Abstract
Background/Aims:
The most common cancer among men and the third most common among women in Saudi Arabia is colon cancer. This study aimed to evaluate the yield of the fecal occult blood test (FOBT) used as a screening tool for colon cancer.
Settings and Design:
Medical records of all patients above the age of 50 years who had visited the Family Medicine clinic at King Faisal Specialist Hospital and Research Centre in Riyadh, Saudi Arabia and who had undergone the FOBT screening between January 2002 and March 2017 were retrospectively reviewed.
Materials and Methods:
We collected patients’ demographic data, FOBT results, and colonoscopy pathology reports results.
Statistical Analysis:
Data were analyzed using the Statistical Package for Social Science Version 25 (IBM Corp., Armonk, NY, USA).
Results:
We included 2179 patients in this study. The mean age was 59.8 ± 8 years. As a result of the FOBT screening, 19.7% of the patients had a positive FOBT. Colon cancer accounted for 3.5% of all abnormal colonoscopy pathology reports. The Saudi age-standardized incidence rate (ASIR) in our study was 26.56 per 100,000 persons.
Conclusion:
FOBT is a feasible tool that can be used for colon cancer screening in primary care settings. The Saudi ASIR in our study was higher than the Saudi Cancer Registry, but it was still lower in comparison to other countries. We recommend the establishment of a national community-based colon cancer screening program.
Keywords: Colon cancer, colonic polyps, colonoscopy, FOBT, Saudi Arabia, screening
Background
Colon cancer is one of the most common cancers worldwide. It is ranked the third most common cancer among men and the second most common type of cancer among women.[1] In Saudi Arabia, colon cancer has become the most common cancer among men and the third most common cancer among women.[2] The overall age-standardized incidence rate (ASIR) for colon cancer in Saudi Arabia was reported to be 9.6 per 100,000,[3] which is considered to be very low in comparison to several developed and developing countries.[4] The 5-year overall survival rate is low in Saudi Arabia in comparison to developed countries; this has been attributed to the higher proportion of cancer presenting at an advanced stage and the absence of screening programs.[3]
Multiple modalities have been used as screening tools for colon cancer, ranging from simple methods to more invasive techniques, including fecal occult blood test (FOBT), sigmoidoscopy, computed tomography (CT) colonography, and colonoscopy.[5,6] Primary care physicians play a significant role in selecting the screening modalities when screening for colon cancer. The literature provides evidence of the effectiveness of FOBT usage in comparison to colonoscopy for the early detection of colon cancer.[7,8] FOBT is a simple, widely accepted, non-invasive, and efficient method for colon cancer screening.[9,10]
The most recent updated guidelines for colon cancer screening, which were published by the American College of Gastroenterology in 2017, recommend screening asymptomatic patients starting at the age of 50 years by using either a colonoscopy every ten years or a fecal immunochemical test (FIT) annually as first-tier options.[6] The second-tier options were either 5-yearly CT colonography, 3-yearly FIT–fecal DNA, or a flexible sigmoidoscopy every 5–10 years.[6] Capsule endoscopy should be offered as a third-tier option if the patient refuses all the previously mentioned modalities.[6] Community-based studies evaluating FOBT screening in Saudi Arabia are lacking, we conducted this study to evaluate the yield of FOBT used as a screening tool for colon cancer.
Methods
Primary objectives
To evaluate the yield of FOBT in colon cancer screening.
A total of 2179 patients were included in the study. Data for all patients over the age of 50 years, who visited the Family Medicine clinics linked to King Faisal Specialist Hospital and Research Centre (KFSH&RC) in Riyadh, Saudi Arabia, and who underwent FOBT from January 2002 until March 2017 were reviewed retrospectively from their electronic medical records. We collected the patients’ demographic data and lab results, including the FOBT results. In our study, the laboratory utilized the Hemosure one-step immunological FOBT, an immunospecific sandwich assay that is not affected by dietary peroxidases, animal blood, or ascorbic acid. If a patient had more than one FOBT, we considered the result to be positive if one sample came back as positive.
Patient age, date, stage of colon cancer at diagnosis, and treatment received (including medical, surgical, and radiotherapy treatments) were all reviewed. We referred to the tumor-node-metastasis (TNM) staging system for colon cancer staging.[11]
The clinical pathway for colon screening in our clinics follows the United States Preventive Services Task Force guidelines; a patient with a positive FOBT is referred for a colonoscopy; a patient with a negative FOBT undergoes an annual FOBT screening. Patients diagnosed with colon cancer are referred to and followed up by the oncology services, in addition to being seen by their primary care physicians.
Ethical considerations
This research was conducted in accordance with international ethical principles experimental research, as referenced in the Declaration of Helsinki (2000) of the World Medical Association. The research proposal was approved by the Research Advisory Committee (RAC), which is the institutional review board of KFSH&RC. The research complies with the international and national research guidelines of the Kingdom of Saudi Arabia (RAC# 2171-058).
Statistical analysis
Descriptive analysis was used for the continuous variables, including median, mean, and standard deviation; the categorical variables were represented as frequencies and percentages. The continuous variables were analyzed using the t test. The Chi-squared test was used to compare the categorical variables. Univariate and multivariate regression analyses were used to evaluate the role of different risk factors in the development of colon cancer. Data were analyzed using the Statistical Package for Social Science (SPSS) Version 25 (IBM Corp., Armonk, NY, USA). The adenoma detection rate (ADR) is the percentage of patients who underwent a colonoscopy and had one or more adenomas detected and removed. The person-years at risk was defined as the difference in time between the date of the first FOBT and the date of either the colon cancer diagnosis, the patient’s death, or the patient’s last hospital visit. The Saudi ASIR was calculated based on the Sagi 1996 world population.[12] The level of statistical significance was set at P < 0.05.
Results
The mean age of the 2179 patients in our study was 59.8 ± 8 years; of those, 44.7% were males. Saudi nationals represented 73% of the study population. The mean body mass index (BMI) of the study population was 30 ± 6 kg/m2 [Table 1].
Table 1.
Patient demographics
| Gender | Nationality | ||
|---|---|---|---|
|
|
|
||
| Males | Females | Saudis | Non-Saudis |
| 44.7% | 55.3% | 73% | 23% |
| Age | 59.8±8 | ||
| BMI* | 30±6 kg/m2 | ||
Body Max Index*
Among the study population, 19.7% had a positive FOBT. The average number of FOBT tests per patient was 4. The median time between the first positive FOBT and colonoscopy was 5 months. Among all patients who had a positive FOBT, 52% actually underwent colonoscopy. The other 48% either did not adhere to the plan (no show, did not confirm the appointment, or declined the procedure) or had a future appointment. Colonoscopy was the most commonly ordered investigation for patients that underwent FOBT [Table 2]. Of the study population, 18% (n = 394) of the patients underwent colonoscopy; the findings were abnormal in 67% of those patients. Colonic polyps accounted for 47.9% of all abnormal colonoscopy pathology reports [Figure 1]. Tubular adenoma with low grade dysplasia was the most common type of colonic polyp, followed by hyperplastic polyps. Among the patients that underwent screening colonoscopy in our study, the ADR was 34%. Of those who had an abnormal colonoscopy, 23.7% underwent a follow-up colonoscopy. The rate of follow-up colonoscopy in patients with a normal colonoscopy was 7.5%. Only one patient had bowel perforation as a complication of the colonoscopy procedure.
Table 2.
The most common investigations yielded from the fecal occult blood test (FOBT)
| Type | n | % |
|---|---|---|
| Colonoscopy | 394 | 18.1% |
| Gastroscopy | 243 | 11.2% |
| CT* Scan | 157 | 7.2% |
| CT* Colonography | 19 | 0.9% |
| Barium Enema | 5 | 0.5% |
*Computed tomography
Figure 1.
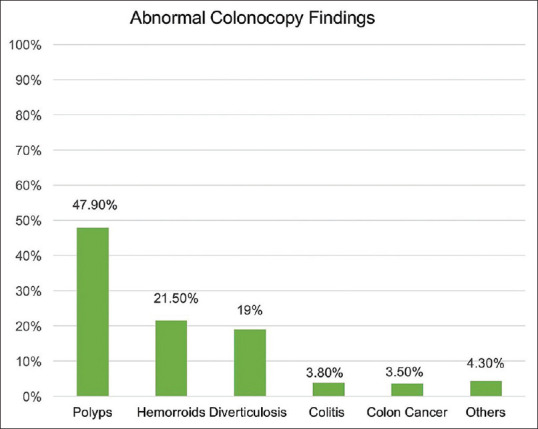
Pathology report of abnormal colonoscopy findings
The prevalence of colon cancer diagnosis in our screened population was 0.5%. In our study, the median age at presentation for colon cancer was 65.7 ± 7 years (males: 66.1 ± 9 years, females: 62.4 ± 7 years). The Saudi ASIR for colon cancer in our study population was 26.56 per 100,000. Adenocarcinoma was found exclusively on the histopathological colonoscopy reports. According to the TNM cancer staging system, five patients had T4 cancer, five patients had T3 cancer, and one patient had T2 cancer. All patients who were diagnosed with colon cancer received treatment with either chemotherapy, radiotherapy, surgery, or a specific combination of these treatments. The Saudi 5-year overall survival rate for colon cancer in our study was 49.72%.
Discussion
In our study, we found that the ASIR for colon cancer in Saudi nationals was 26.56 per 100,000. This finding is higher than the ASIR for colon cancer reported by the Saudi Cancer Registry, which was 9.6 per 100,000 in 2010.[3] The Saudi ASIR reported in our study is still in the lower range in comparison to several developed and developing countries[3,4] [Figure 2]. This finding could be related to the absence of structured screening programs in the community as well as under-reporting and under-diagnosing, especially from peripheral hospitals in the country.
Figure 2.
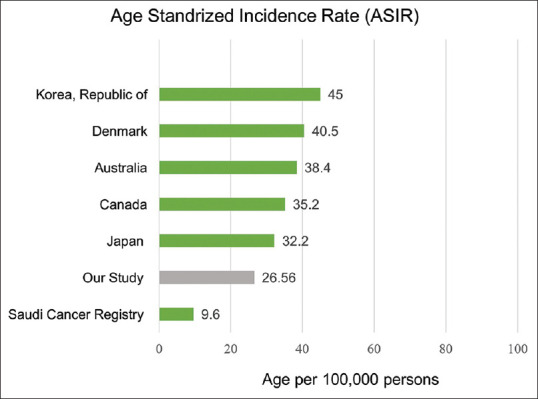
The age standardized incidence rate of Colorectal cancer per 100,000 persons as compared to other countries[3,4]
ADR is a well-established, gold-standard quality indicator for colonoscopy.[13] It has been reported that the benchmark for ADR is 25% overall, 30% for men, and 20% for women.[14] However, ADR can range from 7.4% to 52.5% in screening colonoscopy.[15] A study conducted by Almadi et al.[16] that evaluated the prevalence and characteristics of colonic polyps in Saudi Arabia showed that the ADR in screening colonoscopy was 22.9%. Almadi et al.[16] also reviewed the ADR in Saudi Arabia locally and regionally and found it to be low in comparison to populations in Western countries. However, similar rates were reported in Brazil.[17] Figure 3 illustrates the comparison between the ADR in our study and the ADR from other studies undertaken within our region and worldwide.[16,17,18,19,20,21,22] The ADR in our study was 34%, which is considered to be above the standard. The ADR reported in a study conducted in Portugal was also found to be above the standard ADR; it was slightly higher than the 34% ADR found in our study.[18] One reason that might explain a higher-than-expected ADR in our study is the patient population that underwent colonoscopy. The majority of colonoscopies were performed on FOBT-positive patients; 52% of FOBT-positive patients underwent colonoscopy, whereas only 9.8% of FOBT-negative patients proceeded to colonoscopy. This would mean that the colonoscopies were carried out on a higher risk population; thus, the ADR would be higher than expected in a normal screening population.
Figure 3.
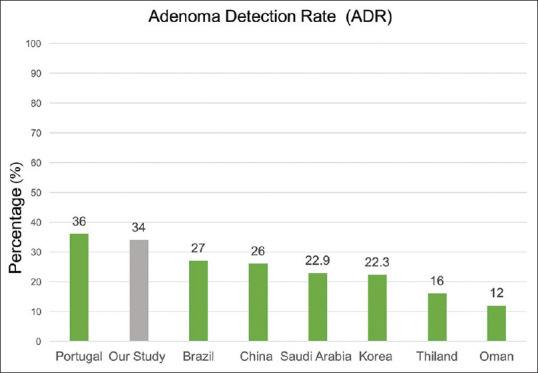
Comparison of Adenoma Detection Rate percentage reported in different countries[16,17,18,19,20,21,22]
Adenocarcinoma was found to be the most common histopathological finding in colon cancer patients.[23] Our findings are consistent with the results reported in the literature regarding the histopathological nature of colon cancer. Several authors have reported that the rate of adenocarcinoma in patients diagnosed with colon cancer can be 95% or higher.[24,25] A retrospective study conducted at a referral center in Tabuk, Saudi Arabia found that 98.6% of the patients diagnosed with colon cancer had adenocarcinoma.[24] Similarly, a population-based study conducted in the United States showed that 96% of the colon cancer cases were adenocarcinoma.[25]
During our study period (2002–2017), the Saudi 5-year survival for colon cancer was 49.72%. The 5-year survival reported by Alsanea et al.[3] in the 1994–2004 time period was 44.6%. Thus, our findings are reasonably consistent with that study’s findings; the 5-year survival was below 50% in both studies, which is considered low in comparison to developed countries. We observed that our patients were often reluctant to proceed with a colonoscopy once they had positive FOBT results. We believe this to be a contributing factor to the advanced cancer stage at diagnosis in our study, as well as the low survival rate. In Western countries, the survival rate is much higher. For example, the 5-year survival in the United States was 65% between 2006 and 2012.[26]
Conclusion
The Saudi ASIR in our study is higher than the ASIR reported in the Saudi Cancer Registry,[3] but it is still less than the ASIR in other countries.[4] There is a need to establish structured screening programs for colon cancer in Saudi Arabia. Moreover, there is a need for larger, national studies to investigate the prevalence and incidence of colon cancer in Saudi Arabia.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
- 1.World Health Organization and International Agency for Research on Cancer. Estimated Cancer Incidence, Mortality and Prevalence Worldwide in 2012. Lyon, France: Lung Cancer GLOBOCAN; 2012. [Google Scholar]
- 2.Bazarbashi S, Al Eid H, Minguet J. Cancer incidence in Saudi Arabia: 2012 data from the saudi cancer registry. Asian Pac J Cancer Prev. 2017;18:2437–44. doi: 10.22034/APJCP.2017.18.9.2437. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Alsanea N, Abduljabbar AS, Alhomoud S, Ashari LH, Hibbert D, Bazarbashi S. Colorectal cancer in Saudi Arabia: Incidence, survival, demographics and implications for national policies. Ann Saudi Med. 2015;35:196–202. doi: 10.5144/0256-4947.2015.196. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Ferlay J, Soerjomataram I, Dikshit R, Eser S, Mathers C, Rebelo M, et al. Cancer incidence and mortality worldwide: Sources, methods and major patterns in GLOBOCAN 2012. Int J Cancer. 2015;136:E359–86. doi: 10.1002/ijc.29210. [DOI] [PubMed] [Google Scholar]
- 5.Alsanea N, Almadi MA, Abduljabbar AS, Alhomoud S, Alshaban TA, Alsuhaibani A, et al. National guidelines for colorectal cancer screening in Saudi Arabia with strength of recommendations and quality of evidence. Ann Saudi Med. 2015;35:189–95. doi: 10.5144/0256-4947.2015.189. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Rex DK, Boland CR, Dominitz JA, Giardiello FM, Johnson DA, Kaltenbach T, et al. Colorectal cancer screening: Recommendations for physicians and patients from the US multi-society task force on colorectal cancer. Am J Gastroenterol. 2017;112:1016–30. doi: 10.1038/ajg.2017.174. [DOI] [PubMed] [Google Scholar]
- 7.Van Kleek E, Liu S, Conn LM, Hoadley A, Ho SB. Improving the effectiveness of fecal occult blood testing in a primary care clinic by direct colonoscopy referral for positive tests. J Healthc Qual. 2010;32:62–9. doi: 10.1111/j.1945-1474.2009.00071.x. [DOI] [PubMed] [Google Scholar]
- 8.Elmunzer BJ, Singal AG, Sussman JB, Deshpande AR, Sussman DA, Conte ML, et al. Comparing the effectiveness of competing tests for reducing colorectal cancer mortality: A network meta-analysis. Gastrointest Endosc. 2015;81:700–9.e3. doi: 10.1016/j.gie.2014.10.033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Wong CK, Lam CL, Wan YF, Fong DY. Cost-effectiveness simulation and analysis of colorectal cancer screening in Hong Kong Chinese population: Comparison amongst colonoscopy, guaiac and immunologic fecal occult blood testing. BMC Cancer. 2015;15:705. doi: 10.1186/s12885-015-1730-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.van Ballegooijen M, Habbema JD, Boer R, Zauber AG, Brown ML. Rockville MD, USA: Agency for Healthcare Research and Quality; 2003. A Comparison of the Cost-Effectiveness of Fecal Occult Blood Tests with Different Test Characteristics in the Context of Annual Screening in the Medicare Population. [PubMed] [Google Scholar]
- 11.Cancer Staging by National Cancer Institute. (n.d.) Retrieved from: https://www.cancer.gov/about-cancer/diagnosis-staging/staging .
- 12.Segi M, Kurihara M, Tsukahara Y. Sendai, Japan: Tohoku University, School of Medicine. Department of Public Health; 1966. Cancer Mortality for Selected Sites in 24 Countries: No 4 (1962-63) [Google Scholar]
- 13.Liem B, Gupta N. Adenoma detection rate: The perfect colonoscopy quality measure or is there more? Transl Gastroenterol Hepatol. 2018;3:19. doi: 10.21037/tgh.2018.03.04. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Rex DK, Schoenfeld PS, Cohen J, Pike IM, Adler DG, Fennerty MB, et al. Quality indicators for colonoscopy. Gastrointest Endosc. 2015;81:31–53. doi: 10.1016/j.gie.2014.07.058. [DOI] [PubMed] [Google Scholar]
- 15.Corley DA, Jensen CD, Marks AR, Zhao WK, Lee JK, Doubeni CA, et al. Adenoma detection rate and risk of colorectal cancer and death. N Engl J Med. 2014;370:1298–306. doi: 10.1056/NEJMoa1309086. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Almadi MA, Alharbi O, Azzam N, Wadera J, Sadaf N, Aljebreen AM. Prevalence and characteristics of colonic polyps and adenomas in 2654 colonoscopies in Saudi Arabia. Saudi J Gastroenterol. 2014;20:154–61. doi: 10.4103/1319-3767.132986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Cardoso DM, Botacin MA, Mekdessi MA. Botacin MA, Mekdessi MA. Adenoma detection rate evaluation and quality of colonoscopy in the Center-West region of Brazil. Arq Gastroenterol. 2017;54:315–20. doi: 10.1590/S0004-2803.201700000-47. [DOI] [PubMed] [Google Scholar]
- 18.Oliveira Ferreira A, Fidalgo C, Palmela C, Costa Santos MP, Torres J, Nunes J, et al. Adenoma detection rate: I will show you mine if you show me yours. GE Port J Gastroenterol. 2017;24:61–7. doi: 10.1159/000450901. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Leung WK, Tang V, Lui PC. Detection rates of proximal or large serrated polyps in Chinese patients undergoing screening colonoscopy. J Dig Dis. 2012;13:466–71. doi: 10.1111/j.1751-2980.2012.00621.x. [DOI] [PubMed] [Google Scholar]
- 20.Choi YS, Suh JP, Lee DS, Youk EG, Lee IT, Lee SH, et al. Colonoscopy screening for individuals aged 40-49 years with a family history of stomach cancer in Korea. Int J Colorectal Dis. 2010;25:443–7. doi: 10.1007/s00384-009-0855-3. [DOI] [PubMed] [Google Scholar]
- 21.Aswakul P, Prachayakul V, Lohsiriwat V, Bunyaarunnate T, Kachintorn U. Screening colonoscopy from a large single center of Thailand-something needs to be changed? Asian Pac J Cancer Prev. 2012;13:1361–4. doi: 10.7314/apjcp.2012.13.4.1361. [DOI] [PubMed] [Google Scholar]
- 22.Ashktorab H, Brim H, Al-Riyami M, Date A, Al-Mawaly K, Kashoub M, et al. Sporadic colon cancer: Mismatch repair immunohistochemistry and microsatellite instability in Omani subjects. Dig Dis Sci. 2008;53:2723–31. doi: 10.1007/s10620-007-0189-3. [DOI] [PubMed] [Google Scholar]
- 23.Marzouk O, Schofield J. Review of histopathological and molecular prognostic features in colorectal cancer. Cancers (Basel) 2011;3:2767–810. doi: 10.3390/cancers3022767. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Abdullah Omer AA. The clinical and pathological features of colorectal cancer in Tabuk region, Saudi Arabia: Trends in the young and elderly patients. Int J Surg Surgical Porced. 2017;2:122. [Google Scholar]
- 25.Stewart SL, Wike JM, Kato I, Lewis DR, Michaud F. A population-based study of colorectal cancer histology in the United States 1998-2001. Cancer. 2006;107(5 Suppl):1128–41. doi: 10.1002/cncr.22010. [DOI] [PubMed] [Google Scholar]
- 26.Siegel RL, Miller KD, Jemal A. Cancer statistics 2017. CA Cancer J Clin. 2017;67:7–30. doi: 10.3322/caac.21387. [DOI] [PubMed] [Google Scholar]


