Abstract
Introduction
Ammonia is a highly toxic irritant gas, and its toxicity usually occurs from occupational exposure. Most are unintentional toxicity. It causes tissue damage via exothermic reaction with body tissues causing liquefactive necrosis. Presentation depends on the level and duration of exposure. Management is supportive and its antidote is not available yet.
Presentation of case
We report a case of a 22-year-old male with accidental workplace exposure from coolant damage and leakage. He had altered mental status with visible injury to the nasal cavity, and ulcerated laryngeal mucosa with asymmetric vocal cords on bronchoscopy. Computed tomography showed right lower lobe consolidations. A muti-disciplinary approach and prompt management with respiratory support along with inhaled steroids and bronchodilators, antibiotics, electrolyte replacement, analgesics, and supportive eye and oral treatments were initiated. He had to be managed with a high-flow nasal cannula with subsequent tapering of the oxygen supplementation with titration.
Discussion
Inhalation injury due to ammonia toxicity can have varied presentations from mild to severe life-threatening respiratory distress and neurological deterioration. Complications can range from upper airway edema to pulmonary edema. Most of these are usual findings except for the presence of pneumothorax. It could be due to the pre-existing lung status or oxygen therapy but the literature is inadequate.
Conclusion
Ammonia inhalational injury may be associated with pneumothorax. It is important to have a multi-disciplinary approach and in-hospital management of ammonia toxicity as well as to raise awareness regarding the work environment and timely recognition of the possible occupational health hazards.
Keywords: Case report, Ammonia inhalation, Pneumothorax, Multidisciplinary approach, Occupational awareness
Highlights
-
•
Pneumothorax in a case of inhalation ammonia injury.
-
•
Early recognition and a multispecialty approach are vital for the outcome of the toxicity.
-
•
Awareness of occupational safety measures in the workplace can help minimize the associated health hazards.
1. Introduction
Ammonia is a colorless pungent odor highly water soluble irritant gas producing ammonium ion in water [1,2]. Ammonia is synthesized de-novo within the body as part of many biochemical reactions including bacterial enzymatic activities and is quickly metabolized to non-toxic urea by the liver and excreted by kidneys to neutralize its high toxicity effect. The normal blood level of ammonia in a healthy adult is in the range of 15–45 μg/dL and its toxicity occurs once its blood level surpasses the liver's ability to detoxify it [3]. Ammonia exposure occurs from occupational ammonia gas exposure like in production storage, transportation facilities, exposure to ammonia-based fertilizers, household and industrial cleaners, cooling refrigerants, decaying manure via ingestion, inhalation, direct contact with skin, or contact with the eye [1,2]. As per the annual report of the American Association of Poison Control Centre National Poison Data system annual report of the American Association of Poison Control Center's National Poison Data System, there were 1846 single exposures to ammonia with 15 major events and no mortality in 2017 [4]. Most of the ammonia toxicities are unintentional with only 9.2% being intentional ones and accidental ingestion was the most common mode of toxicities among children [1]. Ammonia causes tissue damage via exothermic reaction with body tissues producing ammonium ions and liquefactive necrosis with deeper burns [5]. So, exposure to ammonia gas can damage eyes, skin, lungs, and gastrointestinal tract causing mucosal injuries, scarring, and even perforation. The presentation of a patient varies with acute vs chronic intoxication, causing tracheobronchitis, asphyxiation, and even death on acute exposure and bronchiectasis, optic neuropathy on chronic exposure via inhalation [1,2,6,7]. Its diagnosis is done based on circumstances of possible exposure, physical examination findings, and basic and body system-focused lab tests while there are no tests for assessing the extent of its toxicity [1]. The management of ammonia toxicity is symptomatic and supportive as there's no antidote for it [1]. Here, we present a case of accidental workplace acute ammonia intoxication. This case report has been reported as per SCARE 2020 criteria [8] (see Fig. 1, Fig. 3, Fig. 2, Fig. 4).
Fig. 1.
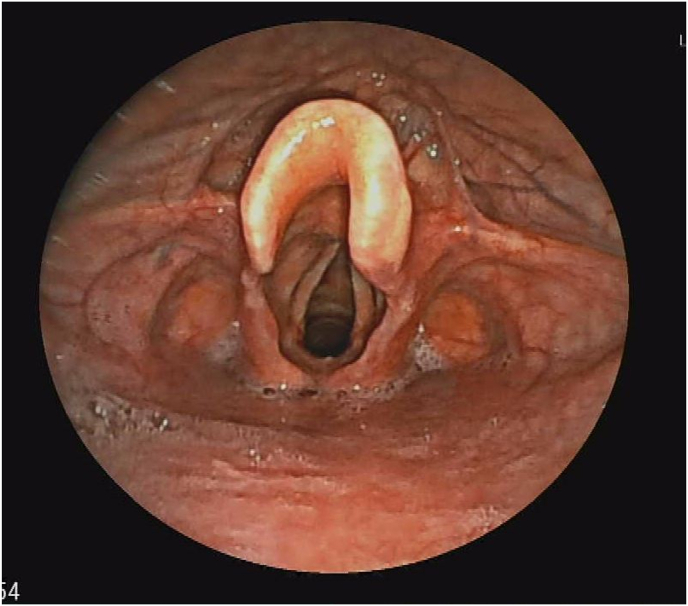
Laryngoscopy image showing laryngeal mucosal lacerations and asymmetric vocal cords.
Fig. 3.
Contrast-enhanced CT chest shows consolidation in right lower lobe (white arrow) as well as patchy consolidation in the left lower lobe.
Fig. 2.
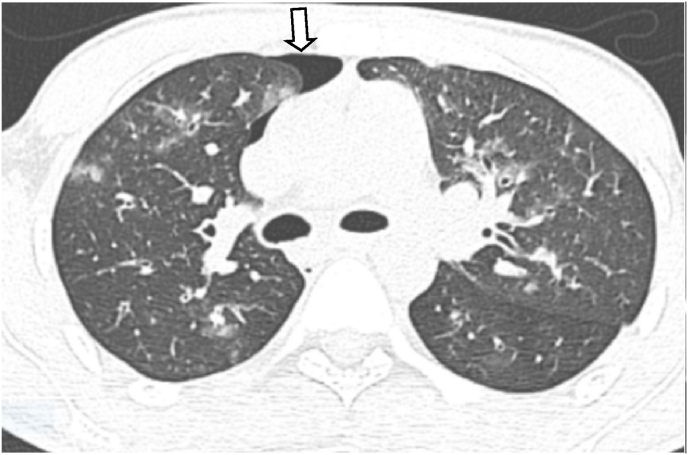
High-resolution computed tomography (HRCT) axial image shows pneumothorax in the right side (white arrow).
Fig. 4.
Follow up laryngoscopy image showing healed mucosal lacerations and resolution of vocal cord edema.
2. Case presentation
Twenty-two-year-old male, a known smoker for eight years, with no other co-morbid conditions was referred from a medical teaching hospital for ammonia intoxication with possible need for mechanical ventilatory support. As per the patient's attendants, he was sleeping at his workplace, a potato storage plant, where there was leakage of ammonia containing coolant from a broken pipeline. He was there for about 20–30 minutes before he was rescued by his colleagues who took him to the first treatment center. Following the exposure, he had an altered level of consciousness, persistent drooling of oral secretions, and desquamation of both of his eyelids and lips. There was no history of fever, abnormal body movements, vomiting, and upward rolling of the eyes. He received supportive treatment at the initial treatment center with intravenous medications of ceftriaxone, hydrocortisone, frusemide, ondansetron, and intravenous fluids, and then referred to our center.
On arrival at the emergency department at our institute, his vitals were stable with a blood pressure of 110/80 mmHg without inotropes and vasopressors and oxygen saturation of 94% with 5 L per minute O2 via face mask. He was fully conscious with intact orientation, and normal gag reflex with no significant findings in full body system examination. He was transferred to ICU for further management. On the first day of admission, he had sloughing of bilateral nasal cavity, bilateral pyriform sinus and lateral pharyngeal wall, swollen and congested epiglottis, and uvula. He was managed with supportive measures like nil per oral, supplemental oxygenation via high flow nasal cannula with 40% FiO2, injection meropenem 1 gm three times a day (TDS), tablet N-acetylcysteine 600 mg twice a day, injection acetaminophen 1 gm four times a day, benzydamine mouth gargle TDS, Nasivion drop in bilateral nostrils TDS, ointment occupol-D on both eyes one a day, gt fluorometholone, and nebulization with normal saline/fluticasone/salbutamol (3/2/1 ml respectively). His echocardiography showed normal left ventricle and right ventricle function without regional wall motion abnormalities and pericardial effusion. On the second day of intensive care unit admission, oxygenation was maintained via venturi at 35% FiO2 at 8 L oxygen per minute. Contrast-enhanced computed tomography neck and chest was performed the following day which showed mild irregularity in the outline of oropharyngeal mucosa, patchy and lobar consolidation was seen in superior and basal segments of bilateral lower lobes, right more than left. These findings were consistent with the toxic effects of anhydrous ammonia on pulmonary tissue. However, we also noted pneumothorax in the right side of the lung. He was reviewed by an otorhinolaryngologist, and feeding was started.
However, on the third day of admission, supplemental oxygen increased and had to be changed to a high flow nasal cannula at 50% FiO2. Following this, a combination injection of amoxicillin and clavulanate of 1.2 g every 8 hours was added, and magnesium was supplemented. A day later, on the fourth day of admission, tapering of oxygen supplementation was done and maintained via Venturi mask at 31% FiO2. Ophthalmology review advised for follow-up in a week with a continuation of current management. With continued supportive management, the patient's oxygenation was titrated and maintained via nasal prongs at 4 L oxygen per minute on the fifth day showing substantial improvement despite the deterioration on the third day. Injectable antibiotics were changed to oral antibiotics, and analgesics were provided on a need basis. The patient was shifted to the general ward after complete hemodynamic stabilization. And was subsequently discharged after a couple of days with a follow-up in 2 weeks.
Follow-up after 2 weeks showed improved clinical status. The laryngoscopy finding was indicative of healed laryngeal and vocal cord lacerations. A pulmonary function test is planned for him in his next follow-up.
3. Discussion
Ammonia toxicity is not an uncommon poisoning that is often seen in the occupational setting as accidental exposure [9], and so in this case. This is a severe ammonia gas intoxication following several minutes of exposure resulting into irritation syndrome of the upper airways, acute toxic lobar consolidation, and irritant conjunctivitis. But in our patient HRCT also showed pneumothorax in the right lung. It is relatively a new finding in a case of ammonia inhalation injury.
Severe acute exposure to ammonia gas may cause significant damage to respiratory tissues which may be fatal or lead to severe pulmonary impairment often requiring mechanical ventilatory assistance to rarely needing lung transplantation [1,2]. Our patient did not require such measures despite severe injuries resulting from the prolonged inhalation exposure and managed with supportive and symptomatic treatment including nebulized glucocorticoids and prophylactic antibiotic therapy to prevent secondary bacterial infection. The use of steroids is debated however, in patient with respiratory symptoms such as ours, may be justified with the clinical improvement with its use. In a case report by Bhalla et al., they discussed a case of accidental ammonia gas intoxication who was managed with steroids, prophylactic antibiotics, and other supportive measures only and was discharged after 72 hours of ICU stay [10]. Our patient initially showed some improvement but there was some deterioration on the third day which subsequently improved after additional antibiotics indicating that superinfection infection should be considered and managed accordingly in such cases. Similarly, a case reported by Maries et al. developed irritation syndrome of the upper airways, occlusive bronchiolitis, and acute toxic pulmonary edema and managed symptomatically including steroids and prophylactic antibiotics, and did not require ventilatory assistance [2]. While cases reported by Köksal et al. [11] and Lalić et al. [12] required mechanical ventilation and lung transplantation respectively. In our patient, patchy and lobar consolidation was seen in superior and basal segments of bilateral lower lobes, more in the right following the toxicity. Pneumothorax was also seen in our patient which does not have supportive datas in the literatures. It can possibly be due to barotrauma related to oxygen therapy or any pre-existing lung conditions like subpleural blebs.
Acute respiratory distress syndrome (ARDS) and acute lung injury are common complications following acute moderate to severe exposure which may progress to chronic obstructive airway disease, this often occurs following chronic mild to moderate ammonia gas exposure Thus, following ammonia gas toxic exposure, the long-term follow-up of a patient is required to diagnose and manage the long-term effect of toxic exposure as in cases reported by Maries et al. [2], Köksal et al. [11] and Lalić et al. [12]. In follow-up 2 weeks after discharge, our case had improved both clinically and in the findings of repeat bronchoscopy. He has been planned for a pulmonary function test during his next visit to access the possible obstructive and/or restrictive sequelae of ammonia gas intoxication.
4. Conclusion
Ammonia gas intoxication is not uncommon and mostly happens as accidental exposure in an occupational setting and has a debilitating effect on different body systems and might cause long morbidity as it lacks a proper antidote. Hence, it is important to raise awareness regarding the environment and occupational safety measures such as proper ventilation of workplace for prevention and availability of hospital facilities to promptly manage such cases. In-hospital management involving specialist like pulmonologist, ophthalmologist, otorhinolaryngologist, gastroenterologist, etc., is the most effective management protocol that can prevent the disease burden on patients.
Ethical approval
N/A.
Sources of funding
None.
Author contributions
Author 1: Led data collection, concept of study, contributed in writing the case information,
Author 2: Literature review, writing initial draft, revising, and editing the manuscript.
Author 3: Literature review and writing case information,
Author 4: Literature review, revising and editing the manuscript,
All authors were involved in manuscript drafting and revising, and approved the final version.
Registration of research studies
-
1.
Name of the registry: N/A.
-
2.
Unique Identifying number or registration ID: N/A.
-
3.
Hyperlink to your specific registration (must be publicly accessible and will be checked): N/A.
Guarantor
Sulochana Khadka, Nepalese Army Institute of Health Sciences – College of Medicine, Kathmandu, Nepal. Email: suloachankhadka1@gmail.com, Phone: +977–9848559942.
Consent
Written informed consent was obtained from the patient for publication of this case report and accompanying images. A copy of the written consent is available for review by the Editor-in-Chief of this journal on request.
Provenance and peer review
Not commissioned, externally peer reviewed.
Declaration of competing interest
None.
Acknowledgement
None.
Footnotes
Supplementary data to this article can be found online at https://doi.org/10.1016/j.amsu.2022.104741.
Appendix A. Supplementary data
The following are the Supplementary data to this article:
References
- 1.Padappayil R.P., Borger J. Ammonia toxicity. Compare. Physiol. Nat. Animal Model. Clinic. Med. 2022 Feb 7:149–189. [Google Scholar]
- 2.Maries L., Maries M. Ammonia intoxication at the work place - a case report. J. Ecophysiol. Occup. Health. 2018;18(3 & 4):103–105. doi: 10.18311/jeoh/2018/20030. [DOI] [Google Scholar]
- 3.Braissant O., McLin V.A., Cudalbu C. Ammonia toxicity to the brain. J. Inherit. Metab. Dis. 2013 Jul;36(4):595–612. doi: 10.1007/s10545-012-9546-2. [DOI] [PubMed] [Google Scholar]
- 4.Gummin D.D., Mowry J.B., Spyker D.A., Brooks D.E., Osterthaler K.M., Banner W. Annual report of the American association of Poison Control centers' national Poison data system (NPDS): 35th annual report. Clin. Toxicol. 2017;56(12):1213–1415. doi: 10.1080/15563650.2018.1533727. 2018 Dec. [DOI] [PubMed] [Google Scholar]
- 5.Arwood R., Hammond J., Ward G.G. Ammonia inhalation. J. Trauma. 1985 May;25(5):444–447. doi: 10.1097/00005373-198505000-00014. [DOI] [PubMed] [Google Scholar]
- 6.EPA. Toxicological . Integrated Risk Information System National Center for Environmental Assessment Office of Research and Development US Environmental Protection Agency Washington, DC; 2016;16(September. Review of Ammonia (Noncancer Inhalation): Executive Summary; p. 10. [Google Scholar]
- 7.Murphy D.B. Public Health England; 2007;(November. Ammonia: Toxicological Overview; pp. 1–15. [Google Scholar]
- 8.Agha R.A., Franchi T., Sohrabi C., Mathew G., Kerwan A., SCARE Group The SCARE 2020 guideline: updating consensus surgical CAse REport (SCARE) guidelines. Int. J. Surg. 2020 Dec;84:226–230. doi: 10.1016/j.ijsu.2020.10.034. [DOI] [PubMed] [Google Scholar]
- 9.O'Kane G.J. Inhalation of ammonia vapour. A report on the management of eight patients during the acute stages. Anaesthesia. 1983 Dec;38(12):1208–1213. doi: 10.1111/j.1365-2044.1983.tb12527.x. [DOI] [PubMed] [Google Scholar]
- 10.Bhalla A., Mahi S., Sharma N., Singh S. Glycopyrrolate in toxic exposure to ammonia gas. J. Emergencies, Trauma, Shock. 2011;4(1):140–141. doi: 10.4103/0974-2700.76830. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Koksal O., Almacioglu M.L., Ozdemir F., Bulut M., Akkose Aydin S. A case report of ammonium sulfate inhalation. J. Acad. Emergency Med. 2011:180–182. [Google Scholar]
- 12.Lalić H., Djindjić-Pavičić M., Kukuljan M. Ammonia intoxication on workplace - case report and a review of literature. Coll. Antropol. 2009;33(3):945–949. PMID: 19860130. [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.