Abstract
Root meristem is a reserve of undifferentiated cells which guide root development. To maintain root meristem identity and therefore continuous root growth, the rate of cell differentiation must coordinate with the rate of generation of new cells. The E2 promoter-binding factor a (E2Fa) has been shown to regulate root growth through controlling G1/S cell cycle transitions in Arabidopsis thaliana. Here, we found that NAC1, a member of the NAM/ATAF/CUC family of transcription factors, regulated root growth by directly repressing the transcription of E2Fa. Loss of NAC1 triggers an up-regulation of the E2Fa expression and causes a reduced meristem size and short-root phenotype, which are largely rescued by mutation of E2Fa. Further analysis showed that NAC1 was shown to regulate root meristem by controlling endopolyploidy levels in an E2Fa-dependent manner. This study provides evidence to show that NAC1 maintains root meristem size and root growth by directly repressing the transcription of E2Fa in Arabidopsis.
Keywords: root, meristem, NAC1, E2Fa, endoreduplication
1. Introduction
Plant meristem is a reserve of undifferentiated cells which guide postembryonic development [1,2,3,4]. In the root meristem of Arabidopsis, all root tissues originate from a stem cell niche which includes a small group of slowly dividing cells, quiescent center (QC), and the surrounding stem cells [5]. Stem cell daughters undergo a certain number of cell divisions until they reach the transition zone where divisions cease and cells start to differentiate [5,6]. To maintain root meristem and therefore continuous root growth, the rate of cell differentiation must coordinate with the rate of generation of new cells [1]. A delayed or accelerated transition to elongation causes either an increase or a decrease in root meristem, respectively [1,2].
The root meristem is located at the distal part of the growing root and continuously generates new cells [6]. Two sets of transcriptional routes have been shown to play a key role in the establishment and maintenance of root meristem activity through specifying the QC and stem cell identity [1]. One is the plant hormone auxin, which is transduced through AP2 transcription factors PLETHORAs (PLTs) to control root stem cell niche identity and root meristem activity [1,7]. The other one involves the GRAS family transcription factors SHORTROOT/SCARECROW (SHR/SCR) and their interacting proteins [8,9,10,11]. Mutating each of the key regulators causes the loss of the root QC and stem cell identity, eventually affecting root growth and development [1,12,13]. In addition, other plant hormones such as cytokinin (CK), gibberellin (GA), and brassinosteroids (BR) also play important roles in the maintenance of root meristem identity [14,15,16].
Eukaryotic E2 promoter-binding factors (E2Fs) are transcription factors that are major regulators of cell division, DNA repair, and cell differentiation [17,18]. Among the six E2F transcription factors (E2Fa, E2Fb, E2Fc, EDL1/E2Fe, EDL2/E2Fd, EDL3/E2Ff) in Arabidopsis [19], both E2Fa and E2Fb are transcriptional activators, while E2Fc acts as a repressor of the cell cycle [17,20,21]. The other three E2F proteins, repressing E2F-regulated reporter genes, are also functional repressors [18,22]. The E2Fa plays an important role in switching cell division to endoreduplication [23]. Overexpression of both E2Fa and its partner DIMERIZATION PARTNER (DPa) in Arabidopsis can stimulate cell proliferation and induce extra rounds of DNA replication, which result cell endoreduplication [17]. The E2Fa/DPa complex, controlling the expression of essential genes during the G1/S transition, can be dissociated by SUMOylation E3 METHYL METHANESULFONATE SENSITIVITY GENE21 (AtMMS21) [24]. In particular, the phenotype of defective root development in 35s::E2Fa-DPa transgenic seedlings is completely recovered when AtMMS21 is overexpressed [24]. Additionally, rapamycin (TOR) kinase is essential for the maintenance of mitotic activity by directly phosphorylating E2Fa and E2Fb in both the shoot and root apexes [25,26]. All these results suggest that E2Fa plays an important role in root growth and development. However, the underlying molecular mechanism regarding how E2Fa is finely regulated at the transcriptional level to ensure root meristem identity and root growth is not well understood.
NAC (NAM, ATAF1/2, CUC) domain proteins are unique to plants and comprise large gene families [27]. There are around 105 NAC members which are involved in various aspects of plant development [27,28]. NAC transcription factors are characterized by a highly conserved NAM DNA-binding domain in the N-terminal region, accompanied by diverse C-terminal domains [29]. During lateral root initiation, NAC1 acts as a transcription activator to mediate auxin signaling, and microRNA (miRNA) 164 guides the cleavage of endogenous NAC1 mRNA in an auxin-dependent manner [30,31,32]. Furthermore, the NAC1 proteins are ubiquitinated and degraded by the 26S proteasome in a SINAT5 E3 controlled manner [31]. Novel functions are described in that NAC1 is induced in response to wounding and functions in promotion of root rip emergence [33]. However, the mechanistic studies of NAC1 on the functions of plant root growth are still unknown.
In this study, we found that the NAC domain protein NAC1 plays an important role in root growth. NAC1 maintains root meristem size and therefore root growth through directly repressing the transcription of E2Fa. The stunted root growth and reduced root meristem size, which result from the decreased cell numbers in root meristem, were largely rescued by mutation of E2Fa in the nac1 mutant. This study not only expands our knowledge about the biological roles of NAC1, but it also improves our understanding of how the cell cycle regulator E2Fa is delicately regulated by NAC1 to maintain root meristem and root growth.
2. Results
2.1. NAC1 Is Required for Primary Root Growth
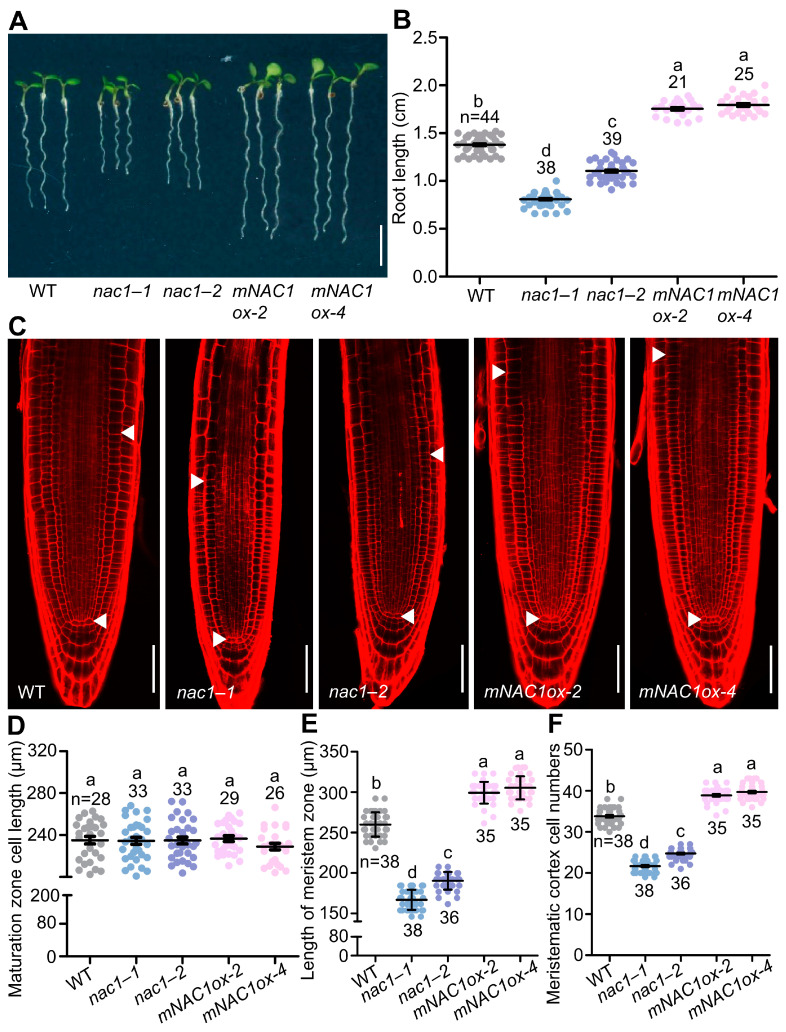
In contrast to the other members of the family whose expression is restricted to shoot meristem and flower (NAM, CUC2, NAP) or to vascular tissues (CmNACP), NAC1 is one NAC family member which is expressed in the root [32]. Although some expression can be detected in leaf primordia, wounding in the leaf explant, or in expanding cotyledons, the highest NAC1 expression is restricted to the lateral root initiation regions and root meristem [32,33]. The role of NAC1 in lateral root development has been highlighted by transducing auxin signals downstream of the F-box protein TIR1 [32], and whether NAC1 also plays a role in primary root growth was unknown. To test this hypothesis, two T-DNA insertion mutants, nac1–1 and nac1–2, were examined and showed defective root growth (Figure 1A). Considering that microRNAs (miRNAs) guised the cleavage of endogenous and transgenic NAC1 mRNA, we also generated overexpression transgenic plants expressing a cleavage-resistant form of NAC1 mRNA (mNAC1) according to the previous report [30].
Figure 1.
NAC1 is required for primary root growth. (A) The root growth phenotypes of WT, nac1–1, nac1–2, and two independent overexpression transgenic lines of mNAC1ox-2 or mNAC1ox-4 grown on 1/2 MS medium. The seedlings were photographed at 5 days post-germination (dpg). Scale bar represents 0.5 cm. (B) Measurements of primary root length of WT, nac1–1, nac1–2, and two independent overexpression transgenic lines as shown in (A). (C) Confocal images of root tips of WT, nac1–1, nac1–2, and two independent overexpression transgenic lines as shown in (A). The seedlings were used at 5 dpg for imaging. White arrows indicate the boundary between root meristem and transition zones. White arrows below indicate the quiescent center (QC). Scale bars represent 50 μm. (D) Root cortical cell length in the maturation zone of 5 dpg seedlings as shown in (C). (E) Length of meristem zone of 5 dpg seedlings as shown in (C). (F) Cell numbers in the proliferation domain of 5 dpg seedlings as shown in (C). Data information: in (B,D–F), data represent mean ± SD, n denotes the total number of scored samples. Individual values (black dots) are shown. Different lowercase letters indicate significant differences by one-way ANOVA followed by Tukey’s multiple comparison test (p < 0.05).
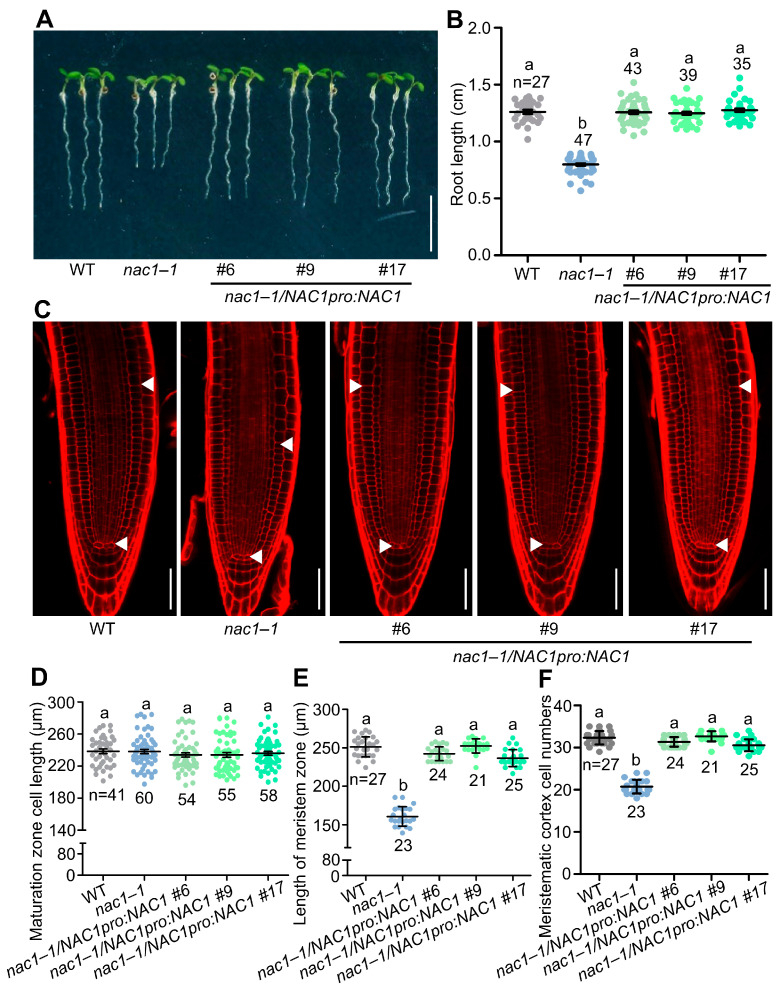
Compared to wild-type plants, both nac1 mutants and nac1–1/nac1–2 F1 generation mutants had shorter roots, whereas the mNAC1 overexpression lines displayed longer roots (Figure 1A,B and Figure S1A,B). Similarly, the size of the root apical meristem and meristem cell number were reduced in the nac1, nac1–1/nac1–2 F1 generation mutants and increased in the mNAC1 overexpressing lines, whereas the cortical cell length in the root maturation zone was unaltered compared to the wild type (Figure 1C–F and Figure S1C–F). Likewise, root length, root apical meristem size, and meristem cell number were completely or largely restored in nac1–1/NAC1pro:NAC1 compared to nac1–1 (Figure 2A–F), confirming that these phenotypes were caused by a mutation in NAC1.
Figure 2.
NAC1pro:NAC1 rescues the short-root-length phenotype in nac1–1. (A) The root growth phenotypes of WT, nac1–1, and three biological translational transgenic lines of NAC1pro:NAC1 in nac1–1 mutant background grown on 1/2 MS medium. The seedlings were photographed at 5 dpg. Scale bar represents 0.5 cm. (B) Measurements of primary root length of WT, nac1–1, and three biological translational transgenic lines of NAC1pro:NAC1 in nac1–1 mutant background as shown in (A). (C) Confocal images of root tips of WT, nac1–1, and three biological translational transgenic lines of NAC1pro:NAC1 in nac1–1 mutant background as shown in (A). The seedlings were used at 5 dpg for imaging. White arrows indicate the boundary between root meristem and transition zones. White arrows below indicate the QC. Scale bars represent 50 μm. (D) Root cortical cell length in the maturation zone of 5 dpg seedlings as shown in (C). (E) Length of meristem zone of 5 dpg seedlings as shown in (C). (F) Cell numbers in the proliferation domain of 5 dpg seedlings as shown in (C). Data information: in (B,D–F) data represent mean ± SD, n denotes the total number of scored samples. Individual values (black dots) are shown. Different lowercase letters indicate significant differences by one-way ANOVA followed by Tukey’s multiple comparison test (p < 0.05).
2.2. NAC1 Inhibits the Transcription of the Cell Cycle Regulator E2Fa
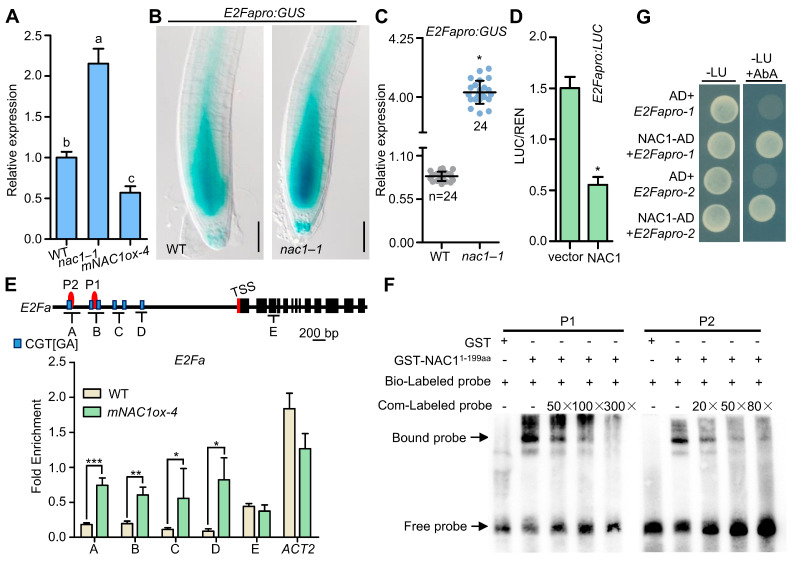
Considering the essential role of the E2Fa gene in controlling cell division and differentiation during the G1/S cell cycle [17,24,34], we first examined the expression of E2Fa by RT-qPCR since meristem cell numbers were decreased in the nac1 mutant (Figure 3A). We observed a higher transcript level of E2Fa in nac1–1, and a decrease in mNAC1ox-4 overexpression lines compared to wild type (Figure 3A). Consistently, the expression of E2Fapro:GUS, which is highly expressed in the meristem of wild-type roots, was dramatically accumulated in nac1–1 roots (Figure 3B,C). Furthermore, to examine whether the NAC1 affects the transcriptional activity of E2Fa in plants, we then used a transient transformation LUC reporter gene (E2Fapro:LUC) and performed transient expression assays in Arabidopsis leaf mesophyll protoplasts. The results showed that the LUC expression of the E2Fa promoter was specifically repressed by NAC1 compared with control (Figure 3D). Taken together, these results suggest that NAC1 inhibits the transcriptional activity of E2Fa.
Figure 3.
NAC1 directly binds to E2Fa promoter and represses its expression. (A) RT-qPCR analysis of E2Fa expression in WT, nac1–1, and mNAC1ox-4. Total RNA was extracted from primary root of 5 dpg seedlings. The expression level in seedlings was defined as “1”. Three biological replicates with three technical replicates for each biological replicate were performed with similar results. Data represent mean ± SE of three biological replicates. Different letters above bars indicate a significant difference (one-way ANOVA, Tukey’ multiple comparisons test, p < 0.05). (B) Expression of the E2Fapro:GUS in WT, nac1–1 roots. E2Fapro:GUS crossed with nac1–1 seedling. The seedlings were photographed at 5 dpg. Scale bar represents 50 μm. (C) Statistical analysis of E2Fapro:GUS expression in (B). Data represent mean ± SD, n denotes the total number of scored samples. Individual values (black dots) are shown (Student’s t test, * p < 0.05). (D) NAC1 transrepresses the E2Fa promoter in Arabidopsis leaf protoplasts. The effector (35s::NAC1) and reporter (E2Fapro:LUC) constructs. The empty vector pBI221 was used as a negative control. Three biological replicates with three technical replicates for each biological replicate were performed with similar results. Data represent mean ± SE of three biological replicates. * means differ significantly (p < 0.05) from the negative control. (E) Schematic diagram of the E2Fa and PCR amplicons (indicated as letters A–E) used for ChIP-qPCR. TSS, transcription start site. ChIP-qPCR results show the enrichment of NAC1 on the chromatin of E2Fa. Sonicated chromatins from 5 dpg seedlings (35s::MYC-mNAC1) were precipitated with anti-Myc antibodies. The precipitated DNA was used as a template for qPCR analysis, with primers targeting different regions of the E2Fa as shown in (E). Three biological replicates with three technical replicates for each biological replicate were performed with similar results. Data represent mean ± SE of three biological replicates (*** p < 0.001, ** p < 0.01, * p < 0.05, Student’s t test). (F) Electrophoretic mobility shift assay (EMSA) shows that NAC1 (1–199aa) binds the putative motif in the E2Fa promoter. The biotin-labeled probes (P1 and P2) are indicated in (E). Unlabeled probes were used in the competition assay. (G) Yeast one-hybrid binding assay involving NAC1 and E2Fa promoters. The yeast transformants were dropped onto SD-L-U (-Leu, -Ura) media. Aureobasidin A (AbA) concentration with 290 ng/mL. E2Fapro-1 spans from −1211 to −647 bp; E2Fapro-2 spans from −646 to −1 bp.
2.3. NAC1 Directly Binds to E2Fa Promoter
To further investigate whether NAC1 directly binds to the E2Fa promoter in vivo, we performed a chromatin immunoprecipitation (ChIP) followed by a quantitative real-time PCR assay (RT-qPCR) using NAC1 overexpression seedlings (mNAC1ox-4). It was previously reported that the DNA-binding specificity of NAC proteins contain the core CGT[GA] [29]. The global genomic DNA-binding site analysis revealed that the E2Fa promoter contains the predicated transcriptional sites of NAC proteins which marked A–D and E negative controls (Figure 3E). Compared with the wild-type seedlings, the A–D but not E fragments of the E2Fa promoter were highly enriched in the mNAC1ox-4 seedlings, indicating that the NAC1 proteins were associated with the genomic regions of the E2Fa promoter in vivo (Figure 3E). Furthermore, the electrophoretic mobility shift assays (EMSA) showed that NAC1 bound to the E2Fa ChIP-positive fragment (P1 and P2) specifically in vitro, and this interaction could be abolished by adding specific competitor probes (Figure 3F). Consistently, using yeast one-hybrid assays, we observed the interaction between NAC1 and the E2Fa promoter in yeast cells, suggesting that NAC1 directly binds to the E2Fa promoter (Figure 3G).
Considering that three Arabidopsis E2F proteins E2Fa, E2Fb, and E2Fc, but not other E2F factors, have been shown to interact with the retinoblastoma-related (RBR) proteins and play the same roles in G1/S transition [19,35,36], we also studied the relationship between NAC1 and E2Fb or E2Fc. Similarly, we performed transient expression assays in Arabidopsis leaf protoplasts and yeast one-hybrid assays to text whether NAC1 regulated E2Fb or E2Fc expression (Figure S2A–D). The results showed that NAC1 neither regulates the expression of E2Fb nor E2Fc, nor binds to their promoter directly in yeast one-hybrid assays, indicating NAC1 specifically inhibits E2Fa expression during root development.
2.4. E2Fa Acts Downstream of NAC1 to Control Primary Root Growth
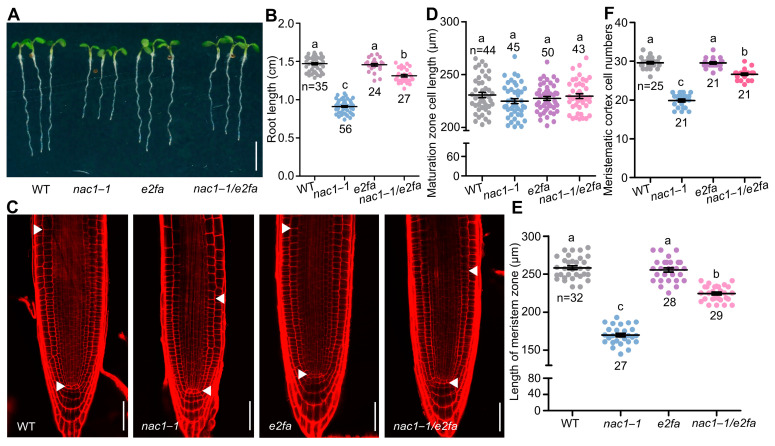
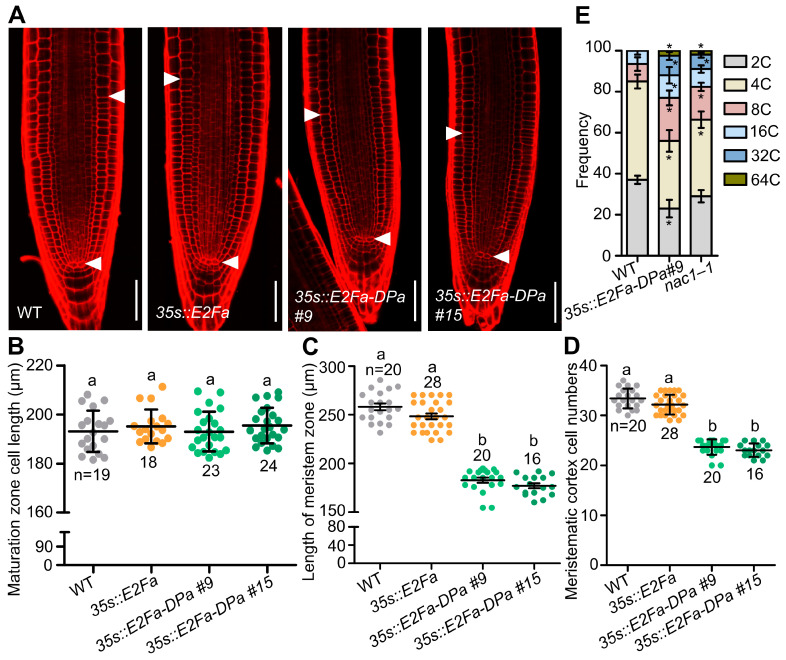
To further investigate the role of NAC1-mediated inhibition of E2Fa in cell division and root growth, we crossed an e2fa mutant with the nac1–1 mutant. We found that several phenotypes of the nac1–1 mutant were largely recovered in the nac1–1/e2fa double mutant, including the shorter root, shorter root apical meristem, and reduced meristem cell number (Figure 4A–F). Significantly, co-overexpression E2Fa and DPa lines displayed a shorter root meristem and reduced root length compared with the WT [24] (Figure 5A–D). We also examined root meristem cell numbers in co-overexpression E2Fa and DPa lines. As shown in Figure 5A–D, root meristem cell numbers were reduced in the 35s::E2Fa-DPa seedlings but not in the E2Fa overexpressing lines, implying that the reduced root meristem size and root growth resulted from the decreased root meristem cell numbers in nac1–1. Taken together, these results indicated that E2Fa acts as a downstream signaling component of NAC1 to maintain root meristem size and root growth.
Figure 4.
E2Fa acts downstream of NAC1 to control root growth. (A) The root growth phenotypes of WT, nac1–1, e2fa, and nac1–1/e2fa grown on 1/2 MS medium. The seedlings were photographed at 5 dpg. Scale bar represents 0.5 cm. (B) Measurements of primary root length of WT, nac1–1, e2fa, and nac1–1/e2fa as shown in (A). (C) Confocal images of root tips of WT, nac1–1, e2fa, and nac1–1/e2fa as shown in (A). The seedlings were used at 5 dpg for imaging. White arrows indicate the boundary between root meristem and transition zones. White arrows below indicate the QC. Scale bars represent 50 μm. (D) Root cortical cell length in the maturation zone of 5 dpg seedlings as shown in (C). (E) Length of meristem zone of 5 dpg seedlings as shown in (C). (F) Cell numbers in the proliferation domain of 5 dpg seedlings as shown in (C). Data information: in (B,D–F), data represent mean ± SD, n denotes the total number of scored samples. Individual values (black dots) are shown. Different lowercase letters indicate significant differences by one-way ANOVA followed by Tukey’s multiple comparison test (p < 0.05).
Figure 5.
Overexpression E2Fa-Dpa complex affects root length by promoting endoreduplication. (A) Confocal images of root tips of WT, 35s::E2Fa, and two independent overexpression transgenic lines of E2Fa-DPa. The seedlings were used at 5 dpg for imaging. White arrows indicate the boundary between root meristem and transition zones. Scale bars represent 50 μm. (B) Root cortical cell length in the maturation zone of 5 dpg seedlings as shown in (A). (C) Length of meristem zone of 5 dpg seedlings as shown in (A). (D) Cell numbers in the proliferation domain of 5 dpg seedlings as shown in (A). (E) Distribution of nuclear ploidy in the root meristem cells of WT, 35s::E2Fa-DPa #9, and nac1–1, using flow cytometry at 4 dpg. Two independent experiments were performed, and representative results are presented (Student’s t test, * p < 0.05). Data information: in (B–D), data represent mean ± SD, n denotes the total number of scored samples. Individual values (black dots) are shown. Different lowercase letters indicate significant differences by one-way ANOVA followed by Tukey’s multiple comparison test (p < 0.05).
The previous study showed that E2Fa-DPa was a key regulator of the endocycle [17]. To further study if the elevated E2Fa expression caused root growth arrest and might be a result of the regulation of endoreduplication in the nac1–1 mutant, we performed flow cytometric analysis in the WT, 35s::E2Fa-DPa, and nac1–1 plants. The results showed that, similar to the 35s::E2Fa-Dpa line, the 4 dpg seedling roots of nac1–1 mutants also showed higher endopolyploidy levels compared with the WT (Figure 5E). These findings indicate that endoreduplication was accelerated by the loss-of-function mutations of NAC1, which affected cell division and thus reduced meristem cells in the nac1 mutant.
3. Discussion
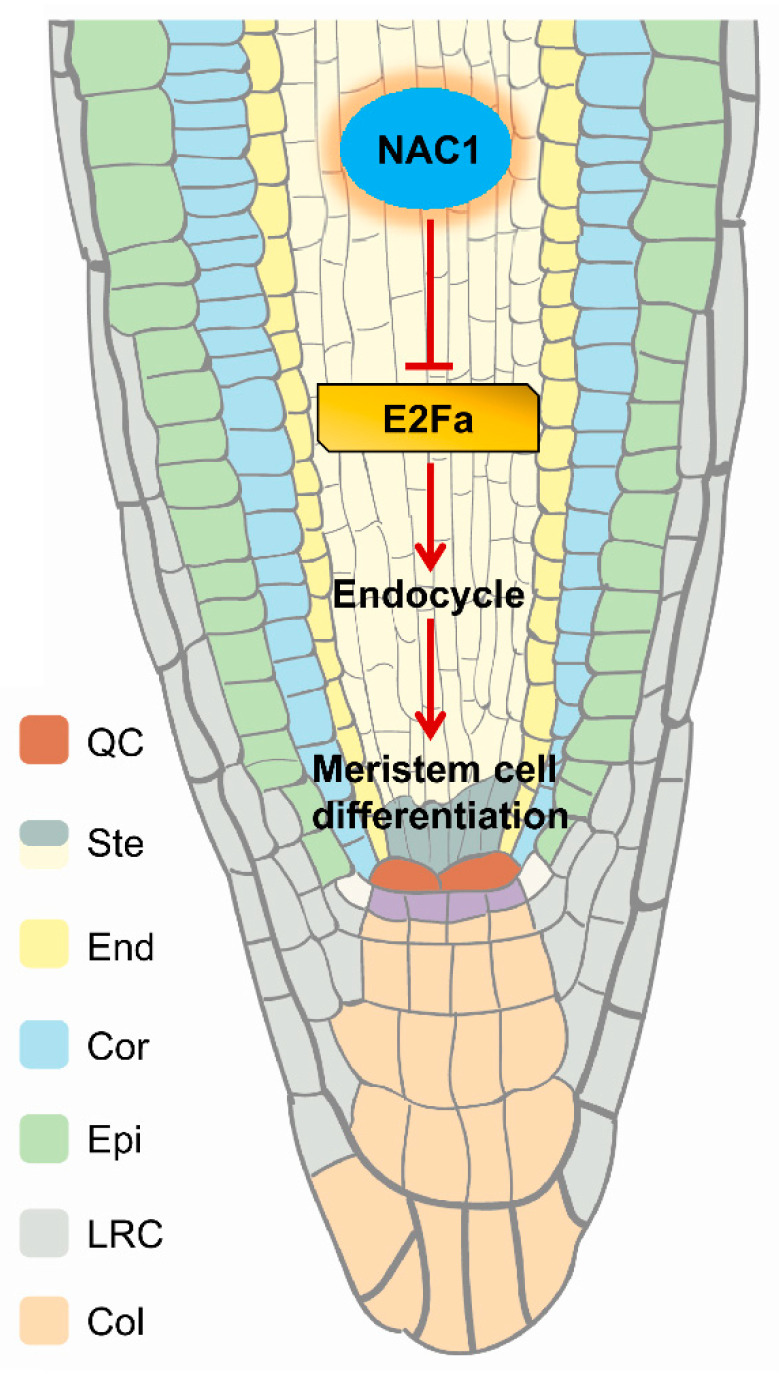
In Petunia, the NAC family gene NAM was expressed at the primordial and meristem boundaries, and the mutants failed to develop apical shoots [37]; Arabidopsis cuc1 cuc2 double mutants displayed highly affected shoot apical meristem development and also exhibited fused cotyledons, sepals, and stamens [38,39]. In contrast to the other members of the NAC family, whose expression is restricted to shoot meristem and flower (NAM, CUC2, NAP) or to vascular tissues (such as, CmNACP), as a member of NAC family, NAC1 is highly expressed in roots, and especially, the highest NAC1 expression is restricted to lateral root initiation regions and the root tip (meristem and elongation zone) [32]. Previously data identify NAC1 as a transcription activator in the auxin signaling pathway that regulates genes encoding molecules involved in the specification of lateral root formation [32]. During de novo root organogenesis in Arabidopsis, the NAC1 pathway functioned independently of auxin-mediated explant-specific wounding and root tip emergence [33]. However, the mechanism by which NAC1 regulates root meristem and root growth is unknown. Here, we provide evidence to show that NAC1 maintains root meristem and root growth through directly repressing E2Fa transcription, and loss of NAC1 triggers up-regulation of the E2Fa gene expression, which affects root meristem cell division through the regulation of the endocycle (Figure 6).
Figure 6.
Hypothetical model for the regulation of primary root development by NAC1. NAC1 plays a critical role in primary root growth by directly binding to the promoter and thus repressing the transcription of E2Fa. Loss of NAC1 triggers the up-regulation of E2Fa expression, which affects root meristem cell differentiation through the regulation of endocycle.
E2F/DP transcription factors are well-established targets of the universal CYC-CDK-RBR cascade and key regulators of S-phase genes governing cell cycle progression and DNA replication during postembryonic development, especially in root meristem cell divisions [23,40,41]. On the other hand, the E2Fa-DPa complex not only regulates the mitotic cell cycle progression but also plays a role in the endocycle [17,24]. A previous study showed that E2Fa-DPa was a key regulator of the endocycle [17], and overexpression of E2Fa and DPa in Arabidopsis showed a shorter primary root (Figure 5) [24], which is consistent with the elevated E2Fa expression that caused root growth arrest in the nac1–1 mutant (Figure 4). Furthermore, the transgenic lines overexpressing the NAC1 were bigger, with larger leaves, thicker stems, and more lateral roots compared with control plants [32]. Considering that endoreduplication is often associated with cell differentiation such as cell growth and organ enlargement, it is reasonable to speculate that the function of NAC1, as a negative regulator of E2Fa expression, in maintaining the root meristem size and root growth that are achieved through repressing E2Fa-mediated endoreduplication.
From all the results presented in this study, we put forward an NAC1-E2Fa signaling pathway that controls root meristem growth. However, there are still a couple of unknown questions. For instance, the hormone auxin plays a pivotal role in establishing the root proximodistal axis including meristem, acting as a local signaling factor [3,4], and the NAC1 gene is induced by auxin [31,32]. Interestingly, the relationship between the NAC1-E2Fa signaling pathway and auxin needs to be clarified in the future. On the other hand, NAC1 acts as a transcription activator in promoting lateral root development [32]. Unexpectedly, we found that NAC1 directly binds to the E2Fa promoter to repress its expression; therefore, it should be interesting to find out if NAC1 might regulate specific gene expression through transcriptional activation or repression that is dependent on its partner factors.
4. Materials and Methods
4.1. Plant Materials and Growth Conditions
In this study, all wild-type, mutant, and transgenic lines are in the Col-0 background; e2fa [25], E2Fapro:GUS [34], 35s::E2Fa, and 35s::E2Fa-DPa [24], as described previously. Mutant seed stocks used in this study are listed in Supplementary Materials. The nac1–1/e2fa double mutant was obtained by crossing of nac1–1 and e2fa. The nac1–1/nac1–2 F1 double mutant was obtained by crossing of nac1–1 and nac1–2. The nac1–1/E2Fapro:GUS transgenic line was obtained by crossing of nac1–1 and E2Fapro:GUS.
Plants were sowed on half-strength Murashige and Skoog medium, and then stratified at 4 °C for 2 days in the dark and then transferred to a phytotron set at 22 °C with a 16 h light/8 h dark photoperiod in vertically oriented Petri dishes. Roots were examined at 5 dpg.
4.2. PCR, RNA Extraction, and RT-qPCR
The promoter region and coding sequence of mNAC1 were amplified with Gateway compatible primers (Supplemental Table S1) [30]. The PCR products were first cloned to pEntry vector and then recombined with the binary vector pGWB18 (35s promoter, N-4 × Myc) to generate the 35s::Myc-mNAC1 (mNAC1ox) construct. For the NAC1pro:NAC1 construction, the NAC1 coding region or promoter regions were first cloned to the pEntry vector and then recombined with the binary vector pGWB1 (no promoter, no tag). All the constructs were transformed into Agrobacterium tumefaciens strain GV3101, which was used for transformation of Arabidopsis plants by the floral dip method. Transgenes were selected based on their resistance to hygromycin. Homozygous T3 transgenic plants were used for further experiments.
For RT-qPCR analysis, total RNA was extracted form 5 dpg roots using the RNeasy Plant Mini Kit (Qiagen, Hilden, Germany), and cDNA was prepared form 1 μg of total RNA with Hiscript III Reverse Transcriptase (Vazyme) [42,43]. The expression levels of target genes were normalized against ACT2. Primers are listed in the Supplementary Materials.
4.3. Expression Analysis and Microscopy
For histochemical β-glucuronidase [44] staining, seedlings were infiltrated with 100 mM sodium phosphate buffer (pH 7.2), 0.1% Triton X-100, 2 mM potassium fericyanide and potassium ferrocyanide, 10 mM EDTA, and 2 mM 5-bromo-4-chloro-3-indolyl-β-giucuronide (X-gluc), and incubated at 37 °C for overnight. Samples were cleared in chloral hydrate and visualized with Olympus BX53 microscopy. The process was performed according to Lv et al. [45].
4.4. Yeast One-Hybrid Assay
The full-length coding sequences of NAC1 were amplified with the primers listed in the table in Supplementary Materials and cloned into pGADT7 vector, and the promoter sequences of E2Fa, E2Fb, or E2Fc were cloned into the pAbAi vector. All the constructs used for testing the interactions were transformed into Y1Hgold. The presence of transgenes was confirmed by growth on SD-Ura-Leu plates. Protein interactions were assessed by dropping the yeast transformants on SD-Ura-Leu with Aureobasidin A (AbA) plates. Interactions were observed at 30 °C after 2 days of incubation.
4.5. Fluorescence Imaging and Quantification
Root meristems were imaged by a Zeiss LSM 900 laser scanning microscope with a 20× objective. For confocal laser scanning microscopy, root meristems were mounted in 10 μg/mL propidium iodide. The process was performed according to the method described by Tian et al. [44]. In addition, to determine the number of cells belonging to the root meristem, root meristematic cortex cells were counted in a file extending from the QC to the first elongated cell excluded [46]. We quantified root cortical cell length in the maturation zone which has root hairs using 20 to 50 cells from 15 to 20 roots for each genetic background with Image J. Image processing was performed with the LSM image-processing software (Zeiss, Jena, Germany). We determined statistical significance by Student’s t test or one-way ANOVA (Tukey’s multiple comparison tests).
4.6. EMSA
The GST-NAC1 (1–199 aa) (pGEX-4T-1) protein was expressed in E. coil BL21 (DE3). We grew BL21 cells at 37 °C in Luria–Bertani (LB) medium in the presence of antibiotics to an OD600 of 0.3 to 0.5. We induced protein accumulation by adding IPTG to a final concentration of 0.3 mM and purified with Glutathione Sepharose 4B (GE Healthcare, Chicago, IL, USA, 17-0756-01) according to the manufacturer’s instructions. EMSA was performed using the LightShift Chemiluminescent EMSA kit (Thermo Scientific, Waltham, MA, USA, 20148) according to the manufacturer’s instructions.
4.7. ChIP
ChIP was performed according to the Anne-Valérie Gendrel et al. [47] protocol. Seedlings at 5 dpg were harvested and crossed-linked with 4% formaldehyde under vacuum infiltration, then halted in 2M Gly. Immunoprecipitated chromatin was analyzed by qPCR. Enrichment was calculated as a ratio of bound sequence over input. The expression levels of target genes were normalized against ACT2. Primers are listed in Table S1.
4.8. Transient Expression
The NAC1 coding sequences were amplified, and the resulting sequences introduced into pBI221 to place them under the control of the CaMV 35s promoter. The E2Fa, E2Fb, and E2Fc promoter sequences were amplified and introduced into the pGreenII0800-LUC vector. Both recombinant plasmids were then transferred into Arabidopsis protoplasts. The process was performed according to the method described by Yoo et al. [48]. Primers are listed in the Table S1.
4.9. Nuclear Ploidy Analysis
For extraction of nuclei, 4 dpg meristem roots were quickly, finely chopped with a sharp razor blade in 1 mL buffer 1 (100 mM citric acid, 0.5% (v/v) polysorbate-20, pH 2–3). The process was performed according to the method described by Li et al. [49].
Acknowledgments
We thank Qi Xie (University of Chinese Academy of Sciences), Yan Xiong (Basic Forestry and Proteomics Research Centre, Haixia Institute of Science and Technology, Fujian Agriculture and Forestry University), and Yongxiu Liu (Institute of Botany, the Chinese Academy of Sciences) for sharing published materials.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/ijms232012258/s1.
Author Contributions
Conceived the study and designed the experiments, Z.D.; wrote the article, C.X. and Z.D. All authors have read and agreed to the published version of the manuscript.
Data Availability Statement
All data are available in the main text or the Supplementary Materials.
Conflicts of Interest
The authors declare no conflict of interests.
Funding Statement
This work is supported by the National Natural Science Foundation of China, grant numbers 32170338 and 32061143005, by the Youth Interdisciplinary Science and Innovative Research Groups of Shandong University, grant number 2020QNQT014, and by the Shandong Province Funds for Excellent Young Scholars, grant number ZR2020YQ19.
Footnotes
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Petricka J.J., Winter C.M., Benfey P.N. Control of Arabidopsis root development. Annu. Rev. Plant Biol. 2012;63:563–590. doi: 10.1146/annurev-arplant-042811-105501. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Sharma V.K., Carles C., Fletcher J.C. Maintenance of stem cell populations in plants. Proc. Natl. Acad. Sci. USA. 2003;100((Suppl. S1)):11823–11829. doi: 10.1073/pnas.1834206100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Leyser O. Auxin distribution and plant pattern formation: How many angels can dance on the point of PIN? Cell. 2005;121:819–822. doi: 10.1016/j.cell.2005.06.005. [DOI] [PubMed] [Google Scholar]
- 4.Sabatini S., Beis D., Wolkenfelt H., Murfett J., Guilfoyle T., Malamy J., Benfey P., Leyser O., Bechtold N., Weisbeek P., et al. An auxin-dependent distal organizer of pattern and polarity in the Arabidopsis root. Cell. 1999;99:463–472. doi: 10.1016/S0092-8674(00)81535-4. [DOI] [PubMed] [Google Scholar]
- 5.Aichinger E., Kornet N., Friedrich T., Laux T. Plant stem cell niches. Annu. Rev. Plant Biol. 2012;63:615–636. doi: 10.1146/annurev-arplant-042811-105555. [DOI] [PubMed] [Google Scholar]
- 6.Scheres B. Stem-cell niches: Nursery rhymes across kingdoms. Nat. Rev. Mol. Cell Biol. 2007;8:345–354. doi: 10.1038/nrm2164. [DOI] [PubMed] [Google Scholar]
- 7.Aida M., Beis D., Heidstra R., Willemsen V., Blilou I., Galinha C., Nussaume L., Noh Y.S., Amasino R., Scheres B. The PLETHORA genes mediate patterning of the Arabidopsis root stem cell niche. Cell. 2004;119:109–120. doi: 10.1016/j.cell.2004.09.018. [DOI] [PubMed] [Google Scholar]
- 8.Levesque M.P., Vernoux T., Busch W., Cui H., Wang J.Y., Blilou I., Hassan H., Nakajima K., Matsumoto N., Lohmann J.U., et al. Whole-genome analysis of the SHORT-ROOT developmental pathway in Arabidopsis. PLoS Biol. 2006;4:e143. doi: 10.1371/journal.pbio.0040143. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Sabatini S., Heidstra R., Wildwater M., Scheres B. SCARECROW is involved in positioning the stem cell niche in the Arabidopsis root meristem. Genes Dev. 2003;17:354–358. doi: 10.1101/gad.252503. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Sozzani R., Cui H., Moreno-Risueno M.A., Busch W., Van Norman J.M., Vernoux T., Brady S.M., Dewitte W., Murray J.A.H., Benfey P.N. Spatiotemporal regulation of cell-cycle genes by SHORTROOT links patterning and growth. Nature. 2010;466:128–132. doi: 10.1038/nature09143. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Welch D., Hassan H., Blilou I., Immink R., Heidstra R., Scheres B. Arabidopsis JACKDAW and MAGPIE zinc finger proteins delimit asymmetric cell division and stabilize tissue boundaries by restricting SHORT-ROOT action. Genes Dev. 2007;21:2196–2204. doi: 10.1101/gad.440307. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Heidstra R., Sabatini S. Plant and animal stem cells: Similar yet different. Nat. Rev. Mol. Cell Biol. 2014;15:301–312. doi: 10.1038/nrm3790. [DOI] [PubMed] [Google Scholar]
- 13.Shimotohno A., Heidstra R., Blilou I., Scheres B. Root stem cell niche organizer specification by molecular convergence of PLETHORA and SCARECROW transcription factor modules. Genes Dev. 2018;32:1085–1100. doi: 10.1101/gad.314096.118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Dello Ioio R., Linhares F.S., Scacchi E., Casamitjana-Martinez E., Heidstra R., Costantino P., Sabatini S. Cytokinins determine Arabidopsis root-meristem size by controlling cell differentiation. Curr. Biol. 2007;17:678–682. doi: 10.1016/j.cub.2007.02.047. [DOI] [PubMed] [Google Scholar]
- 15.Dello Ioio R., Nakamura K., Moubayidin L., Perilli S., Taniguchi M., Morita M.T., Aoyama T., Costantino P., Sabatini S. A genetic framework for the control of cell division and differentiation in the root meristem. Science. 2008;322:1380–1384. doi: 10.1126/science.1164147. [DOI] [PubMed] [Google Scholar]
- 16.Moubayidin L., Perilli S., Dello Ioio R., Di Mambro R., Costantino P., Sabatini S. The rate of cell differentiation controls the Arabidopsis root meristem growth phase. Curr. Biol. 2010;20:1138–1143. doi: 10.1016/j.cub.2010.05.035. [DOI] [PubMed] [Google Scholar]
- 17.De Veylder L., Beeckman T., Beemster G.T.S., de Almeida Engler J., Ormenese S., Maes S., Naudts M., Van Der Schueren E., Jacqmard A., Engler G., et al. Control of proliferation, endoreduplication and differentiation by the Arabidopsis E2Fa-DPa transcription factor. EMBO J. 2002;21:1360–1368. doi: 10.1093/emboj/21.6.1360. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Mariconti L., Pellegrini B., Cantoni R., Stevens R., Bergounioux C., Cella R., Albani D. The E2F Family of Transcription Factors from Arabidopsis thaliana: Novel and conserved components of the retinoblastoma/E2F pathway in plants. J. Biol. Chem. 2002;277:9911–9919. doi: 10.1074/jbc.M110616200. [DOI] [PubMed] [Google Scholar]
- 19.Gutierrez C. The Arabidopsis cell division cycle. Arab. Book. 2009;7:e0120. doi: 10.1199/tab.0120. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Del Pozo J.C., Boniotti M.B., Gutierrez C. Arabidopsis E2Fc functions in cell division and is degraded by the ubiquitin-SCF(AtSKP2) pathway in response to light. Plant Cell. 2002;14:3057–3071. doi: 10.1105/tpc.006791. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Sozzani R., Maggio C., Varotto S., Canova S., Bergounioux C., Albani D., Cella R. Interplay between Arabidopsis activating factors E2Fb and E2Fa in cell cycle progression and development. Plant Physiol. 2006;140:1355–1366. doi: 10.1104/pp.106.077990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Kosugi S., Ohashi Y. Interaction of the Arabidopsis E2F and DP proteins confers their concomitant nuclear translocation and transactivation. Plant Physiol. 2002;128:833–843. doi: 10.1104/pp.010642. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Magyar Z., Horváth B., Khan S., Mohammed B., Henriques R., De Veylder L., Bakó L., Scheres B., Bögre L. Arabidopsis E2FA stimulates proliferation and endocycle separately through RBR-bound and RBR-free complexes. EMBO J. 2012;31:1480–1493. doi: 10.1038/emboj.2012.13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Liu Y., Lai J., Yu M., Wang F., Zhang J., Jiang J., Hu H., Wu Q., Lu G., Xu P., et al. The Arabidopsis SUMO E3 Ligase AtMMS21 Dissociates the E2Fa/DPa Complex in Cell Cycle Regulation. Plant Cell. 2016;28:2225–2237. doi: 10.1105/tpc.16.00439. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Li X., Cai W., Liu Y., Li H., Fu L., Liu Z., Xu L. Differential TOR activation and cell proliferation in Arabidopsis root and shoot apexes. Proc. Natl. Acad. Sci. USA. 2017;114:2765–2770. doi: 10.1073/pnas.1618782114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Xiong Y., McCormack M., Li L., Hall Q., Xiang C., Sheen J. Glucose-TOR signalling reprograms the transcriptome and activates meristems. Nature. 2013;496:181–186. doi: 10.1038/nature12030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Kikuchi K., Ueguchi-Tanaka M., Yoshida K.T., Nagato Y., Matsusoka M., Hirano H.Y. Molecular analysis of the NAC gene family in rice. Mol. Gen. Genet. MGG. 2000;262:1047–1051. doi: 10.1007/PL00008647. [DOI] [PubMed] [Google Scholar]
- 28.Ooka H., Satoh K., Doi K., Nagata T., Otomo Y., Murakami K., Matsubara K., Osato N., Kawai J., Carninci P., et al. Comprehensive analysis of NAC family genes in Oryza sativa and Arabidopsis thaliana. DNA Res. Int. J. Rapid Publ. Rep. Genes Genomes. 2003;10:239–247. doi: 10.1093/dnares/10.6.239. [DOI] [PubMed] [Google Scholar]
- 29.Olsen A.N., Ernst H.A., Leggio L.L., Skriver K. DNA-binding specificity and molecular functions of NAC transcription factors. Plant Sci. 2005;169:785–797. doi: 10.1016/j.plantsci.2005.05.035. [DOI] [PubMed] [Google Scholar]
- 30.Guo H.-S., Xie Q., Fei J.-F., Chua N.-H. MicroRNA directs mRNA cleavage of the transcription factor NAC1 to downregulate auxin signals for arabidopsis lateral root development. Plant Cell. 2005;17:1376–1386. doi: 10.1105/tpc.105.030841. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Xie Q., Guo H.S., Dallman G., Fang S., Weissman A.M., Chua N.H. SINAT5 promotes ubiquitin-related degradation of NAC1 to attenuate auxin signals. Nature. 2002;419:167–170. doi: 10.1038/nature00998. [DOI] [PubMed] [Google Scholar]
- 32.Xie Q., Frugis G., Colgan D., Chua N.H. Arabidopsis NAC1 transduces auxin signal downstream of TIR1 to promote lateral root development. Genes Dev. 2000;14:3024–3036. doi: 10.1101/gad.852200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Chen X., Cheng J., Chen L., Zhang G., Huang H., Zhang Y., Xu L. Auxin-Independent NAC Pathway Acts in Response to Explant-Specific Wounding and Promotes Root Tip Emergence during de novo Root Organogenesis in Arabidopsis. Plant Physiol. 2016;170:2136–2145. doi: 10.1104/pp.15.01733. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Berckmans B., Vassileva V., Schmid S.P., Maes S., Parizot B., Naramoto S., Magyar Z., Alvim Kamei C.L., Koncz C., Bögre L., et al. Auxin-dependent cell cycle reactivation through transcriptional regulation of Arabidopsis E2Fa by lateral organ boundary proteins. Plant Cell. 2011;23:3671–3683. doi: 10.1105/tpc.111.088377. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Müller H., Helin K. The E2F transcription factors: Key regulators of cell proliferation. Biochim. Biophys. Acta. 2000;1470:M1–M12. doi: 10.1016/S0304-419X(99)00030-X. [DOI] [PubMed] [Google Scholar]
- 36.Vandepoele K., Vlieghe K., Florquin K., Hennig L., Beemster G.T., Gruissem W., Van de Peer Y., Inzé D., De Veylder L. Genome-wide identification of potential plant E2F target genes. Plant Physiol. 2005;139:316–328. doi: 10.1104/pp.105.066290. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Souer E., van Houwelingen A., Kloos D., Mol J., Koes R. The no apical meristem gene of Petunia is required for pattern formation in embryos and flowers and is expressed at meristem and primordia boundaries. Cell. 1996;85:159–170. doi: 10.1016/S0092-8674(00)81093-4. [DOI] [PubMed] [Google Scholar]
- 38.Aida M., Ishida T., Fukaki H., Fujisawa H., Tasaka M. Genes involved in organ separation in Arabidopsis: An analysis of the cup-shaped cotyledon mutant. Plant Cell. 1997;9:841–857. doi: 10.1105/tpc.9.6.841. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Aida M., Ishida T., Tasaka M. Shoot apical meristem and cotyledon formation during Arabidopsis embryogenesis: Interaction among the CUP-SHAPED COTYLEDON and SHOOT MERISTEMLESS genes. Development. 1999;126:1563–1570. doi: 10.1242/dev.126.8.1563. [DOI] [PubMed] [Google Scholar]
- 40.De Veylder L., Joubès J., Inzé D. Plant cell cycle transitions. Curr. Opin. Plant Biol. 2003;6:536–543. doi: 10.1016/j.pbi.2003.09.001. [DOI] [PubMed] [Google Scholar]
- 41.Satyanarayana A., Kaldis P. Mammalian cell-cycle regulation: Several Cdks, numerous cyclins and diverse compensatory mechanisms. Oncogene. 2009;28:2925–2939. doi: 10.1038/onc.2009.170. [DOI] [PubMed] [Google Scholar]
- 42.Zhang L., Yu Z., Xu Y., Yu M., Ren Y., Zhang S., Yang G., Huang J., Yan K., Zheng C., et al. Regulation of the stability and ABA import activity of NRT1.2/NPF4.6 by CEPR2-mediated phosphorylation in Arabidopsis. Mol. Plant. 2021;14:633–646. doi: 10.1016/j.molp.2021.01.009. [DOI] [PubMed] [Google Scholar]
- 43.Yu Z., Zhang D., Xu Y., Jin S., Zhang L., Zhang S., Yang G., Huang J., Yan K., Wu C., et al. CEPR2 phosphorylates and accelerates the degradation of PYR/PYLs in Arabidopsis. J. Exp. Bot. 2019;70:5457–5469. doi: 10.1093/jxb/erz302. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Tian H., Wabnik K., Niu T., Li H., Yu Q., Pollmann S., Vanneste S., Govaerts W., Rolcík J., Geisler M., et al. WOX5-IAA17 feedback circuit-mediated cellular auxin response is crucial for the patterning of root stem cell niches in Arabidopsis. Mol. Plant. 2014;7:277–289. doi: 10.1093/mp/sst118. [DOI] [PubMed] [Google Scholar]
- 45.Lv B., Wei K., Hu K., Tian T., Zhang F., Yu Z., Zhang D., Su Y., Sang Y., Zhang X., et al. MPK14-mediated auxin signaling controls lateral root development via ERF13-regulated very-long-chain fatty acid biosynthesis. Mol. Plant. 2021;14:285–297. doi: 10.1016/j.molp.2020.11.011. [DOI] [PubMed] [Google Scholar]
- 46.Di Mambro R., Sabatini S. Developmental Analysis of Arabidopsis Root Meristem. Methods Mol. Biol. 2018;1761:33–45. doi: 10.1007/978-1-4939-7747-5_3. [DOI] [PubMed] [Google Scholar]
- 47.Gendrel A.V., Lippman Z., Martienssen R., Colot V. Profiling histone modification patterns in plants using genomic tiling microarrays. Nat. Methods. 2005;2:213–218. doi: 10.1038/nmeth0305-213. [DOI] [PubMed] [Google Scholar]
- 48.Yoo S.D., Cho Y.H., Sheen J. Arabidopsis mesophyll protoplasts: A versatile cell system for transient gene expression analysis. Nat. Protoc. 2007;2:1565–1572. doi: 10.1038/nprot.2007.199. [DOI] [PubMed] [Google Scholar]
- 49.Li P.-C., Li K., Wang J., Zhao C.-Z., Zhao S.-Z., Hou L., Xia H., Ma C.-L., Wang X.-J. The AAA-ATPase MIDASIN 1 Functions in Ribosome Biogenesis and Is Essential for Embryo and Root Development. Plant Physiol. 2019;180:289–304. doi: 10.1104/pp.18.01225. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
All data are available in the main text or the Supplementary Materials.