Abstract
Patient: Female, 36-year-old
Final Diagnosis: Severe ARDS secondary to COVID-19 leading to severe PAH and RV systolic function impairment
Symptoms: Hypoxemia
Medication: —
Clinical Procedure: —
Specialty: Critical Care Medicine
Objective:
Rare disease
Background:
Inhaled nitric oxide (iNO) is used as a treatment for pulmonary arterial hypertension (PAH). Severe hypoxia with hypoxic vasoconstriction caused by severe acute respiratory distress syndrome (ARDS) can induce pulmonary hypertension with hemodynamic implications, mainly secondary to right ventricle (RV) systolic function impairment.
We report the case of the use of iNO in a critically ill patient with bilateral SARS-CoV-2 pneumonia and severe ARDS and hypoxemia leading to acute severe PAH, causing a ventilation/perfusion mismatch, RV pressure overload, and RV systolic dysfunction.
Case Report:
A 36-year-old woman was admitted to the Intensive Care Unit with a severe ARDS associated with SARS-CoV-2 pneumonia requiring invasive mechanical ventilation. Severe hypoxia and hypoxic vasoconstriction developed, leading to an acute increase in pulmonary vascular resistance, severe to moderate tricuspid regurgitation, RV pressure overload, RV systolic function impairment, and RV dilatation. Following 24 h of treatment with iNO at 15 ppm, significant oxygenation and hemodynamic improvement were noted, allowing vasopressors to be stopped. After 24 h of iNO treatment, echocardiography showed very mild tricuspid regurgitation, a non-dilated RV, no impairment of transverse free wall contractility, and no paradoxical septal motion. iNO was maintained for 7 days. The dose of iNO was progressively decreased with no adverse effects and maintaining an improvement of oxygenation and hemodynamic status, allowing respiratory weaning.
Conclusions:
Sustained acute hypoxia in ARDS secondary to SARS-CoV-2 pneumonia can lead to PAH, causing a ventilation/perfusion mismatch and RV systolic impairment. iNO can be considered in patients with significant PAH causing hypoxemia and RV dysfunction.
Keywords: Cardiac Output, Low; COVID-19; Familial Primary Pulmonary Hypertension; Hypoxia; Nitric Oxide; Tricuspid Valve Insufficiency
Background
COVID-19 pneumonia can be associated with severe acute respiratory distress syndrome (ARDS) [1]. No specific antiviral therapy is currently approved for SARS-CoV-2. Therefore, treatment is supportive. Severe hypoxemia and ARDS can lead to hypoxic vasoconstriction and an acute increase in pulmonary vascular resistance (PVR), leading to right ventricle (RV) pressure overload and RV systolic function impairment.
In human physiology, nitric oxide (NO) has a very significant role as a signal molecule. It is delivered into the well-ventilated alveoli, leading to local vasodilatation. Therefore, it vaso-dilates pulmonary arterioles in well-ventilated alveoli, increasing blood flow in well-ventilated alveoli and decreasing flow in poorly ventilated alveoli, enhancing V/Q matching and increasing oxygenation [1].
As NO goes into the blood circulation, it reacts quickly with hemoglobin, leading to its inactivation and therefore limiting its systemic effects. Bronchodilation and neutrophil-mediated oxidative burst inhibition are 2 known effects of inhaled nitric oxide (iNO) [1]. ARDS, newborn persistent pulmonary hypertension, RV failure after cardiac surgery, and pulmonary fibrosis treated with long-term oxygen are the main indications for the use of iNO and acute pulmonary embolism [1–4].
Data from a matched cohort study on patients with SARS showed an association between the use of iNO and an improvement in oxygenation [5]. Furthermore, exogenous NO has been associated with the inhibition of SARS-CoV-2 viral replication [6]. With respect to ARDS, the use of iNO in adults has not been associated with a decreased mortality rate or a decrease in mechanical ventilation duration. However, the use of iNO leads to an important oxygenation improvement and a decrease in PVR [1].
We report a case of the use of iNO in a critically ill patient with bilateral COVID-19 pneumonia and severe ARDS and hypoxemia leading to acute severe pulmonary arterial hypertension (PAH), RV pressure overload, and RV systolic function impairment.
Case Report
A 36-year-old woman affected with severe ARDS, which was associated with COVID-19 pneumonia and required invasive mechanical ventilation, was admitted to the Intensive Care Unit (ICU) in August 2021. A COVID-19 diagnosis was confirmed by reverse transcription PCR testing (Xpert® Xpress SARS-CoV-2, Cepheid, Switzerland). A written informed consent was signed by the family for publication of this case report. Laboratory analysis at ICU admission can be found in Table 1.
Table 1.
Laboratory analysis at Intensive Care Unit admission.
| ICU admission | |
|---|---|
| PaO2/FiO2 ratio | 182 mmHg |
| Ventilatory mode | VC |
| Leucocyte | 6.5×109/L |
| CRP | 5.8 mg/L |
| Hb | 9.5 g/dL |
| Platelets | 606×109/L |
| FBG | 947 mg/dL |
| PCT | 0.02 ng/mL |
CRP – C-reactive protein; D11 – day 11; FBG – fibrinogen; Hb – hemoglobin; PCT – procalcitonin; VC – volume controlled.
The patient had received a single dose of tocilizumab 8 mg/kg intravenously before being admitted to the ICU. On ICU admission, dexamethasone 6 mg per day was given for the first period of 10 days. Moderate hypoxemia with a PaO2/FiO2 ratio of 182 mmHg was present on admission. A chest X-ray at ICU admission showed a bilateral pulmonary infiltration (Figure 1). Severe hypoxemia was treated with prone positioning for 16 to 20 h per day for 10 days. Initially, the hypoxemia improved by the prone position strategy. However, after 10 days, the hypoxemia started being refractory to prone positioning. Hence, a further prone position was not performed and the PaO2/FiO2 ratio was maintained between 100 and 110 for 4 days. On day 12, a tracheostomy was performed. On day 14, the second period of treatment with dexamethasone 6 mg per day was given for another 10 days. A chest computed tomography (CT) scan was performed, showing bilateral, peripheral, and basal predominant ground-glass opacities (Figures 2, 3).
Figure 1.
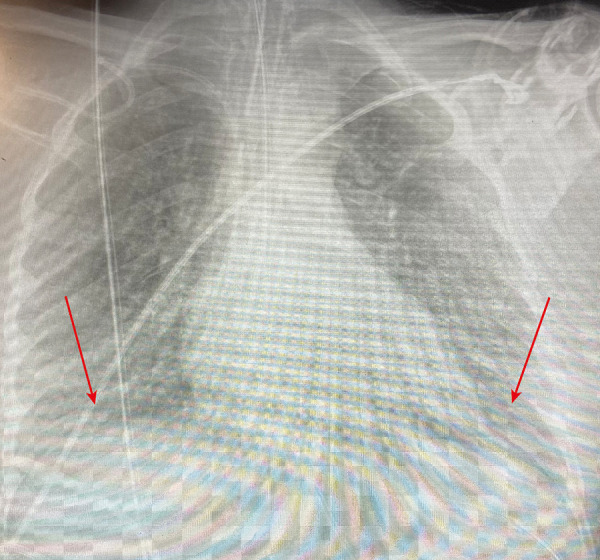
Arrow showing a bilateral pulmonary infiltration in the chest X-ray at Intensive Care Unit admission.
Figure 2.
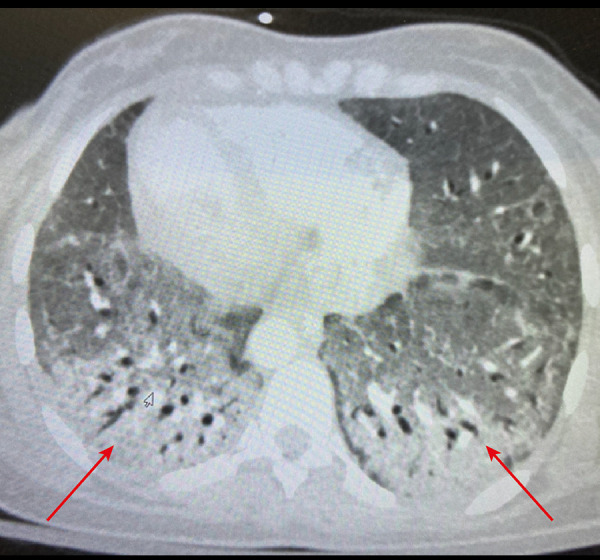
Arrow showing bilateral, peripheral, and basal predominant ground-glass opacities in the basal chest computed tomography scan.
Figure 3.
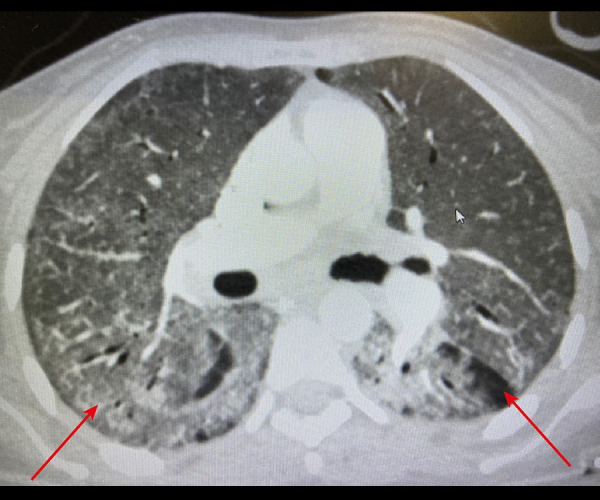
Arrow showing bilateral, peripheral, and basal predominant ground-glass opacities in the apical chest computed tomography.
On day 14 of the ICU stay, the patient presented hemodynamic instability that required an increased dose of vasopressors. A transthoracic echocardiography was performed. A moderate to severe tricuspid regurgitation was observed, and a systolic pulmonary artery pressure of 74 mmHg was estimated. An RV to LV end-diastolic area ratio >1 was observed, which suggested a severe RV dilatation (Figure 4). The RV systolic function was impaired, particularly the free-wall RV transversal contractility. A D-shape of the interventricular septum at endsystole was observed, which suggested an RV pressure overload (Figure 5). The chest X-ray on the day of iNO treatment initiation showed a bilateral infiltration (Figure 6).
Figure 4.
Arrow showing right ventricle moderate dilatation in the transthoracic echocardiography apical 4 chambers view.
Figure 5.
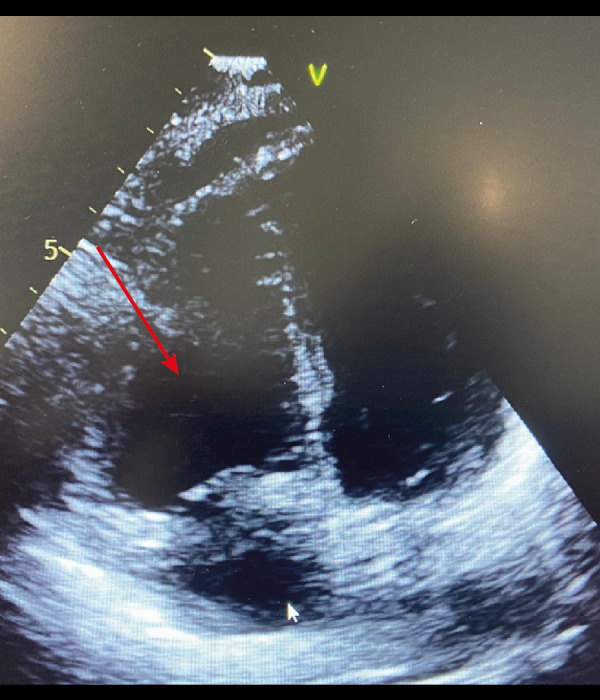
Arrow showing a D-shape of the interventricular septum at end-systole in the transthoracic echocardiography short parasternal axis view.
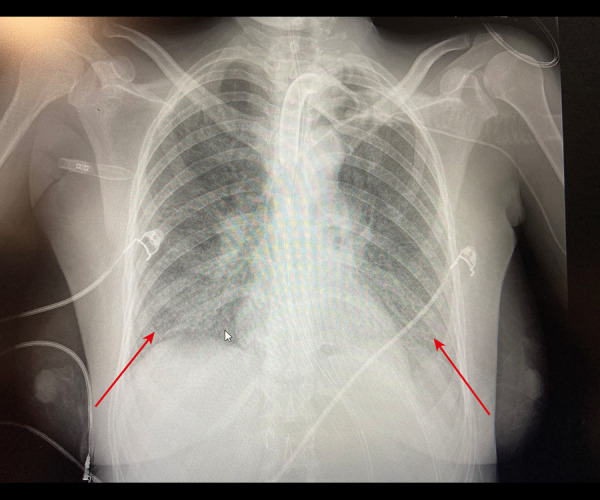
Figure 6.
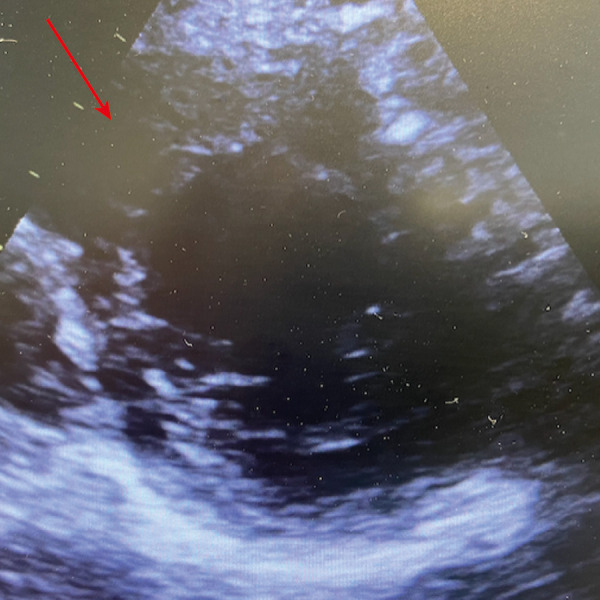
Arrow showing bilateral pulmonary infiltration, on the day of inhaled nitric oxide treatment in the chest X-Ray.
Neither RV hypertrophy nor right atrium dilatation was noted, suggestive of an acute episode. The LV systolic function was not impaired, and the LV filling pressure was not increased, suggesting that the PAH was not due to left heart disease. The PaO2/FiO2 ratio was 100 mmHg. The presence of a pulmonary embolism was ruled out by CT pulmonary angiogram.
Given the presence of severe PAH and severe hypoxemia leading to RV systolic dysfunction, we decided to initiate iNO at 15 ppm. A significant improvement in oxygenation and hemodynamics were observed, allowing us to stop the vasopressors. After 24 h of treatment, the PaO2/FiO2 ratio increased from 100 mmHg to 191 mmHg (Table 2). The transthoracic echocardiography was repeated, and we observed mild tricuspid regurgitation, normal RV transversal/longitudinal contractility, a non-dilated RV (Figure 7), and normal C-shape of the interventricular septum (Figure 8). The iNO was decreased to 10 ppm on day 5, and was gradually decreased until it was stopped on day 7. The PaO2/FiO2 ratio continuously increased from 100 mmHg to 321 mmHg after 7 days of iNO treatment (Table 2, Figure 9). After the iNO was stopped, no incidences were observed and an improvement in oxygenation and hemodynamic status were preserved (Table 3), allowing for respiratory weaning. Finally, on day 35, the patient was discharged from the ICU.
Table 2.
Laboratory analysis during inhaled nitric oxide treatment period.
| D0 before iNO | D1 after iNO | D2 after iNO | D3 after iNO | D4 after iNO | |
|---|---|---|---|---|---|
| PaO2/FiO2 (mmHg) | 100 | 216 | 234 | 140 | 170 |
| Ventilatory mode | VC | VC | PS | CPAP | PS |
| Leukocyte (×109/L) | 14.3 | 10.1 | 13 | 16 | 17 |
| CRP (mg/L) | 10.38 | 4.21 | 2.47 | 2.45 | 2.4 |
| Hb (g/dL) | 9.4 | 8.8 | 9.5 | 9.3 | 8.8 |
| Platelets (×109/L) | 426 | 495 | 591 | 602 | 561 |
| FBG (mg/dL) | 1061 | 892 | 770 | 725 | 710 |
| PCT (ng/mL) | <0.02 | <0.02 | <0.02 | <0.02 | <0.02 |
| D5 after iNO | D6 after iNO | D7 after iNO | D8 STOP iNO | D9 | D10 | |
|---|---|---|---|---|---|---|
| PaO2/FiO2 (mmHg) | 124 | 192 | 321 | 315 | 200 | 190 |
| Ventilatory mode | PS | PS | PS | PS | VC | VC |
| Leukocyte (×109/L) | 17.8 | 18 | 21 | 22 | 18 | 21 |
| CRP (mg/L) | 2.4 | 2.4 | 2.55 | 16.7 | 7.7 | 3.7 |
| Hb (g/dL) | 8.6 | 8.8 | 9 | 7.7 | 8 | 8.7 |
| Platelets (×109/L) | 513 | 518 | 618 | 442 | 468 | 611 |
| FBG (mg/dL) | 700 | 690 | 680 | 820 | 744 | 756 |
| PCT (ng/mL) | <0.02 | <0.02 | <0.02 | 0.03 | 0.04 | 0.05 |
CPAP – continuous positive airway pressure; CRP – C-reactive protein; D0 – day 0; D1 – day 01; D2 – day 2; D3 – day 3; D4 – day 4; D5 – day 5; D6 – day 6; D7 – day 7; D8 – day 8; D9 – day 9; D10 – day 10; FBG – fibrinogen; Hb – hemoglobin; iNO – inhaled nitric oxide; PCT – procalcitonin; PS – pressure support; VC – volume controlled.
Figure 7.
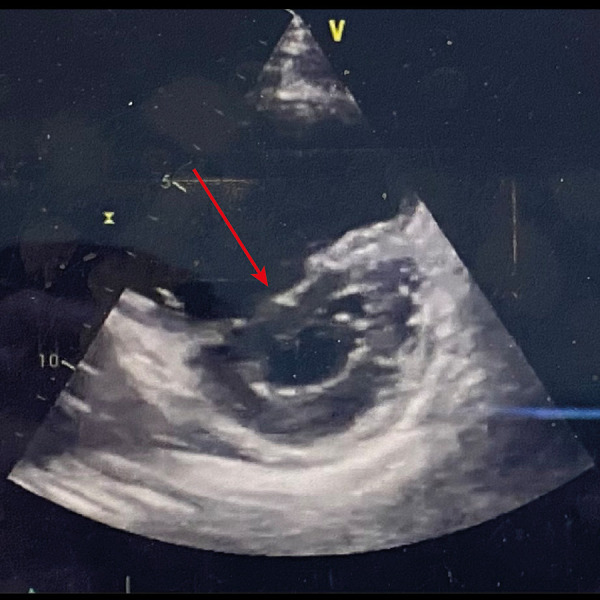
Arrow showing a normal C-shape of the interventricular septum at end-systole after inhaled nitric oxide treatment in the transthoracic echocardiography short parasternal axis view.
Figure 8.
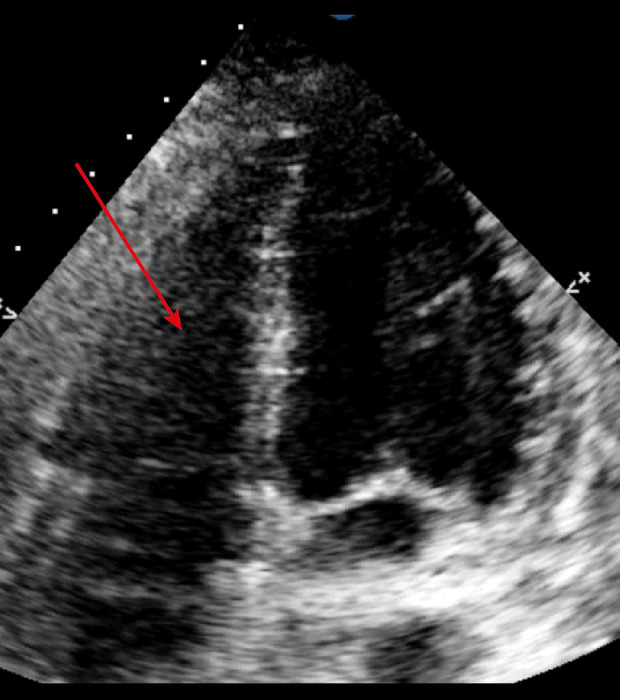
Arrow showing a non-dilated right ventricle after inhaled nitric oxide treatment. In the transthoracic echocardiography apical 4 chambers view.
Figure 9.
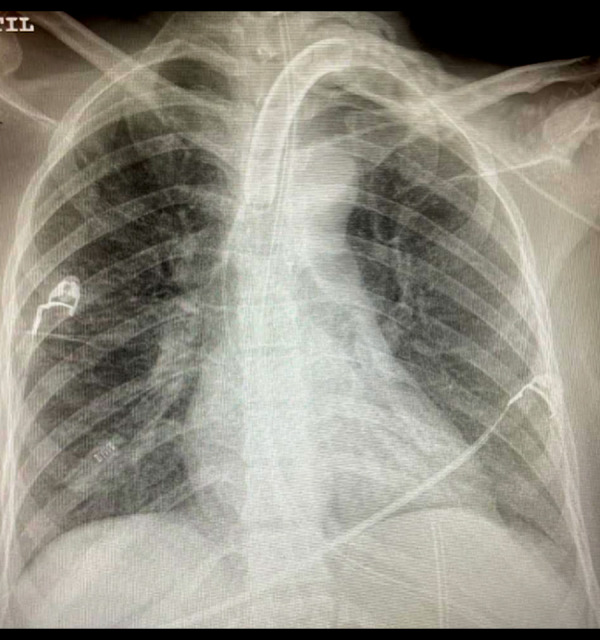
Chest X-ray showing an improvement of bilateral pulmonary infiltration after inhaled nitric oxide treatment.
Table 3.
Laboratory analysis after inhaled nitric oxide treatment period.
| D11 | D12 | D13 | D14 | D15 | |
|---|---|---|---|---|---|
| PaO2/FiO2 (mmHg) | 200 | 205 | 212 | 215 | 210 |
| Ventilatory mode | PS | PS | PS | PS | PS |
| Leukocytes (×109/L) | 16 | 16.8 | 26 | 19 | 17 |
| CRP (mg/L) | 6.2 | 10.7 | 13.7 | 16.9 | 7.4 |
| Hb (g/dL) | 8.9 | 9.1 | 7.9 | 7.8 | 7.8 |
| Platelets (×109/L) | 598 | 583 | 430 | 399 | 403 |
| FBG (mg/dL) | 630 | 878 | 880 | 992 | 862 |
| PCT (ng/mL) | 0.07 | <0.02 | <0.02 | <0.02 | <0.02 |
| D16 | D17 | D18 | D19 | D20 | D21 | |
|---|---|---|---|---|---|---|
| PaO2/FiO2 (mmHg) | 208 | 220 | 225 | 300 | 330 | 270 |
| Ventilatory mode | PS | PS | PS | HFNC by tracheo | HFNC by tracheo | Mask |
| Leukocytes (×109/L) | 17 | 13.8 | 12 | 13 | 12 | 10 |
| CRP (mg/L) | 5.2 | 3.3 | 1.88 | 2.98 | 2.12 | 1 |
| Hb (g/dL) | 7.3 | 8 | 8.4 | 8.8 | 8.3 | 8.7 |
| Platelets (×109/L) | 383 | 353 | 376 | 447 | 351 | 301 |
| FBG (mg/dL) | 780 | 710 | 687 | 667 | 603 | 546 |
| PCT (ng/mL) | <0.02 | <0.02 | <0.02 | <0.02 | <0.02 | <0.02 |
CRP – C-reactive protein; D11 – day 11; D12 – day 12; D13 – day 13; D14 – day 14; D15 – day 15; D16 – day 16; D17 – day 17; D18 – day 18; D19 – day 19; D20 – day 20; D21 – day 21; FBG – fibrinogen; Hb – hemoglobin; HFNC – high flow nasal cannula; PCT – procalcitonin; PS – pressure support; trachea – tracheostomy.
Discussion
This is a single case report on the usefulness of iNO for a critically ill patient with severe ARDS secondary to SARS-CoV-2 infection that led to severe PAH and RV systolic function impairment, along with RV pressure overload and severe RV dilatation. Data on the benefits of using iNO for patients with COVID-19 are controversial [7–18].
In our case report, a significant oxygenation improvement was observed. The PaO2/FiO2 ratio increased from 100 mmHg to 321 mmHg after 7 days of treatment. Our case is interesting because this was a patient with not only COVID-19-associated ARDS but also severe PAH, RV dilatation, and RV systolic function impairment who responded extremely well to iNO.
A systematic review on the efficacy and safety of iNO in the treatment of patients with severe COVID-19 was recently published [7]. The review included 423 patients with COVID-19 in 14 retrospective cohort studies and observed that the use of iNO was associated with a slight increase in the PaO2/FiO2 ratio, with no effect on mortality. Data from a retrospective cohort study on 47 patients on invasive mechanical ventilation showed that iNO increased oxygenation more significantly in non-COVID-19-related ARDS than in COVID-19-related ARDS. However, none of the patients in this cohort had moderate to severe PAH with RV systolic dysfunction [8]. In a prospective cohort study on 9 patients with COVID-19-related ARDS on invasive mechanical ventilation, a statistically significant oxygenation improvement associated with the use iNO was observed, with a PaO2/FiO2 ratio increase from 65 (range 67–73) to 72 (range 67–73), P=0.015. However, a PaO2/FiO2 ratio increase of 7 mmHg might be statistically significant but it is not clinically significant. None of these patients had severe PAH with RV systolic dysfunction [9].
Another retrospective cohort study on 7 patients with COVID-19-related moderate to severe ARDS on invasive mechanical ventilation showed a significant oxygenation improvement; the PaO2/FiO2 ratio was increased from 78.2 (range 64.5–101.5) to 105.0 (range 78.5–144.5), P=0.0313; and a decrease in PVR from 184.1 (range 153.5–237.4) to 155.3 (range 153.1–227.5), P=0.125, was associated with the use of iNO [10]. In a case series of 6 patients with COVID-19 who were pregnant, an improvement in oxygenation with the use of iNO through the mask was observed [11]. An observational prospective cohort study compared the use of iNO with the use of iNO plus almitrine in 10 patients with COVID-19 and severe ARDS. Oxygenation improvement was significantly increased in the group of patients receiving iNO plus almitrine compared with iNO alone. The PaO2/FiO2 ratio increased from 102 mmHg at baseline to 124 mmHg after iNO (P=0.13) and to 180 mmHg after iNO and almitrine (P<0.01) [12].
In another retrospective cohort study, 20 patients with COVID-19 pneumonia were consecutively included. All patients had moderate to severe ARDS with a PaO2/FiO2 ratio <120 mmHg on invasive mechanical ventilation. Neither iNO nor almitrine nor iNO combined with almitrine increased the PaO2/FiO2 ratio by more than 20% (defined as responders). iNO alone increased the PaO2/FiO2 ratio by 2.2%, and almitrine alone increased the PaO2/FiO2 ratio by 1.9%; whereas, the combination of iNO plus almitrine increased the PaO2/ FiO2 ratio by 5%. However, none of the patients included had moderate to severe PAH associated with RV dilatation or RV systolic dysfunction [13]. A case series of 10 patients on invasive mechanical ventilation with severe ARDS (PaO2/FiO2 ratio <100) who required at least 12 to 16 h of ventilation in a prone position observed no response to a 30-min test of iNO at 20 ppm. In this case series, no mention is given to whether the patients had PAH and/or RV pressure overload or systolic dysfunction [14].
A single-center retrospective observational study on 32 consecutive patients observed that 66% of the patients were responders to almitrine combined with iNO. The patients who were responders (21 responders vs 11 non-responders) had a 67% improvement of PaO2/FiO2 ratio, while there was a trend toward a decreased mortality rate for responders compared with non-responders (47% vs 63.6%; P=0.39) [15]. A prospective observational study on 34 patients with moderate to severe COVID-19-related ARDS (PaO2/FiO2 ratio <150) showed that 65% (22/34) were responders and 35% (12/34) were nonresponders to a 30-min test of 10 ppm of iNO. Responders were defined as a 20% increase in PaO2/FiO2 ratio. The rate of acute cor pulmonale did not differ between groups. The rate of PAH was not reported [16]. Data from an observational cohort study on 16 patients with severe ARDS (PaO2/FiO2 <100 mmHg) on invasive mechanical ventilation showed that only a subgroup of patients (4/16, 25%) responded to 25 ppm of iNO. Three of these 4 patients had RV dysfunction requiring inotropics [17]. A case series of 5 patients with PAH and COVID-19-related moderate ARDS observed a continuous significant improvement of PaO2/FiO2 ratio from 146 to 244 mmHg. iNO was associated with a decreased in PVR and a decrease of RV dys-function rate [18]. Our patient had similar characteristics as the patients (25%) who responded to iNO in the observational cohort published by Tavazzi et al [17] and the case series by Feng et al [18]. Our patient had COVID-19-related moderate to severe ARDS and was on invasive mechanical ventilation associated with severe to moderate PAH and RV dysfunction that required vasopressors.
Conclusions
Sustained and severe acute hypoxia in patients with ARDS due to COVID-19 pneumonia might lead to PAH and a ventilation/perfusion mismatch, along with RV dysfunction. NO might have a role in patients with acute hypoxia that is associated with severe ARDS due to COVID-19 pneumonia and severe PAH with RV impairment while the acute episode leading to deterioration is treated.
This case report does not prove the benefit of using iNO for treating hypoxemia in the general population with SARS-CoV-2. Well-powered clinical trials are needed to determine the efficacy of iNO in the setting of SARS-CoV-2 and moderate to severe ARDS, PAH, and RV systolic dysfunction.
Footnotes
Declaration of Figures’ Authenticity
All figures submitted have been created by the authors who confirm that the images are original with no duplication and have not been previously published in whole or in part.
References:
- 1.Griffiths MJ, Evans TW. Inhaled nitric oxide therapy in adults. N Engl J Med. 2005;353:2683–95. doi: 10.1056/NEJMra051884. [DOI] [PubMed] [Google Scholar]
- 2.Gladwin MT, Kato GJ, Weiner D, et al. DeNOVO Investigators. Nitric oxide for inhalation in the acute treatment of sickle cell pain crisis: A randomized controlled trial. JAMA. 2011;305:893–902. doi: 10.1001/jama.2011.235. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Kline JA, Puskarich MA, Jones AE, et al. Inhaled nitric oxide to treat intermediate risk pulmonary embolism: A multicenter randomized controlled trial. Nitric Oxide. 2019;84:60–68. doi: 10.1016/j.niox.2019.01.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Nathan SD, Flaherty KR, Glassberg MK, et al. A randomized, double-blind, placebo-controlled study to assess the safety and efficacy of pulsed, inhaled nitric oxide at a dose of 30 mg/Kg ideal body weight/hr in subjects at risk of pulmonary hypertension associated with pulmonary fibrosis receiving oxygen therapy. Chest. 2020;158(2):637–45. doi: 10.1016/j.chest.2020.02.016. [DOI] [PubMed] [Google Scholar]
- 5.Chen L, Liu P, Gao H, et al. Inhalation of nitric oxide in the treatment of severe acute respiratory syndrome: A rescue trial in Beijing. Clin Infect Dis. 2004;39:1531–35. doi: 10.1086/425357. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Akerstrom S, Mousavi-Jazi M, Klingstrom J, et al. Nitric oxide inhibits the replication cycle of severe acute respiratory syndrome coronavirus. J Virol. 2005;79:1966–69. doi: 10.1128/JVI.79.3.1966-1969.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Prakash A, Kaur S, Kaur C, et al. Efficacy and safety of inhaled nitric oxide in the treatment of severe/critical COVID-19 patients: A systematic review. Indian J Pharmacol. 2021;53(3):236–43. doi: 10.4103/ijp.ijp_382_21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Longobardo A, Montanari C, Shulman R, et al. Inhaled nitric oxide minimally improves oxygenation in COVID-19 related acute respiratory distress syndrome. Br J Anaesth. 2021;126:e44–46. doi: 10.1016/j.bja.2020.10.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Robba C, Ball L, Battaglini D, et al. Early effects of ventilatory rescue therapies on systemic and cerebral oxygenation in mechanically ventilated COVID-19 patients with acute respiratory distress syndrome: A prospective observational study. Crit Care. 2021;25:111. doi: 10.1186/s13054-021-03537-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Lotz C, Muellenbach RM, Meybohm P, et al. Effects of inhaled nitric oxide in COVID-19-induced ARDS – is it worthwhile? Acta Anaesthesiol Scand. 2021;65:629–32. doi: 10.1111/aas.13757. [DOI] [PubMed] [Google Scholar]
- 11.Safaee Fakhr B, Wiegand SB, Pinciroli R, et al. High concentrations of nitric oxide inhalation therapy in pregnant patients with severe coronavirus disease 2019 (COVID-19) Obstet Gynecol. 2020;136:1109–13. doi: 10.1097/AOG.0000000000004128. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Bagate F, Tuffet S, Masi P, et al. Rescue therapy with inhaled nitric oxide and almitrine in COVID-19 patients with severe acute respiratory distress syndrome. Ann Intensive Care. 2020;10:151. doi: 10.1186/s13613-020-00769-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Cardinale M, Esnault P, Cotte J, et al. Effect of almitrine bismesylate and inhaled nitric oxide on oxygenation in COVID-19 acute respiratory distress syndrome. Anaesth Crit Care Pain Med. 2020;39:471–72. doi: 10.1016/j.accpm.2020.05.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Ferrari M, Santini A, Protti A, et al. Inhaled nitric oxide in mechanically ventilated patients with COVID-19. J Crit Care. 2020;60:159–60. doi: 10.1016/j.jcrc.2020.08.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Caplan M, Goutay J, Bignon A, et al. Almitrine infusion in severe acute respiratory syndrome coronavirus 2-induced acute respiratory distress syndrome: A single-center observational study. Crit Care Med. 2021;49:e191–98. doi: 10.1097/CCM.0000000000004711. [DOI] [PubMed] [Google Scholar]
- 16.Abou-Arab O, Huette P, Debouvries F, et al. Inhaled nitric oxide for critically ill COVID-19 patients: A prospective study. Crit Care. 2020;24:645. doi: 10.1186/s13054-020-03371-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Tavazzi G, Pozzi M, Mongodi S, et al. Inhaled nitric oxide in patients admitted to intensive care unit with COVID-19 pneumonia. Crit Care. 2020;24:508. doi: 10.1186/s13054-020-03222-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Feng WX, Yang Y, Wen J, et al. Implication of inhaled nitric oxide for the treatment of critically ill COVID-19 patients with pulmonary hypertension. ESC Heart Fail. 2021;8:714–18. doi: 10.1002/ehf2.13023. [DOI] [PMC free article] [PubMed] [Google Scholar]


