trimming abilities. Just as HLA molecules affect disease susceptibility, ERAP polymorphisms also affect susceptibility to specific diseases.8 The contribution of ERAP to disease susceptibility is generally epistatic with HLA class I. Epistasis describes a gene–gene interaction. For example, for ERAP1 to affect susceptibility to ankylosing spondylitis, HLA-B27 must be present as well. The 11 diseases with arguably the best documented association between an ERAP polymorphism and susceptibility are listed in Table 1. The list includes BSCR, a disease that by definition always includes uveitis; acute anterior uveitis associated with HLA-B27, which is reportedly the most common form of uveitis in Europe and North America; and Behçet’s disease, which causes uveitis in most people with this diagnosis. It also includes Kawasaki’s disease, a rare disease whose best-known ocular manifestation is conjunctivitis, but anterior uveitis often is present as well.10 The other 7 diseases included in Table 1 are also associated with uveitis, just not as consistently as the 4 prototypes. Surely, this is a clue to the pathogenesis of uveitis.
Table 1.
Associations with Disease, HLA Class I, and Endoplasmic Reticulum Aminopeptidase
| Disease | HLA Association Class I | Endoplasmic Reticulum Aminopeptidase Association | Comment |
|---|---|---|---|
| Ankylosing spondylitis8,9 | B27 | rs26653 rs2287987 rs30187 rs10050860∗ rs17482078∗ rs27044 |
Association holds for B27-associated acute anterior uveitis as well as for ankylosing spondylitis |
| Enthesitis-related arthritis11 | B27 | rs30187 | |
| Behçet’s disease8,9 | B51 | rs10050860∗ rs17482078∗ |
|
| Type 1 diabetes9 | A∗02:01 A∗24:02 |
rs30187 | Diabetes is associated weakly with uveitis12 |
| Psoriasis8,9 | C∗06:02 | rs26653 rs30187 rs27044 |
Psoriasis is associated weakly with uveitis13 |
| Psoriatic arthritis8,14 | B27 negative B27 positive |
rs2248374 (ERAP2) rs30187 |
|
| Multiple sclerosis8,9 | C∗05 (protective) | rs30187 | |
| Inflammatory bowel disease8,9 | C∗07 | rs30187 | |
| Birdshot chorioretinopathy8,9,15,16 | A∗29:02 | rs10044354 (ERAP2) rs27432 rs2287987 |
|
| Kawasaki’s disease8,17,18 | Various | rs149481 |
Uveitis is an accepted association with all the listed diseases, but the association with uveitis is weak for diabetes and cutaneous psoriasis, as referenced. All associations are with ERAP1 polymorphisms except for birdshot chorioretinopathy and psoriatic arthritis, which also have an association with ERAP2. The table indicates 11 diseases (counting ankylosing spondylitis and acute anterior uveitis as diseases that can occur separately) with 3 common traits: an association with HLA class I, an association with an endoplasmic reticulum aminopeptidase polymorphism, and uveitis. Arthritis occurs in most of the listed diseases, but it is not typical of birdshot chorioretinopathy or multiple sclerosis.
ERAP1 variants rs100050860 and rs17482078 are in linkage disequilibrium with each other. These variants as well as rs30187 are associated with increased ERAP1 activity.
Functions beyond antigen presentation have been ascribed to both ERAP and HLA class I. For example, ERAP could modify receptors for cytokines.9 Polymorphisms in ERAP affect the immune response to viruses, including the coronavirus responsible for coronavirus disease 2019.19 Viruses are a potential trigger to cause immune-mediated disease. HLA class I could misfold and activate a cascade of biochemical events known as the unfolded protein response.20 HLA class I is recognized by a type of lymphocyte called the natural killer cell, which has been implicated in uveitis. Although it is certainly possible that HLA class I, ERAP, or both affect the likelihood of disease by a mechanism other than antigen presentation, the epistatic effect between the 2 genes strongly suggests that antigen presentation is the key function.21
An additional possibility is that antigen presentation is the critical function, but perhaps the antigen presentation is at the thymic epithelium, where ERAP is known to be active.22 The thymic epithelium is responsible for deleting clones of autoreactive T cells. A failure to achieve this central tolerance predisposes a person to uveitis, as is known to occur in a mouse model called autoimmune regulator (AIRE).23
Uveitis is not a single disease, so one would not expect that 1 factor contributes to all forms of uveitis. However, it is also reasonable to believe that many forms of uveitis would share common mechanisms.24 Both ERAP1 and ERAP2 are detected in the retina (Brooks MJ, Swaroop A, unpublished data, July 30, 2021; accessed via NEI Commons). Antigen presentation within the eye could represent a simple explanation as to why uveitis is so common in diseases associated with an ERAP polymorphism. Such an antigen could be an autoantigen, that is, endogenous, or it could be a foreign antigen such as one introduced by an infection, diet, or respiration. The peptide VTLGILVSY is contained within S-arrestin, which is uveitogenic. Because this peptide is trimmed by ERAP2, it is a potential contributor to BSCR.7 One study found that 10 of 10 patients with BSCR harbored autoantibodies to recoverin, a retinal protein,25 although it is unclear if such antibodies are related causally to the disease or merely are epiphenomena secondary to inflammation.
The role of ERAP in predisposing an eye to uveitis could involve antigen presentation without invoking a role for an antigen within the eye. The intestinal microbiome, the collection of micro-organisms that coexist within humans, educates the immune system such that laboratory animals that are raised in a germ-free environment have an impoverished immune system.26 Changes in the microbiome have been implicated in a vast array of immune-mediated diseases. We recently reported that HLA-A29 helps to shape the intestinal microbiome.27 Other HLA alleles such as HLA-B2728 and DRB128 also impact the microbiome. This seems predictable because the microbiome expresses an immense variety of antigens. In any ecosystem, altering the prevalence of a single species inevitably affects other species downstream. Recently, Paun et al29 linked the HLA-controlled immune response to commensal bacteria and the development of juvenile type I diabetes or Crohn’s disease. Pianta et al30 reported that an HLA-regulated immune response to Prevotella correlates with generating autoantibody to cyclic citrullinated peptide, which is characteristic of rheumatoid arthritis. Furthermore, lymphocytes migrate from the gut to the eye and other target organs.31,32 Accordingly, ERAP conceivably could increase the likelihood of uveitis developing via an effect on the microbiome.
Although ERAP polymorphisms such as rs10044354 for ERAP2 increase the activity of the enzyme, other polymorphisms such as rs27342 for ERAP1 reduce the activity of the enzyme.15,16 The mechanism by which reduced activity predisposes a person to an autoimmune disease is speculative, but multiple potential explanations exist. For example, if ERAP contributes to tolerance induced by the thymus, reduced activity could lead to a greater immune response. Some reciprocity exists between ERAP1 and ERAP2, so reduced activity of ERAP1 might increase the activity of ERAP2. If the disease results from an immune response to a virus, a hypomorphic allele could result in more viral antigen, and if the mechanism for disease predisposition involves the intestinal microbiome, a reduced immune response could skew the ecosystem within the gut.
A recent genome-wide association study of 286 patients with BSCR found that HLA-A29, HLA-Aw19 (a supertype that is detectable in A29, A30, A31, and A33), and polymorphisms in both ERAP1 (rs27342) and ERAP2 (rs10044354) could synergize to increase the risk of BSCR developing well beyond the effect of HLA-A29 alone.15 A prior study found the rs27342 disease-associated polymorphism in 42% of European control participants and 63% of those affected by BSCR.33 The same study found the rs10044354 disease-associated polymorphism in 65% of control participants and 83% of patients with BSCR. The rs27342 polymorphism increases the risk of BSCR developing with an odds ratio of 2.46.15 The rs10044354 polymorphism increases the risk with an odds ratio of 1.95.15 Two copies of the Aw19 supertype increase the risk of BSCR with an odds ratio of 4.44.15 All 3 of these risk factors together increase the odds ratio to 13.53.15 However, even when all such genetic risk factors are present, the chance of BSCR developing is still less than 1 in 2000, meaning that ERAP typing currently does not have clinical usefulness. Further research should focus not only on how these genetic factors predispose people to uveitis, but also on why the vast majority of people with these predisposing factors still do not demonstrate uveitis.
We find the link relating 11 diseases, ERAP, HLA class I, and uveitis unlikely to represent a mere coincidence. However, it is a coincidence that ERAP exists within therapy and seraphim. We are thus “enraptured” that deciphering the mechanism by which ERAP is linked to uveitis will provide vital clues to several forms of uveitis.
Acknowledgment
The authors thank Kimberly Ogle for contributing her playfulness with words to this editorial.
Footnotes
Further research should focus not only on how these genetic factors predispose people to uveitis, but also on why the vast majority of people with these predisposing factors still do not demonstrate uveitis.
Disclosure(s):
All authors have completed and submitted the ICMJE disclosures form.
The author(s) have made the following disclosure(s): J.T.R.: Consultant – Gilead, AbbVie, Novartis, Roche, UCB, Corvus, Roivant, Revolo, Affibody, Neoleukin, Horizon Pharmaceuticals, Santen; Financial support – Pfizer, Horizon; Data monitoring board – Celgene-Bristol Myers; Royalties – UpToDate
Supported by the National Institutes of Health, Bethesda, Maryland (grant nos.: R01 EY026572, RO1EY029266, and P30 EY010572); Research to Prevent Blindness, Inc, New York, New York; the Grandmaison Fund for Autoimmunity Research (J.T.R.); the Stan and Madelle Rosenfeld Family Trust (J.T.R.); and the William and Mary Bauman Foundation (J.T.R.).
References
- 1.Benacerraf B., McDevitt H.O. Histocompatibility-linked immune response genes. Science. 1972;175(19):273–279. doi: 10.1126/science.175.4019.273. [DOI] [PubMed] [Google Scholar]
- 2.Brewerton D.A., Hart F.D., Nicholls A., et al. Ankylosing spondylitis and HL-A 27. Lancet. 1973;1(7809):904–907. doi: 10.1016/s0140-6736(73)91360-3. [DOI] [PubMed] [Google Scholar]
- 3.Shiina T., Inoko H., Kulski J.K. An update of the HLA genomic region, locus information and disease associations: 2004. Tissue Antigens. 2004;64(6):631–649. doi: 10.1111/j.1399-0039.2004.00327.x. [DOI] [PubMed] [Google Scholar]
- 4.McGonagle D., McDermott M.F. A proposed classification of the immunological diseases. PLoS Med. 2006;3(8):e297. doi: 10.1371/journal.pmed.0030297. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Vader W., Stepniak D., Kooy Y., et al. The HLA-DQ2 gene dose effect in celiac disease is directly related to the magnitude and breadth of gluten-specific T cell responses. Proc Natl Acad Sci U S A. 2003;100(21):12390–12395. doi: 10.1073/pnas.2135229100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Davey M.P., Rosenbaum J.T. The human leukocyte antigen complex and chronic ocular inflammatory disorders. Am J Ophthalmol. 2000;129(2):235–243. doi: 10.1016/s0002-9394(99)00433-x. [DOI] [PubMed] [Google Scholar]
- 7.Kuiper J., Rothova A., de Boer J., Radstake T. The immunopathogenesis of birdshot chorioretinopathy; a bird of many feathers. Prog Retin Eye Res. 2015;44:99–110. doi: 10.1016/j.preteyeres.2014.11.003. [DOI] [PubMed] [Google Scholar]
- 8.Yao Y., Liu N., Zhou Z., Shi L. Influence of ERAP1 and ERAP2 gene polymorphisms on disease susceptibility in different populations. Hum Immunol. 2019;80(5):325–334. doi: 10.1016/j.humimm.2019.02.011. [DOI] [PubMed] [Google Scholar]
- 9.Reeves E., James E. The role of polymorphic ERAP1 in autoinflammatory disease. Biosci Rep. 2018;38(4) doi: 10.1042/BSR20171503. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Shiari R., Jari M., Karimi S., et al. Relationship between ocular involvement and clinical manifestations, laboratory findings, and coronary artery dilatation in Kawasaki disease. Eye (Lond) 2020;34(10):1883–1887. doi: 10.1038/s41433-019-0762-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Hinks A., Martin P., Flynn E., et al. Subtype specific genetic associations for juvenile idiopathic arthritis: ERAP1 with the enthesitis related arthritis subtype and IL23R with juvenile psoriatic arthritis. Arthritis Res Ther. 2011;13(1):R12. doi: 10.1186/ar3235. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Watanabe T., Keino H., Nakayama K., et al. Clinical features of patients with diabetic anterior uveitis. Br J Ophthalmol. 2019;103(1):78–82. doi: 10.1136/bjophthalmol-2017-311453. [DOI] [PubMed] [Google Scholar]
- 13.Chi C.C., Tung T.H., Wang J., et al. Risk of uveitis among people with psoriasis: a nationwide cohort study. JAMA Ophthalmol. 2017;135(5):415–422. doi: 10.1001/jamaophthalmol.2017.0569. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Popa O.M., Cherciu M., Cherciu L.I., et al. ERAP1 and ERAP2 gene variations influence the risk of psoriatic arthritis in Romanian population. Arch Immunol Ther Exp (Warsz) 2016;64(Suppl 1):123–129. doi: 10.1007/s00005-016-0444-4. [DOI] [PubMed] [Google Scholar]
- 15.Gelfman S., Monnet D., Ligocki A.J., et al. medRxiv; 2021. ERAP1, ERAP2, and two copies of HLA-Aw19 alleles increase the risk for birdshot chorioretinopathy in HLA-A29 carriers. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Kuiper J.J.W., Setten J.V., Devall M., et al. Functionally distinct ERAP1 and ERAP2 are a hallmark of HLA-A29-(birdshot) uveitis. Hum Mol Genet. 2018;27(24):4333–4343. doi: 10.1093/hmg/ddy319. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Onouchi Y. Molecular genetics of Kawasaki disease. Pediatr Res. 2009;65(5 pt 2):46R–54R. doi: 10.1203/PDR.0b013e31819dba60. [DOI] [PubMed] [Google Scholar]
- 18.Tsai F.J., Lee Y.C., Chang J.S., et al. Identification of novel susceptibility loci for Kawasaki disease in a Han chinese population by a genome-wide association study. PLoS One. 2011;6(2) doi: 10.1371/journal.pone.0016853. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Saulle I., Vicentini C., Clerici M., Biasin M. Antigen presentation in SARS-CoV-2 infection: the role of class I HLA and ERAP polymorphisms. Hum Immunol. 2021;82(8):551–560. doi: 10.1016/j.humimm.2021.05.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Colbert R.A., Delay M.L., Layh-Schmitt G., Sowders D.P. HLA-B27 misfolding and spondyloarthropathies. Adv Exp Med Biol. 2009;649:217–234. doi: 10.1007/978-1-4419-0298-6_16. [DOI] [PubMed] [Google Scholar]
- 21.Venema W.J., Hiddingh S., de Boer J.H. ERAP2 increases the abundance of a peptide submotif highly selective for the birdshot uveitis-associated HLA-A29. Front Immunol. 2021;12:634441. doi: 10.3389/fimmu.2021.634441. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Gabrielsen I.S., Viken M.K., Amundsen S.S., et al. Autoimmune risk variants in ERAP2 are associated with gene-expression levels in thymus. Genes Immun. 2016;17(7):406–411. doi: 10.1038/gene.2016.39. [DOI] [PubMed] [Google Scholar]
- 23.DeVoss J., Hou Y., Johannes K., et al. Spontaneous autoimmunity prevented by thymic expression of a single self-antigen. J Exp Med. 2006;203(12):2727–2735. doi: 10.1084/jem.20061864. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Rosenbaum J.T., Harrington C.A., Searles R.P., et al. Identifying RNA biomarkers and molecular pathways involved in multiple subtypes of uveitis. Am J Ophthalmol. 2021;226:226–234. doi: 10.1016/j.ajo.2021.01.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Gibbs E., Matsubara J., Cao S., Cui J., Forooghian F. Antigen-specificity of antiretinal antibodies in patients with noninfectious uveitis. Can J Ophthalmol. 2017;52(5):463–467. doi: 10.1016/j.jcjo.2017.03.010. [DOI] [PubMed] [Google Scholar]
- 26.Hill D.A., Artis D. Intestinal bacteria and the regulation of immune cell homeostasis. Annu Rev Immunol. 2010;28:623–667. doi: 10.1146/annurev-immunol-030409-101330. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Sternes P.R., Martin T.M., Paley M., et al. HLA-A alleles including HLA-A29 affect the composition of the gut microbiome: a potential clue to the pathogenesis of birdshot retinochoroidopathy. Sci Rep. 2020;10(1):17636. doi: 10.1038/s41598-020-74751-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Asquith M., Sternes P.R., Costello M.E., et al. HLA alleles associated with risk of ankylosing spondylitis and rheumatoid arthritis influence the gut microbiome. Arthritis Rheumatol. 2019;71(10):1642–1650. doi: 10.1002/art.40917. [DOI] [PubMed] [Google Scholar]
- 29.Paun A., Yau C., Meshkibaf S., et al. Association of HLA-dependent islet autoimmunity with systemic antibody responses to intestinal commensal bacteria in children. Sci Immunol. 2019;4(32) doi: 10.1126/sciimmunol.aau8125. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Pianta A., Chiumento G., Ramsden K., et al. Identification of novel, immunogenic HLA-DR-presented Prevotella copri peptides in patients with rheumatoid arthritis: patients with rheumatoid arthritis. Arthritis Rheumatol. 2021 May 27 doi: 10.1002/art.41807. Online ahead of print. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Ashrafi M., Kuhn K.A., Weisman M.H. The arthritis connection to inflammatory bowel disease (IBD): why has it taken so long to understand it? RMD Open. 2021;7(1) doi: 10.1136/rmdopen-2020-001558. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Nakamura Y.K., Janowitz C., Metea C., et al. Short chain fatty acids ameliorate immune-mediated uveitis partially by altering migration of lymphocytes from the intestine. Sci Rep. 2017;7(1):11745. doi: 10.1038/s41598-017-12163-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Kuiper J.J., Van Setten J., Ripke S., et al. A genome-wide association study identifies a functional ERAP2 haplotype associated with birdshot chorioretinopathy. Hum Mol Genet. 2014;23(22):6081–6087. doi: 10.1093/hmg/ddu307. [DOI] [PMC free article] [PubMed] [Google Scholar]