Abstract
Chinese Cordyceps is a valuable source of natural products with various therapeutic effects. It is rich in various active components, of which adenosine, cordycepin and polysaccharides have been confirmed with significant immunomodulatory and antitumor functions. However, the underlying antitumor mechanism remains poorly understood. In this review, we summarized and analyzed the chemical characteristics of the main components and their pharmacological effects and mechanism on immunomodulatory and antitumor functions. The analysis revealed that Chinese Cordyceps promotes immune cells’ antitumor function by via upregulating immune responses and downregulating immunosuppression in the tumor microenvironment and resetting the immune cells’ phenotype. Moreover, Chinese Cordyceps can inhibit the growth and metastasis of tumor cells by death (including apoptosis and autophagy) induction, cell-cycle arrest, and angiogenesis inhibition. Recent evidence has revealed that the signal pathways of mitogen-activated protein kinases (MAPKs), nuclear factor kappaB (NF-κB), cysteine–aspartic proteases (caspases) and serine/threonine kinase Akt were involved in the antitumor mechanisms. In conclusion, Chinese Cordyceps, one type of magic mushroom, can be potentially developed as immunomodulator and anticancer therapeutic agents.
Keywords: Chinese Cordyceps, antitumor, immunomodulatory, bioactive components, mechanism
1. Bioactive Components
According to the epidemiological studies, a high intake of foods rich in bioactive compounds is beneficial to human health. Well-known as medicinal mushrooms, both natural and cultured products of Chinese Cordyceps many bioactive components. Over 20 bioactive ingredients have been isolated and their bioactivities/pharmacological effects have been proven (Table 1), of which, adenosine, cordycepin and polysaccharides are of significance in antitumor and immune regulation and considered to be the most important properties of Chinese Cordyceps [1,2]. In addition, Chinese Cordyceps is rich in fatty acids (including saturated and unsaturated fatty acids), vitamins, metal elements and other components, which also have positive effects on reducing blood lipids, preventing cardiovascular diseases, protecting kidneys and improving the essence of life [3,4].
Table 1.
Bioactive components and bioactivities of Chinese Cordyceps.
| No. | Bioactive Components | Pharmacological Effects | Ref. |
|---|---|---|---|
| 1 | Adenosine | Antitumor activity | [5,6,7,8] |
| Attenuation of chronic heart failure | [9] | ||
| Anti-inflammation | [10,11,12,13,14] | ||
| Immunomodulatory activity | [12,15] | ||
| 2 | Inosine | Anti-inflammation | [16] |
| 3 | Guanosine | Seizure prevention | [17] |
| Immunomodulatory activity | [15] | ||
| 4 | Cordycepin | Antitumor activity | [18,19,20,21,22,23,24,25,26,27,28,29] |
| Antibacterial activity | [20] | ||
| Treatment for ischemic/reperfusion (IR) injury | [21] | ||
| Anti-inflammation | [22,23,24,25,26,27,28,29,30,31,32,33,34,35,36,37] | ||
| Immunomodulatory activity | [38,39,40] | ||
| Antioxidant activity | [41,42] | ||
| Cholesterol lowering effect | [43] | ||
| Anti-fibroblast activity | [44] | ||
| 5 | Cordycepic acid | Diuretic effect | [45] |
| Attenuating postreperfusion syndrome | [46] | ||
| Anti-fibrosis and anti-inflammation | [47] | ||
| 6 | Polysaccharides | Antitumor activity | [48,49,50,51,52] |
| Immunomodulatory activity | [48,53,54,55,56,57,58,59,60,61] | ||
| Anti-inflammation | [56,62,63] | ||
| Antioxidant activity | [61,64,65] | ||
| Antiviral activity | [66] | ||
| Protective effects on kidney | [67,68] | ||
| Hypoglycemic effect | [69] | ||
| 7 | Cordymin | Analgesic effect | [70] |
| Anti-inflammation | [71] | ||
| Antioxidant | [71] | ||
| Hypoglycemic effect | [72] | ||
| 8 | Cordycedipeptide A | Antitumor activity | [73] |
| 9 | Tryptophan | Sedative hypnotic effect | [74] |
| 10 | Fibrinolytic enzymes | Treatment for thrombosis | [75,76] |
| 11 | Ergosterol | Cytotoxicity | [77] |
| Anti-inflammation | [78] | ||
| Anti-fibroblast activity | [79] | ||
| Antiviral activity | [80] | ||
| 12 | β-Sitosterol | Cytotoxicity | [77,81] |
| 13 | 5α,8α-epidioxy-22E-ergosta-6,22-dien-3β-ol | Cytotoxicity | [77] |
| 14 | 5α,8α-epidioxy-22E-ergosta-6,9(11),22-trien-3β-ol | Cytotoxicity | [77] |
| 15 | 5α,6α-epoxy-5α-ergosta-7,22-dien-3β-ol | Cytotoxicity | [77] |
| 16 | H1-A | Cytotoxicity | [82] |
| 17 | Cordysinin A | Anti-inflammatory | [83] |
| 18 | Cordysinin B | Anti-inflammatory | [83] |
| 19 | Cordysinin C | Anti-inflammatory | [83] |
| 20 | Cordysinin D | Anti-inflammatory | [83] |
| 21 | Cordysinin E | Anti-inflammatory | [83] |
1.1. Adenosine and Cordycepin
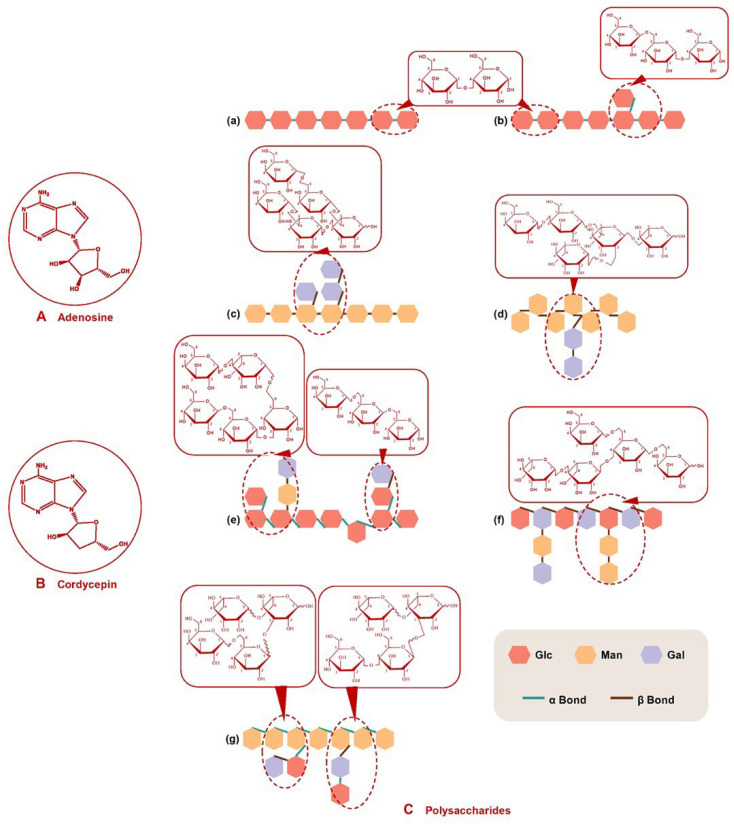
Adenosine is regarded as an important marker reflecting the quality of Chinese Cordyceps in Chinese Pharmacopoeia [84,85]. The molecular weight (MW) of cordycepin is 267.245 Da, molecular formula C10H13N5O4 and structure is shown in Figure 1A. The content of adenosine in Chinese Cordyceps ranges from 0.28 to 14.15 mg/g [86]. Cordycepin, another important nucleoside, is a purine alkaloid and confirmed as 3′-deoxyadenosine, similar in structure to adenosine except for lacking a hydroxyl group in the 3′ position of its ribose moiety (Figure 1B). The MW of cordycepin is 251.24 Da, molecular formula C10H13N5O3. The content of cordycepin ranges from 0.006 to 6.36 mg/g in O. sinensis [87] and can reach 2.28 mg/g in cultured C. militaris [88].
Figure 1.
Chemical structure of adenosine (A), cordycepin (B) and polysaccharides (C) of Cordyceps spp. Illustration of the chemical structure of several Cordyceps polysaccharides: (a) (α1→4)-glucan; (b) (α1→6)-branched, (α1→4)-glucan; (c) (β1→6)-branched, (β1→4)-galactomannan; (d) (β1→4)-(β1→6)-branched, (β1→2)-(β1→6)-galactomannan; (e) (β1→4)-(β1→6)-(α1→6)-branched, (α1→3)-galactoglucmannan; (f) (β1→4)-branched, (β1→6)-galactoglucmannan; (g) (α1→4)-(β1→6)-branched, (α1→6)-galactoglucmannan.
Both adenosine and cordycepin belong to nucleosides, and nucleosides can be dissolved with water, methanol and ethanol [84], while they are insoluble in benzene, ether or chloroform. Usually, Chinese Cordyceps is ground into powder and extracted with methanol aqueous solution or distilled water using an ultrasonic machine or a soxhlet extractor to obtain nucleosides [80,89]. Additionally, phosphate-buffered saline (PBS) is used as the solvent.
1.2. Polysaccharides
Polysaccharides are another important biologically active component of Chinese Cordyceps, with a content of 3–8% of the total weight [47]. The structure of polysaccharides is currently inconclusive, and a large amount of work was performed to try to explore its structure characteristic. It is reported that medicinal fungal polysaccharides with immunomodulatory and antitumor effects are most common glucans linked by various glycosidic bonds, such as (1→3)-, (1→6)-β, or (1→3)-, (1→4)-, (1→6)-α-glucans [90], and heteropolysaccharides are very important bioactive polysaccharides in Chinese Cordyceps [91]. Figure 1C shows the common structures of Chinese Cordyceps polysaccharides. Moreover, the pharmacological activity of polysaccharides is corelated to its MW. The larger the MW (10–1000 kDa) of the polysaccharides, the greater the water solubility and the better the biological activity [3]. It is reported that fungi polysaccharides have antitumor activity only when the MW is greater than 16,000 Da [47]. Usually, polysaccharides are colorless and odorless, and stably dissolved in water [92]. Polysaccharides have good water solubility, and water extraction combined with alcohol precipitation is a very effective method to extract active polysaccharides. Cordyceps polysaccharides include two forms: extracellular polysaccharide (EPS), mainly sourced from the fermentation broth of submerged Cordyceps spp., and intracellular polysaccharide (IPS), mainly resourcing from the fruiting bodies of Chinses Cordyceps and cultured mycelium. The chemical structure and immunomodulatory and antitumor activities of Polysaccharides originated from Cordyceps spp. are shown in Table 2.
Table 2.
Polysaccharides that originated from Cordyceps spp.: chemical structure and immunomodulatory and antitumor activities.
| No. | Name | MW | Components | Glycosyl Linkage and Branches (Characteristic Signals) | Bioactivities | Source | Ref. |
|---|---|---|---|---|---|---|---|
| 1 | AEPS-1 | 36 kDa | Glcp:GlcUp = 8:1 (M ratio), plus a trace amount of mannose |
A main chain of (1→3)-linked α-d-Glcp with α-d-Glcp and α-d-GlcUp branches attached to the main chain by (1→6) glycosidic bonds at every seventh α-d-Glcp unit | Anti-inflammatory; immunomodulatory | Mycelial fermentation of C. sinensis (Cs-HK1) | [93] |
| 2 | EPS | 104 kDa | Man:Glc:Gal = 23:1:2.6 (M ratio) | Immunomodulatory | Mycelial fermentation of C. sinensis (G1) | [58] | |
| 3 | NCSP-50 | 976 kDa | Glucose | A main chain of (1→4)-linked α-d-Glcp with α-d-Glcp branch attached to the C-6 | Immunomodulatory | C. sinensis | [59] |
| 4 | CSP | 28 kDa | Gal:Glc:Man:Ara:GalA = 36.40:28.99:24.81:3.34:7.55 (percentage ratio) | A main chain of (1→4)-linked α-d-Glc and (1→4)-linked α-d-Gal | Antitumor | Cultured mycelia of C. sinensis | [51,65] |
| 5 | CME-1 | 27.6 kDa | Man:Gal:Glc = 39.1:59.2:1.7 (M ratio) | A backbone of (1→4)-linked β-d-Man with Gal branches attached to the O- 6 | Antitumor | Cultured mycelia of C. sinensis | [49] |
| 6 | APSP | Man:Glc:Gal = 3.5:1:1.5 (M ratio) | Immunomodulatory | Mycelia of liquid cultured C. sinensis | [53] | ||
| 7 | PLCM (CPSN Fr II) |
36 kDa | Man:Gal:Glc:Protein:Hexosamine:Uronic acid = 65.12:28.72:6.12:0.20:0.06:0.29 (percentage ratio) | A backbone of (1→2)-, (1→6)-linked β-d-Man with (1→4)-linked β-d-Gal branches attached to the O- 6 | Immunomodulatory | C. militaris liquid culture broth | [55,94] |
| 8 | CMP-III | 4.796 × 104 kDa | Glc:Man:Gal = 8.09:1.00:0.25 (M ratio) | A backbone of (1→4)-linked α-d-Glc with (1→4,6)-linked α-d-Man and (1→2,6)-α-d-Man branches attached to the O- 6 | Immunomodulatory | Cultured fruiting bodies of C. militaris | [54] |
| 9 | CMPB90-1 | 5.8 kD | Gal:Glc:Man = 3.04:1:1.45 (M ratio) | A main chain of (1→6)-linked α-d-Glc and (1→3)- linked α-d-Glc, with branching at O-6, which consists of (1→4)-linked β-d-Man and (1→6)-linked α-d-Glc, respectively, and β-d-Man as the terminal unit | Immunomodulatory | Cultured fruiting bodies of C. militaris | [95] |
| 10 | CPMN Fr III | 210 kDa | Glc:Gal:Man = 9.17:18.61:72.22 (M ratio) | A backbone of (1→6)- linked β-d-Man and (1→6)- linked β-d-Glc with branches of (1→4)- linked β-d-Man terminated with d-Gal and d-Man, respectively | Immunomodulatory | Cultured mycelia of C. militaris | [50] |
| 11 | HS002-II | 44 kDa | D-Man:D-Rib:L-Rha:D-GlcUA:D-GalUA:D-Glc:D-Gal:D-Xyl:L-Ara = 6.47:2.27:1.25:0.69:0.42:65.89:16.17:2.13:4.26 (M ratio) polysaccharide:protein = 57.9:42.1 (percentage ratio) |
A long backbone of (1→3)-linked α-d-Ribf, (1→4)-linked α-d-Xylp and approximately 1/31 of (1→4)-linked β-d-Glcp, which was substituted at C-6. The two branches were (1→6)-linked β-d-Manp and (1→6)-linked β-d-Galp terminated with α-L-Arap, respectively | Immunomodulatory | Mycelial fermentation of Hirsutella sinensis Liu, Guo, Yu and Zeng | [96] |
| 12 | P70-1 | 42 k Da | Man:Gal:Glc = 3.12:1.45:1.00 (M ratio) | A backbone of (1→6)-linked α-d-Manp with branching points at O-3, and the branches composed of (1→4)-linked α-d-Glcp and (1→6)-linked β-d-Galp, and terminated with β-d-Galp and α-d-Glcp | Antioxidant | Fruiting bodies of cultured C. militaris | [97] |
| 13 | CPS-1 | 23 kDa | Rha:Xyl:Man:Glc:Gla = 1:6.43:25.6:16.0:13.8 (M ratio) | Composed of (1→2)-linked Man, (1→4)-linked Xyl and (1→2)-linked or (1→3)-linked Rha or Gal | Anti-inflammatory | Cultured C. militaris | [63] |
| 14 | AIPS | 1.15 × 103 kDa | Glucose | α-d-(1→4) glucan | Antitumor | Mycelial fermentation of C. sinensis (Cs-HK1) | [98] |
Ara, arabinose; Arap, arabinopyranosyl; Gal, galactose; GalA, galacturonic acid; GalUA, galacturonic acid; Glc, glucose; Glcp, glucopyranose; GlcUA, glucuronic acid; GlcUp, pyrano-glucuronic acid; Man, mannose; Manp, mannopyranosyl; MW, molecular weight; Rha, rhamnose; Rib, ribose; Ribf, ribofuranosyl; Xyl, Xylose; Xylp, xylopyranosyl.
2. Antitumor Activities and Their Participation in Molecular Mechanisms
2.1. Enhancing Antitumor Immune Responses
Studies have shown that the occurrence and development of tumors are closely related to immune surveillance. Immunotherapy has been proved to be an effective method to treat a variety of cancers [99] and increasing data has shown that antitumor effects of enhancing immunity may be associated with its action for the regulation of the tumor immune environment [100,101]. Immune cells show different phenotypes in response to various environmental cues (microbial products, damaged cells, cytokines, etc.). Chinese Cordyceps has a biphasic regulatory effect on the immune-cell phenotype and can increase antitumor immune activity in the tumor immune microenvironment (TIM): increasing the proinflammatory phenotypic while reversing the suppressive phenotype (Table 3).
Table 3.
Antitumor immunity effects and mechanisms of Chinese Cordyceps in various models.
| Bioactive Component | Pharmacological Effects | Models | Major Mediating Signaling Pathways | Mechanism of Action | Ref. |
|---|---|---|---|---|---|
| Cordycepin | ↑Antitumor immunity responses ↓CT 26 cell migration ↑CT 26 cell apoptosis |
CT 26 cells in mice | ↑CD4+ T, CD8+ T cells ↑NK cells ↑M1 macrophages ↑CD11b+, F4/80+ ↓CD47 |
[38] | |
| JLM 0636 (cordycepin-enriched extract of C. militaris) |
↑Th 1 cells ↑Immune responses ↓Treg cells ↓Immunosuppression |
FM3A murine breast cancer cells, derived from C3H/He mouse | ↑CD8+ T cells ↑IFN-γ ↓CD4+CD25+ T cells ↓IL-2 ↓TGF-β |
[39] | |
| WECS (Nucleoside extract of C. sinensis) |
↓MDA-MB-231 cells ↓4T1 cells ↑M1 macrophages ↑Immune responses |
MDA-MB-231, 4T1 breast cancer cells co-cultured with macrophages | NF-κB | ↑CD38 ↑iNOS ↑IL-1β ↑IL-12p70 ↑TNF-α ↑IL-6 ↑IFN-γ ↑NO |
[102] |
| EPSP | ↑M1 macrophages ↑Spleen lymphocyte ↑Immune response ↓Tumor migration |
B16 melanoma-bearing mice | ↓Bcl-2 | [52] | |
| APSF | ↑M1 macrophages ↑Immune response ↓M2 macrophages ↓Immunosuppression |
Ana-1 mouse macrophages co-cultured with H22 cells | NF-κB | ↑TNF-α ↑IL-12 ↑iNOS ↓IL-10 ↓SR ↓MR |
[103] |
| CMPB90-1 | ↑M1 macrophages ↑Immune response ↓M2 macrophages ↓Immunosuppression |
IL-4, tumor cell supernatant-induced RAW264.7 cells | NF-κB Akt MAPK (p38 and ERK) |
↓IL-10 ↓TGF-β ↓Arg-1 ↑IL-12 ↑iNOS |
[48] |
Akt, serine/threonine kinase; Arg-1, arginase-1; ERK, extracellular-signal-regulated kinases; IFN-γ, interferon-gamma; IL, interleukin; iNOS, inducible nitric oxide synthase; IPS, intracellular polysaccharide; MAPKs, mitogen-activated protein kinases; MR, mannose receptor; NK, natural killer cell; NF-κB, nuclear factor kappaB; NO, nitric oxide; TGF-β, transforming growth factor-beta; TNF-α, tumor necrosis factor-alpha; SR, scavenger receptor.
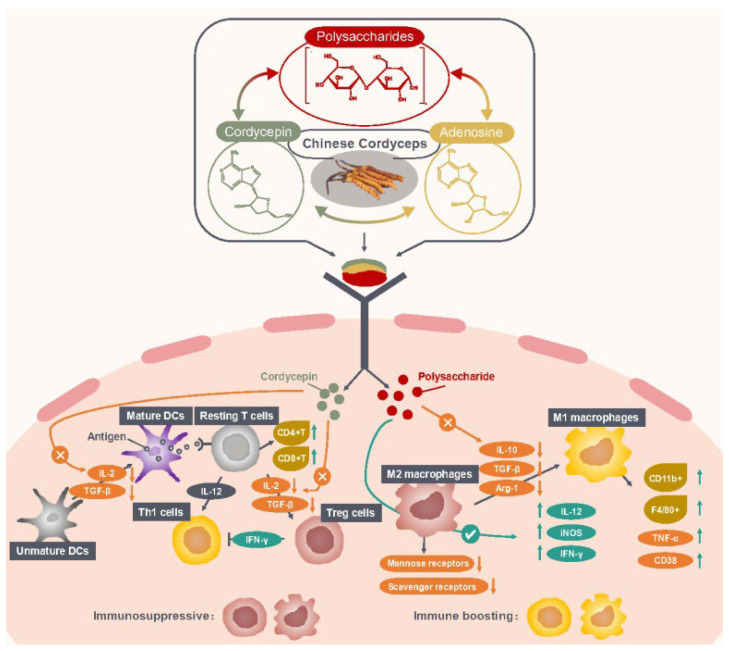
Chinese Cordyceps as an immunomodulator has suppressive effects on the immune system. Cordycepin has demonstrated to inhibit the differentiation of T cells into regulatory T cells (Treg, a suppressive phenotype of T cells) and delay tumor growth in tumor-bearing mice [39]. The further investigation reveals that cordycepin decreased the secretions of interleukin-2 (IL-2) and transforming growth factor-β (TGF-β) which were essential for Treg cells’ proliferation and differentiation. In addition, macrophages are the key player in the immune system which can engulf and destroy foreign pathogens and cancer cells. Tumor-associated macrophages (TAM), taking up 50% of the infiltrated cells at the tumor site, can be differentiated into two phenotypes: M1 phenotype (classic activation polarization) or M2 phenotype (alternative activation polarization), based on the stimulatory signals from the tumor microenvironment (TME) [104]. Macrophages in the TME are predominantly in an M2 state [105], and currently M2 macrophages are potential targets for the treatment of cancer. Activated M2 macrophages would suppress the immune system and promote tumor progression by releasing immunosuppressive cytokines (i.e., IL-4 and IL-10) and recruiting Th2 and Treg cells [106,107]. Clinical studies have proposed a new strategy where reversing M2 into the M1 phenotype is an effective approach to enhance antitumor immunity [108,109]. Chinese Cordyceps are capable to of resetting the macrophage phenotype repolarizing M2 to M1 macrophages. A study by Chen et al. [103] showed that APSF, a polysaccharide isolated from the fruiting bodies of O. sinensis, reversed M2 to the M1 phenotype through reducing the expression of IL-10 and increasing the expression of tumor necrosis factor-alpha (TNF-α), IL-12 and inducible nitric oxide synthase (iNOS), and downregulating the expressions of SR and MR (Scavenger Receptor and Mannose Receptor, M2 markers), in Ana-1 mouse macrophages co-cultured with a supernatant of H22 cells. Additionally, a novel polysaccharide CMPB90-1 from C. militaris was found to remodel TAMs from M2 to the M1 phenotype through decreasing the mRNA expression level of immunosuppressive cytokines (IL-10, TGF-β and Arg-1 (arginase 1), M2 markers) while increasing the mRNA expression levels of IL-12 and iNOS (M1 markers). Additionally, a further investigation revealed that the signaling pathways of p38, extracellular-signal-regulated kinases (ERK), Akt and nuclear factor kappaB (NF-κB) were activated [48]. These findings demonstrated that Chinese Cordyceps and its bioactive constituents could promote immune cells’ antitumor function by enhancing immune responses and downregulating immune suppression (Figure 2).
Figure 2.
Mechanism of Chinese Cordyceps regulating immune cells in TIM. Abbreviations: Arg-1, arginase-1; CD38, CD11b+ and F4/80+, M1 macrophage markers; IFN-γ, interferon-gamma; IL, interleukin; iNOS, inducible nitric oxide synthase; TGF-β, transforming growth factor-beta; TIM, tumor immune microenvironment; TNF-α, tumor necrosis factor-alpha.
2.2. Direct Antitumor Effects
To date, increasing studies have shown that Chinese Cordyceps has significant antitumor effects. Although the mechanisms of action are complicated, the possible mechanisms of antitumor action of Chinese Cordyceps are summarized and presented in Table 4.
Table 4.
Antitumor effects and mechanisms of Chinese Cordyceps in various cancer models.
| Cancer | Bioactive Component | Pharmacological Effects | Cell line | Major Mediating Signaling Pathways | Mechanism of Action | Ref. |
|---|---|---|---|---|---|---|
| Bladder cancer | ||||||
| Cordycepin | ↓Migration and invasion | TNF-α-induced 5637 and T-24 cells | NF-κB AP-1 |
↓MMP-9 | [23] | |
| Breast cancer | ||||||
| Cordycepin | ↑Apoptosis | MDA-MB-231 cells | Caspase | ↑Bax (mitochondria) ↑Cytochrome c (cytosol) ↑PARP ↑c-caspases-9, -3 ↑DNA fragmentation |
[19] | |
| Cordycepin | ↑Autophagy | MCF-7 cells | Autophagy | ↑LC3-II ↑Autophagosome-like structure |
[19] | |
| Cordycepin | ↑Apoptosis | MDA-MB-435 and T47D cells | ↑DNA fragmentation ↑Histone γH2AX ↓RNA synthesis |
[26] | ||
| C. militaris extract | ↑Apoptosis | MCF-7 cells | Caspase | ↑Bax/Bcl-2 ↑c-caspase-7, -8 |
[25] | |
| Cordycepin | ↑Apoptosis | C6 glioma cells | A2AR Caspase |
↑Caspase-7 ↑p-p53 ↑PARP |
[110] | |
| Cervical cancer | ||||||
| Cordycepin | ↑Apoptosis ↓Cell cycle |
SiHa cells HeLa cells |
↓CDK-2 ↓Cyclin-E1 ↓Cyclin-A2 ↑ROS |
[111] | ||
| CCP (C. cicadae polysaccharides) |
↑Apoptosis ↓Cell cycle |
hela cells | Akt | ↑Bak ↑Bax ↑Caspase-3, -7, -9 ↓P21 ↓P27 ↓CDK2 ↓Cyclin E1 ↓Cyclin A2 ↓Bcl-2 ↓Bcl-xl ↓PARP |
[112] | |
| Colon cancer | ||||||
| CSP | ↑Autophagy, ↑Apoptosis |
HCT116 cells | Autophagy mTOR Caspase |
↑LC3B-II ↑Caspase-8, -3 |
[51,65] | |
| Cordycepin | ↓Cell cycle | HCT116 cells | JNK MAPK | ↑p21WAF1 ↓Cyclin B1 ↓Cdc25c ↓Cdc2 |
[24] | |
| Colorectal cancer | ||||||
| C. militaris extract | ↓Cell cycle | RKO cells | ↑Bax ↑Bim ↑Bak ↑Bad ↑PARP ↑p-p53 ↑c-caspase -9, -3 |
[113] | ||
| Gastric cancer | ||||||
| Cordycepin | ↑Apoptosis | AGS cells | PI3K/Akt | ↑Caspase-9, -3, -7 ↑Bax ↓Bcl-2 |
[114] | |
| CECJ (C. jiangxiensis extract) |
↑Apoptosis ↓Cell cycle |
SGC-7901 cells | Caspase | ↑Caspase-3 | [115] | |
| Liver cancer | ||||||
| Adenosine | ↑Apoptosis | HepG2 cells | Caspase | ↑TNF ↑TRADD ↑TRAIL-R2 ↑FADD ↑Caspase-9, -8, -3 |
[8] | |
| Adenosine | ↑Apoptosis | BEL-7404 cells | Caspase | ↑Caspase-8, -9, -3 ↑c-PARP ↑Bak ↑Mcl1 ↑Bcl-xl |
[6] | |
| Adenosine | ↑Apoptosis | HuH-7 Fas-deficient cells | Caspase | ↑AMP ↓Caspase-3, -8 ↓c-FLIP |
[7] | |
| CMF (C. militaris extract) |
↓Migration and invasion ↓Tumor growth |
SMMC-7721 cells | Akt ERK |
↓p-VEGFR2 ↓p-Akt ↓p-ERK |
[116] | |
| Lung cancer | ||||||
| AECS1, AECS2 (C. sinensis nucleosides extract) |
↓Tumor growth | Lewis xenograft mouse | Akt NF-κB |
↓p-Akt ↓MMP2 ↓MMP9 ↓p-IκBα ↓TNF-α ↓COX-2 ↓Bcl2 ↓Bcl-xl ↑Bax |
[117] | |
| CS (C. sinensis extract) |
↑Apoptosis | H157 NSCLC cells | ↓VEGF ↓bFGF |
[118] | ||
| C. militaris extract | ↑Apoptosis ↓Cell cycle |
NCI-H460 cells | ↑P53 ↑P21 ↑53BP1 |
[119] | ||
| Mouse melanoma | ||||||
| Cordycepin | ↓Proliferation | B16-BL6 cells | A3R | ↑GSK-3β ↓Cyclin D1 protein |
[28] | |
| CME-1 | ↓Tumor migration | B16-F10 cells | NF-κB MAPK (ERK and p38) |
↓MMP-1 | [49] | |
| EPSP | ↓Tumor migration | B16 cells | ↓c-Myc ↓c-Fos ↓VEGF |
[120] | ||
| Myeloma cancer | ||||||
| Cordycepin | ↑Apoptosis | MM.1S cells | Caspase | ↑Caspase-9, -3, -8 ↓RNA synthesis |
[8] | |
| Oral cancer | ||||||
| CMP (C. militaris polysaccharides) |
↑Apoptosis ↓Cell cycle |
4NAOC-1 cells | STAT3 ERK |
↓ki-67 ↓EGFR ↓IL-17A ↓Cyclin B1 ↓DNA synthesis |
[121] | |
| WECM (C. militaris extract) |
Apoptosis ↓Cell cycle |
SCC-4 cells | ↓PCNA ↓VEGF ↓Caspase-3 ↓c-fos |
[122] | ||
| Ovarian cancer | ||||||
| CME (C. militaris extract) |
↑Apoptosis ↓Migration |
SKOV-3 cells | NF-κB | ↓TNF-1R ↓Bcl-2 ↑Bcl-xl |
[123] | |
| ↑Autophagy ↓Tumor growth |
A2780 and OVCAR3 cells | ENT1-AMPK-mTOR | ↑LC3II/LC3I ↑p-AMPK |
[124] | ||
| Prostate cancer | ||||||
| Cordycepin | ↓Migration and invasion | LNCaP cells | PI3K/Akt | ↑TIMP-1 ↑TIMP-2 ↓MMP-2 ↓MMP-9 |
[21] | |
| Testicular cancer | ||||||
| Cordycepin | ↑Apoptosis ↓Cell cycle |
MA-10 cells | Caspase | ↑Caspase-9, -3, -7 ↑DNA fragmentation |
[20] |
AMP, activated protein kinase; A2AR, adenine 2A receptor; A3R, adenine 3 receptor; c-FLIP, cellular FADD-like interleukin-1β-converting enzyme inhibitory protein; c-PARP, cleaved-poly ADP-ribose polymerase; COX-2, cyclooxygenase-2; c-Fos, c-Myc, cellular proto-oncogenes; cyclin B1, Cdc25c and Cdc2, cell cycle regulatory proteins; ERK, extracellular signal-regulated kinases; FADD, fas-associated death domain; JNK, Jun N terminal kinase; LC3-II, the lipidated form of LC3B; MMP, mitochondrial membrane potential; mTOR, mechanistic target of rapamycin; MAPK, mitogen-activated protein kinases; p-Akt, phosphorylated serine/threonine kinase; p21WAF1, cyclin-dependent kinase inhibitor; TNF, tumor necrosis factor; TRADD, TNF receptor-associated death domain; TRAIL-R2, TNF-related apoptosis-inducing ligand receptor 2; VEGF, vascular endothelial growth factor.
2.2.1. Inducing Apoptosis and Autophagy
Apoptosis is a form of programmed cell death and essential for the development and homeostasis of organisms, and its abnormal regulation is perhaps related to tumor formulation. Inducing apoptosis involves two major pathways: the intrinsic pathway (particularly mitochondrial stress) and extrinsic signal pathway. The Fas/FasL system plays an important role in apoptosis regulation. Fas and its ligand FasL are mainly expressed on the cell membrane surface. When external FasL is expressed by cytotoxic T lymphocytes and combines with Fas which is expressed by target cells, Fas-associated death domain (FADD) will be formed. FADD triggers apoptosis through recruiting extrinsic stimuli and death receptors (DRs) [125,126]. A study by Lee et al. [127] showed that cordycepin inhibited proliferation and induced HT-29 colon cancer cells’ apoptosis by increasing expression of DR3, caspase-8, -1 and -3. Caspases is a family of cysteine proteases and acts on proteins or enzymes related to the cytoskeleton or DNA and is responsible for apoptosis. DR3 activated apoptosis through triggering TRADD, FADD and caspase-8 [128], and caspase-8 further activated downstream effectors caspase-1 and caspase-3, resulting in cell death [129,130]. Similarly, cordycepin induced apoptosis in human prostate carcinoma LNCaP cells via the caspase pathway by increasing the expression of Fas, DR5, caspase-8, -9 and -3, and causing a dose-dependent increase in pro-apoptotic Bax and decrease in anti-apoptotic Bcl-2 [131]. Changes in Bax and Bcl-2 levels trigger a collapse of mitochondrial membrane potential and activation of caspase-9 and -3. A study by Balk et al. [132] revealed that cordycepin increased the levels of Fas, FasL and TRAIL (related apoptosis-inducing ligand) of U87MG cells, and decreased Bcl-2 level, indicating that cordycepin induced apoptosis via the Fas/FasL pathway. Lee et al. [25] found that C. militaris extract induced apoptosis by increasing the protein expression ratio of Bax/Bcl-2 and the cleavage of caspase-7, -8 and -9 in MCF-7 cells. In addition, cordycepin has been demonstrated to inhibit the proliferation of B16-BL6 mouse melanoma cells through combination with adenosine A3 receptor (A3R) on the B16 cell membrane, reducing expression of cyclin D1 protein and activating glycogen synthase kinase-3β (GSK-3β) [28]. In addition, cordycepin is also thought to induce apoptosis through A3R and A2AR. The apoptosis induction of cordycepin is possibly mediated by A3R in human bladder cancer T24 cells since both overexpression of A3R and cordycepin treatment decreased cell survival since the apoptosis-inducing effect of cordycepin is abolished with the depletion of adenosine receptors [133]. Moreover, adenosine was found to induce apoptosis and upregulate mRNAs of TNF, FADD, TRADD, and TRAIL-2 by activating caspase-3, -8 and -9 in human hepatoma HepG2 cells [8]. Ma et al. [6] found a novel apoptosis mechanism that extracellular adenosine could trigger apoptosis by increasing reactive oxygen species (ROS) production and mitochondrial membrane dysfunction in BEL-7404 liver cancer cells. Choi et al. [19] found that cordycepin induced MDA-MB-231 cells’ apoptosis through increasing translocation of Bax in the mitochondrion and triggering cytosolic release of cytochrome c and activation of caspases-9 and -3. These studies indicate that Chinese Cordyceps induces apoptosis via both the mitochondrion-mediated intrinsic pathway and extrinsic Fas/FasL and ARs pathways.
Autophagy, mediated by an intracellular suicide program, plays important roles in antitumor responses. A study by Qi et al. [51] showed that a polysaccharide named CSP from O. sinensis mycelia inhibited the proliferation of HCT116 human colon cancer cells through inducing apoptosis and autophagy. On one hand, CSP induced apoptosis by activating caspase-8 and -3, on the other hand, it inhibited lysosome formation, blocked autophagy flux and accumulated autophagosomes, resulting in autophagy. The further investigation revealed that signaling pathways of PI3K-AKT-mTOR and AMPK-mTOR-ULK1 were all involved. Cordycepin was also found to induce autophagic cell death and formation of a large membranous vacuole in MCF-7 human breast cancer cells, accompanied with the increase in autophagosome marker LC3-II levels [19]. Generally, autophagy occurs before apoptosis under certain stress stimuli, and autophagy will be inactivated when stress exceeds the intensity threshold or critical duration, followed by the activation of apoptosis [134].
2.2.2. Blocking Cell Cycle
The cell cycle can be mainly divided into four phases, of which G1, S and G2/M phases are crucial checkpoints in the cell cycle processes. Cell cycle arrest in the G1, S and G2/M phases can lead to the inhibition of tumor cells’ proliferation and induction of apoptosis. Adenosine, cordycepin and polysaccharides were found to cause cell cycle arrest at certain checkpoints. Cordycepin inhibited the growth of 5637 and T-24 bladder cancer cells and HCT116 colon cancer cells, through G2/M cell-cycle arrest. The expression of p21WAF1 (a universal key inhibitor in regulating cell-cycle progression) was upregulated and cyclin B1, Cdc25c and Cdc2 (G2/M cell-cycle regulatory proteins) were downregulated, through the JNK1 signal pathway [50,135]. Cells blocked in the G2/M phase failed to enter mitosis, resulting in cell growth inhibition. Moreover, cordycepin induced an increase in subG1 cell number and the decrease in G1 and G2/M cell numbers and cell viability through inducing caspase-9, -3 and -7 expression as subG1 phase accumulation could be partly suppressed using a caspase inhibitor, which indicated that the caspase-9 pathway is involved in cordycepin-induced subG1 phase arrest [20].
In addition, cordycepin is a transcription and polyadenylation inhibitor and affects RNA synthesis. A study showed that cordycepin caused accumulation of the corresponding triphosphate derivative, 3′dATP, which might lead to the incorporation of analogue into nascent nucleic acid oligonucleotides and RNA synthesis inhibition [18,136]. This might illustrate that cordycepin affects the cell cycle from another perspective. An extract of Cordyceps cicadae was identified as a nucleoside mixture containing adenine, adenosine, uridine and N6-(2-Hydroxyethyl)-adenosine, induced S phase arrest in human gastric cancer SGC-7901 cells, which was related to downregulation of CDK2 expression and upregulation of expression of transcription factor E2F1 (cyclin/CDK complexes, regulating G1/S phase transition), cyclin A2 and cyclin E [137].
2.2.3. Inhibiting Migration, Invasion and Metastasis
Metastasis refers to the movement of cancer cells from primary tumor sites to other organs and tissues and is the end result of multiple interactions including invasion between the tumor and host, indicating the uncontrolled spread of the tumor cells. Epithelial–mesenchymal transition (EMT)-related proteins such as matrix metalloproteinases (MMPs) play an important role in metastasis. For example, MMP-2 and MMP-9 can lead to the degradation of extracellular matrix (ECM) components and tissue invasion [138,139,140]. A study showed that cordycepin inhibited 5637 and T-24 cells’ invasion through decreasing MMP-9 expression and the transcriptional activity of activator protein-1 (AP-1), which were identified by gel-shift assay as cis-elements for TNF-α activation of the MMP-9 promoter via the NF-κB/MMP-9 pathway [94]. In addition, a novel polysaccharide CME-1 isolated from O. sinensis was found to inhibit migration of B16-F10 melanoma cells, and the mechanism was that CME-1 reduced MMP-1 expression and downregulated the phosphorylation level of ERK1/2 and p38 MAPK [49].
Angiogenesis is vital for organ growth and repair, and essential for tumor growth. The vascular endothelial growth factor (VEGF) family plays an important role in angiogenesis. VEGF, a key angiogenic growth factor, has a higher expression level in tumor tissues and can accelerate the differentiation, proliferation, and migration of endothelial cells. Chinese Cordyceps has been demonstrated to inhibit the VEGF/VEGFR2 signaling pathway and exert antiangiogenesis function [116]. Moreover, the overexpression of proto-oncogenes c-Myc and c-Fos may promote tumor cell proliferation under growth-promoting stimulation. c-Myc, encoding a ubiquitous transcription factor and promoting cell division, is related to apoptosis and the occurrence and development of various tumors. c-Fos, essential for cell proliferation, can upregulate the cell cycle by induction of cyclin D1 [141]. c-Fos is expressed at a low level in normal cells while it is overexpressed in tumor cells. Yang et al. [120] found that EPSF isolated from C.sinensis could downregulate the expression of VEGF, c-Myc and c-Fos, which was the important factor to inhibit tumor growth, invasion and metastasis.
3. Discussion
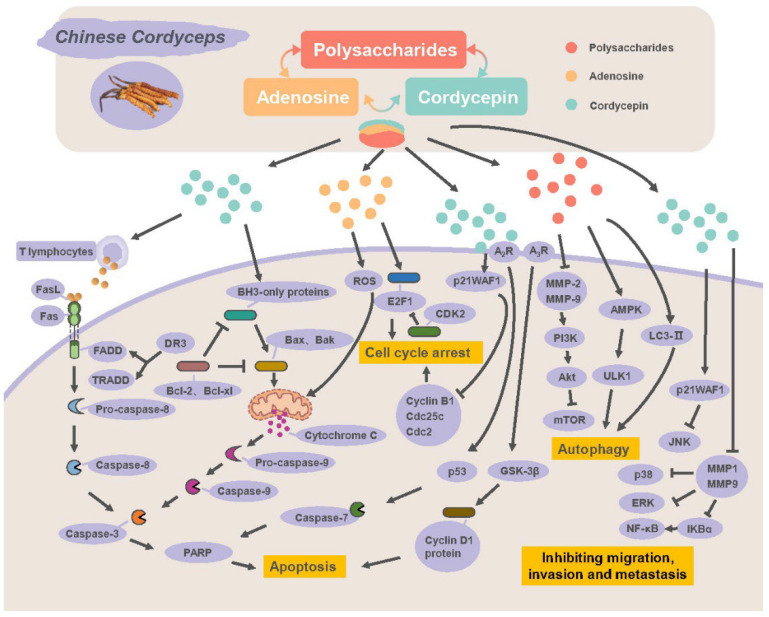
In recent years, the traditional therapy for cancer has become an attention direction of researchers, and many researchers believe that traditional therapy is a potential new therapy. The pathogenesis of cancer is diverse and complex, and Chinese Cordyceps has many active ingredients and diverse extracts, which can inhibit the growth of various tumors and prevent or overcome metastasis through various pathways (Figure 3). It is well known that improving self-immunity can lay a good foundation for fighting and treating many diseases. Chinese Cordyceps has a long history of use in China, and much evidence suggests that Chinese Cordyceps, acting as an immune response activator, is used for the treatment of a variety of diseases including cancer. Increasing studies have shown that Chinese Cordyceps has immunomodulatory, anti-inflammatory and antioxidant activities that affect the immune system and TME in various ways. The polarization and remodeling of the phenotype of immune cells (such as T cells and macrophages) by Chinese Cordyceps have effects on cytokine production in TME, which may affect tumor progression. The anticancer ability of Chinese Cordyceps has been the subject of research for nearly 60 years and its antitumor effect has been confirmed in cancer cells or mouse cancer models alone or in combination with other drugs. The research on clinical application still needs more attention.
Figure 3.
Possible mechanisms of antitumor activity of Chinese Cordyceps.
4. Conclusions
In conclusion, Chinese Cordyceps has significant antitumor activity and immunomodulatory activity. On the one hand, it can directly act on tumor cells to kill tumor cells or inhibit tumor growth and effectively attenuate tumor cell metastasis. On the other hand, Chinese Cordyceps can change the tumor microenvironment and enhance antitumor immune responses by downregulating the expression of immunosuppressive factors and upregulating the expression of pro-inflammatory factors, thereby improving the antitumor function. These findings may provide therapeutic strategies for treating cancer.
Author Contributions
Conceptualization, Y.L. and X.-W.Z.; writing—original draft preparation, Y.L.; data curation, Y.L.; drawing, Z.-J.G.; writing—review and editing, X.-W.Z.; supervision, X.-W.Z. All authors have read and agreed to the published version of the manuscript.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Conflicts of Interest
The authors declare no conflict of interest.
Sample Availability
Not applicable.
Funding Statement
This study was supported by Open Project Funding of the Key Laboratory of Fermentation Engineering (Ministry of Education) grant number 202105FE02.
Footnotes
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Bhetwal S., Chattarjee S., Rijal R., Rana M., Srivastava S. Cordyceps sinensis: Peculiar caterpillar mushroom, salutary in its medicinal and restorative capabilities. J. Pharm. Innov. 2021;10:1045–1054. [Google Scholar]
- 2.Xiao G., Miyazato A., Abe Y., Zhang T., Nakamura K., Inden K., Tanaka M., Tanno D., Miyasaka T., Ishii K., et al. Activation of myeloid dendritic cells by deoxynucleic acids from Cordyceps sinensis via a Toll-like receptor 9-dependent pathway. Cell. Immunol. 2010;263:241–250. doi: 10.1016/j.cellimm.2010.04.006. [DOI] [PubMed] [Google Scholar]
- 3.Zhou X.W., Gong Z.H., Su Y., Lin J., Tang K.X. Cordyceps fungi: Natural products, pharmacological functions and developmental products. J. Pharm. Pharmacol. 2009;61:279–291. doi: 10.1211/jpp.61.03.0002. [DOI] [PubMed] [Google Scholar]
- 4.Yue K., Ye M., Zhou Z., Sun W., Lin X. The genus Cordyceps: A chemical and pharmacological review. Pharm. Pharmacol. 2013;65:474–493. doi: 10.1111/j.2042-7158.2012.01601.x. [DOI] [PubMed] [Google Scholar]
- 5.Haskó G., Cronstein B.N. Adenosine: An endogenous regulator of innate immunity. Trends Immunol. 2004;25:33–39. doi: 10.1016/j.it.2003.11.003. [DOI] [PubMed] [Google Scholar]
- 6.Ma Y.F., Zhang J., Zhang Q., Chen P., Song J., Yu S., Liu H., Liu F., Song C., Yang D., et al. Adenosine induces apoptosis in human liver cancer cells through ROS production and mitochondrial dysfunction. Biochem. Biophys. Res. Commun. 2014;448:8–14. doi: 10.1016/j.bbrc.2014.04.007. [DOI] [PubMed] [Google Scholar]
- 7.Yang D., Yaguchi T., Yamamoto H., Nishizaki T. Intracellularly transported adenosine induces apoptosis in HuH-7 human hepatoma cells by downregulating c-FLIP expression causing caspase-3/-8 activation. Biochem. Pharmacol. 2007;73:1665–1675. doi: 10.1016/j.bcp.2007.01.020. [DOI] [PubMed] [Google Scholar]
- 8.Yang D., Yaguchi T., Lim C.R., Ishizawa Y., Nakano T., Nishizaki T. Tuning of apoptosis-mediator gene transcription in HepG2 human hepatoma cells through an adenosine signal. Cancer Lett. 2010;291:225–229. doi: 10.1016/j.canlet.2009.10.016. [DOI] [PubMed] [Google Scholar]
- 9.Kitakaze M., Hori M. Adenosine therapy: A new approach to chronic heart failure. Expert Opin. Investig. Drugs. 2000;9:2519–2535. doi: 10.1517/13543784.9.11.2519. [DOI] [PubMed] [Google Scholar]
- 10.Cronstein B.N., Sitkovsky M. Adenosine and adenosine receptors in the pathogenesis and treatment of rheumatic diseases. Nat. Rev. Rheumatol. 2017;13:41–51. doi: 10.1038/nrrheum.2016.178. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Csoka B., Selmeczy Z., Koscso B., Nemeth Z.H., Pacher P., Murray P.J., Kepka-Lenhart D., Morris S.M., Jr., Gause W.C., Leibovich J.S., et al. Adenosine promotes alternative macrophage activation via A2A and A2B receptors. FASEB J. 2012;26:376–386. doi: 10.1096/fj.11-190934. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Da Rocha Lapa F., da Silva M.D., de Almeida Cabrini D., Santos A.R.S. Anti-inflammatory effects of purine nucleosides, adenosine and inosine, in a mouse model of pleurisy: Evidence for the role of adenosine A2 receptors. Purinergic Signal. 2012;8:693–704. doi: 10.1007/s11302-012-9299-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Safarzadeh E., Jadidi-Niaragh F., Motallebnezhad M., Yousefi M. The role of adenosine and adenosine receptors in the immunopathogenesis of multiple sclerosis. Inflamm. Res. 2016;65:511–520. doi: 10.1007/s00011-016-0936-z. [DOI] [PubMed] [Google Scholar]
- 14.Subramanian M., Kini R., Madasu M., Ohta A., Nowak M., Exley M., Sitkovsky M., Ohta A. Extracellular adenosine controls NKT-cell-dependent hepatitis induction. Eur. J. Immunol. 2014;44:1119–1129. doi: 10.1002/eji.201343866. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Yu L., Zhao J., Zhu Q., Li S.P. Macrophage biospecific extraction and high performance liquid chromatography for hypothesis of immunological active components. J. Pharm. Biomed. 2007;44:439–443. doi: 10.1016/j.jpba.2007.01.003. [DOI] [PubMed] [Google Scholar]
- 16.Welihinda A.A., Kaur M., Greene K., Zhai Y., Amento E.P. The adenosine metabolite inosine is a functional agonist of the adenosine A2A receptor with a unique signaling bias. Cell. Signal. 2016;28:552–560. doi: 10.1016/j.cellsig.2016.02.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Vinadé E.R., Schmidt A.P., Frizzo M.E.S., Izquierdo I., Elisabetsky E., Souza D.O. Chronically administered guanosine is anticonvulsant, amnesic and anxiolytic in mice. Brain Res. 2003;977:97–102. doi: 10.1016/S0006-8993(03)02769-0. [DOI] [PubMed] [Google Scholar]
- 18.Chen L.S., Stellrecht C.M., Gandhi V. RNA-directed agent, cordycepin, induces cell death in multiple myeloma cells. Br. J. Haematol. 2008;140:682–687. doi: 10.1111/j.1365-2141.2007.06955.x. [DOI] [PubMed] [Google Scholar]
- 19.Choi S., Lim M.H., Kim K.M., Jeon B.H., Song W.O., Kim T.W. Cordycepin-induced apoptosis and autophagy in breast cancer cells are independent of the estrogen receptor. Toxicol. Appl. Pharmacol. 2011;257:165–173. doi: 10.1016/j.taap.2011.08.030. [DOI] [PubMed] [Google Scholar]
- 20.Jen C.Y., Lin C.Y., Huang B.M., Leu S.F. Cordycepin induced MA-10 mouse Leydig tumor cell apoptosis through caspase-9 pathway. Evid. Based Complement. Altern. Med. 2011;2011:984537. doi: 10.1093/ecam/nen084. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Jeong J.W., Jin C.Y., Park C., Han M.H., Kim G.Y., Moon S.K., Kim C.G., Jeong Y.K., Kim W.J., Lee J.D., et al. Inhibition of migration and invasion of LNCaP human prostate carcinoma cells by cordycepin through inactivation of Akt. Int. J. Oncol. 2012;40:1697–1704. doi: 10.3892/ijo.2012.1332. [DOI] [PubMed] [Google Scholar]
- 22.Jin Y., Meng X., Qiu Z., Su Y., Yu P., Qu P. Antitumor and antimetastatic roles of cordycepin, one bioactive compound of Cordyceps militaris. Saudi J. Biol. Sci. 2018;25:991–995. doi: 10.1016/j.sjbs.2018.05.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Lee E.J., Kim W.J., Moon S.K. Cordycepin suppresses TNF-alpha-induced invasion, migration and matrix metalloproteinase-9 expression in human bladder cancer cells. Phytother. Res. 2010;24:1755–1761. doi: 10.1002/ptr.3132. [DOI] [PubMed] [Google Scholar]
- 24.Lee S.J., Moon G.S., Jung K.H., Kim W.J., Moon S.K. c-Jun N-terminal kinase 1 is required for cordycepin-mediated induction of G2/M cell-cycle arrest via p21WAF1 expression in human colon cancer cells. Food Chem. Toxicol. 2010;48:277–283. doi: 10.1016/j.fct.2009.09.042. [DOI] [PubMed] [Google Scholar]
- 25.Lee D., Lee W.Y., Jung K., Kwon Y.S., Kim D., Hwang G.S., Kim C.E., Lee S., Kang K.S. The inhibitory effect of cordycepin on the proliferation of MCF-7 breast cancer cells, and its mechanism/ an investigation using network pharmacology-based analysis. Biomology. 2019;9:414. doi: 10.3390/biom9090414. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Lee H.J., Burger P., Vogel M., Friese K., Bruening A. The nucleoside antagonist cordycepin causes DNA double strand breaks in breast cancer cells. Investig. New Drugs. 2012;30:1917–1925. doi: 10.1007/s10637-012-9859-x. [DOI] [PubMed] [Google Scholar]
- 27.Leu S.F., Poon S.L., Pao H.Y., Huang B.M. The in vivo and in vitro stimulatory effects of cordycepin on mouse Leydig cell steroidogenesis. Biosci. Biotechnol. Biochem. 2011;75:723–731. doi: 10.1271/bbb.100853. [DOI] [PubMed] [Google Scholar]
- 28.Yoshikawa N., Yamada S., Takeuchi C., Kagota S., Shinozuka K., Kunitomo M., Nakamura K. Cordycepin (3′-deoxyadenosine) inhibits the growth of B16-BL6 mouse melanoma cells through the stimulation of adenosine A3 receptor followed by glycogen synthase kinase-3β activation and cyclin D1 suppression. Naunyn-Schmiedeberg’s Arch. Pharmacol. 2008;377:591–595. doi: 10.1007/s00210-007-0218-y. [DOI] [PubMed] [Google Scholar]
- 29.Zheng Q., Sun J., Li W., Li S., Zhang K. Cordycepin induces apoptosis in human tongue cancer cells in vitro and has anti- tumor effects in vivo. Arch. Oral Biol. 2020;118:104846. doi: 10.1016/j.archoralbio.2020.104846. [DOI] [PubMed] [Google Scholar]
- 30.Jiang Q., Lou Z., Wang H., Chen C. Antimicrobial effect and proposed action mechanism of cordycepin against Escherichia coli and Bacillus subtilis. J. Microbiol. 2019;57:288–297. doi: 10.1007/s12275-019-8113-z. [DOI] [PubMed] [Google Scholar]
- 31.Park E.S., Kang D.H., Yang M.K., Kang J.C., Jang Y.C., Park J.S., Shin H.S. Cordycepin, 3’-deoxyadenosine, prevents rat hearts from ischemia/reperfusion injury via activation of Akt/GSK-3β/p70S6K signaling pathway and HO-1 expression. Cardiovasc. Toxicol. 2014;14:1–9. doi: 10.1007/s12012-013-9232-0. [DOI] [PubMed] [Google Scholar]
- 32.Choi Y.H., Kim G.Y., Lee H.H. Anti-inflammatory effects of cordycepin in lipopolysaccharide-stimulated RAW 264.7 macrophages through Toll-like receptor 4-mediated suppression of mitogen-activated protein kinases and NF-κB signaling pathways. Drug Des. Dev. Ther. 2014;8:1941–1953. doi: 10.2147/DDDT.S71957. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Jeong J.W., Jin C.Y., Kim G.Y., Lee J.D., Park C., Kim G.D., Kim W.J., Jung W.K., Seo S.K., Choi I.W., et al. Anti-inflammatory effects of cordycepin via suppression of inflammatory mediators in BV2 microglial cells. Int. Immunopharmacol. 2010;10:1580–1586. doi: 10.1016/j.intimp.2010.09.011. [DOI] [PubMed] [Google Scholar]
- 34.Kim H., Naura A.S., Boulares H. Cordycepin blocks lung injury-associated inflammation and promotes BRCA1-deficient breast cancer cell killing by effectively inhibiting PARP. Mol. Med. 2011;17:893–897. doi: 10.2119/molmed.2011.00032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Xiong Y., Zhang S., Xu L., Song B., Huang G., Lu J., Guan S. Suppression of T-cell activation in vitro and in vivo by cordycepin from Cordyceps militaris. J. Surg. Res. 2013;185:912–922. doi: 10.1016/j.jss.2013.06.057. [DOI] [PubMed] [Google Scholar]
- 36.Yang X., Li Y., He Y., Li T., Wang W., Zhang J., Wei J., Deng Y., Lin R. Cordycepin alleviates airway hyperreactivity in a murine model of asthma by attenuating the inflammatory process. Int. Immunopharm. 2015;26:401–408. doi: 10.1016/j.intimp.2015.04.017. [DOI] [PubMed] [Google Scholar]
- 37.Yang R., Wang X., Xi D., Mo J., Wang K., Luo S., Wei J., Ren Z., Pang H., Luo Y. Cordycepin attenuates IFN-γ-induced macrophage IP-10 and mig expressions by inhibiting STAT1 activity in CFA-induced inflammation mice model. Inflammtion. 2020;43:752–764. doi: 10.1007/s10753-019-01162-3. [DOI] [PubMed] [Google Scholar]
- 38.Deng Q., Li X., Fang C., Li X., Zhang J., Xi Q., Li Y., Zhang R. Cordycepin enhances anti-tumor immunity in colon cancer by inhibiting phagocytosis immune checkpoint CD47 expression. Int. Immunopharmacol. 2022;107:108695. doi: 10.1016/j.intimp.2022.108695. [DOI] [PubMed] [Google Scholar]
- 39.Jeong M.H., Lee C.M., Lee S.W., Seo S.Y., Seo M.J., Kang B.W., Jeong Y.K., Choi Y.J., Yang K.M., Jo W.S. Cordycepin-enriched Cordyceps militaris induces immunomodulation and tumor growth delay in mouse-derived breast cancer. Oncol. Rep. 2013;30:1996–2002. doi: 10.3892/or.2013.2660. [DOI] [PubMed] [Google Scholar]
- 40.Lai X., Ning F., Yao Z., Wang T., Zhang L., Fang J., Ma J., Li G., Xu L., Guo Y., et al. Ethylene carbodiimide-fixed donor splenocytes combined with cordycepin induce long-term protection to mice cardiac allografts. Trans. Immunol. 2019;56:101196. doi: 10.1016/j.trim.2019.02.001. [DOI] [PubMed] [Google Scholar]
- 41.Ahn H.Y., Cho H.D., Cho Y.S. Antioxidant and antihyperlipidemic effects of cordycepin-rich Cordyceps militaris in a Sprague-Dawley rat model of alcohol-induced hyperlipidemia and oxidative stress. Bioresour. Bioprocess. 2020;7:104846. doi: 10.1186/s40643-020-00323-9. [DOI] [Google Scholar]
- 42.Xiao L., Ge Y., Sun L., Xu X., Xie P., Zhan M., Wang M., Dong Z., Li J., Duan S., et al. Cordycepin inhibits albumin-induced epithelial-mesenchymal transition of renal tubular epithelial cells by reducing reactive oxygen species production. Free Radic. Res. 2012;46:174–183. doi: 10.3109/10715762.2011.647688. [DOI] [PubMed] [Google Scholar]
- 43.Guo P., Kai Q., Gao J., Lian Z.Q., Wu C.M., Wu C.A., Zhu H.B. Cordycepin prevents hyperlipidemia in hamsters fed a high-fat diet via activation of AMP-activated protein kinase. J. Pharmacol. Sci. 2010;113:395–403. doi: 10.1254/jphs.10041FP. [DOI] [PubMed] [Google Scholar]
- 44.Wong Y.Y., Moon A., Duffin R., Barthet-Barateig A., Meijer H.A., Clemens M.J., de Moor C.H. Cordycepin inhibits protein synthesis and cell adhesion through effects on signal transduction. J. Biol. Chem. 2010;285:2610–2621. doi: 10.1074/jbc.M109.071159. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Nomani A.Z., Nabi Z., Rashid H., Janjua J., Nomani H., Majeed A., Chaudry S.R., Mazhar A.S. Osmotic nephrosis with mannitol: Review article. Ren. Fail. 2014;36:1169–1176. doi: 10.3109/0886022X.2014.926758. [DOI] [PubMed] [Google Scholar]
- 46.Sahmeddini M.A., Zahiri S., Khosravi M.B., Ghaffaripour S., Eghbal M.H., Shokrizadeh S. Effect of mannitol on postreperfusion cardiac output and central venous oxygen saturation during orthotopic liver transplant: A double-blind randomized clinical trial. Prog. Transplant. 2014;24:121–125. doi: 10.7182/pit2014483. [DOI] [PubMed] [Google Scholar]
- 47.Liu Y., Wang J., Wang W., Zhang H., Zhang X., Han C. The chemical constituents and pharmacological actions of Cordyceps sinensis. Evid. Based Complement. Altern. Med. 2015;2015:575063. doi: 10.1155/2015/575063. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Bi S., Huang W., Chen S., Huang C., Li C., Guo Z., Yang J., Zhu J., Song L., Yu R. Cordyceps militaris polysaccharide converts immunosuppressive macrophages into M1-like phenotype and activates T lymphocytes by inhibiting the PD-L1/PD-1 axis between TAMs and T lymphocytes. Int. J. Biol. Macromol. 2020;150:261–280. doi: 10.1016/j.ijbiomac.2020.02.050. [DOI] [PubMed] [Google Scholar]
- 49.Jayakumar T., Chiu C.C., Wang S.H., Chou D.S., Huang Y.K., Sheu J.R. Anti-cancer effects of CME-1, a novel polysaccharide, purified from the mycelia of Cordyceps sinensis against B16-F10 melanoma cells. J. Cancer Res. Ther. 2014;10:43–49. doi: 10.4103/0973-1482.131365. [DOI] [PubMed] [Google Scholar]
- 50.Lee J.S., Kwon J.S., Yun J.S., Pahk J.W., Shin W.C., Lee S.Y., Hong E.K. Structural characterization of immunostimulating polysaccharide from cultured mycelia of Cordyceps militaris. Carbohydr. Polym. 2010;80:1011–1017. doi: 10.1016/j.carbpol.2010.01.017. [DOI] [Google Scholar]
- 51.Qi W., Zhou X., Wang J., Zhang K., Zhou Y., Chen S., Nie S., Xie M. Cordyceps sinensis polysaccharide inhibits colon cancer cells growth by inducing apoptosis and autophagy flux blockage via mTOR signaling. Carbohydr. Polym. 2020;237:116113. doi: 10.1016/j.carbpol.2020.116113. [DOI] [PubMed] [Google Scholar]
- 52.Zhang W.Y., Yang J.Y., Chen J.P., Hou Y.Y., Han X.D. Immunomodulatory and antitumour effects of an exopolysaccharide fraction from cultivated Cordyceps sinensis (Chinese caterpillar fungus) on tumor-bearing mice. Biotechnol. Appl. Biochem. 2005;42:9–15. doi: 10.1042/BA20040183. [DOI] [PubMed] [Google Scholar]
- 53.Chen W.X., Zhang W.Y., Shen W.B., Wang K. Effects of the acid polysaccharide fraction isolated from a cultivated C. sinensis on macrophages in vitro. Cell. Immunol. 2010;262:69–74. doi: 10.1016/j.cellimm.2010.01.001. [DOI] [PubMed] [Google Scholar]
- 54.He B.L., Zheng Q.W., Guo L.Q., Huang J.Y., Yun F., Huang S.S., Lin J.F. Structural characterization and immune-enhancing activity of a novel high-molecular-weight polysaccharide from Cordyceps militaris. Int. J. Biol. Macromol. 2020;145:11–20. doi: 10.1016/j.ijbiomac.2019.12.115. [DOI] [PubMed] [Google Scholar]
- 55.Lee J.S., Kwon D.S., Lee K.R., Park J.M., Ha S.J., Hong E.K. Mechanism of macrophage activation induced by polysaccharide from Cordyceps militaris culture broth. Carbohydr. Polym. 2015;120:29–37. doi: 10.1016/j.carbpol.2014.11.059. [DOI] [PubMed] [Google Scholar]
- 56.Liu Y., Li Q.Z., Li L.D.J., Zhou X.W. Immunostimulatory effects of the intracellular polysaccharides isolated from liquid culture of Ophiocordyceps sinensis (Ascomycetes) on RAW264.7 cells via the MAPK and PI3K/Akt signaling pathways. J. Ethnopharmacol. 2021;275:114130. doi: 10.1016/j.jep.2021.114130. [DOI] [PubMed] [Google Scholar]
- 57.Song D., Lin J.Y., Yuan F.J., Zhang W.Y. Ex vivo stimulation of murine dendritic cells by an exopolysaccharide from one of the anamorph of C. sinensis. Cell Biochem. Funct. 2011;29:555–561. doi: 10.1002/cbf.1787. [DOI] [PubMed] [Google Scholar]
- 58.Sheng L., Chen J., Li J., Zhang W. An exopolysaccharide from cultivated Cordyceps sinensis and its effects on cytokine expressions of immunocytes. Appl. Microbiol. Biotechnol. 2011;163:669–678. doi: 10.1007/s12010-010-9072-3. [DOI] [PubMed] [Google Scholar]
- 59.Wang J., Nie S., Cui S.W., Wang Z., Phillips A.O., Phillips G.O., Li Y., Xie M. Structural characterization and immunostimulatory activity of a glucan from natural Cordyceps sinensis. Food Hydrocoll. 2017;67:139–147. doi: 10.1016/j.foodhyd.2017.01.010. [DOI] [Google Scholar]
- 60.Wang M., Meng X., Yang R., Qin T., Li Y., Zhang L., Hu Y. Cordyceps militaris polysaccharides can improve the immune efficacy of Newcastle disease vaccine in chicken. Int. J. Biol. Macromol. 2013;59:178–183. doi: 10.1016/j.ijbiomac.2013.04.007. [DOI] [PubMed] [Google Scholar]
- 61.Zhang J., Yu Y., Zhang Z., Ding Y., Dai X., Li Y. Effect of polysaccharide from cultured Cordyceps sinensis on immune function and anti-oxidation activity of mice exposed to 60Co. Int. Immunopharmacol. 2011;11:2251–2257. doi: 10.1016/j.intimp.2011.09.019. [DOI] [PubMed] [Google Scholar]
- 62.Li C.Y., Chiang C.S., Tsai M.L., Hseu R.S., Shu W.Y., Chuang C.Y., Sun Y.C., Chang Y.S., Lin J.G., Chen C.S., et al. Two-sided effect of Cordyceps sinensis on dendritic cells in different physiological stages. J. Leukocyte Biol. 2009;85:987–995. doi: 10.1189/jlb.0908573. [DOI] [PubMed] [Google Scholar]
- 63.Yu R., Song L., Zhao Y., Bin W., Wang L., Zhang H., Wu Y., Ye W., Yao X. Isolation and biological properties of polysaccharide CPS-1 from cultured Cordyceps militaris. Fitoterapia. 2004;75:465–472. doi: 10.1016/j.fitote.2004.04.003. [DOI] [PubMed] [Google Scholar]
- 64.Lin R., Liu H., Wu S., Pang L., Jia M., Fan K., Jia L. Production and in vitro antioxidant activity of exopolysaccharide by a mutant, Cordyceps militaris SU5-08. Int. J. Biol. Macromol. 2012;51:153–157. doi: 10.1016/j.ijbiomac.2012.04.011. [DOI] [PubMed] [Google Scholar]
- 65.Wang J., Nie S., Kan L., Chen H., Cui S.W., Phillips A.O., Phillips G.O., Xie M. Comparison of structural features and antioxidant activity of polysaccharides from natural and cultured Cordyceps sinensis. Food Sci. Biotechnol. 2017;26:55–62. doi: 10.1007/s10068-017-0008-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Ohta Y., Lee J.B., Hayashi K., Fujita A., Park D.K., Hayashi T. In vivo anti-influenza virus activity of an immunomodulatory acidic polysaccharide isolated from Cordyceps militaris grown on germinated soybeans. J. Agric. Food Chem. 2007;55:10194–10199. doi: 10.1021/jf0721287. [DOI] [PubMed] [Google Scholar]
- 67.Wang Y., Liu D., Zhao H., Jiang H., Luo C., Wang M., Yin H. Cordyceps sinensis polysaccha-ride CPS-2 protects human mesangial cells from PDGF-BB-induced proliferation through the PDGF/ERK and TGF-β1/Smad pathways. Mol. Cell. Endocrinol. 2014;382:979–988. doi: 10.1016/j.mce.2013.11.018. [DOI] [PubMed] [Google Scholar]
- 68.Wang Y., Wang M., Ling Y., Fan W., Wang Y., Yin H. Structural determination and antioxidant activity of a polysaccharide from the fruiting bodies of cultured Cordyceps sinensis. Am. J. Chin. Med. 2009;37:977–989. doi: 10.1142/S0192415X09007387. [DOI] [PubMed] [Google Scholar]
- 69.Li S.P., Zhang G.H., Zeng Q., Huang Z.G., Wang Y.T., Dong T.T., Tsim K.W. Hypoglycemic activity of polysaccharide, with antioxidation, isolated from cultured Cordyceps mycelia. Phytomedicine. 2006;13:428–433. doi: 10.1016/j.phymed.2005.02.002. [DOI] [PubMed] [Google Scholar]
- 70.Qian G.M., Pan G.F., Guo J.Y. Anti-inflammatory and antinociceptive effects of cordymin, a peptide purified from the medicinal mushroom Cordyceps sinensis. Nat. Prod. Res. 2012;26:2358–2362. doi: 10.1080/14786419.2012.658800. [DOI] [PubMed] [Google Scholar]
- 71.Wang J., Liu Y.M., Cao W., Yao K.W., Liu Z.Q., Guo J.Y. Anti-inflammation and antioxidant effect of cordymin, a peptide purified from the medicinal mushroom Cordyceps sinensis, in middle cerebral artery occlusion-induced focal cerebral ischemia in rats. Metab. Brain Dis. 2012;27:159–165. doi: 10.1007/s11011-012-9282-1. [DOI] [PubMed] [Google Scholar]
- 72.Qi W., Zhang Y., Yan Y.B., Lei W., Wu Z.X., Liu N., Liu S., Shi L., Fan Y. The protective effect of cordymin, a peptide purified from the medicinal mushroom Cordyceps sinensis, on diabetic osteopenia in alloxan-induced diabetic rats. Evid. Based Complement. Altern. Med. 2013;2013:985636. doi: 10.1155/2013/985636. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Jia J.M., Ma X.C., Wu C.F., Wu L.J., Hu G.S. Cordycedipeptide A, a new cyclodipeptide from the culture liquid of Cordyceps sinensis (BERK.) SACC. Chem. Pharmaceut. Bull. 2005;53:582–583. doi: 10.1248/cpb.53.582. [DOI] [PubMed] [Google Scholar]
- 74.Zhang S.S., Zhang D.S., Zhu T.J., Chen X.Y. A pharmacological analysis of the amino acid components of Cordyceps sinensis Sacc. Yao Xue Xue Bao. 1991;26:326–330. (In Chinese) [PubMed] [Google Scholar]
- 75.Choi D., Cha W.S., Park N., Kim H.W., Lee J.H., Park J.S., Park S.S. Purification and characterization of a novel fibrinolytic enzyme from fruiting bodies of Korean Cordyceps militaris. Bioresour. Technol. 2011;102:3279–3285. doi: 10.1016/j.biortech.2010.10.002. [DOI] [PubMed] [Google Scholar]
- 76.Cui L., Dong M.S., Chen X.H., Jiang M., Lv X., Yan G. A novel fibrinolytic enzyme from Cordyceps militaris, a Chinese traditional medicinal mushroom. World J. Microbiol. Biotechnol. 2008;24:483–489. doi: 10.1007/s11274-007-9497-1. [DOI] [Google Scholar]
- 77.Matsuda H., Akaki J., Nakamura S., Okazaki Y., Kojima H., Tamesada M., Yoshikawa M. Apoptosis-inducing effects of sterols from the dried powder of cultured mycelium of Cordyceps sinensis. Chem. Pharm. Bull. 2009;57:411–414. doi: 10.1248/cpb.57.411. [DOI] [PubMed] [Google Scholar]
- 78.Sun X., Feng X., Zheng D., Li A., Li C., Li S., Zhao Z. Ergosterol attenuates cigarette smoke extract-induced COPD by modulating inflammation, oxidative stress and apoptosis In Vitro and In Vivo. Clin. Sci. 2019;133:1523–1536. doi: 10.1042/CS20190331. [DOI] [PubMed] [Google Scholar]
- 79.Peng Y., Tao Y., Wang Q., Shen L., Yang T., Liu Z., Liu C. Ergosterol is the active compound of cultured mycelium Cordyceps sinensis on antiliver fibrosis. Evid. Based Complement. Altern. Med. 2014;2014:537234. doi: 10.1155/2014/537234. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Li S.P., Yang F.Q., Tsim K.W.K. Quality control of Cordyceps sinensis, a valued traditional Chinese medicine. J. Pharm. Biomed. Anal. 2006;41:1571–1584. doi: 10.1016/j.jpba.2006.01.046. [DOI] [PubMed] [Google Scholar]
- 81.Wu J., Zhang Q., Leung P. Inhibitory effects of ethyl acetate extract of Cordyceps sinensis mycelium on various cancer cells in culture and B16 melanoma in C57BL/6 mice. Phytomedicines. 2007;14:43–49. doi: 10.1016/j.phymed.2005.11.005. [DOI] [PubMed] [Google Scholar]
- 82.Yang L.Y., Huang W.J., Hsieh H.G., Lin C.Y. H1-A extracted from Cordyceps sinensis, suppresses the proliferation of human mesangial cells and promotes apoptosis, probably by inhibiting the tyrosine phosphorylation of Bcl-2 and Bcl-XL. J. Lab. Clin. Med. 2003;141:74–83. doi: 10.1067/mlc.2003.6. [DOI] [PubMed] [Google Scholar]
- 83.Yang M.L., Kuo P.C., Hwang T.L., Wu T.S. Anti-inflammatory principles from Cordyceps sinensis. J. Nat. Prod. 2011;74:1996–2000. doi: 10.1021/np100902f. [DOI] [PubMed] [Google Scholar]
- 84.Shashidhar M.G., Giridhar P., Sankar K.U., Manohar B. Bioactive principles from Cordyceps sinensis: A potent food supplement—A review. J. Funct. Foods. 2013;5:1013–1030. doi: 10.1016/j.jff.2013.04.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Zhou Y.J., Wang M., Zhang H., Huang Z., Ma J. Comparative study of the composition of cultivated, naturally grown Cordyceps sinensis, and stiff worms across different sampling years. PLoS ONE. 2019;14:e0225750. doi: 10.1371/journal.pone.0225750. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Li J., Feng C.Q., Ni X.M., Zhang W.S. Determination of nucleosides of natural Cordyceps sinensis in Qinghai province by capillary electrophoresis. Chin. Pharm. J. 2008;43:1105–1107. doi: 10.1016/S1872-2075(08)60042-4. (In Chinese) [DOI] [Google Scholar]
- 87.Ikeda R., Nishimura M., Sun Y., Wada M., Nakashima K. Simple HPLC-UV determination of nucleosides and its application to the authentication of Cordyceps and its allies. Biomed. Chromatogr. 2008;22:630–636. doi: 10.1002/bmc.980. [DOI] [PubMed] [Google Scholar]
- 88.Yang F.Q., Ge L., Yong J.W., Tan S.N., Li S.P. Determination of nucleosides and nucleobases in different species of Cordyceps by capillary electrophoresis-mass spectrometry. J. Pharm. Biomed. 2009;50:307–314. doi: 10.1016/j.jpba.2009.04.027. [DOI] [PubMed] [Google Scholar]
- 89.Yuan Y.S., Zhang L., Xu X.F., Zhou Y.X., Wei L.X. Determination of nucleosides in Cordyceps by RP-HPLC. Chin. Pharm. J. 2002;37:776–778. doi: 10.1016/j.fct.2008.08.005. [DOI] [Google Scholar]
- 90.Vannucci L., Krizan J., Sima P., Stakheev D., Caja F., Rajsiglova L., Horak V., Saieh M. Immunostimulatory properties and antitumor activities of glucans (review) Int. J. Oncol. 2013;43:357–364. doi: 10.3892/ijo.2013.1974. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Yan J.K., Wang W.Q., Li L., Wu J. Recent advances in Cordyceps sinensis polysaccharides: Mycelial fermentation, isolation, structure, and bioactivities: A review. J. Funct. Foods. 2014;6:33–47. doi: 10.1016/j.jff.2013.11.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Xu J., Huang Y., Chen X.X., Zheng S.C., Chen P., Mo M.H. The mechanisms of pharmacological activities of Ophiocordyceps sinensis fungi. Phytother. Res. 2016;30:1572–1583. doi: 10.1002/ptr.5673. [DOI] [PubMed] [Google Scholar]
- 93.Wang Z.M., Peng X., Lee K.L., Tang J.C., Cheung P.C., Wu J.Y. Structural characterization and immunomodulatory property of an acidic polysaccharide from mycelial culture of Cordyceps sinensis fungus Cs-HK1. Food Chem. 2011;125:637–643. doi: 10.1016/j.foodchem.2010.09.052. [DOI] [Google Scholar]
- 94.Lee S.J., Cho J.Y., Hong E.K. Study on macrophage activation and structural characteristics of purified polysaccharide from the liquid culture broth of Cordyceps militaris. Carbohydr. Polym. 2010;82:982–988. doi: 10.1016/j.carbpol.2010.06.025. [DOI] [Google Scholar]
- 95.Bi S., Jing Y., Zhou Q., Hu X., Zhu J., Guo Z., Song L., Yu R. Structural elucidation and immunostimulatory activity of a new polysaccharide from Cordyceps militaris. Food Funct. 2018;9:279–293. doi: 10.1039/C7FO01147D. [DOI] [PubMed] [Google Scholar]
- 96.He L., Ji P., Cheng J., Wang Y., Qian H., Li W., Gong X., Wang Z. Structural characterization and immunostimulatory activity of a novel protein-bound polysaccharide produced by Hirsutella sinensis Liu, Guo, Yu & Zeng. Food Chem. 2013;141:946–953. doi: 10.1016/j.foodchem.2013.04.053. [DOI] [PubMed] [Google Scholar]
- 97.Yu R.M., Yang W., Song L., Yan C., Zhang Z., Zhao Y. Structural characterization and antioxidant activity of a polysaccharide from the fruiting bodies of cultured Cordyceps militaris. Carbohydr. Polym. 2007;70:430–436. doi: 10.1016/j.carbpol.2007.05.005. [DOI] [Google Scholar]
- 98.Yan J.K., Wang W.Q., Li L., Wu J.Y. Physiochemical properties and antitumor activities of two α-glucans isolated from hot water and alkaline extracts of Cordyceps (Cs-HK1) fungal mycelia. Carbohydr. Polym. 2011;85:753–758. doi: 10.1016/j.carbpol.2011.03.043. [DOI] [Google Scholar]
- 99.Zigler M., Shir A., Levitzki A. Targeted cancer immunotherapy. Curr. Opin. Pharmacol. 2013;13:504–510. doi: 10.1016/j.coph.2013.04.003. [DOI] [PubMed] [Google Scholar]
- 100.Xu L., Feng J.M., Li J.X., Zhu J.M., Song S.S., Tong L.J., Chen Y., Yang X.Y., Shen Y.Y., Lian F.L., et al. Tanshinone-1 induces tumor cell killing, enhanced by inhibition of secondary activation of signaling networks. Cell Death Dis. 2013;4:e905. doi: 10.1038/cddis.2013.443. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Jia Y., Guan Q., Guo Y., Du C. Reduction of inflammatory hyperplasia in the intestine in colon cancer-prone mice by water-extract of Cistanche deserticola. Phytother. Res. 2012;26:812–819. doi: 10.1002/ptr.3637. [DOI] [PubMed] [Google Scholar]
- 102.Li J., Cai H., Sun H., Qu J., Zhao B., Hu X., Li W., Qian Z., Yu X., Kang F., et al. Extracts of Cordyceps sinensis inhibit breast cancer growth through promoting M1 macrophage polarization via NF-κB pathway activation. J. Ethnopharmacol. 2020;260:112969. doi: 10.1016/j.jep.2020.112969. [DOI] [PubMed] [Google Scholar]
- 103.Chen W., Yuan F., Wang K., Song D., Zhang W. Modulatory effects of the acid polysaccharide fraction from one of anamorph of Cordyceps sinensis on Ana-1 cells. J. Ethnopharmacol. 2012;142:739–745. doi: 10.1016/j.jep.2012.05.055. [DOI] [PubMed] [Google Scholar]
- 104.Guo Q., Li J., Lin H. Effect and molecular mechanisms of traditional Chinese medicine on regulating tumor immunosuppressive microenvironment. Biomed. Res. Int. 2015;2015:261620. doi: 10.1155/2015/261620. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Liu M., Luo F., Ding C., Albeituni S., Hu X., Ma Y., Cai Y., Mcnally L., Sanders M.A., Jain D., et al. Dectin-1 activation by a natural product β-glucan converts immunosuppressive macrophages into an M1-like phenotype. J. Immunol. 2015;195:5055–5065. doi: 10.4049/jimmunol.1501158. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Ji Y., Sun S., Xu A., Bhargava P., Yang L., Lam K.S.L., Gao B., Lee C.H., Kersten S., Qi L. Activation of natural killer T cells promotes M2 macrophage polarization in adipose tissue and improves systemic glucose tolerance via interleukin-4 (IL-4)/STAT6 protein signaling axis in obesity. J. Biol. Chem. 2012;287:13561–13571. doi: 10.1074/jbc.M112.350066. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Yuan F., Fu X., Shi H., Chen G., Dong P., Zhang W. Induction of murine macrophage M2 polarization by cigarette smoke extract via the JAK2/STAT3 pathway. PLoS ONE. 2014;9:e107063. doi: 10.1371/journal.pone.0107063. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Isidro R.A., Appleyard C.B. Colonic macrophage polarization in homeostasis, inflammation, and cancer. Am. J. Physiol. Gastrointest. Liver Physiol. 2016;311:G59–G73. doi: 10.1152/ajpgi.00123.2016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Tariq M., Zhang J., Liang G., Ding L., He Q., Yang B. Macrophage polarization: Anti-cancer strategies to target tumor-associated macrophage in breast cancer. J. Cell. Biochem. 2017;118:2484–2501. doi: 10.1002/jcb.25895. [DOI] [PubMed] [Google Scholar]
- 110.Chen Y., Yang S.H., Hueng D.Y., Syu J.P., Liao C.C., Wu Y.C. Cordycepin induces apoptosis of C6 glioma cells through the adenosine 2A receptor-p53-caspase-7-PARP pathway. Chem. Biol. Interact. 2014;216:17–25. doi: 10.1016/j.cbi.2014.03.010. [DOI] [PubMed] [Google Scholar]
- 111.Tania M., Shawon J., Saif K., Kiefer R., Khorram M.S., Halim M.A., Khan M.A. Cordycepin Downregulates Cdk-2 to Interfere with Cell Cycle and Increases Apoptosis by Generating ROS in Cervical Cancer Cells: In vitro and in silico Study. Curr. Cancer Drug Targets. 2019;19:152–159. doi: 10.2174/1568009618666180905095356. [DOI] [PubMed] [Google Scholar]
- 112.Xu J., Tan Z.C., Shen Z.Y., Shen X.J., Tang S.M. Cordyceps cicadae polysaccharides inhibit human cervical cancer hela cells proliferation via apoptosis and cell cycle arrest. Food Chem. Toxicol. 2021;148:111971. doi: 10.1016/j.fct.2021.111971. [DOI] [PubMed] [Google Scholar]
- 113.Lee W.H., Lee S., Lee K., Shin Y.S., Kang H., Cho H. Anti-cancer effect of Cordyceps militaris in human colorectal carcinoma RKO cells via cell cycle arrest and mitochondrial apoptosis. DARU J. Pharm. Sci. 2015;23:35. doi: 10.1186/s40199-015-0117-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Lee H.H., Jeong J.W., Choi Y.H. Inhibition of PI3K/AKT signaling pathway enhances cordycepin-induced apoptosis in human gastric cancer cells. J. Korean Soc. Food Sci. Nutr. 2016;45:835–842. doi: 10.3746/jkfn.2016.45.6.835. [DOI] [Google Scholar]
- 115.Xiao J.H., Chen D.X., Fang N., Liu Z.L., Zhang T. Growth arrest of human gastric adenocarcinoma cells by bioactive compounds of Cordyceps jiangxiensis (CaoMuWang) through induction of apoptosis. J. Food Agric. Environ. 2006;4:66–73. [Google Scholar]
- 116.Li Z., Guo Z., Zhu J., Bi S., Luo Y., Yu R., Huang W., Song L. Cordyceps militaris fraction inhibits angiogenesis of hepatocellular carcinoma in vitro and in vivo. Pharmacogn. Mag. 2020;16:169–176. doi: 10.4103/pm.pm_347_19. [DOI] [Google Scholar]
- 117.Huo X., Liu C., Bai X., Li W., Li J., Hu X., Cao L. Aqueous extract of Cordyceps sinensis potentiates the antitumor effect of DDP and attenuates therapy-associated toxicity in non-small cell lung cancer via IkBa/NFkB and AKT/MMP2/MMP9 pathways. RSC Adv. 2017;66:37743–37754. doi: 10.1039/C7RA04716A. [DOI] [Google Scholar]
- 118.Yao L.S., Li Y., He W., Yi K.S., Huang M. Polysaccharide of Cordyceps sinensis Enhances Cisplatin Cytotoxicity in Non–Small Cell Lung Cancer H157 Cell Line. Integr. Cancer Ther. 2011;10:359–367. doi: 10.1177/1534735410392. [DOI] [PubMed] [Google Scholar]
- 119.Bizarro A., Ferreira I.C.F.R., Soković M., Van G., Leo J.L.D., Sousa D., Vasconcelos M.H., Lima R.T. Cordyceps militaris (L.) Link Fruiting Body Reduces the Growth of a Non-Small Cell Lung Cancer Cell Line by Increasing Cellular Levels of p53 and p21. Molecules. 2015;20:13927–13940. doi: 10.3390/molecules200813927. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Yang J., Zhang W., Shi P., Chen J., Han X., Wang Y. Effects of exopolysaccharide fraction (EPSF) from a cultivated Cordyceps sinensis fungus on c-Myc, c-Fos, and VEGF expression in B16 melanoma-bearing mice. Pathol. Res. Pract. 2005;201:745–750. doi: 10.1016/j.prp.2005.08.007. [DOI] [PubMed] [Google Scholar]
- 121.Hsu P.Y., Lin Y.H., Yeh E.L., Lo H.C., Hsu T.H., Su C.C. Cordycepin and a preparation from Cordyceps militaris inhibit malignant transformation and proliferation by decreasing EGFR and IL-17RA signaling in a murine oral cancer model. Oncotarget. 2017;8:93712–93728. doi: 10.18632/oncotarget.21477. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Lin L.T., Lai Y.J., Wu S.C., Hsu W.H., Tai C.J. Optimal conditions for cordycepin production in surface liquid-cultured Cordyceps militaris treated with porcine liver extracts for suppression of oral cancer. J. Food Drug Anal. 2018;26:135–144. doi: 10.1016/j.jfda.2016.11.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Jo E., Jang H.J., Yang K.E., Jang M.S., Huh Y.H., Yoo H.S., Park J.S., Jang I.S., Park S.J. Cordyceps militaris induces apoptosis in ovarian cancer cells through TNF-α/TNFR1-mediated inhibition of NF-κB phosphorylation. BMC Complement. Med. Ther. 2020;20:1. doi: 10.1186/s12906-019-2780-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Yoon S.Y., Lindroth A.M., Kwon S., Park S.J., Par Y.J. Adenosine derivatives from Cordyceps exert antitumor effects against ovarian cancer cells through ENT1-mediated transport, induction of AMPK signaling, and consequent autophagic cell death. Biomed. Pharmcother. 2022;153:113491. doi: 10.1016/j.biopha.2022.113491. [DOI] [PubMed] [Google Scholar]
- 125.Algeciras-Schimnich A., Shen L., Barnhart B.C. Molecular ordering of the initial signaling events of CD95. Mol. Cell. Biol. 2002;22:207–220. doi: 10.1128/MCB.22.1.207-220.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Villa-Morales M., Fernandez-Piqueras J. Targeting the Fas FasL signaling pathway in cancer therapy. Expert Opin. Ther. Targets. 2012;16:85–101. doi: 10.1517/14728222.2011.628937. [DOI] [PubMed] [Google Scholar]
- 127.Lee S.Y., Debnath T., Kim S.K., Lim B.O. Anti-cancer effect and apoptosis induction of cordycepin through DR3 pathway in the human colonic cancer cell HT-29. Food Chem. Toxicol. 2013;60:439–447. doi: 10.1016/j.fct.2013.07.068. [DOI] [PubMed] [Google Scholar]
- 128.Ashkenazi A., Dixit V.M. Death receptors: Signaling and modulation. Science. 1998;281:1305–1308. doi: 10.1126/science.281.5381.1305. [DOI] [PubMed] [Google Scholar]
- 129.Gao L., Abu K.Y. Hijacking of apoptotic pathways by bacterial pathogens. Microbes Infect. 2002;2:1705–1719. doi: 10.1016/S1286-4579(00)01326-5. [DOI] [PubMed] [Google Scholar]
- 130.Detjen K.M., Farwig K., Welzel M., Wiedenmann B., Rosewicz S. Interferon gamma inhibits growth of human pancreatic carcinoma cells via caspase-1 dependent induction of apoptosis. Gut. 2001;49:251–262. doi: 10.1136/gut.49.2.251. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Lee H.H., Kim S.O., Kim G.Y., Moon S.K., Kim W.J., Jeong Y.K., Yoo Y.H., Choi Y.H. Involvement of autophagy in cordycepin-induced apoptosis in human prostate carcinoma LNCaP cells. Environ. Toxicol. Pharmacol. 2014;38:239–250. doi: 10.1016/j.etap.2014.06.003. [DOI] [PubMed] [Google Scholar]
- 132.Balk J.S., Mun S.W., Kim K.S., Park S.J., Yoon H.K., Kim D.H., Park M.K., Kim C.H., Lee Y.C. Apoptotic effects of cordycepin through the extrinsic pathway and p38 MAPK activation in human glioblastoma U87MG cells. J. Microbiol. Biotechnol. 2016;26:309–314. doi: 10.4014/jmb.1507.07090. [DOI] [PubMed] [Google Scholar]
- 133.Cao H.L., Liu Z.J., Chang Z. Cordycepin induces apoptosis in human bladder cancer cells via activation of A3 adenosine receptors. Tumor Biol. 2017;39:7. doi: 10.1177/1010428317706915. [DOI] [PubMed] [Google Scholar]
- 134.Marino G., Niso-Santano M., Baehrecke E.H., Guido K. Self-consumption: The interplay of autophagy and apoptosis. Nat. Rev. Mol. Cell Biol. 2014;15:81–94. doi: 10.1038/nrm3735. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Lee S.J., Kim S.K., Choi W.S., Kim W.J., Moon S.K. Cordycepin causes p21WAF1-mediated G2/M cell-cycle arrest by regulating c-Jun N-terminal kinase activation in human bladder cancer cells. Arch. Biochem. Biophys. 2009;490:103–109. doi: 10.1016/j.abb.2009.09.001. [DOI] [PubMed] [Google Scholar]
- 136.Holbein S., Wengi A., Decourty L., Freimoser F.M., Jacquier A., Dichtl B. Cordycepin interferes with 3′ end formation in yeast independently of its potential to terminate RNA chain elongation. RNA. 2009;15:837–849. doi: 10.1261/rna.1458909. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Xie H., Li X., Chen Y., Lang M., Shen Z., Shi L. Ethanolic extract of Cordyceps cicadae exerts antitumor effect on human gastric cancer SGC-7901 cells by inducing apoptosis, cell cycle arrest and endoplasmic reticulum stress. J. Ethnopharmacol. 2019;231:230–240. doi: 10.1016/j.jep.2018.11.028. [DOI] [PubMed] [Google Scholar]
- 138.Hunter K.W., Crawford N.P.S., Alsarraj J. Mechanisms of metastasis. Breast Cancer Res. 2008;10:S2. doi: 10.1186/bcr1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Tao X., Ning Y., Zhao X., Pan T. The effects of cordycepin on the cell proliferation, migration and apoptosis in human lung cancer cell lines A549 and NCI-H460. J. Pharm. Pharmacol. 2016;68:901–911. doi: 10.1111/jphp.12544. [DOI] [PubMed] [Google Scholar]
- 140.Nakamura K., Shinozuka K., Yoshikawa N. Anticancer and antimetastatic effects of cordycepin, an active component of Cordyceps sinensis. J. Pharm. Sci. 2015;127:53–56. doi: 10.1016/j.jphs.2014.09.001. [DOI] [PubMed] [Google Scholar]
- 141.Phuchareon J., Tokuhisa T. Deregulated c-Fos/AP-1 modulates expression of the cyclin and the cdk gene in splenic B cells stimulated with lipopolysaccharide. Cancer Lett. 1995;92:203–208. doi: 10.1016/0304-3835(95)03780-Z. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Not applicable.