Abstract
Background:
Hypertension and angiotensin-converting enzyme inhibitors (ACEIs)/angiotensin receptor blockers (ARBs) have been reported to be associated with the prognosis of COVID-19, but the findings remain controversial. Here, we conducted a systematic review to summarize the current evidence.
Methods:
We retrieved all the studies by MEDLINE via PubMed, CENTRAL, and Embase using the MeSH terms until 30 April 2021. A fixed or random effect model was applied to calculate pooled adjusted odds ratio (AOR) with 95% confidence interval (CI). Interactive analysis was performed to identify the interaction effect of hypertension and age on in-hospital mortality.
Results:
In total, 86 articles with 18 775 387 COVID-19 patients from 18 countries were included in this study. The pooled analysis showed that the COVID-19 patients with hypertension had increased risks of in-hospital mortality and other adverse outcomes, compared with those without hypertension, with an AOR (95% CI) of 1.36 (1.28–1.45) and 1.32 (1.24–1.41), respectively. The results were mostly repeated in countries with more than three independent studies. Furthermore, the effect of hypertension on in-hospital mortality is more evident in younger and older COVID-19 patients than in 60–69-year-old patients. ACEI/ARBs did not significantly affect the mortality and adverse outcomes of COVID-19 patients, compared with those receiving other antihypertensive treatments.
Conclusion:
Hypertension is significantly associated with an increased risk of in-hospital mortality and adverse outcomes in COVID-19. The effect of hypertension on in-hospital mortality among consecutive age groups followed a U-shaped curve. ACEI/ARB treatments do not increase in-hospital mortality and other poor outcomes of COVID-19 patients with hypertension.
Keywords: adverse outcomes, angiotensin-converting enzymes/angiotensin–renin blocker treatment, coronavirus disease 2019, hypertension, mortality
BACKGROUND
The outbreak of coronavirus disease 2019 (COVID-19) caused by the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection resulted in public health crisis worldwide. Globally, as of 1747 h. CET, 29 April 2022, there have been 510 270 667 confirmed COVID-19 cases, including 6 233 526 deaths, reported to WHO [1]. The overall crude fatality rate was approximately 1.22%. A number of studies have shown that ageing, immune and inflammation, and underlying comorbidities are the most common factors associated with the mortality and severity of COVID-19 [2–4]. Hypertension is a common health issue in adults, especially in elderly populations. However, the findings from the published studies about the effect of hypertension on the prognosis of COVID-19 remain controversial. Some studies showed COVID-19 patients with hypertension had a higher risk of developing severe COVID-19 or mortality, compared with those without hypertension [5–7]. Conversely, hypertension did not have a significant effect on the prognosis of COVID-19 in another group of researches [8–10]. Furthermore, as SARS-CoV-2 infected patients of all ages were at risk of dying from COVID-19, few studies illustrated the effect of comorbidities on the mortality in different age groups. Thus, an interactive effect of hypertension and age on the mortality of COVID-19 remains to be determined. In addition, evidence regarding the effect of antihypertensive treatments on clinical outcomes is still limited in real world, especially for angiotensin-converting enzyme inhibitors (ACEIs) and angiotensin receptor blockers (ARBs). ACEIs and ARBs are the two types of renin–angiotensin–aldosterone system (RAAS) inhibitors widely used for the treatment of hypertension. Previous researches showed that ACEI/ARBs could increase the expression of angiotensin-converting enzyme 2 (ACE2), which is required for SARS-CoV-2 entry and propagation [11,12]. Hence, it has been speculated that COVID-19 patients receiving RAAS inhibitors had an increased risk of adverse clinical outcome [13,14], although this hypothesis was not supported by some other studies [15,16]. To clarify the effects of hypertension and age as well as the use of ACEI/ARB treatment on the outcomes of COVID-19, we conducted a systematic review and meta-analysis to provide more reliable and comprehensive evidence.
METHODS
Protocol registration
We previously registered the protocol for this systematic review on PROSPERO (https://www.crd.york.ac.uk/PROSPERO, ID: CRD42021274578).
Search strategy
A systematic literature searching was performed using MEDLINE via PubMed, the Cochrane Central Register of Controlled Trials (CENTRAL), and Embase using the medical subject heading (MeSH) terms. The relevant literature published before 30 April 2021 was all retrieved. There were no restrictions on study design, publication type, or language. Non-English studies were translated into English after the inclusion. The searching terms were as follows: Corona Virus Disease-2019, 2019 novel coronavirus, SARS-CoV-2, COVID-19, 2019-nCoV, antihypertensive treatment, hypertension with COVID-19, ACEI, ARB, and RAAS.
Selection criteria
The inclusion criteria for the articles were as follows: studies that included COVID-19 patients with or without hypertension/antihypertensive treatment; endpoints of the studies related to in-hospital mortality and/or other adverse outcomes relevant to COVID-19, such as ICU admission. The exclusion criteria were as follows: patients younger than 18 years old; the studies with sample size of smaller than 20; the studies not published as the full reports or lack of necessary information for data extraction.
Definition of exposure and endpoints
Hypertension and the use of ACEI/ARB therapy were defined as those COVID-19 patients diagnosed as or noted with ‘hypertension’, ‘Arterial hypertension’, ‘High blood pressure’, ‘Antihypertensive medication’, ‘RAAS inhibitors’, ‘ACEI(s)’, ‘ARB(s)’ from the electronic medical record system. Non-RAAS inhibitors were defined as antihypertensive treatment with beta-blockers, calcium channel blockers, or diuretics. The primary endpoint was all-cause mortality within 28 days or longer after hospital admission, labeled as ‘dead’, ‘died’, ‘death’, ‘not survivor’, or ‘deceased’, while the survival was defined as those patients with an outcome labeled as ‘alive’, ‘survivor’, ‘discharged’, ‘recovered’. The secondary endpoints were the adverse outcomes related to COVID-19: ICU admission, invasive mechanical intubation requirement, and severe COVID-19. The severity of COVID-19 disease was evaluated according to WHO's interim guidelines [17].
Study selection
Our systematic searching followed the Preferred Reporting Items for Systematic Reviews and Meta-Analysis (PRISMA, http://www.prisma-statement.org) purposely for recommendations. All the potential related records have been exported into the Endnote Library for further screening and management by order of author names and years. We also reviewed the reference lists of the studies and established cross citation searching in Google Scholar. We also searched the conference proceedings to acquire the relevant articles for the meta-analysis. If the duration and sources of study population recruited overlapped more than 30% in two or more articles written by the same authors, we only included the most recent study or the study with the largest sample size. The studies were also recruited when they shared different outcomes, although the participants of these studies were from the same source. Three reviewers (A.U.K., Y.L., and Y.F.) independently established study selection, including screening of titles and abstracts, and ultimately resolved disagreements. The complicated articles, which could not be identified by those reviewers were arbitrated by the third experienced reviewer (G.C. or P.L.).
Data extraction
Data were independently extracted by three authors (A.U.K., Y.L., and Y.F.), and then checked by the other co-authors. The concordance rate between the three authors was 95%. The extracted data were managed in a standardized data extraction form. The information as followed was extracted from all included publications: author, study design, sample collection time, country, sample size, other demographic characteristics, description of prognostic elements, outcomes, and their definitions. The effects of hypertension or ACEI/ARB treatment on the outcomes of COVID-19 patients were evaluated by odds ratio (OR) and 95% confidence interval (CI). Specifically, the adjusted OR (AOR) was preferred when the AOR and the crude OR were both presented in the studies, in order to minimize confounding effects.
Statistical analysis
The baseline information of the included studies was synthesized and presented in a tabular form. The items including the sample size, data collection time, demographic characteristics, and the risk of bias in each study were summarized. Categorical variables were presented as counts (%) and continuous variables were described using means. A fixed-effect model with the Mentel–Haenszel method was applied to estimate pooled OR (95% CI) if no evidence of significant heterogeneity existed; otherwise, a random-effect model with the DerSimonian–Laird method was applied. A P value of less than 0.05 was considered significant. The heterogeneity was assessed by I2 statistics, I2 less than 25 indicating low heterogeneity, 25≤I2 < 75 moderate heterogeneity, and I2 at least 75 high heterogeneity. Generally, I2 statistic of greater than 50% was considered significant heterogeneity. The subgroup analysis was conducted to figure out the source of heterogeneity according to the basic characteristics of patients or other features. The interactive analysis was performed to identify the interaction effect of hypertension and age on in-hospital mortality. Additionally, the sensitivity analysis was also performed using the leave-one-out method to track the source of heterogeneity and evaluate the statistical robustness. The publication bias of the included studies was evaluated qualitatively by funnel plot and quantitatively by the Egger's test. When the number of the studies was less than 10, the publication bias assessment was not applicable and discarded. These statistical analyses and plots were performed using Meta package from R software, version 3.6.2 (R Foundation for Statistical Computing, Vienna, Austria).
Quality assessment
We used the Cochrane's Collaboration Tool to assess the risk of bias in the included studies. Newcastle–Ottawa Scale (NOS) was recommended to evaluate cohort and case–control studies by Agency for Healthcare Research and Quality (AHRQ) [18,19]. A study could be awarded a maximum of nine stars in three sections. Quality assessment for the randomized trials was waived because of the absence of eligible randomized trials after study selection. The methodological quality of cross-sectional studies was assessed using an 11-item checklist, which was recommended by AHRQ [19]. The quality of the articles was judged as follows: low quality = 0–3; moderate quality = 4–6; high quality at least 7. Two reviewers (A.U.K. and and P.L.) independently assessed the risk of bias of each study twice.
RESULTS
Characteristics of studies and coronavirus disease 2019 participants
A total of 7087 potentially relevant articles were identified by our searching strategy. After excluding 6001 unqualified articles, 86 articles were involved in the subsequent analysis [2,4–7,9,10,14–16,20–95]. Of these, 23 studies contained multiple endpoints with two more kinds of comparisons (Fig. 1). A summary of baseline characteristics of the 86 studies was illustrated in Table 1. A total of 18 775 387 COVD-19 patients from 18 countries worldwide were enrolled in this study. Of these, the top three counties were China, the United States of America (USA), and Mexico. The duration of most population collection was from January 2020 to July 2020. The mean age of the total population was approximately 60 years. The proportion of hypertension in each included study varied from 7.9 to 100%. Of the 86 included studies, 24 targeted particular populations, such as the severe and elderly COVID-19 patients. All the included studies were observational, and there were no randomized clinical trials (RCTs). Of these, 60 were retrospective studies; 17 were prospective cohort studies; and nine were cross-sectional studies. For quality assessment, the risk of bias in the most studies was low or moderate.
FIGURE 1.
Flowchart of study selection.
TABLE 1.
Characteristics of studies and coronavirus disease 2019 participants included in the meta-analysis
| Study (First author, year [ref]) | Study Design | Country | Sample collection time | Sample size | Age, years (mean) | Gender, male (n, %) | Hypertension (n, %) | Physical status of population | Risk of bias |
| Abadía, 2020 [20] | Prospective study | Spain | Within April 2020 | 83 | 82 | 35 (42.2) | 63 (75.9) | General | 3 |
| Agarwal, 2021 [21] | Cross-sectional study | India | 25 April to 12 July 2020 | 95 | 47 | 196 (69.8) | 39 (13.8) | Severe | 9 |
| Albitar, 2020 [22] | Cross-sectional study | Worldwide data | On 21 April 2020 | 828 | 49 | 489 (59.1) | 90 (10.9) | General | 7 |
| Altunok, 2020 [23] | Retrospective study | Turkey | 15 March to 1 May 2020 | 722 | 57 | 371 (51.4) | 251 (34.8) | General | 7 |
| An, 2020 [24] | Retrospective study | South Korea | 23 January to 2 April 2020 | 10 237 | 44 | 4088 (39.9) | 1864 (18.2) | General | 7 |
| Atkins, 2020 [4] | Prospective study | UK | 16 March to 26 April 2020 | 269 070 | 73 | 311 (61.3) | 302 (59.6) | General | 6 |
| Bae, 2020 [16] | Retrospective study | South Korea | 20 January to 31 March 2020 | 864 | _ | 444 (51.4) | 846 (97.9) | With CVD | 7 |
| Barrett, 2020 [25] | Retrospective study | USA | 9 March to 4 April 2020 | 1122 | 62 | _ | _ | General | _ |
| Besharat, 2020 [26] | Cross-sectional study | Iran | Within April 2020 | 62 | 67 | 44 (71.0) | 5 (11.1) | General | 9 |
| Boulle, 2021 [27] | Retrospective study | South Africa | 1 March to 9 June 2020 | 2978 | 40 | 1 455 144 (42.0) | 563 908 (16.0) | General | 7 |
| Bousquet, 2020 [28] | Prospective study | French | March to April 2020 | 108 | 78 | 60 (55.5) | 77 (71.0) | Elderly | 4 |
| Burrell, 2020 [29] | Prospective study | Australia | 27 February to 30 June 2020 | 204 | 63 | 140 (69.0) | 29 (24.0) | Severe | 4 |
| Caliskan, 2020 [30] | Retrospective study | Turkey | 15 March and 10 May 2020 | 565 | 48 | _ | 128 (22.7) | General | 6 |
| Cao, 2020 [31] | Retrospective study | China | 5 January to 22 February 2020 | 101 | 56 | 67 (66.3) | 38 (37.6) | Severe | 6 |
| Carrillo-Vega, 2020 [32] | Cross-sectional study | Mexico | Ended on 23 April 2020 | 10 544 | 46 | 6082 (57.7) | 2272 (21.7) | General | 7 |
| Cetinkal, 2020 [33] | Retrospective study | _ | 10 March to 10 May 2020 | 349 | 69 | 176 (44.7) | 349 (100.0) | General | 6 |
| Gao, 2020 [34] | Retrospective study | China | 5 February to 15 March 2020 | 2877 | _ | 1470 (51.1) | 850 (29.5) | General | 6 |
| Chaudhri, 2020 [35] | Retrospective study | USA | 7 March to 1 April 2020 | 300 | _ | 166 (55.3) | 133 (44.3) | General | 6 |
| Chilimuri, 2020 [2] | Retrospective study | USA | 9 March to 9 April 2020 | 375 | 63 | 236 (63.0) | 225 (60.0) | General | 7 |
| Cho, 2020 [36] | Retrospective study | South Korea | 1 February to 15 May 2020 | 7327 | 47 | 2964 (40.5) | 1559 (21.3) | General | 8 |
| Crouse, 2020 [37] | Retrospective study | USA | 25 February to 22 June 2020 | 604 | 60 | 272 (45.0) | 420 (69.5) | General | 6 |
| Cummings, 2020 [38] | Prospective study | USA | 2 March to 1 April 2020 | 257 | 62 | 171 (67.0) | 86 (33.0) | Severe | 6 |
| Dai, 2020 [39] | Retrospective study | China | 29 January to 25 February 2020 | 492 | _ | 226 (45.9) | 141 (28.7) | General | 5 |
| de Souza, 2020 [40] | Cross-sectional study | Brazil | On 2 August 2020 | 9807 | >60 | 4662 (47.5) | 597 (6.1) | Elderly | 6 |
| Denova-Gutiérrez, 2020 [41] | Cross-sectional study | Mexico | 27 February to 10 April 2020 | 3844 | 45.4 | 2230 (58.0) | 727 (18.9) | General | 7 |
| Dublin, 2021 [42] | Retrospective study | USA | Ended on February 2020 | 322 044 | _ | 148 330 (46.1) | 66 443 (20.6) | General | 8 |
| Felice, 2020 [43] | Retrospective study | Italy | 9 March to 31 March 2020 | 133 | _ | 86 (64.7) | 133 (100.0) | General | 6 |
| Galloway, 2020 [44] | Retrospective study | UK | 1 March to 17 April 2020 | 1157 | 71 | 666 (57.6) | 611 (52.9) | General | 6 |
| Gottlieb, 2020 [45] | Retrospective study | USA | 4 March to 21 June 2020 | 8673 | 41 | 4045 (46.6) | 1917 (22.1) | General | 7 |
| Guisado-Vasco, 2020 [46] | Retrospective study | Spain | 10 March to 15 April 2020 | 607 | 69 | 394 (65.0) | 276 (46.9) | Severe | 7 |
| Gupta, 2020 [47] | Prospective study | USA | 4 March to 4 April 2020 | 2215 | 60.5 | 1436 (64.8) | 390 (18.3) | Severe | 7 |
| Hernández-Galdamez, 2021 [48] | Cross-sectional study | Mexico | Ended on 27 June 2020 | 211 003 | 45.7 | 115 442 (54.7) | 42 453 (20.1) | General | 8 |
| Hernández-Vásquez, 2020 [49] | Cross-sectional study | Mexico | Ended on 18 May 2020 | 51 053 | 46 | 29 422 (57.6) | 11 090 (21.7) | General | 6 |
| Hu, 2020 [50] | Retrospective study | China | January to March 2020 | 105 | 58 | 66 (62.9) | 18 (17.1) | Severe | 6 |
| Hu, 2020 [51] | Retrospective study | China | 24 January to 16 February 2020 | 213 | 44 | 102 (47.8) | 30 (14.0) | General | 7 |
| Iaccarino, 2020 [52] | Cross-sectional study | Italy | 9 March to 29 April 2020 | 2378 | 68.2 | 1489 (62.6) | 1391 (58.5) | General | 8 |
| Imam, 2020 [53] | Retrospective study | USA | 1 March to 1 April 2020 | 1305 | 61 | 702 (53.8) | 734 (56.2) | General | 7 |
| Ioannou, 2020 [10] | Retrospective study | USA | 28 February to 14 May 2020 | 10 131 | 63 | 9221 (91.0) | 3837 (37.9) | General | 8 |
| Jiang, 2021 [54] | Retrospective study | China | 30 January to 8 March 2020 | 281 | 60 | 143 (50.9) | 137 (48.8) | Elderly and severe | 6 |
| Josef Smolen, 2020 [55] | Retrospective study | French | Ended on 18 May 2020 | 694 | 73 | 199 (32.2) | 133 (21.6) | Rheumatic and inflammatory diseases | 6 |
| Kabarriti, 2020 [56] | Retrospective study | USA | 14 March to 15 April 2020 | 5902 | 58 | 2768 (46.9) | _ | General | 7 |
| Kammar-García, 2020 [57] | Retrospective study | Mexico | 1 January and 25 April 2020 | 13 842 | 46 | 5853 (42.3) | 2969 (21.4) | General | 6 |
| Kim, 2020 [6] | Retrospective study | South Korea | 18 February to 10 July 2020 | 2254 | 58 | 808 (35.8) | 646 (28.7) | General | 7 |
| Klang, 2020 [58] | Retrospective study | USA | 1 March to 9 May 2020 | 1320 | 75 | 772 (58.5) | 1003 (76.0) | Elderly | 7 |
| Kocayigit, 2020 [59] | Cross-sectional study | Turkey | 20 March to 10 April 2020 | 169 | 65.8 | 79 (46.7) | 169 (100.0) | General | 6 |
| Kvale, 2021 [60] | Cross-sectional study | Norwegian | 30 June to 4 December 2020 | 8809 | 60 | 4365 (49.5) | 645 (7.3) | General | 9 |
| Lee, 2020 [61] | Retrospective study | South Korea | Ended on 15 May 2020 | 1609 | _ | 699 (43.4) | _ | General | 7 |
| Li, 2020 [62] | Retrospective study | China | 26 January 2020 to 5 February 2020 | 548 | 60 | 279 (50.9) | 166 (30.3) | General | 7 |
| Liu, 2020 [63] | Retrospective study | China | 1 January to 4 March 2020 | 665 | 57 | 318 (47.8) | 158 (23.8) | General | 6 |
| Lohia, 2020 [64] | Retrospective study | USA | 10 March to 30 June 2020 | 1871 | 66 | 965 (51.6) | 1485 (79.4) | Elderly | 7 |
| Lu, 2020 [65] | Retrospective study | China | 19 January to 8 March 2020 | 20 | 69 | 394 (65.0) | 276 (46.9) | Severe | 7 |
| Mancilla-Galindo, 2020 [7] | Retrospective study | Mexico | 1 June to Jul 23, 2020 | 83 779 | 46 | 47 386 (56.6) | 17 609 (21.0) | General | 7 |
| Martínez-del, 2020 [66] | Retrospective study | Spain | 1 March to 30 April 2020 | 921 | 78 | 500 (54.3) | 545 (59.2) | General | 6 |
| Martos-Benítez, 2020 [67] | Retrospective study | Mexico | 1 January to 12 May 2020 | 65 535 | 44 | 22,362 (58.3) | _ | General | 7 |
| Mehta, 2020 [68] | Retrospective study | USA | 8 March 8 to 12 April 2020 | 18 472 | 49 | 7384 (40.0) | 1235 (94.0) | General | 7 |
| Mithal, 2020 [69] | Cross-sectional study | India | 9 July to 8 August 2020 | 401 | _ | 276 (68.8) | 164 (40.9) | General | 7 |
| Mora, 2020 [70] | Prospective study | Italy | 12 March to 11 April 2020 | 431 | 65 | 263 (61.0) | 221 (51.0) | Elderly | 9 |
| Muhammad, 2020 [71] | Retrospective study | USA | 1 March to 30 May 2020 | 200 | 58.9 | 121 (60.5) | 130 (65.0) | General | 6 |
| Olivas-Martínez, 2020 [72] | Prospective study | Mexico | 26 February to 5 June 2020 | 800 | 51 | 488 (61.0) | 240 (30.0) | Severe | 5 |
| Palaiodimos, 2020 [73] | Retrospective study | USA | 9 March to 12 April 2020 | 200 | 64 | 98 (49.0) | 152 (76.0) | General | 5 |
| Pantea Stoian, 2020 [15] | Retrospective study | Romania | 26 February to 20 April 2020 | 432 | 67 | 282 (65.3) | 162 (37.5) | General | 7 |
| Pena, 2020 [74] | Cross-sectional study | Mexico | February to 13 November 2020 | 323 671 | _ | 12 118 (38.6) | 25 587 (7.9) | General | 9 |
| Rossi, 2020 [75] | Prospective study | Italy | 27 February to 2 April 2020 | 2653 | _ | 1328 (50.1) | 430 (18.1) | General | 6 |
| Rozaliyani, 2020 [76] | Retrospective study | Jakarta | 2 March to 27 April 2020 | 4052 | 45 | 2169 (53.5) | 390 (18.3) | General | 7 |
| Rubio-Rivas, 2021 [77] | Retrospective study | Spain | 1 March to 31 July 2020 | 12 066 | 67 | 7052 (58.5) | 6030 (50.0) | Elderly | 7 |
| Ruocco, 2020 [78] | Cross-sectional study | Italy | On or after 22 February 2020 | 864 | 65 | 537 (62.2) | 420 (48.6) | General | 7 |
| Selçuk, 2020 [14] | Retrospective study | Turkey | _ | 113 | _ | 59 (52.2) | 113 (100.0) | General | 6 |
| Seo, 2020 [79] | Cross-sectional study | South Korea | On 15 May 2020 | 423 | 78 | 100 (47.0) | _ | General | 7 |
| Shah, 2020 [80] | Retrospective study | USA | 2 March to 6 May 2020 | 522 | 63 | 218 (41.8) | 416 (79.7) | General | 7 |
| Simonnet, 2020 [81] | Retrospective study | French | 27 February to 5 April 2020 | 124 | 60 | 90 (73) | 60 (49) | Severe | 6 |
| Suleyman, 2020 [82] | Cross-sectional study | USA | 9 March to 27 March 2020 | 463 | 57.5 | 204 (44.1) | 295 (63.7) | General | 7 |
| Sun, 2020 [9] | Retrospective study | China | January to April 2020 | 3400 | 61 | 1649 (48.5) | 1782 (52.4) | General | 7 |
| Thompson, 2020 [83] | Retrospective study | UK | 12 March to 19 May 2020 | 470 | 71 | 255 (54.3) | 218 (46.4) | General | 6 |
| Vicenzi, 2020 [84] | Retrospective study | Italy | _ | 69 | 61 | 50 (72.0) | 31 (45.0) | Severe | 6 |
| Wang, 2020 [85] | Retrospective study | China | 7 January to 11 February 2020 | 296 | 47 | 140 (47.3) | 42 (14.2) | General | 6 |
| Wang, 2020 [86] | Retrospective study | China | 1 January to 6 February 2020 | 339 | 69 | 173 (51.0) | 138 (40.8) | General | 5 |
| Williamson, 2020 [5] | Retrospective study | UK | Ended on 6 May 2020 | 17 278 392 | 50 | 8 647 989 (50.1) | 5 925 492 (34.3) | General | 6 |
| Wu, 2020 [87] | Retrospective study | China | 25 December 2019 to 26 January 2020 | 201 | 51 | 128 (63.7) | 39 (19.4) | General | 6 |
| Xie, 2020 [88] | Retrospective study | USA | 30 March to 5 April 2020 | 287 | 61.5 | 124 (43.2) | 230 (80.1) | General | 8 |
| Xu, 2020 [89] | Retrospective study | China | 22 January to 1 April 2020 | 659 | 50 | 332 (50.4) | 167 (26.1) | General | 6 |
| Yang, 2020 [90] | Retrospective study | China | 1 January to 29 February 2020 | 226 | 70 | 113 (50.0) | 137 (48.8) | Elderly | 5 |
| Zangrillo, 2020 [91] | Retrospective study | Italy | 20 February to 2 April 2020 | 73 | 54 | 61 (83.6) | 38 (52.9) | Severe | 6 |
| Qian, 2020 [92] | Retrospective study | China | December 2019 to February 2020 | 582 | 64 | 289 (50.3) | 194 (33.0) | Severe | 6 |
| Zhang, 2021 [93] | Retrospective study | China | 31 December 2019 to 20 February 2020 | 1128 | 64 | 608 (53.5) | 1,128 (100.0) | General | 7 |
| Zhou, 2020 [94] | Retrospective study | China | 29 December 2019 to 31 January 2020 | 191 | 56 | 119 (62.0) | 58 (30.0) | General | 6 |
| Zhu, 2020 [95] | Cross-sectional study | China | 23 January to 28 March 2020 | 136 | 50.2 | 51 (37.5) | 33 (24.3) | General | 6 |
The effect of hypertension on the mortality of coronavirus disease 2019 inpatients
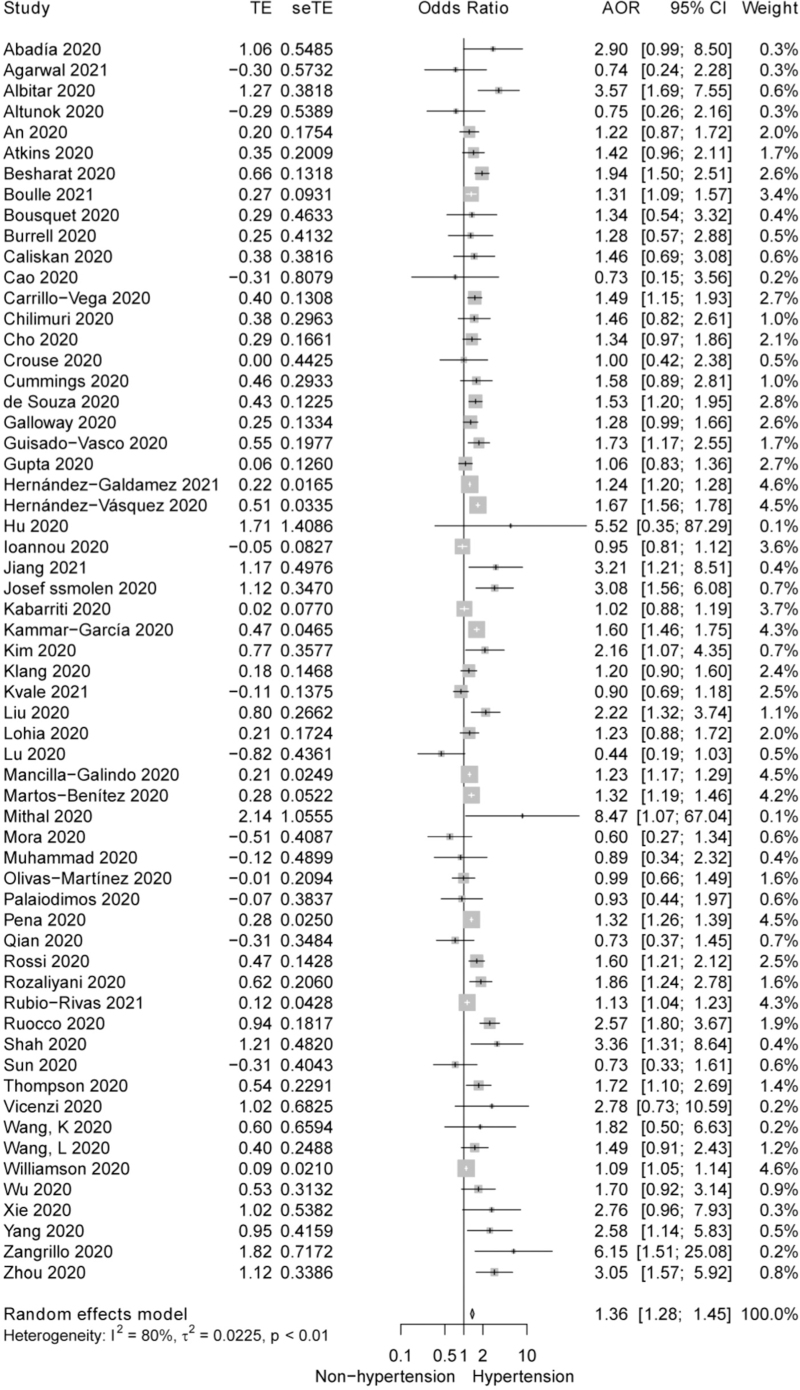
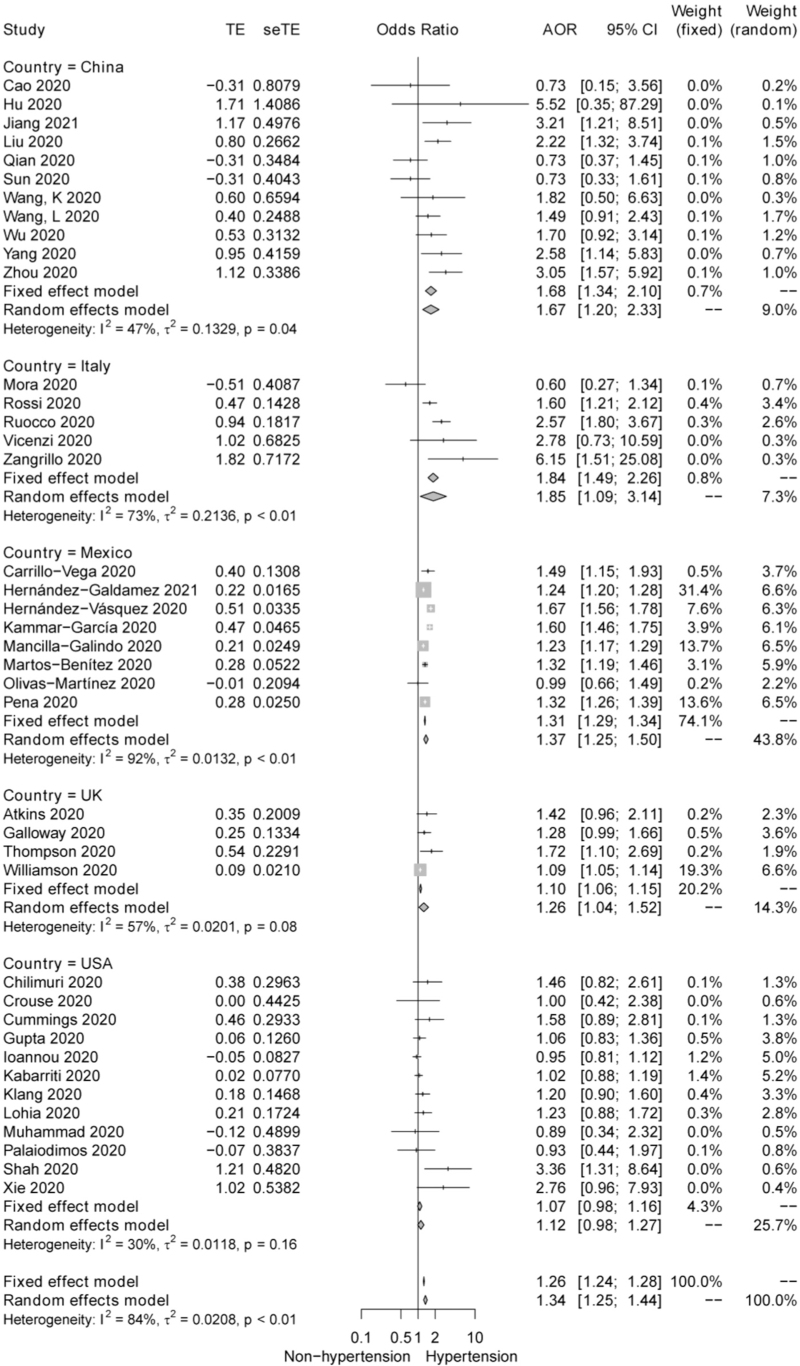
Of 60 studies evaluating the impact of hypertension on the risk of mortality in COVID-19 patients, 50 exhibited AORs (95% CI). The crude ORs were reported in the rest 10 studies because of the nonsignificant results from the univariate or multivariate analysis. Our pooled analysis indicated that COVID-19 patients with hypertension had a higher risk of mortality, compared with those without hypertension (Random effects model, AOR: 1.36, 95% CI 1.28–1.45; P < 0.001), although the heterogeneity was statistically significant (I2 = 80%). Consequent subgroup analysis stratified by country, age, and demographic characteristics of the population was conducted to seek for the source of heterogeneity. Of these factors, country partly explained the heterogeneity (the counties with more than three studies were incorporated into this analysis). The subgroup effects were significant in China, Italy, Mexico, and the United Kingdom (UK) but marginally insignificant in USA [China: 1.68 (1.34–2.10), Italy: 1.85 (1.09–3.14), Mexico: 1.37 (1.25–1.50), UK: 1.26 (1.04–1.52) and USA: 1.07 (0.98–1.16)]. The heterogeneity was moderate in China, UK, and USA (I2 = 47%, I2 = 57%, I2 = 30%) but high in Italy (I2 = 73%) and Mexico (I2 = 92%) (Figs 2 and 3). Furthermore, the sensitivity analysis indicated that the association of hypertension with a higher mortality risk in COVID-19 was consistently evident, after removal of any single study (Figure S1).
FIGURE 2.
Forest plot for the effect of hypertension on in-hospital mortality of coronavirus disease 2019 patients. AOR, adjusted odds ratio; COVID-19, coronavirus disease 2019.
FIGURE 3.
Forest plot of subgroup analysis by country for the effect of hypertension on in-hospital mortality of coronavirus disease 2019 patients. AOR, adjusted odds ratio; COVID-19, coronavirus disease 2019.
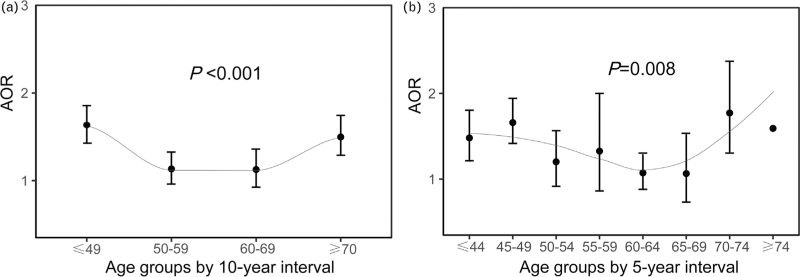
Interactive effect of hypertension and age on the mortality of coronavirus disease 2019 inpatients
Regarding nonlinear association between age and hypertension on the mortality, mean age was transformed into age groups to identify the interactive effect. AOR appeared to have an ascent trend after an initial decline with age, divided by a 10-year interval (Fig. 4a). Specifically, the effect of hypertension on the mortality was higher in COVID-19 patients aged 49 or less and at least 70 years than that in those aged 50–69 years (P < 0.001). To further determine this interactive effect, age was divided into eight groups by 5-year interval. The effects of hypertension on in-hospital mortality among those age groups followed a similar U-shaped curve. A decreased trend was evident in COVID-19 patients younger than 60 years and then increased in patients older than 69 years (Fig. 4b).
FIGURE 4.
Interactive effect of hypertension and age on in-hospital mortality of coronavirus disease 2019 patients. Dark points represent AORs and line bars represent 95% CIs. 95% CI was discarded in the oldest age group (≥74 years), as only two studies were included. AOR, adjusted odds ratio; CI, confidence interval; COVID-19, coronavirus disease 2019.
Effect of hypertension on the adverse outcomes of coronavirus disease 2019 patients
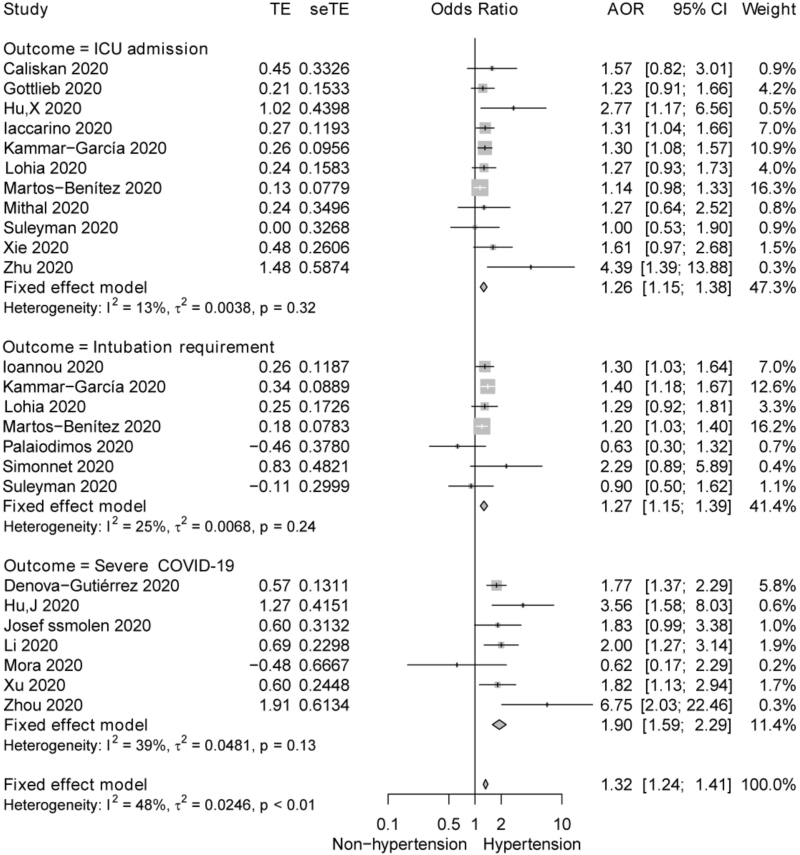
In total, 25 eligible studies were included in this analysis. On the basis of fixed-effect model, hypertension had an adjusted OR of 1.32 (1.24–1.41) for the adverse outcomes in COVID-19 patients and the heterogeneity was generally acceptable (I2 = 48%, τ2 = 0.0246, P < 0.01). Subsequently, the analysis of subgroup stratified by the outcomes was performed. The subgroup effects for ICU admission (n = 11, AOR: 1.26, 95% CI 1.15–1.38, I2 = 13%) and invasive intubation requirement (n = 7, AOR: 1.27, 95% CI 1.15–1.39, I2 = 24%) were similar. However, the subgroup effect of hypertension on the severe COVID-19 (n = 7, AOR: 1.90, 95% CI 1.59–2.29, I2 = 39%) was higher than that of other two adverse outcomes. Generally, the effects of hypertension on ICU admission, mechanical intubation requirement, and severe COVID-19 were consistently significant (Fig. 5).
FIGURE 5.
Forest plot for the effect of hypertension on ICU admission, mechanical intubation requirement, and severe disease in coronavirus disease 2019 patients. AOR, adjusted odds ratio; COVID-19, coronavirus disease 2019.
Effect of angiotensin-converting enzyme-inhibitors/angiotensin–renin blockers on the mortality of coronavirus disease 2019 inpatients
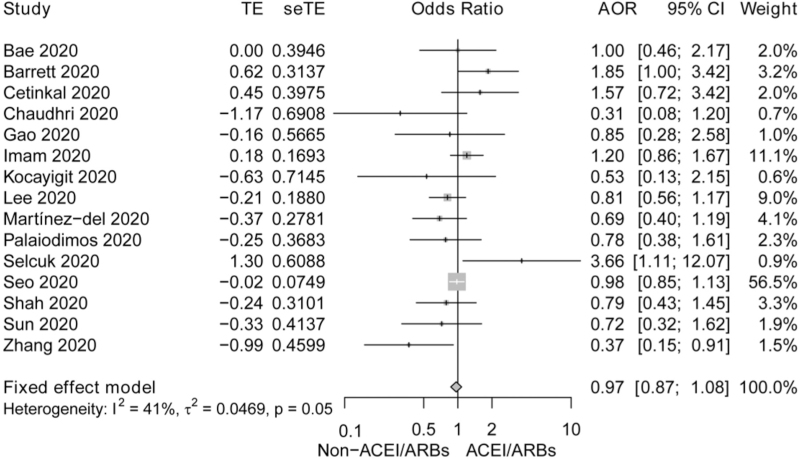
In total, 15 studies were included to investigate the relationship between ACEI/ARB medication and the mortality of COVID-19 patients with hypertension. The fixed-effect model showed that the use of ACEI/ARBs was not significantly associated with the risk of in-hospital mortality [0.97 (0.87–1.08)], compared with the hypertensive COVID-19 patients taking non-RAAS inhibitors. In addition, the moderate heterogeneity (I2 = 41%) indicated an acceptable systematic difference between those studies (Fig. 6).
FIGURE 6.
Forest plot for the effect of angiotensin-converting enzyme inhibitor/angiotensin receptor blocker treatment on in-hospital mortality of coronavirus disease 2019 patients with hypertension. ACEI, angiotensin-converting enzyme inhibitor; AOR, adjusted odds ratio; ARB, angiotensin receptor blocker; COVID-19, coronavirus disease 2019.
Effect of angiotensin-converting enzyme-inhibitors/angiotensin–renin blockers on the adverse outcomes in coronavirus disease 2019 patients
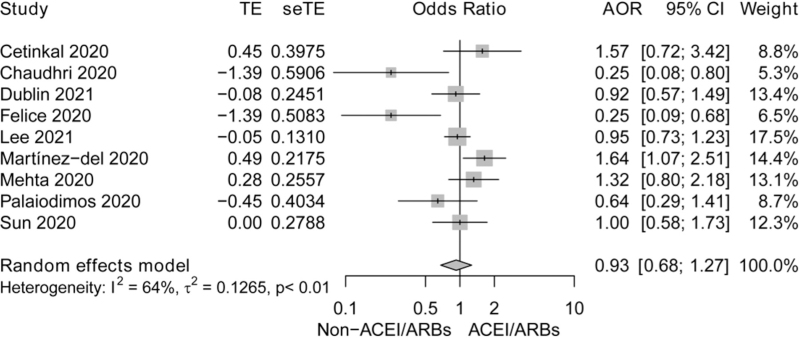
In total, nine eligible studies were analyzed to evaluate the impact of antihypertensive RAAS inhibitors on the adverse outcomes. The random-effect model revealed that no significant association was detected between ACEI/ARB use and the adverse outcomes in hypertensive patients with COVID-19 (AOR: 0.93, 95% CI: 0.68–1.27; P = 0.636; I2 = 64%). Although the subgroup analysis stratified by the adverse outcome was not performed because of the limited studies, the robustness of our findings was demonstrated by the sensitivity analysis with leave-one-out method (Fig. 7, Figure S2).
FIGURE 7.
Forest plot for the effect of coronavirus disease 2019 treatment on ICU admission, mechanical intubation requirement, and severe disease in coronavirus disease 2019 patients with hypertension. ACEI, angiotensin-converting enzyme inhibitor; AOR, adjusted odds ratio; ARB, angiotensin receptor blocker; COVID-19, coronavirus disease 2019.
Publication bias analysis
Funnel plots were symmetrical for the effects of hypertension and ACEI/ARB use on the mortality of COVID-19 inpatients (P = 0.062, P = 0.567, respectively), but slightly asymmetrical for the impact of hypertension on the adverse outcomes (P = 0.028). Overall, strong evidence of publication bias was not detected. Nevertheless, the assessment of the publication bias in the studies evaluating the effect of ACEI/ARB use on the adverse outcomes was waived as the number of studies was less than 10 (Figure S3).
DISCUSSION
In this study, we investigated the effects of hypertension and the use of ACEI/ARB treatment on the clinical outcomes in COVID-19 inpatients. A significant association of the mortality with hypertension was found in those inpatients. However, the use of ACEI/ARBs was not associated with increased mortality risk or the severity in the hypertensive inpatients with COVID-19 compared with the treatment with non-RAAS inhibitors.
In an earlier systematic review, the risk of getting serious clinical outcomes (including ICU admission, ARDS, and mechanical ventilation) in hypertensive patients with COVID-19 was 2.95 (2.21–3.94) times higher than those without hypertension, based on eight studies and the univariate analysis [96]. By contrast, another systematic review reported the association between hypertension and the mortality as well as mechanical ventilation was not statistically significant, based on 29 studies and age-adjusted model [OR, 95% CI 1 (0.94–1.06) and 1.01 (0.93–1.09), respectively) [97]. Our pooled results based on the multivariate analysis of 86 studies suggested that hypertension was an independent risk factor for the mortality and the adverse outcomes in COVID-19 inpatients, despite that nearly half of the studies presented insignificant results (Figs 2 and 5). Hence, it is reasonably considered that the inconsistent evidence is probably attributable to the quantity and the sample sizes of eligible studies, or the adjustment for potential confounders. More importantly, seeking out the source of heterogeneity among the diverse populations exhibited a greater value. In our study, the subgroup analysis stratified by country could account for part of the heterogeneity. Specifically, the influence of hypertension on the mortality risk was significantly different in domestic regions of Mexico and Italy. We also found interactive effects of hypertension and age on the mortality among COVID-19 inpatients, following a U-shaped curve (Fig. 4). The results indicated that antihypertension treatment needs to be emphasized in younger (<60 years) and older patients (>69 years). Although the prevalence of hypertension is relatively lower in younger patients but this regimen is crucial to prevent young patients from developing severe illness. This finding was supported by a large, prospective, and multicenter cohort study in the UK in which younger people with comorbidities were identified to have a much higher mortality rate than those who had complications, compared with those at the same age without complications [98].
This pooled analysis did not identify the significant effects of the ACEI/ARB treatment on in-hospital mortality and the adverse outcomes [AOR, 95% CI 0.97 (0.87–1.08) and 0.93 (0.68–1.27), respectively] (Figs 5 and 6). Our results are consistent with the findings of WHO's rapid review. There is low-certainty evidence that patients on long-term treatment with ACE inhibitors or ARBs are not at higher risk of poor outcomes from COVID-19 [99].
The underlying mechanisms by which hypertension worsens the prognosis of COVID-19 inpatients remain unknown. There are two widely recognized explanations: RAAS activation of hypertensive patients was assumed to be associated with severe COVID-19, which could trigger the inflammation response and vasoconstriction [100–103]; it has been well documented that the prevalence of hypertension increased in line with old age and comorbidities, which may distort this correlation [2–5]. For the latter point, our meta-analysis showed hypertension was an independent predictor of severe COVID-19 via adjusting for those confounders (Figs. 2 and 5). It is worth noting that younger people have relatively well developed immune system and fewer comorbidities, thus cytokine storm was presumed to play a prominent role in aggravating COVID-19 in younger patients. Thus, hypertension management should be emphasized to control inflammation and enhance host immunity.
The possible mechanisms by which ACEI/ARBs affect the prognosis of COVID-19 have been explained [104]. However, the hypothesis is still controversial, because of the complicated molecular mechanism and absence of high-certainty evidence in humans. At the early stage of COVID-19 outbreak, ACEI/ARB treatment was considered to aggravate lung injury, as some studies reported this treatment could promote the expression of ACE2 [11,105], which linked with SARS-CoV-2 entry and propagation [11,12]. However, most clinical studies presented that ACEI/ARB treatment was neutral and even beneficial to critical COVID-19 (Fig. 6). An RCT also showed no significant difference in the mean number of days alive and out of the hospital for those assigned to discontinue versus continue ACEI/ARB medications [106]. Specially, it was reported that ACEI treatment dampened COVID-19-related hyperinflammation and increased cell-intrinsic antiviral responses, whereas ARB treatment enhanced epithelial–immune cell interactions, based on the clinical data (n = 144) and single-cell sequencing data (n = 48) [107]. Therefore, it is reasonable to believe that ACEI/ARB treatment has positive or neutral effects on clinical outcomes in patients with COVID-19, although the negative impact exists theoretically.
To the best of our knowledge, this systematic review is the most comprehensive exploration and analysis of the existing literature on this topic to date. More importantly, it was the first systematic review illustrating the interactive effect of hypertension and age on the mortality of COVID-19 patients. However, our study also has limitations. First, no relevant RCTs were available, even though we tried our best to mitigate this by establishing broad searching terms and enrolling in the repositories and databases as many as possible. Second, the heterogeneity was not probed enough, although the subgroup and sensitivity analyses were performed. Further investigations, including RCTs, are highly recommended.
In conclusion, the pooled analysis of 86 articles included in our study showed that hypertension is independently associated with an increased risk of mortality and adverse outcomes for COVID-19 inpatients. Younger and older hypertensive patients have a higher mortality risk compared with those of the same age without hypertension. Hence, hypertension management should be emphasized in those patients. However, ACEI/ARB treatments do not increase the mortality and other poor outcomes of hypertensive COVID-19 patients. Thus, ACEI/ARBs are suggested to be applied to treat hypertension in patients with COVID-19.
ACKNOWLEDGEMENTS
We acknowledge all the workers involved in study searching, data extraction, statistical analysis and writing.
Ethics approval and consent to participate: not applicable.
Funding: this study was supported by National Natural Science Foundation of China (82041022 to G.C.), Ministry of Science and Technology of the People's Republic of China (2018ZX10101003–001-003 to G.C.), Scientific Research Project of Shanghai Science and Technology Commission (20JC1410202 and 20431900404 to G.C.), Key discipline from the ‘3-year public health promotion’ program of Shanghai Municipal Health Commission (GWV-10.1-XK17 to: G.C.), and the institutional research projects for natural-focus infectious diseases and COVID-19 (to G.C.).
Authors’ contributions: A.U.K., Y.L., Y.F., K.Y., and M.H.; study search, data extraction, and arrangement. A.U.K., P.L, Z.J., and D.J.: quality control, data analysis, and composition of figures and table. G.C.: conceptualization, supervision, funding acquisition. G.C., X.T., and P.L.: article writing.
Conflicts of interest
There are no conflicts of interest.
Supplementary Material
A.U.K., P.L., Z.J., and X.T. contributed equally to this work.
Abbreviations: ACEI, angiotensin-converting enzymes; ARB, angiotensin–renin blockers; COVID-19, coronavirus disease 2019; RAAS, renin–angiotensin–aldosterone system; SARS-CoV-2, severe acute respiratory syndrome coronavirus 2
Supplemental digital content is available for this article.
REFERENCES
- 1.WHO. WHO Coronavirus Disease (COVID-19) Dashboard. Available at: https://covid19.who.int. [Accessed 30 April 2022] [Google Scholar]
- 2.Chilimuri S, Sun H, Alemam A, Mantri N, Shehi E, Tejada J, et al. Predictors of mortality in adults admitted with COVID-19: retrospective cohort study from New York City. West J Emerg Med 2020; 21:779. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Yang PH, Ding YB, Xu Z, Pu R, Li P, Yan J, et al. Increased circulating level of interleukin-6 and CD8+ T cell exhaustion are associated with progression of COVID-19. Infect Dis Poverty 2020; 9:161. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Atkins JL, Masoli JAH, Delgado J, Pilling LC, Kuo CL, Kuchel GA, et al. Preexisting comorbidities predicting COVID-19 and mortality in the UK biobank community cohort. J Gerontol A Biol Sci Med Sci 2020; 75:2224–2230. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Williamson EJ, Walker AJ, Bhaskaran K, Bacon S, Bates C, Morton CE, et al. Factors associated with COVID-19-related death using OpenSAFELY. Nature 2020; 584:430–436. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Kim SW, Kim SM, Kim YK, Kim JY, Lee YM, Kim BO, et al. Clinical characteristics and outcomes of COVID-19 cohort patients in Daegu Metropolitan City Outbreak in 2020. J Korean Med Sci 2021; 36:e12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Mancilla-Galindo J, Vera-Zertuche JM, Navarro-Cruz AR, Segura-Badilla O, Reyes-Velázquez G, Tepepa-Lopez FJ, et al. Development and validation of the patient history COVID-19 (PH-Covid19) scoring system: a multivariable prediction model of death in Mexican patients with COVID-19. Epidemiol Infect 2020; 148:e286. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Kabarriti R, Brodin NP, Maron MI, Guha C, Kalnicki S, Garg MK, et al. Association of race and ethnicity with comorbidities and survival among patients with COVID-19 at an Urban Medical Center in New York. JAMA Netw Open 2020; 3:e2019795. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Sun Y, Guan X, Jia L, Xing N, Cheng L, Liu B, et al. Independent and combined effects of hypertension and diabetes on clinical outcomes in patients with COVID-19: A retrospective cohort study of Huoshen Mountain Hospital and Guanggu Fangcang Shelter Hospital. J Clin Hypertens (Greenwich) 2021; 23:218–231. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Ioannou GN, Locke E, Green P, Berry K, O’Hare AM, Shah JA, et al. Risk factors for hospitalization, mechanical ventilation, or death among 10 131 US veterans with SARS-CoV-2 infection. JAMA Network Open 2020; 3:e2022310. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Ferrario CM, Jessup J, Chappell MC, Averill DB, Brosnihan KB, Tallant EA, et al. Effect of angiotensin-converting enzyme inhibition and angiotensin II receptor blockers on cardiac angiotensin-converting enzyme 2. Circulation 2005; 111:2605–2610. [DOI] [PubMed] [Google Scholar]
- 12.Hoffmann M, Kleine-Weber H, Schroeder S, Krüger N, Herrler T, Erichsen S, et al. SARS-CoV-2 cell entry depends on ACE2 and TMPRSS2 and is blocked by a clinically proven protease inhibitor. Cell 2020; 181:271–280. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Fang L, Karakiulakis G, Roth M. Are patients with hypertension and diabetes mellitus at increased risk for COVID-19 infection? Lancet Respir Med 2020; 8:e21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Selçuk M, Çinar T, Keskin M, Çiçek V, Kiliç Ş, Kenan B, et al. Is the use of ACE inb/ARBs associated with higher in-hospital mortality in COVID-19 pneumonia patients? Clin Exp Hypertens 2020; 42:738–742. [DOI] [PubMed] [Google Scholar]
- 15.Stoian AP, Pricop-Jeckstadt M, Pana A, Ileanu BV, Schitea R, Geanta M, et al. Death by SARS-CoV 2: a Romanian COVID-19 multicentre comorbidity study. Sci Rep 2020; 10:1–11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Bae S, Kim JH, Kim YJ, Lim JS, Yun SC, Kim YH, et al. Effects of recent use of renin-angiotensin system inhibitors on mortality of patients with coronavirus disease 2019. Open Forum Infect Dis 2020; 7:ofaa519. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.World Health Organization. Clinical management of severe acute respiratory infection when Novel Coronavirus (2019-nCoV) infection is suspected: interim guidance. Available at: https://apps.who.int/iris/bitstream/handle/10665/330893/WHO-nCoV-Clinical-2020.3-eng.pdf?sequence=1&isAllowed=y (2020). [Accessed 18 May 2021] [Google Scholar]
- 18. Wells GA, Shea B, O’Connell D, Peterson J, Welch V, Losos M, et al. The Newcastle-Ottawa Scale (NOS) for assessing the quality of nonrandomised studies in meta-analyses. Professor GA Wells, Department of Epidemiology and Commuunity Medicine, University of Ottawa, Ontario K1J 8M5, Canada. 2020. Available at: http://www.ohri.ca/programs/clinical_epidemiology/oxford.asp. [Accessed 1 June 2021] [Google Scholar]
- 19.Rostom A, Dube C, Cranney A, Saloojee N, Sy R, Garritty C, et al. Celiac disease. Rockville (MD): Agency for Healthcare Research and Quality (US); 2004 September (Evidence Reports/Technology Assessments, No. 104.) Appendix D. Quality Assessment Forms. [Google Scholar]
- 20.Abadía Otero J, Briongos Figuero LS, Gabella Mattín M, Usategui Martín I, Cubero Morais P, Cuellar Olmedo L, et al. The nutritional status of the elderly patient infected with COVID-19: the forgotten risk factor? Curr Med Res Opin 2021; 37:549–554. [DOI] [PubMed] [Google Scholar]
- 21.Agarwal N, Biswas B, Lohani P. Epidemiological determinants of COVID-19 infection and mortality: A study among patients presenting with severe acute respiratory illness during the pandemic in Bihar, India. Niger Postgrad Med J 2020; 27:293. [DOI] [PubMed] [Google Scholar]
- 22.Albitar O, Ballouze R, Ooi JP, Sheikh Ghadzi SM. Risk factors for mortality among COVID-19 patients. Diabetes Res Clin Pract 2020; 166:108293. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Altunok ES, Alkan M, Kamat S, Demirok B, Satici C, Demirkol MA, et al. Clinical characteristics of adult patients hospitalized with laboratory-confirmed COVID-19 pneumonia. J Infect Chemother 2021; 27:306–311. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.An C, Lim H, Kim DW, Chang JH, Choi YJ, Kim SW. Machine learning prediction for mortality of patients diagnosed with COVID-19: a nationwide Korean cohort study. Sci Rep 2020; 10:1–11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Barrett B, Pamphile S, Yang FJ, Friedman B. 149 association of ACE-I and ARB prescriptions with mortality in patients admitted to the hospital with COVID-19 in New York City. Ann Emerg Med 2020; 76:S58. [Google Scholar]
- 26.Besharat S, Alamda NM, Dadashzadeh N, Talaie R, Mousavi SS, Barzegar A. Clinical and demographic characteristics of patients with COVID-19 who died in modarres hospital. Open Access Maced J Med Sci 2020; 8 (T1):144–149. [Google Scholar]
- 27.Boulle A, Davies MA, Hussey H, Ismail M, Morden E, Vundle Z, et al. Risk factors for COVID-19 death in a population cohort study from the Western Cape Province, South Africa. Clin Infect Dis 2021; 73:e2005–e2015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Bousquet G, Falgarone G, Deutsch D, Derolez S, Lopez-Sublet M, Goudot FX, et al. ADL-dependency, D-Dimers, LDH and absence of anticoagulation are independently associated with one-month mortality in older inpatients with COVID-19. Aging (Albany, NY) 2020; 12:11306. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Burrell AJC, Pellegrini B, Salimi F, Begum H, Broadley T, Campbell LT, et al. Outcomes for patients with COVID-19 admitted to Australian intensive care units during the first four months of the pandemic. Med J Aust 2021; 214:23–30. [DOI] [PubMed] [Google Scholar]
- 30.Caliskan T, Saylan B. Smoking and comorbidities are associated with COVID-19 severity and mortality in 565 patients treated in Turkey: a retrospective observational study. Rev Assoc Med Bras 2020; 66:1679–1684. [DOI] [PubMed] [Google Scholar]
- 31.Cao Y, Han X, Gu J, Li Y, Liu J, Alwalid O, et al. Prognostic value of baseline clinical and HRCT findings in 101 patients with severe COVID-19 in Wuhan, China. Sci Rep 2020; 10:1–13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Carrillo-Vega MF, Salinas-Escudero G, García-Peña C, Gutiérrez-Robledo LM, Parra-Rodríguez L. Early estimation of the risk factors for hospitalization and mortality by COVID-19 in Mexico. PLoS One 2020; 15:e0238905. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Cetinkal G, Kocas BB, Ser OS, Kilci H, Yildiz SS, Ozcan SN. The Association between chronic use of renin-angiotensin-aldosterone system blockers and in-hospital adverse events among covid-19 patients with hypertension. Sisli Etfal Hastan Tip Bul 2020; 54:399. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Gao C, Cai Y, Zhang K, Zhou L, Zhang Y, Zhang X, et al. Association of hypertension and antihypertensive treatment with COVID-19 mortality: a retrospective observational study. Eur Heart J 2020; 41:2058–2066. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Chaudhri I, Koraishy FM, Bolotova O, Yoo J, Marcos LA, Taub E, et al. Outcomes associated with the use of renin-angiotensin-aldosterone system blockade in hospitalized patients with SARS-CoV-2 infection. Kidney360 2020; 1:801. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Cho KH, Kim SW, Park JW, Do JY, Kang SH. Effect of sex on clinical outcomes in patients with coronavirus disease: a population-based study. J Clin Med 2021; 10:38. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Crouse AB, Grimes T, Li P, Might M, Ovalle F, Shalev A. Metformin use is associated with reduced mortality in a diverse population with COVID-19 and diabetes. Front Endocrinol 2021; 11:1081. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Cummings MJ, Baldwin MR, Abrams D, Jacobson SD, Meyer BJ, Balough EM, et al. Epidemiology, clinical course, and outcomes of critically ill adults with COVID-19 in New York City: a prospective cohort study. Lancet 2020; 395:1763–1770. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Dai Y, Liu Z, Du X, Wei H, Wu Y, Li H, et al. Acute kidney injury in hospitalized patients infected with COVID-19 from Wuhan, China: a retrospective study. Biomed Res Int 2021; 2021:6655185. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.de Souza CD, de Arruda Magalhães AJ, Lima AJ, Nunes DN, de Fátima Machado Soares É, de Castro Silva L, et al. Clinical manifestations and factors associated with mortality from COVID-19 in older adults: retrospective population-based study with 9807 older Brazilian COVID-19 patients. Geriatr Gerontol Int 2020; 20:1177–1181. [DOI] [PubMed] [Google Scholar]
- 41.Denova-Gutiérrez E, Lopez-Gatell H, Alomia-Zegarra JL, López-Ridaura R, Zaragoza-Jimenez CA, Dyer-Leal DD, et al. The association of obesity, type 2 diabetes, and hypertension with severe coronavirus disease 2019 on admission among Mexican patients. Obesity 2020; 28:1826–1832. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Dublin S, Walker RL, Floyd JS, Shortreed SM, Fuller S, Albertson-Junkans L, et al. Renin-angiotensin-aldosterone system inhibitors and COVID-19 infection or hospitalization: a cohort study. Am J Hypertens 2021; 34:339–347. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Felice C, Nardin C, Di Tanna GL, Grossi U, Bernardi E, Scaldaferri L, et al. Use of RAAS inhibitors and risk of clinical deterioration in COVID-19: results from an Italian cohort of 133 hypertensives. Am J Hypertens 2020; 33:944–948. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Galloway JB, Norton S, Barker RD, Brookes A, Carey I, Clarke BD, et al. A clinical risk score to identify patients with COVID-19 at high risk of critical care admission or death: an observational cohort study. J Infect 2020; 81:282–288. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Gottlieb M, Sansom S, Frankenberger C, Ward E, Hota B. Clinical course and factors associated with hospitalization and critical illness among COVID-19 patients in Chicago, Illinois. Acad Emerg Med 2020; 27:963–973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Guisado-Vasco P, Valderas-Ortega S, Carralón-González MM, Roda-Santacruz A, González-Cortijo L, Sotres-Fernández G, et al. Clinical characteristics and outcomes among hospitalized adults with severe COVID-19 admitted to a tertiary medical center and receiving antiviral, antimalarials, glucocorticoids, or immunomodulation with tocilizumab or cyclosporine: a retrospective observational study (COQUIMA cohort). EClinicalMedicine 2020; 28:100591. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Gupta S, Hayek SS, Wang W, Chan L, Mathews KS, Melamed ML, et al. Factors associated with death in critically ill patients with coronavirus disease 2019 in the US. JAMA Intern Med 2020; 180:1436–1446. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Hernández-Galdamez DR, González-Block MÁ, Romo-Dueñas DK, Lima-Morales R, Hernández-Vicente IA, Lumbreras-Guzmán M, et al. Increased risk of hospitalization and death in patients with COVID-19 and preexisting noncommunicable diseases and modifiable risk factors in Mexico. Arch Med Res 2020; 51:683–689. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Hernández-Vásquez A, Azañedo D, Vargas-Fernández R, Bendezu-Quispe G. Association of comorbidities with pneumonia and death among COVID-19 patients in Mexico: a nationwide cross-sectional study. J Prev Med Public Health 2020; 53:211. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Hu K, Li B. Logistic regression analysis of death risk factors of patients with severe and critical coronavirus disease 2019 and their predictive value. Zhonghua wei Zhong Bing ji jiu yi xue (Chinese) 2020; 32:544–547. [DOI] [PubMed] [Google Scholar]
- 51.Hu X, Hu C, Yang Y, Chen J, Zhong P, Wen Y, et al. Clinical characteristics and risk factors for severity of COVID-19 outside Wuhan: a double-center retrospective cohort study of 213 cases in Hunan, China. Ther Adv Respir Dis 2020; 14:1753466620963035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Iaccarino G, Grassi G, Borghi C, Carugo S, Fallo F, Ferri C, et al. Gender differences in predictors of intensive care units admission among COVID-19 patients: the results of the SARS-RAS study of the Italian Society of Hypertension. PLoS One 2020; 15:e0237297. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Imam Z, Odish F, Gill I, O’Connor D, Armstrong J, Vanood A, et al. Older age and comorbidity are independent mortality predictors in a large cohort of 1305 COVID-19 patients in Michigan, United States. J Intern Med 2020; 288:469–476. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Jiang Y, Abudurexiti S, An MM, Cao D, Wei J, Gong P. Risk factors associated with 28-day all-cause mortality in older severe COVID-19 patients in Wuhan, China: a retrospective observational study. Sci Rep 2020; 10:1–13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.FAI2R/SFR/SNFMI/SOFREMIP/CRI/IMIDIATE consortium and contributors. Severity of COVID-19 and survival in patients with rheumatic and inflammatory diseases: data from the French RMD COVID-19 cohort of 694 patients. Ann Rheum Dis 2021; 80:527–538. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Kabarriti R, Brodin NP, Maron MI, Guha C, Kalnicki S, Garg MK, et al. Association of race and ethnicity with comorbidities and survival among patients with COVID-19 at an urban medical center in New York. JAMA Network Open 2020; 3:e2019795. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Kammar-García A, Vidal-Mayo JJ, Vera-Zertuche JM, Lazcano-Hernández M, Vera-López O, Segura-Badilla O, et al. Impact of comorbidities in Mexican SARS-CoV-2-positive patients: a retrospective analysis in a national cohort. Rev Invest Clin 2020; 72:151–158. [DOI] [PubMed] [Google Scholar]
- 58.Klang E, Soffer S, Nadkarni G, Glicksberg B, Freeman R, Horowitz C, et al. Sex differences in age and comorbidities for COVID-19 mortality in urban New York City. SN Compr Clin Med 2020; 2:1319–1322. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Kocayigit I, Kocayigit H, Yaylaci S, Can Y, Erdem AF, Karabay O. Impact of antihypertensive agents on clinical course and in-hospital mortality: analysis of 169 hypertensive patients hospitalized for COVID-19. Rev Assoc Med Bras 2020; 66:71–76. [DOI] [PubMed] [Google Scholar]
- 60.Kvåle R, Bønaa KH, Forster RB, Gravningen K, Júlíusson PB, Myklebust TÅ. Does a history of cardiovascular disease or cancer affect mortality after SARS-CoV-2 infection? Tidsskr Nor Laegeforen 2021; 140: [DOI] [PubMed] [Google Scholar]
- 61.Lee J, Jo SJ, Cho Y, Lee JH, Oh IY, Park JJ, et al. Effects of renin-angiotensin system blockers on the risk and outcomes of severe acute respiratory syndrome coronavirus 2 infection in patients with hypertension. Korean J Intern Med 2021; 36: (Suppl 1): S123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Li X, Xu S, Yu M, Wang K, Tao Y, Zhou Y, et al. Risk factors for severity and mortality in adult COVID-19 inpatients in Wuhan. J Allergy Clin Immunol 2020; 146:110–118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Liu M, Han S, Liao Q, Chang L, Tan Y, Jia P, et al. Outcomes and prognostic factors in 70 nonsurvivors and 595 survivors with COVID-19 in Wuhan, China. Transbound Emerg Dis 2021; 68:3611–3623. [DOI] [PubMed] [Google Scholar]
- 64.Lohia P, Kapur S, Benjaram S, Pandey A, Mir T, Seyoum B. Metabolic syndrome and clinical outcomes in patients infected with COVID-19: Does age, sex, and race of the patient with metabolic syndrome matter? J Diabetes 2021; 13:420–429. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Lu J, Zhang Y, Cheng G, He J, Wu F, Hu H, et al. Clinical characteristics and outcomes of adult critically ill patients with COVID-19 in Honghu, Hubei Province [in Chinese]. Nan fang yi ke da xue xue bao 2020; 40:778–785. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Martínez-del Río J, Piqueras-Flores J, Negreira-Caamaño M, Negreira-Caamaño M, Águila-Gordo D, Mateo-Gómez C, et al. Comparative analysis between the use of renin-angiotensin system antagonists and clinical outcomes of hospitalized patients with COVID-19 respiratory infection. Med Clin (Barc) 2020; 155:473–481. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Martos-Benítez FD, Soler-Morejón CD, García-del Barco D. Chronic comorbidities and clinical outcomes in patients with and without COVID-19: a large population-based study using national administrative healthcare open data of Mexico. Intern Emerg Med 2021; 16:1507–1517. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Mehta N, Kalra A, Nowacki AS, Anjewierden S, Han Z, Bhat P, et al. Association of use of angiotensin-converting enzyme inhibitors and angiotensin II receptor blockers with testing positive for coronavirus disease 2019 (COVID-19). JAMA Cardiol 2020; 5:1020–1026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Mithal A, Jevalikar G, Sharma R, Singh A, Farooqui KJ, Mahendru S, et al. High prevalence of diabetes and other comorbidities in hospitalized patients with COVID-19 in Delhi, India, and their association with outcomes. Diabetes Metab Syndr 2021; 15:169–175. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Anzola GP, Bartolaminelli C, Gregorini GA, Coazzoli C, Gatti F, Mora A, et al. Neither ACEI nor ARBs are associated with respiratory distress or mortality in COVID-19 results of a prospective study on a hospital-based cohort. Intern Emerg Med 2020; 15:1477–1484. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Muhammad R, Ogunti R, Ahmed B, Munawar M, Donaldson S, Sumon M, et al. Clinical characteristics and predictors of mortality in minority patients hospitalized with COVID-19 infection. J Racial Ethn Health Disparities 2021; 9:335–345. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Olivas-Martínez A, Cárdenas-Fragoso JL, Jiménez JV, Lozano-Cruz OA, Ortiz-Brizuela E, Tovar-Méndez VH, et al. In-hospital mortality from severe COVID-19 in a tertiary care center in Mexico City; causes of death, risk factors and the impact of hospital saturation. PLoS One 2021; 16:e0245772. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Palaiodimos L, Kokkinidis DG, Li W, Karamanis D, Ognibene J, Arora S, et al. Severe obesity, increasing age and male sex are independently associated with worse in-hospital outcomes, and higher in-hospital mortality, in a cohort of patients with COVID-19 in the Bronx, New York. Metabolism 2020; 108:154262. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Peña JE, Rascón-Pacheco RA, Ascencio-Montiel IJ, González-Figueroa E, Fernández-Gárate JE, Medina-Gómez OS, et al. Hypertension, diabetes and obesity, major risk factors for death in patients with COVID-19 in Mexico. Arch Med Res 2021; 52:443–449. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Giorgi Rossi P, Marino M, Formisano D, Venturelli F, Vicentini M, Grilli R, et al. Characteristics and outcomes of a cohort of COVID-19 patients in the Province of Reggio Emilia, Italy. PloS one 2020; 15:e0238281. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Rozaliyani A, Savitri AI, Setianingrum F, Shelly TN, Ratnasari V, Kuswindarti R, et al. Factors Associated with Death in COVID-19 Patients in Jakarta, Indonesia: an epidemiological study. Acta Med Indones 2020; 52:246–254. [PubMed] [Google Scholar]
- 77.Rubio-Rivas M, Corbella X, Mora-Luján JM, Loureiro-Amigo J, López Sampalo A, Yera Bergua C, et al. Predicting clinical outcome with phenotypic clusters in COVID-19 pneumonia: an analysis of 12,066 hospitalized patients from the Spanish registry SEMI-COVID-19. J Clin Med 2020; 9:3488. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Ruocco G, McCullough PA, Tecson KM, Mancone M, De Ferrari GM, D’Ascenzo F, et al. Mortality risk assessment using CHA (2) DS (2)-VASc scores in patients hospitalized with coronavirus disease 2019 infection. Am J Cardiol 2020; 137:111–117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Seo J, Son M. Update on association between exposure to renin-angiotensin-aldosterone system inhibitors and coronavirus disease 2019 in South Korea. Korean J Intern Med 2021; 36: (Suppl 1): S114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Shah P, Owens J, Franklin J, Mehta A, Heymann W, Sewell W, et al. Demographics, comorbidities and outcomes in hospitalized COVID-19 patients in rural southwest Georgia. Ann Med 2020; 52:354–360. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Simonnet A, Chetboun M, Poissy J, Raverdy V, Noulette J, Duhamel A, et al. High prevalence of obesity in severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2) requiring invasive mechanical ventilation. Obesity 2020; 28:1195–1199. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Suleyman G, Fadel RA, Malette KM, Hammond C, Abdulla H, Entz A, et al. Clinical characteristics and morbidity associated with coronavirus disease 2019 in a series of patients in metropolitan Detroit. JAMA Network Open 2020; 3:e2012270. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Thompson JV, Meghani NJ, Powell BM, Newell I, Craven R, Skilton G, et al. Patient characteristics and predictors of mortality in 470 adults admitted to a district general hospital in England with COVID-19. Epidemiol Infect 2020; 48:e285. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Vicenzi M, Ruscica M, Iodice S, Rota I, Ratti A, Di Cosola R, et al. The efficacy of the mineralcorticoid receptor antagonist canrenone in COVID-19 patients. J Clin Med 2020; 9:2943. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Wang K, Zuo P, Liu Y, Zhang M, Zhao X, Xie S, et al. Clinical and laboratory predictors of in-hospital mortality in patients with coronavirus disease-2019: a cohort study in Wuhan, China. Clin Infect Dis 2020; 71:2079–2088. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Wang L, He W, Yu X, Hu D, Bao M, Liu H, et al. Coronavirus disease 2019 in elderly patients: characteristics and prognostic factors based on 4-week follow-up. J Infect 2020; 80:639–645. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Wu C, Chen X, Cai Y, Xia J, Zhou X, Xu S, et al. Risk factors associated with acute respiratory distress syndrome and death in patients with coronavirus disease 2019 pneumonia in Wuhan, China. JAMA Intern Med 2020; 180:934–943. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Xie J, Zu Y, Alkhatib A, Pham TT, Gill F, Jang A, et al. Metabolic syndrome and COVID-19 mortality among adult black patients in New Orleans. Diabetes Care 2021; 44:188–193. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Xu W, Sun NN, Gao HN, Chen ZY, Yang Y, Ju B, et al. Risk factors analysis of COVID-19 patients with ARDS and prediction based on machine learning. Sci Rep 2021; 11:1–12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Yang Q, Zhou Y, Wang X, Gao S, Xiao Y, Zhang W, et al. Effect of hypertension on outcomes of adult inpatients with COVID-19 in Wuhan, China: a propensity score-matching analysis. Respir Res 2020; 21:1–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Zangrillo A, Beretta L, Scandroglio AM, Monti G, Fominskiy E, Colombo S, et al. Characteristics, treatment, outcomes and cause of death of invasively ventilated patients with COVID-19 ARDS in Milan, Italy. Crit Care Resusc 2020; 22:200–211. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Qin W, Hu BZ, Zhang Z, Chen S, Li FJ, Zhu ZY, et al. Clinical characteristics and death risk factors of severe COVID-19. Zhonghua Jie He He Hu Xi Za Zhi (Chinese) 2020; 43:648–653. [DOI] [PubMed] [Google Scholar]
- 93.Zhang P, Zhu L, Cai J, Lei F, Qin JJ, Xie J, et al. Association of inpatient use of angiotensin-converting enzyme inhibitors and angiotensin II receptor blockers with mortality among patients with hypertension hospitalized with COVID-19. Circ Res 2020; 126:1671–1681. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Zhou F, Yu T, Du R, Fan G, Liu Y, Liu Z, et al. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: a retrospective cohort study. Lancet 2020; 395:1054–1062. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Zhu Z, Cai T, Fan L, Lou K, Hua X, Huang Z, et al. The potential role of serum angiotensin-converting enzyme in coronavirus disease 2019. BMC Infect Dis 2020; 20:1–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Nandy K, Salunke A, Pathak SK, Pandey A, Doctor C, Puj K, et al. Coronavirus disease (COVID-19): a systematic review and meta-analysis to evaluate the impact of various comorbidities on serious events. BMC Infect Dis 2020; 20:1017–1025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Patel U, Malik P, Usman MS, Mehta D, Sharma A, Malik FA, et al. Age-adjusted risk factors associated with mortality and mechanical ventilation utilization amongst COVID-19 hospitalizations-a systematic review and meta-analysis. SN Compr Clin Med 2020; 2:1740–1749. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Drake TM, Riad AM, Fairfield CJ, Egan C, Knight SR, Pius R, et al. Characterisation of in-hospital complications associated with COVID-19 using the ISARIC WHO Clinical Characterisation Protocol UK: a prospective, multicentre cohort study. Lancet 2021; 398:223–237. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.WHO. COVID-19 and the use of angiotensin-converting enzyme inhibitors and receptor blockers. Available at: https://www.who.int/news-room/commentaries/detail/covid-19-and-the-use-of-angiotensin-converting-enzyme-inhibitors-and-receptor-blockers [Accessed 14 June 2021] [Google Scholar]
- 100.Savoia C, Sada L, Zezza L, Pucci L, Lauri FM, Befani A, et al. Vascular inflammation and endothelial dysfunction in experimental hypertension. Int J Hypertens 2011; 2011:281240. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Harrison DG, Guzik TJ, Lob HE, Madhur MS, Marvar PJ, Thabet SR, et al. Inflammation, immunity, and hypertension. Hypertension 2011; 57:132–140. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Lin L, Lu L, Cao W, Li T. Hypothesis for potential pathogenesis of SARS-CoV-2 infection-a review of immune changes in patients with viral pneumonia. Emerg Microbes Infect 2020; 9:727–732. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Tay MZ, Poh CM, Rénia L, MacAry PA, Ng LFP. The trinity of COVID-19: immunity, inflammation and intervention. Nat Rev Immunol 2020; 20:363–374. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Lüscher Thomas F. COVID-19: (mis)managing an announced Black Swan. Eur Heart J 2020; 41:1779–1782. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Furuhashi M, Moniwa N, Mita T, Fuseya T, Ishimura S, Ohno K, et al. Urinary angiotensin-converting enzyme 2 in hypertensive patients may be increased by olmesartan, an angiotensin II receptor blocker. Am J Hypertens 2015; 28:15–21. [DOI] [PubMed] [Google Scholar]
- 106.Lopes RD, Macedo AV, Silva PGMBE, Moll-Bernardes RJ, Dos Santos TM, Mazza L. Effect of discontinuing vs continuing angiotensin-converting enzyme inhibitors and angiotensin II receptor blockers on days alive and out of the hospital in patients admitted with COVID-19: a randomized clinical trial. Jama 2021; 325:254–264. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Trump S, Lukassen S, Anker MS, Chua RL, Liebig J, Thürmann L, et al. Hypertension delays viral clearance and exacerbates airway hyperinflammation in patients with COVID-19. Nat Biotechnol 2021; 39:705–716. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.