Abstract
Aims
Magnetic resonance imaging (MRI) studies have identified structural and functional differences in salience network nodes of individuals with alcohol use disorders (AUDs) after chronic exposure to alcohol. However, no studies have investigated cerebral blood flow (CBF) in nontreatment-seeking (NTS) individuals with AUD.
Methods
In this work, we sought to quantify putative CBF deficits in NTS individuals relative to social drinking (SD) controls and determine if CBF in the salience network is associated with AUD severity. Fifteen NTS (36.5 ± 11.2 years old, 30.0 ± 22.7 drinks/week) and 22 SD (35.6 ± 11.9 years old, 9.1 ± 5.7 drinks/week) underwent pseudocontinuous arterial spin labeling MRI.
Results
Compared with social drinkers, NTS individuals had significantly lower CBF in the right and left dorsal anterior insula, and the left ventral anterior and posterior insula. The Alcohol Use Disorder Identification Test (AUDIT) score showed a significant negative relationship with CBF in the bilateral caudal anterior cingulate cortex. In addition, a significant negative correlation was present between number of standard drinks consumed per week and the left frontal opercular CBF.
Conclusion
These results provide evidence that insular CBF is negatively associated with heavy drinking, and that severity of alcohol use is related to CBF deficits in key nodes of the salience network. Longitudinal data are needed to understand if disruptions of CBF in the insula and the salience network are a predisposition for or a consequence of chronic AUD.
Short Summary: Non-treatment seeking alcohol use disorder participants show cerebral blood flow (CBF) deficits in the insular cortex compared with social drinkers. In addition, CBF in nodes of the salience network inversely correlates with degree of alcohol risk behaviors.
INTRODUCTION
According to the 2019 National Survey on Drug Use and Health, ~15 million Americans had an alcohol use disorder (AUD; Dept of Health and Human Services, 2019). Chronic exposure to alcohol is known to have a multitude of deleterious effects on the human body (Oscar-Berman and Marinkovic, 2007), and the number of alcohol-related deaths has doubled in the last two decades, from 35,914 in 1999 to 72,558 in 2017 (White et al., 2020). Thus, any information about the neural substrates of AUD may ultimately become useful in prevention and/or mitigation of personal and societal damage from AUD. Neuroimaging techniques such as magnetic resonance imaging (MRI) provide important opportunities to understand the neurobiological signatures of chronic alcohol exposure. There is a substantial, well-established literature on the negative effects of AUD on gray matter (Fein et al., 2002; Cardenas et al., 2007), white matter volume (Pfefferbaum and Sullivan, 2002; Cardenas et al., 2007) and white matter integrity (Pfefferbaum et al., 2000; Pfefferbaum et al., 2014), which recently has been extended to assessments of white matter connectivity (Chumin et al., 2018, 2019), resting state connectivity (Galandra et al., 2019; Halcomb et al., 2019) and task-based functional activation and connectivity (Courtney et al., 2013; Zhu et al., 2016). However, targeted assessments of regional blood flow effects in chronic alcohol use are sparse. A positron emission tomography study in older adults found that global cerebral blood flow (CBF) was lower in those who consumed >15 drinks per week compared with lighter drinkers (<1 drink per week; Christie et al., 2008). Considering the prior literature on loss of GM, disruption of WM integrity and putative alterations in blood flow in AUD (see also below), we hypothesized that perfusion deficits would be observed in AUD.
The salience network is of particular interest in the study of addiction, due to its role in recognizing relevant and/or important (salient) internal and external stimuli and subsequently eliciting an appropriate behavioral response (Goulden et al., 2014). The key regions involved in this cingulo-opercular network include the anterior insula (AI) and the anterior cingulate cortex (ACC). During the presence of salient stimuli, the AI sends a ‘bottom-up’ signal to the ACC. This signal initiates a ‘switching’ between other functional networks such as the default mode network (internally focused mode of thought), and the networks implicated in the executive control (extrinsically focused mode of thought; Goulden et al., 2014) and reward (Arienzo et al., 2020). Given that alcohol intoxication is likely a powerful internal stimulus, it is possible that functional alterations of the salience network in AUD may contribute to persistent hazardous alcohol consumption (Halcomb et al., 2019).
Several studies have demonstrated that, in nontreatment-seeking (NTS), detoxified, and abstinent AUD persons, there are deficits in cortical volume and thickness in nodes of the salience network, specifically the AI and the ACC (Durazzo et al., 2011; Galandra et al., 2018). Decrements in CBF have also been observed in these regions in detoxified AUD individuals (Sullivan et al., 2013, 2021). In addition, Durazzo et al. (2010) observed CBF deficits in the ACC of treatment-seeking individuals who had relapsed, relative to abstainers and light-drinking controls. In our previous work, we observed lower resting-state insular connectivity to frontal regions in currently-drinking AUD (Halcomb et al., 2019), which is consistent with findings in detoxified males with AUD (Sullivan et al., 2013). Taken together, the literature suggests that the structural and functional integrity of the salience network may be compromised in individuals with AUD.
At present, there is no information regarding CBF in individuals with AUD who are currently-drinking and not seeking treatment. Therefore, the aim of the present study was to use arterial spin labeling (ASL) MRI to test for differences in CBF in the salience network in a sample of NTS AUD compared with social drinking (SD) controls. Our primary hypothesis was that CBF would be lower in key regions of the salience network in NTS compared with SD. Furthermore, we hypothesized that, in nodes of the salience network, CBF would be negatively related to the degree of alcohol consumption.
MATERIALS AND METHODS
Participants
All procedures were approved by the Indiana University Institutional Review Board in accordance with the Belmont Report. Participants were recruited from the greater Indianapolis area using local advertisement and social media. All participants gave written informed consent prior to participation in the study after confirming that breath alcohol concentration was zero. Exclusion criteria were: history or presence of any psychiatric, neurological or other major medical disorder (except tobacco use disorder), current use of any psychotropic medication, positive urine pregnancy test, self-reported persistent illicit drug use at the time of interview, and contraindications of safety in the MRI scanner. NTS participants met DSM-IV criteria for alcohol dependence, had not received treatment in the last year, and were not actively seeking nor expressed interest in seeking treatment. See Table 1 for participant characteristics by group.
Table 1.
Participant demographics and characteristics
| NTS (n = 15) | SD (n = 22) | Significance | |
|---|---|---|---|
| Age | 36.5 ± 11.2 | 35.6 ± 11.9 | t(35) = −0.24, P = 0.81 |
| Education (years) | 13.9 ± 2.5 | 15.5 ± 2.3 | t(35) = 2.07, P = 0.51 |
| Gender | 9 M (60%) / 6F | 9 M (41%) / 13F | X 2 (1) = 1.30, P = 0.33 |
| Race | 10 W; 4 B; 1 AI | 15 W; 6 B; 1 As | X 2 (3) = 2.15, P = 0.54 |
| BMI | 30.9 ± 8.3 | 29.3 ± 7.0 | t(35) = 0.59, P = 0.38 |
| Smokers | 7 (47%) | 4 (18%) | X 2 (1) = 3.46, P = 0.08 |
| Age first drink | 17 ± 2.8 | 16 ± 2.8 | t(35) = 1.11, P = 0.27 |
| Drinks/week (TLFB) | 30.0 ± 22.7 | 9.1 ± 5.7 | t(35) = −4.16, P < 0.001*** |
| AUDIT | 13.1 ± 6.1 | 6.5 ± 3.5 | t(35) = −4.14, P < 0.001*** |
Data are mean ± standard deviation. NTS: nontreatment-seeking AUD; SD: social drinking control; M: male; F: female; W: White; B: Black; AI: American Indian; As: Asian; BMI: body mass index; TLFB: timeline follow-back; AUDIT: Alcohol Use Disorder Identification Test.
***< 0.001.
Participants underwent an extensive structured interview, which included: the Semi-Structured Assessment for the Genetics of Alcoholism (Bucholz et al., 1994), Alcohol Use Disorders Identification Test (AUDIT; Saunders et al., 1993), 90-day Timeline Follow-Back for alcohol use (TLFB; Sobell et al., 1988); a medical history and demographics questionnaire, an in-house substance use questionnaire, Fagerstrom Test for Nicotine Dependence (Pomerleau et al., 1994) and the Edinburgh Handedness Inventory (Oldfield, 1971). Body mass index (BMI) was calculated according to https://www.cdc.gov/nccdphp/dnpao/growthcharts/training/bmiage/page5_1.html.
Resting state fMRI data from a subset of the participants in the present study have been published elsewhere (Halcomb et al., 2019).
Image acquisition
MRI acquisition sequences were identical for all participants. Imaging was performed on a 3T Siemens MRI PRISMA scanner with a 64-channel head coil array (Siemens, Erlangen, Germany). CBF data were acquired using pseudocontinuous ASL (pCASL) labeling scheme (Wu et al., 2007) and a 3D gradient and spin echo (GRASE) acquisition (Kilroy et al., 2014) with background subtraction and the following parameters: repetition time (TR) 3840 ms, echo time (TE) 40.7 ms, flip angle 120°, turbo spin echo factor (TF) 10, labeling duration ( post label delay time (PLD) 1800 ms, field of view (FOV) 240 mm with 96 × 71 resolution, 54 axial slices, reconstructed into 2.5 × 2.5 × 2.5 mm3 voxels. The equilibrium brain tissue magnetization reference image and seven label-control measurement pairs were obtained in 7 minutes and 18 seconds. A high-resolution, T1-weighted, whole-brain magnetization prepared rapid gradient echo (MP-RAGE) image was also acquired for co-registration and normalization to the Montreal Neurological Institutes (MNI) coordinate system. Prior to the pCASL scan, participants were instructed to ‘think about nothing in particular’, to keep their eyes closed, and to remain awake.
post label delay time (PLD) 1800 ms, field of view (FOV) 240 mm with 96 × 71 resolution, 54 axial slices, reconstructed into 2.5 × 2.5 × 2.5 mm3 voxels. The equilibrium brain tissue magnetization reference image and seven label-control measurement pairs were obtained in 7 minutes and 18 seconds. A high-resolution, T1-weighted, whole-brain magnetization prepared rapid gradient echo (MP-RAGE) image was also acquired for co-registration and normalization to the Montreal Neurological Institutes (MNI) coordinate system. Prior to the pCASL scan, participants were instructed to ‘think about nothing in particular’, to keep their eyes closed, and to remain awake.
Image preprocessing and CBF quantification
pCASL images were visually inspected for artifacts, motion, and signal dropout and all passed visual inspection. Image processing was performed with Statistical Parametric Mapping SPM12, implemented in Matlab (release 18b). For each participant, pCASL data were co-registered to the first image to correct for motion, followed by co-registration to each participant’s MP-RAGE image. A participant-specific GM mask was generated with a 2-step procedure: (a) using the SPM Segment function, a GM tissue-type probability map was generated from the MP-RAGE, which was then thresholded to retain voxels with greater than 75% probability GM; and (b) the resultant GM mask was smoothed with a 6-mm full-width half-max kernel and thresholded to remove non-GM voxels (< 0.2 probability). Perfusion measures were obtained by subtracting labeled from control images as described in Aguirre et al. (2002), implemented in Matlab (https://cfn.upenn.edu/perfusion/software.htm). CBF was then calculated as described in Wang et al. (2003) and Alsop et al. (2015):
 |
where factor of 6000 converts the units from ml/g/s to customary ml/(100 g)/min, the brain/blood partition coefficient 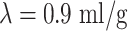 ,
, 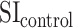 and
and  are time-averaged signal intensities in the control and label images, respectively, whereas
are time-averaged signal intensities in the control and label images, respectively, whereas  is the signal intensity of a proton density weighted image (here acquired as the first image of a pCASL scan). The labeling time parameters (
is the signal intensity of a proton density weighted image (here acquired as the first image of a pCASL scan). The labeling time parameters (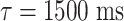 , PLD = 1800 ms) are defined in the image acquisition paragraph, the longitudinal relaxation time of blood
, PLD = 1800 ms) are defined in the image acquisition paragraph, the longitudinal relaxation time of blood  and labeling efficiency
and labeling efficiency 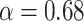 .
.
For each participant, the seven CBF measurements were averaged into a single mean volume masked by GM (mask as described above; Jahng et al., 2005). Due to a ghosting and incomplete fat suppression artifact affecting posterior areas of the brain in all participants, we constrained our analyses to a fronto-insular mask comprised of a priori Harvard-Oxford parcellation (available in FSL https://fsl.fmrib.ox.ac.uk/fsl/fslwiki/Atlases) regions of interest (ROIs) not affected by the artifact (Fig. S1). The a priori ROIs (see Section ‘Regions of interest’) were not affected by the artifact in any of the participants. Voxels with non-physiological CBF values (< 20 ml/100 g/min; Leenders et al., 1990; or > 175 ml/100 g/min) were set to zero. The latter value corresponded to the 99.5 percentile (~3.5 standard deviations above the mean) of the CBF distribution of all GM voxels across the whole sample. Next, for each participant, we calculated the mean GM CBF within the fronto-insular mask (Fig. S1; 54.8 ± 13.1 ml/100 g/min across the whole sample) for use as a covariate in the analyses to control for the natural variability in CBF. Within the fronto-insular mask, groups did not differ in mean CBF (independent t-test). Finally, deformation fields obtained from the segmentation of the MP-RAGE images were used to spatially transform the final CBF images of each participant into common (MNI) space.
Regions of interest
The insula is a large, structurally heterogenous region that is implicated in many processes. Chang et al. (2013) used a data-driven approach to divide the insular cortex into three functional subdivisions: dorsal anterior insular cortex (dAIC; higher cognitive tasks and executive control), ventral anterior insular cortex (vAIC; emotional, chemosensory and autonomic function) and posterior insular cortex (PIC; pain, sensorimotor and language). To approximate these three subdivisions for our a priori insular ROIs, we used Chang et al.’s (2013) coherence maps (thresholded to >0.8 for minimal overlap among the insular subdivisions) from the NeuroVault repository (https://neurovault.org/collections/13/; Fig. 1A). Three additional regions from the Harvard-Oxford parcellations served as negative controls: orbital frontal cortex (OFC), inferior frontal gyrus (IFG) and middle frontal gyrus (MFG). For each participant, the mean CBF values from each region (left and right hemispheres separately) were extracted for subsequent statistical analyses.
Fig. 1.

Insula and salience network masks. (A) Functional subdivisions of the insular cortex used for region of interest analysis overlaid on the Montreal Neurological Institute (MNI152) template with the respective slice coordinates. Green: posterior insula, blue: dorsal AI and yellow: ventral AI. (B) Mask of dorsal anterior insular coactivation used for SVC in the voxel-wise correlation analysis overlaid on the Montreal Neurological Institute (MNI152) template with the respective slice coordinates.
Regions of interest statistical analyses
Multivariate analysis of covariance (MANCOVA) tested for group differences in CBF, with group as the independent variable and CBF values from the six insular regions as the dependent variables. A separate MANCOVA was conducted as a negative control, and included CBF from the OFC, IFG and MFG as the dependent variables to assess whether the effects were specific to the insula. Age, education, sex, cigarette smoking status, and each participant’s mean global GM CBF values were included as covariates (IBM Statistical Package for Social Sciences—SPSS version 25). The above-listed covariates were selected due to their known effects on CBF. Where main effects were found, MANCOVA was followed by Bonferroni-corrected individual ANCOVAs (P < 0.01) to assess which insular subdivisions differed between groups.
Independent t-tests were performed to test for group differences in age, education, and recent drinking metrics (drinks consumed per week), AUDIT and age at first drink. Chi-squared tests were used to test for group differences in the ratios of sex, race and cigarette smoking status.
Voxel-wise association with AUD severity
Putative relationships between CBF and AUDIT and drinks per week were assessed in SPM12 with voxel-wise linear regression across all participants. All participants were included to ensure a sufficiently wide range of AUDIT scores and drinking behavior. Age, education, sex, cigarette smoking status and each participant’s mean GM CBF values were used as covariates in the regression. As our a priori hypotheses involved the insula, a major node in the salience network (Goulden et al., 2014), we were interested in the cortical regions that coactivate with the dorsal AI. To create an a priori mask for small volume correction (SVC), the Chang dAIC coactivation map (Chang et al., 2013) was thresholded to retain the most prominent voxels (i.e. the top 20% of values within the map; Fig. 1B). This 19,344 mm3 (2418 voxels) binary mask included the bilateral insular and anterior cingulate cortices, the major hubs of the salience network (Menon and Uddin, 2010) and the bilateral thalamus. Statistical inferences were made at voxel-level significance with family wise error (FWE, P < 0.05), corrected for multiple comparisons (SVC) within the dAIC coactivation mask.
RESULTS
Demographics
There were no significant group differences in age, education, sex, race, ethnicity, BMI, age at first drink or cigarette smoking status (Table 1). As expected, NTS reported greater number of drinks per week and scored higher on the AUDIT (Table 1).
On the day of scan, two participants tested positive for amphetamines, one tested positive for cocaine and six tested positive for THC. The distribution of those who tested positive for illicit substances did not differ between groups (four NTS and five SD).
CBF group differences—ROI analysis
MANCOVA revealed a main effect of group, such that NTS had lower CBF in the insular cortex relative to the SD group (F[6, 25] = 3.86, P = 0.007, Wilk’s Λ = 0.52). Follow-up ANCOVAs revealed that CBF was significantly lower in the left and right dAIC of NTS compared with SD (left, F[1, 30] = 21.66, P < 0.001; right, F[1, 30] = 9.12, P = 0.005). NTS also had lower CBF in the left vAIC (F[1, 30] = 8.25, P = 0.007) and left PIC (F[1, 30] = 12.55, P = 0.001) ROIs (Table 2). To ensure that these findings were not due to group differences in the fronto-insular mask volume, we calculated the number of GM voxels included in the mean CBF calculation for each participant and found no group differences in GM mask volumes. In addition, when the analysis was conducted without participants who tested positive for illicit substances on the day of the scan, the results remained significant (Table 2).
Table 2.
Group differences in CBF: univariate follow-up tests
| (A) Insular subdivision | SD (n = 22) | NTS (n = 15) | F | df | Significance (P) | Partial η2 |
|---|---|---|---|---|---|---|
| L dAIC | 61.30 ± 15.96 | 51.39 ± 15.14 | 21.66 | 1, 30 | <0.001*** | 0.42 |
| L vAIC | 55.94 ± 13.44 | 48.49 ± 14.07 | 8.25 | 1, 30 | 0.007** | 0.22 |
| L PIC | 59.00 ± 16.48 | 48.93 ± 14.38 | 12.55 | 1, 30 | 0.001** | 0.30 |
| R dAIC | 53.53 ± 13.12 | 45.90 ± 12.86 | 9.12 | 1, 30 | 0.005** | 0.23 |
| R vAIC | 44.89 ± 10.78 | 39.97 ± 11.97 | 2.32 | 1, 30 | 0.139 | 0.07 |
| R PIC | 46.21 ± 11.74 | 41.03 ± 12.28 | 3.41 | 1, 30 | 0.075 | 0.10 |
| (B) Insular subdivision | SD (n = 17) | NTS (n = 11) | F | df | Significance (P) | Partial η2 |
| L dAIC | 61.56 ± 17.78 | 52.00 ± 16.47 | 22.15 | 1, 21 | < 0.001*** | 0.51 |
| L vAIC | 56.06 ± 15.17 | 49.83 ± 14.65 | 4.25 | 1, 21 | 0.05* | 0.17 |
| L PIC | 59.71 ± 18.04 | 48.15 ± 14.66 | 19.67 | 1, 21 | < 0.001*** | 0.48 |
| R dAIC | 53.59 ± 14.54 | 46.07 ± 13.20 | 8.82 | 1, 21 | 0.007** | 0.30 |
| R vAIC | 44.61 ± 12.24 | 40.29 ± 12.58 | 1.47 | 1, 21 | 0.24 | 0.07 |
| R PIC | 45.73 ± 13.28 | 41.81 ± 13.09 | 1.25 | 1, 21 | 0.28 | 0.06 |
(A) Univariate follow-up tests in the full sample (n = 37) reveal that all functional regions of the left insula differ between groups, as well as the dorsal region of the right AI, with NTS participants with AUD having lower blood flow than SD participants. (B) Univariate follow-up tests excluding participants who tested positive for illicit drugs on scan day (n = 28). Results did not change. Data are mean ± standard deviation. AIC: anterior insular cortex; PIC: posterior insular cortex; d: dorsal; v: ventral; L: left hemisphere; R: right hemisphere.
*≤ 0.05.
**< 0.01.
***< 0.001.
MANCOVA of the CBF values from OFC, inferior frontal gyrus and middle frontal gyrus did not reveal any group differences (F[4, 27] = 1.48, P = 0.24, Wilk’s Λ = 0.18).
For data visualization purposes, we performed a voxel-wise two-sample t-test in SPM12. Consistent with our ROI findings, the maximal group differences in CBF were localized to the left dorsal AI (Fig. 2A and B).
Fig. 2.

Voxel-wise illustration of group differences in CBF. (A) Visualization of group differences in CBF in the insular cortex (display height, P < 0.005, uncorrected). The peak effect [−28 26 4] in the dorsal AI is circled. (B) Violin plot of mean CBF values for each participant from the cluster (k = 320) containing the peak effect. NTS AUD; SD controls and CBF values are displayed in units of milliliters per 100-g tissue per minute.
Associations between CBF and alcohol consumption—voxel-wise analyses
Voxel-wise linear regression within the dAIC coactivation mask was assessed using a SVC in SPM12. Across the whole sample, there was a significant negative relationship between CBF and AUDIT score in the caudal ACC (pFWE = 0.04, k = 63, peak voxel [−2 10 44]; Fig. 3A and B). We also observed a significant negative correlation between CBF and standard drinks per week in the left frontal operculum (pFWE = 0.01, k = 76, peak voxel at [−38 20 −12]; Fig. 3C and D). There were no positive correlations between CBF and either AUDIT or drinks per week.
Fig. 3.

CBF in salience network regions inversely correlates with degree of alcohol risk behaviors. (A) Linear regression between CBF and AUDIT in the caudal ACC. Peak effect at [−2 10 44] is significant (pFWE = 0.045, k = 63) after correcting for FWE within the SVC mask (see text for detail). (B) Mean CBF values from the cluster containing the peak effect plotted against AUDIT score. (C) Linear regression between CBF and standard drinks per week in the left frontal operculum (peak voxel at [−38 20 –12], pFWE = 0.013, k = 76). (D) Mean CBF values from the cluster containing the peak effect plotted against standard drinks per week. Display height, P < 0.001. AUDIT: Alcohol Use Disorder Identification Test; CBF: cerebral blood flow in units of milliliters per 100-g tissue per minute.
DISCUSSION
The aim of the present study was to explore putative CBF deficits in the salience network of NTS AUD individuals compared with SD controls. In line with our hypotheses, the NTS group had significantly lower CBF in the left and right dorsal AI, as well as the left ventral anterior and posterior insular cortices. In addition, we found that across all participants, AUDIT score was negatively correlated with CBF in the left and right caudal ACC, and that standard drinks per week was inversely associated with CBF in the left frontal operculum (FO) of the dAIC.
The present findings of lower insular blood flow in NTS individuals with AUD are consistent with the data from those recovering from AUD (Sullivan et al., 2013, 2021). Taken together, these findings suggest that deficits in anterior insular blood flow are present during active AUD and persist during detoxification and abstinence.
The insula is involved in processes that likely underlie the development and maintenance of AUD, such as impulse control (Li et al., 2009), self-regulation, stimulus salience attribution (Craig, 2002) and reward processing (Arienzo et al., 2020). Thus, it is plausible that lower insular CBF might contribute to hazardous drinking behaviors. Given that cross-sectional studies cannot provide information about causality, longitudinal studies are needed to understand whether low insular CBF is a predisposing factor for AUD or whether compromised CBF emerges as a consequence of years of heavy drinking.
To our knowledge, this is the first study to test for a putative relationship between degree of alcohol risk behavior, alcohol consumption and CBF. We observed inverse correlations between CBF in key nodes of the salience network and behavioral markers of risky drinking patterns. The negative correlation between right and left caudal ACC CBF and AUDIT scores is interesting in the context of the ACC’s role in pursuit of reward, such as focusing attention on stimuli that predict reward (Rushworth and Behrens, 2008). This result is also consistent with the finding that CBF changes in response to alcohol in this region are predictive of future alcohol problems (Courtney et al., 2019). We also observed a negative correlation between CBF in the left FO/dAIC and standard drinks consumed per week. Severity of alcohol use has been associated with lower cortical thickness in this region, such that treatment-seeking individuals who relapse have diminished cortical thickness in comparison with abstainers (Durazzo et al., 2011). Taken together, the negative association of lower blood flow in the salience network with markers of hazardous drinking may be relevant for the development and/or maintenance of AUD.
We recognize that there are limitations to this study. First, the dataset had a relatively small sample size. Second, the ghosting and incomplete fat suppression artifact present in all participants precluded our ability to investigate possible group differences in blood flow in areas such as the posterior parietal, temporal, and occipital cortices of the brain. Third, although participants were sober on the day of the MRI, recent drinking behavior could have impacted CBF. We were unable to assess any putative effects of acute withdrawal, as time since last drink prior to scan procedures was not acquired. Fourth, although no participants reported consistent use of illicit drugs, several tested positive for illicit substances on the day of scan, which hinders the attribution of observed effects solely to chronic alcohol misuse. However, exclusion of these participants from the analyses did not alter the results (P = 0.02; see Table 2). Inclusion of these participants is also ecologically valid, as many people with substance use disorders often use other substances concurrently (or recreationally) with their substance of choice. Fifth, although no participants reported falling asleep in the scanner, it is impossible to completely rule this out as a potential confound. Lastly, we must address the possibility that vascular differences between groups due to conditions such as hypercholesterolemia or atherosclerosis could have influenced our results. Although we did not measure cerebral blood volume (CBV) in the insula, the lack of group differences in CBF in negative control regions does not support the concept that a global effect of chronic drinking or smoking on CBV posed a significant confound on our CBF measurements. This is also consistent with (Desmond et al., 2020), which reported no differences in CBF or CBV in the motor cortex of chronic drinkers relative to controls.
In summary, these results provide evidence that insular CBF is compromised in currently-drinking individuals with alcohol use disorder, and that degree of hazardous drinking behavior is negatively related to blood flow in the salience network. Larger cross-sectional studies are needed to replicate this work, and longitudinal data are needed to disentangle whether blood flow in the hubs of the salience network represents a predisposition to AUD or is a consequence of protracted hazardous drinking.
Contributors
KKY designed the study; secured all regulatory approvals; obtained funding for the study; supervised all aspects of the study, including but not limited to, participant recruitment, data acquisition and processing and statistical analysis; contributed to the intellectual content of the manuscript; and approved the final version of the manuscript.
TJB assisted with participant recruitment; conducted imaging processing and quality assurance (QA) procedures; conducted the statistical analyses and provided initial drafts of the manuscript.
EJC supervised the data processing, QA, and statistical analyses, and contributed to the intellectual content of the manuscript.
JDW provided initial code for data processing as well as contributed to data processing and QA procedures.
MD supervised image preprocessing and QA; assisted with the design of the statistical analyses; and contributed intellectual content to the manuscript.
Supplementary Material
ACKNOWLEDGEMENTS
The authors would like to thank Danny JJ Wang (University of Southern California) for implementing the pCASL sequence and script reconstructing the perfusion images. We would also like to thank Dr Yu-Chien Wu and research MRI technologists of the Indiana Institute of Biomedical Imaging Sciences, Michele Dragoo, Traci Day and Robert Bryant for their assistance with imaging. We would also like to acknowledge the assistance of study coordinators Karen Hile and James Walters.
Contributor Information
Tarah J Butcher, Department of Radiology and Imaging Sciences, Indiana University School of Medicine, 355 W. 16th St., Indianapolis, IN 46202, USA; Department of Radiology and Imaging Sciences, Indiana University Center for Neuroimaging, Indiana University School of Medicine, 355 W. 16th St., Indianapolis, IN 46202, USA; Department of Psychiatry, Indiana University School of Medicine, 355 W. 16th St., Indianapolis, IN 46202, USA; Department of Psychology, Indiana University Purdue University Indianapolis, 402 N. Blackford St, Indianapolis, IN 46202, USA.
Evgeny J Chumin, Department of Radiology and Imaging Sciences, Indiana University School of Medicine, 355 W. 16th St., Indianapolis, IN 46202, USA; Department of Radiology and Imaging Sciences, Indiana University Center for Neuroimaging, Indiana University School of Medicine, 355 W. 16th St., Indianapolis, IN 46202, USA; Stark Neurosciences Research Institute, Indiana University School of Medicine, 320 W. 15th St., Indianapolis, IN 46202, USA; Department of Psychological and Brain Sciences, Indiana University, 1101 E 10th St, Bloomington, IN 47405, USA; Indiana University Network Science Institute, Indiana University, 1001 IN-45, Bloomington, IN 47408, USA.
John D West, Department of Radiology and Imaging Sciences, Indiana University School of Medicine, 355 W. 16th St., Indianapolis, IN 46202, USA; Department of Radiology and Imaging Sciences, Indiana University Center for Neuroimaging, Indiana University School of Medicine, 355 W. 16th St., Indianapolis, IN 46202, USA; Stark Neurosciences Research Institute, Indiana University School of Medicine, 320 W. 15th St., Indianapolis, IN 46202, USA.
Mario Dzemidzic, Department of Radiology and Imaging Sciences, Indiana University School of Medicine, 355 W. 16th St., Indianapolis, IN 46202, USA; Department of Radiology and Imaging Sciences, Indiana University Center for Neuroimaging, Indiana University School of Medicine, 355 W. 16th St., Indianapolis, IN 46202, USA; Department of Neurology, Indiana University School of Medicine, 355. W. 16th St., Indianapolis, IN 46202, USA.
Karmen K Yoder, Department of Radiology and Imaging Sciences, Indiana University School of Medicine, 355 W. 16th St., Indianapolis, IN 46202, USA; Department of Radiology and Imaging Sciences, Indiana University Center for Neuroimaging, Indiana University School of Medicine, 355 W. 16th St., Indianapolis, IN 46202, USA; Stark Neurosciences Research Institute, Indiana University School of Medicine, 320 W. 15th St., Indianapolis, IN 46202, USA; Department of Psychology, Indiana University Purdue University Indianapolis, 402 N. Blackford St, Indianapolis, IN 46202, USA.
Data Availability
The data underlying this article will be shared on reasonable request to the corresponding author.
Funding
This work was supported by the National Institute of Alcohol Abuse and Alcoholism grants R01 AA01835402 (KKY), R21 AA02265002 (KKY) and F31 AA025518 (EJC).
Conflict of interest statement
None of the authors have any conflicts of interest to declare.
References
- Aguirre GK, Detre JA, Zarahn E et al. (2002) Experimental design and the relative sensitivity of BOLD and perfusion fMRI. Neuroimage 15:488–500. [DOI] [PubMed] [Google Scholar]
- Alsop DC, Detre JA, Golay X et al. (2015) Recommended implementation of arterial spin-labeled perfusion MRI for clinical applications: a consensus of the ISMRM perfusion study group and the European consortium for ASL in dementia. Magn Reson Med 73:102–16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Arienzo D, Happer JP, Molnar SM et al. (2020) Binge drinking is associated with altered resting state functional connectivity of reward-salience and top down control networks. Brain Imaging Behav 14:1731–46. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bucholz KK, Cadoret R, Cloninger CR et al. (1994) A new, semi-structured psychiatric interview for use in genetic linkage studies: a report on the reliability of the SSAGA. J Stud Alcohol 55:149–58. [DOI] [PubMed] [Google Scholar]
- Cardenas VA, Studholme C, Gazdzinski S et al. (2007) Deformation-based morphometry of brain changes in alcohol dependence and abstinence. Neuroimage 34:879–87. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chang LJ, Yarkoni T, Khaw MW et al. (2013) Decoding the role of the insula in human cognition: functional parcellation and large-scale reverse inference. Cereb Cortex 23:739–49. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Christie IC, Price J, Edwards L et al. (2008) Alcohol consumption and cerebral blood flow among older adults. Alcohol 42:269–75. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chumin EJ, Goni J, Halcomb ME et al. (2018) Differences in white matter microstructure and connectivity in nontreatment-seeking individuals with alcohol use disorder. Alcohol Clin Exp Res 42:889–96. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chumin EJ, Grecco GG, Dzemidzic M et al. (2019) Alterations in white matter microstructure and connectivity in young adults with alcohol use disorder. Alcohol Clin Exp Res 43:1170–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Courtney KE, Ghahremani DG, Ray LA. (2013) Fronto-striatal functional connectivity during response inhibition in alcohol dependence. Addict Biol 18:593–604. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Courtney KE, Infante MA, Brown GG et al. (2019) The relationship between regional cerebral blood flow estimates and alcohol problems at 5-year follow-up: the role of level of response. Alcohol Clin Exp Res 43:812–21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Craig AD. (2002) How do you feel? Interoception: the sense of the physiological condition of the body. Nat Rev Neurosci 3:655–66. [DOI] [PubMed] [Google Scholar]
- Desmond JE, Rice LC, Cheng DT et al. (2020) Changes in hemodynamic response function resulting from chronic alcohol consumption. Alcohol Clin Exp Res 44:1099–111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Durazzo TC, Gazdzinski S, Mon A et al. (2010) Cortical perfusion in alcohol-dependent individuals during short-term abstinence: relationships to resumption of hazardous drinking after treatment. Alcohol 44:201–10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Durazzo TC, Tosun D, Buckley S et al. (2011) Cortical thickness, surface area, and volume of the brain reward system in alcohol dependence: relationships to relapse and extended abstinence. Alcohol Clin Exp Res 35:1187–200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fein G, Di Sclafani V, Cardenas VA et al. (2002) Cortical gray matter loss in treatment-naive alcohol dependent individuals. Alcohol Clin Exp Res 26:558–64. [PMC free article] [PubMed] [Google Scholar]
- Galandra C, Basso G, Manera M et al. (2018) Salience network structural integrity predicts executive impairment in alcohol use disorders. Sci Rep 8:14481. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Galandra C, Basso G, Manera M et al. (2019) Abnormal fronto-striatal intrinsic connectivity reflects executive dysfunction in alcohol use disorders. Cortex 115:27–42. [DOI] [PubMed] [Google Scholar]
- Goulden N, Khusnulina A, Davis NJ et al. (2014) The salience network is responsible for switching between the default mode network and the central executive network: replication from DCM. Neuroimage 99:180–90. [DOI] [PubMed] [Google Scholar]
- Halcomb ME, Chumin EJ, Goni J et al. (2019) Aberrations of anterior insular cortex functional connectivity in nontreatment-seeking alcoholics. Psychiatry Res Neuroimaging 284:21–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jahng GH, Song E, Zhu XP et al. (2005) Human brain: reliability and reproducibility of pulsed arterial spin-labeling perfusion MR imaging. Radiology 234:909–16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kilroy E, Apostolova L, Liu C et al. (2014) Reliability of two-dimensional and three-dimensional pseudo-continuous arterial spin labeling perfusion MRI in elderly populations: comparison with 15O-water positron emission tomography. J Magn Reson Imaging 39:931–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Leenders KL, Perani D, Lammertsma AA et al. (1990) Cerebral blood flow, blood volume and oxygen utilization. Normal values and effect of age. Brain 113:27–47. [DOI] [PubMed] [Google Scholar]
- Li CS, Luo X, Yan P et al. (2009) Altered impulse control in alcohol dependence: neural measures of stop signal performance. Alcohol Clin Exp Res 33:740–50. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Menon V, Uddin LQ. (2010) Saliency, switching, attention and control: a network model of insula function. Brain Struct Funct 214:655–67. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oldfield RC. (1971) The assessment and analysis of handedness: the Edinburgh inventory. Neuropsychologia 9:97–113. [DOI] [PubMed] [Google Scholar]
- Oscar-Berman M, Marinkovic K. (2007) Alcohol: effects on neurobehavioral functions and the brain. Neuropsychol Rev 17:239–57. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pfefferbaum A, Rosenbloom MJ, Chu W et al. (2014) White matter microstructural recovery with abstinence and decline with relapse in alcohol dependence interacts with normal ageing: a controlled longitudinal DTI study. Lancet Psychiatry 1:202–12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pfefferbaum A, Sullivan EV. (2002) Microstructural but not macrostructural disruption of white matter in women with chronic alcoholism. Neuroimage 15:708–18. [DOI] [PubMed] [Google Scholar]
- Pfefferbaum A, Sullivan EV, Hedehus M et al. (2000) In vivo detection and functional correlates of white matter microstructural disruption in chronic alcoholism. Alcohol Clin Exp Res 24:1214–21. [PubMed] [Google Scholar]
- Pomerleau CS, Carton SM, Lutzke ML et al. (1994) Reliability of the Fagerstrom tolerance questionnaire and the Fagerstrom test for nicotine dependence. Addict Behav 19:33–9. [DOI] [PubMed] [Google Scholar]
- Rushworth MF, Behrens TE. (2008) Choice, uncertainty and value in prefrontal and cingulate cortex. Nat Neurosci 11:389–97. [DOI] [PubMed] [Google Scholar]
- Saunders JB, Aasland OG, Babor TF et al. (1993) Development of the alcohol use disorders identification test (AUDIT): WHO collaborative project on early detection of persons with harmful alcohol consumption--II. Addiction 88:791–804. [DOI] [PubMed] [Google Scholar]
- Services, DoHaH (2019) National Survey on Drug Use and Health: Department of Health and Human Services.
- Sobell LC, Sobell MB, Leo GI et al. (1988) Reliability of a timeline method: assessing normal drinkers' reports of recent drinking and a comparative evaluation across several populations. Br J Addict 83:393–402. [DOI] [PubMed] [Google Scholar]
- Sullivan EV, Muller-Oehring E, Pitel AL et al. (2013) A selective insular perfusion deficit contributes to compromised salience network connectivity in recovering alcoholic men. Biol Psychiatry 74:547–55. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sullivan EV, Zhao Q, Pohl KM et al. (2021) Attenuated cerebral blood flow in frontolimbic and insular cortices in alcohol use disorder: relation to working memory. J Psychiatr Res 136:140–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang J, Alsop DC, Song HK et al. (2003) Arterial transit time imaging with flow encoding arterial spin tagging (FEAST). Magn Reson Med 50:599–607. [DOI] [PubMed] [Google Scholar]
- White AM, Castle I-JP, Hingson RW et al. (2020) Using death certificates to explore changes in alcohol related mortality in the United States, 1999 to 2017. Alcohol Clin Exp Res 44:178–87. [DOI] [PubMed] [Google Scholar]
- Wu WC, Fernandez-Seara M, Detre JA et al. (2007) A theoretical and experimental investigation of the tagging efficiency of pseudocontinuous arterial spin labeling. Magn Reson Med 58:1020–7. [DOI] [PubMed] [Google Scholar]
- Zhu X, Sundby K, Bjork JM et al. (2016) Alcohol dependence and altered engagement of brain networks in risky decisions. Front Hum Neurosci 10:142. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
The data underlying this article will be shared on reasonable request to the corresponding author.


