Abstract
Purpose
Carbapenem-resistant Gram-negative bacteria bloodstream infection (CRGNB-BSI) has gradually become a major threat worldwide due to its treatment difficulty and high mortality. This study aimed to determine the risk factors for CRGNB-BSI in immunosuppressed patients.
Patients and Methods
A total of 427 immunosuppressed patients with CRGNB-BSI were retrospectively investigated from 2015 to 2021. Both univariate and multivariate logistic regression analyses were applied to evaluate independent risk factors for CRGNB-BSI.
Results
The most common etiology was Klebsiella Pneumoniae (50.59%; 216/427), while the Acinetobacillus baumannii infection was associated with the highest mortality (58.25%) among all etiologies. The 60-day mortality of immunosuppressed patients with CRGNB-BSI was 52.48% (224/427). Procalcitonin (PCT) > 0.5 μg/L (OR = 2.32, 95% CI: 1.28–4.19, P = 0.005) and age > 55 years (OR = 2.06, 95% CI: 1.17–3.64, P = 0.012) were found to be predictors of 60-day mortality of CRGNB-BSI, and tigecycline regimen (OR = 3.20, 95% CI: 1.81–5.67, P < 0.001) was associated with higher mortality. Multivariate analysis also revealed that patients who developed acute kidney injury (AKI) (OR = 2.19, 95% CI: 1.11–4.30, P = 0.023), gastrointestinal bleeding (OR = 3.18, 95% CI: 1.10–9.16, P = 0.032), multiple organ dysfunction syndrome (MODS) (OR = 12.11, 95% CI: 2.61–56.19, P = 0.001), and septic shock (OR = 3.24, 95% CI: 1.77–5.94, P < 0.001) showed worse outcomes. The risk factors were also significantly associated with mortality in the different subgroups.
Conclusion
This study demonstrated that PCT > 0.5 μg/L, age > 55 years, and the tigecycline regimen were significantly associated with higher 60-day mortality among immunosuppressed patients with CRGNB- BSI. Patients developing MODS, septic shock, or AKI had worse clinical outcomes.
Keywords: bloodstream infection, carbapenem-resistant, immunosuppressed patients, subgroups, prognosis
Introduction
Carbapenem-resistant Gram-negative bacteria (CRGNB) currently pose a great threat worldwide. Compared with other types of CRGNB infection, such as respiratory infection, urinary tract infection, and wound infection, CRGNB Bloodstream Infection has the highest mortality.1,2 According to the China Antimicrobial Surveillance Network 2021 report, there were 301,917 bacterial samples in China in 2021, 14.4% of which came from blood, and the rate is rising each year. Moreover, there is an increasing trend in CRGNB.3 Carbapenem-resistant Gram-negative bacteria bloodstream infections (CRGNB-BSI), including carbapenem-resistant Klebsiella pneumoniae (CRKP), Acinetobacter baumannii (CRAB), and Pseudomonas aeruginosa (CRPA), have emerged as a global public health problem with few effective treatments and are associated with high mortality.4,5 Immunosuppressive status refers to low immune response or immunodeficiency.6 Immunosuppressed patients having infections may present with atypical symptoms and worse prognosis.7,8 They include patients receiving immunosuppressive therapy, such as oral immunosuppressive medications, chemotherapy, or radiotherapy; patients under transplantation states; patients with diseases such as diabetes, cancer, liver cirrhosis, and burns; and patients having a postoperative critical condition.9,10 Previous studies explored certain specific immunosuppressed groups such as patients with cancers, transplantation status, hematological diseases, and diabetes,5,11–13 but there is still a lack of research on CRGNB-BSI in the overall immunosuppressed population.
In this retrospective study that aimed to determine the risk factors for CRGNB-BSI mortality in immunosuppressed patients, we explored the clinical characteristics, antibiotic strategies, microbiological types, and clinical outcomes of immunosuppressed patients with CRGNB-BSI in Shanghai Ruijin hospital.
Materials and Methods
Patients and Definition
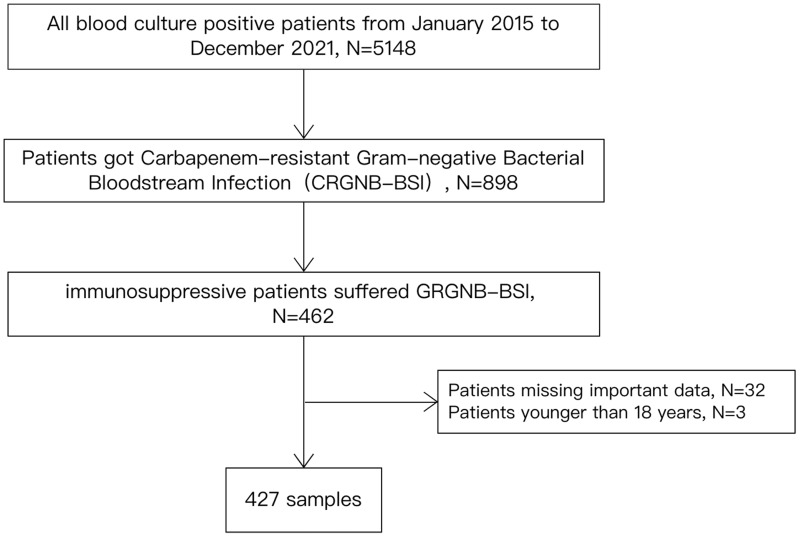
This retrospective cohort study was conducted from January 2015 to December 2021 in Ruijin Hospital, Shanghai Jiaotong University School of Medicine, Shanghai, China, a 2139-bed academic tertiary-care hospital that ranks among the top five hospitals in China. The inclusion criteria for the study were age ≥ 18 years and diagnosis of CRGNB-BSI. CRGNB was defined as Gram-negative isolates demonstrating resistance to at least one of the carbapenems, including ertapenem, meropenem, or imipenem, according to the results of antimicrobial susceptibility testing. BSI was defined according to the US Centers for Disease Control and Prevention (CDC) criteria.14 Immunosuppressed patients included patients receiving immunosuppressive therapy, chemotherapy, or radiotherapy; patients under transplantation states; patients having diseases such as cancer, burns, diabetes, and liver cirrhosis; and patients having a postoperative critical condition. Immunosuppressive therapy is defined as the use of drugs such as corticosteroids (prednisone equivalent > 20 mg/day) for at least 14 days, or methotrexate, cyclosporine, azathioprine, or biological modifiers for up to 3 months. Transplantation states include solid-organ, stem cell, or bone marrow transplantation. Cancer refers to both solid organ and hematological system tumors. Patients with a postoperative critical condition were identified when their blood culture was positive within 1 week following thoracotomy and laparotomy9,10 (Figure 1). Regarding the therapeutic strategy, monotherapy was defined as the use of one intravenous antibiotic, and combination therapy was defined as the concomitant use of at least two intravenous antibiotics.
Figure 1.
Study flow chart.
Data Collection
Data regarding age, sex, precipitating factors, complications, etiologies, and treatment outcomes were collected. The evaluation of outcomes was based on 60-day survival or death after blood cultures. Procalcitonin (PCT) level was recorded at the onset of infection. Body Mass Index (BMI) was collected at hospitalization. All data in this study were collected through the electronic medical record system, and two authors independently cleaned and analyzed the data.
Statistical Analysis
Data were analyzed using Stata 14.1 (StataCorp LLC, College Station, TX, USA) on Mac OS. All samples from patients in this study were categorized into 60-day death and 60-day survival groups. Each factor was first summarized and then compared between different groups. For continuous variables, the clinical characteristics are presented as the mean ± standard deviation, and Student’s t-test was applied for the comparison between the two groups. For categorical variables, the number of samples and proportion (%) are presented, and Chi-square test was applied. Risk factors related to 60-day mortality were identified by a two-step logistic regression. First, all characteristics were tested by univariate regression; then, those with P-value < 0.05 were selected and included in the multivariate analysis. A Kaplan–Meier survival curve was plotted for variables that showed significance in the multivariate logistic regression. The same regression analysis procedure was then applied in different subgroups (ICU admission, acute kidney injury, septic shock, burns, tumors, diabetes, CRAB, CRKP, tigecycline).
Result
Patients’ Clinical Characteristics and Laboratory Data
A total of 427 patients were included in this retrospective cohort study, of whom 203 were categorized as 60-day survival and 224 as 60-day death. The mortality rate in the whole population was 52.48%. In the 60-day survival group, approximately 74% (151/203) were male, with mean age of 50.82 ± 1.23 years and mean BMI of 22.98 ± 0.25 kg/m2. In the 60-day death group, approximately 71% (158/224) were male, with mean age of 58.40 ± 1.04 years and mean BMI of 23.40 ± 0.27 kg/m2. Table 1 presents the general characteristics of patients in both groups. The results showed that there were approximately 84% (178/213) of patients in the 60-day death group with PCT > 0.5 μg/L, while the percentage in the 60-day survival group was 67% (126/188). In addition, PCT > 0.5 μg/L, age > 55 years, ICU admission, hypertension, diabetes, coronary heart disease, and hypoproteinemia all showed significant differences between the two groups, whereas no clear differences in sex, smoking, and drinking were found (Table 1).
Table 1.
The Clinical Characteristics of Carbapenem-Resistant Gram-Negative Bloodstream Infections in Immunosuppressed Patients
| 60-Day Survivals | 60-Day Death | P-value | |
|---|---|---|---|
| Age | 50.82(1.23) | 58.40(1.04) | <0.001 |
| BMIa | 22.98(0.25) | 23.40(0.27) | 0.263 |
| PCTb>0.5 ug/L | 126(67%) | 178(84%) | <0.001 |
| Male | 151(74%) | 158(71%) | 0.374 |
| Smoking | 58(29%) | 53(24%) | 0.248 |
| Drinking | 45(22%) | 50(22%) | 0.970 |
| Age>55 Years | 86(42%) | 141(63%) | <0.001 |
| ICUc-Stay | 73(36%) | 127(57%) | <0.001 |
| Hypertension | 55(27%) | 111(50%) | <0.001 |
| Diabetes | 40(20%) | 66(29%) | 0.020 |
| Asthma | 2(1%) | 2(1%) | 0.921 |
| COPDd | 1(0%) | 3(1%) | 0.364 |
| Chronic Bronchitis | 2(1%) | 1(0%) | 0.506 |
| Cerebral Infarction | 11(5%) | 14(6%) | 0.715 |
| Coronary Heart Disease | 17(8%) | 37(17%) | 0.011 |
| Anemia | 24(12%) | 30(13%) | 0.626 |
| Hypoproteinemia | 24(12%) | 48(21%) | 0.008 |
| Hyperlipidemia | 10(5%) | 7(3%) | 0.342 |
| Chronic Renal Failure | 9(4%) | 9(4%) | 0.831 |
| Chronic Kidney Disease | 2(1%) | 5(2%) | 0.311 |
| Underlying Conditions (n,%) | |||
| Liver Cirrhosis | 4(2%) | 11(5%) | 0.099 |
| Acute Myocardial Infarction | 2(1%) | 9(4%) | 0.048 |
| Acute Pancreatitis | 20(10%) | 33(15%) | 0.127 |
| Rheumatic Heart Disease | 1(0%) | 3(1%) | 0.364 |
| Valvular Heart Disease | 9(4%) | 23(10%) | 0.022 |
| Rheumatic Systemic Diseases | 5(2%) | 11(5%) | 0.184 |
| Skin Disease | 2(1%) | 5(2%) | 0.311 |
| Burning | 69(34%) | 49(22%) | 0.005 |
| Biliary Infection | 15(7%) | 24(11%) | 0.234 |
| Aortic Dissection | 0(0%) | 5(2%) | 0.032 |
| Aortic Aneurysm | 2(1%) | 4(2%) | 0.483 |
| Tumors | 70(34%) | 60(27%) | 0.084 |
| Haematological Diseases | 19(9%) | 22(10%) | 0.871 |
| Gastrointestinal Cancer | 35(17%) | 33(15%) | 0.479 |
| Respiratory System Tumors | 0(0%) | 1(0%) | 0.341 |
| Urinary System Tumors | 4(2%) | 1(0%) | 0.144 |
| Complications (n, %) | |||
| AKIe | 22(11%) | 63(28%) | <0.001 |
| Cerebral Hemorrhage | 5(2%) | 2(1%) | 0.202 |
| Abdominal Infection | 29(14%) | 53(24%) | 0.014 |
| Abdominal Hemorrhage | 10(5%) | 17(8%) | 0.259 |
| Acute Respiratory Failure | 12(6%) | 25(11%) | 0.054 |
| Gastrointestinal Bleeding | 8(4%) | 24(11%) | 0.008 |
| MODSf | 4(2%) | 27(12%) | <0.001 |
| Shock | 33(16%) | 95(42%) | <0.001 |
| Inducement (n, %) | |||
| Surgery | 75(37%) | 104(46%) | 0.047 |
| Chemotherapy | 18(9%) | 20(9%) | 0.982 |
| Burning | 68(33%) | 48(21%) | 0.005 |
| Catheter Placement | 6(3%) | 20(9%) | 0.010 |
| Transplant Status | 9(4%) | 3(1%) | 0.053 |
| Hormone | 2(1%) | 6(3%) | 0.197 |
| Hormone+ Immunosuppressant | 4(2%) | 5(2%) | 0.851 |
| Pathogen Sources (n, %) | |||
| Respiratory Tract | 52(26%) | 85(38%) | 0.006 |
| Digestive Tract | 48(24%) | 53(24%) | 0.997 |
| Skin | 48(24%) | 39(17%) | 0.110 |
| Urinary System | 6(3%) | 4(2%) | 0.425 |
| Catheter | 21(10%) | 18(8%) | 0.408 |
| Etiologies (n, %) | |||
| ECOg | 10(5%) | 3(1%) | 0.031 |
| ECLh | 4(2%) | 0(0%) | 0.035 |
| KPNi | 96(47%) | 120(54%) | 0.195 |
| PAEj | 29(14%) | 22(10%) | 0.155 |
| ABAk | 43(21%) | 60(27%) | 0.177 |
| Others | 16(8%) | 11(5%) | 0.208 |
| Antibacterial therapy | |||
| Third Generation Cephalosporin | 33(16%) | 29(13%) | 0.332 |
| Quinolone | 26(13%) | 23(10%) | 0.411 |
| Fourth Generation Cephalosporin | 2(1%) | 4(2%) | 0.483 |
| Tetracycline | 6(3%) | 4(2%) | 0.425 |
| Polymyxin | 11(5%) | 26(12%) | 0.023 |
| Macrolides | 11(5%) | 16(7%) | 0.465 |
| Ceftazidime-Avibactam | 3(1%) | 2(1%) | 0.575 |
| Oxazolidinone | 31(15%) | 40(18%) | 0.474 |
| Tigecycline | 36(18%) | 79(35%) | <0.001 |
| Aminoglycosides | 22(11%) | 17(8%) | 0.245 |
| Cyclo-Lipopeptide | 0(0%) | 5(2%) | 0.032 |
| Nitroimidazole | 6(3%) | 8(4%) | 0.721 |
| Carbapenem | 134(66%) | 147(66%) | 0.933 |
| Sulfonamides | 4(2%) | 10(4%) | 0.148 |
| Glycopeptide | 67(33%) | 72(32%) | 0.849 |
| Penicillin | 7(3%) | 6(3%) | 0.644 |
| Antifungal Agents | 79(39%) | 104(46%) | 0.117 |
| Mono Carbapenem | 37(18%) | 26(12%) | 0.054 |
| Mono Tigecycline | 0(0%) | 7(3%) | 0.011 |
| Mono Polymyxins | 0(0%) | 2(1%) | 0.177 |
| Carbapenem combination | 97(48%) | 121(54%) | 0.198 |
| Tigecycline combination | 36(18%) | 72(32%) | 0.001 |
| Polymyxins combination | 11(5%) | 24(11%) | 0.046 |
| Mono Ceftazidime-Avibactam | 0(0%) | 0(0%) | NA |
| Ceftazidime-Avibactam combination | 3(1%) | 2(1%) | 0.575 |
Notes: aBody Mass Index, bProcalcitonin, cIntensive Care Unit, dChronic Obstructive Pulmonary Disease, eAcute kidney injury, fMultiple Organ Dysfunction Syndrome, gEscherichia Coli, hEnterobacter cloacae, iKlebsiella pneumoniae, jPseudomonas aeruginosa, kAcinetobacter baumannii.
Underlying conditions that were significant included acute myocardial infarction (1% vs 4%), valvular heart disease (4% vs 10%), burns (34% vs 22%), and aortic dissection (0% vs 2%). There were approximately 34% (70/203) and 27% (60/224) of patients with tumors in the 60-day survival and 60-day death groups, respectively. The percentages in the different tumor types were hematological diseases (9% vs 10%), gastrointestinal cancer (17% vs 15%), respiratory system tumors (0% vs 0%), and urinary system tumors (2% vs 0%) (Table 1).
Complications that showed significant differences were acute kidney injury (11% vs 28%), abdominal infection (14% vs 24%), gastrointestinal bleeding (4% vs 11%), multiple organ dysfunction syndrome (MODS) (2% vs 12%), and septic shock (16% vs 42%) (Table 1).
The participating factors were surgery (37% vs 46%), burns (33% vs 21%), and catheter placement (3% vs 9%), which showed significant differences, and chemotherapy, hormone, transplant status, and hormone combination immunosuppressant, which showed no significant differences. The sources of infection included respiratory tract, digestive tract, skin, urinary system, and catheter, of which only respiratory tract (26% vs 38%) showed significant differences (Table 1, Supplementary Figure 1).
The etiologies of bloodstream infections that showed significant differences were ECO (5% vs 1%) and ECL (2% vs 0%), whereas CRKP, CRAB, and CRPA showed no significant differences. The major antibiotics were polymyxin (5% vs 12%), tigecycline (18% vs 35%), and cyclo-lipopeptide (0% vs 2%), others such as third-generation cephalosporin, fourth-generation cephalosporin, and carbapenem were not significant (Table 1, Supplementary Figure 2).
The antibiotic therapies were carbapenem, polymyxin, and tigecycline. Only tigecycline (0% vs 3%) showed significance in both monotherapy and combination therapy (Table 1). For all patients, 88 (21%) received monotherapy, 325 (76%) received combination therapy, and monotherapy (27% vs 15%) was associated with a higher survival rate (P < 0.01). In the CRE subgroup, tigecycline-based combining therapy (23% vs 37%) and polymyxin-based combining therapy (3% vs 11%) were found to be significant (Table 2).
Table 2.
Therapeutic Regimen of CRGNB BSI Patients in Immunosuppressive Status
| 60-Day Survivals | 60-Day Death | P-value | |
|---|---|---|---|
| Mono-therapy | 54(27%) | 34(15%) | 0.004 |
| Combination-therapy | 140(69%) | 185(83%) | 0.001 |
| CRABa | |||
| Mono-therapy | |||
| Carbapenem | 6(14%) | 9(15%) | 0.882 |
| Tigecycline | 0(0%) | 2(3%) | 0.227 |
| Polymyxins | 0(0%) | 1(2%) | 0.395 |
| Combination-therapy | |||
| Carbapenem Combination | 21(49%) | 33(55%) | 0.537 |
| Tigecycline Combination | 8(19%) | 13(22%) | 0.704 |
| Polymyxins Combination | 4(9%) | 5(8%) | 0.864 |
| CREb | |||
| Mono-therapy | |||
| Carbapenem | 21(20%) | 13(11%) | 0.050 |
| Tigecycline | 0(0%) | 4(3%) | 0.061 |
| Polymyxins | 0(0%) | 1(1%) | 0.352 |
| Ceftazidime-avibactam | 0(0%) | 0(0%) | NA |
| Combination-therapy | |||
| Carbapenem Combination | 56(53%) | 68(55%) | 0.710 |
| Tigecycline Combination | 24(23%) | 46(37%) | 0.016 |
| Polymyxins Combination | 3(3%) | 13(11%) | 0.022 |
| Ceftazidime-avibactam Combination | 3(3%) | 2(2%) | 0.534 |
| CRPAc | |||
| Mono-therapy | |||
| Carbapenem | 4(14%) | 3(14%) | 0.987 |
| Ceftazidime-avibactam | 0(0%) | 0(0%) | NA |
| Polymyxins | 0(0%) | 0(0%) | NA |
| Combination-therapy | |||
| Carbapenem Combination | 13(45%) | 8(36%) | 0.543 |
| Ceftazidime-avibactam Combination | 0(0%) | 0(0%) | NA |
| Polymyxins Combination | 3(10%) | 4(18%) | 0.421 |
Notes: aCarbapenem-resistant Gram-negative Acinetobacillus baumannii. bCarbapenem-resistant Gram-negative Enterobacteriaceae, cCarbapenem-resistant Gram-negative Pseudomonas aeruginosa.
Risk Factors Associated with 60-Day Mortality
As revealed by the univariate logistic regression, 20 variables including PCT > 0.5 μg/L (OR = 2.50, 95% CI: 1.56–4.02, P < 0.001) and age > 55 years (OR = 2.31, 95% CI: 1.57–3.41, P < 0.01) showed statistical significance and were included in the multivariate analysis. Table 3 shows both the univariate and multivariate regression results. The multivariate logistic regression showed that PCT > 0.5 μg/L (OR = 2.32, 95% CI: 1.28–4.19, P = 0.005), age > 55 years (OR = 2.06, 95% CI: 1.17–3.64, P = 0.012), AKI (OR = 2.19, 95% CI: 1.11–4.30, P = 0.023), gastrointestinal bleeding (OR = 3.18, 95% CI: 1.10–9.16, P = 0.032), MODS (OR = 12.11, 95% CI: 2.61–56.19, P = 0.001), septic shock (OR = 3.24, 95% CI: 1.77–5.94, P < 0.001), and tigecycline regimen (OR = 3.20, 95% CI: 1.81–5.67, P < 0.001) were considered as significant factors for 60-day mortality.
Table 3.
Univariate and Multivariate Analysis of Factors Related to Over 60-Day Survivals
| Univariate Analysis | Multivariate Analysis | |||
|---|---|---|---|---|
| OR (95% CI) | P value | OR (95% CI) | P value | |
| PCT>0.5 ug/L | 2.50 (1.56,4.02) | <0.001 | 2.32 (1.28,4.19) | 0.005 |
| Age>55 years | 2.31 (1.57,3.41) | <0.001 | 2.06 (1.17,3.64) | 0.012 |
| ICU-Stay | 2.33 (1.58,3.44) | <0.001 | NA | 0.511 |
| Hypertension | 2.64 (1.76,3.96) | <0.001 | NA | 0.299 |
| Diabetes | 1.70 (1.09,2.67) | 0.020 | NA | 0.862 |
| Coronary Heart Disease | 2.16 (1.18,3.98) | 0.013 | NA | 0.848 |
| Hypoproteinemia | 2.03 (1.19,3.46) | 0.009 | NA | 0.469 |
| Valvular Heart Disease | 2.47 (1.11,5.46) | 0.026 | NA | 0.407 |
| Burning | 0.54 (0.35,0.84) | 0.005 | NA | 0.955 |
| Surgery | 1.48 (1.00,2.18) | 0.048 | NA | 0.822 |
| Catheter Placement | 3.22 (1.27,8.18) | 0.014 | NA | 0.142 |
| AKI | 3.22 (1.90,5.47) | <0.001 | 2.19 (1.11,4.30) | 0.023 |
| Abdominal Infection | 1.86 (1.13,3.06) | 0.015 | NA | 0.851 |
| Gastrointestinal Bleeding | 2.92 (1.28,6.67) | 0.011 | 3.18 (1.10,9.16) | 0.032 |
| MODS | 6.82 (2.34,19.85) | <0.001 | 12.11 (2.61,56.19) | 0.001 |
| Shock | 3.79 (2.40,5.99) | <0.001 | 3.24 (1.77,5.94) | <0.001 |
| Respiratory Tract | 1.78 (1.17,2.69) | 0.007 | NA | 0.899 |
| ECOa | 0.26 (0.07,0.97) | 0.044 | NA | 0.294 |
| Polymyxin | 2.29 (1.10,4.77) | 0.026 | NA | 0.182 |
| Tigecycline | 2.53 (1.61,3.97) | <0.001 | 3.20 (1.81,5.67) | <0.001 |
Notes: aEscherichia coli.
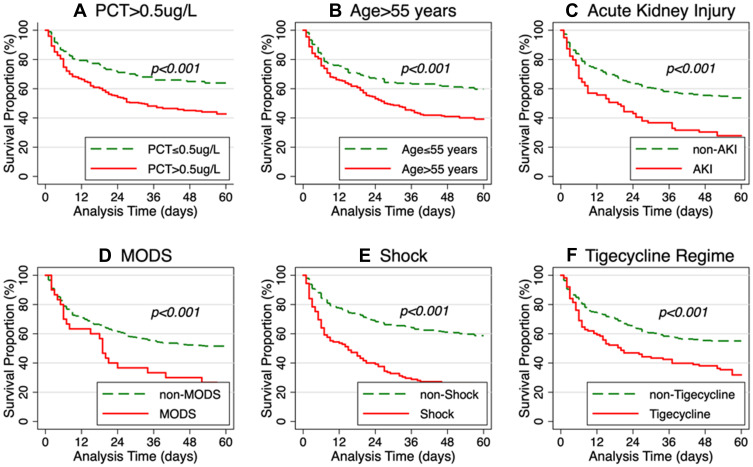
The Kaplan–Meier survival curve is shown in Figure 2. The Log rank test was applied to compare the survival proportion between the treatment and control groups. The results showed that the 60-day mortality was significantly higher for patients with PCT > 0.5 μg/L, age > 55 years, AKI, septic shock, and tigecycline regimen.
Figure 2.
Survival analysis of patients with carbapenem-resistant Gram-negative bloodstream infections in immunosuppressed patients, Kaplan–Meier curves showing the impact on 60-day mortality of (A) Procalcitonin > or <=0.5ug/L, (B) Age> or <=55 years, (C) patients with or without acute kidney injury, (D) patients with or without multiple organ dysfunction syndrome (E) patients with or without Shock, (F) patients took or did not take tigecycline.
Risk Factors Associated with 60-Day Mortality in Different Subgroups
Table 4 summarizes risk factors of the subgroups associated with ICU admission, diabetes, CRAB infection, and CRKP infection. The results of both univariate and multivariate regression analyses were displayed, and significant variables in each subgroup were listed. Moreover, we summarized risk factors of the subgroups incorporating acute kidney injury, septic shock, burns, tumors, and tigecycline regimen (Supplementary Table 1).
Table 4.
Univariate and Multivariate Analysis of Factors Related to Over 60-Day Survivals in Different Subgroups
| Univariate Analysis | Multivariate Analysis | |||
|---|---|---|---|---|
| ICU-Admission Subgroup | OR (95% CI) | P value | OR (95% CI) | P value |
| PCT>0.5 ug/L | 3.06 (1.49,6.26) | 0.002 | 2.23 (1.02,4.88) | 0.046 |
| Age>55 years | 2.22 (1.21,4.09) | 0.010 | 2.88 (1.39,5.98) | 0.005 |
| Hypertension | 2.04 (1.14,3.66) | 0.017 | NA | 0.206 |
| AKI | 2.65 (1.34,5.24) | 0.005 | 2.91 (1.27,6.70) | 0.012 |
| Shock | 2.21 (1.21,4.04) | 0.010 | 2.49 (1.22,5.05) | 0.012 |
| Tigecycline | 5.59 (2.09,14.98) | 0.001 | 4.96 (1.70,14.44) | 0.003 |
| Diabetes Subgroup | OR (95% CI) | P value | OR (95% CI) | P value |
| Age>55 years | 3.36 (1.33,8.50) | 0.011 | NA | 0.057 |
| ICU Stay | 2.51 (1.07,5.89) | 0.034 | NA | 0.119 |
| Hyperlipidemia | 0.22 (0.05,0.93) | 0.039 | NA | 0.290 |
| Shock | 2.50 (1.05,5.93) | 0.038 | 3.89 (1.20,12.64) | 0.024 |
| CRAB | 4.20 (1.32,13.34) | 0.015 | 7.20 (1.85,27.93) | 0.004 |
| Tigecycline | 22.29 (2.88,172.65) | 0.003 | 34.16 (4.05,288.06) | 0.001 |
| CRAB Infection Subgroup | OR (95% CI) | P value | OR (95% CI) | P value |
| PCT>0.5 ug/L | 4.48 (1.63,12.31) | 0.004 | 3.77 (1.20,11.85) | 0.023 |
| ICU Stay | 2.29 (1.03,5.11) | 0.042 | NA | 0.750 |
| Hypertension | 3.31 (1.35,8.07) | 0.009 | NA | 0.391 |
| Diabetes | 5.25 (1.65,16.71) | 0.005 | NA | 0.063 |
| Coronary Heart Disease | 3.26 (1.10,9.63) | 0.033 | NA | 0.814 |
| Hypoproteinemia | 5.67 (1.21,26.62) | 0.028 | 7.28 (1.16,45.49) | 0.034 |
| Tumors | 0.36 (0.13,0.95) | 0.040 | NA | 0.125 |
| AKI | 4.40 (1.51,12.83) | 0.007 | NA | 0.251 |
| CRKP Infection Subgroup | OR (95% CI) | P value | OR (95% CI) | P value |
| Age>55 years | 2.40 (1.39,4.16) | 0.002 | 2.35 (1.13,4.91) | 0.022 |
| ICU-Stay | 2.09 (1.21,3.60) | 0.008 | NA | 0.772 |
| Hypertension | 2.43 (1.39,4.26) | 0.002 | NA | 0.528 |
| Coronary Heart Disease | 3.54 (1.14,10.96) | 0.029 | NA | 0.173 |
| Burning | 0.41 (0.22,0.77) | 0.005 | NA | 0.997 |
| AKI | 2.69 (1.27,5.69) | 0.010 | NA | 0.323 |
| Abdominal Infection | 2.15 (1.12,4.13) | 0.021 | NA | 0.132 |
| MODS | 7.76 (1.75,34.47) | 0.007 | 7.49 (1.47,38.23) | 0.015 |
| Shock | 4.52 (2.37,8.63) | <0.001 | 3.49 (1.63,7.49) | 0.001 |
| Respiratory Tract | 2.09 (1.15,3.82) | 0.016 | NA | 0.683 |
| Skin | 0.46 (0.24,0.88) | 0.020 | NA | 0.952 |
| Polymyxin | 3.77 (1.04,13.62) | 0.043 | NA | 0.219 |
| Tigecycline | 2.27 (1.25,4.10) | 0.007 | 2.63 (1.29,5.37) | 0.008 |
There were 200 observations in the ICU admission subgroup. In the univariate analysis, seven variables were significant, of which PCT > 0.5 μg/L (OR = 2.23, 95% CI: 1.02–4.88, P = 0.046), age > 55 years (OR = 2.88, 95% CI: 1.39–5.98, P = 0.005), AKI (OR = 2.91, 95% CI: 1.27–6.70, P = 0.012), septic shock (OR = 2.49, 95% CI: 1.22–5.05, P = 0.012), and tigecycline regimen (OR = 4.96, 95% CI: 1.70–14.44, P = 0.003) were considered as significant factors in the multivariate analysis.
There were 117 observations in the Diabetes subgroup. In the univariate analysis, six variables were significant, of which septic shock (OR = 3.89, 95% CI: 2.041.20–12.64, P = 0.024), CRAB infection (OR = 7.2, 95% CI: 1.85–27.93, P = 0.001), and tigecycline regimen (OR = 34.16, 95% CI: 4.05–288.06, P = 0.001) were considered as significant factors in the multivariate analysis.
There were 103 observations in the CRAB subgroup. In the univariate analysis, nine variables were significant, of which PCT > 0.5 μg/L (OR = 3.77, 95% CI: 1.20–11.85, P = 0.023) and hypoproteinemia (OR = 7.28, 95% CI: 1.16–45.49, P = 0.034) were considered as significant factors in the multivariate analysis.
There were 216 observations in the CAKP subgroup. In the univariate analysis, 14 variables were significant, of which age > 55 years (OR = 2.35, 95% CI: 1.13–4.91, P = 0.022), MODS (OR = 7.49, 95% CI: 1.647–38.23, P = 0.015), septic shock (OR = 3.49, 95% CI: 1.63–7.49, P = 0.001), and tigecycline regimen (OR = 2.63, 95% CI: 1.29–5.37, P = 0.008) were considered as significant factors in the multivariate analysis.
Discussion
CRGNB infection has become a significant problem worldwide, with poorer outcomes in patients who are immunocompromised.15 In the CHINET resistance report 2021, the prevalence of resistance to meropenem of Klebsiella pneumoniae, Acinetobacter baumannii, and Pseudomonas aeruginosa were reported to be 24.4%, 72.3%, and 18.9%, respectively.3 In the present study, the clinical features and prognosis of CRGNB-BSI in immunosuppressed patients were reported. PCT level > 0.5 μg/L, use of tigecycline, age > 55 years, and development of MODS, shock, and AKI were all found to be associated with poor prognosis. To date, many studies have evaluated risk factors for different microbial infections in immunosuppressed patients. However, those studies were conducted in certain groups of immunosuppressed patients or for specific microbial infections in different types of immunosuppression.1,16–19 Studies related to CRGNB-BSI in the overall immunosuppressed population are still scarce. This study evaluated the clinical characteristics of immunosuppressed patients with CRGNB-BSI and investigated the prognostic factors in immunosuppression status for the overall population and different subgroups. A total of 427 patients were included, which is larger than previous studies on immunosuppressed patients with CRGNB-BSI.
In our study, the most common etiologies were CRKP, CRAB, and CRPA (Supplementary Figure 3), which is consistent with previous studies.20 The incidence of carbapenem-resistant Klebsiella pneumoniae (CRKP) infection is increasing, and it is an important source of infection in critically ill patients, such as patients with ICU-acquired infection and hematological malignancies, transplant patients, post-surgery patients, and patients on long-term corticosteroids. Furthermore, CRKP infection often suggests worse prognosis.19,21 Moreover, the mortality of CRKP bloodstream infection (BSI) is higher than that of other types of CRKP infection.20,21 The median time to death after a positive blood culture was found to be 46 days; therefore, we chose 60-day mortality as the outcome of our study. We found that the 30-, 60-, and 90-day mortality rates of the immunosuppressed patients were significantly affected by the risk factors for death mentioned earlier (Table 3, Supplementary Tables 2 and 3). The 60-day mortality of CRKP infection was 55.56% from our results, consistent with that of previous studies on CRKB-BSI.1 Besides, tigecycline regimen and septic shock development resulted in a higher mortality rate in the CRKP infection subgroup, being consistent with other studies.22 Moreover, age > 55 years and MODS were found to be independent risk factors for this population. Another remarkable etiology is CRAB; CRAB bacteremia has emerged as a major cause of nosocomial infections in recent decades and tops the World Health Organization’s threat to human health list.23 In our study, the 60-day mortality of the CRAB infection subgroup attained 58.25%, higher than that of CRKP and CRPA infection. Increased PCT level and hypoproteinemia were found to be independent risk factors for CRAB infection. Hypoproteinemia indicates poor nutritional status; previous studies have pointed out that hypoproteinemia is a risk factor for BSI death.24 Furthermore, CRAB infection is a noteworthy risk factor in the diabetes subgroup. Studies have shown that patients with diabetes have a higher mortality when Acinetobacter baumannii infection or CRAB bacteremia occurs, and a worse outcome can be expected when patients with bacteremia develop septic shock.13,25 In addition, we noticed that the use of tigecycline also led to worse outcomes in the diabetes subgroup (Table 4).
Immunosuppressed patients are often admitted to the ICU due to multiple infections, and infectious complications lead to high mortality in immunosuppressed patients.7 In previous studies, PCT and age were considered as independent risk factors for ICU admission,9 consistent with the results of our study. Furthermore, it was found that use of tigecycline and development of AKI and septic shock could be prognostic predictors of ICU admission for immunosuppressed patients with CRGNB-BSI (Table 4).
This study demonstrated that age > 55 years, PCT > 0.5 μg/L, and the tigecycline regimen are independent risks factors for CRGNB-BSI in immunosuppressed patients, and that a history of transplantation and having acute kidney injury or gastrointestinal bleeding are also associated with a higher 60-day mortality.
This study showed that PCT was closely related to poor prognosis in immunosuppressed patients with CRGNB-BSI. Previous studies showed that increased PCT often suggests poor outcomes in both immunosuppressed patients with BSI and patients with CRGNB-BSI,9,24 similar to the conclusion of our study. PCT is a predictor of poor prognosis in immunosuppressed patients with CRGNB infection. The results showed that PCT > 0.5 μg/L is an independent risk factor in immunosuppressed patients. PCT suggested a higher mortality in some subgroups such as the ICU admission, AKI, and CRAB infection subgroups (Table 4, Supplementary Table 1).
Studies have shown that older people compared to young individuals have an increased risk of Gram-negative bacilli infection and antibiotic resistance, and the highest mortality from BSI.24 Previous studies have shown that age could be used as an indicator of bloodstream infection in immunosuppressed patients and patients with severe infection.8,26 The mean age of our population is 54.8 years, and the median is 57 years. Therefore, we set age > 55 years as a factor and found that it could be an independent risk factor for 60-day mortality in immunosuppressed patients. Age > 55 years also emerged as an important prognostic factor in different subgroups of patients with burns, septic shock, and ICU hospitalization (Table 4, Supplementary Table 1).
Tigecycline is a new class of broad-spectrum glycylcycline antibiotics. Although the efficacy remains controversial, tigecycline is often recommended as an option for the treatment of resistant bacterial infections because it is active against multiple drug-resistant bacteria. The FDA has issued a black box warning that tigecycline use for FDA-approved or unapproved indications is associated with increased risk of death.27 Previous investigations also showed that tigecycline regimen was associated with higher mortality and caused worse outcomes than another antibiotic regimen in patients with CRGNB-BSI,22,28,29 consistent with the results of our study. Tigecycline regimen is not only an independent risk factor for immunosuppressed patients with CRGNB-BSI but also is regarded as a reason for the increased risk of 60-day mortality in the CRKP infection, diabetes, tumor, septic shock, and ICU stay subgroups. Moreover, many studies have shown that the use of tigecycline benefits immunosuppressed patients having CRGNB-BSI or other types infection.30–32 Some studies pointed out that high-dosage tigecycline therapy (200 mg loading dose followed by 100 mg every 12 h) has better outcomes in the treatment of severe infections compared with standard-dosage tigecycline therapy (100 mg loading dose followed by 50 mg every 12 h) and other non-tigecycline-containing regimens.33 In our study, only 20.87% accepted the high-dosage tigecycline therapy, and low serum levels of the drug may lead to an unfavorable microbiological response. Thus, more relevant research is needed.
BSI is associated with a high complication rate among severely ill patients, including those with AKI, which is a complication of critical illness that has long been recognized as being independently associated with mortality.34,35 Our study reached a consistent conclusion that AKI is one of the major complications among all immunosuppressed patients and that the occurrence of AKI remarkably increases mortality. Furthermore, AKI development was a significant prognostic risk factor in the ICU admission and septic shock subgroups (Table 4, Supplementary Table 1).
The results of the multivariate analysis indicated that MODS and septic shock were closely associated with poor outcomes in immunosuppressed patients with CRGNB-BSI, indicating that the severity of the patient’s disease was closely related to death, which was consistent with some previous studies.5,16,21,36 In this study, gastrointestinal bleeding often indicated a poor prognosis. A study by Amy et al showed that gastrointestinal bleeding had a negative impact on the short-term survival of patients with BSI.37 However, the underlying mechanism remains unclear, and more studies are needed in the future.
This study has several limitations. First, the clinical data in the present study were collected from a single center, indicating that the results may not be applied to other medical institutions. Second, novel antibacterial medications such as polymyxins and ceftazidime-avibactam have only been used more frequently in the last 2 years; therefore, the clinical outcome of the treatment could be biased. Third, although we provide features of CRGNB-BSI in the largest number of immunosuppressed patients so far, the number of cases is still small, and large-scale research may be needed in the future.
Conclusion
This study demonstrated that PCT > 0.5 μg/L, age > 55 years, and the tigecycline regimen were significantly associated with higher 60-day mortality among immunosuppressed patients with CRGNB-BSI. Patients developing MODS, septic shock, or AKI had poor clinical outcomes. The etiological characteristics of the underlying diseases or the occurrence of ICU admission vary little between the 60-day mortality and 60-day survival groups.
Funding Statement
The present study was supported by the National Natural Science Foundation of China (No.82170086), Shanghai Shenkang Hospital Development Center Clinical Science and Technology Innovation Project (SHDC12018102), Shanghai Municipal Key Clinical Specialty (shslczdzk02202), Shanghai Key Laboratory of Emergency Prevention, Diagnosis and Treatment of Respiratory Infectious Diseases (20dz2261100).
Ethics Statements
All analyses were based on previous clinical data and the study obtained ethical clearance from Ruijin Hospital Affiliated to Shanghai Jiaotong University School of Medicine Ethic committee with the approval of patient informed consent exemption. And this study followed the guidelines outlined in the Declaration of Helsinki.
Author Contributions
All authors made a significant contribution to the work reported, whether that is in the conception, study design, execution, acquisition of data, analysis and interpretation, or in all these areas; took part in drafting, revising or critically reviewing the article; gave final approval of the version to be published; have agreed on the journal to which the article has been submitted; and agree to be accountable for all aspects of the work.
Disclosure
Yulian Gao, Hongxia Lin, Yumin Xu, and Yijin Yao are co-first authors for this study. The authors report no conflicts of interest in this work.
References
- 1.Zhang P, Wang J, Hu H, et al. Clinical characteristics and risk factors for bloodstream infection due to carbapenem-resistant Klebsiella pneumoniae in patients with hematologic malignancies. Infect Drug Resist. 2020;13:3233–3242. doi: 10.2147/IDR.S272217 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Babiker A, Clarke LG, Saul M, et al. Changing epidemiology and decreased mortality associated with carbapenem-resistant gram-negative bacteria, 2000–2017. Clin Infect Dis. 2020;73(11):e4521–e4530. doi: 10.1093/cid/ciaa1464 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Hu FP China Antimicrobial Surveillance Network, the results of bacterial resistance in 2021[DB/OL]; 2022. Available from: https://www.chinets.com/Document. Accessed October 28, 2022.
- 4.Birru M, Woldemariam M, Manilal A, et al. Bacterial profile, antimicrobial susceptibility patterns, and associated factors among bloodstream infection suspected patients attending arba minch general hospital, Ethiopia. Sci Rep. 2021;11:1. doi: 10.1038/s41598-021-95314-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Wu D, Chen C, Liu T, et al. Epidemiology, susceptibility, and risk factors associated with mortality in carbapenem-resistant gram-negative bacterial infections among abdominal solid organ transplant recipients: a retrospective C. Infect Dis Ther. 2021;10(1):559–573. doi: 10.1007/s40121-021-00411-z [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Minotti C, Tirelli F, Barbieri E, Giaquinto C, Donà D. How is immunosuppressive status affecting children and adults in SARS-CoV-2 infection? A systematic review. Int J Infect. 2020;81(1):e61–e66. doi: 10.1016/j.jinf.2020.04.026 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Azoulay E, Russell L, Louw AV, et al. Diagnosis of severe respiratory infections in immunocompromised patients. Intensive Care Med. 2020;46(2):298–314. doi: 10.1007/s00134-019-05906-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Chai J, Han X, Mei Q, et al. Clinical characteristics and mortality of non-tuberculous mycobacterial infection in immunocompromised vs. Immunocompetent Hosts Front Med. 2022;9. doi: 10.3389/fmed.2022.884446 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Lin H, Yang L, Fang J, et al. Clinical characteristics of bloodstream infection in immunosuppressed patients: a 5-year retrospective cohort study. Front Cell Infect Microbiol. 2022;12. doi: 10.3389/fcimb.2022.796656 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Bar-Yoseph H, Cohen N, Korytny A, et al. Risk factors for mortality among carbapenem-resistant enterobacteriaceae carriers with focus on immunosuppression. Int J Infect. 2019;78(2):101–105. doi: 10.1016/j.jinf.2018.10.003 [DOI] [PubMed] [Google Scholar]
- 11.Baier C, Beck M, Panagiota V, et al. Infection control management and surveillance of carbapenem-resistant gram-negative bacteria in hematopoietic stem cell recipients. Antimicrob Resist Infect Control. 2019;8:1. doi: 10.1186/s13756-019-0606-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Lan P, Lu Y, Chen Z, et al. Emergence of high-level cefiderocol resistance in carbapenem-resistant Klebsiella pneumoniae from bloodstream infections in patients with hematologic malignancies in China. Microbiol Spectr. 2022;10:2. doi: 10.1128/spectrum.00084-22 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Leung C-H, Liu C-P. Diabetic status and the relationship of blood glucose to mortality in adults with carbapenem-resistant Acinetobacter baumannii complex bacteremia. J Microbiol Immunol Infect. 2019;52(4):654–662. doi: 10.1016/j.jmii.2018.06.005 [DOI] [PubMed] [Google Scholar]
- 14.Horan TC, Andrus M, Dudeck MA. CDC/NHSN surveillance definition of health care–associated infection and criteria for specific types of infections in the acute care setting. Am J Infect Control. 2008;36(5):309–332. doi: 10.1016/j.ajic.2008.03.002 [DOI] [PubMed] [Google Scholar]
- 15.Andria N, Henig O, Kotler O, et al. Mortality burden related to infection with carbapenem-resistant gram-negative bacteria among haematological cancer patients: a retrospective cohort study. J Antimicrob Chemother. 2015;70(11):3146–3153. doi: 10.1093/jac/dkv218 [DOI] [PubMed] [Google Scholar]
- 16.Hu Y, Li D, Xu L, et al. Epidemiology and outcomes of bloodstream infections in severe burn patients: a six-year retrospective study. Antimicrob Resist Infect Control. 2021;10:1. doi: 10.1186/s13756-021-00969-w [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Amanati A, Sajedianfard S, Khajeh S, et al. Bloodstream infections in adult patients with malignancy, epidemiology, microbiology, and risk factors associated with mortality and multi-drug resistance. BMC Infect Dis. 2021;21:1. doi: 10.1186/s12879-021-06243-z [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Kim SY, Jung JY, Kang YA, et al. Risk factors for occurrence and 30-day mortality for carbapenem-resistant Acinetobacter baumannii bacteremia in an intensive care unit. J Korean Med Sci. 2012;27(8):939. doi: 10.3346/jkms.2012.27.8.939 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Pereira MR, Scully BF, Pouch SM, et al. Risk factors and outcomes of carbapenem-resistant Klebsiella pneumoniae infections in liver transplant recipients. Liver Transplant. 2015;21(12):1511–1519. doi: 10.1002/lt.24207 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Zhang X, Qi S, Duan X, et al. Clinical outcomes and safety of polymyxin B in the treatment of carbapenem-resistant gram-negative bacterial infections: a real-world multicenter study. J Transl Med. 2021;19:1. doi: 10.1186/s12967-021-03111-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Chen J, Ma H, Li Y, et al. Risk factors and mortality of carbapenem-resistant Klebsiella pneumoniae bloodstream infection in a tertiary-care hospital in China: an eight-year retrospective study; 2020. [DOI] [PMC free article] [PubMed]
- 22.Xiao T, Zhu Y, Zhang S, et al. A retrospective analysis of risk factors and outcomes of carbapenem-resistant Klebsiella pneumoniae bacteremia in nontransplant patients. J Infect Dis. 2020;221(Supplement_2):S174–S183. doi: 10.1093/infdis/jiz559 [DOI] [PubMed] [Google Scholar]
- 23.Du X, Xu X, Yao J, et al. Predictors of mortality in patients infected with carbapenem-resistant Acinetobacter baumannii: a systematic review and meta-analysis. Am J Infect Control. 2019;47(9):1140–1145. doi: 10.1016/j.ajic.2019.03.003 [DOI] [PubMed] [Google Scholar]
- 24.Xie ZY, Meng GL, Xiong Y, et al. Prognostic factors of central venous catheter-related bloodstream infections. Zhongguo Yi Xue Ke Xue Yuan Xue Bao. 2020;42(6):789–794. doi: 10.3881/j.issn.1000-503X.12266 [DOI] [PubMed] [Google Scholar]
- 25.Perera D, Kleinstein SE, Hanson B, et al. Impaired host response and the presence of Acinetobacter baumannii in the serum microbiome of type-II diabetic patients. IScience. 2021;24(1):101941. doi: 10.1016/j.isci.2020.101941 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Martin GS, Mannino DM, Moss M. The effect of age on the development and outcome of adult sepsis. Crit Care Med. 2006;34(1):15–21. doi: 10.1097/01.ccm.0000194535 [DOI] [PubMed] [Google Scholar]
- 27.Vardakas KZ, Rafailidis PI, Falagas ME. Effectiveness and safety of tigecycline: focus on use for approved indications. Clin Infect Dis. 2012;54(11):1672–1674. doi: 10.1093/cid/cis239 [DOI] [PubMed] [Google Scholar]
- 28.Niu T, Xiao T, Guo L, et al. Retrospective comparative analysis of risk factors and outcomes in patients with carbapenem resistant Acinetobacter baumannii bloodstream infections: cefoperazone&Ndash; Sulbac. Infect Drug Resist. 2018;11:2021–2030. doi: 10.2147/IDR.S169432 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Niu T, Luo Q, Li Y, et al. Comparison of tigecycline or cefoperazone/sulbactam therapy for bloodstream infection due to carbapenem-resistant Acinetobacter baumannii. Antimicrob Resist Infect Control. 2019;8:1. doi: 10.1186/s13756-019-0502-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Modemann F, Härterich S, Wiesch JS, et al. Efficacy of tigecycline as salvage therapy in multidrug-resistant febrile neutropenia in patients with acute leukemia—A single center analysis. Antibiotics. 2022;11(2):128. doi: 10.3390/antibiotics11020128 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Geng -T-T, Xu X, Huang M. High-Dose Tigecycline for the Treatment of Nosocomial Carbapenem-Resistant Klebsiella Pneumoniae Bloodstream Infections. Medicine. 2018;97(8):e9961. doi: 10.1097/MD.0000000000009961 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Wu Y, Hu Z, Xing L, et al. 替加环素治疗血液病患者继发感染的疗效分析 [Efficacy of tigecycline in treating severe infections of patients with hematological diseases]. Zhonghua Yi Xue Za Zhi. 2014;94(34):2669–2672. Chinese. [PubMed] [Google Scholar]
- 33.Zha L, Pan L, Guo J, et al. Effectiveness and safety of high dose tigecycline for the treatment of severe infections: a systematic review and meta-analysis. Adv Ther. 2020;37(3):1049–1064. doi: 10.1007/s12325-020-01235-y [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Yu SN, Kim T, Park SY, et al. Predictors of acute kidney injury and 28-day mortality in carbapenem-resistant Acinetobacter baumannii complex bacteremia. Microb Drug Resist. 2021;27(8):1029–1036. doi: 10.1089/mdr.2020.0312 [DOI] [PubMed] [Google Scholar]
- 35.Papadimitriou-Olivgeris M, Assimakopoulos SF, Kolonitsiou F, et al. Risk factors for acute kidney injury in critically ill patients with bacteraemia by carbapenem non-s. Infez Med. 2019;27:380–392. [PubMed] [Google Scholar]
- 36.Freire MP, Garcia D, Garcia CP, et al. Bloodstream infection caused by extensively drug-resistant Acinetobacter baumannii in cancer patients: high mortality associated with delayed treatment rather than with the degree of neutropenia. Clin Microbiol Infect. 2016;22(4):352–358. doi: 10.1016/j.cmi.2015.12.010 [DOI] [PubMed] [Google Scholar]
- 37.Huang AH, Liu Y, Hsien Y-C, et al. Survival impact and clinical predictors of acute gastrointestinal bleeding in patients with bloodstream infection. J Intensive Care Med. 2019;36(1):63–69. doi: 10.1177/0885066619884896 [DOI] [PubMed] [Google Scholar]