Take Home Message
Patients receiving percutaneous cryoablation (PCA) had lower baseline scores on physical functioning but were comparable with those receiving partial nephrectomy (PN) with regard to Global Health Status.
Significant differences in health-related quality of life were reported from baseline to 14 d postoperatively between PN and PCA, but were no longer evident after 90 d.
Keywords: Renal cell carcinoma, Nephron-sparing treatment, Partial nephrectomy, Cryoablation, Health-related quality of life, Quality of life, Patient-reported outcome
Abstract
Background
Partial nephrectomy (PN) is the gold standard for the treatment of stage cT1 renal cell carcinoma (RCC). However, the increasing incidence of RCC in the elderly population calls for alternative minimally invasive treatments to reduce the negative effects on patients’ health-related quality of life (HRQoL) and subsequent healthy life expectancy.
Objective
To assess and compare short-term HRQoL and self-reported health status after PN and percutaneous cryoablation (PCA) of patients treated for RCC stage cT1.
Design, setting, and participants
Patients who underwent PN or PCA between 2019 and 2021 for RCC stage cT1 at two university hospitals in Denmark were assessed. The exclusion criteria included insufficient understanding of the Danish language, dementia, metastatic RCC, conversion to nephrectomy, and salvage procedures.
Intervention
PN and PCA.
Outcome measurements and statistical analysis
The European Organisation for the Research and Treatment of Cancer Quality of Life Questionnaire was distributed before treatment, and 14 and 90 d postoperatively. A linear mixed-effect model was used to analyze the changes from baseline to follow-up between PN and PCA treatment.
Results and limitations
The cohort included 165 patients (PN: 79; PCA: 86). The completion rate was 96–98%. Patients receiving PCA were significantly older (median 69.1 vs 62.1 yr) and had lower scores on physical (p < 0.001) and role functioning (p = 0.009) than PN. A statistically significant change from baseline to 14 d was found for several HRQoL scales, which favored PCA over PN. However, the observed change was no longer significant at 90-d follow-up. Limitations include sample size and confounding by indication.
Conclusions
This study found a significant difference between baseline and 14-d follow-up in several HRQoL and symptoms scales, favoring PCA over PN. However, no significant differences were observed in any HRQoL scales between PN and PCA of RCC stage cT1 from baseline to 90-d follow-up.
Patient summary
Surgical removal and percutaneous cryoablation (freezing) of small tumors in the kidney had a similar impact on quality of life after 90 d.
1. Introduction
In recent decades, increases in the incidence of renal cell carcinoma (RCC) stage cT1 have been reported, with the majority being incidental findings, with increasing age [1]. Partial nephrectomy (PN) is considered the gold standard treatment; however, ablation therapy, including percutaneous cryoablation (PCA), is an alternative minimally invasive treatment mostly preferred in elderly patients or those with severe comorbidity and not suitable candidates for surgery [2]. The increasing pace of an aging population highlights the need to expand the surgical treatment paradigm [3]. Treatment of small renal masses should take into account several factors, including oncological status, health-related quality of life (HRQoL), and healthy life expectancy. Patient-reported HRQoL contributes valuable information on factors otherwise challenging to measure, including emotional and cognitive functioning.
Previous studies on RCC have focused on HRQoL after PN [4], [5], [6] or compared PN with radical nephrectomy (RN) [7], [8], [9], [10]. However, only a few studies have assessed HRQoL after cryoablation, and these often pooled ablative therapies (eg, cryoablation and radiofrequency ablation) [11] and minimally invasive procedures (eg, percutaneous and laparoscopic) [12]. Furthermore, many studies were retrospective, and either these used HRQoL as a secondary outcome or HRQoL was inconsistently measured or reported [13]. Thus, it remains challenging to discuss HRQoL outcomes when advising patients with RCC stage cT1 on treatment decisions.
The aim of this prospective study was to assess and compare short-term HRQoL and self-reported health status after PN and PCA in patients treated for RCC stage cT1.
2. Patients and methods
The details of this comparative, prospective cohort study have been reported according to the “Strengthening the Reporting of Observational Studies in Epidemiology” (STROBE) guidelines [14]. In addition, this study is registered on ClinicalTrials.gov (NCT04040530).
2.1. Setting and participants
Patients were recruited from June 2019 to February 2021 from two university hospitals, Odense University Hospital (OUH) and Zeeland University Hospital (ZUH), covering one-third of the Danish population of 5.8 million people. Prior to treatment, all patients were discussed at a multidisciplinary team (MDT) conference that included a cancer course coordinator, urologists, radiologists, pathologists, and oncologists. If nephron-sparing surgery (NSS) was recommended at the MDT, consensus on treatment (PN or PCA) was reached between the patient and the urologist, based on the principles of shared decision-making. Thus, patient allocation was not decided by the physicians but was a result of shared decision-making, following standard clinical practice at the institutions.
The inclusion criteria were determined as follows: PN or PCA of histologically verified primary RCC clinical stage cT1, >18 yr of age, and sufficient understanding of the Danish language to self-report HRQoL. Exclusion criteria were the following: patients with diagnosed dementia or under evaluation for dementia, and patients with multiple tumors treated with NSS more than once within 3 mo. One patient treated with NSS for a new primary tumor after 3 mo was included in the study as a new patient with a new baseline.
For PN, we included both robot-assisted PN (RAPN) and open PN (OPN), with RAPN as the preferred choice if surgically possible. Experienced urologists performed PN at both institutions, and the surgical procedure has been described previously [15]. PCA was performed by specialized interventional radiologists under computed tomography guidance, with the patient under sedation, if possible. The PCA procedure is described in the Supplementary material. PCA was performed only at OUH, so in the case of a patient from ZUH choosing PCA, that patient was referred to OUH.
2.2. Variables and data collection
HRQoL was measured using the Danish-language translation of the validated European Organization for the Research and Treatment of Cancer (EORTC) Quality of Life Questionnaire version 3.0 [16]. The EORTC questionnaire includes 30 items, distributed across the following subscales: global health status, five functioning scales (physical [PF], role [RF], emotional [EF], cognitive [CF], and social [SF]), and eight symptom scales (fatigue, nausea/vomiting, pain, dyspnea, insomnia, appetite loss, constipation, and diarrhea), and finally, a scale describing the financial impact of cancer [17]. The latter scale was not analyzed in this study. The scores were calculated based on the EORTC scoring manual [16]. All scale scores range from 0 to 100, with a high score on the functional scales indicating a high level of functioning and a high score on the symptom scales indicating greater severity of individual symptoms. The primary outcome was the change in HRQoL between PN and PCA from baseline to follow-up. The questionnaire was distributed before treatment (baseline), and at 14 and 90 d after treatment. The follow-up cutoffs of 14 and 90 d were chosen to reflect our standard follow-ups and convalescence after treatment. The questionnaire was distributed either electronically or manually (as a paper version), according to patient preferences. With the paper version, the patient filled out the questionnaire and then provided their answers through structured telephone interviews conducted by a third-party research assistant to minimize interviewer bias. Patients who chose the electronic version received automatic e-mail reminders if they did not respond within 72 h. Patients completing the baseline questionnaire were consecutively included in the study cohort. In addition to HRQoL, demographic data were collected with additional questions. Clinical information was collected from patients’ medical records and through patient interviews, including: ECOG performance status [18], complications graded according to the Clavien-Dindo classification [19], histopathology according to the American Joint Committee on Cancer eighth edition for RCC tumor node metastasis (TNM) staging system [20], Charlson Comorbidity Index (CCI) [21], and radius-endophytic-nearness-anterior-location (RENAL) nephrometry score [22].
2.3. Ethical considerations
Prior to inclusion, patients were informed about the purpose of the study, the anonymity and confidentiality of their personal data, and their right to withdraw from the study at any time. The study was approved by the Danish Data Protection Agency (18/52479), and the National Committee on Health Research Ethics waived the need for ethical approval (cf. 0182000-96).
2.4. Statistical analysis
Descriptive statistics are presented as means and standard deviation, medians with interquartile range (IQR), or frequencies. Fisher’s exact test, chi-square test, Student t test, or Wilcoxon rank-sum test were used for comparative analyses. The primary outcome was analyzed by a linear mixed-effect model (restricted maximum likelihood estimation [REML]) with patients as random effects, and treatment (PN vs PCA) and visits (baseline, 14 d, and 90 d) as fixed effects. Standard model selection was performed using the likelihood ratio test. Standard model assumptions were not violated. Adjusted models included age and sex as covariates as these were considered potential confounding factors. Statistical significance was set at 0.05. All analyses were performed using the STATA 16 software (StataCorp LLC, College Station, TX, USA).
3. Results
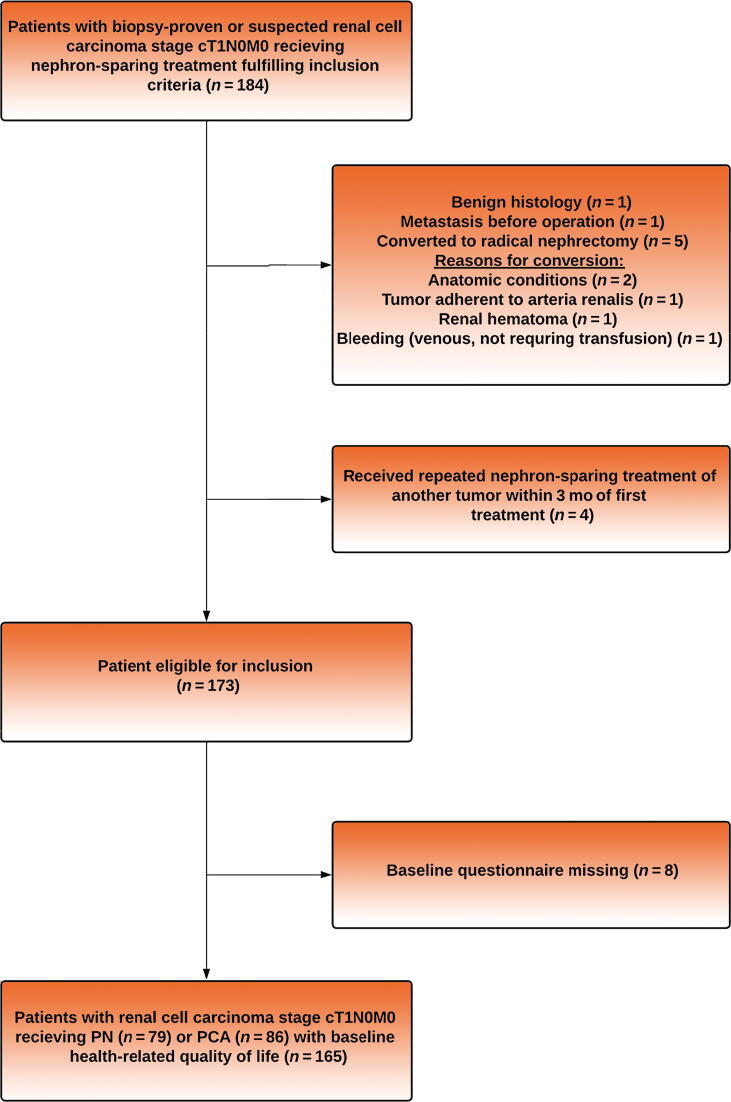
A total of 184 patients with RCC stage cT1N0M0 were scheduled to undergo NSS during the inclusion period. Seven patients, who planned to receive PN, were excluded due to conversion to RN, benign tumor pathology, or metastasis before operation. Three patients who planned to receive PCA had repeated NSS for another tumor within 3 mo of the first treatment, leaving 173 patients eligible to participate in the study. Eight patients either misplaced or did not return the baseline questionnaire, resulting in 165 remaining patients (PN, n = 79 and PCA, n = 86) included in the analysis (CONSORT flowchart, Fig. 1). Patients receiving PCA were significantly older, were more frequently unemployed due to retirement, had worse performance status, had higher CCI, and were more likely to have other malignities at the time of treatment than patients who underwent PN (Table 1). Patients receiving PCA had significantly smaller tumors than those receiving PN. RENAL scores and histological subtypes were comparable between PN and PCA.
Fig. 1.
CONSORT flowchart showing patient inclusion. PCA = percutaneous cryoablation; PN = partial nephrectomy.
Table 1.
Baseline characteristics of patients treated with partial nephrectomy (PN) or percutaneous cryoablation (PCA)
| Patient characteristics | PN (n = 79) | PCA (n = 86) | Difference (p value) |
|---|---|---|---|
| Age (yr), median (IQR) | 62.1 (52.7–71.0) | 69.1 (62.2–76.1) | 0.0005 |
| Male gender, n (%) | 58 (73) | 62 (72) | NS |
| PSa, n (%) | |||
| 0 | 67 (85) | 51 (59) | 0.001 |
| 1 | 10 (13) | 23 (27) | |
| 2 | 2 (3) | 11 (13) | |
| 3 | – | 1 (1) | |
| ASA, n (%) | |||
| 1 | 8 (10) | 6 (7) | NS |
| 2 | 45 (57) | 40 (47) | |
| 3 | 26 (33) | 40 (47) | |
| CCI, median (IQR) | 2 (1–3) | 3 (2–5) | <0.001 |
| Other malignity, n (%) | |||
| None | 68 (86) | 54 (63) | 0.002 |
| Ongoing treatment | 1 (1) | 6 (7) | |
| Follow-up/surveillance | 10 (13) | 26 (30) | |
| Multiple RCC at diagnosis, n (%) | 1 (1) | 5 (6) | NS |
| Symptoms of RCC, n (%) | |||
| None | 67 (85) | 70 (81) | NS |
| Pain | 2 (3) | 2 (2) | NS |
| Gross hematuria | 3 (4) | 6 (7) | NS |
| Fatigue | 3 (4) | 1 (1) | NS |
| Weight loss | 6 (8) | 3 (3) | NS |
| Not registered | 1 (1) | 6 (7) | NS |
| Living alone, n (%) | |||
| Yes | 21 (27) | 21 (24) | NS |
| No | 58 (73) | 65 (76) | |
| Current occupation, n (%) | |||
| Employed | 36 (46) | 19 (22) | 0.001 |
| Not employed | 39 (49) | 66 (77) | |
| Not registered | 4 (5) | 1 (1) | |
| Highest achieved education (yr), n (%) | |||
| Short (7–10) | 22 (28) | 29 (34) | NS |
| Medium (11–13) | 37 (47) | 29 (34) | |
| Long (>13) | 18 (23) | 28 (33) | |
| Not registered | 2 (3) | – | |
| Tumor characteristics | PN (n = 80) | PCA (n = 90) | |
| Tumor size (cm), mean (SD) | 3.75 (1.25) | 3.04 (0.93) | <0.001 |
| Clinical tumor stage, n (%) | 0.001 | ||
| cT1a | 53 (66) | 79 (88) | |
| cT1b | 27 (34) | 11 (12) | |
| Tumor placement, n | |||
| Right/left | 29/51 | 41/49 | NS |
| RENAL score, median (IQR) | 7 (6–9) | 8 (6–9) | NS |
| RENAL score group, n (%) | |||
| 4–6 (low) | 27 (34) | 28 (31) | NS |
| 7–9 (medium) | 47 (59) | 51 (57) | |
| 10–12 (high) | 6 (8) | 11 (12) | |
| Histological subtypeb, n (%) | |||
| Unclassified RCC | 1 (1) | 2 (2) | NS |
| Clear cell | 59 (74) | 57 (63) | |
| Papillary | 11 (14) | 25 (28) | |
| Chromophobe | 5 (6) | 5 (6) | |
| Epithelioid angiomyolipoma | 3 (4) | 1(1) | |
| 1 (1) | – | ||
| Treatment characteristics | PN (n = 79) | PCA (n = 86) | |
| Outpatient procedure, n (%) | |||
| Yes | NA | 81 (94) | |
| No | NA | 5 (6) | |
| Length of stay (d), median (IQR) | 2 (1–2) | 2 (1–3)c | NS |
| Procedure, n (%) | |||
| Robot assisted | 74 (94) | NA | |
| Open | 2 (3) | NA | |
| Converted from robot to open | 3 (4) | NA | |
| Hydro displacement, n (%) | |||
| Yes | NA | 55 (64) | |
| No | NA | 31 (36) | |
| Double-J catheter, n (%) | |||
| Yes | NA | 10 (12) | |
| No | NA | 76 (88) | |
| Anesthesia, n (%) | |||
| Sedation | NA | 81 (94) | |
| GA | 79 (100) | 5 (6) | |
| Multiple tumors treated, n (%) | 1 (1) | 4 (5) | NS |
| Postoperative complications within 90 d, n (%) | 18 (23) | 16 (19) | NS |
| Major postoperative complications (Clavien-Dindo grade ≥3), n (%) | 2 (3) | 5 (6) | NS |
| Mean ± SD | Mean ± SD | ||
| Health-related quality of life outcomes | |||
| Global health status | 66.7 ± 21.2d | 61.8 ± 24.0 | NS |
| Physical functioning | 88.4 ± 16.7 | 76.4 ± 25.0 | 0.0005 |
| Role functioning | 82.7 ± 26.5d | 69.6 ± 35.8 | 0.009 |
| Emotional functioning | 76.3 ± 21.8 | 72.3 ± 22.6 | NS |
| Cognitive functioning | 84.0 ± 20.1 | 82.9 ± 22.0 | NS |
| Social functioning | 90.5 ± 18.0 | 84.3 ± 25.9e | NS |
| Symptoms | |||
| Fatigue | 27.1 ± 24.2d | 35.5 ± 29.0 | 0.0470 |
| Nausea and vomiting | 3.6 ± 9.9 | 4.3 ± 12.0 | NS |
| Pain | 18.6 ± 27.7 | 25.8 ± 30.6 | NS |
| Dyspnea | 14.5 ± 22.5d | 18.0 ± 25.0e | NS |
| Insomnia | 25.7 ± 29.7 | 36.4 ± 35.3 | NS |
| Appetite loss | 9.3 ± 19.9 | 13.2 ± 25.2 | NS |
| Constipation | 7.3 ± 21.9 | 8.9 ± 16.5 | NS |
| Diarrhea | 10.5 ± 19.6 | 16.7 ± 27.1 | NS |
ASA = American Society of Anesthesiologists; CCI = Charlson Comorbidity Index; GA = general anesthesia; IQR = interquartile range; NA = not applicable; NS = not significant; PS = performance status; RCC = renal cell carcinoma; RENAL score = radius-endophytic-nearness-anterior-location nephrometry score; SD = standard deviation.
Performance score according to Eastern Cooperative Oncology Group [18].
Histological subtype based on biopsy for PCA and tumor pathology after PN.
Based on the five patients who did not leave the hospital within 12 h after the procedure.
Seventy eight observations.
Eighty five observations.
Patients receiving PN had significantly higher scores on PF and RF, and reported significantly lower levels of fatigue and insomnia at baseline than those who received PCA. Otherwise, the groups were comparable on HRQoL scores and self-reported symptoms at baseline (Table 1). In addition, 90-d postoperative complications were comparable between PCA and PN.
The completion rates of the questionnaire were 98% at 14-d and 96% at 90-d follow-up. Patients lost to follow-up were considered random for both PN and PCA.
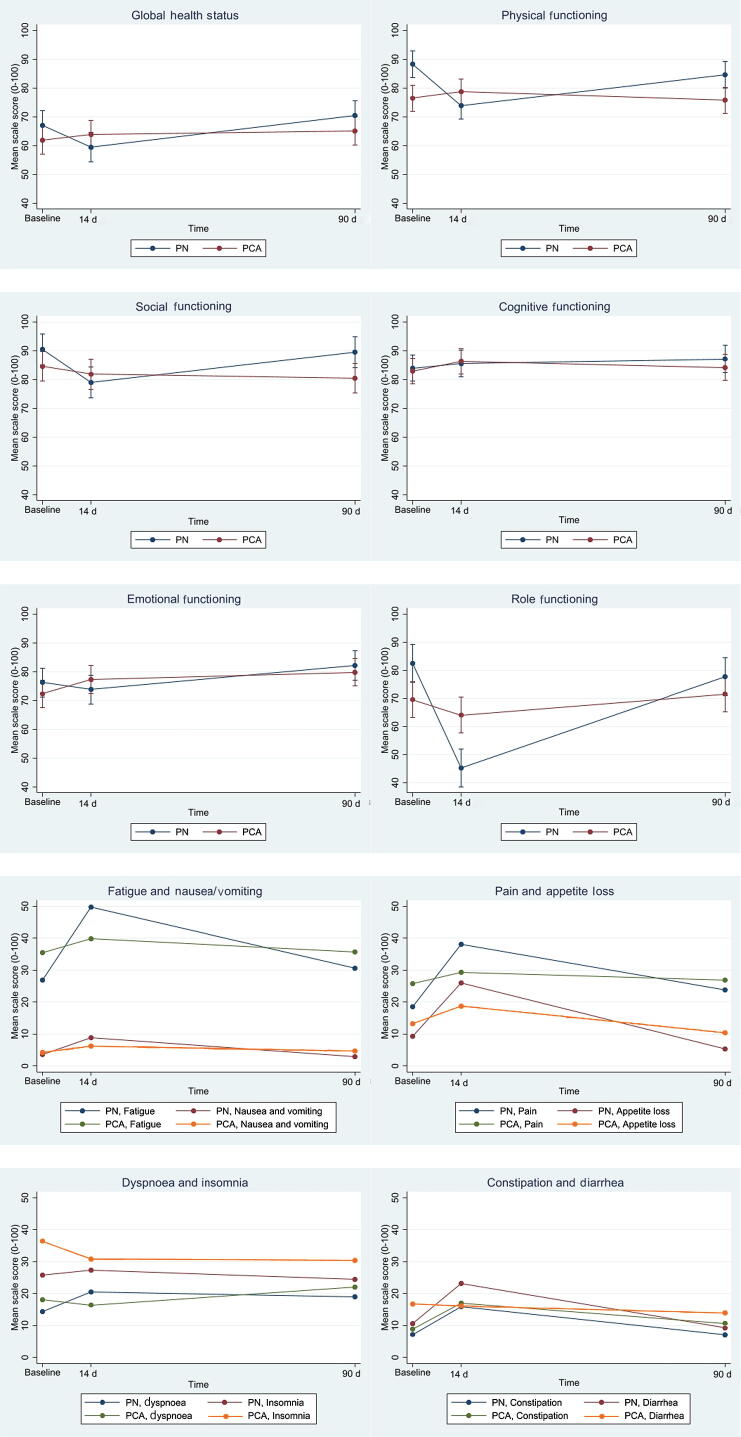
A statistically significant difference between baseline and 14-d follow-up was found for Global health status, PF, RF, and EF, suggesting that PCA either increased or did not decrease as much as PN. Likewise, patients who underwent PN had a significant increase in fatigue, pain, dyspnea, appetite loss, and diarrhea at 14-d follow-up compared with PCA patients (Fig. 2). Overall, the data show that all significant differences observed from baseline to 14-d follow-up were not significant compared with those from baseline to 90-d follow-up. Adjusting for age and sex did not change the results on any scales or symptoms. Table 2 shows the differences from baseline to follow-up between PN and PCA in global health status, the five functioning scales, and the eight symptom scales after 14 and 90 d. For the mean scores for PN and PCA for each scale, the difference between the groups, and the difference from baseline to each follow-up, see Supplementary Table 1.
Fig. 2.
HRQoL scales and self-reported symptoms from baseline up to 90 d postoperatively following partial nephrectomy (PN) and percutaneous cryoablation (PCA). HRQoL = health-related quality of life.
Table 2.
The differences from baseline to follow-up between PN and PCA on GHS, the five functioning scales, and the eight symptom scales after 14 and 90 d
| Difference (95% CI) from baseline to 14 d (PN vs PCA)a | Difference (95% CI) from baseline to 90 d (PN vs PCA)a | |
|---|---|---|
| Overall HRQoL | ||
| Global health status (GHS) | 9.6 (3.2; 16.0) | –0.26 (–6.7; 6.2) |
| Functional scales | ||
| Physical functioning | 16.7 (11.6; 21.8) | 3.1 (–2.0; 8.3) |
| Role functioning | 31.7 (22.6; 40.8) | 6.7 (–2.4; 15.9) |
| Emotional functioning | 7.4 (1.3; 13.6) | 1.6 (–4.5; 7.8) |
| Cognitive functioning | 1.9 (–3.5; 7.4) | –1.9 (–7.4; 3.6) |
| Social functioning | 8.9 (1.9; 16.0) | –3.1 (–10.2; 4.0) |
| Symptoms | ||
| Fatigue | –18.6 (–25.2; –12.0) | –3.4 (–10.1; 3.2) |
| Nausea and vomiting | –3.2 (–8.2; 1.8) | 1.1 (–3.9; 6.2) |
| Pain | –16.0 (–24.5; –7.4) | –4.1 (–12.8; 4.5) |
| Dyspnea | –7.7 (–15.4; –0.02) | –0.7 (–8.4; 7.0) |
| Insomnia | –7.3 (–16.5; 2.0) | –4.8 (–14.1; 4.5) |
| Appetite loss | –11.2 (–19.5; –2.9) | 1.2 (–7.1; 9.5) |
| Constipation | –0.6 (–8.5; 7.3) | 2.0 (–6.0; 9.9) |
| Diarrhea | –13.2 (–20.9; –5.5) | –1.3 (–9.0; 6.4) |
CI = confidence interval; HRQoL = health-related quality of life; PN = partial nephrectomy; PCA = percutaneous cryoablation.
Bold font indicates significant results.
Adjusted for age and sex.
4. Discussion
In this prospective comparative study, we found that patients receiving PN had a statistically significant decrease in global health status, PF, RF, and EF at 14-d follow-up compared with patients receiving PCA. This finding was accompanied by PN having a significant increase in symptom scores on fatigue, pain, dyspnea, appetite loss, and diarrhea at 14-d follow-up. However, this difference was not present at 90-d follow-up in any of the HRQoL scales or self-reported symptoms scores. These results corroborate those reported by Sandbergen et al. [12], who found an initial decrease in HRQoL following surgical treatment of RCC after 1 mo, which gradually improved up to or above baseline after 1 yr, regardless of procedural approaches or treatment modality. The same findings were reported by Becker et al. [23], who found no significant difference in long-term HRQoL outcomes following laparoscopic PN and OPN. Moreover, Breau et al. [6] reported that patients receiving OPN recovered to baseline HRQoL after 12 mo. These previous findings suggest that treatment procedure has a smaller effect on long-term HRQoL. In the present study, we confirmed these findings at 90-d follow-up. Watanabe et al. [5] recently reported a significant or small improvement in HRQoL scores following RAPN after 6–12 mo except for mental health, which improved significantly at 3-mo follow-up.
The significant differences observed at 14-d follow-up in the present study may reflect the nature of the treatment, as PCA is less invasive than PN. Similar results were reported by Sandbergen et al. [12], who found that short-term HRQoL favors minimally invasive procedures. The more significant change in symptom burden observed in the PN group could also affect the changes observed in the functional scales. However, statistical significance does not necessarily indicate a clinically relevant change [16]. Several authors have investigated how to interpret the meaning of small and significant changes in HRQoL scores measured by the EORTC. Osoba et al. [24] used an anchor-based approach that investigated the correlation between patients’ subjective significance and changes in mean scores in the EORTC questionnaire when administered to a population of women treated for breast cancer. The authors reported that a mean change in scores between 5 and 10 corresponded to “a little” change, a mean change in scores between 10 and 20 corresponded to “a moderate” change, and a mean change above 20 corresponded to “very much” change. They did not distinguish between the HRQoL scales [24]. Cocks et al. [25], by contrast, reported that changes in mean scores on the EORTC scales should be interpreted differently with regard to mean changes over time when associated with the minimal clinically important difference (MCID). Furthermore, they showed that estimates of improvements were smaller than those of worsening. Applying the thresholds by Cocks et al. [25] to the results in this study, the changes at 14-d follow-up in the domains of global health status, PF, RF, and pain were medium, whereas the changes in EF, dyspnea, appetite loss, and diarrhea were small. Thus, only the difference in fatigue could be classified as a large change. We found a significant difference within the range of MCID between PN and PCA at 14-d follow-up. A finding that could, however, be considered less relevant for patients when compared with that over a lifespan. Nevertheless, it could aid patients and clinicians in decision-making. Furthermore, given the nonrandomized design, this study cannot conclude whether a comorbid and elderly patient receiving PN would recover to baseline HRQoL after 90 d. Previous reviews exploring quality of life following surgical treatment of RCC have observed that evidence in this field is sparse, partially due to the over-representation of retrospective studies [13], [26]. This situation creates an issue when comparing treatments such as PN and PCA in which, as we have reported in this study, patients have different baseline HRQoL scores. Another factor, when comparing HRQoL results across the literature, is the variety of instruments used, as we recently addressed in a systematic review [27]. To our knowledge, a disease-specific instrument is not yet available to assess HRQoL in patients with localized RCC. Therefore, we chose the EORTC questionnaire based on relevance and the recall period that fit the follow-up period. The high response compliance by patients in this study (98% and 96%) suggests that patients found the EORTC questionnaire a meaningful assessment tool for reporting HRQoL.
Studies have shown that PCA could be associated with an increased risk of treatment failure or recurrence of RCC [28], [29]. Given that this study focused on short-term HRQoL, we did not include information regarding oncological outcomes. However, we reported a mean difference between PN and PCA at 90-d follow-up below the MCID on all HRQoL scales. Thus, a possibly more significant concern regarding whether treatment was successful or not in the PCA group was not detectable in the HRQoL outcome measured in this study.
Patient selection and decision-making in the treatment of RCC stage cT1 have been debated since the use of ablation therapies have become more widespread [30], [31], [32]. We found that patients receiving PCA were significantly older and had worse performance status and more severe comorbidity than patients receiving PN. In addition, patient frailty has been reported to impact perioperative and oncological outcomes [33]. Therefore, preoperative frailty assessment could optimize patient outcomes and is a factor to consider in decision-making. Whereas the most discussed factors revolve around clinical outcomes, this study proposes that patient-reported outcomes in terms of HRQoL should also be considered as part of clinical decision-making when treating RCC stage cT1.
4.1. Limitations
A limitation of the present study is that patients were not randomized, which poses a risk of confounding by indication, potentially compromising internal validity. However, the design followed the clinical practice of providing information about HRQoL outcomes from a broader patient perspective. We argue that this feature, combined with a high response rate throughout the study, contributes to the external and internal validity. Therefore, the magnitude of the indication bias is limited. The inclusion of open procedures poses another limitation since postoperative outcomes after minimally invasive procedures such as RAPN, compared with open PN, may differ, especially in the short-term outcome. However, this study reflects the standard clinical practice and real-life data.
It would have strengthened the results if more patients had been included, generating a larger sample size. However, patients treated with NSS were consecutively recruited for almost 2 yr, from an uptake area covering 2 million people.
5. Conclusions
This prospective multicenter study found a significant difference between baseline and a 14-d follow-up in several HRQoL and symptom scales, favoring PCA over PN. However, we found no significant differences between baseline and 90-d follow-up in HRQoL scales and self-reported symptoms between patients who underwent PN and those who received PCA to treat stage cT1 RCC.
Author contributions: Theresa Junker had full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis.
Study concept and design: Junker, Duus, Rasmussen, Azawi, Nørgaard, Lund, Graumann.
Acquisition of data: Junker, Duus.
Analysis and interpretation of data: Junker, Duus, Nørgaard, Graumann.
Drafting of the manuscript: Junker.
Critical revision of the manuscript for important intellectual content: Junker, Duus, Rasmussen, Azawi, Nørgaard, Lund, Graumann.
Statistical analysis: Junker.
Obtaining funding: Graumann.
Administrative, technical, or material support: None.
Supervision: Rasmussen, Azawi, Nørgaard, Lund, Graumann.
Other: None.
Financial disclosures: Theresa Junker certifies that all conflicts of interest, including specific financial interests and relationships and affiliations relevant to the subject matter or materials discussed in the manuscript (eg, employment/affiliation, grants or funding, consultancies, honoraria, stock ownership or options, expert testimony, royalties, or patents filed, received, or pending), are the following: Theresa Junker: research grant from Boston Scientific. Ole Graumann: speaker honoraria from, advisory board member of, and research grant from Boston Scientific. Others: none declared.
Funding/Support and role of the sponsor: This work was supported by the Region of Southern Denmark, The University of Southern Denmark.
Acknowledgments: The authors gratefully acknowledge all patients who participated in the study.
Associate Editor: M. Carmen Mir
Footnotes
Supplementary data to this article can be found online at https://doi.org/10.1016/j.euros.2022.09.013.
Appendix A. Supplementary data
The following are the Supplementary data to this article:
References
- 1.Padala S.A., Barsouk A., Thandra K.C., et al. Epidemiology of renal cell carcinoma. World J Oncol. 2020;11:79–87. doi: 10.14740/wjon1279. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Ljungberg B, Albiges L, Bensalah K, et al. EAU guidelines on renal cell carcinoma. 2020. https://uroweb.org/wp-content/uploads/EAU-Guidelines-on-Renal-Cell-Carcinoma-2020V3-1.pdf.
- 3.World Health Organization. Ageing and health. 2021. https://www.who.int/news-room/fact-sheets/detail/ageing-and-health.
- 4.Wang Y., Shao J., Lu Y., Li X. Thulium laser-assisted versus conventional laparoscopic partial nephrectomy for the small renal mass. Lasers Surg Med. 2019;52:402–407. doi: 10.1002/lsm.23153. [DOI] [PubMed] [Google Scholar]
- 5.Watanabe H., Motoyama D., Sato R., et al. Health-related quality of life in patients with small renal mass who underwent robot-assisted partial nephrectomy: a prospective evaluation. J Endourol. 2021;35:1644–1649. doi: 10.1089/end.2021.0254. [DOI] [PubMed] [Google Scholar]
- 6.Breau R.H., Fergusson D.A., Knoll G., et al. Hypothermia during partial nephrectomy for patients with renal tumors: a randomized controlled trial. J Urol. 2021;205:1303–1309. doi: 10.1097/JU.0000000000001517. [DOI] [PubMed] [Google Scholar]
- 7.Clark P.E., Schover L.R., Uzzo R.G., Hafez K.S., Rybicki L.A., Novick A.C. Quality of life and psychological adaptation after surgical treatment for localized renal cell carcinoma: impact of the amount of remaining renal tissue. Urology. 2001;57:252–256. doi: 10.1016/s0090-4295(00)00927-4. [DOI] [PubMed] [Google Scholar]
- 8.Shinohara N., Harabayashi T., Sato S., Hioka T., Tsuchiya K., Koyanagi T. Impact of nephron-sparing surgery on quality of life in patients with localized renal cell carcinoma. Eur Urol. 2001;39:114–119. doi: 10.1159/000052422. [DOI] [PubMed] [Google Scholar]
- 9.Ficarra V., Novella G., Sarti A., et al. Psycho-social well-being and general health status after surgical treatment for localized renal cell carcinoma. Int Urol Nephrol. 2002;34:441–446. doi: 10.1023/a:1025683306449. [DOI] [PubMed] [Google Scholar]
- 10.Althaus A.B., Chang P., Mao J., et al. Patient-reported quality of life and convalescence after minimally invasive kidney cancer surgery. Urology. 2020;144:123–129. doi: 10.1016/j.urology.2020.06.020. [DOI] [PubMed] [Google Scholar]
- 11.Alam R., Patel H.D., Osumah T., et al. Comparative effectiveness of management options for patients with small renal masses: a prospective cohort study. BJU Int. 2019;123:42–50. doi: 10.1111/bju.14490. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Sandbergen L., Spriensma A.S., de la Rosette J.J., Laguna M.P. Health-related quality of life in localized renal masses: a matter of sparing nephrons or minimizing the incision? Urol Oncol. 2020;38:43.e1–43.e11. doi: 10.1016/j.urolonc.2019.09.017. [DOI] [PubMed] [Google Scholar]
- 13.MacLennan S., Imamura M., Lapitan M.C., et al. Systematic review of perioperative and quality-of-life outcomes following surgical management of localised renal cancer. Eur Urol. 2012;62:1097–1117. doi: 10.1016/j.eururo.2012.07.028. [DOI] [PubMed] [Google Scholar]
- 14.von Elm E., Altman D.G., Egger M., Pocock S.J., Gøtzsche P.C., Vandenbroucke J.P. Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement: guidelines for reporting observational studies. BMJ. 2007;335:806–808. doi: 10.1136/bmj.39335.541782.AD. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Dam M., Hansen C., Poulsen T.D., et al. Transmuscular quadratus lumborum block reduces opioid consumption and prolongs time to first opioid demand after laparoscopic nephrectomy. Reg Anesth Pain Med. 2021;46:18–24. doi: 10.1136/rapm-2020-101745. [DOI] [PubMed] [Google Scholar]
- 16.Fayers PM, Aaronson NK, Bjordal K, et al. The EORTC QLQ-C30 scoring manual. ed. 3. 2001. https://www.eortc.org/app/uploads/sites/2/2018/02/SCmanual.pdf.
- 17.Aaronson N.K., Ahmedzai S., Bergman B., et al. The European Organization for Research and Treatment of Cancer QLQ-C30: a quality-of-life instrument for use in international clinical trials in oncology. J Natl Cancer Inst. 1993;85:365–376. doi: 10.1093/jnci/85.5.365. [DOI] [PubMed] [Google Scholar]
- 18.Oken M.M., Creech R.H., Tormey D.C., et al. Toxicity and response criteria of the Eastern Cooperative Oncology Group. Am J Clin Oncol. 1982;5:649–655. [PubMed] [Google Scholar]
- 19.Dindo D., Demartines N., Clavien P.-A. Classification of surgical complications: a new proposal with evaluation in a cohort of 6336 patients and results of a survey. Ann Surg. 2004;240:205–213. doi: 10.1097/01.sla.0000133083.54934.ae. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Rini B.I., McKiernan J., Chang S.S., et al. In: AJCC cancer staging manual. ed. 8. Amin M.B., Edge S.B., Greene F.L., editors. Springer; Berlin, Germany: 2017. Kidney; pp. 739–755. [Google Scholar]
- 21.Charlson M.E., Pompei P., Ales K.L., MacKenzie C.R. A new method of classifying prognostic comorbidity in longitudinal studies: development and validation. J Chronic Dis. 1987;40:373–383. doi: 10.1016/0021-9681(87)90171-8. [DOI] [PubMed] [Google Scholar]
- 22.Kutikov A., Uzzo R.G. The R.E.N.A.L. nephrometry score: a comprehensive standardized system for quantitating renal tumor size, location and depth. J Urol. 2009;182:844–853. doi: 10.1016/j.juro.2009.05.035. [DOI] [PubMed] [Google Scholar]
- 23.Becker A., Kluth L.A., Schmid M., et al. Laparoscopic vs open partial nephrectomy for clinical T1 renal masses: no impact of surgical approach on perioperative complications and long term post-operative quality of life. Eur Urol Suppl. 2014;13:e426. doi: 10.1007/s00345-014-1318-1. [DOI] [PubMed] [Google Scholar]
- 24.Osoba D., Rodrigues G., Myles J., Zee B., Pater J. Interpreting the significance of changes in health-related quality-of-life scores. J Clin Oncol. 1998;16:139–144. doi: 10.1200/JCO.1998.16.1.139. [DOI] [PubMed] [Google Scholar]
- 25.Cocks K., King M.T., Velikova G., et al. Evidence-based guidelines for interpreting change scores for the European Organisation for the Research and Treatment of Cancer Quality of Life Questionnaire Core 30. Eur J Cancer. 2012;48:1713–1721. doi: 10.1016/j.ejca.2012.02.059. [DOI] [PubMed] [Google Scholar]
- 26.Rossi S.H., Klatte T., Stewart G.D. Quality of life outcomes in patients with localised renal cancer: a literature review. World J Urol. 2018;36:1961–1972. doi: 10.1007/s00345-018-2415-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Junker T., Duus L., Rasmussen B.S., et al. Quality of life and complications after nephron sparing treatment of renal cell carcinoma stage T1—a systematic review. BMC Syst Rev. 2022;11:4. doi: 10.1186/s13643-021-01868-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Zargar H., Atwell T.D., Cadeddu J.A., et al. Cryoablation for small renal masses: selection criteria, complications, and functional and oncologic results. Eur Urol. 2016;69:116–128. doi: 10.1016/j.eururo.2015.03.027. [DOI] [PubMed] [Google Scholar]
- 29.Turna B., Kaouk J.H., Frota R., et al. Minimally invasive nephron sparing management for renal tumors in solitary kidneys. J Urol. 2009;182:2150–2157. doi: 10.1016/j.juro.2009.07.066. [DOI] [PubMed] [Google Scholar]
- 30.Campi R., Mari A., Minervini A., Serni S. L'Essentiel est Invisible pour les Yeux: the art of decision-making and the mission of patient-centred care for patients with localised renal masses. Eur Urol. 2021;80:589–591. doi: 10.1016/j.eururo.2021.02.027. [DOI] [PubMed] [Google Scholar]
- 31.Chandrasekar T., Boorjian S.A., Capitanio U., Gershman B., Mir M.C., Kutikov A. Collaborative review: factors influencing treatment decisions for patients with a localized solid renal mass. Eur Urol. 2021;80:575–588. doi: 10.1016/j.eururo.2021.01.021. [DOI] [PubMed] [Google Scholar]
- 32.Zondervan P.J., van Lienden K.P., van Delden O.M., de la Rosette J.J., Laguna M.P. Preoperative decision making for nephron-sparing procedure in the renal mass: time for using standard tools? J Endourol. 2016;30:128–134. doi: 10.1089/end.2015.0472. [DOI] [PubMed] [Google Scholar]
- 33.Campi R., Berni A., Amparore D., et al. Impact of frailty on perioperative and oncologic outcomes in patients undergoing surgery or ablation for renal cancer: a systematic review. Minerva Urol Nephrol. 2022;74:146–160. doi: 10.23736/S2724-6051.21.04583-3. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.