Abstract
Altered neurocognitive functioning is a key feature of attention-deficit/hyperactivity disorder (ADHD), and increasing numbers of studies assess task-based functional connectivity in the disorder. We systematically reviewed and critically appraised functional magnetic resonance imaging (fMRI) task-based functional connectivity studies in ADHD. A systematic search conducted up to September 2020 found 34 studies, including 51 comparisons. Comparisons were divided into investigations of ADHD neuropathology (37 comparing ADHD and typical development, 2 comparing individuals with ADHD and their nonsymptomatic siblings, 2 comparing remitted and persistent ADHD, and 1 exploring ADHD symptom severity) and the effects of interventions (8 investigations of stimulant effects and 1 study of fMRI neurofeedback). Large heterogeneity in study methodologies prevented a meta-analysis; thus, the data were summarized as a narrative synthesis. Across cognitive domains, functional connectivity in the cingulo-opercular, sensorimotor, visual, subcortical, and executive control networks in ADHD consistently differed from neurotypical populations. Furthermore, literature comparing individuals with ADHD and their nonsymptomatic siblings as well as adults with ADHD and their remitted peers showed ADHD-related abnormalities in similar sensorimotor and subcortical (primarily striatal) networks. Interventions modulated those dysfunctional networks, with the most consistent action on functional connections with the striatum, anterior cingulate cortex, occipital regions, and midline default mode network structures. Although methodological issues limited many of the reviewed studies, the use of task-based functional connectivity approaches has the potential to broaden the understanding of the neural underpinnings of ADHD and the mechanisms of action of ADHD treatments.
Keywords: ADHD, Attention-deficit/hyperactivity disorder, Connectivity, fMRI, Functional magnetic resonance imaging, Task-based functional connectivity
Attention-deficit/hyperactivity disorder (ADHD) is a neurodevelopmental disorder defined by age-inappropriate levels of hyperactivity, impulsivity, and/or inattention (1). ADHD is associated with impairments in various “hot” and “cool” executive functions (2, 3, 4, 5). The neural underpinnings of these behavioral problems include hypoactivation in frontostriatal and temporoparietal domain-relevant regions (6, 7, 8, 9, 10, 11, 12), which have been associated with disorder severity (13, 14, 15), cognitive performance (13,16), and symptomatic improvement with treatment (17,18) and can be modulated with pharmacotherapy (19). However, a recent meta-analysis highlighted the lack of convergence of brain activation alterations in ADHD (20), perhaps reflecting a failure to consider the interconnected nature of neural processing.
As most complex cognitive functions depend on information processing in multiple regions, studying regional interactions is crucial in characterizing brain function. Furthermore, given the large-scale neural reorganization in youth, investigations of functional connectivity may provide a better understanding of neurodevelopmental disorders (21, 22, 23). Consequently, many studies in ADHD focused on network-wide alterations in resting-state connectivity to characterize domain-independent neural function (24, 25, 26). Assessments of task-based functional connectivity, however, allow these findings to be extended by investigating functional connections specific to distinct cognitive processes (27). Given the presence of discrete cognitive deficits in ADHD, studies of task-based connectivity in ADHD are becoming increasingly common.
Several systematic reviews and meta-analyses examined differences in cognition-related activation (6, 7, 8, 9, 10, 11, 12,20,28,29) and connectivity during resting-state paradigms in ADHD (24, 25, 26). Although reviews of functional connectivity have been published (30, 31, 32, 33, 34), there have been no systematic evaluations of task-based functional connectivity literature of ADHD or its quality. Consequently, this review focused on functional networks in ADHD aiming to provide a framework for considering the neural correlates of the disorder accommodating context-dependent, correlated activity across brain regions and its modulation with interventions. Furthermore, given the recent advances in understanding the limitations of functional magnetic resonance imaging (fMRI), this review aimed to appraise the quality of studies and reporting practices in the field.
Methods and Materials
This preregistered review (https://www.crd.york.ac.uk/PROSPERO/display_record.php?RecordID=205500) was conducted according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines (35).
Information Sources and Search Strategy
A systematic search was conducted using the Cochrane Library, Embase, PubMed/MEDLINE, PsycINFO, and Web of Science Core Collection identifying fMRI studies of task-based functional connectivity in ADHD. The search was undertaken by one investigator (OSK) with keywords approved by the study team. The search string included (functional connectivity or connecti∗) and (ADHD or attention deficit hyperactivity disorder or attention deficit disorder or hyperkinetic) and (functional magnetic resonance imaging or fMRI or BOLD or blood oxygen level dependent). The search was limited to articles published in English between January 1990 and September 2020. Additionally, reference lists of past reviews focusing on functional connectivity in ADHD (30, 31, 32, 33, 34) were screened for relevant publications.
Study Selection Criteria
The identified citations were uploaded onto CADIMA (36). Duplicates were removed semiautomatically using CADIMA’s inbuilt function and reviewed manually by one investigator (OSK). Titles and abstracts and subsequently full texts of surviving records were screened for eligibility in parallel by two investigators (OSK and MC). A screening exercise was conducted on 20 randomly selected records ensuring good reliability between investigators [κ = 0.63, calculated according to measuring agreement of Cochrane Version 5.1 (37)]. Only peer-reviewed fMRI studies of task-based functional connectivity in patients of all ages, sexes, and races/ethnicities where ADHD (per DSM or ICD) was the primary diagnosis were retained. Discrepancies were resolved by consensus.
Exclusion Criteria
Studies were excluded if they did not assess fMRI task-based functional connectivity, did not present primary data, or were not published in a peer-reviewed journal. Studies comparing ADHD solely with other psychiatric/neurodevelopmental disorders, including participants without a formal ADHD diagnosis, recruiting only ADHD remitters, or including participants for whom ADHD was not the primary diagnosis were excluded.
Data Extraction and Critical Appraisal
Data were extracted by two investigators (OSK and MC). Records were divided into two equal-sized batches, one for each investigator. The investigators independently extracted data from their allocated studies and cross-checked the accuracy of the other investigator’s extraction. Data pertaining to 1) the study sample (sample size, age, sex, medication history, ADHD presentation, comorbidities); 2) study methods (connectivity estimation method, motion correction [method and exclusion criteria], drug washout period, task, case-control matching criteria); and 3) functional connectivity findings (changes of connectivity [increases/decreases] and their manuscript-defined location in the brain and justification of method used [e.g., choice of seed region]) were extracted and critically appraised. We defined decreased or increased functional connectivity if a hub/network was found in the group contrast in at least two comparisons. Findings were defined as mixed when the hub/network was observed in increased and decreased connectivity.
Additionally, risk of bias in intervention studies was examined using the Cochrane Collaboration risk of bias tool (37) across selection, performance, detection, attrition, and reporting biases. Two investigators (OSK and MC) independently conducted critical appraisal. Records were divided into two equal-sized batches, each assigned to one investigator.
Results
Study Selection
The search yielded 946 unique records, of which 802 were excluded during title and abstract screening. A further 110 were excluded after full-text screening for one or more of the following reasons: 1) not measuring fMRI task-based connectivity (n = 87), 2) no peer review (n = 39), 3) not assessing individuals with current primary ADHD diagnosis (n = 25), 4) not presenting an empirical investigation (n = 20), and 5) no available full text (n = 1). Following the selection process, 34 studies remained (Figure 1; Supplement lists included studies).
Figure 1.
Study selection flow chart. ADHD, attention-deficit/hyperactivity disorder; fMRI, functional magnetic resonance imaging.
These 34 studies included 51 comparisons. Of these, 37 investigated differences between ADHD and neurotypical groups, 9 tested effects of interventions in patients, 2 compared individuals with ADHD and their nonsymptomatic siblings, 2 compared remitted and persistent ADHD, and 1 explored ADHD symptom severity (investigations of siblings, remitters, and disorder severity are described in the Supplement and Table 2). Across all studies, this review included 981 individuals with ADHD, 38 ADHD remitters, 134 nonsymptomatic siblings of individuals with ADHD, and 774 neurotypical controls.
Table 2.
Studies Investigating fMRI Functional Connectivity Differences Between Individuals With ADHD and Nonsymptomatic Siblings and Persisters and Remitters and Exploring the Impact of Symptom Severity
| Study | Analysis Method | Task (Contrast) | NADHD (% Male) | AgeADHD, Years, Mean (SD) | Medication History | Medication Washout | ADHD Comorbidities | Comparison Group | NComparison (% Male) | AgeComparison, Years, Mean (SD) | Comparison > ADHD | ADHD > Comparison |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Clerkin et al., 2013 (66) | PPISPM | Cued reaction time task (cues > noncues) | 16 (75%) | 24.44 (2.02) | Current stimulant use (6%); past stimulant use but medication-free at time of study (71%) | >48 hours | Mood disorder (23%), anxiety disorder (23%), substance use disorder (43%) | Remitters | 19 (90%) | 24.74 (2.1) | R thalamus ↔ BIL frontal pole, L DLPFC | None |
| Kolodny et al., 2020 (114) | gPPIFSLa | GNG (rare no-go > prevalent no-go) | 37 (41%) | 26.6 (4) | Current stimulant use (84%); medication-free (16%) | >24 hours | None | – | – | – | L IPS ↔ R IFC, postcentral/SPG (negatively related to symptom severity) | |
| Luo et al., 2018 (40) | GTTs | Cued attention task (cues) | 17 (77%) | 24.55 (2.2) | Current stimulant use (12%); past stimulant use (unspecified) | 48 hours | None | Remitters | 19 (84%) | 24.79 (2.2) | Acting network hubs in R MFG, globus pallidus, putamen; nodal efficiency in BIL MFG | Acting network hubs in L MFG and precentral |
| Mulder et al., 2011 (50) | SBC | GNG (unspecified) | Sample 1: 11 (100%) Sample 2: 12 (100%) |
Sample 1: 13.97 (3.14) Sample 2: 14.9 (2.3) |
Sample 1: current stimulant use (55%); medication-free (45%) Sample 2: current stimulant use (58%); medication-free (42%) |
>24 hours | Sample 1: ODD (27%) Sample 2: ODD (33%) |
Nonsymptomatic siblings | Sample 1: 11 (100%) Sample 2: 12 (100%) |
Sample 1: 14.45 (2.58) Sample 2: 14.1 (2.7) |
Motor cortex ↔ striatum | None |
| van Rooij et al., 2015 (51) | PPIFSLa,b | SST: 1) successful stop > go; 2) failed stop > go | 185 (70%) | 17.3 (3.2) | Current medication use, class unspecified (77%); medication-free (23%) | Unspecified | ODD (30%), CD (7%), reading disability (18%) | Nonsymptomatic siblings | 111 (43%) | 17.3 (4) | 1) L IFC ↔ R putamen; L SFG ↔ L thalamus, operculum; 2) L IFC ↔ L occipital cortex, MTG, R IFC, MFG | 1) L IFC ↔ L cerebellum, precuneus, MTG; L SFG ↔ BIL precentral, precuneus, R frontal pole, ACC, cerebellum; 2) L IFC ↔ R medial frontal, ACC; L SFG ↔ L MTG |
ACC, anterior cingulate cortex; ADHD, attention-deficit/hyperactivity disorder; BIL, bilateral; CD, conduct disorder; DLPFC, dorsolateral prefrontal cortex; fMRI, functional magnetic resonance imaging; GNG, go/no-go; gPPI, generalized psychophysiological interaction; GTT, graph theoretic technique; IFC, inferior frontal cortex; IPS, intraparietal sulcus; L, left; MFG, middle frontal gyrus; MTG, middle temporal gyrus; ODD, oppositional defiant disorder; PPI, psychophysiological interaction; R, right; SBC, seed-based correlation; SFG, superior frontal gyrus; SPG, superior parietal gyrus; SST, stop signal task.
Scrubbing regressors included in the model for volumes with excessive motion in addition to standard motion parameters.
White matter and cerebrospinal fluid signal regressors included in the model in addition to standard motion parameters.
The heterogeneity of methodologies of this literature prevented a meta-analysis. Consequently, the comparisons were summarized as a narrative synthesis.
Functional Connectivity in ADHD
The differences in connectivity between ADHD and neurotypical groups were investigated in 37 comparisons (youths = 23, adults = 14) (Table 1). Based on the collective descriptions in the literature (38), the following cognitive domains emerged: attention (n = 4) (39, 40, 41, 42), cognitive control (n = 6) (15,43, 44, 45, 46, 47), response inhibition (n = 5) (15,48, 49, 50, 51), reward processing (n = 5) (52, 53, 54, 55, 56), working memory (n = 5) (55,57, 58, 59, 60), and emotion processing (n = 6) (43,55,61, 62, 63, 64). Additionally, 6 comparisons that could not be classified into the above domains included error monitoring (65), response preparation (66), motor response (55), social cognition/relational processing (55), and time discrimination (46).
Table 1.
Studies Investigating fMRI Functional Connectivity Differences Between ADHD and Typical Development Grouped by Cognitive Domain
| Study | Analysis Method | Task (Contrast) | NADHD (% Male) | AgeADHD, Years, Mean (SD) | Medication History | Medication Washout | ADHD Comorbidities | NControl (% Male) | AgeControl, Years, Mean (SD) | Control > ADHD | ADHD > Control |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Attention | |||||||||||
| Li et al., 2012 (39) | SBC | CPT (unspecified) | 22 (55%) | 11.6 (2.86) | Current MPH use (41%); medication-free (59%) | 48 hours | None | 22 (45%) | 12.1 (2.23) | L pulvinar nuclei ↔ R IFC, MFG; R pulvinar nuclei ↔ R PFC | R pulvinar nuclei ↔ BIL occipital lobe |
| Luo et al., 2018 (40) | GTTs | Cued attention task (cues) | 17 (77%) | 24.69 (2.1) | Current stimulant use (12%); past stimulant use (unspecified) | 48 hours | None | 33 (85%) | 24.27 (2.2) | Acting network hubs in BIL IPL; L IPL ↔ L SFG; degree in R MFG; betweenness centrality in L SFG, MFG, precentral, R IFC | Acting network hubs in L MFG and precentral |
| Rubia et al., 2009 (41) | SBC | CPT (targets > nontargets) | 13 (100%) | 12.5 (1.3) | Medication-naïve (100%) | – | ODD/CD (8%) | 13 (100%) | 13 (1.7) | L IFC ↔ striatum, cerebellum; R IFC ↔ striatum, BIL cerebellum; BIL thalamus/striatum ↔ striatum, R cerebellum; L striatum ↔ BIL cerebellum, R striatum; ACC ↔ cerebellum, cerebellar vermis; R IPL ↔ cerebellum, L IPL; cerebellar vermis ↔ PCC; R cerebellum ↔ PCC, L IPL; L cerebellum ↔ L IPL | None |
| Xia et al., 2014 (42) |
GTTs |
CPT (unspecified) |
22 (55%) |
11.6 (2.86) |
Current MPH use (41%); medication-free (59%) |
48 hours |
None |
22 (45%) |
12.1 (2.23) |
Nodal efficiency in L superior OFG and R SOG; degree and betweenness centrality in BIL occipital lobes, R temporal lobe, L paracentral, SMG |
Nodal efficiency in L cuneus; degree and betweenness centrality in ACC |
| Cognitive Control | |||||||||||
| Cubillo et al., 2010 (15) | SBC | Switch task (unspecified) | 11 (100%) | 29 (1) | Medication-naïve (100%) | – | Anxiety disorder (9%), mood disorder (27%), CD (9%), substance use disorder (18%) | 13 (100%) | 28 (1) | None | None |
| Hwang et al., 2015 (43) | gPPIAFNI | Affective Stroop task (incongruent > congruent stimuli) | 26 (65%) | 14.53 (unspecified) | Current stimulant use (42%); medication-free (58%) | >24 hours | ODD (4%), substance use disorder (8%) | 35 (51%) | 13.91 (unspecified) | L DMFG ↔ R lateral frontal, claustruma | L DMFG ↔ L posterior insula |
| Querne et al., 2017 (44) | ICA | Flanker task (unspecified) | 11 (unspecified) | 9.8 (1.7) | Medication-naïve (100%) | – | None | 11 (unspecified) | 10.8 (1.7) | Anticorrelation between DMN and frontotemporoparietal regions (direct group comparison not reported) | None |
| Plessen et al., 2016 (45) | ICA | Flanker task (post-error > post-correct trials) | 25 (68%) | 10.75 (1.09) | Medication-naïve (100%) | – | ODD (40%), ODD+CD (8%), phobia (16%), tics (4%), separation anxiety disorder (4%), elimination disorder (4%) | 29 (52%) | 10.15 (1.04) | None | Cingulo-opercular network ↔ VANa |
| Vloet et al., 2010 (46) | PPISPM | Time discrimination + stimulus-response compatibility task (stimulus-response compatibility) | 14 (100%) | 11.3 (2) | Past or current stimulant use (100%) | >48 hours | None | 14 (100%) | 11.9 (1.4) | L IFC ↔ L SPG; R IFC ↔ R SPG | None |
| Zamorano et al., 2017 (47) |
PPIFSL |
MSIT (incongruent > congruent conditions) |
17 (100%) |
11.6 (0.86) |
Current MPH use (100%) |
Medication not taken on study day |
None |
17 (100%) |
11.7 (0.67) |
Not reported |
R MFG + R IFC ↔ BIL OFC, striatum |
| Emotion Processing | |||||||||||
| Hafeman et al., 2017 (61) | gPPISPM | Emotional dynamic faces task (emotional faces > shapes) | 30 (67%) | 14.1 (1.8) | Current use of stimulants (43%), antipsychotics (10%), antidepressants (10%) | Unspecified | ODD (53%), CD (3%), depressive disorder (53%), anxiety disorder (3%) | 26 (46%) | 13.2 (2.2) | None | BIL amygdala ↔ subgenual cingulate; BIL amygdala ↔ R SFG |
| Hwang et al., 2015 (43) | gPPISPM | Affective Stroop task: 1) positive > neutral stimuli; 2) positive > neutral incongruent stimuli; 3) negative > neutral stimuli) | 26 (65%) | 14.53 (unspecified) | Current stimulant use (42%); medication-free (58%) | >24 hours | ODD (4%), substance use disorder (8%) | 35 (51%) | 13.91 (unspecified) | 1) R amygdala ↔ R MOG, L lentiform nucleus; 2) R amygdala ↔ BIL postcentrala; 3) none | None |
| Park et al., 2016 (55) | GTTsb | Emotive faces task (unspecified) | 34 (59%) | 27.88 (3.37) | Unspecified | Unspecified | Unspecified | 34 (62%) | 29.44 (3.57) | Degree in BIL medial frontal, L ACC, L postcentral, R caudate, L insula | Degree in L MFG, R SMG, R IPL, L MOG, L IOG, R cerebellum |
| Posner et al., 2011 (62) | DCM | Fearful faces task with priming (fearful faces) | 15 (87%) | 13.5 (1.2) | Current stimulant use (100%) | >48 hours | ODD/CD (% unspecified) | 15 (87%) | 13.4 (1.2) | None | R amygdala ↔ R lateral PFC |
| Schulz et al., 2014 (63) | PPISPM | Face emotion GNG (correct no-go > go) | 14 (100%) | 23.3 (2.3) | Medication-naïve (29%); past stimulant use but medication-free at time of study (71%) | – | Mood disorder (14%), anxiety disorder (14%), substance use disorder (36%) | 14 (100%) | 22.8 (2.7) | R DLPFC ↔ L IFC, putamen, BIL subgenual cingulate | None |
| Stoddard et al., 2017 (64) |
gPPIAFNI |
Implicit face emotion processing task (150% intensity across emotions) |
24 (75%) |
13.5 (2.9) |
Unspecified |
Unspecified |
Unspecified |
22 (41%) |
14.2 (2.1) |
None |
L amygdala ↔ L insula |
| Response Inhibition | |||||||||||
| Cai et al., 2021 (48) | gPPISPM | GNG (correct no-go) | 27 (78%) | 13.95 (2.62) | Medication-free during testing (100%) | >5 half-lives of drug | Unspecified | 30 (73%) | 13.65 (2.47) | R DLPFC ↔ R posterior parietal | None |
| Cubillo et al., 2010 (15) | SBC | SST (unspecified) | 10 (100%) | 28 (1) | Medication-naïve (100%) | – | Anxiety disorder (10%), mood disorder (30%), CD (10%), substance use disorder (20%) | 14 (100%) | 28 (2) | R IFC ↔ L IFC, R MFG, ACC, PCC, SMA, thalamus, striatum, BIL parietal/temporal/occipital; R ACC/PCC/SMA ↔ R thalamus, striatum | None |
| Massat et al., 2018 (49) | PPISPM | SST (successful > failed stop) | 18 (44%) | 10.6 (1.13) | Medication-naïve (100%) | – | None | 19 (47%) | 10 (1.35) | R IFC ↔ R OFC, L MFG, IFC | R dorsal caudate ↔ R IPL, SPG, L MFG, middle cingulate, precentral, postcentral |
| Mulder et al., 2011 (50) | SBC | GNG (unspecified) | Sample 1: 11 (100%) Sample 2: 12 (100%) |
Sample 1: 13.97 (3.14) Sample 2: 14.9 (2.3) | Sample 1: current stimulant use (55%); medication-free (45%) Sample 2: current stimulant use (58%); medication-free (42%) |
>24 hours | Sample 1: ODD (27%) Sample 2: ODD (33%) |
Sample 1: 11 (100%) Sample 2: 12 (100%) | Sample 1: 15.27 (1.92) Sample 2: 15 (2.1) | Samples 1 and 2: ACC ↔ cerebellum Sample 1: motor cortex ↔ striatuma Sample 2: not reported |
None |
| van Rooij et al., 2015 (51) |
PPIFSLb,c |
SST: 1) successful stop > go; 2) failed stop > go) |
185 (70%) |
17.3 (3.2) |
Current medication use, class unspecified (77%); medication-free (23%) |
Unspecified |
ODD (30%), CD (7%), reading disability (18%) |
125 (44%) |
16.5 (3.3) |
1) L IFC ↔ R putamen; L SFG ↔ L thalamus, operculum; 2) L IFC ↔ R IFC, BIL SFG/preSMA, L occipital cortex, MTG; L SFG ↔ L IFC |
1) L IFC ↔ L MTG, cerebellum; L SFG ↔ R ACC, frontal pole, BIL precuneus, L precentral, R cerebellum; 2) L IFC ↔ BIL temporal pole, L cerebellum, R SMG; L SFG ↔ L MTG |
| Reward Processing | |||||||||||
| Ceceli et al., 2020 (52) | PPIFSL | Free operant task with food rewards (late > early phase) | 25 (56%) | 22.31 (4.69) | Current or previous stimulant use (72%); past stimulant use but medication-free at time of study (16%); medication-naïve (12%) | 36 hours | None | 25 (56%) | 21.48 (2.92) | L posterior putamen ↔ dorsal ACC, medial frontal | None |
| Ma et al., 2016 (53) | gPPISPM | Rewarded Stroop task (rewarded > neutral Stroop) | 25 (76%) | 15.36 (1.08) | Current MPH use (60%); medication-free (40%) | 24 hours | ODD and CD (% unspecified) | 33 (67%) | 15.3 (1.05) | None | L ventral striatum ↔ R precentral |
| Mowinckel et al., 2017 (54) | Bayesian hierarchical mixed model | Value-based decision-making task (unspecified) | 20 (35%) | 29.9 (1.41) | Current stimulant use (100%) | >20 hours | None | 27 (30%) | 27.42 (1.23) | Within VIS, FPN, ECN, subcortical network, L VAN; ECN ↔ FPN ↔ sensorimotor network; DAN ↔ sensorimotor network; DAN ↔ VIS | VAN ↔ DMN; VAN ↔ ECN; DMN ↔ ECN |
| Park et al., 2016 (55) | GTTsb | Gambling task: 1) gambling reward; 2) gambling punishment | 34 (59%) | 27.88 (3.37) | Unspecified | Unspecified | Unspecified | 34 (62%) | 29.44 (3.57) | 1) Degree in BIL SFG, MTG; 2) degree in R medial frontal, MFG, insula, BIL SFG, L IPL, thalamus, parahippocampal | 1) Degree in R ACC, L PCC, lingual, thalamus, BIL insula, cerebellum; 2) R precentral, MTG, L postcentral, STG, BIL cerebellum |
| von Rhein et al., 2017 (56) |
ICA |
MID task (unspecified) |
150 (70%) |
17.7 (3) |
Unspecified |
>48 hours |
ODD (23%), CD (5%) |
48 (69%) |
16.9 (3.2) |
Within SAL (R ITG), ECN (R IFC, L cerebellum) |
Within SAL (R cerebellum) |
| Working Memory | |||||||||||
| Bédard et al., 2014 (57) | PPISPM | Visuospatial n-back task: 1) 1-back > 0-back; 2) 2-back > 0-back | 24 (88%) | 13.07 (1.93) | Current stimulant use (4%); current nonstimulant use (4%); past stimulant/nonstimulant use but medication-free at time of study (29%); medication-naïve (63%) | 2 weeks | ODD (8%), CD (4%), anxiety disorder (17%) | 21 (76%) | 12.44 (1.95) | 1) L DLPFC ↔ L PCC; 2) L DLPFC ↔ L midcingulate, PCC | 1) L DLPFC ↔ BIL posterior insula, R temporal cortex; 2) L DLPFC ↔ L intraparietal sulcus, cerebellum |
| Massat et al., 2012 (58) | gPPISPM | Verbal n-back task (2-back > 0-back) | 19 (47%) | 10.75 (1.31) | Medication-naïve (100%) | – | None | 14 (57%) | 10.05 (1.28) | None | R cerebellum ↔ red nucleus, R amygdalaa, hippocampusa, linguala, precuneusa, L IFCa, MFGa, postcentrala, cerebelluma; L occipital ↔ BIL MFGa, R MTGa, STGa, fusiforma, putamena, L cerebelluma; L IPL ↔ BIL IFCa, MFGa, STGa, L ACCa, SMAa; R caudate ↔ BIL MFGa, R SFGa, putamena, insulaa |
| Park et al., 2016 (55) | GTTsb | Visuospatial n-back task (unspecified) | 34 (59%) | 27.88 (3.37) | Unspecified | Unspecified | Unspecified | 34 (62%) | 29.44 (3.57) | Degree in L precuneus, MTG, cuneus, insula | Degree in L precentral, IPL, cerebellum, R MFG, IFC, STG, BIL SFG, caudate |
| Wolf et al., 2009 (59) | ICA | Verbal working memory task (unspecified) | 12 (100%) | 22.2 (4.4) | Current MPH use (50%); past MPH use but medication-free at time of study (50%) | 72 hours | None | 12 (100%) | 21.6 (4.7) | Within BIL IFC, SFG, SPG, cerebellum, L ACC, medial frontal | Within L dorsal cingulate, cuneus, R IFC, SFG |
| Wu et al., 2017 (60) |
ICA |
Verbal n-back task (2-back > 0-back) |
22 (100%) |
12.71 (1.55) |
Past stimulant use but medication-free at time of study (23%); medication-naïve (77%) |
>4 weeks |
ODD (18%) |
30 (100%) |
11.96 (1.72) |
Within ECN (L SMG, insula) |
Within FPN (L postcentral, SPG), auditory network (R cuneus, occipital pole, supracalcarine, intracalcarine, lateral SOG, precuneus) |
| Other Cognitive Functions | |||||||||||
| Chevrier et al., 2019 (65) | SBC | SST: 1) error detection; 2) post-error slowing | 14 (50%) | 13.7 (2.1) | Current stimulant use (43%); medication-free (57%) | 24 hours | ODD (14%) | 14 (64%) | 15.4 (1.6) | 1) SN ↔ medial septal; 2) LC ↔ L amygdala, L hypothalamus; medial septal nuclei ↔ R amygdala, LC, R hypothalamus; raphe nucleus ↔ R SN/parahippocampal | 1) Dorsal striatum ↔ R IPL; SN ↔ R hypothalamus; SN ↔ L amygdala, LC, raphe nucleus; 2) ventral pallidum ↔ SN/parahippocampal, R dorsal pallidum, L amygdala; SN ↔ L hypothalamus; LC ↔ R IFC; medial septal nuclei ↔ BIL amygdala, L SN, BIL basal forebrain; raphe nucleus ↔ BIL amygdala, R SN, BIL hypothalamus |
| Clerkin et al., 2013 (66) | PPISPM | Cued reaction time task (cues > noncues) | 35 (83%) | 24.6 (2.04) | Current stimulant use (6%); past stimulant use but medication-free at time of study (71%) | >48 hours | Mood disorder (23%), anxiety disorder (23%), substance use disorder (43%) | 32 (84%) | 24.38 (2.4) | R thalamus ↔ pons | None |
| Park et al., 2016 (55) | GTTsb | Motor task (unspecified) | 34 (59%) | 27.88 (3.37) | Unspecified | Unspecified | Unspecified | 34 (62%) | 29.44 (3.57) | Degree in R precentral, medial frontal, SMG, L MFG, precuneus, cuneus, parahippocampal, cerebellum, BIL MTG, MOG | Degree in BIL SFG, PCC, R MFG, ACC, L postcentral |
| GTTsb | Relational processing task (unspecified) | 34 (59%) | 27.88 (3.37) | Unspecified | Unspecified | Unspecified | 34 (62%) | 29.44 (3.57) | Degree in R medial frontal, SFG, BIL ACC, L lingual, cerebellum | Degree in R PCC, cuneus, BIL IPL, STG, L MTG | |
| GTTsb | Social cognition task (unspecified) | 35 (59%) | 27.88 (3.37) | Unspecified | Unspecified | Unspecified | 34 (62%) | 29.44 (3.57) | Degree in BIL SFG, R PCC, L cuneus | Degree in L precentral, postcentral, cerebellum, BIL precuneus, R MTG | |
| Vloet et al., 2010 (46) | PPISPM | Time discrimination + stimulus-response compatibility task (time discrimination) | 14 (100%) | 11.3 (2) | Past or current stimulant use (100%) | >48 hours | None | 14 (100%) | 11.9 (1.4) | R IFC ↔ R cerebellum | None |
ACC, anterior cingulate cortex; ADHD, attention-deficit/hyperactivity disorder; BIL, bilateral; CD, conduct disorder; CPT, continuous performance task; DAN, dorsal attention network; DCM, dynamic causal modeling; DLPFC, dorsolateral prefrontal cortex; DMFG, dorsomedial frontal gyrus; DMN, default mode network; ECN, executive control network; fMRI, functional magnetic resonance imaging; FPN, frontoparietal network; GNG, go/no-go; gPPI, generalized psychophysiological interaction; GTT, graph theoretic technique; ICA, independent component analysis; IFC, inferior frontal cortex; IOG, inferior occipital gyrus; IPL, inferior parietal lobule; ITG, inferior temporal gyrus; L, left; LC, locus coeruleus; MFG, middle frontal gyrus; MID, monetary incentive delay; MOG, middle occipital gyrus; MPH, methylphenidate; MSIT, multi-source interference task; MTG, middle temporal gyrus; ODD, oppositional defiant disorder; OFG, orbitofrontal gyrus; PCC, posterior cingulate cortex; PFC, prefrontal cortex; PPI, psychophysiological interaction; preSMA, pre–supplementary motor area; R, right; SAL, salience network; SBC, seed-based correlation; SFG, superior frontal gyrus; SMA, supplementary motor area; SMG, supramarginal gyrus; SN, substantia nigra; SOG, superior occipital gyrus; SPG, superior parietal gyrus; SST, stop signal task; STG, superior temporal gyrus; VAN, ventral attention network; VIS, visual network.
Correction for multiple comparisons not specified.
White matter and cerebrospinal fluid signal regressors included in the model in addition to standard motion parameters.
Scrubbing regressors included in the model for volumes with excessive motion in addition to standard motion parameters.
Differences Between Individuals With ADHD and Neurotypical Populations by Cognitive Domain
There was an overall decrease of connectivity in ADHD compared with neurotypical control subjects during attention tasks (74 patients and 90 control subjects across 4 comparisons). The right inferior frontal cortex (IFC) and bilateral inferior parietal lobules (IPLs) were indicated as hubs of connectivity decreases in ADHD, whereas the anterior cingulate cortex (ACC), left middle frontal gyrus (MFG), precentral gyrus, and bilateral occipital lobes showed both increases and decreases of connectivity, all with a 1:1 ratio indicating equal number of increases and decreases.
The cognitive control results were heterogeneous, not yielding many common case-control differences (104 patients and 119 control subjects across 6 comparisons). Only the right IFC consistently showed abnormalities, with both increases and decreases (1:1 ratio) of functional connectivity.
ADHD was related to predominantly decreased functional connectivity compared with neurotypical control subjects during response inhibition (263 patients and 211 control subjects across 5 comparisons). The right IFC, supplementary motor complex, and parieto-occipital regions showed decreased connectivity in ADHD, while the left precentral gyrus exhibited increased connectivity. Conversely, the right striatum (2:1 ratio, decreases-to-increases ratio), left IFC (3:1), MFG (2:1), superior frontal gyrus (SFG) (1:1) middle temporal gyrus (1:1), ACC (1:1), and cerebellum (1:1) were hubs of increased and decreased connectivity in patients.
ADHD was associated with an overall increase in connectivity compared with control subjects during working memory (111 patients and 111 control subjects across 5 comparisons). The right insula, superior temporal gyrus, striatum, and left MFG and IPL showed increased connectivity in patients. Bilateral IFCs, SFG, left insula, cingulate, precuneus, cuneus, and cerebellum showed both increases and decreases of connectivity in ADHD, all with a 1:1 ratio except for the cerebellum, which showed more increases (3:1).
During reward processing, the medial frontal cortex showed decreased functional connectivity, while the precentral gyrus was a hub of increased connectivity in ADHD (254 patients and 167 control subjects across 5 comparisons). The right insula, middle temporal gyrus, left thalamus, striatum, bilateral ACC, and cerebellum exhibited increases and decreases of connectivity in patients, all with a 1:1 ratio.
During emotion processing, the left postcentral gyrus showed decreased connectivity in ADHD compared with control subjects (143 patients and 146 control subjects across 6 comparisons). Additionally, the right amygdala, left insula, and ACC formed hubs of increased and decreased connectivity in ADHD, all with a 1:1 ratio.
Differences Between Individuals With ADHD and Neurotypical Populations by Functional Network
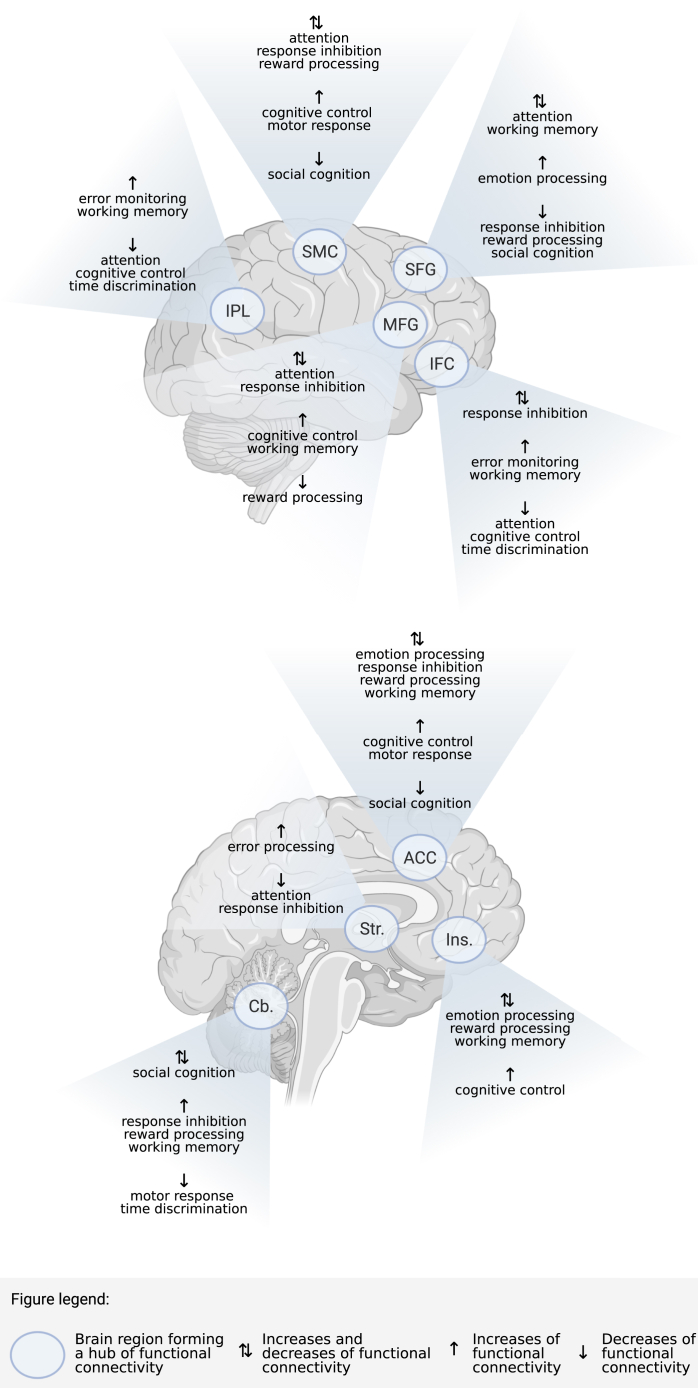
We also aimed to identify hubs and networks exhibiting common connectivity differences in ADHD across cognitive functions. Regions that formed hubs of connectivity differences between patients and control subjects included the ACC (6:7, decreases-to-increases ratio), IFC (4:3), MFG (3:4), SFG (5:3), insula (3:4), sensorimotor cortex (1:1 ratio), IPL (1:2), striatum (3:1), and cerebellum (3:4). These regions exhibited increases and decreases of connectivity across tasks (Figure 2).
Figure 2.
Regions that formed core hubs of functional connectivity differences between individuals with ADHD and neurotypical control subjects across cognitive domains. ACC, anterior cingulate cortex; Cb., cerebellum; IFC, inferior frontal cortex; Ins., insula; IPL, inferior parietal lobule; MFG, middle frontal gyrus; SFG, superior frontal gyrus; SMC, sensorimotor cortex; Str., striatum. (Figure created with BioRender; https://biorender.com/.)
Several studies performed formal analyses of established functional networks, often described in resting-state literature (67,68), finding both within- and between-network differences. Relative to control subjects, patients showed reduced connectivity in visual (VIS), frontoparietal (FPN), executive control (ECN), ventral attention (VAN), subcortical, and salience networks during reward processing as well as in the ECN during working memory. Individuals with ADHD also showed increased connectivity within the salience network during reward processing and within the FPN and auditory networks during working memory compared with control subjects. Furthermore, ADHD was associated with decreased functional connectivity between the default mode (DMN) and frontotemporoparietal networks during cognitive control as well as between the ECN and both the FPN and the sensorimotor network (SMN), between the dorsal attention network (DAN) and SMN, and between the DAN and VIS during reward processing. Increased functional connectivity in ADHD was observed between the cingulo-opercular network (CON) and VAN in cognitive control and between the VAN and DMN, between the VAN and ECN, and between the DMN and ECN across cognitive domains.
These studies suggest that the functional network architecture differs in ADHD. Alterations of functional connectivity were observed primarily in SMN, VIS, ECN, DMN, CON, and subcortical networks across cognitive domains. Nonetheless, both increases and decreases of connectivity were observed in ADHD across all implicated networks.
Effects of Interventions on Functional Connectivity in ADHD
Nine studies tested the effects of interventions on functional connectivity, with 8 studies investigating stimulants (youths = 6, adults = 2) (41,44,54,60,62,69, 70, 71) and 1 study evaluating fMRI neurofeedback of the right IFC (73). The intervention studies investigated various cognitive domains, and thus findings were synthesized across cognitive functions and within treatment type (Table 3).
Table 3.
Studies Investigating the Impact of Interventions on fMRI Functional Connectivity in ADHD
| Study | Analysis Method | Task (Contrast) | NADHD (% Male) | AgeADHD, Years, Mean (SD) | Medication History | Medication Washout | ADHD Comorbidities | Intervention/Comparison | Design | On Intervention > Off Intervention | Off Intervention > On Intervention |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Mowinckel et al., 2017 (54) | Bayesian hierarchical mixed model | Value-based decision making task (unspecified) | 20 (35%) | 29.9 (1.41) | Current stimulant use (100%) | >20 hours | None | Acute MPH (10–40 mg of regularly prescribed formulation)/placebo | Randomized, double-blind, crossover | Auditory network ↔ ECN | Within DMN and VIS |
| Posner et al., 2011 (62) | DCM | Fearful faces task with priming (fearful faces) | 15 (87%) | 13.5 (1.2) | Current stimulant use (100%) | >48 hours | ODD/CD (% unspecified) | Acute stimulant (regularly prescribed formulation and dose)/off medication | Cross-over | None | None (main group comparison); BIL amygdala ↔ BIL lateral PFC (secondary nonparametric analysis) |
| Querne et al., 2017 (44) | ICA | Flanker task (unspecified) | 11 (unspecified) | 9.8 (1.7) | Medication-naïve (100%) | – | None | 4 weeks MPH (20–30 mg extended release)/off medication | Crossover (off medication → MPH) | DMN composed of anterior and posterior regions; anticorrelation between DMN and BIL anterior frontal, striatum, dorsal ACC, R occipitoparietal cortex, L cerebellum (direct group comparison not reported) | DMN composed posterior regions only (direct group comparison not reported) |
| Rubia et al., 2009 (41) | SBC | CPT (targets > nontargets) | 13 (100%) | 12.5 (1.3) | Medication-naïve (100%) | – | ODD/CD (8%) | Acute MPH (0.3 mg/kg)/placebo | Randomized, double-blind, crossover | L caudate/putamen ↔ R caudate/putamen | None |
| Rubia et al., 2019 (72) | SBC | Neurofeedback | Active group: 18 (100%); control group: 13 (100%) | Active group: 14 (2); control group: 14 (2) | Active group: current use of stimulants (83%), withdrew from medication for duration of study (17%); control group: current stimulant use (69%), withdrew from medication for duration of study (23%), medication-naïve (8%) | >7 days for those willing to withdraw from medication | ODD/CD (% unspecified) | fMRI neurofeedback of R IFC/fMRI neurofeedback of L parahippocampal gyrus | 11-run parallel groups active control (randomized single-blind control trial) | Relative to first run and control: R IFC (BA 45) ↔ R caudate, ACC; R IFC (BA 44) ↔ R ACC | Relative to first run: R IFC (BA 45) ↔ L parahippocampal, hippocampus, lingual, BIL PCC, precuneus, calcarine, thalamus, caudate, putamen, pallidum; R IFC (BA 44) ↔ BIL precuneus, PCC, hippocampus, parahippocampal, lingual, thalamus; relative to control: R IFC (BA 45) ↔ BIL PCC, precuneus, calcarine; R IFC (BA 44) ↔ BIL PCC, precuneus, hippocampus, parahippocampal, lingual, thalamus |
| Schulz et al., 2018 (69) | PPISPM | Emotional GNG (correct go trials cued by sad faces) | 25 (56%) | 34.8 (9.8) | Current use of medication, class unspecified (8%); past stimulant and/or nonstimulant use but medication-free at time of study (36%); medication-naïve (56%) | 2 weeks | None | 5 weeks LDX (30–70 mg)/placebo | Randomized, single-blind, crossover | None | L amygdala ↔ R SPG, L STG; R amygdala ↔ L IFC, STG, R SPG |
| Sheridan et al., 2010 (70) | BSC | Delayed match to sample task (encoding) | 5 (0%) | 14.8 (2.4) | Current stimulant use (60%); current stimulant and nonstimulant use (20%); current stimulant and SSRI use (20%) | 24 hours (for stimulants only) | Unspecified | Acute stimulant (regularly prescribed formulation and dose)/off medication | Crossover | BIL MFG ↔ cerebellar vermisa | BIL MFG ↔ striatuma, L MFGa, medial PFCa, hippocampusa, ITGa, R TPJa, insulaa, linguala |
| Wong and Stevens, 2012 (71) | ICA | Sternberg item recognition task (unspecified) | 18 (83%) | 14.6 (2) | Current stimulant use (100%) | 48 hours | ODD (6%) | Acute stimulant (regularly prescribed formulation and dose)/placebo | Randomized, double-blind, crossover | Within ACC, medial frontal, PCC, precuneus, cuneus, lingual, SFG, cingulate, R postcentral, precentral, L IFC, SMG, MTG, angular regions | Within PCC, precuneus |
| Wu et al., 2017 (60) | ICA | Verbal n-back task (2-back > 0-back) | 22 (100%) | 12.71 (1.55) | Past stimulant use but medication-free at time of study (23%); medication-naïve (77%) | >4 weeks | ODD (18%) | Acute MPH (10 mg)/placebo | Randomized, double-blind, crossover | Within ECN (R precuneus, L PCC) | None |
ACC, anterior cingulate cortex; ADHD, attention-deficit/hyperactivity disorder; BIL, bilateral; BA, Brodmann area; BSC, beta series correlation; CD, conduct disorder; CPT, continuous performance task; DCM, dynamic causal modeling; DMN, default mode network; ECN, executive control network; fMRI, functional magnetic resonance imaging; GNG, go/no-go; gPPI, generalized psychophysiologic interaction; ICA, independent component analysis; IFC, inferior frontal cortex; ITG, inferior temporal gyrus; L, left; LDX, lisdexamfetamine; MFG, middle frontal gyrus; MPH, methylphenidate; MTG, middle temporal gyrus; ODD, oppositional defiant disorder; PCC, posterior cingulate cortex; PFC, prefrontal cortex; PPI, psychophysiological interaction; R, right; SBC, seed-based correlation; SFG, superior frontal gyrus; SMG, supramarginal gyrus; SPG, superior parietal gyrus; SSRI, selective serotonin reuptake inhibitor; STG, superior temporal gyrus; TPJ, temporoparietal junction; VIS, visual network.
Correction for multiple comparisons not specified.
Stimulants increased connectivity of the striatum (although decreases were seen in one study), ACC, and cerebellum across tasks and decreased connectivity of the amygdala in emotion paradigms compared with no intervention/placebo. MFG, IFC, medial frontal cortex, posterior cingulate cortex (PCC), occipital cortex, and precuneus showed both increased and decreased connectivity with stimulants, all with 1:1 ratio. Additionally, network analyses showed decreased connectivity within DMN and VIS with stimulants relative to no treatment/placebo. Stimulants enhanced connections within the ECN and between the ECN and auditory networks.
The neurofeedback study showed increased functional connectivity between the right IFC and the right striatum and ACC relative to both baseline and control subjects. Additionally, neurofeedback was associated with decreased connectivity between the right IFC and various PCC-occipital, striatothalamic, and hippocampal regions. Overall, interventions modulated functional connectivity of the striatum, ACC, occipital regions, and midline DMN areas.
Critical Appraisal
Across all 51 included comparisons, 28 specified a motion cutoff. All comparisons included motion correction, with 36 comparisons applying standard methods (e.g., default software options) and 15 comparisons using more advanced approaches.
Average sample size of patient groups across all comparisons was 28, with larger samples in case-control than intervention comparisons (31 relative to 16, respectively). Independent samples were tested in 42 comparisons. Within those, studies reported matching groups on age in 40 comparisons, sex in 35 comparisons, handedness in 26 comparisons, motion in 21 comparisons, IQ in 21 comparisons, race/ethnicity in 9 comparisons, socioeconomic status in 7 comparisons, presence of unrelated symptoms in 7 comparisons, education level in 6 comparisons, working memory capacity in 1 comparison, and pubertal status in 1 comparison. Additionally, of all 51 comparisons, 42 reported information about ADHD presentation. On average, 72% of patients had combined ADHD, 22% had inattentive presentation, 3% had hyperactive-impulsive presentation, and 0.5% were classified as ADHD not otherwise specified.
The reviewed studies used heterogeneous methods to assess connectivity. Of all 51 comparisons, 20 used psychophysiological interaction (psychophysiological interaction = 12, generalized psychophysiological interaction = 8), 9 used seed-based correlations, 9 used graph theoretic techniques, 8 used independent component analysis, 2 used dynamic causal modeling, 2 used Bayesian hierarchical mixed models, and 1 used beta series correlation. Of all comparisons, 41 were seed-based and required definition of seed regions used in analysis. Within those, 21 used seeds defined independently of the dataset studied (based on past research or anatomical atlases), while 20 used seeds based on the same dataset (e.g., regions of peak activation in the same cohort). Furthermore, while most comparisons reported multiple comparisons correction, 6 of all 51 comparisons did not (indicated by a footnote symbol in Tables 1 and 3).
Of 51 comparisons, 34 recruited samples currently receiving pharmacotherapy, 9 recruited medication-naïve participants, 1 recruited participants who were medication-naïve or had a history of pharmacotherapy, and 7 did not specify medication history. Within the 34 comparisons recruiting currently medicated participants, 31 specified a washout period. Washout periods ranged from 20 hours to 4 weeks (20 hours = 2; 24 hours = 8; 36 hours = 1; 48 hours = 12; 72 hours = 1; 1 week = 1; 2 weeks = 2; 4 weeks = 2). Additionally, 2 comparisons used washout periods without specifying their exact duration (Tables 1 and 2).
The effects of interventions were tested in 9 comparisons. Selection bias (random sequence generation and allocation concealment) was deemed low in 6 comparisons, unclear in 2 comparisons, and high in 1 comparison. Performance (blinding of participants/personnel) and detection (blinding of outcome assessment) biases were rated low in 3 comparisons, unclear in 3 comparisons, and high in 3 comparisons. Attrition (incomplete outcome data) and reporting (selective reporting) biases were deemed low in all 9 comparisons (Supplement).
Discussion
Task-Based Connectivity in ADHD
Across cognitive domains, changes of functional connectivity were observed in ADHD relative to neurotypical populations, with core hubs of connectivity differences in the ACC, IFC, MFG, SFG, sensorimotor cortex, insula, IPL, striatum, and cerebellum. Although changes of connectivity were observed when cognitive domains were considered individually, inhibition and attention were associated primarily with reductions in connectivity, whereas working memory was related to enhanced connectivity in ADHD relative to typical development.
Additional differences were observed in between-network connectivity. Across cognitive domains, individuals with ADHD showed stronger connections between VAN and both DMN and ECN as well as between ECN and DMN. During cognitive control, decreased connectivity was observed between DMN and frontotemporoparietal networks, while increased connectivity was seen between CON and VAN. During reward processing, only decreases of connectivity were observed between ECN and both FPN and SMN as well as between DAN and both SMN and VIS. Furthermore, for individuals with ADHD and their nonsymptomatic siblings and adults with ADHD and ADHD remitters, a limited literature showed connectivity differences similar to those seen between ADHD and neurotypical populations, specifically in striatal and sensorimotor regions (Table 2).
This review compiled findings estimated with several methods. Although these methods have fundamental differences and their outcomes may not represent the same aspects of connectivity, they reflect abnormal functioning of discrete networks in ADHD. This heterogeneity of methods prevents a synthesis yielding mechanistic insight into network-level pathophysiology of ADHD, although there is value in highlighting the cumulative evidence implicating certain neural systems.
The observations of abnormalities in task-relevant functional networks in ADHD bolster evidence of largely decreased local activation in core executive function–relevant areas, including ventrolateral, dorsolateral, and medial prefrontal, temporoparietal, and striatal regions in meta-analyses of fMRI studies in ADHD (6, 7, 8, 9, 10, 11, 12,28). Consequently, these findings support the presence of abnormalities in core task-positive networks and DMN in ADHD, and the high prevalence of abnormal sensorimotor connectivity resonates with similar observations in resting-state studies (24,73, 74, 75), which may reflect the previously proposed hypothesis of deviant maturational trajectories within these networks in ADHD (73). Nonetheless, the current literature largely focused on pediatric samples, and more exploration of adults and longitudinal cohorts is needed to better characterize the developmental trajectories of ADHD.
Our review also extends the knowledge base of resting-state connectivity alterations in ADHD in DMN, ECN, DAN, VAN, and salience networks (24,26,30,76,77) in two important ways. First, during different tasks, both increases and decreases of connectivity in ADHD were observed. Relative to connectivity under unconstrained context (resting state), which may reflect underlying anatomical or long-term functional plasticity differences, task-based literature indicates that connectivity alterations in ADHD may reflect differences in adaptability of functional circuits to changing demands. These context-dependent changes may be related to arousal systems that respond differently under distinct tasks (78). Such explanations of ADHD pathophysiology move beyond seeing the brain as a static system and suggest a conceptualization of ADHD as a disorder of dynamic neurocognitive processes.
Second, the review emphasizes that even within tasks results to date are mixed. With small numbers of studies in some areas, it was not possible to assess whether these mixed findings were due to low power or specific task or patient factors. Although ADHD heterogeneity can contribute to the mixed findings (79), the association between neurocognitive phenotypes and individual differences is still poorly understood (Supplement). Task factors, however, are supported by a recent study that found that youths with ADHD engage more task-specific than generic networks, showing hypoconnectivity in executive and reward circuits relative to neurotypical control subjects and nonsymptomatic siblings of individuals with ADHD (80). These findings suggest that the inconsistencies in the literature may reflect inefficient task-specific networks in ADHD, with greater variability in functional connections.
This review summarizes impairments of functional connectivity in ADHD across several cognitive domains. The included studies used different tasks to elicit specific cognitive processes. However, there is a risk of nonspecificity in tasks. While this review indicates context-specific alterations, efforts have been made to understand the underlying processes key in explaining ADHD pathophysiology, with some investigators proposing executive dysfunction (3,81), while others argue for poor deployment of resources (78,82). As yet, the precise neurofunctional manifestation of these explanations is poorly understood in patients. While cross-sectional imaging studies cannot clearly address questions of multifinality or equifinality in ADHD, they demonstrate the context-dependent nature of the dysfunction. How this relates to symptoms, clinical presentation, and treatment effects can help determine the degree to which ADHD is associated with one set of dysfunctions that differentially manifest across patients or whether true biological subtypes exist. Such efforts show promise (83,84) but have not yet been applied to context-dependent connectivity.
Effects of Interventions on Task-Based Connectivity in ADHD
Most intervention studies investigated stimulant medications, while one addressed the effects of fMRI neurofeedback. All interventions modulated connections of the striatum, ACC, occipital regions, and midline DMN structures. Furthermore, stimulants increased connectivity of cerebellar hubs across task paradigms and decreased amygdala connectivity during emotion processing. Additionally, stimulants led to increases and decreases of connectivity with IFC, MFG, medial frontal cortex, PCC, precuneus, and occipital regions across cognitive functions. Network-wide modulation with stimulants was also observed, with decreased connectivity within DMN and auditory networks and increased connectivity within ECN as well as between ECN and auditory networks.
Our findings align with individual resting-state studies showing that stimulants modulate spontaneous brain activity in similar ventrolateral frontal, occipital, and cerebellar regions, along with connectivity within ECN, VIS, and DMN (85, 86, 87, 88). Our findings also complement evidence of stimulant-related modulation of activation in areas dysfunctional in ADHD (7,9, 10, 11,19). These results highlight that stimulants also act on context-dependent network reorganization, potentially facilitating task performance.
One study explored the effects of fMRI neurofeedback. The modulation of connectivity of striatal, ventrolateral frontal, cingulate, and occipital regions observed with the intervention mirrored the changes seen with stimulant use, suggesting that neurofeedback of the right IFC may offer similar benefits as stimulants; however, more research is needed.
Limitations and Recommendations
Although this review supports the presence of network-wide dysfunction in ADHD and its modulation with treatment, a meta-analysis was not possible owing to the methodological heterogeneity of the literature. Consequently, it is difficult to quantify the degree of convergence across studies. A similar problem was noted in a recent systematic review of pharmacological effects on resting-state connectivity in ADHD (89). This is particularly relevant as recent task-based activation (20) and resting-state (25) meta-analyses of ADHD fMRI literature showed no spatial convergence across studies. Within the current review, eight different methods of estimating functional connectivity were used. Although most studies used seed-based methods, these comprised seven distinct approaches and different ways of defining seed regions. Furthermore, only approximately half of the studies used seeds defined independently of the dataset studied, thus avoiding the potential biases of circular analyses (90). Overall, while diverse methods provide different ways of characterizing the data and avoid potential issues stemming from one specific method, these benefits come at the cost of limiting the quantitative synthesis of findings across studies.
Past and current medication history represented another source of heterogeneity. Most studies included previously medicated participants. As stimulant use has been associated with structural (9,91,92), functional (7, 8, 9,11,93), and neurochemical changes (94), studying neural networks in currently or previously medicated individuals may confound pathophysiology of the disorder with the long-term impact of treatment. Another issue is the variability in the drug washout periods used (20 hours to 1 month). A minimum washout of 5 half-lives of the drug is recommended (95); however, discontinuing treatment can lead to withdrawal or rebound effects (96), and the length of the washout period may influence the level of neural differences between ADHD and neurotypical populations (10). Therefore, aside from the confounding effects of medication, some of the variability within the observed findings may be attributed to variable washout periods.
Small sample sizes, particularly in the intervention literature, which are linked to lower replicability of findings (97, 98, 99, 100, 101), are a limitation of this literature. Such issues have prompted recommendations such as a minimum sample size of 20 (100) and development of software allowing power calculations for fMRI studies (102). Consequently, these findings need to be interpreted with caution given that many were likely underpowered.
Some limitations of the reviewed studies involve the transparency of reporting, data quality assurance, and processing pipelines. For instance, only approximately half of the comparisons specified a motion cutoff. Given that ADHD is characterized by increased movement (103,104) and lower tolerability of the scanner environment (105) and that functional connectivity methods are particularly sensitive to motion artifacts (106, 107, 108), appropriate checks of data quality are essential. Issues with transparent reporting and data processing were also evident in studies not specifying multiple comparisons correction. False-positive rates in fMRI analyses are notorious without adjustment for multiple comparisons (98,109,110), and thus publications not reporting application of multiple comparisons correction should be interpreted with caution.
Further, the reviewed studies differed in general methodology, including study design, acquisition parameters, and data processing. Such heterogeneity further complicates cross-study synthesis of findings. Although these factors are not specific to this field and assessment of their impact was beyond the scope of this review, future studies should carefully consider and outline justification of their methodological choices.
This literature was also limited by other patient-specific factors frequently present in ADHD research, including male predominance, presence of comorbidities, variability of clinical presentation, and age-related differences (Supplement). Finally, ADHD is an inherently heterogeneous disorder with variable severity and class of symptoms, genetic and environmental risk factors, and profiles of associated pathophysiology (3,111, 112, 113). Consequently, it is likely that the heterogeneity of findings can be partly explained by the interindividual differences of ADHD groups. The impact of these factors should thus be explored further.
Overall, the limitations of the current literature illustrate the need for improved standards of study methodology and reporting. We propose that researchers prioritize recruiting larger, more diverse, and medication-naïve samples; implement greater control of in-scan motion and motion-related artifacts; use state-of-the-art data processing pipelines; and promote reporting transparency and openness [see Pereira-Sanchez et al. (89) for an in-depth discussion].
Conclusions
This is the first systematic review appraising the task-based functional connectivity literature of ADHD. We reviewed studies describing ADHD and the impact of interventions on task-relevant functional networks involved in the pathophysiology of the disorder. Our review supports the presence of CON, SMN, VIS, subcortical, ECN, and DMN network abnormalities in ADHD and shows that interventions can modulate the functional reorganization of those circuits. Overall, this review highlights the utility of task-based connectivity studies in broadening the understanding of the neural underpinnings of ADHD and in studying the mechanisms of action of ADHD treatments, but advocates for improvements to methodological quality of this line of research.
Acknowledgments and Disclosures
This work was supported by the National Institute for Health Research Maudsley Biomedical Research Centre at South London and Maudsley National Health Service Foundation Trust and King’s College London, and the Medical Research Council (postdoctoral appointment to MC).
The views expressed are those of the authors and not necessarily those of the National Health Service, National Institute for Health Research, Medical Research Council, or Department of Health and Social Care.
The authors report no biomedical financial interests or potential conflicts of interest.
Footnotes
Supplementary material cited in this article is available online at https://doi.org/10.1016/j.bpsgos.2021.10.006.
Supplementary Material
References
- 1.American Psychiatric Association . 5th ed. American Psychiatric Association; Arlington, VA: 2013. Diagnostic and Statistical Manual of Mental Disorders. [Google Scholar]
- 2.Pievsky M.A., McGrath R.E. The neurocognitive profile of attention-deficit/hyperactivity disorder: A review of meta-analyses. Arch Clin Neuropsychol. 2018;33:143–157. doi: 10.1093/arclin/acx055. [DOI] [PubMed] [Google Scholar]
- 3.Willcutt E.G., Doyle A.E., Nigg J.T., Faraone S., Pennington B.F. Validity of the executive function theory of attention-deficit/hyperactivity disorder: A meta-analytic review. Biol Psychiatry. 2005;57:1336–1346. doi: 10.1016/j.biopsych.2005.02.006. [DOI] [PubMed] [Google Scholar]
- 4.Balogh L., Czobor P. Post-error slowing in patients with ADHD: A meta-analysis. J Atten Disord. 2016;20:1004–1016. doi: 10.1177/1087054714528043. [DOI] [PubMed] [Google Scholar]
- 5.Noreika V., Falter C.M., Rubia K. Timing deficits in attention-deficit/hyperactivity disorder (ADHD): Evidence from neurocognitive and neuroimaging studies. Neuropsychologia. 2013;51:235–266. doi: 10.1016/j.neuropsychologia.2012.09.036. [DOI] [PubMed] [Google Scholar]
- 6.Cortese S., Kelly C., Chabernaud C., Proal E., Di Martino A., Milham M.P., Castellanos F.X. Toward systems neuroscience of ADHD: A meta-analysis of 55 fMRI studies. Am J Psychiatry. 2012;169:1038–1055. doi: 10.1176/appi.ajp.2012.11101521. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Hart H., Radua J., Nakao T., Mataix-Cols D., Rubia K. Meta-analysis of functional magnetic resonance imaging studies of inhibition and attention in attention-deficit/hyperactivity disorder. JAMA Psychiatry. 2013;70:185. doi: 10.1001/jamapsychiatry.2013.277. [DOI] [PubMed] [Google Scholar]
- 8.Hart H., Radua J., Mataix-Cols D., Rubia K. Meta-analysis of fMRI studies of timing in attention-deficit hyperactivity disorder (ADHD) Neurosci Biobehav Rev. 2012;36:2248–2256. doi: 10.1016/j.neubiorev.2012.08.003. [DOI] [PubMed] [Google Scholar]
- 9.Lukito S., Norman L., Carlisi C., Radua J., Hart H., Simonoff E., Rubia K. Comparative meta-analyses of brain structural and functional abnormalities during cognitive control in attention-deficit/hyperactivity disorder and autism spectrum disorder. Psychol Med. 2020;50:894–919. doi: 10.1017/S0033291720000574. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.McCarthy H., Skokauskas N., Frodl T. Identifying a consistent pattern of neural function in attention deficit hyperactivity disorder: A meta-analysis. Psychol Med. 2014;44:869–880. doi: 10.1017/S0033291713001037. [DOI] [PubMed] [Google Scholar]
- 11.Norman L., Carlisi C., Lukito S., Hart H., Mataix-Cols D., Radua J., Rubia K. Structural and functional brain abnormalities in attention-deficit/hyperactivity disorder and obsessive-compulsive disorder. JAMA Psychiatry. 2016;73:815. doi: 10.1001/jamapsychiatry.2016.0700. [DOI] [PubMed] [Google Scholar]
- 12.Dickstein S.G., Bannon K., Castellanos F.X., Milham M.P. The neural correlates of attention deficit hyperactivity disorder: An ALE meta-analysis. J Child Psychol Psychiatry. 2006;47:1051–1062. doi: 10.1111/j.1469-7610.2006.01671.x. [DOI] [PubMed] [Google Scholar]
- 13.van Rooij D., Hoekstra P.J., Mennes M., von Rhein D., Thissen A.J., Heslenfeld D., et al. Neural activation patterns during response inhibition distinguish adolescents with ADHD, their unaffected siblings, and healthy controls. Am J Psychiatry. 2015;172:674–683. doi: 10.1176/appi.ajp.2014.13121635. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Rubia K., Smith A.B., Brammer M., Toone B., Taylor E. Abnormal brain activation during inhibition and error detection in medication-naive adolescents with ADHD. Am J Psychiatry. 2005;162:1067–1075. doi: 10.1176/appi.ajp.162.6.1067. [DOI] [PubMed] [Google Scholar]
- 15.Cubillo A.I., Halari R., Ecker C., Giampietro V., Taylor E., Rubia K. Reduced activation and inter-regional functional connectivity of fronto-striatal networks in adults with childhood attention-deficit hyperactivity disorder (ADHD) and persisting symptoms during tasks of motor inhibition and cognitive switching. J Psychiatr Res. 2010;44:629–639. doi: 10.1016/j.jpsychires.2009.11.016. [DOI] [PubMed] [Google Scholar]
- 16.Vaidya C.J., Bunge S.A., Dudukovic N.M., Zalecki C.A., Elliott G.R., Gabrieli J.D. Altered neural substrates of cognitive control in childhood ADHD: Evidence from functional magnetic resonance imaging. Am J Psychiatry. 2005;162:1605–1613. doi: 10.1176/appi.ajp.162.9.1605. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Schulz K.P., Fan J., Bédard A.C., Clerkin S.M., Ivanov I., Tang C.Y., et al. Common and unique therapeutic mechanisms of stimulant and nonstimulant treatments for attention-deficit/hyperactivity disorder. Arch Gen Psychiatry. 2012;69:952–961. doi: 10.1001/archgenpsychiatry.2011.2053. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Schulz K.P., Bédard A.V., Fan J., Hildebrandt T.B., Stein M.A., Ivanov I., et al. Striatal activation predicts differential therapeutic responses to methylphenidate and atomoxetine. J Am Acad Child Adolesc Psychiatry. 2017;56:602–609.e2. doi: 10.1016/j.jaac.2017.04.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Rubia K., Alegria A.A., Cubillo A.I., Smith A.B., Brammer M., Radua J. Effects of stimulants on brain function in attention-deficit/hyperactivity disorder: A systematic review and meta-analysis. Biol Psychiatry. 2014;76:616–628. doi: 10.1016/j.biopsych.2013.10.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Samea F., Soluki S., Nejati V., Zarei M., Cortese S., Eickhoff S.B., et al. Brain alterations in children/adolescents with ADHD revisited: A neuroimaging meta-analysis of 96 structural and functional studies. Neurosci Biobehav Rev. 2019;100:1–8. doi: 10.1016/j.neubiorev.2019.02.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Stevens M.C. The developmental cognitive neuroscience of functional connectivity. Brain Cogn. 2009;70:1–12. doi: 10.1016/j.bandc.2008.12.009. [DOI] [PubMed] [Google Scholar]
- 22.Vink M., Zandbelt B.B., Gladwin T., Hillegers M., Hoogendam J.M., van den Wildenberg W.P.M., et al. Frontostriatal activity and connectivity increase during proactive inhibition across adolescence and early adulthood. Hum Brain Mapp. 2014;35:4415–4427. doi: 10.1002/hbm.22483. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Wang H., Fan L., Song M., Liu B., Wu D., Jiang R., et al. Functional connectivity predicts individual development of inhibitory control during adolescence. Cereb Cortex. 2021;31:2686–2700. doi: 10.1093/cercor/bhaa383. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Gao Y., Shuai D., Bu X., Hu X., Tang S., Zhang L., et al. Impairments of large-scale functional networks in attention-deficit/hyperactivity disorder: A meta-analysis of resting-state functional connectivity. Psychol Med. 2019;49:2475–2485. doi: 10.1017/S003329171900237X. [DOI] [PubMed] [Google Scholar]
- 25.Cortese S., Aoki Y.Y., Itahashi T., Castellanos F.X., Eickhoff S.B. Systematic review and meta-analysis: Resting state functional magnetic resonance imaging studies of attention-deficit/hyperactivity disorder. J Am Acad Child Adolesc Psychiatry. 2021;60:61–75. doi: 10.1016/j.jaac.2020.08.014. [DOI] [PubMed] [Google Scholar]
- 26.Sutcubasi B., Metin B., Kurban M.K., Metin Z.E., Beser B., Sonuga-Barke E. Resting-state network dysconnectivity in ADHD: A system-neuroscience-based meta-analysis. World J Biol Psychiatry. 2020;21:662–672. doi: 10.1080/15622975.2020.1775889. [DOI] [PubMed] [Google Scholar]
- 27.Stevens M.C. The contributions of resting state and task-based functional connectivity studies to our understanding of adolescent brain network maturation. Neurosci Biobehav Rev. 2016;70:13–32. doi: 10.1016/j.neubiorev.2016.07.027. [DOI] [PubMed] [Google Scholar]
- 28.Lei D., Du M., Wu M., Chen T., Huang X., Du X., et al. Functional MRI reveals different response inhibition between adults and children with ADHD. Neuropsychology. 2015;29:874–881. doi: 10.1037/neu0000200. [DOI] [PubMed] [Google Scholar]
- 29.Plichta M.M., Scheres A. Ventral-striatal responsiveness during reward anticipation in ADHD and its relation to trait impulsivity in the healthy population: A meta-analytic review of the fMRI literature. Neurosci Biobehav Rev. 2014;38:125–134. doi: 10.1016/j.neubiorev.2013.07.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Posner J., Park C., Wang Z. Connecting the dots: A review of resting connectivity MRI studies in attention-deficit/hyperactivity disorder. Neuropsychol Rev. 2014;24:3–15. doi: 10.1007/s11065-014-9251-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.De La Fuente A., Xia S., Branch C., Li X. A review of attention-deficit/hyperactivity disorder from the perspective of brain networks. Front Hum Neurosci. 2013;7:192. doi: 10.3389/fnhum.2013.00192. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Konrad K., Eickhoff S.B. Is the ADHD brain wired differently? A review on structural and functional connectivity in attention deficit hyperactivity disorder. Hum Brain Mapp. 2010;31:904–916. doi: 10.1002/hbm.21058. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Cao M., Shu N., Cao Q., Wang Y., He Y. Imaging functional and structural brain connectomics in attention-deficit/hyperactivity disorder. Mol Neurobiol. 2014;50:1111–1123. doi: 10.1007/s12035-014-8685-x. [DOI] [PubMed] [Google Scholar]
- 34.Rubia K. Cognitive neuroscience of attention deficit hyperactivity disorder (ADHD) and its clinical translation. Front Hum Neurosci. 2018;12:100. doi: 10.3389/fnhum.2018.00100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Moher D., Liberati A., Tetzlaff J., Altman D.G., Prisma Group Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. PLoS Med. 2009;6 [PMC free article] [PubMed] [Google Scholar]
- 36.Kohl C., McIntosh E.J., Unger S., Haddaway N.R., Kecke S., Schiemann J., Wilhelm R. Online tools supporting the conduct and reporting of systematic reviews and systematic maps: A case study on CADIMA and review of existing tools [published correction appears in Environ Evid 2018; 7:12] Environ Evid. 2018;7:8. [Google Scholar]
- 37.Higgins J.P.T., Green S., The Cochrane Collaboration Cochrane Handbook for Systematic Reviews of Interventions Version 5.1.0 [updated March 2011] 2011. www.handbook.cochrane.org Available at:
- 38.Coghill D.R., Toplak M., Rhodes S., Adamo N. In: Oxford Textbook of Attention Deficit Hyperactivity Disorder. Banaschewski T., Coghill D., Zuddas A., editors. Oxford University Press; Oxford, UK: 2018. Cognitive functioning in ADHD: Inhibition, memory, temporal discounting, decision-making, timing and reaction time variability; pp. 94–102. [Google Scholar]
- 39.Li X., Sroubek A., Kelly M.S., Lesser I., Sussman E., He Y., et al. Atypical pulvinar-cortical pathways during sustained attention performance in children with attention-deficit/hyperactivity disorder. J Am Acad Child Adolesc Psychiatry. 2012;51:1197–1207.e4. doi: 10.1016/j.jaac.2012.08.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Luo Y., Schulz K.P., Alvarez T.L., Halperin J.M., Li X. Distinct topological properties of cue-evoked attention processing network in persisters and remitters of childhood ADHD. Cortex. 2018;109:234–244. doi: 10.1016/j.cortex.2018.09.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Rubia K., Halari R., Cubillo A.I., Mohammad A., Brammer M., Taylor E. Methylphenidate normalises activation and functional connectivity deficits in attention and motivation networks in medication-naïve children with ADHD during a rewarded continuous performance task. Neuropharmacology. 2009;57:640–652. doi: 10.1016/j.neuropharm.2009.08.013. [DOI] [PubMed] [Google Scholar]
- 42.Xia S., Foxe J.J., Sroubek A.E., Branch C., Li X. Topological organization of the “small-world” visual attention network in children with attention deficit/hyperactivity disorder (ADHD) Front Hum Neurosci. 2014;8:162. doi: 10.3389/fnhum.2014.00162. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Hwang S., White S.F., Nolan Z.T., Craig Williams W., Sinclair S., Blair R.J. Executive attention control and emotional responding in attention-deficit/hyperactivity disorder—a functional MRI study. Neuroimage Clin. 2015;9:545–554. doi: 10.1016/j.nicl.2015.10.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Querne L., Fall S., Le Moing A.G., Bourel-Ponchel E., Delignieres A., Simonnot A., et al. Effects of methylphenidate on default-mode network/task-positive network synchronization in children with ADHD. J Atten Disord. 2017;21:1208–1220. doi: 10.1177/1087054713517542. [DOI] [PubMed] [Google Scholar]
- 45.Plessen K.J., Allen E.A., Eichele H., van Wageningen H., Hovik M.F., Sorensen L., et al. Reduced error signalling in medication-naive children with ADHD: Associations with behavioural variability and post-error adaptations. J Psychiatry Neurosci. 2016;41:77–87. doi: 10.1503/jpn.140353. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Vloet T.D., Gilsbach S., Neufang S., Fink G.R., Herpertz-Dahlmann B., Konrad K. Neural mechanisms of interference control and time discrimination in attention-deficit/hyperactivity disorder. J Am Acad Child Adolesc Psychiatry. 2010;49:356–367. [PubMed] [Google Scholar]
- 47.Zamorano F., Billeke P., Kausel L., Larrain J., Stecher X., Hurtado J.M., et al. Lateral prefrontal activity as a compensatory strategy for deficits of cortical processing in attention deficit hyperactivity disorder. Sci Rep. 2017;7:7181. doi: 10.1038/s41598-017-07681-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Cai W., Griffiths K., Korgaonkar M.S., Williams L.M., Menon V. Inhibition-related modulation of salience and frontoparietal networks predicts cognitive control ability and inattention symptoms in children with ADHD. Mol Psychiatry. 2021;26:4016–4025. doi: 10.1038/s41380-019-0564-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Massat I., Slama H., Villemonteix T., Mary A., Baijot S., Albajara Sáenz A., et al. Hyperactivity in motor response inhibition networks in unmedicated children with attention deficit-hyperactivity disorder. World J Biol Psychiatry. 2018;19:101–111. doi: 10.1080/15622975.2016.1237040. [DOI] [PubMed] [Google Scholar]
- 50.Mulder M.J., van Belle J., van Engeland H., Durston S. Functional connectivity between cognitive control regions is sensitive to familial risk for ADHD. Hum Brain Mapp. 2011;32:1511–1518. doi: 10.1002/hbm.21141. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.van Rooij D., Hartman C.A., Mennes M., Oosterlaan J., Franke B., Rommelse N., et al. Altered neural connectivity during response inhibition in adolescents with attention-deficit/hyperactivity disorder and their unaffected siblings. Neuroimage Clin. 2015;7:325–335. doi: 10.1016/j.nicl.2015.01.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Ceceli A.O., Natsheh J.Y., Cruz D., Tricomi E. The neurobehavioral mechanisms of motivational control in attention-deficit/hyperactivity disorder. Cortex. 2020;127:191–207. doi: 10.1016/j.cortex.2020.02.009. [DOI] [PubMed] [Google Scholar]
- 53.Ma I., van Holstein M., Mies G.W., Mennes M., Buitelaar J., Cools R., et al. Ventral striatal hyperconnectivity during rewarded interference control in adolescents with ADHD. Cortex. 2016;82:225–236. doi: 10.1016/j.cortex.2016.05.021. [DOI] [PubMed] [Google Scholar]
- 54.Mowinckel A.M., Alnaes D., Pedersen M.L., Ziegler S., Fredriksen M., Kaufmann T., et al. Increased default-mode variability is related to reduced task-performance and is evident in adults with ADHD. Neuroimage Clin. 2017;16:369–382. doi: 10.1016/j.nicl.2017.03.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Park B.Y., Kim M., Seo J., Lee J.M., Park H. Connectivity analysis and feature classification in attention deficit hyperactivity disorder sub-types: A task functional magnetic resonance imaging study. Brain Topogr. 2016;29:429–439. doi: 10.1007/s10548-015-0463-1. [DOI] [PubMed] [Google Scholar]
- 56.von Rhein D., Beckmann C.F., Franke B., Oosterlaan J., Heslenfeld D.J., Hoekstra P.J., et al. Network-level assessment of reward-related activation in patients with ADHD and healthy individuals. Hum Brain Mapp. 2017;38:2359–2369. doi: 10.1002/hbm.23522. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Bédard A.C., Newcorn J.H., Clerkin S.M., Krone B., Fan J., Halperin J.M., Schulz K.P. Reduced prefrontal efficiency for visuospatial working memory in attention-deficit/hyperactivity disorder. J Am Acad Child Adolesc Psychiatry. 2014;53:1020–1030.e6. doi: 10.1016/j.jaac.2014.05.011. [DOI] [PubMed] [Google Scholar]
- 58.Massat I., Slama H., Kavec M., Linotte S., Mary A., Baleriaux D., et al. Working memory-related functional brain patterns in never medicated children with ADHD. PLoS One. 2012;7 doi: 10.1371/journal.pone.0049392. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Wolf R.C., Plichta M.M., Sambataro F., Fallgatter A.J., Jacob C., Lesch K.P., et al. Regional brain activation changes and abnormal functional connectivity of the ventrolateral prefrontal cortex during working memory processing in adults with attention-deficit/hyperactivity disorder. Hum Brain Mapp. 2009;30:2252–2266. doi: 10.1002/hbm.20665. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Wu Z.M., Bralten J., An L., Cao Q.J., Cao X.H., Sun L., et al. Verbal working memory-related functional connectivity alterations in boys with attention-deficit/hyperactivity disorder and the effects of methylphenidate. J Psychopharmacol. 2017;31:1061–1069. doi: 10.1177/0269881117715607. [DOI] [PubMed] [Google Scholar]
- 61.Hafeman D., Bebko G., Bertocci M.A., Fournier J.C., Chase H.W., Bonar L., et al. Amygdala-prefrontal cortical functional connectivity during implicit emotion processing differentiates youth with bipolar spectrum from youth with externalizing disorders. J Affect Disord. 2017;208:94–100. doi: 10.1016/j.jad.2016.09.064. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Posner J., Nagel B.J., Maia T.V., Mechling A., Oh M., Wang Z., Peterson B.S. Abnormal amygdalar activation and connectivity in adolescents with attention-deficit/hyperactivity disorder. J Am Acad Child Adolesc Psychiatry. 2011;50:828–837.e3. doi: 10.1016/j.jaac.2011.05.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Schulz K.P., Bedard A.C., Fan J., Clerkin S.M., Dima D., Newcorn J.H., Halperin J.M. Emotional bias of cognitive control in adults with childhood attention-deficit/hyperactivity disorder. Neuroimage Clin. 2014;5:1–9. doi: 10.1016/j.nicl.2014.05.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Stoddard J., Tseng W.L., Kim P., Chen G., Yi J., Donahue L., et al. Association of irritability and anxiety with the neural mechanisms of implicit face emotion processing in youths with psychopathology. JAMA Psychiatry. 2017;74:95–103. doi: 10.1001/jamapsychiatry.2016.3282. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Chevrier A., Bhaijiwala M., Lipszyc J., Cheyne D., Graham S., Schachar R. Disrupted reinforcement learning during post-error slowing in ADHD. PLoS One. 2019;14 doi: 10.1371/journal.pone.0206780. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Clerkin S.M., Schulz K.P., Berwid O.G., Fan J., Newcorn J.H., Tang C.Y., Halperin J.M. Thalamo-cortical activation and connectivity during response preparation in adults with persistent and remitted ADHD. Am J Psychiatry. 2013;170:1011–1019. doi: 10.1176/appi.ajp.2013.12070880. [DOI] [PubMed] [Google Scholar]
- 67.Fox M.D., Snyder A.Z., Vincent J.L., Corbetta M., Van Essen D.C., Raichle M.E. The human brain is intrinsically organized into dynamic, anticorrelated functional networks. Proc Natl Acad Sci U S A. 2005;102:9673–9678. doi: 10.1073/pnas.0504136102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Smith S.M., Vidaurre D., Beckmann C.F., Glasser M.F., Jenkinson M., Miller K.L., et al. Functional connectomics from resting-state fMRI. Trends Cogn Sci. 2013;17:666–682. doi: 10.1016/j.tics.2013.09.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Schulz K.P., Krone B., Adler L.A., Bédard A.V., Duhoux S., Pedraza J., et al. Lisdexamfetamine targets amygdala mechanisms that bias cognitive control in attention-deficit/hyperactivity disorder. Biol Psychiatry Cogn Neurosci Neuroimaging. 2018;3:686–693. doi: 10.1016/j.bpsc.2018.03.004. [DOI] [PubMed] [Google Scholar]
- 70.Sheridan M.A., Hinshaw S., D’Esposito M. Stimulant medication and prefrontal functional connectivity during working memory in ADHD. J Atten Disord. 2010;14:69–78. doi: 10.1177/1087054709347444. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Wong C.G., Stevens M.C. The effects of stimulant medication on working memory functional connectivity in attention-deficit/hyperactivity disorder. Biol Psychiatry. 2012;71:458–466. doi: 10.1016/j.biopsych.2011.11.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Rubia K., Criaud M., Wulff M., Alegria A., Brinson H., Barker G., et al. Functional connectivity changes associated with fMRI neurofeedback of right inferior frontal cortex in adolescents with ADHD. Neuroimage. 2019;188:43–58. doi: 10.1016/j.neuroimage.2018.11.055. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Marcos-Vidal L., Martinez-Garcia M., Pretus C., Garcia-Garcia D., Martinez K., Janssen J., et al. Local functional connectivity suggests functional immaturity in children with attention-deficit/hyperactivity disorder. Hum Brain Mapp. 2018;39:2442–2454. doi: 10.1002/hbm.24013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Pretus C., Marcos-Vidal L., Martinez-García M., Picado M., Ramos-Quiroga J.A., Richarte V., et al. Stepwise functional connectivity reveals altered sensory-multimodal integration in medication-naive adults with attention deficit hyperactivity disorder. Hum Brain Mapp. 2019;40:4645–4656. doi: 10.1002/hbm.24727. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.McLeod K.R., Langevin L.M., Goodyear B.G., Dewey D. Functional connectivity of neural motor networks is disrupted in children with developmental coordination disorder and attention-deficit/hyperactivity disorder. Neuroimage Clin. 2014;4:566–575. doi: 10.1016/j.nicl.2014.03.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Sripada C., Kessler D., Fang Y., Welsh R.C., Prem Kumar K., Angstadt M. Disrupted network architecture of the resting brain in attention-deficit/hyperactivity disorder. Hum Brain Mapp. 2014;35:4693–4705. doi: 10.1002/hbm.22504. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Castellanos F.X., Aoki Y. Intrinsic functional connectivity in attention-deficit/hyperactivity disorder: A science in development. Biol Psychiatry Cogn Neurosci Neuroimaging. 2016;1:253–261. doi: 10.1016/j.bpsc.2016.03.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Sergeant J. The cognitive-energetic model: An empirical approach to attention-deficit hyperactivity disorder. Neurosci Biobehav Rev. 2000;24:7–12. doi: 10.1016/s0149-7634(99)00060-3. [DOI] [PubMed] [Google Scholar]
- 79.Ghaderi A.H., Nazari M.A., Shahrokhi H., Darooneh A.H. Functional brain connectivity differences between different ADHD presentations: Impaired functional segregation in ADHD-combined presentation but not in ADHD-inattentive presentation. Basic Clin Neurosci. 2017;8:267–278. doi: 10.18869/nirp.bcn.8.4.267. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Chauvin R.J., Buitelaar J.K., Sprooten E., Oldehinkel M., Franke B., Hartman C., et al. Task-generic and task-specific connectivity modulations in the ADHD brain: An integrated analysis across multiple tasks. Transl Psychiatry. 2021;11:159. doi: 10.1038/s41398-021-01284-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Barkley R.A. Behavioral inhibition, sustained attention, and executive functions: Constructing a unifying theory of ADHD. Psychol Bull. 1997;121:65–94. doi: 10.1037/0033-2909.121.1.65. [DOI] [PubMed] [Google Scholar]
- 82.Martella D., Aldunate N., Fuentes L.J., Sánchez-Pérez N. Arousal and executive alterations in attention deficit hyperactivity disorder (ADHD) Front Psychol. 2020;11:1991. doi: 10.3389/fpsyg.2020.01991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Fair D.A., Bathula D., Nikolas M.A., Nigg J.T. Distinct neuropsychological subgroups in typically developing youth inform heterogeneity in children with ADHD. Proc Natl Acad Sci U S A. 2012;109:6769–6774. doi: 10.1073/pnas.1115365109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Fair D.A., Nigg J.T., Iyer S., Bathula D., Mills K.L., Dosenbach N.U.F., et al. Distinct neural signatures detected for ADHD subtypes after controlling for micro-movements in resting state functional connectivity MRI data. Front Syst Neurosci. 2013;6:80. doi: 10.3389/fnsys.2012.00080. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Silk T.J., Malpas C., Vance A., Bellgrove M.A. The effect of single-dose methylphenidate on resting-state network functional connectivity in ADHD. Brain Imaging Behav. 2017;11:1422–1431. doi: 10.1007/s11682-016-9620-8. [DOI] [PubMed] [Google Scholar]
- 86.Yoo J.H., Kim D., Choi J., Jeong B. Treatment effect of methylphenidate on intrinsic functional brain network in medication-naïve ADHD children: A multivariate analysis. Brain Imaging Behav. 2018;12:518–531. doi: 10.1007/s11682-017-9713-z. [DOI] [PubMed] [Google Scholar]
- 87.Picon F.A., Sato J.R., Anes M., Vedolin L.M., Mazzola A.A., Valentini B.B., et al. Methylphenidate alters functional connectivity of default mode network in drug-naive male adults with ADHD. J Atten Disord. 2020;24:447–455. doi: 10.1177/1087054718816822. [DOI] [PubMed] [Google Scholar]
- 88.Cary R.P., Ray S., Grayson D.S., Painter J., Carpenter S., Maron L., et al. Network structure among brain systems in adult ADHD is uniquely modified by stimulant administration. Cereb Cortex. 2017;27:3970–3979. doi: 10.1093/cercor/bhw209. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Pereira-Sanchez V., Franco A.R., Vieira D., de Castro-Manglano P., Soutullo C., Milham M.P., Castellanos F.X. Systematic review: Medication effects on brain intrinsic functional connectivity in patients with attention-deficit/hyperactivity disorder. J Am Acad Child Adolesc Psychiatry. 2021;60:222–235. doi: 10.1016/j.jaac.2020.10.013. [DOI] [PubMed] [Google Scholar]
- 90.Kriegeskorte N., Simmons W.K., Bellgowan P.S., Baker C.I. Circular analysis in systems neuroscience: The dangers of double dipping. Nat Neurosci. 2009;12:535–540. doi: 10.1038/nn.2303. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Nakao T., Radua J., Rubia K., Mataix-Cols D. Gray matter volume abnormalities in ADHD: Voxel-based meta-analysis exploring the effects of age and stimulant medication. Am J Psychiatry. 2011;168:1154–1163. doi: 10.1176/appi.ajp.2011.11020281. [DOI] [PubMed] [Google Scholar]
- 92.Frodl T., Skokauskas N. Meta-analysis of structural MRI studies in children and adults with attention deficit hyperactivity disorder indicates treatment effects. Acta Psychiatr Scand. 2012;125:114–126. doi: 10.1111/j.1600-0447.2011.01786.x. [DOI] [PubMed] [Google Scholar]
- 93.Konrad K., Neufang S., Fink G.R., Herpertz-Dahlmann B. Long-term effects of methylphenidate on neural networks associated with executive attention in children with ADHD: Results from a longitudinal functional MRI study. J Am Acad Child Adolesc Psychiatry. 2007;46:1633–1641. doi: 10.1097/chi.0b013e318157cb3b. [DOI] [PubMed] [Google Scholar]
- 94.Fusar-Poli P., Rubia K., Rossi G., Sartori G., Balottin U. Striatal dopamine transporter alterations in ADHD: Pathophysiology or adaptation to psychostimulants? A meta-analysis. Am J Psychiatry. 2012;169:264–272. doi: 10.1176/appi.ajp.2011.11060940. [DOI] [PubMed] [Google Scholar]
- 95.Dhariwal K., Jackson A. Effect of length of sampling schedule and washout interval on magnitude of drug carryover from period 1 to period 2 in two-period, two-treatment bioequivalence studies and its attendant effects on determination of bioequivalence. Biopharm Drug Dispos. 2003;24:219–228. doi: 10.1002/bdd.359. [DOI] [PubMed] [Google Scholar]
- 96.Buitelaar J.K., Asherson P., Soutullo C., Colla M., Adams D.H., Tanaka Y., et al. Differences in maintenance of response upon discontinuation across medication treatments in attention-deficit/hyperactivity disorder. Eur Neuropsychopharmacol. 2015;25:1611–1621. doi: 10.1016/j.euroneuro.2015.06.003. [DOI] [PubMed] [Google Scholar]
- 97.Button K.S., Ioannidis J.P., Mokrysz C., Nosek B.A., Flint J., Robinson E.S., Munafò M.R. Power failure: Why small sample size undermines the reliability of neuroscience. Nat Rev Neurosci. 2013;14:365–376. doi: 10.1038/nrn3475. [DOI] [PubMed] [Google Scholar]
- 98.Poldrack R.A., Baker C.I., Durnez J., Gorgolewski K.J., Matthews P.M., Munafò M.R., et al. Scanning the horizon: Towards transparent and reproducible neuroimaging research. Nat Rev Neurosci. 2017;18:115–126. doi: 10.1038/nrn.2016.167. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Szucs D., Ioannidis J.P. Empirical assessment of published effect sizes and power in the recent cognitive neuroscience and psychology literature. PLoS Biol. 2017;15 doi: 10.1371/journal.pbio.2000797. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Thirion B., Pinel P., Mériaux S., Roche A., Dehaene S., Poline J.B. Analysis of a large fMRI cohort: Statistical and methodological issues for group analyses. Neuroimage. 2007;35:105–120. doi: 10.1016/j.neuroimage.2006.11.054. [DOI] [PubMed] [Google Scholar]
- 101.Turner B.O., Paul E.J., Miller M.B., Barbey A.K. Small sample sizes reduce the replicability of task-based fMRI studies. Commun Biol. 2018;1:62. doi: 10.1038/s42003-018-0073-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Mumford J.A. A power calculation guide for fMRI studies. Soc Cogn Affect Neurosci. 2012;7:738–742. doi: 10.1093/scan/nss059. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Epstein J.N., Casey B.J., Tonev S.T., Davidson M., Reiss A.L., Garrett A., et al. Assessment and prevention of head motion during imaging of patients with attention deficit hyperactivity disorder. Psychiatry Res Neuroimaging. 2007;155:75–82. doi: 10.1016/j.pscychresns.2006.12.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Pardoe H.R., Kucharsky Hiess R., Kuzniecky R. Motion and morphometry in clinical and nonclinical populations. Neuroimage. 2016;135:177–185. doi: 10.1016/j.neuroimage.2016.05.005. [DOI] [PubMed] [Google Scholar]
- 105.Yerys B.E., Jankowski K.F., Shook D., Rosenberger L.R., Barnes K.A., Berl M.M., et al. The fMRI success rate of children and adolescents: Typical development, epilepsy, attention deficit/hyperactivity disorder, and autism spectrum disorders. Hum Brain Mapp. 2009;30:3426–3435. doi: 10.1002/hbm.20767. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Goto M., Abe O., Miyati T., Yamasue H., Gomi T., Takeda T. Head motion and correction methods in resting-state functional MRI. Magn Reson Med Sci. 2015;15:178–186. doi: 10.2463/mrms.rev.2015-0060. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Spisák T., Jakab A., Kis S.A., Opposits G., Aranyi C., Berényi E., Emri M. Voxel-wise motion artifacts in population-level whole-brain connectivity analysis of resting-state fMRI. PLoS One. 2014;9 doi: 10.1371/journal.pone.0104947. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Satterthwaite T.D., Ciric R., Roalf D.R., Davatzikos C., Bassett D.S., Wolf D.H. Motion artifact in studies of functional connectivity: Characteristics and mitigation strategies. Hum Brain Mapp. 2019;40:2033–2051. doi: 10.1002/hbm.23665. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Bennett C.M., Wolford G.L., Miller M.B. The principled control of false positives in neuroimaging. Soc Cogn Affect Neurosci. 2009;4:417–422. doi: 10.1093/scan/nsp053. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Poldrack R.A. The future of fMRI in cognitive neuroscience. Neuroimage. 2012;62:1216–1220. doi: 10.1016/j.neuroimage.2011.08.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Faraone S., Asherson P., Banaschewski T., Biederman J., Buitelaar J., Ramos-Quiroga J., et al. Attention-deficit/hyperactivity disorder. Nat Rev Dis Primer. 2015;1:15020. doi: 10.1038/nrdp.2015.20. [DOI] [PubMed] [Google Scholar]
- 112.Castellanos F.X., Tannock R. Neuroscience of attention-deficit/hyperactivity disorder: The search for endophenotypes. Nat Rev Neurosci. 2002;3:617–628. doi: 10.1038/nrn896. [DOI] [PubMed] [Google Scholar]
- 113.Saad J.F., Griffiths K.R., Korgaonkar M.S. A systematic review of imaging studies in the combined and inattentive subtypes of attention deficit hyperactivity disorder. Front Integr Neurosci. 2020;14:31. doi: 10.3389/fnint.2020.00031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Kolodny T., Mevorach C., Stern P., Biderman N., Ankaoua M., Tsafrir S., Shalev L. Fronto-parietal engagement in response inhibition is inversely scaled with attention-deficit/hyperactivity disorder symptom severity. Neuroimage Clin. 2020;25:102119. doi: 10.1016/j.nicl.2019.102119. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.