Abstract
Neutrophils are the most abundant leukocyte population in the human circulatory system and are rapidly recruited to sites of inflammation. Neutrophils play a multifaceted role in intestinal inflammation, as they contribute to the elimination of invading pathogens. Recently, their role in epithelial restitution has been widely recognized; however, they are also associated with bystander tissue damage. The intestinal epithelium provides a physical barrier to prevent direct contact of luminal contents with subepithelial tissues, which is extremely important for the maintenance of intestinal homeostasis. Numerous studies have demonstrated that transepithelial migration of neutrophils is closely related to disease symptoms and disruption of crypt architecture in inflammatory bowel disease and experimental colitis. There has been growing interest in how neutrophils interact with the epithelium under inflammatory conditions. Most studies focus on the effects of neutrophils on intestinal epithelial cells; however, the effects of intestinal epithelial cells on neutrophils during intestinal inflammation need to be well-established. Based on these data, we have summarized recent articles on the role of neutrophil–epithelial interactions in intestinal inflammation, particularly highlighting the epithelium-derived molecular regulators that mediate neutrophil recruitment, transepithelial migration, and detachment from the epithelium, as well as the functional consequences of their crosstalk. A better understanding of these molecular events may help develop novel therapeutic targets for mitigating the deleterious effects of neutrophils in inflammatory bowel disease.
Keywords: Inflammatory Bowel Disease, Intestinal Epithelium, Neutrophil–Epithelial Interactions
Abbreviations used in this paper: A2B (Adora2b), adenosine receptor A2b; Ado, adenosine; CAR, Coxsackie and adenovirus receptor; DSS, dextran sulfate sodium; HIF, hypoxia-inducible factor; HXA3, hepoxilin A3; IBD, inflammatory bowel disease; ICAM-1, intercellular adhesion molecule-1; IEC, intestinal epithelial cell; IL, interleukin; JAML, junction adhesion molecule-like molecule; LTB4, leukotriene B4; MIP-2, macrophage inflammatory protein-2; MMPs, metalloproteinases; NETs, neutrophil extracellular traps; PHD, prolyl hydroxylase; ROS, reactive oxygen species; SIRP-α, signal-regulatory protein-α; TJs, tight junctions; TNF, tumor necrosis factor
Summary.
Neutrophil–epithelial crosstalk functions as a double-edged sword in intestinal inflammation. Our review summarizes the molecular basis and functional consequences of these interactions during intestinal inflammation, aiming to improve the clinical treatment of intestinal inflammation.
The intestinal epithelium consists of a single layer of cells that provides a physical barrier to prevent the direct contact of gut luminal contents, such as bacteria and food particles, with subepithelial tissues.1,2 Maintenance of intestinal homeostasis is closely related to the integrity and permeability of the epithelium, which relies on the controlled proliferation, differentiation, and renewal of intestinal epithelial cells (IECs) and intercellular junctions.3,4 However, both genetic and environmental events can lead to epithelial injury, followed by a cascade of inflammatory responses wherein circulating neutrophils are rapidly recruited to the site of injury.5,6
Neutrophils are the most abundant leukocytes in human blood and are thought to be the first to respond to inflammation.7 They play a vital role in eliminating invasive pathogens.7,8 After neutrophils recognize and phagocytose pathogens, they produce a large amount of toxic reactive oxygen species (ROS), such as superoxide anions (O2-) and hydrogen peroxide (H2O2), through respiratory bursts to destroy the invading bacteria.9,10 They can also release a large number of bactericidal enzymes, such as antimicrobial peptides (defensins and LL37), hydrolases (collagenase, lysozyme, and sialidase), myeloperoxidase, proteases (elastase and cathepsin G), metal chelators, and metalloproteinases, through degranulation.7,11,12 Recently, the novel bactericidal form of neutrophils, termed neutrophil extracellular traps (NETs), a mesh-like structure containing condensed chromatin, DNA, and granular components, has received considerable attention.13, 14, 15, 16 In addition to their pathogen-destroying functions, neutrophils have also been shown to aid in wound healing and resolution of inflammation by producing vascular endothelial growth factors and pro-resolving lipid mediators such as protectin D1 and resolvin E1, which inhibit the recruitment of neutrophils and enhance the phagocytosis of apoptotic neutrophils by macrophages. Moreover, neutrophils decrease the cell debris at the site of inflammation by phagocytosis.16, 17, 18, 19 In addition to their favorable effects, neutrophils can directly destroy the intestinal epithelium by releasing a series of proteases, such as matrix metalloproteinases (MMPs) and neutrophil elastase, and by producing a large amount of ROS from respiratory bursts.10,20 Moreover, neutrophil-derived secretagogues are closely related to goblet cell depletion, which is one of the histological features of intestinal inflammation.21, 22, 23 It is well-accepted that uncontrolled accumulation of hyperactivated neutrophils leads to the deformation of crypt architecture and crypt abscess formation accompanied by an excessive enzymatic reaction; the production of pro-inflammatory cytokines such as tumor necrosis factor (TNF)-α and interleukin (IL)-1β; and the release of non-cytokine inflammatory mediators such as α defensins and calprotectin, which have been demonstrated to recruit monocytes, T lymphocytes, and more neutrophils to the sites of inflammation and to be closely related to the pathogenesis of inflammatory bowel disease (IBD).24, 25, 26, 27, 28 IBD, including ulcerative colitis and Crohn’s disease, is a group of chronic and recurrent gastrointestinal inflammatory conditions. These conditions are characterized by massive neutrophilic infiltration, mucosal damage, increased epithelial permeability, and transepithelial translocation of commensal microorganisms into the underlying tissues.29,30
As highlighted above, neutrophils function as a double-edged sword in intestinal inflammation.31 Recent studies have shown that the intestinal epithelium plays a vital role in the recruitment, maintenance, and elimination of neutrophils at sites of inflammation.32 Accordingly, neutrophil–epithelial crosstalk determines the prognosis of intestinal inflammation. This review aims to determine the role of neutrophil–epithelial interactions in intestinal inflammation, particularly the effects of IECs on neutrophils during inflammatory processes. An in-depth understanding of neutrophil–epithelial interactions in intestinal inflammation, particularly the molecular basis, is imperative in ultimately identifying precise therapeutic targets of IBD.
The Intestinal Epithelium Recruits Neutrophils to Sites of Inflammation
At the onset of intestinal inflammation, neutrophils are rapidly recruited from microcirculation to the gut through a series of chemotactic gradients. Numerous studies have revealed that immune cells, such as macrophages and Th17 cells, release chemotactic cytokines (IL-1β, IL-6, TNF-α, and chemokines [CXCL-8, CXCL-10], and macrophage inflammatory proteins [MIP]-2) and growth factors (granulocyte-macrophage colony-stimulating factor and granulocyte colony-stimulating factor), which play critical roles in the recruitment of neutrophils during IBD.33, 34, 35 Moreover, bacterial products (formyl-methionyl-leucyl-phenylalanine and short-chain fatty acids) have also been shown to attract neutrophils into the gut.36, 37, 38 However, little is known about the function of the epithelium in attracting neutrophils to the site of inflammation. Based on the few studies conducted to date, we summarized the chemotactic substances that are released by the intestinal epithelium to enhance neutrophil migration.
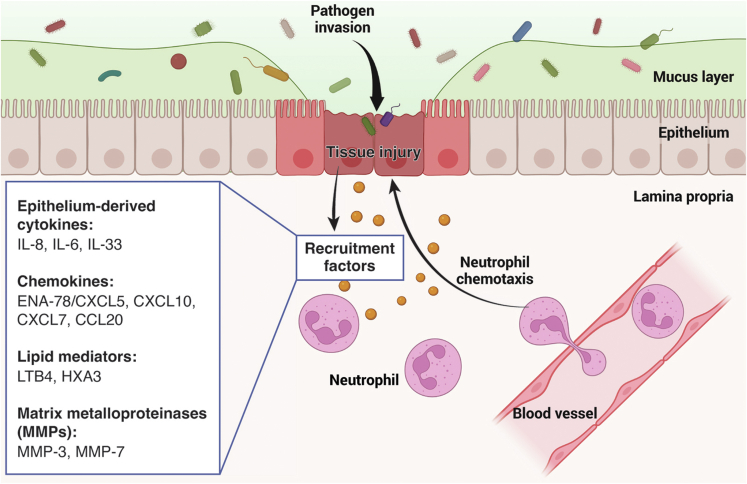
Cytokines are key regulators of neutrophil recruitment during intestinal inflammation.7 It is widely accepted that IL-8, a powerful chemoattractant released by IECs, effectively attracts neutrophils to the basolateral surface of the epithelium.39,40 Notably, the gene expression and protein concentration of IL-8 in the intestinal mucosa are significantly increased during IBD.41,42 Moreover, under chronic inflammatory conditions, Prevotella stimulates IECs to secrete IL-6, which facilitates neutrophil migration across the lamina propria to the intestinal epithelium.43 In addition, Chen et al demonstrated that the intestinal epithelial-derived IL-33 indirectly recruits neutrophils to sites of inflammation via enhanced platelet activity (Figure 1).44
Figure 1.
Epithelium-derived recruitment factors for neutrophils that enable neutrophil migration to inflammation sites. Schematic diagram outlining the recruitment signals released by inflammatory epithelial cells that attract neutrophils to migrate from blood vessels to sites of inflammation across the lamina propria. Epithelium-derived chemokines include cytokines (IL-8, IL-6, IL-33), chemokines (ENA-78/CXCL5, CXCL10, CXCL7, CCL20), lipid mediators (LTB4, HXA3), and matrix metalloproteinases (MMP-3, MMP-7). Figure created with BioRender.
In addition to cytokines, epithelium-derived chemokines are potent chemoattractants of neutrophils. Previous studies have indicated that compared with that in healthy tissues, the expression of neutrophil-activating protein-78 (ENA-78/CXCL5) is elevated in the colonic tissues of patients with ulcerative colitis and have established that enterocytes are the source of ENA-78.45 Furthermore, CXCL10 is abundantly secreted by murine IECs. In a previous study, the genetic depletion of CXCR3, which is the receptor of CXCL10, could alleviate dextran sulfate sodium (DSS)-induced colitis; however, CXCR3-knockout mice had significantly fewer intestinal neutrophils than wild-type mice did, which confirmed that CXCL10 is a vital neutrophil chemotactic molecule.46,47 Moreover, other epithelial-derived chemokines, such as CXCL7 and CCL20, have been reported to play important roles in neutrophil recruitment and infiltration during IBD and experimental colitis (Figure 1).48,49
Leukotriene B4 (LTB4) belongs to the eicosanoid family and is an important epithelium-derived lipid mediator that attracts neutrophils to the sites of intestinal inflammation.50 LTB4 is biosynthesized from arachidonic acid. Notably, the activities of the 3 key enzymes participating in the leukotriene pathway, namely, 5-lipoxygenase, 5-lipoxygenase-activating protein, and leukotriene A4 hydrolase, were markedly increased in the lesions of patients with active IBD, indicating that an elevated LTB4 release may contribute to neutrophil accumulation during IBD.50,51 In addition, genetic depletion of 5-lipoxygenase or neutralization of 5-lipoxygenase with zileuton is associated with significant amelioration of experimental colitis.52 Another arachidonic acid metabolite, hepoxilin A3 (HXA3), a well-known neutrophil chemotactic agent, is released from the apical surface of the epithelium to drive neutrophils to migrate across the epithelium into the gut lumen.40 Previous studies have shown that the suppression of key enzymes essential for HXA3 synthesis inhibits HXA3-mediated neutrophil infiltration.53,54 These observations indicate that HXA3 is a potent lipid mediator of epithelial origin that promotes neutrophil chemotaxis (Figure 1).
Epithelium-derived MMPs are also important regulators of neutrophil recruitment.7 MMPs are zinc- or calcium-dependent endoproteases that potently degrade the extracellular matrix to form channels for neutrophil transmigration.17 In addition, previous studies have revealed that MMPs can indirectly regulate neutrophil migration through processing chemokines. For instance, MMP-3 can enhance neutrophil attraction via proteolytic processing and activation of the IEC-released CXCL7 precursor. Interestingly, in lesions of patients with ulcerative colitis, the expression of CXCL7 is increased.49 Furthermore, epithelium-derived MMP-7 is important for neutrophil recruitment. Compared with wild-type mice, MMP-7-knockout mice show less neutrophil infiltration in the gut after DSS treatment, and this phenotype is likely to be related to the proteolytic effect of MMP7 on CXCL8 and macrophage inflammatory protein-2.55 Nevertheless, the specific mechanism of the interactions between MMP7 and these 2 chemokines remains unclear and needs to be further verified (Figure 1).
Taken together, these examples suggest that intestinal epithelial cells are also an important source of neutrophil trafficking to the intestinal epithelium, and these epithelium-derived neutrophil recruitment factors are promising novel targets for the treatment of IBD (Figure 1).
Adhesion Molecules Act as “Rungs on the Ladder” for Neutrophils Squeezing Through IECs
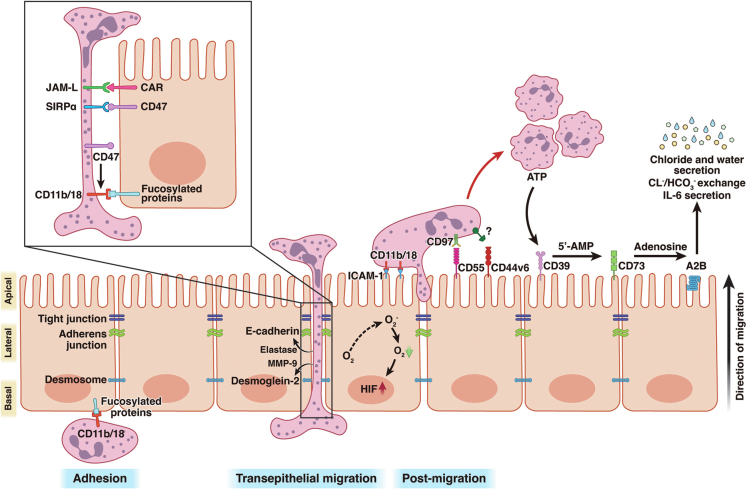
Neutrophils need to cross the epithelium into the gut lumen in a process termed transepithelial migration, and they participate in the pathogenesis of intestinal inflammation.32,56 In this multistep process, neutrophils coordinate with the epithelium through a series of molecules to enable adhesion and transmigration. First, neutrophils adhere to the basolateral surface of the epithelium, and this process exclusively relies on CD11b/CD18 (also known as β-2 integrin). Although the ligand for CD11b/CD18 on IECs remains ambiguous, fucosylated glycoproteins are speculated to be potential ligands.57,58 In addition, it is well-established that CD11b/CD18 is an important mediator of the transepithelial migration of neutrophils; however, CD11b/CD18 antibodies can only partially inhibit neutrophil migration across the T84 cell monolayer, suggesting the existence of CD11b/CD18-independent mechanisms of neutrophil epithelial trafficking into the gut lumen (Figure 2).58, 59, 60
Figure 2.
Molecular basis and functional consequences of neutrophil–epithelial crosstalk in transepithelial migration. The initiation of the neutrophil adhesion step is mediated by CD11b/18 binding to the fucosylated proteins of the IEC basolateral membrane. The movement of neutrophils through the IEC intercellular space is regulated by neutrophil–epithelial cell (SIRP-α/CD47 and JAM-L/CAR) molecular interactions; in addition, CD47 expressed by neutrophils can promote neutrophil migration in a CD11b/18-dependent manner. Neutrophil-produced enzymes such as elastase and MMP-9 can disrupt cell junctions by cleaving E-cadherin (the core protein of adhesion junctions) and desmocore 2 (a key protein of desmosomes), respectively. Neutrophil transepithelial migration can stabilize HIF in IECs by promoting epithelial restitution through increased oxygen consumption. When neutrophils rise to the apical surface of the epithelium, their retention is regulated by the interaction of neutrophil-expressed CD11b/18 and epithelium-derived ICAM-1. In contrast, epithelial CD55 (which binds to the CD97 expressed on neutrophils) and CD44v6 are responsible for the detachment of migrating neutrophils. Neutrophils produce a large amount of ATP that is converted into Ado and adenosine by epithelial CD39 and CD73, respectively. Subsequently, adenosine binds to its receptor A2B (expressed on the apical membrane of IECs), which results in the secretion of chloride and water, CL-/HCO3- exchange, and IL-6 production by IECs. Figure created with BioRender
After the adhesion step, neutrophils squeeze through adjacent IECs, where they challenge desmosomes and adherent and tight junctions (TJs) in the paracellular route; this process requires broad cooperation between the epithelium and neutrophils.7,61 Although little is known about neutrophil transepithelial migration, a few candidate molecules have been identified. For instance, a transmembrane glycoprotein termed CD47, which is predominantly expressed on the lateral membrane of IECs binds to the signal-regulatory protein-α (SIRP-α) expressed on neutrophils and mediates the post-adhesive events required for neutrophil transepithelial migration in the gut (Figure 2).62,63 In contrast, Azcutia et al indicated that the recently discovered neutrophil-derived CD47, and not the CD47 expressed by IEC, modulated the passage of CD11b/CD18-dependent neutrophils through the intercellular space of the epithelium (Figure 2).64
As neutrophils continue to rise to the TJ level, the Coxsackie and adenovirus receptor (CAR) expressed on IECs interact with the JAM-like (JAML) molecules expressed on neutrophils to help them migrate across TJs.65 Moreover, the recombinant ectodomain of CAR suppresses the transepithelial migration of neutrophils (Figure 2).65 It is well-known that TJs play a central role in controlling the epithelial barrier function; however, it should be noted that the JAML/CAR interaction creates gaps between IECs, which results in reduced transepithelial resistance, leading to the development of enteritis.66,67 Furthermore, occludin has been reported to regulate this migration process in Madin-Darby canine kidney cells; however, no evidence has been found for IECs in humans or experimental animals.68
It should be noted that the molecular details regulating neutrophil transepithelial migration are still only partially understood. To date, most of the observations are derived from transwell-based experiments in vitro; however, they cannot completely mimic the process of neutrophil epithelial trafficking in vivo. Of note, Flemming et al provided a novel approach called the murine colonic loop model, which enables the quantification of neutrophils migrating across the colonic epithelium in vivo. This approach may aid in identifying the target receptors responsible for the transepithelial migration of neutrophils.69 Furthermore, other in vivo methods, such as live-cell imaging and 2-photon microscopy, can be utilized to track the crosstalk between migrating neutrophils and IECs.
Neutrophil Transepithelial Migration Regulates Epithelial Barrier Function
For transepithelial migration, neutrophils not only directly interact with the above-mentioned adhesion molecules but also target structural molecules called cadherins and catenins by producing elastase, proteinase 3, and MMPs. For example, the core adhesion junction protein E-cadherin maintains mucosal homeostasis by binding to the adapter proteins β-catenin and p120-catenin and is a target of neutrophil elastase (Figure 2).70 Indeed, anti-elastase treatment has been shown to ameliorate DSS-induced colitis in BALB/c mice.71 Intriguingly, genetic depletion of p120-catenin in the mouse gut results in massive neutrophil infiltration and dysfunction of the epithelial barrier, which induces a phenotype similar to that of patients with IBD.70 In addition, neutrophil-derived microparticles containing MMP-9 disrupt epithelial adhesion by cleaving desmoglein-2 rather than E-cadherin, thereby enhancing neutrophil trafficking (Figure 2). It should be emphasized that the marked enhancement of epithelial permeability increases leukocyte exposure to invasive antigens and stimulates immune responses, whereas the persistent transepithelial migration of neutrophils is associated with mucosal damage and altered barrier function, which are the characteristics of IBD.32
In addition to increasing the epithelial permeability, neutrophil transepithelial migration also influences the proliferation, apoptosis, and function of IECs. For instance, the JAML/CAR combination has been demonstrated to disrupt mucosal wound healing by inhibiting IEC proliferation.72 Moreover, the binding of neutrophils to intercellular adhesion molecule-1 (ICAM-1) activates β-catenin and Akt signaling to promote IEC proliferation and mucosal wound healing.73 In contrast, neutrophil-derived microparticles hinder intestinal wound healing by suppressing epithelial cell proliferation.74 Furthermore, the neutrophils in lesions from patients with IBD and experimental mice impede mucosal wound closure by releasing pro-inflammatory miR-23a and miR-155.75 Recently, NETs have been shown to induce apoptosis of IECs, and they destroy the integrity of cell junctions in experimental colitis. NETs have been shown to positively correlate with disease activity, as demonstrated by their increased presence in the lesions of patients with IBD and their direct role in perpetuating intestinal inflammation in IBD.76, 77, 78, 79 However, the underlying mechanism remains poorly understood. Additionally, transepithelial migration of neutrophils has been shown to increase the expression of MMP ADAM17 in IECs, which activates the pro-inflammatory cytokine TNF-α by cleaving its precursor. This upregulated ADAM17 expression has been found in lesions of patients with IBD in the active phase but not in the chronic phase. Based on this, transepithelial migration can lead to colitis by promoting the production of inflammatory cytokines by IECs.80, 81, 82
Recent studies have revealed that IECs regulate hypoxia-inducible factor (HIF), an important transcription factor that regulates gene expression in hypoxic microenvironments.83, 84, 85 HIF targets various genes, such as those encoding mucins, antimicrobial peptides, and others associated with inflammation resolution, which are critical for maintaining the mucosal barrier.86,87 Numerous experimental colitis studies have shown that during acute intestinal inflammation, transmigrating neutrophils consume oxygen within the epithelial microenvironment in a reduced nicotinamide adenine dinucleotide phosphate-oxidase-dependent manner, thereby significantly stabilizing HIF.56,88 Subsequently, the stabilization of HIF triggers the transcription of a set of genes that enable IECs to promote mucosal restitution (Figure 2).88,89 Given that HIF stabilization is an essential component of mucosal recovery during inflammation, a growing number of pharmacological molecules have been developed to achieve HIF stabilization; currently, this is primarily achieved by inhibiting prolyl hydroxylase (PHD).6,90 A PHD inhibitor, FG-4497, has been shown to significantly promote inflammatory resolution in experimental colitis, with similar pro-resolution effects observed in dimethyloxalylglycine-treated mice.91,92 However, HIF stabilizers need to be developed for clinical translation in the treatment of IBD.
Neutrophil–Epithelial Interactions at the Lumen Surface of the Gut
Normally, neutrophils undergo cell death after completing their tasks and detach from the intestinal epithelium to resolve inflammation and restore tissue function. Otherwise, excessive accumulation of neutrophils in the intestinal epithelium leads to crypt abscess formation and tissue damage, a hallmark of IBD, through the release of numerous pro-inflammatory mediators, ROS, and enzymes.93 Although infiltrated neutrophils mainly undergo apoptosis and are phagocytosed by macrophages, recent studies have shown that IECs also aid in the clearance of neutrophils after migration.7,17 As neutrophils ascend to the apical surface of the intestinal epithelium, an important epithelial-expressed adhesion molecule, termed ICAM-1, interacts with CD11b/CD18 on neutrophils. Furthermore, ICAM-1 is thought to be involved in neutrophil retention on the apical membranes of IECs.94 In contrast, another adhesion molecule, CD44v6, is specifically expressed on the apical surface of the epithelium in the inflammatory state and is responsible for the detachment of neutrophils into the gut lumen.95,96 Likewise, the increased expression of CD55 (known as a decay-accelerating factor) also facilitates the apoptosis of migrated neutrophils through direct contact with neutrophil-derived CD97.97 These adhesion molecules (ie, CD44v6 and CD55) appear to play contrasting roles to ICAM-1 in the release of post-migrated neutrophils, and the balance between CD44v6/CD55 and ICAM-1 is critical in crypt abscess formation (Figure 2).
Furthermore, migrating neutrophils are known to release large amounts of ATP, which are in turn converted into adenosine (Ado) by CD39 and CD73, 2 ectonucleotidases expressed on the apical membrane of the intestinal epithelium.98,99 Ado subsequently activates the cyclic adenosine monophosphate signaling pathway by binding to A2B (Adora2b), an adenosine receptor expressed on IECs.100,101 The combination of Ado and A2B plays a key role in wound healing, inflammation resolution, and maintenance of intestinal mucosal homeostasis, and it has received extensive attention in recent years.100,102,103 For instance, their interaction with the lumen surface of the gut leads to electrogenic chloride secretion into the gut lumen and passive water transport. This passive water flux helps to remove noxious antigens from the surface of the intestinal epithelium. Colitis severity is significantly increased in mice lacking A2B in the gut.100 However, passive water transport is also the pathological basis of diarrhea in patients with IBD.31,58,104 Except for their direct role in releasing ATP at the apical membrane of the intestinal epithelium, the translocated neutrophils can indirectly alter fluid homeostasis by facilitating the transepithelial migration of platelets, which produce even higher levels of ATP in the intestinal lumen than neutrophils do.105 In addition, previous studies have shown that the combination of Ado and A2B helps maintain pH homeostasis in the gut.56,106,107 In murine and human colonic organoids, neutrophil-derived Ado can significantly upregulate the expression of SLC26A3, a major CL-/HCO3- exchanger on the apical membrane of IECs via CREB/cAMP signaling, which subsequently regulates the local pH to facilitate the adaptation of IECs to the inflammation-induced acidity.106 Furthermore, it has been shown that stimulation of the intestinal epithelial cell line T84 monolayer with adenosine induces increased polarized secretion of IL-6 (a pro-inflammatory cytokine associated with neutrophil degranulation) into the intestinal lumen. Similar to SLC26A3, Ado-induced IL-6 secretion is also dependent on CREB/cAMP signaling, which induces an intracellular calcium flux in neutrophils and subsequently promotes neutrophil degranulation to enhance the bactericidal effect of neutrophils upon entry into the intestinal lumen (Figure 2).108
Collectively, the interaction between neutrophils and IECs on the apical surface of the epithelium plays a key role not only in inflammation resolution but also in tissue damage. More research is needed in the future to gain a deeper understanding of how this interaction affects the prognosis of intestinal inflammation, which will facilitate clinical translation (Figure 2).
Conclusion
Our understanding of the double-edged sword role of neutrophil–epithelial interactions in intestinal inflammation has increased significantly in recent years. It is well-known that neutrophils are essential for mucosal immunity; however, excessive recruitment and activation of neutrophils is a direct cause of intestinal mucosal crypt abscesses and extensive mucosal damage. Numerous studies have revealed the effects of neutrophils on IECs; however, the effects of IECs on neutrophils in colitis need to be well-elucidated. Our review provides insights into the recent research on the neutrophil–epithelial crosstalk in colitis and highlights the important roles of IECs in regulating neutrophil recruitment, transepithelial migration, cell death, and clearance. Despite mounting evidence showing that neutrophil–epithelial crosstalk can determine the prognosis of intestinal inflammation, the molecular events modulating their interactions and the underlying regulatory mechanisms are only partially understood, and they should be the major targets in further research. More appropriate methods, such as live-cell imaging and 2-photon microscopy, should be utilized to track neutrophil transepithelial trafficking in vivo. In vitro, an air–liquid interface culture of colonic organoids, similar to the normal monolayer cell culture system but comprising diverse intestinal epithelial cells, can be used to investigate how neutrophils interact with various epithelial cells rather than with enterocytes alone. New technologies such as single-cell RNA sequencing and spatial transcriptomics can improve our understanding of the distribution of cells during neutrophil transepithelial migration, reveal cell-to-cell communication, and predict the potential signals that mediate the interaction between neutrophils and IECs. In addition, exosomes containing functional components, such as proteins and RNAs, have been shown to play an important role in mediating intercellular communication in IBD. They are worth investigating in further studies on the crosstalk between neutrophils and IECs. In conclusion, a better understanding of the complex interactions between neutrophils and IECs during intestinal inflammation may provide new avenues for tissue-specific treatment of IBD.
Acknowledgments
CRediT Authorship Contributions
Le Kang, (Conceptualization: Lead; Writing – original draft: Lead; Visualization: Lead)
Xue Fang (Conceptualization: Equal; Visualization: Lead; Writing – original draft: Equal)
Yi-Hang Song (Visualization: Supporting)
Zi-Xuan He (Visualization: Supporting)
Zhi-Jie Wang (Visualization: Supporting)
Shu-Ling Wang (Writing – review & editing: Equal)
Zhao-Shen Li (Conceptualization: Lead; Writing – review & editing: Lead)
Yu Bai (Conceptualization: Lead; Funding acquisition: Lead; Writing – review & editing: Lead)
Footnotes
Conflicts of interest The authors disclose no conflicts.
Funding This study was supported by the National Natural Science Foundation of China (Nos. 81873546, 82170567, and 82100587); the Shu Guang project of Shanghai Municipal Education Commission and Shanghai Education Development Foundation (No. 19SG30); the Shanghai Sailing Program (No. 21YF1458700); 234 Discipline Climbing Plan of Changhai Hospital, Naval Medical University (No. 2019YXK004); the Program of Shanghai Academic/Technology Research Leader (No. 22XD1425000); and the Youth Start-up Fund of Changhai Hospital, Naval Medical University (No. 2019QNB02).
Contributor Information
Shu-Ling Wang, Email: wangshuling0000@126.com.
Zhao-Shen Li, Email: li.zhaoshen@hotmail.com.
Yu Bai, Email: md.baiyu@foxmail.com.
References
- 1.Soderholm A.T., Pedicord V.A. Intestinal epithelial cells: at the interface of the microbiota and mucosal immunity. Immunology. 2019;158:267–280. doi: 10.1111/imm.13117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Fassarella M., Blaak E.E., Penders J., Nauta A., Smidt H., Zoetendal E.G. Gut microbiome stability and resilience: elucidating the response to perturbations in order to modulate gut health. Gut. 2021;70:595–605. doi: 10.1136/gutjnl-2020-321747. [DOI] [PubMed] [Google Scholar]
- 3.Degirmenci B., Valenta T., Dimitrieva S., Hausmann G., Basler K. GLI1-expressing mesenchymal cells form the essential Wnt-secreting niche for colon stem cells. Nature. 2018;558:449–453. doi: 10.1038/s41586-018-0190-3. [DOI] [PubMed] [Google Scholar]
- 4.Borrelli C., Valenta T., Handler K., Velez K., Gurtner A., Moro G., Lafzi A., Roditi L.V., Hausmann G., Arnold I.C., Moor A.E., Basler K. Differential regulation of beta-catenin-mediated transcription via N- and C-terminal co-factors governs identity of murine intestinal epithelial stem cells. Nat Commun. 2021;12:1368. doi: 10.1038/s41467-021-21591-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Honarpisheh M., Foresto-Neto O., Desai J., Steiger S., Gomez L.A., Popper B., Boor P., Anders H.J., Mulay S.R. Phagocytosis of environmental or metabolic crystalline particles induces cytotoxicity by triggering necroptosis across a broad range of particle size and shape. Sci Rep. 2017;7 doi: 10.1038/s41598-017-15804-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Hall C.H.T., Campbell E.L., Colgan S.P. Neutrophils as components of mucosal homeostasis. Cell Mol Gastroenterol Hepatol. 2017;4:329–337. doi: 10.1016/j.jcmgh.2017.07.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Zhou G., Yu L., Fang L., Yang W., Yu T., Miao Y., Chen M., Wu K., Chen F., Cong Y., Liu Z. CD177(+) neutrophils as functionally activated neutrophils negatively regulate IBD. Gut. 2018;67:1052–1063. doi: 10.1136/gutjnl-2016-313535. [DOI] [PubMed] [Google Scholar]
- 8.Cai S., Paudel S., Jin L., Ghimire L., Taylor C.M., Wakamatsu N., Bhattarai D., Jeyaseelan S. NLRP6 modulates neutrophil homeostasis in bacterial pneumonia-derived sepsis. Mucosal Immunol. 2021;14:574–584. doi: 10.1038/s41385-020-00357-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Amara N., Cooper M.P., Voronkova M.A., Webb B.A., Lynch E.M., Kollman J.M., Ma T., Yu K., Lai Z., Sangaraju D., Kayagaki N., Newton K., Bogyo M., Staben S.T., Dixit V.M. Selective activation of PFKL suppresses the phagocytic oxidative burst. Cell. 2021;184:4480–4494.e15. doi: 10.1016/j.cell.2021.07.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Denson L.A., Jurickova I., Karns R., Shaw K.A., Cutler D.J., Okou D.T., Dodd A., Quinn K., Mondal K., Aronow B.J., Haberman Y., Linn A., Price A., Bezold R., Lake K., Jackson K., Walters T.D., Griffiths A., Baldassano R.N., Noe J.D., Hyams J.S., Crandall W.V., Kirschner B.S., Heyman M.B., Snapper S., Guthery S.L., Dubinsky M.C., Leleiko N.S., Otley A.R., Xavier R.J., Stevens C., Daly M.J., Zwick M.E., Kugathasan S. Clinical and genomic correlates of neutrophil reactive oxygen species production in pediatric patients with Crohn’s disease. Gastroenterology. 2018;154:2097–2110. doi: 10.1053/j.gastro.2018.02.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Kienle K., Glaser K.M., Eickhoff S., Mihlan M., Knopper K., Reategui E., Epple M.W., Gunzer M., Baumeister R., Tarrant T.K., Germain R.N., Irimia D., Kastenmuller W., Lammermann T. Neutrophils self-limit swarming to contain bacterial growth in vivo. Science. 2021;372 doi: 10.1126/science.abe7729. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Bedouhène S., Dang P.M.-C., Hurtado-Nedelec M., El-Benna J. In: Neutrophil: Methods and Protocols. Quinn M.T., DeLeo F.R., editors. Springer US; 2020. Neutrophil degranulation of azurophil and specific granules; pp. 215–222. [DOI] [PubMed] [Google Scholar]
- 13.Leppkes M., Lindemann A., Gosswein S., Paulus S., Roth D., Hartung A., Liebing E., Zundler S., Gonzalez-Acera M., Patankar J.V., Mascia F., Scheibe K., Hoffmann M., Uderhardt S., Schauer C., Foersch S., Neufert C., Vieth M., Schett G., Atreya R., Kuhl A.A., Bleich A., Becker C., Herrmann M., Neurath M.F. Neutrophils prevent rectal bleeding in ulcerative colitis by peptidyl-arginine deiminase-4-dependent immunothrombosis. Gut. 2021 doi: 10.1136/gutjnl-2021-324725. Online ahead of print. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Lin E.Y., Lai H.J., Cheng Y.K., Leong K.Q., Cheng L.C., Chou Y.C., Peng Y.C., Hsu Y.H., Chiang H.S. Neutrophil extracellular traps impair intestinal barrier function during experimental colitis. Biomedicines. 2020;8:275. doi: 10.3390/biomedicines8080275. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Yang L., Liu Q., Zhang X., Liu X., Zhou B., Chen J., Huang D., Li J., Li H., Chen F., Liu J., Xing Y., Chen X., Su S., Song E. DNA of neutrophil extracellular traps promotes cancer metastasis via CCDC25. Nature. 2020;583:133–138. doi: 10.1038/s41586-020-2394-6. [DOI] [PubMed] [Google Scholar]
- 16.Seo D.H., Che X., Kim S., Kim D.H., Ma H.W., Kim J.H., Kim T.I., Kim W.H., Kim S.W., Cheon J.H. Triggering receptor expressed on myeloid cells-1 agonist regulates intestinal inflammation via Cd177(+) neutrophils. Front Immunol. 2021;12 doi: 10.3389/fimmu.2021.650864. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Phillipson M., Kubes P. The healing power of neutrophils. Trends Immunol. 2019;40:635–647. doi: 10.1016/j.it.2019.05.001. [DOI] [PubMed] [Google Scholar]
- 18.McCourt M., Wang J.H., Sookhai S., Redmond H.P. Proinflammatory mediators stimulate neutrophil-directed angiogenesis. Arch Surg. 1999;134:1325–1331. doi: 10.1001/archsurg.134.12.1325. discussion: 1331–1332. [DOI] [PubMed] [Google Scholar]
- 19.Schwab J.M., Chiang N., Arita M., Serhan C.N. Resolvin E1 and protectin D1 activate inflammation-resolution programmes. Nature. 2007;447:869–874. doi: 10.1038/nature05877. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Tarlton J.F., Whiting C.V., Tunmore D., Bregenholt S., Reimann J., Claesson M.H., Bland P.W. The role of up-regulated serine proteases and matrix metalloproteinases in the pathogenesis of a murine model of colitis. Am J Pathol. 2000;157:1927–1935. doi: 10.1016/S0002-9440(10)64831-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Leiper K., Campbell B.J., Jenkinson M.D., Milton J., Yu L.G., Democratis J., Rhodes J.M. Interaction between bacterial peptides, neutrophils and goblet cells: a possible mechanism for neutrophil recruitment and goblet cell depletion in colitis. Clin Sci. 2001;101:395–402. [PubMed] [Google Scholar]
- 22.Shinoda M., Shin-Ya M., Naito Y., Kishida T., Ito R., Suzuki N., Yasuda H., Sakagami J., Imanishi J., Kataoka K., Mazda O., Yoshikawa T. Early-stage blocking of Notch signaling inhibits the depletion of goblet cells in dextran sodium sulfate-induced colitis in mice. J Gastroenterol. 2010;45:608–617. doi: 10.1007/s00535-010-0210-z. [DOI] [PubMed] [Google Scholar]
- 23.Surawicz C.M., Haggitt R.C., Husseman M., McFarland L.V. Mucosal biopsy diagnosis of colitis: Acute self-limited colitis and idiopathic inflammatory bowel disease. Gastroenterology. 1994;107:755–763. doi: 10.1016/0016-5085(94)90124-4. [DOI] [PubMed] [Google Scholar]
- 24.Ozaki R., Kobayashi T., Okabayashi S., Nakano M., Morinaga S., Hara A., Ohbu M., Matsuoka K., Toyonaga T., Saito E., Hisamatsu T., Hibi T. Histological risk factors to predict clinical relapse in ulcerative colitis with endoscopically normal mucosa. J Crohns Colitis. 2018;12:1288–1294. doi: 10.1093/ecco-jcc/jjy092. [DOI] [PubMed] [Google Scholar]
- 25.Karmakar M., Minns M., Greenberg E.N., Diaz-Aponte J., Pestonjamasp K., Johnson J.L., Rathkey J.K., Abbott D.W., Wang K., Shao F., Catz S.D., Dubyak G.R., Pearlman E. N-GSDMD trafficking to neutrophil organelles facilitates IL-1beta release independently of plasma membrane pores and pyroptosis. Nat Commun. 2020;11:2212. doi: 10.1038/s41467-020-16043-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Cunliffe R.N., Kamal M., Rose F.R., James P.D., Mahida Y.R. Expression of antimicrobial neutrophil defensins in epithelial cells of active inflammatory bowel disease mucosa. J Clin Pathol. 2002;55:298–304. doi: 10.1136/jcp.55.4.298. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Yang D., Chen Q., Chertov O., Oppenheim J.J. Human neutrophil defensins selectively chemoattract naive T and immature dendritic cells. J Leukoc Biol. 2000;68:9–14. [PubMed] [Google Scholar]
- 28.Yui S., Nakatani Y., Mikami M. Calprotectin (S100A8/S100A9), an inflammatory protein complex from neutrophils with a broad apoptosis-inducing activity. Biol Pharm Bull. 2003;26:753–760. doi: 10.1248/bpb.26.753. [DOI] [PubMed] [Google Scholar]
- 29.Friedrich M., Pohin M., Jackson M.A., Korsunsky I., Bullers S.J., Rue-Albrecht K., Christoforidou Z., Sathananthan D., Thomas T., Ravindran R., Tandon R., Peres R.S., Sharpe H., Wei K., Watts G.F.M., Mann E.H., Geremia A., Attar M., Oxford IBD Cohort Investigators, Roche Fibroblast Network Consortium, McCuaig S., Thomas L., Collantes E., Uhlig H.H., Sansom S.N., Easton A., Raychaudhuri S., Travis S.P., Powrie F.M. IL-1-driven stromal-neutrophil interactions define a subset of patients with inflammatory bowel disease that does not respond to therapies. Nat Med. 2021;27:1970–1981. doi: 10.1038/s41591-021-01520-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Li G., Lin J., Zhang C., Gao H., Lu H., Gao X., Zhu R., Li Z., Li M., Liu Z. Microbiota metabolite butyrate constrains neutrophil functions and ameliorates mucosal inflammation in inflammatory bowel disease. Gut Microbes. 2021;13 doi: 10.1080/19490976.2021.1968257. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Parkos C.A. Neutrophil-epithelial interactions: a double-edged sword. Am J Pathol. 2016;186:1404–1416. doi: 10.1016/j.ajpath.2016.02.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Lin Y., Cheng L., Liu Y., Wang Y., Wang Q., Wang H.L., Shi G., Li J.S., Wang Q.N., Yang Q.M., Chen S., Su X.L., Yang Y., Jiang M., Hu X., Fan P., Fang C., Zhou Z.G., Dai L., Deng H.X. Intestinal epithelium-derived BATF3 promotes colitis-associated colon cancer through facilitating CXCL5-mediated neutrophils recruitment. Mucosal Immunol. 2021;14:187–198. doi: 10.1038/s41385-020-0297-3. [DOI] [PubMed] [Google Scholar]
- 33.Salem M., El Azreq M.A., Pelletier J., Robaye B., Aoudjit F., Sevigny J. Exacerbated intestinal inflammation in P2Y6 deficient mice is associated with Th17 activation. Biochim Biophys Acta Mol Basis Dis. 2019;1865:2595–2605. doi: 10.1016/j.bbadis.2019.06.019. [DOI] [PubMed] [Google Scholar]
- 34.Park H., Li Z., Yang X.O., Chang S.H., Nurieva R., Wang Y.H., Wang Y., Hood L., Zhu Z., Tian Q., Dong C. A distinct lineage of CD4 T cells regulates tissue inflammation by producing interleukin 17. Nat Immunol. 2005;6:1133–1141. doi: 10.1038/ni1261. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Mateer S.W., Mathe A., Bruce J., Liu G., Maltby S., Fricker M., Goggins B.J., Tay H.L., Marks E., Burns G., Kim R.Y., Minahan K., Walker M.M., Callister R.C., Foster P.S., Horvat J.C., Hansbro P.M., Keely S. IL-6 drives neutrophil-mediated pulmonary inflammation associated with bacteremia in murine models of colitis. Am J Pathol. 2018;188:1625–1639. doi: 10.1016/j.ajpath.2018.03.016. [DOI] [PubMed] [Google Scholar]
- 36.Bedouhene S., Liu M., Senani N., Boussetta T., Pintard C., Dang P.M., El-Benna J. Prolyl-isomerase Pin1 controls key fMLP-induced neutrophil functions. Biomedicines. 2021;9:1130. doi: 10.3390/biomedicines9091130. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Correa R.O., Vieira A., Sernaglia E.M., Lancellotti M., Vieira A.T., Avila-Campos M.J., Rodrigues H.G., Vinolo M.A.R. Bacterial short-chain fatty acid metabolites modulate the inflammatory response against infectious bacteria. Cell Microbiol. 2017;19 doi: 10.1111/cmi.12720. [DOI] [PubMed] [Google Scholar]
- 38.Kamp M.E., Shim R., Nicholls A.J., Oliveira A.C., Mason L.J., Binge L., Mackay C.R., Wong C.H. G protein-coupled receptor 43 modulates neutrophil recruitment during acute inflammation. PLoS One. 2016;11 doi: 10.1371/journal.pone.0163750. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Kucharzik T., Hudson J.T., 3rd, Lugering A., Abbas J.A., Bettini M., Lake J.G., Evans M.E., Ziegler T.R., Merlin D., Madara J.L., Williams I.R. Acute induction of human IL-8 production by intestinal epithelium triggers neutrophil infiltration without mucosal injury. Gut. 2005;54:1565–1572. doi: 10.1136/gut.2004.061168. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Holani R., Babbar A., Blyth G.A.D., Lopes F., Jijon H., McKay D.M., Hollenberg M.D., Cobo E.R. Cathelicidin-mediated lipopolysaccharide signaling via intracellular TLR4 in colonic epithelial cells evokes CXCL8 production. Gut Microbes. 2020;12 doi: 10.1080/19490976.2020.1785802. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Jeffrey M.P., Strap J.L., Jones Taggart H., Green-Johnson J.M. Suppression of intestinal epithelial cell chemokine production by Lactobacillus rhamnosus R0011 and Lactobacillus helveticus R0389 is mediated by secreted bioactive molecules. Front Immunol. 2018;9:2639. doi: 10.3389/fimmu.2018.02639. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Kim H., Venancio V.P., Fang C., Dupont A.W., Talcott S.T., Mertens-Talcott S.U. Mango (Mangifera indica L.) polyphenols reduce IL-8, GRO, and GM-SCF plasma levels and increase Lactobacillus species in a pilot study in patients with inflammatory bowel disease. Nutr Res. 2020;75:85–94. doi: 10.1016/j.nutres.2020.01.002. [DOI] [PubMed] [Google Scholar]
- 43.Larsen J.M. The immune response to Prevotella bacteria in chronic inflammatory disease. Immunology. 2017;151:363–374. doi: 10.1111/imm.12760. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Chen Z., Luo J., Li J., Kim G., Stewart A., Huang Y., Wu C. Intestinal IL-33 promotes platelet activity for neutrophil recruitment during acute inflammation. Blood. 2022;139:1878–1891. doi: 10.1182/blood.2021013474. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Keates S., Keates A.C., Mizoguchi E., Bhan A., Kelly C.P. Enterocytes are the primary source of the chemokine ENA-78 in normal colon and ulcerative colitis. Am J Physiol. 1997;273(1 Pt 1):G75–G82. doi: 10.1152/ajpgi.1997.273.1.G75. [DOI] [PubMed] [Google Scholar]
- 46.Chami B., Yeung A.W., van Vreden C., King N.J., Bao S. The role of CXCR3 in DSS-induced colitis. PLoS One. 2014;9 doi: 10.1371/journal.pone.0101622. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Salem M., Tremblay A., Pelletier J., Robaye B., Sevigny J. P2Y6 receptors regulate CXCL10 expression and secretion in mouse intestinal epithelial cells. Front Pharmacol. 2018;9:149. doi: 10.3389/fphar.2018.00149. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Vergara A.F., Vidal R.M., Torres A.G., Farfan M.J. Long polar fimbriae participates in the induction of neutrophils transepithelial migration across intestinal cells infected with enterohemorrhagic E. coli O157:H7. Front Cell Infect Microbiol. 2014;4:185. doi: 10.3389/fcimb.2014.00185. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Kruidenier L., MacDonald T.T., Collins J.E., Pender S.L., Sanderson I.R. Myofibroblast matrix metalloproteinases activate the neutrophil chemoattractant CXCL7 from intestinal epithelial cells. Gastroenterology. 2006;130:127–136. doi: 10.1053/j.gastro.2005.09.032. [DOI] [PubMed] [Google Scholar]
- 50.Kang S.A., Yu H.S. Acceleration of Trichinella spiralis worm expulsion by leukotriene B4 receptor binding inhibition. Parasite Immunol. 2021;43 doi: 10.1111/pim.12843. [DOI] [PubMed] [Google Scholar]
- 51.Mohan S., Mok S., Judge T. Identification of novel therapeutic molecular targets in inflammatory bowel disease by using genetic databases. Clin Exp Gastroenterol. 2020;13:467–473. doi: 10.2147/CEG.S264812. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Pallio G., Bitto A., Pizzino G., Galfo F., Irrera N., Minutoli L., Arcoraci V., Squadrito G., Macri A., Squadrito F., Altavilla D. Use of a balanced dual cyclooxygenase-1/2 and 5-lypoxygenase inhibitor in experimental colitis. Eur J Pharmacol. 2016;789:152–162. doi: 10.1016/j.ejphar.2016.07.033. [DOI] [PubMed] [Google Scholar]
- 53.Agbor T.A., Demma Z., Mrsny R.J., Castillo A., Boll E.J., McCormick B.A. The oxido-reductase enzyme glutathione peroxidase 4 (GPX4) governs Salmonella Typhimurium-induced neutrophil transepithelial migration. Cell Microbiol. 2014;16:1339–1353. doi: 10.1111/cmi.12290. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Hurley B.P., Sin A., McCormick B.A. Adhesion molecules involved in hepoxilin A3-mediated neutrophil transepithelial migration. Clin Exp Immunol. 2008;151:297–305. doi: 10.1111/j.1365-2249.2007.03551.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Swee M., Wilson C.L., Wang Y., McGuire J.K., Parks W.C. Matrix metalloproteinase-7 (matrilysin) controls neutrophil egress by generating chemokine gradients. J Leukoc Biol. 2008;83:1404–1412. doi: 10.1189/jlb.0108016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Campbell E.L., Bruyninckx W.J., Kelly C.J., Glover L.E., McNamee E.N., Bowers B.E., Bayless A.J., Scully M., Saeedi B.J., Golden-Mason L., Ehrentraut S.F., Curtis V.F., Burgess A., Garvey J.F., Sorensen A., Nemenoff R., Jedlicka P., Taylor C.T., Kominsky D.J., Colgan S.P. Transmigrating neutrophils shape the mucosal microenvironment through localized oxygen depletion to influence resolution of inflammation. Immunity. 2014;40:66–77. doi: 10.1016/j.immuni.2013.11.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Zen K., Liu Y., Cairo D., Parkos C.A. CD11b/CD18-dependent interactions of neutrophils with intestinal epithelium are mediated by fucosylated proteoglycans. J Immunol. 2002;169:5270–5278. doi: 10.4049/jimmunol.169.9.5270. [DOI] [PubMed] [Google Scholar]
- 58.Overbeek S.A., Kleinjan M., Henricks P.A., Kamp V.M., Ricciardolo F.L., Georgiou N.A., Garssen J., Kraneveld A.D., Folkerts G. Chemo-attractant N-acetyl proline-glycine-proline induces CD11b/CD18-dependent neutrophil adhesion. Biochim Biophys Acta. 2013;1830:2188–2193. doi: 10.1016/j.bbagen.2012.09.023. [DOI] [PubMed] [Google Scholar]
- 59.Kelm M., Lehoux S., Azcutia V., Cummings R.D., Nusrat A., Parkos C.A., Brazil J.C. Regulation of neutrophil function by selective targeting of glycan epitopes expressed on the integrin CD11b/CD18. FASEB J. 2020;34:2326–2343. doi: 10.1096/fj.201902542R. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Blake K.M., Carrigan S.O., Issekutz A.C., Stadnyk A.W. Neutrophils migrate across intestinal epithelium using beta2 integrin (CD11b/CD18)-independent mechanisms. Clin Exp Immunol. 2004;136:262–268. doi: 10.1111/j.1365-2249.2004.02429.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Boerner K., Luissint A.C., Parkos C.A. Functional assessment of intestinal permeability and neutrophil transepithelial migration in mice using a standardized intestinal loop model. J Vis Exp. 2021:168. doi: 10.3791/62093. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Liu Y., Merlin D., Burst S.L., Pochet M., Madara J.L., Parkos C.A. The role of CD47 in neutrophil transmigration. Increased rate of migration correlates with increased cell surface expression of CD47. J Biol Chem. 2001;276:40156–40166. doi: 10.1074/jbc.M104138200. [DOI] [PubMed] [Google Scholar]
- 63.Parkos C.A., Colgan S.P., Liang T.W., Nusrat A., Bacarra A.E., Carnes D.K., Madara J.L. CD47 mediates post-adhesive events required for neutrophil migration across polarized intestinal epithelia. J Cell Biol. 1996;132:437–450. doi: 10.1083/jcb.132.3.437. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Azcutia V., Kelm M., Luissint A.C., Boerner K., Flemming S., Quiros M., Newton G., Nusrat A., Luscinskas F.W., Parkos C.A. Neutrophil expressed CD47 regulates CD11b/CD18-dependent neutrophil transepithelial migration in the intestine in vivo. Mucosal Immunol. 2021;14:331–341. doi: 10.1038/s41385-020-0316-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Zen K., Liu Y., McCall I.C., Wu T., Lee W., Babbin B.A., Nusrat A., Parkos C.A. Neutrophil migration across tight junctions is mediated by adhesive interactions between epithelial coxsackie and adenovirus receptors and a junctional adhesion molecule-like protein on neutrophils. Mol Biol Cell. 2005;16:2694–2703. doi: 10.1091/mbc.E05-01-0036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Kuo W.T., Zuo L., Odenwald M.A., Madha S., Singh G., Gurniak C.B., Abraham C., Turner J.R. The tight junction protein ZO-1 is dispensable for barrier function but critical for effective mucosal repair. Gastroenterology. 2021;161:1924–1939. doi: 10.1053/j.gastro.2021.08.047. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Chen X., Liu R., Liu X., Xu C., Wang X. Protective role of coxsackie-adenovirus receptor in the pathogenesis of inflammatory bowel diseases. Biomed Res Int. 2018;2018 doi: 10.1155/2018/7207268. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Huber D., Balda M.S., Matter K. Occludin modulates transepithelial migration of neutrophils. J Biol Chem. 2000;275:5773–5778. doi: 10.1074/jbc.275.8.5773. [DOI] [PubMed] [Google Scholar]
- 69.Flemming S., Luissint A.C., Nusrat A., Parkos C.A. Analysis of leukocyte transepithelial migration using an in vivo murine colonic loop model. JCI Insight. 2018;3 doi: 10.1172/jci.insight.99722. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Smalley-Freed W.G., Efimov A., Burnett P.E., Short S.P., Davis M.A., Gumucio D.L., Washington M.K., Coffey R.J., Reynolds A.B. p120-catenin is essential for maintenance of barrier function and intestinal homeostasis in mice. J Clin Invest. 2010;120:1824–1835. doi: 10.1172/JCI41414. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Shioya Y., Katakura K., Ohira H. Neutrophil elastase inhibitor suppresses IL-17 based inflammation of murine experimental colitis. Fukushima J Med Sci. 2014;60:14–21. doi: 10.5387/fms.2013-2. [DOI] [PubMed] [Google Scholar]
- 72.Weber D.A., Sumagin R., McCall I.C., Leoni G., Neumann P.A., Andargachew R., Brazil J.C., Medina-Contreras O., Denning T.L., Nusrat A., Parkos C.A. Neutrophil-derived JAML inhibits repair of intestinal epithelial injury during acute inflammation. Mucosal Immunol. 2014;7:1221–1232. doi: 10.1038/mi.2014.12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Sumagin R., Brazil J.C., Nava P., Nishio H., Alam A., Luissint A.C., Weber D.A., Neish A.S., Nusrat A., Parkos C.A. Neutrophil interactions with epithelial-expressed ICAM-1 enhances intestinal mucosal wound healing. Mucosal Immunol. 2016;9:1151–1162. doi: 10.1038/mi.2015.135. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Slater T.W., Finkielsztein A., Mascarenhas L.A., Mehl L.C., Butin-Israeli V., Sumagin R. Neutrophil microparticles deliver active myeloperoxidase to injured mucosa to inhibit epithelial wound healing. J Immunol. 2017;198:2886–2897. doi: 10.4049/jimmunol.1601810. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Butin-Israeli V., Bui T.M., Wiesolek H.L., Mascarenhas L., Lee J.J., Mehl L.C., Knutson K.R., Adam S.A., Goldman R.D., Beyder A., Wiesmuller L., Hanauer S.B., Sumagin R. Neutrophil-induced genomic instability impedes resolution of inflammation and wound healing. J Clin Invest. 2019;129:712–726. doi: 10.1172/JCI122085. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Zhang T., Mei Y., Dong W., Wang J., Huang F., Wu J. Evaluation of protein arginine deiminase-4 inhibitor in TNBS- induced colitis in mice. Int Immunopharmacol. 2020;84 doi: 10.1016/j.intimp.2020.106583. [DOI] [PubMed] [Google Scholar]
- 77.Li T., Wang C., Liu Y., Li B., Zhang W., Wang L., Yu M., Zhao X., Du J., Zhang J., Dong Z., Jiang T., Xie R., Ma R., Fang S., Zhou J., Shi J. Neutrophil extracellular traps induce intestinal damage and thrombotic tendency in inflammatory bowel disease. J Crohns Colitis. 2020;14:240–253. doi: 10.1093/ecco-jcc/jjz132. [DOI] [PubMed] [Google Scholar]
- 78.Brinkmann V., Zychlinsky A. Beneficial suicide: why neutrophils die to make NETs. Nat Rev Microbiol. 2007;5:577–582. doi: 10.1038/nrmicro1710. [DOI] [PubMed] [Google Scholar]
- 79.Yipp B.G., Petri B., Salina D., Jenne C.N., Scott B.N., Zbytnuik L.D., Pittman K., Asaduzzaman M., Wu K., Meijndert H.C., Malawista S.E., de Boisfleury Chevance A., Zhang K., Conly J., Kubes P. Infection-induced NETosis is a dynamic process involving neutrophil multitasking in vivo. Nat Med. 2012;18:1386–1393. doi: 10.1038/nm.2847. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Quarta S., Mitric M., Kalpachidou T., Mair N., Schiefermeier-Mach N., Andratsch M., Qi Y., Langeslag M., Malsch P., Rose-John S., Kress M. Impaired mechanical, heat, and cold nociception in a murine model of genetic TACE/ADAM17 knockdown. FASEB J. 2019;33:4418–4431. doi: 10.1096/fj.201801901R. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Shimoda M., Horiuchi K., Sasaki A., Tsukamoto T., Okabayashi K., Hasegawa H., Kitagawa Y., Okada Y. Epithelial cell-derived a disintegrin and metalloproteinase-17 confers resistance to colonic inflammation through EGFR activation. EBioMedicine. 2016;5:114–124. doi: 10.1016/j.ebiom.2016.02.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Giese A.A., Babendreyer A., Krappen P., Gross A., Strnad P., Dusterhoft S., Ludwig A. Inflammatory activation of surface molecule shedding by upregulation of the pseudoprotease iRhom2 in colon epithelial cells. Sci Rep. 2021;11 doi: 10.1038/s41598-021-03522-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Sun L., Li T., Tang H., Yu K., Ma Y., Yu M., Qiu Y., Xu P., Xiao W., Yang H. Intestinal epithelial cells-derived hypoxia-inducible factor-1alpha is essential for the homeostasis of intestinal intraepithelial lymphocytes. Front Immunol. 2019;10:806. doi: 10.3389/fimmu.2019.00806. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Dowdell A.S., Cartwright I.M., Goldberg M.S., Kostelecky R., Ross T., Welch N., Glover L.E., Colgan S.P. The HIF target ATG9A is essential for epithelial barrier function and tight junction biogenesis. Mol Biol Cell. 2020;31:2249–2258. doi: 10.1091/mbc.E20-05-0291. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Islam S.M.T., Won J., Khan M., Mannie M.D., Singh I. Hypoxia-inducible factor-1 drives divergent immunomodulatory functions in the pathogenesis of autoimmune diseases. Immunology. 2021;164:31–42. doi: 10.1111/imm.13335. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Fachi J.L., Felipe J.S., Pral L.P., da Silva B.K., Correa R.O., de Andrade M.C.P., da Fonseca D.M., Basso P.J., Camara N.O.S., de Sales E.S.E.L., Dos Santos Martins F., Guima S.E.S., Thomas A.M., Setubal J.C., Magalhaes Y.T., Forti F.L., Candreva T., Rodrigues H.G., de Jesus M.B., Consonni S.R., Farias A.D.S., Varga-Weisz P., Vinolo M.A.R. Butyrate protects mice from clostridium difficile-induced colitis through an HIF-1-dependent mechanism. Cell Rep. 2019;27:750–761.e7. doi: 10.1016/j.celrep.2019.03.054. [DOI] [PubMed] [Google Scholar]
- 87.Yin J., Zhou C., Yang K., Ren Y., Qiu Y., Xu P., Xiao W., Yang H. Mutual regulation between butyrate and hypoxia-inducible factor-1alpha in epithelial cell promotes expression of tight junction proteins. Cell Biol Int. 2020;44:1405–1414. doi: 10.1002/cbin.11336. [DOI] [PubMed] [Google Scholar]
- 88.Lin N., Shay J.E.S., Xie H., Lee D.S.M., Skuli N., Tang Q., Zhou Z., Azzam A., Meng H., Wang H., FitzGerald G.A., Simon M.C. Myeloid cell hypoxia-inducible factors promote resolution of inflammation in experimental colitis. Front Immunol. 2018;9:2565. doi: 10.3389/fimmu.2018.02565. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Goggins B.J., Minahan K., Sherwin S., Soh W.S., Pryor J., Bruce J., Liu G., Mathe A., Knight D., Horvat J.C., Walker M.M., Keely S. Pharmacological HIF-1 stabilization promotes intestinal epithelial healing through regulation of alpha-integrin expression and function. Am J Physiol Gastrointest Liver Physiol. 2021;320:G420–G438. doi: 10.1152/ajpgi.00192.2020. [DOI] [PubMed] [Google Scholar]
- 90.Wu M.M., Wang Q.M., Huang B.Y., Mai C.T., Wang C.L., Wang T.T., Zhang X.J. Dioscin ameliorates murine ulcerative colitis by regulating macrophage polarization. Pharmacol Res. 2021;172 doi: 10.1016/j.phrs.2021.105796. [DOI] [PubMed] [Google Scholar]
- 91.Bakshi H.A., Mishra V., Satija S., Mehta M., Hakkim F.L., Kesharwani P., Dua K., Chellappan D.K., Charbe N.B., Shrivastava G., Rajeshkumar S., Aljabali A.A., Al-Trad B., Pabreja K., Tambuwala M.M. Dynamics of prolyl hydroxylases levels during disease progression in experimental colitis. Inflammation. 2019;42:2032–2036. doi: 10.1007/s10753-019-01065-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Liu Y., Zhu F., Li H., Fan H., Wu H., Dong Y., Chu S., Tan C., Wang Q., He H., Gao F., Leng X., Zhou Q., Zhu X. MiR-155 contributes to intestinal barrier dysfunction in DSS-induced mice colitis via targeting HIF-1alpha/TFF-3 axis. Aging. 2020;12:14966–14977. doi: 10.18632/aging.103555. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Perez-Figueroa E., Alvarez-Carrasco P., Ortega E. Maldonado-Bernal C. Neutrophils: many ways to die. Front Immunol. 2021;12 doi: 10.3389/fimmu.2021.631821. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Huang G.T., Eckmann L., Savidge T.C., Kagnoff M.F. Infection of human intestinal epithelial cells with invasive bacteria upregulates apical intercellular adhesion molecule-1 (ICAM)-1) expression and neutrophil adhesion. J Clin Invest. 1996;98:572–583. doi: 10.1172/JCI118825. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Brazil J.C., Liu R., Sumagin R., Kolegraff K.N., Nusrat A., Cummings R.D., Parkos C.A., Louis N.A. alpha3/4 Fucosyltransferase 3-dependent synthesis of Sialyl Lewis A on CD44 variant containing exon 6 mediates polymorphonuclear leukocyte detachment from intestinal epithelium during transepithelial migration. J Immunol. 2013;191:4804–4817. doi: 10.4049/jimmunol.1301307. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Brazil J.C., Lee W.Y., Kolegraff K.N., Nusrat A., Parkos C.A., Louis N.A. Neutrophil migration across intestinal epithelium: evidence for a role of CD44 in regulating detachment of migrating cells from the luminal surface. J Immunol. 2010;185:7026–7036. doi: 10.4049/jimmunol.1001293. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Lawrence D.W., Bruyninckx W.J., Louis N.A., Lublin D.M., Stahl G.L., Parkos C.A., Colgan S.P. Antiadhesive role of apical decay-accelerating factor (CD55) in human neutrophil transmigration across mucosal epithelia. J Exp Med. 2003;198:999–1010. doi: 10.1084/jem.20030380. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Eltzschig H.K., Ibla J.C., Furuta G.T., Leonard M.O., Jacobson K.A., Enjyoji K., Robson S.C., Colgan S.P. Coordinated adenine nucleotide phosphohydrolysis and nucleoside signaling in posthypoxic endothelium: role of ectonucleotidases and adenosine A2B receptors. J Exp Med. 2003;198:783–796. doi: 10.1084/jem.20030891. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Zhu Y., Zhuang Z., Wu Q., Lin S., Zhao N., Zhang Q., Xie L., Yu S. CD39/CD73/A2a adenosine metabolic pathway: targets for moxibustion in treating DSS-induced ulcerative colitis. Am J Chinese Med. 2021;49:661–676. doi: 10.1142/S0192415X21500300. [DOI] [PubMed] [Google Scholar]
- 100.Antonioli L., El-Tayeb A., Pellegrini C., Fornai M., Awwad O., Giustarini G., Natale G., Ryskalin L., Nemeth Z.H., Muller C.E., Blandizzi C., Colucci R. Anti-inflammatory effect of a novel locally acting A2A receptor agonist in a rat model of oxazolone-induced colitis. Purinergic Signal. 2018;14:27–36. doi: 10.1007/s11302-017-9591-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Schenck L.P., Hirota S.A., Hirota C.L., Boasquevisque P., Tulk S.E., Li Y., Wadhwani A., Doktorchik C.T., Macnaughton W.K., Beck P.L., MacDonald J.A. Attenuation of Clostridium difficile toxin-induced damage to epithelial barrier by ecto-5'-nucleotidase (CD73) and adenosine receptor signaling. Neurogastroenterol Motil. 2013;25:e441–e453. doi: 10.1111/nmo.12139. [DOI] [PubMed] [Google Scholar]
- 102.Li X., Li C., Li Y., Liu C., Liang X., Liu T., Liu Z. Sodium nitroprusside protects HFD induced gut dysfunction via activating AMPKalpha/SIRT1 signaling. BMC Gastroenterol. 2021;21:359. doi: 10.1186/s12876-021-01934-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Huang L., Fan J., Chen Y.X., Wang J.H. Inhibition of A2B adenosine receptor attenuates intestinal injury in a rat model of necrotizing enterocolitis. Mediators Inflamm. 2020;2020 doi: 10.1155/2020/1562973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Asano T., Noda Y., Tanaka K.I., Yamakawa N., Wada M., Mashimo T., Fukunishi Y., Mizushima T., Takenaga M. A2B adenosine receptor inhibition by the dihydropyridine calcium channel blocker nifedipine involves colonic fluid secretion. Sci Rep. 2020;10:3555. doi: 10.1038/s41598-020-60147-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Weissmuller T., Campbell E.L., Rosenberger P., Scully M., Beck P.L., Furuta G.T., Colgan S.P. PMNs facilitate translocation of platelets across human and mouse epithelium and together alter fluid homeostasis via epithelial cell-expressed ecto-NTPDases. J Clin Invest. 2008;118:3682–3692. doi: 10.1172/JCI35874. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Cartwright I.M., Curtis V.F., Lanis J.M., Alexeev E.E., Welch N., Goldberg M.S., Schaefer R.E.M., Gao R.Y., Chun C., Fennimore B., Onyiah J.C., Gerich M.E., Dempsey P.J., Colgan S.P. Adaptation to inflammatory acidity through neutrophil-derived adenosine regulation of SLC26A3. Mucosal Immunol. 2020;13:230–244. doi: 10.1038/s41385-019-0237-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Jung D., Alshaikh A., Ratakonda S., Bashir M., Amin R., Jeon S., Stevens J., Sharma S., Ahmed W., Musch M., Hassan H. Adenosinergic signaling inhibits oxalate transport by human intestinal Caco2-BBE cells through the A2B adenosine receptor. Am J Physiol Cell Physiol. 2018;315:C687–C698. doi: 10.1152/ajpcell.00024.2017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Sitaraman S.V., Merlin D., Wang L., Wong M., Gewirtz A.T., Si-Tahar M., Madara J.L. Neutrophil-epithelial crosstalk at the intestinal lumenal surface mediated by reciprocal secretion of adenosine and IL-6. J Clin Invest. 2001;107:861–869. doi: 10.1172/JCI11783. [DOI] [PMC free article] [PubMed] [Google Scholar]