Abstract
Human immunodeficiency virus (HIV) infection causes extensive phenotypic alterations in lymphocytes. Cellular markers that are normally absent or expressed at low levels on quiescent cells are upregulated throughout the disease course. The transmembrane form of regeneration and tolerance factor (RTF) is expressed at negligible levels on resting T cells but is quickly upregulated following in vitro stimulation and activation. Recently, we reported that expression of RTF was significantly higher in cells from HIV-seropositive (HIV+) individuals than in cells from HIV-seronegative (HIV−) individuals. Because T cells from HIV+ individuals express markers reflecting chronic activation, we hypothesized that these in vivo-activated cells would coexpress RTF. Flow cytometry was used to assess RTF expression on activated (CD38+ and HLA-DR+) CD4+ and CD8+ T cells. HIV+ individuals had higher percentages of RTF+ CD38+ (P < 0.0001) or RTF+ HLA-DR+ (P = 0.0001) CD4+ T cells than HIV− individuals. In HIV+ individuals, increased percentages of CD4+ T cells that were RTF+, RTF+ CD38+, and RTF+ HLA-DR+ correlated inversely with the absolute number and percentage of CD4+ T cells and correlated positively with plasma β2-microglobulin concentrations. HIV+ individuals had higher percentages of CD8+ T cells that were RTF+ CD38+ (P = 0.0001) or RTF+ HLA-DR+ (P = 0.0010). In HIV+ individuals, increased percentages of CD8+ T cells that were RTF+ HLA-DR+ correlated inversely with the percentage of CD4+ T cells, and high percentages of CD8+ T cells that were RTF+ CD38+ correlated positively with plasma β2-microglobulin levels. These findings strongly suggest that increased RTF expression is a correlate of HIV-associated immune system activation.
Regeneration and tolerance factor (RTF) is a protein capable of modulating the immune system during a variety of conditions. RTF is encoded by the gene TJ6, and sequence and hydrophobicity analysis of a murine cDNA clone revealed a hydrophobic carboxy terminus with seven transmembrane-spanning domains and a hydrophilic external amino terminus (3). RTF shares amino acid homology with a vacuolar H+-ATPase (21) and the recently identified 75-kDa TIRC7 (28). RTF also has protein kinase C sites and serine protease-sensitive sites (3). RTF and TIRC7 are of similar size, are transiently upregulated in early T-cell activation, and are involved in allograft survival and modulation of the immune response.
The native 70-kDa RTF protein is posttranslationally modified to yield a 50-kDa integral membrane protein and a 20-kDa soluble protein (3, 20). The 50-kDa transmembrane protein exhibits a unique expression pattern on a variety of developing cells and regenerating tissues. During the course of pregnancy, RTF is expressed within the developing fetal placental unit (26, 27). It is believed that RTF is involved in regulating the TH1/TH2 cytokine environment at the maternal-fetal interface and plays a crucial role in fetal allograft acceptance or rejection during pregnancy. Establishment of peripheral tolerance and a successful pregnancy is characterized by a shift from a TH1 cytokine profile to a TH2 profile (17, 29). It has been demonstrated that RTF is a marker for predicting the outcome of pregnancy because of its distinctive expression pattern (6, 23). RTF is abundantly expressed on the peripheral blood B lymphocytes during normal pregnancies but is predominantly expressed by natural killer (NK) cells during pregnancies that end abruptly due to spontaneous abortion. Interestingly, T cells express negligible or undetectable levels of RTF in normal individuals (7). These studies suggest that RTF is an important immunoregulatory marker during pregnancy.
Recently, RTF has been shown to also play a role in tumor survival and growth in vivo (1). RTF is expressed at high levels by leukemic B-cell lines representing various stages of differentiation as well as by peripheral blood B cells of patients with chronic lymphocytic leukemia. Tumor progression is also related to predominance of TH2 cytokines (16, 22). It is believed that RTF expression may allow B-cell tumors to evade immune response destruction by suppression of tumor-specific killer cells (1).
Interestingly, human immunodeficiency virus (HIV) disease progression, like pregnancy, has been linked to cytokine dysregulation or a TH1-to-TH2 cytokine switch (4, 5). Cytokines are potent immunoregulatory molecules, and aberrant cytokine production as a result of immune activation perpetuates the chronic immune system activation, thereby greatly enhancing HIV replication and disease progression. Since it appears that RTF may be expressed during situations in which there is a bias toward TH2 cytokines, it was postulated that cells from HIV-seropositive (HIV+) individuals might express increased amounts of RTF. In a previous study, HIV-associated induction of RTF expression was observed on both B and T cells of HIV+ individuals (7). Therefore, the goal of the present study was to determine whether elevated levels of RTF are coordinately expressed on the surface of T cells bearing markers of activation (e.g., CD38 and HLA-DR). RTF may mark a unique subset of activated lymphocytes in HIV+ individuals. The data presented herein support the contention that high levels of RTF expression correlate with T-cell subset activation in HIV+ individuals.
MATERIALS AND METHODS
Study participants and specimen collection.
Peripheral venous blood collected in sodium heparin anticoagulant was obtained from 26 HIV+ individuals (15 males and 11 females) at Mt. Sinai Hospital, Chicago, Ill. The HIV+ individuals were between the ages of 21 and 64 years (mean, 35.9 years). Of the HIV+ subjects, five had CD4+ counts of >500/mm3, 14 had CD4+ counts between 200 and 499/mm3, and nine had CD4+ counts of <200 mm3. CD4+-T-cell determinations were made as previously described (7). Also, of the HIV+ individuals, 12 were inpatients and 14 were outpatients. Clinical information was obtained through retrospective analysis of medical records for inpatients only. No clinical data were available for outpatients. Five of the nine HIV+ individuals with CD4+-T-cell counts of <200/mm3 also had documented conditions listed in the 1993 AIDS surveillance case definition. Nine HIV+ individuals received antiretroviral treatment with combinations of reverse transcriptase inhibitors with (n = 6) or without (n = 3) a protease inhibitor. Plasma HIV RNA levels were measured for only 3 of the 26 HIV+ individuals (of which all were undergoing therapy with reverse transcriptase and protease inhibitors) and were <100 copies/ml for each individual. Sodium heparin-anticoagulated peripheral blood samples were also obtained from 18 HIV-seronegative (HIV−) healthy donors (8 males and 10 females) at Finch University of Health Sciences/The Chicago Medical School, North Chicago, Ill. The HIV− donors were between the ages of 25 and 58 years (mean, 38.2 years). This study was approved by the institutional review boards at Mt. Sinai Hospital and Finch University of Health Sciences/The Chicago Medical School.
Purification and evaluation of anti-RTF MAb specificity.
Monoclonal antibody (MAb) against the membrane form of RTF was purified and conjugated to fluorescein isothiocyanate (FITC) as previously described (7, 20). Following conjugation, the FITC (495 nm)-to-protein (280 nm) ratio of the anti-RTF-FITC MAb was determined spectrophotometrically to be 1.1. The protein concentration was adjusted to 0.77 mg/ml. Finally, the reactivity of the purified MAb to a synthetic peptide representing amino acids 488 to 514 of the RTF gene sequence was confirmed by an enzyme-linked immunosorbent assay (ELISA). The specificity of the anti-RTF-FITC MAb was also confirmed by ELISA and demonstration that preincubation of the antibody with the peptide abolished membrane binding. Also, the specificity of anti-RTF-FITC MAb was evaluated by comparison to fluorescence obtained by labeling with mouse immunoglobulin G2a (IgG2a; Ortho Diagnostics, Raritan, N.J.) and IgG1 (Becton Dickinson, San Jose, Calif.) isotype control antibodies conjugated to FITC and phycoerythrin (PE) and unconjugated mouse IgG1 (Pharmingen, San Diego, Calif.).
Lymphocyte immunophenotyping and flow cytometric analysis.
Three-color flow cytometric analysis was performed on sodium heparin-anticoagulated peripheral blood samples. The combinations of surface marker-specific MAbs were conjugated to different fluorescent dyes used, including FITC, PE, and PE-cyanin 5.1 (PC5). Within 24 h after collection, peripheral blood samples from HIV+ and HIV− individuals were prepared for flow cytometric analysis with the Coulter (Miami, Fla.) Clone Immuno-Lyse system. Briefly, 100-μl aliquots of peripheral blood were incubated for 15 min in the dark with 10 μl of commercial antibodies to either: CD45-FITC–CD14-PE (Coulter) IgG1-FITC–IgG1-PE (Coulter), CD4-PC5 (Coulter), CD8-PC5 (Coulter), CD38-PE (Immunotech, Westbrook, Maine), and HLA-DR–PE (Pharmingen). Next, 25 μl of FITC-conjugated anti-RTF MAb was added to tubes containing antibody combinations of CD4-CD38, CD4–HLA-DR, CD8-CD38, and CD8–HLA-DR. After labeling, the samples were immediately analyzed with a Coulter Epics XL-MCL flow cytometer. Data from 5,000 gated (CD4+ or CD8+ T lymphocytes) events were acquired and analyzed with histogram and dot plot profiles of FITC and PE fluorescence. Lymphocytes were identified based on forward and side scatter parameters. Antibodies conjugated to CD45-FITC–CD14-PE were used to validate the established lymphocyte gate. CD4+ and CD8+ T cells were identified by PC5 fluorescence. A second gate was positioned around the CD4+ and CD8+ T cells, and CD38, HLA-DR, and RTF expression levels of these cells were evaluated. Results are expressed as either the mean channel fluorescence (MCF) of activation marker expression over the whole histogram or the percentage of CD4+ or CD8+ T cells that are RTF+ CD38+ or RTF+ HLA-DR+. Compensation for the three conjugates used in the study was determined as previously described (1, 7). To demonstrate differences in RTF expression between infected and noninfected individuals, flow cytometric markers for RTF expression were defined by using conjugated isotype control antibodies and samples obtained from both infected and uninfected individuals.
Measurement of plasma β2-microglobulin concentrations.
Sodium heparin-anticoagulated peripheral blood samples were collected and centrifuged at 1,000 × g for 10 min. Plasma was then collected and sent to SmithKline Beecham Clinical Laboratories (Schaumburg, Ill.) for determination of β2-microglobulin levels.
Statistical analysis.
The data were analyzed by using the StatView statistical software package (ABACUS Concepts, Berkeley, Calif.) and Sigma Plot (SPSS, Chicago, Ill.). Descriptive statistics are expressed as mean values plus or minus the standard error. Comparisons between HIV− and HIV+ individuals were performed by the unpaired t test or the Mann-Whitney U test (two sided). Significance was defined as a P of <0.05. Correlation analyses were performed by linear regression analysis or the Spearman rank correlation coefficient test.
RESULTS
Flow cytometric analysis of RTF expression on T-cell subsets.
We have previously demonstrated that CD3+ T cells from HIV+ individuals expressed significantly higher levels of RTF than T cells from HIV− individuals (7). Here we used flow cytometry to examine RTF expression on T-cell subsets from HIV+ and HIV− persons. As shown in Table 1, the average MCF of RTF expression on CD4+ and CD8+ T cells from HIV+ individuals was significantly higher than the average MCF of RTF expression in HIV− persons (P < 0.0001).
TABLE 1.
RTF expression is upregulated on T-cell subsets of HIV-infected individuals
| Variable | CD4+ T
cells
|
CD8+ T cells
|
||||
|---|---|---|---|---|---|---|
| HIV+ | HIV− | P valuea | HIV+ | HIV− | P valuea | |
| RTF MCF | 2.916 ± 0.439 | 0.871 ± 0.046 | <0.0001 | 2.408 ± 0.082 | 1.308 ± 0.080 | <0.0001 |
| % RTF+ | 26.01 ± 2.97 | 9.23 ± 1.04 | <0.0001 | 33.79 ± 3.17 | 16.77 ± 2.67 | 0.0007 |
| % RTF+ CD38+ | 17.90 ± 2.52 | 5.02 ± 1.10 | <0.0001 | 27.69 ± 2.93 | 11.06 ± 2.02 | 0.0001 |
| % RTF+ HLA-DR+ | 11.30 ± 3.11 | 2.22 ± 0.25 | 0.0001 | 16.97 ± 2.68 | 5.34 ± 1.04 | 0.0010 |
Significance is defined as a P value of <0.05 as determined by the Mann-Whitney U test.
The percentages of T-cell subsets expressing RTF were also determined by gating on either CD4+ or CD8+ T cells and by measuring anti-RTF-FITC positivity. As indicated in Table 1, HIV+ individuals had significantly greater percentages of CD4+ (P < 0.0001) and CD8+ (P = 0.0007) T lymphocytes expressing high levels of RTF. Therefore, within each T-cell compartment, RTF expression was significantly higher on cells from HIV+ individuals than on cells from HIV− individuals.
RTF expression on activated T-cell subsets.
Cell surface expression of RTF by T cells is induced following activation (3). Therefore, we determined whether RTF was coexpressed by CD4+ and CD8+ T cells bearing known markers of activation. Using three-color flow cytometric analysis, the percentages of CD4+ and CD8+ T cells that were double positive for RTF expression and either CD38 or HLA-DR expression were determined. Consistent with published reports, T-cell subsets (particularly CD8+) from HIV+ individuals expressed higher amounts of CD38 and HLA-DR than did T cells from HIV− individuals (Table 1). As also indicated in Table 1, significantly more CD4+ T cells from HIV+ individuals were RTF+ CD38+ (P < 0.0001) and RTF+ HLA− DR+ (P = 0.0001) as compared to CD4+ T cells from HIV− individuals. Similarly, as indicated in Table 1, significantly more CD8+ T cells from HIV+ individuals than CD8+ T cells from HIV− individuals were RTF+ CD38+ (P = 0.0001) and RTF+ HLA-DR+ (P = 0.0010).
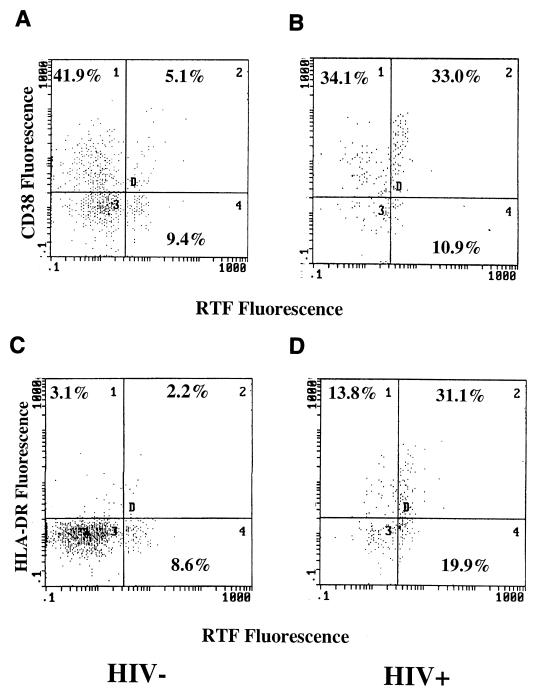
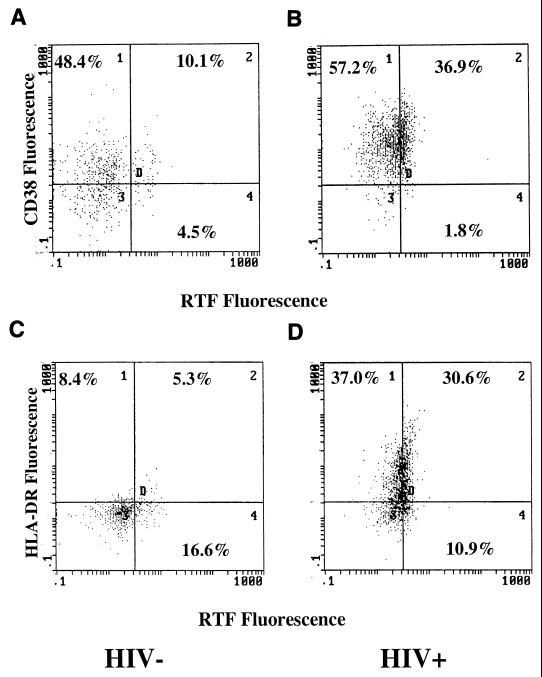
To illustrate the differences in coordinate expression of RTF with activation markers CD38 and HLA-DR, flow cytometric dot plots from HIV+ individuals with elevated percentages of CD4+ and CD8+ T cells expressing these markers were compared and contrasted to dot plots from HIV− individuals. As illustrated in Fig. 1 and 2, flow cytometric dot plots show increased coexpression of RTF on activated CD4+ and CD8+ T cell subsets from HIV+ individuals, respectively. Interestingly, although HIV+ individuals had higher percentages of T-cell subsets expressing RTF and either CD38 or HLA-DR, not all activated T cells expressed RTF.
FIG. 1.
Flow cytometric dot plots of CD4+ T cells from HIV− (A and C) and HIV+ (B and D) individuals showing dual expression of RTF and CD38 (A and B) or RTF and HLA-DR (C and D). A gate was drawn around the CD4+ cells, and the percentages of double-positive (RTF+ CD38+ or RTF+ HLA-DR+) cells were determined by FITC and PE fluorescence.
FIG. 2.
Flow cytometric dot plots of CD8+ T cells from HIV− (A and C) and HIV+ (B and D) individuals showing dual expression of RTF and CD38 (A and B) or RTF and HLA-DR (C and D). A gate was drawn around the CD8+ cells, and the percentages of double-positive (RTF+ CD38+ or RTF+ HLA-DR+) cells were determined by FITC and PE fluorescence.
As shown in Fig. 1 and 2, a large proportion of CD38+ and HLA-DR+ T cells did not express RTF, suggesting that RTF expression is unique to a specific subset of activated T cells in HIV+ individuals. In HIV+ individuals on average, 30.2% ± 22.1% of CD4+ CD38+ cells were RTF+, 61.8% ± 18.3% of CD4+ HLA-DR+ cells were RTF+, 35.7% ± 19.5% of CD8+ CD38+ cells were RTF+, and 49.1% ± 17.3% of CD8+ HLA-DR+ cells were RTF+ (data not shown). In comparison, among HIV− individuals on average, 6.94% ± 3.55% of CD4+ CD38+ cells were RTF+, 45.9% ± 12.5% of CD4+ HLA-DR+ cells were RTF+, 19.5% ± 13.9% of CD8+ CD38+ cells were RTF+, and 33.4% ± 16.8% of CD8+ HLA-DR+ cells were RTF+ (data not shown).
Relationship between T-cell subset expression of RTF and the percentage and absolute number of CD4+ cells.
The percentage and absolute number of CD4+ T cells were determined for each peripheral blood sample obtained from each HIV+ individual. Relationships between the percentage and absolute number of CD4+ T cells with expression of RTF and activation markers were determined by using the Spearman rank correlation coefficient (Spearman’s rho, rs). As indicated in Table 2, the MCF of RTF on CD4+ T cells, the percentage of CD4+ RTF+ cells, the percentage of CD4+ T cells that were RTF+ CD38+, and the percentage of CD4+ T cells that were RTF+ HLA-DR+ had significant and inverse correlations with both the percentage and absolute number of CD4+ T cells in HIV+ persons. These data suggest that increased RTF expression on CD4+ T cells is related to activation and depletion of CD4+ T cells in HIV+ individuals. The MCF of CD38 expression on CD8+ T cells did not correlate with either the percentage (rs = −0.205) or absolute number (rs = −0.361) of CD4+ T cells in HIV+ individuals. The MCF of HLA-DR expression on CD4+ T cells correlated closely with both the percentage (rs = −0.705; P = 0.0004) and absolute number (rs = −0.725; P = 0.0004) of CD4+ T cells in HIV+ individuals.
TABLE 2.
Relationship between CD4+ T cells and β2-microglobulin levels and expression of RTF and activation markers in HIV+ individuals
| Variable | % CD4+
cells
|
No. of CD4+
cells
|
β2-Microglobulin
|
|||
|---|---|---|---|---|---|---|
| rsa | P valueb | rsa | P valueb | rsa | P valueb | |
| MCF of RTF on CD4+ cells | −0.667 | 0.0011 | −0.694 | 0.0001 | +0.473 | 0.0204 |
| % RTF+ CD4+ cells | −0.694 | 0.0007 | −0.661 | 0.0012 | +0.469 | 0.0216 |
| % RTF+ CD38+ CD4+ cells | −0.530 | 0.0081 | −0.470 | 0.0212 | +0.402 | 0.0490 |
| % RTF+ HLA-DR+ CD4+ cells | −0.785 | <0.0001 | −0.739 | 0.0003 | +0.471 | 0.0211 |
| MCF of RTF on CD8+ cells | −0.364 | NS | −0.369 | NS | +0.288 | NS |
| % RTF+ CD8+ cells | −0.185 | NS | −0.205 | NS | +0.163 | NS |
| % RTF+ CD38+ CD8+ cells | −0.342 | NS | −0.300 | NS | +0.425 | 0.0335 |
| % RTF+ HLA-DR+ CD8+ cells | −0.464 | 0.0204 | −0.400 | NS | +0.180 | NS |
Spearman rank correlation coefficient. Absolute values greater than ±0.4 are in boldface type.
Significance is defined as a P value of <0.05. NS, not significant.
In contrast, within the CD8+ T-cell subset, only the percentage of RTF+ HLA-DR+ cells correlated with the percentage of CD4+ T cells in HIV+ individuals (Table 2). The MCF of CD38 expression on CD8+ T cells correlated slightly with the percentage (rs = −0.385; P = 0.0495) but not the absolute number of CD4+ T cells in HIV+ individuals. The MCF of HLA-DR expression on CD8+ T cells did not correlate with either the percentage or absolute number of CD4+ T cells in HIV+ individuals (data not shown). Also, RTF expression on CD8+ T cells from HIV+ persons did not correlate with either the percentage or absolute number of CD8+ T cells in these individuals (data not shown).
Relationship between RTF expression and plasma levels of β2-microglobulin.
To further determine the relationship between RTF expression and immune system activation during HIV infection, plasma levels of β2-microglobulin were measured for each HIV+ individual. Elevated plasma concentrations of β2-microglobulin had significant positive correlations with the increase in MCF of RTF on CD4+ cells and increasing percentages of CD4+ RTF+ cells, CD4+ T cells that were RTF+ CD38+, and CD4+ T cells that were RTF+ HLA-DR+ (Table 2). However, only the percentage of CD8+ T cells that were RTF+ CD38+ correlated in a positive manner with plasma β2-microglobulin levels in HIV+ individuals (Table 2). The MCF of CD38 on CD4+ T cells and the MCF of HLA-DR on CD4+ and CD8+ T cells did not correlate with plasma β2-microglobulin concentrations in HIV+ individuals (data not shown). The MCF of CD38 on CD8+ T cells (rs = +0.615; P = 0.0021) correlated closely with plasma β2-microglobulin levels. Collectively, these findings suggest that RTF expression is associated with the generalized immune system activation that occurs during HIV infection.
DISCUSSION
It has been demonstrated that increased RTF expression can be observed when the cellular immune response is suppressed (e.g., during pregnancy [6], HIV infection [7], and tumor progression [1]). Here we provide further evidence of significant upregulation of RTF expression during HIV infection associated with immune response activation. The MCF of RTF expression was significantly greater in HIV+ individuals than in HIV− individuals.
Persistent activation of T-cell subpopulations implies that cellular activation is a key component of HIV disease pathogenesis (12). Two markers of activation that are often studied are HLA-DR and CD38. HLA-DR is a major histocompatibility complex class II molecule that is expressed on activated T cells and is an important feature of HIV infection (19, 24). CD38 is expressed at specific developmental stages and is a marker of cellular immaturity and activation (9). Consistent with published reports (2, 10, 24, 25), we also found that CD4+ and CD8+ T cells from HIV+ individuals expressed higher levels of CD38 and HLA-DR. Increased activation of CD8 cells, mainly manifested by expression of CD38, strongly correlates with progression to AIDS and death (11–13, 18). We extended the findings of previous studies by showing that these activated T-cell subsets also express RTF during HIV infection.
It is a major finding that a significant percentage of both CD4+ and CD8+ T lymphocytes that express CD38 also coexpress RTF. Both CD4+ and CD8+ T cells from some HIV+ individuals exhibited a very pronounced increase in RTF+ CD38+ and RTF+ HLA-DR+ T cells. This observation suggests that RTF expression may be an additional marker for T-cell activation, particularly in HIV infection. Although no single cell surface activation marker is unique to HIV-associated immune activation, many markers are used to assess disease progression. Thus far, only CD38 expression on CD8+ T cells has been proven to have predictive value independent of CD4+-T-cell counts and viral load (11, 13, 18). The prominent dual expression of RTF with other activation markers suggests that RTF may also be a useful marker of immune status in HIV infection.
Interestingly, upregulation of RTF expression on CD4+ T lymphocytes during HIV infection could have dual implications. One possibility is that RTF could be an additional cell activation marker that is induced following activation by HIV antigens and/or opportunistic pathogens. On the other hand, since chronic immune activation results in a significant degree of lymphocyte apoptosis (8, 14), RTF may be involved in some way in loss of T-cell homeostasis and depletion of CD4+ T cells during HIV disease progression. Cell activation does not always lead to proliferation. Rather, programmed cell death or a physiologic suicide mechanism that preserves homeostasis may result (15). RTF may contribute to or at least mark enhanced sensitivity of T cells to apoptosis. If such is the case, RTF may serve as a tag to identify those CD4+ T cells that are being eliminated by activation-induced apoptosis during HIV infection. Resolution of these issues may be important to understanding how cell membrane-associated RTF functions in association with other cell surface molecules involved in T-cell activation and apoptosis.
Unlike RTF expression on CD4+ T lymphocytes, RTF expression on CD8+-T-cell subsets did not correlate with either the percentage or absolute numbers of CD4+ or CD8+ T cells with the exception of the percentage of CD8+ T cells that were positive for both RTF and HLA-DR. This finding may explain, in part, the lack of correlation between RTF expression on CD3+ T cells and the absolute numbers of CD4+ T cells in HIV+ individuals in our previous study (7). However, the MCF of RTF expression on CD8+ T cells correlated strongly with the MCF of CD38 (rs = 0.621) and HLA-DR (rs = 0.737) expression on CD8+ T cells (data not shown). These data imply that RTF expression on CD8+ T cells is linked to the activation of this subset of T cells during HIV infection.
In summary, we have shown that RTF expression on T lymphocytes, particularly CD4+ T cells, correlates with immune system activation in HIV infection. This study is part of an ongoing investigation to elucidate the role of RTF in T-lymphocyte activation in HIV infection. Further studies are needed to more precisely delineate the exact function(s) of RTF. The decrease in CD4+ T cells is strongly related to the increase in expression of RTF and activation markers, indicating a link between RTF and CD4+-T-cell activation and elimination. Future studies will focus on examining RTF expression on T-cell subsets from HIV+ individuals that are susceptible to or are undergoing apoptosis.
ACKNOWLEDGMENTS
We thank Catrina M. Crociani and Gail Hoppe at The Chicago Medical School, North Chicago, Ill., for their technical and clerical assistance, respectively. We also thank Sondra Allen at Mount Sinai Hospital for her assistance with accessing patient medical records.
REFERENCES
- 1.Aslakson C J, Lee G W, Gilman-Sachs A, Kucuk O, Beaman K D. Regeneration and tolerance factor is expressed on leukemias and may be a mechanism for leukemias to escape immune surveillance. Am J Hematol. 1999;61:46–52. doi: 10.1002/(sici)1096-8652(199905)61:1<46::aid-ajh9>3.0.co;2-g. [DOI] [PubMed] [Google Scholar]
- 2.Bass H Z, Fahey J L, Nishanian P, Detels R, Cumberland W, Kemeny M, Plaeger S. Relation of impaired lymphocyte proliferation function to other major human immunodeficiency virus type 1-induced immunological changes. Clin Diagn Lab Immunol. 1997;4:64–69. doi: 10.1128/cdli.4.1.64-69.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Beaman K D, Angkachatchai V, Gilman-Sachs A. TJ6: The pregnancy-associated cytokine. Am J Reprod Immunol. 1996;35:338–341. doi: 10.1111/j.1600-0897.1996.tb00490.x. [DOI] [PubMed] [Google Scholar]
- 4.Clerici M, Shearer G M. The Th1-Th2 hypothesis of HIV infection: new insights. Immunol Today. 1994;15:575–581. doi: 10.1016/0167-5699(94)90220-8. [DOI] [PubMed] [Google Scholar]
- 5.Clerici M, Shearer G M. A Th1-Th2 switch is a critical step in the etiology of HIV infection. Immunol Today. 1993;14:107–111. doi: 10.1016/0167-5699(93)90208-3. [DOI] [PubMed] [Google Scholar]
- 6.Coulam C B, Beaman K D. Reciprocal alteration in circulating TJ6+ CD19+ and TJ6+ CD56+ leukocytes in early pregnancy predicts success or miscarriage. Am J Reprod Immunol. 1995;34:219–224. doi: 10.1111/j.1600-0897.1995.tb00944.x. [DOI] [PubMed] [Google Scholar]
- 7.DuChateau B K, Lee G W, Westerman M P, Beaman K D. Increased expression of regeneration and tolerance factor in individuals with human immunodeficiency virus infection. Clin Diagn Lab Immunol. 1999;6:193–198. doi: 10.1128/cdli.6.2.193-198.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Estaquier J, Idziorek T, Zou W, Emilie D, Farber C M, Bourez J M, Ameisen J C. T helper type 1/T helper type 2 cytokines and T cell death: preventive effect of interleukin 12 on activation-induced and CD95 (FAS/APO-1)-mediated apoptosis of CD4+ T cells from human immunodeficiency virus-infected persons. J Exp Med. 1995;182:1759–1967. doi: 10.1084/jem.182.6.1759. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Ferrero E, Malavasi F. The metamorphosis of a molecule: from soluble enzyme to the leukocyte receptor CD38. J Leukoc Biol. 1999;65:151–161. doi: 10.1002/jlb.65.2.151. [DOI] [PubMed] [Google Scholar]
- 10.Ginaldi L, DeMartinis M, D’Ostilio A, DiGennaro A, Martini L, Quaglino D. Altered lymphocyte antigen expression in HIV infection. Am J Clin Pathol. 1997;108:585–592. doi: 10.1093/ajcp/108.5.585. [DOI] [PubMed] [Google Scholar]
- 11.Giorgi J V, Ho H N, Hirji K, Chou C C, Hultin L E, O’Rouke S, Park L, Margolick J B, Ferbas J, Phair J P. CD8+ lymphocyte activation at human immunodeficiency virus type 1 seroconversion: development of HLA− DR+ CD38− CD8+ cells is associated with subsequent stable CD4+ cell levels. The Multicenter AIDS Cohort Study Group. J Infect Dis. 1994;170:775–781. doi: 10.1093/infdis/170.4.775. [DOI] [PubMed] [Google Scholar]
- 12.Giorgi J V, Hultin L E, McKeating J A, Johnson T D, Owens B, Jacobson L P, Shih R, Lewis J, Wiley D J, Phair J P, Wolinsky S M, Detels R. Shorter survival in advanced human immunodeficiency virus type 1 infection is more closely associated with T lymphocyte activation than with plasma virus burden or virus chemokine coreceptor usage. J Infect Dis. 1999;179:859–870. doi: 10.1086/314660. [DOI] [PubMed] [Google Scholar]
- 13.Giorgi J V, Liu Z, Hultin L E, Cumberland W G, Hennessey K, Detels R. Elevated levels of CD38+ CD8+ T cells in HIV infection add to the prognostic value of low CD4+ T cell levels: results of 6 years of follow-up. The Los Angeles Center, Multicenter AIDS Cohort Study. J Acquir Immune Defic Syndr. 1993;6:904–912. [PubMed] [Google Scholar]
- 14.Gougeon M L, Lecoeur H, Dulioust A, Enouf M G, Crouvoiser M, Goujard C, Debord T, Montagnier L. Programmed cell death in peripheral lymphocytes from HIV-infected persons: increased susceptibility to apoptosis of CD4 and CD8 T cells correlates with lymphocyte activation and with disease progression. J Immunol. 1996;159:3509–3520. [PubMed] [Google Scholar]
- 15.Gougeon M L, Montagnier L. Apoptosis in AIDS. Science. 1993;260:1269–1270. doi: 10.1126/science.8098552. [DOI] [PubMed] [Google Scholar]
- 16.Knoefel B, Nuske K, Steiner T, Junker K, Kosmehl H, Rebstock K, Reinhold D, Junker U. Renal cell carcinomas produce IL-6, IL-10, IL-11, and TGF-beta 1 in primary cultures and modulate T lymphocyte blast transformation. J Interferon Cytokine Res. 1997;17:95–102. doi: 10.1089/jir.1997.17.95. [DOI] [PubMed] [Google Scholar]
- 17.Lin H, Mosmann T, Guilbert L, Tuntipopipat S, Wegmann T. Synthesis of T helper 2-type cytokines at the maternal-fetal interface. J Immunol. 1993;151:4562–4573. [PubMed] [Google Scholar]
- 18.Liu Z, Cumberland W G, Hultin L E, Kaplan A H, Detels R, Giorgi J V. CD8+ T-lymphocyte activation in HIV-1 disease reflects an aspect of pathogenesis distinct from viral burden and immunodeficiency. J Acquir Immune Defic Syndr Hum Retrovirol. 1998;18:332–340. doi: 10.1097/00042560-199808010-00004. [DOI] [PubMed] [Google Scholar]
- 19.Mahalingam M, Peackman M, Davies E T, Pozniak A, McManus T J, Vergani D. T cell activation and disease severity in HIV infection. Clin Exp Immunol. 1993;93:337–343. doi: 10.1111/j.1365-2249.1993.tb08182.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Mandal M, Beaman K D. Purification and characterization of a pregnancy-associated protein: TJ6s. Am J Reprod Immunol. 1995;33:60–67. doi: 10.1111/j.1600-0897.1995.tb01139.x. [DOI] [PubMed] [Google Scholar]
- 21.Manolson M F, Proteau D, Preston R A, Stenbit A, Roberts B T, Hoyt M A, Preuss D, Mulholland J, Botstein D, Jones E W. The VPHI gene encodes a 95-kDa integral membrane polypeptide required for in vivo assembly and activity of the yeast H+-ATPase. J Biol Chem. 1992;267:14294. [PubMed] [Google Scholar]
- 22.Merogi A J, Marrogi A J, Ramesh R, Robinson W R, Fermin C D, Freeman S M. Tumor-host interaction; analysis of cytokines, growth factors, and tumor-infiltrating lymphocytes in ovarian carcinomas. Hum Pathol. 1997;28:321–331. doi: 10.1016/s0046-8177(97)90131-3. [DOI] [PubMed] [Google Scholar]
- 23.Nichols T C, Kang J A, Angkachatchai V, Beer A E, Beaman K D. Expression of a membrane form of the pregnancy-associate protein TJ6 on lymphocytes. Cell Immunol. 1994;155:219–229. doi: 10.1006/cimm.1994.1114. [DOI] [PubMed] [Google Scholar]
- 24.Peakman M, Mahalingam M, Pozniak A, McManus T J, Phillips A N, Vergani D. Markers of immune cell activation and disease progression. Cell activation in HIV disease. Adv Exp Med Biol. 1995;374:17–26. doi: 10.1007/978-1-4615-1995-9_2. [DOI] [PubMed] [Google Scholar]
- 25.Plaeger S, Bass H Z, Nishanian P, Thomas J, Aziz N, Detels R, King J, Cumberland W, Kemeny M, Fahey J L. The prognostic significance in HIV infection of immune activation represented by cell surface antigen and plasma activation marker changes. Clin Immunol. 1999;90:238–246. doi: 10.1006/clim.1998.4646. [DOI] [PubMed] [Google Scholar]
- 26.Ribbing S L, Hoversland R C, Beaman K D. T-cell suppressor factors play an integral role in preventing fetal rejection. J Reprod Immunol. 1988;14:83–95. doi: 10.1016/0165-0378(88)90038-1. [DOI] [PubMed] [Google Scholar]
- 27.Rubesa G, Beaman K D, Lucin P, Beer A E, Rukavina D. Expression of TJ6 protein in the human first trimester decidual lymphocytes. Reg Immunol. 1994;6:331–333. [Google Scholar]
- 28.Utku N, Heinemann T, Tullius S G, Bulwin G-C, Beinke S, Blumberg R S, Beato F, Randall J, Kojima R, Busconi L, Robertson E S, Schulein R, Volk H-D, Milford E L, Guilans S R. Prevention of acute allograft rejection by antibody targeting TIRC7, a novel T cell membrane protein. Immunity. 1998;9:509. doi: 10.1016/s1074-7613(00)80634-2. [DOI] [PubMed] [Google Scholar]
- 29.Wegmann T G, Lin H, Guilbert L, Mosmann T R. Bidirectional cytokine interactions in the maternal-fetal relationship: is successful pregnancy a TH2 phenomenon? Immunol Today. 1993;14:353–356. doi: 10.1016/0167-5699(93)90235-D. [DOI] [PubMed] [Google Scholar]