Abstract
Regeneration and tolerance factor (RTF) plays a pivotal role in successful pregnancy outcome and has potent immunomodulating properties. During pregnancy, it is abundantly expressed in the placenta and on peripheral B lymphocytes. Several lines of evidence suggest that both successful pregnancy outcome and progression from human immunodeficiency virus (HIV) infection to AIDS are associated with a Th2-type response. As a result, we hypothesized that the cellular expression of RTF may also be increased during infection with HIV. Using flow cytometric analysis, we showed a significantly (P < 0.01) increased expression of RTF on CD3+ cells obtained from individuals with HIV over that for individuals without HIV. On average, 32.1% of the CD3+ cells from individuals with HIV expressed high levels of RTF. In contrast, an average of only 6.7% of the CD3+ cells from individuals without HIV expressed high levels of RTF. Similar results were obtained when CD19+ cells from individuals with (mean, 44.1%) and without (mean, 25.8%) HIV were evaluated. Linear regression analysis suggested that high levels of RTF expression by CD3+ cells correlated better with viral load (r value, 0.46) than with absolute CD4 count (r value, 0.09). While additional experiments are necessary to delineate the precise immunologic role of RTF, our current data suggest that RTF expression during HIV infection may be a useful marker of immune activation.
Regeneration and tolerance factor (RTF) is a recently described 70-kDa protein that exists as a 50-kDa membrane-associated form or an extracellularly cleaved 20-kDa soluble form (2, 20). It is abundantly expressed within the developing fetal placental unit and by decidual lymphocytes (24, 29). Peripheral blood B lymphocytes of pregnant women (8, 24) and often the NK cells of pregnant women with histories of recurrent spontaneous abortion also express RTF (8). We demonstrated previously that the pregnancies of Swiss Webster mice were completely ablated when treated with monoclonal antibody (MAb) to RTF (1, 26). Collectively, these studies suggest that RTF plays a pivotal role in pregnancy. However, the precise immunologic mechanisms by which the fetal allograft survives during pregnancy are not well elucidated. Th2-type cytokines produced at the maternal fetal interface act to promote fetal growth (4, 13, 16), while Th1-type cytokines have a deleterious effect on successful pregnancy outcome (14, 32). RTF does possess potent immunomodulating properties. It inhibits the allogeneic response in a mixed lymphocyte reaction (15). These data suggest that RTF may be involved in potentiating or maintaining the Th2-type response that is essential to successful pregnancy outcome (33).
It has been proposed that progression from infection with human immunodeficiency virus (HIV) to AIDS may also be associated with a shift from a Th1- to a Th2-type response (5–7, 19). However, the findings of some studies do not support this hypothesis (10). In the current study, we investigated lymphocyte surface expression of RTF in individuals with HIV. We showed an increase in RTF expression on peripheral blood CD3+ and CD19+ cells obtained from individuals infected with HIV over that for individuals not infected with HIV.
MATERIALS AND METHODS
Study subjects.
HIV-infected individuals did not have to meet any special criteria to be included in the study. During a 12-week period, all available sodium-heparin-anticoagulated peripheral blood samples obtained from HIV-infected individuals at Mount Sinai Hospital, Chicago, Ill., were included in the study. During this time, 25 samples were collected from individuals aged 25.4 to 64.6 years (mean, 39.5 years). Of the 25 patients evaluated, 3 were inpatients and 22 were outpatients. Furthermore, all three AIDS surveillance case definitions (3) based on absolute numbers of CD4+ T lymphocytes were included in this study (category 1, >500 CD4+ T lymphocytes/μl of blood; category 2, 499 to 200 CD4+ T lymphocytes/μl of blood; and category 3, <200 CD4+ T lymphocytes/μl of blood). Sodium-heparin-anticoagulated peripheral blood samples were also obtained from 13 uninfected, healthy donors (aged 22.3 to 50.8 years; mean, 32.5 years) at Finch University of Health Sciences/The Chicago Medical School, North Chicago, Ill. The appropriate local institutional review boards approved this study.
Determination of absolute CD4 counts and acquisition of clinical information.
The percentages of CD4+ and CD8+ T lymphocytes present in the lymphocyte population of samples obtained from HIV-infected individuals were determined by using flow cytometry and appropriately conjugated antibodies. The number of lymphocytes present in the samples was determined with an automated hematology machine. Absolute CD4 count was then calculated by multiplying the percentage of CD4+ T lymphocytes by the absolute lymphocyte count. Absolute CD4 counts are expressed as the number of cells per microliter of blood. Viral load determinations were obtained by a retrospective analysis of hospital-patient records.
Antibody reagents.
Peripheral blood T and B lymphocytes were identified with phycoerythrin (PE)-conjugated antibodies to CD3 (Coulter, Miami, Fla.) and CD19 (Coulter). A combination of fluorescein isothiocyanate (FITC)-conjugated MAb to CD45 and PE-conjugated MAb to CD14 (Ortho Diagnostics, Raritan, N.J.) was also used. The murine hybridoma cell line 2C1 was used to generate anti-RTF MAb. This hybridoma was generated with a synthetic peptide (Clontech, Palo Alto, Calif.) representing amino acids 488 to 514 of the RTF gene sequence (15) and produces a MAb of the immunoglobulin G1 (IgG1) isotype (unpublished data). For ascites production, hybridoma cell line 2C1 was grown in vitro and injected into the peritoneal cavities of pristane (Sigma, St. Louis, Mo.)-primed BALB/c mice (Charles River, Wilmington, Mass.). Ascites fluid containing anti-RTF MAb was collected 14 to 21 days after injection, purified, and conjugated by standard methods (11). Following conjugation, the FITC (495 nm)-to-protein (280 nm) ratios of the MAb solutions were determined spectrophotometrically to be 1.0 and 1.1. Protein concentrations were adjusted to 0.5 mg/ml. The specificity of the anti-RTF MAb was evaluated by using murine IgG2a (Ortho Diagnostics) and IgG1 (Becton Dickinson, San Jose, Calif.) isotype control antibodies conjugated with PE and FITC. The specificity of the anti-RTF MAb was further evaluated with unconjugated mouse IgG1 (PharMingen, San Diego, Calif.).
Analysis of peripheral blood samples by flow cytometry.
Within 24 h after collection, peripheral blood samples from HIV-infected and uninfected individuals were prepared for flow cytometric analysis with the Coulter Clone Immuno-Lyse system. One-hundred-microliter samples of peripheral blood were labeled with 10 μl of antibody to either CD45/CD14, CD3, or CD19 or isotype control antibodies. Then, 30 μl of FITC-conjugated anti-RTF MAb was added to tubes containing antibody to CD3 and CD19. After labeling, samples were immediately analyzed with a Coulter Epics XL-MCL flow cytometer. Data from 10,000 cells were acquired and analyzed with histogram and dot plot profiles of PE and FITC fluorescence. Lymphocytes were identified with forward and side light scatter parameters. Conjugated antibodies to CD45/CD14 were used to validate the established lymphocyte gate. CD3+ and CD19+ cells were identified by PE fluorescence. A second gate was established around these cells, and RTF expression of CD3+ and CD19+ cells was determined by FITC fluorescence. Fluorescence negatives were defined with conjugated isotype control antibodies. Markers established with isotype control antibodies suggested that all individuals evaluated expressed RTF. To demonstrate differences in RTF expression between infected and uninfected individuals, all subsequent markers were established with samples obtained from uninfected individuals. Marker placement was established such that <12% of the CD3+ cells from >90% of all uninfected individuals evaluated expressed high levels of RTF. As a result, percentages indicate cells expressing high levels of RTF in individuals not infected and infected with HIV.
Statistical evaluation.
Statistical analysis was conducted with the Statworks computer program. P values were determined by an unpaired t test. r values were determined by linear regression analysis. The alpha level was set at 0.05 before the experiments were started. The standard error of the mean was also calculated for each of the groups represented in Fig. 3 and 4.
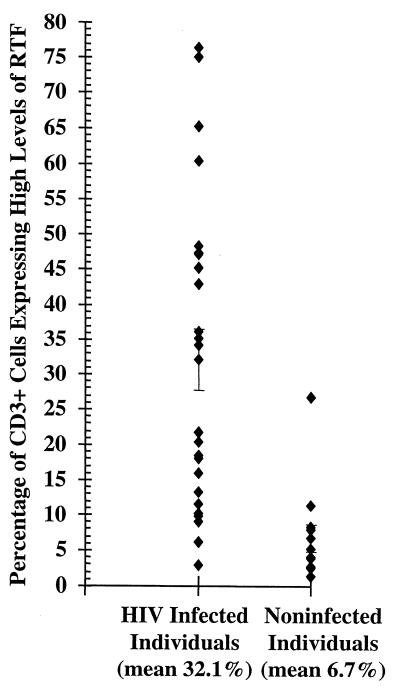
FIG. 3.
Flow cytometric analysis of CD3+ peripheral blood cells expressing high levels of RTF in individuals infected (n = 25) or not infected (n = 13) with HIV. The bars represent standard errors of the means. Mean percentages of high-level RTF expression for each group are indicated under the grouping labels on the x axis.
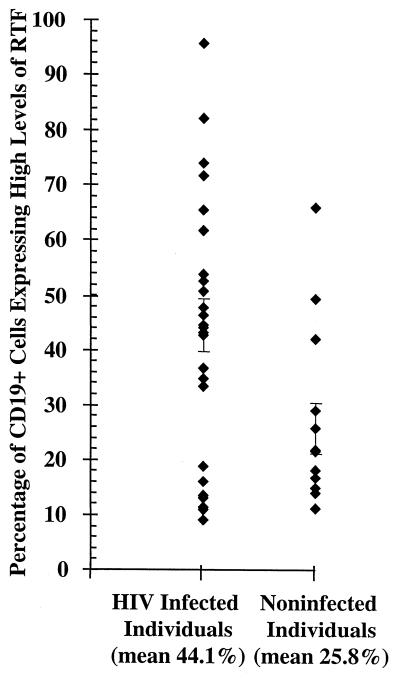
FIG. 4.
Flow cytometric analysis of CD19+ peripheral blood cells expressing high levels of RTF in individuals infected (n = 25) or not infected (n = 13) with HIV. The bars represent standard errors of the means. Mean percentages of high-level RTF expression for each group are indicated under the grouping labels on the x axis.
RESULTS
Evaluation of anti-RTF MAb specificity.
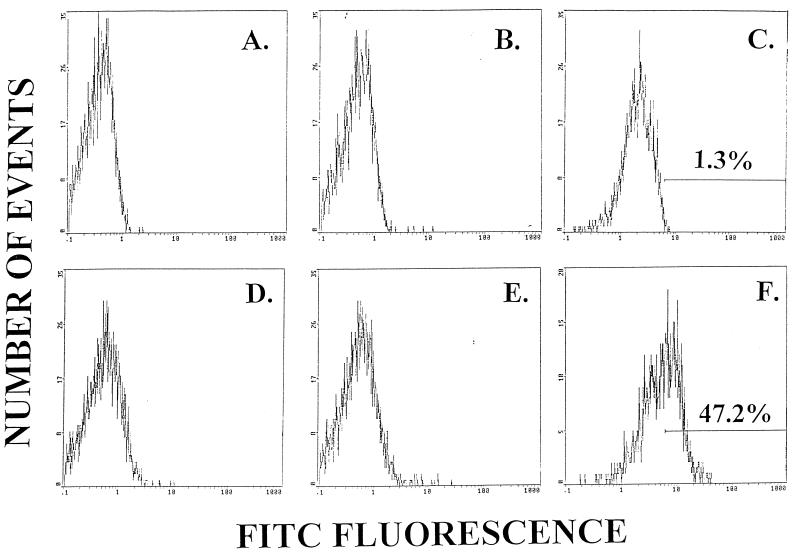
The specificity of the anti-RTF MAb was evaluated with two different isotype control antibodies. Neither IgG2a (Fig. 1A and D)- nor IgG1 (Fig. 1B and E)-conjugated isotype control antibodies bound to the surface of cells obtained from individuals without (Fig. 1A and B) or with (Fig. 1D and E) HIV infection. Furthermore, preincubation of peripheral blood samples with unconjugated mouse IgG1 (15 μg) did not decrease the amount of RTF expression detected in individuals with or without HIV infection (data not shown). Flow cytometric histograms also suggested that CD3+ T lymphocytes obtained from uninfected individuals expressed low levels of RTF (Fig. 1C). To demonstrate differences in RTF expression between infected and uninfected individuals, markers were established with samples obtained from uninfected individuals. As a result, percentages indicate cells expressing high levels of RTF in individuals not infected (Fig. 1C) and infected (Fig. 1F) with HIV.
FIG. 1.
Histograms were generated by flow cytometric analysis and with either IgG2a (A and D) or IgG1 (B and E) isotype control antibodies or anti-RTF MAb (C and F). Peripheral blood cells were obtained from an individual not infected (A to C) or infected (D to F) with HIV. Percentages indicate cells expressing high levels of RTF.
Flow cytometric analysis of RTF expression on CD3+ and CD19+ cells.
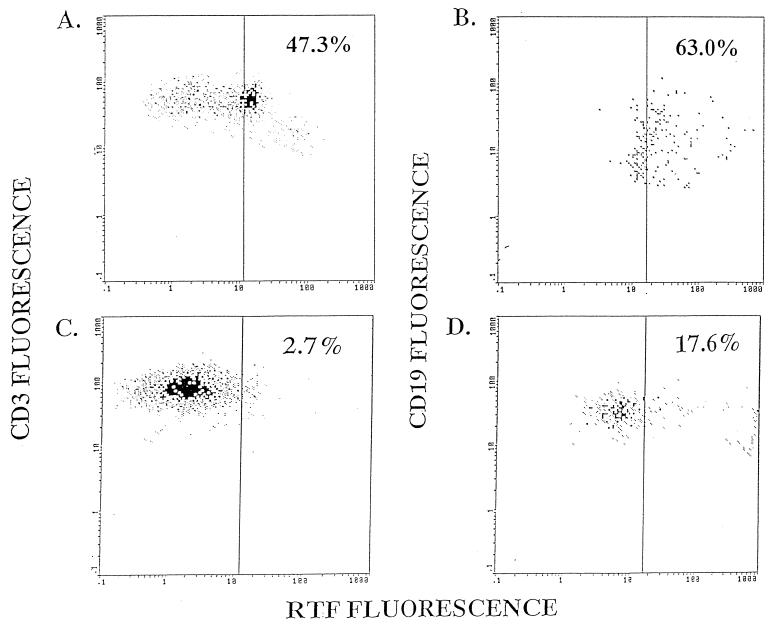
We used flow cytometric analysis to evaluate RTF expression by peripheral blood CD3+ and CD19+ cells from individuals infected and not infected with HIV. Representative flow cytometric dot plots demonstrate the increased expression of RTF on CD3+ and CD19+ cells from an individual with (Fig. 2A and B) and without (Fig. 2C and D) HIV infection. When additional individuals were evaluated, we demonstrated a significantly (P < 0.01) increased expression of RTF on CD3+ and CD19+ cells from individuals with HIV over that for uninfected individuals. On average, 32.1% of the CD3+ cells expressed high levels of RTF in individuals infected with HIV (Fig. 3). In contrast, an average of only 6.7% of the CD3+ cells expressed high levels of RTF in individuals not infected with HIV (Fig. 3). We further demonstrated that a significantly (P < 0.03) greater percentage of CD19+ cells expressed high levels of RTF in individuals infected with HIV (mean, 44.1%) than in individuals not infected with HIV (mean, 25.8%) (Fig. 4).
FIG. 2.
Flow cytometric analysis of either CD3+ (A and C) or CD19+ (B and D) peripheral blood cells expressing RTF from an individual infected (A and B) or not infected (C and D) with HIV. CD3+ and CD19+ cells were identified by PE fluorescence. These cells were gated on, and RTF expression was determined by, FITC fluorescence. Percentages indicate cells expressing high levels of RTF.
Relationship of RTF expression on CD3+ cells to other clinical parameters.
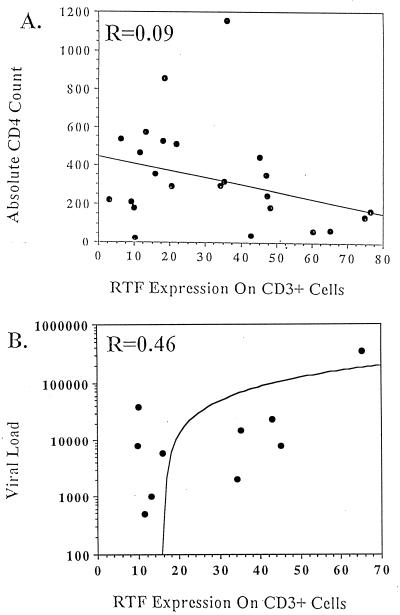
Absolute numbers of CD4+ T lymphocytes and the percentages of both CD4+ and CD8+ T lymphocytes were determined for each individual at the same time that RTF expression was evaluated (Table 1). Individuals representing all three AIDS surveillance case definitions (3) were included in the study. Linear regression analysis suggested that RTF expression by CD3+ cells did not correlate well with absolute CD4 count (Fig. 5A), as this comparison yielded an r value of 0.09. However, the five individuals with the greatest percentage of CD3+ cells expressing RTF did have absolute CD4 counts below 200 cells/μl of blood (Table 1, individuals 1 to 5). In addition, RTF expression on CD3+ cells did increase as absolute CD4 counts decreased. Viral load determinations were also obtained for a subset of HIV-infected individuals included in the study (Table 1). In most cases, viral load determinations were made at the same time that RTF expression was evaluated. Linear regression analysis yielded an r value of 0.46 when RTF expression by CD3+ cells was compared with viral load (Fig. 5B). Furthermore, RTF expression on CD3+ cells did increase as viral burden increased.
TABLE 1.
Clinical information for individuals with HIV expressing RTF on the surface of peripheral blood T lymphocytesa
| Individual | % of T lymphocytes expressing high levels of RTF | % of CD4+/CD8+ T lymphocytes | Absolute CD4 count | Viral load | Admission status |
|---|---|---|---|---|---|
| 1 | 76.4 | 22.0/47.2 | 170 | 340.0 | In (pneumonia) |
| 2 | 75.0 | 13.0/67.6 | 143 | ND | Out |
| 3 | 65.2 | 0.9/18.4 | 68 | ND | Out |
| 4 | 60.3 | 4.6/79.5 | 63 | ND | Out |
| 5 | 48.2 | 17.0/62.2 | 187 | ND | Out (h/o hepatitis C) |
| 6 | 47.3 | 6.8/10.5 | 246 | ND | Out |
| 7 | 47.2 | 37.6/38.9 | 350 | ND | Out |
| 8 | 45.2 | 27.5/65.1 | 447 | 8.2 | In (peritonitis) |
| 9 | 42.9 | 2.3/73.1 | 40 | 24.7 | Out (h/o hepatitis C) |
| 10 | 36.1 | 41.5/33.3 | 1,157 | ND | Out |
| 11 | 35.2 | 30.0/36.7 | 317 | 15.0 | Out |
| 12 | 34.3 | 60.0/21.0 | 295 | 2.1 | Out (influenza-like symptoms) |
| 13 | 32.1 | 0.2/52.5 | 2 | ND | In (cytomegalovirus retinitis) |
| 14 | 21.8 | 18.7/62.5 | 513 | ND | Out |
| 15 | 20.4 | 39.0/44.2 | 290 | ND | Out |
| 16 | 18.5 | 36.8/41.3 | 852 | ND | Out |
| 17 | 18.1 | 26.6/51.0 | 528 | ND | Out (influenza-like symptoms) |
| 18 | 16.0 | 25.9/49.7 | 356 | 5.9 | Out |
| 19 | 13.2 | 40.2/33.1 | 571 | 1.0 | Out |
| 20 | 11.5 | 21.5/60.3 | 465 | <0.5 | Out (influenza-like symptoms) |
| 21 | 10.2 | 0.9/26.2 | 23 | 40.0 | Out |
| 22 | 9.9 | 13.9/71.0 | 182 | 8.1 | Out |
| 23 | 9.1 | 20.4/64.8 | 212 | ND | Out |
| 24 | 6.1 | 27.8/51.3 | 535 | ND | Out |
| 25 | 2.9 | 18.8/42.5 | 221 | ND | Out |
The percentages of CD4+ and CD8+ T lymphocytes present in the lymphocyte population of the individuals evaluated were determined by flow cytometric analysis and with appropriately conjugated antibodies. Absolute CD4 counts were determined at the same time that RTF expression was evaluated and are expressed as the number of cells per microliter of blood. In most cases, viral load determinations were also made at the same time that RTF expression was evaluated. Viral load determinations are expressed as the number of HIV RNA copies per milliliter of blood (103). ND, not determined. Admission status refers to in- or outpatient and was recorded when RTF expression was evaluated. When known, the reason for the individual’s stay in or visit to the hospital is indicated in parentheses. h/o, history of. The majority of evaluated individuals were asymptomatic outpatients visiting the hospital for routine evaluation.
FIG. 5.
Linear regression comparison of high-level RTF expression on CD3+ cells obtained from individuals infected with HIV and either absolute CD4 count (n = 25) (A) or viral load (n = 10) (B). Linear regression coefficients (r values) are indicated in the upper left corner of each linear regression graph. Lines on graphs represent the mathematical best fit for the input data.
DISCUSSION
Human infection with HIV results in a multitude of complex immunologic changes that are often manifested by the modulation of different immune markers. In the current study, we demonstrated an increased cellular expression of RTF on CD3+ and CD19+ cells from individuals with HIV over that for uninfected individuals. Specifically, a greater percentage of CD3+ cells expressed high levels of RTF in individuals infected with HIV (mean, 32.1%) than in individuals not infected with HIV (mean, 6.7%). Similar results were obtained when CD19+ cells from individuals infected (mean, 44.1%) and not infected (mean, 25.8%) with HIV were evaluated for high levels of RTF expression. Divergent T-lymphocyte expression of RTF in these different groups of individuals may be due to the skewing of different T-lymphocyte subpopulations. The CD3+ cell populations in HIV-infected and uninfected individuals are composed of very different T-lymphocyte subpopulations. However, in preliminary studies we found that individuals with HIV expressed high levels of RTF on both CD4+ and CD8+ T lymphocytes (data not shown). Further studies are needed to conclusively delineate which T-lymphocyte subsets express RTF.
It is also possible that divergent RTF expression may be reflective of individual risk group differences. The majority of HIV-infected individuals included in the study contracted disease by heterosexual means and belong to the same risk group as the uninfected control individuals. However, a subset of HIV-infected individuals included in the study contracted disease by intravenous drug use. This risk group is not reflective of the uninfected control group.
Linear regression analysis suggested that high levels of RTF expression by CD3+ cells correlated better with viral load (r value, 0.46) than with absolute CD4 count (r value, 0.09). These results may suggest that increased RTF expression is designative of an immunologic change(s) not indicated by an absolute CD4 count. These results may also indicate that the relationship between RTF expression and absolute CD4 count is nonlinear. That is, expression of RTF may not correlate with absolute CD4 count until the health of an individual is in an advanced deleterious state. In support, we showed that the five individuals with the greatest percentage of CD3+ cells expressing RTF all had absolute CD4 counts below 200 cells/μl of blood. More-comprehensive studies may provide more-definitive information regarding the relationship of RTF expression to absolute CD4 count and viral load.
It is also possible that RTF expression may not correlate with the degree of immune deficiency but may be coordinately upregulated during the states of immune activation that exist during bacterial and viral infections. Infection with HIV is known to result in immune activation and the subsequent modulation of several different immune activation markers (9, 12, 17, 18, 27, 30). However, it is possible that increased expression of RTF could be attributed to the presence of a secondary infection and not to infection with HIV. Three of the 25 individuals evaluated for RTF expression in this study were hospitalized with documented secondary infections (Table 1). In addition, two of the outpatients evaluated had histories of hepatitis C virus infection and three had influenza-like symptoms (Table 1). However, a more plausible explanation is that RTF expression is coordinately upregulated during states of immune activation including infection with HIV. In support, the majority of HIV-infected individuals evaluated in this study were asymptomatic outpatients but still had increased RTF expression. Although no immune activation marker is specific to infection with HIV, several have been shown to have prognostic value (12, 17, 18, 30). The very pronounced increase in RTF expression detected in individuals infected with HIV over that for uninfected individuals suggests that evaluation of RTF expression may also be a potentially useful immunologic marker. Long-term evaluation of HIV-infected individuals is necessary to determine if RTF expression changes with clinical status during both remission and progression of disease.
Our current findings also raise intriguing questions regarding the precise immunologic role of RTF expression during HIV infection. We have shown previously that RTF plays a pivotal role in successful pregnancy outcome (1, 8, 26). Successful pregnancy is known to be associated with a polarization from a Th1- to a Th2-type response (33). Similarly, the progression from infection with HIV to AIDS may also be driven by a shift from a Th1- to a Th2-type response (5–7, 19). The results of our current study demonstrate a very pronounced increase in the percentage of cells expressing high levels of RTF during infection with HIV. Collectively, the results of these studies suggest that expression of RTF may be coordinately upregulated during Th2-type responses. However, other possible immunologic roles for RTF expression during HIV infection exist. Direct infection of T lymphocytes with HIV can result in cell death by a variety of mechanisms (28). Apoptosis has been suggested as a mechanism of T-lymphocyte depletion (21, 31). Cells from HIV-infected individuals do have an increased propensity to undergo apoptosis (21–23, 25), particularly in individuals with progressed disease (22, 25). Our unpublished data suggest that RTF is coordinately expressed on the surface of apoptotic cells. Further studies are needed to determine if T lymphocytes expressing RTF during HIV infection are also apoptotic.
ACKNOWLEDGMENTS
We thank Sondra Allen at Mount Sinai Hospital, Chicago, Ill., for assistance with compiling patient information. We also thank Gail Hoppe and Catrina Crociani at The Chicago Medical School, North Chicago, Ill., for their clerical and technical assistance.
REFERENCES
- 1.Beaman K D, Hoversland R C. Induction of abortion in mice with a monoclonal antibody specific for suppressor T-lymphocyte molecules. J Reprod Fertil. 1988;82:691–696. doi: 10.1530/jrf.0.0820691. [DOI] [PubMed] [Google Scholar]
- 2.Beaman K D, Angkachatchai V, Gilman-Sachs A. TJ6: the pregnancy-associated cytokine. Am J Reprod Immunol. 1996;35:338–341. doi: 10.1111/j.1600-0897.1996.tb00490.x. [DOI] [PubMed] [Google Scholar]
- 3.Centers for Disease Control. 1993 revised classification system for HIV infection. Morbid Mortal Weekly Rep. 1992;44:RR–17. [PubMed] [Google Scholar]
- 4.Chaouat G, Meliani A A, Martal J, Raghupathy R, Elliot J, Mosmann T, Wegmann T G. IL-10 prevents naturally occurring fetal loss in the CBA X DBA/2 mating combination, and local defect in IL-10 production in this abortion-prone combination is corrected by in vivo injection of IFN-γ. J Immunol. 1995;154:4261–4268. [PubMed] [Google Scholar]
- 5.Clerici M, Shearer G M. A Th1-Th2 switch is a critical step in the etiology of HIV infection. Immunol Today. 1993;14:107–111. doi: 10.1016/0167-5699(93)90208-3. [DOI] [PubMed] [Google Scholar]
- 6.Clerici M, Shearer G M. The Th1-Th2 hypothesis of HIV infection: new insights. Immunol Today. 1994;15:575–581. doi: 10.1016/0167-5699(94)90220-8. [DOI] [PubMed] [Google Scholar]
- 7.Clerici M, Wynn T A, Berzofsky J A, Blatt S P, Hendrix C W, Sher A, Coffman R L, Shearer G M. Role of interleukin-10 in T helper cell dysfunction in asymptomatic individuals with the human immunodeficiency virus. J Clin Invest. 1994;93:768–775. doi: 10.1172/JCI117031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Coulam C B, Beaman K D. Reciprocal alteration in circulating TJ6+ CD19+ and TJ6+ CD56+ leukocytes in early pregnancy predicts success or miscarriage. Am J Reprod Immunol. 1995;34:219–224. doi: 10.1111/j.1600-0897.1995.tb00944.x. [DOI] [PubMed] [Google Scholar]
- 9.Ginaldi L, DeMartinis M, D’Ostilio A, DiGennaro A, Martini L, Quaglino D. Altered lymphocyte antigen expression in HIV infection. Hematol Pathol. 1997;108:585–592. doi: 10.1093/ajcp/108.5.585. [DOI] [PubMed] [Google Scholar]
- 10.Graziosi C, Pantaleo G, Gantt K R, Fortin J P, Demarest J F, Cohen O J, Sekaly R P, Fauci A S. Lack of evidence for the dichotomy of TH1 and TH2 predominance in HIV-infected individuals. Science. 1994;265:248–252. doi: 10.1126/science.8023143. [DOI] [PubMed] [Google Scholar]
- 11.Harlow E, Lane D. Antibodies: a laboratory manual. Cold Spring Harbor, N.Y: Cold Spring Harbor Laboratory; 1988. p. 354. [Google Scholar]
- 12.Henne V, Frei P, Burgisser P. Beta-2-microglobulin—a rapid and automated determination for a broad range of clinical applications. Anticancer Res. 1997;17:2915–2918. [PubMed] [Google Scholar]
- 13.Krishnan L, Guilbert L, Russell A, Wegmann T, Mosmann T, Belosevic M. Pregnancy impairs resistance of C57BL/6 mice to Leishmania major infection and causes decreased antigen specific IFN-γ response and increased production of T helper 2 cytokines. J Immunol. 1996;156:644–652. [PubMed] [Google Scholar]
- 14.Krishnan L, Guilbert L, Wegmann T, Belosevic M, Mosmann T. T helper 1 response against Leishmania major in pregnant CD57BL/6 mice increases implantation failure and fetal resorptions. Correlation with increased IFN-γ and TNF and reduced IL-10 production by placental cells. J Immunol. 1996;156:653–662. [PubMed] [Google Scholar]
- 15.Lee C, Ghoshal K, Beaman K D. Cloning of a cDNA for a T cell produced molecule with a putative immune regulatory role. Mol Immunol. 1990;27:1137–1144. doi: 10.1016/0161-5890(90)90102-6. [DOI] [PubMed] [Google Scholar]
- 16.Lin H, Mosmann T, Guilbert L, Tuntipopipat S, Wegmann T. Synthesis of T helper 2-type cytokines at the maternal-fetal interface. J Immunol. 1993;151:4562–4573. [PubMed] [Google Scholar]
- 17.Liu Z, Cumberland W G, Hultin L E, Prince H E, Detels R, Giorgi J V. Elevated CD38 antigen expression on CD8+ T cells is a stronger marker for the risk of chronic HIV disease progression to AIDS and death in the Multicenter AIDS Cohort Study than CD4+ cell count, soluble immune activation markers, or combinations of HLA-DR and CD38 expression. J Acquired Immune Defic Syndr Hum Retrovirol. 1997;16:83–92. doi: 10.1097/00042560-199710010-00003. [DOI] [PubMed] [Google Scholar]
- 18.Liu Z, Hultin L E, Cumberland W G, Hultin P, Schmid I, Matud J L, Detels R, Giorgi J V. Elevated relative fluorescence intensity of CD38 antigen expression on CD8+ T cells is a marker of poor prognosis in HIV infection: results of 6 years of follow-up. Cytometry. 1996;26:1–7. doi: 10.1002/(SICI)1097-0320(19960315)26:1<1::AID-CYTO1>3.0.CO;2-L. [DOI] [PubMed] [Google Scholar]
- 19.Maggi E, Giudizi M G, Biagiotti R, Annunziato F, Manetti R, Piccinni M-P, Parronchi P, Sampognaro S, Giannarini L, Zuccati G, Romagnani S. Th2-like CD8+ T cells showing B cell helper function and reduced cytolytic activity in human immunodeficiency virus type 1 infection. J Exp Med. 1994;180:489–495. doi: 10.1084/jem.180.2.489. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Mandal M, Beaman K D. Purification and characterization of a pregnancy-associated protein: TJ6s. Am J Reprod Immunol. 1995;33:60–67. doi: 10.1111/j.1600-0897.1995.tb01139.x. [DOI] [PubMed] [Google Scholar]
- 21.Martin S J, Green D R. Apoptosis during HIV infection. A cytopathic effect of HIV or an important host-defense mechanism against viruses in general? Adv Exp Med Biol. 1995;374:129–138. [PubMed] [Google Scholar]
- 22.McCloskey T W, Oyaizu N, Kaplan M, Pahwa S. Expression of the Fas antigen in patients infected with human immunodeficiency virus. Cytometry. 1995;22:111–114. doi: 10.1002/cyto.990220206. [DOI] [PubMed] [Google Scholar]
- 23.Muro-Cacho C A, Pantaleo G, Fauci A S. Analysis of apoptosis in lymph nodes of HIV-infected persons. Intensity of apoptosis correlates with the general state of activation of the lymphoid tissue and not with stage of disease or viral burden. J Immunol. 1995;154:5555–5566. [PubMed] [Google Scholar]
- 24.Nichols T C, Kang J A, Angkachatchai V, Beer A E, Beaman K D. Expression of a membrane form of the pregnancy-associated protein TJ6 on lymphocytes. Cell Immunol. 1994;155:219–229. doi: 10.1006/cimm.1994.1114. [DOI] [PubMed] [Google Scholar]
- 25.Patki A H, Georges D L, Lederman M M. CD4+-T-cell counts, spontaneous apoptosis, and Fas expression in peripheral blood mononuclear cells obtained from human immunodeficiency virus type 1-infected subjects. Clin Diagn Lab Immunol. 1997;4:736–741. doi: 10.1128/cdli.4.6.736-741.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Ribbing S L, Hoversland R C, Beaman K D. T-cell suppressor factors play an integral role in preventing fetal rejection. J Reprod Immunol. 1988;14:83–95. doi: 10.1016/0165-0378(88)90038-1. [DOI] [PubMed] [Google Scholar]
- 27.Rizzardi G P, Tambussi G, Barcellini W, Capiluppi B, Clerici E, Maestra L L, Lillo F, Pantaleo G, Lazzarin A. Soluble CD30, tumour necrosis factor (TNF)-alpha, and TNF receptors in primary HIV-1 infection: relationship with HIV-1, RNA, clinical outcome and early antiviral therapy. J Biol Regul Homeost Agents. 1997;11:43–49. [PubMed] [Google Scholar]
- 28.Rosenberg Z F, Fauci A S. The immunopathogenesis of HIV infection. Adv Immunol. 1989;47:377–431. doi: 10.1016/s0065-2776(08)60665-3. [DOI] [PubMed] [Google Scholar]
- 29.Rubesa G, Beaman K D, Lucin P, Beer A E, Rukavina D. Expression of TJ6 protein in the human first trimester decidual lymphocytes. Reg Immunol. 1994;6:331–333. [Google Scholar]
- 30.Stein D S, Lyles R H, Graham N M, Tassoni C J, Margolick J B, Phair J P, Rinaldo C, Detels R, Saah A, Bilello J. Predicting clinical progression or death in subjects with early-stage human immunodeficiency virus (HIV) infection: a comparative analysis of quantification of HIV RNA, soluble tumor necrosis factor type II receptors, neopterin, and beta2-microglobulin. J Infect Dis. 1997;176:1161–1167. doi: 10.1086/514108. [DOI] [PubMed] [Google Scholar]
- 31.Sunila I, Vaccarezza M, Pantaleo G, Fauci A S, Orenstein J M. gp120 is present on the plasma membrane of apoptotic CD4 cells prepared from lymph nodes of HIV-1-infected individuals: an immunoelectron microscopic study. AIDS. 1997;11:27–32. doi: 10.1097/00002030-199701000-00005. [DOI] [PubMed] [Google Scholar]
- 32.Tezabwala B, Johnson P, Rees R. Inhibition of pregnancy viability in mice following IL-2 administration. Immunology. 1989;67:115–119. [PMC free article] [PubMed] [Google Scholar]
- 33.Wegmann T G, Lin H, Guilbert L, Mosmann T R. Bidirectional cytokine interactions in the maternal-fetal relationship: is successful pregnancy a TH2 phenomenon? Immunol Today. 1993;14:353–356. doi: 10.1016/0167-5699(93)90235-D. [DOI] [PubMed] [Google Scholar]