Abstract
Background
People living with HIV (PLWH) are at increased risk of developing cancer. Cancer diagnoses are often incompletely captured at antiretroviral therapy (ART) clinics.
Aim
To estimate the incidence and explore risk factors of cancer in a cohort of PLWH in Harare using probabilistic record linkage (PRL).
Methods
We conducted a retrospective cohort study that included PLWH aged ≥16 years starting ART between 2004 and 2017. We used PRL to match records from the Zimbabwe National Cancer Registry (ZNCR) with electronic medical records from an ART clinic in Harare to investigate the incidence of cancer among PLWH initiating ART. We matched records based on demographic data followed by manual clerical review. We followed PLWH up until first cancer diagnosis, death, loss to follow‐up, or 31 December 2017, whichever came first.
Results
We included 3442 PLWH (64.9% female) with 19 346 person‐years (PY) of follow‐up. Median CD4 count at ART initiation was 169 cells/mm3 (interquartile range [IQR]: 82–275), median age was 36.6 years (IQR: 30.6–43.4). There were 66 incident cancer cases for an overall incidence rate of 341/100 000 PY (95% confidence interval [CI]: 268–434). Twenty‐two of these cases were recorded in the ZNCR only. The most common cancers were cervical cancer (n = 16; 123/100 000 PY; 95% CI: 75–201), Kaposi sarcoma, and lymphoma (both n = 12; 62/100 000 PY; 95% CI: 35–109). Cancer incidence increased with age and decreased with higher CD4 cell counts at ART initiation.
Conclusion
PRL was key to correct for cancer under‐ascertainment in this cohort. The most common cancers were infection‐related types, reinforcing the role of early HIV treatment, human papillomavirus vaccination, and cervical cancer screening for cancer prevention in this setting.
Keywords: cancer, HIV, incidence, record linkage, Zimbabwe
Abbreviations
- aHR
adjusted hazard ratio
- AIDS
acquired immune deficiency syndrome
- ART
antiretroviral therapy
- CI
confidence interval
- HIV
human immunodeficiency virus
- HPV
human papillomavirus
- HR
hazard ratio
- IeDEA
International Epidemiology Database to Evaluate AIDS
- IQR
interquartile range
- MRCZ
Medical Research Council of Zimbabwe
- PLWH
people living with HIV
- PY
person‐years
- RL
record linkage
- USA
United States of America
- WHO
World Health Organization
- ZNCR
Zimbabwe National Cancer Registry
1. INTRODUCTION
People living with HIV (PLWH) are at increased risk of developing cancer. 1 , 2 Incidence rates of AIDS‐defining cancers such as Kaposi sarcoma and non‐Hodgkin lymphoma have been reduced by widespread access to antiretroviral therapy (ART), but late presentation for ART initiation is still a challenge despite current guidelines promoting a test‐and‐treat approach to ART initiation. 3 , 4 , 5 PLWH who initiate ART at CD4 counts less than 350 cells/mm3 or with an AIDS defining condition are known to be at increased risk of developing cancer. 6 Furthermore, HIV infection diminishes the ability to clear human papillomavirus (HPV) infection, which increases the risk of HPV related cancers, such as cervical cancer, among PLWH. 7
The impact of HIV on cancer risk in sub‐Saharan Africa is still poorly understood. 8 Reporting of cancer incidence among PLWH is often hampered by the absence of HIV serostatus information in national cancer databases, and cancer diagnoses are often incompletely captured at ART clinics. 9 Record linkage methods have been widely used in high‐income countries but are not commonly used in low‐ to middle‐income countries due to low uptake of electronic health records systems by health institutions. 9 , 10 , 11 Probabilistic record linkage (PRL), a method of linking records from different systems in the absence of a common unique identifier, have been used to improve under ascertainment of cancer incidence in sub‐Saharan Africa. 9 , 10 , 12
In the current study, we estimated the incidence of cancer in a cohort of PLWH in Harare using PRL of records from the Zimbabwe National Cancer Registry (ZNCR) and an ART clinic, and explored risk factors for incident cancer in this population.
2. METHODS
We conducted a retrospective cohort study using datasets from the ZNCR and Newlands Clinic.
2.1. Study setting
The ZNCR is a population‐based registry for Harare. It is operated by the Ministry of Health and Child Care in Zimbabwe with the support of the International Agency for Research on Cancer. The ZNCR employs a combination of active and passive methods of case finding, with staff visiting institutions within the healthcare delivery system of Harare that are involved in the diagnosis and management of cancer patients to register cases. Death certificates of cancer patients who die in the greater Harare area are also used to identify cancer cases that may have been missed before death. 13 A detailed description of the ZNCR operations is available elsewhere. 14
The International epidemiology Databases to Evaluate AIDS Southern Africa (IeDEA) is an international research consortium of HIV observational databases, and the Southern African region (IeDEA‐SA, www.iedea-sa.org) includes ART programs located in seven countries (Botswana, Lesotho, Malawi, Zambia, Mozambique, Zimbabwe, and Republic of South Africa). 15 , 16 In this study, we used data from Newlands Clinic, one of the IeDEA‐SA sites situated in Harare, Zimbabwe. Newlands Clinic is a referral HIV management facility founded in 2004. It is a private voluntary organization that provides comprehensive HIV care to PLWH predominantly from the Greater Harare area, and is in a Public‐Private‐Partnership with the Ministry of Health and Child Care. It is operated by the Ruedi Luethy Foundation, a Swiss based charitable organization. Newlands Clinic's model of care comprises ART management, laboratory monitoring, mental and social health support, as well as sexual reproductive health (SRH) services. Under SRH services women who are sexually active are screened for cervical cancer. At the time of the study, cervical cancer screening was conducted using visual inspection with acetic acid (VIA). Women with a positive VIA screen were treated with cryotherapy or booked for a loop electrosurgical excision procedure (LEEP). Further details of Newlands Clinic's operations are available in a previous publication. 17
2.2. Data collection and pre‐processing
We included all records available in the ZNCR and Newlands Clinic databases. The ZNCR database contained records up to December 2017. The ZNCR dataset stored in CanReg4 software (International Association of Cancer Registries, http://www.iacr.com.fr/) and Newlands Clinic's electronic medical records were exported and uploaded into KNIME Analytics Platform (Version 3.7.2, Build 18 April 2019). Pre‐processing of data was conducted using KNIME workflows built using core and custom KNIME nodes. Pre‐processing involved standardization of all strings to lower‐case, removal of special characters (other than alphabet characters and digits), stripping off white spaces and transformation of numerical values to strings. We used names, dates of birth, and national identification (ID) numbers, where available, for PRL.
2.3. Deduplication and record linkage
We used PRL to identify duplicate entries of the same patient in each of the two datasets. This involved linking each dataset to itself by comparing each record to other records in the same dataset using first names, middle names, last names, national identification numbers, year of birth, month of birth, and day of birth. There were no additional variables common to both databases that could be used for PRL. We compared names as complete texts as well as n‐grams. The variables used for record linkage were assessed for completeness. However, we added all records available into the deduplication and record linkage workflows regardless of their completeness. We used probability scores that were computed based on matching variables to classify records into matches, probable matches, and mismatches. We considered match pairs with a score of less than 12 as mismatches and those with scores >25 as definite matches.
We then linked deduplicated datasets from the ZNCR and Newlands Clinic to each other using the same variables. Records classified as matches or probable matches underwent another clerical review process. We used KNIME software for deduplication and PRL. Details of the record linkage parameters are shown in Appendix S1. Records were anonymized after PRL.
2.4. Statistical analysis and definitions
We used frequencies to describe patient characteristics and the spectrum of incident cancers diagnosed in PLWH starting ART between 2004 and 2017. We described continuous variables using medians and interquartile ranges (IQRs). We defined incident cancers as new cancer cases diagnosed more than 30 days after initiating ART. We categorized cancers that are typically associated with Group 1 infectious agents (classified as carcinogenic to humans by the International Agency for Research on Cancer) as infection related. 18
For the analysis of cancer incidence, we included all patients aged ≥16 years, who started ART at Newlands Clinic between 2004 and 2017. We excluded patients diagnosed with cancer before ART initiation or within 30 days after (considered as prevalent cancer cases), and all patients with follow‐up time of <30 days from ART initiation. We calculated person‐years (PY) at risk from 30 days after ART initiation until the date of first cancer diagnosis, death, 6 months after the last visit for patients who did not die, or 31 December 2017, whichever occurred first. We present crude cancer incidence rates per 100 000 PY with 95% confidence intervals (CI). The confidence intervals were calculated by quadratic approximation to the Poisson log likelihood for the log‐rate parameter. 19
We used univariable and multivariable Cox proportional hazards models to assess the following potential risk factors (exposure variables) for developing cancer: sex, age at ART initiation (continuous variable), ART initiation period, education, employment status, CD4 count category at ART initiation (<100, 100–199, ≥200 cells/mm3) and WHO clinical stage at ART initiation. Demographic characteristics were as collected at patient registration into the Newlands Clinic cohort. The outcome variable (cancer diagnosis) was defined as having a cancer diagnosis recorded in either the ZNCR or Newlands Clinic database. In the multivariable model, we included sex, education and CD4 count at the time of ART initiation. We did not include WHO clinical stage at ART initiation in the multivariable model due to collinearity with CD4 cell count. We tested proportional hazard assumptions individually for each covariate using Schoenfeld residuals. All statistical analyses were conducted using Stata version 13.1 (StataCorp, College Station, Texas 77 845 USA).
3. RESULTS
After deduplication, 8792 records from the Newlands Clinic and 95 254 records from the ZNCR were probabilistically linked resulting in 414 confirmed matches (Figure 1). Details of the PRL dataset completeness are given in Appendix S2. Within this linked dataset, 5102 PLWH had newly initiated ART at Newlands Clinic between 2004 and 2017. We excluded 1161 children younger than 16 years, 413 individuals with less than 30 days of follow‐up after ART initiation, and 86 patients with a prevalent cancer diagnosis (Figure 1).
FIGURE 1.
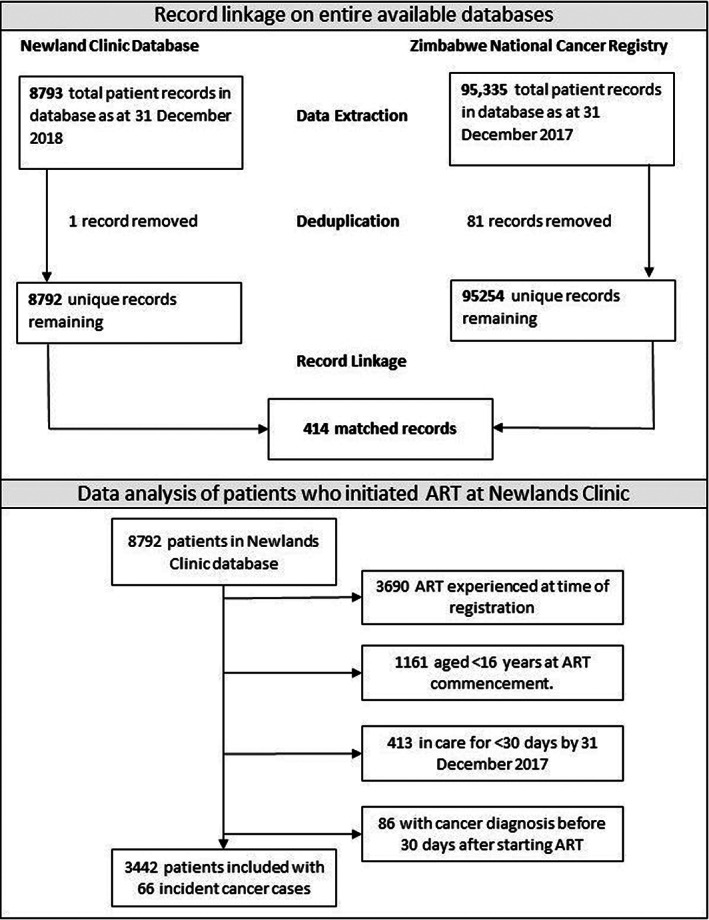
Top: Flow chart of record linkage between Zimbabwe National Cancer Registry and Newlands Clinic; Bottom: Flow diagram of patients included in the study
For the cancer incidence analysis, we included 3442 PLWH with 66 incident cancers. Thirty cancer cases (45%) were recorded in both databases, whereas 22 cases (33%) were documented only in the ZNCR database, and 14 cases (21%) were found only in the Newlands Clinic database (Appendix S3). Of the 14 cases found only in the Newlands Clinic database, 10 had documented biopsy information while visit notes were not available for the other four. Two‐thirds of the included PLWH were female (2235, 64.9%) and the median age at ART initiation was 36.6 years (IQR 30.6–43.4). About a 10th were aged over 50 years (374, 10.9%) and most had secondary or tertiary education (2609, 75.8%), although only 1261 (36.6%) were employed. Most of the included PLWH were severely immune compromised with a median CD4 cell count of 169 cells/mm3 (IQR 82–275) at ART initiation. Almost a third (1039, 30.2%) had CD4 cell counts less than 100 cells/mm3. Forty‐one percent (n = 1404) were classified as WHO clinical stage three or four at ART initiation (Table 1).
TABLE 1.
Characteristics and cancer incidence rates of patients initiating antiretroviral therapy at Newlands Clinic between 2004 and 2017 (N = 3442)
| Characteristic | Patients n (%) | Person‐years at risk | Cancer cases (n) | Incidence rate per 100 000 PY (95% CI) |
|---|---|---|---|---|
| All | 3442 | 19 346 | 66 | 341 (268–434) |
| Sex | ||||
| Female | 2235 (64.9) | 12 994 | 50 | 385 (292–508) |
| Male | 1207 (35.1) | 6351 | 16 | 252 (154–411) |
| Age group (years) | ||||
| 16–35 | 1489 (43.3) | 8275 | 22 | 265 (175–404) |
| 36–50 | 1579 (45.9) | 9053 | 34 | 376 (268–526) |
| > 50 | 374 (10.9) | 2018 | 10 | 496 (266–921) |
| ART initiation period (years) | ||||
| 2004–2007 | 653 (19.0) | 6344 | 20 | 315 (203–489) |
| 2008–2010 | 944 (27.4) | 7025 | 26 | 370 (252–544) |
| 2011–2013 | 761 (22.1) | 3834 | 13 | 339 (197–584) |
| 2014–2017 | 1084 (31.5) | 2143 | 7 | 327 (156–685) |
| Education | ||||
| Primary or none | 833 (24.2) | 5478 | 26 | 475 (323–697) |
| Secondary or tertiary | 2609 (75.8) | 13 868 | 40 | 288 (212–393) |
| Employment | ||||
| Employed | 1261 (36.6) | 6564 | 23 | 350 (233–527) |
| Unemployed | 1970 (57.2) | 11 396 | 40 | 351 (257–479) |
| Unknown | 211 (6.1) | 1386 | 3 | ‐ |
| CD4 count categories at ART initiation (cells/mm3) | ||||
| <100 | 1039 (30.2) | 6271 | 33 | 526 (374–740) |
| 100–199 | 974 (28.3) | 6397 | 17 | 266 (165–427) |
| ≥200 | 1410 (41.0) | 6656 | 16 | 240 (147–392) |
| Unknown | 19 (0.6) | 21 | 0 | ‐ |
| WHO clinical stage for HIV disease at ART initiation | ||||
| Stage 1 | 1144 (33.2) | 4915 | 10 | 203 (109–378) |
| Stage 2 | 890 (25.9) | 5488 | 17 | 310 (193–498) |
| Stage 3 | 1061 (30.8) | 6952 | 29 | 417 (290–600) |
| Stage 4 | 343 (10.0) | 1977 | 10 | 506 (272–940) |
| Unknown | 4 (0.1) | 14 | 0 | ‐ |
Note: Incidence rates are unadjusted. Data source: Linked Zimbabwe National Cancer Registry and Newlands Clinic electronic records.
Abbreviations: ART, antiretroviral therapy; CI, confidence interval; PY, person‐years.
Patients who developed cancer were slightly older at ART initiation (median age 39.1 years, IQR 33.8–45.5) compared with those who did not develop cancer (median age 36.5 years, IQR 30.4–43.3). The median baseline CD4 count among patients who developed cancer was lower (106 cells/mm3, IQR 54–196) than among those who did not (170 cells/mm3, IQR 84–276), p = .001. Half of the PLWH who developed an incident cancer (n = 33, 50.0%) had initiated ART with CD4 counts below 100 cells/mm3.
Overall, there were 66 incident cancer cases during 19 346 PY of follow‐up for an incidence rate of 341/100 000 PY (95% CI 268–434). The cancer incidence rate increased with more advanced WHO clinical stage and decreasing CD4 counts at ART initiation (Table 2). PLWH who initiated ART at a CD4 count of <100 cells/mm3 had a particularly high cancer incidence rate of 526/100 000 PY (95% CI 374–740). The most common cancer was cervical cancer (n = 16) with an incidence rate of 123/100 000 female PY (95% CI 75–201) followed by Kaposi sarcoma and lymphoma with 12 cases each (62/100 000 PY; 95% CI 35–109). The distribution of the remaining cancer cases was as follows: five non‐cervical anogenital cancer (26/100 000 PY; 95% CI 11–62), five breast, three conjunctiva, two central nervous system, five gastrointestinal, and one each of kidney, larynx, lung, plasma cell tumor, soft tissue sarcoma, and uterus.
TABLE 2.
Risk factors for cancer diagnosis after antiretroviral therapy initiation
| Characteristic | HR (95% CI) | aHR (95% CI) | p‐Value |
|---|---|---|---|
| Sex | |||
| Male | 1 | 1 | 0.053 |
| Female | 1.56 (0.89–2.74) | 1.76 (0.99–3.12) | |
| Age at ART initiation | 1.03 (1.01–1.05) | 1.03 (1.00–1.05) | 0.024 |
| ART initiation period | |||
| 2004–2007 | 1 | ‐ | |
| 2008–2010 | 1.14 (0.61–2.12) | ‐ | |
| 2011–2013 | 0.90 (0.43–1.90) | ‐ | |
| 2014–2017 | 0.61 (0.24–1.54) | ‐ | |
| Education | |||
| Primary or none | 1 | 1 | 0.231 |
| Secondary or tertiary | 0.57 (0.35–0.94) | 0.73 (0.44–1.22) | |
| Employment | |||
| Unemployed | 1 | ‐ | |
| Employed | 0.97 (0.58–1.62) | ‐ | |
| CD4 count categories at ART initiation (cells/mm3) | |||
| <100 | 1 | 1 | 0.007 a |
| 100–199 | 0.51 (0.28–0.92) | 0.49 (0.27–0.88) | |
| ≥200 | 0.42 (0.23–0.77) | 0.42 (0.23–0.77) | |
| WHO clinical stage at ART initiation | |||
| Stage 1 | 1 | ‐ | |
| Stage 2 | 1.70 (0.78–3.73) | ‐ | |
| Stage 3 | 2.33 (1.13–4.82) | ‐ | |
| Stage 4 | 2.77 (1.15–6.68) | ‐ |
Note: Covariates without statistically significant results in the univariable model were not included in the multivariable model. WHO stage was not included in the multivariable model due to collinearity with CD4 cell count. Bold type indicates statistically significant results. p‐Values are from the multivariable analysis model.
Abbreviations: aHR, adjusted hazard ratio; ART, antiretroviral therapy; HR, hazard ratio.
From likelihood ratio test.
In adjusted analyses, the risk of developing cancer increased by 3% with each additional year in age at ART initiation (adjusted hazard ratio [aHR] 1.03, 95% CI 1.00–1.05). When compared with PLWH commencing ART with CD4 cell counts below 100 cells/mm3, those with CD4 cell counts between 100 and 199 had a 51% lower risk (aHR 0.49, 95% CI 0.27–0.88) while those with CD4 cell counts ≥200 cells/mm3 had a 58% lower risk of developing cancer (aHR 0.42, 95% CI 0.23–0.77), see Table 2. Cancer risk tended to be higher in female than male PLWH (aHR 1.76, 95% CI 0.99–3.12).
4. DISCUSSION
We report record linkage ascertained cancer incidence rates from an HIV cohort in Zimbabwe showing that cervical cancer was the most common incident malignancy with an incidence rate of 123 per 100 000 PY. Most incident cancer cases were AIDS‐defining malignancies. The risk of being diagnosed with cancer increased with older age and lower CD4 cell count at ART initiation.
The strength of our study was the use of PRL methods to enhance cancer ascertainment at an ART clinic. We matched quality controlled electronic medical records at Newlands Clinic to cancer records from the ZNCR and, thereby, managed to identify an additional 22 cancer cases that were only recorded in the ZNCR. This enabled us to conduct what to our knowledge is the first record linkage ascertained cancer incidence study in Zimbabwe. However, there were some limitations to the study. The Newlands Clinic cohort is relatively small compared with those that have been analyzed in similar studies 9 , 10 and not necessarily representative of the much larger public ART clinics in Zimbabwe. Furthermore, many PLWH who were referred to Newlands Clinic had initiated ART elsewhere and were, therefore, excluded from this analysis, further reducing the sample size. In addition, our results may be an underestimate in view of the high number of records with incomplete key data such as missing national ID numbers, full names, or dates of birth (Appendix 2).
In line with similar studies from Malawi and South Africa, 9 , 10 we found AIDS‐defining cancers to be the most common cancer diagnoses among PLWH. However, our overall cancer incidence rate estimate was lower than that observed in Malawi (age‐adjusted incidence: 689 per 100 000 PY, 95% CI: 610–768) and South Africa (age‐standardized incidence: 877 per 100 000 PY, 95% CI: 744–1041). Of note, the three studies may not be directly comparable due differences in study periods, study populations, and definition of incident cancers. Furthermore, we reported crude incidence rates, which may be lower than age‐adjusted rates given the young age of our cohort. Interestingly, Kaposi sarcoma was by far the most common cancer diagnosis in the other two studies, 9 , 10 but incidence rates in our study were highest for cervical cancer. This is in line with a report that showed four‐fold cumulative incidence of cervical cancer compared with Kaposi sarcoma in the general population of Harare and Bulawayo, Zimbabwe, between 2011 and 2013. 20 Active screening of women for cervical cancer since 2011 may also have contributed to increased case finding at Newlands Clinic.
Infection related cancers such as cervical and other anogenital cancers, Kaposi sarcoma, and lymphoma accounted for most incident cancers in our study. Infectious agents are common causes of cancer in sub‐Saharan Africa, unlike in high‐income settings where lifestyle factors are the predominant causes of cancer. 3 , 8 The high burden of HPV associated cancers in our study reinforces the urgency of accelerating HPV vaccination programs to advance toward the 90‐70‐90 targets of eliminating cervical cancer as a public health problem by 2030. 21 , 22 It also highlights the important role of cervical cancer screening programs to reduce the incidence of cervical cancer. 23 , 24 Furthermore, consistent with other studies, our results showed that initiating ART at higher CD4 cell counts reduced the overall risk of developing cancer. 25 , 26 Enhanced efforts toward HIV testing and early ART initiation as prescribed in the WHO “test‐and‐treat” drive is likely to reduce cancer incidence among PLWH.
Linking records between the ZNCR and Newlands Clinic improved cancer incidence ascertainment by identifying cancer cases that were recorded in only one of either of the databases. However, of 14 patients whose cancer diagnoses were only found in the Newlands Clinic database, 10 patients had histological information available. For these patients, the failure to find matching records in the ZNCR was probably due to missing or incomplete information that impeded the PRL process. Active investment in electronic medical records systems is needed to improve gathering of epidemiologic data in low‐ and middle‐income countries. Reliable epidemiologic data are essential to inform allocation of resources and planning of interventions. However, as observed in our study, complete and accurate capturing of identifying variables such as names and national ID numbers is needed to achieve accurate record linkage results. This effort involves all stakeholders in the chain of care including ART and oncology clinics, laboratories, radiology service providers and cancer registry data capture clerks.
In conclusion, we used PRL methods to improve the estimation of cancer incidence among PLWH in Zimbabwe. Cervical cancer and other infection related cancers were the most common cancer types in this cohort of PLWH on ART. Lower CD4 cell counts at the time of ART initiation and older age were associated with higher risk of developing cancer. Intensified efforts toward HPV vaccinations and the WHO prescribed “test‐and‐treat” approach for early ART initiation may help lower cancer incidence among PLWH.
CONFLICT OF INTEREST
The authors declare no conflict of interest.
AUTHOR CONTRIBUTIONS
Conceptualization, Formal Analysis, Methodology, Project Administration, Supervision, Visualization, E.R.; Conceptualization, Data Curation, Methodology, Resources, E.C.; Data Curation, Methodology, A.M.; Conceptualization, Data Curation, Formal Analysis, Methodology, Software, A.S.; Conceptualization, Methodology, Project Administration, Supervision, C.C.; Conceptualization, Funding Acquisition, Methodology, Resources, Supervision, M.E.; Conceptualization, Funding Acquisition, Methodology, Supervision, J.B.; Conceptualization, Investigation, Methodology, Resources, Supervision, M.B.
ETHICAL STATEMENT
Participants included in this study provided written consent. The study was approved by the Newlands Clinic Research Team and the Medical Research Council of Zimbabwe (MRCZ No. A1336).
Supporting information
Appendix S1. Supporting Information.
Appendix S2. Supporting Information.
Appendix S3. Supporting Information.
ACKNOWLEDGMENTS
The authors acknowledge the ZNCR staff, Newlands Clinic patients and staff, as well Joerg Reiher who built the additional regular expression, string transformation and K‐Link KNIME nodes used in this project.
Shamu T, Rohner E, Chokunonga E, et al. Cancer incidence among people living with HIV in Zimbabwe: A record linkage study. Cancer Reports. 2022;5(10):e1597. doi: 10.1002/cnr2.1597
Funding information National Institute of Allergy and Infectious Diseases of the National Institutes of Health, Grant/Award Number: U01AI069924; National Cancer Institute (NCI); Fogarty International Center (FIC); National Institute on Alcohol Abuse and Alcoholism (NIAAA); National Heart, Lung, and Blood Institute (NHLBI); National Institute on Drug Abuse (NIDA); National Institute of Diabetes and Digestive And Kidney Diseases (NIDDK); Eunice Kennedy Shriver National Institute of Child Health & Human Development (NICHD)
DATA AVAILABILITY STATEMENT
The data that support the findings of this study are available from the corresponding author upon reasonable request.
REFERENCES
- 1. Sigel K, Dubrow R, Silverberg M, Crothers K, Braithwaite S, Justice A. Cancer screening in patients infected with HIV. Curr HIV/AIDS Rep. 2011;8(3):142‐152. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Shiels MS, Cole SR, Kirk GD, Poole C. A meta‐analysis of the incidence of non‐AIDS cancers in HIV‐infected individuals. J Acquir Immune Defic Syndr. 2009;52(5):611‐622. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Sitas F, Parkin DM, Chirenje M, Stein L, Abratt R, Wabinga H. Part II: cancer in indigenous Africans‐causes and control. Lancet Oncol. 2008;9:786‐795. http://www.thelancet.com/article/S1470204508701980/fulltext [DOI] [PubMed] [Google Scholar]
- 4. Darcis G, Lambert I, Sauvage AS, et al. Factors associated with late presentation for HIV care in a single Belgian reference center: 2006–2017. Scientific Rep. 2018;8(1):1‐6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Shiels MS, Engels EA. Evolving epidemiology of HIV‐associated malignancies. Curr Opin HIV AIDS. 2017;12:6‐11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. The Late Presentation Working Groups in EuroSIDA and COHERE . Estimating the burden of HIV late presentation and its attributable morbidity and mortality across Europe 2010‐2016. BMC Infect Dis. 2020;20(1):728. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Lekoane KMB, Kuupiel D, Mashamba‐Thompson TP, Ginindza TG. The interplay of HIV and human papillomavirus‐related cancers in sub‐Saharan Africa: scoping review. System Rev. 2020;9:88. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Mbulaiteye SM, Bhatia K, Adebamowo C, Sasco AJ. HIV and cancer in Africa: mutual collaboration between HIV and cancer programs may provide timely research and public health data. Infect Agents Cancer. 2011;6:16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Sengayi M, Spoerri A, Egger M, et al. Record linkage to correct under‐ascertainment of cancers in HIV cohorts: the Sinikithemba HIV clinic linkage project. Int J Cancer. 2016;139(6):1209‐1216. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Horner MJ, Chasimpha S, Spoerri A, et al. High cancer burden among antiretroviral therapy users in Malawi: a record linkage study of observational human immunodeficiency virus cohorts and cancer registry data. Clin Infect Dis. 2019;69(5):829‐835. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Akanbi MO, Ocheke AN, Agaba PA, et al. Use of electronic health records in sub‐Saharan Africa: progress and challenges. J Med Tropics. 2012;14(1):1‐6. [PMC free article] [PubMed] [Google Scholar]
- 12. Bohlius J, Maxwell N, Spoerri A, et al. Incidence of AIDS‐defining and other cancers in HIV‐positive children in South Africa: record linkage study. Pediatr Infect Dis J. 2016;35(6):e164‐e170. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Chokunonga E, Windridge P, Sasieni P, Borok M, Parkin DM. Black‐white differences in cancer risk in Harare, Zimbabwe, during 1991‐2010. Int J Cancer. 2016;138(6):1416‐1421. [DOI] [PubMed] [Google Scholar]
- 14. Chokunonga E, Borok MZ, Chirenje ZM, Nyakabau AM, Parkin DM. Trends in the incidence of cancer in the black population of Harare, Zimbabwe 1991‐2010. Int J Cancer. 2013;133(3):721‐729. [DOI] [PubMed] [Google Scholar]
- 15. Egger M, Ekouevi DK, Williams C, et al. Cohort profile: the international epidemiological databases to evaluate AIDS (IeDEA) in sub‐Saharan Africa. Int J Epidemiol. 2012;41(5):1256‐1264. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Chammartin F, Dao Ostinelli CH, Anastos K, et al. International epidemiology databases to evaluate AIDS (IeDEA) in sub‐Saharan Africa, 2012–2019. BMJ Open. 2020;10(5):e035246. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Shamu T, Chimbetete C, Shawarira‐Bote S, Mudzviti T, Luthy R. Outcomes of an HIV cohort after a decade of comprehensive care at Newlands Clinic in Harare, Zimbabwe: TENART cohort. PLoS ONE. 2017;12(10):e0186726. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. IARC . Working Group on Evaluation of Carcinogenic Risks to Humans. A Review of Human Carcinogens. International Agency for Research on Cancer; 2009. [Google Scholar]
- 19.StataCorp. 2013. Stata 13 Base Reference Manual. College Station, TX: Stata Press. https://www.stata.com/manuals13/ststrate.pdf (Accessed October 6, 2021)
- 20. Parkin DM, Ferlay J, Jemal A, et al. Cancer in Sub‐Saharan Africa. Vol 167. International Agency for Research on Cancer; 2018:251. [Google Scholar]
- 21. Shinkafi‐Bagudu Z. Global partnerships for HPV vaccine must look beyond national income. JCO Global Oncol. 2020;6:1746‐1748. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Global strategy to accelerate the elimination of cervical cancer as a public health problem. https://www.who.int/publications/i/item/9789240014107 (Accessed May 27, 2021)
- 23. Mandiriri AM, Pascoe MJ, Shamu T, Lowe S. Cervical human papillomavirus prevalence, risk factors and outcomes in a cohort of HIV‐infected women in Harare, Zimbabwe. South Afr J HIV Med. 2020;21(1):8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Schiffman M, Castle PE, Jeronimo J, Rodriguez AC, Wacholder S. Human papillomavirus and cervical cancer. Lancet. 2007;370(9590):890‐907. [DOI] [PubMed] [Google Scholar]
- 25. Ruffieux Y, Muchengeti M, Egger M, et al. Immunodeficiency and cancer in 3.5 million people living with human immunodeficiency virus (HIV): the south African HIV cancer match study. Clin Infect Dis. 2021;73(3):e735‐e744. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Kelly H, Weiss HA, Benavente Y, et al. Association of antiretroviral therapy with high‐risk human papillomavirus, cervical intraepithelial neoplasia, and invasive cervical cancer in women living with HIV: a systematic review and meta‐analysis. Lancet HIV. 2018;5(1):e45‐e58. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Appendix S1. Supporting Information.
Appendix S2. Supporting Information.
Appendix S3. Supporting Information.
Data Availability Statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.


