Abstract
Introduction
5-fluorouracil (5-FU) is the most widely used chemotherapeutic drug in treating colorectal cancer. However, its toxicity to normal tissues and tumour resistance are the main hurdles to efficient cancer treatment. MiR27-a promotes the proliferation of colon cancer cells by stimulating the Wnt/β-catenin pathway. The present study was conducted to examine whether quercetin (Q) combined with 5-FU improves the anti-proliferative effect of 5-FU on HCT-116 and Caco-2 cell lines through detection of the miR-27a/Wnt/β-catenin signalling pathway.
Material and methods
Cell viability in HCT-116 and Caco-2 cell lines following quercetin and 5-FU treatment alone and in combination for 48 hours was determined using the MTT assay. The flow cytometry, quantitative real-time polymerase chain reaction, and ELISA techniques were used.
Results
Our results showed that combination of quercetin and 5-FU exhibited greater cytotoxic efficacy than did 5-FU alone. Co-administration of both drugs either in combination 1 (1 : 1 Q: 5-FU) or in combination 2 (1 : 0.5 Q: 5-FU) enhanced apoptosis in HCT-116 and Caco-2 cells compared with 5-FU alone and significantly inhibited the expression of miR-27a, leading to upregulation of secreted frizzled-related protein 1 and suppression of Wnt/β-catenin signalling, which was confirmed by a significant decrease in cyclin D1 expression.
Conclusions
Quercetin strongly enhanced 5-FU sensitivity via suppression of the miR-27a/Wnt/β-catenin signalling pathway in CRC, which advocates further research of this combination with the lower dose of 5-FU.
Keywords: colorectal cancer, 5-fluorouracil, quercetin, miR-27a, Wnt/β- catenin
Introduction
Colorectal cancer (CRC) is the second leading cause of cancer-related deaths and the third most common malignancy worldwide [1]. Currently, chemotherapy is the most widely used protocol for treating CRC patients [2]. However, its clinical applications are limited due to its toxic effect on normal cells, in addition to the emergence of drug resistance.
It was shown that the Wnt/β-catenin pathway plays an important role in oncogenesis, tumour progression, and chemoresistance [3, 4]. Development, tissue homeostasis, cell differentiation, and cell proliferation are all significantly influenced by the family of glycosylated lipid-modified proteins known as Wnt-secreted proteins [5]. Numerous types of human malignancies, including CRC, have been linked to the pathophysiology of inappropriate Wnt signalling pathway activation [6]. An intracellular transcriptional coactivator known as β-catenin is stabilized as a result of a complex signalling cascade that is started when Wnt proteins connect to frizzled transmembrane receptors. After β-catenin translocation into the nucleus, β-catenin is able to couple with T cell factor or lymphoid enhancing factor (TCF/LEF) transcription factors activating Wnt target gene transcription. Secreted frizzled-related proteins (SFRPs), a class of secreted proteins, have recently been recognized as extracellular regulators of the Wnt signalling pathway [6]. SFRPs have a cysteine-rich domain homologous to the frizzled receptors. Recent studies have demonstrated down-regulation of secreted frizzled-related protein 1 (SFRP1) in CRC [7, 8]. Furthermore, deregulated β-catenin activation has been linked to the development of drug resistance to conventional chemotherapy [9, 10]. Several studies have shown that inhibiting or silencing Wnt/β-catenin signalling in drug-resistant cancer cells reduces p-glycoprotein levels and reverses microb drug resist to chemotherapeutic agents [11–13].
MiRNAs are small, single-stranded, non-coding RNAs (18–22 nucleotides) that negatively regulate target gene expression by interfering with transcription and translation [14]. They contribute to carcinogenesis as tumour suppressors or oncogenes through the regulation of their target genes [15–18].
It was shown that miRNAs can exhibit crosstalk with key cellular signalling networks including the Wnt/β-catenin cascade [19, 20]. For example, miR-27a, located on chromosome 19 [21], has been shown to function as an oncogene in several human cancers [22–25]. According to recent research, miR-27a can directly target SFRPs, a tumour suppressor protein and Wnt signalling pathway regulator, resulting in potentiation of epithelial-mesenchymal transition in oral squamous carcinoma stem cells and the invasion of human osteosarcoma cells [26, 27]. It was shown that miR-27a induces chemoresistance to tamoxifen in human breast cancer cell lines [28].
Recently, there has been increased interest in the clinical utilization of herbal remedies and natural products as a safe, effective, and low-cost alternative to conventional therapeutics [29–31].
Quercetin (Q) is a bioactive component belonging to the flavonol subclass of flavonoids, which is ubiquitous in various foods [32, 33]. Quercetin exhibits a variety of pharmacologic effects such as antiviral, antibacterial, antioxidant, anticancer, and anti-inflammatory [34]. Several studies have demonstrated that the anticancer effects of Q are mediated through induction of cell cycle arrest, apoptosis, and suppression of proliferation in leukaemia, melanoma, breast cancer, ovarian cancer, lung cancer, and colon cancer cell lines [35, 36]. Quercetin has been reported to inhibit cell growth through the inhibition of miR-27a in renal cancer cells [37] and colon cancer cells [38]. Moreover, miR-27a was shown to stimulate the proliferation and invasiveness of HCT-116 colon cancer cells by targeting SFRP1 through the Wnt/β-catenin signalling pathway [39].
The goal of this study was to examine whether Q combined with 5-fluorouracil (5-FU) would improve the antiproliferative effects of chemotherapy in HCT-116 and Caco-2 cells compared with 5-FU alone through the detection of the Wnt/β-catenin signalling pathway. In addition, in light of what was previously published about the effect of Q on oncogenic miR-27a, the present study further examined the potential effect of miR-27a targeting on enhancing the chemotherapeutic effect of 5-FU.
Material and methods
Chemicals and reagents
Quercetin (#SLBV2993), 5-FU (#MKBX3795V), foetal bovine serum (FBS), 3-(4,5-dimethylthiazol-2-yl)-2,5- diphenyltetrazolium bromide (MTT), and dimethyl sulfoxide (DMSO) were purchased from Sigma-Aldrich (St. Louis, MO, USA). Trypsin, Dulbecco’s Modified Eagle’s Medium (DMEM), phosphate-buffered saline (PBS), and penicillin/streptomycin antibiotics were obtained from GIBCO (New York, USA). L-glutamine (2%) was purchased from Invitrogen (New York, USA). Ethanol was obtained from the El Nasr Pharmaceutical Chemicals Co. (Cairo, Egypt).
Cell lines and cell culture
Two human colon cancer cell lines, HCT-116 and Caco-2, were procured from the American Type Culture Collection (ATTC, Manassas, VA, USA). The cells were cultured in DMEM supplemented with 10% FBS and 1% penicillin/streptomycin and were incubated in a 5% carbon dioxide and 95% air atmosphere at 37°C (Thermo Electron Co., Waltham, Massachusetts, USA). Stock solutions of Q and 5-FU were prepared by dissolving in DMSO and diluting with DMEM to the appropriate concentration [40]. Dulbecco’s Modified Eagle’s Medium containing equivalent amounts of DMSO was used as a control for each cell line.
Cell viability by MTT assay
The effect of Q and/or 5-FU on cell viability was evaluated by MTT assay as described by Van Meerloo et al. [41]. Cells were seeded in 96-well plates (1 × 104 cells/well) and maintained overnight at 37°C, old media was aspirated, and 100 µl of treatment media containing Q concentrations (0, 3.125, 6.25, 12.5, 25, 50, and 100 µg/ml) and/or 5-FU (0, 15.6, 31.25, 62.5, 125, 250, and 500 µg/ml) were added and incubated for 48 hours. The cells were then treated with MTT (5 mg/ml) for 4 hour, and the resulting formazan crystals were dissolved in 100 µl DMSO. The optical density (OD) was recorded at 570 nm using a microplate reader. Experiments were performed in triplicate in 3 independent experiments [42–44]. The growth inhibition rate was expressed as: growth inhibition (%) = (1-OD of treated/OD of control) × 100. The linear regression method was used to calculate the IC50 values of both Q and 5-FU.
Experimental design
Cells were divided into the following 5 groups: Group I (control group), cells were treated with 1% DMSO; Group II, cells were treated with 5-FU at its IC50 value; Group III, cells were treated with Q at its IC50 value; Group IV combination 1 (QFH), cells were treated with a combination of Q and 5-FU at concentrations based on IC50 values (1 : 1 Q: 5-FU); and Group V combination 2 (QFL), cells were treated with a combination of Q and 5-FU at concentrations based on IC50 values (1 : 0.5 Q: 5-FU). Experiments were performed in triplicate in 3 independent experiments. All treatments were done using HCT-116 and Caco-2 cells at 70–80% confluence. The cells were incubated in a CO2 incubator for 48 hours, trypsinized, and immediately prepared for molecular analysis. Supernatants were kept at –80ºC for cyclin D1 detection by ELISA.
Annexin V-FITC/PI double staining for flow-cytometric apoptosis detection
Apoptotic cells were quantified using an Annexin V-FITC-propidium iodide (PI) double staining kit (Cat. No. 556547 BD Pharmingen, San Jose, CA, USA) according to the manufacturer’s instructions. Each of the HCT-116 and Caco-2 cells was plated in a 24-well plate (5 ´ 105 cells/well) and then incubated for 24 hours for attachment, and then the cells were treated with Q and/or 5-FU, as in the experimental design, for 48 hours. After cell collection by trypsinization, HCT-116 and Caco-2 cells were washed with PBS and stained by annexin V/propidium (BD Biosciences) based on the manufacturer’s instructions for 25 minutes at room temperature in a dark place. The stained cells were analysed using an Attune flow cytometer (Applied Bio-system, USA). Experiments were performed in triplicate.
Quantitative real-time polymerase chain reaction
Total RNA was extracted from HCT-116 and Caco-2 cells using the miRNA Easy Kit (Cat. No. 217004 Qiagen Strasse, Hilden, Germany), and the RNA concentration and purity were measured using a NanoDrop 1000 Spectrophotometer (Thermo Fisher Scientific, Wilmington, DE, USA). The miScript miRNA assay system (Qiagen Strasse, Hilden, Germany) was used to measure miR-27a-3p expression with U6 snRNA as the internal control. For the measurement of WNT1, SFRP1, and β-catenin mRNA expression, RNA was reverse-transcribed into cDNA using the HiSenScript™ RH (–) cDNA Synthesis Kit (iNtRON Biotechnology, Inc., Korea), and quantitative real-time PCR analysis was done using TOPreal™ qPCR 2X PreMix (Cat. No. RT500S Enzynomics, Korea) along with specific primers (Thermo Fisher Scientific, UK), as shown in Table 1. Glyceraldehyde-3-phosphate dehydrogenase was used as an internal control. Calculation of relative expression was done as described by Livak et al. [45].
Table 1.
Primers of MiR-27a-3p, U6, β-catenin, WNT1, secreted frizzled-related protein 1, and glyceraldehyde-3-phosphate dehydrogenase were designed using the National Centre for Biotechnology Information reference sequence
| Primer name | Sequences | |
|---|---|---|
| MiR-27a-3p | ||
| Tm = 60.73 | F: 5' TGCGGTTCACAGTGGCTAAG 3' | |
| Tm = 59.31 | R: 5' CTCAACTGGTGTCGCTGGA 3' | |
| U6 | ||
| Tm = 59.29 | F: 5' CTCGCTTCGGCAGGCACA 3' | |
| Tm = 60.07 | R: 5' AACGCTTCACGAATTTGCGT 3' | |
| β-catenin | ||
| Tm = 60.39 | F: 5' GGGATCAAACCTGACAGCCA 3' | |
| Tm = 58.13 | R: 5' GAAAACGCCATCACCACGTC 3' | |
| WNT1 | ||
| Tm = 60.83 | F: 5' ATTGTCTGCGCCCCTAACC 3' | |
| Tm = 59.01 | R: 5' CGGCACCGCCTCTTATAGTT 3' | |
| SFRP1 | ||
| Tm = 59.11 | F: 5' CAGGAAGAACTAGAGGAACG 3' | |
| Tm = 59.91 | R: 5' TACTGGCCGTTCTGACCTGT 3' | |
| GAPDH | ||
| Tm = 68.03 | F: 5' AATGGGCAGCCGTTAGGAAA 3' | |
| Tm = 59.21 | R: 5' GCCAATACGACCAATCAGAG 3' | |
GAPDH – glyceraldehyde-3-phosphate dehydrogenase, SFRP1 – secreted frizzled-related protein 1
Detection of cyclin D1 by ELISA
The ELISA technique was used to determine the cyclin D1 levels. A specific antibody for cyclin D1 (Cat. No. LS-F21523, LifeSpan BioSciences, Inc., USA) was precoated separately onto microplates. The standard and samples were then added to the wells to form an immobilized antibody complex. Biotin-conjugated antibody was added, followed by avidin-conjugated horseradish peroxidase. A substrate solution was then added to the wells for colour development. A sulphuric acid stop solution was added to terminate colour development, and the OD was measured for each well at a wavelength of 450 nm. The colour intensity was measured relative to the quantity of cyclin D1.
Statistical analysis
Data are expressed as the mean ± standard deviation (SD). Results were analysed by a one-way ANOVA test. A post hoc Tukey’s multiple comparison test was used for multiple comparison analysis. Significant differences among the means were considered at p < 0.05. Statistical analysis and graphical presentation of the data were done using the Graph Pad Prism® software package version 8.0.2 (GraphPad Software Inc., CA, USA).
The datasets used and/or analysed during the current study are available from the corresponding author on reasonable request.
The study was conducted according to the guidelines of the Declaration of Helsinki and approved by the Institutional Review Board of the Faculty of Pharmacy’s Ethics Committee, Damanhour University.
Results
The effect of quercetin and 5-fluorouracil on HCT-116 and Caco-2 cell growth
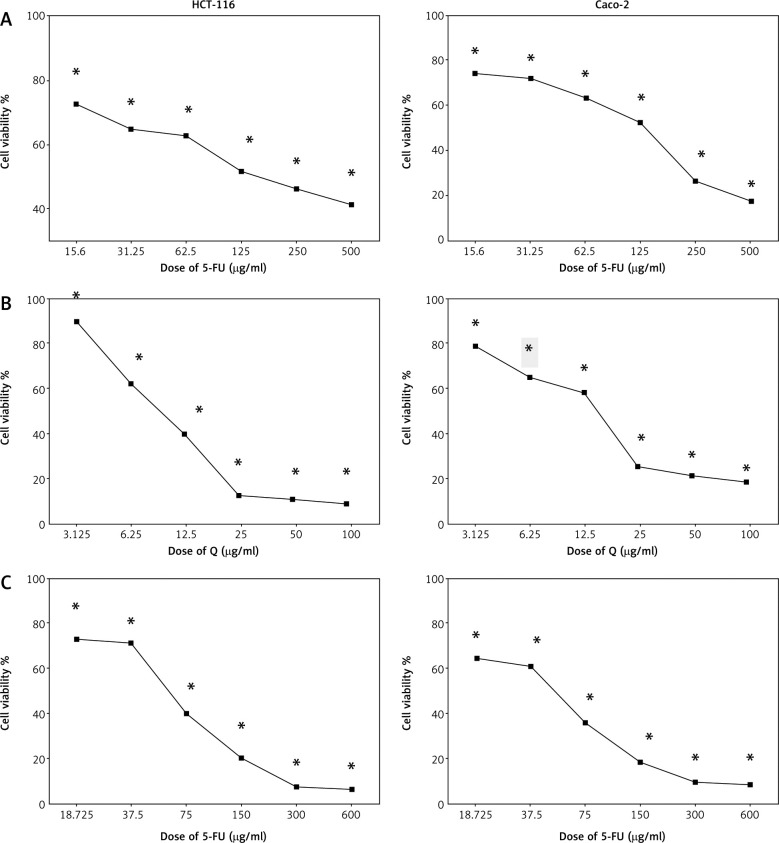
Figure 1 shows the cell viability of HCT-116 and Caco-2 cells treated with different concentrations of Q and/or 5-FU for 48 hours. Both Q and 5-FU decreased the viability of the 2 cell lines depending on the dosage when used alone. Q and 5-FU showed IC50 = 12.36 µg/ml and 125 µg/ml, respectively, on HCT-116, and showed IC50 = 15 µg/ml and 133 µg/ml, respectively, on Caco-2 cells. Furthermore, the combination of Q and 5-FU showed higher antiproliferative activity compared with 5-FU alone in both cell lines.
Fig. 1.
The effect of 5-fluorouracil (5-FU) (15.6–500 µg/ml) (A), Q (3.125–100 µg/ml) (B), and combinations of quercetin and 5-FU on HCT-116 and Caco-2 cells for 48 hours (C)
Data are represented as the mean ±SD; each data point represented an average of 3 independent experiments (triplet). *p < 0.05 indicates a significant difference for Q and/or 5-FU vs. the control group.
The effect of quercetin and 5-fluorouracil on apoptosis of HCT-116 and Caco-2 cells
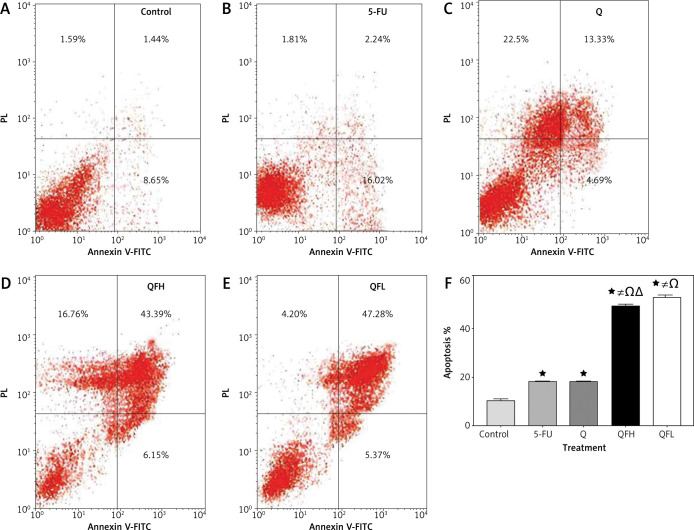
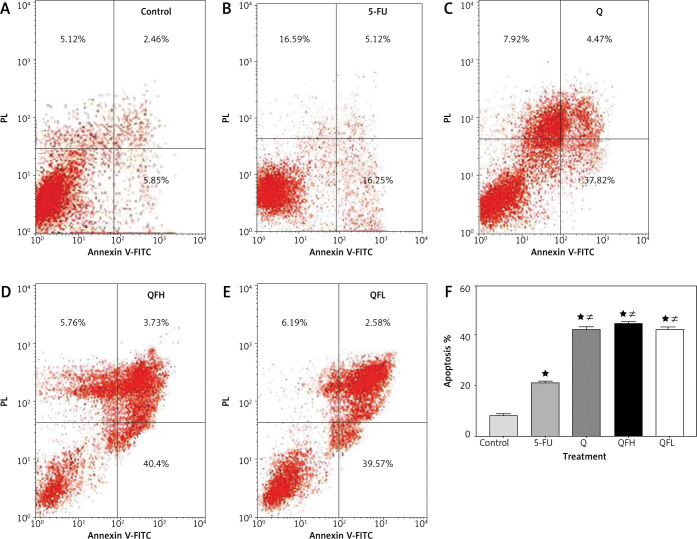
As the pharmacodynamic endpoint of cancer therapy, an apoptosis assay was performed to investigate the Q enhancing effect on 5-FU-induced apoptosis. Figures 2 and 3 show the apoptotic effects of the 5-FU, Q, QFH, and QFL. The percentages of total apoptosis (early apoptosis plus late apoptosis) following treatment with 5-FU, Q, QFH, and QFL, respectively, were 18.25%, 18.1%, 49%, and 52.9% on HCT-116 cells. Compared to the 5-FU group, Q/5-FU combinations significantly (p < 0.001) increased apoptosis (Fig. 2). The Caco-2 cell apoptosis percentages using 5-FU, Q, QFH, and QFL application, respectively, were 21.12%, 42.26%, 44.44%, and 42.38%. Compared to the 5-FU group, Q/5-FU combinations significantly (p < 0.001) increased apoptosis (Fig. 3).
Fig. 2.
The effect of 5-fluorouracil (5-FU) and quercetin (Q) on the apoptosis of HCT-116 cells for 48 hours. Dot-plots from flow cytometric illustrating apoptotic status in HCT-116 cells. Cells in the upper-left quadrant are necrotic, cells in the upper-right quadrant have late apoptosis, the lower-left quadrant shows normal living cells, and the cells in the lower-right quadrant are early apoptotic cells (A–E), the total percentage of apoptosis in HCT-116 cells treated with the indicated concentrations of 5-FU, Q, QFH, and QFL for 48 hours (F)
All data are expressed as the mean ±SD. Statistically significant differences between groups are designated as *p < 0.05 vs. control group, #p < 0.05 vs. 5-FU group and Ωp < 0.05 vs. Q group and ∆p < 0.05 vs. QFL group.
Fig. 3.
The effect of 5-fluorouracil (5-FU) and quercetin (Q) on the apoptosis of Caco-2 cells for 48 hours. Dot-plots from flow cytometric illustrating apoptotic status in Caco-2 cells. Cells in the upper-left quadrant are necrotic, cells in the upper-right quadrant have late apoptosis, the lower-left quadrant shows normal living cells, and the cells in the lower-right quadrant are early apoptotic cells(A–E), the total percentage of apoptosis in Caco-2 cells treated with the indicated concentrations of 5-FU, Q, combination 1 (QFH), and combination 2 (QFL) for 48 hours (F) designated as *p < 0.05 vs. control group, #p < 0.05 vs. 5-FU group and Ωp < 0.05 vs. Q group and ∆p < 0.05 vs. QFL group
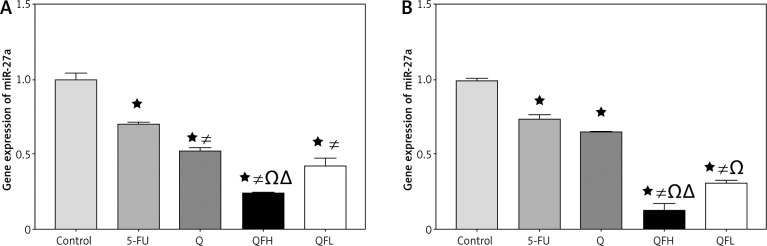
Effect of quercetin and 5-fluorouracil on miR-27a expression
As shown in Figures 4A and B, treatment with Q resulted in a significant downregulation of miR-27a expression compared with the control group in both cell lines. Quercetin and 5-FU co-treatment (combinations 1 and 2) resulted in greater downregulation of miR-27a expression compared with the 5-FU group in both cell lines.
Fig. 4.
The effect of quercetin (Q), 5-fluorouracil (5-FU), combination 1 (QFH) and combination 2 (QFL) on gene expression of miR-27a in HCT-116 cells (A) and Caco-2 cells (B)
Data are expressed as the mean ±SD of 3 samples each performed in triplicate. Data analysis was performed using Graph Pad Prism 8.0.2. Statistically significant differences between groups are designated as *p < 0.05 vs. control, #p < 0.05 vs. 5-FU group, and Ωp < 0.05 vs. Q group and ∆p < 0.05 vs. QFL group.
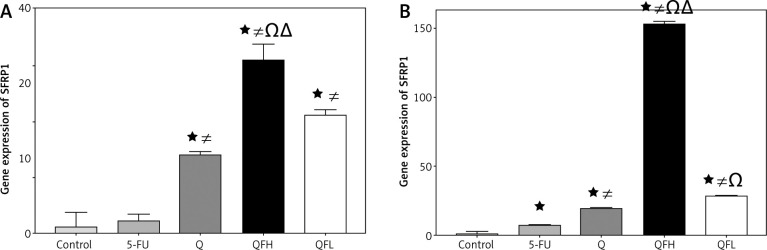
The effect of quercetin and 5-fluorouracil on secreted frizzled-related protein 1 expression
Secreted frizzled-related protein 1 is a negative regulator of Wnt/β-catenin signalling. Secreted frizzled-related protein 1 was significantly upregulated by Q in both cell lines compared with the control groups. Overexpression of SFRP1 was observed following combined treatment with Q and 5-FU (combination 1, 2) compared with the 5-FU group in both cell lines (Figs. 5 A, B).
Fig. 5.
The effect of quercetin (Q), 5-fluorouracil (5-FU), combination 1 (QFH), and combination 2 (QFL) on gene expression of secreted frizzled-related protein 1 in HCT-116 (A) and Caco-2 (B) cells
Data are expressed as the mean ±SD (standard deviation) of 3 samples each performed in triplicate. Data analysis was performed using Graph Pad Prism 8.0.2. Statistically significant differences between group are designated as *p < 0.05 vs. control group, #p < 0.05 vs. 5-FU group and Ωp < 0.05 vs. Q group, and ∆p < 0.05 vs. QFL group.
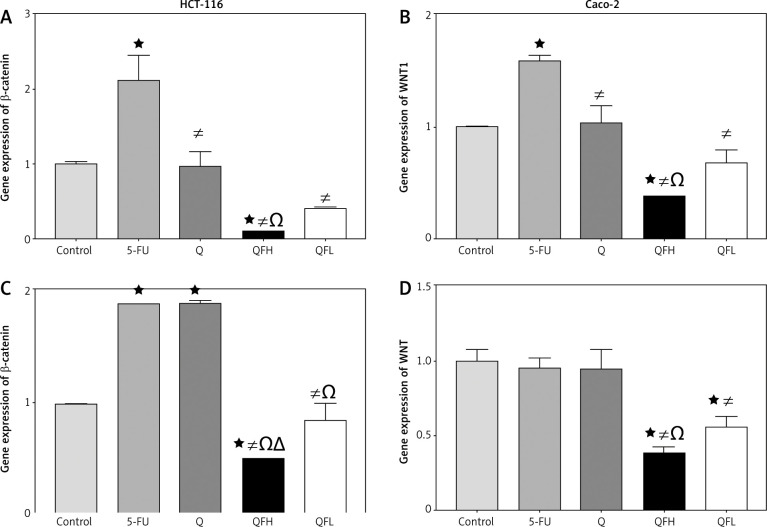
The effect of quercetin and 5-fluorouracil on WNT1 and β-catenin expression
In HCT-116 cells, Q had no significant effect on WNT1 and β-catenin gene expressions compared with the control group. Upon 5-FU treatment, there was a significant upregulation of WNT1 and β-catenin expression compared with the control group. Q and 5-FU co-treatment (combinations 1 and 2) resulted in a significant downregulation of WNT1 and β-catenin expression compared with the 5-FU group (Figs. 6A, B). In the Caco-2 cell line, the quantitative real-time polymerase chain reaction assay showed significant upregulation of β-catenin expression following treatment with Q and 5-FU alone compared with the control group. Q and 5-FU co-treatment (combinations 1 and 2) resulted in a significant downregulation of β-catenin expression compared with 5-FU alone (Fig. 6 C). In Caco-2 cells, Q demonstrated no significant effect on WNT1 expression compared with the control group. Following combined treatment with Q and 5-FU (combinations 1 and 2) there was significant downregulation of WNT1 expression compared with the 5-FU group (Fig. 6 D).
Fig. 6.
The effect of quercetin (Q), 5-fluorouracil (5-FU), combination 1( QFH), and combination 2 (QFL) on gene expression of β-catenin and WNT1 in HCT 116 and Caco-2 cells
Data are expressed as the mean ±SD of 3 samples each performed in triplicate. Data analysis was performed using Graph Pad Prism 8.0.2. Statistically significant differences between groups are designated as *p < 0.05 vs. control group, #p < 0.05 vs. 5-FU group and Ωp < 0.05 vs. Q group, and ∆p < 0.05 vs. QFL group.
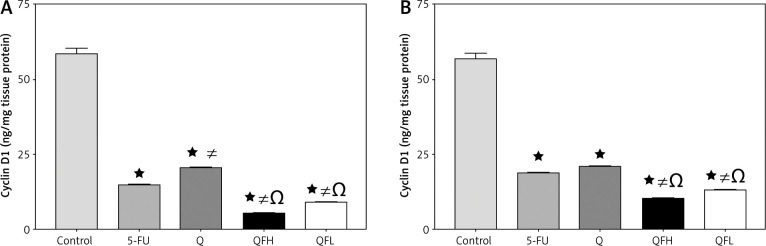
The effect of quercetin and 5-fluorouracil on cyclin D1 expression
The highest level of cyclin D1 was observed in the control groups in both HCT-116 and Caco-2 cell lines. There was a significant reduction in cyclin D1 expression following treatment with Q and 5-FU alone compared with the control group in both cell lines. Significant inhibition of cyclin D1 was observed in the groups treated with QFH and QFL compared with 5-FU alone in both cell lines (p < 0.01) (Figs. 7A, B).
Fig. 7.
The effect of quercetin (Q), 5-fluorouracil (5-FU), combination 1 (QFH), and combination 2 (QFL) on the cyclin D1 level in HCT-116 (A) and Caco-2 (B) cells
Data are expressed as the mean ±SEM (standard error of mean) of 3 samples each performed in triplicate. Data analysis was performed using Graph Pad Prism 8.0.2. Statistically significant differences between groups are designated as *p < 0.05 vs. control group, #p < 0.05 vs. 5-FU group and Ωp < 0.05 vs. Q group, and ∆p < 0.05 vs. QFL group.
Discussion
Colorectal cancer is one of the most frequently occurring malignancies worldwide. 5-fluorouracil is the cornerstone drug used for treating CRC; however, tumour cell resistance and cytotoxicity to normal cells are obstacles to successful treatment [46, 47]. Therefore, new therapeutic strategies are needed to circumvent resistance, alleviate adverse effects, and reduce the dose of chemotherapy [48]. Using natural products in combination with chemotherapy represents a fruitful strategy to increase the sensitivity of cancer cells to chemotherapy (chemo-sensitizers) and reduce the negative effects of chemotherapy (chemo- protectors) [49, 50].
Quercetin either alone or in combination was shown to attenuate the progression of colon cancer through several mechanisms, including cell cycle arrest, decreased cell viability, modulation of oncogenic signalling pathways, induction of apoptosis and autophagy, and inhibition of metastasis. To the best of our knowledge, this is the first study to examine the effect of Q on enhancing the anti-proliferative effects of 5-FU concerning the Wnt/β-catenin signalling pathway through miR-27a modulation in HCT-116 and Caco-2 cell lines.
Our study revealed that a combination of Q ad 5-FU resulted in a higher cytotoxic effect on HCT-116 and Caco-2 cells compared with 5-FU alone. This was shown by a higher apoptotic rate upon treatment with combination 1 and combination 2 compared with 5-FU alone. Our findings are consistent with previous studies. A recent study revealed that a combination of 5-FU and Q enhanced the cytotoxic and apoptotic effects of 5-FU on MCF-7 cells compared to 5-FU alone [51]. As previously reported, 5-FU in combination with Q and melatonin in human liver and colon cancer cells enhances its effect on apoptosis and growth suppression compared to 5-FU alone [52–54]. Curcumin combined with 5-FU stimulated apoptosis and decreased Bcl-2 protein levels in cancer cells [55].
MiR-27a is located on chromosome 19 [21] and has been reported to function as a tumour promoter in different types of human cancers, including breast cancer, gastric adenocarcinoma, HCC, and pancreatic cancer [22–25, 56]. Our results indicated that combination 1 and combination 2 resulted in more significant downregulation in the gene expression of miR-27a than either agent alone. Our findings are in line with previous research examining the effects of a combination of resveratrol and Q (5–20 µg/ml) on cell growth inhibition via the miR-27a reduction in colon cancer cells [38]. Li et al. reported that the combination of Q and hyperoside 5 to 20 µg/ml inhibited miR-27a expression level in 786-O renal cancer cells [37].
The Wnt/β-catenin signalling pathway is one of the main dysregulated pathways in CRC, and it potentiates cell proliferation and drug resistance [57]. Secreted frizzled-related protein 1 is an antagonist of the frizzled receptors and Wnt pathway activation. Previous studies have demonstrated the downregulation of SFRP1 in CRC [7]. Moreover, according to recent research, miR-27a could directly target SFRP1 through Wnt/β-catenin signalling pathway in HCT-116 cells [39]. The present study showed that combinations 1 and 2 inhibited Wnt/β-catenin signalling by significantly upregulating SFRP1 that was a result of higher downregulation of miR-27a expression in both cell lines more than each drug alone. Consistent with previous reports, it was reported that Q increases chemotherapeutic drug sensitivity of K562 and K562R cells by suppressing the Wnt/β-catenin signalling pathway [58]. Another study demonstrated that Q increased the sensitivity of human HCC cells to chemotherapeutic drugs through the FZD7/β-catenin signalling pathway [59]. Another study reported that cardamonin, a natural chalcone, enhanced the antiproliferative effect of 5-FU in gastric cancer cells by targeting the Wnt/β-catenin signalling pathway [60].
Cyclin D1, a target of the β-catenin pathway, is overexpressed in several tumour types and mediates cell cycle progression from the G1 to the S phase [61]. The synergistic effect of combinations 1 and 2 of Q and 5-FU was confirmed by a more significant decline in cyclin D1 levels in both combination groups compared with 5-FU alone. This result is consistent with a previous study that demonstrated that amla extract, which is rich in Q, inhibited human colon cancer stem cells (HCCSC) by targeting the Wnt/β-catenin signalling pathway, resulting in a decrease in cyclin D1 expression [62]. It was noted that the combination of 5-FU and chloroquine caused G1 arrest as well as CDK2 and cyclin D1 downregulation [63].
Conclusions
It was demonstrated that Q combined with 5-FU enhanced the antiproliferative effects of chemotherapy agents in HCT-116 and Caco-2 cells by targeting SFRP1 through the Wnt/β-catenin signalling pathway via miR-27a downregulation. Further studies are recommended for the use of this combination with a reduced dose of chemotherapy.
Footnotes
The authors declare no conflict of interest.
References
- 1.Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin 2018; 68: 394-424. [DOI] [PubMed] [Google Scholar]
- 2.Firouzi Amoodizaj F, Baghaeifar S, Taheri E, et al. Enhanced anticancer potency of doxorubicin in combination with curcumin in gastric adenocarcinoma. J Biochem Mol Toxicol 2020; 34: e22486. [DOI] [PubMed] [Google Scholar]
- 3.Peng C, Zhang X, Yu H, Wu D, Zheng J. Wnt5a as a predictor in poor clinical outcome of patients and a mediator in chemoresistance of ovarian cancer. Int J Gynecol Cancer 2011; 21: 280-288. [DOI] [PubMed] [Google Scholar]
- 4.Vangipuram SD, Buck SA, Lyman WD. Wnt pathway activity confers chemoresistance to cancer stem-like cells in a neuroblastoma cell line. Tumor Biol 2012; 33: 2173-2183. [DOI] [PubMed] [Google Scholar]
- 5.Cadigan KM, Nusse R. Wnt signaling: a common theme in animal development. Genes Dev 1997; 11: 3286-3305. [DOI] [PubMed] [Google Scholar]
- 6.Surana R, Sikka S, Cai W, et al. Secreted frizzled-related proteins: implications in cancers. Biochim Biophys Acta 2014; 1845: 53-65. [DOI] [PubMed] [Google Scholar]
- 7.Qi J, Zhu JQ, Luo J, Tao WH. Hypermethylation and expression regulation of secreted frizzled-related protein genes in colorectal tumor. World J Gastroenterol 2006; 12: 7113-7117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Huang S, Zhong XM, Gao J, et al. Coexpression of SFRP1 and WIF1 as a prognostic predictor of favorable outcomes in patients with colorectal carcinoma. Biomed Res Int 2014; 2014: 256723. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Noda T, Nagano H, Takemasa I, et al. Activation of Wnt/β-catenin signalling pathway induces chemoresistance to interferon-α/5-fluorouracil combination therapy for hepatocellular carcinoma. Br J Cancer 2009; 100: 1647-1658. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Xu N, Shen C, Luo Y, et al. Upregulated miR-130a increases drug resistance by regulating RUNX3 and Wnt signaling in cisplatin-treated HCC cell. Biochem Biophys Res Commun 2012; 425: 468-472. [DOI] [PubMed] [Google Scholar]
- 11.Ma Y, Ren Y, Han EQ, et al. Inhibition of the Wnt-β-catenin and Notch signaling pathways sensitizes osteosarcoma cells to chemotherapy. Biochem Biophys Res Commun 2013; 431: 274-279. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Shen DY, Zhang W, Zeng X, Liu CQ. Inhibition of Wnt/β-catenin signaling downregulates P-glycoprotein and reverses multi-drug resistance of cholangiocarcinoma. Cancer Sci 2013; 104: 1303-1308. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Wei Y, Shen N, Wang Z, et al. Sorafenib sensitizes hepatocellular carcinoma cell to cisplatin via suppression of Wnt/β-catenin signaling. Mol Cell Biochem 2013; 381: 139-144. [DOI] [PubMed] [Google Scholar]
- 14.Calin GA, Cimmino A, Fabbri M, et al. MiR-15a and miR-16-1 cluster functions in human leukemia. Proc Natl Acad Sci 2008; 105: 5166-5171. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Iorio MV, Croce CM. MicroRNAs in cancer: small molecules with a huge impact. J Clin Oncol 2009; 27: 5848. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Kwak PB, Iwasaki S, Tomari Y. The microRNA pathway and cancer. Cancer Sci 2010; 101: 2309-2315. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Ruan K, Fang X, Ouyang G. MicroRNAs: novel regulators in the hallmarks of human cancer. Cancer Lett 2009; 285: 116-126. [DOI] [PubMed] [Google Scholar]
- 18.Sumazin P, Yang X, Chiu HS, et al., An extensive microRNA-mediated network of RNA-RNA interactions regulates established oncogenic pathways in glioblastoma. Cell, 2011. 147(2): p. 370-381. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Wang L, Zhang X, Sheng L, Qiu C, Luo R. LINC00473 promotes the Taxol resistance via miR-15a in colorectal cancer. Biosci Rep 2018; 38: BSR20180790. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Onyido EK, Sweeney E, Nateri AS. Wnt-signalling pathways and microRNAs network in carcinogenesis: experimental and bioinformatics approaches. Mol Cancer 2016; 15: 1-17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Sun Q, Gu H, Zeng Y, et al. Hsa-mir-27a genetic variant contributes to gastric cancer susceptibility through affecting miR-27a and target gene expression. Cancer Sci 2010; 101: 2241-2247. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Mertens-Talcott SU, Chintharlapalli S, Li X, Safe S, et al. The oncogenic microRNA-27a targets genes that regulate specificity protein transcription factors and the G2-M checkpoint in MDA-MB-231 breast cancer cells. Cancer Res 2007; 67: 11001-11011. [DOI] [PubMed] [Google Scholar]
- 23.Guttilla IK, White BA. Coordinate regulation of FOXO1 by miR-27a, miR-96, and miR-182 in breast cancer cells. J Biol Chem 2009; 284: 23204-23216. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Huang S, He X, Ding J, et al. Upregulation of miR-23a approximately 27a approximately 24 decreases transforming growth factor-beta-induced tumor-suppressive activities in human hepatocellular carcinoma cells. Int J Cancer 2008; 123: 972-978. [DOI] [PubMed] [Google Scholar]
- 25.Ma Y, Yu S, Zhao W, Lu Z, Chen J. miR-27a regulates the growth, colony formation and migration of pancreatic cancer cells by targeting Sprouty2. Cancer Lett 2010; 298: 150-158. [DOI] [PubMed] [Google Scholar]
- 26.Mu Y, Zhang L, Chen X, Chen S, Shi Y, Li J. Silencing microRNA-27a inhibits proliferation and invasion of human osteosarcoma cells through the SFRP1-dependent Wnt/β-catenin signaling pathway. Biosci Rep 2019; 39: BSR20182366. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Qiao B, He BX, Cai JH, Tao Q, Lam AKY. MicroRNA-27a-3p modulates the Wnt/β-catenin signaling pathway to promote epithelial-mesenchymal transition in oral squamous carcinoma stem cells by targeting SFRP1. Sci Rep 2017; 7: 1-12. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 28.Ye P, Fang C, Zeng H, et al. Differential microRNA expression profiles in tamoxifen-resistant human breast cancer cell lines induced by two methods. Oncol Lett 2018; 15: 3532-3539. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Badria F, Fathy HM, Fatehe AS, Elimam DMA, Ghazy M. Evaluate the cytotoxic activity of honey, propolis, and bee venom from different localities in Egypt against liver, breast, and colorectal cancer. J Apither 2017; 2: 1-4. [Google Scholar]
- 30.Okda TM, Abd-Elghaffar SK, Katary MA, Abd-Alhaseeb MM. Chemopreventive and anticancer activities of indomethacin and vitamin D combination on colorectal cancer induced by 1, 2-dimethylhydrazine in rats. Biomed Rep 2021; 14: 1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Sabry S, Okda T, Hasan A. Formulation, characterization, and evaluation of the anti-tumor activity of nanosized galangin loaded niosomes on chemically induced hepatocellular carcinoma in rats. J Drug Deliv Sci Technol 2021; 61: 102163. [Google Scholar]
- 32.Zhang H, Zhang H, Zhang M, Yu L, Zhao Y, He N, Yang X. Antitumor activities of quercetin and quercetin-5', 8-disulfonate in human colon and breast cancer cell lines. Food Chem Toxicol 2012; 50: 1589-1599. [DOI] [PubMed] [Google Scholar]
- 33.Chien SY, Wu YC, Chung JG, et al. Quercetin-induced apoptosis acts through mitochondrial-and caspase-3-dependent pathways in human breast cancer MDA-MB-231 cells. Hum Exp Toxicol 2009; 28: 493-503. [DOI] [PubMed] [Google Scholar]
- 34.Erlund I. Review of the flavonoids quercetin, hesperetin, and naringenin. Dietary sources, bioactivities, bioavailability, and epidemiology. Nutrition Res 2004; 24: 851-874. [Google Scholar]
- 35.Samuel T, Fadlalla K, Mosley L, Katkoori V, Turner T, Manne U. Dual-mode interaction between quercetin and DNA-damaging drugs in cancer cells. Anticancer Res 2012; 32: 61-71. [PMC free article] [PubMed] [Google Scholar]
- 36.Hundsberger H, Stierschneider A, Sarne V, et al. Concentration-dependent pro-and antitumor activities of quercetin in human melanoma spheroids: comparative analysis of 2D and 3D cell culture models. Molecules 2021; 26: 717. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Li W, Liu M, Xu YF, et al. Combination of quercetin and hyperoside has anticancer effects on renal cancer cells through inhibition of oncogenic microRNA-27a. Oncol Rep 2014; 31: 117-124. [DOI] [PubMed] [Google Scholar]
- 38.Del Follo-Martinez A, Banerjee N, Li X, Safe S, Mertens-Talcott S. Resveratrol and quercetin in combination have anticancer activity in colon cancer cells and repress oncogenic microRNA-27a. Nutr Cancer 2013; 65: 494-504. [DOI] [PubMed] [Google Scholar]
- 39.Ba S, Xuan Y, Long ZW, Chen CY, Zheng SS. MicroRNA-27a promotes the proliferation and invasiveness of colon cancer cells by targeting SFRP1 through the Wnt/β-catenin signaling pathway. Cell Physiol Biochem 2017; 42: 1920-1933. [DOI] [PubMed] [Google Scholar]
- 40.El-Hanboshy SM, Helmy MW, Abd-Alhaseeb MM. Catalpol synergistically potentiates the anti-tumour effects of regorafenib against hepatocellular carcinoma via dual inhibition of PI3K/Akt/mTOR/NF-κB and VEGF/VEGFR2 signaling pathways. Mol Biol Rep 2021; 48: 7233-7242. [DOI] [PubMed] [Google Scholar]
- 41.Van Meerloo J, Kaspers GJ, Cloos J. Cell sensitivity assays: the MTT assay, in Cancer cell culture. Springer; 2011, 237-245. [DOI] [PubMed] [Google Scholar]
- 42.Helmy MW, Ghoneim AI, Katary MA, Elmahdy RK. The synergistic anti-proliferative effect of the combination of diosmin and BEZ-235 (dactolisib) on the HCT-116 colorectal cancer cell line occurs through inhibition of the PI3K/Akt/mTOR/NF-κB axis. Mol Biol Rep 2020; 47: 2217-2230. [DOI] [PubMed] [Google Scholar]
- 43.Khamis AA, Ali EMM, A Abd El-Moneim M, et al. Hesperidin, piperine and bee venom synergistically potentiate the anticancer effect of tamoxifen against breast cancer cells. Biomed Pharmacother 2018; 105: 1335-1343. [DOI] [PubMed] [Google Scholar]
- 44.Abd-Alhaseeb MM, Massoud SM, Elsayed F, Omran GA, Salahuddin A. Evening primrose oil enhances tamoxifen’s anticancer activity against breast cancer cells by inducing apoptosis, inhibiting angiogenesis, and arresting the cell cycle. Molecules 2022; 27: 2391. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Livak KJ, Schmittgen TD. Analysis of relative gene expression data using real-time quantitative PCR and the 2–∆∆CT method. Methods 2001; 25: 402-408. [DOI] [PubMed] [Google Scholar]
- 46.Abdel Latif Y, El-Bana M, Hussein J, El-Khayat Z, Farrag AR. Effects of resveratrol in combination with 5-fluorouracil on N-methylnitrosourea-induced colon cancer in rats. Comparative Clin Pathol 2019; 28: 1351-1362. [Google Scholar]
- 47.Katona C, Kralovánszky J, Rosta A, Pandi E, Fónyad G, Tóth K, Jeney A. Putative role of dihydropyrimidine dehydrogenase in the toxic side effect of 5-fluorouracil in colorectal cancer patients. Oncology 1998; 55: 468-474. [DOI] [PubMed] [Google Scholar]
- 48.González-Sarrías A, Tomé-Carneiro J, Bellesia A, Tomás-Barberán FA, Espín JC. The ellagic acid-derived gut microbiota metabolite, urolithin A, potentiates the anticancer effects of 5-fluorouracil chemotherapy on human colon cancer cells. Food Funct 2015; 6: 1460-1469. [DOI] [PubMed] [Google Scholar]
- 49.Amiri F, Zarnani AH, Zand H, Koohdani F, Jeddi-Tehrani M, Vafa M. Synergistic anti-proliferative effect of resveratrol and etoposide on human hepatocellular and colon cancer cell lines. Eur J Pharmacol 2013; 718: 34-40. [DOI] [PubMed] [Google Scholar]
- 50.Okda TM, Katry MA, Ragab NM, Shalkami AGS. Phytic acid potentiates oxaliplatin effects in colorectal cancer induced by 1, 2-DMH: the role of miR-224 and miR-200a. Contemp Oncol 2021; 25: 118-124. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Mawalizadeh F, Mohammadzadeh G, Khedri A, Rashidi M. Quercetin potentiates the chemosensitivity of MCF-7 breast cancer cells to 5-fluorouracil. Mol Biol Rep 2021; 48: 7733-7742. [DOI] [PubMed] [Google Scholar]
- 52.Gao Y, Xiao X, Zhang C, et al. Melatonin synergizes the chemotherapeutic effect of 5-fluorouracil in colon cancer by suppressing PI 3K/AKT and NF-κB/iNOS signaling pathways. J Pineal Res 2017; 62: e12380. [DOI] [PubMed] [Google Scholar]
- 53.Bar-Sela G, Epelbaum R, Schaffer M. Curcumin as an anti-cancer agent: review of the gap between basic and clinical applications. Curr Med Chem 2010; 17: 190-197. [DOI] [PubMed] [Google Scholar]
- 54.Dai W, Gao Q, Qiu J, Yuan J, Wu G, Shen G. Quercetin induces apoptosis and enhances 5-FU therapeutic efficacy in hepatocellular carcinoma. Tumor Biol 2016; 37: 6307-6313. [DOI] [PubMed] [Google Scholar]
- 55.Wei Y, Yang P, Cao S, Zhao L. The combination of curcumin and 5-fluorouracil in cancer therapy. Arch Pharm Res 2018; 41: 1-13. [DOI] [PubMed] [Google Scholar]
- 56.Barisciano G, Colangelo T, Rosato V, et al. miR-27a is a master regulator of metabolic reprogramming and chemoresistance in colorectal cancer. Br J Cancer 2020; 122: 1354-1366. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Moradi A, Ghasemi F, Anvari K, et al. The cross-regulation between SOX15 and Wnt signaling pathway. J Cell Physiol 2017; 232: 3221-3225. [DOI] [PubMed] [Google Scholar]
- 58.Li W, Zhao Y, Qiu L, Ma J. Effect of Quercetin on Wnt/β-catenin signal pathway of K562 and K562R cells. Zhongguo Shi Yan Xue Ye Xue Za Zhi 2019; 27: 1409-1415. [DOI] [PubMed] [Google Scholar]
- 59.Chen Z, Huang C, Ma T, et al. Reversal effect of quercetin on multidrug resistance via FZD7/β-catenin pathway in hepatocellular carcinoma cells. Phytomedicine 2018; 43: 37-45. [DOI] [PubMed] [Google Scholar]
- 60.Hou G, Yuan X, Li Y, Hou G, Liu X. Cardamonin, a natural chalcone, reduces 5-fluorouracil resistance of gastric cancer cells through targeting Wnt/β-catenin signal pathway. Invest New Drugs 2020; 38: 329-339. [DOI] [PubMed] [Google Scholar]
- 61.Qiao Q, Jiang Y, Li G. Inhibition of the PI3K/AKT-NF-κB pathway with curcumin enhanced radiation-induced apoptosis in human Burkitt’s lymphoma. J Pharmacol Sci 2013; 121: 247-256. [DOI] [PubMed] [Google Scholar]
- 62.Vadde R, Radhakrishnan S, Eranda H, Kurundu K, Reddivari L, Vanamala JKP. Indian gooseberry (Emblica officinalis Gaertn.) suppresses cell proliferation and induces apoptosis in human colon cancer stem cells independent of p53 status via suppression of c-Myc and cyclin D1.J Functional Foods 2016; 25: 267-278. [Google Scholar]
- 63.Choi JH, Yoon JS, Won JW, Park BB, Lee YY. Chloroquine enhances the chemotherapeutic activity of 5-fluorouracil in a colon cancer cell line via cell cycle alteration. APMIS 2012; 120: 597-604. [DOI] [PubMed] [Google Scholar]