Oral fluids, including saliva, gingival crevicular fluid, and mucosal transudates have been widely used for monitoring drugs, hormones, and a variety of other molecules and chemical substances for over 50 years (34, 35, 53, 54, 68). During the past decade, the use of oral fluids also has been advocated as a noninvasive alternative to the collection of blood for the detection of antibodies to a number of specific bacterial (32, 82), viral (1–4, 18, 40, 61–65, 75), fungal (41), and parasitic (17, 20) agents. Particular attention has been given to the value of oral fluids for the diagnosis of infection with human immunodeficiency virus (HIV) (58, 73).
Oral-fluid testing for HIV antibodies was first reported by Archibald et al. (1–4) and Parry et al. (60, 61). As summarized by Malamud and Friedman (51), in studies with saliva conducted between 1986 and 1991, the concordance between positive serum tests and positive saliva tests for the detection of HIV antibodies ranged from 70 to 100%. The less-than-perfect agreement has led to considerable confusion regarding the efficacy of using oral fluids in screening for HIV antibody. This is due, in part, to variations in the type and volume of oral sample collected, how the sample is handled prior to testing, the concentration of immunoglobulin (Ig) G present, and if testing methods have been modified to accommodate the use of oral fluids. In early studies that reported poor sensitivity, whole saliva was used and there was little consideration for the volume and condition of the sample needed and the choice of screening assays employed. For this reason, investigators have developed specialized collection devices that enhance the level of antibodies, particularly IgG, in oral specimens, ensure sufficient specimen volume, and include reagents to prevent microbial growth and proteolytic breakdown of antibodies. In general, this has been accomplished by collecting oral fluids enriched in gingival crevicular fluid and mucosal transudate, which possess increased levels of IgG relative to saliva (46, 66). In addition, recent modifications to existing HIV antibody assays and the development of extremely sensitive assays specifically designed for oral fluids have greatly improved the accuracy of oral-based diagnostic tests for antibodies to HIV and have compensated for the low levels of antibodies present in oral secretions compared with serum (46, 66).
In this review, we assess the usefulness of saliva and other oral fluids for the detection of HIV antibodies, discuss the devices employed for specimen collection, and analyze the current reliability and accuracy of performing HIV antibody tests on oral secretions compared to serum or plasma.
SPECIMEN OF CHOICE
Whole saliva, glandular-duct saliva, or mucosal transudates are specimens that can be collected for tests to detect antibody to HIV in oral secretions. A basic understanding of these different types of oral fluids, however, is necessary in choosing which oral fluid is the most appropriate and which method of recovery is best suited for the testing system employed. Detailed information on the nomenclature, specimen collection, and immunobiology of oral fluids can be found in references 5, 46, 58, 59, and 66. Best results are obtained with oral fluids that are rich in IgG, since the primary humoral immune response to HIV infection involves mainly this class of antibodies.
Whole saliva.
Whole saliva is the fluid obtained from the mouth by expectoration and includes secretions from the parotid, submandibular, sublingual, and minor salivary glands as well as transudates of the oral mucosa. It contains mostly secretory IgA and low levels of IgG (58). Whole saliva also contains bacteria, leukocytes, mucin, desquamated epithelial cells, and food debris, which may lead to degradation of IgG by bacterial and salivary proteases and makes the specimen difficult to process due to the viscosity. Either unstimulated saliva or saliva secreted in response to exogenous stimulation can be collected. Unstimulated saliva is obtained by tilting the head forward and dribbling saliva from the lower lip into a graduated test tube fitted with a funnel. After 5 min, the subject expectorates any remaining saliva from the mouth. To stimulate saliva, Parafilm, paraffin wax, neutral gum base, or rubber bands can be employed as mechanical stimuli. Dribbled saliva has a stability of 5 days at room temperature but can be stored for longer times at 4 to −20°C (63).
Glandular-duct saliva.
Saliva from the parotid, submandibular, and sublingual glands is obtained directly from the glandular ducts with specially designed collectors. Absorbent filter paper or suction aspiration with a micropipette can be used for the collection of secretions from the minor salivary glands. Glandular-duct saliva contains predominantly secretory IgA and should be stored as described for whole saliva.
Oral mucosal transudates.
Oral mucosal transudates are fluids from the capillaries beneath the buccal mucosa and at the base of the crevice between the teeth and gums. These fluids not only contain secretory IgA but are rich in IgG and IgM that originate in the plasma and are passively transferred to the mouth across the mucosa and through the gingival crevices. The IgG concentration in oral mucosal transudates, however, is less than that in plasma but higher than that in whole saliva (46, 66). Thus, a device is required to collect these fluids in order to efficiently enrich and elute the IgG antibodies; saliva production should not be stimulated during the collection, as this will only decrease the concentration of IgG present in the final specimen. The terms “crevicular fluid,” “gingival crevicular fluid,” and “crevicular fluid saliva” also have been used to describe oral mucosal transudates.
COLLECTION DEVICES
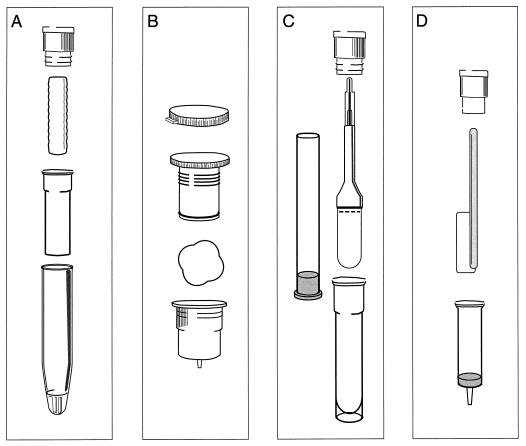
Several devices are commercially available for the collection of oral mucosal transudate specimens for the detection of HIV antibodies (Fig. 1). These include Salivette (Sarstedt Ltd., Leicester, United Kingdom), Orapette (Trinity Biotech, Dublin, Ireland), Omni-SAL (Saliva Diagnostic Systems, Inc., Vancouver, Wash.), and OraSure (Epitope, Inc., Beaverton, Oreg.). The devices are simple, safe, and convenient to use and provide an adequate homogeneous specimen with low viscosity. Specimen collection, however, must be supervised and the instructions must be carefully followed to ensure specimen adequacy. A transport buffer provided with the Omni-SAL and Orasure devices contains antimicrobial agents and protein stabilizers. Specimens may be stored at temperatures of 4 to 37°C for 21 days or at −20°C for longer periods (27, 72, 78).
FIG. 1.
Commercial devices for the collection of oral-fluid specimens. (A) Salivette; (B) Orapette; (C) Omni-SAL; (D) OraSure.
Salivette.
With the Salivette device, a compressed cylinder of cotton is placed in the mouth and is gently chewed for approximately 1 min to enhance the release of oral mucosal transudates. The saturated cotton is then placed inside the provided stoppered inner tube which has a small hole in its base. The inner tube fits into an outer tube which possesses a conical base, and oral fluids are collected by centrifugation.
Orapette.
The Orapette device includes a small, rayon ball and a snapped-cap plunger which screws into a receiving container. The rayon ball is placed in the mouth, and the subject is asked to concentrate on collecting oral fluids from around the teeth and gum area until the rayon is completely saturated. The saturated rayon ball is then placed into the receiving container, and the plunger is screwed into the container, compressing the rayon and releasing drops of oral fluid from a small opening in the container. The fluid is collected in a stoppered tube suitable for transport and testing.
Omni-SAL.
The Omni-SAL device employs a compressed, absorbent cotton pad attached to a plastic stem. The pad is placed under the tongue and absorbs fluid from the floor of the mouth. The device incorporates an indicator on the plastic stem that turns from white to blue when an adequate amount of sample has been collected. The collection pad is then inserted into a stoppered transport tube containing 1.1 ml of phosphate-buffered saline, pH 7.0, protease inhibitors, surfactants, antimicrobial agents, and 0.2% sodium azide as a preservative. In the laboratory, the collection pad is compressed and the eluate is filtered with a piston-style filter.
OraSure.
The OraSure oral-specimen collection device is the only apparatus that has been licensed by the Food and Drug Administration (FDA) for use in the detection of HIV antibodies in oral fluids. The collection device is available only by order of a physician, and certain restrictions apply to its use. The device is intended for subjects 13 years of age and older, it is not to be provided to subjects for home use or to be used to screen blood donors, the use of the device must be administered by properly trained personnel, and test subjects must receive an information pamphlet that explains the limitations of the procedure prior to specimen collection. To obtain the device from the manufacturer, a physician is required to sign a letter of agreement accepting responsibility for the proper training of personnel and use of the collection device, the reporting of test results, and counseling of the subject about the results and attesting that specimen collection and HIV antibody testing are done in accordance with laws and regulations concerning consent and confidentiality. Training materials and proficiency panels are supplied by the manufacturer to physicians or laboratories that want to become qualified to collect and test the oral-fluid specimens.
OraSure uses a flat, absorbent cotton pad which is treated with a salt solution containing 3.5% sodium chloride, 0.3% citric acid, 0.1% potassium sorbate, 0.1% sodium benzoate, and 0.1% gelatin (pH 7.2). The treated pad is then dried, attached to a plastic handle, and packaged for use. For collection of oral specimens, the subject is instructed to place the pad between the lower cheek and gum and to rub back and forth until moist. The pad is then held in place for 2 min, removed from the mouth, and placed into a stoppered transport vial with 0.8 ml of aqueous solution containing 0.5% Tween 20 and 0.01% chlorhexidine digluconate as preservatives. Following transport to the laboratory, the fluid is eluted from the pad and the eluate is recovered by centrifugation.
Foam swab.
Oral-fluid specimens can also be obtained by using a polystyrene foam swab developed at the Public Health Laboratory Service Virus Reference Division, London, United Kingdom (63). The tip of the foam swab is similar to a soft sponge and is saturated with oral mucosal transudates by rubbing the swab along the junction of the teeth and gums for approximately 1 min. The oral fluids are then extracted from the swab into 1 ml of phosphate-buffered saline, pH 7.2, containing 0.2% Tween 20 and 10% fetal calf serum and collected by centrifugation (74). This device is not commercially available.
For each device, a sufficient volume of fluid must be obtained for testing, and this is measured by the laboratory receiving the specimen or, as with the Omni-SAL device, by using an indicator of specimen adequacy. Collections typically yield from 0.5 to 1.5 ml of oral mucosal transudate. It has been reported by Mortimer and Parry (57) that for 5% of subjects it may be difficult to obtain enough specimen to saturate a collection device because of parched mouth. Also, there is no assurance with these devices that the appropriate type of fluid and concentration of IgG has been obtained. It has been suggested that IgG levels be determined for each specimen prior to testing and that only specimens with at least 0.5 mg of IgG per liter be used (57). Current methods for quantifying IgG in oral mucosal transudates, however, are complex and insensitive and add time and expense to any screening system for HIV antibody. Also, this may not be necessary with the development of more sensitive antibody assays. To our knowledge, there have been no published studies that have directly compared all of the different devices for the collection of oral fluids for the detection of HIV antibodies. The cost of using oral-fluid collection devices is comparable to the expense of collecting blood specimens, while there is little cost involved in the collection of dribbled whole saliva.
SCREENING FOR HIV ANTIBODY IN ORAL FLUIDS AND SERUM
Table 1 summarizes the data of 44 studies that compared the use of oral fluids with the use of serum for the detection of HIV antibodies. We have limited our review to data published in full manuscripts, excluding studies published only as abstracts. The mean sensitivities of oral-fluid-based screening for HIV antibody were reported to be 95.2% (range, 50 to 100%) for whole saliva compared with 98.6% (range, 92.7 to 100%) for OraSure, 98.1% (range, 90.7 to 100%) for Omni-SAL, and 97.9% (range, 88 to 100%) for Salivette. The mean specificities of HIV screening tests with oral-fluid samples were uniformly high: 99.0% (range, 84.1 to 100%) for whole saliva, 99.9% (range, 99.2 to 100%) for OraSure, 99.7% (range, 98.7 to 100%) for Omni-SAL, and 98.0% (range, 81.8 to 100%) for Salivette. Single studies involving Orapette or foam swab collection devices reported sensitivities of 100% for both systems and specificities of 99.8 and 100%, respectively. In all reported studies, the sensitivity of oral-fluid testing was measured by using samples collected from known HIV-seropositive individuals and the specificity was determined with specimens obtained from individuals who were at low risk for HIV and were seronegative.
TABLE 1.
Recent studies comparing oral fluids with serum for the detection of antibodies to HIV by screening assays
| Assaya | Sensitivityb (%) | Specificityc (%) | Reference |
|---|---|---|---|
| Whole saliva | |||
| Immune complex transfer EIA | 63/63 (100) | 76/76 (100) | Ishikawa et al., 1995 (39) |
| GACELISA VK 61 HIV-1+2 | 49/50 (98) | 57/57 (100) | Luo et al., 1995 (49) |
| Wellcozyme HIV-1+2 VK54/55 | 47/50 (94) | 57/57 (100) | |
| Wellcozyme HIV-1 VK56/57 | 48/50 (96) | 57/57 (100) | |
| Abbott TestPack HIV-1/HIV-2 | 35/35 (100) | 35/35 (100) | Fernandez et al., 1994 (21) |
| GACELISA HIV-1+2 | 71/71 (100) | 65/65 (100) | de Pilar Azinheira et al., 1994 (18)d |
| DB ELISA HIV-1 | 89/100 (89) | 94/100 (94) | Chamnanput and Phanuphak, 1993 (9) |
| Genelavia HIV-1/HIV-2 | 100/100 (100) | 96/100 (96) | |
| Abbott Recombinant HIV-1/HIV-2 | 100/100 (100) | 99/100 (99) | |
| Abbott TestPack HIV-1/HIV-2 | 100/100 (100) | 100/100 (100) | |
| Recombigen HIV-1/HIV-2 RTD | 20/20 (100) | 20/20 (100) | |
| GACRIA HIV-1 | 17/18 (94.4) | 273/273 (100) | Hunt et al., 1993 (38) |
| GACELISA HIV-1 | 8/8 (100) | 214/214 (100) | |
| Abbott TestPack HIV-1/HIV-2 | 48/50 (96) | 50/50 (100) | Urquia et al., 1993 (77) |
| GACELISA HIV-1+2 | 31/32 (96.9) | 43/43 (100) | Gershy-Damet et al., 1992 (28)e |
| GACELISA HIV-1+2 | 49/49 (100) | 50/50 (100) | Thongcharoen et al., 1992 (76) |
| Vironostika HIV-1-Mixt | 79/79 (100) | 115/115 (100) | van den Akker et al., 1992 (79) |
| Vironostika HIV-1 | 142/145 (97.9) | 313/313 (100) | Behets et al., 1991 (7) |
| Recombigen HIV-1 | 117/119 (98.3) | 429/429 (100) | Major et al, 1991 (50) |
| Vironostika HIV-1 | 35/36 (97.2) | 14/14 (100) | Holmstrom et al., 1990 (37) |
| Passive hemagglutination HIV-1 | 19/22 (86.4) | NDf | Vasudevachari et al., 1989 (81) |
| DuPont HIV-1 ELISA | 11/22 (50) | ND | |
| GACRIA HIV-1 | 196/196 (100) | 459/460 (99.8) | Johnson et al., 1988 (42) |
| Wellcozyme HIV-1 Monoclonal | 150/165 (90.9) | 404/405 (99.8) | |
| Abbott Recombinant HIV-1 | 151/184 (82.1) | 443/443 (100) | |
| Serodia HIV-1 | 175/179 (97.8) | 354/421 (84.1) | |
| GACRIA HIV-1 | 41/41 (100) | 10/10 (100) | Parry et al., 1987 (60) |
| GACELISA HIV-1 | 43/43 (100) | 10/10 (100) | |
| OraSure | |||
| GACELISA HIV-1 | 474/474 (100) | 3,940/3,948 (99.8) | Granade et al., 1998 (31) |
| Oral-fluid Vironostika HIV-1 | 470/474 (99.2) | 3,915/3,948 (99.2) | |
| Vironostika HIV-1 | 468/474 (98.7) | 3,948/3,948 (100) | |
| Oral-fluid Vironostika HIV-1 | 672/673 (99.9) | 2,893/2,897 (99.9) | Gallo et al., 1997 (26) |
| Abbott HIV-1 AB | 195/195 (100) | 198/198 (100) | Emmons et al., 1995 (19) |
| Coulter HIV-1 EIA | 108/109 (99.1) | 16/16 (100) | Gomez et al., 1994 (29) |
| Abbott Recombinant HIV-1/HIV-2 | 38/41 (92.7) | 244/244 (100) | |
| Abbott TestPack HIV-1/HIV-2 | 44/44 (100) | 243/243 (100) | Holm-Hansen et al., 1993 (36) |
| SUDS HIV-1 | 35/36 (97.2) | 230/230 (100) | |
| Organon Teknika HIV-1 EIA | 354/356 (99.4) | 1,524/1,524 (100) | Soto-Ramirez et al., 1992 (71) |
| Omni-SAL | |||
| Detect HIV 1/2 | 51/52 (98.1) | 35/35 (100) | Solomon et al., 1997 (70) |
| Abbott TestPack HIV-1/HIV-2 | 195/205 (95.1) | 295/297 (99.3) | Grant et al., 1996 (33) |
| Organon Teknika HIV-1 | 149/149 (100) | 135/136 (99.3) | Granade et al., 1995 (30) |
| Abbott 3A11 EIA | 149/149 (100) | 135/136 (99.3) | |
| GACELISA HIV-1 | 149/149 (100) | 135/136 (99.3) | |
| Recombigen HIV-1 | 358/368 (97.3) | 888/888 (100) | King et al., 1995 (43) |
| OraScreen Dipstick HIV-1/HIV-2 | 71/75 (94.7) | 195/196 (99.5) | Leow et al., 1995 (47) |
| GACELISA HIV-1+2 | 133/133 (100) | 151/153 (98.7) | Vall Mayans et al., 1995 (78) |
| Detect HIV 1/2 | 168/172 (97.7) | 814/814 (100) | Wongba et al., 1995 (83) |
| GACELISA HIV-1+2 | 115/115 (100) | 451/451 (100) | Chassany et al., 1994 (10) |
| Recombigen HIV-1 | 70/75 (93.3) | 1,404/1,405 (99.9) | Frerichs et al., 1994 (24) |
| Abbott HIV-1 AB | 74/75 (98.7) | 1,393/1,405 (99.1) | |
| GACELISA HIV-1+2 | 75/75 (100) | 1,403/1,405 (99.9) | |
| GACELISA HIV-1+2 | 300/300 (100) | 1,647/1,654 (99.6) | Frerichs et al., 1994 (25) |
| Abbott Recombinant HIV-1/HIV-2 | 139/139 (100) | 52/52 (100) | Lu et al., 1994 (48) |
| Abbott TestPack HIV-1/HIV-2 | 139/139 (100) | 52/52 (100) | |
| Recombigen HIV-1 | 315/315 (100) | 207/207 (100) | Fitzgibbons et al., 1993 (22) |
| Abbott Recombinant HIV-1/HIV-2 | 315/315 (100) | 207/207 (100) | |
| Recombigen HIV-1 | 68/75 (90.7) | 1,424/1,426 (99.9)g | Frerichs et al., 1992 (23) |
| 71/75 (94.7) | 1,423/1,425 (99.9)h | ||
| ImmunoComb II HIV-1 and HIV-2 | 62/62 (100) | 527/527 (100) | Saville et al., 1997 (67)i |
| Salivette | |||
| Abbott TestPack HIV-1/HIV-2 | 103/104 (99) | 137/137 (100) | Martinez et al., 1995 (55) |
| GACELISA HIV-1+2 | 104/104 (100) | 112/137 (81.8) | |
| Assaya | Sensitivityb (%) | Specificityc (%) | Reference |
| GACELISA HIV-1+2 | 50/50 (100) | 127/127 (100) | Connell et al., 1993 (12) |
| GACELISA HIV-1 | 4/4 (100) | 94/94 (100) | Covell et al., 1993 (15) |
| Wellcozyme HIV-1/2 ELISA | 133/135 (98.5) | 87/87 (100) | Stark et al., 1993 (72) |
| Biotest HIV-1/2 ELISA | 132/135 (97.8) | 87/87 (100) | |
| GACELISA HIV-1 | 49/50 (98) | 50/50 (100) | Crofts et al., 1991 (16) |
| Abbott Recombinant HIV-1 | 44/50 (88) | 50/50 (100) | |
| GACELISA HIV-1+2 | 42/42 (100) | 48/48 (100) | Klokke et al., 1991 (44) |
| Orapette, SalivaCard HIV-1/HIV-2 | 114/114 (100) | 501/502 (99.8) | Saville et al., 1997 (67)j |
| Foam swab, GACELISA VK 61 HIV-1+2 | 34/34 (100) | 297/297 (100) | Tess et al., 1996 (74) |
As named by the authors.
Number of reactive oral-fluid specimens/total seropositive specimens.
Number of nonreactive oral-fluid specimens/total seronegative specimens.
Includes 60 saliva specimens from HIV-2-infected individuals.
Includes eight saliva specimens from HIV-2-infected individuals.
ND, not done.
From testing performed in Myanma.
From testing performed in Vancouver, Wash.
Includes four oral-fluid specimens from HIV-2-infected individuals.
Includes five oral-fluid specimens from HIV-2-infected individuals.
McAlpine et al. (56) have reported a sensitivity range of 96.9 to 100% and a specificity range of 89.9 to 100% for 13 blood-based screening assays for antibodies to HIV, and van Kerckhoven et al. (80) have demonstrated sensitivity and specificity ranges of 96.6 to 100% and 85.6 to 100% for 36 commercial blood-based assays. A study of six commercial rapid serological tests for HIV-1 antibody conducted by Malone et al. (52) revealed a sensitivity that ranged from 89.2 to 100% and a specificity ranging from 56.5 to 100%. A more recent study by Silvester et al. (69) reported sensitivity from 99.5 to 99.9% and specificity from 99.5 to 99.9% for four commonly used blood-based enzyme immunoassays that detect antibody to both HIV-1 and HIV-2. Most of the reports in Table 1 clearly demonstrate that antibodies to HIV can be detected in whole saliva and oral mucosal transudate specimens with a sensitivity and a specificity comparable to those of assays which use serum.
The data presented also suggest that the appropriate use of an oral-fluid collection device increases the overall performance of HIV antibody screening assays compared to the collection of dribbled whole saliva, although direct comparative studies of whole saliva and the various collection devices have not been accomplished. This is thought to be due to the increased levels of IgG in oral mucosal transudates and the use of preservatives in the collection devices to maintain antibody integrity. Cordeiro et al. (14) have reported IgG levels to be 7.2 to >100 mg/liter when oral fluids are collected with a collection device. This is well above the recommended level of 0.5 mg/liter needed to accurately detect HIV antibody in oral fluids. Also, Gaudette et al. (27) have demonstrated that preservatives provided within some collection devices effectively inhibit microbial overgrowth of collected oral-fluid specimens and maintain the stability of IgG levels during long-term storage, whereas a marked and progressive decline in IgG was observed in unpreserved whole saliva. Of the collection devices for which multiple studies have been performed, use of the OraSure device resulted in the highest reported sensitivities and specificities for the detection of HIV antibodies in oral fluids.
A number of different screening assays have been employed for the detection of HIV antibodies in oral fluids (Table 1). These include both conventional enzyme immunoassays (EIA) and rapid tests designed for use with serum or plasma samples, as well as an IgG antibody capture radioimmunoassay (GACRIA) and enzyme-linked immunosorbent assay (GACELISA) optimized for the detection of HIV antibody in specimens that contain low concentrations of immunoglobulin (12). With screening assays intended primarily for serum or plasma, modifications to the system must be made for oral-fluid specimens. These have included increasing the sample volume, decreasing the sample diluent volume, increasing the specimen incubation time, and lowering the optical density cutoff. When such changes are made and assay protocols are optimized to accept oral-fluid specimens, the sensitivities and specificities of these assays are equal to those observed with serum. However, not all blood-based assays have produced consistent results for oral fluids.
The GACELISA has been used in 17 of the 44 reported studies in Table 1, and highly sensitive and specific results have been obtained with both whole saliva and specimens obtained with oral-fluid collection devices. The assay also has demonstrated superiority to many of the conventional EIA formats, as capturing HIV-specific antibody to a solid phase allows for the enhanced detection of low levels of HIV antibody in oral-fluid specimens. Recently, Lamey et al. (45) have observed excellent results when using the GACELISA on any component of saliva, including not only whole saliva and oral mucosal transudate but saliva obtained from parotid, submandibular, and minor salivary glands. Testing of oral fluids by the GACELISA has been shown by Tess et al. (74) to be an accurate and acceptable method for assessing the HIV infection status of children older than 12 months of age who were born to infected mothers. Connell and Parry (13) have also shown that the GACELISA can detect HIV antibodies in oral fluids at the same time or within a few days of seroconversion following primary infection, whereas other commercial assays may take up to 4 weeks after seroconversion to detect antibodies in oral fluids. The GACELISA is manufactured by Murex Diagnostics, Ltd., Dartford, United Kingdom, and is currently available only on the international market.
The FDA has recently approved a commercial HIV antibody test that is specifically designed for use with oral-fluid specimens. The oral-fluid Vironostika HIV-1 Microelisa system (Organon Teknika Corporation, Durham, N.C.) has been licensed for use with the associated OraSure collection device. In two large comparative trials using this system, sensitivities and specificities of 99.9% were reported by Gallo et al. (26) and 99.2% by Granade et al. (31), respectively. This EIA is identical to the serum-based Vironostika HIV-1 assay, except that the procedure has been modified by decreasing the sample dilution from 1 to 75 for serum to 1 to 2 for oral fluids. The test system is approved for use in subjects 13 years of age and older.
Rapid and simple “point-of-care” tests which use techniques involving membrane capture or particle agglutination have also been used for the detection of HIV antibody in oral fluids. Similar to the conventional EIAs described above, most of these rapid assays were originally designed to be used with serum or plasma but have been modified to analyze oral-fluid specimens. The performance of serum-based rapid assays for the detection of HIV antibody in oral fluids has been reported by many investigators to be excellent (Table 1). In particular, the TestPack HIV-1/HIV-2 (Abbott Laboratories, Abbott Park, Ill.) rapid assay performed exceedingly well, with a sensitivity of 98.6% (range, 95 to 100%) and a specificity of 99.9% (range, 99.3 to 100%) in seven published studies. Recently, a prototype oral-fluid-based rapid dipstick assay (OraScreen; Beacon Diagnostics, Foster City, Calif.) was examined by Leow et al. (47) and, when used in combination with the Omni-SAL oral-fluid collection device, was found to have a sensitivity of 94.7% and a specificity of 99.5%. The assay consists of a nitrocellulose membrane spotted with HIV-1 and HIV-2 viral lysates and two reference spots used as a negative control and for the detection of human IgG antibody. The membrane is reacted with an oral-fluid sample, followed by the addition of a conjugated secondary antibody and substrate. The assay is completed in 15 min, and a positive result is read as a blue-purple dot in the IgG reference and test wells. Saville et al. (67) have recently studied two combinations of oral-fluid collection devices and rapid assays designed specifically to detect HIV-1 and HIV-2 antibodies in oral fluids. The combinations of the Orapette collection device with SalivaCard HIV-1/HIV-2 assay from Trinity Biotech and the Omni-SAL (Saliva Diagnostics Systems) collection device with the ImmunoComb II HIV-1 and HIV-2 (Orgenics) were shown to be 100% sensitive and 99.8 to 100% specific in testing oral fluids collected from HIV-seropositive and -seronegative individuals. The SalivaCard HIV-1/HIV-2 assay involves the chromatographic diffusion of sample, enzyme-conjugated secondary antibody, and substrate along a solid matrix coated with synthetic HIV peptides. Interaction of HIV-specific antibodies with the peptide antigens results in a blue color of an intensity equal to or greater than that of a provided control. The assay is completed in 12 min. The ImmunoComb II HIV-1 and HIV-2 assay incorporates a plastic comb with 12 projections which are sensitized in three separate spots with either HIV-1 or HIV-2 synthetic peptides or goat antibodies to human IgG as an internal control. By using a modified dipstick technique, the projections are placed in a developing plate that contains six rows of 12 wells each; samples of oral fluids are placed in the first row of 12 wells, and each subsequent row of 12 wells contains a different reagent used for the next step of the assay. The comb is moved from row to row with incubations at each step, and the results are read as a gray-blue spot on the projections of the comb indicating the detection of antibody to HIV-1 or HIV-2 and the presence of human IgG antibody bound to the control spot. A total of 10 specimens and two controls can be tested in approximately 35 min with this method. The main advantages of using rapid assays for the detection of HIV antibody in oral fluids are speed, simplicity, and convenience. The performance of these assays requires limited laboratory resources and training of personnel, and many of the assays are formatted to include built-in quality controls. Unlike conventional EIAs, they also can be performed without electricity and sophisticated equipment, which may be unavailable in developing countries and under certain testing situations in the field. In conjunction with an appropriate oral-fluid collection device, certain rapid assays have demonstrated a degree of sensitivity and specificity for the detection of HIV antibody comparable to those for serum.
CONFIRMATORY TESTING
Available data on the use of Western blot techniques to confirm the presence of HIV antibody in whole saliva and oral-fluid specimens collected with various devices are summarized in Table 2. The performance of confirmatory Western blots on oral-fluid specimens was initially problematic, with many indeterminate and negative reactivities compared to results for matched serum specimens. Although reactivities to the HIV-1 envelope proteins gp160 and gp120 were observed for most oral-fluid specimens, other HIV-specific proteins were not seen with any consistency. Larger specimen volumes were also necessary to compensate for the loss of Western blot sensitivity, but adequate volumes were not always available for all oral-fluid specimens. Modifications in the chemistry and procedures of both commercial and in-house standard Western blot methods, however, have allowed for efficient and accurate detection of antibodies to HIV-1 in oral fluids by Western blot. In particular, Granade et al. (30) have developed an in-house miniaturized Western blot assay that has been optimized by decreasing the specimen dilution and increasing the specimen and conjugate incubation times. This technique requires much smaller specimen volumes than those used in conventional Western blot procedures, and specimen dilution is minimized so as not to decrease the IgG concentration in the oral fluids.
TABLE 2.
Recent studies comparing oral fluids with serum for the confirmation of antibodies to HIV by Western blot
| Western blot assay (no. of patients testeda) | No. of patients with the following oral-fluid WB resultb:
|
Reference | ||
|---|---|---|---|---|
| Posi- tive | Indeter- minate | Nega- tive | ||
| Whole saliva | ||||
| Organon Teknika | van den Akker et al., 1992 (79) | |||
| Recombinant blot (20) | 20 | 0 | 0 | |
| Standard blot (20) | 7 | 12 | 1 | |
| DuPont (142) | 137 | 5 | 0 | Behets et al., 1991 (7) |
| In-house (103) | 103 | 0 | 0 | Major et al., 1991 (50) |
| Organon Teknika (35) | 33 | 2 | 0 | Holmstrom et al., 1990 (37) |
| DuPont (19) | 19 | 0 | 0 | Vasudevachari et al., 1989 (81) |
| OraSure | ||||
| In-house (474) | 473 | 1 | 0 | Granade et al., 1998 (31) |
| Epitope OraSure (673) | 665 | 8 | 0 | Gallo et al., 1997 (26) |
| Cambridge Biotech (195) | 190 | 5 | 0 | Emmons et al., 1995 (19) |
| Pasteur (108) | 108 | 0 | 0 | Gomez et al., 1994 (29) |
| Organon Teknika (355) | 301 | 52 | 2 | Soto-Ramirez et al., 1992 (71) |
| Omni-SAL | ||||
| Bio-Rad (195) | 195 | 0 | 0 | Grant et al., 1996 (33) |
| In-house (149) | 149 | 0 | 0 | Granade et al., 1995 (30) |
| Cambridge Biotech (315) | 282 | 33 | 0 | Fittzgibbons et al., 1993 (22) |
| Salivette | ||||
| Genelabs 2.2 (102) | 102 | 0 | 0 | Martinez et al., 1995 (55) |
| Bio-Rad (47) | 26 | 19 | 2 | Crofts et al., 1991 (16) |
| Foam swab, Genelabs 2.2 (34) | 34 | 0 | 0 | Tess et al., 1996 (74) |
Data includes only matched oral-fluid and serum specimens that were positive for HIV antibodies by a screening assay.
A positive oral-fluid Western blot was defined as one demonstrating at least two envelope bands (gp 160, gp 120, or gp41) according to the World Health Organization (WHO) criteria (84) or at least two of three bands (gp160/gp120, gp41, or p24) as defined by the Centers for Disease Control and Prevention (CDC) and the Association of State and Territorial Public Health Laboratory Directors (8). Ten laboratories used the CDC criteria, three laboratories used the WHO criteria, and the criteria were not stated for two of the laboratories.
A confirmatory test for oral-fluid specimens has been approved by the FDA. The OraSure HIV-1 Western blot kit (Epitope Inc.) is a modification of the manufacturer’s Western blot assay for serum and has been licensed for use with oral-fluid specimens collected with the OraSure collection device and that are found to be repeatedly reactive by the oral-fluid Vironostika HIV-1 Microelisa system. Gallo et al. (26) recently demonstrated that this Western blot procedure has a sensitivity and a specificity comparable to those obtained for matched serum specimens for HIV antibody. The sensitivity of this assay was achieved by decreasing the specimen dilution from 1 to 51 for serum to 1 to 7.7 for oral fluids, increasing the incubation of specimens with Western blot strips from 1 h to 3 h, and using a highly sensitive enzyme-substrate combination as part of the antibody detection strategy.
APPLICATIONS
There are many advantages to using oral fluids instead of serum or plasma specimens in serological assays for antibodies to HIV. The collection of oral fluids is rapid and less invasive and does not require laboratory personnel with special training; patients can easily obtain the samples themselves. Collection of oral-fluid specimens also increases compliance (7, 11, 50) and alleviates the fear that patients may experience when having their blood drawn. It reduces the potential danger to the health professional through blood exposure and provides a safer sample to handle since saliva and oral fluids have been shown to inactivate HIV and possess less infectious virus than blood (51, 85). The use of oral fluids in screening for HIV antibody may also benefit more challenging populations whose blood may be difficult to obtain, including children, hemophiliacs, obese people, and the elderly and infirm, and may permit improved access for the surveillance of intravenous drug users, homeless persons, sex industry workers, and persons in developing countries. It has also been shown that recent food intake, dentition, poor oral hygiene, gingivitis, periodontitis, oral ulcers, tobacco consumption, and the use of anticholinergic drugs have no effect on test results for oral-fluid specimens (6, 19) and that oral-fluid testing is not affected by conditions known to be associated with increased frequency of false-positive blood-based HIV test results, such as autoimmune disease, multiple births, other viral infections, or polyclonal or monoclonal gammopathy (26). Finally, the use of oral fluids may afford a greater opportunity to screen for HIV antibodies in physicians’ and dentists’ offices, point-of-care settings, public health institutions, and community outreach programs. Although oral-fluid-based testing for HIV antibody has been suggested for home use, collection devices and antibody assays have not been approved for this purpose.
SPECIFIC ISSUES AND FUTURE CONSIDERATIONS
We have reviewed the use of oral fluids and the performance of a variety of serological assays for the detection and confirmation of HIV antibody and conclude that testing of oral fluids is a viable alternative to blood for the detection of antibodies to HIV. Although earlier studies showed inconsistent results with less than optimum sensitivity and specificity, results of more recent work are far more encouraging. With an adequate collection of oral fluids and an appropriate choice of antibody assays, testing of oral-fluid specimens is as accurate as screening blood for HIV antibody.
As with screening and confirmatory assays designed for the detection of HIV antibodies in serum or plasma, testing technologies and strategies using oral fluids continue to evolve. Developed procedures for specimen collection and testing of oral fluids require further improvement and standardization, and appropriate quality assurance programs must be established for use of oral fluids. Direct comparisons of the various oral-fluid collection methods are needed to better determine their reliability, appropriateness, ease of use, and cost. More research is also necessary to develop testing strategies and algorithms for use with oral fluids that comply with the accepted standards used for blood testing and to define testing situations where the use of oral fluids is the most appropriate. It would appear that it is only a matter of time before standard protocols are established and implemented for the use of oral fluids in donor screening, monitoring populations for the prevalence of HIV, diagnosing HIV infection, and research.
ACKNOWLEDGMENTS
This work was supported in part by grants from the National Institutes of Health (DE 09569) and World Health Direct.
REFERENCES
- 1.Archibald D W, Zon L I, Groopman J E, Allan J S, McLane M F, Essex M E. Salivary antibodies as a means of detecting human T cell lymphotropic virus type III/lymphadenopathy-associated virus infection. J Clin Microbiol. 1986;24:873–875. doi: 10.1128/jcm.24.5.873-875.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Archibald D W, Zon L, Groopman J E, McLane M F, Essex M. Antibodies to human T-lymphotropic virus type III (HTLV-III) in saliva of acquired immunodeficiency syndrome (AIDS) patients and in persons at risk for AIDS. Blood. 1986;67:831–834. [PubMed] [Google Scholar]
- 3.Archibald D W, Barr C E, Torosian J P, McLane M F, Essex M. Secretory IgA antibodies to human immunodeficiency virus in parotid saliva of patients with AIDS and AIDS-related complex. J Infect Dis. 1987;155:793–796. doi: 10.1093/infdis/155.4.793. [DOI] [PubMed] [Google Scholar]
- 4.Archibald D W, Hebert C A, Sun D, Tacket C O. Salivary antibodies to human immunodeficiency virus type 1 in a phase 1 AIDS vaccine trial. J Acquired Immune Defic Syndr. 1990;3:954–958. [PubMed] [Google Scholar]
- 5.Atkinson J C, Dawes C, Ericson T, Fox P C, Gondara B K, Malamud D, Mandel I D, Navazesh M, Tabak L A. Guidelines for saliva nomenclature and collection. Ann N Y Acad Sci. 1993;694:xi–xii. [Google Scholar]
- 6.Bagg J, Perry K R, Parry J V, Mortimer P P, Peters T J. The influence of dental status on the detection of IgG class anti-viral antibodies in human saliva. Arch Oral Biol. 1991;36:221–226. doi: 10.1016/0003-9969(91)90089-d. [DOI] [PubMed] [Google Scholar]
- 7.Behets F M, Edidi B, Quinn T C, Atikala L, Bishagara K, Nzila N, Laga M, Piot P, Ryder R W, Brown C C. Detection of salivary HIV-1-specific IgG antibodies in high-risk populations in Zaire. J Acquired Immune Defic Syndr. 1991;4:183–187. [PubMed] [Google Scholar]
- 8.Centers for Disease Control and Prevention. Interpretation and use of the Western blot assay for serodiagnosis of human immunodeficiency virus type 1 infections. Morbid Mortal Weekly Rep. 1989;38(S-7):1–7. [Google Scholar]
- 9.Chamnanput J, Phanuphak P. Comparison of eight commercial test kits for detecting anti-HIV antibodies in saliva specimens. AIDS. 1993;7:1026. doi: 10.1097/00002030-199307000-00026. [DOI] [PubMed] [Google Scholar]
- 10.Chassany O, Bergmann J F, Mazeron M C, Cervoni J, Gregoire V, Sanson-Le-Pors M J, Caulin C. Testing of anti-HIV antibodies in saliva. AIDS. 1994;8:713–714. doi: 10.1097/00002030-199405000-00026. [DOI] [PubMed] [Google Scholar]
- 11.Coates R, Millson M, Myers T, et al. The benefits of HIV antibody testing of saliva in field research. Can J Public Health. 1991;82:397–398. [PubMed] [Google Scholar]
- 12.Connell J A, Parry J V, Mortimer P P, Duncan J. Novel assay for the detection of immunoglobulin G antihuman immunodeficiency virus in untreated saliva and urine. J Med Virol. 1993;41:159–164. doi: 10.1002/jmv.1890410212. [DOI] [PubMed] [Google Scholar]
- 13.Connell J A, Parry J V. Detection of anti-HIV in saliva and urine at the time of seroconversion. Clin Diagn Virol. 1994;1:299–311. doi: 10.1016/0928-0197(94)90060-4. [DOI] [PubMed] [Google Scholar]
- 14.Cordeiro M L, Turpin C S, McAdams S A. A comparative study of saliva and OraSure® oral fluid. Ann N Y Acad Sci. 1993;694:330–331. doi: 10.1111/j.1749-6632.1993.tb18380.x. [DOI] [PubMed] [Google Scholar]
- 15.Covell R, Follett E, Coote I, Bloor M, Finlay A, Frischer M, Goldberg D, Green S, Haw S, McKeganey N. HIV testing among injecting drug users in Glasgow. J Infect. 1993;26:27–31. doi: 10.1016/0163-4453(93)96712-y. [DOI] [PubMed] [Google Scholar]
- 16.Crofts N, Nicholson S, Coghlan P, Gust I D. Testing of saliva for antibodies to HIV-1. AIDS. 1991;5:561–563. [PubMed] [Google Scholar]
- 17.del Muro R, Acosta E, Merino E, et al. Diagnosis of intestinal amebiasis using salivary IgA antibody detection. J Infect Dis. 1990;162:1360–1364. doi: 10.1093/infdis/162.6.1360. [DOI] [PubMed] [Google Scholar]
- 18.de Pilar Azinheira M, Santos Pinto A, Prieto E, Albuquerque R, Canas Ferreira W. Detection of antibodies to HIV-2 in the saliva of HIV-2-seropositive subjects. AIDS. 1994;8:849–859. doi: 10.1097/00002030-199406000-00020. [DOI] [PubMed] [Google Scholar]
- 19.Emmons W W, Paparello S F, Decker C F, Sheffield J M, Lowe-Bey F H. A modified ELISA and western blot accurately determine anti-human immunodeficiency virus type 1 antibodies in oral fluids obtained with a special collecting device. J Infect Dis. 1995;171:1406–1410. doi: 10.1093/infdis/171.6.1406. [DOI] [PubMed] [Google Scholar]
- 20.Feldman M, Plancarte A, Sandoval M, Wilson M, Flisser A. Comparison of two assays (EIA and EITB) and two samples (saliva and serum) for the diagnosis of neurocysticercosis. Trans R Soc Trop Med Hyg. 1990;84:559–562. doi: 10.1016/0035-9203(90)90040-l. [DOI] [PubMed] [Google Scholar]
- 21.Fernandez Rodriguez E, Carcaba Fernandez V, Junquera Rodriguez M, Megido Alfonso J, Amorin Garcia Z, Alonso Garcia S. Detection of HIV antibodies in saliva using a rapid diagnostic immunoenzyme assay. Rev Clin Esp. 1994;194:523–525. [PubMed] [Google Scholar]
- 22.Fitzgibbons D, Barriga G, Seymour E, Stramer S. HIV antibody EIA and western blot assay results on paired serum and saliva specimens. Ann N Y Acad Sci. 1993;694:314–316. doi: 10.1111/j.1749-6632.1993.tb18375.x. [DOI] [PubMed] [Google Scholar]
- 23.Frerichs R R, Htoon M T, Eskes N, Lwin S. Comparison of saliva and serum for HIV surveillance in developing countries. Lancet. 1992;340:1496–1499. doi: 10.1016/0140-6736(92)92755-5. [DOI] [PubMed] [Google Scholar]
- 24.Frerichs R R, Eskes N, Htoon M T. Validity of three assays for HIV-1 antibodies in saliva. J Acquired Immune Defic Syndr. 1994;7:522–525. [PubMed] [Google Scholar]
- 25.Frerichs R R, Silarug N, Eskes N, Pagcharoenpol P, Rodklai A, Thangsupachai S, Wongba C. Saliva-based HIV-antibody testing in Thailand. AIDS. 1994;8:885–894. doi: 10.1097/00002030-199407000-00004. [DOI] [PubMed] [Google Scholar]
- 26.Gallo D, George J R, Fitchen J H, Goldstein A S, Hindahl M S the OraSure HIV Clinical Trials Group. Evaluation of a system using oral mucosal transudate for HIV-1 antibody screening and confirmatory testing. JAMA. 1997;277:254–258. [PubMed] [Google Scholar]
- 27.Gaudette D, North L, Hindahl M, Griffin K, Klimkow N, Thieme T. Stability of clinically significant antibodies in saliva and oral fluid. J Clin Immun. 1994;17:171–175. [Google Scholar]
- 28.Gershy-Damet G M, Koffi K, Abouya L, Sasson-Morokro M, Brattegaard K, Parry J V, DeCock K M, Mortimer P P. Salivary and urinary diagnosis of human immunodeficiency viruses 1 and 2 infection in Cote d’Ivoire, using two assays. Trans R Soc Trop Med Hyg. 1992;86:670–671. doi: 10.1016/0035-9203(92)90184-e. [DOI] [PubMed] [Google Scholar]
- 29.Gomez C, Gutierrez M, Martinez-Acacio P, Soriano V. Evaluation of a new saliva collection device for HIV antibody screening purposes. Vox Sang. 1994;66:244. doi: 10.1111/j.1423-0410.1994.tb00319.x. [DOI] [PubMed] [Google Scholar]
- 30.Granade T C, Phillips S K, Parekh B, Pau C-P, George J R. Oral fluid as a specimen for detection and confirmation of antibodies to human immunodeficiency virus type 1. Clin Diagn Lab Immunol. 1995;2:395–399. doi: 10.1128/cdli.2.4.395-399.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Granade T C, Phillips S K, Parekh B, Gomez P, Kitson-Piggott W, Oleander H, Mahabir B, Charles W, Lee-Thomas S. Detection of antibodies to human immunodeficiency virus type 1 in oral fluids: a large-scale evaluation of immunoassay performance. Clin Diagn Lab Immunol. 1998;5:171–175. doi: 10.1128/cdli.5.2.171-175.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Granstrom G, Askelof P, Granstrom M. Specific immunoglobulin A to Bordetella pertussis antigens in mucosal secretion for rapid diagnosis of whooping cough. J Clin Microbiol. 1988;26:869–874. doi: 10.1128/jcm.26.5.869-874.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Grant R M, Piwowar E M, Katongole-Mbidde E, Muzawalu W, Rugera S, Abima J, Stramer S L, Kataaha P, Jackson B. Comparison of saliva and serum for human immunodeficiency virus type 1 antibody testing in Uganda using a rapid recombinant assay. Clin Diagn Lab Immunol. 1996;3:640–644. doi: 10.1128/cdli.3.6.640-644.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Haeckel R. Interpretation of salivary drug concentrations. J Clin Chem Clin Biochem. 1989;27:223–226. [PubMed] [Google Scholar]
- 35.Haeckel R. Saliva, an alternative specimen in clinical chemistry. J Int Fed Clin Chem. 1990;2:208–217. [Google Scholar]
- 36.Holm-Hansen C, Constantine N T, Haukenes G. Detection of antibodies to HIV in homologous sets of plasma, urine, and oral mucosal transudate samples using rapid assays in Tanzania. Clin Diagn Virol. 1993;1:207–214. doi: 10.1016/0928-0197(93)90002-m. [DOI] [PubMed] [Google Scholar]
- 37.Holmstrom P, Syrjanen S, Laine P, Valle S-L, Suni J. HIV antibodies in whole saliva detected by ELISA and western blot assays. J Med Virol. 1990;30:245–248. doi: 10.1002/jmv.1890300403. [DOI] [PubMed] [Google Scholar]
- 38.Hunt A J, Connell J, Christofinis G, Parry J V, Weatherburn P, Hickson F C I, Coxon A P M, Davies P M, McManus T J, Sutherland S. The testing of saliva samples for HIV-1 antibodies: reliability in a non-clinic setting. Genitourin Med. 1993;69:29–30. doi: 10.1136/sti.69.1.29. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Ishikawa S, Hashida S, Hashinaka K, Hirota K, Saitoh A, Takamizawa A, Shinagawa H, Oka S, Shimada K, Ishikawa E. Diagnosis of HIV-1 infection with whole saliva by detection of antibody IgG to HIV-1 with ultrasensitive enzyme immunoassay using recombinant reverse transcriptase as antigen. J Acquired Immune Defic Syndr Human Retrovirol. 1995;10:41–47. [PubMed] [Google Scholar]
- 40.Jayashree S, Bhan M K, Kumar R, Raj P, Glass R, Bhandari N. Serum and salivary antibodies as indicators of rotavirus infection in neonates. J Infect Dis. 1989;158:1117–1120. doi: 10.1093/infdis/158.5.1117. [DOI] [PubMed] [Google Scholar]
- 41.Jeganathan S, Ufomata D, Hobkirk J A, Ivanyi L. Immunoglobulin A1 and A2 subclass of salivary antibodies to Candida albicans in patients with oral candidosis. Clin Exp Immunol. 1987;70:316–321. [PMC free article] [PubMed] [Google Scholar]
- 42.Johnson A M, Parry J V, Best S J, Smith A M, de Silva M, Mortimer P P. HIV surveillance by testing saliva. AIDS. 1988;2:369–371. doi: 10.1097/00002030-198810000-00006. [DOI] [PubMed] [Google Scholar]
- 43.King A, Marion S A, Cook D, Rekart M, Middleton P J, O’Shaughnessy M V, Montaner J S G. Accuracy of a saliva test for HIV antibody. J Acquired Immune Defic Syndr Human Retrovirol. 1995;9:172–175. [PubMed] [Google Scholar]
- 44.Klokke A H, Ocheng D, Kalluvya S E, Nicoll A G, Laukamm-Josten U, Parry J V, Mortimer P P, Connell J A. Field evaluation of immunoglobulin G antibody capture tests for HIV-1 and HIV-2 antibodies in African serum, saliva and urine. AIDS. 1991;5:1391–1392. doi: 10.1097/00002030-199111000-00022. [DOI] [PubMed] [Google Scholar]
- 45.Lamey P J, Nolan A, Follett E A, Coote I, MacFarlane T W, Kennedy D H, Connell A, Parry J V. Anti-HIV antibody in saliva: an assessment of the role of the components of saliva, testing methodologies and collection systems. J Oral Pathol Med. 1996;25:104–107. doi: 10.1111/j.1600-0714.1996.tb00202.x. [DOI] [PubMed] [Google Scholar]
- 46.Lehner T. Immunology of oral diseases. 3rd ed. Oxford, United Kingdom: Blackwell Scientific Publications; 1992. Gingival crevicular fluid; pp. 18–27. [Google Scholar]
- 47.Leow Y H, Goh A, Lim P Y, Chan R K W, Goh C L, Kamarudin B A. Correlation between saliva and serum for human immunodeficiency virus type 1 and 2 antibodies using a rapid test system. Ann Acad Med Singap. 1995;24:537–540. [PubMed] [Google Scholar]
- 48.Lu X S, Delfraissy J F, Grangeot-Keros L, Rannou M T, Pillot J. Rapid and constant detection of HIV antibody response in saliva of HIV-infected patients; selective distribution of anti-HIV activity in the IgG iostype. Res Virol. 1994;145:369–377. doi: 10.1016/s0923-2516(07)80042-2. [DOI] [PubMed] [Google Scholar]
- 49.Luo N, Kasolo F, Ngwenya B-K, du Pont H L, Zumla A. Use of saliva as an alternative to serum for HIV screening in Africa. S Afr Med J. 1995;85:156–157. [PubMed] [Google Scholar]
- 50.Major C J, Read S E, Coates R A, Francis A, McLaughlin B J, Millson M, Shepherd F, Fanning M, Calzavara L, MacFadden D, Johnson J K, O’Shaughnessy M V. Comparison of saliva and blood for human immunodeficiency virus prevalence testing. J Infect Dis. 1991;163:699–702. doi: 10.1093/infdis/163.4.699. [DOI] [PubMed] [Google Scholar]
- 51.Malamud D, Friedman H M. HIV in the oral cavity: virus, viral inhibitory activity, and antiviral antibodies: a review. Crit Rev Oral Biol Med. 1993;4:461–466. doi: 10.1177/10454411930040032901. [DOI] [PubMed] [Google Scholar]
- 52.Malone J D, Smith E S, Sheffield J, Bigelow D, Hyams K C, Beardsley S G, Lewis R S, Roberts C R. Comparative evaluation of six rapid serological tests for HIV-1 antibody. J Acquired Immune Defic Syndr. 1993;6:115–119. [PubMed] [Google Scholar]
- 53.Mandel I D. The diagnostic use of saliva. J Oral Pathol Med. 1990;19:119–125. doi: 10.1111/j.1600-0714.1990.tb00809.x. [DOI] [PubMed] [Google Scholar]
- 54.Mandel I D. Saliva diagnosis: promises, promises. Ann N Y Acad Sci. 1993;694:1–10. doi: 10.1111/j.1749-6632.1993.tb18336.x. [DOI] [PubMed] [Google Scholar]
- 55.Martinez P, Ortiz de Lejarazu R, Eiros J M, Perlado E, Flores M, del Pozo M A, Rodriguez-Torres A. Comparison of two assays for detection of HIV antibodies in saliva. Eur J Clin Microbiol Infect Dis. 1995;14:330–336. doi: 10.1007/BF02116526. [DOI] [PubMed] [Google Scholar]
- 56.McAlpine L, Gandhi J, Parry J V, Mortimer P P. Thirteen current anti-HIV-1/HIV-2 enzyme immunoassays: how accurate are they? J Med Virol. 1994;42:115–118. doi: 10.1002/jmv.1890420203. [DOI] [PubMed] [Google Scholar]
- 57.Mortimer P P, Parry J V. Non-invasive virological diagnosis: are saliva and urine specimens adequate substitutes for blood? Rev Med Virol. 1991;1:73–78. [Google Scholar]
- 58.Mortimer P P, Parry J V. Detection of antibody to HIV in saliva: a brief review. Clin Diagn Virol. 1994;2:231–243. doi: 10.1016/0928-0197(94)90048-5. [DOI] [PubMed] [Google Scholar]
- 59.Navazesh M. Methods for collecting saliva. Ann N Y Acad Sci. 1993;694:72–77. doi: 10.1111/j.1749-6632.1993.tb18343.x. [DOI] [PubMed] [Google Scholar]
- 60.Parry J V, Perry K R, Mortimer P P. Sensitive assays for viral antibodies in saliva: an alternative to tests on serum. Lancet. 1987;ii:72–75. doi: 10.1016/s0140-6736(87)92737-1. [DOI] [PubMed] [Google Scholar]
- 61.Parry J V. Detection of viral antibodies in saliva specimens as an alternative to serum. J Clin Chem Clin Biochem. 1989;27:245–246. [PubMed] [Google Scholar]
- 62.Parry J V, Perry K R, Panday S, Mortimer P P. Diagnosis of hepatitis A and B by testing saliva. J Med Virol. 1989;28:255–260. doi: 10.1002/jmv.1890280410. [DOI] [PubMed] [Google Scholar]
- 63.Parry J V. Simple and reliable salivary tests for HIV and hepatitis A and B virus diagnosis and surveillance. Ann N Y Acad Sci. 1993;694:216–233. doi: 10.1111/j.1749-6632.1993.tb18355.x. [DOI] [PubMed] [Google Scholar]
- 64.Perry K R, Brown D W G, Parry J V, Panday S, Pipkin C, Richards A. Detection of measles, mumps, and rubella antibodies in saliva using antibody capture radioimmunoassay. J Med Virol. 1993;40:235–240. doi: 10.1002/jmv.1890400312. [DOI] [PubMed] [Google Scholar]
- 65.Poli A, Giannelli C, Pistello M, Zaccaro L, Pipracci D, Bendenelli M, Malvaldi G. Detection of salivary antibodies in cats infected with feline immunodeficiency virus. J Clin Microbiol. 1992;30:2038–2041. doi: 10.1128/jcm.30.8.2038-2041.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Roitt I, Lehner T, editors. Immunology of oral diseases. 2nd ed. Oxford, United Kingdom: Blackwell Scientific Publications; 1983. Oral immunity; pp. 279–304. [Google Scholar]
- 67.Saville R D, Constantine N T, Holm-Hansen C, Wisnom C, DePaola L, Falkler W A., Jr Evaluation of two immunoassays designed to detect HIV antibodies in oral fluids. J Clin Lab Anal. 1997;11:63–68. doi: 10.1002/(SICI)1098-2825(1997)11:1<63::AID-JCLA10>3.0.CO;2-N. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Schramm W, Smith R H, Craig P A, Kidwell D A. Drugs of abuse in saliva: a review. J Anal Toxicol. 1992;16:1–9. doi: 10.1093/jat/16.1.1. [DOI] [PubMed] [Google Scholar]
- 69.Silvester C, Healey D S, Cunningham P, Dax E M for the Australian HIV Test Evaluation Group. Multisite evaluation of four anti-HIV-1/HIV-2 enzyme immunoassays. J Acquired Immune Defic Syndr Human Retrovirol. 1995;8:411–419. [PubMed] [Google Scholar]
- 70.Solomon S, Kumarasamy N, Martin S P, Pandian R, Sathiamoorthy K, Thyagarajan S P. Analysis of risk factors and evaluation of HIV testing in saliva and blood samples. J Infect. 1997;34:85–86. doi: 10.1016/s0163-4453(97)80018-7. [DOI] [PubMed] [Google Scholar]
- 71.Soto-Ramírez L E, Hernández-Gómez L, Sifuentes-Osornio J, Barriga-Angulo G, Duarte de Lima D, López-Portillo M, Ruiz-Palacios G M. Detection of specific antibodies in gingival crevicular transudate by enzyme-linked immunosorbent assay for diagnosis of human immunodeficiency virus type 1 infection. J Clin Microbiol. 1992;30:2780–2783. doi: 10.1128/jcm.30.11.2780-2783.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Stark K, Warnecke C, Brinkmann V, Gelderblom H R, Bienzle U, Pauli G. Sensitivity of HIV antibody detection in saliva. Med Microbiol Immunol. 1993;182:147–151. doi: 10.1007/BF00190267. [DOI] [PubMed] [Google Scholar]
- 73.Tamashiro H, Constantine N T. Serological diagnosis of HIV infection using oral fluid samples. Bull W H O. 1994;72:135–143. [PMC free article] [PubMed] [Google Scholar]
- 74.Tess B H, Granato C, Parry J V, Santos V A, Lago T G, Newell M-L, Dunn D T, Rodrigues L C The Sao Paulo Collaborative Study for Vertical Transmission of HIV-1. Salivary testing for human immunodeficiency virus type 1 infection in children born to infected mothers in Sao Paulo, Brazil. Pediatr Infect Dis J. 1996;15:787–790. doi: 10.1097/00006454-199609000-00010. [DOI] [PubMed] [Google Scholar]
- 75.Thieme T, Yoshihara P, Piacentini S, Beller M. Clinical evaluation of oral fluid samples for diagnosis of viral hepatitis. J Clin Microbiol. 1992;30:1076–1079. doi: 10.1128/jcm.30.5.1076-1079.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Thongcharoen P, Wasi C, Louisirirotchanakul S, Parry J, Connell J, Mortimer P. Immunoglobulin G antibody capture enzyme-linked immunosorbent assay: a versatile assay for detection of anti-human immunodeficiency virus type 1 and 2 antibodies in body fluids. J Clin Microbiol. 1992;30:3288–3289. doi: 10.1128/jcm.30.12.3288-3289.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Urquia M, Rodriguez-Archilla A, Gonzales-Moles M A, Ceballos A. Detection of anti-HIV antibodies in saliva. J Oral Pathol Med. 1993;22:153–156. doi: 10.1111/j.1600-0714.1993.tb01048.x. [DOI] [PubMed] [Google Scholar]
- 78.Vall Mayans M, Casabona J, Rabella N, DeMiniac D the Ad Hoc Group for the Comparative Saliva and Serum Study. Testing of saliva and serum for HIV in high-risk populations. Eur J Clin Microbiol Infect Dis. 1995;14:710–713. doi: 10.1007/BF01690881. [DOI] [PubMed] [Google Scholar]
- 79.van den Akker R, Anneke J, van den Hoek R, van den Akker W M R, Kooy H, Vijge E, Roosendaal G, Coutinho R A, van Loon A M. Detection of HIV antibodies in saliva as a tool for epidemiological studies. AIDS. 1992;6:953–957. doi: 10.1097/00002030-199209000-00007. [DOI] [PubMed] [Google Scholar]
- 80.van Kerckhoven I, Vercauteren G, Piot P, van der Groen G. Comparative evaluation of 36 commercial assays for detecting antibodies to HIV. Bull W H O. 1991;69:753–760. [PMC free article] [PubMed] [Google Scholar]
- 81.Vasudevachari M B, Uffelman K, Pierce P F, Lane H C, Salzman N P. Detection of antibodies to human immunodeficiency virus type 1 in whole blood and saliva by using a passive hemagglutination test. J Clin Microbiol. 1989;27:2384–2385. doi: 10.1128/jcm.27.10.2384-2385.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Wienholt M G, Erbling M C, Bennetts R W, Galen E A, Cimler B M. Detection of antibodies to Helicobacter pylori using oral specimens. Ann N Y Acad Sci. 1993;694:340–342. doi: 10.1111/j.1749-6632.1993.tb18384.x. [DOI] [PubMed] [Google Scholar]
- 83.Wongba C, Pagcharoenpol P, Eskes N, Frerichs R R, Silarug N. HIV saliva test for surveillance and surveys. AIDS. 1995;9:1104–1105. [PubMed] [Google Scholar]
- 84.World Health Organization. Acquired immunodeficiency syndrome (AIDS). Proposed WHO criteria for interpreting results from Western blot assays for HIV-1, HIV-2, and HTLV-1/HTLV-II. Weekly Epidemiol Rec. 1990;65:281–283. [PubMed] [Google Scholar]
- 85.Yeung S C H, Kazazi F, Randle C G M, Howard R C, Rizvi N, Downie J C, Donovan B J, Cooper D A, Sekine H, Dwyer D E, Cunningham A L. Patients infected with human immunodeficiency virus type 1 have low levels of virus in saliva even in the presence of periodontal disease. J Infect Dis. 1993;167:803–809. doi: 10.1093/infdis/167.4.803. [DOI] [PubMed] [Google Scholar]