Abstract
As is true for other intracellular pathogens, immunization with live Chlamydia trachomatis generally induces stronger protective immunity than does immunization with inactivated organism. To investigate the basis for such a difference, we studied immune responses in BALB/c mice immunized with viable or UV-killed C. trachomatis mouse pneumonitis (MoPn). Strong, acquired resistance to C. trachomatis infection was elicited by immunization with viable but not dead organisms. Immunization with viable organisms induced high levels of antigen-specific delayed-type hypersensitivity (DTH), gamma interferon production, and immunoglobulin A (IgA) responses. Immunization with inactivated MoPn mainly induced interleukin-10 (IL-10) production and IgG1 antibody without IgA or DTH responses. Analysis of local early cytokine and cellular events at days 3, 5, and 7 after peritoneal cavity immunization showed that high levels of granulocyte-macrophage colony-stimulating factor and IL-12 were detected with viable but not inactivated organisms. Furthermore, enrichment of a dendritic cell (DC)-like population was detected in the peritoneal cavity only among mice immunized with viable organisms. The results suggest that early differences in inducing proinflammatory cytokines and activation and differentiation of DCs may be the key mechanism underlying the difference between viable and inactivated organisms in inducing active immunity to C. trachomatis infection.
Chlamydia trachomatis is a common cause of several sexually transmitted diseases such as urethritis, cervicitis, and salpingitis and is the causative agent of trachoma, the leading cause of preventable blindness worldwide (40). Chlamydial genital infection is also an important risk factor for transmission of human immunodeficiency virus (12, 17). The host defense to chlamydial infection involves both humoral and cell-mediated immunity (CMI) responses (2, 13, 20, 28, 34, 35). In a previous study, we reported that Th1-dependent CMI was the dominant mechanism involved in resolution of C. trachomatis mouse pneumonitis (MoPn) lung infection (41, 42). Gamma interferon (IFN-γ), an immunoregulatory cytokine produced by Th1 cells, is critical in resolution of and resistance to chlamydial infection (7, 15, 26). As well, local antichlamydia immunoglobulin A (IgA) (secretory IgA [sIgA]) antibody in the genital tract has also been associated with resolution of chlamydial infection (20, 29, 30). In one study, local IgA antibodies were inversely correlated with quantitative shedding of the organism during human genital chlamydial infection, suggesting that IgA may play a role in neutralization and/or clearance of the organisms in vivo (3). In support of this conjecture, monoclonal IgA antibody to the major outer membrane protein of the C. trachomatis MoPn biovar was able to protect mice against a chlamydial genital challenge (23). sIgA may thus form a first line of resistance to chlamydial infection. Therefore, efficient induction of the two protective immune mechanisms, CMI and sIgA, are considered to be critical factors in a successful vaccine for prevention of chlamydial infection (32).
It has long been recognized that live vaccines induce stronger protective immunity than do inactivated vaccines, especially for intracellular pathogens (19). Rank et al. (31) showed that guinea pigs immunized with viable C. psittaci, guinea pig inclusion conjunctivitis agent, were more likely to develop resistance to challenge infection than were guinea pigs immunized with inactivated organisms. Subsequent experiments using mice immunized with C. trachomatis MoPn also demonstrated that mice were resistant to vaginal reinfection only if they received live organism; protection was not observed if inactivated MoPn organisms were used as immunogen irrespective of the route of immunization (16, 22). The reason for the striking difference between viable and nonviable chlamydiae in the induction of protective immunity was not clarified in these studies, although several suggestions were entertained. Among these was the notion that viable and nonviable organisms utilized different types of antigen-presenting cells (APCs) to prime naive T cells. Subsequently, Su et al. (36) demonstrated that ex vivo dendritic cells (DCs) pulsed with killed chlamydiae and infused back into the mouse induced strong protective immunity to vaginal infection. Thus, it may be that in vivo immunization with viable chlamydiae preferentially utilizes DCs in the initiation of the immune response, whereas nonviable chlamydiae are unable to use DCs and/or utilize many fewer DCs to initiate the immune response.
In this study, we compared immune responses and protective efficacy following immunization with viable and inactivated C. trachomatis MoPn. We report that immunization with viable but not dead organisms induces significant protection. Using the peritoneal cavity as an immunization site, we demonstrate that the strong protective immunity induced by immunization with viable organisms is associated with early granulocyte-macrophage colony-stimulating factor (GM-CSF) and interleukin-12 (IL-12) cytokine responses and with enrichment for DC-like cells in peritoneal exudate cells. The study provides direct evidence that viable and dead organisms are substantially different immunogens in terms of inducing protective immunity, proinflammatory cytokine production, and DC development.
MATERIALS AND METHODS
Animal and organism.
Female BALB/c mice (4 to 5 weeks old) were purchased from Charles River Canada (Saint Constant, Quebec, Canada). All animals were maintained and used in accordance with the guidelines issued by the Canadian Council on Animal Care.
C. trachomatis MoPn was grown in HeLa cells, and elementary bodies (EBs) were purified by step gradient density centrifugation and kept at −70°C as previously described (13). EBs were inactivated by UV light (G15T8 UV lamp) at a distance of 5 cm for 1 h. No inclusions were measurable when such preparations were cultured in HeLa cells. Both UV-inactivated and viable chlamydiae were separately suspended in sucrose-phosphate-glutamic acid (SPG) buffer (43) for immunization purposes.
Immunization.
For immunization with dead EBs, mice were injected intraperitoneally with 5 × 106 inclusion-forming units (IFU) of UV-inactivated MoPn EBs in 0.1 ml of SPG or with UV-inactivated MoPn EB mixed with an equal volume of incomplete Freund’s adjuvant (IFA) on two occasions at a 2-week interval (0 and 2 weeks). For immunization with viable EBs, groups of mice were injected intraperitoneally with 5 × 106 IFU of viable EBs in 0.1 ml of SPG on two occasions in the same manner as for immunization with dead EBs. Another group of mice was immunized intranasally with 103 IFU of viable MoPn EBs as described previously (43).
Challenge infection and quantification of MoPn.
Mice were challenged intranasally with 5 × 103 IFU of MoPn in 40 μl 60 days after the initial immunization. Body weights were determined daily for 10 days following the challenge infection. Mice were sacrificed, and their lungs were aseptically isolated and homogenized with a cell grinder in cold SPG. The tissue homogenates were centrifuged at 900 × g for 10 min at 4°C to remove coarse tissue and debris. The supernatants were frozen at −70°C until tested. For assessment of MoPn infectivity, lung supernatants were assayed on HeLa-229 cells grown to confluence in flat-bottom 96-well microtiter plates and pretreated with 100 μl of Hanks’ balanced salt solution containing ethylenedinitrilotetraacetic acid (30 ng/ml) for 15 min. After 2 h of incubation at 37°C on a rocker platform, the supernatants were discarded, and the wells were supplemented with 200 μl of Eagle’s minimal essential medium containing 10% heat-inactivated fetal calf serum (FCS), 1.5 μg of cycloheximide per ml, and 12 μg of gentamicin per ml. Plates were incubated for 48 h at 37°C in 5% CO2. The cells were fixed with absolute methanol and stained with a Chlamydia genus-specific murine monoclonal antibody (MAb) followed by goat anti-mouse IgG conjugated to horseradish peroxidase (HRP) to detect inclusion formation. The stained inclusions were developed by 4-chloro-1-naphthol (Sigma, St. Louis, Mo.) and H2O2. The number of inclusions was counted under a light microscope, and chlamydial growth in each lung was calculated based on the dilution titers of the original inocula.
ELISA for antibody test.
Chlamydia-specific serum IgG2a, IgG1, and IgA antibodies collected 6 weeks after the second immunization were determined by using an alkaline phosphatase-based enzyme-linked immunosorbent assay (ELISA) as described previously (43). ELISA plates (catalog no. 25805; Corning Science Products, Corning, N.Y.) were coated with 50 μl of 105 IFU of MoPn EBs in bicarbonate buffer (0.05 M, pH 9.6) overnight at 4°C. Plates were blocked with 2% bovine serum albumin (BSA) dissolved in phosphate-buffered saline (PBS) for 2 h at room temperature and then incubated with serially diluted sera for 4 h at room temperature. After four washes with PBS-Tween 20, biotinylated goat anti-mouse IgG2a, goat anti-mouse IgG1 (Southern Biotechnology Associates Inc., Birmingham, Ala.), or goat anti-mouse IgA (Caltag Laboratories, San Francisco, Calif.) was added to the wells, which were then incubated overnight at 4°C. Alkaline phosphatase-conjugated streptavidin was added and incubated for 45 min at 37°C. After extensive washing, p-nitrophenyl phosphate was added to the plate wells, which were read with a microplate reader at an optical density (OD) of 405 nm.
For examining IgA secretion by spleen cells in vitro, 5 × 106 spleen cells were incubated with or without UV-inactivated MoPn EBs in 2 ml of RPMI 1640 containing 10% FCS at 37°C in 5% CO2 for 24 h. The supernatants were collected following centrifugation at 500 × g for 5 min. Chlamydia-specific IgA antibodies in the supernatants were detected as described above.
MoPn-specific DTH.
Delayed-type hypersensitivity (DTH) was evaluated 6 weeks after the second immunization as previously described (43). Briefly, 25 μl of UV-killed MoPn EBs (2 × 105 IFU) in SPG buffer was injected into one hind footpad of the mouse, and the same volume of SPG buffer was injected into the opposite hind footpad as a negative control. Footpad swelling was measured at 48 and 72 h following the injection, using a dial-gauge caliper (Walter Stern 601; Fisher Scientific, Ottawa, Ontario, Canada). The difference between the thickness of the two footpads was used as a measurement of the DTH response.
Cytokine analysis.
Single-cell suspensions of spleen cells collected from immunized mice were cultured at 5 × 106 cells/ml (2 ml/well) in the presence or absence of UV-killed MoPn EBs (2 × 105IFU/ml), using 24-well plates at 37°C. The culture medium was RPMI 1640 containing 10% FCS, 1% l-glutamine, and 5 × 10−5 M 2-mercaptoethanol. Culture supernatants were harvested at 24, 48, 72, and 96 h.
Cytokines in culture supernatants were detected by a sandwich ELISA using MAbs purchased from PharMingen (San Diego, Calif.). IFN-γ ELISA was conducted with R4-6A2 and XMG1.2 as capture and detection antibodies, respectively. JES5-2A5 and SXC1 as the capture and detection antibodies, respectively, were used for the IL-10 ELISA. The assay conditions were as previously described (13).
GM-CSF and IL-12 in the peritoneal cavity were examined by peritoneal lavage with 2 ml of PBS at selected times after immunization. The lavage fluid was spun down at 500 × g to remove cells and debris. GM-CSF was assayed by an ELISA using MP1-22E9 and MP1-31G6 (PharMingen) as capture and detection antibodies, respectively. IL-12 was assayed with C15.6 (anti-mouse IL-12 p40/p70) and C17.8 (PharMingen) as capture and detection antibodies, respectively. Measurements were performed as recommended by the manufacturer.
Flow cytometry.
Peritoneal cells were collected at various days after intraperitoneal immunization by peritoneal lavage with 2 ml of PBS containing heparin. Cells were washed twice with PBS containing 1% BSA. Then 3 × 105 cells were preincubated with anti-mouse CD16/32 (Fc Block 2.4G2 clone; PharMingen) on ice for 30 min to prevent nonspecific antibody binding through to Fc receptor. Cells were stained with R-phycoerythrin (R-PE)-conjugated hamster anti-mouse CD11c (HL3) and fluorescein isothiocyanate (FITC)-coupled anti-mouse antibodies including anti-I-Ad (39-10-8), anti-CD14 (rmC5-3), anti-CD11b (M1/70), and anti-CD19 (ID3) (PharMingen). The corresponding isotypes were used as negative controls.
All antibodies were used at 1 μg per 106 cells. Ten thousand events were collected on a FACScan cytometer and analyzed with CellQuest software (Becton Dickinson, Mountain View, Calif.). The cells were gated according to scatter, with propidium iodine staining used to eliminate dead cells and debris.
RESULTS
Protective immunity.
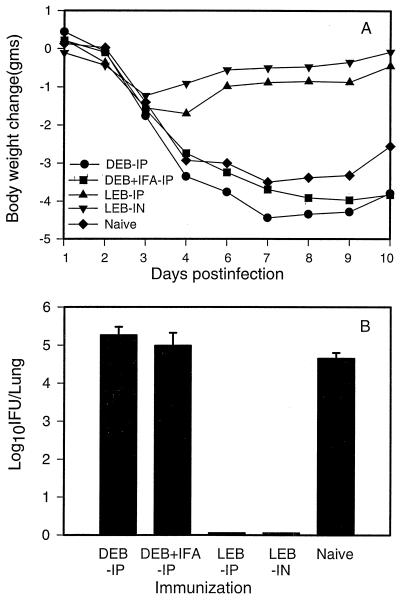
To define protective immunity against MoPn lung infection elicited by viable and dead MoPn EB vaccination, we determined changes in body weight and chlamydial growth in the lungs (plotted as means ± standard errors of the means [SEM]) following intranasal challenge of mice with 5 × 103 IFU of MoPn. As shown in Fig. 1A, dead-EB-immunized mice (with or without IFA) showed levels of body weight loss similar to that observed among unimmunized mice following challenge infection. Chlamydial growth in the lungs of dead-EB-immunized mice was also comparable to that of unimmunized mice (Fig. 1B). The two groups immunized with viable chlamydial EBs either intranasally or intraperitoneally showed only slight body weight loss during the initial 3 to 5 days after challenge infection and then exhibited faster recovery, with body weight returning to near baseline values by day 7 postinfection. Chlamydial growth was virtually undetectable in the lungs of the mice immunized with viable organisms at day 10 following challenge infection. The data indicate that solid resistance to chlamydial challenge was induced by immunization with viable organisms whether the organism was administered intranasally or intraperitoneally. In contrast, intraperitoneal immunization using nonviable organisms with or without adjuvant (IFA) failed to induce significant protective immunity. These data confirm the findings of Kelly et al. (16) and Pal et al. (22).
FIG. 1.
Protective immunity elicited by immunization with live or dead C. trachomatis MoPn EBs. BALB/c mice (five per group) were immunized with UV-inactivated MoPn EBs intraperitoneally (DEB-IP), UV-inactivated MoPn EBs in IFA intraperitoneally (DEB + IFA-IP), or viable EB intraperitoneally (LEB-IP) or intranasally (LEB-IN) on two occasions at 0 and 2 weeks. Mice were then challenged with 5 × 103 IFU of MoPn EBs intranasally at day 60 after the first immunization and sacrificed at day 10 postinfection. (A) Body weight changes measured daily after infection until mice were sacrificed at day 10; (B) MoPn growth in the lung, analyzed by quantitative tissue culture. The data represent means ± SEM of log10 IFU per lung.
Humoral immune responses.
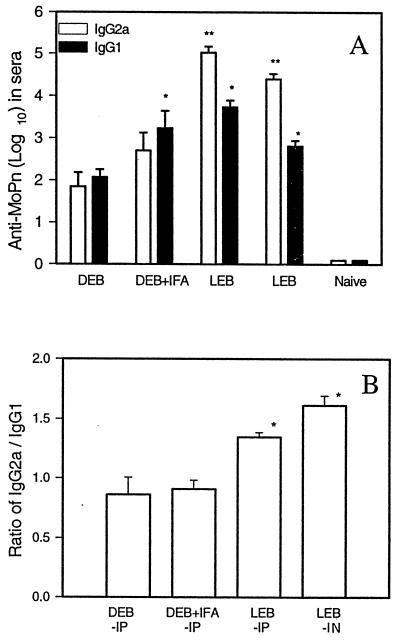
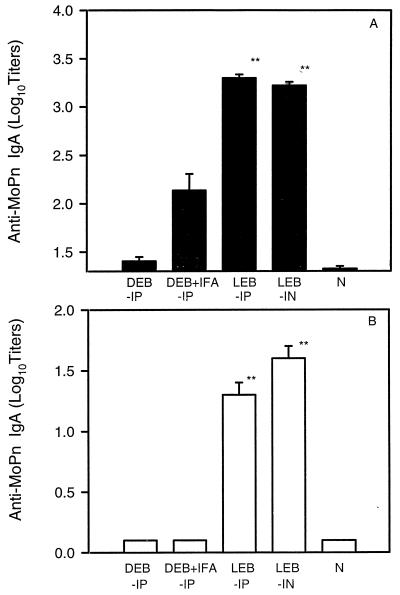
Immune responses evoked by viable and dead organisms were next characterized. Antigen-specific serum antibody responses among immunized mice were assessed prior to challenge infection. Each serum was evaluated for IgG2a, IgG1, and IgA antibodies specific for MoPn EBs. Titers of IgG2a antibodies were 100- to 500-fold higher in mice immunized with viable organisms than in mice immunized with dead EBs (Fig. 2A). As shown in Fig. 2B, IgG2a/IgG1 ratios were greater than 1 among the groups of mice immunized with viable EBs and less than 1 among the groups of mice immunized with dead EBs. Interestingly, serum IgA antibody to MoPn was produced at substantially greater titers by mice immunized with viable organisms than by mice immunized with dead EBs (Fig. 3A). Mice immunized with dead EBs with or without adjuvant showed low or undetectable levels of IgA antibodies. Similarly, MoPn EB-specific IgA antibodies were detectable in the supernatants of cultured splenocytes from viable- but not dead-EB-immunized mice (Figure 3B).
FIG. 2.
Profile of IgG subclasses of MoPn-specific antibodies in the sera of mice immunized with live or dead C. trachomatis MoPn EBs. BALB/c mice (six to nine per group) were immunized with live or dead EBs (LEB or DEB) as described in the legend to Fig. 1A. Sera were collected from immunized mice 6 weeks after the second immunization. MoPn-specific IgG1 and IgG2a antibodies were tested by ELISA. The cutoff point was 0.5 at an OD of 405 nm. (A) Log10 titers (mean ± SEM) of the antibodies; (B) ratios of IgG2a to IgG1 log titers. ∗ represents P < 0.05 and ∗∗ represents P < 0.01 compared with the DEB group.
FIG. 3.
IgA antibodies against C. trachomatis MoPn in sera (A) and in supernatants of cultured splenocytes (B). Mice were immunized and sera were collected as described for Fig. 2. MoPn-specific IgA antibodies were detected by ELISA. The cutoff point is 0.5 at an OD of 405 nm. Spleen cells (5 × 106/ml) from naive mice (N) or mice without antigens immunized with live EBs (LEB) or dead EBs (DEB) were cultured at 37°C in 5% CO2 for 24 h. The supernatants were collected and IgA antibodies against MoPn were detected by ELISA. The columns are labelled as in Fig. 1A. The data represent means ± SEM. ∗∗ represents P < 0.01 compared with the DEB-IP or DEB + IFA-IP group.
In aggregate, the data show that immunization with viable organisms induced strong IgG2a and IgA antibodies whereas dead organisms induced significantly lower levels of IgG2a, with little or no IgA.
CMI and cytokine production.
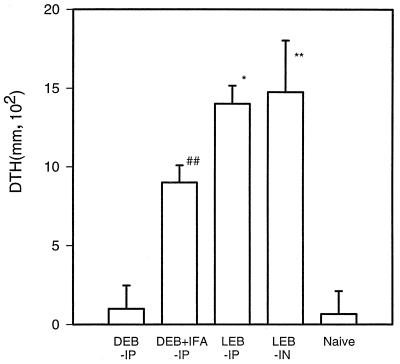
CMI responses following immunization with viable or dead organisms were examined by measuring DTH responses before challenge infection. When viable EBs were used for immunization, high levels of DTH were detected 6 weeks after the second immunization regardless of the route of immunization (intranasal or intraperitoneal) (Fig. 4). In contrast, dead EBs alone did not induce measurable DTH. Immunization with dead EBs plus IFA induced measurable but lower levels of DTH (Fig. 4).
FIG. 4.
DTH responses following immunization with live and dead C. trachomatis MoPn. BALB/c mice (four per group) were immunized as indicated for Fig. 1A. DTH was tested 6 weeks after the second immunization. UV-inactivated MoPn EBs (2 × 105 IFU in 25 μl of SPG buffer) were injected into a hind footpad; 25 μl of SPG buffer was injected into the contralateral hind footpad as a control. Footpad swelling was measured 48 h after the injection. The data represent means ± SEM. ∗ represents P < 0.05 and ∗∗ represents P < 0.01 compared with the DEB + IFA-IP group; ## represents P < 0.01 compared with the DEB-IP group.
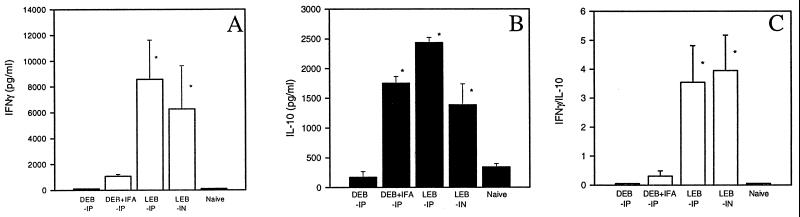
To determine antigen-specific T-cell responses and cytokine profiles, we examined IFN-γ and IL-10 production by cultured spleen cells collected from mice immunized with viable or dead EBs. As shown in Fig. 5A, high levels of antigen driven IFN-γ production were elicited in both groups of mice immunized with viable organisms (intranasally or intraperitoneally). In contrast, there was no measurable IFN-γ produced by spleen cells from mice immunized with dead organisms with or without adjuvant. Antigen-driven IL-10 production was detected in the culture supernatants of splenocytes collected from mice immunized with viable organisms (and was significantly greater for mice immunized intraperitoneally than for mice immunized intranasally (P < 0.05) (Fig. 5B). Interestingly, IFA enhanced IL-10, but not IFN-γ production among mice immunized with dead organisms. As shown in Fig. 5C, the ratio of IFNγ to IL-10 production in groups of mice immunized with viable EBs was greater than 1, while the ratio in mice immunized with dead EBs was less than 1. This finding suggests that immunization with viable EBs induces mainly Th1-like cells whereas intraperitoneal immunization with nonviable EBs with or without IFA induces predominantly Th2-like responses. This interpretation is also consistent with the pattern of specific antibody isotopes (IgG2a and IgG1) induced by viable or dead organisms (Fig. 2B).
FIG. 5.
Profile of IFN-γ and IL-10 production by spleen cells isolated from mice immunized with live or dead C. trachomatis MoPn EBs. BALB/c mice (four per group) were immunized as described for Fig. 1A. Spleen cells were harvested at 6 weeks following the second immunization; 5 × 106/ml spleen cells were incubated with 2 × 105 IFU of UV-killed MoPn EBs in 5% CO2 at 37°C for 96 h. The amounts of IFN-γ (A) and IL-10 (B) in the supernatants of the cell cultures were measured by ELISA. The IFN-γ/IL-10 ratio was also determined (C). The columns are labeled as in Fig. 1. The data represent means ± SEM. ∗ represents P < 0.05 compared with the DEB-IP group (A to C) or DEB + IFA-IP group (A and C).
To elucidate the mechanism(s) underlying the different patterns of immune responses induced by viable and dead EBs, we further examined the cellular and cytokine environment during the initiation of the immune response following immunization with viable or dead organisms.
Cytokine production at the site of immunization.
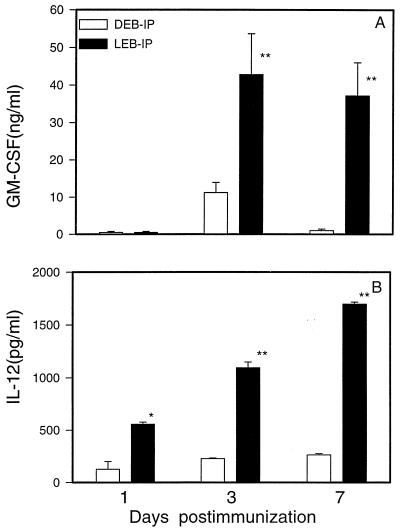
We initially analyzed levels of proinflammatory cytokines (GM-CSF and IL-12) in the peritoneal cavity, the site of immunization, of mice immunized with viable versus dead EBs. GM-CSF is known to be important for DC growth and maturation (1), and epithelial cells infected with C. trachomatis are known to produce substantial amounts of GM-CSF (33). GM-CSF levels in the peritoneal lavage were determined at 1, 3, 7, and 21 days after intraperitoneal immunization with either viable or dead organisms. As shown in Fig. 6A, GM-CSF production was significantly higher and sustained for a longer period in mice immunized with viable organisms than in mice immunized with dead organisms. IL-12 was induced by immunization with viable organisms but not by immunization with dead organisms (Fig. 6B). IL-12 production induced by viable EBs increased in amount over the 7 days of measurement. Notably, IL-12 production among mice immunized with dead EBs at a dose (5 × 106 IFU) 10-fold or more higher than that used for viable EB immunization was still significantly lower (data not shown). The results indicate that viable organisms are significantly more potent stimulators of proinflammatory cytokines than are dead organisms.
FIG. 6.
GM-CSF and IL-12 in peritoneal lavage of mice immunized with live or dead C. trachomatis MoPn EBs. BALB/c mice (four to six per group) were immunized intraperitoneally with viable (LEB-IP) or UV-inactivated (DEB-IP) MoPn EBs. The peritoneal cavity was washed with PBS at days 1, 3, and 7 following immunization. GM-CSF (A) and IL-12 (B) in the supernatants of the peritoneal lavage were detected by ELISA. The data represent means ± SEM. ∗ represents P < 0.05 and ∗∗ represents P < 0.01 compared with the DEB-IP group.
DC-like cell differentiation a the site of immunization with viable organisms.
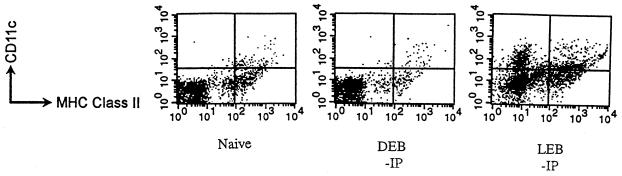
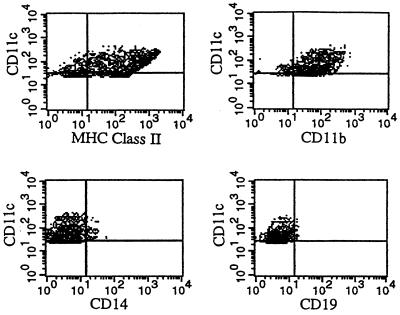
The capacity of DCs to load a large amount of antigenic peptide and to accumulate major histocompatibility complex (MHC) class II peptide complexes on their cell surface following exposure to inflammatory stimuli could favor presentation of antigens from infectious agents that elicit innate production of proinflammatory cytokines such as GM-CSF (4, 33). To identify whether DCs are differentially involved in the induction of immune responses induced by viable versus dead chlamydial EBs, we analyzed peritoneal cells for DC surface markers by flow cytometry. Since DCs typically express high levels of CD11c and MHC class II molecules on the cell surface (8, 11), we examined the expression of these molecules on peritoneal cells collected from immunized mice by using double staining with MAbs. As shown in Fig. 7, more peritoneal cells from mice immunized with viable EBs expressed CD11c molecules at day 3 postimmunization. Two distinct populations of CD11c+ cells, CD11c+ class II− and CD11c+ class II+, were observed (Fig. 7). The CD11c+ class II+ cell population increased from 6.9% ± 2.5% at day 3 to 13.1% ± 0.45% at day 7 as a proportion of the total peritoneal cells from mice immunized with viable organisms (Table 1). In contrast, CD11c+ cells were not measurable in the peritoneal lavage of mice immunized with dead organisms at day 3 postimmunization, and only a small population of CD11c+ class II+ cells was detectable on day 7 (3.2% ± 0.7% of the peritoneal cell population) (Table 1). The enrichment of CD11c+ class II+ cells in the peritoneal cavity of viable-EB-immunized mice was paralleled by the increase in GM-CSF and IL-12 levels in peritoneal lavage (Fig. 6). Since CD11c and MHC class II molecules can also be expressed on other cell populations, such as macrophages and B cells, we also stained peritoneal cells with antibodies which are specific for macrophages (anti-CD14) and B cells (anti-CD19) to clarify the identity of the CD11c+ cells observed in viable-EB-immunized mice. As shown in Fig. 8, the CD11c+ cells collected from viable-EB-immunized mice were CD14− CD19− CD11b+. Because CD14 is rarely expressed on DCs and CD19 is not expressed on DCs (11), the results indicate that the CD11c+ class II+ cells in the peritoneal lavage immunized with viable EBs are most likely DC-like cells.
FIG. 7.
Flow cytometry analysis of DC-like cells in peritoneal cavities of mice immunized with live or dead C. trachomatis MoPn EBs. Mice were immunized with live (LEB) or UV-inactivated dead (DEB) MoPn EBs, and peritoneal cells were collected 3 days postimmunization. Cells were stained with PE-coupled anti-CD11c MAb and FITC-coupled anti-I-Ad MAb. Two-color immunofluorescence analysis dot plots for the peritoneal cells were generated after exclusion of dead cells by using propidium iodide. The quadrant setting was positioned on the isotype control.
TABLE 1.
Distribution of CD11c and MHC class II markers on peritoneal cells obtained from naive mice immunized with live and dead C. trachomatis MoPn EBsa
| Group | Mean % positive ± SEMb
|
||
|---|---|---|---|
| CD11c+ class II− | CD11c+ class II+ | CD11c− class II+ | |
| Day 0 | 0 | 4.1 ± 1.0 | 19.8 ± 3.6 |
| Day 3 | |||
| DEB | 0 | 0.3 ± 0.2 | 5.3 ± 1.6 |
| LEB | 8.2 ± 1.5c | 6.9 ± 2.5c | 11.8 ± 2.9 |
| Day 7 | |||
| DEB | 0 | 3.2 ± 0.75 | 21.2 ± 8.6 |
| LEB | 0 | 13.1 ± 0.45 | 20.3 ± 5.8 |
| Day 21 | |||
| DEB | 0 | 0.75 | 29.7 |
| LEB | 0 | 0.4 | 43.0 |
Four mice for each group were studied on days 0, 3, and 7 postimmunization. One mouse from each group was studied at day 21. Mice were immunized intraperitoneally with dead (UV-killed) or live MoPn EB (DEB or LEB). The cells were double stained with PE-conjugated anti-CD11c and FITC-conjugated anti-MHC class II and analyzed by flow cytometry.
Percentage of surface marker-positive cells as a proportion of total viable peritoneal cell population. Propidium iodine staining and scatter gating were used to exclude dead cells.
P < 0.05 compared with the group of mice immunized with dead EB.
FIG. 8.
Flow cytometry analysis for CD11c+ cells in peritoneal cavities of mice immunized 3 days previously with live C. trachomatis EBs. Peritoneal cells were collected from mice immunized with live EBs by lavaging with PBS, and the cells were double stained by PE-coupled MAb against CD11c and FITC-coupled anti-MHC class II(I-Ad), anti-CD14, anti-CD11b, or anti-CD19 MAbs. The cell population with CD11c+ after exclusion of dead cells was gated according to its isotype control.
DISCUSSION
This study shows that different immune responses are induced by immunization with viable versus dead C. trachomatis MoPn EBs. Intraperitoneal or intranasal immunization with viable chlamydiae elicited IFN-γ dominant responses associated with strong DTH and high levels of EB-specific IgG2a and IgA antibodies. The IFN-γ/IL-10 and IgG2a/IgG1 ratios among viable-EB-immunized mice were significantly higher than the ratios among mice immunized with dead EBs. Inactivated organisms together with IFA induced IL-10-dominant responses, with lower levels of IgG1 and IgG2a antibodies. Interestingly, immunization with viable organisms either given intranasally or intraperitoneally induced high levels of serum IgA antibodies, while immunization with dead organisms with adjuvant induced significantly lower levels of IgA antibodies.
Although we made our observations with C. trachomatis in a single mouse strain (BALB/c), the phenomenon that viable and dead organisms induce different immune responses has been noted with other bacterial pathogens in multiple mouse strains and appears to be especially important for intracellular bacteria such as salmonellae and mycobacteria (5, 10, 38). For instance, one informative study by Thatte et al. (37) demonstrated that intraperitoneal immunization with live Salmonella typhimurium induced an IFN-γ-dominant response associated with strong DTH and IgG2a antibody production, while heat-killed S. typhimurium elicited an IL-4-dominant response with lower DTH and higher levels of IgG1 production.
Why should the immune responses induced by live and dead organisms be so different? Possible explanations include the density of antigenic peptides presented by APCs; also, the types of APC used to prime naive T cells could be different under the two immunization conditions. Since Th1 and Th2 cells can be induced by identical antigenic peptides, T-cell receptor ligand density on the APCs has been considered a determining factor for differential induction of Th1- or Th2-like responses (6, 27). Such experiments show that a high density of MHC-antigen peptide complexes on the surface of APCs tends to stimulate Th1 cell responses, while low ligand density tends to stimulate Th2 responses. Therefore, it may be that infection of APCs by live organism results in higher ligand density, thus inducing Th1-like responses. In the case of chlamydial infection, however, this mechanism may not be valid because recent observations suggest that APCs such as DCs do not readily support the replication of C. trachomatis (21). Nonetheless, we still consider that ligand density may be relevant to the observations we have made and that further experiments are indicated. The ex vivo pulsing of DCs with killed or viable chlamydiae as reported by Su et al. (36) offers an opportunity to evaluate the quantitative importance of this parameter.
Another and possibly more critical factor in determining the differences in immune responses induced by live and dead MoPn EBs is the difference in activation and differentiation of APCs. When viable and dead organisms are introduced into a host, the early innate response that they elicit may be critical in canalizing the subsequent adaptive immune responses. We observed that high levels of GM-CSF and IL-12 were produced in the peritoneal cavity of mice immunized with viable but not dead C. trachomatis. GM-CSF was abundant at the early stages of inflammation (day 3 to day 7) and then declined in amount by day 21 (data not shown). Analysis of peritoneal cell composition demonstrated the enrichment for DC-like cells occurring during the time interval when GM-CSF and IL-12 levels were high (from day 3 to day 7) (Table 1 and Fig. 6). The correlation between GM-CSF production and DC-like cell enrichment suggests that GM-CSF production may be critical for development of DC-like cells following viable EB immunization. C. trachomatis replication in epithelial cells is known to stimulate production of proinflammatory cytokines including GM-CSF (33), and since chlamydiae are able to grow in mesothelial cells (39), it may be that infection of these cells following intraperitoneal immunization with live organism induces the GM-CSF cytokine production that we observed. Since submucosal spaces are known to have rich populations of DCs (24, 25), we speculate that such effects may also occur at epithelial sites of infection. Further studies that more directly test the causal relationships among in vivo C. trachomatis growth, GM-CSF production, DC recruitment, and enhanced immunity are needed.
Matured DCs are the important source of IL-12 and other cytokines that help T-cell activation (18). Since IL-12 is essential to the differentiation of naive T cells into Th1 effector cells, the high levels of IL-12 observed after immunization with viable MoPn EBs may be a mechanism by which viable EBs preferentially induce Th1 immune responses. This speculation is supported by the observation that GM-CSF and IL-12 appeared at only low levels in the peritoneal cavities of mice immunized with inactivated chlamydiae and that immunization with nonviable EBs elicited only marginal levels of CD11c+ DC-like cells (Fig. 6). We speculate that immunization with nonviable organisms may fail to recruit and activate DCs and thus fail to induce strong cellular immune responses.
An interesting finding in this study is the preferential induction of high levels of IgA by immunization with viable MoPn EBs. This observation is not unprecedented since immunization with live but not killed Aro− S. typhimurium also induced strong IgA class switching (10). Based on the Th1/Th2 paradigm, IgA responses are traditionally thought to be associated with Th2 cytokines such as IL-4, IL-5, and IL-6 which have been shown to facilitate IgA class switching. We assayed for IL-4 and IL-5 in the supernatants of antigen-stimulated spleen cells from mice immunized with viable or dead organisms, but neither of these cytokines was readily detected (data not shown). Since we found that mice immunized with viable EBs mounted IFN-γ-dominant Th1 responses and also had high levels of IgA production, this created an apparent paradox. The paradox may be explained by the recently reported observation that DC-like cells regulate IgA isotype switching of CD40-activated human B cells (9). Fayette et al. (9) reported that in these experiments, DCs in the presence of IL-10 and tumor necrosis factor alpha potentiate IgA class switching by CD40-activated naive B cells. Thus, the enrichment for DC-like cells in the peritoneal exudate together with antigen-specific IL-10 secretion could account for the strong IgA responses that were elicited under Th1-dominant circumstances following viable EB immunization.
The finding of difference in protective immunity induced by viable and dead MoPn EBs and its association with proinflammatory cytokine (GM-CSF and IL-12) production and DC-like cell differentiation is helpful for the rational design of a chlamydial vaccine. It is self-evident that a protective vaccine should mimic viable organisms in terms of activating DCs and thus inducing strong protective immunity. Since antigen presentation by DCs can activate both protective Th1-like responses (DTH and IFN-γ production) and IgA responses, it may be possible to develop a chlamydial vaccine which induces both CMI and IgA, the two important lines of defense against chlamydial infection by specifically targetting in vivo vaccine delivery to DCs. The recent observations of Su et al. (36) demonstrating the remarkable immunogenicity of ex vivo chlamydia-laden DCs in producing protective immunity to vaginal chlamydial infection and oviductal pathology are extremely encouraging for this line of research.
ACKNOWLEDGMENTS
This work was supported by the Medical Research Council of Canada University-Industry Program (operating grant UI-14876), Pasteur Mérieux Connaught, and the Canadian Bacterial Diseases Network (grant project VP4).
We are grateful to Shuhong Zhao for technical assistance.
REFERENCES
- 1.Banchereau J, Steinman R M. Dendritic cells and the control of immunity. Nature. 1998;392:245–252. doi: 10.1038/32588. [DOI] [PubMed] [Google Scholar]
- 2.Bavoil P M, Hsia R C, Rank R G. Prospects for a vaccine against chlamydia genital disease. I. Microbiology and pathogenesis. Bull Inst Pasteur. 1996;94:5. [Google Scholar]
- 3.Brunham R C, Kuo C C, Cles L, Holmes K K. Correlation of host immune response with quantitative recovery of Chlamydia trachomatis from the human endocervix. Infect Immun. 1983;39:1491–1494. doi: 10.1128/iai.39.3.1491-1494.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Cella M, Engering A, Pinet V, Pieters J, Lanzavecchia A. Inflammatory stimuli induce accumulation of MHC class II complexes on dendritic cells. Nature. 1997;388:782–787. doi: 10.1038/42030. [DOI] [PubMed] [Google Scholar]
- 5.Chambers M A, Marshall B G, Wangoo A, Bune A, Cook H T, Shaw R J, Young D B. Differential responses to challenge with live and dead Mycobacterium bovis Bacillus Calmette-Guérin. J Immunol. 1997;158:1742–1748. [PubMed] [Google Scholar]
- 6.Constant S, Pfeiffer C, Woodard A, Pasqualini T, Bottomly K. Extent of T cell receptor ligation can determine the functional differentiation of naive CD4 T cells. J Exp Med. 1995;182:591–1596. doi: 10.1084/jem.182.5.1591. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Cotter T W, Ramsey K H, Miranpuri G S, Poulsen C E, Byrne G I. Dissemination of Chlamydia trachomatis chronic genital tract infection in gamma interferon gene knockout mice. Infect Immun. 1997;65:2145–2152. doi: 10.1128/iai.65.6.2145-2152.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.De Smedt T, Pajak B, Muraille E, Lespagnard L, Heiner E, De Baetselier P, Urbain J, Leo O, Moser M. Regulation of dendritic cell numbers and maturation by lipopolysaccharide in vivo. J Exp Med. 1996;184:1413–1424. doi: 10.1084/jem.184.4.1413. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Fayette J, Dubois B, Vandenabeele S, Bridon J-M, Vanbervliet B, Durand I, Banchereau J, Caux C, Briére F. Human dendritic cells skew isotype switching of CD40-activated naive B cells towards IgA1 and IgA2. J Exp Med. 1997;185:1909–1918. doi: 10.1084/jem.185.11.1909. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Harrison J A, Villarreal-Ramos B, Mastroeni P, Demarco de Hormaeche R, Hormaecche C E. Correlates of protection induced by live Aro−Salmonella typhimurium vaccines in the murine typhoid model. Immunology. 1997;90:618–625. doi: 10.1046/j.1365-2567.1997.00158.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Hart D N J. Dendritic cells: unique leukocyte populations which control the primary immune response. Blood. 1997;90:3245–3287. [PubMed] [Google Scholar]
- 12.Ho J L, He S, Hu A, Geng J, Basile F G, Almeida M G, Saito A Y, Laurence J, Johnson W D., Jr Neutrophils from human immunodeficiency virus (HIV)-seronegative donors induce HIV replication from HIV-infected patients’ mononuclear cells and cell line: an in vitro model of HIV transmission facilitated by Chlamydia trachomatis. J Exp Med. 1995;181:1493–1505. [PMC free article] [PubMed] [Google Scholar]
- 13.Igietseme J U D, Magee M, Williams D M, Rank R G. Role for CD8+ T cells in antichlamydial immunity defined by chlamydia-specific T lymphocyte clones. Infect Immun. 1994;62:5195–5197. doi: 10.1128/iai.62.11.5195-5197.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Inaba K, Inaba M, Romani N, Aya H, Deguchi M, Ikehara S, Muramatsu S, Steinman R M. Generation of large numbers of dendritic cells from mouse bone marrow cultures supplemented with granulocyte/macrophage colony-stimulating factor. J Exp Med. 1992;176:1693–1702. doi: 10.1084/jem.176.6.1693. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Johansson M, Schön K, Ward M, Lycke N. Genital tract infection with Chlamydia trachomatis fails to induce protective immunity in gamma interferon receptor-deficient mice despite a strong local immunoglobulin A response. Infect Immun. 1997;65:1032–1044. doi: 10.1128/iai.65.3.1032-1044.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Kelly K A, Robinson E A, Rank R G. Initial route of antigen administration alters the T-cell cytokine profile produced in response to the mouse pneumonitis biovar of Chlamydia trachomatis following genital infection. Infect Immun. 1996;64:4976–4983. doi: 10.1128/iai.64.12.4976-4983.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Laga M, Manoka A, Kivuvu M, Malele B, Tuliza M, Nzila N, Goeman J, Batter V, Alary M. Non-ulcerative sexually transmitted diseases as risk factors for HIV-1 transmission in women: results from a cohort study. AIDS. 1993;7:95–102. doi: 10.1097/00002030-199301000-00015. [DOI] [PubMed] [Google Scholar]
- 18.Macatonia S E, Hosken N A, Litton M, Vieira P, Hsieh C-S, Culpepper J A, Wysocka M, Trinchieri G, Murphy K M, O’Garra A. Dendritic cells produce IL-12 and direct the development of Th1 cells naive from CD4+ T cells. J Immunol. 1995;154:5071–5079. [PubMed] [Google Scholar]
- 19.Mackaness G B. The relationship of delayed hypersensitivity to acquired cellular resistance. Br Med Bull. 1967;23:52–54. doi: 10.1093/oxfordjournals.bmb.a070516. [DOI] [PubMed] [Google Scholar]
- 20.Morrison R P, Feilzer K, Tumas D B. Gene knockout mice establish a primary protective role for major histocompatibility complex class II-restricted responses in Chlamydia trachomatis genital tract infection. Infect Immun. 1995;63:4661–4668. doi: 10.1128/iai.63.12.4661-4668.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Ojcius D M, de Alba Y B, Kanellopoulos J M, Hawkins R A, Kelly K A, Rank R G, Dautry-Varsat A. Internalization of chlamydia by dendritic cells and stimulation of chlamydia-specific T cells. J Immunol. 1998;160:1297–1303. [PubMed] [Google Scholar]
- 22.Pal S, Peterson E M, de la Maza L M. Intranasal immunization induced long-term protection in mice against a Chlamydia trachomatis genital challenge. Infect Immun. 1996;64:5341–5348. doi: 10.1128/iai.64.12.5341-5348.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Pal S, Theodor I, Peterson E M, de la Maza L M. Monoclonal immunoglobulin A antibody to the major outer membrane protein of the Chlamydia trachomatis mouse pneumonitis biovar protects mice against a chlamydial genital challenge. Vaccine. 1997;15:575–582. doi: 10.1016/s0264-410x(97)00206-5. [DOI] [PubMed] [Google Scholar]
- 24.Parr M B, Parr E L. Antigen recognition in the female reproductive tract. I. Uptake of intraluminal protein tracers in the mouse vagina. J Reprod Immunol. 1990;17:101–104. doi: 10.1016/0165-0378(90)90029-6. [DOI] [PubMed] [Google Scholar]
- 25.Parr M B, Parr E L. Langerhans cells and T lymphocyte subsets in the murine vagina and cervix. Biol Reprod. 1991;44:491–498. doi: 10.1095/biolreprod44.3.491. [DOI] [PubMed] [Google Scholar]
- 26.Perry L L, Feilzer K, Caldwell H D. Immunity to Chlamydia trachomatis is mediated by T helper 1 cells through IFNγ-dependent and independent pathways. J Immunol. 1997;158:3344–3355. [PubMed] [Google Scholar]
- 27.Pfeiffer C, Stein J, Southwood S, Ketelaar H, Sette A, Bottomly K. Altered peptide ligands can control CD4 lymphocyte differentiation in vivo. J Exp Med. 1995;181:1569–1574. doi: 10.1084/jem.181.4.1569. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Ramsey K H, Rank R G. Resolution of chlamydial genital infection with antigen-specific T-lymphocyte lines. Infect Immun. 1991;59:925–931. doi: 10.1128/iai.59.3.925-931.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Rank R G, Barron A L. Humoral immune response in acquired immunity to chlamydial genital infection of female guinea pigs. Infect Immun. 1983;39:463–465. doi: 10.1128/iai.39.1.463-465.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Rank R G, Batteiger B E. Protective role of serum antibody in immunity to chlamydial genital infection. Infect Immun. 1989;57:299–301. doi: 10.1128/iai.57.1.299-301.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Rank R G, Batteiger B E, Soderberg L S F. Immunization against chlamydial genital infection in guinea pigs with UV-inactivated and viable chlamydiae administered by different routes. Infect Immun. 1990;58:2599–2605. doi: 10.1128/iai.58.8.2599-2605.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Rank R G, Bavoil P M. Prospects for a vaccine against Chlamydia genital disease. II. Immunity and vaccine development. Bull Inst Pasteur. 1996;94:55–82. [Google Scholar]
- 33.Rasmussen S J, Eckmann L, Quayle A J, Shen L, Zhang Y-X, Anderson D J, Fierer J, Stephens R S, Kagnoff M F. Secretion of proinflammatory cytokines by epithelial cells in response to chlamydia infection suggests a central role for epithelial cells in chlamydial pathogenesis. J Clin Investig. 1997;99:77–87. doi: 10.1172/JCI119136. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Starnbach M N, Bevan M J, Lampe M F. Protective cytotoxic T lymphocytes are induced during murine infection with Chlamydia trachomatis. J Immunol. 1994;153:5183–5189. [PubMed] [Google Scholar]
- 35.Su H, Caldwell H D. CD4+ T cells play a significant role in adoptive immunity to Chlamydia trachomatis infection of the mouse genital tract. Infect Immun. 1995;63:3302–3308. doi: 10.1128/iai.63.9.3302-3308.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Su H, Messer R, Whitmire W, Fischer E, Portis J C, Caldwell H D. Vaccination against chlamydial genital tract infection after immunization with dendritic cells pulsed ex vivo with nonviable chlamydiae. J Exp Med. 1998;188:809–818. doi: 10.1084/jem.188.5.809. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Thatte J, Rath S, Bal V. Immunization with live versus killed Salmonella typhimurium leads to the generation of an IFNγ-dominant versus an IL-4-dominant immune response. Int Immunol. 1993;5:1431–1436. doi: 10.1093/intimm/5.11.1431. [DOI] [PubMed] [Google Scholar]
- 38.VanCott J L, Staats H F, Pascual D W, Roberts M, Chatfield S N, Yamamoto M, Coste M, Carter P B, Kiyono H, McGhee J R. Regulation of mucosal and systemic antibody response by T helper cell subsets, macrophages, and derived cytokines following oral immunization with live recombinant salmonella. J Immunol. 1996;156:1504–1514. [PubMed] [Google Scholar]
- 39.van Westreenen M, Pronk A, Diepersloot R J A, de Groot P G, Leguit P. Chlamydia trachomatis infection of human mesothelial cells alters proinflammatory, procoagulant and fibrinolytic responses. Infect Immun. 1998;66:2352–2355. doi: 10.1128/iai.66.5.2352-2355.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.West S K, Rapoza P, Munoz B, Katala S, Taylor H R. Epidemiology of ocular chlamydial infection in a trachoma-hyperendemic area. J Infect Dis. 1991;163:752–756. doi: 10.1093/infdis/163.4.752. [DOI] [PubMed] [Google Scholar]
- 41.Yang X, Brunham R C. T lymphocyte immunity in host defence against Chlamydia trachomatis and its implication for vaccine development. Can J Infect Dis. 1998;9:99–109. doi: 10.1155/1998/395297. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Yang X, HayGlass K T, Brunham R C. Genetically determined differences in IL-10 and IFNγ responses correlate with clearance of Chlamydia trachomatis mouse pneumonitis infection. J Immunol. 1996;156:4338–4344. [PubMed] [Google Scholar]
- 43.Zhang D-J, Yang X, Berry J, Shen C, McClarty G, Brunham R C. DNA vaccination with the major outer-membrane protein gene induced acquired immunity to Chlamydia trachomatis (mouse pneumonitis) infection. J Infect Dis. 1997;176:1035–1040. doi: 10.1086/516545. [DOI] [PubMed] [Google Scholar]