Abstract
Succinate is a tricarboxylic acid (TCA) cycle intermediate normally confined to the mitochondrial matrix. It is a substrate of succinate dehydrogenase (SDH). Mutation of SDH subunits (SDHD and SDHB) in hereditary tumors such as paraganglioma or reduction of SDHB expression in cancer results in matrix succinate accumulation which is transported to cytoplasma and secreted into the extracellular milieu. Excessive cytosolic succinate is known to stabilize hypoxia inducible factor-1α (HIF-1α) by inhibiting prolyl hydroxylase. Recent reports indicate that cancer-secreted succinate enhances cancer cell migration and promotes cancer metastasis by activating succinate receptor-1 (SUCNR-1)-mediated signaling and transcription pathways. Cancer-derived extracellular succinate enhances cancer cell and macrophage migration through SUCNR-1 → PI-3 K → HIF-1α pathway. Extracellular succinate induces tumor angiogenesis through SUCNR-1-mediated ERK1/2 and STAT3 activation resulting in upregulation of vascular endothelial growth factor (VEGF) expression. Succinate increases SUCNR-1 expression in cancer cells which is considered as a target for developing new anti-metastasis drugs. Furthermore, serum succinate which is elevated in cancer patients may be a theranostic biomarker for selecting patients for SUCNR-1 antagonist therapy.
Keywords: Succinate, Succinate receptor-1, G-protein-coupled receptor 91 (GPR91), Cancer metastasis, Hereditary paraganglioma, Succinate dehydrogenase
Introduction
Succinate is a tricarboxylic acid (TCA) cycle intermediate normally confined to mitochondrial matrix where it serves as a substrate for succinate dehydrogenase (SDH) and an electron donor for electron transport chain (ETC) [1]. When cells are under stressful conditions such as hypoxia, hyperglycemia and endotoxemia, TCA cycle is broken resulting in mitochondrial matrix succinate accumulation. Several mechanisms including SDH dysfunction, fumarate overproduction and/or TCA acceleration contribute to increased succinate [2, 3]. Excessive succinate leaks to cytoplasma and is secreted into extracellular space [3–5]. Cytosolic and extracellular succinate accumulation promotes cancer growth by distinct mechanisms. The intracellular succinate being structurally similar to α-keto-glutarate (α-KG) acts as a competitive inhibitor of a large number of α-KG-dependent enzymes (or 2-oxoglutarate-dependent dioxygenases, 2OGDD) which utilize α-KG as a co-substrate to catalyze diverse reactions and carry out important functional roles in protein hydroxylation, histone and DNA demethylation, collagen biosynthesis and energy metabolism [6]. Several of the α-KG-dependent enzymes including prolyl hydroxylase (PHD), TET family of 5’-methylcytosine hydroxylases, DNA and histone demethylases are dysregulated in cancer [6]. Cytosolic succinate accumulation as a result of SDH mutation was reported to inhibit α-KG-dependent histone and DNA demethylases which contributes to tumorigenesis [7]. Of note, cytosolic succinate stabilizes hypoxia inducible factor-1α (HIF-1α) by inhibiting PHD which is required for HIF-1α degradation via the ubiquitin–proteasome system [8, 9]. HIF-1α mediates transcription of genes important in glycolysis, inflammation and angiogenesis [10, 11]. On the other hand, the extracellular succinate exerts potent biological actions by activating a plasma membrane G protein-coupled receptor, i.e., succinate receptor-1 (SUCNR-1, also known as GRP91) [12]. Extracellular succinate has been shown to play an important role in enhancing inflammation, inducing tissue fibrosis and regulating renin-angiotensin and hypertension [13–16].
Succinate accumulation was detected in tumors with germline mutation of SDH and cytosolic succinate was recognized as a signal molecule to stabilize HIF-1α and alter tumor behavior. However, it was unclear whether cancer cells secrete succinate into the extracellular milieu. Nor was it known that extracellular succinate possesses biological activities to influence cancer behavior and progression. Reports from recent studies start to shed light on succinate secretion by cancer cells and the crucial role that extracellular succinate plays in promoting cancer progression especially cancer metastasis. The purpose of this review is to address the underlying mechanism of succinate accumulation in and secretion from cancer cells, actions of succinate on cancer cell migration, invasion and metastasis and the potential value of SUCNR-1 as a target for developing new anti- metastatic therapy.
Succinate accumulation due to SDH deficiency in tumor cells
SDH is pivotal in mitochondrial metabolism and bioenergetics. It possesses catalytic activity to convert succinate to fumarate in TCA cycle and an electron transfer system for electron transport in ETC. It is a mitochondrial protein complex comprising four subunits i.e., SDHA, B, C and D and assembly factors [17, 18]. SDH subunits and assembly factors are encoded by genes in the nuclear genome [19]. Structurally, SDHC and SDHD subunits are embedded in the inner membrane and serve as the anchor for SDHB and SDHA subunits. In addition, the interface of SDHC and SDHD contains heme and ubiquinone binding sites which are essential for electron transfer. SDHB binds directly to SDHC and SDHD while SDHA binds SDHB. SDHA contains FAD binding site and catalytic site where succinate is oxidized to fumarate. Succinate donates electrons to FAD to form FADH2 which serve as electron donors in ETC. SDHB subunit contains three iron-sulfur clusters to facilitate electron transfer to SDHC and SDHD where ubiquinone is reduced to ubiquinol. In addition to being a TCA cycle enzyme, SDH complex functions as complex II in the ETC. Each of the four subunits is essential for SDH catalytic activity and electron transport function. Mutation or reduced expression of individual subunit disrupts SDH integrity and diminishes SDH catalytic activity leading to succinate and reactive oxygen species (ROS) accumulation and impaired oxidative phosphorylation [20, 21].
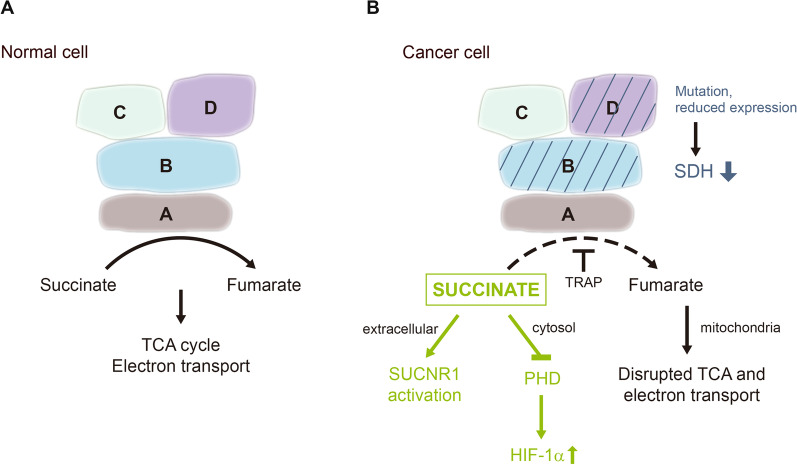
Mammalian cell SDH activity is regulated by extrinsic factors notably TNF receptor associated protein (TRAP). Increased TRAP-1 expression suppresses SDH activity resulting in succinate accumulation. Sirt3, a member of the Sirtuin family is a mitochondrial deacetylase which regulates lysine acetylation of several mitochondrial proteins including SDHA subunit [22]. Sirt3 binds to SDHA and increases SDH electron transfer and catalytic activity. Sirt3 is considered to be a tumor suppressor [23], and modulation of its expression was reported to enhance the effect of sorafenib on hepatocellular carcinoma [24]. Sirt3 suppresses cancer progression and metastasis by controlling acetylation of several mitochondrial proteins including superoxide dismutase II (SOD II) via which it decreases ROS [25]. It remains to be investigated whether Sirt3 regulates cancer cell migration and metastasis through controlling succinate accumulation and/or secretion. Succinate accumulation in tumor cells due to SDH mutation, defective expression and inhibition is summarized in Fig. 1.
Fig. 1.
Succinate dehydrogenase (SDH) in normal and cancer cell. A. Schematic illustration of SDH subunits. It catalyzes conversion of succinate to fumarate in TCA cycle and electron transport in ETC. B Mutation or expression defect of subunit B or D results in reduced SDH activity and accumulation of succinate. TRAP inhibits SDH catalytic activity also resulting in succinate accumulation. Cytosolic succinate stabilizes HIF-1α through inhibition of PHD while extracellular succinate promotes cancer metastasis via SUCNR-1
Succinate accumulation due to SDH mutations in hereditary and sporadic tumors
Germline mutation of SDH was first reported in hereditary paraganglioma [26], and subsequently confirmed in diverse hereditary and sporadic tumors including hereditary and sporadic pheochromocytoma, gastrointestinal stromal tumors and familial renal cell carcinoma [27–30]. Germline mutations of SDHB and SDHD subunits which are commonly detected in hereditary and sporadic tumors result in collapse of SDH catalytic activity and complex II function leading to succinate accumulation and excessive ROS generation [20, 21]. Of note, SDHB mutations are associated with malignant and metastatic tumors such as malignant pheochromocytoma, renal cell carcinoma and neuroendocrine tumors [31, 32].
Succinate accumulation due to reduced SDH expression in cancer cells
In addition to SDH subunit mutations which occur in hereditary tumors and sporadic cancers, loss of SDH function may be the result of loss of heterozygosity [33] or reduction of SDHB or SDHD subunit expression in diverse cancers including colorectal, gastric, hepatocellular, ovarian and clear cell renal cell carcinoma (ccRCC) [34–37]. The mechanism by which SDHB or SDHD expression is reduced in cancer cells is not entirely clear. A recent report on ccRCC suggests that reduced SDHD expression is attributable to degradation of SDHD transcripts mediated by microRNA-210 (miR210) upregulation [34]. miR210 induces SDHD degradation by binding to the 3’-untranslated region of SDHD mRNA. Reduced SDH catalytic activity leads to accumulation of succinate in renal tissues, increased cancer cell invasion and poor prognosis [34]. Reduced SDHB expression in hepatocellular cancer (HCC) is also associated with poor prognosis [35]. Several reports have indicated that SDHB plays an important role in controlling cancer metastasis. Silencing of SDHB in cancer cells with selective siRNA results in phenotypic changes characterized by enhanced migration and invasion, epithelial mesenchymal transition (EMT) and metabolic switch to glycolysis [34, 36, 37]. Aspura et al. reported that knockdown of SDHB results in reduced SDH catalytic activity accompanied by increased cell proliferation, EMT and metabolic switch [36]. Chen et al. reported that overexpression of SDHB in ovarian cancer cells reduces cell proliferation, migration and invasion [37]. By contrast, Cervera et al. reported that silencing of SDHB in HCC and gastric carcinoma cells reduces cell proliferation despite loss of SDH catalytic activity [38]. The reason for the discrepancy has not been resolved. One possible explanation is differences in cell types and experimental conditions. Nevertheless, the reported findings support the notion that reduction of SDH activity in cancer cells has a profound effect on cancer phenotype characterized by increasing cancer cell migration, invasion and cancer metastasis.
Succinate accumulation due to inhibition of SDH activity by TRAP-1
TRAP-1 is a mitochondrial chaperone which forms complex with heat shock protein 90 (HSP90) in the mitochondrial matrix [39, 40]. TRAP-1/HSP90 play an important role in mitochondrial quality control, redox balance, bioenergetics and membrane permeability. TRAP-1 is overexpressed in diverse types of cancers and was reported to accelerate prostate cancer development [41]. One of the mechanisms by which TRAP-1 promotes cancer growth is through binding to SDH thereby inhibiting SDH catalytic activities (Fig. 1) and disrupting electron transfer [42], leading to succinate and ROS accumulation.
Succinate accumulation drives cancer growth and metastasis
Earlier studies recognized that loss-of-function mutation of SDHD in paraganglioma was associated with elevated HIF-1α which led to the proposal that SDH mutation results in activating hypoxia response pathway [20, 43–45]. It was subsequently discovered that HIF-1α elevation is attributed to cytosolic succinate accumulation [6]. Under normoxic conditions, HIF-1α is rapidly degraded by ubiquitin-proteosome system. A pre-requisite for degradation is hydroxylation of HIF-1α prolyl residues catalyzed by PHD [7–9]. Succinate inhibits PHD activity thereby blocking HIF-1α degradation [6] (Fig. 2). HIF-1α is a pleiotropic transcription activator, mediating transcription of tumor promoting genes including pro-metastatic genes [10, 11]. Extracellular succinate augments cancer growth and metastasis through SUCNR-1 mediated signaling pathways and transcription programs. It is of interests to note that cancer cells secrete the glycolytic end product, lactic acid which was shown to be an active extracellular signaling molecule in promoting cancer growth [46, 47]. Wu et al. analyzed metabolites in the conditioned medium (CM) of lung (A549, LLC), prostate (PC3), breast (MCF7), and colon (HT29) cancer cells by liquid chromatography-mass spectrometry (LC–MS). They reported detection of abundant succinate in the CM of all the cancer cells tested [48]. Lactate was also detected but was less abundant. On the other hand, succinate and lactate were undetectable in the CM of unstimulated macrophages [48]. Mu et al. reported detection of succinate in the CM of gastric cancer cells by colorimetry [49]. Cells stimulated by stress signals such as ischemia–reperfusion (I/R) or lipopolysaccharide (LPS) release succinate into the excellular milieu [3, 15] through monocarboxylate transporter-1 (MCT-1) [50, 51]. MCT-1 expression is upregulated in several types of cancer cells which is considered to be involved in tumor growth [52, 53]. Cancer cell may secrete succinate via this membrane transporter. Succinate secreted from LPS-stimulated macrophages and I/R-injured cells amplifies inflammatory responses and induces tissue damage and fibrosis [14, 15, 54–56]. Cancer-derived succinate, on the other hand, promotes cancer metastasis by driving cancer cell migration and invasion, inducing epithelial mesenchymal transition (EMT) and angiogenesis, which will be described below.
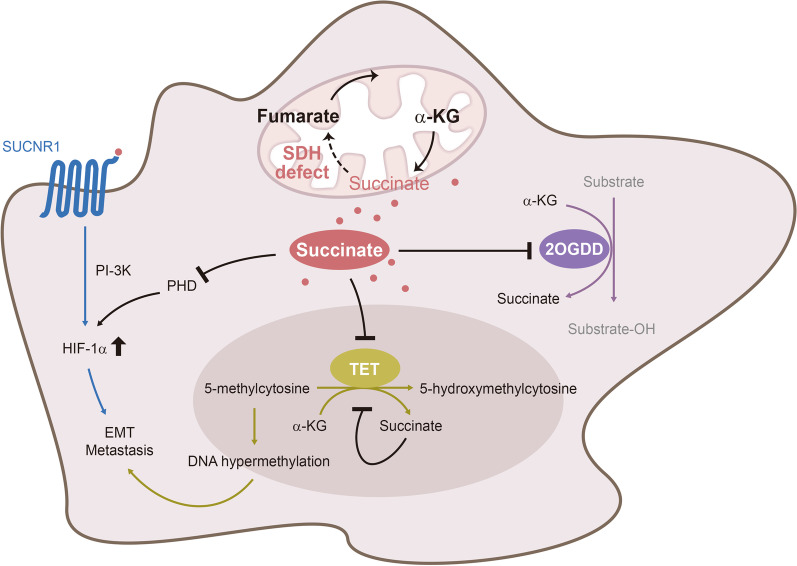
Fig. 2.
Succinate promotes cancer cell growth by inhibiting 2OGDD and through ligating SUCNR-1. Succinate accumulation due to SDH defect leads to increased cytosolic succinate and secretion of succinate. Cytosolic succinate exerts its effect by inhibiting a large number of 2-oxoglutarate-dependent dioxygenases (2OGDD or α-keto glutarate-dependent enzymes) such as prolyl hydroxylase (PHD) and TET (Ten to Eleven Translocation). Inhibition of PHD results in HIF-1α stabilization while inhibition of TET leads to DNA hypermethylation. Extracellular succinate interacts with succinate receptor-1 (SUCNR-1) which signals via PI-3 K to increase HIF-1α expression
Cancer cell-derived succinate enhances cell migration and drives EMT by interaction with SUCNR-1
Succinate released from human lung (A549) or murine lung (LLC) cancer cells enhances cancer cell migration and induces cancer cell EMT [48]. By contrast, cancer-derived succinate does not influence cancer cell viability or proliferation. The specific effect of succinate on cancer cell migration and EMT results in increased cancer metastasis as demonstrated in a syngeneic murine tumor model in which C57BL/6 J mice were implanted with LLC murine lung cancer cells subcutaneously. Administration of succinate in vivo increases cancer metastasis to lungs and other organs, but has no effect on tumor volume [48].
Succinate drives cancer cell migration and enhances cancer metastasis by interaction with SUCNR-1 on cancer cell surface. Cancer cell-derived succinate activates SUCNR-1 resulting in ERK1/2 activation, prostaglandin E2 production and increased intracellular calcium [57, 58]. Furthermore, succinate activates p38 MAPK, Akt and AMPK in lung cancer cells, suggesting that succinate activates multiple signaling pathways via SUCNR-1 interaction. Of note, only PI-3 K/Akt inhibition abrogates succinate-induced cancer cell migration [48]. Interestingly, extracellular succinate induces HIF upregulation via membrane receptor-PI-3 K signaling (Fig. 2). HIF-1α and -2α overexpression promotes cancer cell invasion and drives EMT by a number of biochemical and genetic processes including Twist transcription, Snail nuclear localization and the consequent loss of E-cadherin and increase in mesenchymal markers [59–61]. Experimental results from murine xenograft tumor model support the crucial role of HIF-1α in cancer metastasis. Implantation of HIF-1α silenced A549 cells into nude mice results in reduced lung metastatic nodules accompanied by reversal of E-cadherin and vimentin expression in tumor cells [48].
Succinate accumulation in ccRC with decreased SDHD or SDHB expression was reported to enhance cancer cell invasion and metastasis by increasing DNA 5-methylcytosine (5mC) and suppressing 5-hydroxymethylcytosine (5-hmC) through inhibition of TET-2, resulting in global DNA hypermethylation [34]. TET-2 (Ten to Eleven Translocation-2) is an α-KG-dependent enzyme which requires α-KG as a co-substrate to catalyze conversion of 5mC to 5-hmC (Fig. 2). DNA hypermethylation contributes to cancer invasiveness and EMT by altering binding of transcriptional activators to promoters of genes regulating migration and EMT. For example, HIF-1α binding to the target genes is influenced by DNA methylation [62]. The reported findings suggest that succinate acts in an autocrine and paracrine manners to drive cancer metastasis via the SUCNR-1 → PI-3K → Akt → HIF-1α signaling pathway (Fig. 3).
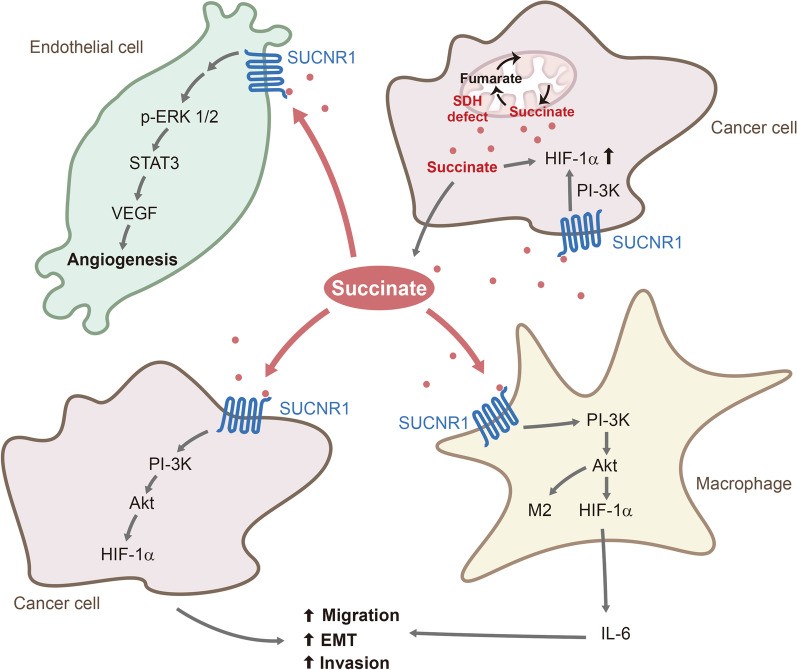
Fig. 3.
Cancer cell-derived succinate acts on macrophages, endothelial cells and cancer cells to drive cancer metastasis
Succinate activates endothelial cells and induces angiogenesis via SUCNR-1 → ERK → VEGF pathway
Cancer metastasis is further enhanced by succinate-mediated tumor angiogenesis which is essential for tumor growth and metastasis [63, 64]. For example, gastric cancer-derived extracellular succinate induces endothelial cell proliferation and angiogenesis by interacting with SUCNR-1 which transmits signals for angiogenesis via STAT3 and ERK1/2 resulting in upregulation of vascular endothelial growth factor (VEGF) [49] (Fig. 3). Tumor angiogenesis induced by gastric cancer-derived succinate is independent of HIF-1α [49]. On the other hand, extracellular succinate induces retinal angiogenesis in retinopathy and synovial angiogenesis in arthritis through signaling and transcriptional pathways depending on HIF-1α [65]. HIF-1α has been incriminated as an important factor for inducing pathological angiogenesis [65, 66]. As cellular and molecular factors participating in cancer metastasis are many and complex, current knowledge on the mechanisms by which succinate promotes cancer metastasis is incomplete. Further investigations are required.
Extracellular succinate promotes macrophage migration and M2 polarization via SUNCR-1 → PI-3 K/Akt → HIF-1α signaling pathway
Cancer cell-derived extracellular succinate not only drives cancer cell migration but also exerts great influence on stromal cells such as macrophages and endothelial cells in the tumor microenvironment (Fig. 3). Succinate was reported to polarize macrophages and induce endothelial cell tube formation by distinct signaling pathways. It converts naïve macrophages into tumor associated macrophages (TAM) through a receptor-mediated signaling pathway similar to that for driving cancer cell migration and EMT [48]. It induces TAM polarization by activating macrophage membrane SUCNR-1 receptor and the downstream PI-3 K/Akt → HIF-1α signaling pathway [48]. Furthermore, it drives macrophage migration via the SUCNR-1 signaling pathway. Succinate-induced macrophage polarization augments cancer cell migration by secreting pro-migratory cytokines such as interleukin-6 (IL-6) (Fig. 3).
Extracellular succinate also targets M2 macrophages and activate M2 macrophage gene transcription via SUCNR1 to create a hyperpolarized M2 macrophage environment [67]. SUCNR-1 is coupled with Gi and/or Gq proteins depending on cell types [68]. Succinate induces hyperpolarized M2 macrophages via SUCNR-1 → Gq pathway [67]. In addition, inhibition of SDH was reported to impair T cell activation and function [69]. It is likely that cancer-derived extracellular succinate is a major player in the tumor microenvironment for immunosuppression and cancer cell immune evasion.
It is to be noted that inflammatory stimuli activate macrophages and induce macrophage secretion of succinate. Macrophage-derived excellular succinate aggravates inflammatory responses and exacerbates inflammatory disorders such as rheumatoid arthritis [15]. Macrophage migration in response to extracellular succinate is crucial for mediating inflammation in adipose tissues and aggravating obesity-induced diabetes [56]. By contrast, macrophage-derived extracellular succinate may activate SUCNR-1 on neural stem cells leading them to release anti-inflammatory factors [70]. Thus, extracellular succinate regulates inflammation in a complex manner depending on cell types and contexts. It remains to be investigated to what extent succinate in tumor microenvironment contributes to inflammation and inflammation-mediated cancer metastasis.
Extracellular succinate promotes cell migration through induction of mitochondrial fission
Normal cells undergo constant mitochondrial fusion and fission to maintain mitochondrial dynamic balance which is vital for mitochondrial metabolism and function [71, 72]. Cancer cells exhibit an imbalanced mitochondrial dynamics with excessive fission and fragmented mitochondria [73, 74]. Several reports reveal that mitochondrial fission promotes cancer cell migration and invasion and contributes to metastasis of diverse cancers including breast, hepatocellular and thyroid cancer [75–78]. Mitochondrial fission and fragmentation in cancer cells is mediated by increased fission proteins including Drp-1, Fis-1 and/or MID49 and decreased fusion protein Mfn-1 [79]. Although extracellular succinate has not been directly linked to induction of mitochondrial fragmentation in cancer cells, a report on human mesenchymal stromal cells suggests that succinate promotes cell migration by a SUCNR-1-mediated Drp-1 phosphorylation [80]. Succinate activates SUCNR-1 coupled Gq and its downstream atypical protein kinase C, PKCδ, which in turn phosphorylates p38 MAPK (Fig. 4). p38 MAPK phosphorylates Drp-1 and induces Drp-1 translocation to mitochondrial outer membrane where it interacts with receptors such as Fis-1 and initiates mitochondrial division. Of note, it was reported that succinate induces mitochondrial fission in cardiomyocytes via SUCNR-1 → PKCδ and ERK1/2 pathway [81].
Fig. 4.
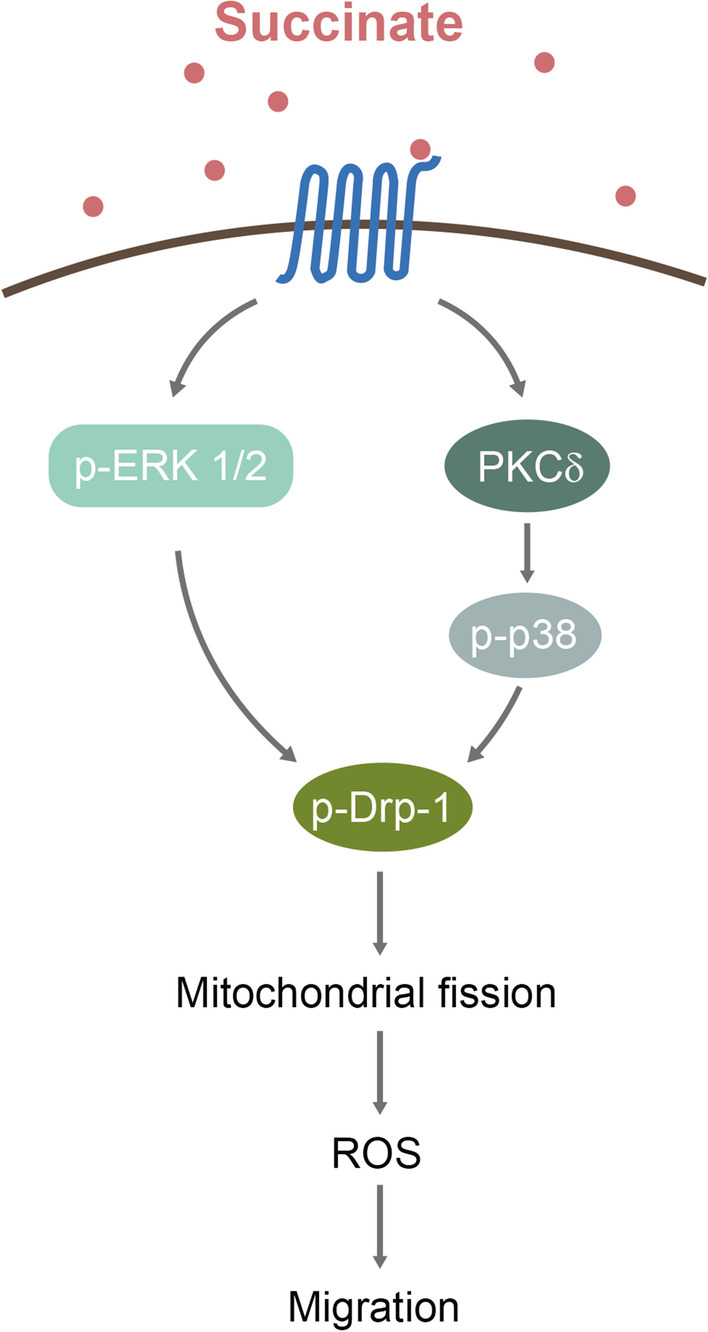
Schematic illustration of the signaling pathway via which succinate phosphorylate Drp-1 and induces mitochondrial fission. ROS generated drive cancer cell migration
Mitochondrial fragmentation is associated with metabolic reprogramming resulting in enhanced glycolysis, and reduced oxidative phosphorylation [72]. It triggers ROS generation and membrane depolarization [82–84]. ROS play an important role in promoting cancer cell migration and invasion [85]. Furthermore, ROS drive EMT and matrix degradation [86–88]. ROS induce mitochondrial fragmentation, creating a vicious cycle [83]. Mitochondrial fission/ROS vicious cycle may be crucial in perpetuating succinate-mediated cancer cell migration and cancer metastasis [88] (Fig. 4).
Succinate receptor is a potential target for controlling cancer metastasis
He et al. made a breakthrough discovery when they reported that succinate is a physiological ligand of an orphan G-protein coupled receptor 91 (GPR91) and linked the receptor activation to renin-angiotensin system and hypertension [12]. Toma et al. confirmed that GRP91 is succinate receptor and noted that high glucose induces extracellular succinate accumulation in kidneys which mediates renin release by activating GRP91 signaling cascade [89]. GRP91 was subsequently renamed SUCNR-1. SUCNR-1 shares with purinergic receptor structural characteristics and was initially thought to be a receptor for purinergic ligands [90, 91]. SCUNR-1 is coupled to Gi and/or Gq depending on cell types [53, 88]. A canonical downstream signal of SUCNR-1 activation is ERK1/2 activation and Ca2+ mobilization [92–94]. Succinate-ligated SUCNR-1 may transmit signals via non-canonical signaling pathways such as PI-3 K [48]. SUCNR-1 signaling is complex and variable depending on stimulation context, succinate concentration and cell types. How are G-proteins and its downstream signaling pathways determined and regulated is poorly understood and requires further investigation. Nevertheless, numerous reports provide evidence that succinate- activated SUCNR-1 mediates diverse pathophysiological conditions [95–99]. A high level of SUCNR-1 is expressed on cancer cells and its silencing by shRNA (short hairpin RNA) results in reducing cancer cell migration and invasion [48]. Furthermore, knockdown of SUCNR-1 in human gastric or pancreatic cancer cells restores mitochondrial function [100]. Subcutaneous implantation of lung cancer cells transfected with SUCNR-1 shRNA into a murine xenograft tumor model results in local tumor growth comparable to that from implantation of lung cancer cells transfected with control vector. However, metastatic lung nodules are significantly reduced in the SCUNR-1 shRNA group [48]. These results suggest that succinate-SUCNR-1 signaling selectively promotes cancer cell migration and cancer metastasis.
SUCNR-1 expression is increased in human SDH-mutated tumors and several common cancers, which is associated with a high risk of metastasis and a high risk of recurrence following surgery [101, 102]. It has been reported that cancer cell SUCNR-1 expression is upregulated by extracellular succinate or SDH subunit silencing, indicative of a feedback regulation [48]. SUCNR-1 was thus considered to be a target for treating SDH-mutated paraganglioma [102]. Small molecule SUCNR-1 inhibitors have been chemically synthesized [67, 103] but drug development has been slow due to concerns of adverse effects as succinate-SUCNR-1 was reported to play important physiological roles such as thermogenesis [104] and skeletal muscle adaptation to exercise [105]. Additional investigations are needed to elucidate the mechanism by which SUCNR-1 is upregulated in cancer cells and to evaluate the effects of suppressing SUCNR-1 upregulation on cancer metastasis.
Elevated serum succinate is a potential biomarker of cancer progression
Hobert et al. were the first to detect an association between SDH mutation and elevation of serum succinate [106]. However, due to small number of patients, the reported association was uncertain and requires confirmation. Wu et al. provided experimental data to show in a syngeneic murine model that tumor growth was associated with elevation of serum succinate [48]. They further reported that serum succinate level is increased in patients with non-small cell lung carcinoma and that serum succinate has the potential to be a biomarker of lung cancer [48]. A recent report reveals that serum succinate is a biomarker of human head and neck squamous carcinoma [107]. Taken together, these findings suggest that cancer cell secrete succinate into circulating blood to raise the blood level of succinate. The biological implications of elevated serum succinate on endothelial dysfunction and cancer cell extravasation are unclear and require further investigation. Preliminary results from animal experiments suggest that elevated blood succinate promotes cancer metastasis, which may serve as a biomarker of cancer progression. It will be important to determine whether serum succinate is a theranostic biomarker for selecting patients for anti-SUCNR-1 therapy.
Conclusion
Succinate is normally located in mitochondrial matrix serving as an intermediate metabolite of TCA cycle. Cancer cells often present with reduced expression of SDH subunits notably SDHB or SDHD resulting in succinate accumulation. Succinate accumulation may also be due to SDH inhibition by TRAP-1 which is often overexpressed in cancer. Succinate accumulated in matrix may leak to the cytosol, where it promotes cancer growth by stabilizing HIF-1α. Excessive succinate in cytoplasm is secreted into the extracellular milieu where it promotes cancer cell migration and cancer metastasis. Extracellular succinate acts in an autocrine and paracrine manner to enhance cancer cell migration by activating a specific G-protein coupled receptor, SUCNR-1 which signals via ERK1/2 and PI-3 K/Akt. Extracellular succinate may also enhance macrophage, endothelial cell and mesenchymal stromal cell migration by activating SUCNR-1 mediated signaling pathways. Cancer cell-derived succinate induces EMT, tumor angiogenesis and matrix metaloprotinase expression via SUCNR-1-mediated signaling and transcriptional pathways as well as epigenetic modification such as hypermethylation. Findings from murine xenograft tumor models support a critical role for succinate/SUCNR-1 in driving cancer metastasis. It is of interests that extracellular succinate induces excessive mitochondrial fission and fragmentation in cells under stresses such as ischemia–reperfusion and lipopolysaccharide via SUCNR-1. Given that mitochondrial fragmentation in cancer cells is associated with increased migration and metastasis, it is possible that succinate/SUCNR-1 → mitochondrial fragmentation pathway may provide additional force to drive cancer metastasis.
Extracellular succinate mediates diverse actions via SUCNR-1. SUCNR-1 is a reasonable target for controlling metastasis. However, targeting SUCNR-1 may be confronted by perturbation of normal physiological functions mediated by SUCNR-1. Further studies are needed to develop therapeutic agents based on differential inhibition of SUCNR-1 on cancer metastasis vs. normal cellular physiological functions.
Cancer cell-secreted succinate contributes to elevation of blood succinate levels. Serum succinate was reported to be a potential biomarker of lung cancer. It is important to determine whether serum succinate is a theranostic biomarker for selecting cancer patients for anti-SUCNR-1 drug therapy.
Acknowledgements
Not applicable.
Abbreviations
- TCA
Tricarboxylic acid
- SDH
Succinate dehydrogenase
- ETC
Electron transport chain
- α-KG
α-Keto-glutarate
- 2OGDD
2-Oxoglutarate-dependent dioxygenases
- PHD
Prolyl hydroxylase
- HIF-1α
Hypoxia inducible factor-1α
- SUCNR-1
Succinate receptor-1
- ROS
Reactive oxygen species
- TRAP
TNF receptor associated protein
- SOD II
Superoxide dismutase II
- ccRCC
Clear cell renal cell carcinoma
- miR210
MicroRNA-210
- HCC
Hepatocellular cancer
- EMT
Epithelial mesenchymal transition
- HSP90
Heat shock protein 90
- CM
Conditioned medium
- LC–MS
Liquid chromatography-mass spectrometry
- I/R
Ischemia–reperfusion
- LPS
Lipopolysaccharide
- MCT-1
Monocarboxylate transporters-1
- EMT
Epithelial mesenchymal transition
- 5mC
5-Methylcytosine
- 5-hmC
5-Hydroxymethylcytosine
- VEGF
Vascular endothelial growth factor
- TAM
Tumor associated macrophages
- IL-6
Interleukin-6
- GPR91
G-protein coupled receptor 91
- Drp-1
Dynamin-related protein-1
Author contributions
KKW wrote the paper and CCK edited and reviewed the paper. JYW contributed to experimental results and analysis. All authors read and approved the final manuscript.
Authors’ information
Dr. CC Kuo is a full Investigator (Professor-equivalent) and Dr. KK Wu is an Emeritus Distinguished Investigator and Past President of NHRI; Dr. JY Wu is a Post-doctoral Fellow at NHRI.
Funding
This work was supported by National Health Research Institutes (Taiwan) intramural Grants and Ministry of Science and Technology Excellent Team Research Program Grant (MOST 108-2321-B-400-009, 109-2326-B-400-002 and 110-2326-B-400-005).
Availability of data and materials
Not applicable.
Declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
NoT applicable.
Competing interests
The authors declare no competing interests.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Contributor Information
Cheng-Chin Kuo, Email: kuocc@nhri.org.tw.
Kenneth K. Wu, Email: kkgo@nhri.org.tw
References
- 1.Martínez-Reyes I, Chandel NS. Mitochondrial TCA cycle metabolites control physiology and disease. Nat Commun. 2020;11:102. doi: 10.1038/s41467-019-13668-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Chouchani ET, Pell VR, Gaude E, Aksentijević D, Sundier SY, Robb EL, et al. Ischaemic accumulation of succinate controls reperfusion injury through mitochondrial ROS. Nature. 2014;515:431–435. doi: 10.1038/nature13909. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Zhang J, Wang YT, Miller JH, Day MM, Munger JC, Brookes PS. Accumulation of succinate in cardiac ischemia primarily occurs via canonical Krebs cycle activity. Cell Rep. 2018;23:2617–2628. doi: 10.1016/j.celrep.2018.04.104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Kohlhauer M, Dawkins S, Costa ASH, Lee R, Young T, Pell VR, et al. Metabolomic profiling in acute ST-segment-elevation myocardial infarction identifies succinate as an early marker of human ischemia-reperfusion injury. J Am Heart Assoc. 2018;7:e007546. doi: 10.1161/JAHA.117.007546. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Nair S, Sobotka KS, Joshi P, Gressens P, Fleiss B, Thornton C, et al. Lipopolysaccharide-induced alteration of mitochondrial morphology induces a metabolic shift in microglia modulating the inflammatory response in vitro and in vivo. Glia. 2019;67:1047–1061. doi: 10.1002/glia.23587. [DOI] [PubMed] [Google Scholar]
- 6.Losman JA, Koivunen P, Kaelin WG., Jr 2-Oxoglutarate-dependent dioxygenases in cancer. Nat Rev Cancer. 2020;20:710–726. doi: 10.1038/s41568-020-00303-3. [DOI] [PubMed] [Google Scholar]
- 7.Smith EH, Janknecht R, Maher LJ., 3rd Succinate inhibition of alpha-ketoglutarate-dependent enzymes in a yeast model of paraganglioma. Hum Mol Genet. 2007;16:3136–3148. doi: 10.1093/hmg/ddm275. [DOI] [PubMed] [Google Scholar]
- 8.Selak MA, Armour SM, MacKenzie ED, Boulahbel H, Watson DG, Mansfield KD, et al. Succinate links TCA cycle dysfunction to oncogenesis by inhibiting HIF-alpha prolyl hydroxylase. Cancer Cell. 2005;7:77–85. doi: 10.1016/j.ccr.2004.11.022. [DOI] [PubMed] [Google Scholar]
- 9.Jaakkola P, Mole DR, Tian YM, Wilson MI, Gielbert J, Gaskell SJ, et al. Targeting of HIF-alpha to the von Hippel-Lindau ubiquitylation complex by O2-regulated prolyl hydroxylation. Science. 2001;292:468–472. doi: 10.1126/science.1059796. [DOI] [PubMed] [Google Scholar]
- 10.Semenza GL. HIF-1: upstream and downstream of cancer metabolism. Curr Opin Genet Dev. 2010;20:51–56. doi: 10.1016/j.gde.2009.10.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Schito L, Semenza GL. Hypoxia-inducible factors: master regulators of cancer progression. Trends Cancer. 2016;2:758–770. doi: 10.1016/j.trecan.2016.10.016. [DOI] [PubMed] [Google Scholar]
- 12.He W, Miao FJ, Lin DC, Schwandner RT, Wang Z, Gao J, et al. Citric acid cycle intermediates as ligands for orphan G-protein-coupled receptors. Nature. 2004;429:188–193. doi: 10.1038/nature02488. [DOI] [PubMed] [Google Scholar]
- 13.Wu B, Luo H, Zhou X, Cheng CY, Lin L, Liu BL, et al. Succinate-induced neuronal mitochondrial fission and hexokinase II malfunction in ischemic stroke: therapeutical effects of kaempferol. Biochim Biophys Acta Mol Basis Dis. 2017;1863:2307–2318. doi: 10.1016/j.bbadis.2017.06.011. [DOI] [PubMed] [Google Scholar]
- 14.Correa PR, Kruglov EA, Thompson M, Leite MF, Dranoff JA, Nathanson MH. Succinate is a paracrine signal for liver damage. J Hepatol. 2007;47:262–269. doi: 10.1016/j.jhep.2007.03.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Littlewood-Evans A, Sarret S, Apfel V, Loesle P, Dawson J, Zhang J, et al. GPR91 senses extracellular succinate released from inflammatory macrophages and exacerbates rheumatoid arthritis. J Exp Med. 2016;213:1655–1662. doi: 10.1084/jem.20160061. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Aguiar CJ, Rocha-Franco JA, Sousa PA, Santos AK, Ladeira M, Rocha-Resende C, et al. Succinate causes pathological cardiomyocyte hypertrophy through GPR91 activation. Cell Commun Signal. 2014;12:78. doi: 10.1186/s12964-014-0078-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Rustin P, Munnich A, Rötig A. Succinate dehydrogenase and human diseases: new insights into a well-known enzyme. Eur J Hum Genet. 2002;10:289–291. doi: 10.1038/sj.ejhg.5200793. [DOI] [PubMed] [Google Scholar]
- 18.Yankovskaya V, Horsefield R, Törnroth S, Luna-Chavez C, Miyoshi H, Léger C, et al. Architecture of succinate dehydrogenase and reactive oxygen species generation. Science. 2003;299:700–704. doi: 10.1126/science.1079605. [DOI] [PubMed] [Google Scholar]
- 19.Rutter J, Winge DR, Schiffman JD. Succinate dehydrogenase—assembly, regulation and role in human disease. Mitochondrion. 2010;10:393–401. doi: 10.1016/j.mito.2010.03.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Gimenez-Roqueplo AP, Favier J, Rustin P, Mourad JJ, Plouin PF, Corvol P, et al. The R22X mutation of the SDHD gene in hereditary paraganglioma abolishes the enzymatic activity of complex II in the mitochondrial respiratory chain and activates the hypoxia pathway. Am J Hum Genet. 2001;69:1186–1197. doi: 10.1086/324413. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Gimenez-Roqueplo AP, Favier J, Rustin P, Rieubland C, Kerlan V, Plouin PF, et al. Functional consequences of a SDHB gene mutation in an apparently sporadic pheochromocytoma. J Clin Endocrinol Metab. 2002;87:4771–4774. doi: 10.1210/jc.2002-020525. [DOI] [PubMed] [Google Scholar]
- 22.Finley LW, Haas W, Desquiret-Dumas V, Wallace DC, Procaccio V, Gygi SP, et al. Succinate dehydrogenase is a direct target of sirtuin 3 deacetylase activity. PLoS ONE. 2011;6:e23295. doi: 10.1371/journal.pone.0023295. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Haigis MC, Deng CX, Finley LW, Kim HS, Gius D. SIRT3 is a mitochondrial tumor suppressor: a scientific tale that connects aberrant cellular ROS, the Warburg effect, and carcinogenesis. Cancer Res. 2012;72:2468–2472. doi: 10.1158/0008-5472.CAN-11-3633. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Jo H, Park Y, Kim T, Kim J, Lee JS, Kim SY, et al. Modulation of SIRT3 expression through CDK4/6 enhances the anti-cancer effect of sorafenib in hepatocellular carcinoma cells. BMC Cancer. 2020;20:332. doi: 10.1186/s12885-020-06822-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Alhazzazi TY, Kamarajan P, Verdin E, Kapila YL. Sirtuin-3 (SIRT3) and the Hallmarks of cancer. Genes Cancer. 2013;4:164–171. doi: 10.1177/1947601913486351. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Baysal BE, Ferrell RE, Willett-Brozick JE, Lawrence EC, Myssiorek D, Bosch A, et al. Mutations in SDHD, a mitochondrial complex II gene, in hereditary paraganglioma. Science. 2000;287:848–851. doi: 10.1126/science.287.5454.848. [DOI] [PubMed] [Google Scholar]
- 27.Ricketts C, Woodward ER, Killick P, Morris MR, Astuti D, Latif F, et al. Germline SDHB mutations and familial renal cell carcinoma. J Natl Cancer Inst. 2008;100:1260–1262. doi: 10.1093/jnci/djn254. [DOI] [PubMed] [Google Scholar]
- 28.Janeway KA, Kim SY, Lodish M, Nosé V, Rustin P, Gaal J, et al. Defects in succinate dehydrogenase in gastrointestinal stromal tumors lacking KIT and PDGFRA mutations. Proc Natl Acad Sci U S A. 2011;108:314–318. doi: 10.1073/pnas.1009199108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.King A, Selak MA, Gottlieb E. Succinate dehydrogenase and fumarate hydratase: linking mitochondrial dysfunction and cancer. Oncogene. 2006;25:4675–4682. doi: 10.1038/sj.onc.1209594. [DOI] [PubMed] [Google Scholar]
- 30.Bayley JP, van Minderhout I, Weiss MM, Jansen JC, Oomen PH, Menko FH, et al. Mutation analysis of SDHB and SDHC: novel germline mutations in sporadic head and neck paraganglioma and familial paraganglioma and/or pheochromocytoma. BMC Med Genet. 2006;7:1. doi: 10.1186/1471-2350-7-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Milione M, Pusceddu S, Gasparini P, Melotti F, Maisonneuve P, Mazzaferro V, et al. Succinate dehydrogenase B subunit immunohistochemical expression predicts aggressiveness in well differentiated neuroendocrine tumors of the ileum. Cancers (Basel) 2012;4:808–820. doi: 10.3390/cancers4030808. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Rapizzi E, Ercolino T, Fucci R, Zampetti B, Felici R, Guasti D, et al. Succinate dehydrogenase subunit B mutations modify human neuroblastoma cell metabolism and proliferation. Horm Cancer. 2014;5:174–184. doi: 10.1007/s12672-014-0172-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Habano W, Sugai T, Nakamura S, Uesugi N, Higuchi T, Terashima M, et al. Reduced expression and loss of heterozygosity of the SDHD gene in colorectal and gastric cancer. Oncol Rep. 2003;10:1375–1380. [PubMed] [Google Scholar]
- 34.Aggarwal RK, Luchtel RA, Machha V, Tischer A, Zou Y, Pradhan K, et al. Functional succinate dehydrogenase deficiency is a common adverse feature of clear cell renal cancer. Proc Natl Acad Sci U S A. 2021;118:e2106947118. doi: 10.1073/pnas.2106947118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Tseng PL, Wu WH, Hu TH, Chen CW, Cheng HC, Li CF, et al. Decreased succinate dehydrogenase B in human hepatocellular carcinoma accelerates tumor malignancy by inducing the Warburg effect. Sci Rep. 2018;8:3081. doi: 10.1038/s41598-018-21361-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Aspuria PP, Lunt SY, Väremo L, Vergnes L, Gozo M, Beach JA, et al. Succinate dehydrogenase inhibition leads to epithelial-mesenchymal transition and reprogrammed carbon metabolism. Cancer Metab. 2014;2:21. doi: 10.1186/2049-3002-2-21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Chen L, Liu T, Zhang S, Zhou J, Wang Y, Di W. Succinate dehydrogenase subunit B inhibits the AMPK-HIF-1α pathway in human ovarian cancer in vitro. J Ovarian Res. 2014;7:115. doi: 10.1186/s13048-014-0115-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Cervera AM, Apostolova N, Crespo FL, Mata M, McCreath KJ. Cells silenced for SDHB expression display characteristic features of the tumor phenotype. Cancer Res. 2008;68:4058–4067. doi: 10.1158/0008-5472.CAN-07-5580. [DOI] [PubMed] [Google Scholar]
- 39.Kang BH, Altieri DC. Compartmentalized cancer drug discovery targeting mitochondrial Hsp90 chaperones. Oncogene. 2009;28:3681–3688. doi: 10.1038/onc.2009.227. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Kang BH, Plescia J, Dohi T, Rosa J, Doxsey SJ, Altieri DC. Regulation of tumor cell mitochondrial homeostasis by an organelle-specific Hsp90 chaperone network. Cell. 2007;131:257–270. doi: 10.1016/j.cell.2007.08.028. [DOI] [PubMed] [Google Scholar]
- 41.Lisanti S, Garlick DS, Bryant KG, Tavecchio M, Mills GB, Lu Y, et al. Transgenic expression of the mitochondrial chaperone TNFR-associated protein 1 (TRAP1) accelerates prostate cancer development. J Biol Chem. 2016;291:25247–25254. doi: 10.1074/jbc.M116.745950. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Sciacovelli M, Guzzo G, Morello V, Frezza C, Zheng L, Nannini N, et al. The mitochondrial chaperone TRAP1 promotes neoplastic growth by inhibiting succinate dehydrogenase. Cell Metab. 2013;17:988–999. doi: 10.1016/j.cmet.2013.04.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Pollard PJ, Brière JJ, Alam NA, Barwell J, Barclay E, Wortham NC, et al. Accumulation of Krebs cycle intermediates and over-expression of HIF1alpha in tumours which result from germline FH and SDH mutations. Hum Mol Genet. 2005;14:2231–2239. doi: 10.1093/hmg/ddi227. [DOI] [PubMed] [Google Scholar]
- 44.Baysal BE. On the association of succinate dehydrogenase mutations with hereditary paraganglioma. Trends Endocrinol Metab. 2003;14:453–459. doi: 10.1016/j.tem.2003.08.004. [DOI] [PubMed] [Google Scholar]
- 45.Pollard PJ, Wortham NC, Tomlinson IP. The TCA cycle and tumorigenesis: the examples of fumarate hydratase and succinate dehydrogenase. Ann Med. 2003;35:632–639. doi: 10.1080/07853890310018458. [DOI] [PubMed] [Google Scholar]
- 46.Colegio OR, Chu NQ, Szabo AL, Chu T, Rhebergen AM, Jairam V, et al. Functional polarization of tumour-associated macrophages by tumour-derived lactic acid. Nature. 2014;513:559–563. doi: 10.1038/nature13490. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Faubert B, Li KY, Cai L, Hensley CT, Kim J, Zacharias LG, et al. Lactate metabolism in human lung tumors. Cell. 2017;171:358–371.e9. doi: 10.1016/j.cell.2017.09.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Wu JY, Huang TW, Hsieh YT, Wang YF, Yen CC, Lee GL, et al. Cancer-derived succinate promotes macrophage polarization and cancer metastasis via succinate receptor. Mol Cell. 2020;77:213–227.e5. doi: 10.1016/j.molcel.2019.10.023. [DOI] [PubMed] [Google Scholar]
- 49.Mu X, Zhao T, Xu C, Shi W, Geng B, Shen J, et al. Oncometabolite succinate promotes angiogenesis by upregulating VEGF expression through GPR91-mediated STAT3 and ERK activation. Oncotarget. 2017;8:13174–13185. doi: 10.18632/oncotarget.14485. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Prag HA, Gruszczyk AV, Huang MM, Beach TE, Young T, Tronci L, et al. Mechanism of succinate efflux upon reperfusion of the ischaemic heart. Cardiovasc Res. 2021;117:1188–1201. doi: 10.1093/cvr/cvaa148. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Payen VL, Mina E, Van Hée VF, Porporato PE, Sonveaux P. Monocarboxylate transporters in cancer. Mol Metab. 2020;33:48–66. doi: 10.1016/j.molmet.2019.07.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Pinheiro C, Longatto-Filho A, Azevedo-Silva J, Casal M, Schmitt FC, Baltazar F. Role of monocarboxylate transporters in human cancers: state of the art. J Bioenerg Biomembr. 2012;44:127–139. doi: 10.1007/s10863-012-9428-1. [DOI] [PubMed] [Google Scholar]
- 53.Halestrap AP, Wilson MC. The monocarboxylate transporter family–role and regulation. IUBMB Life. 2012;64:109–119. doi: 10.1002/iub.572. [DOI] [PubMed] [Google Scholar]
- 54.Cho EH. Succinate as a regulator of hepatic stellate cells in liver fibrosis. Front Endocrinol (Lausanne) 2018;9:455. doi: 10.3389/fendo.2018.00455. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Liu XJ, Xie L, Du K, Liu C, Zhang NP, Gu CJ, Wang Y, et al. Succinate-GPR-91 receptor signalling is responsible for nonalcoholic steatohepatitis-associated fibrosis: effects of DHA supplementation. Liver Int. 2020;40:830–843. doi: 10.1111/liv.14370. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.van Diepen JA, Robben JH, Hooiveld GJ, Carmone C, Alsady M, Boutens L, et al. SUCNR1-mediated chemotaxis of macrophages aggravates obesity-induced inflammation and diabetes. Diabetologia. 2017;60:1304–1313. doi: 10.1007/s00125-017-4261-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Gilissen J, Jouret F, Pirotte B, Hanson J. Insight into SUCNR1 (GPR91) structure and function. Pharmacol Ther. 2016;159:56–65. doi: 10.1016/j.pharmthera.2016.01.008. [DOI] [PubMed] [Google Scholar]
- 58.de Castro FM, Aguiar CJ, da Rocha Franco JA, Gingold RN, Leite MF. GPR91: expanding the frontiers of Krebs cycle intermediates. Cell Commun Signal. 2016;14:3. doi: 10.1186/s12964-016-0126-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Kaelin WG., Jr The von Hippel-Lindau tumour suppressor protein: O2 sensing and cancer. Nat Rev Cancer. 2008;8:865–873. doi: 10.1038/nrc2502. [DOI] [PubMed] [Google Scholar]
- 60.Yang MH, Wu MZ, Chiou SH, Chen PM, Chang SY, Liu CJ, et al. Direct regulation of TWIST by HIF-1alpha promotes metastasis. Nat Cell Biol. 2008;10:295–305. doi: 10.1038/ncb1691. [DOI] [PubMed] [Google Scholar]
- 61.Mak P, Leav I, Pursell B, Bae D, Yang X, Taglienti CA, et al. ERbeta impedes prostate cancer EMT by destabilizing HIF-1alpha and inhibiting VEGF-mediated snail nuclear localization: implications for Gleason grading. Cancer Cell. 2010;17:319–332. doi: 10.1016/j.ccr.2010.02.030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.D'Anna F, Van Dyck L, Xiong J, Zhao H, Berrens RV, Qian J, et al. DNA methylation repels binding of hypoxia-inducible transcription factors to maintain tumor immunotolerance. Genome Biol. 2020;21:182. doi: 10.1186/s13059-020-02087-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Bielenberg DR, Zetter BR. The contribution of angiogenesis to the process of metastasis. Cancer J. 2015;21:267–273. doi: 10.1097/PPO.0000000000000138. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Rouhi P, Lee SL, Cao Z, Hedlund EM, Jensen LD, Cao Y. Pathological angiogenesis facilitates tumor cell dissemination and metastasis. Cell Cycle. 2010;9:913–917. doi: 10.4161/cc.9.5.10853. [DOI] [PubMed] [Google Scholar]
- 65.Li T, Hu J, Gao F, Du X, Chen Y, Wu Q. Transcription factors regulate GPR91-mediated expression of VEGF in hypoxia-induced retinopathy. Sci Rep. 2017;7:45807. doi: 10.1038/srep45807. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Li Y, Liu Y, Wang C, Xia WR, Zheng JY, Yang J, et al. Succinate induces synovial angiogenesis in rheumatoid arthritis through metabolic remodeling and HIF-1α/VEGF axis. Free Radic Biol Med. 2018;126:1–14. doi: 10.1016/j.freeradbiomed.2018.07.009. [DOI] [PubMed] [Google Scholar]
- 67.Trauelsen M, Hiron TK, Lin D, Petersen JE, Breton B, Husted AS, et al. Extracellular succinate hyperpolarizes M2 macrophages through SUCNR1/GPR91-mediated Gq signaling. Cell Rep. 2021;35:109246. doi: 10.1016/j.celrep.2021.109246. [DOI] [PubMed] [Google Scholar]
- 68.Ariza AC, Deen PM, Robben JH. The succinate receptor as a novel therapeutic target for oxidative and metabolic stress-related conditions. Front Endocrinol (Lausanne) 2012;3:22. doi: 10.3389/fendo.2012.00022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Nastasi C, Willerlev-Olsen A, Dalhoff K, Ford SL, Gadsbøll AØ, Buus TB, et al. Inhibition of succinate dehydrogenase activity impairs human T cell activation and function. Sci Rep. 2021;11:1458. doi: 10.1038/s41598-020-80933-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Peruzzotti-Jametti L, Bernstock JD, Vicario N, Costa ASH, Kwok CK, Leonardi T, et al. Macrophage-derived extracellular succinate licenses neural stem cells to suppress chronic neuroinflammation. Cell Stem Cell. 2018;22:355–368.e13. doi: 10.1016/j.stem.2018.01.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Kraus F, Roy K, Pucadyil TJ, Ryan MT. Function and regulation of the divisome for mitochondrial fission. Nature. 2021;590:57–66. doi: 10.1038/s41586-021-03214-x. [DOI] [PubMed] [Google Scholar]
- 72.Liesa M, Shirihai OS. Mitochondrial dynamics in the regulation of nutrient utilization and energy expenditure. Cell Metab. 2013;17:491–506. doi: 10.1016/j.cmet.2013.03.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Senft D, Ronai ZA. Regulators of mitochondrial dynamics in cancer. Curr Opin Cell Biol. 2016;39:43–52. doi: 10.1016/j.ceb.2016.02.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Chen H, Chan DC. Mitochondrial dynamics in regulating the unique phenotypes of cancer and stem cells. Cell Metab. 2017;26:39–48. doi: 10.1016/j.cmet.2017.05.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Zhao J, Zhang J, Yu M, Xie Y, Huang Y, Wolff DW, et al. Mitochondrial dynamics regulates migration and invasion of breast cancer cells. Oncogene. 2013;32:4814–4824. doi: 10.1038/onc.2012.494. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Ferreira-da-Silva A, Valacca C, Rios E, Pópulo H, Soares P, Sobrinho-Simões M, et al. Mitochondrial dynamics protein Drp1 is overexpressed in oncocytic thyroid tumors and regulates cancer cell migration. PLoS ONE. 2015;10:e0122308. doi: 10.1371/journal.pone.0122308. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Han XJ, Yang ZJ, Jiang LP, Wei YF, Liao MF, Qian Y, et al. Mitochondrial dynamics regulates hypoxia-induced migration and antineoplastic activity of cisplatin in breast cancer cells. Int J Oncol. 2015;46:691–700. doi: 10.3892/ijo.2014.2781. [DOI] [PubMed] [Google Scholar]
- 78.Sun X, Cao H, Zhan L, Yin C, Wang G, Liang P, et al. Mitochondrial fission promotes cell migration by Ca2+ /CaMKII/ERK/FAK pathway in hepatocellular carcinoma. Liver Int. 2018;38:1263–1272. doi: 10.1111/liv.13660. [DOI] [PubMed] [Google Scholar]
- 79.Boulton DP, Caino MC. Mitochondrial fission and fusion in tumor progression to metastasis. Front Cell Dev Biol. 2022;10:849962. doi: 10.3389/fcell.2022.849962. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Ko SH, Choi GE, Oh JY, Lee HJ, Kim JS, Chae CW, et al. Succinate promotes stem cell migration through the GPR91-dependent regulation of DRP1-mediated mitochondrial fission. Sci Rep. 2017;7:12582. doi: 10.1038/s41598-017-12692-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Lu YT, Li LZ, Yang YL, Yin X, Liu Q, Zhang L, et al. Succinate induces aberrant mitochondrial fission in cardiomyocytes through GPR91 signaling. Cell Death Dis. 2018;9:672. doi: 10.1038/s41419-018-0708-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Yu T, Robotham JL, Yoon Y. Increased production of reactive oxygen species in hyperglycemic conditions requires dynamic change of mitochondrial morphology. Proc Natl Acad Sci U S A. 2006;103:2653–2658. doi: 10.1073/pnas.0511154103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Willems PH, Rossignol R, Dieteren CE, Murphy MP, Koopman WJ. Redox homeostasis and mitochondrial dynamics. Cell Metab. 2015;22:207–218. doi: 10.1016/j.cmet.2015.06.006. [DOI] [PubMed] [Google Scholar]
- 84.Ježek J, Cooper KF, Strich R. Reactive oxygen species and mitochondrial dynamics: the yin and yang of mitochondrial dysfunction and cancer progression. Antioxidants (Basel) 2018;7:13. doi: 10.3390/antiox7010013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Liao Z, Chua D, Tan NS. Reactive oxygen species: a volatile driver of field cancerization and metastasis. Mol Cancer. 2019;18:65. doi: 10.1186/s12943-019-0961-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Kamiya T, Courtney M, Laukkanen MO. Redox-activated signal transduction pathways mediating cellular functions in inflammation, differentiation, degeneration, transformation, and death. Oxid Med Cell Longev. 2016;2016:8479718. doi: 10.1155/2016/8479718. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Shin DH, Dier U, Melendez JA, Hempel N. Regulation of MMP-1 expression in response to hypoxia is dependent on the intracellular redox status of metastatic bladder cancer cells. Biochim Biophys Acta. 2015;1852:2593–2602. doi: 10.1016/j.bbadis.2015.09.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Tretter L, Patocs A, Chinopoulos C. Succinate, an intermediate in metabolism, signal transduction, ROS, hypoxia, and tumorigenesis. Biochim Biophys Acta. 2016;1857:1086–1101. doi: 10.1016/j.bbabio.2016.03.012. [DOI] [PubMed] [Google Scholar]
- 89.Toma I, Kang JJ, Sipos A, Vargas S, Bansal E, Hanner F, et al. Succinate receptor GPR91 provides a direct link between high glucose levels and renin release in murine and rabbit kidney. J Clin Invest. 2008;118:2526–2534. doi: 10.1172/JCI33293. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Wittenberger T, Schaller HC, Hellebrand S. An expressed sequence tag (EST) data mining strategy succeeding in the discovery of new G-protein coupled receptors. J Mol Biol. 2001;307:799–813. doi: 10.1006/jmbi.2001.4520. [DOI] [PubMed] [Google Scholar]
- 91.Fredriksson R, Lagerström MC, Lundin LG, Schiöth HB. The G-protein-coupled receptors in the human genome form five main families. Phylogenetic analysis, paralogon groups, and fingerprints. Mol Pharmacol. 2003;63:1256–1272. doi: 10.1124/mol.63.6.1256. [DOI] [PubMed] [Google Scholar]
- 92.Sundström L, Greasley PJ, Engberg S, Wallander M, Ryberg E. Succinate receptor GPR91, a Gα(i) coupled receptor that increases intracellular calcium concentrations through PLCβ. FEBS Lett. 2013;587:2399–2404. doi: 10.1016/j.febslet.2013.05.067. [DOI] [PubMed] [Google Scholar]
- 93.Rubic T, Lametschwandtner G, Jost S, Hinteregger S, Kund J, Carballido-Perrig N, et al. Triggering the succinate receptor GPR91 on dendritic cells enhances immunity. Nat Immunol. 2008;9:1261–1269. doi: 10.1038/ni.1657. [DOI] [PubMed] [Google Scholar]
- 94.Gilissen J, Geubelle P, Dupuis N, Laschet C, Pirotte B, Hanson J. Forskolin-free cAMP assay for Gi-coupled receptors. Biochem Pharmacol. 2015;98:381–391. doi: 10.1016/j.bcp.2015.09.010. [DOI] [PubMed] [Google Scholar]
- 95.Hakak Y, Lehmann-Bruinsma K, Phillips S, Le T, Liaw C, Connolly DT, et al. The role of the GPR91 ligand succinate in hematopoiesis. J Leukoc Biol. 2009;85:837–843. doi: 10.1189/jlb.1008618. [DOI] [PubMed] [Google Scholar]
- 96.Högberg C, Gidlöf O, Tan C, Svensson S, Nilsson-Öhman J, Erlinge D, et al. Succinate independently stimulates full platelet activation via cAMP and phosphoinositide 3-kinase-β signaling. J Thromb Haemost. 2011;9:361–372. doi: 10.1111/j.1538-7836.2010.04158.x. [DOI] [PubMed] [Google Scholar]
- 97.Zhang S, Liang Y, Li L, Chen Y, Wu P, Wei D. Succinate: a novel mediator to promote atherosclerotic lesion progression. DNA Cell Biol. 2022;41:285–291. doi: 10.1089/dna.2021.0345. [DOI] [PubMed] [Google Scholar]
- 98.Li YH, Woo SH, Choi DH, Cho EH. Succinate causes α-SMA production through GPR91 activation in hepatic stellate cells. Biochem Biophys Res Commun. 2015;463:853–858. doi: 10.1016/j.bbrc.2015.06.023. [DOI] [PubMed] [Google Scholar]
- 99.Park SY, Le CT, Sung KY, Choi DH, Cho EH. Succinate induces hepatic fibrogenesis by promoting activation, proliferation, and migration, and inhibiting apoptosis of hepatic stellate cells. Biochem Biophys Res Commun. 2018;496:673–678. doi: 10.1016/j.bbrc.2018.01.106. [DOI] [PubMed] [Google Scholar]
- 100.Rabe P, Liebing AD, Krumbholz P, Kraft R, Stäubert C. Succinate receptor 1 inhibits mitochondrial respiration in cancer cells addicted to glutamine. Cancer Lett. 2022;526:91–102. doi: 10.1016/j.canlet.2021.11.024. [DOI] [PubMed] [Google Scholar]
- 101.Shankavaram U, Fliedner SM, Elkahloun AG, Barb JJ, Munson PJ, Huynh TT, et al. Genotype and tumor locus determine expression profile of pseudohypoxic pheochromocytomas and paragangliomas. Neoplasia. 2013;15:435–447. doi: 10.1593/neo.122132. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Matlac DM, Hadrava Vanova K, Bechmann N, Richter S, Folberth J, Ghayee HK, et al. Succinate mediates tumorigenic effects via succinate receptor 1: potential for new targeted treatment strategies in succinate dehydrogenase deficient paragangliomas. Front Endocrinol (Lausanne) 2021;12:589451. doi: 10.3389/fendo.2021.589451. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Bhuniya D, Umrani D, Dave B, Salunke D, Kukreja G, Gundu J, et al. Discovery of a potent and selective small molecule hGPR91 antagonist. Bioorg Med Chem Lett. 2011;21:3596–3602. doi: 10.1016/j.bmcl.2011.04.091. [DOI] [PubMed] [Google Scholar]
- 104.Mills EL, Pierce KA, Jedrychowski MP, Garrity R, Winther S, Vidoni S, et al. Accumulation of succinate controls activation of adipose tissue thermogenesis. Nature. 2018;560:102–106. doi: 10.1038/s41586-018-0353-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Reddy A, Bozi LHM, Yaghi OK, Mills EL, Xiao H, Nicholson HE, et al. pH-gated succinate secretion regulates muscle remodeling in response to exercise. Cell. 2020;183:62–75.e17. doi: 10.1016/j.cell.2020.08.039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Hobert JA, Mester JL, Moline J, Eng C. Elevated plasma succinate in PTEN, SDHB, and SDHD mutation-positive individuals. Genet Med. 2012;14:616–619. doi: 10.1038/gim.2011.63. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Terra X, Ceperuelo-Mallafré V, Merma C, Benaiges E, Bosch R, Castillo P, et al. Succinate pathway in head and neck squamous cell carcinoma: potential as a diagnostic and prognostic marker. Cancers (Basel) 2021;13:1653. doi: 10.3390/cancers13071653. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Not applicable.