Abstract
The retina, owing to its cellular anatomy and physical location, is susceptible to generating reactive oxygen species (ROS), which are associated with several major retinal diseases. When ROS exceeds the body's natural antioxidants, the retina is in a state of oxidative stress, which is recognized as the pathogenesis of retinal diseases. The early stage of the pathogenic process is an adaptive change in which oxidative stress and endogenous defense mechanisms occur. If no treatment is applied, the retinal diseases will progress to the pathological stage with neuronal and vascular dysfunction or damage and even blindness. This review summarizes the role of oxidative stress in several common retinal diseases, including retinitis pigmentosa, age-related macular degeneration, diabetic retinopathy, glaucoma, and retinopathy of prematurity. In addition, we discuss the early intervention strategies for these diseases. An outline is provided to identify potential intervention targets for further research. Early intervention for retinal diseases is necessary and urgent and may offer hope to improve patients' quality of life through functional vision.
1. Introduction
The retina is an extension of the brain and is a highly oxygen-consuming organ in the body, relying more on aerobic glycolysis than the brain; it is also highly sensitive to various stimuli [1]. Furthermore, the retina is extremely metabolically active and rich in polyunsaturated fatty acids, which are vulnerable to lipid peroxidation [2]. Under normal conditions, reactive oxygen species (ROS) in the retina are related to physiological signaling and protective mechanisms via the prosurvival extracellular signal-related kinase 1/2 pathway and endoplasmic reticulum stress signaling. ROS production-induced oxidative stress may contribute to the pathogenesis of several retinal degenerative diseases, including diabetic retinopathy (DR), retinal vascular occlusion, retinitis pigmentosa (RP), age-related macular degeneration (AMD), glaucoma, and retinopathy of prematurity (ROP).
The number of people with retinal diseases, including AMD, DR, and glaucoma, is expected to more than double by 2050 [3]. This prediction indicates that there will be a healthcare crisis that not only affects patients with visual disturbance but also the caregivers and entire healthcare system. Thus, there is an urgent need to focus on the prevention of and protection against retinal diseases. This review includes the necessary recommendations and neuroprotective strategies for preventing the progression of vision loss and blindness in individuals with retinal diseases.
In living cells, oxidation-reduction reactions may generate metabolic byproducts—free radicals, which are ROS and reactive nitrogen species (RNS) [4]. Free radicals are a class of molecules containing one or more unpaired electrons. ROS can be generated via multiple pathways, including endogenous and exogenous substances [4]. ROS are highly active and may react with biomolecules, e.g., proteins, lipid membranes, and DNA, leading to cell damage or functional impairment. Oxidative stress is a condition of oxidation-reduction imbalance characterized by elevated levels of ROS and free radicals or a reduction of antioxidants [5, 6]. Under physiological conditions, the generation of ROS and the scavenging of free radicals may reach a dynamic redox balance. When various excess stimuli (endogenous and/or exogenous) occur, ROS accumulate massively. This may lead to oxidative stress in the corresponding cells and organs [7]. Continuous oxidative stress may ultimately damage cells in target organs. Therefore, the inhibition of ROS generation and scavenging of excessive ROS by various pathways have been used as therapeutic strategies for the treatment of eye diseases. In this review, we will introduce the mechanisms of ROS generation in several retinal diseases, summarize the pathogenetic roles of oxidative stress in their development, and discuss the early intervention pathway of antioxidative stress.
2. Physiological Role of Free Radicals and ROS in Organisms
The free radicals in the human body can be divided into oxygen and nonoxygen radicals. Free oxygen radicals are the predominant category derived from molecular oxygen, and their proportion of oxygen radicals is approximately 95% of the total radicals. Oxygen radicals include hydrogen peroxide (H2O2), hydroxyl radicals, superoxide anions (O2−·), nitroxides, and peroxynitrite, which are defined as ROS and RNS. ROS/RNS can be produced by the metabolism of normal cells and exogenous stimuli, e.g., chemical drugs, high-pressure oxygen, radiation, mental stress, insomnia, smoking, and pollution [8] Free radicals are highly reactive and short-lived intermediates that have one or more unpaired electrons and exist independently on their own [9, 10]. Under normal conditions, free radicals are important for maintaining homeostasis and defending the body against hazardous invasion [11] by (1) enhancing phagocytosis of leukocytes, (2) promoting synthesis of prostaglandin and lipoxygenases, and (3) relaxing vascular smooth muscle and modulating blood pressure. Endogenous ROS are derived from the mitochondria by escaping electrons to molecular oxygen [12]. The generation sources include NADPH oxidases (Nox), xanthine oxidase, and lipoxygenase on the membranes of endothelial cells and phagocytes [2]. In most cases, the terms “free radicals” and “ROS” are regarded as interchangeable [12]. Healthy organisms always generate low levels of free radicals, and antioxidant defense systems may scavenge them rapidly before they cause oxidative damage to the cell. If the dynamic redox balance is imperfect, ROS/RNS-mediated damage may occur continuously. In other words, oxidative stress injury is generated when the speed of free radical production exceeds the capacity of the cellular defense system, for example, exposure to high oxygen pressure or ionizing radiation [8]. Oxidative stress damages biological macromolecules, e.g., nucleic acids, proteins, and lipids by peroxidation, degeneration, crosslinking, and breakage. Finally, oxidative stress can cause injuries to cell structures and functions, as well as to the tissues and organs in the body [11]. Thus far, increasingly more diseases/disorders are recognized gradually by linking them directly or indirectly with oxidative stress [8]. Some of these disorders are due to free radicals, whereas others may be only secondarily involved. Tissue injured by various processes, such as trauma, toxic substances, and infections, may undergo free radical damage more rapidly than healthy tissues. Tissue destruction and degeneration may increase oxidative damage through processes including metal ion release, phagocyte activation, and disruption of mitochondrial electron transport chains. Iron chelation, superoxide dismutase, catalase, vitamins (C and E), and antioxidants (flavonoids) could have protective effects under various experimental conditions, which further indicate the key role of free radicals in many disorders [13–16]. Additionally, an increasing number of small-molecule drugs have been developed and implemented to target ROS [17].
3. Oxidative Stress and Retinal Pathology
Oxidative stress plays a prominent role in the pathogenesis of many degenerative retinal diseases, such as AMD, DR, and RP. Normal, healthy retinal cells are susceptible to significant light exposure, which may lead to the generation of abundant ROS [18]. However, under pathological conditions, the normal homeostatic mechanisms are destroyed. When the relationship of prooxidative stress and antioxidative stress signaling is unbalanced, it may lead to excessive oxidative stress, inflammatory responses, blood-retinal barrier injury, and retinal tissue damage [18, 19]. Retinal diseases, including photoreceptor degeneration, diabetic retinopathy, and retinal ganglion cell injury, always involve the same process of oxidative stress and apoptosis in the final pathological stages. This review will provide a summary of the effects of oxidative stress in several congenital retinal diseases and possible early intervention strategies.
3.1. Oxidative Stress in RP
RP is an inherited retinal disease caused by different genetic mutations. The pathological process leads to photoreceptor cell degeneration (successive rod and cone cell loss) and eventually results in retinal pigmented epithelium (RPE) dysfunction [2]. The prevalence rate is 1 in 4000 persons worldwide, and there are >1.5 million patients with RP worldwide [20]. The early phenotype of RP is difficult to observe at night, and loss of peripheral vision is caused by apoptosis of rod photoreceptors. Then, vision loss becomes more restricted to the central visual field and is eventually lost [20]. Studies have demonstrated that progressive visual loss in RP is associated with the loss of photoreceptor cells and oxidative stress (Figure 1) [21, 22]. Clinical evidence also showed that 8-oxo-7,8-dyhydro-2′-deoxyguanosine (8-oxo-dG) and protein carbonyl contents were increased in the vitreous and aqueous humor of patients with RP [23, 24]. This finding indicates that oxidative stress exists in the ocular tissues of patients with RP. In the outer nuclear layer, rods account for 95% of the cells and are more metabolically active than cones. These cells gradually decrease in number as RP progressed [24]. Subsequently, retinal oxygen demand and usage also decrease. Retinal circulation is not regulated; therefore, the oxygen supply to the outer retina increases spontaneously [24]. Hyperoxia in the outer retina can induce Nox and dual oxidase, but not xanthine oxidase, to produce excessive amounts of ROS and contribute to tissue damage [25]. Hyperoxia in the outer retina induced by rod degeneration may activate Nox enzymes and may also activate the high levels of Nox2 in microglia, which have been demonstrated to migrate into the outer retina in RP. This leads to an increase of superoxide radicals and oxidative damage [25, 26]. High levels of nitric oxide in the outer retina are another indirect oxidative stress factor that may react with superoxide radicals to form highly reactive and damaging peroxynitrite radicals. Hence, inhibition of Nox and nitric oxide synthase (NOS) together with antioxidative stress by scavenging ROS directly should be regarded as a strategy to help maintain photoreceptor function in patients with RP.
Figure 1.
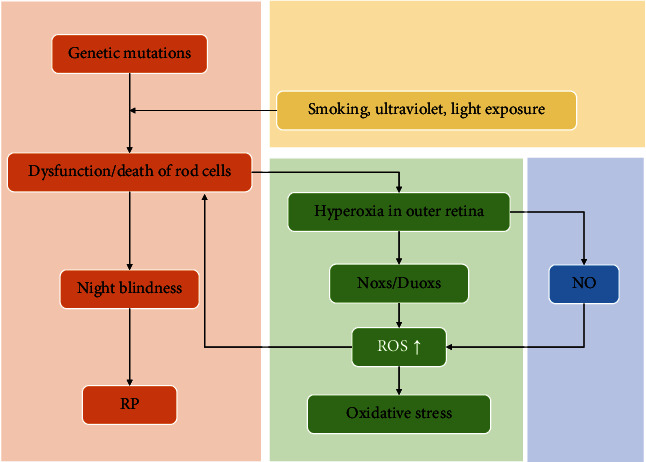
Oxidative stress is involved in the rod cell death-induced clinical feature of RP. ROS: reactive oxygen species; RP: retinitis pigmentosa.
3.1.1. RP: Intervention Strategy at an Early Stage
RP is a family of inherited diseases. Over 250 gene mutations are associated with the cause of rod death, and it is likely the major pathogenesis of RP [27, 28]. Therefore, RP is difficult to cure at the genetic level despite the development of genotype-phenotype technology that may already characterize each causal gene in RP [29]. Currently, there is no specific pharmacological or genetic treatment for RP [27]. Antioxidants have shown promise as an intervention to delay the reduction of cone cell loss and retard the progress of RP [30]. However, the clinical evidence has not been clarified. The application of antioxidant supplements, such as vitamins A and E, beta-carotene, omega-3, docosahexaenoic acid (DHA), zinc, and docosahexaenoic acid, might be able to slow the decline in peripheral field function [20, 30]. The oral antioxidant N-acetyl cysteine (NAC) for RP was studied in a phase 1 clinical trial [31]. However, its efficacy should be addressed in future studies. In addition, a study showed that smoking is detrimental to patients with RP, as it worsens macular function and structural integrity [32]. Hence, smoking may not benefit patients with RP. Furthermore, ultraviolet and blue light exposure may accelerate vision loss in RP [33, 34]. Wearing protective goggles is a simple and effective method to prevent light damage. Regular to moderate physical activity, not endurance exercise without adaptable physical training, may protect against ROS/RNS damage in eye diseases, including RP [35]. Intraocular pressure may affect the circulation of the outer retinal layer. It is possible that an obvious reduction in choroidal blood flow through the elevation of intraocular pressure may prevent sufficient oxygen supply from the choroid [36]. Therefore, it is reasonable to assume that appropriate eyeball massage may be a preconditioning intervention to the retinal outer layer to improve endogenous antioxidative stress during the early stage of RP. Finally, prevention of consanguinity and reinforcement of genetic counseling before birth is very important in reducing the risk of RP. Antioxidant gene modification may be a potential treatment for late stages of RP in the future; although, transgenic overexpression is not applicable to humans.
3.2. Oxidative Stress in AMD
AMD is a chronic, irreversible disease that primarily affects central vision and is an important cause of blindness worldwide. Accumulations of lipid and protein between the RPE and Bruch's membrane (BrM); loss of photoreceptors, RPE, and retinal neurons; and neovascularization are the main pathological processes. Multiple risk factors have been reported for AMD including age, light exposure, smoking, obesity, hypertension, poor antioxidant intake, and a hereditary component [37, 38]. Many studies have demonstrated that oxidative stress is involved in the pathogenesis of AMD [39–42]. The levels of oxidative stress markers, including malondialdehyde, protein carbonyls, and 8-hydroxy-2-deoxyg, are increased significantly in the blood serum of patients with AMD [37, 43]. This finding indicates that systemic oxidative stress is associated with AMD. Additionally, a higher level of carboxyethylpyrrole (CEP) and damaged proteins was detected in the BrM of donor eyes with AMD [44]. CEP is formed from DHA during oxidative stress. The outer segments of photoreceptors are abundantly composed of DHA, which increases their vulnerability to oxidative damage. Under stress conditions, photoreceptors must metabolize continuously to regenerate outer segments, producing a unique source of ROS in RPE cells [45]. RPE cells are responsible for phagocytosis of photoreceptor outer segments. ROS overaccumulation may cause disorders in cell structure and function, which in turn increases ROS generation. Taken together, high oxygen metabolism, continuous light exposure, and high concentrations of polyunsaturated fatty acids make the retina prone to retinal damage. Thus, oxidative stress plays important roles in the pathophysiology of AMD (Figure 2).
Figure 2.
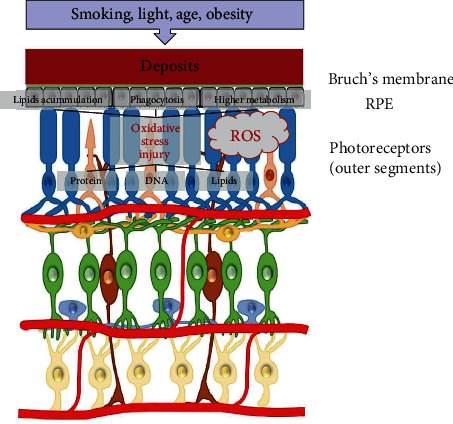
Oxidative stress in AMD. ROS are increased in RPE and photoreceptors since the exposure of environmental risk factors in the early stage of AMD. ROS lead to the deposits of lipid substances, phagocytosis disability, and cell injury in protein, lipids, and DNA, until cell death. AMD: age-related macular degeneration; ROS: reactive oxygen species; RPE: retinal pigmented epithelium.
3.2.1. AMD: Intervention Strategy at an Early Stage
(1) Cigarette smoking has been shown experimentally to be directly linked to the development of AMD and is a major risk factor in epidemiological studies [46, 47]. Therefore, quitting smoking as early as possible is a good strategy to prevent AMD. (2) One of the mechanisms of retinal injury in AMD is the interaction between light exposure and photosensitive molecules (rhodopsin and lipofuscin) [48, 49]. Excessive activation of rhodopsin and light conduction can cause the formation of ROS from the DHA content of the outer segment membranes of the rod and induce photoreceptor cell degeneration [49]. It has been suggested that shorter wavelengths have a higher risk of retinal injury than longer wavelengths [50]. Avoiding exposure to blue light may prevent the increased formation of ROS by photosensitive molecules [51]. (3) Experimental studies have shown that an antioxidation-deficient diet is associated with lipofuscin accumulation and photoreceptor degeneration in the RPE [52]. Increased antioxidants in the diet (vitamins A, C, and E and carotenoids) or serum could protect against AMD progression [49]. A longitudinal clinical study also indicated that the consumption of antioxidants/zinc could decrease the risk of early AMD in a highly susceptible group [53]. Moreover, the topical antioxidant OT-551 (0.45%), investigated in a single-center phase II trial, may improve best-corrected visual acuity [54]. Age-related eye disease studies, antivascular endothelial growth factor (VEGF) injections, and laser therapy are also useful for controlling the progression of wet AMD [55, 56]
3.3. Oxidative Stress in DR
DR, a progressive microvasculature complication of diabetes, is one of the most common causes of blindness in adults of working ages [57, 58]. Approximately 90% of diabetic patients develop DR complications within 25 years of diagnosis [59]. Oxidative stress and inflammation are considered to play key roles in the pathogenesis of DR because the retina has high vascularization and long-term light exposure [60]. Chronic hyperglycemia exposure, resulting in increased ROS production, makes microvessels more vulnerable to oxidative stress. The disturbance of redox homeostasis contributes to the death of neurons in the retina, followed by the rupture of the blood-retinal barrier and increased vascular permeability, leading to advanced DR. In retinal cells under oxidative stress conditions, excessive ROS directly acts on protein and DNA or indirectly acts as a second messenger to affect the pathogenesis of DR (Figure 3) [58, 61, 62]. As the main source of intracellular ROS, mitochondria are abundant in photoreceptors, which are the major O2- contributor in DR [63]. Studies have shown that mitochondrial dysfunction in turn affects the production of ROS in retinal cells, activity of optic nerve cells, and function of photoreceptors. ROS accumulation causes further deterioration [64]. Besides, mitochondrial dysfunction may reduce mitochondrial energy production, leading to optic nerve degeneration [65, 66]. Additionally, the accumulation of byproducts caused by metabolic abnormalities induced by hyperglycemia, e.g., the activation of protein kinase C, hexosamine, polyol flux, and advanced glycation end-products (AGEs), induces oxidative stress through ROS/RNS formation, leading to the death of retinal neurons [58, 67]. Oxygen-derived free radicals, such as hydroperoxyl species, have been shown to cause lipid peroxidation, contributing to the production of ROS to facilitate the senescence of RPE cells, leading to the progression of DR [68–70]. Therefore, oxidative stress plays an important role in DR progression.
Figure 3.
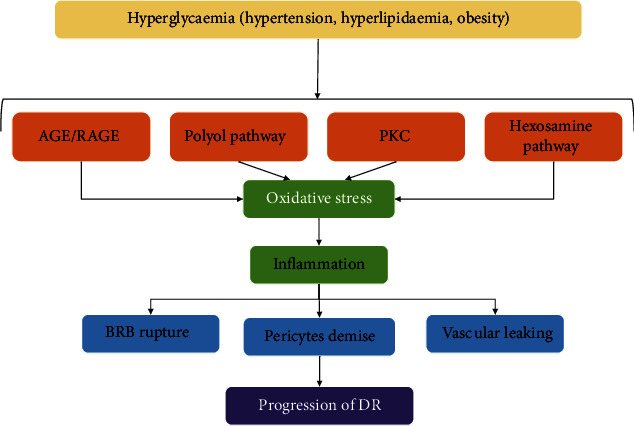
The driving mechanism of oxidative stress in the progression of DR. PKC: protein kinase C; AGEs: advanced glycation end-products; BRB: blood-retinal barrier; DR: diabetic retinopathy.
3.3.1. DR: Intervention Strategy at an Early Stage
Current therapies for DR, such as anti-VEGF treatment, laser therapy, vitrectomy, and glucocorticoids, focus on the late stage, which may reduce visual loss by temporarily protecting retinal vessels [71, 72]. However, it is still difficult for patients with severe vision loss to reestablish normal vision. As a major factor in the progression of DR, oxidative stress is a hot target at an early stage. Nox, the main enzymatic source of ROS, has been demonstrated to be a direct risk factor for DR, and inhibitors of the Nox family have been studied to prevent the development of DR. For example, diphenyleneiodonium (a Nox inhibitor) can suppress ROS generation, alleviate blood-retinal barrier breakdown, and recover the death of retinal capillary endothelial cells [73, 74]. Nox 1/4 specific inhibitors, GKT136901 and GKT137831, have shown potent effects in DR treatment [75]. Polyphenols are antioxidants that are abundant in vegetables, fruits, beverages, whole grains, etc. Studies have demonstrated the protective effects of DR against various polyphenols, including green tea polyphenols, chlorogenic acid, curcumin, beta-glucogallin, and cocoa polyphenols [76–78]. Resveratrol, the most studied polyphenol, has been shown to activate antioxidant enzymes and inactivate NOS activity with a decrease in ROS/RNS in the blood and retina in various experimental models [79–81]. The ROS scavengers NAC and duloxetine are effective in the early stages of DR [82]. Nuclear factor erythroid 2-related factor 2 (NRF2) binds to antioxidant response elements to regulate antioxidant protein levels and fight against oxidative stress. Recent studies have shown that activation of the NRF2 pathway provides new insights for DR treatment. The novel NRF2 activator dh404 provides potential protection to the retina in diabetes, including vision-threatening breakdown of the blood–retinal barrier [83, 84]. Herein, the inhibition or clearance of ROS generation provides an early candidate strategy for blindness caused by DR.
3.4. Oxidative Stress in Glaucoma
Glaucoma is a progressive optic neuropathic disease characterized by retinal ganglion cell (RGCs) degeneration and synaptic loss of dendrites and axon terminals and is the most common cause of irreversible blindness worldwide [85]. It was estimated that the number of people with glaucoma will reach 111.8 million by 2040 globally because of the increase in the number of people of advanced age [86, 87]. Although intraocular pressure is the only known risk factor for glaucoma, increasing studies have shown that oxidative stress plays an important role in the pathogenesis of glaucoma [88, 89]. Increased intraocular pressure and advancing age are closely implicated in the pathogenesis of glaucoma. Oxidative stress occurs in the eyes and may explain the potential mechanism underlying the development of glaucoma [90–92]. Moreover, mitochondrial dysfunction is largely related to oxidative stress in the pathogenesis of glaucoma [93, 94]. Especially in the aging retina, oxidative stress and lipid peroxidation are the major risks to activate the inflammatory response, leading to RGC death and apoptosis, aggravating glaucoma (Figure 4) [95]. Exposure to sunlight and a high-oxygen environment causes higher oxidative stress in the eyes than in other tissues, which can further damage the eye tissue [96, 97]. The trabecular meshwork (TM) is the most sensitive tissue in the eye and is vulnerable to oxidative stress [98]. Studies have shown that increased 8-OH-dG levels are found in the TM of patients with glaucoma, indicating the occurrence of DNA oxidative stress damage [99, 100]. In addition, the accumulation of ROS, particularly H2O2, was also detected in TM cells, which reduced antioxidant activity and inhibited the secretion of adhesion molecules to TM cells, contributing to cytoskeleton reorganization and eventually resulting in cell loss [91, 101, 102].
Figure 4.
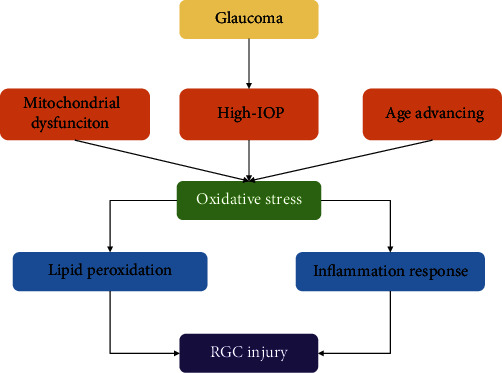
The role of oxidative stress in glaucoma RCG injury. IOP: intraocular pressure; RGC: retinal ganglion cell.
3.4.1. Glaucoma: Intervention Strategy at an Early Stage
Early diagnosis and intervention are important to prevent visual loss in patients with glaucoma due to irreversible blindness [103]. Based on the increasing evidence of oxidative stress in glaucomatous tissues, the levels of biomarker candidates related to oxidative stress, e.g., protein carbonyls and AGEs, have been shown to increase significantly in the blood and aqueous humor samples of patients with glaucoma [104]. Oxidative stress is an important risk factor for ocular hypertension, which is a target of glaucoma treatment [105, 106]. Drugs with antioxidant properties, such as valproic acid and spermidine, have been reported to prevent glaucomatous retinal degeneration in glaucoma mouse models [107, 108]. The grafting of antioxidant molecules to drug carriers can also effectively reduce the intraocular pressure in glaucoma [109]. The activation and recruitment of microglia and astrocytes to the edge of the optic nerve are early characteristics of glaucoma [110–112]. Inhibition of ROS can inhibit the byproducts of electron leakage along the electron transport chain during cell respiration and improve mitochondrial dysfunction, contributing to the delay in glaucoma progression [95, 113, 114]. Additionally, data based on a large population have shown that quitting smoking and moderate-intensity aerobic exercise may reduce the risk of glaucoma [115–117]. A balanced diet, including vegetables, omega fatty acids, and coffee, may help prevent the occurrence or progression of glaucoma [118]. Studies showed that oxidative stress might be a potential target for the prevention and treatment of glaucoma in the early stages; although, there is no direct clinical evidence.
3.5. Oxidative Stress in ROP
ROP is a complex eye disease characterized by retinal neovascularization in low-birth-weight preterm infants (LBWs), and it is the most common cause of blindness in children [119, 120]. Oxidative stress is a physiological redox imbalance caused by excessive ROS/RNS. It plays a key role in the pathogenesis of ROP and significantly increases the mortality and morbidity of ROP in very LBWs (Figure 5) [119, 121, 122]. There are two phases of ROP: (1) abnormal retinal neovascularization motivated by hypoxia and (2) delayed growth of retinal vascularization induced by supplemental oxygen [123–125]. Clinical data showed a connection between the incidence of ROP in LBWs and unrestricted oxygen exposure, and the corresponding findings have shown that reduced oxygen saturation can decrease the incidence of ROP [126]. For newborns, oxidative stress is a challenge during the process of birth itself. The sharp postnatal transition from a lower oxygen content with an intrauterine environment into a higher oxygen content environment causes oxidative stress in infants, and most preterm infants lack antioxidant enzymes and chemical antioxidants because the increase in key antioxidant enzymes such as superoxide dismutase only occurs in late pregnancy, showing a lower antioxidant ability in premature infants [127, 128]. Retinal hyperoxia in LBWs due to the lack of autoregulation of the blood network in the retina causes an imbalance of prooxidants and antioxidants, contributing to the inflammation of retinal tissue, eventually resulting in the development of ROP [129–131]. Moreover, in ROP, damage of the outer retina occurs along with the increased ROS levels in the inner retina [131, 132].
Figure 5.
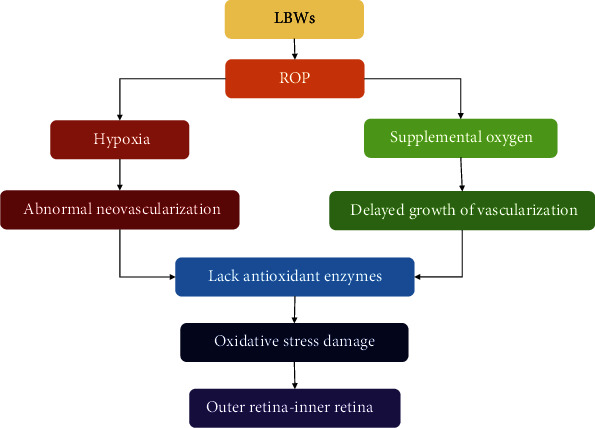
The increased ROS level plays a key role in the pathogenesis of ROP. LBWs: low-birth-weight infants; ROP: retinopathy of prematurity.
3.5.1. ROP: Intervention Strategy at an Early Stage
Early intervention is an efficient strategy to control the incidence of ROP, a preventable ocular disease [133]. According to the early treatment of ROP, cryotherapy and laser therapy are now effective methods to prevent ROP in preterm infants [134–136]. Strict oxygen limitation effectively reduces the incidence of ROP in LBWs. Flavonoids are a group of antioxidants present in the diet [137, 138]. For example, green tee has been demonstrated to prevent ROP through the inhibition of corneal neovascularization in animal models [139, 140]. Bilberries, a natural source of anthocyanins with high antioxidative properties, can significantly render lipid peroxidation and neovascular proliferation and protect the retinal vasculature after high oxygen therapy, contributing to the suppression of ROP progression [140, 141]. Other flavonoids, e.g., baicalin and luteolin, have also been shown to protect against ROP because of their ability to scavenge ROS [142, 143]. Additionally, prenatal supplementation with antioxidants, such as carotenoids and vitamins (A and E), helps to promote normal fetal growth, resulting in a reduction in the birth rate of preterm infants; although, there is no clinical evidence that it can prevent ROP in LBWs [144–147]. Moreover, supplementation with essential fatty acids, e.g., ω-fatty acids, leads to a decrease in ROP development, which can be explained by the antioxidant effect of unsaturated fatty acids that reduces lipid peroxidation [148, 149]. Therefore, it would be a key early intervention strategy to reasonably expect a status with strong antioxidants at birth or provide antioxidant supplementation to reduce the risk of ROP in preterm infants.
4. Summary
An increasing number of people will be diagnosed with retinal diseases in the future. The number of people with vision loss will be 61 million, and that of those with vision impairment will be 470 million by 2050 [3]. The healthcare system will face challenges from millions of people with vision loss and their caregivers. Poor effective treatments currently exist during the advanced disease stage in the clinical setting. Hence, it is important to find ways to detect retinal diseases early and monitor disease progression and treatment efficacy because the adaptive phase and early pathology phase are reversible and are also the most effective phases in retinal diseases (Figure 6) [20].
Figure 6.
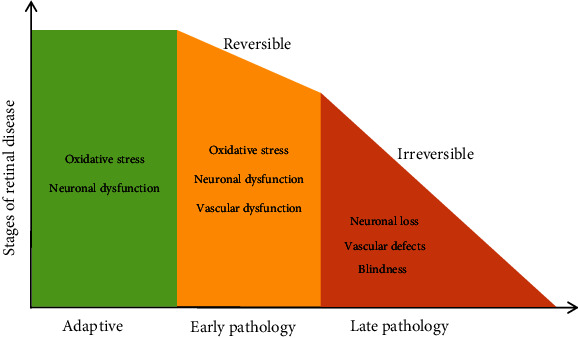
The progression of retinal disease in the hypothetical stages. Effective intervention during the early stage of retinal disease would be most beneficial in protection against vision loss (revised from Machelle T. Pardue, 2018, Prog Retin Eye Res).
Oxidative stress plays a critical role in the pathological processes of many types of retinal dysfunctions [150]. Anatomical features of the eye make the retina more susceptible to ROS production, especially with increasing age [151–153]. The retina, especially the photoreceptors and retinal pigment epithelium, is rich in polyunsaturated fatty acids, which are susceptible to lipid peroxidation [50, 154]. Persistent prooxidant factors and decreased antioxidant capacity with age may accelerate oxidative stress. Thus, the increase in ROS and intracellular Ca2+ concentrations are common pathological changes in the retina [50]. Many studies have indicated that oxidant stress injury is the first step to induce the cell death of retinal neurons [24, 50, 155–157]. Ocular diseases share the same cellular mechanisms. In the final stages of retinal disease, there are limited effective treatments that may rescue lost vision.
Therefore, early intervention for retinal diseases is necessary and urgent, and may offer hope to improve patients' quality of life through functional vision. First, management of the source of ROS generation is a key factor in preventing the progression of retinal disease. Various factors that lead to ROS generation should be suppressed, including hyperglycemia, intraocular hypertension, hyperlipidemia, ultraviolet light exposure, ischemia, and obesity. Early reduction of these stressors may ameliorate ROS-induced retinal damage. Second, the application of dietary antioxidants, antioxidant supplementation, or pharmacological inhibitors could be an effective intervention strategy in the early stages of retinal diseases. This approach may scavenge excess free radicals and reduce oxidative stress injury. Third, physical exercise as a rehabilitation treatment has shown pluripotent benefits in multiple systems of the body, including retinal diseases [158, 159]. Recently, exercise has been demonstrated to have protective effects in animal models of retinal diseases via multiple mechanisms, including the BDNF/TrkB signaling pathway, increased blood flow, and modulation of VEGF and its receptors [160, 161]. Retrospective studies in humans have also indicated that exercise treatment for visual disorders can improve visual function and quality of life. Therefore, exercise interventions may be implemented in the early stage of retinal disease. The retina is a metabolically active tissue that is susceptible to oxidative injury. Therefore, hepatic injury or hyperlipidemia may induce metabolic turbulence and negatively affect retinal health. Maintaining blood and liver health is beneficial to the retina.
4.1. Future Directions
Methods for the early detection of retinal dysfunction must be developed to identify the key treatment window and monitor disease progression
Simple, accessible self-screening is needed to address the problem of visual function and obtain a clinical examination
To explore the differences in the therapeutic effects of nonselective antioxidants, general ROS scavengers, specific inhibitors of Nox isoforms, and molecular/genomic target drugs are necessary to curb the progression of retinal diseases
Acknowledgments
The authors thank Editage Editing Service for editing the English text of a draft of this manuscript. This study was supported by the Key R&D and Promotion Projects of Henan Province No. 182102410099 and No. 182102310452.
Contributor Information
Jun Wang, Email: whb21c@vip.henu.edu.cn.
Dongdong Wu, Email: ddwubiomed2010@163.com.
Yalong Dang, Email: dangyalong@haust.edu.cn.
Conflicts of Interest
The authors report no conflicts of interest.
References
- 1.Hurley J. B., Lindsay K. J., Du J. Glucose, lactate, and shuttling of metabolites in vertebrate retinas. Journal of Neuroscience Research . 2015;93(7):1079–1092. doi: 10.1002/jnr.23583. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Sahnoun Z., Jamoussi K., Zeghal K. M. Free radicals and antioxidants: physiology, human pathology and therapeutic aspects (part II) Thérapie . 1998;53(4):315–339. [PubMed] [Google Scholar]
- 3.Chan T. C., Wilkinson B. J., Deliyanti D., et al. The role of reactive oxygen species in the pathogenesis and treatment of retinal diseases. Experimental Eye Research . 2020;201:p. 108255. doi: 10.1016/j.exer.2020.108255. [DOI] [PubMed] [Google Scholar]
- 4.Trends in prevalence of blindness and distance and near vision impairment over 30 years: an analysis for the Global Burden of Disease Study. The Lancet Global Health . 2021;9(2):e130–e143. doi: 10.1016/S2214-109X(20)30425-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Valko M., Rhodes C. J., Moncol J., Izakovic M., Mazur M. Free radicals, metals and antioxidants in oxidative stress-induced cancer. Chemico-Biological Interactions . 2006;160(1):1–40. doi: 10.1016/j.cbi.2005.12.009. [DOI] [PubMed] [Google Scholar]
- 6.Lushchak V. I. Free radicals, reactive oxygen species, oxidative stress and its classification. Chemico-Biological Interactions . 2014;224:164–175. doi: 10.1016/j.cbi.2014.10.016. [DOI] [PubMed] [Google Scholar]
- 7.Singh A., Kukreti R., Saso L., Kukreti S. Oxidative stress: a key modulator in neurodegenerative diseases. Molecules . 2019;24(8):p. 1583. doi: 10.3390/molecules24081583. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Sies H., Jones D. P. Reactive oxygen species (ROS) as pleiotropic physiological signalling agents. Nature Reviews. Molecular Cell Biology . 2020;21(7):363–383. doi: 10.1038/s41580-020-0230-3. [DOI] [PubMed] [Google Scholar]
- 9.Halliwell B. Antioxidants in human health and disease. Annual Review of Nutrition . 1996;16(1):33–50. doi: 10.1146/annurev.nu.16.070196.000341. [DOI] [PubMed] [Google Scholar]
- 10.Di Meo S., Venditti P. Evolution of the knowledge of free radicals and other oxidants. Oxidative Medicine and Cellular Longevity . 2020;2020:32. doi: 10.1155/2020/9829176.9829176 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Halliwell B. Biochemistry of oxidative stress. Biochemical Society Transactions . 2007;35(5):1147–1150. doi: 10.1042/BST0351147. [DOI] [PubMed] [Google Scholar]
- 12.Halliwell B. Reactive species and antioxidants. Redox biology is a fundamental theme of aerobic life. Plant Physiology . 2006;141(2):312–322. doi: 10.1104/pp.106.077073. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Halliwell B., Gutteridge J. M. The importance of free radicals and catalytic metal ions in human diseases. Molecular Aspects of Medicine . 1985;8(2):89–193. doi: 10.1016/0098-2997(85)90001-9. [DOI] [PubMed] [Google Scholar]
- 14.Amarowicz R., Pegg R. B. Protection of natural antioxidants against low-density lipoprotein oxidation. Advances in Food and Nutrition Research . 2020;93:251–291. doi: 10.1016/bs.afnr.2020.04.002. [DOI] [PubMed] [Google Scholar]
- 15.Zhang Q. J., Li T., Zhan H., Xin Y. M. Inhibitory effects of tea polyphenols and vitamin C on lipid peroxidation induced by FeSO4- cysteine in isolated human plasma and carbon tetrachloride-induced liver free radical injury in mice. Space Medicine & Medical Engineering . 2001;14(1):50–53. [PubMed] [Google Scholar]
- 16.Frei B. Ascorbic acid protects lipids in human plasma and low-density lipoprotein against oxidative damage. The American Journal of Clinical Nutrition . 1991;54(6):1113S–1118S. doi: 10.1093/ajcn/54.6.1113s. [DOI] [PubMed] [Google Scholar]
- 17.Rohowetz L. J., Kraus J. G., Koulen P. Reactive oxygen species-mediated damage of retinal neurons: drug development targets for therapies of chronic neurodegeneration of the retina. International Journal of Molecular Sciences . 2018;19(11):p. 3362. doi: 10.3390/ijms19113362. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Nita M., Grzybowski A. The role of the reactive oxygen species and oxidative stress in the pathomechanism of the age-related ocular diseases and other pathologies of the anterior and posterior eye segments in adults. Oxidative Medicine and Cellular Longevity . 2016;2016:23. doi: 10.1155/2016/3164734.3164734 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Domènech E. B., Marfany G. The relevance of oxidative stress in the pathogenesis and therapy of retinal dystrophies. Antioxidants . 2020;9(4):p. 347. doi: 10.3390/antiox9040347. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Pardue M. T., Allen R. S. Neuroprotective strategies for retinal disease. Progress in Retinal and Eye Research . 2018;65:50–76. doi: 10.1016/j.preteyeres.2018.02.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Campochiaro P. A., Mir T. A. The mechanism of cone cell death in retinitis pigmentosa. Progress in Retinal and Eye Research . 2018;62:24–37. doi: 10.1016/j.preteyeres.2017.08.004. [DOI] [PubMed] [Google Scholar]
- 22.Usui S., Oveson B. C., Lee S. Y., et al. NADPH oxidase plays a central role in cone cell death in retinitis pigmentosa. Journal of Neurochemistry . 2009;110(3):1028–1037. doi: 10.1111/j.1471-4159.2009.06195.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Murakami Y., Ikeda Y., Yoshida N., et al. MutT homolog-1 attenuates oxidative DNA damage and delays photoreceptor cell death in inherited retinal degeneration. The American Journal of Pathology . 2012;181(4):1378–1386. doi: 10.1016/j.ajpath.2012.06.026. [DOI] [PubMed] [Google Scholar]
- 24.Campochiaro P. A., Strauss R. W., Lu L., et al. Is there excess oxidative stress and damage in eyes of patients with retinitis pigmentosa? Antioxidants & Redox Signaling . 2015;23(7):643–648. doi: 10.1089/ars.2015.6327. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Usui S., Komeima K., Lee S. Y., et al. Increased expression of catalase and superoxide dismutase 2 reduces cone cell death in retinitis pigmentosa. Molecular Therapy . 2009;17(5):778–786. doi: 10.1038/mt.2009.47. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Komeima K., Rogers B. S., Campochiaro P. A. Antioxidants slow photoreceptor cell death in mouse models of retinitis pigmentosa. Journal of Cellular Physiology . 2007;213(3):809–815. doi: 10.1002/jcp.21152. [DOI] [PubMed] [Google Scholar]
- 27.Morizane Y., Morimoto N., Fujiwara A., et al. Incidence and causes of visual impairment in Japan: the first nation-wide complete enumeration survey of newly certified visually impaired individuals. Japanese Journal of Ophthalmology . 2019;63(1):26–33. doi: 10.1007/s10384-018-0623-4. [DOI] [PubMed] [Google Scholar]
- 28.Daiger S. P., Sullivan L. S., Bowne S. J. Genes and mutations causing retinitis pigmentosa. Clinical Genetics . 2013;84(2):132–141. doi: 10.1111/cge.12203. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Verbakel S. K., van Huet R., Boon C., et al. Non-syndromic retinitis pigmentosa. Progress in Retinal and Eye Research . 2018;66:157–186. doi: 10.1016/j.preteyeres.2018.03.005. [DOI] [PubMed] [Google Scholar]
- 30.Murakami Y., Nakabeppu Y., Sonoda K. H. Oxidative stress and microglial response in retinitis pigmentosa. International Journal of Molecular Sciences . 2020;21(19):p. 7170. doi: 10.3390/ijms21197170. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Campochiaro P. A., Iftikhar M., Hafiz G., et al. Oral N-acetylcysteine improves cone function in retinitis pigmentosa patients in phase I trial. The Journal of Clinical Investigation . 2020;130(3):1527–1541. doi: 10.1172/JCI132990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Oishi A., Noda K., Birtel J., et al. Effect of smoking on macular function and retinal structure in retinitis pigmentosa. Brain Communications . 2020;2(2, article a117) doi: 10.1093/braincomms/fcaa117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Meer A. V., Berger T., Muller F., Foldenauer A. C., Johnen S., Walter P. Establishment and characterization of a unilateral UV-induced photoreceptor degeneration model in the C57Bl/6J mouse. Translational Vision Science & Technology . 2020;9(9):p. 21. doi: 10.1167/tvst.9.9.21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Schwartz L., Boelle P. Y., D'Hermies F., Ledanois G., Virmont J. Blue light dose distribution and retinitis pigmentosa visual field defects: an hypothesis. Medical Hypotheses . 2003;60(5):644–649. doi: 10.1016/S0306-9877(02)00391-2. [DOI] [PubMed] [Google Scholar]
- 35.Kruk J., Kubasik-Kladna K., Aboul-Enein H. Y. The role oxidative stress in the pathogenesis of eye diseases: current status and a dual role of physical activity. Mini Reviews in Medicinal Chemistry . 2015;16(3):241–257. doi: 10.2174/1389557516666151120114605. [DOI] [PubMed] [Google Scholar]
- 36.Landers M. R. Retinal oxygenation via the choroidal circulation. Transactions of the American Ophthalmological Society . 1978;76:528–556. [PMC free article] [PubMed] [Google Scholar]
- 37.Wei Q., Hu W., Lou Q., Yu J. NAD+ inhibits the metabolic reprogramming of RPE cells in early AMD by upregulating mitophagy. Discovery Medicine . 2019;27(149):189–196. [PubMed] [Google Scholar]
- 38.Hernandez-Zimbron L. F., Zamora-Alvarado R., Ochoa-De L. P. L., et al. Age-related macular degeneration: new paradigms for treatment and management of AMD. Oxidative Medicine and Cellular Longevity . 2018;2018:14. doi: 10.1155/2018/8374647.8374647 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Abokyi S., To C. H., Lam T. T., Tse D. Y. Central role of oxidative stress in age-related macular degeneration: evidence from a review of the molecular mechanisms and animal models. Oxidative Medicine and Cellular Longevity . 2020;2020:19. doi: 10.1155/2020/7901270.7901270 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Nishimura Y., Hara H., Kondo M., Hong S., Matsugi T. Oxidative stress in retinal diseases. Oxidative Medicine and Cellular Longevity . 2017;2017:2. doi: 10.1155/2017/4076518.4076518 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Smith W., Assink J., Klein R., et al. Risk factors for age-related macular degeneration: pooled findings from three continents. Ophthalmology . 2001;108(4):697–704. doi: 10.1016/S0161-6420(00)00580-7. [DOI] [PubMed] [Google Scholar]
- 42.Payne A. J., Kaja S., Sabates N. R., Koulen P. A case for neuroprotection in ophthalmology: developments in translational research. Missouri Medicine . 2013;110(5):429–436. [PMC free article] [PubMed] [Google Scholar]
- 43.Payne A. J., Kaja S., Naumchuk Y., Kunjukunju N., Koulen P. Antioxidant drug therapy approaches for neuroprotection in chronic diseases of the retina. International Journal of Molecular Sciences . 2014;15(2):1865–1886. doi: 10.3390/ijms15021865. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Alge C. S., Priglinger S. G., Neubauer A. S., et al. Retinal pigment epithelium is protected against apoptosis by alphaB-crystallin. Investigative Ophthalmology & Visual Science . 2002;43(11):3575–3582. [PubMed] [Google Scholar]
- 45.Ye L., Yu T., Li Y., et al. Sulforaphane enhances the ability of human retinal pigment epithelial cell against oxidative stress, and its effect on gene expression profile evaluated by microarray analysis. Oxidative Medicine and Cellular Longevity . 2013;2013:13. doi: 10.1155/2013/413024.413024 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Matsuura T., Takayama K., Kaneko H., et al. Nutritional supplementation inhibits the increase in serum malondialdehyde in patients with wet age-related macular degeneration. Oxidative Medicine and Cellular Longevity . 2017;2017:9. doi: 10.1155/2017/9548767.9548767 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Crabb J. W., Miyagi M., Gu X., et al. Drusen proteome analysis: an approach to the etiology of age-related macular degeneration. Proceedings of the National Academy of Sciences of the United States of America . 2002;99(23):14682–14687. doi: 10.1073/pnas.222551899. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Meyer J. G., Garcia T. Y., Schilling B., Gibson B. W., Lamba D. A. Proteome and secretome dynamics of human retinal pigment epithelium in response to reactive oxygen species. Scientific Reports . 2019;9(1):p. 15440. doi: 10.1038/s41598-019-51777-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Velilla S., Garcia-Medina J. J., Garcia-Layana A., et al. Smoking and age-related macular degeneration: review and update. Journal of Ophthalmology . 2013;2013:11. doi: 10.1155/2013/895147.895147 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Wang A. L., Lukas T. J., Yuan M., Du N., Handa J. T., Neufeld A. H. Changes in retinal pigment epithelium related to cigarette smoke: possible relevance to smoking as a risk factor for age-related macular degeneration. PLoS One . 2009;4(4, article e5304) doi: 10.1371/journal.pone.0005304. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Jabbehdari S., Handa J. T. Oxidative stress as a therapeutic target for the prevention and treatment of early age-related macular degeneration. Survey of Ophthalmology . 2021;66(3):423–440. doi: 10.1016/j.survophthal.2020.09.002. [DOI] [PubMed] [Google Scholar]
- 52.Wu J., Seregard S., Algvere P. V. Photochemical damage of the retina. Survey of Ophthalmology . 2006;51(5):461–481. doi: 10.1016/j.survophthal.2006.06.009. [DOI] [PubMed] [Google Scholar]
- 53.Demontis G. C., Longoni B., Marchiafava P. L. Molecular steps involved in light-induced oxidative damage to retinal rods. Investigative Ophthalmology & Visual Science . 2002;43(7):2421–2427. [PubMed] [Google Scholar]
- 54.Wang J. J., Buitendijk G. H., Rochtchina E., et al. Genetic Susceptibility, Dietary Antioxidants, and Long-Term Incidence of Age- Related Macular Degeneration in Two Populations. Ophthalmology . 2014;121(3):667–675. doi: 10.1016/j.ophtha.2013.10.017. [DOI] [PubMed] [Google Scholar]
- 55.Ho L., van Leeuwen R., Witteman J. C., et al. Reducing the genetic risk of age-related macular degeneration with dietary antioxidants, zinc, and ω-3 fatty Acids. Archives of Ophthalmology . 2011;129(6):758–766. doi: 10.1001/archophthalmol.2011.141. [DOI] [PubMed] [Google Scholar]
- 56.Wong W. T., Kam W., Cunningham D., et al. Treatment of geographic atrophy by the topical administration of OT-551: results of a phase II clinical trial. Investigative Ophthalmology & Visual Science . 2010;51(12):6131–6139. doi: 10.1167/iovs.10-5637. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Chew E. Y., Clemons T. E., Agron E., et al. Long-Term Effects of Vitamins C and E, β-Carotene, and Zinc on Age-related Macular Degeneration: AREDS Report No. 35. Ophthalmology . 2013;120(8):1604–1611.e4. doi: 10.1016/j.ophtha.2013.01.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Amoaku W. M., Chakravarthy U., Gale R., et al. Defining response to anti-VEGF therapies in neovascular AMD. Eye (London, England) . 2015;29(6):721–731. doi: 10.1038/eye.2015.48. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Holekamp N. M. Review of neovascular age-related macular degeneration treatment options. The American Journal of Managed Care . 2019;25(10 Suppl):S172–S181. [PubMed] [Google Scholar]
- 60.Ortega A. L. Oxidative Stress in Diabetic Retinopathy. Antioxidants . 2021;10(1):p. 50. doi: 10.3390/antiox10010050. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Kang Q., Yang C. Oxidative stress and diabetic retinopathy: molecular mechanisms, pathogenetic role and therapeutic implications. Redox Biology . 2020;37:p. 101799. doi: 10.1016/j.redox.2020.101799. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Calderon G. D., Juarez O. H., Hernandez G. E., Punzo S. M., De la Cruz Z. D. Oxidative stress and diabetic retinopathy: development and treatment. Eye (London, England) . 2017;31(8):1122–1130. doi: 10.1038/eye.2017.64. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Wang M. H., Hsiao G., Al-Shabrawey M. Eicosanoids and oxidative stress in diabetic retinopathy. Antioxidants . 2020;9(6):p. 520. doi: 10.3390/antiox9060520. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Ahsan H. Diabetic retinopathy - Biomolecules and multiple pathophysiology. Diabetes and Metabolic Syndrome: Clinical Research and Reviews . 2015;9(1):51–54. doi: 10.1016/j.dsx.2014.09.011. [DOI] [PubMed] [Google Scholar]
- 65.Rodriguez M. L., Perez S., Mena-Molla S., Desco M. C., Ortega A. L. Oxidative stress and microvascular alterations in diabetic retinopathy: future therapies. Oxidative Medicine and Cellular Longevity . 2019;2019:18. doi: 10.1155/2019/4940825.4940825 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Du Y., Veenstra A., Palczewski K., Kern T. S. Photoreceptor cells are major contributors to diabetes-induced oxidative stress and local inflammation in the retina. Proceedings of the National Academy of Sciences of the United States of America . 2013;110(41):16586–16591. doi: 10.1073/pnas.1314575110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Bek T. Mitochondrial dysfunction and diabetic retinopathy. Mitochondrion . 2017;36:4–6. doi: 10.1016/j.mito.2016.07.011. [DOI] [PubMed] [Google Scholar]
- 68.Kowluru R. A. Diabetic retinopathy: mitochondrial dysfunction and retinal capillary cell death. Antioxidants & Redox Signaling . 2005;7(11-12):1581–1587. doi: 10.1089/ars.2005.7.1581. [DOI] [PubMed] [Google Scholar]
- 69.Barot M., Gokulgandhi M. R., Mitra A. K. Mitochondrial dysfunction in retinal diseases. Current Eye Research . 2011;36(12):1069–1077. doi: 10.3109/02713683.2011.607536. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Nebbioso M., Lambiase A., Armentano M., et al. Diabetic retinopathy, oxidative stress, and sirtuins: an in depth look in enzymatic patterns and new therapeutic horizons. Survey of Ophthalmology . 2022;67(1):168–183. doi: 10.1016/j.survophthal.2021.04.003. [DOI] [PubMed] [Google Scholar]
- 71.Robles-Rivera R. R., Castellanos-Gonzalez J. A., Olvera-Montano C., et al. Adjuvant therapies in diabetic retinopathy as an early approach to delay its progression: the importance of oxidative stress and inflammation. Oxidative Medicine and Cellular Longevity . 2020;2020:23. doi: 10.1155/2020/3096470.3096470 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Chen Q., Tang L., Xin G., et al. Oxidative stress mediated by lipid metabolism contributes to high glucose- induced senescence in retinal pigment epithelium. Free Radical Biology & Medicine . 2019;130:48–58. doi: 10.1016/j.freeradbiomed.2018.10.419. [DOI] [PubMed] [Google Scholar]
- 73.Gupta M. M., Chari S. Lipid peroxidation and antioxidant status in patients with diabetic retinopathy. Indian Journal of Physiology and Pharmacology . 2005;49(2):187–192. [PubMed] [Google Scholar]
- 74.Wang W., Lo A. Diabetic retinopathy: pathophysiology and treatments. International Journal of Molecular Sciences . 2018;19(6):p. 1816. doi: 10.3390/ijms19061816. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Zhang D., Lv F. L., Wang G. H. Effects of HIF-1α on diabetic retinopathy angiogenesis and VEGF expression. European Review for Medical and Pharmacological Sciences . 2018;22(16):5071–5076. doi: 10.26355/eurrev_201808_15699. [DOI] [PubMed] [Google Scholar]
- 76.Jiao W., Ji J., Li F., et al. Activation of the Notch‑Nox4‑reactive oxygen species signaling pathway induces cell death in high glucose‑treated human retinal endothelial cells. Molecular Medicine Reports . 2019;19(1):667–677. doi: 10.3892/mmr.2018.9637. [DOI] [PubMed] [Google Scholar]
- 77.Li J., Wang J. J., Yu Q., Chen K., Mahadev K., Zhang S. X. Inhibition of reactive oxygen species by lovastatin downregulates vascular endothelial growth factor expression and ameliorates blood-retinal barrier breakdown in db/db mice: role of NADPH oxidase 4. Diabetes . 2010;59(6):1528–1538. doi: 10.2337/db09-1057. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Appukuttan B., Ma Y., Stempel A., et al. Effect of NADPH oxidase 1 and 4 blockade in activated human retinal endothelial cells. Clinical & Experimental Ophthalmology . 2018;46(6):652–660. doi: 10.1111/ceo.13155. [DOI] [PubMed] [Google Scholar]
- 79.Laddha A. P., Kulkarni Y. A. Tannins and vascular complications of diabetes: an update. Phytomedicine . 2019;56:229–245. doi: 10.1016/j.phymed.2018.10.026. [DOI] [PubMed] [Google Scholar]
- 80.Mahmoud A. M., Abd E. S., Abdel-Reheim E. S. Consumption of polyphenol-rich Morus alba leaves extract attenuates early diabetic retinopathy: the underlying mechanism. European Journal of Nutrition . 2017;56(4):1671–1684. doi: 10.1007/s00394-016-1214-0. [DOI] [PubMed] [Google Scholar]
- 81.Kalita D., Holm D. G., LaBarbera D. V., Petrash J. M., Jayanty S. S. Inhibition of α-glucosidase, α-amylase, and aldose reductase by potato polyphenolic compounds. PLoS One . 2018;13(1, article e191025) doi: 10.1371/journal.pone.0191025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Palsamy P., Subramanian S. Resveratrol protects diabetic kidney by attenuating hyperglycemia-mediated oxidative stress and renal inflammatory cytokines via Nrf2-Keap1 signaling. Biochimica et Biophysica Acta . 2011;1812(7):719–731. doi: 10.1016/j.bbadis.2011.03.008. [DOI] [PubMed] [Google Scholar]
- 83.Bungau S., Abdel-Daim M. M., Tit D. M., et al. Health benefits of polyphenols and carotenoids in age-related eye diseases. Oxidative Medicine and Cellular Longevity . 2019;2019:22. doi: 10.1155/2019/9783429.9783429 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Xia N., Daiber A., Habermeier A., et al. Resveratrol reverses endothelial nitric-oxide synthase uncoupling in apolipoprotein E knockout mice. The Journal of Pharmacology and Experimental Therapeutics . 2010;335(1):149–154. doi: 10.1124/jpet.110.168724. [DOI] [PubMed] [Google Scholar]
- 85.Chalke S. D., Kale P. P. Combinational approaches targeting neurodegeneration, oxidative stress, and inflammation in the treatment of diabetic retinopathy. Current Drug Targets . 2021;22(16):1810–1824. doi: 10.2174/1389450122666210319113136. [DOI] [PubMed] [Google Scholar]
- 86.Deliyanti D., Alrashdi S. F., Tan S. M., et al. Nrf2 activation is a potential therapeutic approach to attenuate diabetic retinopathy. Investigative Ophthalmology & Visual Science . 2018;59(2):815–825. doi: 10.1167/iovs.17-22920. [DOI] [PubMed] [Google Scholar]
- 87.Liu Q., Zhang F., Zhang X., et al. Fenofibrate ameliorates diabetic retinopathy by modulating Nrf2 signaling and NLRP3 inflammasome activation. Molecular and Cellular Biochemistry . 2018;445(1-2):105–115. doi: 10.1007/s11010-017-3256-x. [DOI] [PubMed] [Google Scholar]
- 88.Alqawlaq S., Huzil J. T., Ivanova M. V., Foldvari M. Challenges in neuroprotective nanomedicine development: progress towards noninvasive gene therapy of glaucoma. Nanomedicine (London, England) . 2012;7(7):1067–1083. doi: 10.2217/nnm.12.69. [DOI] [PubMed] [Google Scholar]
- 89.Tham Y. C., Li X., Wong T. Y., Quigley H. A., Aung T., Cheng C. Y. Global prevalence of glaucoma and projections of glaucoma burden through 2040: a systematic review and meta-analysis. Ophthalmology . 2014;121(11):2081–2090. doi: 10.1016/j.ophtha.2014.05.013. [DOI] [PubMed] [Google Scholar]
- 90.Kolko M., Horwitz A., Thygesen J., Jeppesen J., Torp-Pedersen C. The prevalence and incidence of glaucoma in Denmark in a fifteen year period: a nationwide study. PLoS One . 2015;10(7, article e132048) doi: 10.1371/journal.pone.0132048. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Jayaram H. Intraocular pressure reduction in glaucoma: does every mmHg count? Taiwan Journal of Ophthalmology . 2020;10(4):255–258. doi: 10.4103/tjo.tjo_63_20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Fan G. J., Shah M. H., Chan E. C. Oxidative stress and the role of NADPH oxidase in glaucoma. Antioxidants . 2021;10(2):p. 238. doi: 10.3390/antiox10020238. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Kikuchi K., Dong Z., Shinmei Y., et al. Cytoprotective effect of astaxanthin in a model of normal intraocular pressure glaucoma. Journal of Ophthalmology . 2020;2020:6. doi: 10.1155/2020/9539681.9539681 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Izzotti A., Bagnis A., Sacca S. C. The role of oxidative stress in glaucoma. Mutation Research . 2006;612(2):105–114. doi: 10.1016/j.mrrev.2005.11.001. [DOI] [PubMed] [Google Scholar]
- 95.Seen S., Tong L. Dry eye disease and oxidative stress. Acta Ophthalmologica . 2018;96(4):e412–e420. doi: 10.1111/aos.13526. [DOI] [PubMed] [Google Scholar]
- 96.Sreekumar P. G., Hinton D. R., Kannan R. The emerging role of senescence in ocular disease. Oxidative Medicine and Cellular Longevity . 2020;2020:19. doi: 10.1155/2020/2583601.2583601 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Eells J. T. Mitochondrial dysfunction in the aging retina. Biology . 2019;8(2):p. 31. doi: 10.3390/biology8020031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Baudouin C., Kolko M., Melik-Parsadaniantz S., Messmer E. M. Inflammation in glaucoma: from the back to the front of the eye, and beyond. Progress in Retinal and Eye Research . 2021;83:p. 100916. doi: 10.1016/j.preteyeres.2020.100916. [DOI] [PubMed] [Google Scholar]
- 99.Shaw P. X., Stiles T., Douglas C., et al. Oxidative stress, innate immunity, and age-related macular degeneration. AIMS Molecular Science . 2016;3(2):196–221. doi: 10.3934/molsci.2016.2.196. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Golestaneh N., Chu Y., Cheng S. K., Cao H., Poliakov E., Berinstein D. M. Repressed SIRT1/PGC-1α pathway and mitochondrial disintegration in iPSC-derived RPE disease model of age-related macular degeneration. Journal of Translational Medicine . 2016;14(1):p. 344. doi: 10.1186/s12967-016-1101-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Zhao J., Wang S., Zhong W., Yang B., Sun L., Zheng Y. Oxidative stress in the trabecular meshwork (review) International Journal of Molecular Medicine . 2016;38(4):995–1002. doi: 10.3892/ijmm.2016.2714. [DOI] [PubMed] [Google Scholar]
- 102.Sacca S. C., Pascotto A., Camicione P., Capris P., Izzotti A. Oxidative DNA damage in the human trabecular Meshwork. Archives of Ophthalmology . 2005;123(4):458–463. doi: 10.1001/archopht.123.4.458. [DOI] [PubMed] [Google Scholar]
- 103.Izzotti A., Sacca S. C., Cartiglia C., De Flora S. Oxidative deoxyribonucleic acid damage in the eyes of glaucoma patients. The American Journal of Medicine . 2003;114(8):638–646. doi: 10.1016/S0002-9343(03)00114-1. [DOI] [PubMed] [Google Scholar]
- 104.Zhou L., Li Y., Yue B. Y. Oxidative stress affects cytoskeletal structure and cell-matrix interactions in cells from an ocular tissue: the trabecular meshwork. Journal of Cellular Physiology . 1999;180(2):182–189. doi: 10.1002/(SICI)1097-4652(199908)180:2<182::AID-JCP6>3.0.CO;2-X. [DOI] [PubMed] [Google Scholar]
- 105.Masihzadeh O., Ammar D. A., Lei T. C., Gibson E. A., Kahook M. Y. Real-time measurements of nicotinamide adenine dinucleotide in live human trabecular meshwork cells: effects of acute oxidative stress. Experimental Eye Research . 2011;93(3):316–320. doi: 10.1016/j.exer.2011.02.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Cook R., Thomas V., Martin R. Laser eye procedure is safe and effective as an early treatment for glaucoma. BMJ . 2019;366:p. l4235. doi: 10.1136/bmj.l4235. [DOI] [PubMed] [Google Scholar]
- 107.Hondur G., Göktas E., Yang X., et al. Oxidative stress-related molecular biomarker candidates for glaucoma. Investigative Ophthalmology & Visual Science . 2017;58(10):4078–4088. doi: 10.1167/iovs.17-22242. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Lanzi C., Lucarini L., Durante M., et al. Role of histamine H3 receptor antagonists on intraocular pressure reduction in rabbit models of transient ocular hypertension and glaucoma. International Journal of Molecular Sciences . 2019;20(4):p. 981. doi: 10.3390/ijms20040981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Liu Q., Ju W. K., Crowston J. G., et al. Oxidative stress is an early event in hydrostatic pressure induced retinal ganglion cell damage. Investigative Ophthalmology & Visual Science . 2007;48(10):4580–4589. doi: 10.1167/iovs.07-0170. [DOI] [PubMed] [Google Scholar]
- 110.Mahalingam K., Chaurasia A. K., Gowtham L., et al. Therapeutic potential of valproic acid in advanced glaucoma: a pilot study. Indian Journal of Ophthalmology . 2018;66(8):1104–1108. doi: 10.4103/ijo.IJO_108_18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Noro T., Namekata K., Azuchi Y., et al. Spermidine ameliorates neurodegeneration in a mouse model of normal tension glaucoma. Investigative Ophthalmology & Visual Science . 2015;56(8):5012–5019. doi: 10.1167/iovs.15-17142. [DOI] [PubMed] [Google Scholar]
- 112.Chou S. F., Luo L. J., Lai J. Y. In vivo Pharmacological Evaluations of Pilocarpine-Loaded Antioxidant- Functionalized Biodegradable Thermogels in Glaucomatous Rabbits. Scientific Reports . 2017;7(1):p. 42344. doi: 10.1038/srep42344. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Oikawa K., Ver H. J., Teixeira L., et al. Sub-region-specific optic nerve head glial activation in glaucoma. Molecular Neurobiology . 2020;57(6):2620–2638. doi: 10.1007/s12035-020-01910-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Sapienza A., Raveu A. L., Reboussin E., et al. Bilateral neuroinflammatory processes in visual pathways induced by unilateral ocular hypertension in the rat. Journal of Neuroinflammation . 2016;13(1):p. 44. doi: 10.1186/s12974-016-0509-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Cooper M. L., Crish S. D., Inman D. M., Horner P. J., Calkins D. J. Early astrocyte redistribution in the optic nerve precedes axonopathy in the DBA/2J mouse model of glaucoma. Experimental Eye Research . 2016;150:22–33. doi: 10.1016/j.exer.2015.11.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Murphy M. P. How mitochondria produce reactive oxygen species. The Biochemical Journal . 2009;417(1):1–13. doi: 10.1042/BJ20081386. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Williams P. A., Harder J. M., Foxworth N. E., et al. Vitamin B3modulates mitochondrial vulnerability and prevents glaucoma in aged mice. Science . 2017;355(6326):756–760. doi: 10.1126/science.aal0092. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Law S. M., Lu X., Yu F., Tseng V., Law S. K., Coleman A. L. Cigarette smoking and glaucoma in the United States population. Eye (London, England) . 2018;32(4):716–725. doi: 10.1038/eye.2017.292. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Fahmideh F., Marchesi N., Barbieri A., Govoni S., Pascale A. Non-drug interventions in glaucoma: putative roles for lifestyle, diet and nutritional supplements. Survey of Ophthalmology . 2022;67(3):675–696. doi: 10.1016/j.survophthal.2021.09.002. [DOI] [PubMed] [Google Scholar]
- 120.Perez C. I., Singh K., Lin S. Relationship of lifestyle, exercise, and nutrition with glaucoma. Current Opinion in Ophthalmology . 2019;30(2):82–88. doi: 10.1097/ICU.0000000000000553. [DOI] [PubMed] [Google Scholar]
- 121.Kim J., Aschard H., Kang J. H., et al. Intraocular pressure, glaucoma, and dietary caffeine consumption: a gene-diet interaction study from the UK biobank. Ophthalmology . 2021;128(6):866–876. doi: 10.1016/j.ophtha.2020.12.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Park J. H., Hwang J. H., Chang Y. S., Lee M. H., Park W. S. Survival rate dependent variations in retinopathy of prematurity treatment rates in very low birth weight infants. Scientific Reports . 2020;10(1):p. 19401. doi: 10.1038/s41598-020-76472-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Li Q. P., Wang Z. H., Li Y. Y., et al. Retinopathy of prematurity screening in 2185 premature infants. Zhonghua Yan Ke Za Zhi . 2012;48(10):903–907. [PubMed] [Google Scholar]
- 124.Stone W. L., Shah D., Hollinger S. M. Retinopathy of prematurity: an oxidative stress neonatal disease. Frontiers in Bioscience . 2016;21(1):165–177. doi: 10.2741/4382. [DOI] [PubMed] [Google Scholar]
- 125.Froysaker T. The influence of constrictive pericarditis on the superior vena caval flow pattern. Scandinavian Journal of Thoracic and Cardiovascular Surgery . 1972;6(3):227–233. doi: 10.3109/14017437209134804. [DOI] [PubMed] [Google Scholar]
- 126.Smith L. E., Hard A. L., Hellstrom A. The biology of retinopathy of prematurity: how knowledge of pathogenesis guides treatment. Clinics in Perinatology . 2013;40(2):201–214. doi: 10.1016/j.clp.2013.02.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Saugstad O. D. Oxygen and retinopathy of prematurity. Journal of Perinatology . 2006;26(Suppl 1):S46–S50. doi: 10.1038/sj.jp.7211475. [DOI] [PubMed] [Google Scholar]
- 128.Good W. V., Hardy R. J., Dobson V., et al. The incidence and course of retinopathy of prematurity: findings from the early treatment for retinopathy of prematurity study. Pediatrics . 2005;116(1):15–23. doi: 10.1542/peds.2004-1413. [DOI] [PubMed] [Google Scholar]
- 129.Fleck B. W., Stenson B. J. Retinopathy of prematurity and the oxygen conundrum: lessons learned from recent randomized trials. Clinics in Perinatology . 2013;40(2):229–240. doi: 10.1016/j.clp.2013.02.010. [DOI] [PubMed] [Google Scholar]
- 130.Buonocore G., Perrone S., Longini M., et al. Oxidative stress in preterm neonates at birth and on the seventh day of life. Pediatric Research . 2002;52(1):46–49. doi: 10.1203/00006450-200207000-00010. [DOI] [PubMed] [Google Scholar]
- 131.Davis J. M., Auten R. L. Maturation of the antioxidant system and the effects on preterm birth. Seminars in Fetal & Neonatal Medicine . 2010;15(4):191–195. doi: 10.1016/j.siny.2010.04.001. [DOI] [PubMed] [Google Scholar]
- 132.Lee J. E. Low IGF-1 suppresses VEGF-survival signaling in retinal endothelial cells: direct correlation with clinical retinopathy of prematurity, by A. Hellström, C. Perruzzi, M. Ju, E. Engström, A Hård, J. Liu, K. Albertson-Wikland, B. Carlsson, A. Niklasson, L. Sjödell, D. LeRoith, D. Senger, and L. Smith. PNAS 98:5804-8, 2001. Survey of Ophthalmology . 2003;48(2):234–235. doi: 10.1073/pnas.101113998. [DOI] [PubMed] [Google Scholar]
- 133.Garg U., Jain A., Singla P., Beri S., Garg R., Saili A. Free radical status in retinopathy of prematurity. Indian Journal of Clinical Biochemistry . 2012;27(2):196–199. doi: 10.1007/s12291-011-0180-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Banjac L., Banjac G., Kotur-Stevuljevic J., et al. Pro-oxidants and antioxidants in retinopathy of prematurity. Acta Clinica Croatica . 2018;57(3):458–463. doi: 10.20471/acc.2018.57.03.08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Zhou T. E., Rivera J. C., Bhosle V. K., et al. Choroidal involution is associated with a progressive degeneration of the outer retinal function in a model of retinopathy of prematurity: early role for IL-1β. The American Journal of Pathology . 2016;186(12):3100–3116. doi: 10.1016/j.ajpath.2016.08.004. [DOI] [PubMed] [Google Scholar]
- 136.Vander J. F., McNamara J. A., Tasman W., Brown G. C. Revised indications for early treatment of retinopathy of prematurity. Archives of Ophthalmology . 2005;123(3):p. 406. doi: 10.1001/archopht.123.3.406-b. [DOI] [PubMed] [Google Scholar]
- 137.Singh J. K., Wymore E. M., Wagner B. D., et al. Relationship between severe bronchopulmonary dysplasia and severe retinopathy of prematurity in premature newborns. Journal of AAPOS . 2019;23(4):209.e1–209.e4. doi: 10.1016/j.jaapos.2019.02.008. [DOI] [PubMed] [Google Scholar]
- 138.Rodriguez S. H., Schechet S. A., Shapiro M. J., Blair M. P. Late visual outcomes in infants treated with primary bevacizumab for type 1 retinopathy of prematurity. Journal of AAPOS . 2020;24(3):149.e1–149.e5. doi: 10.1016/j.jaapos.2020.03.003. [DOI] [PubMed] [Google Scholar]
- 139.Hartnett M. E. Advances in understanding and management of retinopathy of prematurity. Survey of Ophthalmology . 2017;62(3):257–276. doi: 10.1016/j.survophthal.2016.12.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140.Kozlowska A., Szostak-Wegierek D. Flavonoids--food sources and health benefits. Roczniki Państwowego Zakładu Higieny . 2014;65(2):79–85. [PubMed] [Google Scholar]
- 141.Ross J. A., Kasum C. M. DIETARYFLAVONOIDS: bioavailability, metabolic effects, and safety. Annual Review of Nutrition . 2002;22(1):19–34. doi: 10.1146/annurev.nutr.22.111401.144957. [DOI] [PubMed] [Google Scholar]
- 142.Saito Y., Hasebe-Takenaka Y., Ueda T., et al. Effects of green tea fractions on oxygen-induced retinal neovascularization in the neonatal rat. Journal of Clinical Biochemistry and Nutrition . 2007;41(1):43–49. doi: 10.3164/jcbn.2007006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.Wang Y., Zhao L., Lu F., et al. Retinoprotective effects of bilberry anthocyanins via antioxidant, anti-inflammatory, and anti-apoptotic mechanisms in a visible light-induced retinal degeneration model in pigmented rabbits. Molecules . 2015;20(12):22395–22410. doi: 10.3390/molecules201219785. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144.Tsang J., Wolf S. A., Pompoes I. M., et al. Potential Effects of Nutraceuticals in Retinopathy of Prematurity. Lifestyles . 2021;11(2):p. 79. doi: 10.3390/life11020079. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Jo H., Jung S. H., Yim H. B., Lee S. J., Kang K. D. The effect of baicalin in a mouse model of retinopathy of prematurity. BMB Reports . 2015;48(5):271–276. doi: 10.5483/BMBRep.2015.48.5.131. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146.Park S. W., Cho C. S., Jun H. O., et al. Anti-angiogenic effect of luteolin on retinal neovascularization via blockade of reactive oxygen species production. Investigative Ophthalmology & Visual Science . 2012;53(12):7718–7726. doi: 10.1167/iovs.11-8790. [DOI] [PubMed] [Google Scholar]
- 147.Dani C., Lori I., Favelli F., et al. Lutein and zeaxanthin supplementation in preterm infants to prevent retinopathy of prematurity: a randomized controlled study. The Journal of Maternal-Fetal & Neonatal Medicine . 2012;25(5):523–527. doi: 10.3109/14767058.2011.629252. [DOI] [PubMed] [Google Scholar]
- 148.Hanson C., Lyden E., Furtado J., et al. Vitamin E status and associations in maternal-infant dyads in the Midwestern United States. Clinical Nutrition . 2019;38(2):934–939. doi: 10.1016/j.clnu.2018.02.003. [DOI] [PubMed] [Google Scholar]
- 149.Bastos M. S., Rolland S. A., Costa C. M., et al. Vitamin A and Pregnancy: A Narrative Review. Nutrients . 2019;11(3) doi: 10.3390/nu11030681. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 150.de Souza M. L., Mennitti L. V., de Rosso V. V., Pisani L. P. The role of vitamin a and its pro-vitamin carotenoids in fetal and neonatal programming: gaps in knowledge and metabolic pathways. Nutrition Reviews . 2021;79(1):76–87. doi: 10.1093/nutrit/nuaa075. [DOI] [PubMed] [Google Scholar]
- 151.Hellstrom A., Smith L. E., Dammann O. Retinopathy of prematurity. Lancet . 2013;382(9902):1445–1457. doi: 10.1016/S0140-6736(13)60178-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 152.Cayabyab R., Ramanathan R. Retinopathy of prematurity: therapeutic strategies based on pathophysiology. Neonatology . 2016;109(4):369–376. doi: 10.1159/000444901. [DOI] [PubMed] [Google Scholar]
- 153.Di Marco E., Jha J. C., Sharma A., Wilkinson-Berka J. L., Jandeleit-Dahm K. A., de Haan J. B. Are reactive oxygen species still the basis for diabetic complications? Clinical Science (London, England) . 2015;129(2):199–216. doi: 10.1042/CS20150093. [DOI] [PubMed] [Google Scholar]
- 154.Sacca S. C., Roszkowska A. M., Izzotti A. Environmental light and endogenous antioxidants as the main determinants of non-cancer ocular diseases. Mutation Research . 2013;752(2):153–171. doi: 10.1016/j.mrrev.2013.01.001. [DOI] [PubMed] [Google Scholar]
- 155.Finkel T., Holbrook N. J. Oxidants, oxidative stress and the biology of ageing. Nature . 2000;408(6809):239–247. doi: 10.1038/35041687. [DOI] [PubMed] [Google Scholar]
- 156.Sacca S. C., Gandolfi S., Bagnis A., et al. From DNA damage to functional changes of the trabecular meshwork in aging and glaucoma. Ageing Research Reviews . 2016;29:26–41. doi: 10.1016/j.arr.2016.05.012. [DOI] [PubMed] [Google Scholar]
- 157.Hunter J. J., Morgan J. I., Merigan W. H., Sliney D. H., Sparrow J. R., Williams D. R. The susceptibility of the retina to photochemical damage from visible light. Progress in Retinal and Eye Research . 2012;31(1):28–42. doi: 10.1016/j.preteyeres.2011.11.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 158.McGuinness M. B., Karahalios A., Simpson J. A., et al. Past physical activity and age-related macular degeneration: the Melbourne collaborative cohort study. The British Journal of Ophthalmology . 2016;100(10):1353–1358. doi: 10.1136/bjophthalmol-2015-307663. [DOI] [PubMed] [Google Scholar]
- 159.Loprinzi P. D. Association of accelerometer-assessed sedentary behavior with diabetic retinopathy in the United States. JAMA Ophthalmology . 2016;134(10):1197–1198. doi: 10.1001/jamaophthalmol.2016.2400. [DOI] [PubMed] [Google Scholar]
- 160.Chrysostomou V., Galic S., van Wijngaarden P., Trounce I. A., Steinberg G. R., Crowston J. G. Exercise reverses age-related vulnerability of the retina to injury by preventing complement-mediated synapse elimination via a BDNF-dependent pathway. Aging Cell . 2016;15(6):1082–1091. doi: 10.1111/acel.12512. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 161.Hanif A. M., Lawson E. C., Prunty M., et al. Neuroprotective effects of voluntary exercise in an inherited retinal degeneration mouse model. Investigative Ophthalmology & Visual Science . 2015;56(11):6839–6846. doi: 10.1167/iovs.15-16792. [DOI] [PMC free article] [PubMed] [Google Scholar]


