Abstract
Pre-existing Alzheimer's disease is a risk factor for severe/fatal COVID-19 and infection by SARS-CoV2 virus has been associated with an increased incidence of un-masked Alzheimer's disease. The molecular basis whereby SARS-CoV2 may amplify Alzheimer's disease is not well understood. This study analyzed the molecular changes in autopsy brain tissues from people with pre-existing dementia who died of COVID-19 (n = 5) which was compared to equivalent tissues of people who died of COVID-19 with no history of dementia (n = 8), Alzheimer's disease pre-COVID-19 (n = 10) and aged matched controls (n = 10) in a blinded fashion. Immunohistochemistry analyses for hyperphosphorylated tau protein, α-synuclein, and β-amyloid-42 confirmed the diagnoses of Alzheimer's disease (n = 4), and Lewy body dementia (n = 1) in the COVID-19 group. The brain tissues from patients who died of COVID-19 with no history of dementia showed a diffuse microangiopathy marked by endocytosis of spike subunit S1 and S2 in primarily CD31+ endothelia with strong co-localization with ACE2, Caspase-3, IL6, TNFα, and Complement component 6 that was not associated with SARS-CoV2 RNA. Microglial activation marked by increased TMEM119 and MCP1 protein expression closely paralleled the endocytosed spike protein. The COVID-19 tissues from people with no pre-existing dementia showed, compared to controls, 5-10× fold increases in expression of neuronal NOS and NMDAR2 as well as a marked decrease in the expression of proteins whose loss is associated with worsening Alzheimer's disease: MFSD2a, SHIP1, BCL6, BCL10, and BACH1. In COVID-19 tissues from people with dementia the widespread spike-induced microencephalitis with the concomitant microglial activation co-existed in the same areas where neurons had hyperphosphorylated tau protein suggesting that the already dysfunctional neurons were additionally stressed by the SARS-CoV2 induced microangiopathy. ACE2+ human brain endothelial cells treated with high dose (but not vaccine equivalent low dose) spike S1 protein demonstrated each of the molecular changes noted in the in vivo COVID-19 and COVID-19/Alzheimer's disease brain tissues. It is concluded that fatal COVID-19 induces a diffuse microencephalitis and microglial activation in the brain due to endocytosis of circulating viral spike protein that amplifies pre-existing dementia in at least two ways: 1) modulates the expression of proteins that may worsen Alzheimer's disease and 2) stresses the already dysfunctional neurons by causing an acute proinflammatory/hypercoagulable/hypoxic microenvironment in areas with abundant hyperphosphorylated tau protein and/or βA-42.
Keywords: Encephalitis, COVID-19, Endothelialitis, Spike Protein, Microvasculature
1. Introduction
Over one-half billion people have been infected with SARS-CoV2 with nearly 7 million deaths since the pandemic began in Dec 2019. It is now well established that obesity and its related disease type II diabetes mellitus are the key risk factors for severe COVID-19 [1], [2] and that the brain and heart are highly susceptible to pathologic changes [3], [4], [5], [6]. Whereas it was initially assumed that systemic infectious SARS-CoV2 was responsible for non-pulmonary manifestations of severe COVID-19, it is now clear that infectious virus is rarely identified in the blood or organs besides the lung or nasopharyx [7], [8], [9], [10]. Indeed, many investigators have documented that SARS-CoV2 spike protein per se, either in vitro, or in vivo in mouse models, can elicit the cytokine storm and direct CNS and cardiac damage typical of severe COVID-19 [11], [12], [13], [14], [15], [16], [17], [18], [19]. Importantly circulating levels of spike protein post vaccination (50–200 pg/ml) are not associated with disease in these models [20]. Rather spike protein levels of 20,000 to 30,000 pg/ml, as have been found in the sera of people with severe COVID-19 [14], are associated with severe disease in these in vitro and in vivo models. The source of such high circulating levels of SARS-CoV2 spike protein likely is the degeneration of the extremely high viral load in the lung due to the widespread complement mediated microangiopathy [7], [21].
It is well documented that there are strong links between Alzheimer's disease and COVID-19 [22], [23], [24], [25], [26], [27], [28], [29]. COVID-19 can unmask pre-clinical dementia and people with pre-existing Alzheimer's disease are more likely to get severe COVID-19. The molecular bases of these strong clinical correlates are not well understood. N-methyl-d-aspartate receptor (NMDAR) and neuronal nitric oxide synthase (nNOS) have each been implicated in the pathophysiology of Alzheimer's disease. nNOS is over-expressed in GABA producing neurons in Alzheimer's disease which has, in in vitro models of the disease, led to increased neuronal death and, in turn, increased NMDAR-based excitatory glutamatergic neurotransmission [30], [31], [32], [33], [34]. The latter has also been associated with neuronal loss in Alzheimer's disease. The reduction of other proteins, including Src homology region 2 domain-containing phosphatase-1 (SHIP1) [35], [36], BTB Domain And CNC Homolog 1 (BACH1) [37], [38], BCL6 Transcription Repressor (BCL6) [39], BCL10 Immune Signaling Adaptor (BCL10) [40], [41], and Major Facilitator Superfamily Domain Containing 2A (MFSD2A) [42], [43], have each been linked to progressive Alzheimer's disease. It has been shown in in vivo mouse models of COVID-19 that high circulating levels of spike protein can modulate some of these proteins in the brain and we have previously reported altered expression of NMDAR2, nNOS, SHIP1, and MFSD2A in the brains of people who died of COVID-19 who did not have a history of pre-existing dementia [11].
Another clinical link between Alzheimer's disease and COVID-19 is that obesity and type II diabetes have been associated with each disease [1], [2]. It has been postulated that the pro-inflammatory state characteristic of each disease, including increased IL6 expression, may be one of the molecular links [44]. Also, obesity has been associated with increased ACE2 and Furin expression, each of which may facilitate spike protein cell entry [26], [45], [46]. Further, the hypercoagulable state, both in terms of microthrombi and large vessel thrombi, have been extensively reported in both severe COVID-1 and obesity [21], [47].
The purpose of this study was to examine brain tissues from 13 people who either died of COVID-19 with no evidence of dementia or had pre-existing dementia and compare the molecular/histologic/viral findings in a blinded fashion to 10 pre-COVID normal controls and 10 pre-COVID-19 Alzheimer's disease to better define the mechanism(s) whereby SARS-CoV2 can induce or amplify the brain disease in people with pre-existing dementia. The importance of the hematoxylin and eosin findings as well as the in situ based data underscores the importance of the diagnostic pathologist in the interpretation of these data.
2. Materials and methods
2.1. COVID-19 autopsies and controls
Complete autopsy material was available from 13 people who died of COVID-19. They ranged in age from 50 to 92 (mean 75; seven men and six women). The brain tissues from ten aged-matched controls from people who died prior to 2016 and had no history of dementia served as negative controls. Included in the study were ten cases of people with documented Alzheimer's disease who had died prior to the pandemic (2015–2018). In each case from two to eight formalin fixed, paraffin embedded blocks of brain tissue were available that were obtained from either the frontal cortex, the hippocampus, or midbrain/brainstem. Salient clinical information included five of the COVID-19 cases had pre-existing dementia (Alzheimer's disease n = 4 and Lewy body dementia n = 1); only one of these patients (Lewy body dementia) had co-existing obesity. Of the other eight fatal COVID-19 cases with no pre-existing history of dementia, seven had a history of obesity/morbid obesity that in six cases was associated with type II diabetes mellitus. Data from the latter eight cases has been reported previously [11]; however, the data reported in this paper was newly derived after the publication.
2.2. Immunohistochemistry
Immunohistochemistry was done as previously reported [48], [49]. The SARS-CoV2 specific antibodies were from ProSci (Poway, CA) and were directed against spike subunit 1, spike subunit 2, and the nucleocapsid protein. Antibodies directed against ACE2 and Furin were also from ProSci. Antibodies against hyperphosphorylated tau protein, α-synuclein, β-amyloid 42, activated Caspase-3, IL6, TNFα, SHIP1, BACH1, MFSD2A, NMDAR2, CD31, CD41, TMEM119, CD3, CD20, CD11b, CD163, MCP-1, and CD206 were from Abcam (Cambridge, UK). Enzo Life Sciences (Farmingdale, NY) was the source of BCL10 and BCL6, whereas Complement component 6 was from Proteintech Laboratories (Chicago, IL). The protocols for these antibodies have been published [48]. In brief, all required pretreatment (antigen retrieval) for 30 min at 95 °C using an EDTA solution except for hyperphosphorylated tau protein, α-synuclein, and β-amyloid-42 which each was optimized with pre-treatment in proteinase K for 4 min at room temperature.
The immunohistochemistry protocol used the Leica Bond Max automated platform. Both the Fast red (DS 9820) and the DAB (DS 9800) detection kits from Leica Biosystems (Buffalo Grove, IL) were used and gave equivalent results. The HRP conjugate from Enzo Life Sciences was used in cases in place of the equivalent reagent from Leica in the DAB kit as this has been shown to reduce background for some primary antibodies [49].
2.3. In situ hybridization
Detection of SARS-CoV-2 RNA was done using the ACD RNAscope (Newark, California, USA) probe (Cat No. 848561-C3) and the Enzo Life Sciences LOOP RNA assay (ENZ-GEN157 AMPIVIEW™ SARS-CoV-2 RNA Probes and ENZ-ACC152 AMPIVIEW™ Hybridization buffer) using the manufacturer's recommended protocol as previously published [48], [49].
2.4. Cell culture
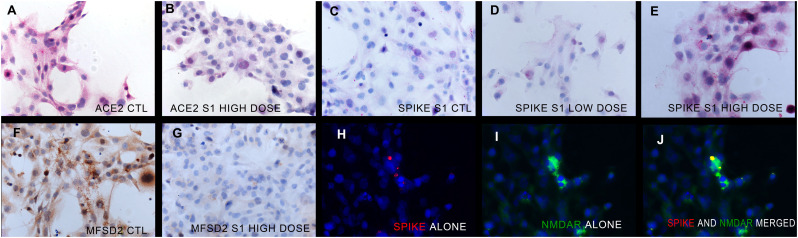
A human cerebral microvascular endothelial cell line (HBEC) prepared from cerebral cortex was purchased from ATCC (ATCC-CRL3245). The THP-1 human monocytic cell line was also purchased from ATCC. HBEC cells were grown in DMEM: F-12 medium supplemented with 10 % FBS, and standard concentration of Penicillin/Streptomycin. THP-1 cells were grown in RPMI-1460 supplemented with 10 % FBS, and standard concentration of Penicillin/Streptomycin. Cells were treated with PBS (mock control), and either 1 ng/ml (low dose), or 1μg/ml (high dose) recombinant spike S1 subunit (ProSci, catalogue # 10–300) for 48 h. Cells were fixed in 4 % formalin and were analyzed for the same molecular events as the human brain samples after spike S1 subunit treatment.
2.5. Quantification, co-expression and statistical analyses
As previously described, co-expression experiments were performed by analyzing a given tissue section for one protein using the DAB (brown) chromogen and analyzing the other protein with Fast Red chromogen; this is possible when the two antibodies optimal pre-treatment windows overlap [48], [49]. Co-expression analyses were done using the Nuance/InForm system whereby each chromogenic signal is separated, converted to a fluorescence-based signal, then mixed to determine what percentage of cells were expressing the two proteins of interest as previously described [49]. Quantification for the signal with either single immunohistochemistry or double-labeled immunohistochemistry was done using either the InForm software or manual counting which yielded equivalent results. In cases where the endothelial cells were quantified, either CD31 or cytologic visualization of microvascular endothelial cells were done to identify the number of microvessels/unit area. Statistical analysis was done using the InStat Statistical Analysis Software (version 3.36) and a paired t-test (also referred to as a “repeated measure t-test”). The null hypothesis was rejected if the significance level was below 5 %.
3. Results
3.1. Clinical/pathologic correlation with documentation of dementia-related changes
A total of 52 formalin fixed, paraffin embedded tissues from the brain were available for study. They were derived from 13 people who died of COVID-19 (n = 32 blocks), 10 age matched pre-2019 controls (n = 10 blocks), and 10 people diagnosed with Alzheimer's disease prior to the pandemic (n = 10 blocks). The fatal COVID-19 cases ranged in age from 50 to 92 (mean 75; seven men and six women). The brain tissues were obtained from either the forebrain (frontal/temporal) cortex or hippocampus (32 blocks), or hindbrain (20 blocks). Salient clinical information included five of the COVID-19 cases had pre-existing dementia (Alzheimer's disease n = 4 with two men (aged 81 and 91) and two women (aged 72 and 76) and Lewy body dementia n = 1, aged 66 woman); only the latter case had co-existing obesity. Of the other eight fatal COVID-19 cases with no pre-existing history of dementia, seven had a history of obesity/morbid obesity that in six cases was associated with type II diabetes mellitus.
To confirm the clinical information, the brain tissues were tested for hyperphosphorylated tau protein, α-synuclein, and β-amyloid-42 blinded to the clinical information. Only one of the controls showed a weak signal for β-amyloid-42. The hyperphosphorylated tau protein signal was evident only in the four cases with Alzheimer's disease where it varied from rare positive cells (1–5/cm2) to many positive cells (>20/cm2), even in a given patient (Fig. 1 ). Lewy bodies positive for α-synuclein were seen in the one case of Lewy body dementia and, rarely, in a case diagnosed as Alzheimer's disease where it co-existed with hyperphosphorylated tau protein.
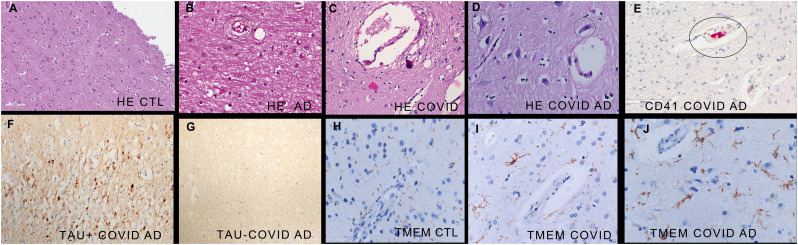
Fig. 1.
Basic pathologic changes in the brain in people with pre-existing Alzheimer's disease who died of COVID-19. The normal brain shows an inconspicuous microvasculature in the brain parenchyma (panel A). The microvasculature is likewise inconspicuous in Alzheimer's disease where retraction artifact around the small vessels can be seen at high magnification (panel B, oval). Note the microvascular changes in the brain in fatal COVID-19, seen as marked perivascular edema and endothelial cell degeneration (swelling or pyknosis) (panel C, COVID-19). Also note the close proximity of the microvasculature changes to neurons (panel D, ovals, COVID-19/Alzheimer's disease). Microthrombi are highlighted with the CD41 immunohistochemistry (signal red, oval). COVID-19 in pre-existing Alzheimer's disease shows the marked heterogeneity of hyperphosphorylated tau protein distribution with sections only 0.5 cm away showing either an intense signal (panel F) or no signal (panel G, signal brown). Panels H-J show the marked increased in microglial TMEM119 signal in COVID-19 (panel I) and COVID-19/Alzheimer's disease (panel J) relative to the control brains (panel H, signal brown). (For interpretation of the references to colour in this figure legend, the reader is referred to the web version of this article.)
The hematoxylin and eosin stains were then reviewed blinded to the clinical information. The following variables were scored: peri-microvascular edema, endothelial cell degeneration (necrotic cells or enlarged nuclei), microthrombi, duplication of microvessels, and mononuclear cell infiltrates. It was evident that the peri-vascular edema and endothelial cell degeneration often co-existed and, thus, these two variables were grouped together as microvascular damage. Microthrombi were limited to the 13 COVID-19 cases, with the density equivalent between the COVID-19 non-dementia cases (5.3+ microvessels/cm2 – SEM 1.5) and the Alzheimer's disease COVID-19 cases (5.9+ microvessels/cm2 – SEM 1.1) (Fig. 1). The one case of Lewy body dementia/COVID-19 showed 4.9+ microvessels/cm2 -SEM 0.8 with microthrombi. No microthrombi were seen in the controls or pre-COVID-19 Alzheimer's disease cases. Fig. 1 demonstrates that the microthrombi were positive for the platelet aggregation marker CD41. Microvascular damage was also limited to the COVID-19 cases where it was seen in 12.9 microvessels/cm2 – SEM 2.0 in the fatal non-dementia cases and 13.8/microvessels/cm2 – SEM 1.8. in the Alzheimer's disease/COVID-19 cases; the one Lewy body dementia case showed 11.3 microvessels/cm2 – SEM 1.4) with microvascular damage (Fig. 1).
3.2. Adaptive and innate immune cell response
The histologic findings were consistent with a diffuse microendothelialitis which would be expected to elicit an immune response especially given how widespread it was in both the forebrain and hindbrain sections from the people who died of COVID-19, irrespective of pre-existing dementia. Thus, the tissues were interrogated for TMEM119, the marker for endogenous microglia, CD3/CD20 (infiltrating T and B cells) as well as the markers of infiltrating macrophages: CD11b, CD163, and CD206 in a blinded fashion. The numbers of B and T cells were extremely low in all cases (<1+ cell/cm2) including the COVID-19 and COVID-19/Alzheimer's disease cases though rare small perivascular infiltrates of CD3+ cells were seen in several COVID-19 cases. Similarly, the numbers of CD11b, CD163, and CD206+ cells were statistically equivalent in the controls versus COVID-19 (with or without dementia) and as follows (COVID cases include with and without pre = existing dementia): CD11b = 11.1/200× field in normal cases versus 12.0/200× field in COVID-19 cases; CD163 = 1.9/200× field in normal cases versus 2.3/200× field in COVID-19 and CD206 = 0.9/200× field in normal cases versus 1.0/200× field in COVID-19 cases. However, the numbers of TMEM119+ cells were dramatically increased in the COVID-19 cases relative to the controls (Fig. 1). Specifically, the density of TMEM119+ processes increased by 4.2 to 6.1× relative to baseline (the normal controls) in the COVID-19 brain tissues with equivalent results in the COVID-19/Alzheimer's disease samples (p, 0.001); note that the TMEM119-positive fibers often intertwine with the microvessels (Fig. 1). We also analyzed COVID-19 brains for hemosiderin extravasation, a sign of vascular leakage, and found that only 1 of 13 cases showed hemosiderin extravasation. Thus, the histologic and basic immune cell immunohistochemistry strongly suggested a diffuse microencephalitis in fatal COVID-19 brain that was based in a microangiopathy, marked by perivascular edema, endothelial cell degeneration, and microthrombi, and where the immune response was primarily seated in the reactive endogenous microglia and not infiltrating immune cells.
3.3. Distribution of SARS-CoV2 proteins and RNA in the brain sections as well as ACE2
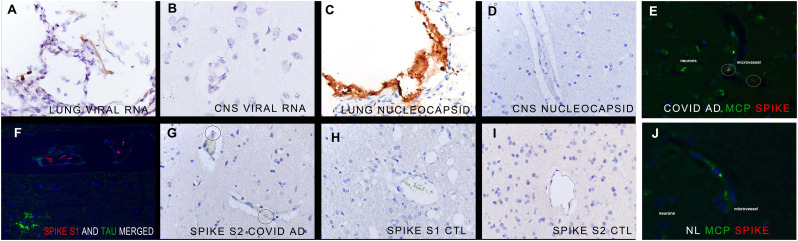
SARS-CoV2 RNA was evident in only 2/13 COVID-19 brain samples (0/5 dementia/COVID-19 cases) and in rare cells in the two positive cases, despite documenting that the lung tissues from most of these people had very high copy SARS-CoV2 RNA (Fig. 2 ). Serial sections of the controls and the COVID-19 cases were then analyzed for the viral spike proteins (subunits 1 and 2, (S1, S2)) and the nucleocapsid protein in a blinded fashion. The nucleocapsid protein was only detected, and rarely, in 2/13 COVID-19 cases (0/5 COVID-19/dementia cases) and none of the controls; these were the same two cases with rare viral RNA. Contrary to the viral RNA and the nucleocapsid, spike S1 and S2 subunits were abundant in the COVID-19 cases and not evident in the controls. Specifically, the percentage of microvessels with at least one cell that stained positive for spike S1 was 21.2 (SEM 4.1) in the COVID-19 non-dementia cases and 23.1 (SEM 5.9) in the COVID-19 Alzheimer's disease cases which was statistically equivalent. The spike density for the one Lewy body dementia case was 19.8 with an of SEM 3.1. The data was equivalent for spike S2 in serial sections: 20.3 % of microvessels (SEM 4.5) in the COVID-19 non-dementia cases and 21.1 % (SEM 5.4) in the COVID-19/Alzheimer's disease cases. Co-localization of spike subunits S1 and S2 was done next where 1–25 % co-expression was defined as weak, 26–49 % co-expression as moderate, and 50 % or greater as strong. The S1 and S2 subunits of SARS-CoV2 spike had the same distribution in serial sections (Fig. 2) as well as strong co-localization (data not shown). Fig. 2 demonstrates the close proximity of the viral spike protein with hyperphosphorylated tau protein in the Alzheimer's disease cases. Fig. 2 also demonstrates the close proximity of spike protein with microglial activation as evidenced by MCP1 expression in the COVID-19 cases which was similar in the cases with no history of dementia and those with pre-existing dementia. Note that the marked increase expression of MCP1 was evident in both the microglia and surrounding neurons. This group of experiments showed that abundant spike but nor viral RNA or nucleocapsid is found in the brain tissue of COVID-19 patients, and that there is no statistical difference in the amount of spike present in COVID-19 cases with or without dementia.
Fig. 2.
Detection of SARS-CoV2 RNA and proteins in the brain in people with pre-existing Alzheimer's disease who died of COVID-19. Although lung sections commonly showed intense signal for SARS-CoV2 RNA (panel A) and nucleocapsid protein (panel C), these signals were usually absent in the corresponding brain tissues (panels B and D; each signal brown). Panel E shows the close proximity of the endocytosed spike protein (circles, fluorescent red) in a microvessel to the MCP1 protein being expressed in microglia and neurons (fluorescent green); panel J shows the lack of a signal for spike and rare MCP1 expression in the normal brain control. Panels F and G are serial sections from the brain of a person with pre-existing Alzheimer's disease who died of COVID-19. Note the localization of the spike S1 signal to the microvasculature (fluorescent red) and its close proximity to neurofibrillary tangles (fluorescent green) after co-expression (panel F). Panel G shows the S2 spike signal has the same distribution (signal red, circles); note the perivascular edema and endothelial cell swelling. Panels H and I show the absence of any signal for spike S1 or S2, respectively, in the normal control brains or Alzheimer's disease brains (not shown). (For interpretation of the references to colour in this figure legend, the reader is referred to the web version of this article.)
Next, the expression of ACE-2, the cellular receptor for the virus where spike binds [50], was compared to that of spike S1 in the brain tissues as defined by the percentage of microvessels that showed a signal for ACE-2 in the endothelia. The controls showed a wide range of ACE-2 expression (9.3 to 33.4 %. mean 19.4) as did the pre-COVID-19 Alzheimer's disease cases (14.3 to 62.4 % mean 29.7). The COVID-19 cases were subdivided into the Alzheimer's disease cases (no patient had co-existing obesity) and the six COVID-19 cases without dementia where there was pre-existing obesity and type II diabetes. The ACE-2 expression value was significantly greater for the latter (76.2 %, SEM 9.2) than for the COVID-19 cases with Alzheimer's disease and no pre-existing obesity (23.3 %, SEM 3,3), p < 0.001. Thus, the data suggested that in the fatal COVID-19 cases that obesity and type II diabetes was strongly correlated with ACE-2 expression in the microvessels of the brain. However, a comparison of the percentage of microvessels with detectable spike S1 showed equivalent results (21.9 % (SEM 4.4) in the COVID-19 non-dementia cases with obesity and 23.1 % (SEM 5.9) in the COVID-19 Alzheimer's disease cases. Co-expression of S1 and ACE2 in both groups did show that >50 % of the spike S1+ cells also expressed ACE 2 (Fig. 3 ).
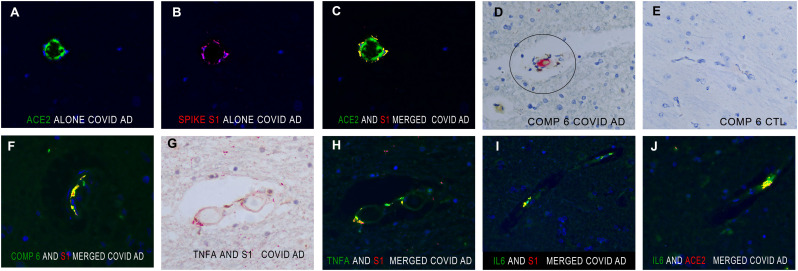
Fig. 3.
Documentation of the host response to the endocytosed spike protein in the brain in people with pre-existing Alzheimer's disease who died of COVID-19. Panels A-C show the co-expression of ACE2 (panel A), spike S1 (panel B) and the merged image in panel C; note the strong co-localization of the two proteins (fluorescent yellow). Panel D shows the strong signal for complement component 6 in a microvessel with marked microangiopathy (oval, signal red); panel E shows the lack of any signal in the control brains. Panel F demonstrates that complement component 6 (fluorescent green) strongly co-localizes with the endocytosed spike protein (fluorescent red merged image shows co-localization as fluorescent yellow). Panel G shows the damaged microvessel after testing for TNFα and spike S1 where panel H shows the fluorescent derived image documenting strong co-localization as fluorescent yellow. Panels I and J show that IL6 likewise co-localizes with spike S1 (panel I) and ACE2 (panel J) in the brains of people with pre-existing Alzheimer's disease who died of COVID-19. (For interpretation of the references to colour in this figure legend, the reader is referred to the web version of this article.)
3.4. Molecular changes associated with SARS-CoV2 endocytosis: pro-inflammatory cytokine/hypercoagulable milieu
The controls and COVID-19 cases were then tested for Caspase-3, IL6, TNFα, CD41 (platelet aggregates) and Complement component 6 using serial sections each four microns apart from the spike S1 data. The documentation of the same distribution of proteins relative to the viral protein is an indicator of specificity [48]. As expected, there was no expression of these proteins in the controls with the exception of minimal background for Caspase-3. Both the COVID-19 cases with no pre-existing dementia and the COVID-19 cases with pre-existing Alzheimer's disease showed equivalent findings: there was strong expression of Caspase-3, IL6, TNFα, CD41 and Complement component 6 that showed the same distribution as spike S1 and S2. Specifically, the percentage of microvessels with at least one cell that stained positive for Caspase-3, IL6, TNFα, or Complement component 6 ranged from 18.9 to 27.9 (mean 22.2) in the COVID-19 non-dementia cases and 17.8 to 29.9 (mean 24.0) in the COVID-19/Alzheimer's disease cases which was statistically equivalent. Co-expression experiments documented a strong co-localization of each of the mentioned proteins with spike S1 (Fig. 3); note the strong association of the protein co-localized with S1 with microvascular damage.
The Alzheimer's disease/COVID-19 cases, as noted, showed marked disparity of hyperphosphorylated tau protein positive neurons in different sections, even from the same person (Fig. 1). Thus, we quantified the spike protein detection in the areas of the brain with abundant hyperphosphorylated tau protein and compared this to areas with little to no hyperphosphorylated tau protein in the Alzheimer's disease cases. Although there was a tendency for higher spike/cytokine density in the hyperphosphorylated tau protein + areas, it did not reach statistical significance (spike density in areas that stained positive for hyperphosphorylated tau protein was 25.5 % of microvessels with SEM of 5.1 versus 22.9 % of microvessels with SEM of 6.1 in sections with minimal hyperphosphorylated tau protein). The neurons with hyperphosphorylated tau protein were often within 500 um of microvessels with spike protein (Fig. 2).
3.5. Molecular changes associated with SARS-CoV2 endocytosis that may augment the CNS damage in Alzheimer's disease
Next, proteins that have been associated with worsening Alzheimer's disease when dysregulated were analyzed to determine if they were altered in fatal COVID-19 disease in the brain. The controls and all COVID-19 samples were tested for Furin, that cleaves spike into S1 and S2 [45], nNOS, NMDAR2, BACH1, BCL6, BCL10, SHIP1, and MFSD2a. The controls showed strong expression of BCL6, BCL10 as well as BACH1, and MFSD2a. BCL6 and BCL10 were expressed mostly in CD31+ endothelial cells in microvessels whereas BACH1 and MFSD2a was present in endothelial and surrounding neurons; SHIP1 was mostly neuronal based. Conversely, nNOS, NMDAR2, and Furin were rarely or not expressed in the control brains (Fig. 4 ).
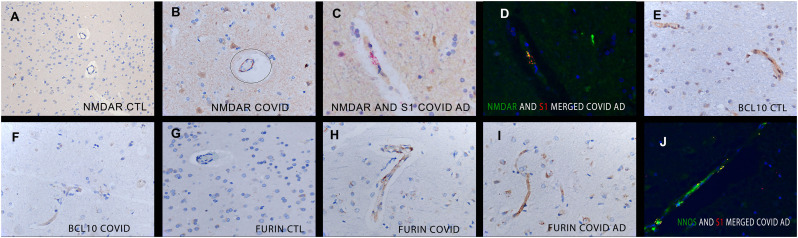
Fig. 4.
Dysregulation of key Alzheimer's disease-related proteins in the brain in people with pre-existing Alzheimer's disease who died of COVID-19. Panel A shows the lack of signal in the control brain for NMDAR2 and the intense signal in the brain of people who died of COVID-19; note the endothelial (oval) and neuron-based signal (brown). Panel C shows the strong co-localization of NMDAR2 (fluorescent green) and the spike (fluorescent red) proteins (fluorescent yellow) in the brain of a case of COVID-19 with pre-existing Alzheimer's disease. Panel E shows the strong microvasculature-based signal for BCL10 (signal is brown) and the loss of the signal in the brains of people who died of COVID-19 with no pre-existing dementia. Panels G-I show the rare expression of Furin in the control brains (panel G) and the marked expression, mostly endothelial cell-based, in the brains from people who died of COVID-19 (panel H) including those with pre-existing Alzheimer's disease (panel I). Panel J shows that the increased nNOS signal (fluorescent green) in the fatal COVID-19 brain strongly co-localizes with spike S1 (fluorescent red). (For interpretation of the references to colour in this figure legend, the reader is referred to the web version of this article.)
The COVID-19 brains with no history of dementia showed a marked reduction (>50 %) in the expression of BCL6, BCL10 (Fig. 4), BACH1, SHIP1, and MFSD2a with no statistically significant difference in the COVID-19 brain tissues from people who had pre-existing Alzheimer's disease. Similarly, there was a marked increase in the expression of nNOS, NMDAR2, and Furin in the brain tissues of people who died of COVID-19 with equivalent results in the group with no history of dementia and in the group with pre-existing Alzheimer's disease. As evident from Fig. 4, the nNOS, NMDAR2, and Furin showed a strong co-localization with the endocytosed spike protein (Fig. 4).
3.6. HUBEC cells treated with spike protein
Given that the in vivo data suggested that SARS-CoV2 spike protein was diffusely endocytosed by ACE-2-expressing endothelial cells in the microvessels of the brain which induced a pro-inflammatory and hypercoagulable state and modulated the expression of proteins that have been correlated with worsening Alzheimer's disease, we re-did these experiments in vitro using a human cortex endothelial cell line purchased from ATCC (HBEC). The data is summarized in Table 1 and Fig. 5 . Note that the low dose of spike S1 (equivalent to the vaccine dose) [20] had no significant effects on these human brain endothelial cells. However, the high dose (equivalent to the reported levels in severe COVID-19) [14] induced the changes reported above for the human COVID-19 brain samples except for increased Furin production which was very high at baseline in these cells. The high dose of spike S1, as evident from Table 1 and Fig. 5, also induced a significant drop in ACE-2 expression. The monocytic THP-1 cells (ATCC) did not express ACE-2 and showed no evidence of spike S1 endocytosis or an inflammatory response (data not shown).
Table 1.
Molecular changes in human brain endothelial cells incubated with low and high dose spike S1 protein.
| Control | S1 low dose | S1 high dose | Significant changea | |
|---|---|---|---|---|
| Spike S1 | 0 | 2.0 (0.2) | 19.9 (5.5) | p < 0.001 |
| Caspase-3 | 7.5 (2.2) | 10.4 (4.9) | 40.3 (4.1) | p < 0.001 |
| ACE-2 | 82.5 (4.4) | 70.1 (5.5) | 11.9 (3.3) | p < 0.001 |
| IL6 | 0 | 7.7 (3.0) | 46.3 (5.5) | p < 0.001 |
| TNFα | 0.1 (0.1) | 0.3 (0.1) | 4.5 (1.0) | p < 0.001 |
| BACH1 | 19.3 (3,3) | 13.1 (4.0) | 5.1 (1.1) | p < 0.001 |
| BCL6 | 65,3 (4.4) | 61.1 (6.9) | 37.3 (5.0) | p < 0.001 |
| BCL10 | 66.3 (6.2) | 50.1 (8.8) | 33.3 (3.3) | p < 0.001 |
| MFSD2A | 71.4 (4.0) | 72.4 (3.4) | 12.0 (2.0) | p < 0.001 |
| CD31 (+ control) | 94.3 | 95.4 | 94.1 | |
| nNOS | 0.5 (0.1) | 1.1 (0.3) | 13.3 (2.0) | p < 0.001 |
| NMDAR2 | 7.2 (1.3) | 8.0 (2.1) | 23.6 (3.9) | p < 0.001 |
| FURIN | 84.5 (2.0) | 85.4 (1.9) | 86.4 (3.0) |
Significant change versus control and low dose spike S1
Fig. 5.
The molecular changes in human brain endothelial cells (HBEC) closely mirror those in the brain in people who died of COVID-19. Panel A shows the strong expression of ACE-2 in the human brain endothelial cells at baseline. Note the strong reduction of the signal induced by high dose spike S1 (panel B, signal is red). Panels C-E show the lack of signal in the brain endothelial cells for spike S1 in the controls (panel C), the weak signal for S1 after incubation with the low dose of S1 (panel D) and the strong signal after incubation with high dose S1 (panel E, signal is red). MFSD2A is highly expressed in the endothelial cells at baseline (panel F) and the protein is nearly lost after 2 days incubation with high dose spike S1 (panel G, signal is brown). NMDAR2 was not expressed by the endothelial cells at baseline (not shown). Panels H-J show the increased expression of NMDAR2 in the cultured brain endothelial cells after high dose S1; note that the NMDAR2 (fluorescent green) co-localizes with the spike protein (fluorescent red). (For interpretation of the references to colour in this figure legend, the reader is referred to the web version of this article.)
4. Discussion
A central question in the central nervous system (CNS) manifestations of COVID-19 is the molecular basis of the disease. Although it is logical to assume there is more than one molecular mechanism, it was initially hypothesized that systemic spread of SARS-CoV2 infection from the lung and nasopharynx to the brain was the primary mechanism. However, many studies have documented that SARS-CoV2 RNA is rarely found in the bloodstream even in fatal COVID-19 and, importantly, the viral RNA is not detected in the brain in such cases [7], [8], [9], [10]. As examples, qRT-PCR assays failed to detect SARS-CoV2 RNA in the CSF of COVID-19 patients with neurological complications [51], and a larger study also indicated that viral RNA was not present in the brain in fatal COVID-19 cases, despite the widespread microvascular damage [52]. A key discovery in understanding the pathogenesis of CNS disease in COVID-19 was the demonstration by many groups, including ours, that spike protein per se when injected into mice or when incubated in vitro with different cell cultures can recapitulate the main pathologic and molecular findings in the brain/brain cells in fatal COVID-19 before any antibody response can be mounted [11], [12], [13], [14], [15], [16], [17], [18], [19]. The data in this paper supports the hypothesis that the endocytosis of circulating spike protein in the ACE-2-expressing endothelial cells of microvessels in the CNS plays a major role in the CNS disease of COVID-19. This is not to say that other factors, such as the cytokine storm and antibody-antigen complexes in the microvessels, do not also play a role. However, it is now clear that endocytosed spike protein in the microvessels of the CNS or in vitro can induce the pro-inflammatory microenvironment and hypercoagulable state well described in the brain for fatal COVID-19 before a systemic cytokine storm or antibody response would be mounted [12], [15], [17].
The source of the circulating spike protein is logically the nasopharynx [53] and, more probably, the lung since the microangiopathy described in the latter [21] leads to marked viral degeneration which could release high concentrations of the spike protein into the circulation. An important finding in this study is that these levels of circulating spike protein induce the same molecular damage in the human brain endothelial cells in vitro. Importantly, however, the spike levels associated with the mRNA vaccines, where the circulating amounts have been reported to be 100–200 pg/ml [20], did not induce any significant molecular changes in the HBEC, underscoring the safety of the vaccine. An interesting part of this study, which corroborated work by others, is that the primary cell type responding to the microangiopathy in the brain is the endogenous microglia cell and, as shown by others, astrocytes [7], [8], [52] and not exogenous inflammatory cells entering the CNS. This immunologically “protected” status of the CNS has been well described and also reported at other sites, such as the placenta [49]. The microglial activation data in this regard was interesting. As shown in a recent report, microglial activation is a prominent feature of the pathophysiology of COVID-19 brain disease [52]. MCP1 is reported to be a specific marker of microglial activation [54], [55], [56]. Although we did note increased MCP1 expression in the brain tissues in all COVID-19 cases relative to the controls, both neurons as well as microglia expressed this protein. MCP1 expression in neurons has been associated with neuronal damage in many diseases including ALS, viral infections, and Alzheimer's disease [54], [55], [56]. Thus, the MCP1 expression in neurons in the COVID-19 brain tissues is consistent with indirect neuronal damage from the associated spike-induced microangiopathy.
The strong correlation between Alzheimer's disease and severe COVID-19 is well documented, including the observation that people with minimal cognitive disorder may develop Alzheimer's disease after contracting COVID-19 [22], [23], [24], [25], [26], [27], [28], [29]. The data in this paper offers possible in vivo molecular mechanisms that may explain this clinical correlation. First, the diffuse microencephalitis involves areas where hyperphosphorylated tau protein is prominent. Thus, the dysfunctional neurons in these areas will be additionally stressed by the strong pro-inflammatory and hypercoagulable state given the importance of the microvasculature unit to neuronal functioning. Second, COVID-19 brain-associated molecular changes included the reduced expression of proteins that has been implicated to worsening Alzheimer's disease. These include BCL6, BCL10, BACH1, SHIP 1 and, importantly, MFSD2A. Reduction of the latter protein, a key player in the blood brain barrier, has been proposed as a biomarker of Alzheimer's disease per se even in the absence of COVID-19 [42]. BCL6, BACH1, SHIP1, and BCL10 were chosen for study as each are targets of miR-155, which we have shown is increased in the cells after the endocytosis of SARS-CoV2 spike protein [9]. Further, though miR-155 is not dysregulated in Alzheimer's disease, there was a relative decrease in BCL6 and BCL10 in the Alzheimer's disease brains versus controls [57] although the decrease was more pronounced in the COVID-19 brain tissues. Proteins such as nNOS and NMDAR2 have been shown to play a direct role in the neuronal dysfunction typical of Alzheimer's disease [30], [31], [32], [33], [34] and, as we showed previously, these are also increased in fatal COVID-19 in the brain [7] though in this study we confirmed that these observations also were observed in the brain of people who died of COVID-19 with pre-existing Alzheimer's disease and that these proteins show a strong co-localization with spike protein.
The increased Furin expression in fatal COVID-19 is interesting and also was seen in the fatal COVID cases with pre-existing Alzheimer's disease. Furin is involved in spike cleavage, and thus, it may facilitate spike endocytosis in the endothelia of the microvessels where both S1 and S2 were detected. With regards to ACE-2, it was not surprising that it was much more highly expressed in the microvessels of the brains of people who had pre-existing obesity versus people without obesity given that other investigators have documented this result [58], [59]. What was surprising however, was that the ACE-2 levels did not correlate with the amount of endocytosed spike protein in the brain of the fatal COVID-19 cases. Further study will be needed to determine if even lower levels of ACE-2 in the brain in the non-obese are sufficient to allow a widespread microangiopathy in fatal COVID-19 and/or if other factors, such as increased Furin expression, may play a role.
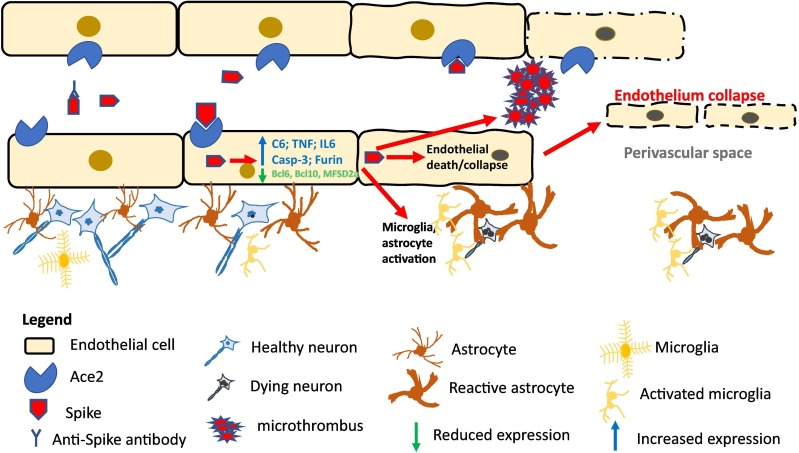
A key study by Lee et al documented that microvascular disease, microthrombi, complement deposition, microglial activation, and endothelial cell damage are essential features of brain damage in COVID-19. Critically, these data were not associated with viral RNA [52]. The results in this study are consistent with their data although they ascribe the changes to antigen-antibody complexes or autoantibodies whereas this study demonstrated that these findings strongly colocalized with endocytosed spike protein. Antigen-antibody complexes typically involve a neutrophil-based response with joint and/or renal involvement [7], [60]. A large study of auto-antibody encephalitis demonstrated a strong CD3 (93 % of cases) and CD20 response (67 % of cases) and it involved the parenchyma, and not the perivascular area, in 29 % of cases which is not consistent with the pathology findings in the brain in fatal COVID-19 [61]. The significance of the hypothesis outlined in Fig. 6 is that it strongly suggests that using anti-spike antibodies to block the endocytosis of the spike protein in the microvasculature endothelia may an effective way to abrogate both severe COVID-19 in the brain and long-haul COVID.
Fig 6.
Schematic of theory.
5. Conclusions
The main conclusion of this work that analyzed molecular differences in the brain of COVID-19 patients including those with dementia is that the endocytosis of SARS-CoV2 spike protein may be a major factor in the CNS disease associated with COVID-19, including long-haul COVID-19. CNS disease in fatal COVID-19 is marked by a dramatic endocytosis of spike protein in the microvascular ACE2+ endothelia and, if one assumes it may persist for months if the disease is not fatal, may also explain long-haul COVID-19. These data suggest that blocking the endocytosis of the spike protein by endothelial cells using antibodies directed against spike may be an effective way of reducing the severity of neurological complications in COVID-19 patients. Once the spike protein has been endocytosed, it is clear that many deleterious pathways are activated which in sum would be difficult to abrogate. The data also offer a likely explanation as to why pre-existing Alzheimer's disease is a risk factor for severe/fatal COVID-19 and why the infection by SARS-CoV-2 is associated with an increased incidence of Alzheimer's disease. Specifically, the data supports a “two hit” hypothesis where spike-induced endothelialitis in patients with Alzheimer's exacerbates the neuronal health located in the vicinity of hyperphosphorylated tau protein and/or βA-42 as well as modulating other proteins that may damage neurons, especially since several play a role in the blood-brain barrier. In vivo mouse models for COVID-19 and Alzheimer's CNS diseases will be one way to document the timing and pharmacodynamics of anti-spike antibody approach which may be beneficial for both the severe CNS disease described in this study as well as long-haul COVID-19.
Declaration of competing interest
The authors have no conflicts of interest to disclose.
Acknowledgements
The authors greatly appreciate the help of Dr. Margaret Nuovo with the photomicroscopy, Dr. Saul Suster who provided many of the samples, Dr. Cynthia Magro who provided samples and advice/comments and Ms. Eva Matys.
Footnotes
The work was supported in part by a grant from the Alzheimer's disease Drug Discovery Foundation #20160204
References
- 1.Feldman E.L., Savelieff M.G., Hayek S.S., et al. COVID-19 and diabetes: a collision and collusion of two diseases. Diabetes. 2020;69:2549–2565. doi: 10.2337/dbi20-0032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Zhou Y., Chi J., Lv W., Wang Y. Obesity and diabetes as high-risk factors for severe coronavirus disease 2019 (Covid-19) Diabetes Metab Res Rev. 2021;37 doi: 10.1002/dmrr.3377. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Abbasi J. The COVID heart—one year after SARS-CoV-2 infection, patients have an Array of increased cardiovascular risks. JAMA. 2022;327:1113–1114. doi: 10.1001/jama.2022.2411. [DOI] [PubMed] [Google Scholar]
- 4.Szelenberger R., Saluk-Bijak J., Bijak M. Ischemic stroke among the symptoms caused by the COVID-19 infection. J Clin Med. 2020;9 doi: 10.3390/jcm9092688. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.AboTaleb H.A. Neurological complications in COVID-19 patients and its implications for associated mortality. Curr Neurovasc Res. 2020;17:522–530. doi: 10.2174/1567202617666200727124704. [DOI] [PubMed] [Google Scholar]
- 6.Pleasure S.J., Green A.J., Josephson S.A. The Spectrum of neurologic disease in the severe acute respiratory syndrome coronavirus 2 pandemic infection: neurologists move to the frontlines. JAMA Neurol. 2020;77:679–680. doi: 10.1001/jamaneurol.2020.1065. [DOI] [PubMed] [Google Scholar]
- 7.Magro C., Mulvey J., Kubiak J., et al. Severe COVID-19: a multifaceted viral vasculopathy syndrome. Ann Diagn Pathol. 2021;50 doi: 10.1016/j.anndiagpath.2020.151645. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Lee M.H., Perl D.P., Nair G., et al. Microvascular injury in the brains of patients with COVID-19. N Engl J Med. 2021;384(5):481–483. doi: 10.1056/NEJMc2033369. Feb 4, NEJMc2033369. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Nuovo G.J., Suster D., Awad H., Michaille J.J., Tili E. The histologic and molecular correlates of liver disease in fatal COVID-19 including with alcohol use disorder. Ann Diagn Pathol. 2022;57 doi: 10.1016/j.anndiagpath.2021.151881. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Mezache L., Nuovo G.J., Suster D., et al. Histologic, viral, and molecular correlates of heart disease in fatal COVID-19. Ann Diagn Pathol. 2022;60 doi: 10.1016/j.anndiagpath.2022.151983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Nuovo G.J., Magro C., Shaffer T., et al. Endothelial cell damage is the central part of COVID-19 in a mouse model induced by injection of the S1 subunit of the spike protein. Ann Diagn Pathol. 2021;51 doi: 10.1016/j.anndiagpath.2020.151682. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Buzhdygan T.P., DeOre B.J., Baldwin-Leclair A., Bullock T.A., McGary H.M., Khan J.A. The SARS-CoV-2 spike protein alters barrier function in 2D static and 3D microfluidic in-vitro models of the human blood-brain barrier. Neurobiol Dis. 2020 Dec;146 doi: 10.1016/j.nbd.2020.105131. Epub 2020 Oct 11. PMID: 33053430; PMCID: PMC7547916. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Ryu J.K., Sozmen E.G., Dixit K. Vol. 2021. bioRxiv [Preprint]; 2021 Oct 13. SARS-CoV-2 spike protein induces abnormal inflammatory blood clots neutralized by fibrin immunotherapy. 10.12.464152. [Google Scholar]
- 14.Avolio E., Carrabba M., Milligan R., et al. The SARS-CoV-2 spike protein disrupts human cardiac pericytes function through CD147 receptor-mediated signalling: a potential non-infective mechanism of COVID-19 microvascular disease. Clin Sci (Lond) 2021;135:2667–2689. doi: 10.1042/CS20210735. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Frank M.G., Nguyen K.H., Ball J.B., et al. SARS-CoV-2 spike S1 subunit induces neuroinflammatory, microglial and behavioral sickness responses: evidence of PAMP-like properties. Brain Behav Immun. 2022;100:267–277. doi: 10.1016/j.bbi.2021.12.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Perico L., Morigi M., Galbusera M., et al. SARS-CoV-2 spike protein 1 activates microvascular endothelial cells and complement system leading to platelet aggregation. Front Immunol. 2022:13. doi: 10.3389/fimmu.2022.827146. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Clough E., Inigo J., Chandra D., Chaves L., Reynolds J.L., Aalinkeel R., et al. Mitochondrial dynamics in SARS-COV2 spike protein treated human microglia: implications for neuro-COVID. J Neuroimmune Pharmacol. 2021 Dec;16(4):770–784. doi: 10.1007/s11481-021-10015-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Bi Z., Hong W., Que H., et al. Inactivated SARS-CoV-2 induces acute respiratory distress syndrome in human ACE2-transgenic mice. Signal Transduct Target Ther. 2021;6:439. doi: 10.1038/s41392-021-00851-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Laubscher G.J., Lourens P.J., Steenkamp J., Kell D.B., Pretorius E. SARS-CoV-2 spike protein S1 induces fibrin(ogen) resistant to fibrinolysis: implications for microclot formation in COVID-19. Biosci Rep. 2021;41(8) doi: 10.1042/BSR20210611. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Ogata A.F., Cheng C.A., Desjardins M., et al. Circulating severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) vaccine antigen detected in the plasma of mRNA-1273 vaccine recipients. Clin Infect Dis. 2022;74:715–718. doi: 10.1093/cid/ciab465. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Magro C., Mulvey J.J., Berlin D., et al. Complement associated microvascular injury and thrombosis in the pathogenesis of severe COVID-19 infection: a report of five cases. Transl Res. 2020;220:1–13. doi: 10.1016/j.trsl.2020.04.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Ciaccio M., Lo Sasso B., Scazzone C., et al. COVID-19 and Alzheimer's disease. Brain Sci. 2021;11(3):305. doi: 10.3390/brainsci11030305. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Matias-Guiu J.A., Pytel V., Matías-Guiu J. Death rate due to COVID-19 in Alzheimer's disease and frontotemporal dementia. J Alzheimers Dis. 2020;78:537–541. doi: 10.3233/JAD-200940. [DOI] [PubMed] [Google Scholar]
- 24.Naughton S.X., Raval U., Pasinetti G.M. Potential novel role of COVID-19 in Alzheimer's disease and preventative mitigation strategies. J Alzheimers Dis. 2020;76:21–25. doi: 10.3233/JAD-200537. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Lennon J.C. Neurologic and immunologic complications of COVID-19: potential long-term risk factors for Alzheimer's disease. J Alzheimers Dis Rep. 2020;4:217–221. doi: 10.3233/ADR-200190. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Rahman M.A., Islam K., Rahman S., Alamin M. Neurobiochemical cross-talk between COVID-19 and Alzheimer's disease. Mol Neurobiol. 2021;58:1017–1023. doi: 10.1007/s12035-020-02177-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Haghighi M.M., Kakhki E.G., Sato C., Ghani M., Rogaeva E. The intersection between COVID-19, the gene family of ACE2 and Alzheimer's disease. Neurosci Insights. 2020;15 doi: 10.1177/2633105520975743. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Xia X., Wang Y., Zheng J. COVID-19 and Alzheimer's disease: how one crisis worsens the other. Transl Neurodegener. 2021;10:15. doi: 10.1186/s40035-021-00237-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Brown E.E., Kumar S., Rajji T.K., Pollock B.G., Mulsant B.H. Anticipating and mitigating the impact of the COVID-19 pandemic on Alzheimer's disease and related dementias. Am J Geriatr Psychiatry. 2020;28:712–721. doi: 10.1016/j.jagp.2020.04.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Wang R., Reddy P.H. Role of glutamate and NMDA receptors in Alzheimer's disease. J Alzheimers Dis. 2017;57(4):1041–1048. doi: 10.3233/JAD-160763. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Uddin M.S., Al Mamun A., Kabir M.T., Ashraf G.M., Bin-Jumah M.N., Abdel-Daim M.M. Multi-target drug candidates for multifactorial Alzheimer's disease: AChE and NMDAR as molecular targets. Mol Neurobiol. 2021 Jan;58(1):281–303. doi: 10.1007/s12035-020-02116-9. [DOI] [PubMed] [Google Scholar]
- 32.Hynd M.R., Scott H.L., Dodd P.R. Glutamate-mediated excitotoxicity and neurodegeneration in Alzheimer's disease. Neurochem Int. 2004 Oct;45(5):583–595. doi: 10.1016/j.neuint.2004.03.007. [DOI] [PubMed] [Google Scholar]
- 33.Malinski T. Nitric oxide and nitroxidative stress in Alzheimer's disease. J Alzheimers Dis. 2007;11(2):207–218. doi: 10.3233/jad-2007-11208. [DOI] [PubMed] [Google Scholar]
- 34.Choi S., Won J.S., Carroll S.L., et al. Pathology of nNOS-expressing GABAergic neurons in mouse model of Alzheimer's disease. Neuroscience. 2018;384:41–53. doi: 10.1016/j.neuroscience.2018.05.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Tsai A.P., Lin P.B., Dong C., et al. INPP5D expression is associated with risk for Alzheimer's disease and induced by plaque-associated microglia. Neurobiol Dis. 2021;153 doi: 10.1016/j.nbd.2021.105303. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Kerr W.G., Pedicone C., Dormann S., Pacherille A., Chisholm J.D. Small molecule targeting of SHIP1 and SHIP2. Biochem Soc Trans. 2020;48:291–300. doi: 10.1042/BST20190775. [DOI] [PubMed] [Google Scholar]
- 37.Di Domenico F., Pupo G., Mancuso C., et al. Bach1 overexpression in down syndrome correlates with the alteration of the HO-1/BVR-a system: insights for transition to Alzheimer's disease. J Alzheimers Dis. 2015;44:1107–1120. doi: 10.3233/JAD-141254. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Perluigi M., Tramutola A., Pagnotta S., Barone E., Butterfield D.A. The BACH1/Nrf2 Axis in brain in down syndrome and transition to Alzheimer disease-like neuropathology and dementia. Antioxidants (Basel) 2020;9:779. doi: 10.3390/antiox9090779. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Baron B.W., Pytel P. Expression pattern of the BCL6 and ITM2B proteins in Normal human brains and in alzheimer disease. Appl Immunohistochem Mol Morphol. 2017;25:489–496. doi: 10.1097/PAI.0000000000000329. [DOI] [PubMed] [Google Scholar]
- 40.Martin D., Galisteo R., Gutkind J.S. CXCL8/IL8 stimulates vascular endothelial growth factor (VEGF) expression and the autocrine activation of VEGFR2 in endothelial cells by activating NFkappaB through the CBM (Carma3/Bcl10/Malt1) complex. J Biol Chem. 2009;284:6038–6042. doi: 10.1074/jbc.C800207200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Franco-Bocanegra D.K., George B., Lau L.C., et al. Microglial motility in Alzheimer’s disease and after Aβ42 immunotherapy: a human post-mortem study. Acta Neuropathol Commun. 2019;7:174. doi: 10.1186/s40478-019-0828-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Sánchez-Campillo M., Ruiz-Pastor M.J., Gázquez A., et al. Decreased blood level of MFSD2a as a potential biomarker of Alzheimer's disease. Int J Mol Sci. 2019;21:70. doi: 10.3390/ijms21010070. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Qu C., Song H., Shen J., et al. Mfsd2a reverses spatial learning and memory impairment caused by chronic cerebral hypoperfusion via protection of the blood-brain barrier. Front Neurosci. 2020;14:461. doi: 10.3389/fnins.2020.00461. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Ng A., Tam W.W., Zhang M.W., et al. IL-1β, IL-6, TNF- α and CRP in elderly patients with depression or Alzheimer's disease: systematic review and meta-analysis. Sci Rep. 2018;8:12050. doi: 10.1038/s41598-018-30487-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Johnson B.A., Xie X., Bailey A.L., et al. Loss of furin cleavage site attenuates SARS-CoV-2 pathogenesis. Nature. 2021;591:293–299. doi: 10.1038/s41586-021-03237-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Marcinkiewicz M. BetaAPP and furin mRNA concentrates in immature senile plaques in the brain of alzheimer patients. J Neuropathol Exp Neurol. 2002;61:815–829. doi: 10.1093/jnen/61.9.915. [DOI] [PubMed] [Google Scholar]
- 47.Herman-Edelstein M., Guetta T., Barnea A., et al. Expression of the SARS-CoV-2 receptor ACE2 in human heart is associated with uncontrolled diabetes, obesity, and activation of the renin angiotensin system. Cardiovasc Diabetol. 2021;20:90. doi: 10.1186/s12933-021-01275-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Nuovo G.J., Suster D., Tili E., Awad H., Magro C. A standardization protocol for the in situ detection of SARS-CoV2 RNA and proteins. Appl Immunohistochem Mol Morphol. 2022;30:83–90. doi: 10.1097/PAI.0000000000000992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Nuovo G.J. 2nd edition. Elsevier; San Diego CA: 2020. In situ molecular pathology and co-expression analyses. [Google Scholar]
- 50.Lan J., Ge J., Yu J., et al. Structure of the SARS-CoV-2 spike receptor-binding domain bound to the ACE2 receptor. Nature. 2020;581:215–220. doi: 10.1038/s41586-020-2180-5. [DOI] [PubMed] [Google Scholar]
- 51.Helms J., Kremer S., Merdji H., et al. Neurologic features in severe SARS-CoV-2 infection. N Engl J Med. 2020;382:2268–2270. doi: 10.1056/NEJMc2008597. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Lee M.H., Perl D.P., Steiner J., Pasternack N., Li W., Maric D., et al. Neurovascular injury with complement activation and inflammation in COVID-19. Brain. 2022 Jul 29;145(7):2555–2568. doi: 10.1093/brain/awac151. PMID: 35788639; PMCID: PMC9278212. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Nuovo G.J., Magro C., Mikhail A. Cytologic and molecular correlates of SARS-CoV-2 infection of the nasopharynx. Ann Diagn Pathol. 2020;48 doi: 10.1016/j.anndiagpath.2020.151565. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Jara J.H., Gautam M., Kocak N., Xie E.F., Mao Q., Bigio E.H., Özdinler P.H. MCP1-CCR2 and neuroinflammation in the ALS motor cortex with TDP-43 pathology. J Neuroinflammation. 2019 Oct 30;16(1):196. doi: 10.1186/s12974-019-1589-y. PMID: 31666087; PMCID: PMC6822373. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Nordengen K., Kirsebom B.E., Henjum K., Selnes P., Gísladóttir B., Wettergreen M., Torsetnes S.B., Grøntvedt G.R., Waterloo K.K., Aarsland D., Nilsson L.N.G., Fladby T. Glial activation and inflammation along the Alzheimer's disease continuum. J Neuroinflammation. 2019 Feb 21;16(1):46. doi: 10.1186/s12974-019-1399-2. PMID: 30791945; PMCID: PMC6383268. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Guo L., Xing Y., Pan R., Jiang M., Gong Z., Lin L., Wang J., Xiong G., Dong J. Curcumin protects microglia and primary rat cortical neurons against HIV-1 gp120-mediated inflammation and apoptosis. PLoS One. 2013 Aug 6;8(8) doi: 10.1371/journal.pone.0070565. PMID: 23936448; PMCID: PMC3735595. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Tili E., Mezache L., Michaille J.J., et al. microRNA 155 up regulation in the CNS is strongly correlated to Down's syndrome dementia. Ann Diagn Pathol. 2018;34:103–109. doi: 10.1016/j.anndiagpath.2018.03.006. [DOI] [PubMed] [Google Scholar]
- 58.Abu-Farha M., Al-Mulla F., Thanaraj T.A., et al. Impact of diabetes in patients diagnosed with COVID-19. Front Immunol. 2020;11 doi: 10.3389/fimmu.2020.576818. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Delorey T.M., Ziegler C.G.K., Heimberg G., et al. COVID-19 tissue atlases reveal SARS-CoV-2 pathology and cellular targets. Nature. 2021;595:107–113. doi: 10.1038/s41586-021-03570-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Maat P., de Beukelaar J.W., Jansen C., Schuur M., van Duijn C.M., van Coevorden M.H., de Graaff E., Titulaer M., Rozemuller A.J., Sillevis Smitt P. Pathologically confirmed autoimmune encephalitis in suspected creutzfeldt-Jakob disease. Neurol Neuroimmunol Neuroinflamm. 2015 Nov 12;2(6) doi: 10.1212/NXI.0000000000000178. PMID: 26601117; PMCID: PMC4645173. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Shmagel K.V., Chereshnev V.A. Molecular bases of immune complex pathology. Biochemistry (Mosc) 2009 May;74(5):469–479. doi: 10.1134/s0006297909050010. PMID: 19538120. [DOI] [PubMed] [Google Scholar]