Abstract
Introduction and importance
Teratomas typically are benign gonadal neoplasms, arising from more than one embryonic germ layer. Extragonadal teratomas are rare and primary adrenal teratomas more so, with few documented cases. We present one such case, diagnosed via CT, resected via laparoscopic adrenalectomy, and confirmed on histology. To the best of our knowledge, this is the first case documented in the Caribbean.
Case presentation
A 38-year-old obese female with restrictive lung disease presented with right back/flank pain due to a non-functional 10.5 cm right adrenal mass on CT, likely a giant myelolipoma. Further radiologic review suggested this was instead a mature adrenal teratoma. She underwent a laparoscopic adrenalectomy and histology confirmed a mature adrenal teratoma.
Clinical discussion
Most adrenal tumours are incidentalomas and are usually benign adenomas. Primary adrenal teratomas account for 1 % of teratomas and 0.13 % of adrenal tumours. They may be mature or immature; the latter carries a greater risk of malignancy. Benign adrenal teratomas are typically non-functional and commonly mistaken for myelolipomas on imaging. Adrenalectomy is required due to the risk of malignant transformation. The laparoscopic approach depends on size, localized tissue invasion and technical considerations, but offers advantages for the patient if possible.
Conclusions
Though uncommon, preoperative radiologic diagnosis of an adrenal teratoma is possible and should be completely resected after a functional workup. A laparoscopic adrenalectomy is preferred once this can be done safely, even when very large, with surgical and oncologic outcomes equivalent to an open approach combined with the known advantages of laparoscopic surgery.
Abbreviations: CECT, contrast-enhanced computed tomography; CT, computed tomography; HU, Hounsfield units
Keywords: Adrenal, Mature, Teratoma, Laparoscopic adrenalectomy, Case report, Myelolipoma
Highlights
-
•
Teratomas are typically gonadal in origin.
-
•
Primary adrenal teratomas are exceedingly rare.
-
•
Accurate pre-operative diagnosis is uncommon.
-
•
The standard of care is laparoscopic adrenalectomy.
-
•
Large size increases difficulty but is not a contraindication.
1. Introduction
Teratomas are typically benign neoplasms, arising from at least two embryonic germ layers, consisting of tissues foreign to the anatomic site in which they are found. They often grow large enough to cause compressive symptoms and pain [1]. Only 15 % of teratomas are extragonadal [2], [3] and occurrence in the adrenals is exceedingly rare, accounting for 1 % of all teratomas, 1–4 % of excised adrenal nodules and 0.13 % of adrenal tumours [4], [5]. There is a paucity of literature on the subject, with no cases reported from the Caribbean. Definitive diagnosis is seldom made before excision, and they were predominantly believed to be myelolipomas [6]. We present a 38-year-old female who underwent laparoscopic adrenalectomy for a giant right adrenal mature teratoma diagnosed on CT.
This case report has been reported in line with the SCARE criteria [7].
2. Case presentation
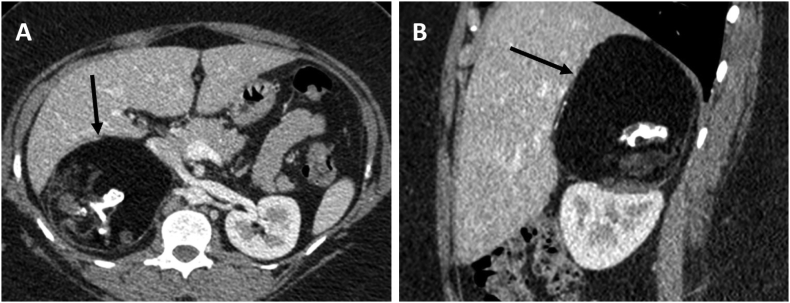
A 38-year-old morbidly obese female (BMI-57.3) with restrictive lung disease secondary to covid-19 and unremarkable family, genetic and psychosocial history presented with new-onset intermittent right flank pain in December 2021 for which she visited her family physician. Abdominopelvic CECT revealed a 10.5 cm × 10 cm × 8.0xm encapsulated right adrenal mass, likely a myelolipoma. Further pre-operative radiologic review noted the mass contained fat with coarse calcifications, rim calcification and mild internal soft tissue density, suggestive of a mature adrenal teratoma (Fig. 1).
Fig. 1.
A - Axial and B - sagittal abdominopelvic CECT demonstrating a well-defined right adrenal gland mass containing predominantly fat density, as well as intra-lesional mural soft-tissue and rim-like peripheral calcification.
Urinary metanephrines, 24 h urinary cortisol, electrolytes, TSH, renal and liver functions and complete blood count were normal. Due to the patient's morbid obesity and the tumour being non-functional and likely benign, a staged procedure of a bariatric operation followed by adrenalectomy was suggested. The patient opted for the adrenalectomy first.
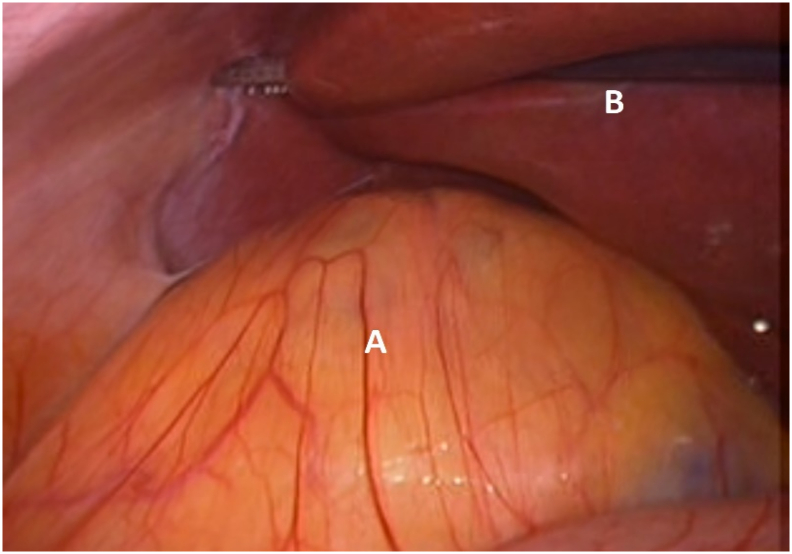
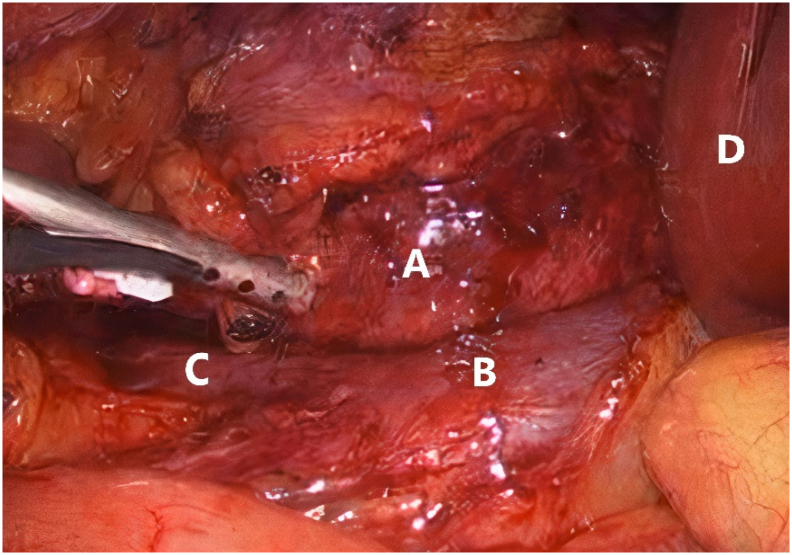
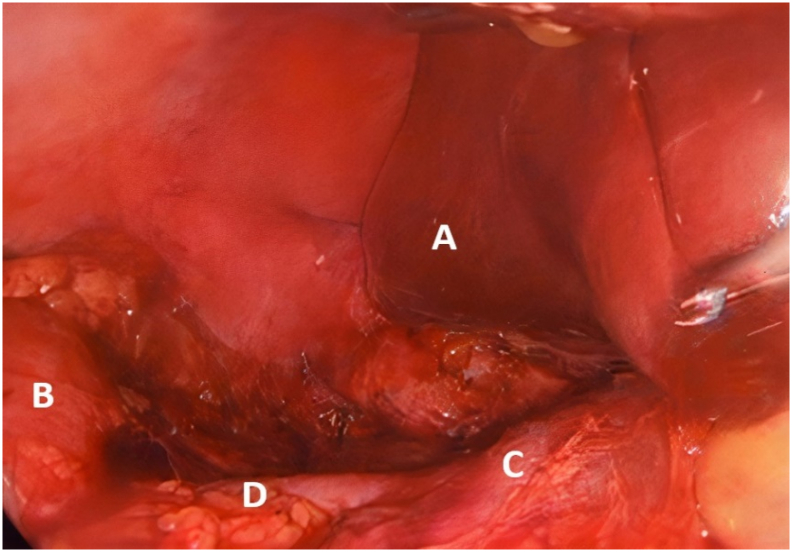
Transperitoneal laparoscopic right adrenalectomy was done by a board-certified laparoscopic surgeon using 4 ports. The tumour displaced the kidney and renal vessels inferiorly, inferior vena cava medially and liver superiorly (Fig. 2). These were all preserved, and the tumour was completely excised (Fig. 3, Fig. 4). It was placed in a bag and removed piecemeal via a 12 mm port site. She was discharged on post-op day one. There were no complications.
Fig. 2.
Adrenal tumour in-tact prior to dissection (A) with liver superiorly (B).
Fig. 3.
Adrenal tumour (A) and its relations to the inferior vena cava (B), right renal vein (C) and liver (D).
Fig. 4.
Tumour bed post-resection showing liver (A), upper pole of right kidney (B), inferior vena cava (C) and right renal vein (D) all intact after complete excision of the tumour.
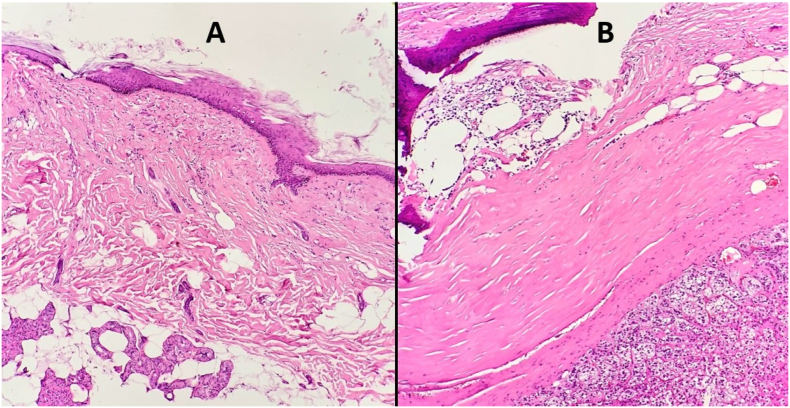
The specimen grossly consisted of tan/brown to tan/white soft tissue with hair-bearing and firm, cartilaginous areas aggregating to 17.0 × 9.0 × 4.0 cm. Histology revealed fragments of adrenal gland with sebaceous glands, cartilage, bone, skin, and hair, most compatible with a mature teratoma with no immature elements and one benign lymph node. Sections are shown in Fig. 5A and B.
Fig. 5.
A - Squamous epithelium-lined soft tissue with sebaceous units; and B - dense fibrosis with bone and bone marrow with maturing trilineage haematopoiesis (top left) and unremarkable adrenal gland parenchyma (bottom right).
3. Discussion
The adrenal glands are retroperitoneal, lying anterosuperior to the upper part of each kidney. They are somewhat asymmetrical, yellowish and lie in their own compartment of renal fascia. The outer cortex (derived from mesoderm) consists of zona glomerulosa, fasciculata and reticularis, producing glucocorticoids, mineralocorticoids and androgens, and the inner medulla (ectodermal in origin) produces catecholamines [8].
Most adrenal tumours are incidentalomas (adrenal mass larger than 1 cm discovered during diagnostic evaluation or treatment of another condition), found in up to 5.0 % of people undergoing abdominal CT. Though typically benign adenomas, they may be functional, causing clinically recognized syndromes. Only 2 % are primary carcinomas, but these account for 25 % of giant adrenal masses (larger than 6 cm) [9], [10].
Teratomas are non-seminomatous germ cell tumours and are common congenital neoplasms. They arise from more than one of three embryonic germ layers and are composed of tissue foreign to the anatomic site in which they are found. They may be solid, cystic, mixed, mature, or immature. Immature teratomas have greater malignant potential [1]. Extragonadal teratomas (15 % of teratomas) may occur anywhere along the midline but most commonly includes the mediastinum, sacrococcygeal region, retroperitoneum, and pineal gland. Only 4 % occur in the retroperitoneum [2], [3].
Teratomas account for 0.13 % of all adrenal tumours [11]. Definitive CT diagnosis is rare; most were reported as myelolipomas [6]. Adrenal myelolipomas are the most common non-functional lipomatous adrenal tumour [12] consisting of fatty tissue and bone marrow-like haematopoietic tissue. They are usually asymptomatic and non-functional, ranging from 2 to 13 cm. They are hyperechoic on ultrasound, but due to their lipid content, they show attenuation <0-HU with foci of hyperattenuating haematopoetic elements which show contrast enhancement [13], [14].
Teratomas, also typically non-functional, are characterized by the development of solid components superimposed on pre-existing cystic components. Due to its derivation from multiple embryonic germ cell layers, gross appearance includes hair, teeth, bone, calcifications, soft tissue, and fat, while microscopic appearances include muscle cells, glandular epithelium, squamous epithelium, and osteocytes. Unenhanced CT reveals a well-defined mixed density mass with low-density cystic and fatty areas and high-density calcifications with visible septations. Egg-shell calcification is a characteristic manifestation. With IV contrast, there is slight enhancement of the substantial part and a hyperdense peripheral rim and internal septations [14].
Mature teratomas are defined by at least a 90 % well-differentiated component. Malignant adrenal teratomas can be immature teratomas, teratomas with other malignant germ cell tumours or mature teratomas with malignant transformation. The latter are rare and usually gonadal. Most adrenal teratomas (90 %) are benign. Significant enhancement of the cyst wall, septations and mural nodules, and abnormal levels of hormones such as vanillylmandelic acid, cortisol, adrenocorticotropic hormone, and aldosterone suggest malignant transformation [5]. Niu et al. report a mature adrenal teratoma with malignant transformation. CT revealed intense enhancement of mural nodules, and biochemical markers as above were elevated (apart from normal aldosterone). These findings may support a diagnosis of an adrenal teratoma with malignant transformation [11].
Primary retroperitoneal teratomas do not originate from any organ but are typically found in the region of the left adrenal gland, carry a 25.8 % risk of malignancy, and can secondarily involve the adrenal, suggesting similar pathogenesis and sparking debate as to whether they are separate entities [6]. It is sometimes difficult to distinguish primary adrenal vs retroperitoneal teratomas radiologically and diagnosis is confirmed surgically or pathologically [15].
Contemporary case series and literature reviews found 18 published cases of primary adrenal teratomas in adults [5], [6], [16]. Other series report larger numbers, but without distinguishing primary adrenal teratomas from primary retroperitoneal teratomas secondarily involving the adrenals. Adrenal teratomas account for 5 % of all paediatric teratomas. Zhou et al. reviewed 7 paediatric and 18 adult cases of primary adrenal teratomas. Of the adults, 72.22 % were female; 44.44 % were incidentalomas, while 50 % experienced pain; 10 tumours were left-sided, the median tumour diameter was 8.25 cm and most underwent Laparoscopic adrenalectomy [16]. A systematic review by Zhong et al. included 12 patients with information on tumour markers, all of whom had normal alpha feto protein and human chorionic gonadotropin levels. No patient has presented with bilateral adrenal teratomas [17].
Most adrenal teratomas, having no specific clinical manifestations, are >6 cm at diagnosis. Laparoscopic adrenalectomy is recommended to relieve symptoms and rule out malignancy pathologically. This is preferred to the open approach due to better visualization of the adrenal gland, decreased likelihood of bleeding, similar efficacy, and shorter hospitalization [12]. The transperitoneal approach is preferred over retroperitoneal for adrenal tumours >5 cm diameter [18]. Sturgeon et al. reported a 10 % risk of malignancy in adrenal tumours 4 cm or greater, and 47 % for tumours 8 cm or greater [19].
Large tumour size, though technically challenging, is not a contraindication to laparoscopic adrenalectomy, but extensive experience in advanced laparoscopic techniques and open adrenal surgery is mandatory to manipulate and excise these tumours [20]. Open adrenalectomy is recommended if one notes local invasion to facilitate lymph node dissection and en bloc resection of adjacent structures. Giordano et al. report 34 patients who underwent laparoscopic adrenalectomy and 16 who underwent open adrenalectomy for giant adrenal tumours. There were no statistically significant differences in postoperative complications, but the laparoscopic approach resulted in less blood loss, and shorter hospital stay and operative time [10]. Similarly, in suspected malignant lesions, recent data shows similar surgical and oncologic outcomes for open and laparoscopic approaches [18].
A biopsy is not necessary once surgical resection is possible and the goal is complete resection to prevent malignant transformation and relieve symptoms. This is the most important prognostic factor and provides the best chance of a cure [2]. They are not responsive to chemotherapy or radiotherapy, and most mature teratomas require no adjuvant therapy, though insufficient data surrounding long-term outcomes mandates close follow-up to detect and treat recurrence or metastasis whether mature or immature [12].
Our patient, despite having a large lesion on CT, had no radiologic features suggestive of malignancy and the tumour was non-functional biochemically. Due to her morbid obesity and restrictive lung disease, open adrenalectomy carried a significant risk of respiratory complications. Therefore, she had laparoscopic adrenalectomy. Due to our confidence of benign pathology based on radiological assessment, we opted for piecemeal extraction to limit the incisions and improve her postoperative course. She was discharged on day 1 post-op and had no complications, but we will continue long-term follow-up.
4. Conclusion
Adrenal teratomas are exceedingly rare typically benign tumours, which require resection, preferably by laparoscopic adrenalectomy. Pre-operative diagnosis, though difficult, is possible and allows operative planning. They should be completely resected since this is curative and necessary to definitively rule out malignancy. Laparoscopic adrenalectomy is a safe option and is associated with oncologic outcomes equivalent to an open approach, coupled with the known advantages of minimally invasive surgery.
Consent
Written informed consent was obtained from the patient for publication of this case report and accompanying images. A copy of the written consent is available for review by the Editor-in-Chief of this journal on request.
Provenance and peer review
Not commissioned, externally peer-reviewed.
Ethical approval
This report is not a research study.
Funding
None.
Guarantor
Dr Ryan Nichol.
Research registration number
-
1.
Name of the registry: N/A
-
2.
Unique identifying number or registration ID: N/A
-
3.
Hyperlink to your specific registration (must be publicly accessible and will be checked): N/A.
CRediT authorship contribution statement
Dr Ryan Nicholas: Conceptualization, Visualization, Writing - original draft, review and editing
Dr Dilip Dan: Conceptualization, Surgeon who performed operations and follow-up, Supervision, Project administration, Writing - Review and editing
Dr Christo Cave: Writing - Review and editing, Visualization
Dr Tanzilah Barrow: Visualization, Radiologist who made the pre-op diagnosis of adrenal teratoma
Dr Melanie Johncilla: Visualization, Pathologist who confirmed diagnosis of adrenal teratoma.
Declaration of competing interest
The authors have no conflicts of interest to declare.
References
- 1.Townsend C.M., Daniel Beauchamp R., Mark Evers B., Mattox Kenneth L. Elsevier Saunders; Philadelphia, PA: 2021. Sabiston Textbook of Surgery: The Biological Basis of Modern Surgical Practice. [Google Scholar]
- 2.el Haddad S., Hessissen L., el Kababri M., Lamalmi N., Kisra M., Allali N., Chat L. Primary mature adrenal teratoma in infant. Pan Afr. Med. J. 2020;37:1–6. doi: 10.11604/pamj.2020.37.27.24016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.He C., Yang Y., Yang Y., Wang F., Hu J., Zhang J., Yin Y., Zeng L., Zhang T., Liu H. Teratoma of the adrenal gland: clinical experience and literature review, gland. Surgery. 2020;9:1056–1064. doi: 10.21037/gs-20-648. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Narla S., Jacob S., Kurian A., Parameswaran A. Primary mature cystic teratoma with carcinoid mimicking an adrenal tumor: report of a rare association and review of literature. Indian J. Pathol. Microbiol. 2016;59:200–202. doi: 10.4103/0377-4929.182012. [DOI] [PubMed] [Google Scholar]
- 5.Ban A., Satapara J., Rathod K., Bahri Nandini. Teratoma involving adrenal gland- a case report and review of the literature. Indian J. Radiol. Imaging. 2019;29(4):452–456. doi: 10.4103/ijri.IJRI_452_18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Kuo E.J., Sisk A.E., Yang Z., Huang J., Yeh M.W., Livhits M.J. Adrenal teratoma: a case series and review of the literature. Endocr. Pathol. 2017;28:152–158. doi: 10.1007/s12022-017-9468-5. [DOI] [PubMed] [Google Scholar]
- 7.Agha R.A., Franchi T., Sohrabi C., Mathew G., Kerwan A., Thoma A., Beamish A.J., Noureldin A., Rao A., Vasudevan B., Challacombe B., Perakath B., Kirshtein B., Ekser B., Pramesh C.S., Laskin D.M., Machado-Aranda D., Miguel D., Pagano D., Millham F.H., Roy G., Kadioglu H., Nixon I.J., Mukhejree I., McCaul J.A., Chi-Yong Ngu J., Albrecht J., Rivas J.G., Raveendran K., Derbyshire L., Ather M.H., Thorat M.A., Valmasoni M., Bashashati M., Chalkoo M., Teo N.Z., Raison N., Muensterer O.J., Bradley P.J., Goel P., Pai P.S., Afifi R.Y., Rosin R.D., Coppola R., Klappenbach R., Wynn R., de Wilde R.L., Surani S., Giordano S., Massarut S., Raja S.G., Basu S., Enam S.A., Manning T.G., Cross T., Karanth V.K., Kasivisvanathan V., Mei Z., The S.C.A.R.E. Guideline: updating consensus surgical CAse REport (SCARE) guidelines. Int. J. Surg. 2020;84(2020):226–230. doi: 10.1016/j.ijsu.2020.10.034. [DOI] [PubMed] [Google Scholar]
- 8.Sinnatamby C.S., Last R.J. Churchill Livingstone; Edinburgh: 2011. Last’s Anatomy: Regional and Applied. [Google Scholar]
- 9.Pinto A., Barletta J.A. Adrenal tumorsinadults. Surg. Pathol. 2015;8:725–749. doi: 10.1016/j.path.2015.07.005. [DOI] [PubMed] [Google Scholar]
- 10.Giordano A., Alemanno G., Bergamini C., Valeri A., Prosperi P. Laparoscopic adrenalectomy for giant adrenal tumours: technical considerations and surgical outcome. J. Minim. Access Surg. 2021;17:76–80. doi: 10.4103/jmas.JMAS_266_19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Niu M., Liu A., Zhao Y., Feng L. Malignant transformation of a mature teratoma of the adrenal gland. Medicine (U. S.) 2017;96 doi: 10.1097/MD.0000000000008333. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Li S., Li H., Ji Z., Yan W., Zhang Y. Primary adrenal teratoma: clinical characteristics and retroperitoneal laparoscopic resection in five adults. Oncol. Lett. 2015;10:2865–2870. doi: 10.3892/ol.2015.3701. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Bhargava P., Sangster G., Haque K., Garrett J., Donato M., D’Agostino H. A multimodality review of adrenal tumors. Curr. Probl. Diagn. Radiol. 2019;48:605–615. doi: 10.1067/j.cpradiol.2018.10.002. [DOI] [PubMed] [Google Scholar]
- 14.Wang F., Liu J., Zhang R., Bai Y., Li C., Li B., Liu H., Zhang T. CT and MRI of adrenal gland pathologies. Quant. Imag. Med. Surg. 2018;8:853–875. doi: 10.21037/qims.2018.09.13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Maenosono R., Saito K., Ibuki N., Takahara K., Inamoto T., Hayahito N., Azuma H. Kyoto University Research Information Repository; 2017. A Case of Retroperitoneal Teratoma Difficult to Disinguish From Adrenal Tumour. [DOI] [PubMed] [Google Scholar]
- 16.Zhou L., Pan X., He T., Lai Y., Li W., Hu Y., Ni L., Yang S., Chen Y., Lai Y. Primary adrenal teratoma: a case series and review of the literature, molecular and clinical. Oncology. 2018:437–442. doi: 10.3892/mco.2018.1687. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Zhong W., Ma R., Cheng S., Tian J., Wang H., Wang T., Zhang Z., Li X., Lu J., Ma L., Zhou L. Clinical characteristics and surgical management of adult adrenal teratoma: a 15-year experience and systematic review of the literature. Urology. 2020;135:71–75. doi: 10.1016/j.urology.2019.05.032. [DOI] [PubMed] [Google Scholar]
- 18.Chuan-Yu S., KX, BG, Y-FH. W-HD. Y-CG. Q-FH. G-WX Laparoscopic adrenalectomy for adrenal tumors. Int J Endocrinol. 2014;2014:1–4. doi: 10.1155/2014/241854. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Sturgeon C., Shen W.T., Clark O.H., Duh Q.Y., Kebebew E. Risk assessment in 457 adrenal cortical carcinomas: how much does tumor size predict the likelihood of malignancy? J. Am. Coll. Surg. 2006;202:423–430. doi: 10.1016/j.jamcollsurg.2005.11.005. [DOI] [PubMed] [Google Scholar]
- 20.Zografos G.N., Farfaras A., Vasiliadis G., Pappa T., Aggeli C., Vassilatou E., Kaltsas G., Piaditis G. Laparoscopic resection of large adrenal tumors. JSLS. 2010;14:364–368. doi: 10.4293/108680810X12924466007160. [DOI] [PMC free article] [PubMed] [Google Scholar]