To the Editor:
Dihydrofolate reductase (DHFR) is a critical enzyme in folate metabolism that reduces folic acid to dihydrofolic and tetrahydrofolic acid and provides an important target for antineoplastic, antimicrobial, and anti-inflammatory drugs. Defective DHFR activity leads to megaloblastic anemia syndrome combined with severe cerebral folate deficiency, and cerebral tetrahydrobiopterin deficiency due to a germ line missense mutation in DHFR has been reported.1,2 Folate represents a large family of water-soluble vitamins that play an important role in DNA synthesis, repair, and transmethylation pathways.3 Folate is also a substrate for purine and thymidine synthesis and a methyl donor for homocysteine to methionine conversion, with low folate status being reflected by elevated plasma homocysteine concentrations.4 Cerebral tetrahydrobiopterin is required for the formation of dopamine, serotonin, and norepinephrine and the hydroxylation of aromatic amino acids as a link to neurodevelopmental symptoms.5
To date, only 6 patients have been reported with DHFR mutations who presented with a spectrum of neurological symptoms, with hematological findings noted in addition to neurological symptoms in some patients.1,2 We report on a Dutch pedigree with a novel homozygous DHFR mutation.
Patient material was obtained with informed consent by the Erasmus Medical Ethics Committee for the Dutch immunodeficiency study (NL40331.078). Whole-genome sequencing and whole-exome sequencing and mutation analysis were performed according to reported procedures.6, 7, 8
Lymphocyte phenotyping CD4+/CD8+ T cells, B cells, naïve/memory T- and B-cell subsets, and NK cells was carried out using immunostaining for flowcytometry as described.9 Enzymatic DHFR activity was defined using Epstein-Barr virus–transformed B-lymphoblastoid cell lines according to previously used methods.2
The index patient presented at 2 months of age. He was the third child of White parents. At his first immunization, he had fever and was admitted to the hospital where he received a diagnosis of moderate megaloblastic anemia and thrombocytopenia. Extensive metabolic tests of blood and urine were noninformative (Table 1). Hydroxocobalamin and folic acid suppletion remained without effect. At 4 months of age, the boy’s health deteriorated, and symptoms such as failure to thrive, dyspnea, coughing, and vomiting were observed. Bronchoalveolar lavage detected Pneumocystis jirovecii, and the infection was treated with high-dose co-trimoxazole (trimethoprim-sulfamethoxazole [TMP-SMX]) and prednisolone. Because of progressive respiratory failure, the index died a week later. Blood erythrocyte sedimentation rate and C-reactive protein levels were low in the presence of leukocytosis, increased lactate dehydrogenase, ferritin, and liver enzyme tests (Table 1). We observed normal absolute lymphocyte counts, including CD4/CD8 ratio and naïve/memory proportions when compared with age-matched controls, normal lymphocyte proliferation, and normal CD40L upregulation upon in vitro activation of T cells (data not shown).9 Despite normal serum folate levels, there was persistent megaloblastic anemia in the bone marrow (Figure 1A,B).
Table 1.
Hematology, chemistry and metabolite parameters in blood and CSF
| At presentation (Blood) | Case 1 | Case 2 | Case 3 | N ranges |
|---|---|---|---|---|
| ESR (mm/1st h) | 12 | N.D. | N.D. | <20 |
| CRP (mg/L) | <3 | 4 | 14 | <5 |
| Hb (mmol/L) | 5.2 | 2.0 | 4.0 | 6.0-8.9 |
| MCV (fL) | 89.7∗ | 110 | 85 | 75-95 |
| Reticulocytes (%) | 1.4 | 2.0 | 0.7 | 0.5-2 |
| Red cell distribution width (%) | 15.2 | 17.2 | 22.4 | 12-16 |
| Leukocytes (109/L) | 29.3 | 68.9 | 11.8 | 6-17 |
| Thrombocytes (109/L) | 147 | 159 | 93 | 150-600 |
| % neutrophils | 50.4 | N.D. | 60 | 35-75 |
| % eosinophils | 26.7 | N.D. | 14 | 2-10 |
| % lymphocytes | 21.7 | N.D. | 22 | 17-42 |
| % monocytes | 2.2 | N.D. | 1 | 5-12 |
| % basophils | 0 | N.D. | 0 | 0-2 |
| % blasts | 0 | N.D. | 0 | neg |
| Coombs test | neg | N.D. | N.D. | neg |
| LDH (U/L) | 2294 | 8298 | 3519 | <300 |
| Haptoglobin (mg/L) | 1.2 | <0.10 | <0.10 | 0-2 |
| Ferritin (μg/L) | 860 | N.D. | 5200 | <250 |
| Folic acid (nmol/L) | 4 | N.D. | 11.6 | 10-50 |
| VitB12 (pmol/L) | 359 | N.D. | 217 | 150-700 |
| Homocysteine (μmol/L) | 14† | N.D. | 15† | 0-9 |
| Transcobalamin | normal | N.D. | N.D. | present |
| Thrombopoietin (U/mL) | 280 | N.D. | N.D. | <40 |
| Creatinine (μmol/L) | 26 | 32 | 32 | 35-80 |
| ASAT (U/L) | 47 | 330 | 22 | <50 |
| ALAT (U/L) | 26 | 131 | 19 | <60 |
| CSF (with folinic acid supplementation) | ||||
| 5-MTHF (nmol/L) | N.D. | N.D. | 374 | 105-233 |
| Biopterin (nmol/L; for BH4+BH2) | N.D. | N.D. | 22 | 10-50 |
Values represent the cell numbers and concentrations of the indicated parameters determined during the first week upon admission. Values outside of the age-related normal reference ranges are marked in bold.
ALAT, alanine amino transferase; ASAT, aspartate amino transferase; CRP, C-reactive protein; ESR, erythrocyte sedimentation rate; Hb, hemoglobin; LDH, lactate dehydrogenase; MCV, mean corpuscular volume; 5-MTHF, 5-methyltetrahydrofolate.
Value at admission at an academic center, after the start of folic acid and vitamin B12 supplementation because of anemia and MCV of 105 fL. At admission, the bone marrow biopsy still showed megaloblastic anemia (Figure 1A).
Mildly increased, not compatible with homozygous MTHFR, CBS, or MS enzyme defect.
Figure 1.
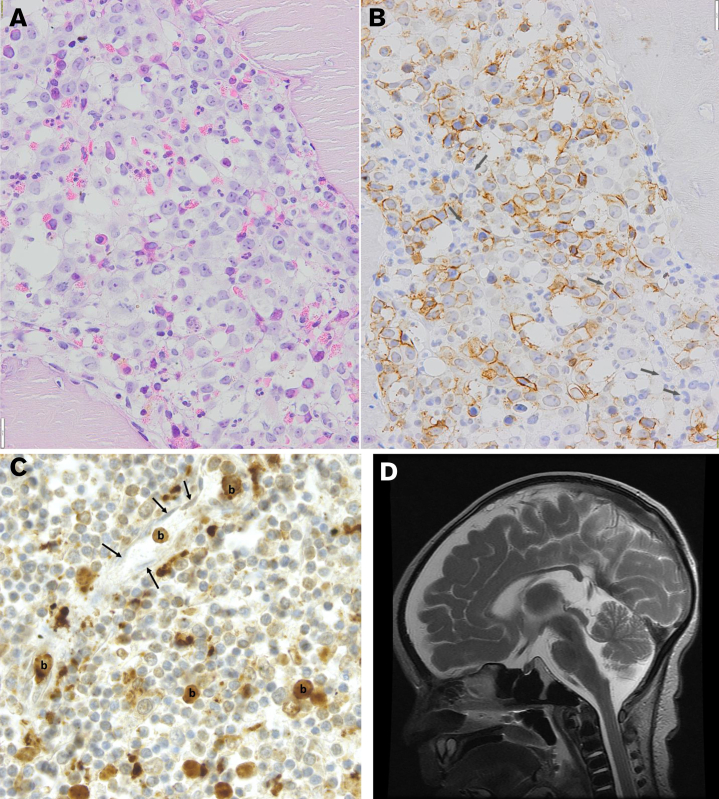
The presence of major symptoms in DHFR deficiency. (A) Bone marrow biopsy showing a severely impaired erythroid outgrowth and a myeloid differentiation defect with marked megaloblastic features (hematoxylin and eosin; bar, 20 μm). (B) Immunohistochemistry with antiglycophorin A, demonstrating the presence of multiple megaloblasts and an almost complete absence of late-stage erythroid precursors. Among the unstained (myeloid) cells, several giant bands are identified (arrows). (C) AML was suggested in the second case. Immunohistochemically stained slide of the spleen. CD33-positive blasts marked as (b) can be observed in lytic tissue of the spleen. Some of these blasts are located in capillaries. The nuclei of the endothelial cells lining the wall of these capillaries are indicated with an arrow. Similar infiltration of MPO/CD33+ myeloid blasts was observed in the liver and bone marrow. The child died at presentation in the emergency room. The diagnosis of infantile AML was made post mortem from the obtained autopsy samples. (D) T2 magnetic resonance imaging in patient 3 demonstrating both cerebral and cerebellar atrophy and hypoplasia of the cerebellar vermis.
The second patient was born as the second child to healthy parents. She presented after a choking incident with hypothermia (34.5°C) at 2 months of age in a moribund state with deep anemia (hemoglobin 2.0 mmol/L). A postmortem examination revealed hepatosplenomegaly and enlarged lymph nodes. Although the diagnosis is limited because of late sampling in a very poor condition and myelodysplasia cannot be formally excluded, the diagnosis of acute myeloid leukemia (AML) was made after extensive discussion of the case, based on CD33 staining and partly myeloperoxidase-positive blasts (Figure 1C).
The third patient was born to the same parents described in the previous instance. At 6 weeks of age, he was treated for herpes stomatitis and developed unexplained anemia for which he received red blood cell transfusions. A bone marrow smear at 7 weeks of age showed decreased erythropoiesis with megaloblasts and dysplastic myelopoiesis with hypersegmented neutrophils. At 13 weeks of age, he was admitted with fever, anemia, hepatomegaly, and dyspnea due to P jirovecii infection. We again observed normal absolute lymphocyte counts, including CD4/CD8 ratio, subsets, and immunoglobulin G (IgG), IgA, and IgM plasma levels (data not shown). High-dose TMP-SMX and prednisolone therapy was initiated, and extracorporeal membrane oxygenation (ECMO) support was provided. An MRI of the brain showed signs of cortical laminar necrosis, hemorrhagic leukomalacia, vermis inferior hypoplasia, and diffuse tissue loss of the supra and infratentorial parenchyma with normal myelinization (Figure 1D), which could be related to not only DHFR deficiency but also ECMO. While the patient was on ECMO support, supplementation of IV folinic acid was initiated after (genetic) diagnosis. The child’s condition improved, and after initial improvement and detubation, pulmonary hypertension and respiratory insufficiency recurred, and the patient died of these pulmonary complications at the age of 4 months.
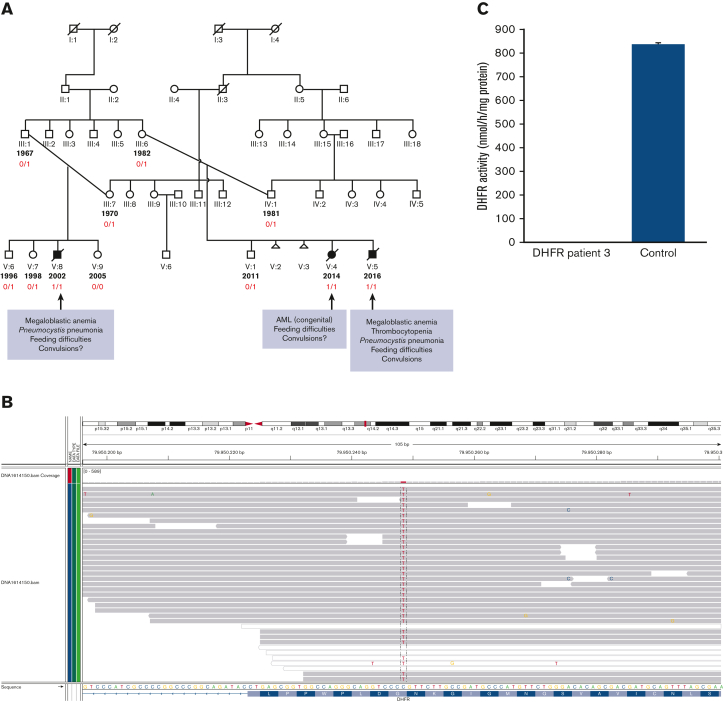
We identified a novel homozygous DHFR mutation in 3 infants by next-generation sequencing, cosegregating with the phenotype across 11 sequenced individuals (Figure 2A) and predicted to have a deleterious effect on protein function, as was biochemically confirmed (Figure 2B,C).
Figure 2.
Pedigree with 3 cases affected by a novel pathogenic DHFR gene mutation. (A) Pedigree of the index case in 1 branch and 2 additional cases diagnosed in another branch of the same family. Segregation studies showed full penetrance in the case of homozygosity, whereas heterozygous carriers had neither clinical symptoms nor any abnormality in the hematological and immunological parameters. Individuals with the year of birth indicated in the extended pedigree have been tested. Filled symbols indicate the 3 homozygous cases in the family (also marked by 1/1 as determined by WGS [whole-genome sequencing]). Carriership of the DHFR mutation is indicated by 0/1, as was confirmed by Sanger sequencing. (B) NGS demonstrating the homozygous mutation in DHFR (NM_000791.3; c.61G>A; p.Gly21Arg) at chromosome 5q14.1 [OMIM 126060]. (C) Absent activity of DHFR in EBV-transformed lymphoblasts from patient 3 vs control (measured in duplicate according to the exact methods as reported by Cario et al).2
Family members were recruited for whole-genome sequencing.7,8 Because of rapid deterioration in the health of the third patient, a clinical single nucleotide polymorphism array analysis and ultrarapid exome sequencing were performed in the meantime.6 Both approaches independently yielded a homozygous novel missense variant in the DHFR gene (NM_000791.3; c.61G>A; p.Gly21Arg), cosegregating with disease status under a recessive mode of inheritance and confirmed by Sanger sequencing (Figure 2). There were no abnormal hematology parameters in the heterozygous carriers (data not shown).
The phenotypes reported thus far were focusing on the neurological aspects of the disease. The first 3 patients reported had severely delayed psychomotor development,1 generalized seizures, and cerebral and cerebellar atrophy, whereas the other 3 siblings were still asymptomatic or had childhood absence epilepsy with eyelid myoclonus and mild learning disabilities.2 Our 3 patients did not have neurological symptoms at the start. The secondary microcephaly reported in the 2 previous publications was absent in our cases.
As expected, neither vitamin B12 nor folic acid in patient 1 improved any of the clinical and laboratory manifestations. In patient 3, we demonstrate that although hematological recovery was seen with treatment administered at the time, ECMO could be stopped. In addition, the supplementation of folinic acid did not prevent the following detrimental course of the disease. Cerebrospinal fluid (CSF) measurement of folate metabolites in this patient showed that folinic acid supplementation led to increased levels of 5- methyl-tetrahydrofolate (THF) in CSF, but a beneficial effect on the neurological outcome could not be acclaimed. This may be related to the complete absence of DHFR activity in this patient (Figure 2C), whereas in previously reported patients, some DHFR activity at rest could still be detected.1,2 A more severe neurological phenotype is less likely to be attenuated by folinic acid supplementation. Therefore, it remains to be shown to what extent folinic acid can affect the course of disease in every case of DHFR deficiency.
The discrepancy in complete recovery of blood but not of cerebral levels has been noted previously.1 The CSF-to-blood folate ratio in healthy humans is 3:1.10 In hereditary folate malabsorption, CSF folate is very low or undetectable, even when blood folate levels in patients are restored to normal or elevated levels through folate supplementation. In addition, in children affected with FOLR1 mutations, a low level of 5- methyl-THF in the CSF with normal serum and erythrocyte folate levels are present.
The active isomers of 5-formyl-THF (leucovorin) or 5-methyl-THF (metafolin) are preferred forms to cross the blood-brain barrier to enter the brain because their affinity for the solute transporter is more than 2 orders of magnitude greater than the affinity of folic acid for this transporter.10
Folate deficiency can cause uracil misincorporation into DNA, leading to chromosome breakage,11 which was proposed to contribute to the increased risk of cancer, consistent with the possible and seemingly congenital AML in the second case in our pedigree.
The increased risk of infections due to an underlying immunodeficiency has been noted before in case of defective folate uptake.10,12,13 Similar to cases 1 and 3 described here, P jiroveci pneumonia has been reported under these folate-deficient conditions.10,12,13 Systemic folate deficiency can be easily mistaken for severe combined immunodeficiency with normal T- and B-cell counts and differentiation,14,15 which may develop before any apparent neurological symptoms to further delay the final diagnosis of an underlying metabolic disorder. However, the high-dose TMP-SMX treatment for P jiroveci may have affected the clinical course in DHFR deficiency because trimethoprim inhibits DHFR.3 The negative contribution of TMP-SMX cannot be excluded, despite the intravenous supplementation of folinic acid and hematological recovery in patient 3.
In summary, we describe a pedigree with previously unrecognized clinical features of DHFR deficiency, an ultrarare inborn error of folate metabolism. The clinical spectrum was characterized by infantile-onset megaloblastic anemia and pneumocystis infection, AML, and pancytopenia with subsequent neurodevelopmental delay unresponsive to treatment. Depending on early diagnosis and the severity of DHFR deficiency, adequate treatment with (parenteral) high-dose 5-formyl-THF may attenuate neurologic features of the disease but could be less effective in the complete absence of any remaining DHFR activity.
Contribution: T.W.K. has written and designed the study; T.W.K., A.C.H.d.V., S.d.P., and H.H.H were clinically involved; A.A.M.E., E.M.v.L., D.E.C.S., and M.M.C.W. were responsible for laboratory studies; S.T.P. contributed with histochemical stainings; M.R.N., H.LA., B.H.B.B., and A.W. were all responsible for genetic testing and counseling; and all authors contributed to the text and critically reviewed the manuscript.
Conflict-of-interest disclosure
The authors declare no competing financial interests.
A complete list of the members of the NBR-RD PID Consortium appears in “Appendix.”
Acknowledgments
The authors are very grateful to King Lam, hemopathologist at Erasmus MC hospital, for sharing the available immunohistochemistry picture of patient 2. The authors also thank the family for supporting and approving the publication to properly describe and illustrate this often unrecognized and unknown disorder. The authors also thank NIHR BioResource volunteers for their participation, and gratefully acknowledge NIHR BioResource centres, NHS Trusts and staff for their contribution. The authors also thank the National Institute for Health Research and NHS Blood and Transplant. The views expressed are those of the author(s) and not necessarily those of the NHS, the NIHR or the Department of Health and Social Care.
This study was supported by The National Institute for Health Research England (grant number RG65966) and the Center of Immunodeficiencies Amsterdam (CIDA-2015).
Footnotes
Data are available on request from the corresponding author, Taco W. Kuijpers (t.w.kuijpers@amsterdamumc.nl).
Appendix
The members of the NBR-RD PID Consortium are: Zoe Adhya, Hana Alachkar, Carl E. Allen, Ariharan Anantharachagan, Richard Antrobus, Gururaj Arumugakani, Chiara Bacchelli, Helen E. Baxendale, Claire Bethune, Shahnaz Bibi, Barbara Boardman, Claire Booth, Matthew Brown, Michael J. Browning, Mary Brownlie, Matthew S. Buckland, Siobhan O. Burns, Oliver S. Burren, Anita Chandra, Ivan K. Chinn, Hayley Clifford, Nichola Cooper, Godelieve J. de Bree, E. Graham Davies, Sarah Deacock, John Dempster, Lisa A. Devlin, Elizabeth Drewe, J. David M. Edgar, William Egner, Shuayb El Khalifa, Tariq El-Shanawany, James H.R. Farmery, H. Bobby Gaspar, Rohit Ghurye, Kimberly C. Gilmour, Sarah Goddard, Pavels Gordins, Sofia Grigoriadou, Scott J. Hackett, Rosie Hague, Lorraine Harper, Grant Hayman, Archana Herwadkar, Stephen Hughes, Aarnoud P. Huissoon, Stephen Jolles, Julie Jones, Yousuf M. Karim, Peter Kelleher, Sorena Kiani, Nigel Klein, Taco W. Kuijpers, Dinakantha S. Kumararatne, James Laffan, Hana Lango Allen, Sara E. Lear, Hilary Longhurst, Lorena E. Lorenzo, Paul A. Lyons, Jesmeen Maimaris, Ania Manson, Elizabeth M. McDermott, Hazel Millar, Anoop Mistry, Valerie Morrisson, Sai H.K. Murng, Iman Nasir, Sergey Nejentsev, Sadia Noorani, Eric Oksenhendler, Mark J. Ponsford, Waseem Qasim, Ellen Quinn, Isabella Quinti, Paula Rayner-Matthews, Alex Richter, Crina Samarghitean, Ravishankar B. Sargur, Sinisa Savic, Suranjith L. Seneviratne, W.A. Carrock Sewell, Fiona Shackley, Olga Shamardina, Ilenia Simeoni, Kenneth G.C. Smith, Emily Staples, Hans Stauss, Cathal L. Steele, Kathleen E. Stirrups, James E. Thaventhiran, David C. Thomas, Moira J. Thomas, Adrian J. Thrasher, John A. Todd, Anton T.J. Tool, Salih Tuna, Rafal D. Urniaz, Steven B. Welch, Lisa Willcocks, Sarita Workman, Austen Worth, Nigel Yeatman, and Patrick F.K. Yong.
References
- 1.Banka S., Blom H.J., Walter J., et al. Identification and characterization of an inborn error of metabolism caused by dihydrofolate reductase deficiency. Am J Hum Genet. 2011;88(2):216–225. doi: 10.1016/j.ajhg.2011.01.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Cario H., Smith D.E., Blom H., et al. Dihydrofolate reductase deficiency due to a homozygous DHFR mutation causes megaloblastic anemia and cerebral folate deficiency leading to severe neurologic disease. Am J Hum Genet. 2011;88(2):226–231. doi: 10.1016/j.ajhg.2011.01.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Zheng Y., Cantley L.C. Toward a better understanding of folate metabolism in health and disease. J Exp Med. 2019;216(2):253–266. doi: 10.1084/jem.20181965. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Blom H.J., Smulders Y. Overview of homocysteine and folate metabolism. With special references to cardiovascular disease and neural tube defects. J Inherit Metab Dis. 2011;34(1):75–81. doi: 10.1007/s10545-010-9177-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Watkins D., Rosenblatt D.S. Lessons in biology from patients with inherited disorders of vitamin B12 and folate metabolism. Biochimie. 2016;126:3–5. doi: 10.1016/j.biochi.2016.05.001. [DOI] [PubMed] [Google Scholar]
- 6.Arts P., Simons A., AlZahrani M.S., et al. Exome sequencing in routine diagnostics: a generic test for 254 patients with primary immunodeficiencies. Genome Med. 2019;11(1):38. doi: 10.1186/s13073-019-0649-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Thaventhiran J.E.D., Lango Allen H., Burren O.S., et al. Whole-genome sequencing of a sporadic primary immunodeficiency cohort. Nature. 2020;583(7814):90–95. doi: 10.1038/s41586-020-2265-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Turro E., Astle W.J., Megy K., et al. Whole-genome sequencing of patients with rare diseases in a national health system. Nature. 2020;583(7814):96–102. doi: 10.1038/s41586-020-2434-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Tuijnenburg P., Lango Allen H., Burns S.O., et al. NIHR BioResource-Rare Diseases Consortium Loss-of-function nuclear factor κB subunit 1 (NFKB1) variants are the most common monogenic cause of common variable immunodeficiency in Europeans. J Allergy Clin Immunol. 2018;142(4):1285–1296. doi: 10.1016/j.jaci.2018.01.039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Zhao R., Min S.H., Qiu A., et al. The spectrum of mutations in the PCFT gene, coding for an intestinal folate transporter, that are the basis for hereditary folate malabsorption. Blood. 2007;110(4):1147–1152. doi: 10.1182/blood-2007-02-077099. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Blount B.C., Mack M.M., Wehr C.M., et al. Folate deficiency causes uracil misincorporation into human DNA and chromosome breakage: implications for cancer and neuronal damage. Proc Natl Acad Sci USA. 1997;94(7):3290–3295. doi: 10.1073/pnas.94.7.3290. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Malatack J.J., Moran M.M., Moughan B. Isolated congenital malabsorption of folic acid in a male infant: insights into treatment and mechanism of defect. Pediatrics. 1999;104(5 Pt 1):1133–1137. doi: 10.1542/peds.104.5.1133. [DOI] [PubMed] [Google Scholar]
- 13.Shin D.S., Mahadeo K., Min S.H., et al. Identification of novel mutations in the proton-coupled folate transporter (PCFT-SLC46A1) associated with hereditary folate malabsorption. Mol Genet Metab. 2011;103(1):33–37. doi: 10.1016/j.ymgme.2011.01.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Borzutzky A., Crompton B., Bergmann A.K., et al. Reversible severe combined immunodeficiency phenotype secondary to a mutation of the proton-coupled folate transporter. Clin Immunol. 2009;133(3):287–294. doi: 10.1016/j.clim.2009.08.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Keller M.D., Ganesh J., Heltzer M., et al. Severe combined immunodeficiency resulting from mutations in MTHFD1. Pediatrics. 2013;131(2):e629–e634. doi: 10.1542/peds.2012-0899. [DOI] [PubMed] [Google Scholar]