Abstract
Although delayed-type hypersensitivity skin testing with tuberculin purified protein derivative (PPD) is the standard for tuberculosis screening, its variability suggests the need for a more sensitive, noninvasive test. An in vitro whole-blood assay has been proposed as an alternative. Using health care worker volunteers, we confirmed the correlation between PPD skin test (PPD-ST) results (positive, induration of >15 mm) and a standardized gamma interferon (IFN-γ) assay, QuantiFERON-TB (Q-IFN), manufactured by CSL Biosciences in Australia, and we evaluated Mycobacterium tuberculosis culture subfractions as potential substitutes for PPD. Twenty healthy volunteers with positive PPD-ST results and 20 PPD-ST-negative controls were enrolled. Whole blood was cultured with human PPD antigens (HuPPD), Mycobacterium avium complex (MAC) PPD, phytohemagglutinin (PHA), and four M. tuberculosis culture subfractions: low-molecular-weight culture, filtrate, culture filtrate without lipoarabinomannan, soluble cell wall proteins, and cytosolic proteins, all developed from M. tuberculosis strain H37RV. Secretion of IFN-γ (expressed as international units per milliliter) was measured by an enzyme immunoassay. The PPD or subculture fraction response as a percentage of the PHA response was used to determine positivity. Sixteen of 20 PPD-ST-positive individuals were classified as M. tuberculosis positive by Q-IFN, and 1 was classified as MAC positive. Sixteen of 20 PPD-ST-negative individuals were M. tuberculosis negative by Q-IFN, 2 were MAC positive, and 2 were M. tuberculosis positive. The tuberculosis culture subfractions stimulated IFN-γ production in PPD-ST-positive volunteers, and significant differences could be seen between the two PPD-ST groups with all subfractions except soluble cell wall protein; however, the response was variable and no better than the Q-IFN PPD. The agreement between the Q-IFN test and the PPD-ST was good (Cohen's kappa = 0.73). The Q-IFN assay can be a useful tool in further studies of immune responses to M. tuberculosis antigens.
The immune response to mycobacterial infection is predominantly cellular (5). Delayed-type hypersensitivity (DTH) skin testing has been a convenient, cost-effective method for assessing cell-mediated immune responses to a variety of antigens, starting with the mycobacterium-derived tuberculin purified protein derivative (PPD) over 100 years ago (28). Also known as the Mantoux test, this method has been the “gold standard” for diagnostic screening for detection of new or asymptomatic Mycobacterium tuberculosis infections. Although the test is reasonably priced, there continue to be multiple factors challenging the accuracy of the PPD skin test (PPD-ST) in different settings. These factors include (but are not limited to) special nursing skill requirements for placement and reading, variability in operator placement and reading, cross-reactivity among mycobacterial species (including M. avium and M. bovis BCG), the need for the patient to return in 48 to 72 h for a reading, and the modulation of the skin response due to underlying illness or immunosuppression (6, 28). Although the specificity of a significant positive test exceeds 95% in cattle, the sensitivity of the test in both animals and humans may be less than 75% (11, 31).
The immune response to M. tuberculosis is highly dependent upon gamma interferon (IFN-γ) production by macrophages and antigen-specific T cells (9). Over the past decade, there has been an increasing interest in the development and application of in vitro culture assays measuring IFN-γ production in response to tuberculin antigen stimulation as diagnostic screening tests substituting for the classic PPD (19). Although it initially used peripheral blood mononuclear cells (PBMC), the methodology evolved to a whole-blood culture technique that was first validated in Australian cattle (24, 31). The whole-blood culture technique requires less incubation time, is technically simpler, and provides a milieu closer to in vivo conditions. A standardized diagnostic kit with a specifically defined data analysis procedure, using human PPD, avian PPD, and the mitogen phytohemagglutinin (PHA), has been marketed by CDL Limited in Australia (QuantiFERON-TB or Q-IFN).
The purpose of this study is twofold: first, to assess the agreement between the Q-IFN assay and the Mantoux skin test (PPD) in classically PPD positive and PPD negative healthy volunteers; second, to evaluate five separate fractional extracts derived from M. tuberculosis cultures in the same Q-IFN assay system and determine concordance with the PPD-ST results.
MATERIALS AND METHODS
Participants and skin testing.
Forty-eight volunteers, all hospital employees, were recruited from the immunization clinic of a tertiary-care hospital in Washington, D.C. The ages of the participants ranged from 25 to 56 years, with a median age of 35. Prior to enrollment, all subjects were skin tested with 0.1 ml of 5-TU (tuberculin units) PPD placed intradermally according to the standard technique (Mantoux technique) (20). Depressed cellular immunity from any cause was ruled out using a panel of recall antigens that define the presence or absence of clinical anergy (13). The antigen panel included aqueous tetanus toxoid (1.6 Lf (limit of flocculation)/ml; Pasteur-Merieux-Connaught, Swiftwater, Pa.), candida (1:100; Hollister-Steir, Spokane, Wash.), and mumps (40 CFU/ml; Pasteur-Merieux-Connaught). Twenty of 28 subjects with a prior history of PPD-ST positivity (PPD-POS) were reproducibly positive at the time of enrollment. The eight with a positive history who tested PPD-ST negative at the time of enrollment were treated as a separate group (Prior-POS). None of these 28 patients had any prior history of BCG vaccination. The PPD-POS subjects were age and gender matched with 20 subjects who had both negative histories and negative PPD-ST results (PPD-NEG) at enrollment. All PPD-STs were placed and read by a certified nurse who performs these duties regularly. All readings were performed with the palpation and ballpoint methods along two axes of the forearm (J. E. Sokal, Editorial, N. Engl. J. Med. 293:501–502, 1975). A positive reading was defined as induration greater than 15 mm in diameter. None of the subjects had any known history of exposure, and all PPD-POS individuals had negative chest X-rays with normal complete blood counts and liver function tests. Informed consent was obtained from all subjects, and the study was approved by the local institutional review board.
Whole-blood culture.
Venous blood was collected from the participants in sodium heparinized tubes prior to intradermal skin testing. Whole-blood culture was performed by aliquoting 1 ml of blood in wells of a 24-well tissue culture plate (Costar) within 2 h after collection. The whole blood was stimulated with either sterile phosphate-buffered saline (Nil control antigen; 3 drops according to the Q-IFN kit instructions), a mitogen (positive control; PHA [3 drops]), human PPD (HuPPD; 3 drops), avian PPD (AvPPD; 3 drops), or an optimal concentration of a tuberculosis-specific antigen as described below. The tissue culture plates were incubated for 18 to 20 h (37°C, 5% CO2 2–95% air, 100% humidity). Plasma was harvested and stored for later quantification of IFN-γ by an enzyme immunoassay (EIA) provided with the Q-IFN kit.
Tuberculosis-specific antigens.
The subfractions were prepared in the Mycobacteria Research Laboratories, Department of Microbiology, Colorado State University. The following subcellular fractions of M. tuberculosis were studied in the whole blood assay: 1- to 10-kDa low-molecular-weight culture filtrate proteins (LMWCFP), whole culture filtrate proteins without lipoarabinomannan (CFP-LAM), soluble cell wall proteins (SCWP), cytosolic proteins (CYT), and M. tuberculosis strain H37Rv PPD. Antigens were prepared from M. tuberculosis strain H37Rv. Culture filtrate proteins were isolated from 14-day mid-log-phase cultures by filtration and concentration as described previously (29), and LAM was removed by partitioning with Triton X-114 (17). The LMWCFP was obtained by passing the sterile culture supernatant over a 10,000-molecular-weight cutoff membrane using an Amicon Beverly, Mass.) apparatus. The effluent (less than 10,000 Da) was collected. This material was concentrated using an Amicon apparatus with a 1,000-molecular-weight cutoff membrane. The concentrate was dialyzed against 10 mM ammonium bicarbonate, and the protein concentration was determined using the bicinchoninic acid (BCA) method (27). The SCWP and cytosol fractions were prepared as previously described (14, 18). The PPD preparation was produced following a standard protocol (25). Protein concentrations were determined by BCA assay (Pierce, Rockford, Ill.). The optimal cell culture subfraction concentrations determined by assaying multiple dilutions were 5 μg/ml for LMWCFP, 5 μg/ml for CFP-LAM, 1 μg/ml for SCWP, 5 μg/ml for CYT, and 10 μg/ml for H37Rv PPD.
IFN-γ EIA.
The EIA was generally performed according to the manufacturer's specifications. Briefly, 96-well plates precoated with an anti-IFN-γ monoclonal antibody were purchased. Each well was filled with 50 μl of anti-human IFN-γ–horseradish peroxidase conjugate and 50 μl of the test specimen. The plate contents were thoroughly mixed and incubated for 1 h at room temperature. The plates were washed for 6 cycles with 300 μl of wash buffer. A 100-μl portion of substrate was added to each well. The admixture was allowed to develop for 30 min (room temperature), at which time 50 μl of enzyme-stopping solution (1 N H2SO4) was added to halt the reaction. Absorbance was measured at 450 nm using a Molecular Devices (Sunnyvale, Calif.) plate reader.
A standard curve was generated by plotting the A450 from four known samples (provided with the Q-IFN kit) against their respective results in international units per milliliter. The IFN-γ values (in international units per milliliter of the unknown samples were determined from the standard curve. We developed our standard curve from the known replicates with a point-to-point methodology rather than the manufacturer's suggested linear best fit. This is described in more detail below. Tuberculin or M. avium complex (MAC) classifications were determined by calculating the following variables and using the criteria as outlined by the manufacturer;
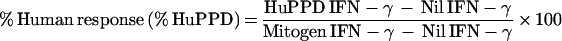 |
1 |
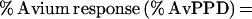 |
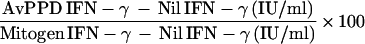 |
2 |
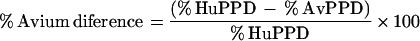 |
3 |
M. tuberculosis infection was defined as a percent HuPPD of >15% and a percent avium difference of >−10%. MAC infection was defined as a percent AvPPD of >20% and a percent avium difference of <−10%.
The percent in vitro response to the M. tuberculosis subfractions was calculated by normalizing the antigen-specific IFN-γ production to the mitogen response and calculating the following for each subfraction:
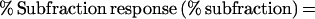 |
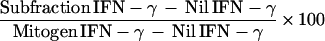 |
4 |
Curve fitting.
A linear fit through the standards is the manufacturer's recommended methodology for generating the EIA standard curve. The Q-IFN kit instructions imply the use of the entire curve without a lower limit cutoff. This often yielded large negative values (in international units per milliliter) for the plasma blank due to extrapolation at lower absorbance readings. The negative values affected the clinical interpretation rendered by the results of equations 1 to 4. Additionally, negative values for diluted samples could not be appropriately adjusted. Therefore, we chose to place our zero standard at the origin and draw the curve from one known replicate to the next (point to point). The values (in international units per milliliter for the unknown samples were determined using the linear portion between two known absorbance values (Microsoft Excel Trend function). The point-to-point curve not only eliminated the negative values but also compensated for any nonlinearity that may have existed in the standards. The two methodologies for generating the standard curve were evaluated with samples containing known quantities of IFN-γ. The point-to-point technique resulted in a more accurate result for determining the actual quantity of IFN-γ contained in the sample and yielded a 0-IU/ml measurement for the blank (see Table 1). As seen in Table 1, the absolute difference between the two methods may not appear to be large; however, the impact of the negative numbers on M. tuberculosis classifications was not insignificant. Also, since a negative IFN-γ value is not a biologic reality, we preferred to consider the blanks to be zero (or greater than zero if some background IFN-γ was observed). The final tuberculin reactivity classifications were determined using both methodologies, and the results were discordant (data not shown). We utilized the point-to-point technique throughout this study because it appears to be more accurate for determining IFN-γ levels while reflecting true biologic reactivity.
TABLE 1.
Comparison of calculated amounts of IFN-γ using the Q-IFN ELISA with two different methods of standard curve development
| Absorbance | Actual concn of IFN-γ (IU/ml) | IFN-γ concn (IU/ml) from the standard curve by the indicated method
|
|
|---|---|---|---|
| Point-to-point | Best linear fit | ||
| 3.531 | 137.2 | 149.3 | 142.6 |
| 2.134 | 69.6 | 75.2 | 80.5 |
| 1.346 | 34.8 | 38.6 | 45.5 |
| 0.49 | 8.4 | 7.9 | 7.5 |
| 0.358 | 4.2 | 4.2 | 1.6 |
| 0.148 | 0 | −0.1 | −7.7 |
Statistics.
Agreement between the PPD-ST and the Q-IFN assay was examined using Cohen's kappa. Kappa values of <0.4, 0.4 to 0.75, and >0.75 are consistent with poor, good, and excellent agreement, respectively (11). The association between the mean skin test size (mean diameter of two axes' induration) and the in vitro IFN-γ response (in international units per milliliter) (IU/ml) was examined using Spearman's rank correlation coefficient (rs). The in vitro percent subfraction responses to the tuberculin-specific subfractions for the PPD-ST-positive and -negative groups were compared using the Mann-Whitney rank sum test. Receiver operating characteristic (ROC) curves were generated for the subfractions which demonstrated a statistically significant difference in response between the two skin test groups. Cutoff points for interpreting in vitro tuberculin positivity and negativity, using tuberculin-specific subfraction stimulants, were determined from the ROCs.
RESULTS
Clinical classification using PPD.
Sixteen of 20 PPD-ST-positive individuals were classified as tuberculin reactors with the Q-IFN kit. Of the remaining four subjects, three were in vitro negative for tuberculosis and one met criteria for MAC. Sixteen of 20 PPD-ST-negative participants were in vitro negative (negative for M. tuberculosis as determined by the Q-IFN kit) for tuberculosis. Of the remaining four skin test-negative subjects, two were tuberculin reactive and two were MAC reactive. The agreement between the PPD-ST and the Q-IFN assay was very good, with a Cohen's kappa of 0.73. The eight Prior-POS patients were all classified as tuberculin positive with the Q-IFN kit (Table 2). The median percent HuPPD response for the PPD-POS subjects was 56.5%, compared to 7.9% for the control group.
TABLE 2.
Agreement between the in vitro measurement of IFN-γ with PPD stimulation using the Q-IFN kit and PPD-ST induration
| PPDST group (n) | In vitro QuantiFERON-TB resulta
|
Cohen's kappa | Median % HuPPD (interquartile range) | ||
|---|---|---|---|---|---|
| TB+ | TB− | MAC+ | |||
| Positive (20) | 16 | 3 | 1 | 56.5 (103.1) | |
| 0.73 | |||||
| Negative (20) | 2 | 16 | 2 | 7.9 (11.9) | |
| Prior positive, (8) now negative | 8 | 0 | 0 | 41.8 (57.5) | |
Plus sign, positive; minus sign, negative; TB, tuberculosis.
Induration versus IFN-γ values.
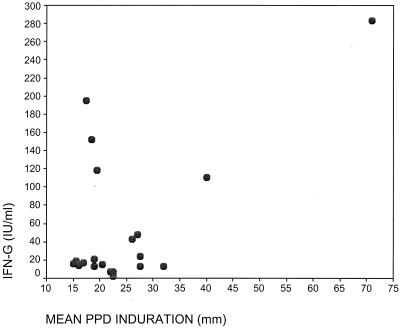
No statistically significant correlation was found between the mean skin test induration (as defined above) and the IFN-γ values of PPD reactors. The PPD reactors had skin test indurations ranging from 15 to 71 mm. The median value of their IFN-γ responses was 56.5 IU/ml, with an interquartile range of 50.2 IU/ml. Spearman's correlation coefficient for this comparison was 0.034, which denotes poor agreement (P = 0.887) (see Fig. 1).
FIG. 1.
PPD skin test induration versus IFN-γ levels of PPD-ST-positive subjects.
Purified M. tuberculosis antigens.
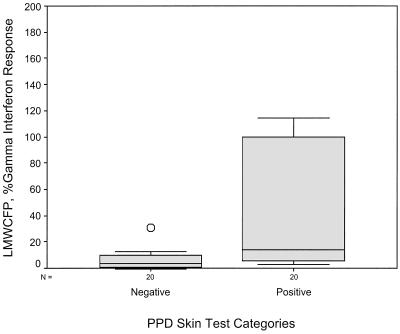
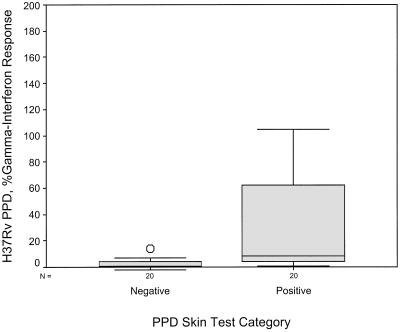
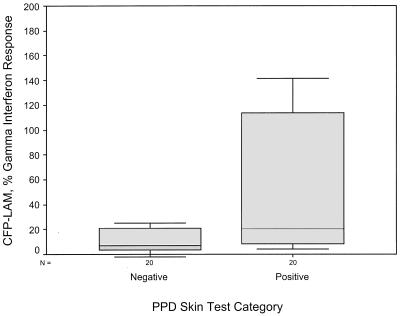
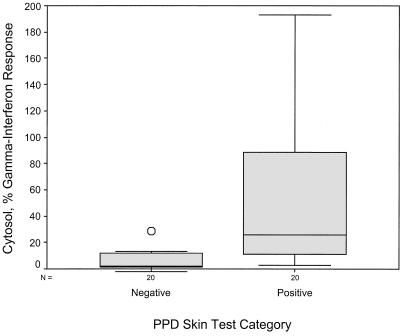
Using equation 4 above (percent subfraction response) to calculate IFN-γ response as a percentage of mitogen response, the mean levels with LMWCFP, CFP-LAM, CYT, and H37Rv PPD were much higher in the PPD-ST-positive group than the PPD-ST-negative group (see Table 3). These four subfractions were able to discriminate between the two skin test groups using the following cutoffs: 8% for LMWCFP, 16% for CFP-LAM, 8% for CYT, and 2% for H37RvPPD (Fig. 2 to 5). For these fractions the agreement (kappa coefficient) with the skin test was 0.45, 0.35, 0.55, and 0.6, respectively. Although the median percent SCWP responses with 1 μg/ml differed between the two skin test groups (8.7 versus 22.8%), the overlap in ranges resulted in no statistical difference (P = 0.076).
TABLE 3.
Agreement between the in vitro measurement of IFN-γ with M. tuberculosis subfraction stimulation using the Q-IFN kit and PPD-ST induration
| Tuberculin subfraction | PPD-ST resulta | Median % IFN-γ response | Interquartile range |
|---|---|---|---|
| LMWCFP | + | 13.6 | 101.0 |
| − | 3.6 | 10.0 | |
| CFP-LAM | + | 20.3 | 120.4 |
| − | 7.0 | 18.4 | |
| SCWP | + | 22.8 | 108.2 |
| − | 8.7 | 42.4 | |
| CYT | + | 25.6 | 82.9 |
| − | 2.0 | 10.6 | |
| H37Rv PPD | + | 8.2 | 74.0 |
| − | 0.6 | 4.8 |
n = 20 for both positive and negative PPD-ST groups. Plus, positive; minus, negative.
FIG. 2.
Comparison of the IFN-γ response to stimulation by LMWCFP in the two PPD-ST groups. The subfraction IFN-γ response is shown as a percentage of the mitogen response.
FIG. 5.
Comparison of the IFN-γ response to stimulation by H37Rv strain PPD in the two PPD-ST groups. The subfraction IFN-γ response is shown as a percentage of the mitogen response.
DISCUSSION
The increase in prevalence of tuberculosis and the emergence of multidrug-resistant strains have created a public health urgency for early identification of M. tuberculosis-infected individuals. The gold standard for detecting exposure remains the Mantoux test, which was developed in the late 1800s and is one of the oldest tests still in clinical use. Despite the long history of clinical application, limitations and controversy with regard to placement and interpretation remain. The PPD-ST has several drawbacks: false-positive reactivity due to nontuberculin strains such as BCG, interobserver variability in reading, false-negative results due to underlying immunosuppression, and variability with repeat testing. Controversies regarding interpretation continue to surface, partly due to changes in the population being tested. Due to changes in disease prevalence and demographics, a heightened need for early detection and better testing methodologies has emerged. Most research has tended toward developing better testing antigens and a diagnostic assay system that would overcome the limitations of the intradermal PPD-ST. In Australia a whole-blood culture kit based on IFN-γ production has recently been approved for in vitro human tuberculosis screening. This kit has not been widely used to date and is currently not approved in the United States. Our study has confirmed the utility of the kit for in vitro PPD testing and has evaluated tuberculin-specific subfractions in lieu of PPD in hopes of finding a more accurate tuberculosis diagnostic system.
The human whole-blood culture assay evolved from earlier bovine studies which compared traditional PBMC stimulation and skin testing to the new whole blood assay. These studies demonstrated that the IFN-γ test had greater diagnostic sensitivity, cost less, and yielded rapid results for cattle tuberculosis screening (24, 31, 32). This concept was extended for human tuberculosis testing by Streeton et al., who reported a sensitivity and specificity of 90.5 and 98%, respectively, for diagnosing M. tuberculosis exposure using the Q-IFN kit (30). In their study the gold standard for diagnosis was the PPD-ST and the study subjects were stratified according to their skin test induration and risk of exposure to tuberculosis. Converse et al. (4) and Kimura et al. (16) used the Q-IFN in studies comparing the IFN release assay with PPD-ST in populations at risk for M. tuberculosis exposure (intravenous drug users with or without human immunodeficiency virus [HIV] infection). They found that the Q-IFN assay detected more reactors than the PPD-ST. Agreement between the two tests was weak (4, 16). In our study of 40 patients there was good agreement between the PPD-ST and the Q-IFN kit (kappa = 0.73). We chose only to evaluate the agreement between the in vivo and in vitro tests and not to refer to sensitivity and specificity because the latter assume comparison to an adequate gold standard, which is currently lacking.
One of the benefits of the in vitro system is the ability to use avian PPD as a stimulant in order to differentiate MAC from tuberculosis infection. We had three individuals test positive for MAC. Davidson et al. evaluated a whole-blood culture system for non-M. tuberculosis adenitis and reported a significant association between IFN-γ response and MAC disease (7). We cannot be certain if the MAC-positive individuals in our study actually carried MAC, since the patients were all healthy. A larger study with a group of pulmonary MAC patients needs to be performed in order to address the positive predictive value for differentiating MAC from M. tuberculosis. Eight patients with previous positive histories of skin test reactivity who were now PPD-ST negative (Prior-POS) were all classified as M. tuberculosis reactive by the Q-IFN kit. In the literature the rate of reversion of the PPD-ST has been estimated at 8% per year (21). This poses an additional limitation on skin testing, as the intradermal response appears to modulate over time. Our eight patients were too small a group to draw any statistically significant conclusions regarding the in vitro response over time, but the results suggest that an individual once exposed may continue to produce IFN-γ upon antigen stimulation.
The immune response to tuberculosis is primarily cell mediated and is an interplay between a variety of T cells, macrophages, and cytokines. Historically, the host immune response to tuberculosis in humans has been measured by the DTH skin test. Sepkowitz reported that the size of the PPD-ST correlated with the risk of developing active tuberculosis (26). The potent responders demonstrated a higher incidence of developing active disease. Pottumarthy et al. showed a correlation between IFN-γ measured by the Q-IFN kit and PPD-ST induration (23). Like Sepkowitz, these authors raised the question whether quantifying the IFN-γ response may predict the future risk of developing disease. In our study we did not see a correlation between IFN-γ and size of skin test induration. The discordance between these results is not unexpected since natural host variability in whole blood culture response due to differing assay conditions (time of sampling, time of incubation) may result in quantitative differences in measurement of the same immunologic mediator (8).
One significant drawback of both the PPD-ST and the Q-IFN kit is the nonspecific response to PPD because of cross-reactivity between tuberculin and other mycobacterial species. False positivity in the M. tuberculosis skin testing due to BCG vaccination is well documented. However, the in vitro IFN-γ production of patients who have received BCG may also be affected by cross-reacting mycobacterial antigens in the PPD preparation. Streeton et al. included BCG vaccinees in their study but were unable to discern the effects of BCG on assay results (30). Therefore, additional studies are needed to delineate the diagnostic value of the Q-IFN kit in this population. The in vitro diagnostic system affords a distinct advantage over the PPD-ST in that one can test tuberculin-specific proteins without unnecessarily exposing the patient. Low-molecular-weight antigens such as ESAT-6 have been shown to differentiate between M. tuberculosis and BCG strains of mycobacteria and thus ESAT-6 may serve as an additional stimulant to determine the effect of BCG. Recently, ESAT-6 (6 kDa) was evaluated in the Q-IFN assay and found to differentiate those infected with tuberculosis from controls with high sensitivity and specificity (15).
LMWCFP contains secreted proteins of less than 10,000 Da. Earlier work with PBMC culture showed the stimulating capacity of proteins in this subfraction (3, 22). We noted higher IFN-γ production in response to LMWCFP in the PPD-POS subjects using the whole-blood assay. This likely represented reactivity not only to ESAT-6 epitopes but also to the other low-molecular-weight proteins. The whole CFP-LAM subfraction contains the majority of secreted proteins, most of which are in the 10,000- to 100,000-Da range. Numerous purified proteins in this subfraction have been evaluated as possible antigens for T-cell stimulation. Boesen et al. showed strong IFN-γ release in patients with active minimal tuberculosis after PBMC stimulation with molecular-mass fractions of CFP (2). Similar results were obtained by Havlir and colleagues, who noted blastogenic activity with the 30-, 37-, 44-, 57-, 64-, and 71-kDa proteins (12). Our data, obtained using a whole-blood assay, show a significantly higher IFN-γ response in PPD reactors than in controls and are consistent with these other studies, which assessed either lymphocyte proliferation or cytokine release in PBMC culture supernatants.
The mycobacterial cell wall is a complex mixture of proteins, carbohydrates, and phospholipids. The SCWP is an extremely heterogeneous mixture of proteins, including low levels of LAM (∼10 ng/ml). The nonspecific response noted in our study in both the PPD-positive and -negative groups is best explained by the impurity of the subfraction. Although specific stimulators are contained in this subfraction, as evident by work performed by Barnes et al., who demonstrated T-cell responses to 10-, 23-, 28-, and 30-kDa proteins, the contribution to the immune response from other uncharacterized protein fragments remains undefined (1). The heterogeneity of our subfraction compared to the purity of the Barnes mixture may explain the discordant results between the two studies.
The cytosolic fraction is similar to the culture filtrate in that some of the cytosolic proteins are released into culture. The CFP can demonstrate considerable variability depending upon temperature and harvesting time. In this study our CFP was late-log-phase culture, and thus there may have been considerable redundancy between the CFP and CYT. This may explain the similar specific IFN-γ responses for these two subfractions.
The discordance in results between the PPD supplied with the Q-IFN kit and H37Rv in terms of degree of IFN-γ response demonstrates the heterogeneity in PPD preparations. The commercial preparations are all standardized to a reference and labeled according to their ability to produce a certain size induration with skin testing (labeling in TU). They are not standardized based on protein concentration. Therefore, different preparations may have variable protein distributions and may result in different in vitro stimulation patterns. We used the H37Rv PPD at 10 μg/ml based on preliminary dose-response studies. We do not know if this is equivalent to the concentration of PPD in the Q-IFN kit, because the latter is proprietary information. This may also explain the differential response between the two PPD preparations.
These data demonstrate the efficacy of tuberculin-specific subfraction preparations in stimulating IFN-γ production in whole-blood culture. However, none of the subfractions performed any better than the whole PPD supplied with the Q-IFN kit. The PPD and the crude preparations used in this study do not differentiate M. tuberculosis-specific responses from non-tuberculin-specific responses. A concurrent test for MAC using AvPPD must be done. Those subfractions demonstrating efficacy in this study should be further isolated and studied in the whole-blood system. The immune response to tuberculosis is complex and is directed to a heterogenous mixture of antigens rather than any one protein. Therefore, a cocktail of native or recombinant antigens may prove to be more specific and sensitive in diagnosing tuberculosis than any one immunodominant protein.
In conclusion, this study is the first to present the stimulating potential of four M. tuberculosis subfractions (LMWCFP, CFP-LAM, SCWP, and CYT) in the Q-IFN kit. This information may prove useful as the quest for a more specific M. tuberculosis testing antigen continues. Additionally, we provide new information regarding the behavior of different PPD formulations in culture, highlighting the importance of using one standardized preparation. Our paper not only corroborates the previously published data confirming the usefulness of Q-IFN as an in vitro test but also critically evaluates the mechanics of the Q-IFN assay and raises several questions regarding data reduction. The advantages of the blood test include the absence of any reading or placement variability and the need for only one office visit. The current disadvantages are the need for more stringent laboratory requirements dealing with blood handling, cell culture, and enzyme-linked immunosorbent assay (ELISA) testing. The areas needing clarification include defining the performance characteristics and the lower-limit cutoff for the EIA. Data are needed on temporal measurements to determine if the time of day or different days have any impact on the whole-blood stimulation response. Also, it is not clear if the interval between the blood draw and processing has any impact on the IFN-γ response. Finally, the percent HuPPD cutoff of 15% needs to be verified based on prevalence of disease in different populations, as has been done with the PPD-ST. These issues should be addressed and followed by large-scale trials to assess the true sensitivity, specificity, and positive predictive value of the Q-IFN kit, prior to its widespread use for clinical M. tuberculosis testing.
FIG. 3.
Comparison of the IFN-γ response to stimulation by CFP-LAM in the two PPD-ST groups. The subfraction IFN-γ response is shown as a percentage of the mitogen response.
FIG. 4.
Comparison of the IFN-γ response to stimulation by CYT in the two PPD-ST groups. The subfraction IFN-γ response is shown as a percentage of the mitogen response.
REFERENCES
- 1.Barnes P, Mehra V, Hirschfield G, Fong S, Abou-Zeid C, Rook G, Hunter S, Brennan P J, Modlin R L. Characterization of T cell antigens associated with the cell wall protein-peptidoglycan complex of Mycobacterium tuberculosis. J Immunol. 1989;143:2656–2662. [PubMed] [Google Scholar]
- 2.Boesen H, Jensen B, Wilcke T, Andersen P. Human T-cell responses to secreted antigen fractions of Mycobacterium tuberculosis. Infect Immun. 1995;63:1491–1497. doi: 10.1128/iai.63.4.1491-1497.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Coler R N, Skeiky Y A, Vedvick T, Bement T, et al. Molecular cloning and immunologic reactivity of a novel low molecular mass antigen of Mycobacterium tuberculosis. J Immunol. 1998;161:2356–2364. [PubMed] [Google Scholar]
- 4.Converse P J, Jones S L, Astemborski J, Vlahov D, Graham N M. Comparison of a tuberculin interferon-gamma assay with the tuberculin skin test in high-risk adults: effect of human immunodeficiency virus infection. J Infect Dis. 1997;176:144–150. doi: 10.1086/514016. [DOI] [PubMed] [Google Scholar]
- 5.Daniel T. The immunology of tuberculosis. Clin Chest Med. 1980;1:189–201. [PubMed] [Google Scholar]
- 6.Daniel T M, Ellner J J. Immunology of tuberculosis. In: Reichman L, Hershfield E S, editors. Tuberculosis: a comprehensive international approach. 1st ed. New York, N.Y: Marcel Dekker, Inc; 1993. pp. 75–101. [Google Scholar]
- 7.Davidson P M, Creati L, Wood P R, Robertson D M, Hosking C S. Lymphocyte production of gamma-interferon as a test for non-tuberculous mycobacterial lymphadenitis in children. Eur J Pediatr. 1993;152:31–35. doi: 10.1007/BF02072513. [DOI] [PubMed] [Google Scholar]
- 8.De Groote D, Zangerle P F, Gevaert Y, Fassotte M F, Beguin Y, Noizat-Pirenne J, Gathy R, Lopez M, Dehart I, et al. Direct stimulation of cytokines (IL-1β, TNF-α, IL-6, IL_2, IFN-γ and GM-CSF) in whole blood. I. Comparison with isolated PBMC stimulation. Cytokine. 1992;4:239–248. doi: 10.1016/1043-4666(92)90062-v. [DOI] [PubMed] [Google Scholar]
- 9.Fenton M, Vermeulen M, Kim S, Burdick M, Strieter R, Kornfeld H. Induction of gamma interferon production in human alveolar macrophages by Mycobacterium tuberculosis. Infect Immun. 1997;65:5149–5156. doi: 10.1128/iai.65.12.5149-5156.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Fleiss J L. Statistical methods for rates and proportions. New York, N.Y: John Wiley & Sons; 1981. p. 218. [Google Scholar]
- 11.Francis J, Seiler R J, Wilkie I W, O'Boyle D, et al. The sensitivity and specificity of various tuberculin tests using bovine PPD and other tuberculins. Vet Rec. 1978;103:420–425. doi: 10.1136/vr.103.19.420. [DOI] [PubMed] [Google Scholar]
- 12.Havlir D, Wallis R S, Boom H, Daniel T, Chervenak K, Ellner J. Human immune response to Mycobacterium tuberculosis antigens. Infect Immun. 1991;59:665–670. doi: 10.1128/iai.59.2.665-670.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Heisser A, DeGuzman R, Brooks J, Veltri N, Carregal V, Smith L S, Carpenter G B, Engler R J M. Delayed-type hypersensitivity testing for the evaluation of cellular immunity: normal responses for adult men and women. J Allergy Clin Immunol. 1996;97(1, part 3):399. [Google Scholar]
- 14.Hirschfield G R, McNeil M, Brennan P J. Peptidoglycan-associated polypeptides of Mycobacterium tuberculosis. J Bacteriol. 1990;172:1005–1013. doi: 10.1128/jb.172.2.1005-1013.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Johnson P D R, Stuart R L, Grayson M L, Olden D, Clancy A, Ravn P, Andersen P, Britton W J, Rothel J S. Tuberculin-purified protein derivative, MPT-64, and ESAT-6 stimulated gamma interferon responses in medical students before and after Mycobacterium bovis BCG vaccination and in patients with tuberculosis. Clin Diagn Lab Immun. 1999;6:934–937. doi: 10.1128/cdli.6.6.934-937.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Kimura M, Converse P J, Astemborski J, Rothel J S, Vlahov D, Comstock G W, Graham N M, Chaisson R E, Bishai W R. Comparison between a whole blood interferon-gamma release assay and tuberculin skin testing for the detection of tuberculosis infection among patients at risk for tuberculosis exposure. J Infect Dis. 1999;179:1297–1300. doi: 10.1086/314707. [DOI] [PubMed] [Google Scholar]
- 17.Laal S, Samanich K M, Sonnenberg M G, Zolla-Pazner S, Phadtare J M, Belisle J T. Human humoral responses to antigens of Mycobacterium tuberculosis: immunodominance of high-molecular-mass antigens. Clin Diagn Lab Immunol. 1997;4:49–56. doi: 10.1128/cdli.4.1.49-56.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Lee B Y, Hefta S A, Brennan P J. Characterization of the major membrane protein of virulent Mycobacterium tuberculosis. Infect Immun. 1992;60:2066–2074. doi: 10.1128/iai.60.5.2066-2074.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Lein D, Von Reyn F. In vitro cellular and cytokine responses to mycobacterial antigens: application to diagnosis of tuberculosis infection and assessment of response to mycobacterial vaccines. Am J Med Sci. 1997;313:364–371. doi: 10.1097/00000441-199706000-00009. [DOI] [PubMed] [Google Scholar]
- 20.Mantoux C. L'intradermo-reaction a la tuberculin et son interpretation clinique. Presse Med. 1910;18:10–13. [Google Scholar]
- 21.Menzies D. Interpretation of repeated tuberculin tests, boosting, conversion and reversion. Am J Respir Crit Care Med. 1999;159:15–21. doi: 10.1164/ajrccm.159.1.9801120. [DOI] [PubMed] [Google Scholar]
- 22.Pais T F, Silv R A, Smedegaard B, Appelberg R, Andersen P. Analysis of T cells recruited during delayed-type hypersensitivity to purified protein derivative versus challenge with tuberculosis infection. Immunology. 1998;95:69–75. doi: 10.1046/j.1365-2567.1998.00561.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Pottumarthy S, Morris A, Harrison A, Wells V. Evaluation of the tuberculin gamma interferon assay: potential to replace the Mantoux skin test. J Clin Microbiol. 1999;37:3229–3232. doi: 10.1128/jcm.37.10.3229-3232.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Rothel J S, Jones S L, Corner L A, Cox J C, Wood P R. The gamma-interferon assay for diagnosis of bovine tuberculosis in cattle: conditions affecting the production of gamma-interferon in whole blood culture. Aust Vet J. 1992;69:1–4. doi: 10.1111/j.1751-0813.1992.tb09848.x. [DOI] [PubMed] [Google Scholar]
- 25.Seibert F B, Glenn J T. Tuberculin purified protein derivative: preparation and analysis of a large quantity for standard. Am Rev Tuber. 1941;44:9–25. [Google Scholar]
- 26.Sepkowitz K A. Tuberculin skin testing and the health care worker: lessons of the Prophit Survey. Tuber Lung Dis. 1996;77:81–85. doi: 10.1016/s0962-8479(96)90081-7. [DOI] [PubMed] [Google Scholar]
- 27.Smith P K, Krohn R I, Hermanson G T, Mallia A K, Gartner F H, Provenzano M D, Fujimoto E K, Goeke N M, Olson B J, Klenk D C. Measurement of protein using bicinchoninic acid. Anal Biochem. 1985;150:76–85. doi: 10.1016/0003-2697(85)90442-7. [DOI] [PubMed] [Google Scholar]
- 28.Snider D. The tuberculin skin test. Am Rev Respir Dis. 1982;125(Suppl.):108–118. doi: 10.1164/arrd.1982.125.3P2.108. [DOI] [PubMed] [Google Scholar]
- 29.Sonnenberg M G, Belisle J T. Definition of Mycobacterium tuberculosis culture filtrate proteins by two-dimensional polyacrylamide gel electrophoresis, N-terminal amino acid sequencing, and electrospray mass spectrometry. Infect Immun. 1997;65:4515–4524. doi: 10.1128/iai.65.11.4515-4524.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Streeton J A, Desem N, Jones S L. Sensitivity and specificity of a gamma interferon blood test for tuberculosis infection. Int J Tuberc Lung Dis. 1998;2:443–450. [PubMed] [Google Scholar]
- 31.Wood P R, Corner L A, Rothel J S, et al. Field comparison of the interferon-gamma assay and the intradermal tuberculin test for the diagnosis of bovine tuberculosis. Aust Vet J. 1991;68:286–290. doi: 10.1111/j.1751-0813.1991.tb03254.x. [DOI] [PubMed] [Google Scholar]
- 32.Wood P R, Rothel J S. In vitro immunodiagnostic assay for bovine tuberculosis. Vet Microbiol. 1994;40:125–135. doi: 10.1016/0378-1135(94)90051-5. [DOI] [PubMed] [Google Scholar]