Abstract
γδ T cells accumulate during Plasmodium infections in both murine and human malarias. The biological role of these cells and the antigens that they recognize are not clearly understood, although recent findings indicate that γδ T cells in general influence both innate and antigen-specific adaptive host responses. We examined the accumulation of γδ T cells elicited during infection with virulent and avirulent Plasmodium yoelii parasites in relatively susceptible and resistant strains of mice. Our results indicated that in nonlethal malaria infections, γδ T cells comprise a larger proportion of splenic T cells than in lethal infections and that only a live infection is capable of inducing an increase in the percentage of γδ T cells in vivo. Furthermore, we demonstrate that γδ T cells elicited during a P. yoelii infection respond by proliferation in vitro to P. falciparum heat shock proteins (HSPs) of 60 and 70 kDa, suggesting a possible immunological involvement of parasite HSPs in this arm of the cellular immune response during malarial infection in mice.
Malaria represents a worldwide health concern and poses a challenge in vaccine development. To develop an effective vaccine, we must first understand the immunological mechanisms involved in the protection against infection. Great advances in the understanding of the humoral and cellular effector arms of the immune response during malaria infection have occurred within the last several years. Many unanswered questions remain, however, such as the role of γδ T cells in infection.
In malaria infections, γδ T cells accumulate in the peripheral blood and spleens of individuals infected with Plasmodium falciparum and P. vivax (22, 23, 28, 32, 34, 38). These γδ T cells are stimulated most strongly by the schizont/merozoite stage of the parasite and are able to inhibit the replication of P. falciparum in vitro (2, 12, 15, 16, 23). During P. chabaudi infections in mice, the splenic γδ T-cell population expanded more than 10-fold; γδ T-cell-depleted mice, however, were unable to control parasitemia during the same time frame as wild-type mice (41–43). More recently, Tsuji et al. have shown by passive transfer experiments that a γδ T-cell clone was able to inhibit the development of P. yoelii during the liver stage of infection in mice (40). γδ T cells are therefore believed to play a beneficial role in disease outcome during malaria infection.
We have examined the biological role of γδ T cells in a rodent malaria system by using P. yoelii strains that cause lethal (17XL) and nonlethal (17XNL) malaria in genetically susceptible and relatively resistant strains of mice. Our results indicate that (i) γδ T cells increase as a percentage of total T cells in the spleens of the infected mice, (ii) γδ T cells comprise a smaller percentage of T cells in more susceptible mouse strains and in infections with more virulent forms of the parasite, (iii) a live infection is required to elicit a γδ T-cell accumulation, and (iv) γδ T cells elicited during infection with the malaria parasite proliferate in vitro in the presence of P. falciparum heat shock proteins (HSPs) of 60 and 70 kDa.
MATERIALS AND METHODS
Mice.
The mice [female BALB/c (H-2d) and C57BL/6 (H-2b)] used in these studies were 6 to 10 weeks old and obtained from the National Cancer Institute, Frederick. Three to five mice were used for any given time point of observation in these studies.
Parasites, infections, and immunizations.
P. yoelii 17XNL and 17XL were maintained by serial passage. Parasites were injected by the intraperitoneal (i.p.) route at 106 parasitized erythrocytes (RBCs) per mouse unless otherwise specified. Parasitemia was determined by microscopic examination of Giemsa-stained thin blood smears every 2 days postinfection until resolution. Results are expressed as a mean of results obtained with three to five mice, and experiments were repeated at least three times with representative data shown. For the immunization experiments, blood was collected in heparin from infected mice. Leukocytes were removed by passage of blood through a glass bead column followed by a cellulose column (1). The parasites were then inactivated in a gamma irradiator (7.2 × 104 rads), and irradiated RBCs were injected i.p. at 1010 infected RBCs.
Flow cytometry.
Spleens were removed from the mice and teased gently in RPMI 1640 to obtain a single-cell suspension. The cells were then treated with ammonium chloride lysing solution to remove RBCs. Washed splenocytes (106) were first incubated at 4°C for 15 min in phosphate-buffered saline containing 1% bovine serum albumin, 0.1% sodium azide (pH 7.2), hamster immunoglobulin G (10 μg/106 cells), and rat immunoglobulin G (10 μg/106 cells) isotype controls (Pharmingen). The cells were then incubated for 30 min at 4°C with the appropriate monoclonal antibodies (MAbs) (0.5 μg). The cells were washed in phosphate-buffered saline, fixed with 1% formaldehyde, and analyzed on a Becton Dickinson FACScan apparatus. The following fluorescein isothiocyanate-conjugated MAbs were used for staining: anti-CD3ɛ (Pharmingen 145-2C11), anti-αβ T-cell receptor (TCR) (Pharmingen H57-597), and anti-CD8α (Gibco-BRL 53-6.7). The following phycoerythrin-conjugated MAbs were used: anti-γδ TCR (Pharmingen GL3), anti-CD3ɛ (Gibco-BRL 29B), and anti-CD4 (Pharmingen GK1.5).
Cytotoxic-cell depletion.
Spleens were removed, teased in RPMI 1640, and treated with an ammonium chloride lysing solution to remove RBCs. Washed cells were resuspended at 108 cells/ml in RPMI 1640 supplemented with 25 mM HEPES and 0.3% bovine serum albumin. The appropriate antibodies (see below) were added, and the cells were treated for 30 min on ice. The cells were pelleted, 100 μl of Low-Tox-M complement (Cedarlane) was added to the RPMI 1640, and the mixture was incubated for 30 min at 37°C. The cells were washed, and the procedure was repeated. Fluorescence-activated cell sorter (FACS) analysis was used to determine the effectiveness of the depletion. Cytotoxic-cell elimination removed more than 95% of the appropriate splenocyte populations. The antibody cocktail of anti-CD4 (GK 1.5 culture supernatant; 0.5 ml), anti-CD8 (2.43 ascites; 20 μl), anti-major histocompatibility complex class II (MHC II) I-Ad (MK-D6 ascites; 20 μl), and anti-αβ TCR (H57 culture supernatant; 0.5 ml) was used to obtain a γδ-T-cell-enriched population. αβ TCR-enriched populations were obtained after treatment with a cocktail of anti-MHC II (MK-D6 ascites; 20 μl) and anti-γδ TCR (GL-3 culture supernatant; 0.5 ml).
Lymphocyte proliferation.
After cytotoxic-cell depletion, cells were resuspended in Dulbecco’s minimal essential medium supplemented with 10% fetal bovine serum, 2 mM l-glutamine, 50 μM 2-mercaptoethanol, 100 U of penicillin per ml, and 100 μg of streptomycin sulfate per ml. Each cell population (unfractionated, γδ enriched, and αβ enriched) was plated at 2 × 105 cells/well in a flat-bottom 96-well plate in the presence of 3 × 105 irradiated antigen-presenting cells (3,000 rads), with triplicate wells used for each antigen. The following antigens were used in the proliferation experiments: medium alone (control), P. yoelii 17XNL extract (106/ml), P. falciparum HSP 60 (10 μg/ml) and HSP 70B (10 μg/ml), and concanavalin A (2.5 μg/ml). The cells were cultured for 5 days at 37°C under 5% CO2 and then pulsed with 1 μCi of [3H]thymidine for 18 h. The cells were harvested with a 96-well plate harvester, and radioactivity was measured in a Packard radioactivity counter. Results are expressed as stimulation index (S.I.), defined as average counts per minute in antigen wells/average counts per minute in negative control wells.
γδ TCR analysis.
Mice were sacrificed on day 10 postinfection, and the spleens were removed and prepared as described above. RNA was isolated with Tri Reagent (Molecular Research Center, Inc.) as specified by the manufacturer. First-strand cDNA was synthesized from total RNA as follows. Briefly, 5 μg of total RNA was mixed with 500 ng of oligo(dT)15 and 1 μl of a 10 mM deoxynucleoside triphosphate (Pharmacia Biotech) mix and incubated for 50 min at 37°C in the presence of 1× first-strand buffer (Gibco-BRL), dithiothreitol, and SuperScript II (Gibco-BRL). PCR was performed with a 1:10 dilution of the cDNA template (5 μl) in a 50-μl reaction mixture containing 5 μl of 10× PCR buffer (Sigma), 400 ng of the appropriate primer, 100 μM deoxynucleoside triphosphate mix (Perkin-Elmer), and 1 U of Taq DNA Polymerase (Sigma). The PCR cycles consisted of an initial denaturation at 94°C for 5 min followed by 30 cycles of 94°C for 45 s, annealing at 45°C for 1 min, and extension at 72°C for 2 min, with a final extension of 10 min. The PCR-amplified products were analyzed on a 1% agarose gel. The specific primers used have been described previously (39) and were designed to amplify any of the rearranged transcripts containing Vγ1.1, Vγ1.2, Vγ2, Jγ1, Jγ2, or Jγ4 and Vδ4, Vδ5, Vδ6, and Jδ1.
Antigens.
P. falciparum HSP 60 and HSP 70B recombinant proteins containing the His6 tag were expressed in Escherichia coli and purified over a Ni2+ column (11, 26), as was the P. falciparum 27-kDa protein (35). The P. yoelii antigen extract was prepared from BALB/c mice infected with P. yoelii (17XNL). Platelets and leukocytes were removed by passing the blood through a glass bead column followed by a cellulose column (1). The cells were then briefly sonicated, sterilized by γ-irradiation, and stored at −70°C.
RESULTS
Parasitemia and splenic T-cell differences in inbred strains of mice.
BALB/c and C57BL/6 mice were infected with 106 P. yoelii 17XNL-infected RBCs and monitored over the course of infection for parasitemia. As seen in Fig. 1, BALB/c mice are relatively more resistant to P. yoelii than are C57BL/6 mice as measured by the percent parasitemia and the duration of infection. In both mouse strains, initial parasitemia increased at relatively the same rate; however, on day 10, when BALB/c mice reached a peak parasitemia of 10 to 15% which resolved by day 13, the parasitemia in C57BL/6 mice continued to rise until it was higher than 50% on day 17 and resolved around day 25.
FIG. 1.
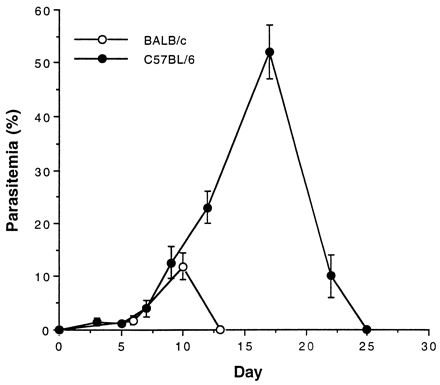
Parasitemia in BALB/c and C57BL/6 mice. Mice were infected i.p. with P. yoelii 17XNL (106 parasitized RBCs). Parasitemia was measured every other day by microscopic examination of a blood smear stained with Giemsa reagent. The values shown are mean and standard deviation.
We next measured changes in the γδ T-cell population in the spleen (Fig. 2) and lymph nodes (data not shown) of both strains of mice. An increase in the percentage of γδ T cells was seen only in the spleens of the infected mice. There were significant differences in the percentage of γδ T cells in the spleens of BALB/c (Fig. 2A) and C57BL/6 (Fig. 2B) mice. In BALB/c mice, γδ T cells accumulated to a level of 20% of total splenic T cells on day 10 before gradually declining to 7.4% by day 24, after the resolution of parasitemia. In contrast, γδ T-cell percentages continued to increase gradually in C57BL/6 mice. On day 10, both strains had roughly the same percent parasitemia (12%), although the γδ T-cell population in C57BL/6 mice comprised only 9% of the total splenic T cells, in contrast to 20% in the BALB/c. These results demonstrated that the more resistant BALB/c mice displayed a higher and faster percent increase in γδ T cells than did the more susceptible C57BL/6 mice.
FIG. 2.
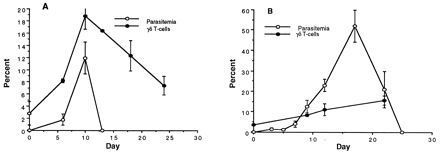
Parasitemia (same data as in Fig. 1) and the percentage of γδ T cells in the spleens of BALB/c (A) and C57BL/6 (B) mice infected with P. yoelii 17XNL (106 parasitized RBCs). Spleens were removed on the appropriate day postinfection. The splenocyte cell suspension was stained with anti-CD3 and anti-γδ TCR for flow cytometry. The values shown are mean and standard deviation.
We also examined changes in the CD4+ and CD8+ splenic T-cell populations. CD8+ cells in both groups of mice showed a similar profile, with an initial population comprising 30 to 35% of the total T cells in the spleen and decreasing to 15% at peak parasitemia in BALB/c mice and later in C57BL/6 mice. In more resistant (BALB/c) mice, the CD4+ population remained constant at 65% of the total T-cell number throughout the infection. In contrast, in C57BL/6 mice, the CD4+ population showed a gradual reduction from an initial 58% to 33% on day 20 (Fig. 3). Therefore, the two major differences observed in the splenic T cells of the more susceptible and less susceptible mice during P. yoelii infection were (i) a lower percentage of γδ T cells in susceptible mice (C57BL/6) and (ii) a decrease in the percentage of CD4+ T cells in the susceptible mice (C57BL/6).
FIG. 3.
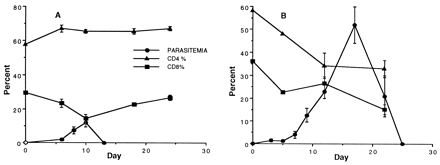
Parasitemia (same data as in Fig. 1) and the percentage of CD4+ and CD8+ cells in the spleens of BALB/c (A) and C57BL/6 (B) mice infected i.p. with P. yoelii 17XNL (106 parasitized RBCs). On the appropriate day spleens were removed and the cell suspension was stained with anti-αβ and anti-CD4 or anti-αβ and anti-CD8 for flow cytometry. The values are expressed as mean and standard deviation.
A secondary infection is associated with a lower percentage of γδ T cells.
To evaluate splenic T-cell populations during a challenge infection, BALB/c mice were infected with 106 P. yoelii 17XNL-infected RBCs and rechallenged with the same dose 3 weeks later when the mice were completely free of any parasitemia. Rechallenged mice were generally resistant to infection since they developed very low parasitemia if any. Eight days after the secondary infection, the mice were sacrificed and the spleens were removed for analysis of γδ, CD4+, CD8+, and αβ T cells by flow cytometry. As shown in Fig. 4, the percentage of γδ T cells did not increase upon rechallenge of the mice, although an elevation in both CD4+ and CD8+ T-cell numbers was observed.
FIG. 4.
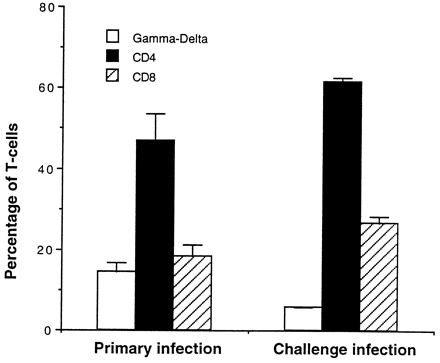
Changes in splenic T-cell populations during primary and challenge infections. BALB/c mice were infected i.p. with P. yoelii 17XNL (106 parasitized RBCs) and rechallenged with the same dose 3 weeks later. Eight days after the secondary infection, the spleen cells were stained with the appropriate combination of antibodies for flow cytometry. Results are expressed as mean and standard deviation.
Comparison of phenotypic differences in the splenic T-cells of mice infected with virulent and avirulent strains of P. yoelii.
To gain further insight into whether γδ T cells play a role in controlling malaria infection, we investigated differences in the splenocyte populations in BALB/c mice infected with virulent and avirulent strains of the parasite. Mice were infected with either 106 P. yoelii 17XL-, 104 P. yoelii 17XL-, or 106 P. yoelii 17XNL-infected RBCs, and parasitemia and splenocyte T-cell populations were measured. As shown in Table 1, parasitemia increased much faster in the lethal infections than in the nonlethal infections and the mice infected with lethal parasites were not able to resolve the infection. Examination of splenic T-cell populations showed no difference in the percentage of CD4+ or CD8+ cells (data not shown). However, significant differences were observed when the percentages of γδ T cells were measured. The γδ T-cell percentage did not change in mice infected with the lethal parasites at 104 and 106 parasitized RBCs, while during the same infection period with the nonlethal parasite the percentage of γδ T cells increased threefold. A possible interpretation of these observations is that accumulation of γδ T cells plays a protective role in less severe malaria caused by P. yoelii 17XNL and that immune system changes during fulminating infection by P. yoelii 17XL do not allow any significant expansion of the percentage of γδ T cells. This observation further suggests a potentially protective role for γδ T cells in less severe malaria infections.
TABLE 1.
Changes in parasitemia and percent γδ T cells during infection with nonlethal and lethal strains of P. yoelii
| P. yoelii strain and dose | Day | % Parasitemia | % γδ T cells |
|---|---|---|---|
| 17XNL (106) | 0 | 0 | 2.8 |
| 3 | 0.1 | 3.3 | |
| 6 | 1.8 | 8.2 | |
| 17XL (104) | 0 | 0 | 2.3 |
| 3 | 0 | 1.8 | |
| 5 | 3.7 | 1.7 | |
| 7 | 61 | 2 | |
| 17XL (106) | 0 | 0 | 2.1 |
| 3 | 1.8 | 2.1 | |
| 5 | 74.6 | 2.4 |
BALB/c mice were infected with 106 P. yoelii 17XNL-parasitized RBCs or 106 or 104 P. yoelii 17XL-parasitized RBCs. Parasitemia was measured by microscopy, and γδ T cells were monitored by the staining of splenocytes with anti-CD3 and anti-γδ TCR MAbs.
A productive infection is required to elicit γδ T cells.
To investigate if a productive infection is required to elicit γδ T-cell expansion, BALB/c mice were infected with live P. yoelii-infected RBCs (106) or immunized with 1010 irradiation-inactivated parasites. The mice were sacrificed on day 8, and γδ T-cell percentages were measured by flow cytometry. Results with the infected group reconfirmed our previous finding in which γδ T cells accounted for roughly 18% of splenic T cells. On the other hand, no change in the percentage of splenic γδ T cells was seen when the mice were inoculated with radiation-inactivated parasites (Fig. 5), demonstrating that only a productive infection is capable of eliciting an increase in the percentage of γδ T cells.
FIG. 5.
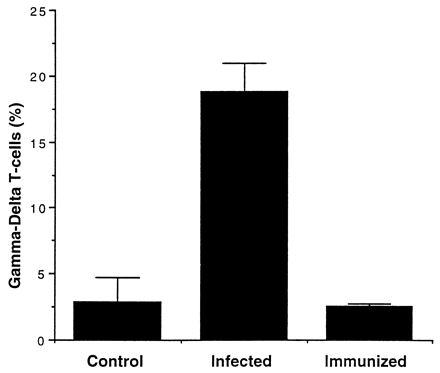
Percentage of γδ T cells in the spleens of control, infected, and immunized mice. Mice were infected with 106 parasitized RBCs, and the γδ T-cell population was measured by staining splenocytes with anti-CD3 and anti-γδ TCR on day 8. Immunized mice were given 1010 irradiated parasitized RBCs i.p., and γδ T-cell percentages were measured on day 8. Control mice were uninfected. Results are expressed as mean and standard deviation.
γδ T cells respond to malarial HSPs.
It has been well documented that γδ T cells respond to Mycobacterium tuberculosis HSPs (5, 14, 19, 33, 38). Therefore, we tested the ability of γδ T cells elicited in a malaria infection to respond to Plasmodium HSPs. BALB/c and C57BL/6 mice were infected with 106 parasitized RBCs, and the spleens were tested in lymphoproliferation assays at three different time points. Splenocytes were separated into three groups: unfractionated, γδ enriched, and αβ enriched. The γδ-enriched and the αβ-enriched T cells were obtained by treatment with a cocktail of antibodies followed by complement treatment (described in Materials and Methods) and were found to be greater than 90% pure as determined by flow cytometry (data not shown). Figure 6 shows the results obtained with BALB/c mice. The total T-cell population did not respond strongly on days 9 and day 12 postinfection, during which time a productive infection was occurring. On day 20, however (8 days after resolution of the infection), there was a strong proliferative response by total T cells to the P. yoelii lysate (SI = 10.5), as well as to Plasmodium recombinant HSPs of 60 kDa (SI = 14.6) and 70 kDa (SI = 15.2). Figure 6B shows the results of the proliferation of the γδ T-cell-enriched population. On day 9 there was little proliferation with all three antigen preparations, but there was a significant proliferative response to P. falciparum HSP 60 (SI = 6.5 on day 12 and 6 on day 20) and HSP 70B (SI = 4.5 on day 12 and 9.4 on day 20) later in infection. These γδ T cells did not proliferate when stimulated in the presence of the P. yoelii extract. Likewise, the αβ-enriched T-cell population showed a proliferative response to HSP 60 (SI = 5.4 on day 12 and 8.0 on day 20) and HSP 70B (SI = 7.7 on day 20). None of these cell populations responded by proliferation (SI < 2.0) in the presence of an unrelated parasite antigen, Pfg27 (35), used as a negative control (data not shown). These results demonstrate that the γδ T cells elicited during infection in BALB/c mice with P. yoelii respond by proliferation in the presence of HSP 60 and HSP 70B of P. falciparum. Similar proliferation studies with C57BL/6 mice were inconclusive (data not shown), and these were consistent with the experience of other investigators with C57BL/6 mice (20).
FIG. 6.
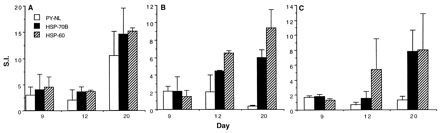
Proliferative responses to P. yoelii extracts and P. falciparum HSP 60 and HSP 70. BALB/c mice were sacrificed on day 9, 12, or 20 after infection with P. yoelii 17XNL. Spleens were removed and treated with a cocktail of antibodies. (A) Total cells (no antibody treatment); (B) γδ T-cell enriched (anti-I-Ad, anti-αβ, anti-CD4, anti-CD8); (C) αβ T-cell enriched (anti-I-Ad, anti-γδ). The cells were plated at 2 × 105/well of a 96-well plate in the presence of P. yoelii extract (106 parasitized RBCs/ml), P. falciparum HSP 60 (10 μg/ml), or P. falciparum HSP 70 (10 μg/ml) with irradiated APCs (3 × 105/well). On day 5, the cells were pulsed with 1 μCi of [3H]thymidine, and 18 h later they were harvested. Radioactive counts in cells incubated in medium alone were 53 (day 9), 87 (day 12), and 103 (day 20) in group A; 115 (day 9), 88 (day 12), and 76 (day 20) in group B; and 108 (day 9), 140 (day 12), and 127 (day 20) in group C. Results are expressed as mean SI and standard deviation.
γδ TCR analysis.
Finally we examined γδ TCR usage in mice that had been infected with P. yoelii 17XNL-infected RBCs 10 days prior to RT-PCR analysis. The results (Table 2) show that the γ chain, consisting of V1.2 with the J2 and J4 segments, and the δ chain, consisting of V4 and V5 with the J1 segment, were predominantly expressed in the splenocytes of infected mice.
TABLE 2.
RT-PCR analysis of γδ TCRa
| TCR chain | Chain expression with:
|
||
|---|---|---|---|
| J1 | J2 | J4 | |
| Gamma | |||
| V1.1 | − | − | − |
| V1.2 | − | + | + |
| V2 | − | − | − |
| Delta | |||
| V4 | + | ||
| V5 | + | ||
| V6 | − | ||
+, PCR amplification (various combinations of V and J primers)-positive results; −, negative results.
DISCUSSION
γδ T cells accumulate in a wide variety of infections, providing both potentially beneficial and pathological roles (6, 8, 17, 18, 21, 23–25, 29, 32, 34, 38, 42). The biological role of these cells in immunity is unclear; however, recent studies indicate that γδ T cells influence both early innate and antigen-specific adaptive host responses. Since the discovery of γδ T cells in 1986 (7), significant advances in our understanding of the immunobiological role of these cells have been made, although this progress has occurred much more slowly than our understanding of the αβ T-cell population. The biological role of γδ T cells seems to vary with the organism causing infection, the anatomical location of the γδ T cell, and the unconventional ways in which these cells recognize antigen (see references 8, 11a, 18, 36, and 38 for reviews).
The studies described in this paper were undertaken to enhance our understanding of the role of the γδ T cell in a malaria infection. Our strategy was to analyze γδ T cells and their antigen specificity during infections with avirulent and virulent strains of P. yoelii by using less susceptible (BALB/c) and more susceptible (C57BL/6) strains of mice. The two inbred strains of mice (BALB/c and C57BL/6) display marked differences in the course of infection, which was extended by 12 days and was associated with four- to fivefold higher levels of parasitemia in the more susceptible C57BL/6 mice than in BALB/c mice. Initial experiments examined the spleen and lymph nodes for changes in γδ T-cell populations. In subsequent studies, we focused on the spleen as the target organ based on our observation that it displayed marked differences in the percentage of γδ T cells. Moreover, previous studies have demonstrated a critical role for spleen in the control of rodent malarias (45). As shown in Fig. 2, there were significant differences in the accumulation of γδ T cells in the two mouse strains, with a much slower accumulation in C57BL/6 (relatively more susceptible) mice.
The increase in the percentage of γδ T cells in both strains of mice coincided with the peak percent parasitemia and declined thereafter. The simplest interpretation of these results may be that in BALB/c mice, γδ T cells could play a role in controlling acute parasite infection (percent parasitemia and duration), resulting in a less virulent (resolving) infection. Further support for such an interpretation was provided by the lack of any such increase during infection with highly lethal parasites, even when a 2-log-lower inoculum was used in an attempt to alter the time course of rising parasitemia. Previous studies have also reported up to a 10-fold expansion of splenic γδ T cells during P. chabaudi infections in mice (41). Antibody depletion of γδ T cells in those studies resulted in exacerbated P. chabaudi malaria, suggesting a beneficial role for these T cells. Colocalization of γδ T cells and parasite infection in the red pulp of the spleen might indicate a possible location for destruction of the parasite (4). More recently, Tsuji et al. have shown a partially protective role for a γδ T-cell clone against the hepatic stage of malaria infection (40). However, this γδ T-cell clone had no effect against the erythrocytic stage of infection. In a different rodent malaria model (P. berghei), γδ T cells were shown to participate in the pathogenesis of malaria leading to cerebral malaria (44). Thus, it appears that γδ T cells might provide a beneficial function in the early phase of infection with less virulent parasites. Other cell-mediated and humoral immune mechanisms might be more effective in chronic phase of infection. Nonetheless, these studies do support the notion that γδ T cells might provide a first line of defense or early innate immune mechanism during the host response to infectious agents.
We also examined CD4+ and CD8+ αβ T-cell changes during infection in these mice. In both strains of mice we observed a decrease in the percentage of CD8+ T cells which coincided with peak parasitemia. This could be due to a direct impact of the rising γδ T-cell percentage on CD8+ cells or could indicate that CD8+ cells are not essential as the primary mediators of the blood stage malaria infection with nonlethal P. yoelii. Several studies have suggested that CD4+ T cells play an important role in the elimination of malaria infections in mice (13, 42). In our studies, the number of CD4+ cells remained relatively constant in the BALB/c mouse infection but decreased in the C57BL/6 mouse infection from roughly 60% to 35% of total cells at peak parasitemia. We do not know if the drop in the CD4+-T-cell population in the more susceptible C57BL/6 mice is directly accountable for our findings of a lower accumulation of γδ T cells. van der Heyde et al. (42) have suggested that CD4+ T cells modulate γδ T-cell responses. Thus, a combination of decreasing numbers of CD4+ T cells and lower accumulation of γδ T cells in C57BL/6 mice might provide one possible immunological mechanism resulting in a more susceptible phenotype, i.e., extended duration of the course of infection and much higher peak parasitemia than in the less susceptible BALB/c mice.
Further support for a beneficial role for γδ T cells was provided by studies in which we compared γδ T cells during infection with lethal and nonlethal parasites. We do not know what triggers the activation of γδ T cells during infection with avirulent P. yoelii. It appears that γδ T cells or their soluble mediators participate in resolving infection caused by nonlethal parasites. Our studies also suggest that these cells participate primarily early in infections in naive mice. Once CD4+ and CD8+ T cells have been primed during primary infections, they might provide additional mechanisms involved in suppressing parasitemia during subsequent infections. Further studies are needed to evaluate the role of the γδ T cell during infections with virulent and avirulent strains of malaria.
Finally, we investigated whether Plasmodium HSPs are recognized as antigens by the γδ T cells elicited during malaria infections. γδ T cells elicited by various infectious organisms recognize a peptide epitope in M. tuberculosis HSP 65 (5, 6, 14, 18, 19, 33) including a γδ T-cell clone with protective activity against the liver stage of infection of P. yoelii (40). More recent studies have demonstrated that γδ T cells also recognize epitopes in nonpeptidic phosphorylated antigens (3, 9, 30). The studies described here demonstrate that γδ T cells elicited during malaria infection proliferate in response to P. falciparum HSPs of 60 and 70 kDa. There was very little proliferative response to any of the antigens tested in total splenocytes, as well as those enriched in γδ T cells and in αβ T cells at the earliest time point, and this was not a result of high background proliferation, since cells incubated in medium alone had comparable counts. Moreover, the cells in all groups tested at all three time points responded vigorously in the presence of concanavalin A (data not shown). Similar suppressed lymphoproliferative responses early during a malaria infection are well documented (10, 37).
Plasmodium HSPs have approximately 60% homology to mouse and other eukaryotic HSPs, and therefore it is conceivable that these γδ T cells can also recognize self HSPs and that this could account for the elevated γδ T-cell levels that remain in the spleen long after the infection has been resolved. Since γδ T cells elicited during infection with P. yoelii respond to epitopes in HSP 70 and HSP 60, we also investigated if immunization with purified HSP 70 will elicit specific γδ T cells. However, the results of these studies were inconclusive, since the incomplete Freund’s adjuvant used during immunization has been shown earlier to elicit γδ T cells, as was also found in our own studies (results not shown). In Toxoplasma gondii infections, γδ T cells play an essential role in the control of the infection, and HSP 65 expression on macrophages was essential for this apparently γδ T-cell-dependent protection. In these studies, virulent forms of the parasite were less able to induce HSP 65 expression on macrophages (31). We do not know if similar mechanisms might participate during malarial infection.
In the present study, we also examined the preferential usage of V and J segments of the TCR of the γδ T cells in the spleens elicited during the infection. Surprisingly, not only did we find γδ T cells that are located primarily in the spleen but we also found VJ gene rearrangements primarily associated with the epidermal areas of the mice. This might suggest that these cells are eventually recruited to the spleen at the time of infection and could account for differences seen in Plasmodium infections in which antibody-mediated γδ T-cell knockout mice show differences in infection compared to gene-disrupted T-cell knockout mice (27, 43).
Overall, the interaction between γδ T cells and malaria appears to be complex, with variations not only among strains of mice used but also among strains of parasites. Additionally, interplay of other T cells (CD4+, CD8+) may greatly affect the course of acute versus chronic infection as well as accumulation of γδ T cells. Since γδ T cells do respond to HSPs of the parasite, perhaps one strategy would be to obtain Plasmodium HSP-specific γδ T-cell clones and directly evaluate their function in vivo and in vitro during infection with the malaria parasite.
ACKNOWLEDGMENTS
This work was supported by National Institutes of Health grant AI 31589.
We thank Hong Zheng and Ashis Das for recombinant clones encoding PfHSP 70 and PfHSP 60 respectively, D. Pardoll for hybridoma GL-3, and Elvia Ramirez for help with the flow cytometry.
REFERENCES
- 1.Baggaley V C, Atkinson E M. Use of CF12 columns for the preparation of DNA from rodent malaria. Trans R Soc Trop Med Hyg. 1972;66:4–5. doi: 10.1016/0035-9203(72)90005-3. [DOI] [PubMed] [Google Scholar]
- 2.Behr C, Dubois P. Preferential expansion of Vγ9 Vδ2 T-cells following stimulation of peripheral blood lymphocytes with extracts of Plasmodium falciparum. Int Immunol. 1991;4:361–366. doi: 10.1093/intimm/4.3.361. [DOI] [PubMed] [Google Scholar]
- 3.Behr C, Poupot R, Peyrat M, Poquet Y, Constant P, Dubois P, Bonneville M, Fournie J. Plasmodium falciparum stimuli for human γδ T-cells are related to phosphorylated antigens of mycobacteria. Infect Immun. 1996;64:2892–2896. doi: 10.1128/iai.64.8.2892-2896.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Bordessoule D, Gaulard P, Mason D Y. Preferential localization of human lymphocytes bearing γδ T-cell receptors to the red pulp of the spleen. J Clin Pathol. 1990;43:461–464. doi: 10.1136/jcp.43.6.461. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Born W, Hall L, Dallas A, Boymel J, Schinnick T, Young D, Brennan P, O’Brien R. Recognition of a peptide antigen by heat shock-reactive γδ T-lymphocytes. Science. 1990;249:67–69. doi: 10.1126/science.1695022. [DOI] [PubMed] [Google Scholar]
- 6.Born W, O’Brien R. The γδ T-cell response to stress: unresolved issues and possible significance. Res Immunol. 1990;141:595–600. doi: 10.1016/0923-2494(90)90063-5. [DOI] [PubMed] [Google Scholar]
- 7.Brenner M B, McLean J, Dialynas D P, Strominger J L, Smith J A, Owen F L, Seidman J G, Ip S, Rosen F, Krangel M S. Identification of a putative second T-cell receptor. Nature. 1986;322:145–149. [PubMed] [Google Scholar]
- 8.Cheng S H, Penninger J M, Ferrick D A, Molina T J, Wallace V A, Mak T W. Biology of murine γδ T cells. Crit Rev Immunol. 1991;11:145–166. [PubMed] [Google Scholar]
- 9.Constant P, Davodeau F, Peyrat M, Poquet Y, Puzo G, Bonneville M, Fournie J. Stimulation of human γδ T cells by nonpeptidic mycobacterial ligands. Science. 1994;264:267–270. doi: 10.1126/science.8146660. [DOI] [PubMed] [Google Scholar]
- 10.Correa M, Narayanan P R, Miller H C. Suppressive activity of splenic adherent cells from P. chabaudi infected mice. J Immunol. 1980;125:749–754. [PubMed] [Google Scholar]
- 11.Das A, Syin C, Fujioka H, Zheng H, Goldman N, Aikawa M, Kumar N. Molecular characterization and ultrastructural localization of Plasmodium falciparum HSP60. Mol Biochem Parasitol. 1997;88:95–105. doi: 10.1016/s0166-6851(97)00081-9. [DOI] [PubMed] [Google Scholar]
- 11a.De Libero G. Sentinel function of broadly reactive human γδ T cells. Immunol Today. 1997;18:22–26. doi: 10.1016/s0167-5699(97)80010-2. [DOI] [PubMed] [Google Scholar]
- 12.Elloso M M, van der Heyde H C, vande Waa J A, Manning D D, Weidanz W P. Inhibition of Plasmodium falciparum in vitro by human γδ T cells. J Immunol. 1994;153:1187–1194. [PubMed] [Google Scholar]
- 13.Elloso M M, van der Heyde H C, Troutt A, Manning D D, Weidanz W P. Human γδ T cell subset-proliferative response to malarial antigen in vitro depends on CD4+ T cells or cytokines that signal through components of the IL-2R. J Immunol. 1996;157:2096–2102. [PubMed] [Google Scholar]
- 14.Fu Y-X, Cranfill R, Vollmer M, van der Zee R, O’brien R L, Born W. In vivo response of murine γδ T cells to a heat shock protein-derived peptide. Proc Natl Acad Sci USA. 1992;90:322–326. doi: 10.1073/pnas.90.1.322. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Goerlich R, Hacker G, Pfeffer K, Heeg K, Wagner H. Plasmodium falciparum merozoites primarily stimulate the Vγ9 subset of human γ/δ T cells. Eur J Immunol. 1991;21:2613–2616. doi: 10.1002/eji.1830211045. [DOI] [PubMed] [Google Scholar]
- 16.Goodier M, Fey P, Eichmann K, Langhorne J. Human peripheral blood γδ T cells respond to antigens of Plasmodium falciparum. Int Immunol. 1991;4:33–41. doi: 10.1093/intimm/4.1.33. [DOI] [PubMed] [Google Scholar]
- 17.Goodier M, Krause-Jauer M, Sanni A, Massougbodji A, Sadeler B-C, Mitchell G H, Modolell M, Eichmann K, Langhorne J. γδ T cells in the peripheral blood of individuals from an area of holoendemic Plasmodium falciparum transmission. Trans R Soc Trop Med Hyg. 1993;87:692–696. doi: 10.1016/0035-9203(93)90299-6. [DOI] [PubMed] [Google Scholar]
- 18.Haas W. Gamma/delta cells. Annu Rev Immunol. 1993;11:637–685. doi: 10.1146/annurev.iy.11.040193.003225. [DOI] [PubMed] [Google Scholar]
- 19.Haregewoin A, Soman G, Hom R C, Finberg R W. Human γδ T cells respond to mycobacterial heat-shock protein. Nature. 1989;340:309–311. doi: 10.1038/340309a0. [DOI] [PubMed] [Google Scholar]
- 20.Hauda, K. M., P. C. Sayles, and D. L. Wassom. 1993. Plasmodium yoelii: cellular immune responses in splenectomized and normal mice. 76:385–393. [DOI] [PubMed]
- 21.Hisaeda H, Nagasawa H, Maeda K-I, Mackawa Y, Ishikawa H, Ito Y, Good R, Himeno K. γδ T-cells play an important role in hsp65 expression and in acquiring protective immune responses against infection with Toxoplasma gondii. J Immunol. 1995;154:244–251. [PubMed] [Google Scholar]
- 22.Ho M, Webster K, Tongtawe P, Pattanapanyasat K, Weidanz W P. Increased γδ T cells in acute Plasmodium falciparum malaria. Immunol Lett. 1990;25:139–142. doi: 10.1016/0165-2478(90)90105-y. [DOI] [PubMed] [Google Scholar]
- 23.Ho M, Tongtawe P, Kriangkum J, Wimonwattrawatee T, Pattanapanyasat K, Bryant L, Shafiq J, Suntharsamai P, Looareesuwan S, Webster H K, Elliott J F. Polyclonal expansion of peripheral γδ T cells in human Plasmodium falciparum malaria. Infect Immun. 1994;62:855–862. doi: 10.1128/iai.62.3.855-862.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Jouen-Beades F, Paris E, Dieulois C, Lemcland J-F, Barre-Dezelus V, Marret S, Humbert G, Leroy J, Tron F. In vivo and in vitro activation and expansion of γδ T cells during Listeria monocytogenes infection in humans. Infect Immun. 1997;65:4267–4272. doi: 10.1128/iai.65.10.4267-4272.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Kasper L H, Matsuura T, Foneseka S, Arruda J, Channon J Y, Khan I A. Induction of γδ T cells during acute murine infection with Toxoplasma gondii. J Immunol. 1996;157:5521–5527. [PubMed] [Google Scholar]
- 26.Kumar N, Zheng H. Evidence for epitope-specific thymus-independent response against a repeat sequence in a protein antigen. Immunology. 1998;94:28–34. doi: 10.1046/j.1365-2567.1998.00486.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Langhorne J, Mombaerts P, Tonegawa S. Alpha beta and gamma delta T cells in the immune response to the erythrocytic stages of malaria in mice. Int Immunol. 1995;7:1005–1011. doi: 10.1093/intimm/7.6.1005. [DOI] [PubMed] [Google Scholar]
- 28.Langhorne J. γδ T cells in malaria infections. Parasitol Today. 1996;12:200–203. doi: 10.1016/0169-4758(96)10009-0. [DOI] [PubMed] [Google Scholar]
- 29.Modlin R L, Pirmez C, Hofman F M, Torigian V, Uyemura K, Rea T H, Bloom B R, Brenner M B. Lymphocytes bearing antigen-specific γδ T cell receptors accumulate in human infectious disease lesions. Nature. 1989;339:544–548. doi: 10.1038/339544a0. [DOI] [PubMed] [Google Scholar]
- 30.Morita C T, Tanaka Y, Bloom B R, Brenner M B. Direct presentation of non-peptide prenyl pyrophosphate antigens to human γδ T cells. Res Immunol. 1996;147:347–353. doi: 10.1016/0923-2494(96)89649-0. [DOI] [PubMed] [Google Scholar]
- 31.Nagasawa H, Hisaeda H, Maekawa Y, Fujioka H, Ito Y, Aikawa M, Himeno K. γδ T cells play a crucial role in the expression of 65 000 MW heat-shock protein in mice immunized with Toxoplasma antigen. Immunology. 1994;83:347–352. [PMC free article] [PubMed] [Google Scholar]
- 32.Nakazawa S, Brown A E, Maeno Y, Smith C D, Aikawa M. Malaria-induced increase of splenic γδ T cells in humans, monkeys, and mice. Exp Parasitol. 1994;79:391–398. doi: 10.1006/expr.1994.1101. [DOI] [PubMed] [Google Scholar]
- 33.O’Brien R L, Fu Y-X, Cranfill R, Dallas A, Ellis C, Reardon C, Lang J, Carding S R, Kubo R, Born W. Heat shock protein Hsp60-reactive γδ cells: a large, diversified T-lymphocyte subset with highly focused specificity. Proc Natl Acad Sci USA. 1992;89:4348–4352. doi: 10.1073/pnas.89.10.4348. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Perera M R, Carter R, Goonewardene R, Mendis K N. Transient increase in circulating γδ T cells during Plasmodium vivax malarial paroxysms. J Exp Med. 1993;179:311–315. doi: 10.1084/jem.179.1.311. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Ploton I N, Wizel B, Viscidi R, Kumar N. Mapping of two overlapping linear epitopes in Pfg27 recognized by Plasmodium falciparum transmission-blocking monoclonal antibodies. Vaccine. 1995;13:1161–1169. doi: 10.1016/0264-410x(95)00033-w. [DOI] [PubMed] [Google Scholar]
- 36.Poquet Y, Halary F, Champagne E, Davodeau F, Gougeon M L, Bonneville M, Fournie J J. Human γδ T cells in tuberculosis. Res Immunol. 1996;147:542–549. doi: 10.1016/s0923-2494(97)85220-0. [DOI] [PubMed] [Google Scholar]
- 37.Riley E, MacLennan C, Wiatkowski D K, Greenwood B M. Suppression of in vitro lymphoproliferative response of acute malaria patients can be partially reversed by indomethacin. Parasite Immunol. 1989;11:509–517. doi: 10.1111/j.1365-3024.1989.tb00685.x. [DOI] [PubMed] [Google Scholar]
- 38.Rzepczyk C M, Anderson K, Stamatiou S, Townsend E, Allworth A, McCormack J, Whitby M. γδ T cells: their immunobiology and role in malaria infections. Int J Parasitol. 1997;27:191–200. doi: 10.1016/s0020-7519(96)00149-x. [DOI] [PubMed] [Google Scholar]
- 39.Sandor M, Sperling A I, Cook G A, Weinstock J V, Lych R G, Bluestone J A. Two waves of γδ T cells expressing different V δ genes are recruited into schistosome-induced liver granulomas. J Immunol. 1995;155:275–284. [PubMed] [Google Scholar]
- 40.Tsuji M, Mombaerts P, Lefrancois L, Nussenzweig R S, Zavala F, Tonegawa S. γδ T cells contribute to immunity against the liver stages of malaria in αβ T-cell-deficient mice. Proc Natl Acad Sci USA. 1994;91:345–349. doi: 10.1073/pnas.91.1.345. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.van der Heyde H C, Elloso M M, Roopenian D C, Manning D D, Weidanz W P. Expansion of the CD4−, CD8− γδ T cell subset in the spleens of mice during non-lethal blood-stage malaria. Eur J Immunol. 1993;23:1846–1850. doi: 10.1002/eji.1830230817. [DOI] [PubMed] [Google Scholar]
- 42.van der Heyde H C, Manning D D, Weidanz W P. Role of CD4+ T cells in the expansion of the CD4−, CD8− γδ T cell subset in the spleens of mice during blood-stage malaria. J Immunol. 1993;151:6311–6317. [PubMed] [Google Scholar]
- 43.van der Heyde H C, Elloso M M, Chang W-L, Kaplan M, Manning D D, Weidanz W P. γδ T-cells function in cell-mediated immunity to acute blood-stage Plasmodium chabaudi adami malaria. J Immunol. 1995;154:3985–3990. [PubMed] [Google Scholar]
- 44.van der Heyde H C, Chang W-L, Weidanz W P. Specific immunity to malaria and the pathogenesis of disease. In: Kaufmann S H E, editor. Host response to intracellular pathogens. New York, N.Y: Chapman & Hall; 1997. pp. 195–226. [Google Scholar]
- 45.Weiss L. The spleen in malaria: the role of barrier cells. Immunol Lett. 1990;25:165–172. doi: 10.1016/0165-2478(90)90109-4. [DOI] [PubMed] [Google Scholar]


