Abstract
Introduction:
Research indicates that prophylactic mesh may help prevent incisional hernia after laparotomy, but best practice patterns in these situations are still evolving. Here, we compare the failure loads (FLs) and biomechanical stiffness (BMS) of 35 porcine abdominal wall laparotomy incisions reinforced with meshes of various widths and fixation distances using biomechanical testing.
Methods:
In each specimen, a 10-cm incision was made and closed using continuous 1-0 Maxon suture. Specimens were randomized to mesh width (none, 2.5 cm, 3 cm, 4 cm,6 cm, 8 cm) and tack separation (1.5 cm, 2 cm apart) and the meshes secured in an onlay fashion. Cyclic loads oscillating from 15 N to 140 N were applied to simulate abdominal wall stress, and the specimens subsequently loaded to failure. FLs (N) and BMS (N/mm) were comparatively analyzed.
Results:
All specimens failed via suture pull-through. FLs and BMS were lowest in specimens with suture-only (421.43 N; 11.69 N/mm). FLs and BMS were significantly higher in 4-cm mesh specimens (567.51 N) than those with suture, 2.5-cm, and 3.0-cm mesh (all P < 0.05). FLs in specimens with a greater number of tacks were consistently higher in meshes of similar sizes, although these did not reach significance.
Conclusions:
A 4-cm mesh reenforcement was superior to suture-only and smaller meshes at preserving strength in laparotomy closure in a porcine model but larger meshes (6 cm,8 cm) did not provide an additional benefit. Meshes with more fixation points may be advantageous, but additional data are needed to make definitive conclusions.
Keywords: Hernia, Laparotomy, Mesh width, Prevention, Prophylactic mesh placement
Introduction
The use of mesh in incisional hernia (IH) repair has been shown to reduce hernia recurrence by providing laminar reinforcement of the musculo-fascial envelope, and mitigating tension across fascial closure, particularly in the early stages of healing.1-4 Recent literature has therefore supported that prophylactic mesh placement (PMP) for high-risk patients during an index abdominal procedure may significantly reduce the development of postoperative IH by shielding the fascia from excessive tension and helping to prevent fascial separation.5,6 Postoperative patient activity for any midline laparotomy incision has been found to generate on average 50 N of force across a repair, exceeding the biomechanical properties of traditional suture closure.7 PMP of the initial suture closure reinforces the anterior fascia and has been found to increase biomechanical strength from 44 N to 57 N.8 With this knowledge, there has been a recent emphasis on clinically investigating the impact of prophylactically reinforcing laparotomy incisions with mesh, particularly in high risk patients. In patients presenting for emergency midline laparotomies,9,10 elective or emergent colorectal procedures,11 open bariatric surgery in the setting of morbid obesity,12 and abdominal aortic aneurysm (AAA) repair,13 PMP has resulted in lower rates of IH than patients repaired with suture-only, with similar complications rates. Results of the PRImary Mesh Closure of Abdominal Midline Wound trial, a multicenter double-blind randomized control trial, found that after randomizing 480 high-risk (body mass index >27 or open AAA) patients to primary suture repair, onlay, or retrorectus mesh placement, rates of IH at 2 y were 30% if repaired with suture alone, 18% if repaired with retrorectus mesh (P = 0.05), and 13% if repaired with onlay mesh (P = 0.0016).14 However, PMP adds time and technical considerations to surgical procedures, which has limited its wide-spread adoption.6,15
The Paradigm Surgical team is currently developing a prophylactic mesh implantation device, the SafeClose Roller System, which automatically fixates mesh onto the anterior abdominal fascia to re-enforce closure.16 Early results with this device have corroborated that mesh reinforcement significantly impacts the biomechanical strength of closure and have further suggested that using a mesh-application system can improve efficiency and consistency of application.16 In designing this device, however, it became evident to the authors that little data exist in the current literature that detail the appropriate mesh size, overlap, and fixation patterns to use in these scenarios.
In this study, we aimed to compare the failure loads (FLs) and biomechanical stiffness (BMS) of laparotomy incisions reinforced with suture and/or onlay meshes of various widths and fixation distances. To accomplish this, we biomechanically tested 35 porcine abdominal walls with midline incisions using a ball-burst apparatus, the Instron electromechanical testing machine. The ball-burst method has been previously used to evaluate mesh characteristics and to test the strength of laparotomy closure by simulating the cyclic and multiaxial forces experienced by the abdominal wall in the immediate postoperative period.17 We sought to determine how altering these factors during stress testing may subsequently affect the FLs and stiffness of a laparotomy incision. Characterizing the effects of varying mesh width and fixation patterns will be essential in advancing our understanding of the ideal relationship between overlap and fascial closure in PMP and in developing optimal mesh-application systems.
Material and Methods
Objectives and porcine tissue preparation
Our primary objectives were to analyze the FL (N) of each specimen (the amount of force it takes for a closure to fail) and the BMS (N/mm) (a measure of how much load a tissue can take before it deforms) and comparatively analyze these among specimens with varying mesh widths and fixation distances. FL was defined as the peak of the load versus extension curve of each specimen during FL testing, and BMS was defined as the slope of the linear portion of these curves.
Previous literature has demonstrated that cadaveric porcine tissue has similar biologic and mechanical characteristics to human tissue18 and therefore porcine abdominal wall specimens were obtained from Animal Biotech Industries (Doylestown, Pennsylvania, https://www.animalbiotech.com/, Order number: 21,912) for use in this study. The specimens were thawed on the morning of testing, and dissected to remove skin and excess adipose tissue, leaving only the rectus muscle, anterior rectus sheath, and posterior rectus sheath. Thirty five specimens were cut to an equal size (22 cm × 18 cm); 10 cm incisions were made using a scalpel in the center of each specimen. Subsequently, one individual (A.N.C.) sutured the incisions closed in a standard fashion using a continuous running 1-0 Maxon suture. Sutures were placed at approximately 1 cm intervals with 1 cm bites of fascia on either side of the midline (Fig. 1). Strips of Bard Phasix mesh were carefully cut to size (Widths: 2.5 cm, 3.0 cm, 4.0 cm, 6.0 cm, and 8.0 cm), with the lengths remaining constant at 12.5 cm. A Phasix mesh was used due to the senior surgeon’s practice patterns, which are supported by existing literature that the biosynthetic properties of Phasix are particularly suited for onlay and complex hernia repair.19,20
Fig. 1 –
Specimen following closure with running 1-0 Maxon suture.
To account for the natural variation in the porcine specimens and the possibility of slight variations in closure techniques, the Google play random number generator was used to assign numbers to each specimen, which dictated tacker distance and mesh size. In addition, the remaining suture length after closure and porcine abdominal wall thickness were comparatively analyzed to assess the degree of variation among closures and specimens. Following randomization, the Bard OptiFix tacker was used to fixate the meshes onto the individual specimens in an onlay fashion using resorbable tacks. The meshes were first tacked in all four corners and then along the long, lateral edge of the mesh strip on either side. Mesh was placed taut, without wrinkling or bunching. Half of the meshes were fixated using a mesh tacked at a distance of 2 cm (seven tacks per side) and the other half were fixated using a mesh tacked at a distance of 1.5 cm (nine tacks per side) (Fig. 2). Two specimens were used for baseline testing (no mesh), three specimens acted as our control specimens (no mesh), and the remaining 30 specimens had mesh placed (Fig. 3).
Fig. 2 –
Specimen following mesh fixation with the Bard OptiFix tacker.
Fig. 3 –
Schematic breakdown of randomization of specimens to suture only, mesh width, and tacker distance.
Ball-burst testing
Biomechanical strength testing was conducted at the Penn Center for Musculoskeletal Disorders’ biomechanics core laboratory using a ball-burst testing apparatus fitted onto the Instron ElectroPuls E3000. Traditionally, a spherical head is attached to the piston. However, as the curvature of the abdominal wall is thought to be about 200 mm,21,22 our group designed and 3-D printed a rectangular, rounded plunger that mimicked this curvature and a tissue clamping fixture to accommodate the specimen and plunger. Custom parts were designed on PTC Creo Parametric Computer Aided Design software and manufactured at Protolabs. Four heavy-duty C-clamps purchased from McMaster Carr were used to secure the tissue specimens (Fig. 4).
Fig. 4 –
Representation of a specimen loaded and secured into the Instron prior to testing.
Baseline testing
Two specimens without mesh were placed in the apparatus and statically loaded until they reached failure. Failure was defined as suture pull-through, where a suture completely separates from the tissue, consistent with previous literature.17 The baseline FL of these two specimens were averaged (Fig. 5).
Fig. 5 –
Failure load of a baseline specimen, determined by peak of the extension versus load graph.
Cyclic load testing
The cyclic load testing protocol was adapted from a study by Sahoo et al. that tested mesh properties23 and has subsequently been used in studies evaluating the strength of laparotomy incisions.17 The purpose of cyclic loading was to represent everyday postoperative stress to the abdominal wall. Using 35% of the baseline average FL calculated in Step 1, we established the maximum cyclic load. The oscillating forces in the cyclic load testing ranged from 15 N to the maximum cyclic load; 15 N was used instead of 0 N to more realistically represent the human intraabdominal pressures, which are never 0 N. The oscillating load was applied for 500 cycles at a rate of 1 Hz.
Failure load testing
Following cyclic loading, the mesh and tacks were removed from each specimen and the specimens were statically loaded to failure. Mesh removal before FL testing was done for two reasons. First, it facilitated our ability to visually analyze for any microfailures at the incision, such as gapping or suture pull-through. Second, it isolated the protective and reinforcing effect that the mesh had during the fatiguing phase rather than testing the fixation strength of the mesh itself. In a real-world mesh application, mesh ingrowth occurs over several months. However, since this is not replicable in the short term, removing the mesh before failure loading is a more accurate way to assess how well the presence of mesh has protected the tissue during the fatiguing phase rather than testing its fixation strength.
Statistical analysis
Descriptive statistics were used to describe report means and standard deviations of suture length and porcine tissue size and to calculate average FLs and BMS. A linear regression of the load versus extension graph was used to calculate the stiffness (slope) of each specimen. Comparative data of FL and stiffness by mesh width and tack distance were tested for normal distribution using Box and Whisker plots, QQ Plots, and by calculating skewness. These tests indicated that the data were not sampled from a normally distributed population; this is likely due to small sample sizes. To accurately evaluate statistical significance of the data and calculate the P values, the nonparametric Mann–Whitney U-test was used.
Results
Controlling for study variation
Of the 35 sutures used in this study, the mean and standard deviation of the remaining suture after each closure was 24.94 cm ± 4.1 cm. In addition, the thickness of each porcine abdominal wall specimen was measured using dial calipers and the measurements were taken at the center of the specimen. The average thickness of each specimen was 1.36 cm ± 0.36 cm.
Failure load analysis
Load testing revealed that in all cases, failure occurred at the suture-line via suture pull-through. The maximum FLs recorded from the peak of each specimen’s Load versus Extension graph are found in Figures 6-8. Of all the specimens, the average FL was lowest in specimens with no mesh reinforcement (421.43 N). In both seven-tack and nine-tack fixated mesh groups, the FL was highest for specimens reinforced with a 4 cm wide mesh (557.03 N and 577.98, respectively). When analyzed independently based on tack fixation, comparison of FLs only reached significance in the nine-tack fixated groups when comparing 4-cm mesh with no mesh (P = 0.025) and 2-cm mesh (P = 0.042) (Fig. 7). Furthermore, when sample size was increased and all mesh specimens regardless of a number of fixation points were analyzed together, the average FL of specimens re-enforced with 4 cm mesh was significantly higher than no mesh, 2.5 cm mesh, and 3.0 cm mesh groups (P = 0.035, P = 0.034, and P = 0.039, respectively) (Fig. 8). While the failure loads for 4 cm mesh were higher than both 6 cm and 8 cm mesh specimens, the differences did not reach statistical significance. A comparative analysis of the average FLs by mesh width and fixation distance revealed that in all cases the average load was not significantly affected by tack distance; however, the specimens reinforced with mesh that had nine tacks per side (524.10 N) were higher than those specimens with only seven tacks per side (480.72 N). The percent improvement in failure loading ranged from 3.76% to 15.3% when comparing the seven-tack and nine-tack mesh, with an average improvement of 9.15%.
Fig. 6 –
Average failure loads in specimens secured with seven tacks per side.
Fig. 8 –
Average failure loads in all specimens.
Fig. 7 –
Average failure loads in specimens secured with nine tacks per side.
Biomechanical stiffness analysis
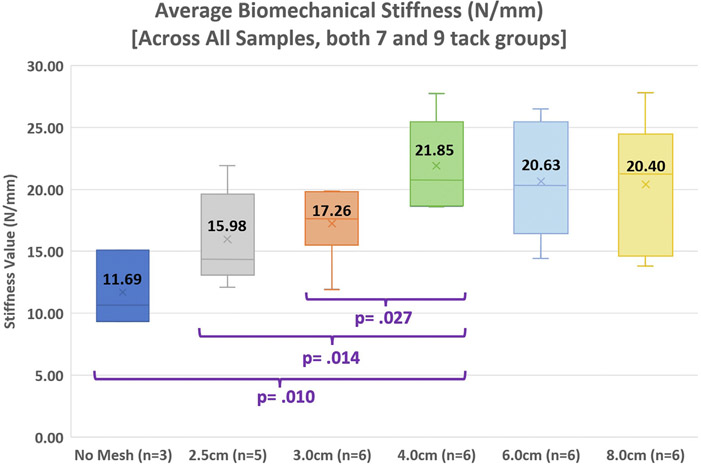
When analyzed regardless of tack distance, BMS in the entire cohort was highest in specimens re-enforced with 4 cm meshes (21.85 N/mm) and significantly higher than specimens with no mesh (P = 0.010), 2.5 cm mesh (P = 0.014), and 3.0 cm mesh (P = 0.027). While the stiffness was higher in 4 cm meshes than in 6 cm or 8 cm meshes, these did not reach statistical significance (P > 0.05) (Fig. 9). Results from the seven-tack group were similar to those from the entire cohort, with 4 cm mesh displaying significantly a higher stiffness than both no mesh (P = 0.025) and 2.5 cm (P = 0.025) groups (Fig. 10). However, when the nine-tack group was independently analyzed, stiffness was linear, with no mesh having the lowest (11.69 N/mm) and 8 cm mesh having the highest (22.05 N/mm) values (Fig. 11). Similar to the FL analysis, the average stiffness in specimens reinforced with nine tacks (19.49 N/mm) was higher than those reinforced with just seven tacks (19.02 N/mm), with an average percent increase of 3.71%.
Fig. 9 –
Average biomechanical stiffness in all specimens.
Fig. 10 –
Average biomechanical stiffness in specimens secured with seven tacks per side.
Fig. 11 –
Average biomechanical stiffness in specimens secured with nine tacks per side.
Discussion
IH continues to affect as many as one in five patients after midline laparotomy.24-26 In this study, we used biomechanical testing to apply cyclic forces of varying intensity to suture and mesh reinforced porcine abdominal wall incisions to simulate the immediate postoperative period. The results illustrated here suggest that prophylactic onlay mesh reinforcement improves the strength/BMS of a closure when compared to suture repair alone and that a 4-cm mesh (2 cm overlap) may provide immediate benefits when compared to smaller and larger mesh sizes and help prevent IH. In an era of preventative and cost-effective medicine, this study has important implications in advancing our understanding of best practice patterns for PMP and the design of future mesh-application devices.
In this porcine study, we simulate the placement of a biosynthetic mesh in the onlay plane. While systematic reviews of the PMP literature support that mesh in both the onlay and retrorectus planes improve IH rates when compared to suture repair alone,5,27 the previously mentioned results of the PRImary Mesh Closure of Abdominal Midline Wound trial demonstrated that mesh placed in the onlay plane was most preventative of IH development.14 Specifically, rates of recurrence for onlay mesh reinforcement and retrorectus mesh reinforcement were 13% and 18%, respectively.14 In some instances, like emergency abdominal surgery, the use of retrorectus repair techniques has actually been associated with an increased risk of postoperative complications, and the onlay plane is recommended in these scenarios.28 In addition, it is technically easier to develop the onlay plane and fixate a mesh to the anterior abdominal wall than it is to dissect the retrorectus plane. This leads to less surgical time and operating room expense and makes adoption of PMP by both plastic and general surgeons more feasible. While the onlay plane has been critiqued for higher incidences of postoperative wound events due to a more extensive soft tissue dissection,14,29,30 incidences may be reduced with smaller mesh sizes in PMP patients which permit less undermining. In terms of mesh choice, a cost utility analysis has shown that PMP is more cost-effective than suture repair alone when mesh costs up to $3700.31 While synthetic mesh is relatively inexpensive, concerns regarding chronic pain, foreign body sensation, and long-term mesh complications make surgeons less inclined to prophylactically place a permanent material. On the other hand, biologic products are significantly more expensive at $4000-$6000 per sheet, making them less cost-effective for PMP.31 Biosynthetic products, which are slowly resorbable and minimize the prosthetic footprint, yet are less costly than biologics, may therefore be ideal for these scenarios.
While the current literature surrounding PMP has been promising, data regarding important technical considerations like appropriate mesh overlap or fixation mechanisms in these unique situations are scant. In a study comparing 200 hernia repairs, the recurrence rate decreased from 9% to 4% when mesh overlap was increased from 3 cm to 5 cm.32 Similarly, in a systematic review and meta-analysis of recurrence rates in patients undergoing hernia repair, hernia recurrence dropped with an increase in mesh overlap from <3 cm to >5 cm in defects of all sizes when repaired laparoscopically; however, altering mesh size in open repairs did not appear to affect recurrence rates.33 In patients with existing abdominal wall defects, a larger mesh with this degree of overlap may better displace the forces applied to a closure that is under a greater amount of tension. In PMP patients, however, the same principles may not apply, and using data from hernia repair patients to guide mesh size for PMP may result in the unnecessary utilization of larger meshes without an increased benefit. When we consider that larger meshes are more expensive and would require greater undermining of skin flaps in the onlay position, we understand why evaluating PMP separately from hernia patients is important to mitigate patient risk and hospital cost. In this study, the 4-cm mesh (2-cm overlap) width group consistently performed better than the other mesh widths, with higher maximum FLs and BMS. Wider meshes have more room to stretch which means they take a long time to reach their maximum load capacity. As the load increases, the mesh is increasingly stretched and therefore the incision is subjected to a portion of that load for a longer period of time. On the contrary, a mesh that is very narrow has very little room to stretch, meaning the maximum load is reached quickly, which in turn increases the load on the incision, thereby stressing and fatiguing it over time. A mesh 4 cm in width seems to represent the “sweet spot,” balancing the relative drawbacks of an excessively wide or narrow mesh.
In addition to proper overlap, adequate mesh fixation plays an important role in the success of mesh placement. Mesh fixation ensures that the mesh is placed under enough tension to avoid ripples or folding. A flat mesh lays in better contact with the tissue underneath, thereby facilitating integration of the mesh over time.34 In addition, a well fixated mesh distributes the tensile forces across the mesh itself, offloading the tension from the fascial closure and leading to more favorable biodynamics in the early stages of healing.35 While in this study we did not see a significant difference in those specimens fixated with seven tacks per side versus nine tacks per side, we did observe that the meshes with more fixation points had higher FLs and BMS than those with just seven tacks. Studies with larger sample sizes and more substantial differences in tack distances are necessary to determine how significantly varying fixation patterns would impact clinical practice. It should also be noted that recent literature has postulated that chemical fixation, like the use of fibrin glue, may decrease operative time, technical difficulty, postoperative pain, and length of stay when compared to suture or tack fixation techniques.36-39 Therefore, despite the addedcost of these chemical sealants, there may be decreased overall hospital utilization when compared to repairs using suture or tacks, making these products more cost-effective.37 Nevertheless, a robust comparative analysis of fixation techniques in the open hernia surgery literature is limited. The authors recommend that regardless of fixation technique, surgeons should take care to meticulously and systematically secure the mesh to the abdominal wall so it lays flat and taut without wrinkling, folding, or bunching.
A major limitation to this work is that immediate mesh fixation followed by load testing using cadaveric porcine specimens does not represent the real-world practice of mesh placement in humans. In patients, the success of mesh to augment a laparotomy incision is not attributed to size or fixation alone but is multifactorial and dependent on incorporation and neovascularization at the mesh/tissue interface, degree of mesh shrinkage and migration, and many patient-level risk factors. Using porcine specimens prevents us from examining all of these contributing factors. Next, to examine the closure for failure and test the ability of the mesh to preserve tissue strength after loading, the mesh was removed during FL testing. Again, this is not a true depiction of testing a real-world repair, in which mesh ingrowth occurs over several months. However, to isolate the protective effect of the mesh itself during cyclic loading, rather than fixation strength, removal was necessary. In addition, knowing that most of the tension in a midline closure is due to lateral distracting forces, mesh overlap was only varied in the horizontal and not the vertical direction. Certainly though, vertical overlap should be a consideration in mesh failure, and not testing for this variation is a limitation of this work. Finally, depending on the mesh product used, inflammatory reactions cause various degrees of mesh shrinkage over time, ranging anywhere from 5% to 50%.40,41 Certainly, the degree of mesh shrinkage may play a role in dictating an ideal overlap and surgeons should be aware of these mesh specific properties.
In conclusion, prophylactic onlay biosynthetic mesh repair over suture closure alone helped preserve strength in the fascial closure of porcine abdominal wall specimens and may help prevent the development of fascial dehiscence and herniation after abdominal surgery. Mesh with 2 cm of overlap may be sufficient to adequately distribute force over these closures, while remaining cost-effective and minimizing the amount of foreign material placed for prophylactic purposes. Future studies that examine the clinical effects of using smaller prophylactic meshes in patients after abdominal surgery to prevent IH are needed to confirm these findings.
Funding
This study was funded by the National Institutes of Health as part of a Phase II Small Business Innovation in Research Grant to Paradigm Surgical for the development of the SafeClose Roller System.
Footnotes
Disclosure
Dr John P. Fischer reports having received consulting fees from Becton Dickinson, Integra, Gore and Baxter. The remaining authors have no conflicts to disclose.
Availability of Data and Code
Data and code available upon request.
Ethical Approval/Patient Consent
Not required for this cadaveric porcine study.
REFERENCES
- 1.Nguyen MT, Berger RL, Hicks SC, et al. Comparison of outcomes of synthetic mesh vs suture repair of elective primary ventral herniorrhaphy : a systematic review and meta-analysis. JAMA Surg. 2014;149:415–421. [DOI] [PubMed] [Google Scholar]
- 2.Mathes T, Walgenbach M, Siegel R. Suture versus mesh repair in primary and incisional ventral hernias: a systematic review and meta-analysis. World J Surg. 2016;40:826–835. [DOI] [PubMed] [Google Scholar]
- 3.Manuel LC, Lidia AMD, Pereira JA, et al. Balancing mesh-related complications and benefits in primary ventral and incisional hernia surgery. A meta-analysis and trial sequential analysis. PLoS One. 2018;13:e0197813. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Luijendijk RW, Hop WCJ, Van Den Tol MP, et al. A comparison of suture repair with mesh repair for incisional hernia. N Engl J Med. 2000;343:392–398. [DOI] [PubMed] [Google Scholar]
- 5.Borab ZM, Shakir S, Lanni MA, et al. Does prophylactic mesh placement in elective, midline laparotomy reduce the incidence of incisional hernia? A systematic review and meta-analysis. Surg (United States). 2017;161:1149–1163. [DOI] [PubMed] [Google Scholar]
- 6.Depuydt M, Allaeys M, Abreu de Carvalho L, et al. Prophylactic mesh after midline laparotomy: evidence is out there, but why do surgeons hesitate? World J Surg. 2021;45:1349–1361. [DOI] [PubMed] [Google Scholar]
- 7.Konerding MA, Bohn M, Wolloscheck T, et al. Maximum forces acting on the abdominal wall: experimental validation of a theoretical modeling in a human cadaver study. Med Eng Phys. 2011;33:789–792. [DOI] [PubMed] [Google Scholar]
- 8.Bellón JM, López-Hervás P, Rodríguez M, et al. Midline abdominal wall closure: a new prophylactic mesh concept. J Am Coll Surg. 2006;203:490–497. [DOI] [PubMed] [Google Scholar]
- 9.Argudo N, Pereira JA, Sancho JJ, et al. Prophylactic synthetic mesh can be safely used to close emergency laparotomies, even in peritonitis. Surg (United States). 2014;156:1238–1244. [DOI] [PubMed] [Google Scholar]
- 10.Kurmann A, Barnetta C, Candinas D, Beldi G. Implantation of prophylactic nonabsorbable intraperitoneal mesh in patients with peritonitis is safe and feasible. World J Surg. 2013;37:1657–1660. [DOI] [PubMed] [Google Scholar]
- 11.Garcia-Urena MA. Preventing incisional ventral hernias: important for patients but ignored by surgical specialities? A critical review. Hernia. 2021;25:13–22. [DOI] [PubMed] [Google Scholar]
- 12.Strzelczyk JM, Szymański D, Nowicki ME, et al. Randomized clinical trial of postoperative hernia prophylaxis in open bariatric surgery. Br J Surg. 2006;93:1347–1350. [DOI] [PubMed] [Google Scholar]
- 13.Bevis PM, Windhaber RAJ, Lear PA, et al. Randomized clinical trial of mesh versus sutured wound closure after open abdominal aortic aneurysm surgery. Br J Surg. 2010;97:1497–1502. [DOI] [PubMed] [Google Scholar]
- 14.Jairam AP, Timmermans L, Eker HH, et al. Prevention of incisional hernia with prophylactic onlay and sublay mesh reinforcement versus primary suture only in midline laparotomies (PRIMA): 2-year follow-up of a multicentre, double-blind, randomised controlled trial. Lancet. 2017;390:567–576. [DOI] [PubMed] [Google Scholar]
- 15.Fischer JP, Harris HW, López-Cano M, Hope WW. Hernia prevention: practice patterns and surgeons’ attitudes about abdominal wall closure and the use of prophylactic mesh. Hernia. 2019;23:329–334. [DOI] [PubMed] [Google Scholar]
- 16.Messa CA, Sanchez J, Kozak GM, et al. Biomechanical parameters of mesh reinforcement and analysis of a novel device for incisional hernia prevention. J Surg Res. 2021;258:153–161. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Scheiber CJ, Kurapaty SS, Goldman SM, et al. Suturable mesh better resists early laparotomy failure in a cyclic ball-burst model. Hernia. 2020;24:559–565. [DOI] [PubMed] [Google Scholar]
- 18.Cooney GM, Lake SP, Thompson DM, et al. The suture pullout characteristics of human and porcine linea alba. J Mech Behav Biomed Mater. 2017;68:103–114. [DOI] [PubMed] [Google Scholar]
- 19.Christopher AN, Morris MP, Patel V, et al. An evaluation of clinical and quality of life outcomes after ventral hernia repair with poly-4-hydroxybutyrate mesh. Hernia. 2021;25:717–726. [DOI] [PubMed] [Google Scholar]
- 20.Christopher AN, Patel V, Othman S, et al. Onlay poly-4-hydroxybutyrate (P4HB) mesh for complex hernia: early clinical and patient reported outcomes. J Surg Res. 2021;264:199–207. [DOI] [PubMed] [Google Scholar]
- 21.Lyons M, Mohan H, Winter DC, Simms CK. Biomechanical abdominal wall model applied to hernia repair. Br J Surg. 2015;102:e133–e139. [DOI] [PubMed] [Google Scholar]
- 22.Song C, Alijani A, Frank T, et al. Mechanical properties of the human abdominal wall measured in vivo during insufflation for laparoscopic surgery. Surg Endosc Other Interv Tech. 2006;20:987–990. [DOI] [PubMed] [Google Scholar]
- 23.Sahoo S, DeLozier KR, Erdemir A, Derwin KA. Clinically relevant mechanical testing of hernia graft constructs. J Mech Behav Biomed Mater. 2015;41:177–188. [DOI] [PubMed] [Google Scholar]
- 24.Fink C, Baumann P, Wente MN, et al. Incisional hernia rate 3 years after midline laparotomy. Br J Surg. 2014;101:51–54. [DOI] [PubMed] [Google Scholar]
- 25.Mudge M, Hughes LE. Incisional hernia: a 10 year prospective study of incidence and attitudes. Br J Surg. 1985;72:70–71. [DOI] [PubMed] [Google Scholar]
- 26.Nieuwenhuizen J, Eker HH, Timmermans L, et al. A double blind randomized controlled trial comparing primary suture closure with mesh augmented closure to reduce incisional hernia incidence. BMC Surg. 2013;13:48. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Tansawet A, Numthavaj P, Techapongsatorn S, et al. Mesh position for hernia prophylaxis after midline laparotomy: a systematic review and network meta-analysis of randomized clinical trials. Int J Surg. 2020;83:144–151. [DOI] [PubMed] [Google Scholar]
- 28.Juul N, Henriksen NA, Jensen KK. Increased risk of postoperative complications with retromuscular mesh placement in emergency incisional hernia repair: a nationwide register-based cohort study. Scand J Surg. 2021;110:193–198. [DOI] [PubMed] [Google Scholar]
- 29.Berger RL, Li LT, Hicks SC, et al. Development and validation of a risk-stratification score for surgical site occurrence and surgical site infection after open ventral hernia repair. J Am Coll Surg. 2013;217:974–982. [DOI] [PubMed] [Google Scholar]
- 30.Binnebösel M, Klink CD, Otto J, et al. Impact of mesh positioning on foreign body reaction and collagenous ingrowth in a rabbit model of open incisional hernia repair. Hernia. 2010;14:71–77. [DOI] [PubMed] [Google Scholar]
- 31.Fischer JP, Basta MN, Wink JD, et al. Cost-utility analysis of the use of prophylactic mesh augmentation compared with primary fascial suture repair in patients at high risk for incisional hernia. In: Surgery. United States: Mosby Inc.; 2015:700–711. [DOI] [PubMed] [Google Scholar]
- 32.LeBlanc KA, Whitaker JM, Bellanger DE, Rhynes VK. Laparoscopic incisional and ventral hernioplasty: lessons learned from 200 patients. Hernia. 2003;7:118–124. [DOI] [PubMed] [Google Scholar]
- 33.LeBlanc K Proper mesh overlap is a key determinant in hernia recurrence following laparoscopic ventral and incisional hernia repair. Hernia. 2016;20:85–99. [DOI] [PubMed] [Google Scholar]
- 34.Khansa I, Janis JE. The 4 principles of complex abdominal wall reconstruction. Plast Reconstr Surg - Glob Open. 2019;7:e2549. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Rangwani S, Kraft CT, Schneeberger SJ, et al. Strategies for mesh fixation in abdominal wall reconstruction: concepts and techniques. Plast Reconstr Surg. 2020;147:484–491. [DOI] [PubMed] [Google Scholar]
- 36.Shahan CP, Stoikes NF, Webb DL, Voeller GR. Sutureless onlay hernia repair: a review of 97 patients. Surg Endosc. 2016;30:3256–3261. [DOI] [PubMed] [Google Scholar]
- 37.Rhemtulla IA, Tecce MG, Broach RB, et al. Retromuscular mesh repair using fibrin glue: early outcomes and cost-effectiveness of an evolving technique. Plast Reconstr Surg Glob Open. 2019;7:e2184. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Weltz AS, Sibia US, Zahiri HR, et al. Operative outcomes after open abdominal wall reconstruction with retromuscular mesh fixation using fibrin glue versus transfascial sutures. Am Surg. 2017;83:937–942. [PubMed] [Google Scholar]
- 39.Stoikes N, Webb D, Powell B, Voeller G. Preliminary report of a sutureless onlay technique for incisional hernia repair using fibrin glue alone for mesh fixation. Am Surg. 2013;79:1177–1180. [PubMed] [Google Scholar]
- 40.Scheidbach H, Tamme C, Tannapfel A, et al. In vivo studies comparing the biocompatibility of various polypropylene meshes and their handling properties during endoscopic total extraperitoneal (TEP) patchplasty: an experimental study in pigs. Surg Endosc. 2004;18:211–220. [DOI] [PubMed] [Google Scholar]
- 41.de Tayrac R, Deffieux X, Gervaise A, et al. Long-term anatomical and functional assessment of trans-vaginal cystocele repair using a tension-free polypropylene mesh. Int Urogynecol J Pelvic Floor Dysfunct. 2006;17:483–488. [DOI] [PubMed] [Google Scholar]