Abstract
There are several bacteria called superbugs that are resistant to multiple antibiotics which can be life threatening specially for critically ill and hospitalized patients. This article provides up-to-date treatment strategies employed against some major superbugs, like methicillin-resistant Staphylococcus aureus, carbapenem-resistant Enterobacteriaceae, vancomycin-resistant Enterococcus, multidrug-resistant Pseudomonas aeruginosa, and multidrug-resistant Escherichia coli. The pathogen-directed therapeutics decrease the toxicity of bacteria by altering their virulence factors by specific processes. On the other hand, the host-directed therapeutics limits these superbugs by modulating immune cells, enhancing host cell functions, and modifying disease pathology. Several new antibiotics against the global priority superbugs are coming to the market or are in the clinical development phase. Medicinal plants possessing potent secondary metabolites can play a key role in the treatment against these superbugs. Nanotechnology has also emerged as a promising option for combatting them. There is urgent need to continuously figure out the best possible treatment strategy against these superbugs as resistance can also be developed against the new and upcoming antibiotics in future. Rational use of antibiotics and maintenance of proper hygiene must be practiced among patients.
Introduction
Antimicrobial resistance is the capability of a microorganism to resist the action of the different antimicrobials. In this type of resistance, microbes can resist the medication that could once be successful against them [1]. When this resistance occurs to multiple drugs, it is known as multidrug resistance (MDR). There are different types of resistance mechanisms observed in microbes, like natural resistance in certain microbes against a particular antimicrobial, genetic mutation, or acquired resistance from other species [2].
Globally, the drug resistance is increasing due to indiscriminate use of antimicrobial agents. Resistance microbes are hard to treat, requiring alternative or higher doses of antimicrobials or lack/shortage of effective antimicrobials, adversely affecting countries at all levels of development. As per the statement of World Health Organization (WHO), MDR pathogens called ‘superbugs’ are one of the major public threats that yearly cause several million deaths globally [3]. In 2021, WHO published the list of antibiotic-resistant pathogens (priority pathogens), especially highlighting the resistant gram-negative bacteria that pose maximum threat to human health [4]. On the basis of urgency for new antibiotics, the list is categorized into three headings, mainly critical, high, and medium priority. The critical group of MDR bacteria includes Pseudomonas aeruginosa, Acinetobacter baumannii, and Enterobacteriaceae, which cause severe infections like pneumonia and blood stream infections in hospital-admitted patients. The high and medium priority group include drug-resistant bacteria like Salmonella that causes common diseases, such as gonorrhoea and food poisoning.
Many review articles have been published about multidrug-resistant bacteria and treatment strategies employed to combat them. However, most of these articles focus on a particular resistant bacteria or a specific line of treatment [5–9]. Moreover, in the continuous fight against emerging MDR bacterial species, knowledge updating is highly recommended for researchers and medical professionals. This article attempts to provide up-to-date treatment strategies employed against different antibiotic-resistant bacteria currently responsible for causing severe infections, mostly in hospital-admitted patients. Our review will focus on the innovative treatment strategies and promising new chemicals currently in the pipeline against multidrug-resistant organisms, such as combinatorial drug approaches, polymeric biomaterials, nanoparticles, and plant-derived products.
Mechanisms of Bacterial Resistance
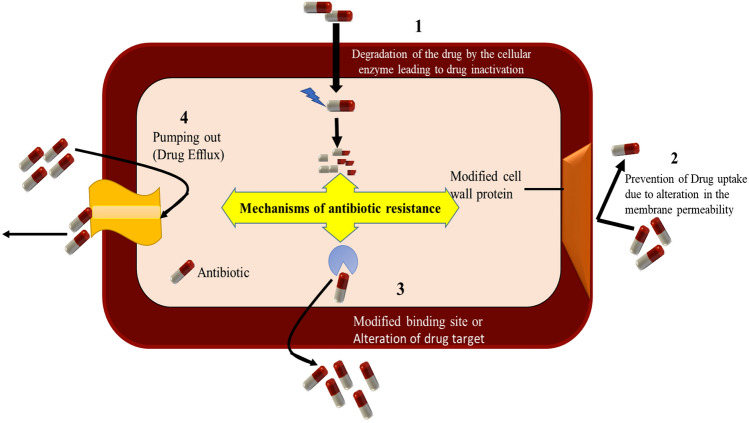
Various bacteria exhibit antibiotics resistance by mainly four mechanisms (Fig. 1). In the drug inactivation process, the antibiotics are deactivated by the protective enzymes of the bacteria cell wall such as Penicillin G inactivation by β-lactamase enzyme released from the penicillin-resistant bacteria [10]. On the other hand, protective protein bound to the ribosomes of the bacterial cell inactivates the antibiotics via ‘target alteration’ by altering their structural confirmation. This leads to the inhibition of their protein synthesis. Such type of resistance can be seen in methicillin-resistant Staphylococcus aureus (MRSA), where protective proteins bound with the ribosomes of the bacterial cell inactivate antibiotics. This prevents antibiotic binding to the ribosome and inhibits bacterial protein synthesis [11].
Fig. 1.
Mechanisms of antimicrobial resistance. (1) Drug inactivation by bacterial enzyme; (2) drug uptake reduction by lowering of bacterial cell permeability; (3) structural modification of drug target by gene mutation; and (4) drug efflux outside the bacterial cell membrane
In metabolic pathway alteration, the bacteria usually modify or protect the target site and restrict the binding of antibiotics to that site, which ultimately decreases the affinity of antibiotic molecules. Bacteria synthesize essential folic acid and nucleic acid from the para-amino benzoic acid (PABA) precursor [12]. However, when an antibiotic targets this pathway, the bacteria adapt themselves using preformed folic acid. This restricts the efficacy of antibiotics like sulphonamide that block PABA pathway, hence inhibiting bacterial growth. Bacteria also use a drug efflux mechanism to increase the expression of active efflux pumps or decrease the drug permeability from the cell surface, which results in low-level susceptibility to antibiotics. Certain bacteria have shown this type of resistance against fluoroquinolones prior to drug penetration by the inbuilt pump present within the bacterial cellular membrane [13].
Gram-negative bacteria use all the above four mechanisms of drug resistance, whereas gram-positive bacteria lack drug efflux mechanism due to the absence of lipopolysaccharides in their outer membrane. This suggests the structure of the outer membrane of bacteria is the key to drug resistance to different types of antibiotics [14]. Any changes in the structure of the outer membrane or mutations in porin channel or increase in hydrophobic properties leads to the development of bacterial drug resistance. The major resistant gram-negative bacteria include Enterobacteriaceae and P. aeruginosa, which can cause serious infections in catheter- and ventilator-related pneumonia and complicated urinary tract infections (cUTI). P. aeruginosa mostly shows resistance due to over-expression of efflux pumps or low membrane permeability or mutations of porin-associated genes, resulting in ventilator-linked pneumonia and blood stream infections [15]. Likewise, Pseudomonas resistance to β-lactam antibiotics was mediated through inactivation of amide bond by β-lactamases enzymes [16]. Similarly, different classes of modifying enzymes like phosphoryltransferase, adenylyltransferase, and acetyltransferase deactivate the aminoglycosides of antibiotics by hindering their binding affinity toward bacterial outer cell membrane. Some gram-negative bacteria also produce plasmid-mediated genes which selectively bind with DNA gyrase or topoisomerase IV, thereby protecting them from the quinolones, like antibiotics. In addition, genetic mutation and gene transfer can also contribute to drug resistance. Enterobacteriaceae resistant to carbapenem and Enterococcus resistant to vancomycin antibiotics come under this category. Major resistance mechanisms against some common antibiotic classes are provided in Table 1.
Table 1.
Drug resistance mechanisms against some commonly used antibiotics
| Antibiotic class | Examples | Target | Modes of resistance |
|---|---|---|---|
| β-Lactams | Penicillin (ampicillin) Cephalosporins, Penem | Peptidoglycan biosynthesis | Hydrolysis, efflux, altered target |
| Aminoglycoside | Gentamycin, Streptomycin | Translation | Phosphorylation, acetylation, efflux, altered target |
| Glycopeptides | Vancomycin, Teicoplanin |
Peptidoglycan Biosynthesis |
Reprogramming Peptidoglycan biosynthesis |
| Tetracyclines | Minocycline, tigecycline | Translation | Mono-oxygenation, altered target |
| Macrolides | Erythromycin, azithromycin | Translation | Hydrolysis, efflux glycosylation, Phosphorylation |
| Phenicols | Chloramphenicol | Translation | Acetylation, efflux |
| Quinolones | Ciprofloxacin | DNA replication | Acetylation, altered target |
| Sulfonamides | Sulfamethoxazole | C1 metabolism | Efflux, altered target |
| Rifamycins | Rifampin | Transcription | ADP-ribosylation, efflux |
Currently, there are mainly two types of therapeutic option to combat superbugs, like pathogen-directed therapeutics (Table 2) and host-directed therapeutics (Table 3). Pathogen-directed therapeutics decrease the toxicity of bacteria by altering their virulence factors. The host-directed therapeutics limit infectious diseases by modulating immune cells, enhancing host cell functions, and modifying disease pathology.
Table 2.
Pathogen-directed therapeutic strategy to combat superbug
| Pathogen-directed therapeutics strategy | Characteristics |
|---|---|
| Neutralization of virulence factors |
• One of the main cause of bacterial infection is the presence of virulence factors in the bacterial surface morphology • This factor helps bacteria in adherence to epithelial mucosal membrane, formation of biofilm, and intracellular invasion of host cells • Therapies that target neutralization of the bacterial virulence factor at genetic level can neutralize its harmful effect such as genetic engineering technique can significantly reduce shiga toxin production in vitro enteric mouse infection model |
| Blockage of epithelial adherence or Biofilm formation |
• The small drug molecules with effective anti-adherence characteristics may help in resisting the bacterial infections and serve as alternative treatment option against these superbugs • In case of Pseudomonas aeruginosa, adhesion-mediated binding of mucosal pathogens with epithelial cell receptor in host organ and their mucosal binding to form a polymeric matrix or biofilm formation is considered as the initiation step of disease pathogenesis • Reduction of biofilm formation and enhancing its elimination could also be a helpful strategy in minimizing host toxicity |
| Monoclonal antibody (mAb) |
• Immunotherapy via monoclonal antibodies are intended for binding to the surface of target site of the specific pathogen or antigen having virulence factors • These antibodies help in the clearance of phagocytic part of microorganism and thus eliminate their pathogenic effect |
| Strategies for toxin neutralization |
• Bacteria toxin can be neutralized either by physical sequestration technique or altering the pathogen binding affinity toward host cell receptor • Pharmacological agents can be prepared that mimic the structural features of the cell membrane of host. This mechanism can sequester the toxins away from the target host cell |
| Reducing bacterial virulence factor changing gene expression |
• Virstatin is a potent virulence inhibition agent active against Vibrio cholera that changes the gene expression by downregulating the dimerization of transcriptional activator • Further, it prevents the biogenesis of pilus, motility, and formation of biofilm in resistant pathogens • Regacin is another similar agent that also inhibits the pathogenic virulence factor by changing its gene expression |
| Interference with bacterial quorum sensing |
• Quorum sensing (QS) is a process in which bacterial species released auto inducers to increase its concentration as a function of cell density • Many resistant bacteria species regulate gene expression within host tissue through quorum sensing • Recent studies found emergence of resistance against tobramycin used against Pseudomonas aeruginosa infections; however, the drugs like 6-gingerol (derived from ginger) is effective in such cases by competitively inhibiting autoinducers of resistant bacteria or inhibits QS. The combination of 6-gingerol derivative and tobramycin provides more effective inhibition of virulence factor of P. aeruginosa by inhibiting QS as well as biofilm production in comparison to single therapy. The anti-infective activity of this combination was further confirmed in an insect model using Tenebrio larvae. The results indicated strong inhibitory activity at very low dose of adjuvant and suggests combined therapy might act as effective strategy for the management of P. aeruginosa infections |
| Sensitization of the pathogen toward host innate immune clearance |
• This can be a beneficial approach in treating the patient suffering resistant bacterial infections • When the multidrug-resistant pathogens cause chronic infection to an individual, it is important to identify the gene that is withdrawn by the resistant bacteria leading to its virulence • Thus, re-sensitization of the said gene can be helpful in initiating its immune destruction |
Table 3.
Host-directed therapeutic strategy to combat superbug
| Host-directed therapeutics strategy | Characteristics |
|---|---|
| Improving bactericidal activity of phagocytic cells |
• Severe bacterial infections weaken immune function due to the regular use of medicine for the treatment of inflammatory disorder that supress innate immunity • Pharmacologically improving bactericidal activity of phagocytic cell could therefore be effective against superbugs |
| Manipulating cytokines and chemokine | • Cytokine or chemokine are the endogenous pro-immune regulating substances responsible for clearance of resistant pathogen. They have been also used as a new treatment strategy to fight superbugs |
| Lipid mediators of innate immunity | • It is reported that macrophages when treated with leukotriene B4 (LTB4) induce production of reactive oxygen species through NADPH oxidase system to fight superbugs |
| Targeting immune regulatory pathways in phagocytes | • Host-directed innate immune boosting approach has been studied in knockout mice and immune modulatory peptides by altering the susceptibility of resistant pathogens |
| Enhancing Toll-like receptor signaling pathways | • Toll-like receptors play an important role in recognizing pathogen-associated microbial patterns to initiate signaling cascade that promote the regulation of innate immune response genes. This results in increase in the production of cytokines leading to reduction in bacterial burdens |
| Drugs having unanticipated immune boosting effects |
• Some drugs can boost immune response that results in elimination of bacterial pathogen • For example, Tamoxifen-boosted immune system produce beneficial effect against systemic MRSA infection • A randomized trial reports that statin drugs, used for reduction of cholesterol through specific mechanism, also help to reduce the infectious disease like pneumonia, sepsis-associated comorbidities, and mortality rate in patients with bacterial resistance |
Treatment Strategies Against Critical Superbugs
The recent advancement in the treatment of antimicrobial resistance applies various options, such as combinatorial drug approaches, antimicrobial polymeric biomaterials products, bio-nanotechnology approaches, and many others. The most effective treatment strategies currently adopted against various critical superbugs like methicillin-resistant S. aureus (MRSA), carbapenem-resistant Enterobacteriaceae (CRE), vancomycin-resistant Enterococcus (VRE), multidrug-resistant P. aeruginosa (MRPA), and multidrug-resistant E. coli (MREC) along with their probable mechanisms are summarized in Table 4.
Table 4.
Current treatment strategies against major superbugs
| Superbug | Mechanism of resistance | Drug of choice | Mechanism of drug action |
|---|---|---|---|
| Methicillin-resistant Staphylococcus aureus (MRSA) | Production of Penicillin-Binding Protein (PBP2a) | Vancomycin | Inhibits cell wall synthesis by binding to the D-Ala-D-Ala terminal of the growing peptide chain [45] |
| Carbapenem-resistant Enterobacteriaceae (CRE) | β-Lactamase activity and production of carbapenemases | Polymyxin, Tigecycline, and Aminoglycosides | Disrupt the membrane by binding to lipopolysaccharide. Inhibition of protein synthesis [49] |
| Vancomycin-resistant Enterococcus (VRE) | Production of low-affinity pentapeptide precursor of cell wall | Daptomycin | Disrupting the membrane potential [51] |
| Multidrug-resistant Pseudomonas aeruginosa | Genetic alterations | Ceftazidime–avibactam and Ceftolozane–tazobactam | Inhibits penicillin-binding proteins and also inhibits many serine β-lactamases. Inhibition of cell wall biosynthesis [61] |
| Multidrug-resistant Escherichia coli | Presence of resistance genes | Mecillinam | Inhibits penicillin-binding protein 2 [65] |
Methicillin-Resistant S. aureus
MRSA is considered as a major cause of community and healthcare-associated infections and is highly resistant to different classes of antibiotics. The possible mechanism of such resistance is the expression of a unique transpeptidase protein penicillin-binding protein (PBP2a) that causes cross-linking among the peptidoglycan polymers. A study has shown that drug-resistant S. aureus acquired mecA genetic factor that exhibited phenotypic expression or mutation of pathogens. This resulted in its resistance to β-lactam antibiotics like methicillin to prevent bactericidal effect of these antibiotics [17].
Polyphenolic antioxidant ethyl gallate in combination with tetracycline or fusidic acid act as synergistic herb-drug combination to delay the development of MRSA resistance. The probable mechanism of such action is the changes in the mutant selection window [18].
Carbapenem-Resistant Enterobacteriaceae
The resistance of CRE toward carbapenems is generally due to its β-lactamase activity in combination with structural mutations to produce carbapenemases enzymes which hydrolyze carbapenem antibiotics. Ursolic acid, a drug prescribed in nosocomial infections caused by Enterobacter cloacae, can be a good choice against CRE. It probably acts by inhibition of biofilm formation and disturbance of integrity of bacterial cell membrane [19].
Certain drug combinations have also been successfully applied against CRE. For example, combination of meropenem and amikacin was introduced to increase the efficacy against CRE. Combination of meropenem and polymyxin was also studied against CRE [20]. The outcome indicated that polymixins should not be administered first in this combination therapy. It was further concluded that isolated treatment with each antimicrobial has proven to be less effective compared to the combination therapy against CRE pathogens.
Vancomycin-Resistant Enterococcus
Enterococcus possess antimicrobial resistance either by the acquired or intrinsic means to transfer resistance genes. Vancomycin resistance generally occurs when low-affinity pentapeptide precursor of the cell wall are formed in place of its high affinity variant [21]. A very limited number of treatment options are available against VRE. In a study, linezolid and bactericidal drug daptomycin were only found to produce in vivo bactericidal effects against it [22]. These antibiotics significantly decreased the mortality rate of the patient. However, the delay in initiating the treatment might result in increase in the mortality rate in patients with severe infections. Therefore, novel strategies are needed to control VRE. Among promising results, the UV-stable bacteriocin like inhibitory substance (BLIS) produced by Enterococcus faecium strain has shown potent bactericidal activity against VRE [23]. A glycopeptide antibiotic named LY333328 has also shown bacteriostatic effect when applied in combination with ampicillin, quinupristin–dalfopristin and gentamicin. Additionally, LY333328, a semisynthetic N-alkyl derivative of LY264826, was found to exert the most potent bactericidal effect against it.
Multidrug-Resistant P. aeruginosa
The resistance of P. aeruginosa pathogen is mainly due to genetic alterations, antibiotic efflux system, or mutation of topoisomerases II/ topoisomerases IV, especially in fluoroquinolone antimicrobial compounds [24]. Derepression of the chromosomal AmpC β-lactamase reduces susceptibility to β-lactam antibiotics like penicillin and cephalosporin against MRPA [25]. Various approaches have been successful in contradicting MRPA. Lysozyme enzymes like Lysins and endolysins are found active against MRPA by disturbing the peptidoglycan structure of the outer membrane of the bacteria [26]. In an experiment, combination of third-generation cephalosporin (ceftolozane) with β-lactamase inhibitor (tazobactam) was found to exhibit low MIC against the P. aeruginosa in ETEST® strips, suggesting its prospective antimicrobial activity against MRPA.
Multidrug-Resistant E. coli
E. coli is a common pathogen associated with serious nosocomial infections, like urinary tract infections (UTI). The emergence of multidrug resistance in E. coli is due to the development of inherent R-plasmid genes in them. This results in failure of the antimicrobial agents that act via dihydropteroate synthase inhibition [27]. Pivmecillinam, a prodrug of mecillinam has proven efficacy against acute lower UTI. It exerts activity against MREC by inhibition of Penicillin-Binding Protein (PBP2a) [28]. Lytic bacteriophage has also emerged as an alternative strategy against resistant bacteria, but its effectiveness relies on appropriate genomic characterization. phiLLS, a unique bacteriophage from the Siphoviridae family, has shown significant lytics properties against resistant E. coli strains. Recently, probiotics have gained interest in combating multidrug-resistant E. coli. In a research, six E. coli strains which are resistant to antibiotics like Ampicillin, Clarithromycin, Amoxicillin–Clavulanic acid, and ceftriaxone, when treated with probiotics have shown effective antimicrobial activities by preventing biofilm formation [29]. The probiotic lactic acid bacteria Lactobacillus plantarum Y3 (LPY3) obtained from fermented dairy product inhibited biofilm production of multidrug-resistant E. coli U12 pathogens. Scanning electron microscope indicated the adherence rate of LPY3 is only 14.5% at urinary catheter, i.e., reduced up to four times.
Drugs Beyond Antibiotics
Natural Products
Recently, secondary plant metabolites have been used for the management of MDR pathogens. A study reported that the secondary plant metabolites like geranial, neral, 1,8-cineole, camphene, β-phellandrene, and α-curcumene obtained from the essential oil of Zingiber officinale have significant antibacterial properties against 18 resistant pathogens with high efficacy against gram-negative MDR pathogens, like carbapenem- and polymyxin-resistant ten Klebsiella pneumoniae and Serratia marcescens [30]. In a carbapenem-resistant K. pneumonia-induced mice sepsis model, researchers exhibited a reduction of bacterial load and increase in survival time of infected animals without any toxicity to the host. This efficacy might be due to the presence of major secondary metabolites, like geranial, neral, and 1,8-cineole [30].
Another study reported the antibacterial efficacy of secondary metabolites (lariciresinol and berberine) of Zingiber officinale Roscoe of the North-Western Himalaya region against MDR Salmonella typhimurium by efflux pump inhibitory action. Results showed that berberine has increased antibacterial activity by 2 to 4 folds, whereas lariciresinol decreased the MIC by 2 to 4 folds. It was concluded that lariciresinol could be a potential efflux pump inhibitor in killing of drug-resistant S. typhimurium [31].
Plant derived secondary metabolites like saponins and bromo-polyphenols obtained from Cassia fistula bark were found effective against MDR pathogens (E. coli and M. tuberculosis) isolated from Ganga river water, milk, and chicken meat. The inhibition of bacterial RNA polymerase was much higher as compared to the standard rifampicin antibiotics. It was concluded that these phytoconstituents could be used in future as therapeutic adjunct in the prevention and treatment of MDR bacterial infections [32].
The flavonoids and polyphenol obtained from aerial parts of the plant Vernonia auriculifera Hiern showed strong antibacterial action against the E. coli, E. aerogenes, K. pneumoniae, P. aeruginosa, and S. aureus with a low MIC value. The antibacterial efficacy was further enhanced in combination therapy with phenylalanine β-naphthylamide (efflux pump inhibitor). It was inferred that these phytoconstituents have a significant bactericidal effect against MDR pathogens by overexpressing efflux pumps [33].
Rolta et al. evaluated bergenin isolated from the rhizome of Bergenia ligulata against MDR E. coli and S. aureus. The high antimicrobial activity was observed against both the bacteria, suggesting that the secondary metabolite of B. ligulata could be utilized in the treatment of drug-resistant bacteria [34].
Pleuromutilin and its derivatives such as valnemulin, tiamulin, azamulin, and retapamulin have also shown significant antibacterial agents against MRSA. The probable mechanism might be the inhibition of protein synthesis by binding with peptidyl transferase constituents of 50S-subunit of ribosomes of bacteria [35]. Phloroglucinol and its acylphloroglucinol derivatives have shown promising anti-MRSA action. They prevent such drug resistance by disturbing bacterial inner membrane permeability [36]. Indole-based natural compound epidithiodioxopiperazine have also demonstrated significant bactericidal action against MRSA [37]. Additionally, the ketone derivative of flavonoids like isoorientin, luteolin, quercetin, 3-o-methylquercetin, and rutin are found to be effective against MRSA. Natural guanidine alkaloids, tetrandrine, and demethyltetrandrine in combination with ampicillin, 8-acetoxycycloberberine, 8-substituted cycloberberines, xanthone derivatives, and polyphenolic compounds like chalcones extracted from the Desmodium congestum have also shown significant anti-MRSA activity [38]. Similarly, bacterial-derived secondary metabolite teixobactin exerts dose-dependent antibacterial action against MRSA due to selective replacement of particular amino acid sequence of resistance pathogens. Muhsinah et al. have reported that the natural flavor obtained from the plant Illicium verum possesses significant antibacterial action against MRSA clinical isolates [39].
Some phytomedicines have been found effective against CRE infection. An essential oil obtained from the seeds of Camellia japonica exhibited significant antibacterial action against CRE. Similarly, the combination of natural compounds curcumin and chitosan showed significant antibacterial and anti-biofilm activities against CRE isolates [40].
The anti-VRE activity of the hydroxyalkenyl salicylic acids, active chemical constituents of Ginkgo biloba, exhibited low MIC against VRE stains [41]. The antibacterial activity of several plant extracts like ethanolic extracts of Ageratum conyzoides, Phyllanthus emblica, Camellia sinensis, and Mentha longoifolia were evaluated against clinically isolated VRE stain by agar well diffusion method. The Mentha longifolia extract showed greatest zone of inhibition against VRE, indicating it to be the more potent antimicrobial agent against VRE compared to the other plant extracts [42]. In another study, the phytocompounds butyryl alkannin obtained from root extract of Arnebia euchroma showed superior antibacterial properties compared to the vancomycin antibiotics, providing scientific validation for the traditional use of these medicinal plants against VRE [43].
Phytochemicals–antibiotics combination have also been tried against resistant MRPA. The promising results were observed by combining the extracts of Rhus coriaria seed, Sarcopoterium spinosum seed, and Rosa damascene flower with selected antimicrobial agents, like oxytetracycline, sulfadimethoxine, and cephalexin [44]. Medicinal plants like Terminalia bellerica, Celastrus paniculatus, Kingiodendron pinnatum, Schleichera oleosa, Melastoma malabathricum, and Garcinia gummi-gutta have also been investigated against MRPA [45]. In that study, the extract of Terminalia bellerica was found most effective by significant reduction of quorum sensing regulated virulence factors and biofilm formation.
Some nutritionally enriched herbs have also been found to have significant role in the treatment of MRPA. Glycyrrhiza glabra, a natural flavoring agent, showed promising bactericidal action against resistant strains of P. aeruginosa in comparison to standard antibiotic amikacin. This bactericidal activity is attributed to the generation of quinones and superoxide ions due to the presence of bioactive phenolic compounds in the plant [46].
Researchers have investigated the antimicrobial activity of Acacia nilotica extract against E. coli on the basis of cell membrane integrity, permeability, and kill time. Experimental findings showed that it exerted bactericidal activity by damaging the cellular membrane of the bacteria [47]. Study reports have shown that aqueous and lime extracts of natural spices like garlic, onion, coriander, pepper, and ginger is also having potent in vitro antimicrobial activity against MREC strains [48].
Phytochemicals like flavonoids have been used to prevent the formation of biofilm of seropathotype E. coli O157:H7 (STEC) by inhibiting the expression of locomotive surface assembly fimbriae. Flavonoid phloretin obtained from apple showed significant inhibition of biofilm of STEC at concentration of 50 μg/ml, confirmed by both genomic qRT-PCR analysis as well as electron microscope studies [49]. Similarly, Ginkgolic acid obtained from Ginkgo biloba and naringenin present in grape seed exhibited significant inhibition of STEC biofilm by the downregulation of curli gene. The polyphenol trans-resveratrol obtained from grape seeds also exhibited anti-biofilm of STEC effect by similar downregulating of curli gene expression. Transcriptional assay indicates trans-resveratrol at the dose of 10 μg/ml prevented the adherence of E. coli cell onto human epithelial tissue [49].
Nanoparticles (NPs)
Nanotechnology has emerged as a promising option for combatting MDR bacteria. Various treatment strategies include amplifying drug affinity toward the bacterial cell membrane utilizing the protective nature of NPs against bacterial enzyme, enhanced tissular diffusion, larger surface to volume ratio and high drug loading capacity. Antibiotics conjugated with NPs in the recommendable size range have the potential to facilitate the drug binding more effectively at the desirable site.
Among their different variants, metal and metal oxide-based NPs exert more antibacterial activity [50]. The antibacterial activity exerted by these NPs is supposed to be due to the exertion of oxidative stress by reactive oxygen species (ROS), restriction in biofilm production, direct contact with bacterial cell wall, triggering of host immune response, and major interaction with DNA fragments and protein molecules [51].
The potential bioactivity and antibacterial properties of selenium nanoparticles (SeNP) provide a unique platform in the treatment of resistant bacterial infection. Further, the antimicrobial action of SeNPs is enhanced when synthesized from the extract of different plants. A recent article explored the antimicrobial activity of synthesized SeNP (245–321 nm) using extract of Allium cepa, Malpighia emarginata, and Gymnanthemum amygdalinum against gram-positive resistant pathogens, like Streptococcus agalactiae, S. aureus, and MRSA. It suggested that SeNP could be a promising option in the treatment of MDR bacteria with high efficacy along with low cytotoxicity in red blood cell [52].
Another noted work studied the effect of ZnO NPs (330–350 nm), prepared from ripe fruits and leave of Limonia acidissima, against resistant UTI pathogens, such as S. paratyphi, Shigella, Streptococcus, Staphylococcus, and K. pneumonia. The result of in vitro well diffusion antibacterial test and biofilm tube ring formation study reveals significant activities against MDR bacteria as well as inhibition of bacterial biofilm formation. The study suggested that the plant-derived ZnO NPs have strong antibacterial properties and could be utilized for the management of resistant bacterial infections [53].
Arshad et al. investigated the effect of Aloe vera-conjugated AgNP (30-80 nm) against resistant gram-positive strains (S. aureus) and Gram-negative strains (E. coli, A. baumannii, and P. pseudomonas) using the Kirby–Bauer agar disc diffusion method. The result indicates promising antibacterial activity against Gram-negative bacteria with maximum MBC confirmed for S. aureus (200 µg/ml), while the minimum MBC was observed for Candida albicans (50 µg/ml). The possible mechanism of antibacterial action of conjugated NP is either due to penetration of NP into the negatively charged cell membrane of resistant pathogens or generation of more ROS. This could lead to inhibition of DNA replication, resulting in the impairment of bacterial cell growth and cell death [54].
The green-synthesized mono-dispersed silver nanoparticles (AgNP) in the size range of 25–50 nm showed potent antibacterial activity against highly resistant MRSA strains [55]. A recent study on chitosan nanoparticles for delivery of a newly discovered antibiotic N′-((5-nitrofuran-2-yl) methylene)-2-benzhydrazide (5-NFB) against MRSA. The study revealed that the drug-loaded NPs (44% encapsulation efficiency) had enhanced antibacterial activity against all S. aureus strains and exerted a great synergistic effect between chitosan nanoparticles and 5-NFB [56].
Vancomycin is the drug of choice against MRSA which acts by inhibiting cell wall synthesis by blocking transpeptidase and thus prevents cross-linking of peptidoglycan. However, after 48 h of administration, free vancomycin starts to lose its antimicrobial potency. Researchers used Hyaluronic acid (HA) with oleylamine (OLA) conjugate (HA-OLA) as nano-drug carrier for Vancomycin. The drug and HA-OLA conjugate showed intrinsic and long-lasting antibacterial properties and sustains drug release up to 72 h with MIC 500 µg/ml [22].
The possible antibacterial action of silver nanoparticle (AgNP) against P. aeruginosa is due to its high penetrability into bacterial membranes, which disrupt the biofilm formation as well as inhibit the bacterial DNA synthesis. Brown et al. have reported that the AgNP-loaded ampicillin at a dose of 4 μg/ml completely killed the ampicillin-resistant P. aeruginosa pathogens at dose of 4 μg/ml [57]. In another investigation by Wahab et al., the antibacterial activity of AgNP synthesized from leaf extract of Mukia scabrella with zeta potential −21.7 mV exhibited promising anti-MRPA activity. It was suggested that the plant extract in AgNP interacted strongly with the negative charge cell membrane of bacteria, leading to a strong antibacterial action [58].
Khalil M. et.al, studied the efficacy of some antibiotics conjugated silver nanoparticles (AgNP) against MRPA. They observed the synergistic effect of neomycin and AgNPs against MDR P. aeruginosa P8 and P14 isolates. The study suggested that AgNP in combination with antibiotics would be effective against MRPA [59].
Gabrielyan et al. reported that the iron oxide nanoparticles (Fe3O4NPs) with unique superparamagnetic properties, greater stability, nontoxicity, and high magnetic susceptibility exhibited strong antibacterial effect on the growth mechanism and membrane activity of E.coli strains. Additionally, they reported that the Fe3O4NPs could successfully reduce the growth rate of bacteria by increasing the lag phase duration. Combination of nanoparticles with antibiotics provided a synergistic effect to inhibit the H2 yield and H+ fluxes, suggesting that the iron oxide nanoparticles could be used to combat bacterial multidrug resistance [60]. Another study confirmed that the silver nanoparticles (AgNPs) synthesized from Arisaema flavum tuber extract having a size range of 12–20 nm had shown significant antibacterial activity against MREC QH4 strains [61].
Memar et al. reported that the Meropenem-loaded mesoporous silica nanoparticles (MSNs) had much lower MIC against CRE than meropenem alone due to the efficient fusion of bacteria cell walls. Additionally, MSNs showed enhanced physicochemical and biocompatibility characteristics with low in vitro cytotoxic activity. They concluded that MSNs could become a promising drug against CRE [62]. All these reports suggest that plant-derived nanoparticles could be a cost-effective and eco-friendly alternative in controlling bacterial MDR.
Approved/Pipeline Drugs Against Critical Superbugs
WHO’s global priority pathogen list has provided a vital tool in finding out newer antibiotics viable against these superbugs. In this context, 32 antibiotics, notified by WHO, have been in the clinical development phase [63]. Currently, there are nearly 43 new antibiotics in clinical development by the top 50 pharmaceutical companies with significant antibacterial properties against serious or life-threatening bacterial infections (Table 4) [63]. Among these antibiotics, 18 have shown potential antimicrobial action against gram-negative resistant pathogens, particularly Enterobacteriaceae and P. aeruginosa. Similarly, 10 antibiotics are used in the treatment of the resistant pathogen Neisseria gonorrhoeae. Some of these drugs are in the clinical pipeline and few are already approved by FDA. Cefiderocol and Eravacycline have recently been approved by FDA, respectively, for the treatment of complicated urinary tract infections (cUTI) and intra-abdominal infections caused by gram-negative microorganisms, including P. aeruginosa. These infections previously had limited treatment options. FDA has also stepped forward for to reduce the threat of AMR infections by approving three new antibiotics, namely oritavancin, dalbavancin, and tedizolid for the treatment of acute skin infections actively caused by methicillin resistant and susceptible S. aureus [64].
Patients affected with serious community-acquired pneumonia are very difficult to treat. To address the challenge, FDA has approved both oral and parenteral Xenleta to treat mild-to-severe lung illness of all age groups [65]. In addition, FDA also approved intravenous Recarbrio, a combination of imipenem–cilastatin and relebactam, for the treatment of various serious antimicrobial-resistant infections, including hospital and ventilator-acquired bacterial pneumonia (due to gram-negative bacteria), cUTI, and complicated intra-abdominal infections (cIBI) [66]. The administration of Recarbrio, tested for 28 days in a randomized controlled clinical trial of 535 hospitalized-acquired bacterial pneumonia patients, reported less percentage of death in comparison to piperacillin–tazobactam-treated patients [66]. Recently, the antibacterial drug Fetroja (Shionogi & Co.) got approval from FDA for the treatment of gram-negative bacteria-mediated cUTI in patients having limited treatment options due to hypersensitive history. The list of antibiotics currently in the phase of clinical development against various critical superbugs is summarized in Table 5 [67–85].
Table 5.
List of antibiotics in the clinical development against various critical superbugs
| Sl no. | Name of antibiotics | Trade name | Developer | Class | Phase of clinical trial | Application | References |
|---|---|---|---|---|---|---|---|
| 1 | Lascufloxacin | Lasvic (iv) | Kyorin Pharmaceutical | Fluoroquinolone | NDA | Lower RTI | [67] |
| 2 | Cefiderocol | Fetroja (iv) | Shionogi & Co., Ltd., Japan | Siderophore cephalosporins | Phase 3 | cUTI, CRE | [68] |
| 3 | Sulopenem etzadroxil/probenecid | Oral Sulopenem | Iterum Therapeutics | Penem | Phase 3 | cUTI | [68] |
| 4 | Durlobactam/sulbactam | SUL-DUR (iv) | Entasis Therapeutics | BL/BLI | Phase 3 | cUTI | [68] |
| 5 | Taniborbactam+cefepime | CERTAIN-1 (iv) | Venatorx’s | BL/BLI | Phase 3 | CRE | [68] |
| 6 | Enmetazobactam+cefepime | Exblifep | Allecra | BL/BLI | Phase 3 | cUTI | [68] |
| 7 | Zoliflodacin | ETX-0914 | Entasis |
Topoisomerase inhibitor (spiropyrimidinetrione) |
Phase 3 | Gonorrhoea infection | [68] |
| 8 | Gepotidacin | GSK2140944 | GSK |
Topoisomerase inhibitor (triazaacenaphthylene) |
Phase 3 | cUTI | [68] |
| 9 | Levonadifloxacin | EMROK O | Wockhardt | Fluoroquinolone | Phase 3 | MRSA | [69] |
| 10 | Cefilavancin | TD-1792 | Theravance and GSK |
Glycopeptide-cephalosporin conjugate |
Phase 3 | Acute bacterial skin and skin structure infections | [70] |
| 11 | Solithromycin | Solithera | Melinta | Macrolide | Phase 3 | Community-acquired pneumonia | [71] |
| 12 | Contezolid acefosamil | MRX 4 | MicuRx | Oxazolidinone | Phase 2/3 | Acute Bacterial Skin and Skin Structure Infection | [72] |
| 13 | Afabicin | AFN-1720 | Debiopharm | Enoyl-acyl carrier protein reductase (FabI) inhibitor | Phase 2 | Bacterial Skin Infection | [73] |
| 14 | LYS-228 | Ancremonam | Boston | Monobactam | Phase 2 | CRE | [74] |
| 15 | Nafithromycin | WCK 4873 | Wockhardt | Macrolide | Phase 2 | Community-acquired pneumonia | [75] |
| 16 | TNP-2092 | CBR-2092 | TenNor | Rifamycin–quinolone hybrid | Phase 2 | Acute Bacterial Skin and Skin Structure Infection | [76] |
| 17 | Benapenem | Benapenum | Sihuan | Carbapenem | Phase 2 | cUTI | [76] |
| 18 | Zidebactam+cefepime | WCK 5222 | Wockhardt | DBO-BLI | Phase 1 | cUTI | [76] |
| 19 | Nacubactam+meropenem | - | NacuGen | DBO-BLI | Phase 1 | Meropenem-resistant P. aeruginosa | [76] |
| 20 | ETX0282+cefpodoxime | ETX0282CPDP | Entasis | DBO-BLI | Phase 1 | CRE | [76] |
| 21 | VNRX-7145+ceftibuten | VNRX-5236 Etzadroxil | VenatoRx | Boronate-BLI | Phase 1 | CRE | [76] |
| 22 | SPR-741+β-lactam | – | Spero | Polymyxin+β-lactam | Phase 1 | Resistance Gram-negative bacterial infection | [76] |
| 23 | SPR-206 | EVER-206 | Spero | Polymyxin | Phase 1 | CRE | [76] |
| 24 | KBP-7072 | – | KBP Biosciences | 3rd-gen tetracycline | Phase 2 | Multi-resistant bacterial infection | [76] |
| 25 | TP-271 | – | Tetraphase | Tetracycline | Phase 1 | CRE | [76] |
| 26 | TP-6076 | – | Tetraphase | Tetracycline | Phase 1 | CRE | [76] |
| 27 | Apramycin (EBL-10031) | – | Juvabis | Aminoglycoside | Phase 1 | CRAB | [77] |
| 28 | AIC-499 | AiCuris | β-lactam | Phase 1 |
CRE, CRPA, CRAB |
[78] | |
| 29 | TNP-2198 | – | TenNor | Rifamycin–nitroimidazole hybrid | Phase 1 | Bacterial vaginosis | [78] |
| 30 | TXA-709 | – | Taxis | FtsZ inhibitor | Phase 1 | MRSA | [78] |
| 31 | BCM-0184 | Biocidium | – | Phase 1 | MRSA, VRE, CRE | [78] | |
| 32 | Ceftazidime-avibactam | Zavicefta | Actavis | BLI+β-lactam | Phase 1 | CRE | [79] |
| 33 | Meropenem and vaborbactam | VABOMERE | Melinta | β-lactam+BLI | US FDA approved | cUTI | [80] |
| 34 | Iclaprim | Mersarex | Motif Bio PLC | Diaminopyrimidine | NDA | Acute Bacterial Skin and Skin Structure Infection | [81] |
| 35 | Omadacycline | Nuzyra | Paratek | Tetracycline | NDA | cUTI, CRAB | [82] |
| 36 | Eravacycline | Xerava | Tetraphase | Tetracycline | NDA | CRE | [82] |
| 37 | Delpazolid | LCB01-0371 |
LegoChem Biosciences |
Oxazolidinone | Phase 1 | TB | [82] |
| 38 | Brilacidin | PMX-30063 | Innovation | Polymer-based antibiotics, Mimetic defensin | Phase 2/NDA | Coronavirus disease 2019(COVID 19), Acute bacterial skin, and skin structure infections | [83] |
| 39 | Nemonoxacin | Taigexyn | TaiGen | Quinolone | Phase 2 | Acute bacterial skin and skin structure infections | [84] |
| 40 | Lefamulin | BC-3781 | Nabriva | Pleuromutilin | USFDA | Acute bacterial skin and skin structure infections | [85] |
| 41 | Murepavadin | POL7080 | Polyphor | Peptidomimetic | Phase 3 | Hospital-acquired bacterial pneumonia | [85] |
| 42 | Ceftobiprole | Zevtera | Basilea | β-lactam | Phase 3 | MRSA | [85] |
| 43 | Ridinilazole | SMT19969 | Summit | Bis-benzimidazole 11 | Phase 2 | Clostridioides difficile infection | [85] |
Conclusion
Antimicrobial resistance has been increasing in the recent years. This study presented updated information on treatment methodologies currently adopted worldwide against major superbugs. The application of novel strategies like nanoparticle-based formulations and new plant-derived antimicrobial agents can eliminate such antimicrobial resistance and may prevent these superbugs causing much damage. Preventive measures must be taken to minimize bacterial MDR by rational use of antimicrobial drugs whenever needed. Healthcare professionals should also promote drug safety awareness to prevent such drug resistance. Instead of broad-spectrum antibiotics, narrow-spectrum antibiotic must be preferred. Additionally, the rational use of antibiotics and maintenance of proper hygiene and disinfectant must be practiced among patients.
Author Contributions
All authors made substantial contributions to the conception and writing of the manuscript. They have also read and agreed to the publication of the manuscript.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or nonprofit sectors.
Data Availability
Not applicable.
Code Availability
Not applicable.
Declarations
Conflict of interest
The authors declare no conflict of interest.
Ethical Approval
Not applicable.
Consent to Participate
Not applicable.
Consent for Publication
Not applicable.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Colson AR, Morton A, Årdal C, Chalkidou K, Davies SC, Garrison LP, Jit M, Laxminarayan R, Megiddo I, Morel C, Nonvignon J. Antimicrobial resistance: is health technology assessment part of the solution or part of the problem? Value Health. 2021;24(12):1828–1834. doi: 10.1016/j.jval.2021.06.002. [DOI] [PubMed] [Google Scholar]
- 2.Catalano A, Iacopetta D, Ceramella J, Scumaci D, Giuzio F, Saturnino C, Aquaro S, Rosano C, Sinicropi MS. Multidrug resistance (MDR): a widespread phenomenon in pharmacological therapies. Molecules. 2022;27(3):616. doi: 10.3390/molecules27030616. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Bloom DE, Black S, Salisbury D, Rappuoli R. Antimicrobial resistance and the role of vaccines. Proc Natl Acad Sci USA. 2018;115(51):12868–12871. doi: 10.1073/pnas.1717157115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.https://www.who.int/medicines/publications/WHO-PPL-Short_Summary_25Feb-ET_NM_WHO.pdf. Accessed 6 Apr 2021
- 5.Billamboz M, Fatima Z, Hameed S, Jawhara S. Promising drug candidates and new strategies for fighting against the emerging superbug Candida auris. Microorganisms. 2021;9(3):634. doi: 10.3390/microorganisms9030634. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Tahmasebi H, Dehbashi S, Arabestani MR. Co-harboring of mcr-1 and β-lactamase genes in Pseudomonas aeruginosa by high-resolution melting curve analysis (HRMA): molecular typing of superbug strains in bloodstream infections (BSI) Infect Genet Evol. 2020;85:104518. doi: 10.1016/j.meegid.2020.104518. [DOI] [PubMed] [Google Scholar]
- 7.Sleiman A, Awada B, Mocadie M, Sherri N, Haraoui LP, Baby V, Araj GF, Kanj SS, Rizk N, Matar GM, Abou Fayad AG. An unequivocal superbug: PDR Klebsiella pneumoniae with an arsenal of resistance and virulence factor genes. J Infect Dev Ctries. 2021;15(03):404–414. doi: 10.3855/jidc.13573. [DOI] [PubMed] [Google Scholar]
- 8.Chawla M, Verma J, Gupta R, Das B. Antibiotic potentiators against multidrug-resistant bacteria: discovery, development, and clinical relevance. Front Microbiol. 2022 doi: 10.3389/fmicb.2022.887251. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Kaur B, Gupta J, Sharma S, Sharma D, Sharma S. Focused review on dual inhibition of quorum sensing and efflux pumps: a potential way to combat multi drug resistant Staphylococcus aureus infections. Int J Biol Macromol. 2021;190:33–43. doi: 10.1016/j.ijbiomac.2021.08.199. [DOI] [PubMed] [Google Scholar]
- 10.Gaglio R, Couto N, Marques C, Lopes MD, Moschetti G, Pomba C, Settanni L. Evaluation of antimicrobial resistance and virulence of enterococci from equipment surfaces, raw materials, and traditional cheeses. Int J Food Microbiol. 2016;236:107–114. doi: 10.1016/j.ijfoodmicro.2016.07.020. [DOI] [PubMed] [Google Scholar]
- 11.Lambert PA. Bacterial resistance to antibiotics: modified target sites. Adv Drug Deliv Rev. 2005;57(10):1471–1485. doi: 10.1016/j.addr.2005.04.003. [DOI] [PubMed] [Google Scholar]
- 12.Lade H, Kim JS. Bacterial targets of antibiotics in methicillin-resistant Staphylococcus aureus. Antibiotics. 2021;10(4):398. doi: 10.3390/antibiotics10040398. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Davin-Regli A, Bolla JM, James CE, Lavigne JP, Chevalier J, Garnotel E, Molitor A. Membrane permeability and regulation of drug “influx and efflux” in enterobacterial pathogens. Curr Drug Targets. 2008;9(9):750–759. doi: 10.2174/138945008785747824. [DOI] [PubMed] [Google Scholar]
- 14.Pang Z, Raudonis R, Glick BR, Lin TJ, Cheng Z. Antibiotic resistance in Pseudomonas aeruginosa: mechanisms and alternative therapeutic strategies. Biotechnol Adv. 2019;37(1):177–192. doi: 10.1016/j.biotechadv.2018.11.013. [DOI] [PubMed] [Google Scholar]
- 15.Pachori P, Gothalwal R, Gandhi P. Emergence of antibiotic resistance Pseudomonas aeruginosa in intensive care unit; a critical review. Genes Dis. 2019;6(2):109–119. doi: 10.1016/j.gendis.2019.04.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Bush K, Bradford PA. β-Lactams and β-lactamase inhibitors: an overview. Cold Spring Harb Perspect Med. 2016;6(8):a025247. doi: 10.1101/cshperspect.a025247. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Shalaby MA, Dokla EM, Serya RA, Abouzid KA. Penicillin binding protein 2a: an overview and a medicinal chemistry perspective. Eur J Med Chem. 2020;199:112312. doi: 10.1016/j.ejmech.2020.112312. [DOI] [PubMed] [Google Scholar]
- 18.Gemmell CG, Edwards DI, Fraise AP, Gould FK, Ridgway GL, Warren RE. Guidelines for the prophylaxis and treatment of methicillin-resistant Staphylococcus aureus (MRSA) infections in the UK. J Antimicrob Chemother. 2006;57(4):589–608. doi: 10.1093/jac/dkl017. [DOI] [PubMed] [Google Scholar]
- 19.Rajput A, Bhamare KT, Mukhopadhyay A, Rastogi A, Kumar M. Efficacy of anti-biofilm agents in targeting ESKAPE pathogens with a focus on antibiotic drug resistance. InQuorum sensing: microbial rules of life 2020. Washington: American Chemical Society; 2020. pp. 177–199. [Google Scholar]
- 20.Kulengowski B, Rutter WC, Campion JJ, Lee GC, Feola DJ, Burgess DS. Effect of increasing meropenem MIC on the killing activity of meropenem in combination with amikacin or polymyxin B against MBL-and KPC-producing Enterobacter cloacae. Diagn Microbiol Infect Dis. 2018;92(3):262–266. doi: 10.1016/j.diagmicrobio.2018.06.013. [DOI] [PubMed] [Google Scholar]
- 21.O’Driscoll T, Crank CW. Vancomycin-resistant enterococcal infections: epidemiology, clinical manifestations, and optimal management. Infect Drug Resist. 2015;8:217. doi: 10.2147/idr.s54125. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Yu W, Zhang J, Tong J, Zhang L, Zhan Y, Huang Y, Qiu Y. In vitro antimicrobial activity of fosfomycin, vancomycin and daptomycin alone, and in combination, against linezolid-resistant Enterococcus faecalis. Infect Dis Ther. 2020;9(4):927–934. doi: 10.1007/s40121-020-00342-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Farias FM, Teixeira LM, Vallim DC, de Freire Bastos MD, Miguel MA, Bonelli RR. Characterization of Enterococcus faecium E86 bacteriocins and their inhibition properties against Listeria monocytogenes and vancomycin-resistant Enterococcus. Braz J Microbiol. 2021;26:1. doi: 10.1007/s42770-021-00494-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Ferreira M, Bessa LJ, Sousa CF, Eaton P, Bongiorno D, Stefani S, Campanile F, Gameiro P. Fluoroquinolone metalloantibiotics: a promising approach against methicillin-resistant Staphylococcus aureus. Int J Environ Res Public Health. 2020;17(9):3127. doi: 10.3390/ijerph17093127. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Spinler JK, Haidacher SJ, Hoch KM, Luna RA, Haag AM. Discerning strain-specific β-lactam drug resistance by clonal isolates of multi-drug resistant Pseudomonas aeruginosa using selected reaction monitoring. Int J Mass Spectrom. 2019;438:36–43. doi: 10.1016/j.ijms.2018.12.013. [DOI] [Google Scholar]
- 26.Gondil VS, Harjai K, Chhibber S. Endolysins as emerging alternative therapeutic agents to counter drug-resistant infections. Int J Antimicrob Agents. 2020;55(2):105844. doi: 10.1016/j.ijantimicag.2019.11.001. [DOI] [PubMed] [Google Scholar]
- 27.Phan MD, Forde BM, Peters KM, Sarkar S, Hancock S, Stanton-Cook M, Ben Zakour NL, Upton M, Beatson SA, Schembri MA. Molecular characterization of a multidrug resistance IncF plasmid from the globally disseminated Escherichia coli ST131 clone. PLoS ONE. 2015;10(4):e0122369. doi: 10.1371/journal.pone.0122369. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Zykov IN, Frimodt-Møller N, Småbrekke L, Sundsfjord A, Samuelsen Ø. Efficacy of mecillinam against clinical multidrug-resistant Escherichia coli in a murine urinary tract infection model. Int J Antimicrob Agents. 2020;55(2):105851. doi: 10.1016/j.ijantimicag.2019.11.008. [DOI] [PubMed] [Google Scholar]
- 29.Abdelhamid AG, Esaam A, Hazaa MM. Cell free preparations of probiotics exerted antibacterial and antibiofilm activities against multidrug resistant E. coli. Saudi Pharm J. 2018;26(5):603–607. doi: 10.1016/j.jsps.2018.03.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Vaz MS, Simionatto E, de Souza GH, Fraga TL, de Oliveira GG, Coutinho EJ, dos Santos MV, Simionatto S. Zingiberofficinale roscoe essential oil: an alternative strategy in the development of novel antimicrobial agents against MDR bacteria. Ind Crops Prod. 2022;185:115065. doi: 10.1016/j.indcrop.2022.115065. [DOI] [Google Scholar]
- 31.Mehta J, Rolta R, Dev K. Role of medicinal plants from North Western Himalayas as an efflux pump inhibitor against MDR AcrAB-TolC Salmonella entericaserovartyphimurium: in vitro and in silico studies. J Ethnopharmacol. 2022;282:114589. doi: 10.1016/j.jep.2021.114589. [DOI] [PubMed] [Google Scholar]
- 32.Chakraborty AK, Saha S, Poria K, Samanta T, Gautam S, Mukhopadhyay J. A saponin-polybromophenol antibiotic (CU1) from Cassia fistula bark against multi-drug resistant bacteria targeting RNA polymerase. Curr Res Pharmacol Drug Discov. 2022;3:100090. doi: 10.1016/j.crphar.2022.100090. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Jepkoech C, Omosa LK, Nchiozem-Ngnitedem VA, Kenanda EO, Guefack MG, Mbaveng AT, Kuete V, Heydenreich M. Antibacterial secondary metabolites from Vernonia auriculifera Hiern (Asteraceae) against MDR phenotypes. Nat Prod Res. 2022;36(12):3203–3206. doi: 10.1080/14786419.2021.1953024. [DOI] [PubMed] [Google Scholar]
- 34.Rolta R, Goyal M, Sharma S, Bharaj D, Salaria D, Upadhyay NK, Lal UR, Dev K, Sourirajan A. Bioassay guided fractionation of phytocompounds from Bergenia ligulata: a synergistic approach to treat drug resistant bacterial and fungal pathogens. Pharmacol Res Modern Chin Med. 2022;3:100076. doi: 10.1016/j.prmcm.2022.100076. [DOI] [Google Scholar]
- 35.Fang HQ, Zeng J, Wang SK, Wang X, Chen F, Li B, Liu J, Jin Z, Liu YH, Tang YZ. Discovery of novel pleuromutilin derivatives as potent antibacterial agents for the treatment of MRSA infection. Molecules. 2022;27(3):931. doi: 10.3390/molecules27030931. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Khan F, Tabassum N, Bamunuarachchi NI, Kim YM. Phloroglucinol and its derivatives: antimicrobial properties toward microbial pathogens. J Agric Food Chem. 2022;70(16):4817–4838. doi: 10.1021/acs.jafc.2c00532. [DOI] [PubMed] [Google Scholar]
- 37.Qin HL, Liu J, Fang WY, Ravindar L, Rakesh KP. Indole-based derivatives as potential antibacterial activity against methicillin-resistance Staphylococcus aureus (MRSA) Eur J Med Chem. 2020;194:112245. doi: 10.1016/j.ejmech.2020.112245. [DOI] [PubMed] [Google Scholar]
- 38.Sasaki H, Kashiwada Y, Shibata H, Takaishi Y. Prenylated flavonoids from Desmodium caudatum and evaluation of their anti-MRSA activity. Phytochemistry. 2012;82:136–142. doi: 10.1016/j.phytochem.2012.06.007. [DOI] [PubMed] [Google Scholar]
- 39.Muhsinah AB, Maqbul MS, Mahnashi MH, Jalal MM, Altayar MA, Saeedi NH, Alshehri OM, Shaikh IA, Khan AA, Iqubal SS, Khan KA. Antibacterial activity of Illicium verum essential oil against MRSA clinical isolates and determination of its phyto-chemical components. J King Saud Univ Sci. 2021 doi: 10.1016/j.jksus.2021.101800. [DOI] [Google Scholar]
- 40.Ramachandran G, Rajivgandhi GN, Murugan S, Alharbi NS, Kadaikunnan S, Khaled JM, Almanaa TN, Manoharan N, Li WJ. Anti-carbapenamase activity of Camellia japonica essential oil against isolated carbapenem resistant Klebsiella pneumoniae (MN396685) Saudi J Biol Sci. 2020;27(9):2269. doi: 10.1016/j.sjbs.2020.06.044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Konai MM, Bhattacharjee B, Ghosh S, Haldar J. Recent progress in polymer research to tackle infections and antimicrobial resistance. Biomacromol. 2018;19(6):1888–1917. doi: 10.1021/acs.biomac.8b00458. [DOI] [PubMed] [Google Scholar]
- 42.Kiani M, Pournajaf A, Zareh T, Karami M, Taghizadeh Armaki M, Gholami M. Antimicrobial activity of ethanolic and methanolic extracts of Urtica dioica, Mentha longifolia, and bacteriocin produced by Lactobacillus casei against antibiotic-resistant bacteria. Res Mol Med. 2020;8(4):163–170. doi: 10.32598/rmm.8.1062.2. [DOI] [Google Scholar]
- 43.Singh LK, Maheshwari DK, Shukla S. Antibacterial effect of butyryl alkannin from Arnebia euchroma against vancomycin-resistant pathogens of Enterococcus faecalis causing urinary tract infections. Nat Prod Res. 2015;29(24):2299–2301. doi: 10.1080/14786419.2015.1004676. [DOI] [PubMed] [Google Scholar]
- 44.Dey DI, Ray R, Hazra B. Antibacterial and antitubercular activity of selected plant products against multi-drug resistant clinical isolates. Res. 2015;1021:1014–1021. doi: 10.1002/ptr.5090. [DOI] [PubMed] [Google Scholar]
- 45.Dharmaratne MP, Manoraj A, Thevanesam V, Ekanayake A, Kumar NS, Liyanapathirana V, Abeyratne E, Bandara BR. Terminalia bellirica fruit extracts: in-vitro antibacterial activity against selected multidrug-resistant bacteria, radical scavenging activity and cytotoxicity study on BHK-21 cells. BMC Complement Altern Med. 2018;18(1):1–2. doi: 10.1186/s12906-018-2382-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Chakotiya AS, Chawla R, Thakur P, Tanwar A, Narula A, Grover SS, Goel R, Arora R, Sharma RK. In vitro bactericidal activity of promising nutraceuticals for targeting multidrug resistant Pseudomonas aeruginosa. Nutrition. 2016;32(7–8):890–897. doi: 10.1016/j.nut.2016.01.024. [DOI] [PubMed] [Google Scholar]
- 47.Sadiq MB, Tarning J, Aye Cho TZ, Anal AK. Antibacterial activities and possible modes of action of Acacia nilotica (L.) Del. against multidrug-resistant Escherichia coli and Salmonella. Molecules. 2017;22(1):47. doi: 10.3390/molecules22010047. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Rahman S, Parvez AK, Islam R, Khan MH. Antibacterial activity of natural spices on multiple drug resistant Escherichia coli isolated from drinking water, Bangladesh. Ann Clin Microbiol Antimicrob. 2011;10(1):1–4. doi: 10.1186/1476-0711-10-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Rohatgi A, Gupta P. Natural and synthetic plant compounds as anti-biofilm agents against Escherichia coli O157: H7 biofilm. Infect Genet Evol. 2021;95:105055. doi: 10.1016/j.meegid.2021.105055. [DOI] [PubMed] [Google Scholar]
- 50.Singh R, Smitha MS, Singh SP. The role of nanotechnology in combating multi-drug resistant bacteria. J Nanosci Nanotechnol. 2014;14(7):4745–4756. doi: 10.1166/jnn.2014.9527. [DOI] [PubMed] [Google Scholar]
- 51.Baptista PV, McCusker MP, Carvalho A, Ferreira DA, Mohan NM, Martins M, Fernandes AR. Nano-strategies to fight multidrug resistant bacteria—“A Battle of the Titans”. Front Microbiol. 2018;9:1441. doi: 10.3389/fmicb.2018.01441. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.dos Santos Souza LM, Dibo M, Sarmiento JJ, Seabra AB, Medeiros LP, Lourenço IM, Kobayashi RK, Nakazato G. Biosynthesis of selenium nanoparticles using combinations of plant extracts and their antibacterial activity. Curr Res Green Sustain Chem. 2022;5:100303. doi: 10.1016/j.crgsc.2022.100303. [DOI] [Google Scholar]
- 53.Murugan M, Rani KB, Wins JA, Ramachandran G, Guo F, Mothana RA, Noman OM, Nasr FA, Siddiqi MZ. Green synthesized ZnO NPs as effective bacterial inhibitor against isolated MDRs and biofilm producing bacteria isolated from urinary tract infections. J King Saud Univ Sci. 2022;34(1):101737. doi: 10.1016/j.jksus.2021.101737. [DOI] [Google Scholar]
- 54.Arshad H, Saleem M, Pasha U, Sadaf S. Synthesis of Aloe vera-conjugated silver nanoparticles for use against multidrug-resistant microorganisms. Electron J Biotechnol. 2022;55:55–64. doi: 10.1016/j.ejbt.2021.11.003. [DOI] [Google Scholar]
- 55.Verma AK, Kumar P. On recent developments in biosynthesis and application of Au and Ag nanoparticles from biological systems. J Nanotechnol. 2022 doi: 10.1155/2022/5560244. [DOI] [Google Scholar]
- 56.de Andrade LF, Apolinário AC, Rangel-Yagui CO, Stephano MA, Tavares LC. Chitosan nanoparticles for the delivery of a new compound active against multidrug-resistant Staphylococcus aureus. J Drug Deliv Sci Technol. 2020;55:101363. doi: 10.1016/j.jddst.2019.101363. [DOI] [Google Scholar]
- 57.Memar MY, Yekani M, Ghanbari H, Shahi S, Sharifi S, Maleki DS. Biocompatibility, cytotoxicity and antibacterial effects of meropenem-loaded mesoporous silica nanoparticles against carbapenem-resistant Enterobacteriaceae. Artif Cells Nanomed Biotechnol. 2020;48(1):1354–1361. doi: 10.1080/21691401.2020.1850466. [DOI] [PubMed] [Google Scholar]
- 58.Brown AN, Smith K, Samuels TA, Lu J, Obare SO, Scott ME. Nanoparticles functionalized with ampicillin destroy multiple-antibiotic-resistant isolates of Pseudomonas aeruginosa and Enterobacter aerogenes and methicillin-resistant Staphylococcus aureus. Appl Environ Microbiol. 2012;78(8):2768–2774. doi: 10.1128/aem.06513-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Khalil MA, El Maghraby GM, Sonbol FI, Allam NG, Ateya PS, Ali SS. Enhanced efficacy of some antibiotics in presence of silver nanoparticles against multidrug resistant Pseudomonas aeruginosa recovered from burn wound infections. Front Microbiol. 2021 doi: 10.3389/fmicb.2021.648560. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Wahab S, Khan T, Adil M, Khan A. Mechanistic aspects of plant-based silver nanoparticles against multi-drug resistant bacteria. Heliyon. 2021;7(7):e07448. doi: 10.1016/j.heliyon.2021.e07448. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Gabrielyan L, Hakobyan L, Hovhannisyan A, Trchounian A. Effects of iron oxide (Fe3O4) nanoparticles on Escherichia coli antibiotic-resistant strains. J Appl Microbiol. 2019;126(4):1108–1116. doi: 10.1111/jam.14214. [DOI] [PubMed] [Google Scholar]
- 62.Rahman AU, Khan AU, Yuan Q, Wei Y, Ahmad A, Ullah S, Khan ZU, Shams S, Tariq M, Ahmad W. Tuber extract of Arisaema flavum eco-benignly and effectively synthesize silver nanoparticles: photocatalytic and antibacterial response against multidrug resistant engineered E. coli QH4. J Photochem Photobiol B. 2019;193:31–38. doi: 10.1016/j.jphotobiol.2019.01.018. [DOI] [PubMed] [Google Scholar]
- 63.World Health Organization. 2019 antibacterial agents in clinical development: an analysis of the antibacterial clinical development pipeline
- 64.Katahira EJ, Davidson SM, Stevens DL, Bolz DD. Subinhibitory concentrations of tedizolid potently inhibit extracellular toxin production by methicillin-sensitive and methicillin-resistant Staphylococcus aureus. J Med Microbiol. 2019;68(2):255. doi: 10.1099/jmm.0.000905. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Eraikhuemen N, Julien D, Kelly A, Lindsay T, Lazaridis D. Treatment of community-acquired pneumonia: a focus on lefamulin. Infect Dis Ther. 2021 doi: 10.1007/s40121-020-00378-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Heo YA. Imipenem/cilastatin/relebactam: a review in gram-negative bacterial infections. Drugs. 2021 doi: 10.1007/s40265-021-01471-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Tanaka K, Vu H, Hayashi M. In vitro activities and spectrum of lascufloxacin (KRP-AM1977) against anaerobes. J Infect Chemother. 2021;27(8):1265–1269. doi: 10.1016/j.jiac.2021.03.026. [DOI] [PubMed] [Google Scholar]
- 68.Kanj SS, Bassetti M, Kiratisin P, Rodrigues C, Villegas MV, Yu Y, van Duin D. Clinical data from studies involving novel antibiotics to treat multidrug-resistant Gram-negative bacterial infections. Int J Antimicrob Agents. 2022 doi: 10.1016/j.ijantimicag.2022.106633. [DOI] [PubMed] [Google Scholar]
- 69.Bakthavatchalam YD, Rao SV, Isaac B, Manesh A, Nambi S, Swaminathan S, Nagvekar V, Nangia V, John PV, Veeraraghavan B. A comparative assessment of clinical, pharmacological and antimicrobial profile of novel anti-methicillin-resistant Staphylococcus aureus agent levonadifloxacin: therapeutic role in nosocomial and community infections. Indian J Med Microbiol. 2019;37(4):478–487. doi: 10.4103/ijmm.IJMM_20_34. [DOI] [PubMed] [Google Scholar]
- 70.Giacobbe DR, Dettori S, Corcione S, Vena A, Sepulcri C, Maraolo AE, De Rosa FG, Bassetti M. Emerging treatment options for acute bacterial skin and skin structure infections and bloodstream infections caused by Staphylococcus aureus: a comprehensive review of the evidence. Infect Drug Resist. 2022;15:2137. doi: 10.2147/IDR.S318322. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Barrera CM, Mykietiuk A, Metev H, Nitu MF, Karimjee N, Doreski PA, Mitha I, Tanaseanu CM, Molina JM, Antonovsky Y, Van Rensburg DJ. Efficacy and safety of oral solithromycin versus oral moxifloxacin for treatment of community-acquired bacterial pneumonia: a global, double-blind, multicentre, randomised, active-controlled, non-inferiority trial (SOLITAIRE-ORAL) Lancet Infect Dis. 2016;16(4):421–430. doi: 10.1016/S1473-3099(16)00017-7. [DOI] [PubMed] [Google Scholar]
- 72.Bassetti M, Del Puente F, Magnasco L, Giacobbe DR. Innovative therapies for acute bacterial skin and skin-structure infections (ABSSSI) caused by methicillin-resistant Staphylococcus aureus: advances in phase I and II trials. Expert Opin Investig Drugs. 2020;29(5):495–506. doi: 10.1080/13543784.2020.1750595. [DOI] [PubMed] [Google Scholar]
- 73.Dyon-Tafani V, Josse J, Dieppois G, Ferry T, Laurent F. Antimicrobial activity of the new FabI inhibitor afabicin desphosphono against intraosteoblastic Staphylococcus aureus. Int J Antimicrob Agents. 2021;57(5):106321. doi: 10.1016/j.ijantimicag.2021.106321. [DOI] [PubMed] [Google Scholar]
- 74.Kong Q, Yang Y. Recent advances in antibacterial agents. Bioorg Med Chem Lett. 2021;35:127799. doi: 10.1016/j.bmcl.2021.127799. [DOI] [PubMed] [Google Scholar]
- 75.Trivedi J, Shaikh J, Chavan N, Thorve D, Chaudhary B, Karade A, Gupta S, Patel A, Bhagwat S. Pretreatment of nafithromycin attenuates inflammatory response in murine lipopolysaccharide induced acute lung injury. Cytokine. 2020;129:155049. doi: 10.1016/j.cyto.2020.155049. [DOI] [PubMed] [Google Scholar]
- 76.Giacobbe DR, Labate L, Vena A, Bassetti M. Potential role of new-generation antibiotics in acute bacterial skin and skin structure infections. Curr Opin Infect Dis. 2021;34(2):109–117. doi: 10.1097/qco.0000000000000708. [DOI] [PubMed] [Google Scholar]
- 77.Berini F, Orlandi V, Gornati R, Bernardini G, Marinelli F. Nanoantibiotics to fight multidrug resistant infections by Gram-positive bacteria: hope or reality? Biotechnol Adv. 2022 doi: 10.1016/j.biotechadv.2022.107948. [DOI] [PubMed] [Google Scholar]
- 78.Isler B, Doi Y, Bonomo RA, Paterson DL. New treatment options against carbapenem-resistant Acinetobacter baumannii infections. Antimicrob Agents Chemother. 2019;63(1):e01110–e01118. doi: 10.1128/aac.01110-18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Hakeam HA, Alsahli H, Albabtain L, Alassaf S, Al Duhailib Z, Althawadi S. Effectiveness of ceftazidime–avibactam versus colistin in treating carbapenem-resistant Enterobacteriaceae bacteremia. Int J Infect Dis. 2021;109:1–7. doi: 10.1016/j.ijid.2021.05.079. [DOI] [PubMed] [Google Scholar]
- 80.Jean SS, Lu MC, Ho MW, Ko WC, Hsueh PR, Grop ST. Non-susceptibilities to antibiotics against important Gram-negative bacteria, and imipenem-relebactam, meropenem-vaborbactam against carbapenem non-susceptible Enterobacterales and Pseudomonas aeruginosa isolates implicated in complicated intra-abdominal and urinary tract infections in Taiwan, 2019. Int J Antimicrob Agents. 2022;59(3):106521. doi: 10.1016/j.ijantimicag.2022.106521. [DOI] [PubMed] [Google Scholar]
- 81.Huang DB, Noviello S, Balser B, Scaramucci A, Corey GR. A Pooled analysis of the safety and efficacy of iclaprim versus vancomycin for the treatment of acute bacterial skin and skin structure infections in patients with intravenous drug use: phase 3 REVIVE studies. Clin Ther. 2019;41(6):1090–1096. doi: 10.1016/j.clinthera.2019.04.004. [DOI] [PubMed] [Google Scholar]
- 82.Nicklas DA, Maggioncalda EC, Story-Roller E, Eichelman B, Tabor C, Serio AW, Keepers TR, Chitra S, Lamichhane G. Potency of omadacycline against Mycobacteroides abscessus clinical isolates in vitro and in a mouse model of pulmonary infection. Antimicrob Agents Chemother. 2022;66(1):e01704–e01721. doi: 10.1128/AAC.01704-21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Xu C, Wang A, Honnen W, Pinter A, Weston WK, Harness JA, Narayanan A, Chang TL. Brilacidin, a non-peptide defensin-mimetic molecule, inhibits SARS-CoV-2 infection by blocking viral entry. EC Microbiol. 2022;18(4):1. [PMC free article] [PubMed] [Google Scholar]
- 84.Zhao M, Chi Z, Pan X, Yin Y, Tang W. Economic evaluation of nemonoxacin, moxifloxacin and levofloxacin in the treatment of early community-acquired pneumonia with possible pulmonary tuberculosis. Int J Environ Res Public Health. 2022;19(8):4816. doi: 10.3390/ijerph19084816. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Paukner S, Goldberg L, Alexander E, Das AF, Heinrich S, Patel P, Moran GJ, Sandrock C, File TM, Jr, Vidal JE, Waites KB. Pooled microbiological findings and efficacy outcomes by pathogen in adults with community-acquired bacterial pneumonia from the lefamulin evaluation against pneumonia (LEAP) 1 and LEAP 2 phase 3 trials of lefamulin versus moxifloxacin. J Glob Antimicrob Resist. 2022;29:434–443. doi: 10.1016/j.jgar.2021.10.021. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Not applicable.
Not applicable.