Abstract
New mutations conferring resistance to SARS-CoV-2 therapeutics have important clinical implications. We describe the first cases of an independently acquired V792I RNA-dependent RNA polymerase mutation developing in renal transplant recipients after remdesivir exposure. Our work underscores the need for augmented efforts to identify concerning mutations and address their clinical implications.
Keywords: antiviral resistance, severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), immunocompromise, remdesivir, solid-organ transplant
Although immunization against severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) results in a markedly decreased risk of severe infection in immunocompetent hosts, a diminished vaccination response translates to a greater risk of severe disease and death in immunocompromised individuals—for example, solid-organ-transplant (SOT) recipients [1, 2]. Given the substantial risk associated with SARS-CoV-2 infection in SOT recipients, proactive therapy is warranted in patients requiring hospitalization. Remdesivir, a prodrug of the nucleoside analog GS-441524 that inhibits viral RNA-dependent–RNA polymerase (RdRp) activity, represents the first Food and Drug Administration (FDA)–approved direct-acting antiviral for coronavirus disease 2019 (COVID-19) treatment [3]. Recently, a case of remdesivir resistance was described in a patient who experienced a protracted course of COVID-19 while receiving rituximab and bendamustine for lymphoma [4]. The risk of developing resistance to COVID-19 therapeutics during treatment and the precise prevalence of clinically significant mutations in the community are unknown.
CASE DESCRIPTIONS
Case 1 is a patient in their 60’s with a history of diabetes who underwent deceased donor kidney transplant (DDKT) addressing end-stage renal disease (ESRD). Prior to transplant, the patient received 2 doses of the Pfizer BNT162b2 vaccine. Induction immunosuppression included basiliximab and methylprednisolone. Blood-type mismatch prompted the use of anti-thymocyte globulin (ATG). Delayed graft function required augmented corticosteroids. Graft biopsy on postoperative day 7 identified acute cellular and antibody-mediated rejection. The patient was treated with ATG and plasmapheresis and was ultimately transitioned to maintenance immunosuppression including prednisone, mycophenolate, and belatacept.
Six months after transplantation, the patient developed malaise, cough, and fever. On admission, reverse transcriptase–polymerase chain reaction (RT-PCR) was positive for SARS-CoV-2 (cycle threshold [Ct] value of 27). Genomic sequencing identified the B.1.529 (Omicron) subvariant BA.1.1. Not requiring oxygen at that time, the patient received a 5-day course of remdesivir, experienced improvement in symptomatology, and was discharged.
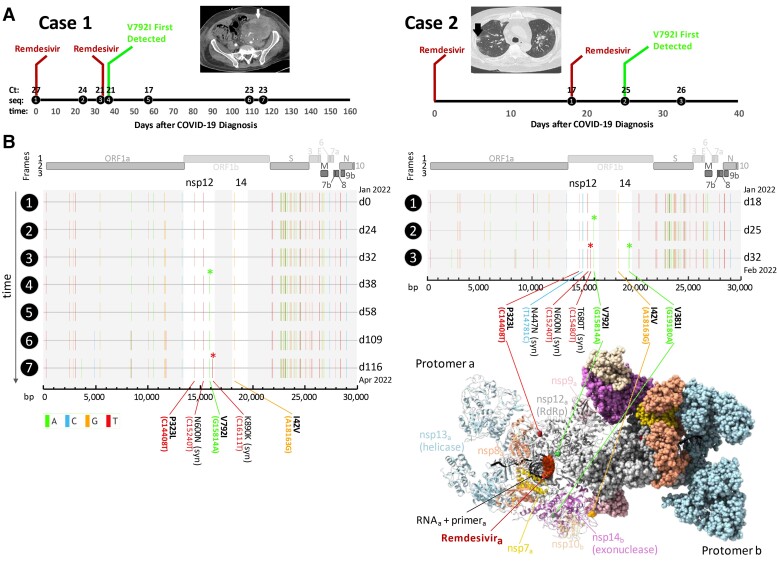
Twenty-four days after initial COVID-19 diagnosis, the patient was readmitted with fatigue, cough, dyspnea, abdominal discomfort, and fever. SARS-CoV-2 RT-PCR was positive (Ct 24), and sequencing identified Omicron BA.1.1. In the setting of substantial oxygen requirement, the patient was treated with another 5-day course of remdesivir and a 10-day course of dexamethasone. Genomic sequencing 38 days after initial COVID-19 diagnosis identified the de novo RdRp mutation V792I (G15814A) (Figure 1A and B).
Figure 1.
A, Timeline of SARS-CoV-2 infection and relation between remdesivir exposure and the subsequent development of the de novo RdRp V792I mutation in Case 1 and Case 2. Ct values are provided at points when the patient was symptomatic and a high-quality SARS-CoV-2 genomic sequence was obtained (circles). For Case 1, a Ct of 34.1 was obtained 153 days after the diagnosis of COVID-19 when the patient experienced durable resolution of all symptoms associated with SARS-CoV-2 infection; and for case 2, a Ct value of 26 was obtained 32 days after the diagnosis of COVID-19 when the patient experienced marked improvement in symptoms and their oxygen requirement had resolved. Case 1 CT of the abdomen demonstrating mass-like thickening along the renal graft (white arrow) contiguous with the abdominal wall is shown above the timeline (left); and Case 2 CT of the chest (right) demonstrating multifocal nodules, many of which are surrounded by ground-glass opacities. The black arrow indicates a cavitary lesion. An elevated galactomannan level from bronchoalveolar lavage fluid suggested the diagnosis of pulmonary aspergillosis. B, Full genome mutation profiles of SARS-CoV-2 viruses in longitudinal specimens of 2 immunocompromised patients treated with remdesivir (Case 1, left; Case 2, right). Base-pair (bp) mutations compared with the Wuhan-Hu-1 reference are shown as ticks, color-coded according to the legend on the lower left. The bp and corresponding amino acid (aa) mutations in nsp12 (RNA-dependent RNA polymerase [RdRp]) and nsp14 (containing 3′-to-5′ exoribonuclease proofreading activity) are labeled and shown in bold if nonsynonymous. The longitudinal acquisition of mutations in nsp12 and 14 is highlighted by colored asterisks. Full genome maps are shown on top. The timeline is shown on the y-axis where time points are indicated on the left as black circles and days (d) elapsed since the first COVID-19 sampling per patient on the right of each plot. A 3D protein structure of the multidomain polymerase complex is shown in its active dimeric form (bottom right). Each domain is colored differently and labeled in the protomer that is shown in ribbon representation, whereas the other domains are shown in sphere representation, respectively. Nsp14 (exonuclease activity) and its cofactor nsp10 convey RNA proofreading in trans and are thus highlighted/shown as ribbons in protomer b together with the other domains in protomer a. The nonsynonymous mutations in nsp12 and 14 as well as remdesivir are highlighted and labeled. Abbreviations: COVID-19, coronavirus disease 2019; Ct, cycle threshold; CT, computed tomography; SARS-CoV-2, severe acute respiratory syndrome coronavirus 2; seq, sequence.
Computed tomography (CT) performed shortly after admission demonstrated a pleural effusion and mass-like soft tissue infiltration along the renal graft (Figure 1A). Serum Epstein Barr virus (EBV) viral load was elevated (645 000 IU/mL). Flow cytometry from pleural fluid and retroperitoneal lymph node biopsy demonstrated EBV-positive diffuse large B-cell lymphoma consistent with monomorphic post-transplant lymphoproliferative disorder. The patient was treated with multiple cycles of antineoplastic therapy including rituximab, cyclophosphamide, doxorubicin, vincristine, and prednisone. During the course of chemotherapy, the patient experienced severe cytopenias, gastrointestinal bleeding, and hemorrhagic shock. The patient progressed to ESRD, and belatacept and mycophenolate were discontinued. Over the course of a 3-month admission, the patient's cough, fever, and hypoxemia resolved; EBV viral load decreased dramatically; and interval reimaging demonstrated decreased size of the renal graft and associated lymphadenopathy. Three months after the initial diagnosis of COVID-19, SARS-CoV-2 nucleocapsid immunoglobulin G (IgG) was detectable, the Ct increased (29.3), and the patient remained free of symptoms of active respiratory infection.
One hundred ten days after COVID-19 diagnosis, the patient developed new dry cough and rhinorrhea. RT-PCR was positive for SARS-CoV-2 (Ct of 23.3, Omicron BA.1.1). RT-PCR for other respiratory pathogens was negative. Repeat RT-PCR 1 week later yielded a Ct of 22.6, and genomic sequencing at that time identified a de novo synonymous mutation in RdRp at K890 (Figure 1B). The patient’s mild symptoms gradually improved over the course of weeks, and no further treatment was given. One hundred fifty-three days after COVID-19 diagnosis when the patient was asymptomatic, Ct was 34.1. During the patient's prolonged course of infection, 2 additional de novo nonsynonymous mutations were also identified in nsp6 and orf3 (Supplementary Appendix, Supplementary Table 1).
Case 2 is a patient in their 50’s with a history of splenectomy and diabetes who underwent DDKT addressing ESRD. Prior to transplantation, the patient received 2 doses of the Moderna mRNA-1273 vaccine. Induction immunosuppression included methylprednisolone and ATG, and the patient was subsequently maintained on prednisone, mycophenolate, and tacrolimus. Delayed graft function prompted empiric methylprednisolone. Graft biopsy 1 week after transplant demonstrated evidence of tacrolimus toxicity, and the patient transitioned to everolimus. Although graft function improved, the patient experienced a series of complications, including significant cytopenias, breakthrough cytomegalovirus viremia, and bacterial infections over several months.
Fourteen months after transplant, the patient developed malaise, dyspnea, and cough. RT-PCR was positive for SARS-CoV-2; however no Ct values and no specimens were available for sequencing at that time. Because of pulmonary infiltrates and hypoxemia, the patient received a 3-day course of remdesivir and a 4-day course of baricitinib, and improved. Eighteen days post–COVID-19 diagnosis, the patient was readmitted with worsening cough and hypoxia and was SARS-CoV-2 positive (Ct of 17.4). X-ray demonstrated worsening patchy infiltrates. The patient required high-flow oxygen, and CT identified multiple cavitary lung lesions (Figure 1A). The patient was managed with methylprednisolone and a 5-day course of remdesivir, and also received voriconazole for pulmonary aspergillosis based on elevated galactomannan from bronchoalveolar lavage and high-dose corticosteroids for biopsy-proven organizing pneumonia. Genomic sequencing on day 25 of illness identified a de novo V792I mutation in RdRp (Figure 1A and B). De novo mutations in the nsp14 exonuclease (Figure 1B) and spike protein (Supplementary Table 1) were also identified at day 32. The patient's symptoms eventually improved significantly, hypoxia resolved, and the patient was discharged.
DISCUSSION
As the COVID-19 pandemic progressed, clinicians have recognized the role that immunosuppression plays in complicating the course of infection. Despite undergoing vaccination prior to transplant, Case 1 experienced a protracted course of symptomatic infection spanning several months. Similarly striking presentations have occurred in other patients treated with rituximab [4, 5]. The increased risk of mortality attributed to rituximab extends to patients with rheumatologic and oncologic indications [6]. Like Case 1, the previously vaccinated Case 2 also experienced life-threatening complications related to COVID-19, including severe pulmonary aspergillosis. RT-PCR analysis and sequencing in both cases confirmed the presence of viable SARS-CoV-2 infection contributing to ongoing symptoms.
Ineffective immune clearance contributes to persistent viral replication in immunocompromised hosts and increased opportunities for mutation. The nonstructural protein nsp12 is the catalytic subunit of the SARS-CoV-2 RdRp. While the exonuclease nsp14 and its cofactor nsp10 offset the low fidelity of nsp12, the mutation frequency of SARS-CoV-2 remains substantial [7]. In the setting of remdesivir exposure and profound immune deficiency allowing protracted infection, genomic sequencing from both patients independently revealed a de novo V792I substitution in ORF1ab, the open reading frame encoding nsp12. A recent study showed that V792I readily develops in vitro in the presence of increasing remdesivir concentrations [3]. Out of the 11 million genomes deposited in the Global Initiative on Sharing Avian Influenza Database (GISAID) globally, fewer than 300 isolates contain the V792I substitution [8]. Interestingly, a recent prepublication identified V792I in 7% of immunocompromised patients [9]. V792I alone increases the remdesivir half-maximal effective concentration (EC50) by 2.6-fold [3]. Other in vitro mutations may complement V792I to further increase its EC50 [3]. Notably, SARS-CoV-2 isolated throughout the course of infection in both our cases also carried P323L, an Omicron-defining mutation, in ORF1ab that has been associated with a modest increase in remdesivir EC50 [10]. The combined effect of these mutations may limit the clinical efficacy of remdesivir.
As remdesivir use has become widespread, and we show that mutations associated with remdesivir resistance arise in vivo, our work emphasizes surveillance efforts to detect mutations in immunocompromised patients. Potentially foreshadowing an “end game” scenario for the COVID-19 pandemic, complex cases like the ones we describe highlight the need for more advanced molecular diagnostics at the onset of illness to guide therapeutic decisions.
Additionally, our work emphasizes the risk of immune escape in immunocompromised hosts, with novel mutations contributing to recrudescence of infection. Equally concerning are recent reports in immunocompromised hosts highlighting the development of SARS-CoV-2 spike mutations conferring resistance to immunotherapeutics after treatment with monoclonal antibodies [11].
Finally, the prolonged nature of the infections outlined in this work emphasizes the critical need for thoughtful input from specialists when considering isolation precautions in immunocompromised hosts. Despite a Ct value approaching 30, detectable anti–SARS-CoV-2 IgG, and markedly improved symptoms 3 months after COVID-19 diagnosis, the patient in Case 1 eventually experienced relapsed symptomatic infection and additional mutations in ORF1ab. Multiple authors have posited that variants of concern may have originated in immunocompromised hosts [12, 13]. The failure to appropriately identify, treat, and control the spread of mutated SARS-CoV-2 isolates could have far-reaching consequences.
METHODS
SARS-CoV-2 Sequencing
Viral genome sequencing and bioinformatic analysis were carried out as described [14] using the XGen SARS-CoV-2 amplicon-based method (Integrated DNA Technologies). Details can be found in the Supplementary Appendix.
Structural Analysis
Three-dimensional structures were created with UCSF ChimeraX 1.4 [15]. Structural overlay was done using MatchMaker in ChimeraX. Chain pairing was performed according to the best aligning pairs of chains between reference and match structure.
Supplementary Data
Supplementary materials are available at Clinical Infectious Diseases online. Consisting of data provided by the authors to benefit the reader, the posted materials are not copyedited and are the sole responsibility of the authors, so questions or comments should be addressed to the corresponding author.
Supplementary Material
Contributor Information
John I Hogan, Department of Medicine, NYU Long Island School of Medicine, Mineola, New York, USA.
Ralf Duerr, Department of Microbiology, NYU Grossman School of Medicine, New York, New York, USA.
Dacia Dimartino, Genome Technology Center, NYU Langone Health, New York, New York, USA.
Christian Marier, Genome Technology Center, NYU Langone Health, New York, New York, USA.
Sarah E Hochman, Department of Medicine, NYU Grossman School of Medicine, New York, New York, USA.
Sapna Mehta, Department of Medicine, NYU Grossman School of Medicine, New York, New York, USA.
Guiqing Wang, Department of Pathology, NYU Grossman School of Medicine, New York, New York, USA.
Adriana Heguy, Genome Technology Center, NYU Langone Health, New York, New York, USA; Department of Pathology, NYU Grossman School of Medicine, New York, New York, USA.
Notes
Acknowledgments. The authors thank Dr. Joan Cangiarella for her continuous support of genomic surveillance for SARS-CoV-2 at NYU Langone Health, including providing institutional funding for this study, and Joanna Fung for assistance with testing, saving, and retrieving specimens.
Financial support. This work was supported by NYU Langone Health. The Genome Technology Center is partially supported by grant number P30CA016087.
Ethics/study approval. This study was approved by the NYU Langone Health Institutional Review Board, protocols i21-00493 and i21-00561. Both patients consented to publication, including use of clinical images.
References
- 1. Saharia KK, Anjan S, Streit J, et al. . Clinical characteristics of COVID-19 in solid organ transplant recipients following COVID-19 vaccination: a multicenter case series. Transpl Infect Dis 2022; 24:e13774. [DOI] [PubMed] [Google Scholar]
- 2. Manothummetha K, Chuleerarux N, Sanguankeo A, et al. . Immunogenicity and risk factors associated with poor humoral immune response of SARS-CoV-2 vaccines in recipients of solid organ transplant: a systematic review and meta-analysis. JAMA Netw Open 2022; 5:e226822. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Stevens LJ, Pruijssers AJ, Lee HW, et al. . Mutations in the SARS-CoV-2 RNA dependent RNA polymerase confer resistance to remdesivir by distinct mechanisms. Sci Transl Med 2022; 14:eabo0718. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Gandhi S, Klein J, Robertson AJ, et al. . De novo emergence of a remdesivir resistance mutation during treatment of persistent SARS-CoV-2 infection in an immunocompromised patient: a case report. Nat Commun 2022; 13:1547. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Clerico M, Dogliotti I, Calcagno A, et al. . COVID-19 in a post-transplant heart recipient who developed aggressive lymphoma: a biphasic course during rituximab treatment. Hemasphere 2021; 5:e592. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Andersen KM, Bates BA, Rashidi ES, et al. . Long-term use of immunosuppressive medicines and in-hospital COVID-19 outcomes: a retrospective cohort study using data from the National COVID Cohort Collaborative. Lancet Rheumatol 2022; 4:e33–41. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Moeller NH, Shi K, Demir O, et al. . Structure and dynamics of SARS-CoV-2 proofreading exoribonuclease ExoN. Proc Natl Acad Sci USA 2022; 119:e2106379119. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Global Initiative on Sharing Avian Influenza Database (GISAID) . Home page. 2022. Available at: https://www.gisaid.org/(Accessed July 2022).
- 9. Wilkinson SA, Richter A, Casey A, et al. . Recurrent SARS-CoV-2 mutations in immunodeficient patients. medRxiv [Preprint]. [cited 2 March 2022]. Available at: 10.1101/2022.03.02.22271697. [DOI] [PMC free article] [PubMed]
- 10. Checkmahomed L, Pont VD, Riola NC, et al. . In vitro selection of remdesivir-resistant SARS-CoV-2 demonstrates high barrier to resistance. bioRxiv [Preprint]. [cited 12 February 2022]. Available at: 10.1101/2022.02.07.479493. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Rockett R, Basile K, Maddocks S, et al. . Resistance mutations in SARS-CoV-2 delta variant after sotrovimab use. N Engl J Med 2022; 386:1477–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Dennehy JJ, Gupta RK, Hanage WP, Johnson MC, Peacock TP. Where is the next SARS-CoV-2 variant of concern? Lancet 2022; 399:1938–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Burki T. The origin of SARS-CoV-2 variants of concern. Lancet Infect Dis 2022; 22:174–5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Duerr R, Dimartino D, Marier C, et al. . Dominance of alpha and iota variants in SARS-CoV-2 vaccine breakthrough infections in New York City. J Clin Invest 2021; 131:e152702. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Goddard TD, Huang CC, Meng EC, et al. . UCSF ChimeraX: meeting modern challenges in visualization and analysis. Protein Sci 2018; 27:14–25. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.