Abstract
Cystic fibrosis (CF) is a genetic disease caused by mutations in the cystic fibrosis transmembrane conductance regulator (CFTR) protein, a plasma membrane protein expressed on the apical surface of secretory epithelia of the airways. In the airways, defective or absent function of the CFTR protein determines abnormalities of chloride and bicarbonate secretion and, in general, of the transepithelial homeostasis that lead to alterations of airway surface liquid (ASL) composition and properties. The reduction of ASL volume impairs ciliary beating with the consequent accumulation of a sticky mucus. This situation prevents normal mucociliary clearance, favoring the survival and proliferation of bacteria and contributing to the genesis of the CF pulmonary disease. We explored the potential of some CFTR modulators, namely ivacaftor, tezacaftor, elexacaftor and their combination KaftrioTM, capable of partially recovering the basic defects of the CFTR protein, to ameliorate the transepithelial fluid transport and the viscoelastic properties of the mucus when used singly or in combination. Primary human bronchial epithelial cells obtained from CF and non-CF patients were differentiated into a mucociliated epithelia in order to assess the effects of correctors tezacaftor, elexacaftor and their combination with potentiator ivacaftor on the key properties of ASL, such as fluid reabsorption, viscosity, protein content and pH. The treatment of airway epithelia bearing the deletion of a phenylalanine at position 508 (F508del) in the CFTR gene with tezacaftor and elexacaftor significantly improved the pericilial fluid composition, reducing the fluid reabsorption, correcting the ASL pH and reducing the viscosity of the mucus. KaftrioTM was more effective than single modulators in improving all the evaluated parameters, demonstrating once more that this combination recently approved for patients 6 years and older with cystic fibrosis who have at least one F508del mutation in the CFTR gene represents a valuable tool to defeat CF.
Keywords: cystic fibrosis, correctors, bronchial epithelial cell culture, ion transport, pericilial mucus properties
1. Introduction
Mutations in the cystic fibrosis transmembrane conductance regulator (CFTR) gene cause cystic fibrosis (CF), the most common life-threatening monogenic disease among Caucasians [1]. CFTR is an anion channel that transports chloride and bicarbonate across the apical plasma membrane of epithelial cells. CFTR gating is regulated by the binding of ATP to its intracellular binding domains and cAMP-dependent phosphorylation is mandatory for channel activity [2,3]. Its absence or dysfunction impairs the transepithelial flux of ions and fluid in airway cells. The respiratory disorder is the major cause of morbidity and mortality of CF patients. Most of the health problems in CF pulmonary disease are determined by the buildup of a viscous and tenacious mucus primarily induced by the post-secretional dysregulation of fluid and ion transport of the airway surface liquid (ASL) that coats the epithelial cells of the airways. Mucus accumulation constitutes the environment for recurrent infections and chronic inflammation leading to epithelial damage, tissue remodeling and progressive deterioration of lung function, ultimately climaxing in respiratory failure [4].
More than 2000 mutations in the CFTR gene have been described [5], of which at least 352 are disease-causing [6,7]. CFTR mutations have been grouped according to the molecular mechanism they disturb: protein synthesis (class I), maturation (class II), gating (class III), conductance (class IV), plasma membrane abundance (class V) and stability (class VI) [8,9]. Many mutations are pleiotropic, thus, they fit in more than one class [8]. The most prevalent CFTR mutation, the deletion of a phenylalanine at position 508 (F508del) [10,11], is primarily characterised by an incomplete folding (class II) [12,13,14] but also presents gating defects (class III) [15,16,17] and a reduced cell surface permanence (class VI) [12,18,19].
In recent years, a great deal of effort has been devoted to the identification of compounds, particularly potentiators of gating mutations and correctors of processing defects, able to recover the underlying defects of CFTR. This process culminated in the regulatory approval of TrikaftaTM (USA) or KaftrioTM (EU), the combination of correctors tezacaftor and elexacaftor with the potentiator ivacaftor, that has radically changed the health outcomes for many CF patients [20]. Indeed, modulator therapy has been shown to significantly improve sweat chloride, pulmonary function, body weight, and overall quality of life in patients with CF [21]. While the current research has provided unequivocal evidence that treatment with CFTR modulators leads to positive clinical outcomes, we do not yet truly understand their mechanism of action at the molecular level and provide evidence of the link between the pharmacological correction of mutant CFTR and the clinical benefits observed in patients. The elucidation of these aspects could provide useful information to further improve the efficacy of modulator therapy, especially in light of the extension of its use in patients with rare class II CFTR mutations.
ASL thickness depletion and mucus viscoelastic abnormalities have long been postulated to be the underlying causes of lung deterioration, inadequate host defence capability, lung deterioration and increased susceptibility to bacterial infections [22,23]. We have previously demonstrated that the pharmacological correction of F508del CFTR with lumacaftor significantly reduced fluid reabsorption and mucus viscosity in human bronchial epithelial cell (HBEC) monolayers [24]. Recently, a study by Birket and collaborators demonstrated that the treatment with the potentiator ivacaftor positively impacted the transport rate, viscosity and layer depth of the mucus recovered from the lungs of a rat model harbouring the ivacaftor-sensitive G551D (class III) mutation [25]. Similarly, in HBEC from subjects with G551D (class III) or F508del (class II) mutations, the rehydration of the ASL retrieved by treatment with CFTR modulators determined a decrease of mucus concentration, a relaxation of mucin network ultrastructure and a significant improvement of mucus clearance [26].
In this work, we aimed to demonstrate that CFTR modulators that compose the drug KaftrioTM (the potentiator ivacaftor and the correctors tezacaftor and elezacaftor) can recover some physical-chemical and visco-elastic properties of the ASL that lines the surface of the CF airway epithelium.
To achieve this goal, we analysed the effect of KaftrioTM and each of its components on the properties of the ASL recovered from monolayers formed by HBEC obtained from CF subjects [27]. We found that treatment of F508del-CFTR epithelia with the single correctors leads to the fluidification of the mucus, the alkalinisation of the ASL and increased fluid reabsorption, leading to values closer to those measured in non-CF control epithelia. This improvement was found to be greater with KaftrioTM treatment.
2. Results
2.1. Transepitelial Conductance
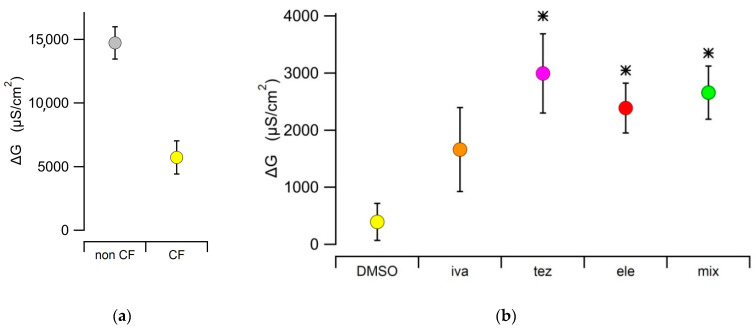
The conductance (G) values obtained in non-CF epithelia (14,732 ± 1267 µS/cm2, n = 10) were significantly higher than those obtained in CF epithelia (5727 ± 1299 µS/cm2, n = 10), consistently with the virtually null expression of CFTR in the apical membrane of mutant cells (Figure 1a). The effect of the different treatments on CF HBEC was assessed in terms of difference in conductance measurement before and after treatment (ΔG). As expected, the DMSO vehicle treatment was not significantly different from zero in either non-CF (ΔG = −338 ± 689 µS/cm2, n = 6) and F508del mutant CF HBEC (ΔG = 396 ± 325 µS/cm2, n = 6). In CF HBEC, treatment with elezacaftor or tezacaftor, or with the triple combination KaftrioTM, significantly increased ΔG to 2387 ± 437 µS/cm2 (n = 9), 2996 ± 692 µS/cm2 (n = 9) and 2558 ± 464 µS/cm2 (n = 9), respectively. The virtual absence of CFTR in the apical membrane of CF epithelium is corroborated by the absence of any effect of the potentiator ivacaftor (ΔG = 1662 ± 737 µS/cm2, n = 9), whose ΔG value was not statistically different from that of the DMSO control (Figure 1b).
Figure 1.
(a) Transepithelial conductance, G, in HBEC epithelial monolayers expressing WT (non-CF) and F508del mutant CFTR (CF). Measurements (n = 10 for both conditions) were obtained after treatment with DMSO as vehicle. (b) Difference between transepithelial conductances measured before and after the different treatments (∆G), measured in epithelia expressing the F508del mutation. (DMSO: 0.1% DMSO (vehicle control), iva: 1 µM of ivacaftor, tez: 5 µM of tezacaftor, ele: 5 µM of elexacaftor, mix: 1 µM of ivacaftor + 5 µM of tezacaftor + 5 µM of elexacaftor). The sample size, n, was comprised between 6 ≤ n ≤ 9. Asterisks indicate that the ∆G value is significantly different from that measured in CF HBEC treated with the DMSO vehicle.
2.2. Transepithelial Fluid Reabsorption and Protein Content of Airway Surface Liquid (ASL)
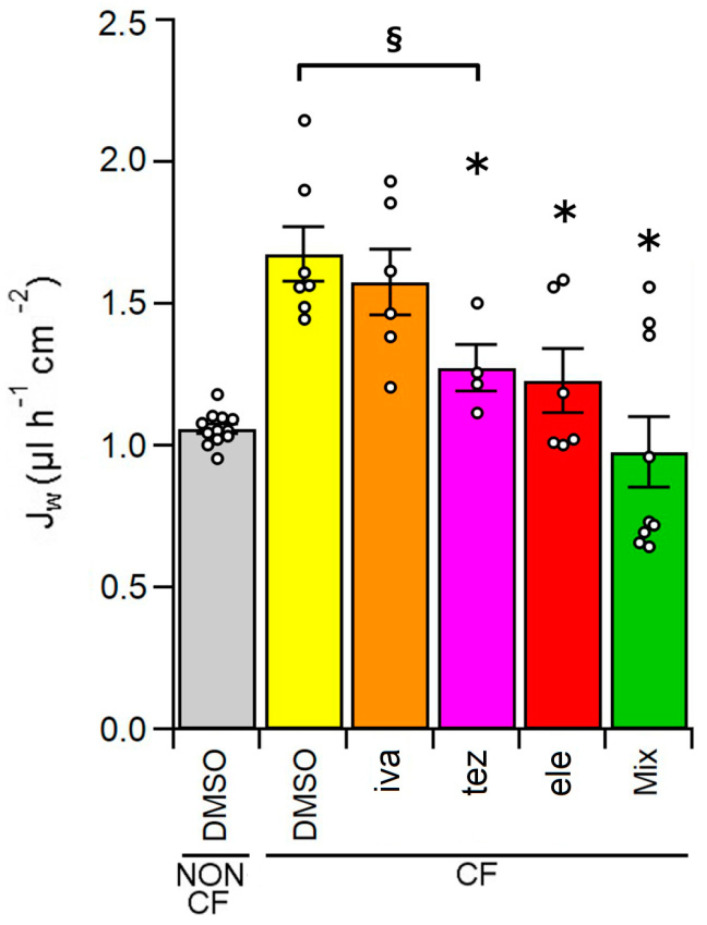
The fluid reabsorption rate of HBEC non-CF epithelia incubated with DMSO for 48 h was 1.06 ± 0.02 µL h−1 cm−2 (n = 12), as shown by Figure 2. Consistently with the presence of a dehydrated ASL [28,29,30], fluid reabsorption of control homozygous F508del HBEC treated with the vehicle DMSO was significantly higher, with an average value of 1.67 ± 0.10 µL h−1 cm−2 (n = 7). Treatment with the potentiator ivacaftor did not significantly modify Jw of CF HBEC (1.58 ± 0.11 µL h−1 cm−2, n = 6), while incubation with correctors tezacaftor, elexacaftor and the combination tezacaftor + elexacaftor + ivacaftor decreased fluid reabsorption in HBEC CF epithelia to 1.27 ± 0.08 (n = 4), 1.23 ± 0.11 (n = 6), and 0.98 ± 0.13 (n = 9) µL h−1 cm−2, respectively (Figure 2).
Figure 2.
Fluid reabsorption, JW, of HBEC monolayers, cultured on Transwell permeable supports, from non-CF cells treated with 0.1% DMSO (vehicle control) and CF HBEC treated with 0.1% DMSO (vehicle control), 1 µM of ivacaftor (iva), 5 µM of tezacaftor (tez), 5 µM of elexacaftor (ele), 1 µM of ivacaftor + 5 µM of tezacaftor + 5 µM of elexacaftor (Mix) for 48 h. Fluid reabsorption of each sample was evaluated gravimetrically as described in the Section 4. The white circles represent the values of each individual measure (6 ≤ n ≤ 12). The section mark (§) indicates data that are statistically different from non-CF control cells, while asterisks (*) indicate a statistical significance versus control CF cells.
The protein concentration in the ASL of non-CF and CF HBEC was not significantly different in all examined conditions as shown by Table 1.
Table 1.
Protein concentration in ASL samples collected from the apical side of non-CF treated with 0.1 % DMSO (vehicle control) and CF HBEC treated with 0.1 % DMSO (vehicle control) or 1 µM of ivacaftor (iva), 5 µM of tezacaftor (tez), 5 µM of elexacaftor (ele) and 1 µM of ivacafotor + 5 µM of tezacaftor + 5 µM of elexacaftor (Mix) for 48 h. Protein concentration (µg/mL) was measured with the Bradford assay. Data are expressed as mean ± sem (standard error of the mean). n indicates the sample size.
| Non-CF | CF | |||||
|---|---|---|---|---|---|---|
| DMSO | DMSO | iva | tez | ele | mix | |
| Mean ± sem (µg/mL) |
1.08 ± 0.03 | 1.18 ± 0.23 | 1.22 ± 0.21 | 1.12 ± 0.09 | 1.09 ± 0.10 | 1.00 ± 0.07 |
| n | 12 | 7 | 7 | 7 | 7 | 7 |
2.3. ASL Rheological Properties
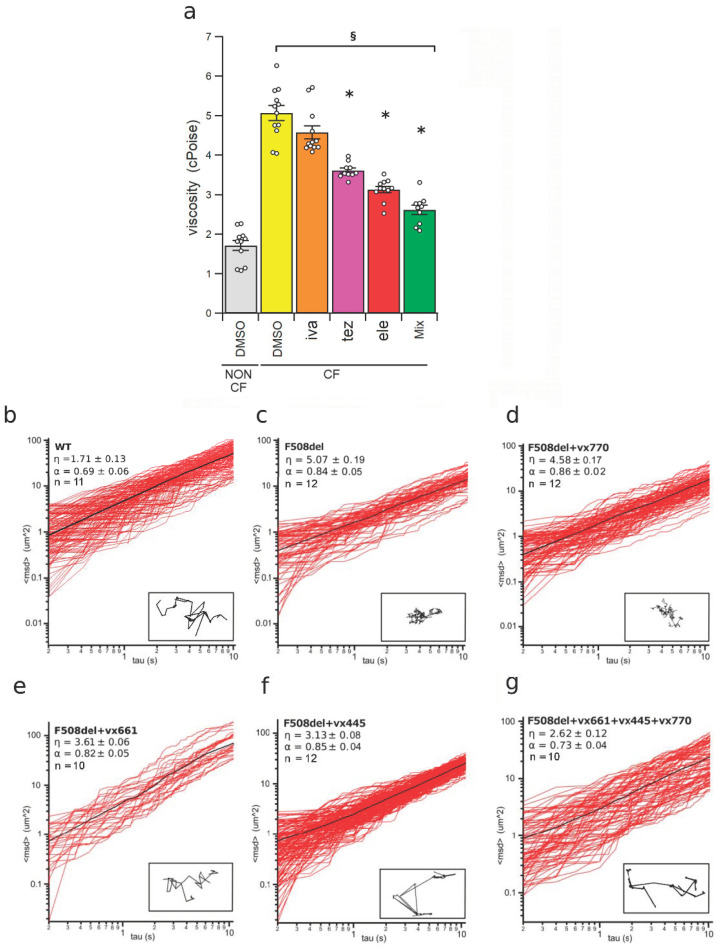
The micro-rheological properties of the fluid recovered from the apical side of HBEC epithelia were analysed using the Multiple Particle Tracking (MPT) [24,31,32,33]. As shown by Figure 3a,c,d, the viscosity of the ASL of non-CF HBEC, calculated from the plot of the mean square displacement against the time interval (1.72 ± 0.13 cPoises, n = 11), was significantly lower than that of CF epithelia (5.07 ± 0.19 cPoises, n = 12).
Figure 3.
Micro-rheology of the airway surface liquid (ASL) recovered from HBEC monolayers measured by the multiple particle technique (MPT). (a) Viscosity of the ASL collected from the apical side of non-CF and CF epithelia. Non-CF HBEC epithelia were incubated with a solution containing 0.1% DMSO (vehicle control) while CF HBEC epithelia were treated with 0.1% DMSO (vehicle control), 1 µM of ivacaftor (iva), 5 µM of tezacaftor (tez), 5 µM of elexacaftor (ele), 1 µM of ivacaftor + 5 µM of tezacaftor + 5 µM of elexacaftor (mix), respectively, administered on the apical side of the monolayers. The white circles in (a) represent the values of each single measure (10 ≤ n ≤ 12). The section mark (§) indicates that data are statistically different from non-CF control cells, while asterisks (*) indicate a statistical significance versus control CF cells. (b–g) Plots of the square displacement, <msd> against the time interval of ≤100 beads in the ASL samples from non-CF HBEC (WT) treated with DMSO (b) and from F508del mutant CF HBEC (F508del) treated with DMSO (c), ivacaftor (VX770) (d), tezacaftor (VX661) (e), elexacaftor (VX445) (f), and ivacaftor + tezacaftor + elexacaftor (VX661 + VX445 + VX770) (g). The average <msd> is shown as a black solid line. The values of viscosity, η, the elastic modulus, α, and the sample size, n, are indicated in each panel. The insets in panels (b–g) show the trajectory path of a bead recorded in each ASL sample.
The viscosity of the ASL of CF HBEC epithelia treated with the potentiator ivacaftor (4.58 ± 0.17, cPoises, n = 12) was not different from that of DMSO-treated CF HBEC (Figure 3a–d). Conversely, treatment with tezacaftor, elexacaftor and tezacaftor + elexacaftor + ivacaftor reduced the viscosity of the ASL of F508del HBEC epithelia to 3.61 ± 0.06 cPoises (n = 10), 3.13 ± 0.08 cPoises (n = 11) and 2.62 ± 0.12 cPoises (n = 10), respectively (Figure 3a,e–g). Analogous results were obtained when comparing the path of a bead in the mucus samples of mutant-CFTR epithelia treated with 1 µM of ivacaftor, 5 µM of tezacaftor, 5 µM of elexacaftor and 1 µM of ivacaftor + 5 µM of tezacaftor + 5 µM of elexacaftor, respectively (insets of Figure 3b–g).
The elastic component of the ASL, expressed by the coefficient α, represents the non-linearity of the mean square displacement of the beads in the mucus samples. The α values of ASL from non-CF HBEC (0.69 ± 0.06, n = 11) was not statistically different from that of DMSO-treated CF HBEC (0.84 ± 0.05, n = 12). Also, treatment with CFTR modulators did not cause any significant change of α, that resulted 0.86 ± 0.02 (n = 12), 0.82 ± 0.05 (n = 10), 0.85 ± 0.05 (n = 12) and 0.73 ± 0.04 (n = 10), in ivacaftor, tezacaftor, elexacaftor, and tezacaftor + elexacaftor + ivacaftor CF HBEC mucus samples, respectively (Figure 3b–g).
2.4. ASL pH
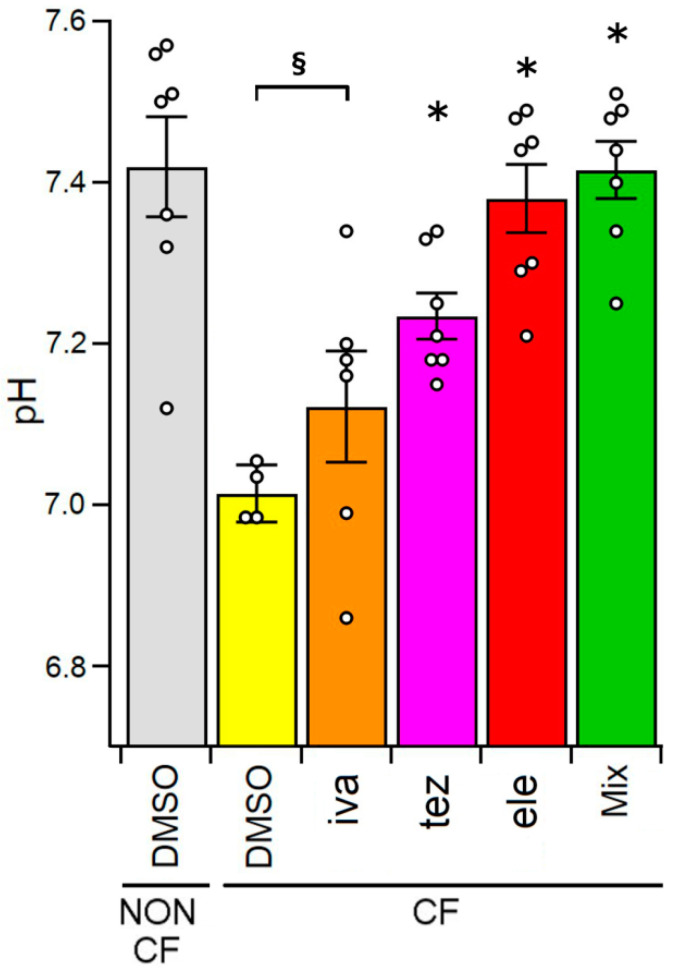
The data presented in Figure 4 show that the pH of the ASL of non-CF HBEC (7.42 ± 0.06, n = 7) was significantly more alkaline than that of CF cells (7.03 ± 0.11, n = 5). In CF HBEC epithelia, the treatment with potentiator ivacaftor did not change the ASL pH (7.12 ± 0.07, n = 6). Conversely, the incubation with CFTR correctors significantly alkalinised the ASL pH (7.23 ± 0.03, n = 7; 7.38 ± 0.04, n = 7 and 7.42 ± 0.04, n = 7 for tezacaftor, elexacaftor and tezacaftor + elexacaftor + ivacaftor, respectively).
Figure 4.
ASL pH measured from non-CF HBEC epithelia treated with 0.1% DMSO (vehicle control) and from CF HBEC epithelia incubated with 0.1% DMSO (vehicle control), 1 µM of ivacaftor (iva), 5 µM of tezacaftor (tez), 5 µM of elexacaftor (ele), and 1 µM of ivacaftor + 5 µM of tezacaftor + 5 µM of elexacaftor (mix), respectively. The white circles represent the values of each single measure. The section mark (§) indicates that data are statistically different from non-CF control cells, while asterisks (*) indicate a statistical significance compared to control CF cells.
3. Discussion
ASL is a thin fluid layer that forms a continuous barrier between the lungs and the external environment, lining the surface of the airway epithelia. ASL mucus performs several important defence functions: it traps particulates and microorganisms; it constitutes a moving medium that can be pushed by the cilia toward the oropharynx; it is a physical barrier that reduces loss of fluid through the airways; and, finally, it acts as a transport medium for secreted substances such as enzymes, defensins, collectins, antiproteases and immunoglobulins [34]. To ensure its homeostasis, the composition of ASL is finely regulated thanks to the transport of ions and water through a variety of ion channels and transporters differentially distributed along the apical and basolateral membranes of epithelial cells. The airway epithelium is basically absorptive. The driving force for transcellular sodium reabsorption is provided by the Na+/K+-ATPase located along the basolateral membrane [35], while the main mediator of apical transport is the epithelial sodium channel (ENaC) found in the apical membrane. Most of the chloride secretion in human airways is provided by CFTR [36] and to a lesser extent by other channels, such as the Ca2+-activated chloride channels (CaCCs), including TMEM16A (ANO1) channels [37,38,39]. The transepithelial transport of water is passive, being generally driven by the osmotic gradient [40]. In CF, the equilibrium of the system is compromised by CFTR mutations that cause the diminution of the CFTR-dependent chloride secretion and the lack of the CFTR-mediated inhibition of ENaC activity [41], ultimately leading to a decrease of the ASL volume. ASL dehydration causes the collapse of the mucociliary clearance and reflects negatively on the innate immune system [41,42,43]. The lack of CFTR function also reduces the secretion of bicarbonate whose release is crucial for mucins post-secretional expansion [35,44,45,46,47,48].
Great advances in the pharmacological treatment of CF have been made since the discovery of CFTR modulators, mainly potentiators and correctors, which can recover the defects underlying the disease. The approval of the triple combination (TrikaftaTM/KaftrioTM) tezacaftor, elexacaftor and ivacaftor for the treatment of CF patients 6 years and older who have at least one F508del mutation represents a milestone in the fight against the disease. The use of this CFTR modulator formulation expands the cohort of patients eligible for treatment to the 90% of the CF population, demonstrating significant improvements in lung function and overall patient quality of life [49,50]. The adequate characterisation of the effect of CFTR modulators paves the way to the possibility to improve their efficacy and to define new formulations in order to expand the population of CF patients who can benefit from the pharmacological treatment of the disease.
This work is aimed to explore the impact of KaftrioTM and its individual components on key properties of the ASL from HBEC: fluid reabsorption, mucus micro-rheology and pH. To achieve this end, we used HBEC epithelia, which are pseudo-stratified, ciliated, produce mucus and express the main transport systems of in vivo airways epithelia [27]. It has been established that HBEC monolayer cultures grown under the ALI condition constitutes an excellent ex vivo model that closely mimics most of the characteristics of human airway epithelium [27,51,52,53].
It has been demonstrated that correctors tezacaftor and elexacaftor and their combination with potentiator ivacaftor significantly increase the CFTR-mediated ion transport in cells expressing F508del CFTR [54]. Our measurements in F508del HBEC preparations of the transepithelial electrical resistance (TEER), expressed as transepithelial conductance (G = 1/TEER, see Figure 1a,b), are consistent with an increase of the anionic transport across the membranes as also shown in isolated cells and epithelium models [55,56]. Hence, our observations on the electrical resistance let us argue that the improvement in the CFTR-mediated ion transport, correlates with corrector’s capability to recover F508del-CFTR protein abnormal maturation, membrane localisation and function, when administrated singly or in combination [56,57].
Our data showed that fluid reabsorption of CF epithelia was almost 1.6 times higher than that of non-CF epithelia, which is consistent with the decrease of the thickness of the ASL layer [29,30,58,59]. Differently from lumacaftor, the first corrector approved for the use of CF patients, that did not influence the fluid reabsorption in HBEC [24], treatment of CF HBEC epithelia with new generation CFTR correctors tezacaftor and elexacaftor and, with the triple combination, TrikaftaTM/KaftrioTM, reduced fluid reabsorption to values closer to those of non-CF-epithelia (Figure 2). The less reabsorption, the greater the ASL volume. This result should therefore positively influence the mucociliary clearance of CF epithelia as well as ASL antibacterial defense capability in vivo. We also noted that treatment with the potentiator ivacaftor did not change fluid reabsorption in CF epithelia. These data highlight once again that the activity of the fraction of F508del protein that manages to reach the plasma membrane [60] is not capable of producing a change in fluid secretion comparable to that of healthy HBEC even after stimulation with ivacaftor.
Next, we focused our attention on the concentration of the proteins, almost mucins, collected from CF and non-CF epithelia. Aware that the measure of the concentration of mucins in the airway epithelium of CF patients is complex and could generate contradictory results [61,62,63,64], we used the Bradford assay to assess the protein content in the ASL samples recovered from the apical side of non-CF and CF monolayers treated with ivacaftor, tezacaftor, elexacaftor or their triple combination KaftrioTM. Contrarily to previous works reporting that the concentration of mucins in the secretions from CF patients was higher than that of healthy patient secretions [65], we found that the protein concentration was similar in all CF and non-CF mucus samples (Table 1). Retrieved results are in agreement with those obtained by Finkbeiner and co-workers, who applied different biochemical and biomolecular techniques to quantify mucins secretion in human airway gland mucous primary cells [66]. This finding allowed us to hypothesise that in our epithelium model, the mucin content is independent from the presence of the CF pathological condition and also by the treatment with CFTR modulators. The impact of the treatment with CFTR modulators on the viscosity of the mucus secreted by HBEC was evaluated by MPT, a technique that permits to analyse the rheological properties of mucus from a relatively small amount of sample (≤8 µL, [24,31,32,33,67]). Similarly to what was observed with lumacaftor, administered alone or in combination with ivacaftor [24], the pharmacological correction by tezacaftor, elexacaftor and almost all by their combination with potentiator ivacaftor significantly improved the viscoelastic properties of the mucus (Figure 3). The triple combination of modulators was confirmed to be more effective than single CFTR modulators in decreasing mucus viscosity of CF cells. In CF epithelia, the potentiator ivacaftor had any effect on mucus viscosity, due to little or no F508del in the uncorrected cell membrane. This further confirms that CFTR channel function, especially bicarbonate secretion [52], plays a central role in regulating ASL mucus homeostasis.
Although the value and the role of ASL pH from CF subjects is still matter of discussion among researchers [68], it is generally accepted that CF airway secretions have a lower pH than the slightly alkaline mucus of normal airway, in partly due to impaired bicarbonate secretion and in partly to inflammation [69,70,71]. While our results could be biased toward slightly more alkaline values caused by the free diffusion of carbonic dioxide from the fluid, we observed significant differences in the pH of the ASL from CF and non-CF primary human airway epithelia, being more acidic than the ASL from homozygous F508del HBEC. Treatment with correctors resulted in a significant increase of the pH of the CF ASL, whose value in the case of administration of triple combination of modulators approached to that of non-CF ASL (Figure 4). The alkalinisation of pH after treatment with correctors could be linked to the increase of bicarbonate permeability of the corrector-rescued CFTR [47,67,72,73] and could translate in vivo into an amelioration of the innate defense of CF epithelia, as already demonstrated in CF-null pigs [74].
In conclusion, we have shown that correctors tezacaftor, elexacaftor and their combination with potentiator ivacaftor (TrikaftaTM/KaftrioTM) are able not only to recover some of the basic defects of F508del CFTR, such as plasma membrane expression (correctors) and transport activity (potentiator), but also to improve the microrheological properties of the mucus, influencing three features that participate in the regulation of ASL mucus homeostasis: hydration, viscosity and pH. Importantly, the combination of the new-generation correctors elexacaftor and tezacaftor with the potentiator ivacaftor increased the microrheological properties of ASL to a greater extent than each compound alone. As already mentioned, these results correlated well with the ability of the correctors to improve the processing, trafficking and function of the mutant CFTR observed in vitro [54,57,75]. In summary, our outcomes suggest that the administration of combinations of CFTR modulators capable to target different CFTR defects is the best strategy to address the CF pulmonary disease [49,76,77].
The effectiveness of the triple-combination elexacaftor, tezacaftor and ivacaftor to clinically improve lung function and respiratory-related quality of life of patients with at least one F508del copy has greatly impacted the life of the 90% of the CF patient population [55,78]. The results of this work provide further evidence of the effectiveness of KaftrioTM. In fact, tezacaftor and elexacaftor, the two correctors that compose it, have been shown to significantly improve some key properties of the ASL that lines airway epithelial cells, such as fluid reabsorption, pH and viscosity. It remains to be determined whether KaftrioTM or other combinations of CFTR modulators will be able to recover the processing defects of other rare CFTR mutations.
4. Materials and Methods
4.1. Chemicals
Culture media and supplements used in this article were those described in Gianotti and co-workers [27]. Ivacaftor (VX770), tezacaftor (VX-661) and elexacaftor (VX-445) were purchased from Selleck Chemicals (Munich, Germany). If not explicitly indicated in the text all other chemicals were provided by Merck (Milan, Italy). CFTR modulators were dissolved in DMSO to have a final concentration of the vehicle in the solutions in contact with the cells ≤ 0.1%.
4.2. Human Bronchial Epithelial Cell Culture
F508del/F508del and non-CF HBEC, from two non-CF and CF donors, were provided by the “Primary Cultures Service” of the Italian Cystic Fibrosis Research Foundation, isolated by following the procedures described elsewhere [27]. For use in these experiments, HBEC cells were thawed and plate cultured in a 1:1 mixture of LHC-9 and RPMI 1640 (LHC9-RPMI) media for two additional passages. To obtain differentiated epithelia, HBEC were seeded at high density on Transwell permeable supports (Corning, code 3379, New York, NY, USA) with bilateral addition of serum-free LHC9-RPMI medium. After 48 h the medium was replaced only on the basolateral side (air-liquid-interface condition, ALI) with DMEM/F12 (1:1) plus 2% New Zealand fetal bovine serum (Life Technologies, Monza, Italy), hormones and supplements. The cells were maintained in ALI condition for 4–5 weeks before performing the experiments in order to promote further differentiation of the epithelium. The complete maturation of epithelia was checked by measuring the transepithelial electrical resistance (TEER) (see below). Seven- to 8-day-old epithelia with TEER ≥ 1 KΩ∙cm2 and a potential difference ≈ −20 mV were considered as completely differentiated [27]. To achieve the highest effect on the parameters that we intended to assess, monolayers were treated for 48 h [79,80,81,82,83,84] with 80 µL of a solution containing (in mM): 150 NaCl, 5 KCl, 1.2 CaCl2, 0.5 MgCl2 and 0.1 HEPES (pH 7.4) and 1 µM of ivacaftor, 5 µM of tezacaftor, 5 µM of elexacaftor, and 1 µM of ivacaftor + 5 µM of tezacaftor + 5 µM of elexacaftor or 0.1% DMSO as control vehicle, administered on the apical side of the monolayers.
4.3. Transepithelial Electrical Measurements
Transepithelial electrical resistance (TEER) was measured with 4 mm Ag|AgCl chopstick electrodes with an epithelial voltohmmeter EVOM2 (World Precision Instruments, Sarasota, FL, USA). To monitor the electrical properties of the treated epithelia, we measured the TEER immediately before the beginning and at the end (48 h) of the pharmacological treatment. The measurements were expressed in terms of conductance (G = 1/TEER; µS/cm2).
4.4. Measurement of Fluid Reabsorption and Protein Content
At the end of incubation with CFTR modulators, the fluid lining the apical surface of each epithelium was carefully aspirated and collected. The volume of each sample was evaluated gravimetrically. The net flux of fluid across the epithelium, JW, was calculated as:
| Jw = (Vi − Vf)/(A × t) | (1) |
where Vi and Vf are the initial and final apical fluid volumes, A is the area of the epithelium (for Transwell support: 0.33 cm2) and t is the time interval between the addition of Vi and recovering of the remaining fluid Vf. The protein content of each sample was determined by using the Bradford assay. If not tested immediately, samples were frozen and stored at −80 °C up to three weeks.
4.5. Micro-Rheology
The micro-rheological properties of the fluid recovered from the apical side of epithelia was evaluated using the multiple particle tracking (MPT) method [24,27,30,31,32,33]. The method has been controlled by measuring the viscosity of glycerol solutions with known viscosity (see Figure S1 and Table S1). In MPT, the time course of the position of the beads in suspension inside the medium to be studied is recorded. A 25-µL aliquot of mucus and 1 µL of solution containing 200 nm diameter yellow/green fluorescent polystyrene carboxylated beads (λexc = 488 nm, λem = 505–515 nm; Life Technologies) were mixed, and a sample of 8 µL of mucus containing ≤100 beads/field was deposited between two glass cover slips, and the borders were sealed to avoid evaporation. After an equilibration at room temperature for 20 min, beads were focused on the mid-height of the sample to exclude beads that might be interacting with the cover slips with a 60× (N.A. = 1.42) objective connected to a CCD video camera. Images of 1280 × 960 pixels were captured at a rate of 5 frames/s. The trajectory of the Brownian motion of the beads was recorded using the Multitracker plug-in of ImageJ [85]. About 400 beads were tracked in four to eight fields per sample. The movement of the fluorescent beads within the mucus in a given time interval, τ, is described by its mean squared displacement, <msd>, from which it is possible to calculate the diffusion coefficient D0, according to the equation:
| <msd(τ)> = 4D0 τα | (2) |
where α is the elastic contribution of the fluid whose value is 0 < α ≤ 1. The viscosity, η, is calculated from the Stokes–Einstein equation, as:
| η = kB T/6π D0 r | (3) |
where kB is the Boltzmann constant, T is the absolute temperature and r is the radius of the microspheres.
4.6. pH Measurement
After 48 h of incubation with CFTR modulators, mucus samples (≥40 µL) were quickly collected from the apical surface of each epithelium and their pH was immediately measured (within 2 min) using a microelectrode (SevenCompact-Mettler Toledo, Novate Milanese, Italy). The measurements were repeated at least six-fold.
4.7. Statistics
Analysis of variance was applied to verify that data obtained from the two non-CF and CF donors had the same fluctuation. Once verified that there was no difference among intra- and inter-group replicates, the replicates under each condition (5 to 12) were pooled as a single population. Data are shown as mean ± standard error of the mean (sem). All statistical and MPT analysis was done with IgorPro 9 software (Lake Oswego, OR, USA). The Kruskal–Wallis non-parametric analysis of variance followed by Dunn’s post hoc test were used to compare data sets. A value of p < 0.05 was considered statistically significant.
Acknowledgments
We thank the “Servizio Colture Primarie” of the Italian Cystic Fibrosis Research Foundation for providing CF primary cells.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/ijms231911396/s1.
Author Contributions
D.B. and O.M. designed the study. D.B. and A.L. conducted the experiments and analysed the results. D.B. and O.M. interpreted the results and wrote the manuscript. All authors have read and agreed to the published version of the manuscript.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
The datasets used and/or analysed during the current study are available from the corresponding author on reasonable request.
Conflicts of Interest
The authors declare no conflict of interest.
Funding Statement
This work was partially supported by Regione Liguria with grant Programma Operativo Regione Liguria Fondo Sociale Europeo 2014–2020 (DB).
Footnotes
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Bobadilla J.L., Macek M., Jr., Fine J.P., Farrell P.M. Cystic fibrosis: A worldwide analysis of CFTR mutations—Correlation with incidence data and application to screening. Hum. Mutat. 2002;19:575–606. doi: 10.1002/humu.10041. [DOI] [PubMed] [Google Scholar]
- 2.Riordan J.R. Assembly of functional CFTR chloride channels. Annu. Rev. Physiol. 2005;67:701–718. doi: 10.1146/annurev.physiol.67.032003.154107. [DOI] [PubMed] [Google Scholar]
- 3.Saint-Criq V., Gray M.A. Role of CFTR in epithelial physiology. Cell. Mol. Life Sci. CMLS. 2017;74:93–115. doi: 10.1007/s00018-016-2391-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Ratjen F., Bell S.C., Rowe S.M., Goss C.H., Quittner A.L., Bush A. Cystic fibrosis. Nat. Rev. Dis. Prim. 2015;1:15010. doi: 10.1038/nrdp.2015.10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. [(accessed on 10 June 2022)]. Available online: www.genet.sickkids.on.ca.
- 6.Castellani C. CFTR2: How will it help care? Paediatr. Respir. Rev. 2013;14:2–5. doi: 10.1016/j.prrv.2013.01.006. [DOI] [PubMed] [Google Scholar]
- 7.De Boeck K., Zolin A., Cuppens H., Olesen H.V., Viviani L. The relative frequency of CFTR mutation classes in European patients with cystic fibrosis. J. Cyst. Fibros. 2014;13:403–409. doi: 10.1016/j.jcf.2013.12.003. [DOI] [PubMed] [Google Scholar]
- 8.Veit G., Avramescu R.G., Chiang A.N., Houck S.A., Cai Z., Peters K.W., Hong J.S., Pollard H.B., Guggino W.B., Balch W.E., et al. From CFTR biology toward combinatorial pharmacotherapy: Expanded classification of cystic fibrosis mutations. Mol. Biol. Cell. 2016;27:424–433. doi: 10.1091/mbc.e14-04-0935. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.De Boeck K., Amaral M.D. Progress in therapies for cystic fibrosis. Lancet Respir. Med. 2016;4:662–674. doi: 10.1016/S2213-2600(16)00023-0. [DOI] [PubMed] [Google Scholar]
- 10.Kerem B., Rommens J.M., Buchanan J.A., Markiewicz D., Cox T.K., Chakravarti A., Buchwald M., Tsui L.C. Identification of the cystic fibrosis gene: Genetic analysis. Science. 1989;245:1073–1080. doi: 10.1126/science.2570460. [DOI] [PubMed] [Google Scholar]
- 11.Riordan J.R., Rommens J.M., Kerem B., Alon N., Rozmahel R., Grzelczak Z., Zielenski J., Lok S., Plavsic N., Chou J.L., et al. Identification of the cystic fibrosis gene: Cloning and characterization of complementary DNA. Science. 1989;245:1066–1073. doi: 10.1126/science.2475911. [DOI] [PubMed] [Google Scholar]
- 12.Cheng S.H., Gregory R.J., Marshall J., Paul S., Souza D.W., White G.A., O’Riordan C.R., Smith A.E. Defective intracellular transport and processing of CFTR is the molecular basis of most cystic fibrosis. Cell. 1990;63:827–834. doi: 10.1016/0092-8674(90)90148-8. [DOI] [PubMed] [Google Scholar]
- 13.Jensen T.J., Loo M.A., Pind S., Williams D.B., Goldberg A.L., Riordan J.R. Multiple proteolytic systems, including the proteasome, contribute to CFTR processing. Cell. 1995;83:129–135. doi: 10.1016/0092-8674(95)90241-4. [DOI] [PubMed] [Google Scholar]
- 14.Lukacs G.L., Verkman A.S. CFTR: Folding, misfolding and correcting the ΔF508 conformational defect. Trends Mol. Med. 2012;18:81–91. doi: 10.1016/j.molmed.2011.10.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Dalemans W., Barbry P., Champigny G., Jallat S., Dott K., Dreyer D., Crystal R.G., Pavirani A., Lecocq J.P., Lazdunski M. Altered chloride ion channel kinetics associated with the delta F508 cystic fibrosis mutation. Nature. 1991;354:526–528. doi: 10.1038/354526a0. [DOI] [PubMed] [Google Scholar]
- 16.Serohijos A.W., Hegedus T., Aleksandrov A.A., He L., Cui L., Dokholyan N.V., Riordan J.R. Phenylalanine-508 mediates a cytoplasmic-membrane domain contact in the CFTR 3D structure crucial to assembly and channel function. Proc. Natl. Acad. Sci. USA. 2008;105:3256–3261. doi: 10.1073/pnas.0800254105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Mendoza J.L., Schmidt A., Li Q., Nuvaga E., Barrett T., Bridges R.J., Feranchak A.P., Brautigam C.A., Thomas P.J. Requirements for efficient correction of ΔF508 CFTR revealed by analyses of evolved sequences. Cell. 2012;148:164–174. doi: 10.1016/j.cell.2011.11.023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Okiyoneda T., Barrière H., Bagdány M., Rabeh W.M., Du K., Höhfeld J., Young J.C., Lukacs G.L. Peripheral protein quality control removes unfolded CFTR from the plasma membrane. Science. 2010;329:805–810. doi: 10.1126/science.1191542. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Swiatecka-Urban A., Brown A., Moreau-Marquis S., Renuka J., Coutermarsh B., Barnaby R., Karlson K.H., Flotte T.R., Fukuda M., Langford G.M., et al. The short apical membrane half-life of rescued ΔF508-cystic fibrosis transmembrane conductance regulator (CFTR) results from accelerated endocytosis of ΔF508-CFTR in polarized human airway epithelial cells. J. Biol. Chem. 2005;280:36762–36772. doi: 10.1074/jbc.M508944200. [DOI] [PubMed] [Google Scholar]
- 20.Barry P.J., Mall M.A., Álvarez A., Colombo C., de Winter-de Groot K.M., Fajac I., McBennett K.A., McKone E.F., Ramsey B.W., Sutharsan S., et al. Triple Therapy for Cystic Fibrosis Phe508del-Gating and Residual Function Genotypes. N. Engl. J. Med. 2021;385:815–825. doi: 10.1056/NEJMoa2100665. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Shteinberg M., Taylor-Cousar J.L. Impact of CFTR modulator use on outcomes in people with severe cystic fibrosis lung disease. Eur. Respir. Rev. 2020;29:190112. doi: 10.1183/16000617.0112-2019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Pezzulo A.A., Tang X.X., Hoegger M.J., Abou Alaiwa M.H., Ramachandran S., Moninger T.O., Karp P.H., Wohlford-Lenane C.L., Haagsman H.P., van Eijk M., et al. Reduced airway surface pH impairs bacterial killing in the porcine cystic fibrosis lung. Nature. 2012;487:109–113. doi: 10.1038/nature11130. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Matsui H., Wagner V.E., Hill D.B., Schwab U.E., Rogers T.D., Button B. A physical linkage between cystic fibrosis airway surface dehydration and Pseudomonas aeruginosa biofilms. Proc. Natl. Acad. Sci. USA. 2006;103:18131–18136. doi: 10.1073/pnas.0606428103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Gianotti A., Capurro V., Scudieri P., Galietta L.J., Moran O., Zegarra-Moran O. Pharmacological rescue of mutant CFTR protein improves the viscoelastic properties of CF mucus. J. Cyst. Fibros. 2016;15:295–301. doi: 10.1016/j.jcf.2015.11.003. [DOI] [PubMed] [Google Scholar]
- 25.Birket S.E., Davis J.M., Fernandez-Petty C.M., Henderson A.G., Oden A.M., Tang L., Wen H., Hong J., Fu L., Chambers A., et al. Ivacaftor Reverses Airway Mucus Abnormalities in a Rat Model Harboring a Humanized G551D-CFTR. Am. J. Respir. Crit. Care Med. 2020;202:1271–1282. doi: 10.1164/rccm.202002-0369OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Morrison C.B., Shaffer K.M., Araba K.C., Markovetz M.R., Wykoff J.A., Quinney N.L., Hao S., Delion M.F., Flen A.L., Morton L.C., et al. Treatment of cystic fibrosis airway cells with CFTR modulators reverses aberrant mucus properties via hydration. Eur. Respir. J. 2022;59:2100185. doi: 10.1183/13993003.00185-2021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Gianotti A., Delpiano L., Caci E. In vitro methods for the development and analysis of human primary airway epithelia. Front. Pharmacol. 2018;9:1176–1188. doi: 10.3389/fphar.2018.01176. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Tarran R., Trout L., Donaldson S.H., Boucher R.C. Soluble mediators, not cilia, determine airway surface liquid volume in normal and cystic fibrosis superficial airway epithelia. J. Gen. Physiol. 2006;127:591–604. doi: 10.1085/jgp.200509468. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Harvey P.R., Tarran R., Garoff S., Myerburg M.M. Measurement of the airway surface liquid volume with simple light refraction microscopy. Am. J. Respir. Cell Mol. Biol. 2011;45:592–599. doi: 10.1165/rcmb.2010-0484OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Gianotti A., Melani R., Caci E., Sondo E., Ravazzolo R., Galietta L.J.V., Zegarra-Moran O. Epithelial sodium channel silencing as a strategy to correct the airway surface fluid deficit in cystic fibrosis. Am. J. Respir. Cell. Mol. Biol. 2013;49:445–452. doi: 10.1165/rcmb.2012-0408OC. [DOI] [PubMed] [Google Scholar]
- 31.Qian H., Sheetz M.P., Elson E.L. Single particle tracking. Analysis of diffusion and flow in two-dimensional systems. Biophys. J. 1991;60:910–921. doi: 10.1016/S0006-3495(91)82125-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Wirtz D. Particle-tracking microrheology of living cells: Principles and applications. Annu. Rev. Biophys. 2009;38:301–326. doi: 10.1146/annurev.biophys.050708.133724. [DOI] [PubMed] [Google Scholar]
- 33.Gianotti A., Capurro V., Delpiano L., Mielczarek M., García-Valverde M., Carreira-Barral I., Ludovico A., Fiore M., Baroni D., Moran O., et al. Small Molecule Anion Carriers Correct Abnormal Airway Surface Liquid Properties in Cystic Fibrosis Airway Epithelia. Int. J. Mol Sci. 2020;21:1488. doi: 10.3390/ijms21041488. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Lillehoj E.R., Kim K.C. Airway mucus: Its components and function. Arch. Pharmacal Res. 2002;25:770–780. doi: 10.1007/BF02976990. [DOI] [PubMed] [Google Scholar]
- 35.Widdicombe J.H. Regulation of the depth and composition of airway surface liquid. J. Anat. 2002;201:313–318. doi: 10.1046/j.1469-7580.2002.00098.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Collawn J.F., Matalon S. CFTR and lung homeostasis. Am. J. Physiol. Lung Cell. Mol. Physiol. 2014;307:L917–L923. doi: 10.1152/ajplung.00326.2014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Gallos G., Remy K.E., Danielsson J., Funayama H., Fu X.W., Chang H.Y., Yim P., Xu D., Emala C.W., Sr. Functional expression of the TMEM16 family of calcium-activated chloride channels in airway smooth muscle. Am. J. Physiol. Lung Cell. Mol. Physiol. 2013;305:L625–L634. doi: 10.1152/ajplung.00068.2013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Rock J.R., O’Neal W.K., Gabriel S.E., Randell S.H., Harfe B.D., Boucher R.C., Grubb B.R. Transmembrane protein 16A (TMEM16A) is a Ca2+-regulated Cl− secretory channel in mouse airways. J. Biol. Chem. 2009;284:14875–14880. doi: 10.1074/jbc.C109.000869. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Schiffhauer E.S., Vij N., Kovbasnjuk O., Kang P.W., Walker D., Lee S., Zeitlin P.L. Dual activation of CFTR and CLCN2 by lubiprostone in murine nasal epithelia. Am. J. Physiol. Lung Cell. Mol. Physiol. 2013;304:L324–L331. doi: 10.1152/ajplung.00277.2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Folkesson H.G., Matthay M.A., Frigeri A., Verkman A.S. Transepithelial water permeability in microperfused distal airways. Evidence for channel-mediated water transport. J. Clin. Investig. 1996;97:664–671. doi: 10.1172/JCI118463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Boucher R.C. Cystic fibrosis: A disease of vulnerability to airway surface dehydration. Trends Mol. Med. 2007;13:231–240. doi: 10.1016/j.molmed.2007.05.001. [DOI] [PubMed] [Google Scholar]
- 42.Krouse M.E. Is cystic fibrosis lung disease caused by abnormal ion composition or abnormal volume? J. Gen. Physiol. 2001;118:219–222. doi: 10.1085/jgp.118.2.219. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Cantin A.M., Hartl D., Konstan M.W., Chmiel J.F. Inflammation in cystic fibrosis lung disease: Pathogenesis and therapy. J. Cyst. Fibros. 2015;14:419–430. doi: 10.1016/j.jcf.2015.03.003. [DOI] [PubMed] [Google Scholar]
- 44.Matsui H., Randell S.H., Peretti S.W., Davis C.W., Boucher R.C. Coordinated clearance of periciliary liquid and mucus from airway surfaces. J. Clin. Investig. 1998;102:1125–1131. doi: 10.1172/JCI2687. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Song Y., Thiagarajah J., Verkman A.S. Sodium and Chloride Concentrations, pH, and Depth of Airway Surface Liquid in Distal Airways. J. Gen. Physiol. 2003;122:511–519. doi: 10.1085/jgp.200308866. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Rose M.C., Voynow J.A. Respiratory Tract Mucin Genes and Mucin Glycoproteins in Health and Disease. Physiol. Rev. 2006;86:245–278. doi: 10.1152/physrev.00010.2005. [DOI] [PubMed] [Google Scholar]
- 47.Gustafsson J.K., Ermund A., Ambort D., Johansson M.E.V., Nilsson H.E., Thorell K., Hebert H., Sjövall H., Hansson G.C. Bicarbonate and functional CFTR channel are required for proper mucin secretion and link cystic fibrosis with its mucus phenotype. J. Exp. Med. 2012;209:1263–1272. doi: 10.1084/jem.20120562. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Yang N., Garcia M.A.S., Quinton P.M. Normal mucus formation requires cAMP-dependent HCO3− secretion and Ca2+-mediated mucin exocytosis. J. Physiol. 2013;591:4581–4593. doi: 10.1113/jphysiol.2013.257436. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Middleton P.G., Mall M.A., Dřevínek P., Lands L.C., McKone E.F., Polineni D., Ramsey B.W., Taylor-Cousar J.L., Tullis E., Vermeulen F., et al. Elexacaftor–tezacaftor–ivacaftor for cystic fibrosis with a single Phe508del allele. N. Engl. J. Med. 2019;381:1809–1819. doi: 10.1056/NEJMoa1908639. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Heijerman H.G.M., McKone E.F., Downey D.G. Efficacy and safety of the elexacaftor plus tezacaftor plus ivacaftor combination regimen in people with cystic fibrosis homozygous for the F508del mutation: A double-blind, randomised, phase 3 trial. Lancet. 2019;394:1940–1948. doi: 10.1016/S0140-6736(19)32597-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Fulcher M.L., Gabriel S., Burns K.A., Yankaskas J.R., Randell S.H. Well-differentiated human airway epithelial cell cultures. Methods Mol. Med. 2005;107:183–206. doi: 10.1385/1-59259-861-7:183. [DOI] [PubMed] [Google Scholar]
- 52.Galietta L.J., Lantero S., Gazzolo A., Sacco O., Romano L., Rossi G.A. An improved method to obtain highly differentiated monolayers of human bronchial epithelial cells. In Vitro Cell. Dev. Biol. Anim. 1998;34:478–481. doi: 10.1007/s11626-998-0081-2. [DOI] [PubMed] [Google Scholar]
- 53.Neuberger T., Burton T., Clark H., Van Goor F. Use of primary cultures of human bronchial epithelial cells isolated from cystic fibrosis patients for the pre-clinical testing of CFTR modulators. Methods Mol. Biol. 2011;741:39–54. doi: 10.1007/978-1-61779-117-8_4. [DOI] [PubMed] [Google Scholar]
- 54.Veit G., Roldan A., Hancock M.A., Da Fonte D.F., Xu H., Hussein M., Frenkiel S., Matouk E., Velkov T., Lukacs G.L. Allosteric folding correction of F508del and rare CFTR mutants by elexacaftor-tezacaftor-ivacaftor (Trikafta) combination. JCI Insight. 2020;5:e139983. doi: 10.1172/jci.insight.139983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Pranke I.M., Hatton A., Simonin J., Jais J.P., Le Pimpec-Barthes F., Carsin A., Bonnette P., Fayon M., Stremler-Le Bel N., Grenet D., et al. Correction of CFTR function in nasal epithelial cells from cystic fibrosis patients predicts improvement of respiratory function by CFTR modulators. Sci. Rep. 2017;7:7375. doi: 10.1038/s41598-017-07504-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Amico G., Brandas C., Moran O., Baroni D. Unravelling the Regions of Mutant F508del-CFTR More Susceptible to the Action of Four Cystic Fibrosis Correctors. Int. J. Mol. Sci. 2019;20:5463. doi: 10.3390/ijms20215463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Becq F., Mirval S., Carrez T., Lévêque M., Billet A., Coraux C., Sage E., Cantereau A. The rescue of F508del-CFTR by elexacaftor/tezacaftor/ivacaftor (Trikafta) in human airway epithelial cells is underestimated due to the presence of ivacaftor. Eur. Respir. J. 2022;59:2100671. doi: 10.1183/13993003.00671-2021. [DOI] [PubMed] [Google Scholar]
- 58.Choi H.C., Kim C.S.K., Tarran R. Automated acquisition and analysis of airway surface liquid height by confocal microscopy. Am. J. Physiol. Cell. Mol. Physiol. 2015;309:109–118. doi: 10.1152/ajplung.00027.2015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Olivença D.V., Fonseca L.L., Voit E.O., Pinto F.R. Thickness of the airway surface liquid layer in the lung is affected in cystic fibrosis by compromised synergistic regulation of the ENaC ion channel. J. R. Soc. Interface. 2019;16:20190187. doi: 10.1098/rsif.2019.0187. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Zhang L., Button B., Gabriel S.E., Burkett S., Yan Y., Skiadopoulos M.H., Dang Y.L., Vogel L.N., McKay T., Mengos A., et al. CFTR delivery to 25% of surface epithelial cells restores normal rates of mucus transport to human cystic fibrosis airway epithelium. PLoS Biol. 2009;7:e1000155. doi: 10.1371/journal.pbio.1000155. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Hoegger M.J., Fischer A.J., McMenimen J.D., Ostedgaard L.S., Tucker A.J., Awadalla M.A., Moninger T.O., Michalski A.S., Hoffman E.A., Zabner J., et al. Impaired mucus detachment disrupts mucociliary transport in a piglet model of cystic fibrosis. Science. 2014;345:818–822. doi: 10.1126/science.1255825. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Niv Y., Ho S.B., Rokkas T. Mucin Secretion in Cystic Fibrosis: A Systematic Review. Dig. Dis. 2021;39:375–381. doi: 10.1159/000512268. [DOI] [PubMed] [Google Scholar]
- 63.Burgel P.R., Montani D., Danel C., Dusser D.J., Nadel J.A. A morphometric study of mucins and small airway plugging in cystic fibrosis. Thorax. 2007;62:153–161. doi: 10.1136/thx.2006.062190. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Henke M.O., Renner A., Huber R.M. MUC5AC and MUC5B mucins are decreased in cystic fibrosis airway secretions. Am. J. Respir. Cell Mol. Biol. 2004;31:86–91. doi: 10.1165/rcmb.2003-0345OC. [DOI] [PubMed] [Google Scholar]
- 65.Henderson A.G., Ehre C., Button B., Abdullah L.H., Cai L.H., Leigh M.W., DeMaria G.C., Matsui H., Donaldson S.H., Davis C.W., et al. Cystic fibrosis airway secretions exhibit mucin hyperconcentration and increased osmotic pressure. J. Clin. Investig. 2014;124:3047–3060. doi: 10.1172/JCI73469. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Finkbeiner W.E., Zlock L.T., Morikawa M., Lao A.Y., Dasari V., Widdicombe J.H. Cystic fibrosis and the relationship between mucin and chloride secretion by cultures of human airway gland mucous cells. Am. J. Physiol. Lung Cell. Mol. Physiol. 2011;301:L402–L414. doi: 10.1152/ajplung.00210.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Ferrera L., Capurro V., Delpiano L., Gianotti A., Moran O. The Application of Bicarbonate Recovers the Chemical-Physical Properties of Airway Surface Liquid in Cystic Fibrosis Epithelia Models. Biology. 2021;10:278. doi: 10.3390/biology10040278. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Marquette C.R., Luckie D.B. Dissection of a Mechanistic Controversy in Cystic Fibrosis. JSM Genet. Genom. 2016;3:1017. [Google Scholar]
- 69.Tang X.X., Ostedgaard L.S., Hoegger M.J., Moninger T.O., Karp P.H., McMenimen J.D., Choudhury B., Varki A., Stoltz D.A., Welsh M.J. Acidic pH increases airway surface liquid viscosity in cystic fibrosis. J. Clin. Investig. 2016;126:879–891. doi: 10.1172/JCI83922. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Shah V.S., Meyerholz D.K., Tang X.X., Reznikov L., Abou Alaiwa M., Ernst S.E., Karp P.H., Wohlford-Lenane C.L., Heilmann K.P., Leidinger M.R., et al. Airway acidification initiates host defense abnormalities in cystic fibrosis mice. Science. 2016;351:503–507. doi: 10.1126/science.aad5589. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Ferrera L., Baroni D., Moran O. Lumacaftor-rescued F508del-CFTR has a modified bicarbonate permeability. J. Cyst. Fibros. 2019;18:602–605. doi: 10.1016/j.jcf.2019.01.012. [DOI] [PubMed] [Google Scholar]
- 72.Fiore M., Picco C., Moran O. Correctors modify the bicarbonate permeability of F508del-CFTR. Sci. Rep. 2020;10:8440. doi: 10.1038/s41598-020-65287-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Shah V.S., Ernst S., Tang X.X., Karp P.H., Parker C.P., Ostedgaard L.S., Welsh M.J. Relationships among CFTR expression, HCO3- secretion, and host defense may inform gene- and cell-based cystic fibrosis therapies. Proc. Natl. Acad. Sci. USA. 2016;113:5382–5387. doi: 10.1073/pnas.1604905113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Davies J.C., Moskowitz S.M., Brown C., Horsley A., Mall M.A., McKone E.F., Plant B.J., Prais D., Ramsey B.W., Taylor-Cousar J.L., et al. VX-659-Tezacaftor-Ivacaftor in Patients with Cystic Fibrosis and One or Two Phe508del Alleles. N. Engl. J. Med. 2018;379:1599–1611. doi: 10.1056/NEJMoa1807119. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Lopes-Pacheco M. CFTR Modulators: The Changing Face of Cystic Fibrosis in the Era of Precision Medicine. Front. Pharmacol. 2020;10:1662. doi: 10.3389/fphar.2019.01662. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Farinha C.M., King-Underwood J., Sousa M., Correia A.R., Henriques B.J., Roxo-Rosa M., Da Paula A.C., Williams J., Hirst S., Gomes C.M., et al. Revertants, low temperature, and correctors reveal the mechanism of F508del-CFTR rescue by VX-809 and suggest multiple agents for full correction. Chem. Biol. 2013;20:943–955. doi: 10.1016/j.chembiol.2013.06.004. [DOI] [PubMed] [Google Scholar]
- 77.Okiyoneda T., Veit G., Dekkers J.F., Bagdany M., Soya N., Xu H., Roldan A., Verkman A.S., Kurth M., Simon A., et al. Mechanism-based corrector combination restores ΔF508-CFTR folding and function. Nat. Chem. Biol. 2013;9:444–454. doi: 10.1038/nchembio.1253. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Sutharsan S., McKone E.F., Downey D.G., Duckers J., MacGregor G., Tullis E., Van Braeckel E., Wainwright C.E., Watson D., Ahluwalia N., et al. Efficacy and safety of elexacaftor plus tezacaftor plus ivacaftor versus tezacaftor plus ivacaftor in people with cystic fibrosis homozygous for F508del-CFTR: A 24-week, multicentre, randomised, double-blind, active-controlled, phase 3b trial. Lancet Respir. Med. 2022;10:267–277. doi: 10.1016/S2213-2600(21)00454-9. [DOI] [PubMed] [Google Scholar]
- 79.Cholon D.M., Quinney N.L., Fulcher M.L., Esther C.R., Das J., Jr., Dokholyan N.V., Randell S.H., Boucher R.C., Gentzsch M. Potentiator ivacaftor abrogates pharmacological correction of ΔF508 CFTR in cystic fibrosis. Sci. Transl. Med. 2014;6:246ra96. doi: 10.1126/scitranslmed.3008680. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Stanton B.A., Coutermarsh B., Barnaby R., Hogan D. Pseudomonas aeruginosa Reduces VX-809 Stimulated F508del-CFTR Chloride Secretion by Airway Epithelial Cells. PLoS ONE. 2015;10:e0127742. doi: 10.1371/journal.pone.0127742. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Xue X., Mutyam V., Tang L., Biswas S., Du M., Jackson L.A., Dai Y., Belakhov V., Shalev M., Chen F., et al. Synthetic aminoglycosides efficiently suppress cystic fibrosis transmembrane conductance regulator nonsense mutations and are enhanced by ivacaftor. Am. J. Respir. Cell. Mol. Biol. 2014;50:805–816. doi: 10.1165/rcmb.2013-0282OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Wu Y.S., Jiang J., Ahmadi S., Lew A., Laselva O., Xia S., Bartlett C., Ip W., Wellhauser L., Ouyang H., et al. ORKAMBI-Mediated Rescue of Mucociliary Clearance in Cystic Fibrosis Primary Respiratory Cultures Is Enhanced by Arginine Uptake, Arginase Inhibition, and Promotion of Nitric Oxide Signaling to the Cystic Fibrosis Transmembrane Conductance Regulator Channel. Mol. Pharmacol. 2019;96:515–525. doi: 10.1124/mol.119.117143. [DOI] [PubMed] [Google Scholar]
- 83.Borcherding D.C., Siefert M.E., Lin S., Brewington J., Sadek H., Clancy J.P., Plafker S.M., Ziady A.G. Clinically-approved CFTR modulators rescue Nrf2 dysfunction in cystic fibrosis airway epithelia. J. Clin. Investig. 2019;129:3448–3463. doi: 10.1172/JCI96273. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Gentzsch M., Ren H.Y., Houck S.A., Quinney N.L., Cholon D.M., Sopha P., Chaudhry I.G., Das J., Dokholyan N.V., Randell S.H., et al. Restoration of R117H CFTR folding and function in human airway cells through combination treatment with VX-809 and VX-770. Am. J. Physiol. Lung Cell. Mol. Physiol. 2016;311:L550–L559. doi: 10.1152/ajplung.00186.2016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Schneider C.A., Rasband W.S., Eliceiri K.W. NIH Image to ImageJ: 25 years of image analysis. Nat. Methods. 2012;9:671–675. doi: 10.1038/nmeth.2089. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
The datasets used and/or analysed during the current study are available from the corresponding author on reasonable request.