Abstract
In this study, the effects of orally administered viable Lactobacillus casei Shirota strain YIT9029 on the immunity parameters of Wistar and Brown Norway rats were examined. For this purpose, we used the Trichinella spiralis host resistance model. Two weeks before and during T. spiralis infection, rats were fed 109 viable L. casei bacteria 5 days per week. The T. spiralis-specific delayed-type hypersensitivity (DTH) response was significantly enhanced in both Wistar and Brown Norway rats given L. casei. In both rat strains fed L. casei, serum T. spiralis-specific immunoglobulin G2b (IgG2b) concentrations were also significantly increased. In the model, no significant effects of L. casei on larval counts or inflammatory reactions in the tongue musculature, body weights, or lymphoid organ weights were observed. Serum specific antibody responses, other than IgG2b, were not changed by feeding of L. casei. In contrast to L. casei, it was shown that orally administered Bifidobacterium breve or Bifidobacterium bifidum had no influence on the measured infection and immunity indices in the rat infection model. Since the rat DTH response is considered to be a manifestation of Th1 cell-mediated immunity and the IgG2b isotype has been associated with Th1 activity, it was concluded that Th1 cells could play an active role in the immunomodulatory effects of orally administered L. casei. Furthermore, our data do not indicate that the effect of oral supplementation with L. casei is dependent on the genetic background of the host.
Various strains of lactobacilli and bifidobacteria have been reported to bestow an array of health-promoting activities after either parenteral or oral administration, including improved resistance to (intestinal) infections, antimutagenic activity, control of serum cholesterol, alleviation of lactose intolerance, and positive effects on diarrhea, allergies, and autoimmunity (10, 22, 27, 28, 30, 36; for an extensive review, see reference 18). The beneficial effects of oral supplementation with lactobacilli and bifidobacteria may be accomplished by improving the gut mucosal barrier and/or innate and acquired immune responses. Improvement of the gut barrier can be due to (i) competitive exclusion, (ii) production of inhibitory compounds, and (iii) rebalancing of disturbed gastrointestinal microbial composition and metabolism. Dependent on their intrinsic properties, orally applied lactobacilli have been reported to affect T-helper 1 and T-helper 2 pathways by local cytokine production in the gut and systemic specific antibody formation (13, 14). Because of these T-helper activity-skewing properties, lactobacilli have gained a lot of interest in the research fields of oral vaccine development and immunomodulation (for a review, see reference 5). Oral administration of lactobacilli has also been shown to enhance the phagocytic activity of macrophages (7, 23). From an immunological point of view, lactobacilli may form important new strategies to combat or prevent infections with any of the increasing numbers of pathogenic bacteria with multiple resistance to antibiotics (3). The reported immunomodulating effects of orally administered lactobacilli and bifidobacteria in vivo are still rather limited but increasing.
Many studies regarding the effects of orally administered lactobacilli and bifidobacteria on the immune system have been performed in animal models (e.g., tumor, infection, and allergy models). It is feasible that the results of these studies are at least partly dependent on the models themselves; in terms of the animal species, genetic background, and type of tumor, infection, or allergy studied. For instance, the immune modulating capacity of Lactobacillus casei Shirota has been investigated mainly in various mouse models (15) and less in other species. The aim of the present study was to gain insight into the potential immunomodulating properties of orally administered L. casei Shirota strain YIT9029 in the rat. In identical parallel studies, the effects of Bifidobacterium breve YIT4065 and Bifidobacterium bifidum YIT4007 were also examined. To establish the influence of genetic background on the outcome, two genetically different rat strains were used, namely, Wistar and Brown Norway (BN) rats. Besides other genetically determined differences, BN rats have been reported to be more Th2 skewed than Wistar rats (9). The helminth Trichinella spiralis infection model was used to investigate which components of the immune system are potentially modulated by orally supplementation with L. casei. In the life cycle of T. spiralis, the encapsulated muscle larvae excyst in the acid-pepsin environment of the stomach, pass toward the jejunum, and mature within 3 to 4 days. Viviparous females penetrate the small intestinal epithelium and produce larvae. Newborn larvae migrate through the intestinal mucosa via lymphatic and blood vessels toward host striated muscle tissue, where they are encapsulated within a host-derived structure. A major advantage of the T. spiralis model is that immunity to the parasite depends on multiple cell types at the mucosal and systemic, specific and nonspecific, and humoral and cellular levels. Immunomodulation on nearly any level of the immune system can be monitored in the T. spiralis infection model (33). We report that in both Wistar and BN rats, oral administration of L. casei is able to enhance antigen-specific cellular immunity, as measured by the delayed-type hypersensitivity (DTH) response and immunoglobulin G2b (IgG2b) anti-T. spiralis antigen titers.
MATERIALS AND METHODS
Animals.
Four-week-old specific-pathogen-free male outbred Wistar (U:Wu) rats were obtained from the Utrecht University animal facilities, and inbred BN (Rij:Hsd) rats were purchased from Harlan Netherlands B.V. (Horst, The Netherlands). The rats were housed in a barrier unit under standard conditions (50 to 60% humidity, 12-h dark–12-h light cycle). The rats (two per cage) were randomly allocated and kept in filter-topped Macrolon cages with free access to standard rat chow and drinking water.
Formal permission for the animal experiments was granted by an independent ethics committee of the Utrecht University Faculty of Veterinary Medicine, as required by Dutch law.
Parasite and parasitological methods.
T. spiralis was originally isolated from an infected pig in Poland and maintained since 1960 by 6-week passages in Swiss mice at the National Institute of Public Health and the Environment, Bilthoven, The Netherlands (24). For experimental oral infections, muscle larvae were isolated from infected Swiss mice after digestion of muscle in HCl-pepsin as described elsewhere (33).
Preparation of crude T. spiralis antigen was performed as follows (6). T. spiralis larvae were homogenized in 5 ml of extraction buffer (10 mM Tris-HCl, pH 8.0; 2 mM EDTA; 2 mM phenylmethylsulfonyl fluoride; 1 μg of leupeptin per ml; 1 μg of pepstatin per ml) in Potter tubes. After centrifugation (1,000 × g) of the homogenate, the supernatant was stored. New extraction buffer (5 ml) was added, and the same procedure was repeated four times. After the last preparation, the homogenate was added to stored supernatants. This suspension was stirred for 30 min in a rotator. Subsequently, the suspension was centrifuged for 60 min at 50,000 × g. The supernatant was used as T. spiralis antigen crude extract.
Lactobacilli and bifidobacteria.
Freeze-dried powders containing L. casei Shirota strain YIT9029, B. breve strain YIT4065, and B. bifidum strain YIT4007 were obtained from the Yakult Central Institute for Microbiological Research (Tokyo, Japan). L. casei Shirota is a commercial probiotic strain which was described extensively elsewhere (15). Besides the bacteria, the prepared vehicle powder consisted of skim milk, protease, sodium glutamate, ascorbic acid, and cornstarch. In the control powder, the lactic acid bacteria were replaced with extra cornstarch. The powder was suspended in sterile distilled water, and the viability of the lactic acid bacteria (lactobacilli and bifidobacteria) was determined by aerobic culturing of rehydrated powder on MRS plates (CM361; Oxoid, Haarlem, The Netherlands). The viability of the rehydrated lactic acid bacteria was over 90%.
Experimental design.
In four separate sets of experiments the effect of orally administered lactic acid bacteria (lactobacilli and bifidobacteria) in T. spiralis-infected rats was studied (eight animals per group). In two sets of experiments, the effect of L. casei on Wistar rats was studied (the two independent studies are named experiments 1 and 2, respectively). In one set of experiments, the influence of B. breve and B. bifidum on Wistar rats was examined. In another set of experiments, the effect of L. casei on BN rats was studied.
In each study, the rats received an oral dosage of lactic acid bacteria or control medium five times per week for 6 consecutive weeks, starting 2 weeks before parasitic infection. Each dosage consisted of 109 viable lactic acid bacteria in 0.5 ml of distilled water and was administered by placing a feeding tube into the esophagus.
In all infection studies, 2 weeks after the first administration of lactic acid bacteria, the rats were infected per os with 103 viable T. spiralis muscle larvae. An infectious dose of 103 larvae has been shown to induce a subclinical infection state which is useful for monitoring of immunostimulation or immunosuppression (33). A T. spiralis antigen DTH assay was done 3 weeks after infection. Four weeks after infection, the rats were sacrificed and analyzed for serum T. spiralis-specific antibodies, number of larvae in tongue muscle tissue, severity of inflammation around muscle larvae, and body, thymus, and spleen weights.
DTH.
Three weeks after T. spiralis infection, rats were challenged with 2.5 μg of T. spiralis antigen–25 μl of phosphate-buffered saline (PBS) by intradermal injection into one ear pinna and 25 μl of PBS into the other ear pinna after determination of the initial thickness of the ear with an engineer's micrometer (Mitutoyo, Veenendaal, The Netherlands). Twenty-four hours later, the thickness of both ears was measured. In one of the two identical studies with Wistar rats and L. casei, the DTH response was also measured 48 h after an ear challenge. The swelling was calculated according to the following equation: net swelling = (T24 − T0) − (P24 − P0), where T0 is ear thickness before T. spiralis antigen challenge, T24 is ear thickness 24 h after T. spiralis antigen challenge, P0 is ear thickness before PBS injection, and P24 is ear thickness 24 h after PBS injection. The same type of equation for DTH response measurements at 48 h after challenge was performed. In noninfected rats administered L. casei or control medium, a DTH assay was also done with T. spiralis antigen to detect possible cross-reactivity of T. spiralis antigens with L. casei antigens. No net swelling was measured after a T. spiralis antigen challenge of noninfected animals (data not shown).
Histology.
The thymus, spleen, mesenteric lymph nodes, liver, and so-called Swiss rolls (17) of the duodenum, jejunum, ileum (including Peyer's patches), and colon of T. spiralis-infected rats were fixed in 10% buffered formalin and embedded in paraffin. Sections 2 to 3 μm thick were stained with hematoxylin-eosin in accordance with standard procedures and microscopically examined. The microscopic analysis was based on histopathological indices as described elsewhere (12, 35).
The larvae in two longitudinal sections through the tongue of each rat were counted under a microscope, the surface of the section was subsequently determined by a morphometric analysis system, and the result was expressed as the number of larvae per square centimeter. Histological assessment of numbers of encapsulated larvae has been found to correlate with the muscle digestion method decribed by Vos et al. (34).
The same section was scored for the severity of the inflammatory reaction around the encysted larvae based on the distribution of different cell types in the infiltrate and the number of inflammatory cells (35). A score of 0 indicates no inflammation, a score of 1 indicates minimal inflammation, a score of 2 indicates slight inflammation, a score of 3 indicates moderate inflammation, and a score of 4 indicates marked inflammation.
ELISA.
T. spiralis antigen-specific log2 serum immunoglobulin titers were measured by enzyme-linked immunosorbent assay in accordance with a slightly modified version of a procedure described previously (33). Briefly, for IgM, IgG1, IgG2a, IgG2b, and IgA, plastic 96-well microtiter plates (Costar, Cambridge, Mass.) were coated with T. spiralis antigen (2.5 μg/ml in 0.1 M bicarbonate buffer, pH 9.6) and incubated overnight at room temperature. For determination of IgE, the plates were coated with T. spiralis antigen at a concentration of 20 μg/ml. Subsequently, plates were blocked with 2% skim milk (Protifar; Nutricia, Zoetermeer, The Netherlands) in PBS–0.05% Tween 20 for 1 h at 37°C. Rat sera were diluted in PBS–0.1% skim milk–0.05% Tween 20 and incubated for 1 h at 37°C. Biotinylated mouse anti-rat IgM, IgG1, IgG2a, IgG2b, IgA, and IgE (Zymed, San Francisco, Calif.) were used as second-step antibodies, followed by incubation with streptavidin-peroxidase (Jackson ImmunoResearch Laboratories, West Grove, Pa.) and 3,3′,5,5′-tetramethylbenzidine substrate solution. Optical densities at 450 nm were read with a Titertek Multiscan apparatus (Flow Laboratories, Zwanenburg, The Netherlands).
Statistics.
To determine differences between experimental and control groups, data were analyzed with the Student t test (one sided). To analyze correlations between immunoglobulin titers and DTH response values of the same animal, the Pearson correlation test was applied. All statistical analyses were performed with SPPS software (SPPS version 7.5.2.)
RESULTS
Body weights and relative organ weights.
Oral administration of L. casei (experiments 1 and 2), B. breve, and B. bifidum had no effect on the terminal body weights and thymus-body weight and spleen-body weight ratios of T. spiralis-infected Wistar and BN rats compared to those of control animals (data not shown).
Cell-mediated immunity: T. spiralis-specific DTH response.
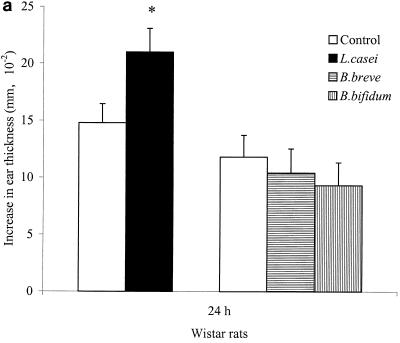
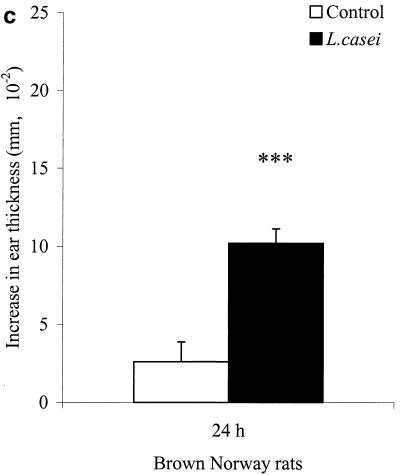
Figure 1A shows that oral L. casei administration is able to enhance the T. spiralis antigen-specific DTH response of Wistar rats at 24 h significantly (P < 0.05) compared to that of control animals (experiment 1). Oral administration of B. breve and B. bifidum did not have an effect on the T. spiralis antigen-specific DTH response of infected Wistar rats. In a second experiment with Wistar rats (experiment 2) which were fed L. casei, again a significantly augmented T. spiralis antigen-specific DTH response was observed at 24 h (P < 0.05) but also at 48 h (P < 0.01, as shown in Fig. 1B. The difference in DTH responses between L. casei-fed and control animals was even larger at 48 h.
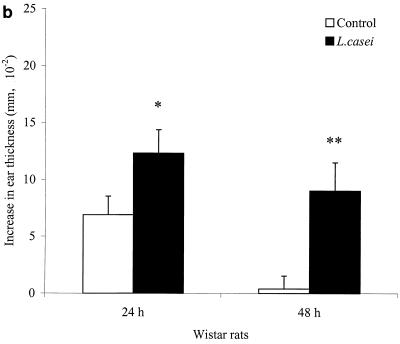
FIG. 1.
Effects of L. casei, B. breve, and B. bifidum ingestion on the T. spiralis-specific DTH response, at 24 h in experiment 1 (a) and at 24 and 48 h in experiment 2 (b), after a challenge of Wistar rats and BN rats (c). Following 3 weeks after oral T. spiralis infection, DTH responses were measured as the differences in ear thickness before and 24 to 48 h after an intradermal challenge. Bars represent the means ± the standard errors of eight rats per group for control rats and L. casei-, B. breve-, and B. bifidum-fed rats. Some values are significantly different from those of control rats (∗, P < 0.05; ∗∗, P < 0.01; ∗∗∗, P < 0.001), as calculated with Student’s t test.
The effects of L. casei were also studied in BN rats to examine possible rat strain-dependent immunological influences. An enhanced DTH response was also obtained with BN rats, as shown in Fig. 1C. L. casei-fed BN rats showed a significantly increased DTH response (P < 0.001) compared to the control group. The DTH response values of BN rats showed a tendency to be lower than those of Wistar rats.
Histology.
Microscopic analysis of the thymus, spleen, mesenteric lymph nodes, and gastrointestinal tract (including Peyer's patches) showed no influence of L. casei (experiments 1 and 2), B. breve, and B. bifidum on infected Wistar and BN rats compared to control rats.
Humoral immunity: serum T. spiralis-specific immunoglobulins.
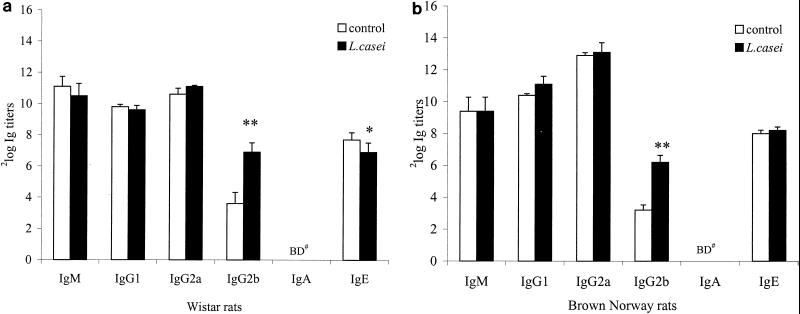
In both Wistar (experiments 1 and 2) and BN rats, it was shown that oral administration of L. casei significantly enhanced serum IgG2b anti-T. spiralis titers (P < 0.01). The results for Wistar (experiment 1) and BN rats are shown in Fig. 2A and B). Other isotypes were not different between the experimental and control animals of both strains. In only one (experiment 1) of two experiments with Wistar rats was it found that ingestion of L. casei significantly decreased T. spiralis-specific IgE titers (Fig. 2A, P < 0.05). Further, it was found that consumption of B. breve and B. bifidum did not influence serum T. spiralis antigen-specific antibody titers compared to those of the control group (data not shown). Enhancement of the antigen-specific IgG2b concentration in serum was significantly correlated with the enhanced DTH responses of Wistar rats at 48 h (experiment 2, r = 0.563, P < 0.05), but not at 24 h (experiments 1 and 2), and for BN rats at 24 h (r = 0.693, P < 0.01). No correlation between the other isotypes and the DTH response of either rat strain was observed.
FIG. 2.
T. spiralis antigen-specific serum antibody titers. In two sets of experiments, Wistar (A) and BN rats (B) were orally infected with 1,000 T. spiralis muscle larvae. The results of experiment 1 (A) are representative of both Wistar rat experiments 1 and 2. Each group consisted of eight rats that were fed L. casei or control medium. Four weeks after infection, serum was collected and T. spiralis antigen-specific log2 IgM, IgG1, IgG2a, IgG2b, IgA, and IgE titers were measured. Data are presented as means ± standard errors. ∗ and ∗∗, P <0.05 and P < 0.01, respectively, versus the control group, as calculated by Student's t test. BD#, below detection limit (<2 log2 titer).
Host resistance: pathology and number of muscle larvae.
In general, no effect of L. casei ingestion on parasite load was found, although in experiment 2, a small increase in the number of encysted muscle larvae was obeserved (Table 1, P < 0.05), which was not reproducible. Also, B. breve and B. bifidum failed to significantly affect the parasite load (Table 1). In all Wistar rat experiments, no effect of L. casei (experiments 1 and 2), B. breve, or B. bifidum on inflammatory reactions around the encysted larvae was found. In T. spiralis-infected BN rats, we observed no effect of oral L. casei administration on encysted muscle larva counts and inflammation scores compared to those of control animals. It was shown that the number of muscle larvae in BN rats was markedly higher than that in Wistar rats.
TABLE 1.
Parasite pathology of tongue musculature of T. spiralis-infected ratsa
| Rat strain and treatment | Mean result for indicated expt and group ± SD
|
|
|---|---|---|
| No. of larvae/cm2 | Inflammation score | |
| Wistar (expt 1) | ||
| Control | 41 ± 21 | 2.9 ± 0.8 |
| L. casei | 53 ± 27 | 2.8 ± 0.9 |
| Wistar (expt 2) | ||
| Control | 61 ± 11 | 2.9 ± 0.7 |
| L. casei | 80 ± 24b | 3.0 ± 0.8 |
| Wistar | ||
| Control | 114 ± 20 | 2.8 ± 0.9 |
| B. breve | 141 ± 32 | 2.8 ± 0.7 |
| B. bifidum | 153 ± 38 | 2.9 ± 0.9 |
| BN | ||
| Control | 436 ± 88 | 2.9 ± 0.7 |
| L. casei | 454 ± 116 | 2.9 ± 0.9 |
Wistar and BN rats were orally infected with T. spiralis muscle larvae. Each group consisted of eight rats that were fed L. casei, B. breve, B. bifidum, or control medium. At 28 days after infection, the number of encysted T. spiralis larvae and the severity of the inflammatory reaction around encysted larvae in the tongue musculature were measured.
Significantly different (P < 0.05) from the control group value as calculated by Student's t test.
DISCUSSION
Immunomodulation by orally administered lactobacilli or bifidobacteria can be studied by the application of so-called animal host resistance infection models in which the infection and host resistance indices very due to immune enhancement or suppression. In the rat model of T. spiralis, we studied the immunomodulating capacity of orally administered L. casei Shirota YIT9029, as well as two strains of bifidobacteria. Since it likely that the effects of orally administered bacteria on the immune system are at least partly dependent on host genetics, we used two genetically different strains of rats in our studies, namely, Wistar and BN rats. In rats, as well as mice and humans, immunological responsiveness is largely determined by T-helper cells. T-helper cells have been divided into Th1 and Th2 cells based on different cytokine expression profiles. The balance between Th1 and Th2 cells is dependent on genetic and environmental factors. In rats, Th1 cells produce interleukin-2 and gamma interferon and preferentially induce the production of IgG2b whereas Th2 cells produce interleukin-4 and stimulate IgG1 and IgE production (8, 25, 26). BN rats are representative of rats which tend to respond immunologically in a more Th2-skewed fashion than Wistar rats (9, 32). In the present study, it was shown that BN and Wistar rats respond differently to equivalent T. spiralis infections. Control BN rats have higher muscle larva yields and lower DTH responses at 24 h compared to control Wistar rats.
Five times per week, oral supplementation with 109 viable L. casei bacteria, in contrast to B. breve and B. bifidum, significantly enhanced antigen-specific cell-mediated immunity, as measured by the DTH reaction. Also, it was shown that ingestion of L. casei significantly enhanced antigen-specific humoral immunity, as measured by increased serum IgG2b levels. The enhanced antigen-specific DTH reaction and serum IgG2b response appeared not to be dependent on the genetic background of the rats, since the effects were found in both Wistar and BN rats.
The interaction of lactobacilli with T. spiralis infection has been described in earlier studies. Stefanski and Przyjalkowski showed that monoassociation of mice with various rat lactobacilli was not favorable to the establishment of T. spiralis in the intestines compared to those of control conventional animals (29). Moreover, in a recent study, intraperitoneal administration of L. casei has been demonstrated to enhance protective immunity to T. spiralis in NIH mice (2). In our studies, in which the lactobacilli and bifidobacteria were administered orally instead of intraperitoneally, as described in the study by Bautista-Garfias et al., no definite effects on pathology could be found, despite the enhanced DTH and IgG2b responses directed against T. spiralis antigen. This might support the view that protective immunity and DTH are not necessarily coupled, in contrast to reports that the two responses appear to be linked. In Listeria monocytogenes- and Mycobacterium tuberculosis-infected mice, a dissociation was observed between cells mediating cellular acquired resistance and those mediating DTH (20, 31). In the parasite model with Schistosoma mansoni, it was shown that hypersensitivity and resistance are also not coupled since UVB suppressed the hypersensitivity response but not resistance to the parasite (19). Recently, Orme and Cooper proposed that protective immune reactions and DTH responses are based upon separate mechanisms, which are interleukin and chemokine driven, respectively (21). Nevertheless, it is important to consider that the DTH reaction is an effector response, which in our studies cannot result in enhanced protection, since the rats are killed after a primary infection. It is known that a challenge infection will induce rapid expulsion of T. spiralis from the intestinal tract. T cells and immunoglobulins are important mediators of rapid expulsion (1). Reinfection of the rats with T. spiralis might possibly elucidate a protective role for the cells involved in the enhanced DTH reaction and elevated serum IgG2b.
Our data strongly suggest that ingestion of L. casei Shirota augments cellular, as well as humoral, adaptive immunity by enhancement of Th1 cell activity. The enhancement of antigen-specific IgG2b in serum indicated that ingestion of L. casei elevated Th1 activity. It has been reported that in rats, switching to IgG2b is dependent on Th1 cytokines (8). The enhanced activity of Th1 cells was also reflected in the significantly reduced IgG1/IgG2b ratios (P < 0.01) after oral administration of L. casei to both Wistar (experiments 1 and 2) and BN rats. No significant effects of L. casei on Th2 cell-related immunoglobulin isotypes IgG1 and IgE were found. Only in experiment 1 with Wistar rats was a significantly decreased T. spiralis-specific serum IgE response observed in the experimental L. casei-fed group. The DTH response is a complex Th1 cell-mediated immune response characterized by swelling, induration, and erythema which appears only several hours after a challenge and comes to a maximum after 24 to 48 h (4). Local memory Th1 cells which release cytokines and chemokines upon an antigen challenge activate monocytes/macrophages and other phagocytic immunocytes and attract them to the site of the challenge. In previous studies with T. spiralis-infected rats, it was shown that the T. spiralis-specific DTH response is predominately a Th1-type reaction (32). Our data do not contradict the notion that the T. spiralis-specific DTH reaction is a Th1-type cell-mediated response. The DTH responses in infected control BN rats are lower compared to those of control Wistar rats with respect to the immunological skewedness of those rat strains. Furthermore, DTH and IgG2b values were significantly correlated in both Wistar and BN rats after oral L. casei administration, which might imply a common cause of the enhanced Th1 activity observed.
It remains to be determined which immune cells and factors (e.g., cytokines) contribute to the established increase in the DTH and serum IgG2b responses that occurs after oral administration of L. casei. L. casei could have a direct effect on the number and activity of antigen-specific Th1 memory cells and/or Th1 cell activity is indirectly enhanced by the modulation of other innate immune cell types, such as phagocytes (7, 23).
Next to our rat studies, in previous in vivo and in vitro studies with mice, it also has been reported that L. casei Shirota has the capacity to stimulate Th1 activity (11, 16, 27). These studies did not indicate a direct effect of L. casei Shirota on T cells but rather suggest that oral administration of L. casei leads to a prominent state of cellular innate immunity via phagocyte activation, which subsequently enhances Th1 cell activity (11). It can be concluded from studies with mice and our data obtained with two rat strains that L. casei Shirota has a genuine, direct or indirect, effect on Th1 cell activity. This effect appears to be independent of the host species and its genetic background.
ACKNOWLEDGMENTS
This work was partly supported by the Dutch Ministry of Economic Affairs.
We commemorate stimulating discussions with the late J. H. J. Huis in ‘t Veld and thank J. E. van Dijk, E. Claassen, and M. V. Herias for critically reviewing the manuscript.
REFERENCES
- 1.Ahmad A, Bell R G, Wang C H, Sacuto F R. Characterization of the thoracic duct T-helper cells that co-mediate, with antibody, the rapid expulsion of Trichinella spiralis in adult rats. Parasite Immunol. 1991;13:147–159. doi: 10.1111/j.1365-3024.1991.tb00271.x. [DOI] [PubMed] [Google Scholar]
- 2.Bautista-Garfias C R, Ixta O, Orduna M, Martinez F, Aguilar B, Cortes A. Enhancement of resistance in mice treated with Lactobacillus casei: effect on Trichinella spiralis infection. Vet Parasitol. 1999;80:251–260. doi: 10.1016/s0304-4017(98)00210-6. [DOI] [PubMed] [Google Scholar]
- 3.Bengmark S. Ecological control of the gastrointestinal tract. The role of probiotic flora. Gut. 1998;42:2–7. doi: 10.1136/gut.42.1.2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Black C A. Delayed type hypersensitivity: current theories with an historic perspective. Dermatol Online J. 1999;5:7. [PubMed] [Google Scholar]
- 5.Boersma W J A, Shaw M, Claassen E. Probiotic bacteria as live oral vaccines. Lactobacillus as the versatile delivery system. In: Fuller R, Perdigon G, editors. Probiotics 3: immunomodulation by the gut microflora and probiotics. Dordrecht, The Netherlands: Kluwer Academic Publishers; 2000. pp. 234–270. [Google Scholar]
- 6.Garssen J, Norval M, Crosby J, Dortant P, Van Loveren H. The role of urocanic acid in UVB-induced suppression of immunity to Trichinella spiralis infection in the rat. Immunology. 1999;96:298–306. doi: 10.1046/j.1365-2567.1999.00698.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Gill H S, Rutherfurd K J, Prasad J, Gopal P K. Enhancement of natural and acquired immunity by Lactobacillus rhamnosus (HN001), Lactobacillus acidophilus (HN017) and Bifidobacterium lactis (HN019) Br J Nutr. 2000;83:167–176. doi: 10.1017/s0007114500000210. [DOI] [PubMed] [Google Scholar]
- 8.Gracie J A, Bradley J A. Interleukin-12 induces interferon-gamma-dependent switching of IgG alloantibody subclass. Eur J Immunol. 1996;26:1217–1221. doi: 10.1002/eji.1830260605. [DOI] [PubMed] [Google Scholar]
- 9.Hylkema M N, van der Deen M, Pater J M, Kampinga J, Nieuwenhuis P, Groen H. Single expression of CD45RC and RT6 in correlation with T-helper 1 and T-helper 2 cytokine patterns in the rat. Cell Immunol. 2000;199:89–96. doi: 10.1006/cimm.1999.1607. [DOI] [PubMed] [Google Scholar]
- 10.Kato I, Endo-Tanaka K, Yokokura T. Suppressive effects of the oral administration of Lactobacillus casei on type II collagen-induced arthritis in DBA/1 mice. Life Sci. 1998;63:635–644. doi: 10.1016/s0024-3205(98)00315-4. [DOI] [PubMed] [Google Scholar]
- 11.Kato I, Tanaka K, Yokokura T. Lactic acid bacterium potently induces the production of interleukin-12 and interferon-gamma by mouse splenocytes. Int J Immunopharmacol. 1999;21:121–131. doi: 10.1016/s0192-0561(98)00072-1. [DOI] [PubMed] [Google Scholar]
- 12.Kuper C F, Harleman J H, Richter-Reichelm H B, Vos J G. Histopathologic approaches to detect changes indicative of immunotoxicity. Toxicol Pathol. 2000;28:454–466. doi: 10.1177/019262330002800317. [DOI] [PubMed] [Google Scholar]
- 13.Maassen C B, van Holten J C, Balk F, Heijne den Bak-Glashouwer M J, Leer R, Laman J D, Boersma W J, Claassen E. Orally administered Lactobacillus strains differentially affect the direction and efficacy of the immune response. Vet Q. 1998;20(Suppl. 3):S81–S83. [PubMed] [Google Scholar]
- 14.Maassen C B, van Holten-Neelen C, Balk F, Heijne den Bak-Glashouwer M, Leer R J, Laman J D, Boersma W J, Claassen E. Strain-dependent induction of cytokine profiles in the gut by orally administered Lactobacillus strains. Vaccine. 2000;18:2613–2623. doi: 10.1016/s0264-410x(99)00378-3. [DOI] [PubMed] [Google Scholar]
- 15.Matsuzaki T. Immunomodulation by treatment with Lactobacillus casei strain Shirota. Int J Food Microbiol. 1998;41:133–140. doi: 10.1016/s0168-1605(98)00046-4. [DOI] [PubMed] [Google Scholar]
- 16.Matsuzaki T, Yamazaki R, Hashimoto S, Yokokura T. The effect of oral feeding of Lactobacillus casei strain Shirota on immunoglobulin E production in mice. J Dairy Sci. 1998;81:48–53. doi: 10.3168/jds.S0022-0302(98)75549-3. [DOI] [PubMed] [Google Scholar]
- 17.Moolenbeek C, Ruitenberg E J. The “Swiss roll:” a simple technique for histological studies of the rodent intestine. Lab Anim. 1981;15:57–59. doi: 10.1258/002367781780958577. [DOI] [PubMed] [Google Scholar]
- 18.Naidu A S, Bidlack W R, Clemens R A. Probiotic spectra of lactic acid bacteria (LAB) Crit Rev Food Sci Nutr. 1999;39:13–126. doi: 10.1080/10408699991279187. [DOI] [PubMed] [Google Scholar]
- 19.Noonan F P, Lewis F A. UVB-induced immune suppression and infection with Schistosoma mansoni. Photochem Photobiol. 1995;61:99–105. doi: 10.1111/j.1751-1097.1995.tb09249.x. [DOI] [PubMed] [Google Scholar]
- 20.Orme I M, Collins F M. Adoptive protection of the Mycobacterium tuberculosis-infected lung. Dissociation between cells that passively transfer protective immunity and those that transfer delayed-type hypersensitivity to tuberculin. Cell Immunol. 1984;84:113–120. doi: 10.1016/0008-8749(84)90082-0. [DOI] [PubMed] [Google Scholar]
- 21.Orme I M, Cooper A M. Cytokine/chemokine cascades in immunity to tuberculosis. Immunol Today. 1999;20:307–312. doi: 10.1016/s0167-5699(98)01438-8. [DOI] [PubMed] [Google Scholar]
- 22.Perdigon G, Alvarez S, Rachid M, Aguero G, Gobbato N. Immune system stimulation by probiotics. J Dairy Sci. 1995;78:1597–1606. doi: 10.3168/jds.S0022-0302(95)76784-4. [DOI] [PubMed] [Google Scholar]
- 23.Perdigon G, de Macias M E, Alvarez S, Oliver G, de Ruiz Holgado A A. Effect of perorally administered lactobacilli on macrophage activation in mice. Infect Immun. 1986;53:404–410. doi: 10.1128/iai.53.2.404-410.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Ruitenberg E J, Elgersma A, Kruizinga W, Leenstra F. Trichinella spiralis infection in congenitally athymic (nude) mice. Parasitological, serological and haematological studies with observations on intestinal pathology. Immunology. 1977;33:581–587. [PMC free article] [PubMed] [Google Scholar]
- 25.Saoudi A, Bernard I, Hoedemaekers A, Cautain B, Martinez K, Druet P, De Baets M, Guery J C. Experimental autoimmune myasthenia gravis may occur in the context of a polarized Th1- or Th2-type immune response in rats. J Immunol. 1999;162:7189–7197. [PubMed] [Google Scholar]
- 26.Saoudi A, Kuhn J, Huygen K, de Kozak Y, Velu T, Goldman M, Druet P, Bellon B. TH2 activated cells prevent experimental autoimmune uveoretinitis, a TH1-dependent autoimmune disease. Eur J Immunol. 1993;23:3096–3103. doi: 10.1002/eji.1830231208. [DOI] [PubMed] [Google Scholar]
- 27.Shida K, Makino K, Morishita A, Takamizawa K, Hachimura S, Ametani A, Sato T, Kumagai Y, Habu S, Kaminogawa S. Lactobacillus casei inhibits antigen-induced IgE secretion through regulation of cytokine production in murine splenocyte cultures. Int Arch Allergy Immunol. 1998;115:278–287. doi: 10.1159/000069458. [DOI] [PubMed] [Google Scholar]
- 28.Shu Q, Lin H, Rutherfurd K J, Fenwick S G, Prasad J, Gopal P K, Gill H S. Dietary Bifidobacterium lactis (HN019) enhances resistance to oral Salmonella typhimurium infection in mice. Microbiol Immunol. 2000;44:213–222. doi: 10.1111/j.1348-0421.2000.tb02486.x. [DOI] [PubMed] [Google Scholar]
- 29.Stefanski W, Przyjalkowski Z. Effect of alimentary tract microorganisms on the development of Trichinella spiralis in mice. II. Exp Parasitol. 1966;18:92–98. doi: 10.1016/0014-4894(66)90011-7. [DOI] [PubMed] [Google Scholar]
- 30.Takagi A, Matsuzaki T, Sato M, Nomoto K, Morotomi M, Yokokura T. Inhibitory effect of oral administration of Lactobacillus casei on 3-methylcholanthrene-induced carcinogenesis in mice. Med Microbiol Immunol. 1999;188:111–116. doi: 10.1007/s004300050112. [DOI] [PubMed] [Google Scholar]
- 31.Tsukada H, Kawamura I, Arakawa M, Nomoto K, Mitsuyama M. Dissociated development of T cells mediating delayed-type hypersensitivity and protective T cells against Listeria monocytogenes and their functional difference in lymphokine production. Infect Immun. 1991;59:3589–3595. doi: 10.1128/iai.59.10.3589-3595.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Vandebriel R J, Meredith C, Scott M P, van Dijk M, van Loveren H. Interleukin-10 is an unequivocal Th2 parameter in the rat, whereas interleukin-4 is not. Scand J Immunol. 2000;52:519–524. doi: 10.1046/j.1365-3083.2000.00804.x. [DOI] [PubMed] [Google Scholar]
- 33.Van Loveren H, Luebke R W, Vos J G. Assessment of immunotoxicity with the parasitic infection model Trichinella spiralis. In: Burleson G R, Dean J H, Munson A E, editors. Methods in immunotoxicology. Vol. 2. New York, N.Y: Wiley-Liss; 1995. pp. 243–274. [Google Scholar]
- 34.Vos J G, De Klerk A, Krajnc E I, Van Loveren H, Rozing J. Immunotoxicity of bis(tri-n-butyltin)oxide in the rat: effects on thymus-dependent immunity and on nonspecific resistance following long-term exposure in young versus aged rats. Toxicol Appl Pharmacol. 1990;105:144–155. doi: 10.1016/0041-008x(90)90366-3. [DOI] [PubMed] [Google Scholar]
- 35.Vos J G, Ruitenberg E J, Van Basten N, Buys J, Elgersma A, Kruizinga W. The athymic nude rat. IV. Immunocytochemical study to detect T-cells, and immunological and histopathological reactions against Trichinella spiralis. Parasite Immunol. 1983;5:195–215. doi: 10.1111/j.1365-3024.1983.tb00737.x. [DOI] [PubMed] [Google Scholar]
- 36.Yasui H, Kiyoshima J, Hori T, Shida K. Protection against influenza virus infection of mice fed Bifidobacterium breve YIT4064. Clin Diagn Lab Immunol. 1999;6:186–192. doi: 10.1128/cdli.6.2.186-192.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]