Abstract
The SARS-CoV-2 Omicron BA.2.75 variant emerged in May 2022. BA.2.75 is a BA.2 descendant but is phylogenetically distinct from BA.5, the currently predominant BA.2 descendant. Here, we show that BA.2.75 has a greater effective reproduction number and different immunogenicity profile than BA.5. We determined the sensitivity of BA.2.75 to vaccinee and convalescent sera as well as a panel of clinically available antiviral drugs and antibodies. Antiviral drugs largely retained potency, but antibody sensitivity varied depending on several key BA.2.75-specific substitutions. The BA.2.75 spike exhibited a profoundly higher affinity for its human receptor, ACE2. Additionally, the fusogenicity, growth efficiency in human alveolar epithelial cells, and intrinsic pathogenicity in hamsters of BA.2.75 were greater than those of BA.2. Our multilevel investigations suggest that BA.2.75 acquired virological properties independent of BA.5, and the potential risk of BA.2.75 to global health is greater than that of BA.5.
Keywords: SARS-CoV-2, COVID-19, Omicron, BA.2.75, transmissibility, immune resistance, antiviral drug resistance, pathogenicity
Graphical abstract
Saito and G2P-Japan Consortium et al. elucidate the virological properties of the SARS-CoV-2 Omicron BA.2.75 variant. BA.2.75 is more transmissible than BA.5 and exhibits different antigenicity than BA.2 and BA.5. The BA.2.75 spike exhibits higher affinity to ACE2 and higher fusogenicity, and BA.2.75 is more pathogenic than BA.2 in hamsters.
Introduction
Newly emerging SARS-CoV-2 variants need to be carefully and rapidly assessed for a potential increase in their growth efficiency in the human population (i.e., relative effective reproduction number [Re]), their evasion from antiviral immunity, and their pathogenicity. Resistance to antiviral humoral immunity can be mainly determined by substitutions in the spike (S) protein. For instance, Omicron BA.1 (Cao et al., 2021; Cele et al., 2021; Dejnirattisai et al., 2022; Garcia-Beltran et al., 2021; Liu et al., 2021; Meng et al., 2022; Planas et al., 2021; Takashita et al., 2022; VanBlargan et al., 2022), BA.2 (Bruel et al., 2022; Takashita et al., 2022; Yamasoba et al., 2022b), and BA.5 (Yamasoba et al., 2022b; Khan et al., 2022; Wang et al., 2022; Qu et al., 2022; Hachmann et al., 2022; Tuekprakhon et al., 2022; Cao et al., 2022; Arora et al., 2022; Lyke et al., 2022; Gruell et al., 2022; Kimura et al., 2022c) exhibit profound resistance to neutralizing antibodies induced by vaccination, natural SARS-CoV-2 infection, and therapeutic monoclonal antibodies. In particular, newly spreading SARS-CoV-2 variants tend to be resistant to the humoral immunity induced by infection with a prior variant; for instance, BA.2 is resistant to BA.1 breakthrough infection sera (Qu et al., 2022; Tuekprakhon et al., 2022; Yamasoba et al., 2022a), and BA.5 is resistant to BA.2 breakthrough infection sera (Wang et al., 2022; Hachmann et al., 2022; Kimura et al., 2022c). Therefore, acquiring immune resistance to previously dominant variants is a key factor in outcompeting previous variants, thereby obtaining relatively increased Re compared with the previously dominant variant. In addition to the evasion of humoral immunity induced by vaccination and infection, substitutions in the S protein can affect sensitivity to therapeutic monoclonal antibodies; for instance, BA.5 exhibits higher resistance to certain therapeutic antibodies than BA.2 (Yamasoba et al., 2022b; Wang et al., 2022; Cao et al., 2022). Furthermore, viral pathogenicity is closely associated with the phenotype of the viral S protein. In particular, we have proposed that the fusogenicity of the viral S protein in in vitro cell cultures is associated with viral pathogenicity in vivo (Kimura et al., 2022c; Yamasoba et al., 2022a; Suzuki et al., 2022; Saito et al., 2022).
As mentioned above, major SARS-CoV-2 phenotypes can be defined by the function of the viral S protein. The SARS-CoV-2 S protein has two major domains, the receptor-binding domain (RBD) and the N-terminal domain (NTD) (Mittal et al., 2022; Harvey et al., 2021). The RBD is crucial for the binding to the human angiotensin-converting enzyme 2 (ACE2) receptor for cell attachment and entry; therefore, this domain has been considered a major target for neutralizing antibodies to block viral infection (Harvey et al., 2021; Jackson et al., 2022; Barnes et al., 2020). The NTD can also be recognized by antibodies, and some antibodies targeting the NTD potentially neutralize viral infection (Lok, 2021; Voss et al., 2021; Cerutti et al., 2021; Suryadevara et al., 2021; McCallum et al., 2021; Liu et al., 2020; Chi et al., 2020), despite our limited understanding of its virological function.
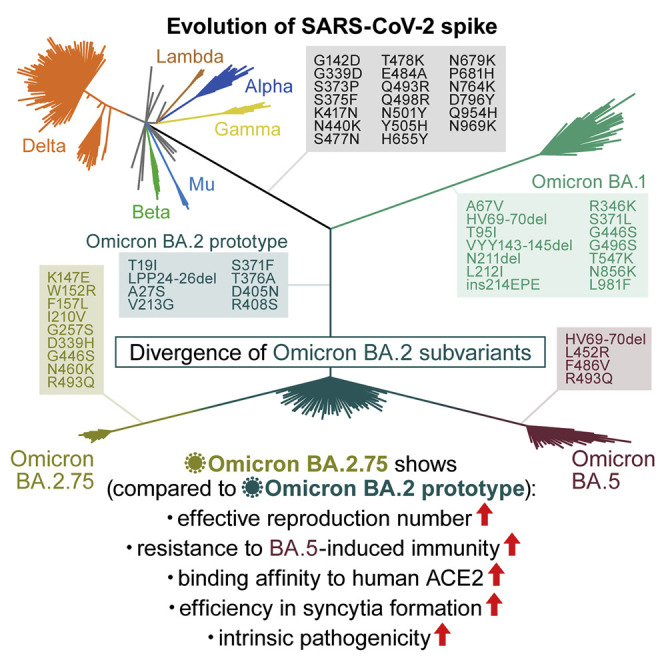
The Omicron BA.2.75 variant, a new BA.2 subvariant, was first detected in India in May 2022 (WHO, 2022). Because an early preliminary investigation suggested the potential increase in the relative Re value of BA.2.75 compared with that of BA.5 and the original BA.2 (GitHub, 2022), BA.2.75 has been flagged as the most concerning variant that can potentially outcompete BA.5 and become the next predominant variant in the future. On July 19, 2022, the WHO classified this variant as a variant of concern lineage under monitoring (WHO, 2022). Compared with the BA.2 S, BA.5 S has four mutations (Kimura et al., 2022c; Yamasoba et al., 2022a), whereas BA.2.75 S has nine mutations. These findings suggest that the virological phenotype of BA.2.75 is critically different from that of previous BA.2 subvariants. Here, we elucidate the features of a newly emerging SARS-CoV-2 Omicron BA.2.75 subvariant.
Results
Epidemics of BA.2.75 in India
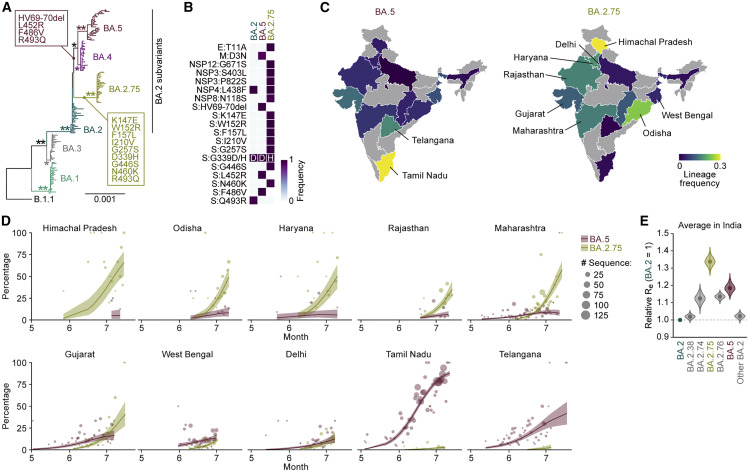
As of the beginning of August 2022, the Omicron BA.5 variant is predominant worldwide. However, a novel BA.2 subvariant, BA.2.75, emerged and rapidly spread in India since May 2022. Although BA.2.75 and BA.5 belong to the BA.2 subvariant clade, BA.2.75 is phylogenetically distinct from the BA.5 clade (Figure 1 A). Compared with BA.2, BA.2.75 harbors 14-amino-acid substitutions, including nine substitutions in the S protein (Figures 1B and S1A). In India, BA.5 and BA.2.75 spread in different regions: BA.5 spread in the southern area, including the Tamil Nadu and Telangana states, whereas BA.2.75 spread to the other areas, including the Himachal Pradesh, Odisha, Haryana, Rajasthan, and Maharashtra states (Figures 1C and 1D). To compare the relative Re between BA.5 and BA.2.75 in India upon adjusting the regional differences, we constructed a Bayesian hierarchical model that can estimate both state-specific Re values and the value averaged in India (Figures 1E and S1B; Table S1). The Re value of BA.5 is 1.19-fold higher than that of BA.2 on average in India (Figure 1E). This value is comparable with the relative Re value of BA.5 in South Africa (1.21) estimated in our recent study (Kimura et al., 2022c). Of note, the Re value of BA.2.75 was 1.34-fold higher than that of BA.2, and the Re value of BA.2.75 was 1.13-fold higher than that of BA.5 (Figures 1E and S1C). Furthermore, in the Indian states analyzed, the Re value of BA.2.75 was greater than that of BA.5 (Figures S1B and S1C). Together, our data suggest that BA.2.75 has the potential to spread more rapidly than BA.5 and will be predominant in some regions including India in the near future.
Figure 1.
Epidemics of BA.2.75 in India
(A) A maximum likelihood tree of Omicron sublineages. Sequences of BA.1-BA.5 sampled from South Africa and BA.2.75 are included. The mutations acquired in the S protein of BA.2.75 are indicated. Note that R493Q is a reversion (i.e., back mutation from the BA.1–BA.3 lineages [R493] to the B.1.1 lineage [Q493]). Bootstrap values, ∗≥0.8; ∗∗≥0.95.
(B) Amino acid differences among BA.2, BA.2.75, and BA.5. Heatmap indicates the frequency of amino acid substitutions.
(C) Lineage frequencies of BA.5 (left) and BA.2.75 (right) in each Indian state. SARS-CoV-2 sequences collected from June 15 to July 15, 2022 were analyzed.
(D) Epidemic dynamics of SARS-CoV-2 lineages in Indian states. The results for BA.2.75 and BA.5 are shown. The observed daily sequence frequency (dot) and the dynamics (posterior mean, line; 95% CI, ribbon) are shown. The dot size is proportional to the number of sequences.
(E) Estimated relative Re of each viral lineage, assuming a fixed generation time of 2.1 days. The Re value of BA.2 is set at 1. The posterior (violin), posterior mean (dot), and 95% CI (line) are shown. The average values across India estimated by a Bayesian hierarchical model are shown, and the state-specific Re values are shown in Figure S1B.
Sensitivity of BA.2.75 to antiviral humoral immunity
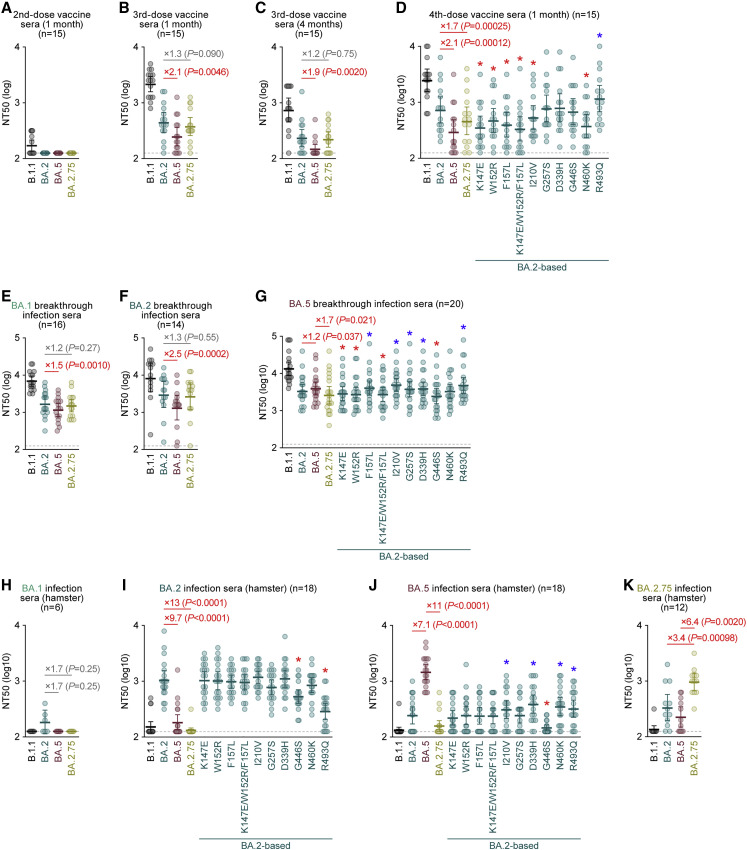
To investigate the sensitivity of BA.2.75 to antiviral humoral immunity, we prepared pseudoviruses bearing the S proteins of D614G-bearing ancestral B.1.1, BA.2, BA.5, and BA.2.75. Human sera were collected from vaccinated and infected individuals (Table S2). The 2-dose vaccine sera were ineffective against all Omicron subvariants tested, including BA.2.75 (Figure 2 A). Although BA.5 was significantly more resistant to 3-dose vaccine sera than BA.2, which is consistent with previous studies (Wang et al., 2022; Hachmann et al., 2022; Kimura et al., 2022c), the sensitivity of BA.2.75 to these sera was comparable with that of BA.2 (Figures 2B and 2C). However, BA.5 (2.1-fold) and BA.2.75 (1.7-fold) were significantly more resistant to 4-dose vaccine sera than BA.2 (Figure 2D). To identify the substitution(s) responsible for the higher resistance of BA.2.75 to 4-dose vaccine sera than BA.2, we prepared BA.2 S-based derivatives with the BA.2.75 substitutions. As shown in Figure 2D, four substitutions in the NTD, K147E, W152R, F157L, and I210V, and a substitution in the RBD, N460K, are responsible for the resistance to 4-dose vaccine sera. On the other hand, R493Q increased the sensitivity to 4-dose vaccine sera (Figure 2D). Since R493Q is a reversion substitution (R493 in BA.2 but Q493 in B.1.1, BA.5, and BA.2.75; Figure S1A), these results suggest that this substitution recovered the epitope recognized by vaccine-induced humoral immunity.
Figure 2.
Immune resistance of BA.2.75
Neutralization assays were performed with pseudoviruses harboring the S proteins of B.1.1, BA.1, BA.2, and BA.2.75.
The BA.2 S-based derivatives are included in (D), (G), (I), and (J). The following sera were used.
(A–D) mRNA vaccine sera (15 donors) collected 1 month after the 2nd-dose vaccination (A), 1 month after the 3rd-dose vaccination (B), 4 months after the 3rd-dose vaccination (C), and 1 month after the 4th-dose vaccination (D).
(E–G) Convalescent sera from fully vaccinated individuals who had been infected with BA.1 after full vaccination (16 donors) (E), BA.2 after full vaccination (14 donors) (F), and BA.5 after full vaccination (20 donors) (G).
(H–K) Sera from hamsters infected with BA.1 (6 hamsters; H), BA.2 (18 hamsters; I), BA.5 (18 hamsters; J), and BA.2.75 (12 hamsters; K).
Assays for each serum sample were performed in triplicate to determine the 50% neutralization titer (NT50). Each dot represents one NT50 value, and the geometric mean and 95% CI are shown. Statistically significant differences were determined by two-sided Wilcoxon signed-rank tests. The p values versus BA.2 (B–F, H, and I), BA.5 (G and J), or BA.2.75 (K) are indicated in the panels. The horizontal dashed line indicates the detection limit (120-fold). For the BA.2 derivatives (D, G, I, and J), statistically significant differences versus BA.2 (p < 0.05) are indicated with asterisks. Red and blue asterisks indicate decreased and increased NT50s, respectively. Information on the vaccinated/convalescent donors is summarized in Table S2.
See also Table S2.
We then assessed the sensitivity of BA.2.75 to the convalescent sera from individuals who were infected with BA.1 or BA.2 after 2-dose or 3-dose vaccination (i.e., breakthrough infection). Similar to the previous reports including ours (Wang et al., 2022; Hachmann et al., 2022; Kimura et al., 2022c), BA.5 exhibited significant resistance to breakthrough infection sera compared with BA.2. In contrast, the sensitivity of BA.2.75 to these sera was comparable with that of BA.2 (Figures 2E and 2F), suggesting that BA.2.75 is not resistant to the humoral immunity induced by infection with prior Omicron subvariants, including BA.1 and BA.2. In the case of BA.5 breakthrough infection sera, both BA.2 (1.2-fold) and BA.2.75 (1.7-fold) were significantly more resistant than BA.5 (Figure 2G). The neutralization assay to screen the substitution(s) responsible for the higher resistance of BA.2.75 to breakthrough BA.5 infection sera showed that two substitutions in the NTD, K147E and W152R, and a substitution in the RBD, G446S, were responsible for the resistance to breakthrough BA.5 infection sera (Figure 2G).
To further address the difference in immunogenicity among Omicron subvariants, we used the sera obtained from infected hamsters at 16 days postinfection (d.p.i.) (Kimura et al., 2022c; Yamasoba et al., 2022a; Suzuki et al., 2022). Although the sera obtained from the BA.1-infected hamsters were ineffective against BA.2, BA.5, and BA.2.75 (Figure 2H), both BA.5 (9.7-fold) and BA.2.75 (13-fold) exhibited significantly greater resistance to the sera obtained from the BA.2-infected hamsters than BA.2 (Figure 2I). These results suggest that the immunogenicity of BA.5 and BA.2.75 is different from that of BA.2. Notably, BA.2 (7.1-fold) and BA.2.75 (11-fold) exhibited significant resistance to the sera obtained from BA.5-infected hamsters (Figure 2J). The assay using BA.2-based derivatives showed that G446S contributes to the resistance of BA.2.75 to the humoral immunity induced by BA.2 and BA.5 infections in hamsters (Figures 2I and 2J). Additionally, R493Q is associated with resistance to BA.2-induced hamster immunity (Figure 2I). Because the R493Q substitution is shared with BA.5 (Figures 1B and S1A), this substitution may contribute to the resistance of BA.5 to BA.2-induced immunity (Figure 2I). Finally, we used the sera obtained from hamsters infected with BA.2.75 at 16 d.p.i. for the neutralization assay. Compared with BA.2.75, BA.2 (3.4-fold) and BA.5 (6.4-fold) exhibited significant resistance to the sera obtained from the BA.2.75-infected hamsters (Figure 2K). Altogether, these results suggest that the immunogenicity of BA.2, BA.5, and BA.2.75 is different from each other.
Sensitivity of BA.2.75 to antiviral monoclonal antibodies and drugs
To analyze the sensitivity of BA.2.75 toward therapeutic monoclonal antibodies, we prepared ten monoclonal antibodies and three antibody cocktails. Adintrevimab, bamlanivimab, casirivimab, etesevimab, and imdevimab did not work against BA.2, BA.5, and BA.2.75 (Table 1 ; Figure S2A). Importantly, although regdanvimab, sotrovimab, and tixagevimab did not exhibit antiviral effects against BA.2 and BA.5, these three antibodies were functional against BA.2.75 (Table 1), suggesting that these antibodies can be used for the therapy and prevention of BA.2.75 infection. Consistent with our recent study (Yamasoba et al., 2022b), cilgavimab was less effective against BA.5 than BA.2, and BA.2.75 exhibited 24.4-fold higher resistance to cilgavimab than BA.2 (familywise error rate [FWER] = 0.04) (Table 1). Notably, although bebtelovimab exhibited robust antiviral effects against BA.2 and BA.5 (Yamasoba et al., 2022b), BA.2.75 was significantly more resistant to this antibody than BA.2 (21.2-fold, FWER = 0.01) and BA.5 (25.6-fold, FWER = 0.01) (Table 1).
Table 1.
IC50 values of ten therapeutic monoclonal antibodies against BA.2.75
| B.1.1 | BA.2 | BA.4/5 | BA.2.75 | |
|---|---|---|---|---|
| Adintrevimab | 6.3 ± 1.7 | >2,750 | >2,750 | >2,750 |
| Bamlanivimab | 6.7 ± 1.1 | >4,725 | >4,725 | >4,725 |
| Bebtelovimab | 2.4 ± 0.9 | 1.7 ± 0.8 | 1.3 ± 0.3 | 34 ± 6.9 ∗† |
| Casirivimab | 3.4 ± 1.2 | >5,042 | >5,042 | 2,303 ± 2,570 |
| Cilgavimab | 14 ± 1.7 | 21 ± 7.9 | 305 ± 127 | 479 ± 154 ∗ |
| Etesevimab | 12 ± 1.6 | >4,600 | >4,600 | >4,600 |
| Imdevimab | 8.0 ± 3.1 | >5,000 | >5,000 | >5,000 |
| Regdanvimab | 1.0 ± 0.4 | >4,025 | >4,025 | 42 ± 14 ∗† |
| Sotrovimab | 47 ± 50 | 1,213 ± 224 | 1,149 ± 159 | 240 ± 56 ∗† |
| Tixagevimab | 1.5 ± 0.6 | 3,815 ± 1,032 | >4,375 | 45 ± 8.2 ∗† |
| Ronapreve (casirivimab + imdevimab) | 3.9 ± 2.3 | >5,000 | >5,000 | >5,000 |
| Evusheld (cilgavimab + tixagevimab) | 4.7 ± 1.1 | 42 ± 17 | 586 ± 193 | 113 ± 31 ∗ |
| Etesevimab + bamlanivimab | 8.3 ± 1.0 | >4,600 | >4,600 | >4,600 |
The average 50% inhibitory concentration (IC50; ng/mL) ± 95% confidence interval is shown. Statistical significance was evaluated by the Welch t test with multiple testing corrections by the Holm method. An asterisk (∗) and dagger (†) denote FWER < 0.05 for the BA.2.75 versus BA.2 and BA.2.75 versus BA.4/5 comparisons, respectively. Representative neutralization curves are shown in Figure S2.
Because BA.2.75 exhibited different sensitivities to five monoclonal antibodies, bebtelovimab, cilgavimab, regdanvimab, sotrovimab, and tixgevimab, from BA.2 (Table 1), we assessed the BA.2.75-specific substitution(s) that determine the sensitivity/resistance of BA.2.75. As shown in Figure S2B and Table S3, the resistance of BA.2.75 to bebtelovimab and cilgavimab is determined by the G446S substitution, which is present in the epitope of these two antibodies (Westendorf et al., 2022; Dong et al., 2021). The increased sensitivity of BA.2.75 to sotrovimab is attributed to the D339H substitution, whereas those to regdanvimab and tixagevimab are attributed to the R493Q reversion substitution (Figure S2B; Table S3). These observations are consistent with the fact that D339H and R493Q are present in the known epitopes of these antibodies (Dong et al., 2021; Pinto et al., 2020; Kim et al., 2021).
To evaluate the sensitivity of BA.2.75 to three antiviral drugs, remdesivir, EIDD-1931, and nirmatrelvir, we used clinical isolates of BA.2.75, B.1.1 (Suzuki et al., 2022), BA.2 (Kimura et al., 2022c), and BA.5 (Tamura et al., 2022). These viruses were inoculated into human airway organoids (AOs), a physiologically relevant model (Sano et al., 2022), and treated with three antiviral drugs. As shown in Figure S3 and Table 2 , remdesivir had a stronger antiviral effect against BA.2.75 than against the other variants, B.1.1, BA.2, and BA.5. EIDD-1931 inhibited BA.2 and BA.2.75 more potently than B.1.1 and BA.5. For nirmatrelvir, no differences in antiviral efficacy were observed among the four variants. These results suggest that all three drugs exhibit antiviral effects against BA.2.75, and EIDD-1931 is particularly effective against BA.2.75.
Table 2.
Effects of three antiviral drugs against BA.2.75 in AOs
| EC50 (μM) |
EC50 ratio |
||||||||
|---|---|---|---|---|---|---|---|---|---|
| B.1.1 | BA.2 | BA.5 | BA.2.75 | BA.5/BA.2 | BA.2.75/BA.2 | BA.2/B.1.1 | BA.5/B.1.1 | BA.2.75/B.1.1 | |
| Remdesivir | 1.08 | 1.89 | 1.31 | 0.63 | 0.70 | 0.34 | 1.75 | 1.21 | 0.59 |
| EIDD-1931 | 0.24 | 0.02 | 0.21 | 0.08 | 8.82 | 3.53 | 0.10 | 0.89 | 0.36 |
| Nirmatrelvir | 0.84 | 0.85 | 0.63 | 0.81 | 0.74 | 0.95 | 1.02 | 0.75 | 0.97 |
Representative inhibition curves are shown in Figure S3.
Virological characteristics of BA.2.75 S in vitro
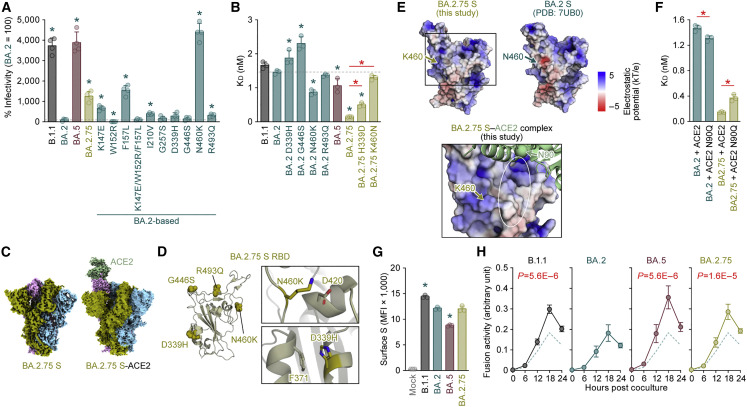
To investigate the virological properties of BA.2.75 S, we measured pseudovirus infectivity. As shown in Figure 3 A, the pseudovirus infectivity of BA.2.75 was significantly (12.5-fold) higher than that of BA.2. To assess the association of TMPRSS2 usage with the increased pseudovirus infectivity of BA.2.75, we used both HEK293-ACE2/TMPRSS2 cells and HEK293-ACE2 cells, on which endogenous surface TMPRSS2 is undetectable (Yamasoba et al., 2022a), as target cells. As shown in Figure S4A, the infectivity of BA.2.75 pseudovirus was not increased by TMPRSS2 expression (Figure S4A), suggesting that TMPRSS2 is not associated with an increase in pseudovirus infectivity of BA.2.75. To determine the substitutions that are responsible for the increased pseudovirus infectivity of BA.2.75, we used a series of BA.2 derivatives that bear the BA.2.75-specific substitutions. Three substitutions in the NTD, K147E, F157L, and I210V, and two substitutions in the RBD, N460K and R493Q, significantly increased infectivity (Figure 3A). Notably, the N460K substitution increased infectivity by 44-fold (Figure 3A). However, a substitution in the NTD, W152R, significantly (8.9-fold) decreased infectivity (Figure 3A). The BA.2 derivative bearing the three substitutions in the NTD in close proximity to each other, K147E, W152R, and F157L, exhibited comparable infectivity with BA.2 (Figure 3A).
Figure 3.
Virological features of BA.2.75 S in vitro
(A) Pseudovirus assay. The percent infectivity compared with that of the virus pseudotyped with the BA.2 S protein are shown.
(B) Binding affinity of the RBD of SARS-CoV-2 S protein to ACE2 by yeast surface display. The KD value indicating the binding affinity of the RBD of the SARS-CoV-2 S protein to soluble ACE2 when expressed on yeast is shown.
(C) Overall cryo-EM map of SARS-CoV-2 BA.2.75 S (closed state 1, left) and BA.2.75 S (open state) bound to ACE2 receptor (right).
(D) Left, position of the four substitutions, D339H, G446S, N460K, and R493Q, in BA.2.75 S RBD. Right, close-up views of the interaction details of the two independent interfaces of BA.2.75 S.
(E) (Top) Electrostatic potential of BA.2.75 RBD (left) and BA.2 RBD (PDB: 7UB0, right) (Lan et al., 2020). The area indicated in square is shown in the bottom panel. (Bottom) BA.2.75 S RBD-human ACE2 complex. BA.2.75 S RBD is shown in surface. Human ACE2 is shown in green cartoon, and a putative N90-glycan of ACE2 is indicated in a circle. Electrostatic potential surface depictions calculated by APBS electrostatic plug-in (Jurrus et al., 2018) of PyMOL. The scale bar shows the electrostatic charge (kT/e).
(F) Binding affinity of the RBD of SARS-CoV-2 S protein to the ACE2 bearing the N90Q substitution by yeast surface display.
(G and H) S-based fusion assay. (G) S protein expression on the cell surface. The summarized data are shown. (H) S-based fusion assay in Calu-3 cells. The recorded fusion activity (arbitrary units) is shown. The dashed green line indicates the results of BA.2.
Assays were performed in quadruplicate (A and H) or triplicate (B, F, and G), and the presented data are expressed as the average ± SD. In (A) and (B), the dashed horizontal lines indicate the value of BA.2. In (A), (B), (F), and (G), each dot indicates the result of an individual replicate. In (A), (B), and (G), statistically significant differences between BA.2 and other variants (∗p < 0.05) were determined by two-sided Student’s t tests. In (B) and (F), red asterisks indicate statistically significant differences (p < 0.05) determined by two-sided Student’s t tests. In (H), statistically significant differences between BA.2 and other variants across time points were determined by multiple regression. FWERs calculated using the Holm method are indicated in the figures.
We next measured the ACE2 binding affinity of the RBDs of BA.2.75 as well as those of BA.2 derivatives bearing D339H, G446S, N460K, and R493Q substitutions by an enhanced yeast surface display system (Zahradník et al., 2021a). Intriguingly, the BA.2.75 RBD showed a strong tight binding with an affinity of 146 ± 6 pM (Figure 3B). Consistent with the results of the pseudovirus assay (Figure 3A), the BA.2 N460K substitution exhibited a significantly increased binding affinity compared with BA.2 (Figure 3B). To assess the impact of N460K in BA.2.75 S, we prepared the BA.2.75 RBD derivative bearing the K460N reversion substitution. As shown in Figure 3B, the BA.2.75 K460N substitution exhibited a significantly reduced binding affinity compared with BA.2.75. These observations suggest that N460K is a critical substitution in the BA.2.75 S to increase viral infectivity by enhancing binding affinity to ACE2.
To reveal the structural effect of the N460K substitution, we performed cryoelectron microscopy (cryo-EM) analysis of the BA.2.75 S ectodomain and the complex of the BA.2.75 S ectodomain and human ACE2, which determined cryo-EM maps with C1 symmetry at resolutions of 2.86 Å (closed 1) and 3.48 Å, respectively (Figure 3C; Table S4). The ectodomain of BA.2.75 S showed three different conformational states (mol ratio, 4.5:2.5:3.0), with two closed states with all RBDs “down,” but these maps were reconstructed with slightly different RBD conformations, and an open state with one RBD “up” and two RBD “down” (Figures S4B and S4C; Table S4). There are four-amino-acid substitutions in the RBD of BA.2.75 S compared with BA.2. G446S and R493Q are located on the receptor-binding motif (RBM), whereas N460K and D339H are located distal to the RBM (Figure 3D, left). N460K forms an intramolecular salt bridge with D420, suggesting that N460K may contribute to RBD folding/flexibility (Figure 3D, upper right). However, at a low resolution, the electron density map of the BA.2.75 S and ACE2 complex showed the same binding mode as that of ACE2 bound to the RBDs of other SARS-CoV-2 variants (Figures 3C right, S4B, and S4C; Table S4). Therefore, this binding mode suggested that N460K is in a position close to the N-linked glycan on the N90 residue of ACE2 (Figure 3E). Interestingly, a previous study showed that the glycan linked to the N90 residue of ACE2 exhibits an inhibitory effect on binding to the S proteins of previous SARS-CoV-2 variants (Chan et al., 2020). To test the possible association of the N-linked glycan on the N90 residue of ACE2 with the increased binding affinity of BA.2.75 S to ACE2, we performed an additional round of binding experiments using the human ACE2 receptor bearing the N90Q substitution, which ablates the N-linked glycosylation. Consistent with the previous study (Chan et al., 2020), the ACE2 N90Q substitution increased the binding affinity of BA.2 S RBD (Figure 3F). On the other hand, the binding affinity of BA.2.75 RBD was significantly reduced by the N90Q substitution of ACE2 (Figure 3F). Altogether, our findings suggest the increased binding affinity of BA.2.75 S is partly attributed to the interaction of BA.2.75 S K460 residue to the N-linked glycan on the N90 residue of ACE2.
In addition to N460K, the structure of the BA.2.75 S ectodomain showed that D339H forms an intramolecular interaction with F371 (Figure 3D, bottom right). This observation suggests that D339H possibly contributes to the improved folding/flexibility of RBD. In particular, D339H requires two nucleotide changes in the codon to occur. Such changes are still relatively rare in the evolution of SARS-CoV-2, reinforcing the importance and corresponding fitness advantage. To analyze the potential impact of this substitution, we additionally prepared the BA.2.75 RBD derivative bearing H339D and measured its affinity. As shown in Figure 3B, the KD value of this derivative was significantly (3-fold) higher than that of the parental BA.2.75, suggesting that D339H appears to indirectly affect the affinity for ACE2.
To further reveal the virological property of BA.2.75 S, we performed a cell-based fusion assay (Kimura et al., 2022b, 2022c; Yamasoba et al., 2022a; Suzuki et al., 2022; Saito et al., 2022; Motozono et al., 2021) using Calu-3 cells as target cells. Flow cytometry analysis showed that the surface expression level of BA.2.75 was comparable with that of BA.2 (Figure 3G). Consistent with our recent study (Kimura et al., 2022c), the fusogenicity of BA.5 was significantly higher than that of BA.2, and notably, BA.2.75 S was also significantly more fusogenic than BA.2 S (Figure 3H). Moreover, a coculture experiment using HEK293-ACE2/TMPRSS2 cells as the target cells (Kimura et al., 2022c; Yamasoba et al., 2022a; Suzuki et al., 2022) showed that the S proteins of BA.5 and BA.2.75 showed significantly increased fusogenicity compared with that of BA.2 S (Figure S4D). Altogether, these results suggest that BA.2.75 S exhibits higher binding affinity to human ACE2 and higher fusogenicity.
Virological characteristics of the BA.2.75 clinical isolate in vitro
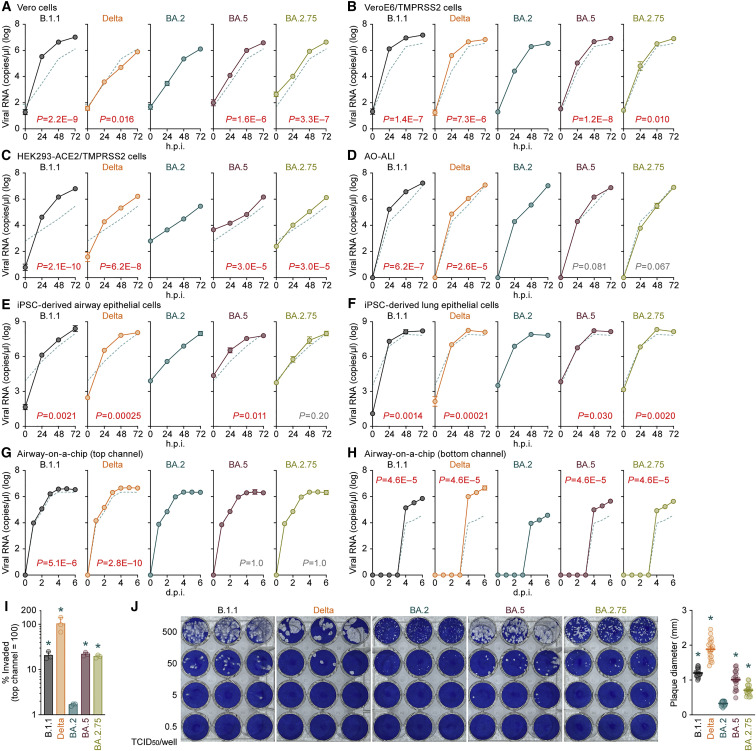
To evaluate the growth capacity of BA.2.75, we inoculated clinical isolates of BA.2.75, B.1.1 (Suzuki et al., 2022), Delta (Saito et al., 2022), BA.2 (Kimura et al., 2022c), and BA.5 (Tamura et al., 2022) in a variety of in vitro cell culture systems. The growth efficacy of B.1.1 and Delta was significantly higher than that of BA.2 in Vero cells (Figure 4 A), VeroE6/TMPRSS2 cells (Figure 4B), HEK293-ACE2/TMPRSS2 cells (Figure 4C), the AO-derived air-liquid interface (AO-ALI) model (Figure 4D), human induced pluripotent stem cell (iPSC)-derived airway epithelial cells (Figure 4E), and lung epithelial cells (Figure 4F). BA.5 replicated more efficiently than BA.2 with statistically significant differences in all cell culture systems except AO-ALI (Figures 4A–4F). The growth efficacy of BA.2.75 was significantly higher than that of BA.2 in Vero cells (Figure 4A), VeroE6/TMPRSS2 cells (Figure 4B), HEK293-ACE2/TMPRSS2 cells (Figure 4C), and iPSC-derived lung epithelial cells (Figure 4F), whereas the growth efficacy of BA.2.75 and BA.2 was comparable in the two airway epithelial cell systems (Figures 4D and 4E).
Figure 4.
Growth capacity of BA.2.75 in vitro
(A–I) Growth kinetics of B.1.1, Delta, BA.2, BA.5, and BA.2.75. Clinical isolates of B.1.1, Delta, BA.2, BA.5, and BA.2.75 were inoculated into Vero cells (A), VeroE6/TMPRSS2 cells (B), HEK293-ACE2/TMPRSS2 cells (C), AO-ALI (D), iPSC-derived airway epithelial cells (E), iPSC-derived lung epithelial cells (F), and an airway-on-a-chip system (G and H; see also Figure S3C). The copy numbers of viral RNA in the culture supernatant (A–C), the apical sides of cultures (D–F), and the top (G) and bottom (H) channels of an airway-on-a-chip were routinely quantified by RT-qPCR. The dashed green line in each panel indicates the results of BA.2. In (I), the percentage of viral RNA load in the bottom channel per top channel at 6 d.p.i. (i.e., % invaded virus from the top channel to the bottom channel) is shown.
(J) Plaque assay. Representative panels (left) and a summary of the recorded plaque diameters (20 plaques per virus) (right) are shown.
Assays were performed in quadruplicate, and the presented data are expressed as the average ± SD. In (A)–(H), statistically significant differences between BA.2 and the other variants across time points were determined by multiple regression. FWERs calculated using the Holm method are indicated in the figures. In (I) and (J) (right), statistically significant differences versus BA.2 (∗p < 0.05) were determined by two-sided Mann-Whitney U tests. Each dot indicates the result of an individual replicate.
See also Figure S4.
To evaluate the effect of BA.2.75 on the airway epithelial and endothelial barriers, we used airway-on-a-chips system (Figure S4E; Hashimoto et al., 2022). By measuring the amount of virus that invades from the top channel (Figure 4G) to the bottom channel (Figure 4H), we could evaluate the ability of viruses to disrupt the airway epithelial and endothelial barriers. Notably, the amount of virus that invaded the blood vessel channel of BA.2.75-, BA.5-, and B.1.1-infected airway-on-chips was significantly higher than that of BA.2-infected airway-on-chips (Figure 4I). These results suggest that BA.2.75 exhibits more severe airway epithelial and endothelial barrier disruption than BA.2.
To further address the fusogenic capacity of BA.2.75, we performed a plaque assay using VeroE6/TMPRSS2 cells. Consistent with our previous studies with a Delta isolate (Saito et al., 2022) as well as the recombinant SARS-CoV-2 bearing the B.1.1 S (Yamasoba et al., 2022a), BA.2 S (Yamasoba et al., 2022a), and BA.5 S (Kimura et al., 2022c), the plaques formed by the infections of clinical isolates of B.1.1, Delta, and BA.5 were significantly larger than those formed by the infection of BA.2 (Figure 4J). Notably, BA.2.75 infection also resulted in significantly larger plaques than BA.2 infection (Figure 4J). Together with the results of the cell-based fusion assay (Figure 3H) and airway-on-a-chip infection experiments (Figures 4G–4I), these observations suggest that BA.2.75 is more fusogenic than BA.2, and the fusogenicity of BA.2.75 is comparable with that of BA.5.
Virological characteristics of BA.2.75 in vivo
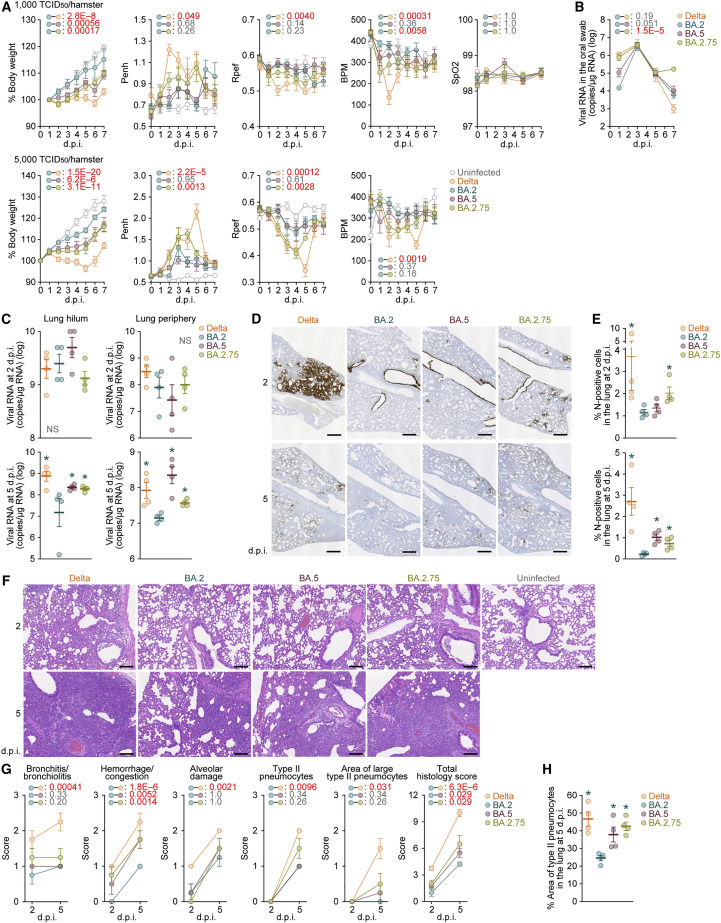
As we proposed in our prior studies (Kimura et al., 2022c; Yamasoba et al., 2022a; Suzuki et al., 2022; Saito et al., 2022), the fusogenicity of the S proteins of SARS-CoV-2 variants is closely associated with the intrinsic pathogenicity in an experimental hamster model. Given that BA.2.75 is more fusogenic than BA.2 in the in vitro cell culture systems (Figures 3 and 4), it is hypothesized that BA.2.75 is intrinsically more pathogenic than BA.2. To address this possibility, we intranasally inoculated a BA.2.75 isolate into hamsters. As controls, we also used clinical isolates of Delta, BA.2, and BA.5. Although we followed our established experimental protocol (Kimura et al., 2022c; Yamasoba et al., 2022a; Suzuki et al., 2022; Saito et al., 2022), the viral titers of clinical isolates of Omicron subvariants were relatively low. Therefore, we set out to conduct animal experiments in this study with relatively lower inocula (1,000 50% tissue culture infectious dose [TCID50] [Figure 5 A, top] or 5,000 TCID50 [Figure 5A, bottom] per hamster) than our previous studies (10,000 TCID50 per hamster) (Kimura et al., 2022c; Yamasoba et al., 2022a; Suzuki et al., 2022; Saito et al., 2022). Nevertheless, consistent with our previous study (Saito et al., 2022), Delta infection resulted in weight loss in the infection group with a higher inoculum (Figure 5A, bottom). At both challenge doses, the body weights of the BA.5- and BA.2.75-infected hamsters were significantly lower than those of the BA.2-infected hamsters (Figure 5A).
Figure 5.
Virological characteristics of BA.2.75 in vivo
Syrian hamsters were intranasally inoculated with Delta, BA.2, BA.5, and BA.2.75. Two different doses of inoculum (1,000 TCID50/hamster [A, top and B–G] or 5,000 TCID50/hamster [A, bottom]) were used. Six hamsters per infection group were used to routinely measure the respective parameters (A and B). Four hamsters per infection group at a lower inoculum (1,000 TCID50/hamster) were euthanized at 2 and 5 d.p.i. and used for virological and pathological analysis (C–G).
(A) Body weight, Penh, Rpef, BPM, and SpO2 values of infected hamsters (n = 6 each). The results at a low inoculum (1,000 TCID50/hamster) and a high inoculum (5,000 TCID50/hamster) are shown in the top and bottom panels, respectively.
(B) Viral RNA loads in the oral swab (n = 6 each).
(C) Viral RNA loads in the lung hilum (left) and lung periphery (right) of infected hamsters (n = 4 each) at 2 d.p.i. (top) and 5 d.p.i. (bottom).
(D and E) IHC of the viral N protein in the lungs at 2 d.p.i. (top) and 5 d.p.i. (bottom) of all infected hamsters. (D) Representative figures. N-positive cells are shown in brown. (E) Percentage of N-positive cells in whole lung lobes (n = 4 each). The raw data are shown in Figures S5B and S5C.
(F and G) (F) H&E staining of the lungs of infected hamsters. Representative figures are shown. Uninfected lung alveolar space and bronchioles are also shown. (G) Histopathological scoring of lung lesions (n = 4 each). Representative pathological features are reported in our previous studies (Kimura et al., 2022c; Yamasoba et al., 2022a; Suzuki et al., 2022; Saito et al., 2022).
(H) Type II pneumocytes in the lungs of infected hamsters. The percentage of the area of type II pneumocytes in the lung at 5 d.p.i. is summarized. The raw data are shown in Figure S5D.
In (A)–(C), (E), (G), and (H), data are presented as the average ± SEM. In (C), (E), and (H), each dot indicates the result of an individual hamster.
In (A), (B), and (G), statistically significant differences between BA.2 and other variants across time points were determined by multiple regression. In (A), the 0 d.p.i. data were excluded from the analyses. FWERs calculated using the Holm method are indicated in the figures.
In (C), (E), and (G), the statistically significant differences between BA.2 and other variants were determined by a two-sided Mann-Whitney U test.
In (D) and (F), each panel shows a representative result from an individual infected hamster. Scale bars: 500 μm in (D) and 200 μm in (F).
See also Figure S5.
We then analyzed the pulmonary function of infected hamsters as reflected by three parameters, enhanced pause (Penh), the ratio of time to peak expiratory flow relative to the total expiratory time (Rpef), and breath per minute (BPM), which are surrogate markers for bronchoconstriction or airway obstruction. Subcutaneous oxygen saturation (SpO2) was routinely measured in the group with the lower inoculum (Figure 5A, top). Although the SpO2 values were comparable among the five groups, Delta infection resulted in significant differences in the other three respiratory parameters compared with BA.2 (Figure 5A), suggesting that Delta is more pathogenic than BA.2. In the group with a lower inoculum, although the values of Penh and Rpef of the hamsters infected with BA.2, BA.5, and BA.2.75 were comparable, the BPM value of BA.2.75 was significantly lower than that of BA.2 (Figure 5A, top). In the group with a higher inoculum, although the values of Penh and Rpef of the BA.5-infected hamsters were comparable with those of the BA.2-infected hamsters, these two values indicated that BA.2.75 infection leads to more severe disorder compared with BA.2 infection with statistical significance (Figure 5A, bottom). These results suggest that BA.2.75 is more pathogenic than BA.2.
To address the viral spread in infected hamsters, we routinely measured the viral RNA load in the oral swab. Although the viral RNA loads of the hamsters infected with Delta, BA.2, and BA.5 were comparable, the viral load in the swabs of the BA.2.75-infected hamsters was relatively highly maintained by 7 d.p.i. and was significantly higher than that of the BA.2-infected hamsters (Figure 5B). To address the possibility that BA.2.75 more efficiently spread in the respiratory tissues, we collected the lungs of infected hamsters at 2 and 5 d.p.i., and the collected tissues were separated into the hilum and periphery regions. Although the viral RNA loads in both the hilum and periphery of the four infection groups were comparable at 2 d.p.i. (Figure 5C, top), those of the hamsters infected with Delta, BA.5, and BA.2.75 were significantly higher than those infected with BA.2 at 5 d.p.i. (Figure 5C, bottom).
To further address virus spread in the respiratory tissues, we performed immunohistochemical (IHC) analysis targeting viral nucleocapsid (N) protein. Similar to our previous studies (Kimura et al., 2022c; Yamasoba et al., 2022a; Suzuki et al., 2022), epithelial cells in the upper tracheae of infected hamsters were sporadically positive for viral N protein at 2 d.p.i., but there were no significant differences among the four viruses, including BA.2.75 (Figure S5A). In the alveolar space around the bronchi/bronchioles at 2 d.p.i., N-positive cells were detected in Delta-infected hamsters. However, the N proteins strongly remained in the lobar bronchi in BA.5- and BA.2.75-infected hamsters (Figures 5D, top and S5B). Although few N-positive cells were detected in the alveolar space of the BA.2- and BA.5-infected hamsters, the N positivity spread into the alveolar space in the BA.2.75-infected hamsters (Figures 5D, top and S5B). The quantification of the N-positive area in four lung lobes at 2 d.p.i. (Figure S5B) showed that the N-positive areas of the Delta- and BA.2.75-infected hamsters were significantly greater than those of the BA.2-infected hamsters (Figure 5E, top). At 5 d.p.i., although the N-positive cells were barely detected in the lungs infected with BA.2, a few N-positive cells were detected in the peripheral alveolar space in the Delta, BA.5, and BA.2.75 groups (Figures 5D, bottom and S5C). The quantification of the N-positive area in the four lung lobes at 5 d.p.i. (Figure S5C) further showed that the N-positive areas of the Delta-, BA.5-, and BA.2.75-infected hamsters were significantly greater than those of the BA.2-infected hamsters (Figure 5E, bottom). These data suggest that BA.2 infects a smaller portion of the bronchial/bronchiolar epithelium because it is less efficiently transmitted to neighboring epithelial cells. In contrast, BA.5 and BA.2.75 infections seemed to persist in the bronchial/bronchiolar epithelium, and in particular, BA.2.75 invaded the alveolar space more efficiently than BA.5 at the early stage of infection. Altogether, the IHC data suggest that among Omicron subvariants, BA.2.75 more efficiently spread into the alveolar space than BA.2 and BA.5, with persistent infection in the bronchi/bronchioles.
Pathogenicity of BA.2.75
To investigate the intrinsic pathogenicity of BA.2.75, we analyzed the formalin-fixed right lungs of infected hamsters at 2 and 5 d.p.i. by carefully identifying the four lobules and main bronchus and lobar bronchi sectioning each lobe along with the bronchial branches. Histopathological scoring was performed according to the criteria described in our previous studies (Kimura et al., 2022c; Yamasoba et al., 2022a; Suzuki et al., 2022; Saito et al., 2022). Consistent with our previous studies (Suzuki et al., 2022; Saito et al., 2022), all five parameters as well as the total score of the Delta-infected hamsters were significantly higher than those of the BA.2-infected hamsters (Figures 5F and 5G). When we compared the histopathological scores of Omicron subvariants, the scores indicating hemorrhage or congestion and total histology scores of BA.5 and BA.2.75 were significantly greater than those of BA.2 (Figures 5F and 5G). Similar to our recent studies (Kimura et al., 2022c; Tamura et al., 2022), BA.5 is intrinsically more pathogenic than BA.2, and notably, our results suggest that BA.2.75 exhibits more significant inflammation than BA.2. For determination of the area of pneumonia, the inflammatory area was termed the area of type II pneumocytes and was morphometrically analyzed (Figure S5D). As summarized in Figure 5H, at 5 d.p.i., the percentages of the area of type II pneumocytes of Delta, BA.5, and BA.2.75 were significantly higher than that of BA.2. Altogether, these findings suggest that BA.2.75 infection intrinsically induces greater inflammation and exhibits higher pathogenicity than BA.2.
Discussion
Here, we characterized the properties of the Omicron BA.2.75 variant, such as the growth rate in the human population, resistance to antiviral humoral immunity and antiviral drugs, functions of the S protein in vitro, and intrinsic pathogenicity. In terms of the emergence geography and phylogeny, BA.5 and BA.2.75 emerged independently. Nevertheless, the results of the cell-based fusion assay, airway-on-a-chip assay, and plaque assay suggested that both BA.5 and BA.2.75 acquired higher fusogenicity after divergence from BA.2. Our data, including a recent study (Kimura et al., 2022c), suggest that the critical substitution responsible for the higher fusogenicity of the BA.5 and BA.2.75 S proteins are different: the L452R substitution for BA.5 S and the D339H/N460K substitutions for BA.2.75 S.
In our previous studies focusing on Delta (Saito et al., 2022), Omicron BA.1 (Suzuki et al., 2022), BA.2 (Yamasoba et al., 2022a), and BA.5 (Kimura et al., 2022c), we proposed a close association between S-mediated fusogenicity in vitro and pathogenicity in a hamster model. Consistent with our hypothesis, we demonstrated that compared with BA.2, BA.2.75 exhibits higher fusogenicity in vitro and efficient viral spread in the lungs of infected hamsters, which leads to enhanced inflammation in the lung and higher pathogenicity in vivo. However, viral load is not necessarily associated with pathogenicity. Moreover, in vitro experiments using a variety of cell culture systems showed that BA.2.75 replicates more efficiently than BA.2 in alveolar epithelial cells but not in airway epithelial cells. Altogether, our results suggest that BA.2.75 exhibits higher fusogenicity and pathogenicity via evolution of its S protein independent of BA.5.
Using hamster sera, we demonstrated that the immunogenicity of BA.5 and BA.2.75 is different from each other, whereas BA.2.75 and BA.5 are the descendants of BA.2. However, the antiviral effect of BA.5 breakthrough infection sera is comparable between BA.2 and BA.2.75. In this regard, a recent study showed that BA.2.75 exhibits a pronounced resistance to BA.5 breakthrough infection sera compared with BA.2 (Cao et al., 2022). This discrepancy may be explained by the type of vaccine used: a cohort in the present study was vaccinated with mRNA vaccines (BNT162b2 or mRNA-1273), whereas the cohort in the previous study (Cao et al., 2022) was vaccinated with an inactivated vaccine (CoronaVac). In fact, another study on a cohort of mRNA-1273 vaccination showed results consistent with ours (Shen et al., 2022). These observations suggest that the basal immunity induced by vaccination is different by the type of vaccine used, and thereby, the immunity induced by breakthrough BA.5 infection is different.
We showed that G446S in BA.2.75 S was closely associated with resistance to various antisera: 4-dose vaccinated sera, breakthrough BA.5 infection sera, and BA.2- and BA.5-infected hamster sera. Additionally, G446S was responsible for the increased resistance to therapeutic antibodies, such as bebtelovimab and cilgavimab. These results suggest that G446S is critical to evade diverse antibodies. Importantly, our receptor-binding assay showed that G446S significantly decreased the binding affinity of BA.2 RBD to human ACE2, and this effect was compensated by the other substitutions in the BA.2.75 S RBD, particularly by N460K. Altogether, these results suggest that G446S was acquired to evade antiviral immunity, and the other substitutions in the RBD, such as N460K, were acquired likely to compensate for the ACE2 binding affinity reduced by G446S. This is reminiscent of our recent study focusing on Omicron BA.5 (Kimura et al., 2022c): in the case of the BA.5 S, F486V resulted in immune evasion and decreased binding affinity to ACE2, whereas L452R compensated the decreased ACE2 binding affinity. Although the mechanisms of action are different between BA.5 and BA.2.75, the acquisition of two types of substitutions—one leads to immune evasion, which tends to decrease ACE2 affinity, and the other leads to increased ACE2 affinity for compensation—might be a common strategy of SARS-CoV-2 evolution.
Our investigation using viral genome surveillance data reported from India suggested that BA.2.75 has the potential to outcompete BA.2 as well as BA.5, the most predominant variant in the world as of August 2022. Following the worldwide spread of BA.5, it is probable that the number of individuals infected with BA.5 will increase. Additionally, we showed that the intrinsic pathogenicity of BA.2.75 in hamsters is comparable with that of BA.5 and higher than that of BA.2. Since a recent study showed that the hospitalization risk of BA.5 was significantly higher than that of BA.2 in the once-boosted vaccinated population (Kislaya et al., 2022), it is not unreasonable to infer that the intrinsic pathogenicity in infected hamsters reflects the severity and outcome in infected humans to a meaningful extent.
In summary, our multisystem investigations revealed that the growth rate in the human population, fusogenicity, and intrinsic pathogenicity of BA.2.75 are greater than those of BA.2. These features of BA.2.75 suggest the potential risk of this variant to global health. Since BA.2.75 shows significantly higher Re than BA.2 and BA.5 in India, this variant will probably transmit to and outcompete BA.2 and BA.5 in some countries other than India in the near future. For the assessment of the potential risk of BA.2.75 to global health, this variant should be under monitoring carefully and continuously through worldwide cooperation of in-depth viral genomic surveillance.
Consortia
Mai Kishimoto, Marie Kato, Zannatul Ferdous, Hiromi Mouri, Kenji Shishido, Naoko Misawa, Mai Suganami, Mika Chiba, Ryo Yoshimura, So Nakagawa, Jiaqi Wu, Yasuhiro Kazuma, Ryosuke Nomura, Yoshihito Horisawa, Yusuke Tashiro, Yugo Kawai, Ryoko Kawabata, Otowa Takahashi, Kimiko Ichihara, Chihiro Motozono, Yuri L. Tanaka, Erika P. Butlertanaka, Rina Hashimoto, Tateki Suzuki, Kanako Kimura, Jiei Sasaki, and Yukari Nakajima
STAR★Methods
Key resources table
| REAGENT or RESOURCE | SOURCE | IDENTIFIER |
|---|---|---|
| Antibodies | ||
| Adintrevimab | This study | N/A |
| Bamlanivimab | Yamasoba et al., 2022b | N/A |
| Bebtelovimab | Yamasoba et al., 2022b | N/A |
| Casirivimab | Yamasoba et al., 2022b | N/A |
| Cilgavimab | Yamasoba et al., 2022b | N/A |
| Etesevimab | Yamasoba et al., 2022b | N/A |
| Imdevimab | Yamasoba et al., 2022b | N/A |
| Regdanvimab | This study | N/A |
| Sotrovimab | Yamasoba et al., 2022b | N/A |
| Tixagevimab | Yamasoba et al., 2022b | N/A |
| Rabbit anti-SARS-CoV-2 S S1/S2 polyclonal antibody | Thermo Fisher Scientific | Cat# PA5-112048; RRID: AB_2866784 |
| Normal rabbit IgG | Southern Biotech | Cat# 0111-01; RRID: AB_2732899 |
| APC-conjugated goat anti-rabbit IgG polyclonal antibody | Jackson ImmunoResearch | Cat# 111-136-144; RRID: AB_2337987 |
| Bacterial and virus strains | ||
| SARS-CoV-2 B.1.1 (strain TKYE610670) | Saito et al., 2022; Suzuki et al., 2022 | N/A |
| SARS-CoV-2 Delta (strain TKYTK1734) | Saito et al., 2022; Suzuki et al., 2022 | N/A |
| SARS-CoV-2 BA.2 (strain TY40-385) | Kimura et al., 2022b; Tamura et al., 2022 | N/A |
| SARS-CoV-2 BA.5 (strain TKYS14631) | Kimura et al., 2022b; Tamura et al., 2022 | N/A |
| SARS-CoV-2 BA.2.75 (strain TY41-716) | This study | N/A |
| Biological samples | ||
| Human sera | This study | N/A |
| Human airway epithelial cells derived from human iPSCs | Yamamoto et al., 2017 | N/A |
| Human alveolar epithelial cells derived from human iPSCs | Yamamoto et al., 2017 | N/A |
| Human airway organoids | Sano et al., 2022 | N/A |
| Airway-on-a-chips | Hashimoto et al., 2022 | N/A |
| Chemicals, peptides, and recombinant proteins | ||
| TransIT-LT1 | Takara | Cat# MIR2300 |
| Recombinant RNase inhibitor | Takara | Cat# 2313B |
| Carboxymethyl cellulose | Wako | Cat# 039-01335 |
| 4% Paraformaldehyde in PBS | Nacalai Tesque | Cat# 09154-85 |
| Methylene blue | Nacalai Tesque | Cat# 22412-14 |
| Fetal bovine serum | Sigma-Aldrich | Cat# 172012-500ML |
| Poly-L-lysine | Sigma | Cat# P4832 |
| Hoechst 33342 | Thermo Fisher Scientific | Cat# H3570 |
| Fluoromount-G | Southern Biotech | Cat# 0100-01 |
| Penicillin-streptomycin | Sigma-Aldrich | Cat# P4333-100ML |
| DMEM (high glucose) | Sigma-Aldrich | Cat# 6429-500ML |
| DMEM (high glucose) | Nacalai Tesque | Cat# 08458-16 |
| DMEM (low glucose) | Wako | Cat# 041-29775 |
| EMEM | Sigma-Aldrich | Cat# M4655-500ML |
| EMEM | Wako | Cat# 056-08385 |
| EGM-2-MV medium | Lonza | Cat# CC-3202 |
| SD-CAA medium | Zahradník et al., 2021a | N/A |
| 1/9 medium | Zahradník et al., 2021a | N/A |
| Glucose | Sigma-Aldrich | Cat# G8270 |
| Galactose | Sigma-Aldrich | Cat# G0750 |
| Yeast nitrogen base | Sigma-Aldrich | Cat# Y0626 |
| Casamino acids | Sigma-Aldrich | Cat# 2240 |
| Sodium phosphate dibasic | Sigma-Aldrich | Cat# S9763 |
| Sodium phosphate monobasic | Sigma-Aldrich | Cat# S3139 |
| PneumaCult ALI medium | STEMCELL Technologies | Cat# ST-05001 |
| Heparin | Nacalai Tesque | Cat# 17513-96 |
| Y-27632 | LC Laboratories | Cat# Y-5301 |
| Hydrocortisone | Sigma-Aldrich | Cat# H0135 |
| Puromycin | InvivoGen | Cat# ant-pr-1 |
| Hygromycin | Nacalai Tesque | Cat# 09287-84 |
| G418 | Nacalai Tesque | Cat# G8168-10ML |
| KpnI | New England Biolab | Cat# R0142S |
| NotI | New England Biolab | Cat# R1089S |
| PEI Max | Polysciences | Cat# 24765-1 |
| Fibronectin | Sigma-Aldrich | Cat# F1141 |
| Opti-MEM | Thermo Fisher Scientific | Cat# 11058021 |
| Triton X-100 | Nacalai Tesque | Cat# 35501-15 |
| EnduRen live cell substrate | Promega | Cat# E6481 |
| Hoechst 33342 | Thermo Fisher Scientific | Cat# H3570 |
| Fluoromount-G | SouthernBiotech | Cat# 0100-01 |
| Dexamethasone | Sigma-Aldrich | Cat# D4902 |
| KGF | PeproTech | Cat# 100-19 |
| 8-Br-cAMP | Biolog | Cat# B007 |
| 3-Isobutyl 1-methylxanthine (IBMX) | Wako | Cat# 095-03413 |
| CHIR99021 | Axon Medchem | Cat# 1386 |
| SB431542 | Wako | Cat# 198-16543 |
| Soluble human ACE2 (residues 18-618 for binding assay) | Yamasoba et al., 2022b | N/A |
| Soluble human ACE2 (residues 19-617 for structural analysis) | Yamasoba et al., 2022b | N/A |
| SARS-CoV-2 B.1.1 RBD | Kimura et al., 2022a; Motozono et al., 2021 | N/A |
| SARS-CoV-2 BA.1 RBD | This study | N/A |
| SARS-CoV-2 BA.2 RBD | This study | N/A |
| SARS-CoV-2 BA.5 RBD | This study | N/A |
| SARS-CoV-2 BA.2.75 RBD | This study | N/A |
| SARS-CoV-2 BA.2.75 S ectodomain | This study | N/A |
| Bilirubin | Sigma-Aldrich | Cat# 14370-1G |
| cOmplete His-Tag Purification Resin | Roche | Cat# 5893682001 |
| Medetomidine hydrochloride (Domitor®) | Nippon Zenyaku Kogyo | N/A |
| Midazolam | Fujifilm Wako | Cat# 135-13791 |
| Butorphanol (Vetorphale®) | Meiji Seika Pharma | N/A |
| Alphaxaone (Alfaxan®) | Jurox | N/A |
| Isoflurane | Sumitomo Dainippon Pharma | N/A |
| EnVision FLEX target retrieval solution high pH | Agilent | Cat# K8004 |
| Matrigel growth factor reduced basement membrane matrix | Corning | Cat# 354230 |
| poly-L-lysine | Sigma-Aldrich | Cat# P4832 |
| Remdesivir | Clinisciences | Cat# A17170 |
| EIDD-1931 | Cell Signalling Technology | Cat# 81178S |
| Nirmatrelvir | MedChemExpress | Cat# HY-138687 |
| n-octyl-β-D-glucoside | Dojindo | Cat# O001 |
| Critical commercial assays | ||
| QIAamp viral RNA mini kit | Qiagen | Cat# 52906 |
| NEBNext Ultra RNA Library Prep Kit for Illumina | New England Biolabs | Cat# E7530 |
| MiSeq reagent kit v3 | Illumina | Cat# MS-102-3001 |
| One Step TB Green PrimeScript PLUS RT-PCR kit | Takara | Cat# RR096A |
| SARS-CoV-2 direct detection RT-qPCR kit | Takara | Cat# RC300A |
| Nano Glo HiBiT lytic detection system | Promega | Cat# N3040 |
| Bright-Glo luciferase assay system | Promega | Cat# E2650 |
| One-Glo luciferase assay system | Promega | Cat# E6130 |
| Cell counting kit-8 | Dojindo | Cat# CK04-11 |
| Deposited data | ||
| Viral genome sequencing data of working viral stocks (see also Table S6) | This paper | SRA: PRJDB14324 (https://www.ncbi.nlm.nih.gov/sra) |
| Cryo-EM map of SARS-CoV-2 BA.2.75 S (closed state 1) | This study | EMD-34221 |
| Cryo-EM map of SARS-CoV-2 BA.2.75 S (closed state 2) | This study | EMD-34222 |
| Cryo-EM map of SARS-CoV-2 BA.2.75 S (1-up state) | This study | EMD-34223 |
| Cryo-EM map of SARS-CoV-2 BA.2.75 S in complex with ACE2 | This study | EMD-34224. |
| Structure of the SARS-CoV-2 BA.2.75 S (close-1 state) | This study | PDB: 8GS6 |
| Experimental models: Cell lines | ||
| Human: HEK293T cells | ATCC | CRL-3216 |
| Human: HEK293 cells | ATCC | CRL-1573 |
| Human: HEK293-ACE2 cells | Motozono et al., 2021 | N/A |
| Human: HEK293-ACE2/TMPRSS2 cells | Motozono et al., 2021 | N/A |
| Human: HEK293S GnTI(-) cells | Reeves et al., 2002 | N/A |
| Human: HOS-ACE2/TMPRSS2 cells | Ferreira et al., 2021; Ozono et al., 2021 | N/A |
| Human: Calu-3/DSP1-7 cells | Yamamoto et al., 2020 | N/A |
| African green monkey (Chlorocebus sabaeus): Vero cells | JCRB Cell Bank | JCRB0111 |
| African green monkey (Chlorocebus sabaeus): VeroE6/TMPRSS2 cells | JCRB Cell Bank | JCRB1819 |
| Yeast (Saccharomyces cerevisiae): strain EBY100 | ATCC | MYA-4941 |
| Experimental models: Organisms/strains | ||
| Human lung microvascular endothelial cells (HMVEC-L) | Lonza | Cat# CC-2527 |
| Normal human bronchial epithelial cells (NHBE) | Lonza | Cat# CC-2540 |
| Slc:Syrian hamsters (male, 4 weeks old) | Japan SLC Inc. | http://www.jslc.co.jp/pdf/hamster/2020/028_Slc_Syrian.pdf |
| Oligonucleotides | ||
| Primers for the construction of plasmids expressing the codon-optimized S proteins of BA.2-bearing variants, see Table S5 | This study | N/A |
| RT-qPCR, forward: AGC CTC TTC TCG TTC CTC ATC AC | Kimura et al., 2022b, 2022c; Meng et al., 2022; Motozono et al., 2021; Saito et al., 2022; Suzuki et al., 2022; Yamasoba et al., 2022b | N/A |
| RT-qPCR, reverse: CCG CCA TTG CCA GCC ATT C | Kimura et al., 2022b, 2022c; Meng et al., 2022; Motozono et al., 2021; Saito et al., 2022; Suzuki et al., 2022; Yamasoba et al., 2022b | N/A |
| Primers for the construction of yeast-optimized SARS-CoV-2 BA.2 RBD expression plasmid, see Table S5 | This study | N/A |
| Recombinant DNA | ||
| Plasmid: pCAGGS | Niwa et al., 1991 | N/A |
| Plasmid: psPAX2-IN/HiBiT | Ozono et al., 2020 | N/A |
| Plasmid: pWPI-Luc2 | Ozono et al., 2020 | N/A |
| Plasmid: pJYDC1 | Addgene | Cat# 162458 |
| Plasmid: pHL-sec | Addgene | Cat# 99845 |
| Plasmid: pDSP1-7 | Kondo et al., 2011 | N/A |
| Plasmid: pDSP8-11 | Kondo et al., 2011 | N/A |
| Plasmid: pC-B.1.1 S | Motozono et al., 2021; Ozono et al., 2021 | N/A |
| Plasmid: pC-BA.2 S | Yamasoba et al., 2022b | N/A |
| Plasmid: pC-BA.4/5 S | Kimura et al., 2022c | N/A |
| Plasmid: pC-BA.2.75 S | Yamasoba et al., 2022a | N/A |
| Plasmid: pC-BA.2 K147E S | This study | N/A |
| Plasmid: pC-BA.2 W152R S | This study | N/A |
| Plasmid: pC-BA.2 F157L S | This study | N/A |
| Plasmid: pC-BA.2 K147E/W152R/F157L S | This study | N/A |
| Plasmid: pC-BA.2 I210V S | This study | N/A |
| Plasmid: pC-BA.2 G257S S | This study | N/A |
| Plasmid: pC-BA.2 D339H S | This study | N/A |
| Plasmid: pC-BA.2 G446S S | This study | N/A |
| Plasmid: pC-BA.2 N460K S | This study | N/A |
| Plasmid: pC-BA.2 R439Q S | This study | N/A |
| Plasmid: pHLsec BA.2.75 S ectodomain | This study | N/A |
| Plasmid: pHLsec soluble human ACE2 | This study | N/A |
| Software and algorithms | ||
| fastp v0.21.0 | Chen et al., 2018 | https://github.com/OpenGene/fastp |
| BWA-MEM v0.7.17 | Li and Durbin, 2009 | http://bio-bwa.sourceforge.net |
| SAMtools v1.9 | Li et al., 2009 | http://www.htslib.org |
| snpEff v5.0e | Cingolani et al., 2012 | http://pcingola.github.io/SnpEff |
| roblanf/sarscov2phylo: 13-11-20 (GISAID phylogenetic analysis pipeline) | GitHub | https://github.com/roblanf/sarscov2phylo |
| Minimap2 v2.17 | Li, 2018 | https://github.com/lh3/minimap2 |
| trimAl v1.2 | Capella-Gutiérrez et al., 2009 | http://trimal.cgenomics.org |
| RAxML v8.2.12 | Stamatakis, 2014 | https://cme.h-its.org/exelixis/web/software/raxml |
| CmdStan v2.28.1 | The Stan Development Team | https://mc-stan.org |
| CmdStanr v0.4.0 | The Stan Development Team | https://mc-stan.org/cmdstanr/ |
| R v4.1.3 | The R Foundation | https://www.r-project.org/ |
| Sequencher v5.1 software | Gene Codes Corporation | N/A |
| In-house scripts | This study | https://github.com/TheSatoLab/Omicron_BA.2.75 |
| Prism 9 software v9.1.1 | GraphPad Software | https://www.graphpad.com/scientific-software/prism/ |
| Fiji software v2.2.0 | ImageJ | https://fiji.sc |
| FlowJo software v10.7.1 | BD Biosciences | https://www.flowjo.com/solutions/flowjo |
| Python v3.7 | Python Software Foundation | https://www.python.org |
| FinePointe Station and Review softwares v2.9.2.12849 | DSI | https://www.datasci.com/products/software/finepointe-software |
| NDP.scan software v3.2.4 | Hamamatsu Photonics | https://nanozoomer.hamamatsu.com/jp/en/why_nanozoomer/scan.html |
| PyMOL v2.1.1 or v2.3.3 | Schrödinger | https://pymol.org/2/ |
| CytExpert software v2.4 | Beckman Coulter | https://www.beckman.pt/flow-cytometry/research-flow-cytometers/cytoflex/software |
| ColabFold | Mirdita et al., 2022 | https://colab.research.google.com/github/sokrypton/ColabFold/blob/main/AlphaFold2.ipynb#scrollTo=kOblAo-xetgx |
| MolProbity | Williams et al., 2018 | http://molprobity.biochem.duke.edu/ |
| PHENIX v1.20 | Adams et al., 2010 | http://www.phenix-online.org/ |
| Coot v0.9.6 | Emsley and Cowtan, 2004 | https://www2.mrc-lmb.cam.ac.uk/personal/pemsley/coot/o |
| EPU v2.14 | Thermo Fisher Scientific | N/A |
| cryoSPARC v3.3.1 | Punjani et al., 2017 | https://cryosparc.com/ |
| UCSF Chimera v1.15 | Pettersen et al., 2004 | https://www.cgl.ucsf.edu/chimera/ |
| UCSF Chimera X v1.1 | Goddard et al., 2018 | https://www.cgl.ucsf.edu/chimerax/ |
| Other | ||
| Centro XS3 LB960 | Berthhold Technologies | N/A |
| GloMax explorer multimode microplate reader 3500 | Promega | N/A |
| FACS Canto II | BD Biosciences | N/A |
| GISAID database | Khare et al., 2021 | https://www.gisaid.org/ |
| 24-well Cell Culture Insert | Falcon | Cat# 353104 |
| 96-well black plate | PerkinElmer | Cat# 6005225 |
| 3,3’-diaminobenzidine tetrahydrochloride | Dako | Cat# DM827 |
| MAS-GP-coated glass slides | Matsunami Glass | Cat# S9901 |
| A1Rsi Confocal Microscope | Nikon | N/A |
| QuantStudio 1 Real-Time PCR system | Thermo Fisher Scientific | N/A |
| QuantStudio 3 Real-Time PCR system | Thermo Fisher Scientific | N/A |
| QuantStudio 5 Real-Time PCR system | Thermo Fisher Scientific | N/A |
| StepOne Plus Real-Time PCR system | Thermo Fisher Scientific | N/A |
| Thermal Cycler Dice Real Time System III | Takara | N/A |
| CFX Connect Real-Time PCR Detection system | Bio-Rad | N/A |
| Eco Real-Time PCR System | Illumina | N/A |
| qTOWER3 G Real-Time System | Analytik Jena | N/A |
| 7500 Real-Time PCR System | Thermo Fisher Scientific | N/A |
| CF®640R succinimidyl ester | Biotium | Cat# 92108 |
| 96 well plate | Thermo Fisher Scientific | Cat# 268200 |
| CytoFLEX S Flow Cytometer | Beckman Coulter | Cat# N0-V4-B2-Y4 |
| Autostainer Link 48 | Dako | N/A |
| Cell-Culture Treated Multidishes (24-well plate) | Thermo Fisher Scientific | Cat# 142475 |
| Superose 6 Increase 10/300 GL | Cytiva | Cat# 29091596 |
| Buxco Small Animal Whole Body Plethysmography | DSI | https://www.datasci.com/products/buxco-respiratory-products/finepointe-whole-body-plethysmography |
| MouseOx PLUS | STARR | https://www.starrlifesciences.com/product-category/pulse-oximetry/ |
| Multiskan FC | Thermo Fisher Scientific | N/A |
| Transwell 6.5mm Polycarbonate Membrane Inserts Pre-Loaded in 24-Well Culture Plates | Corning | Cat# 3413 |
| PDMS (Silicone Elastomer Kit) | Dow Corning | Cat# SYLGARD 184 |
| SU-8 2150 | MicroChem | Cat# SU-8 2150 |
| Kai Medical Biopsy Punch 6mm | Kai Corporation | Cat# BP-L60K |
| Cell Culture Inserts, 3.0-μm pore size inserts, 6-well, Transparent PET | Falcon | Cat# 353091 |
| Quantifoil R2.0/2.0 Cu 300 mesh grid | Quantifoil Micro Tools GmbH | N/A |
| Vitrobot | Thermo Fisher Scientific | N/A |
| Krios G4 | Thermo Fisher Scientific | N/A |
| GIF-Biocontinuum energy filter | Gatan | N/A |
Resource availability
Lead contact
Further information and requests for resources and reagents should be directed to and will be fulfilled by the lead contact, Kei Sato (keisato@g.ecc.u-tokyo.ac.jp).
Materials availability
All unique reagents generated in this study are listed in the key resources table and available from the lead contact with a completed Materials Transfer Agreement.
Experimental model and subject details
Ethics statement
All experiments with hamsters were performed in accordance with the Science Council of Japan’s Guidelines for the Proper Conduct of Animal Experiments. The protocols were approved by the Institutional Animal Care and Use Committee of National University Corporation Hokkaido University (approval ID: 20-0123 and 20-0060). All protocols involving specimens from human subjects recruited at Kyoto University and Interpark Kuramochi Clinic were reviewed and approved by the Institutional Review Boards of Kyoto University (approval ID: G1309), Chiba University (approval ID: HS202103-03) and Interpark Kuramochi Clinic (approval ID: G2021-004). All human subjects provided written informed consent. All protocols for the use of human specimens were reviewed and approved by the Institutional Review Boards of The Institute of Medical Science, The University of Tokyo (approval IDs: 2021-1-0416 and 2021-18-0617), Kyoto University (approval ID: G0697), Kumamoto University (approval IDs: 2066 and 2074), and University of Miyazaki (approval ID: O-1021).
Human serum collection
Vaccine sera of fifteen individuals who had the BNT162b2 vaccine (Pfizer/BioNTech) (average age: 38 years, range: 24–48 years; 53% male) (Figures 2A–2D) were obtained at one month after the second dose, one month after the third dose, and four months after the third dose. Vaccine sera of fifteen individuals who had the BNT162b2 vaccine (Pfizer/BioNTech) for the first, second, and third doses of vaccination and mRNA-1273 (Moderna) for the fourth dose of vaccination (average age: 42 years, range: 32–56 years; 33% male) (Figure 2D) were obtained at one month after the fourth dose. The details of the vaccine sera are summarized in Table S2.
Convalescent sera were collected from fully vaccinated individuals who had been infected with BA.1 (16 2-dose vaccinated; 10–27 days after testing; average age: 48 years, range: 20–76 years, 44% male) (Figure 2E), fully vaccinated individuals who had been infected with BA.2 (9 2-dose vaccinated and 5 3-dose vaccinated; 11–61 days after testing. n=14 in total; average age: 47 years, range: 24–84 years, 64% male) (Figure 2F), and fully vaccinated individuals who had been infected with BA.5 (2 2-dose vaccinated, 17 3-dose vaccinated and 1 4-dose vaccinated; 10–23 days after testing. n=20 in total; average age: 51 years, range: 25–73 years, 45% male) (Figure 2G). The SARS-CoV-2 variants were identified as previously described (Kimura et al., 2022c; Yamasoba et al., 2022a). Sera were inactivated at 56°C for 30 minutes and stored at –80°C until use. The details of the convalescent sera are summarized in Table S2.
Cell culture
HEK293T cells (a human embryonic kidney cell line; ATCC, CRL-3216), HEK293 cells (a human embryonic kidney cell line; ATCC, CRL-1573) and HOS-ACE2/TMPRSS2 cells (HOS cells stably expressing human ACE2 and TMPRSS2) (Ozono et al., 2021; Ferreira et al., 2021) were maintained in DMEM (high glucose) (Sigma-Aldrich, Cat# 6429-500ML) containing 10% fetal bovine serum (FBS, Sigma-Aldrich Cat# 172012-500ML) and 1% penicillin–streptomycin (PS) (Sigma-Aldrich, Cat# P4333-100ML). HEK293-ACE2 cells (HEK293 cells stably expressing human ACE2) (Motozono et al., 2021) were maintained in DMEM (high glucose) containing 10% FBS, 1 μg/ml puromycin (InvivoGen, Cat# ant-pr-1) and 1% PS. HEK293-ACE2/TMPRSS2 cells (HEK293 cells stably expressing human ACE2 and TMPRSS2) (Motozono et al., 2021) were maintained in DMEM (high glucose) containing 10% FBS, 1 μg/ml puromycin, 200 μg/ml hygromycin (Nacalai Tesque, Cat# 09287-84) and 1% PS. Vero cells [an African green monkey (Chlorocebus sabaeus) kidney cell line; JCRB Cell Bank, JCRB0111] were maintained in Eagle’s minimum essential medium (EMEM) (Sigma-Aldrich, Cat# M4655-500ML) containing 10% FBS and 1% PS. VeroE6/TMPRSS2 cells (VeroE6 cells stably expressing human TMPRSS2; JCRB Cell Bank, JCRB1819) (Matsuyama et al., 2020) were maintained in DMEM (low glucose) (Wako, Cat# 041-29775) containing 10% FBS, G418 (1 mg/ml; Nacalai Tesque, Cat# G8168-10ML) and 1% PS. Calu-3/DSP1-7 cells (Calu-3 cells stably expressing DSP1-7) (Yamamoto et al., 2020) were maintained in EMEM (Wako, Cat# 056-08385) containing 20% FBS and 1% PS. HEK293S GnTI(-) cells (HEK293S cells lacking N-acetylglucosaminyltransferase) (Reeves et al., 2002) were maintained in DMEM (Nacalai Tesque, Cat# 08458-16) containing 2% FBS without PS. Human airway and lung epithelial cells derived from human induced pluripotent stem cells (iPSCs) were manufactured according to established protocols as described below (see “preparation of human airway and lung epithelial cells from human iPSCs” section) and provided by HiLung Inc. AOs and AO-ALI model were generated according to established protocols as described below (see “Airway organoids” and “AO-ALI model” sections).
Method details
Viral genome sequencing
Viral genome sequencing was performed as previously described (Meng et al., 2022; Yamasoba et al., 2022a; Suzuki et al., 2022; Saito et al., 2022; Motozono et al., 2021). Briefly, the virus sequences were verified by viral RNA-sequencing analysis. Viral RNA was extracted using a QIAamp viral RNA mini kit (Qiagen, Cat# 52906). The sequencing library employed for total RNA sequencing was prepared using the NEBNext Ultra RNA Library Prep Kit for Illumina (New England Biolabs, Cat# E7530). Paired-end 76-bp sequencing was performed using a MiSeq system (Illumina) with MiSeq reagent kit v3 (Illumina, Cat# MS-102-3001). Sequencing reads were trimmed using fastp v0.21.0 (Chen et al., 2018) and subsequently mapped to the viral genome sequences of a lineage B isolate (strain Wuhan-Hu-1; GenBank accession number: NC_045512.2) (Matsuyama et al., 2020) using BWA-MEM v0.7.17 (Li and Durbin, 2009). Variant calling, filtering, and annotation were performed using SAMtools v1.9 (Li et al., 2009) and snpEff v5.0e (Cingolani et al., 2012).
Phylogenetic analyses
For construction of an ML tree of Omicron lineages (BA.1–BA.5) sampled from South Africa and BA.2.75 (shown in Figure 1A), the genome sequence data of SARS-CoV-2 and its metadata were downloaded from the GISAID database (https://www.gisaid.org/) (Khare et al., 2021) on July 23, 2022. We excluded the data of viral strains with the following features from the analysis: i) a lack of collection date information; ii) sampling from animals other than humans, iii) >2% undetermined nucleotide characters, or iv) sampling by quarantine. From each viral lineage, 30 sequences were randomly sampled and used for tree construction, in addition to an outgroup sequence, EPI_ISL_466615, representing the oldest isolate of B.1.1 obtained in the UK. The viral genome sequences were mapped to the reference sequence of Wuhan-Hu-1 (GenBank accession number: NC_045512.2) using Minimap2 v2.17 (Li, 2018) and subsequently converted to a multiple sequence alignment according to the GISAID phylogenetic analysis pipeline (https://github.com/roblanf/sarscov2phylo). The alignment sites corresponding to the 1–265 and 29674–29903 positions in the reference genome were masked (i.e., converted to NNN). Alignment sites at which >50% of sequences contained a gap or undetermined/ambiguous nucleotide were trimmed using trimAl v1.2 (Capella-Gutiérrez et al., 2009). Phylogenetic tree construction was performed via a three-step protocol: i) the first tree was constructed; ii) tips with longer external branches (Z score > 4) were removed from the dataset; iii) and the final tree was constructed. Tree reconstruction was performed by RAxML v8.2.12 (Stamatakis, 2014) under the GTRCAT substitution model. The node support value was calculated by 100 bootstrap analyses.
Modeling the epidemic dynamics of SARS-CoV-2 lineages
To quantify the spread rate of each SARS-CoV-2 lineage in the human population in India, we estimated the relative Re of each viral lineage according to the epidemic dynamics, calculated on the basis of viral genomic surveillance data. The data were downloaded from the GISAID database (https://www.gisaid.org/) on August 1, 2022. We excluded the data of viral strains with the following features from the analysis: i) a lack of collection date information; ii) sampling in animals other than humans; or iii) sampling by quarantine. We analyzed the datasets of the ten states of India, where ≥20 sequences of either BA.2.75 or BA.5 are reported (i.e., Himachal Pradesh, Odisha, Haryana, Rajasthan, and Maharashtra, Gujarat, West Bengal, Delhi, Tamil Nadu, and Telangana). BA.5 sublineages are summarized as “BA.5”, and BA.2 sublineages with ≤400 sequences are summarized as “other BA.2”. Subsequently, the dynamics of the top seven predominant lineages in India were estimated from April 24, 2022, to August 1, 2022, were analyzed. The number of viral sequences of each viral lineage collected each day in each country was counted, and the count matrix was constructed as an input for the statistical model below.
We constructed a Bayesian hierarchical model to represent relative lineage growth dynamics with multinomial logistic regression as described in our previous study (Yamasoba et al., 2022a). In brief, we incorporated a hierarchical structure into the slope parameter over time, which enabled us to estimate the global average relative Re of each viral lineage in India as well as the average value for each country. Arrays in the model index over one or more indices: L = 7 viral lineages ; S = 10 states ; and T = 100 days . The model is as follows:
The explanatory variable was time, , and the outcome variable was , which represented the count of viral lineage in state at time . The slope parameter of lineage in state , , was generated from a Student’s t distribution with hyperparameters of the mean, , and the standard deviation, . As the distribution generated , we used a Student’s t distribution with six degrees of freedom instead of a normal distribution to reduce the effects of outlier values of . In the model, the linear estimator , consisting of the intercept and the slope , was converted to the simplex , which represented the probability of occurrence of each viral lineage at time in state , based on the softmax link function defined as follows:
is generated from and the total count of all lineages at time in state according to a multinomial distribution.
The relative Re of each viral lineage in each county was calculated according to the slope parameter as follows:
where is the average viral generation time (2.1 days) (http://sonorouschocolate.com/covid19/index.php?title=Estimating_Generation_Time_Of_Omicron). Similarly, the global average relative Re of each viral lineage was calculated according to the slope hyperparameter as follows:
For parameter estimation, the intercept and slope parameters of the BA.2 variant were fixed at 0. Consequently, the relative Re of BA.2 was fixed at 1, and those of the other lineages were estimated relative to that of BA.2.
Parameter estimation was performed via the MCMC approach implemented in CmdStan v2.28.1 (https://mc-stan.org) with CmdStanr v0.4.0 (https://mc-stan.org/cmdstanr/). Noninformative priors were set for all parameters. Four independent MCMC chains were run with 1,000 and 2,000 steps in the warmup and sampling iterations, respectively. We confirmed that all estimated parameters showed <1.01 R-hat convergence diagnostic values and >200 effective sampling size values, indicating that the MCMC runs were successfully convergent. The above analyses were performed in R v4.1.3 (https://www.r-project.org/). Information on the relative Re estimated in the present study is summarized in Table S1.
Plasmid construction
Plasmids expressing the codon-optimized SARS-CoV-2 S proteins of B.1.1 (the parental D614G-bearing variant), BA.2 and BA.5 were prepared in our previous studies (Yamasoba et al., 2022a; Suzuki et al., 2022; Saito et al., 2022; Ozono et al., 2021; Kimura et al., 2022a). Plasmids expressing the codon-optimized S proteins of BA.2.75 and BA.2 S-based derivatives were generated by site-directed overlap extension PCR using the primers listed in Table S5. The resulting PCR fragment was digested with KpnI (New England Biolabs, Cat# R0142S) and NotI (New England Biolabs, Cat# R1089S) and inserted into the corresponding site of the pCAGGS vector (Niwa et al., 1991). Nucleotide sequences were determined by DNA sequencing services (Eurofins), and the sequence data were analyzed by Sequencher v5.1 software (Gene Codes Corporation).
To construct plasmids expressing anti-SARS-CoV-2 monoclonal antibodies (adintrevimab, bamlanivimab, bebtelovimab, casirivimab, cilgavimab, etesevimab, imdevimab, regdanvimab, sotrovimab and tixagevimab), we obtained the sequences of the variable regions of these antibodies from the KEGG Drug Database (https://www.genome.jp/kegg/drug/) and The Structural Antibody Database (http://opig.stats.ox.ac.uk/webapps/newsabdab/sabdab/) and artificially synthesized them by Fasmac. The obtained coding sequences of the variable regions of the heavy and light chains were cloned into the pCAGGS vector containing the sequences of human immunoglobulin 1 and kappa constant region (kindly provided by Dr. Hisashi Arase). Plasmids expressing the SARS-CoV-2 spike proteins of the parental D614G (B.1.1), Omicron BA.2 and BA.4/5 were prepared in our previous studies (Yamasoba et al., 2022b; Kimura et al., 2022c; Yamasoba et al., 2022a; Ozono et al., 2021). Plasmids expressing the spike protein of Omicron variants BA.2.75 (GISAID ID: EPI_ISL_13471039) were generated by site-directed overlap extension PCR using pC-SARS2-S BA.2 (Yamasoba et al., 2022a, 2022b) as the template and the primers listed in Table S5. The resulting PCR fragment was subcloned into the KpnI-NotI site of the pCAGGS vector (Niwa et al., 1991) using an In-Fusion® HD cloning kit (Takara, Cat# Z9650N). Nucleotide sequences were determined by DNA sequencing services (Eurofins), and the sequence data were analyzed by Sequencher v5.1 software (Gene Codes Corporation).
Preparation of monoclonal antibodies
Ten monoclonal antibodies (adintrevimab, bamlanivimab, bebtelovimab, casirivimab, cilgavimab, etesevimab, imdevimab, regdanvimab, sotrovimab and tixagevimab) were prepared as previously described (Meng et al., 2022; Yamasoba et al., 2022b; Liu et al., 2021). Briefly, the pCAGGS vectors containing the sequences encoding the immunoglobulin heavy and light chains were cotransfected into HEK293T cells at a 1:1 ratio using PEI Max (Polysciences, Cat# 24765-1). The culture medium was refreshed with DMEM (low glucose) (Wako, Cat# 041-29775) containing 10% FBS without PS. At 72 hours posttransfection, the culture medium was harvested, and the antibodies were purified using an NAb protein A plus spin kit (Thermo Fisher Scientific, Cat# 89948) according to the manufacturer’s protocol.
Neutralization assay
Pseudoviruses were prepared as previously described (Yamasoba et al., 2022a, 2022b; Kimura et al., 2022a, 2022c; Motozono et al., 2021; Ferreira et al., 2021; Uriu et al., 2021, 2022). Briefly, lentivirus (HIV-1)-based, luciferase-expressing reporter viruses were pseudotyped with SARS-CoV-2 S proteins. HEK293T cells (1,000,000 cells) were cotransfected with 1 μg psPAX2-IN/HiBiT (Ozono et al., 2020), 1 μg pWPI-Luc2 (Ozono et al., 2020), and 500 ng plasmids expressing parental S or its derivatives using PEI Max (Polysciences, Cat# 24765-1) according to the manufacturer's protocol. Two days posttransfection, the culture supernatants were harvested and centrifuged. The pseudoviruses were stored at –80°C until use.
The neutralization assay (Figures 2 and S2; Tables 1 and S3) was prepared as previously described (Yamasoba et al., 2022a, 2022b; Kimura et al., 2022a, 2022c; Ferreira et al., 2021; Uriu et al., 2021; Uriu et al., 2022). Briefly, the SARS-CoV-2 S pseudoviruses (counting ∼20,000 relative light units) were incubated with serially diluted (120-fold to 87,480-fold dilution at the final concentration) heat-inactivated sera or monoclonal antibodies (the concentrations of serially diluted antibodies are indicated in Figure S2) at 37°C for 1 hour. Pseudoviruses without sera were included as controls. Then, a 40 μl mixture of pseudovirus and serum/antibody was added to HOS-ACE2/TMPRSS2 cells (10,000 cells/50 μl) in a 96-well white plate. At 2 d.p.i., the infected cells were lysed with a One-Glo luciferase assay system (Promega, Cat# E6130) or a Bright-Glo luciferase assay system (Promega, Cat# E2650), and the luminescent signal was measured using a GloMax explorer multimode microplate reader 3500 (Promega) or CentroXS3 (Berthhold Technologies). The assay of each serum sample was performed in triplicate, and the 50% neutralization titer (NT50) was calculated using Prism 9 software v9.1.1 (GraphPad Software).
Airway organoids
An airway organoid (AO) model was generated according to our previous report (Sano et al., 2022). Briefly, normal human bronchial epithelial cells (NHBEs, Cat# CC-2540, Lonza) were used to generate AOs. NHBEs were suspended in 10 mg/ml cold Matrigel growth factor reduced basement membrane matrix (Corning, Cat# 354230). Fifty microliters of cell suspension were solidified on prewarmed cell culture-treated multiple dishes (24-well plates; Thermo Fisher Scientific, Cat# 142475) at 37 °C for 10 min, and then, 500 μl of expansion medium was added to each well. AOs were cultured with AO expansion medium for 10 days. For maturation of the AOs, expanded AOs were cultured with AO differentiation medium for 5 days. In experiments evaluating the antiviral drugs (see “Antiviral drug assay using SARS-CoV-2 clinical isolates and AOs” section below), AOs were dissociated into single cells and then were seeded into a 96-well plate.
SARS-CoV-2 preparation and titration
The working virus stocks of SARS-CoV-2 were prepared and titrated as previously described (Meng et al., 2022; Kimura et al., 2022b, 2022c; Yamasoba et al., 2022a; Suzuki et al., 2022; Saito et al., 2022; Motozono et al., 2021). In this study, clinical isolates of B.1.1 (strain TKYE610670; GISAID ID: EPI_ISL_479681) (Suzuki et al., 2022), Delta (B.1.617.2, strain TKYTK1734; GISAID ID: EPI_ISL_2378732) (Saito et al., 2022), BA.2 (strain TY40-385; GISAID ID: EPI_ISL_9595859) (Kimura et al., 2022c) and BA.5 (strain TKYS14631; GISAID ID: EPI_ISL_12812500) (Tamura et al., 2022), and BA.2.75 (strain TY41-716; GISAID ID: EPI_ISL_13969765) were used. In brief, 20 μl of the seed virus was inoculated into VeroE6/TMPRSS2 cells (5,000,000 cells in a T-75 flask). One h.p.i., the culture medium was replaced with DMEM (low glucose) (Wako, Cat# 041-29775) containing 2% FBS and 1% PS. At 3 d.p.i., the culture medium was harvested and centrifuged, and the supernatants were collected as the working virus stock.
The titer of the prepared working virus was measured as the 50% tissue culture infectious dose (TCID50). Briefly, one day before infection, VeroE6/TMPRSS2 cells (10,000 cells) were seeded into a 96-well plate. Serially diluted virus stocks were inoculated into the cells and incubated at 37°C for 4 days. The cells were observed under a microscope to judge the CPE appearance. The value of TCID50/ml was calculated with the Reed–Muench method (Reed and Muench, 1938).
For verification of the sequences of SARS-CoV-2 working viruses, viral RNA was extracted from the working viruses using a QIAamp viral RNA mini kit (Qiagen, Cat# 52906) and viral genome sequences were analyzed as described above (see "Viral genome sequencing" section). Information on the unexpected substitutions detected is summarized in Table S6, and the raw data are deposited in the GitHub repository (https://github.com/TheSatoLab/Omicron_BA.2.75).
Antiviral drug assay using SARS-CoV-2 clinical isolates and AOs
Antiviral drug assays (Table 2; Figure S3A) were performed as previously described (Meng et al., 2022). Briefly, one day before infection, AOs (10,000 cells) were dissociated, and then seeded into a 96-well plate. The cells were infected with either B.1.1, BA.2, BA.5 or BA.2.75 isolate (100 TCID50) at 37 °C for 2 hours. The cells were washed with DMEM and cultured in DMEM supplemented with 10% FCS, 1% PS and serially diluted remdesivir (Clinisciences, Cat# A17170), EIDD-1931 (an active metabolite of molnupiravir; Cell Signaling Technology, Cat# 81178S), or nirmatrelvir (PF-07321332; MedChemExpress, Cat# HY-138687). At 24 hours after infection, the culture supernatants were collected, and viral RNA was quantified using RT–qPCR (see “RT–qPCR” section below). The assay of each compound was performed in quadruplicate, and the 50% effective concentration (EC50) was calculated using Prism 9 software v9.1.1 (GraphPad Software).
Cytotoxicity assay
The cytotoxicity of remdesivir, EIDD-1931 or nirmatrelvir (Figure S3B) was assessed as previously described (Meng et al., 2022). Briefly, one day before the assay, AOs (10,000 cells) were dissociated and then seeded into a 96-well plate. The cells were cultured with the serially diluted antiviral drugs for 24 hours. The Cell Counting Kit-8 (Dojindo, Cat# CK04-11) solution (10 μl) was added to each well, and the cells were incubated at 37 °C for 90 minutes. Absorbance was measured at 450 nm using the a Multiskan FC (Thermo Fisher Scientific). The assay of each compound was performed in quadruplicate, and the 50% cytotoxic concentration (CC50) was calculated using Prism 9 software v9.1.1 (GraphPad Software).
Pseudovirus infection
Pseudovirus infection (Figure 3A) was performed as previously described (Yamasoba et al., 2022a, 2022b; Kimura et al., 2022a, 2022c; Motozono et al., 2021; Ferreira et al., 2021; Uriu et al., 2021, 2022). Briefly, the amount of pseudoviruses prepared was quantified by the HiBiT assay using a Nano Glo HiBiT lytic detection system (Promega, Cat# N3040) as previously described (Ozono et al., 2021, 2020). For measurement of pseudovirus infectivity, the same amount of pseudoviruses (normalized to the HiBiT value, which indicates the amount of HIV-1 p24 antigen) was inoculated into HOS-ACE2/TMPRSS2 cells, HEK293-ACE2 cells or HEK293-ACE2/TMPRSS2 cells and viral infectivity was measured as described above (see “Neutralization assay” section). For analysis of the effect of TMPRSS2 on pseudovirus infectivity (Figure S4A), the fold change of the values of HEK293-ACE2/TMPRSS2 to HEK293-ACE2 was calculated.
Yeast surface display
Yeast surface display (Figure 3B) was performed as previously described (Dejnirattisai et al., 2022; Kimura et al., 2022a, 2022b, 2022c; Yamasoba et al., 2022a; Motozono et al., 2021; Zahradník et al., 2021b). Briefly, the RBD genes [“construct 3” in Zahradník et al. (2021b), covering residues 330–528] in the pJYDC1 plasmid were cloned by restriction enzyme-free cloning and transformed into the EBY100 Saccharomyces cerevisiae. The primers are listed in Table S5. The expression media 1/9 (Zahradník et al., 2021a) was inoculated (OD 1) by overnight (220 rpm, 30°C, SD-CAA media) grown culture, followed by cultivation for 24 hours at 20°C. The medium was supplemented with 10 mM DMSO solubilized bilirubin (Sigma-Aldrich, Cat# 14370-1G) for expression cocultivation labeling [pJYDC1, eUnaG2 reporter holo-form formation, green/yellow fluorescence (excitation at 498 nm, emission at 527 nm)]. Cells (100 μl aliquots) were collected by centrifugation (3000 g, 3 minutes), washed in ice-cold PBSB buffer (PBS with 1 mg/ml BSA), and resuspended in an analysis solution with a series of CF®640R succinimidyl ester labeled (Biotium, Cat# 92108) ACE2 peptidase domain (residues 18–740) concentrations. The peptidase domain of wild-type ACE2 and ACE2 N90Q were produced and purified as previously described (Zahradník et al., 2021b). The reaction volume was adjusted (1–100 ml) to avoid the ligand depletion effect, and the suspension was incubated overnight in a rotator shaker (10 rpm, 4°C). Incubated samples were washed with PBSB buffer, transferred into a 96-well plate (Thermo Fisher Scientific, Cat# 268200), and analyzed by a CytoFLEX S Flow Cytometer (Beckman Coulter, USA, Cat#. N0-V4-B2-Y4) with the gating strategy described previously (Zahradník et al., 2021b). The eUnaG2 signals were compensated by CytExpert software (Beckman Coulter). The mean binding signal (FL4-A) values of RBD-expressing cells, subtracted by signals of nonexpressing populations, were subjected to the determination of the dissociation constant KD, YD by a noncooperative Hill equation fitted by nonlinear least-squares regression using Python v3.7 fitted together with two additional parameters describing the titration curve (Zahradník et al., 2021b).
Protein expression and purification for cryo-EM
Protein expression and purification were performed as previously described (Hashiguchi et al., 2011). Briefly, the expression plasmid, pHLsec, encoding the BA.2.75 S ectodomain bearing six proline substitutions (F817P, A892P, A899P, A942P, K986P and V987P) (Hsieh et al., 2020) and the deletion of the furin cleavage site (i.e., RRAR to GSAG substitution) with a T4-fold trimerization domain or soluble human ACE2 ectodomain were transfected into HEK293S GnTI(-) cells. The proteins in the culture supernatant were purified with a cOmplete His-Tag Purification Resin (Roche, Cat# 5893682001) affinity column, followed by Superose 6 Increase 10/300 GL size-exclusion chromatography (Cytiva, Cat# 29091596) with calcium- and magnesium-free PBS buffer.
Cryo-EM sample preparation and data collection
For the preparation of a complex sample for cryo-EM, BA.2.75 S ectodomain solution was incubated at 37°C for 1 hour before use. Purified ACE2 was incubated with BA.2.75 S ectodomain at a molar ratio of 1:3.2 at 18°C for 1 hour. A 0.1% (w/v) n-octyl-β-D-glucoside solution (Dojindo, Cat# O001) was added to BA.2.75 S ectodomain solution with or without ACE2 to a final concentration of 0.01%. The sample was then applied to a Quantifoil R2.0/2.0 Cu 300 mesh grid (Quantifoil Micro Tools GmbH), which had been freshly glow-discharged for 120 seconds at 10 mA using PIB-10 (Vacuum Device). The samples were plunged into liquid ethane using a Vitrobot mark IV (Thermo Fisher Scientific) with the following settings: temperature 18°C, humidity 100%, blotting time 5 seconds, and blotting force 5. Micrographs were collected on a Krios G4 system (Thermo Fisher Scientific) operated at 300 kV with a K3 direct electron detector (Gatan) at a nominal magnification of 130,000 (0.67 per physical pixel) using a GIF-Biocontinuum energy filter (Gatan) with a 20-eV slit width. Each micrograph was collected with a total exposure of 1.5 s and a total dose of 50 e/Å2 over 50 frames. A total of 3308 and 3248 micrographs of S and S-ACE2 complexes, respectively, were collected. All micrographs were collected at a nominal defocus range of 0.8–1.8 μm using EPU software v2.14 (Thermo Fisher Scientific).
Cryo-EM image processing
For the BA.2.75 S ectodomain without ACE2, micrograph movie frames were aligned, dose-weighted, and CTF-estimated using Patch Motion correction and Patch CTF in cryoSPARC v3.3.1 (Punjani et al., 2017). A total of 754,589 particles were blob-picked, and reference-free 2D classification (K=150, batch=200, iteration=30) was performed to remove junk particles. Heterogeneous refinement was carried out using the Ab initio model that was reconstructed with 467,029 selected particles after 2D classification in cryoSPARC. The particles belonging to the class of S ectodomains were selected, and five rounds of heterogeneous refinement were performed to isolate the S ectodomain in different conformation states, and each state was selected and processed by nonuniform refinement with CTF refinement in cryoSPARC to generate the final maps. For BA.2.75 S ectodomain with the ACE2 complex, the dataset was preprocessed to 2D classification (K=150, batch=200, iteration=30), in the same way as the BA.2.75 S ectodomain without ACE2, with 682,973 picked particles. A total of 338,745 particles belonging cto lasses that show a representative S ectodomain were selected and subsequently refined against a map of SARS-CoV-2 S in complex with ACE2 (EMDB: EMD-22891) (Xiao et al., 2021) and processed by nonuniform refinement with CTF refinement in cryoSPARC to generate the final maps. The reported global resolutions are based on the gold-standard Fourier shell correlation curve (FSC=0.143) criterion. Local resolutions were calculated with cryoSPARC (Cardone et al., 2013). Workflows of data processing are shown in Figure S4B. Figures related to data processing and final reconstructed maps were prepared with UCSF Chimera v1.15 (Pettersen et al., 2004) and UCSF Chimera X v1.1 (Goddard et al., 2018).
Cryo-EM model building and analysis
The SARS-CoV-2 BA.2 S ectodomain (PDB: 7UB0) (Stalls et al., 2022) was fitted to the corresponding maps using UCSF Chimera v1.15 (Pettersen et al., 2004). Iterative rounds of manual fitting in Coot v0.9.6 (Emsley and Cowtan, 2004) and real-space refinement in Phenix v1.20 (Adams et al., 2010) were carried out to improve nonideal rotamers, bond angles, and Ramachandran outliers. The final model was validated with MolProbity (Williams et al., 2018). Electrostatic potentials were calculated using APBS Electrostatic plugins (Jurrus et al., 2018) for PyMOL v2.3.3. The structure models shown in the surface, cartoon, and stick presentation in the figures were prepared with PyMOL.
SARS-CoV-2 S-based fusion assay
A SARS-CoV-2 S-based fusion assay (Figures 3G and 3H) was performed as previously described (Kimura et al., 2022b, 2022c; Yamasoba et al., 2022a; Suzuki et al., 2022; Saito et al., 2022; Motozono et al., 2021). Briefly, on day 1, effector cells (i.e., S-expressing cells) and target cells (Calu-3/DSP1-7 cells) were prepared at a density of 0.6–0.8 × 106 cells in a 6-well plate. On day 2, for the preparation of effector cells, HEK293 cells were cotransfected with the S expression plasmids (400 ng) and pDSP8-11 (Kondo et al., 2011) (400 ng) using TransIT-LT1 (Takara, Cat# MIR2300). On day 3 (24 hours posttransfection), 16,000 effector cells were detached and reseeded into a 96-well black plate (PerkinElmer, Cat# 6005225), and target cells were reseeded at a density of 1,000,000 cells/2 ml/well in 6-well plates. On day 4 (48 hours posttransfection), target cells were incubated with EnduRen live cell substrate (Promega, Cat# E6481) for 3 hours and then detached, and 32,000 target cells were added to a 96-well plate with effector cells. Renilla luciferase activity was measured at the indicated time points using Centro XS3 LB960 (Berthhold Technologies). For measurement of the surface expression level of the S protein, effector cells were stained with rabbit anti-SARS-CoV-2 S S1/S2 polyclonal antibody (Thermo Fisher Scientific, Cat# PA5-112048, 1:100). Normal rabbit IgG (Southern Biotech, Cat# 0111-01, 1:100) was used as a negative control, and APC-conjugated goat anti-rabbit IgG polyclonal antibody (Jackson ImmunoResearch, Cat# 111-136-144, 1:50) was used as a secondary antibody. The surface expression level of S proteins (Figure 3G) was measured using a FACS Canto II (BD Biosciences) and the data were analyzed using FlowJo software v10.7.1 (BD Biosciences). For calculation of fusion activity, Renilla luciferase activity was normalized to the MFI of surface S proteins. The normalized value (i.e., Renilla luciferase activity per the surface S MFI) is shown as fusion activity.
Coculture experiment
A coculture experiment (Figure S4D) was performed as previously described (Kimura et al., 2022c; Yamasoba et al., 2022a; Suzuki et al., 2022). This assay utilizes a dual split protein (DSP) encoding Renilla luciferase and GFP genes; the respective split proteins, DSP8-11 and DSP1-7, are expressed in effector and target cells by transfection. Briefly, one day before transfection, effector cells (i.e., S-expressing cells) were seeded on the coverslips coated with poly-L-lysine (Sigma, Cat# P4832) and placed in a 12-well plate, and target cells were prepared at a density of 100,000 cells in a 12-well plate. For preparation of effector cells, HEK293 cells were cotransfected with the S-expression plasmids (500 ng) and pDSP8-11 (500 ng) using PEI Max (Polysciences, Cat# 24765-1). For preparation of target cells, HEK293 and HEK293-ACE2/TMPRSS2 cells were transfected with pDSP1-7 (500 ng) (Kondo et al., 2011). At 24 hours posttransfection, target cells were detached and cocultured with effector cells in a 1:2 ratio. At 24 h post-coculture, cells were fixed with 4% paraformaldehyde in PBS (Nacalai Tesque, Cat# 09154-85) for 15 minutes at room temperature. Nuclei were stained with Hoechst 33342 (Thermo Fisher Scientific, Cat# H3570). The coverslips were mounted on glass slides using Fluoromount-G (SouthernBiotech, Cat# 0100-01) with Hoechst 33342 and observed using an A1Rsi Confocal Microscope (Nikon). The syncytium size (GFP-positive area) was measured using Fiji software v2.2.0 (ImageJ) as previously described (Kimura et al., 2022c; Yamasoba et al., 2022a; Suzuki et al., 2022).
AO-ALI model
The AO-ALI model (Figure 4D) was generated according to our previous report (Sano et al., 2022). For generation of AO-ALI, expanding AOs were dissociated into single cells, and then were seeded into Transwell inserts (Corning, Cat# 3413) in a 24-well plate. AO-ALI was cultured with AO differentiation medium for 5 days to promote their maturation. AO-ALI was infected with SARS-CoV-2 from the apical side.
Preparation of human airway and lung epithelial cells from human iPSCs
The air-liquid interface culture of airway and lung epithelial cells (Figures 4E and 4F) was differentiated from human iPSC-derived lung progenitor cells as previously described (Kimura et al., 2022c; Tamura et al., 2022; Yamamoto et al., 2017; Konishi et al., 2016; Gotoh et al., 2014). Briefly, lung progenitor cells were induced stepwise from human iPSCs according to a 21-day and 4-step protocol (Yamamoto et al., 2017). At day 21, lung progenitor cells were isolated with the specific surface antigen carboxypeptidase M and seeded onto the upper chamber of a 24-well Cell Culture Insert (Falcon, #353104), followed by 28-day and 7-day differentiation of airway and lung epithelial cells, respectively. Alveolar differentiation medium with dexamethasone (Sigma-Aldrich, Cat# D4902), KGF (PeproTech, Cat# 100-19), 8-Br-cAMP (Biolog, Cat# B007), 3-isobutyl 1-methylxanthine (IBMX) (Fujifilm Wako, Cat# 095-03413), CHIR99021 (Axon Medchem, Cat# 1386), and SB431542 (Fujifilm Wako, Cat# 198-16543) was used for the induction of lung epithelial cells. PneumaCult ALI (STEMCELL Technologies, Cat# ST-05001) with heparin (Nacalai Tesque, Cat# 17513-96) and Y-27632 (LC Laboratories, Cat# Y-5301) hydrocortisone (Sigma-Aldrich, Cat# H0135) was used for induction of airway epithelial cells.
Airway-on-a-chips
Airway-on-a-chips (Figures 4G–4I and S4E) were prepared as previously described (Hashimoto et al., 2022). Human lung microvascular endothelial cells (HMVEC-L) were obtained from Lonza (Cat# CC-2527) and cultured with EGM-2-MV medium (Lonza, Cat# CC-3202). For preparation of the airway-on-a-chip, first, the bottom channel of a polydimethylsiloxane (PDMS) device was precoated with fibronectin (3 μg/ml, Sigma-Aldrich, Cat# F1141). The microfluidic device was generated according to our previous report (Deguchi et al., 2021). HMVEC-L cells were suspended at 5,000,000 cells/ml in EGM2-MV medium. Then, 10 μl of suspension medium was injected into the fibronectin-coated bottom channel of the PDMS device. Then, the PDMS device was turned upside down and incubated for 1 h. After 1 hour, the device was turned over, and the EGM2-MV medium was added into the bottom channel. After 4 days, AOs were dissociated and seeded into the top channel. AOs were generated according to our previous report (Sano et al., 2022). AOs were dissociated into single cells and then suspended at 5,000,000 cells/ml in the AO differentiation medium. Ten microliter suspension medium was injected into the top channel. After 1 hour, the AO differentiation medium was added to the top channel. In the infection experiments (Figures 4G–4I), the AO differentiation medium containing either B.1.1, Delta, BA.2, BA.5 or BA.2.75 isolate (500 TCID50) was inoculated into the top channel (Figure S4E). At 2 h.p.i., the top and bottom channels were washed and cultured with AO differentiation and EGM2-MV medium, respectively. The culture supernatants were collected, and viral RNA was quantified using RT–qPCR (see “RT–qPCR” section above).
Microfluidic device
A microfluidic device was generated according to our previous report (Deguchi et al., 2021). Briefly, the microfluidic device consisted of two layers of microchannels separated by a semipermeable membrane. The microchannel layers were fabricated from PDMS using a soft lithographic method. PDMS prepolymer (Dow Corning, Cat# SYLGARD 184) at a base to curing agent ratio of 10:1 was cast against a mold composed of SU-8 2150 (MicroChem, Cat# SU-8 2150) patterns formed on a silicon wafer. The cross-sectional size of the microchannels was 1 mm in width and 330 μm in height. Access holes were punched through the PDMS using a 6-mm biopsy punch (Kai Corporation, Cat# BP-L60K) to introduce solutions into the microchannels. Two PDMS layers were bonded to a PET membrane containing 3.0-μm pores (Falcon, Cat# 353091) using a thin layer of liquid PDMS prepolymer as the mortar. PDMS prepolymer was spin-coated (4000 rpm for 60 sec) onto a glass slide. Subsequently, both the top and bottom channel layers were placed on the glass slide to transfer the thin layer of PDMS prepolymer onto the embossed PDMS surfaces. The membrane was then placed onto the bottom layer and sandwiched with the top layer. The combined layers were left at room temperature for 1 day to remove air bubbles and then placed in an oven at 60°C overnight to cure the PDMS glue. The PDMS devices were sterilized by placing them under UV light for 1 hour before the cell culture.
SARS-CoV-2 infection
One day before infection, Vero cells (10,000 cells), VeroE6/TMPRSS2 cells (10,000 cells), and HEK293-ACE2/TMPRSS2 cells were seeded into a 96-well plate. SARS-CoV-2 [1,000 TCID50 for Vero cells [Figure 4A]; 100 TCID50 for VeroE6/TMPRSS2 cells [Figure 4B] and HEK293-ACE2/TMPRSS2 cells [Figure 4C]] was inoculated and incubated at 37°C for 1 hour. The infected cells were washed, and 180 μl of culture medium was added. The culture supernatant (10 μl) was harvested at the indicated timepoints and used for RT–qPCR to quantify the viral RNA copy number (see “RT–qPCR” section below). In the infection experiments using human iPSC-derived airway and lung epithelial cells (Figures 4E and 4F), working viruses were diluted with Opti-MEM (Thermo Fisher Scientific, 11058021). The diluted viruses (1,000 TCID50 in 100 μl) were inoculated onto the apical side of the culture and incubated at 37 °C for 1 hour. The inoculated viruses were removed and washed twice with Opti-MEM. For collection of the viruses, 100 μl Opti-MEM was applied onto the apical side of the culture and incubated at 37 °C for 10 minutes. The Opti-MEM was collected and used for RT–qPCR to quantify the viral RNA copy number (see “RT–qPCR” section below). The infection experiments using an airway-on-a-chip system (Figures 4G–4I) were performed as described above (see “Airway-on-a-chips” section).
RT–qPCR
RT–qPCR was performed as previously described (Meng et al., 2022; Kimura et al., 2022b, 2022c; Yamasoba et al., 2022a; Suzuki et al., 2022; Saito et al., 2022; Motozono et al., 2021). Briefly, 5 μl culture supernatant was mixed with 5 μl of 2 × RNA lysis buffer [2% Triton X-100 (Nacalai Tesque, Cat# 35501-15), 50 mM KCl, 100 mM Tris-HCl (pH 7.4), 40% glycerol, 0.8 U/μl recombinant RNase inhibitor (Takara, Cat# 2313B)] and incubated at room temperature for 10 min. RNase-free water (90 μl) was added, and the diluted sample (2.5 μl) was used as the template for real-time RT-PCR performed according to the manufacturer’s protocol using One Step TB Green PrimeScript PLUS RT-PCR kit (Takara, Cat# RR096A) and the following primers: Forward N, 5'-AGC CTC TTC TCG TTC CTC ATC AC-3'; and Reverse N, 5'-CCG CCA TTG CCA GCC ATT C-3'. The viral RNA copy number was standardized with a SARS-CoV-2 direct detection RT-qPCR kit (Takara, Cat# RC300A). Fluorescent signals were acquired using a QuantStudio 1 Real-Time PCR system (Thermo Fisher Scientific), QuantStudio 3 Real-Time PCR system (Thermo Fisher Scientific), QuantStudio 5 Real-Time PCR system (Thermo Fisher Scientific), StepOne Plus Real-Time PCR system (Thermo Fisher Scientific), CFX Connect Real-Time PCR Detection system (Bio-Rad), Eco Real-Time PCR System (Illumina), qTOWER3 G Real-Time System (Analytik Jena) Thermal Cycler Dice Real Time System III (Takara) or 7500 Real-Time PCR System (Thermo Fisher Scientific).
Plaque assay
A plaque assay (Figure 4J) was performed as previously described (Kimura et al., 2022b, 2022c; Yamasoba et al., 2022a; Suzuki et al., 2022). Briefly, one day before infection, VeroE6/TMPRSS2 cells (100,000 cells) were seeded into a 24-well plate and infected with SARS-CoV-2 (0.5, 5, 50 and 500 TCID50) at 37°C for 1 hour. Mounting solution containing 3% FBS and 1.5% carboxymethyl cellulose (Wako, Cat# 039-01335) was overlaid, followed by incubation at 37°C. At 3 d.p.i., the culture medium was removed, and the cells were washed with PBS three times and fixed with 4% paraformaldehyde phosphate (Nacalai Tesque, Cat# 09154-85). The fixed cells were washed with tap water, dried, and stained with staining solution [0.1% methylene blue (Nacalai Tesque, Cat# 22412-14) in water] for 30 minutes. The stained cells were washed with tap water and dried, and the size of plaques was measured using Fiji software v2.2.0 (ImageJ).
Animal experiments
Animal experiments (Figures 5 and S5) were performed as previously described (Kimura et al., 2022b, 2022c; Yamasoba et al., 2022a; Suzuki et al., 2022; Saito et al., 2022; Tamura et al., 2022). Syrian hamsters (male, 4 weeks old) were purchased from Japan SLC Inc. (Shizuoka, Japan). For the virus infection experiments, hamsters were euthanized by intramuscular injection of a mixture of 0.15 mg/kg medetomidine hydrochloride (Domitor®, Nippon Zenyaku Kogyo), 2.0 mg/kg midazolam (Dormicum®, Fujifilm Wako, Cat# 135-13791) and 2.5 mg/kg butorphanol (Vetorphale®, Meiji Seika Pharma) or 0.15 mg/kg medetomidine hydrochloride, 4.0 mg/kg alphaxaone (Alfaxan®, Jurox) and 2.5 mg/kg butorphanol. Delta, BA.2, BA.5 and BA.2.75 (1,000 TCID50 or 5,000 TCID50 in 100 μl) or saline (100 μl) was intranasally inoculated under anesthesia. Oral swabs were collected at the indicated timepoints for the infection group with a lower inoculum (1,000 TCID50 per hamster). Body weight was recorded daily by 7 d.p.i. Enhanced pause (Penh), the ratio of time to peak expiratory follow relative to the total expiratory time (Rpef), and BPM were measured every day until 7 d.p.i. (see below). Subcutaneous oxygen saturation (SpO2, see below) was monitored at 0, 1, 3, 5, and 7 d.p.i. Lung tissues were anatomically collected at 2 and 5 d.p.i. The viral RNA load in the oral swabs and respiratory tissues was determined by RT–qPCR. These tissues were also used for IHC and histopathological analyses (see below).
Lung function test
Lung function tests (Figure 5A) were routinely performed as previously described (Kimura et al., 2022b, 2022c; Yamasoba et al., 2022a; Suzuki et al., 2022; Saito et al., 2022; Tamura et al., 2022). The three respiratory parameters (Penh, Rpef and BPM) were measured by using a Buxco Small Animal Whole Body Plethysmography system (DSI) according to the manufacturer's instructions. In brief, a hamster was placed in an unrestrained plethysmography chamber and allowed to acclimatize for 30 seconds. Then, data were acquired over a 2.5-minute period by using FinePointe Station and Review software v2.9.2.12849 (DSI). The state of oxygenation was examined by measuring SpO2 using a mouseOx PLUS pulse oximeter (STARR). SpO2 was measured for the infection group with a lower inoculum (1,000 TCID50 per hamster) by attaching a measuring chip to the neck of hamsters sedated by with 0.25 mg/kg medetomidine hydrochloride.
Immunohistochemistry
Immunohistochemistry (IHC) (Figures 5D and S5A–S5C) was performed as previously described (Kimura et al., 2022b, 2022c; Yamasoba et al., 2022a; Suzuki et al., 2022; Saito et al., 2022; Tamura et al., 2022) using an Autostainer Link 48 (Dako). The deparaffinized sections were exposed to EnVision FLEX target retrieval solution high pH (Agilent, Cat# K8004) for 20 minutes at 97°C for activation, and a mouse anti-SARS-CoV-2 N monoclonal antibody (clone 1035111, R&D Systems, Cat# MAB10474-SP, 1:400) was used as a primary antibody. The sections were sensitized using EnVision FLEX for 15 minutes and visualized by peroxidase-based enzymatic reaction with 3,3’-diaminobenzidine tetrahydrochloride (Dako, Cat# DM827) as substrate for 5 minutes. The N protein positivity (Figures 5D and S5A–S5C) was evaluated by certificated pathologists as previously described (Kimura et al., 2022b, 2022c; Yamasoba et al., 2022a; Suzuki et al., 2022; Saito et al., 2022; Tamura et al., 2022). Images were incorporated as virtual slides by NDP.scan software v3.2.4 (Hamamatsu Photonics). The N-protein positivity was measured as the area using Fiji software v2.2.0 (ImageJ).
H&E staining
H&E staining (Figures 5F and S5D) was performed as previously described (Kimura et al., 2022b, 2022c; Yamasoba et al., 2022a; Suzuki et al., 2022; Saito et al., 2022; Tamura et al., 2022). Briefly, excised animal tissues were fixed with 10% formalin neutral buffer solution and processed for paraffin embedding. The paraffin blocks were sectioned at a thickness of 3 μm and then mounted on MAS-GP-coated glass slides (Matsunami Glass, Cat# S9901). H&E staining was performed according to a standard protocol.
Histopathological scoring
Histopathological scoring (Figure 5G) was performed as previously described (Kimura et al., 2022b, 2022c; Yamasoba et al., 2022a; Suzuki et al., 2022; Saito et al., 2022; Tamura et al., 2022). Pathological features, including (i) bronchitis or bronchiolitis, (ii) hemorrhage with congestive edema, (iii) alveolar damage with epithelial apoptosis and macrophage infiltration, (iv) hyperplasia of type II pneumocytes, and (v) the area of hyperplasia of large type II pneumocytes, were evaluated by certified pathologists, and the degree of these pathological findings was arbitrarily scored using a four-tiered system as 0 (negative), 1 (weak), 2 (moderate), and 3 (severe). The "large type II pneumocytes" are type II pneumocytes with hyperplasia exhibiting more than 10-μm-diameter nuclei. We described "large type II pneumocytes" as one of the notable histopathological features of SARS-CoV-2 infection in our previous studies (Kimura et al., 2022b, 2022c; Yamasoba et al., 2022a; Suzuki et al., 2022; Saito et al., 2022; Tamura et al., 2022). The total histological score is the sum of these five indices.
For measurement of the inflammatory area in the infected lungs (Figures 5H and S5D), four hamsters infected with each virus were sacrificed at 5 d.p.i., and all four right lung lobes, the upper (anterior/cranial), middle, lower (posterior/caudal), and accessory lobes, were sectioned along with their bronchi. The tissue sections were stained by H&E, and the digital microscopic images were incorporated into virtual slides using NDP.scan software v3.2.4 (Hamamatsu Photonics). The inflammatory area including type II pneumocyte hyperplasia in the infected whole lungs was morphometrically analyzed using Fiji software v2.2.0 (ImageJ).
Quantification and statistical analysis
Statistical significance was tested using a two-sided Mann–Whitney U test, a two-sided Student's t test, a two-sided Welch’s t test, or a two-sided paired t-test unless otherwise noted. The tests above were performed using Prism 9 software v9.1.1 (GraphPad Software).
In the time-course experiments (Figures 3H, 4A–4H, 5A, 5B, and 5G), a multiple regression analysis including experimental conditions (i.e., the types of infected viruses) as explanatory variables and timepoints as qualitative control variables was performed to evaluate the difference between experimental conditions thorough all timepoints. The initial time point was removed from the analysis. The P value was calculated by a two-sided Wald test. Subsequently, familywise error rates (FWERs) were calculated by the Holm method. These analyses were performed in R v4.1.2 (https://www.r-project.org/).
In Figures 5D, 5F, and S5, photographs shown are the representative areas of at least two independent experiments by using four hamsters at each timepoint. In Figure S4D, photographs shown are the representatives of >20 fields of view taken for each sample.
Acknowledgments
We would like to thank all members belonging to G2P-Japan Consortium. We thank National Institute for Infectious Diseases, Japan, for providing clinical isolates of BA.2.75 and BA.2 and the technical assistance from The Research Support Center, Research Center for Human Disease Modeling, and Kyushu University Graduate School of Medical Sciences. We appreciate all data contributors, i.e., the authors and their originating laboratories responsible for obtaining the specimens, and their submitting laboratories for generating the genetic sequence and metadata and sharing via the GISAID Initiative, on which this research is based. The super-computing resource was provided by Human Genome Center at The University of Tokyo. This study was supported in-part by AMED Program on R&D of new generation vaccine including new modality application (JP223fa727002 to K. Sato); AMED Research Program on Emerging and Re-emerging Infectious Diseases (JP21fk0108574 to H.N.; JP21fk0108465 to A.S.; JP21fk0108493 to T.F.; JP22fk0108617 to T.F.; JP22fk0108146 to K. Sato; JP21fk0108494 to H.F., G2P-Japan Consortium, K. Shirakawa, T. Irie, K. Matsuno, S.T., T. Ikeda, T.F., and K. Sato); AMED CRDF Global Grant (JP22jk0210039 to A.S.); AMED Japan Program for Infectious Diseases Research and Infrastructure (JP22wm0325009 to A.S.; JP22wm0125008 to K. Matsuno); AMED BINDS (JP21am0101093 and JP22ama121037 to K. Maenaka); AMED CREST (JP21gm1610005 to K. Takayama); JST CREST (JPMJCR20H8 to T.H. and H.F.; JPMJCR20H4 to K. Sato); JSPS KAKENHI Scientific Research C (22K07089 to M. Toyoda; 22K07103, to T. Ikeda); JSPS Scientific Research B (21H02736 to T.F.); JSPS Early-Career Scientists (22K16375 to H.N.; 20K15767 to J.I.); JSPS Core-to-Core Program (A. Advanced Research Networks) (JPJSCCA20190008 to T.H. and K. Sato); JSPS Transformative Research Areas B JP20H05773 (to T.H.); JSPS Transformative Research Areas A JP20H05873 (to K. Maenaka); JSPS Research Fellow DC2 (22J11578 to K.U.); JSPS Leading Initiative for Excellent Young Researchers (LEADER) (to T. Ikeda); World-leading Innovative and Smart Education (WISE) Program 1801 from the Ministry of Education, Culture, Sports, Science and Technology (to N.N.); Tokyo Biochemical Research Foundation (to K. Sato); Takeda Science Foundation (to K. Maenaka and T. Ikeda); Naito Foundation (to T.H.); Shin-Nihon Foundation of Advanced Medical Research (to M. Toyoda and T. Ikeda); Waksman Foundation of Japan (to T. Ikeda); Hokkaido University, Global Facility Center, Pharma Science Open Unit (to K. Maenaka); Cooperative Research Program, Institute for Life and Medical Sciences, Kyoto University (to K. Maenaka); an intramural grant from Kumamoto University COVID-19 Research Projects (AMABIE) (to T. Ikeda); and the project of National Institute of Virology and Bacteriology, Programme EXCELES, funded by the European Union, Next Generation EU (LX22NPO5103 to J.Z.).
Author contributions
A.S., S.D., I.K., H.N., M. Toyoda, K.N., K.U., Y.K., S.F., D.Y., M.S., M.M.B., R. Shimizu, T. Irie, T.U., and T. Ikeda performed cell culture experiments. T.T., K. Tabata, R. Suzuki, H.I., N.N., K. Yoshimatsu, H.S., K. Matsuno, and T.F. performed animal experiments. Y.O., L.W., M. Tsuda, and S.T. performed histopathological analysis. J.Z. and G.S. performed yeast surface display assay. Y.A., S.K., K.S.-T., H.F., K. Maenaka, and T.H. performed structural analysis. S.D. and K. Takayama prepared AO, AO-ALI, and airway-on-a-chip systems. Y.Y. and T.N. performed generation and provision of human iPSC-derived airway and alveolar epithelial cells. H.A., M.N., K. Sadamasu, and K. Yoshimura performed viral genome sequencing analysis. J.K., A.T.-K., and K. Shirakawa contributed clinical sample collection. J.I. performed statistical, modeling, and bioinformatics analyses. J.I., K. Takayama, K.M., S.T., T. Ikeda, T.F., and K. Sato designed the experiments and interpreted the results. J.I., T.I., T.F., and K. Sato wrote the original manuscript. All authors reviewed and proofread the manuscript. The Genotype to Phenotype Japan (G2P-Japan) Consortium contributed to the project administration.
Declaration of interests
Y.Y. and T.N. are founders and shareholders of HiLung, Inc. Y.Y. is a co-inventor of patents (PCT/JP2016/057254; “method for inducing differentiation of alveolar epithelial cells,” PCT/JP2016/059786, “method of producing airway epithelial cells”).
Published: October 9, 2022
Footnotes
Supplemental information can be found online at https://doi.org/10.1016/j.chom.2022.10.003.
Supplemental information
Data and code availability
All databases/datasets used in this study are available from the GISAID database (https://www.gisaid.org) and GenBank database (https://www.gisaid.org; EPI_SET ID: EPI_SET_220804hy).
The atomic coordinates and cryo-EM maps for the structures of the BA.2.75 S protein closed state 1 (PDB code: 8GS6, EMDB code: 34221), closed state 2 (EMDB code: 34222), 1-up state (EMDB code: 34223) and in complex with hACE2 (EMDB code: 34224) have been deposited in the Protein Data Bank (www.rcsb.org) and Electron Microscopy Data Bank (www.ebi.ac.uk/emdb/). The computational codes used in the present study and the GISAID supplemental table for EPI_SET ID: EPI_SET_220804hy are available in the GitHub repository (https://github.com/TheSatoLab/Omicron_BA.2.75). Viral genome sequencing data for working viral stocks are available in the Sequence Read Archive (accession ID: PRJDB14324).
Additional Supplemental Items are available from Mendeley Data at https://data.mendeley.com/datasets/nrs6t42wbs [https://doi.org/10.17632/nrs6t42wbs.1]. Any additional information required to reanalyze the data reported in this work paper is available from the lead contact upon request.
References
- Adams P.D., Afonine P.V., Bunkóczi G., Chen V.B., Davis I.W., Echols N., Headd J.J., Hung L.W., Kapral G.J., Grosse-Kunstleve R.W., et al. Phenix: a comprehensive Python-based system for macromolecular structure solution. Acta Crystallogr. D Biol. Crystallogr. 2010;66:213–221. doi: 10.1107/S0907444909052925. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Arora P., Kempf A., Nehlmeier I., Schulz S.R., Cossmann A., Stankov M.V., Jäck H.M., Behrens G.M.N., Pöhlmann S., Hoffmann M. Augmented neutralisation resistance of emerging omicron subvariants BA.2.12.1, BA.4, and BA.5. Lancet Infect. Dis. 2022;22:1117–1118. doi: 10.1016/S1473-3099(22)00422-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Barnes C.O., Jette C.A., Abernathy M.E., Dam K.A., Esswein S.R., Gristick H.B., Malyutin A.G., Sharaf N.G., Huey-Tubman K.E., Lee Y.E., et al. SARS-CoV-2 neutralizing antibody structures inform therapeutic strategies. Nature. 2020;588:682–687. doi: 10.1038/s41586-020-2852-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bruel T., Hadjadj J., Maes P., Planas D., Seve A., Staropoli I., Guivel-Benhassine F., Porrot F., Bolland W.H., Nguyen Y., et al. Serum neutralization of SARS-CoV-2 Omicron sublineages BA.1 and BA.2 in patients receiving monoclonal antibodies. Nat. Med. 2022;28:1297–1302. doi: 10.1038/s41591-022-01792-5. [DOI] [PubMed] [Google Scholar]
- Cao Y., Song W., Wang L., Liu P., Yue C., Jian F., Yu Y., Yisimayi A., Wang P., Wang Y., et al. Characterizations of enhanced infectivity and antibody evasion of Omicron BA.2.75. Preprint at bioRxiv. 2022 doi: 10.1101/2022.07.18.500332. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cao Y., Wang J., Jian F., Xiao T., Song W., Yisimayi A., Huang W., Li Q., Wang P., An R., et al. Omicron escapes the majority of existing SARS-CoV-2 neutralizing antibodies. Preprint at bioRxiv. 2021 doi: 10.1101/2021.12.07.470392. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cao Y., Yisimayi A., Jian F., Song W., Xiao T., Wang L., Du S., Wang J., Li Q., Chen X., et al. BA.2.12.1, BA.4 and BA.5 escape antibodies elicited by Omicron infection. Nature. 2022;608:593–602. doi: 10.1038/s41586-022-04980-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Capella-Gutiérrez S., Silla-Martínez J.M., Gabaldón T. trimAl: a tool for automated alignment trimming in large-scale phylogenetic analyses. Bioinformatics. 2009;25:1972–1973. doi: 10.1093/bioinformatics/btp348. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cardone G., Heymann J.B., Steven A.C. One number does not fit all: mapping local variations in resolution in cryo-EM reconstructions. J. Struct. Biol. 2013;184:226–236. doi: 10.1016/j.jsb.2013.08.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cele S., Jackson L., Khoury D.S., Khan K., Moyo-Gwete T., Tegally H., San J.E., Cromer D., Scheepers C., Amoako D.G., et al. Omicron extensively but incompletely escapes Pfizer BNT162b2 neutralization. Nature. 2021;602:654–656. doi: 10.1038/d41586-41021-03824-41585. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cerutti G., Guo Y., Zhou T., Gorman J., Lee M., Rapp M., Reddem E.R., Yu J., Bahna F., Bimela J., et al. Potent SARS-CoV-2 neutralizing antibodies directed against spike N-terminal domain target a single Supersite. Cell Host Microbe. 2021;29:819–833.e7. doi: 10.1016/j.chom.2021.03.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chan K.K., Dorosky D., Sharma P., Abbasi S.A., Dye J.M., Kranz D.M., Herbert A.S., Procko E. Engineering human ACE2 to optimize binding to the spike protein of SARS coronavirus 2. Science. 2020;369:1261–1265. doi: 10.1126/science.abc0870. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen S., Zhou Y., Chen Y., Gu J. fastp: an ultra-fast all-in-one FASTQ preprocessor. Bioinformatics. 2018;34:i884–i890. doi: 10.1093/bioinformatics/bty560. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chi X., Yan R., Zhang J., Zhang G., Zhang Y., Hao M., Zhang Z., Fan P., Dong Y., Yang Y., et al. A neutralizing human antibody binds to the N-terminal domain of the Spike protein of SARS-CoV-2. Science. 2020;369:650–655. doi: 10.1126/science.abc6952. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cingolani P., Platts A., Wang L.L., Coon M., Nguyen T., Wang L., Land S.J., Lu X., Ruden D.M. A program for annotating and predicting the effects of single nucleotide polymorphisms, SnpEff: SNPs in the genome of Drosophila melanogaster strain w1118; iso-2; iso-3. Fly (Austin) 2012;6:80–92. doi: 10.4161/fly.19695. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Deguchi S., Tsuda M., Kosugi K., Sakamoto A., Mimura N., Negoro R., Sano E., Nobe T., Maeda K., Kusuhara H., et al. Usability of polydimethylsiloxane-based microfluidic devices in pharmaceutical research using human hepatocytes. ACS Biomater. Sci. Eng. 2021;7:3648–3657. doi: 10.1021/acsbiomaterials.1c00642. [DOI] [PubMed] [Google Scholar]
- Dejnirattisai W., Huo J., Zhou D., Zahradník J., Supasa P., Liu C., Duyvesteyn H.M.E., Ginn H.M., Mentzer A.J., Tuekprakhon A., et al. SARS-CoV-2 Omicron-B.1.1.529 leads to widespread escape from neutralizing antibody responses. Cell. 2022;185:467–484.e15. doi: 10.1016/j.cell.2021.12.046. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dong J., Zost S.J., Greaney A.J., Starr T.N., Dingens A.S., Chen E.C., Chen R.E., Case J.B., Sutton R.E., Gilchuk P., et al. Genetic and structural basis for SARS-CoV-2 variant neutralization by a two-antibody cocktail. Nat. Microbiol. 2021;6:1233–1244. doi: 10.1038/s41564-021-00972-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Emsley P., Cowtan K. Coot: model-building tools for molecular graphics. Acta Crystallogr. D Biol. Crystallogr. 2004;60:2126–2132. doi: 10.1107/S0907444904019158. [DOI] [PubMed] [Google Scholar]
- Ferreira I.A.T.M., Kemp S.A., Datir R., Saito A., Meng B., Rakshit P., Takaori-Kondo A., Kosugi Y., Uriu K., Kimura I., et al. SARS-CoV-2 B.1.617 Mutations L452R and E484Q Are Not Synergistic for Antibody Evasion. J. Infect. Dis. 2021;224:989–994. doi: 10.1093/infdis/jiab368. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Garcia-Beltran W.F., Lam E.C., St. Denis K., Nitido A.D., Garcia Z.H., Hauser B.M., Feldman J., Pavlovic M.N., Gregory D.J., Poznansky M.C., et al. Multiple SARS-CoV-2 variants escape neutralization by vaccine-induced humoral immunity. Cell. 2021;184:2372–2383.e9. doi: 10.1016/j.cell.2021.03.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- GitHub BA.2 sublineage with S:K147E, W152R, F157L, I210V, G257S, D339H, G446S, N460K, R493Q (73 seq as of 2022-06-29, mainly India) (June 21, 2022) 2022. https://github.com/cov-lineages/pango-designation/issues/773
- Goddard T.D., Huang C.C., Meng E.C., Pettersen E.F., Couch G.S., Morris J.H., Ferrin T.E. UCSF ChimeraX: meeting modern challenges in visualization and analysis. Protein Sci. 2018;27:14–25. doi: 10.1002/pro.3235. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gotoh S., Ito I., Nagasaki T., Yamamoto Y., Konishi S., Korogi Y., Matsumoto H., Muro S., Hirai T., Funato M., et al. Generation of alveolar epithelial spheroids via isolated progenitor cells from human pluripotent stem cells. Stem Cell Rep. 2014;3:394–403. doi: 10.1016/j.stemcr.2014.07.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gruell H., Vanshylla K., Korenkov M., Tober-Lau P., Zehner M., Münn F., Janicki H., Augustin M., Schommers P., Sander L.E., et al. SARS-CoV-2 Omicron sublineages exhibit distinct antibody escape patterns. Cell Host Microbe. 2022;30:1231–1241.e6. doi: 10.1016/j.chom.2022.07.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hachmann N.P., Miller J., Collier A.Y., Ventura J.D., Yu J., Rowe M., Bondzie E.A., Powers O., Surve N., Hall K., et al. Neutralization escape by SARS-CoV-2 Omicron subvariants BA.2.12.1, BA.4, and BA.5. N. Engl. J. Med. 2022;387:86–88. doi: 10.1056/NEJMc2206576. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Harvey W.T., Carabelli A.M., Jackson B., Gupta R.K., Thomson E.C., Harrison E.M., Ludden C., Reeve R., Rambaut A., et al. COVID-19 Genomics UK (COG-UK) Consortium SARS-CoV-2 variants, spike mutations and immune escape. Nat. Rev. Microbiol. 2021;19:409–424. doi: 10.1038/s41579-021-00573-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hashiguchi T., Ose T., Kubota M., Maita N., Kamishikiryo J., Maenaka K., Yanagi Y. Structure of the measles virus hemagglutinin bound to its cellular receptor SLAM. Nat. Struct. Mol. Biol. 2011;18:135–141. doi: 10.1038/nsmb.1969. [DOI] [PubMed] [Google Scholar]
- Hashimoto R., Takahashi J., Shirakura K., Funatsu R., Kosugi K., Deguchi S., Yamamoto M., Tsunoda Y., Morita M., Muraoka K., et al. SARS-CoV-2 disrupts the respiratory vascular barrier by suppressing Claudin-5 expression. Sci. Adv. 2022;8:eabo6783. doi: 10.1126/sciadv.abo6783. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hsieh C.L., Goldsmith J.A., Schaub J.M., DiVenere A.M., Kuo H.C., Javanmardi K., Le K.C., Wrapp D., Lee A.G., Liu Y., et al. Structure-based design of prefusion-stabilized SARS-CoV-2 spikes. Science. 2020;369:1501–1505. doi: 10.1126/science.abd0826. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jackson C.B., Farzan M., Chen B., Choe H. Mechanisms of SARS-CoV-2 entry into cells. Nat. Rev. Mol. Cell Biol. 2022;23:3–20. doi: 10.1038/s41580-021-00418-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jurrus E., Engel D., Star K., Monson K., Brandi J., Felberg L.E., Brookes D.H., Wilson L., Chen J., Liles K., et al. Improvements to the APBS biomolecular solvation software suite. Protein Sci. 2018;27:112–128. doi: 10.1002/pro.3280. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Khan K., Karim F., Ganga Y., Bernstein M., Jule Z., Reedoy K., Cele S., Lustig G., Amoako D., Wolter N., et al. Omicron sub-lineages BA.4/BA.5 escape BA.1 infection elicited neutralizing immunity. Preprint at medRxiv. 2022 doi: 10.1101/2022.1104.1129.22274477. [DOI] [Google Scholar]
- Khare S., Gurry C., Freitas L., Schultz M.B., Bach G., Diallo A., Akite N., Ho J., Lee R.T., Yeo W., et al. GISAID's role in pandemic response. China CDC Wkly. 2021;3:1049–1051. doi: 10.46234/ccdcw2021.255. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kim C., Ryu D.K., Lee J., Kim Y.I., Seo J.M., Kim Y.G., Jeong J.H., Kim M., Kim J.I., Kim P., et al. A therapeutic neutralizing antibody targeting receptor binding domain of SARS-CoV-2 spike protein. Nat. Commun. 2021;12:288. doi: 10.1038/s41467-020-20602-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kimura I., Kosugi Y., Wu J., Zahradnik J., Yamasoba D., Butlertanaka E.P., Tanaka Y.L., Uriu K., Liu Y., Morizako N., et al. The SARS-CoV-2 Lambda variant exhibits enhanced infectivity and immune resistance. Cell Rep. 2022;38:110218. doi: 10.1016/j.celrep.2021.110218. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kimura I., Yamasoba D., Nasser H., Zahradnik J., Kosugi Y., Wu J., Nagata K., Uriu K., Tanaka Y.L., Ito J., et al. SARS-CoV-2 spike S375F mutation characterizes the Omicron BA.1 variant. Preprint at bioRxiv. 2022 doi: 10.1101/2022.1104.1103.486864. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kimura I., Yamasoba D., Tamura T., Nao N., Suzuki T., Oda Y., Mitoma S., Ito J., Nasser H., Zahradnik J., et al. Virological characteristics of the novel SARS-CoV-2 Omicron variants including BA.4 and BA.5. Cell. 2022;185:2103–2115.e19. doi: 10.1016/j.cell.2022.1009.1018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kislaya I., Casaca P., Borges V., et al. SARS-CoV-2 BA.5 vaccine breakthrough risk and severity compared with BA.2: a case-case and cohort study using electronic health records in Portugal. Preprint at medRxiv. 2022 doi: 10.1101/2022.1107.1125.22277996. [DOI] [Google Scholar]
- Kondo N., Miyauchi K., Matsuda Z. Monitoring viral-mediated membrane fusion using fluorescent reporter methods. Curr. Protoc. Cell Biol. 2011;Chapter doi: 10.1002/0471143030.cb2609s50. Unit 26.9. [DOI] [PubMed] [Google Scholar]
- Konishi S., Gotoh S., Tateishi K., Yamamoto Y., Korogi Y., Nagasaki T., Matsumoto H., Muro S., Hirai T., Ito I., et al. Directed induction of functional multi-ciliated cells in proximal airway epithelial spheroids from human pluripotent stem cells. Stem Cell Rep. 2016;6:18–25. doi: 10.1016/j.stemcr.2015.11.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lan J., Ge J., Yu J., Shan S., Zhou H., Fan S., Zhang Q., Shi X., Wang Q., Zhang L., et al. Structure of the SARS-CoV-2 spike receptor-binding domain bound to the ACE2 receptor. Nature. 2020;581:215–220. doi: 10.1038/s41586-020-2180-5. [DOI] [PubMed] [Google Scholar]
- Li H. Minimap2: pairwise alignment for nucleotide sequences. Bioinformatics. 2018;34:3094–3100. doi: 10.1093/bioinformatics/bty191. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li H., Durbin R. Fast and accurate short read alignment with Burrows-Wheeler transform. Bioinformatics. 2009;25:1754–1760. doi: 10.1093/bioinformatics/btp324. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li H., Handsaker B., Wysoker A., Fennell T., Ruan J., Homer N., Marth G., Abecasis G., Durbin R., 1000 Genome Project Data Processing Subgroup The sequence alignment/map format and SAMtools. Bioinformatics. 2009;25:2078–2079. doi: 10.1093/bioinformatics/btp352. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu L., Iketani S., Guo Y., Chan J.F., Wang M., Liu L., Luo Y., Chu H., Huang Y., Nair M.S., et al. Striking antibody evasion manifested by the Omicron variant of SARS-CoV-2. Nature. 2021;602:676–681. doi: 10.1038/d41586-41021-03826-41583. [DOI] [PubMed] [Google Scholar]
- Liu L., Wang P., Nair M.S., Yu J., Rapp M., Wang Q., Luo Y., Chan J.F., Sahi V., Figueroa A., et al. Potent neutralizing antibodies against multiple epitopes on SARS-CoV-2 spike. Nature. 2020;584:450–456. doi: 10.1038/s41586-020-2571-7. [DOI] [PubMed] [Google Scholar]
- Liu Y., Arase N., Kishikawa J.-i., Hirose M., Li S., Tada A., Matsuoka S., Arakawa A., Akamatsu K., Ono C., et al. The SARS-CoV-2 Delta variant is poised to acquire complete resistance to wild-type spike vaccines. Preprint at bioRxiv. 2021 doi: 10.1101/2021.1108.1122.457114. [DOI] [Google Scholar]
- Lok S.M. An NTD Supersite of attack. Cell Host Microbe. 2021;29:744–746. doi: 10.1016/j.chom.2021.04.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lyke K.E., Atmar R.L., Islas C.D., Posavad C.M., Szydlo D., Paul Chourdhury R., Deming M.E., Eaton A., Jackson L.A., Branche A.R., et al. Rapid decline in vaccine-boosted neutralizing antibodies against SARS-CoV-2 Omicron variant. Cell Rep. Med. 2022;3:100679. doi: 10.1016/j.xcrm.2022.100679. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Matsuyama S., Nao N., Shirato K., Kawase M., Saito S., Takayama I., Nagata N., Sekizuka T., Katoh H., Kato F., et al. Enhanced isolation of SARS-CoV-2 by TMPRSS2-expressing cells. Proc. Natl. Acad. Sci. USA. 2020;117:7001–7003. doi: 10.1073/pnas.2002589117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McCallum M., De Marco A., Lempp F.A., Tortorici M.A., Pinto D., Walls A.C., Beltramello M., Chen A., Liu Z., Zatta F., et al. N-terminal domain antigenic mapping reveals a site of vulnerability for SARS-CoV-2. Cell. 2021;184:2332–2347.e16. doi: 10.1016/j.cell.2021.03.028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Meng B., Abdullahi A., Ferreira I.A.T.M., Goonawardane N., Saito A., Kimura I., Yamasoba D., Gerber P.P., Fatihi S., Rathore S., et al. Altered TMPRSS2 usage by SARS-CoV-2 Omicron impacts tropism and fusogenicity. Nature. 2022;603:706–714. doi: 10.1038/s41586-022-04474-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mirdita M., Schütze K., Moriwaki Y., Heo L., Ovchinnikov S., Steinegger M. ColabFold: making protein folding accessible to all. Nat. Methods. 2022;19:679–682. doi: 10.1038/s41592-022-01488-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mittal A., Khattri A., Verma V. Structural and antigenic variations in the spike protein of emerging SARS-CoV-2 variants. PLOS Pathog. 2022;18 doi: 10.1371/journal.ppat.1010260. e1010260. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Motozono C., Toyoda M., Zahradnik J., Saito A., Nasser H., Tan T.S., Ngare I., Kimura I., Uriu K., Kosugi Y., et al. SARS-CoV-2 spike L452R variant evades cellular immunity and increases infectivity. Cell Host Microbe. 2021;29:1124–1136.e11. doi: 10.1016/j.chom.2021.06.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Niwa H., Yamamura K., Miyazaki J. Efficient selection for high-expression transfectants with a novel eukaryotic vector. Gene. 1991;108:193–199. doi: 10.1016/0378-1119(91)90434-d. [DOI] [PubMed] [Google Scholar]
- Ozono S., Zhang Y., Ode H., Sano K., Tan T.S., Imai K., Miyoshi K., Kishigami S., Ueno T., Iwatani Y., et al. SARS-CoV-2 D614G spike mutation increases entry efficiency with enhanced ACE2-binding affinity. Nat. Commun. 2021;12:848. doi: 10.1038/s41467-021-21118-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ozono S., Zhang Y., Tobiume M., Kishigami S., Tokunaga K. Super-rapid quantitation of the production of HIV-1 harboring a luminescent peptide tag. J. Biol. Chem. 2020;295:13023–13030. doi: 10.1074/jbc.RA120.013887. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pettersen E.F., Goddard T.D., Huang C.C., Couch G.S., Greenblatt D.M., Meng E.C., Ferrin T.E. UCSF Chimera—a visualization system for exploratory research and analysis. J. Comput. Chem. 2004;25:1605–1612. doi: 10.1002/jcc.20084. [DOI] [PubMed] [Google Scholar]
- Pinto D., Park Y.J., Beltramello M., Walls A.C., Tortorici M.A., Bianchi S., Jaconi S., Culap K., Zatta F., De Marco A., et al. Cross-neutralization of SARS-CoV-2 by a human monoclonal SARS-CoV antibody. Nature. 2020;583:290–295. doi: 10.1038/s41586-020-2349-y. [DOI] [PubMed] [Google Scholar]
- Planas D., Saunders N., Maes P., Guivel-Benhassine F., Planchais C., Buchrieser J., Bolland W.H., Porrot F., Staropoli I., Lemoine F., et al. Considerable escape of SARS-CoV-2 Omicron to antibody neutralization. Nature. 2021;602:671–675. doi: 10.1038/d41586-41021-03827-41582. [DOI] [PubMed] [Google Scholar]
- Punjani A., Rubinstein J.L., Fleet D.J., Brubaker M.A. cryoSPARC: algorithms for rapid unsupervised cryo-EM structure determination. Nat. Methods. 2017;14:290–296. doi: 10.1038/nmeth.4169. [DOI] [PubMed] [Google Scholar]
- Qu P., Faraone J., Evans J.P., Zou X., Zheng Y.M., Carlin C., Bednash J.S., Lozanski G., Mallampalli R.K., Saif L.J., et al. Neutralization of the SARS-CoV-2 omicron BA.4/5 and BA.2.12.1 Subvariants. N. Engl. J. Med. 2022;386:2526–2528. doi: 10.1056/NEJMc2206725. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reed L.J., Muench H. A simple method of estimating fifty percent endpoints. Am. J. Hyg. 1938;27:493–497. [Google Scholar]
- Reeves P.J., Callewaert N., Contreras R., Khorana H.G. Structure and function in rhodopsin: high-level expression of rhodopsin with restricted and homogeneous N-glycosylation by a tetracycline-inducible N-acetylglucosaminyltransferase I-negative HEK293S stable mammalian cell line. Proc. Natl. Acad. Sci. USA. 2002;99:13419–13424. doi: 10.1073/pnas.212519299. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Saito A., Irie T., Suzuki R., Maemura T., Nasser H., Uriu K., Kosugi Y., Shirakawa K., Sadamasu K., Kimura I., et al. Enhanced fusogenicity and pathogenicity of SARS-CoV-2 Delta P681R mutation. Nature. 2022;602:300–306. doi: 10.1038/s41586-021-04266-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sano E., Suzuki T., Hashimoto R., Itoh Y., Sakamoto A., Sakai Y., Saito A., Okuzaki D., Motooka D., Muramoto Y., et al. Cell response analysis in SARS-CoV-2 infected bronchial organoids. Commun. Biol. 2022;5:516. doi: 10.1038/s42003-022-03499-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shen X., Chalkias S., Feng J., Chen X., Zhou H., Marshall J.C., Girard B., Tomassini J.E., Aunins A., Das R., et al. Neutralization of SARS-CoV-2 omicron BA.2.75 after mRNA-1273 Vaccination. N. Engl. J. Med. 2022;387:1234–1236. doi: 10.1056/NEJMc2210648. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stalls V., Lindenberger J., Gobeil S.M., Henderson R., Parks R., Barr M., Deyton M., Martin M., Janowska K., Huang X., et al. Cryo-EM structures of SARS-CoV-2 Omicron BA.2 spike. Cell Rep. 2022;39:111009. doi: 10.1016/j.celrep.2022.111009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stamatakis A. RAxML version 8: a tool for phylogenetic analysis and post-analysis of large phylogenies. Bioinformatics. 2014;30:1312–1313. doi: 10.1093/bioinformatics/btu033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Suryadevara N., Shrihari S., Gilchuk P., VanBlargan L.A., Binshtein E., Zost S.J., Nargi R.S., Sutton R.E., Winkler E.S., Chen E.C., et al. Neutralizing and protective human monoclonal antibodies recognizing the N-terminal domain of the SARS-CoV-2 spike protein. Cell. 2021;184:2316–2331.e15. doi: 10.1016/j.cell.2021.03.029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Suzuki R., Yamasoba D., Kimura I., Wang L., Kishimoto M., Ito J., Morioka Y., Nao N., Nasser H., Uriu K., et al. Attenuated fusogenicity and pathogenicity of SARS-CoV-2 Omicron variant. Nature. 2022;603:700–705. doi: 10.1038/s41586-022-04462-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Takashita E., Kinoshita N., Yamayoshi S., Sakai-Tagawa Y., Fujisaki S., Ito M., Iwatsuki-Horimoto K., Chiba S., Halfmann P., Nagai H., et al. Efficacy of antibodies and antiviral drugs against Covid-19 Omicron variant. N. Engl. J. Med. 2022;386:995–998. doi: 10.1056/NEJMc2119407. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Takashita E., Kinoshita N., Yamayoshi S., Sakai-Tagawa Y., Fujisaki S., Ito M., Iwatsuki-Horimoto K., Halfmann P., Watanabe S., Maeda K., et al. Efficacy of antiviral agents against the SARS-CoV-2 Omicron subvariant BA.2. N. Engl. J. Med. 2022;386:1475–1477. doi: 10.1056/NEJMc2201933. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tamura T., Yamasoba D., Oda Y., Ito J., Kamasaki T., Nao N., Hashimoto R., Fujioka Y., Suzuki R., Wang L., et al. Comparative pathogenicity of SARS-CoV-2 Omicron subvariants including BA.1, BA.2, and BA.5. Preprint at bioRxiv. 2022 doi: 10.1101/2022.1108.1105.502758. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tuekprakhon A., Nutalai R., Dijokaite-Guraliuc A., Zhou D., Ginn H.M., Selvaraj M., Liu C., Mentzer A.J., Supasa P., Duyvesteyn H.M.E., et al. Antibody escape of SARS-CoV-2 Omicron BA.4 and BA.5 from vaccine and BA.1 serum. Cell. 2022;185:2422–2433.e13. doi: 10.1016/j.cell.2022.06.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Uriu K., Cárdenas P., Muñoz E., Barragan V., Kosugi Y., Shirakawa K., Takaori-Kondo A., Ecuador-COVID19 Consortium, The Genotype to Phenotype Japan (G2P-Japan) Consortium. Sato K. Characterization of the immune resistance of SARS-CoV-2 Mu variant and the robust immunity induced by Mu infection. J. Infect. Dis. 2022;226:1200–1203. doi: 10.1093/infdis/jiac053. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Uriu K., Kimura I., Shirakawa K., Takaori-Kondo A., Nakada T.A., Kaneda A., Nakagawa S., Sato K., Genotype to Phenotype Japan (G2P-Japan) Consortium Neutralization of the SARS-CoV-2 Mu variant by convalescent and vaccine serum. N. Engl. J. Med. 2021;385:2397–2399. doi: 10.1056/NEJMc2114706. [DOI] [PMC free article] [PubMed] [Google Scholar]
- VanBlargan L.A., Errico J.M., Halfmann P.J., Zost S.J., Crowe J.E., Purcell L.A., Kawaoka Y., Corti D., Fremont D.H., Diamond M.S. An infectious SARS-CoV-2 B.1.1.529 Omicron virus escapes neutralization by therapeutic monoclonal antibodies. Nat. Med. 2022;28:490–495. doi: 10.1038/s41591-021-01678-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Voss W.N., Hou Y.J., Johnson N.V., Delidakis G., Kim J.E., Javanmardi K., Horton A.P., Bartzoka F., Paresi C.J., Tanno Y., et al. Prevalent, protective, and convergent IgG recognition of SARS-CoV-2 non-RBD spike epitopes. Science. 2021;372:1108–1112. doi: 10.1126/science.abg5268. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang Q., Guo Y., Iketani S., Nair M.S., Li Z., Mohri H., Wang M., Yu J., Bowen A.D., Chang J.Y., et al. Antibody evasion by SARS-CoV-2 Omicron subvariants BA.2.12.1, BA.4 and BA.5, & BA.5. Nature. 2022;608:603–608. doi: 10.1038/s41586-022-05053-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Westendorf K., Žentelis S., Wang L., Foster D., Vaillancourt P., Wiggin M., Lovett E., van der Lee R., Hendle J., Pustilnik A., et al. LY-CoV1404 (bebtelovimab) potently neutralizes SARS-CoV-2 variants. Cell Rep. 2022;39:110812. doi: 10.1016/j.celrep.2022.110812. [DOI] [PMC free article] [PubMed] [Google Scholar]
- WHO Tracking SARS-CoV-2 variants (July 19, 2022) 2022. https://www.who.int/en/activities/tracking-SARS-CoV-2-variants
- Williams C.J., Headd J.J., Moriarty N.W., Prisant M.G., Videau L.L., Deis L.N., Verma V., Keedy D.A., Hintze B.J., Chen V.B., et al. MolProbity: more and better reference data for improved all-atom structure validation. Protein Sci. 2018;27:293–315. doi: 10.1002/pro.3330. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xiao T., Lu J., Zhang J., Johnson R.I., McKay L.G.A., Storm N., Lavine C.L., Peng H., Cai Y., Rits-Volloch S., et al. A trimeric human angiotensin-converting enzyme 2 as an anti-SARS-CoV-2 agent. Nat. Struct. Mol. Biol. 2021;28:202–209. doi: 10.1038/s41594-020-00549-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yamamoto M., Kiso M., Sakai-Tagawa Y., Iwatsuki-Horimoto K., Imai M., Takeda M., Kinoshita N., Ohmagari N., Gohda J., Semba K., et al. The anticoagulant nafamostat potently inhibits SARS-CoV-2 S protein-mediated fusion in a cell fusion assay system and viral infection in vitro in a cell-yype-dependent manner. Viruses. 2020;12:629. doi: 10.3390/v12060629. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yamamoto Y., Gotoh S., Korogi Y., Seki M., Konishi S., Ikeo S., Sone N., Nagasaki T., Matsumoto H., Muro S., et al. Long-term expansion of alveolar stem cells derived from human iPS cells in organoids. Nat. Methods. 2017;14:1097–1106. doi: 10.1038/nmeth.4448. [DOI] [PubMed] [Google Scholar]
- Yamasoba D., Kimura I., Nasser H., Morioka Y., Nao N., Ito J., Uriu K., Tsuda M., Zahradnik J., Shirakawa K., et al. Virological characteristics of the SARS-CoV-2 Omicron BA.2 spike. Cell. 2022;185:2103–2115.e19. doi: 10.1016/j.cell.2022.04.035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yamasoba D., Kosugi Y., Kimura I., Fujita S., Uriu K., Ito J., Sato K., Genotype to Phenotype Japan (G2P-Japan) Consortium Neutralisation sensitivity of SARS-CoV-2 omicron subvariants to therapeutic monoclonal antibodies. Lancet Infect. Dis. 2022;22:942–943. doi: 10.1016/S1473-3099(22)00365-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zahradník J., Dey D., Marciano S., Kolářová L., Charendoff C.I., Subtil A., Schreiber G. A protein-engineered, enhanced yeast display platform for rapid evolution of challenging targets. ACS Synth. Biol. 2021;10:3445–3460. doi: 10.1021/acssynbio.1c00395. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zahradník J., Marciano S., Shemesh M., Zoler E., Harari D., Chiaravalli J., Meyer B., Rudich Y., Li C., Marton I., et al. SARS-CoV-2 variant prediction and antiviral drug design are enabled by RBD in vitro evolution. Nat. Microbiol. 2021;6:1188–1198. doi: 10.1038/s41564-021-00954-4. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
All databases/datasets used in this study are available from the GISAID database (https://www.gisaid.org) and GenBank database (https://www.gisaid.org; EPI_SET ID: EPI_SET_220804hy).
The atomic coordinates and cryo-EM maps for the structures of the BA.2.75 S protein closed state 1 (PDB code: 8GS6, EMDB code: 34221), closed state 2 (EMDB code: 34222), 1-up state (EMDB code: 34223) and in complex with hACE2 (EMDB code: 34224) have been deposited in the Protein Data Bank (www.rcsb.org) and Electron Microscopy Data Bank (www.ebi.ac.uk/emdb/). The computational codes used in the present study and the GISAID supplemental table for EPI_SET ID: EPI_SET_220804hy are available in the GitHub repository (https://github.com/TheSatoLab/Omicron_BA.2.75). Viral genome sequencing data for working viral stocks are available in the Sequence Read Archive (accession ID: PRJDB14324).
Additional Supplemental Items are available from Mendeley Data at https://data.mendeley.com/datasets/nrs6t42wbs [https://doi.org/10.17632/nrs6t42wbs.1]. Any additional information required to reanalyze the data reported in this work paper is available from the lead contact upon request.