Abstract
Patients with acute myeloid leukemia with myelodysplasia-related changes (AML-MRC) are insensitive to conventional chemotherapy and, therefore, have a poor prognosis. To evaluate the clinical efficacy and safety of low-dose decitabine in combination with small-dose CAG regimen (D-CAG regimen) in treating AML-MRC, a total of 80 patients with newly diagnosed AML-MRC from September 2015 to January 2020 in our center were included in the study. Amongst these patients, 43 and 37 patients received two courses of the D-CAG and CAG regimens, respectively. The complete remission (CR) and complete remission with incomplete blood count recovery (CRi) rate in the D-CAG group was higher than that of the CAG group (62.2% vs. 48.8%, P = 0.013). Among patients with less than 9 months of myelodysplastic syndrome (MDS) history and poor karyotypes, the (CR+CRi) rate of the D-CAG group was statistically higher than that of the CAG group. Except for patients receiving hematopoietic cell transplantation, among patients with less than 9 months of MDS history, the D-CAG group showed a better probability of overall survival than the CAG group did. In conclusion, patients with AML-MRC may benefit from the D-CAG regimen as an induction therapy, especially patients with less than 9 months of MDS history or with poor karyotypes.
Keywords: Decitabine, CAG regimen, D-CAG regimen, AML-MRC
1. Introduction
Acute myeloid leukemia with myelodysplasia-related changes (AML-MRC) is a specific acute leukemia with morphological features of myelodysplasia, or occurring in patients with a prior history of a myelodysplastic syndrome (MDS) or myelodysplastic/myeloproliferative neoplasm (MDS/MPN), or with MDS related cytogenetic abnormalities[1]. Therefore, AML-MRC can be classified into one of the following subtypes: AML arising from previous MDS or MDS/MPN, AML with an MDS-related cytogenetic abnormality, and AML with multilineage dysplasia. Patients with AML-MRC have biological characteristics similar to those of patients with MDS with excessive blasts and are not responsive to conventional chemotherapy. No specific induction chemotherapy, other than that used for AML, is given in the current guidelines[2], [3], [4]. The efficacy of decitabine in MDS has been approved firstly and then the efficacy of hypomethylating agent (HMA; decitabine and azacitidine)when used alone in treating AML were reported later[5]. In 2006, J. S. Welch, et al. [6] reported that AML patients with an unfavorable-risk cytogenetic profile benefited more from a short course of decitabine compared to patients with an intermediate-risk or favorable-risk cytogenetic profile. The application of HMA alone in old patients achieved comparable complete remission (CR) rate to the induction chemotherapy (27% v.s. 39%)[7].The CAG regimen (cytarabine, aclarubicin, and G-CSF) has been widely used to treat patients with MDS-EB and AML-MRC in China and Japan, with complete remission (CR) rates of 57.9% and 45.7%, respectively[8]. In Chinese guidelines, the CAG regimen is recommended to treat MDS-EB and hypoplastic AML Chinese guidelines for the diagnosis and treatment of adult acute myeloid leukemia (not APL)[4]. Decitabine in combination with a small-dose CAG regimen (D-CAG regimen) is widely used in Asia, with a reported CR rate of 50–83% [9], [10], [11], [12]. Our previous retrospective study showed that patients with MDS-EB and AML-MRC achieved a higher CR rate after two courses of the D-CAG regimen than those undergoing the CAG regimen (81.0% vs. 52.4%, P = 0.05) [13]. However, most studies have focused on patients with relapsed and refractory AML or MDS. Furthermore, limited information is available regarding the efficacy of the D-CAG regimen for the treatment of AML-MRC. Thus, we aimed to evaluate the clinical efficacy and safety of the D-CAG regimen for the treatment of AML-MRC.
2. Materials and methods
2.1. Study design and patients
This study analyzed 80 patients with newly diagnosed AML-MRC who received the D-CAG or CAG regimens as induction therapies at Peking University People's Hospital between September 2015 and January 2020. Patients chose their initial therapeutic regimen (D-CAG or CAG regimens) according to their desire and financial capability. Patient follow-ups occurred through our outpatient clinic, hospital medical records, and telephone (Fig. 1).
Fig. 1.
The flowchart of study design.
This study was approved by the ethics committee of Peking University People's Hospital (ChiCTR1800019603) in accordance with the Declaration of Helsinki. All data were collected following written informed consent from the patients or their relatives in accordance with the centers’ ethical research guidelines.
2.2. Treatment regimens
Patients in the CAG regimen group were initially treated with G-CSF (Kyowa Kirin China Pharma) for priming, in combination with cytarabine (10 mg/m2 q12h for 14 days, Pfizer Inc.) and aclarubicin (20 mg/day for 4 days, Aosaikang Pharma). Patients in the D-CAG regimen group were initially treated with 20 mg/m2 decitabine (ChiataiTianqing Pharma) for 5 days and a small-dose CAG regimen of 10 mg/m2 q12h cytarabine (Pfizer Inc.) for 7 days, 10 mg/day aclarubicin (Aosaikang Pharma) for 4 days, and G-CSF (Kyowa Kirin China Pharma) for priming. After induction chemotherapy, patients who did not achieve complete or partial remission (CR/PR) after the CAG regimen were transferred to the D-CAG regimen group. After two courses of the D-CAG regimen, patients who achieved CR received consolidation chemotherapy or hematopoietic stem cell transplantation (HSCT), and patients who did not respond to the treatment received other chemotherapies or salvaged HSCT.
2.3. Response criteria
Response criteria were determined according to the revised recommendations of the International Working Group for AML[14,15]. The overall response rate (ORR) was defined as involving those patients who achieved CR, CRi plus partial remission (PR). High-risk patients were identified according to the Southwest Oncology Group (SWOG) criteria. The durations of overall survival (OS) and leukemia-free survival (LFS) were both defined from the induction chemotherapy.
2.4. Safety assessments
Adverse events (AEs) that occurred throughout the treatment were recorded and graded according to the National Cancer Institute Common Toxicity Criteria (version 2). AEs that led to the discontinuation of treatment were also recorded. Hematological assessments and biochemical tests were conducted throughout the study.
2.5. Endpoints
The primary endpoint of the study was the (CR+CRi) rate. The secondary endpoints were the ORR and the probabilities of OS and LFS. Patient follow-ups occurred, as stated in Section 2.1, either until endpoints were met or up until November 2020.
2.6. Statistical analysis
Patient characteristics were compared using the χ2 test for categorical variables and the t-test for continuous variables. The (CR+CRi) rate and ORR were compared using the χ2 test. The probabilities of OS and LFS were estimated using the Kaplan-Meier method. All the variables shown in Table 1 were included in the univariate analysis. Furthermore, variables with p < 0.1 were included in a Cox proportional hazards model. Unless otherwise stated, a p-value ≤ 0.05 was considered significant. Statistical analyses were performed using the SPSS program (version 21.0, Mathsoft, Seattle, WA, USA).
Table 1.
Patient characteristics.
| DCAG group | CAG group | P value | |
|---|---|---|---|
| Number | 37 | 43 | |
| Age, years | 51(18–67) | 54(24–69) | 0.571 |
| Sex(Male/Female) | 25/12 | 22/21 | 0.137 |
| WBC | 2.46(0.81–28.23) | 1.90(0.51–66.41) | 0.593 |
| Hb | 77.5(50.0–122.0) | 72.0(52.0–122.0) | 0.095 |
| PLT | 52.0(6.0–309.0) | 51.0(5.0–300.0) | 0.946 |
| Risk stratification | 0.495 | ||
| Unavailable | 1(2.7%) | 0 | |
| Intermediate | 21(56.8%) | 20(46.5%) | |
| High | 15(40.5%) | 23(53.5%) | |
| MDS, AML-MRC history | 0.368 | ||
| ≤9months | 27(73.0%) | 35(81.4%) | |
| >9months | 10(27.0%) | 8(18.6%) | |
| AML-MRC subtypes | 0.334 | ||
| MDS-AML | 21(56.8%) | 21(48.8%) | |
| MDS related cytogenetic abnormality | 6(16.2%) | 13(30.2%) | |
| Multilineage dysplasia | 10(27.0%) | 9(20.9%) | |
| HSCT | 17(47.2%) | 23(53.5%) | 0.525 |
| Median follow-up (months) | 13.0(2.6–48.0) | 16.1(1.0–54.0) | 0.686 |
Abbreviations: WBC (white blood cells); Hb (hemoglobin); PLT (Platelet); HSCT (hematopoietic stem cell transplantation); MDS (myelodysplastic syndrome); AML-MRC (acute myeloid leukemia (AML) with myelodysplasia-related changes).
3. Results
3.1. Patient characteristics
This study included 80 newly diagnosed patients with AML-MRC who received the D-CAG or CAG regimens from September 2015 to January 2020 at the Peking University People's Hospital. Among these patients, 37 received the D-CAG regimen, and 43 received the CAG regimen. Table 1 summarizes the characteristics of the patients who received the regimens. The demographic and clinical characteristics of the patients at baseline were comparable between the two groups.
3.2. Response to regimens
After two courses of chemotherapy, 23 patients in the D-CAG group (23/37, 62.2%) and 22 patients in the CAG group (22/43, 51.2%) achieved CR and CRi respectively. The (CR+CRi) rate for the patients in the D-CAG group was higher than that for the CAG group after two courses of chemotherapy (62.2% vs. 48.8%, χ2 = 8.727, P = 0.013, bilateral). Amongst the seven patients who did not respond to the CAG regimen and then underwent the D-CAG regimen, six achieved CR and CRi followed by allogeneic HSCT.
Among the patients with less than 9 months of AML-MRC and/or MDS history, the (CR+CRi) rate (74.1%, 20/27) in the D-CAG group was significantly higher than the (CR+CRi) rate (42.9%, 15/35) in the CAG group (χ2 = 1.909, p = 0.008). However, no significant difference in MLSF rate between the two groups was observed among patients with more than 9 months of MDS history. According to the SWOG criteria, 38 patients were divided into the high-risk subgroup due to poor karyotypes. Compared to that of high-risk patients in the CAG group, high-risk patients in the D-CAG group achieved a significantly better (CR+CRi) rate (80.0% vs. 26.1%, χ2 = 11.392, P = 0.003, bilateral). (Table 2)
Table 2.
The MLFS rate of chemotherapy between the D-CAG group and the CAG group.
| Subgroup | No. | D-CAG | CAG | χ2 | P |
|---|---|---|---|---|---|
| All | 80 | n = 37 | n = 43 | ||
| 2nd DCAG/CAG (CR+CRi) rate |
23/37(62.2%) | 21/43(48.8%) | 8.727 | 0.013 | |
| PR rate | 5/37(13.5%) | 5/43(11.6%) | |||
| ORR | 28/37(75.7%) | 26/43(60.5%) | 2.097 | 0.148 | |
| ≤9 months | 62 | n = 27 | n = 35 | ||
| 2nd DCAG/CAG (CR+CRi) rate |
20/27 (74.1%) | 15/35(42.9%) | 1.909 | 0.008 | |
| PR rate | 3/27(11.1%) | 4/35(11.4%) | |||
| ORR | 23/27 (85.2%) | 19/35(54.3%) | 6.660 | 0.010 | |
| High risk(SWOG) | 38 | n = 15 | n = 23 | ||
| 2nd DCAG/CAG (CR+CRi) rate |
12/15(80.0%) | 6/23(26.1%) | 11.392 | 0.003 | |
| PR rate | 1/15(6.7%) | 4/23(17.4%) | |||
| ORR | 13/15(86.7%) | 10/23(43.5%) | 7.088 | 0.008 | |
| MDS-AML | 42 | n = 21 | n = 21 | ||
| 2nd DCAG/CAG (CR+CRi) rate |
13/21(61.9%) | 11/21(52.4%) | 3.233 | 0.199 | |
| PR rate | 4/21(19.1%) | 4/21(19.0%) | |||
| ORR | 17/21(81.0%) | 15/21(71.4%) | 0.525 | 0.469 | |
| MDS related cytogenetic abnormality | 19 | n = 6 | n = 13 | ||
| 2nd DCAG/CAG (CR+CRi) rate |
4/6(66.7%) | 3/13(23.1%) | 4.124 | 0.127 | |
| PR rate | 0/6(0%) | 1(7.7%) | |||
| ORR | 4/6(66.7%) | 4/13(30.8%) | 2.170 | 0.141 | |
| Multilineage dysplasia | 19 | n = 10 | n = 9 | ||
| 2nd DCAG/CAG (CR+CRi) rate |
6/10(60.0%) | 7/9(77.8%) | 6.041 | 0.049 | |
| PR rate | 1/10(10.0%) | 0(0%) | |||
| ORR | 7/10(70.0%) | 7/9(77.8%) | 0.148 | 0.701 |
Abbreviations:CR: complete remission; CRi: complete remission with incomplete blood count recovery; PR: partial remission; ORR: overall response rate.
The AML-MRC population was heterogeneous. Among the 42 AML-MRC patients with a history of MDS, 21 received the D-CAG regimen and 13 achieved CR and CRi (61.9%), while 21 received the CAG regimen and 11 achieved CR and CRi (52.4%). For the patients diagnosed with AML-MRC with MDS-related cytogenetic abnormalities, the (CR+CRi) rate in the D-CAG group was significantly higher than that in the CAG group (8/9, 88.9% vs. 5/13, 38.5%; p = 0.018). Among the 19 AML-MRC patients with multilineage dysplasia, six achieved CR and CRi out of the ten who received the D-CAG regimen (60%), while seven achieved CR and CRi out of the nine who received the CAG regimen (77.8%).
3.3. Overall survival
Up until November 2020, the median follow-up with patients in the D-CAG and CAG groups was 13.0 (95%CI:12.8–20.7) and 16.1 (95%CI:13.5–22.3) months, respectively. Seventeen patients in the D-CAG group received allo-HSCT, six died of the primary disease, and one died of transplant-related complications in the follow-up. In the CAG group, 23 patients received allo-HSCT, three died of transplant-related complications, and three died of the primary disease. Among the 40 patients that did not receive HSCT, the 2.5-year OS probability was comparable between the two groups (D-CAG: 40.4% ± 14.6%, CAG: 14.5% ± 12.1%, P = 0.109); however, the median survival time of the D-CAG group was higher than that of the CAG group (19.5 months vs. 9.3 months, p = 0.109).
However, among the patients with less than 9 months of MDS history, 29 did not undergo allo-HSCT, of which 12 were in the D-CAG group. For these patients, the 2.5-year OS probability in the D-CAG group was significantly higher than that in the CAG group (58.3% ± 18.6% vs. 11.3% ± 10.3%, p = 0.006, Fig. 2).
Fig. 2.
Comparison of survival rate between the D-CAG group and the CAG group.
(a) Comparison of survival rate of patients without receiving HSCT between the D-CAG group and the CAG group (b) Comparison of survival rate of patients with less than 9 months of MDS history without receiving HSCT between the D-CAG group and the CAG group.
The multivariate analysis showed that a longer history of MDS was associated with a worse OS, but HSCT was beneficial for improving the OS (Table 3).
Table 3.
Multivariate analysis with all variables.
| Covariate | Univariate analysis | Multivariate analysis | ||||
|---|---|---|---|---|---|---|
| HR | 95%CI | P value | HR | 95%CI | P value | |
| OS | ||||||
| Regimen | 0.840 | |||||
| Age | 1.028 | 1.000–1.056 | 0.051 | |||
| Sex | 2.867 | 1.289–6.377 | 0.010 | 2.637 | 1.173–5.932 | 0.019 |
| WBC | 0.877 | |||||
| Hb | 0.676 | |||||
| PLT | 0.385 | |||||
| Risk stratification | 2.402 | 1.195–4.831 | 0.014 | |||
| MDS/AML-MRC history | 1.007 | 1.003–1.011 | 0.001 | 1.006 | 1.002–1.010 | 0.007 |
| Subtypes | 0.062 | |||||
| HSCT | 0.378 | 0.187–0.767 | 0.007 | 0.368 | 0.179–0.754 | 0.006 |
Abbreviations:OS (overall survival); WBC (white blood cells); Hb (hemoglobin); PLT (Platelet); HSCT (hematopoietic stem cell transplantation); HR (hazard ratio); CI ( confidence-interval); MDS (myelodysplastic syndrome); AML-MRC (acute myeloid leukemia (AML) with myelodysplasia-related changes).
3.4. Leukemia-free survival
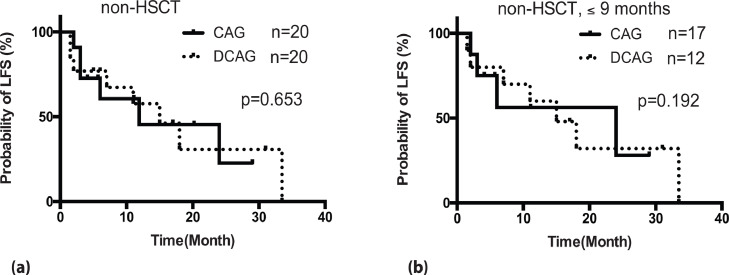
During the follow-up period, the 2.5-year LFS rates for the D-CAG and CAG group patients were 35.9%±9.3% and 32.5%±9.4%, respectively, without any significant difference (χ2 = 0.000, P = 0.995, bilateral). The median LFS for the patients was 11.0 months (95% CI 3.8–18.2) in the D-CAG group and 11.9 months (95% CI 0–28.2) in the CAG group. Among the 40 patients that did not receive HSCT, the 2.5-year LFS probability was comparable between the D-CAG and CAG groups (20.0% ± 11.1% vs. 12.5% ± 10.4%, P = 0.653). For the 29 non–HSCT patients with less than 9 months of MDS history, the 2.5-year LFS probability was also comparable between the D-CAG and CAG groups (26.7% ± 14.6% vs. 13.2% ± 11.0%, p = 0.192, Fig. 3)
Fig. 3.
Comparison of LFS between the D-CAG group and the CAG group.
(a) Comparison of LFS of patients without receiving HSCT between the D-CAG group and the CAG group (b) Comparison of LFS of patients with less than 9 months of MDS history without receiving HSCT between the D-CAG group and the CAG group.
3.5. Adverse events
The most frequent adverse event observed during the induction therapy was myelosuppression. Other common adverse events included pneumonia, skin and soft tissue infections, fever, hypotension, and impaired liver function. Among the 37 patients in the D-CAG group, 14 had pneumonia (37.8%), one had skin and soft tissue infections (2.7%), and 3 had fever during the neutropenia stage but without obvious infection (8.1%) after chemotherapy. Among the 43 patients in the CAG group, 10 (23.3%) had pneumonia, one (2.3%) had skin and soft tissue infections, 11 (25.6%) had fever during the neutropenia stage without infection, and one (2.3%) had septicemia. None of the patients discontinued treatment because of adverse events. There were no significant differences in the incidence of adverse events between the two regimen groups (P = 0.211). (see supplement)
4. Discussion
Our study compared the efficacy and safety of the D-CAG and CAG regimens for the treatment of AML-MRC. For all AML-MRC patients, the (CR+CRi) rate in the D-CAG group was higher than that in the CAG group after two courses but (62.2% v. vs. 48.8%, χ2 = 8.727, P = 0.013, bilateral). For the patients with less than 9 months of MDS history or the high-risk patients, the (CR+CRi) rate in the D-CAG group was significantly statistically higher than that in the CAG group. However, this difference was not observed across the entire study population. The probabilities of OS and LFS were similar between the two groups.
Patients with AML-MRC are unresponsive to conventional chemotherapy, and treatment still remains an issue. However, there are several rationales guiding us behind designing a D-CAG regimen for AML-MRC treatment. Decitabine, as a nucleoside analog, can inhibit DNA methyltransferase 1, which could lead to a significant loss of methyltransferase activity, demethylation of DNA, and intra-S-phase arrest of DNA replication. Decitabine has been approved for the treatment of all MDS subtypes[16,17], but limited reports are available on the efficacy of decitabine alone for the treatment of AML. G-CSF priming induces leukemia cell division and causes leukemia cells to respond to decitabine. The CAG regimen is currently used as an AML-MRC treatment, but it achieved only a 57.9% CR rate in AML treatment and a 45.7% CR rate in MDS-RAEB treatment[8]. In recent years, decitabine in combination with a CAG regimen has been reported to be used for treating MDS and relapsed or refractory AML with an overall response rate of 50–83%[[10], [11], [12],18] in Asian countries. Our previous study reported that patients with MDS-EB and AML-MRC achieved a higher CR rate after treatment with D-CAG than they did after treatment with CAG (81.0% vs. 52.4%, P = 0.050)[12]. However, published studies have focused on relapsed or refractory AML, elderly AML patients, and MDS, and few have used the D-CAG regimen as an induction therapy in treating newly diagnosed AML-MRC.
Thus, we evaluated the efficacy and safety of the D-CAG regimen for the treatment of newly diagnosed AML-MRC. Our study showed that the D-CAG regimen achieved a 75.7% ORR and a 62.2% (CR+CRi) rate in AML-MRC treatment, which was significantly higher compared to a 60.5% ORR and a 48.8% (CR+CRi) rate with the CAG regimen. However, this difference was not observed across the entire study population, and the OS and LFS probabilities were similar between the two groups. Nonetheless, for patients with less than 9 months of MDS history or high-risk patients, the (CR+CRi) rate in the D-CAG group was significantly higher than that in the CAG group. This is in accordance with the study by Li et al.[18], in which 15 patients with complex karyotypes (defined as ≥3 abnormalities) who received the D-CAG regimen had an ORR of 92.9% and a CR rate of 78.6%. These studies suggest that D-CAG regimen as an induction therapy may be important for patients with poor karyotypes.
However, our study had some limitations. Some patients involved in our study who were diagnosed before 2018 chose their initial therapeutic regimen according to their desire and financial capability, while those diagnosed from 2018 were eligible for a prospective trial. This may have influenced the randomness of this study. Additionally, the sample size was not large enough, and more patients are required to be enrolled in the prospective study to provide more reliable conclusions.
5. Conclusions
Patients with AML-MRC have a poor prognosis and may benefit from the D-CAG regimen as an induction therapy, especially patients with less than 9 months of MDS history, or patients with poor karyotypes. However, the OS and LFS probabilities did not improve significantly after treatment with the D-CAG regimen. Therefore, this study highlights the need for further research to develop new or improve on existing treatment regimens for AML-MRC.
CRediT authorship contribution statement
Hao Jiang designed the study; Jing Liu, Xiao-hong Liu, and Hao Jiang collected data; Jing Liu and Hao Jiang analyzed the data; Jing Liu, Xiao-hong Liu, and Hao Jiang drafted the manuscript; all authors contributed to data interpretation, manuscript preparation, and approval of the final version.
Funding
This work was supported by the Peking Specific Clinic Project (no. Z181100001718126) and the Peking University People's Hospital Research and Development Fund (no. RDY2019-35).
Declaration of Competing Interest
All authors have no relevant financial or non-financial interests to disclose.
Acknowledgments
We thank all the faculty members, all patients and their families that participated in these studies.
Footnotes
Supplementary material associated with this article can be found, in the online version, at doi:10.1016/j.lrr.2022.100354.
Appendix. Supplementary materials
References
- 1.Miesner M, Haferlach C, Bacher U, Weiss T, Macijewski K, Kohlmann A, et al. Multilineage dysplasia (MLD) in acute myeloid leukemia (AML) correlates with MDS-related cytogenetic abnormalities and a prior history of MDS or MDS/MPN but has no independent prognostic relevance: a comparison of 408 cases classified as "AML not otherwise specified" (AML-NOS) or "AML with myelodysplasia-related changes" (AML-MRC) Blood. 2010;116:2742–2751. doi: 10.1182/blood-2010-04-279794. [DOI] [PubMed] [Google Scholar]
- 2.Granfeldt Ostgard LS, Medeiros BC, Sengelov H, Norgaard M, Andersen MK, Dufva IH, et al. Epidemiology and clinical significance of secondary and therapy-related acute myeloid leukemia: a national population-based cohort study. J. Clin. Oncol. 2015;33:3641–3649. doi: 10.1200/JCO.2014.60.0890. [DOI] [PubMed] [Google Scholar]
- 3.Grimwade D, Hills RK, Moorman AV, Walker H, Chatters S, Goldstone AH, et al. Refinement of cytogenetic classification in acute myeloid leukemia: determination of prognostic significance of rare recurring chromosomal abnormalities among 5876 younger adult patients treated in the United Kingdom Medical Research Council trials. Blood. 2010;116:354–365. doi: 10.1182/blood-2009-11-254441. [DOI] [PubMed] [Google Scholar]
- 4.Leukemia & Lymphoma Group CSoH Chinese Medical Association. Chinese guidelines for the diagnosis and treatment of adult acute myeloid leukemia (not APL) (2017) Chin. J. Hematol. 2017;38:171–182. doi: 10.3760/cma.j.issn.0253-2727.2017.03.001. [DOI] [Google Scholar]
- 5.Zeidan AM, Jayade S, Schmier J, Botteman M, Hassan A, Ruiters D, et al. Injectable hypomethylating agents for management of myelodysplastic syndromes: patients' perspectives on treatment. Clin. Lymphoma Myeloma Leuk. 2021 doi: 10.1016/j.clml.2021.09.009. [DOI] [PubMed] [Google Scholar]
- 6.Welch JS, Petti AA, Miller CA, Fronick CC, O'Laughlin M, Fulton RS, et al. TP53 and decitabine in acute myeloid leukemia and myelodysplastic syndromes. N. Engl. J. Med. 2016;375:2023–2036. doi: 10.1056/NEJMoa1605949. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Vachhani P, Al Yacoub R, Miller A, Zhang F, Cronin TL, Ontiveros EP, et al. Intensive chemotherapy vs. hypomethylating agents in older adults with newly diagnosed high-risk acute myeloid leukemia: a single center experience. Leuk. Res. 2018;75:29–35. doi: 10.1016/j.leukres.2018.10.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Wei G, Ni W, Chiao JW, Cai Z, Huang H, Liu D. A meta-analysis of CAG (cytarabine, aclarubicin, G-CSF) regimen for the treatment of 1029 patients with acute myeloid leukemia and myelodysplastic syndrome. J. Hematol. Oncol. 2011;4:46. doi: 10.1186/1756-8722-4-46. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Y-p Zhang, W-z Wu, G-x Cui. Comparison of clinical efficacy between decitabine combined with CAG regimen and CAG regimen alone in patients with intermediate to high-risk myelodysplastic syndromes. J. Exp. Hematol. 2014;22:1341–1344. doi: 10.7534/j.issn.1009-2137.2014.05.030. [DOI] [PubMed] [Google Scholar]
- 10.Wang Y, Li W, Chen S, Qiu H, Sun A, Wu D. Salvage chemotherapy with low-dose cytarabine and aclarubicin in combination with granulocyte colony-stimulating factor priming in patients with refractory or relapsed acute myeloid leukemia with translocation (8;21) Leuk. Res. 2011;35:604–607. doi: 10.1016/j.leukres.2010.11.003. [DOI] [PubMed] [Google Scholar]
- 11.Liu L, Qu Q, Jiao W, Zhang Y, Li X, Ding C, et al. Increasing aclarubicin dose in low-dose cytarabine and aclarubicin in combination with granulocyte colony-stimulating factor (CAG regimen) is efficacious as salvage chemotherapy for relapsed/refractory mixed-phenotype acute leukemia. Leuk. Res. 2015;39:805–811. doi: 10.1016/j.leukres.2015.04.006. [DOI] [PubMed] [Google Scholar]
- 12.Saito K, Nakamura Y, Aoyagi M, Waga K, Yamamoto K, Aoyagi A, et al. Low-dose cytarabine and aclarubicin in combination with granulocyte colony-stimulating factor (CAG regimen) for previously treated patients with relapsed or primary resistant acute myelogenous leukemia (AML) and previously untreated elderly patients with AML, secondary AML, and refractory anemia with excess blasts in transformation. Int. J. Hematol. 2000;71:238–244. [PubMed] [Google Scholar]
- 13.Liu J, Jia J, Gong L, Lu S, Zhu H, Huang X, et al. Efficacy and safety of decitabine in combination with G-CSF, low-dose cytarabine and aclarubicin in MDS-EB and AML-MRC. Chin. J. Hematol. 2018;39:734–738. doi: 10.3760/cma.j.issn.0253-2727.2018.09.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Cheson BD, Bennett JM, Kopecky KJ, Buchner T, Willman CL, Estey EH, et al. Revised recommendations of the International Working Group for Diagnosis, standardization of response criteria, treatment outcomes, and reporting standards for therapeutic trials in acute myeloid leukemia. J. Clin. Oncol. 2003;21:4642–4649. doi: 10.1200/JCO.2003.04.036. [DOI] [PubMed] [Google Scholar]
- 15.Dohner H, Estey E, Grimwade D, Amadori S, Appelbaum FR, Buchner T, et al. Diagnosis and management of AML in adults: 2017 ELN recommendations from an international expert panel. Blood. 2017;129:424–447. doi: 10.1182/blood-2016-08-733196. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Steensma DP, Baer MR, Slack JL, Buckstein R, Godley LA, Garcia-Manero G, et al. Multicenter study of decitabine administered daily for 5 days every 4 weeks to adults with myelodysplastic syndromes: the alternative dosing for outpatient treatment (ADOPT) trial. J. Clin. Oncol. 2009;27:3842–3848. doi: 10.1200/JCO.2008.19.6550. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Lubbert M, Suciu S, Baila L, Ruter BH, Platzbecker U, Giagounidis A, et al. Low-dose decitabine versus best supportive care in elderly patients with intermediate- or high-risk myelodysplastic syndrome (MDS) ineligible for intensive chemotherapy: final results of the randomized phase III study of the European Organisation for Research and Treatment of Cancer Leukemia Group and the German MDS Study Group. J. Clin. Oncol. 2011;29:1987–1996. doi: 10.1200/JCO.2010.30.9245. [DOI] [PubMed] [Google Scholar]
- 18.Li J, Chen Y, Zhu Y, Zhou J, Xu Y, Li Y, et al. Efficacy and safety of decitabine in combination with G-CSF, low-dose cytarabine and aclarubicin in newly diagnosed elderly patients with acute myeloid leukemia. Oncotarget. 2015;6:6448–6458. doi: 10.18632/oncotarget.3361. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.