Purpose of review
To describe different strategies adopted during coronavirus disease 2019 pandemic to cope with the shortage of mechanical ventilators.
Recent findings
Short-term interventions aimed to increase ventilator supply and decrease demand. They included: redistributing and centralizing patients, repurposing operating rooms into intensive care units (ICUs) and boosting ventilator production and using stocks and back-ups; support by the critical care outreach team to optimize treatment of patients in the ward and permit early discharge from the ICU, ethical allocation of mechanical ventilators to patients who could benefit more from intensive treatment and short term ICU trials for selected patients with uncertain prognosis, respectively. Long-term strategies included education and training of non-ICU physicians and nurses to the care of critically-ill patients and measures to decrease viral spread among the population and the progression from mild to severe disease.
Summary
The experience and evidence gained during the current pandemic is of paramount importance for physicians and law-makers to plan in advance an appropriate response to any future similar crisis. Intensive care unit, hospital, national and international policies can all be improved to build systems capable of treating an unexpectedly large number of patients, while keeping a high standard of safety.
Keywords: coronavirus disease 2019, intensive care unit, mechanical ventilators, resource limitation, triage
INTRODUCTION
In the summer of 1952, a wave of poliomyelitis infections hit Copenhagen metropolitan area, bringing an unprecedented number of patients in need for artificial ventilation to the attention of Danish physicians. At the peak of the epidemic, 70 patients requiring artificial ventilation were simultaneously treated at Blegdam Hospital, where only six ventilators were available, and medical students were hired to provide manual bag ventilation [1].
Almost 70 years later, despite huge technological advances, intensive care physicians faced another tremendous imbalance between need to ventilate patients and availability of mechanical ventilators. In February 2020, the first cases of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection appeared in Italy, the first European Country to be hit by the pandemic. In the next few days, cases increased exponentially and so did hospital and intensive care unit (ICU) admissions for respiratory failure due to SARS-CoV-2 pneumonia (coronavirus disease 2019) [2]. The existing ICU bed capacity (in the Lombardy region 738 ICU beds [3]) was not enough to face the spread of the epidemic. With an unprecedented effort, regional healthcare resources were increased, repurposed and optimized in order to be able to cope with the escalating demand of coronavirus disease 2019 (COVID-19) admissions and to continue the nondeferrable care of non-COVID-19 patients [4].
In this review, we will focus on strategies to deal with the shortage of mechanical ventilators, which represents a bottleneck during a pandemic of this sort. We will describe strategies to increase mechanical ventilators supply and to decrease demand, using our past experience and published evidence to guide future logistical and operational decisions.
Box 1.
no caption available
INCREASING VENTILATOR SUPPLY
At the beginning of an epidemic, forecasts of hospital and ICU admissions in the next few weeks should be made on a worst-case scenario basis [2]. This requires a basic knowledge of speed of viral spread and rate of severe disease. Standard resources available (free hospital and ICU beds) will give a gross idea of the gap between demand and supply. When this is wide, immediate action is required at different levels (Table 1):
Table 1.
Strategies to cope with mechanical ventilators demand/supply mismatch
| Strategies to ↓ demand | Strategies to ↑ supply | ||
| Immediate action | Long-term action | Immediate action | Long-term action |
| Optimize treatment of patients in the ward | ↑ ICU beds | ||
| Critical-care outreach team • support to ward staff ∘ NIV/CPAP use ∘ awake pronation ∘ sepsis management • triage patients ∘ escalation of treatment ∘ ceiling-of-care • care of patients discharged from ICU ∘ tracheostomy ∘ CPAP/HFNC | Education and training of ward staff (physicians and nurses) to the care of respiratory failure patients with noninvasive respiratory support or tracheostomy | Non-COVID-19 patients reallocation • within region/state • outside region/state COVID-19 patients centralization | Create new cohorted ICUs for COVID-19 patients • inside hospital • outside hospital |
| Optimize allocation of mechanical ventilators | Optimize use of existing ventilators | ||
| ICU consultants committee • triage of patients • end-of-life decisions • communications with family Multidimensional evaluation • severity of disease (gas exchange, CT scan, other infections, response to treatment) • capacity to recover (age, comorbidities, frailty) • outcome prediction models (APACHE, SAPS, MPM) Time-limited ICU trial and withdrawal of care | Specific education of more experienced ICU consultants on end-of-life decisions and communication with family | Repurposing of operating rooms • ORs into ICUs • use anesthesia ventilators and staff ↓ elective surgery • hub hospitals for nondeferrable surgical cases Use 1 mechanical ventilator for 2 patients | Plan in advance • paths for COVID-19 and non-COVID-19 patients • hub hospitals for oncologic surgery and time-dependent diseases (e.g. stroke, acute myocardial infarction) |
| ↓ burden on disease on the general population | ↑ mechanical ventilators/staff | ||
| Social distancing policies | Vaccination programs Therapies to ↓ progression to severe disease Research funding | Obtain new mechanical ventilators • ventilators in stock from vendors • backups, transport ventilators | Money investment in industry to boost production Hiring extra staff Specific training of residents/non-ICU physicians/nurses/respiratory therapists for critically-ill COVID-19 patients care |
APACHE, Acute Physiology and Chronic Health Evaluation; CPAP, continuous positive airway pressure; CT, computed tomography; HFNC, high flow nasal cannula; ICU, intensive care unit; MPM, Mortality Probability Model; NIV, noninvasive ventilation; OR, operating room; SAPS, Simplified Acute Physiology Score.
-
(1)
increase number of ICU beds by reallocating and centralizing patients (region-wide),
-
(2)
optimize use of existing ventilators by repurposing operating rooms (center-wide),
-
(3)
expand the pool of mechanical ventilators available using stocks/backups (ICU-wide).
Reallocation and centralization of patients
The first case of severe COVID-19 pneumonia in Italy occurred in Lombardy (a northern Italian region) on February 20, 2020, soon followed by 36 more cases. On the following day, a regional emergency task force (COVID-19 Lombardy ICU network) identified 15 hospitals as hub centers for critically-ill COVID-19 patients. Centers were recruited based on experience in severe ARDS patients’ care and were required to create dedicated cohorted ICUs for COVID-19 patients [4]. With this simple logistical measure, 130 COVID-19 ICU beds were made available in 48 h. Even so, saturation of resources did occur [5▪]. By March 7, the number of COVID-19 ICU beds was 482 and the number of centers involved 55. Eventually, all hospitals of the regions were involved.
Centralization of patients is possible only if a larger-scale reorganization of the delivery of care is pursued. In Lombardy, non-COVID-19 patients needing urgent or nondeferrable care (e.g. oncologic and vascular surgery, acute coronary syndromes) were diverted to few centers, not or marginally involved in the care of COVID-19 cases [6–8]. Transfer of non-COVID-19 critically-ill patients to receptive hospitals outside the region or Country was another emergency measure undertaken [9].
Repurposing operating rooms and anesthesia ventilators
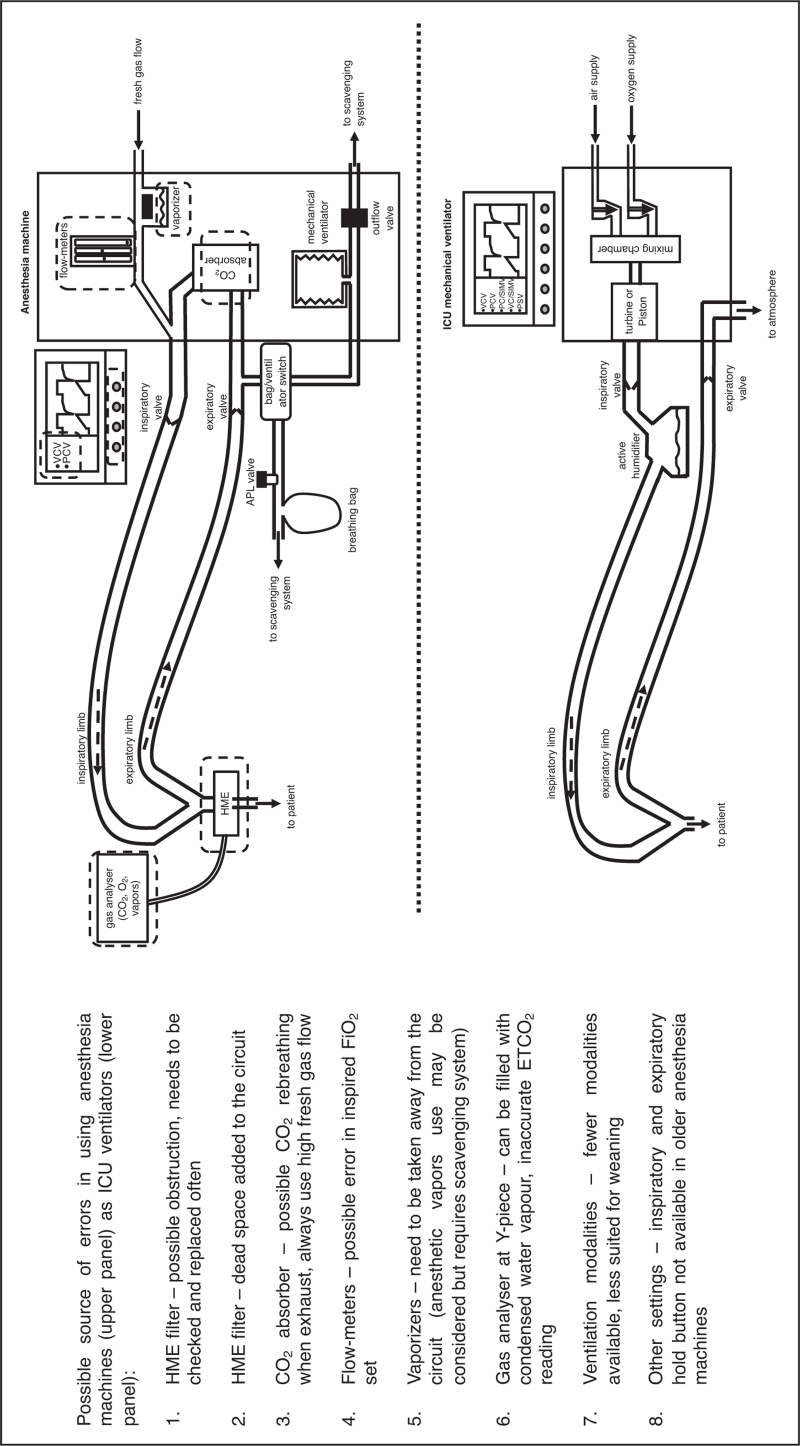
The conversion of operating theatres and postanesthesia care units (PACU) in COVID-19 ICUs was one of the first responses to the crisis [10,11] and presents some advantages. First, the partial or total shut-down of elective surgery creates ready-to-use spaces to cohort COVID-19 patients and frees personnel (anesthesiologists, nurses) trained to take care of intubated patients; second, it allows physical separation of COVID-19 ICU spaces from the conventional ICU; third, it provides an environment already equipped with mechanical ventilators, airway management and invasive cardiovascular monitoring material, pressurized medical gases and power supply [12]. Even if anesthesia ventilators do not have the same features as ICU ventilators, most of them permit basic ventilation modalities and monitoring sufficient to safely ventilate critically-ill patients deeply sedated and curarized. These ventilators lack some important features, need extra care and monitoring and are not usually suitable for weaning patients [13] (Fig. 1), nonetheless they proved useful to expand the number of available mechanical ventilators. During the first wave of the pandemic in March 2020, the first 16-beds cohorted ICU we opened was located in a closed PACU [9]. Others have successfully used operating rooms, housing up to three patients per room [14▪].
FIGURE 1.
Differences between anesthesia and ICU mechanical ventilators. A schematic representation of an anesthesia machine (upper panel) and an intensive care unit mechanical ventilator (lower panel) is shown. Major differences in ventilator set-up and functionalities are indicated with dashed rectangles and discussed further in the caption.
Expanding the pool of mechanical ventilators
All mechanical ventilators available in each hospital should be sought, checked for functioning and presence of appropriate disposables (circuit, valves) and made available. These include back-ups and transport ventilators, which while not suited for long term ventilation, can save time (and lives) while appropriate machines are sought. These may come from the industry (hospital suppliers with overstocked/unpurchased ventilators), other regions less affected by the epidemic [15] or national stocks [16].
Money investment to ensure contracts with manufacturers and boost production and redistribution of resources within and between Countries should be pursued [17]. It is a fact that the number of ICU beds pro capita varies widely across Europe [18]. It is thus not surprising that during the first pandemic wave mechanical ventilators were transferred between differently-affected Countries and that the World Health Organization has indicated supply chains as one of the pillars of COVID-19 preparedness plans [19].
An extreme measure to increase the number of treatable patients is the use of a single ventilator to treat two (or more) patients simultaneously. This technique involves splitting the inspiratory and expiratory limbs, in order to have two circuits connected to a single ventilator [20], and has been recently proposed [21] and actually tested [22▪] in COVID-19 patients pairs. Although the idea is in itself worthy and the setup relatively easy, its realization requires actually more resources (trained staff and additional monitoring) rather than less, for the following reasons: patients need to be carefully matched for respiratory system mechanics to avoid differences in tidal volume delivered; even after correct matching, patients’ respiratory mechanics can rapidly change (e.g. for respiratory secretions worsening resistance in one patient) making patients’ ventilation unpredictable; the connection of two patients to a single ventilator increases the risk of infections and tube dislodgement. The technique has been discouraged by a joint statement of five American Scientific Societies [23]. It is our opinion that efforts and money should be invested in more efficient and safe measures to increase ventilators supply, such as the ones reported above.
STRATEGIES TO DECREASE VENTILATOR NEED
Appropriate allocation of (scarce) resources should always guide ethical and clinical choices, and ICU beds are one of the scarcest resources in any healthcare system. The strategies described below do not aim simply at “saving” mechanical ventilators, but at correctly distributing ventilators in order to maximize benefit among the (many) in need [24▪▪].
Outreach team
The response plan to the COVID-19 waves was initially based on three pathways: cohorting infected subjects at the ward or unit level, creating dedicated COVID-19 wards and ICU – level-3 beds [2]; increasing ICU beds; supporting high dependency units (level-2 beds) outside the ICU.
These beds are managed by non-ICU medical teams equipped with multiparametric monitors, blood gas analyzers, and interfaces for noninvasive ventilatory assistance, that is continuous positive airway pressure (CPAP), noninvasive ventilation (NIV) and high-flow nasal cannula (HFNC). Providing intensive care support to whoever needed by implementing a COVID-19 Critical Care Outreach Team (CCOT) in level-2 areas has been crucial to match the massive influx of critically ill unstable subjects with the limited capacity in our level-3 areas. Specifically, CCOT would support on modalities of oxygen delivery, awake pronation, sepsis management and assessment of patients with respiratory failure. Furthermore, the CCOT team helped in triaging patients considered for an escalation of cure and in the multidisciplinary decision-making process regarding end-of-life, by individualizing each subject's pathway of care according to the predicted benefit of ICU admission. The CCOT team also supported ward staff in caring for patients discharged from the ICU with some residual form of respiratory support (HFNC or tracheostomy), allowing early discharge and optimizing level-3 beds occupation.
In our center during the first wave, the CCOT was composed by two senior ICU consultants working on a 12-h 7/7 schedule, alternately on call during the night. This resulted in a low rate of cardiac arrest calls, no emergency intubations in the ward, and appropriate palliative care for patients having a ceiling of care decision [25]. This subgroup of patients is often treated with forms of noninvasive respiratory support (NIRS) outside the ICU and almost one third of patients with acute respiratory failure (with NIV or HFNC) received a do-not-intubate (DNI) order in the prepandemic setting [26] (Table 2). Ceiling-of-care decisions should consider several factors including not only the clinical characteristics of the patients and the expectations of the physicians, but promoting the participation of patients’ family members in life-sustaining therapies withdrawal [34].
Table 2.
“Do-not-intubate” orders in patients receiving noninvasive respiratory support in different studies
| “Do-not-intubate” orders criteria | ||||||||||
| Study | Pt. overall NIV/CPAP/HFNC (n) | Pt. DNI (%) | Setting | Clinical judgment | Patient refusal | Low predicted probability of hospital survival | Frailty | Comorbidity | Age | Disease severity |
| Franco et. [27] | 678 | 4.1 | PU | X | X | |||||
| Aliberti et al.[28] | 157 | 41.4 | HDU | X | X | X | X | |||
| Di Domenico et al.[29] | 90 | 30.0 | RICU/ED | X | X | X | ||||
| Faraone et al.[30] | 50 | 50.0 | CCU | X | X | X | ||||
| Vaschetto et al.[31] | 537 | 26.1 | RICU | X | X | X | ||||
| Menzella et al.[32] | 79 | 22.8 | PU | X | X | |||||
| Ramirez et al.[33] | 159 | 23.9 | INCU | X | X | |||||
| Messina et al.[25] | 74 | 41.8 | HDU | X | X | X | X | |||
CCU, COVID-19 care unit; ED, emergency department; HDU, high-dependency unit; INCU, intermediate care unit; Pt, patients; PU, pulmonology unit; RICU, respiratory intermediate care unit.
Interestingly, a recent meta-analysis on COVID-19 patients treated with NIRS outside the ICU showed an overall intra-hospital mortality of 36% [30–41%]; only a minority of these patients had DNI orders [35]. In our experience during the first wave, 19 of 61 patients who received a ceiling-of care-decision were finally discharged from the hospital [25]. This is consistent with data from a large (12 850 patients) multinational, observational study, on adult ICU patients showing that one in five patients with treatment limitations survived hospitalization [36▪]. This confirms the feasibility and safety of NIRS outside the ICU in selected patients, and shows that limiting the level of cure does not imply abandoning the patient, rather focusing on those treatments with the highest chances of success, balancing invasiveness, comfort and clinical efficacy.
Appropriate allocation of mechanical ventilators
An important goal of medicine is to offer beneficial treatments [37]. Patients who are extremely unlikely to survive outside the acute care setting should not receive prolonged mechanical ventilation, but palliative and comfort care. This principle is always valid, and even more so when resources are limited.
Outcome prediction models traditionally used in the ICU consider variables related to the principal diagnosis, the degree of acute physiological dysfunction, and the premorbid health status [38]. In evaluating critically ill patients with COVID-19, we used a multidimensional approach, where the severity of disease was weighed against the individual capacity to recover. A committee of two senior consultants not directly involved in providing care and the Head of Department was responsible for triage, end-of-life decisions, and communication with patients and family members.
The severity of disease was assessed based on signs of respiratory distress, gas exchange impairment, and findings at lung computed tomography; other vital organs function [39]; signs of other infection; illness trajectory and response to therapy. The individual capacity to recover was evaluated based on age, comorbidities, premorbid functional status, and frailty. Advanced age was considered a negative factor, as in all prognostic models developed before the pandemic [38]. In some reports, mortality of COVID-19 patients requiring invasive mechanical ventilation were as high as 77.1 (95% confidence intervals: 76.2–78.0) % in patients aged 70 years or more, and 84.4 (83.3–85.4) % in those aged 80 years or more [40]. Even so, age was not the sole criterion for resource allocation [41]. The overall burden of chronic disease was assessed with the Charlson comorbidity index [42], which considers the number and severity of comorbidities to predict a baseline risk of death. The premorbid functional status was evaluated based on the daily tasks that the patient could perform before getting sick [43]. Frailty is a syndrome characterized by an increased vulnerability to stressor events due to the accumulation of several physiological deficits. In response to a small insult, frail subjects typically experience a dramatic decline in health status: they become dependent on others for daily living, or die [44]. In subjects aged 65 years or older, we quantified frailty with the Clinical Frailty Scale, an assessment tool based on the premorbid physical activity, cognition, social relations, nutritional status, and comorbidities [45]. Increasing frailty more strongly predicts a worse outcome in elderly critically ill patients [46], including those with COVID-19 [47▪], independently from age.
The appropriateness of life sustaining interventions was reassessed during the ICU stay. Withdrawal of mechanical ventilation was considered in patients who deteriorated, developing major complications and becoming progressively more fragile, so that the disease became too severe for their residual capacity to recover. Time-limited ICU trials were offered to patients initially perceived to have a poor prognosis, but with some uncertainty [48]. They served to evaluate the response to therapy over the first few days and inform a decision to prolong treatment or not. Of note, withholding or withdrawing treatments that are determined to be futile were part of routine practice in the ICU even before COVID-19 [36▪].
LONG-TERM STRATEGIES TO INCREASE SUPPLY/DECREASE DEMAND
The immediate expansion of the pool of mechanical ventilators is a necessary step to keep the healthcare system working and patients cared for during a pandemic. At the same time, medium-long-term measures to increase preparedness to, and reduce severity of, future pandemics need to be urgently planned. These include the creation of new, cohorted ICUs within or outside the hospital walls (e.g. Ospedale Fiera Milano, a temporary hospital with up to 250 ICU beds capacity inaugurated in March 2020 in Milan [4]), strategic plans [5▪] to increase money investment in mechanical ventilators and other essential equipment, and hiring extra staff trained to care for critically-ill patients [17]. This is of paramount importance, as every “surge” ICU bed would be useless without specifically-trained “surge” personnel. The European Society of Intensive Care promoted a training program for residents, nurses and non-ICU physicians to increase the pool of professionals to be potentially involved in COVID-19 ICUs [49]. A recent large international study did not find differences in mortality rates between patients treated in “surge” versus “standard” ICU beds [50▪]. This underlines the need to extend quality – thus patients’ safety – while we are expanding numbers of ventilators and beds.
Different measures will (and have) in the long term reduce the burden of (severe) COVID-19 on the population and hence on the healthcare system. These include social distancing policies [5▪], vaccination programs and therapies to reduce the progression from infection to severe disease [51,52].
CONCLUSION
COVID-19 pandemic posed healthcare systems around the world on the verge of collapse, forcing physicians and politicians to make difficult choices to treat an overwhelming number of patients and to tackle viral spread. Western countries learnt what scarcity of resources compared to immediate need means. Even if as intensive care physicians we are confronted daily with the need to appropriately allocate treatments (and ventilators), COVID-19 has exposed the weaknesses of our system. Building from this experience shall ameliorate our organization at the hospital and ICU level, ultimately positively impacting on patients’ outcomes.
Acknowledgements
None.
Financial support and sponsorship
None.
Conflicts of interest
Dr Messina received travel expenses and registration for meetings, congresses, and courses and lecture fees from Vygon, Edwards and Philips. Prof. Cecconi is a consultant for Edwards Lifesciences Consultancy, Directed Systems Consultancy. The other authors have no conflict of interest to declare.
REFERENCES AND RECOMMENDED READING
Papers of particular interest, published within the annual period of review, have been highlighted as:
▪ of special interest
▪▪ of outstanding interest
REFERENCES
- 1.Lassen H, A preliminary report on the 1952 epidemic of poliomyelitis in Copenhagen with special reference to the treatment of acute respiratory insufficiency. Fink M, Hayes M, Soni N, editors. The Lancet. London: Springer London; 1953. [DOI] [PubMed] [Google Scholar]
- 2.Grasselli G, Pesenti A, Cecconi M. Critical care utilization for the COVID-19 outbreak in Lombardy, Italy. JAMA 2020; 323:1545–1546. [DOI] [PubMed] [Google Scholar]
- 3.Pecoraro F, Clemente F, Luzi D. The efficiency in the ordinary hospital bed management in Italy: an in-depth analysis of intensive care unit in the areas affected by COVID-19 before the outbreak. PLoS One 2020; 15:e0239249. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Rezoagli E, Magliocca A, Bellani G, et al. Development of a critical care response – experiences from Italy during the coronavirus disease 2019 pandemic. Anesthesiol Clin 2021; 39:265–284. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5▪.Berardi C, Antonini M, Genie MG, et al. The COVID-19 pandemic in Italy: policy and technology impact on health and nonhealth outcomes. Health Policy Technol 2020; 9:454–487. [DOI] [PMC free article] [PubMed] [Google Scholar]; A paper analyzing policies adopted by the Italian government to tackle virus spread during the first wave of the pandemic in the period March-May 2020 and their outcome. Interventions are analyzed from a health-care system (ICU saturation, mortality) and economic system point of view.
- 6.Bertolaccini L, Spaggiari L. Thoracic surgical oncology in lombardy: how to do it during COVID-19 time? The annals of thoracic surgery. Soc Thorac Surg 2020; 110:2108–2109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Kahlberg A, Mascia D, Bellosta R, et al. Vascular surgery during COVID-19 emergency in hub hospitals of lombardy: experience on 305 patients. Eur J Vasc Endovasc Surg 2021; 61:306–315. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Carugo S, Ferlini M, Castini D, et al. Management of acute coronary syndromes during the COVID-19 outbreak in Lombardy: the “macro-hub” experience. IJC Heart Vascul 2020; 31:100662. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Sammito S, Turc J, Post J, et al. Analysis of European air medical evacuation flights of coronavirus disease 2019 patients. Air Med J 2021; 40:211–215. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Carenzo L, Costantini E, Greco M, et al. Hospital surge capacity in a tertiary emergency referral centre during the COVID-19 outbreak in Italy. Anaesthesia 2020; 75:928–934. [DOI] [PubMed] [Google Scholar]
- 11.Peters AW, Chawla KS, Turnbull ZA. Transforming ORs into ICUs. N Engl J Med 2020; 382:e52. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Tan Z, Phoon PHY, Tien CJ-C, et al. Practical considerations for converting operating rooms and postanaesthesia care units into intensive care units in the COVID-19 pandemic – experience from a large Singapore Tertiary Hospital. Ann Acad Med Singap 2020; 49:1009–1012. [PubMed] [Google Scholar]
- 13.Austin PN, Branson RD. Using anesthesia machines as critical care ventilators during the COVID-19 pandemic. Respir Care 2021; 66:1184–1195. [DOI] [PubMed] [Google Scholar]
- 14▪.Mittel AM, Panzer O, Wang DS, et al. Logistical considerations and clinical outcomes associated with converting operating rooms into an intensive care unit during the coronavirus disease 2019 pandemic in a New York City hospital. Anesth Analg 2021; 132:1182–1190. [DOI] [PubMed] [Google Scholar]; A paper describing the repurposing of operating rooms (and anesthesia machines) into cohorted COVID-19 ICUs as a response to the surge of patients during the first wave of the pandemic in a large academic Center in New York. Outcomes of patients treated in the new ICUs were in line with outcomes of COVID-19 patients treated in “regular” ICUs.
- 15.Ramachandran P, Swamy L, Kaul V, Agrawal A. A national strategy for ventilator and ICU resource allocation during the coronavirus disease 2019 pandemic. Chest 2020; 158:887–889. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Halpern NA, Tan KS. United States resource availability for COVID-19, version 3, revised 5/12/2020. Available at: www.sccmorg.com [Accessed May 27, 2022]. [Google Scholar]
- 17.Armocida B, Formenti B, Ussai S, et al. The Italian health system and the COVID-19 challenge. Lancet Public Health 2020; 5:e253. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Bauer J, Brüggmann D, Klingelhöfer D, et al. Access to intensive care in 14 European countries: a spatial analysis of intensive care need and capacity in the light of COVID-19. Intensive Care Me 2020; 46:2026–2034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.World Health Organization. COVID-19 strategic preparedness and response plan: 1 February 2021 to 31 January 2022. 2021. [Google Scholar]
- 20.Neyman G, Irvin CB. A single ventilator for multiple simulated patients to meet disaster surge. Acad Emerg Med 2006; 13:1246–1249. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Tonetti T, Zanella A, Pizzilli G, et al. One ventilator for two patients: feasibility and considerations of a last resort solution in case of equipment shortage. Thorax 2020; 75:517–519. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22▪.Beitler JR, Mittel AM, Kallet R, et al. Ventilator sharing during an acute shortage caused by the COVID-19 pandemic. Am J Respir Crit Care Med 2020; 202:600–604. [DOI] [PMC free article] [PubMed] [Google Scholar]; This is the first report of “ventilator sharing” for up to 48 h in three couples of COVID-19 patients matched for baseline respiratory mechanics and gas exchange. All six patients were successfully weaned off the ventilator and discharged from hospital.
- 23. COVID-19 - SCCM Joint Statement. Consensus statement on multiple patients per ventilator. March 26, 2020. www.sccm.org, Last Accessed 10/07/2022. [Google Scholar]
- 24▪▪.Emanuel EJ, Persad G, Upshur R, et al. Fair allocation of scarce medical resources in the time of COVID-19. N Engl J Med 2020; 382:2049–2055. [DOI] [PubMed] [Google Scholar]; This outstanding paper tries to set a series of recommendations for fair and ethical allocation of treatments in times of resource-limitation such as the ones faced during COVID-19 pandemic. The six recommendations proposed may be used for developing guidelines for helping clinicians make difficult choices in times of crisis.
- 25.Messina A, Pradella A, Alicino V, et al. Critical care outreach team during COVID-19: ventilatory support in the ward and outcomes. Respir Care 2021; 66:928–935. [DOI] [PubMed] [Google Scholar]
- 26.Wilson ME, Mittal A, Karki B, et al. Do-not-intubate orders in patients with acute respiratory failure: a systematic review and meta-analysis. Intensive Care Med 2019; 46:1–10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Franco C, Facciolongo N, Tonelli R, et al. Feasibility and clinical impact of out-of-ICU noninvasive respiratory support in patients with COVID-19-related pneumonia. Eur Respir J 2020; 56: [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Aliberti S, Radovanovic D, Billi F, et al. Helmet CPAP treatment in patients with COVID-19 pneumonia: a multicentre cohort study. Eur Respir J 2020; 56.: [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Di Domenico SL, Coen D, Bergamaschi M, et al. Clinical characteristics and respiratory support of 310 COVID-19 patients, diagnosed at the emergency room: a single-center retrospective study. Intern Emerg Med 2021; 16:1051–1060. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Faraone A, Beltrame C, Crociani A, et al. Effectiveness and safety of noninvasive positive pressure ventilation in the treatment of COVID-19-associated acute hypoxemic respiratory failure: a single center, non-ICU setting experience. Intern Emerg Med 2021; 16:1183–1190. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Vaschetto R, Barone-Adesi F, Racca F, et al. Outcomes of COVID-19 patients treated with continuous positive airway pressure outside the intensive care unit. ERJ Open Res 2021; 7:00541–2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Menzella F, Barbieri C, Fontana M, et al. Effectiveness of noninvasive ventilation in COVID-19 related-acute respiratory distress syndrome. Clin Respir J 2021; 15:779–787. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Ramirez GA, Bozzolo EP, Gobbi A, et al. Outcomes of noninvasive ventilation as the ceiling of treatment in patients with COVID-19. Panminerva Med 2021; doi: 10.23736/S0031-0808.21.04280-4. [Online ahead of print]. [DOI] [PubMed] [Google Scholar]
- 34.Sprung CL, Truog RD, Curtis JR, et al. Seeking worldwide professional consensus on the principles of end-of-life care for the critically ill. The consensus for Worldwide End-of-Life Practice for Patients in Intensive Care Units (WELPICUS) study 2014; 855–866. [DOI] [PubMed] [Google Scholar]
- 35.Cammarota G, Esposito T, Azzolina D, et al. Noninvasive respiratory support outside the intensive care unit for acute respiratory failure related to coronavirus-19 disease: a systematic review and meta-analysis. Crit Care 2021; 25:1–14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36▪.Avidan A, Sprung CL, Schefold JC, et al. Variations in end-of-life practices in intensive care units worldwide (Ethicus-2): a prospective observational study. Lancet Respir Med 2021; 9:1–10. [DOI] [PubMed] [Google Scholar]; Observational international study on end-of-life practices in ICU patients in different countries. Withholding, followed by withdrawing, life-sustaining treatment was the most common treatment limitation with great variability between different countries.
- 37.Luce JM. Ethical principles in critical care. JAMA 1990; 263:696–700. [PubMed] [Google Scholar]
- 38.Vincent J-L, Moreno R. Clinical review: scoring systems in the critically ill. Crit Care 2010; 14:207–209. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Springer-Verlag, Vincent JL, Moreno R, Takala J, et al. The SOFA (Sepsis-related Organ Failure Assessment) score to describe organ dysfunction/failure. On behalf of the working group on sepsis-related problems of the European Society of Intensive Care Medicine 1996; 707–710. [DOI] [PubMed] [Google Scholar]
- 40.Lim ZJ, Subramaniam A, Ponnapa Reddy M, et al. Case fatality rates for patients with COVID-19 requiring invasive mechanical ventilation. A meta-analysis. Am J Respir Crit Care Med 2021; 203:54–66. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Cesari M, Proietti M. COVID-19 in Italy: ageism and decision making in a pandemic. J Am Med Dir Assoc 2020; 21:576–577. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Charlson ME, Pompei P, Ales KL, MacKenzie CR. A new method of classifying prognostic comorbidity in longitudinal studies: development and validation. J Chronic Dis 1987; 40:373–383. [DOI] [PubMed] [Google Scholar]
- 43.Jette AM, Davies AR, Cleary PD, et al. The Functional Status Questionnaire: reliability and validity when used in primary care. J Gen Intern Med 1986; 1:143–149. [DOI] [PubMed] [Google Scholar]
- 44.Clegg A, Young J, Iliffe S, et al. Frailty in elderly people. Lancet 2013; 381:752–762. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Rockwood K, Song X, MacKnight C, et al. A global clinical measure of fitness and frailty in elderly people. CMAJ 2005; 173:489–495. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Muscedere J, Waters B, Varambally A, et al. The impact of frailty on intensive care unit outcomes: a systematic review and meta-analysis. Intensive Care Med 2017; 43:1105–1122. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47▪.Jung C, Flaatten H, Fjølner J, et al. The impact of frailty on survival in elderly intensive care patients with COVID-19: the COVIP study. Crit Care 2021; 25:1–9. [DOI] [PMC free article] [PubMed] [Google Scholar]; Observational multicentre study collectively enrolling 1348 patients ≥70 years of age with COVID-19. Frailty emerged as a predictor of 30-day mortality independent from sex, age, disease severity, treatment limitations and comorbidities.
- 48.Vincent J-L. Withdrawing may be preferable to withholding. Crit Care 2005; 9:226–229. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Mellinghoff J, Cecconi M, Maurizio C, et al. Education to save lives: C19SPACE, the COVID19 Skills PrepAration CoursE. Intensive Care Med 2022; 48:1–4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50▪.Greco M, De Corte T, Ercole A, et al. Clinical and organizational factors associated with mortality during the peak of first COVID-19 wave: the global UNITE-COVID study. Intensive Care Med 2022; 48:1–16. [DOI] [PMC free article] [PubMed] [Google Scholar]; A large multicenter international point-prevalence study describing clinical and demographical characteristics, treatments received and outcomes of patients admitted to the ICU of 46 countries both in surge and standard ICU beds. Non-ICU physician and nurses were employed in the majority of centers and admission to surge ICU beds was not associated with mortality.
- 51.RECOVERY Collaborative Group. Tocilizumab in patients admitted to hospital with COVID-19 (RECOVERY): a randomised, controlled, open-label, platform trial. Lancet 2021; 397:1637–1645. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Beigel JH, Tomashek KM, Dodd LE, et al. Remdesivir for the treatment of COVID-19 – final report. N Engl J Med 2020; 383:1813–1826. [DOI] [PMC free article] [PubMed] [Google Scholar]