Abstract
Background
Iron deficiency (ID) is a frequent condition in patients with inflammatory bowel disease (IBD).
Aim
Our aim was to investigate the prevalence of ID in patients with IBD.
Methods
This was a prospective multicenter cross-sectional study conducted in 21 gastroenterology departments in France between January and March 2020. All adult patients with confirmed IBD who were admitted to the hospital were eligible for inclusion. ID was defined as ferritinemia ≤ 100 μg/L in patients with signs of inflammation (C-reactive protein (CRP) ≥ 5 mg/L) or ferritinemia < 30 μg/L in the absence of inflammation.
Results
In total, 1036 IBD (685 Crohn’s disease and 351 ulcerative colitis) patients (52.1% women) with a mean age of 41.8 ± 15.5 years were recruited. Approximately half of the patients (504, 51.1%) were in disease remission at the time of enrollment. Systematic monitoring of iron status was performed in 12/21 (57%) participating centers, including measurements of ferritin (12/12, 100%), hemoglobin (11/12, 92%), transferrin saturation (TSAT) (6/12, 50.0%), and serum iron (5/12, 42%). About one-fifth of the patients had been treated with intravenous iron (218, 21.0%), whereas only a small percentage received oral iron (36, 3.5%). ID occurred in 97 patients (23.7% CI 95% 19.8–28.1). Patients with moderate/severe IBD activity (OR: 3.66; CI 95% 24.4–61.2; p = 0.007) or concomitant anemia (OR: 3.66; CI 95% 1.97–6.78; p < 0.001) had an increased likelihood of having ID.
Conclusion
Patients with moderate/severe IBD activity or concomitant anemia are at increased risk of ID. Early detection and management of ID in patients with IBD is recommended.
Keywords: Iron deficiency, Prevalence, Inflammatory bowel disease, Ulcerative colitis, Crohn’s disease
Introduction
Iron deficiency is a common condition in patients with inflammatory diseases [1]. Specifically, up to three quarters of patients with chronic inflammatory bowel disease (IBD) experience iron deficiency, resulting in frequent IBD extra-intestinal manifestations [2, 3]. Iron deficiency has a multifactorial origin as it depends on blood loss, a reduced intake, or a reduced absorption of iron [4, 5].
Iron is essential to many cellular mechanisms [3]. A disruption of its metabolism often leads to fatigue, anemia, a decrease in immune defenses, and an impairment of cognitive and physical performance, significantly impacting patients’ quality of life and disease prognosis [6].
However, iron deficiency remains insufficiently diagnosed and rarely managed [7, 8]. To date, based on the recommendations of the European Crohn’s and Colitis Organization (ECCO), iron deficiency is mainly investigated to exclude the presence of anemia [9]. It is important to emphasize that anemia is a relatively late manifestation of iron deficiency and it is not experienced by all iron-deficient patients [8].
Iron deficiency is an underdiagnosed problem as there is no commonly validated definition [10, 11]. Furthermore, there is no standardized algorithm for its monitoring in the absence of anemia, making differential diagnosis difficult [10]. Growing evidence shows that martial-deficient patients have a lower risk of hospitalization and anemia after iron supplementation, leading to a significant reduction in healthcare costs [12].
Studies specifically designed to investigate martial deficiency and define the optimal management of these patients are needed. To overcome this problem, we conducted the CARENFER study with the aim of assessing the prevalence of iron deficiency in patients with IBD through a French multicenter cross-sectional study.
Methods
Study Design and Inclusion Criteria
This was a prospective multicenter cross-sectional study conducted in 21 gastroenterology departments in France between January and March 2020.
All adult patients with confirmed diagnosis of IBD who were present in the medical unit during the study period, whether inpatient or outpatient, were eligible for inclusion and were selected based on a voluntary basis at a national level.
Few inclusion criteria were considered in order to limit selection bias: 18 years old or more, registration with a Social Security system, and signed written informed consent. Pregnant women, breastfeeding women, patients under guardianship or curatorship as well as patients hospitalized without consent were not included.
Patients underwent evaluation of the iron status within 7 days of signing informed consent. However, for patients who had a recent (i.e., within 7 days before their inclusion in the present study) determination of Hb level and iron stores, no additional biochemical assessment was performed at inclusion. Iron balance monitoring included evaluation of serum iron, ferritin, transferrin saturation (TSAT), and hemoglobin (Hb).
Iron deficiency was defined as ferritinemia ≤ 100 μg/L in patients with biological signs of inflammation (C-reactive protein (CRP) ≥ 5 mg/L) or ferritinemia < 30 μg/L in the absence of inflammation (CRP < 5 mg/L) [9].
Iron deficiency was also divided into absolute or functional. Serum ferritin < 30 μg/L and TSAT < 16% were associated with absolute iron deficiency, while ferritin < 100 μg/L and TSAT < 16% were attributable to a functional deficiency [13, 14].
A threshold Hb concentration of 12 g/dL for women and 13 g/dL for men was used to define anemia. Anemia was then classified into mild (Hb 10–12 g/dl for women and 10–13 g/dl for men), moderate (Hb 8–10 g/dl), and severe (Hb < 8 g/dL) [15].
Finally, healthcare professionals were asked to answer a questionnaire in order to investigate the type of screening for iron deficiency and the duration and the type of iron therapies.
Data Collection
The clinical and biochemical data were extracted from the patient’s electronic medical records from each participating hospital. The following data were collected for each patient: gender, date of birth, weight, height, body mass index (BMI), IBD type [Crohn’s disease (CD) or ulcerative colitis (UC)], smoking status, disease extent based on the Montreal classification (the severity of relapse was classified into four disease activity/severity as clinical remission mild, moderate, and severe, as previously described), IBD medical treatments (including oral and local 5-aminosalicyclic acid [5-ASA], corticosteroids, antibiotics, immunosuppressants, tumor necrosis factor-α inhibitors [anti-TNF], vedolizumab, ustekinumab, and tofacitinib), previous or ongoing iron deficiency therapies (oral or intravenous, dosage, and duration), reason for hospitalization, type of hospitalization (traditional, diurnal, weekday, or following a visit), and CRP concentrations [16].
Statistical Analysis and Ethic Committee Approval
Quantitative variables were expressed as mean, standard deviation, median, and interquartile value. The qualitative variables were expressed in number and frequency for each modality. The prevalence of events (iron deficiency and anemia) was estimated (number of patients with the event/number of patients analyzed) with their 95% confidence interval (CI) [17].
A profile likelihood model was used to evaluate the association between characteristics of patients at baseline and the risk of iron deficiency.
The effect size was estimated by crude and adjusted hazard ratios (HR) and their 95% CI. The analysis was performed using SAS v9.4 (SAS Institute Inc., Cary, NC, USA).
The study protocol was approved by the ethic committee and French authorities in accordance with French legislation on non-interventional studies. The ethical approval code was reported to the Commission Nationale de l’Informatique et des Libertés (number 1404720).
Results
Characteristics of Patients
In total, 1036 patients were included (52.1% women, with a mean age of 41.8 ± 15.5 years) (Table 1).
Table 1.
Characteristics of patients depending of the forms of IBD (Crohn’s disease (Crohn) or ulcerative colitis (UC))
| Crohn N = 685 (%) | UC N = 351 (%) | Study population N = 1036 (%) | |||
|---|---|---|---|---|---|
| Sex, n(%) | |||||
| Male | 318 | (46.4) | 178 | (50.7) | 496 |
| Female | 367 | (53.6) | 173 | (49.3) | 540 |
| Age (years), n | 685 | 351 | 1036 | ||
| Mean (SD) | 40.2 | (14.9) | 44.9 | (16.0) | 41.8 |
| Median (Q1–Q3) | 38.0 | (29.0;49.0) | 43.0 | (32.0;57.0) | 39.0 |
| [Min–Max] | [18.0–95.0] | [18.0–87.0] | |||
| Age (classes), n(%) | |||||
| < 55 years | 560 | (81.8) | 247 | (70.4) | 807 |
| ≤ 55 years | 125 | (18,2) | 104 | (29,6) | 229 |
| Height (cm), n | 678 | 344 | 1022 | ||
| Mean (SD) | 169.0 | (9.0) | 170.8 | (8.9) | 169.6 |
| Median (Q1–Q3) | 169.0 | (163.0;175.0) | 170.0 | (164.0;177.0) | 170.0 |
| [Min–Max] | [148.0–192.0] | [150.0–198.0] | |||
| Weight (Kg), n | 682 | 347 | 1029 | ||
| Mean (SD) | 71.2 | (17.2) | 73.1 | (16.2) | 71.8 |
| Median (Q1–Q3) | 69.0 | (59.0;80.0) | 70.0 | (61.0;83.0) | 70.0 |
| [Min–Max] | [35.0–140.0] | [42.0–122.0] | |||
| BMI (Kg/m2), n | 678 | 343 | 1021 | ||
| Mean (SD) | 24.8 | (5.4) | 25.0 | (4.8) | 24.9 |
| Median (Q1–Q3) | 23.7 | (21.1;27.4) | 24.2 | (21.8;27.5) | 23.9 |
| [Min–Max] | [13.7–49.6] | [14.0–43.0] | |||
| BMI (classes), n (%) | |||||
| < 18,5 kg/m2 | 46 | (6.8) | 17 | (5.0) | 63 |
| 18,5–25,0 kg/m2 | 367 | (54.1) | 184 | (53.6) | 551 |
| 25,0–30,0 kg/m2 | 157 | (23.2) | 91 | (26.5) | 248 |
| > = 30 kg/m2 | 108 | (15.9) | 51 | (14.9) | 159 |
| Ongoing treatment, n (%) | 636 | (92.8) | 336 | (95.7) | 972 |
| 5-ASA, n (%) | 22 | (3.5) | 73 | (21.8) | 95 |
| Anti-TNF, n (%) | 469 | (73.7) | 153 | (45.7) | 622 |
| Anti-interleukins, n (%) | 48 | (7.5) | 6 | (1.8) | 54 |
| Anti-integrins, n (%) | 64 | (10.1) | 101 | (30.1) | 165 |
| Antibiotics n (%) | 2 | (0.3) | 1 | (0.3) | 3 |
| Steroids, n (%) | 27 | (4.2) | 20 | (6.0) | 47 |
| Immunosuppressants, n (%) | 156 | (24.5) | 78 | (23.3) | 234 |
| Other, n (%) | 7 | (1.1) | 2 | (0.6) | 9 |
UC ulcerative colitis; n number; BMI body mass index; TNF tumor necrosis factor; SD standard deviation; Q quartile;
The mean height was 169.6 ± 9.0 cm, with a mean weight of 71.8 ± 16.9 kg. More than half of the subjects had a BMI between 18.5 and 25 kg/m2 (551, 54.0%), while only a small percentage of subjects had a BMI < 18.5 (63, 6.2%).
Two-thirds of patients were diagnosed with CD (685, 66.1%) and one-third with UC (351, 33.9%). The reasons for hospital admission were IBD treatment administration (586, 56.6%), follow-up consultation (406, 39.2%), and disease flare (44, 4.2%).
Approximately half of the patients (504, 51.1%) were in disease remission at the time of enrollment. When IBD was considered as active, there was predominantly mild (294, 29.8%) or moderate (139, 14.1%) activity, while only a small percentage of subjects had severe activity (50, 5.1%).
Almost all patients (972, 93.8%) had ongoing treatment for IBD, including anti-TNF agents (622, 64.1%), immunosuppressants (234, 24.1%), vedolizumab (1165, 17.0%), 5-ASA (95, 9.8%), steroids (47, 4.8%), or other.
History of Iron Workup
Systematic monitoring of the iron balance was performed in most of the hospitals that participated in the study (12/21, 57.1%). The iron balance was investigated before admission to the hospital by most of these centers (10/12, 83.3%), during admission to the hospital in half of the cases (6, 50.0%), and after admission to the hospital only in one center (8.3%). The mean period time since the last iron status assessment was 7 ± 14.2 months (Table 2). Most patients (528, 60.8%) had such an assessment within 3 months, and only a small portion of subjects had an evaluation older than 12 months (147, 16.9%) (Table 2). The iron status evaluation included the measurement of ferritin in all cases (12/12, 100%) and the dosage of Hb (11, 91.7%), TSAT (6, 50.0%), and serum iron (5, 41.7%) in a lower percentage.
Table 2.
Timing, duration of the iron assessment, and evaluation of iron deficiency and anemia in patients with IBD (Crohn’s disease (Crohn) or ulcerative colitis (UC))
| Crohn | UC | Study Population | |||
|---|---|---|---|---|---|
| (N = 685) | (N = 351) | (N = 1036) | |||
| Number of months since the last iron evaluation, n | 580 | 288 | 868 | ||
| Mean (SD) | 6.9 | (13.6) | 7.2 | (15.3) | 7.0 |
| Median (Q1–Q3) | 2.0 | (1.0;6.0) | 2.0 | (1.0;6.0) | 2.0 |
| [Min–Max] | [0.0–117.0] | [0.0–131.0] | |||
| Number of months since the last iron evaluation (classes), n(%) | |||||
| < 3 months | 347 | (59.8) | 181 | (62.8) | 528 |
| 3–6 months | 80 | (13.8) | 32 | (11.1) | 112 |
| 6–12 months | 58 | (10.0) | 23 | (8.0) | 81 |
| > = 12 months | 95 | (16.4) | 52 | (18.1) | 147 |
| Iron evaluation: | |||||
| Serum iron, n (%) | 372 | (64.1) | 194 | (67.4) | 566 |
| Serum ferritin, n (%) | 572 | (98.6) | 282 | (97.9) | 854 |
| TSAT, n (%) | 364 | (62.8) | 192 | (66.7) | 556 |
| Serum iron (mg/L), n | 648 | 325 | 973 | ||
| Mean (SD) | 0.80 | (0.40) | 1.00 | (0.40) | 0.80 |
| Median (Q1–Q3) | 0.84 | (0.59;1.12) | 0.90 | (0.68;1.12) | 0.86 |
| [Min–Max] | [0.00–2.60] | [0.20–2.60] | |||
| Hb (g/dL), n | 670 | 343 | 1013 | ||
| Mean (SD) | 13.7 | (1.5) | 13.9 | (1.5) | 13.8 |
| Median (Q1–Q3) | 13.8 | (12.9;14.8) | 13.8 | (12.9;15.0) | 13.8 |
| [Min–Max] | [7.2–17.4] | [8.8–17.6] | |||
| Hb (classes), n | |||||
| < 8 g/dl | 1 | (0.1) | 0 | (0.0) | 1 |
| 8–10 g/dl | 10 | (1.5) | 4 | (1.2) | 14 |
| 10–12 g/dl for females and 10–13 g/dl for males | 86 | (12.8) | 48 | (14.0) | 134 |
| > 12 g/dl for females and > 13 g/dl for males | 573 | (85.5) | 291 | (84.8) | 864 |
| Serum ferritin (µg/L), n | 674 | 345 | 1019 | ||
| Mean (SD) | 139.1 | (140.9) | 131.7 | (115.7) | 136.6 |
| Median (Q1–Q3) | 97.0 | (51.0;174.0) | 100.0 | (49.0;180.0) | 98.0 |
| [Min–Max] | [4.0–992.0] | [1.8–624.0] | |||
| Serum ferritin (classes), n | |||||
| < 30 µg/L | 87 | (12.9) | 53 | (15.4) | 140 |
| 30–100 µg/L | 260 | (38.6) | 117 | (33.9) | 377 |
| ≥ 100 µg/L | 327 | (48.5) | 175 | (50.7) | 502 |
| TSAT (%), n | 651 | 329 | 980 | ||
| Mean (SD) | 25.8 | (12.6) | 26.9 | (11.2) | 26.2 |
| Median (Q1–Q3) | 24.0 | (17.0;33.0) | 26.0 | (20.0;33.0) | 25.0 |
| [Min–Max] | [1.0–92.0] | [3.0–74.0] | |||
| TSAT (classes), n | |||||
| < 16% | 141 | (21.7) | 44 | (13.4) | 185 |
| ≥ 16% | 510 | (78.3) | 285 | (86.6) | 795 |
UC ulcerative colitis; n number; Hb hemoglobin; TSAT transferrin saturation; SD standard deviation; Q quartile
About one-fifth of the patients had been treated with intravenous iron (218, 21.0%) (Table 3). The most frequently used formulations were ferric carboxymaltose (169, 77.5%), iron sucrose (44, 21.8%), and iron isomaltose (2, 1.0%). The intravenous iron dosage ranged from 100 to 9000 mg with the most frequent dosages represented by iron 1000 mg (118, 55.1%) and iron 500 mg (29, 13.6%). Instead, only a small percentage of patients had been treated with oral iron (36, 3.5%). The mean duration of oral iron treatment was 3.1 ± 2.9 months, and the daily dosage ranged from 5 to 500 mg with 80 mg (12, 42.9%), 50 mg (5, 17.9%), and 160 mg (5, 17.9%) being the most frequent dosages.
Table 3.
Iron treatment: duration, type of treatment (oral vs. intravenous iron), and dosage
| Crohn (N = 685) | UC (N = 351) | Study Population (N = 1036) | |||
|---|---|---|---|---|---|
| Oral iron, n(%) | 28 | (4.1) | 8 | (2.3) | 36 |
| Daily dosage (mg), n(%) | |||||
| 5 | 1 | (4.8) | 0 | (0.0) | 1 |
| 50 | 2 | (9.5) | 3 | (42.9) | 5 |
| 66 | 1 | (4.8) | 0 | (0.0) | 1 |
| 80 | 10 | (47.6) | 2 | (28.6) | 12 |
| 100 | 1 | (4.8) | 1 | (14.3) | 2 |
| 160 | 4 | (19.0) | 1 | (14.3) | 5 |
| 200 | 1 | (4.8) | 0 | (0.0) | 1 |
| 500 | 1 | (4.8) | 0 | (0.0) | 1 |
| Treatment duration (months), n | 22 | 5 | 27 | ||
| Mean (SD) | 2.8 | (2.5) | 4.6 | (4.4) | 3.1 |
| Median (Q1–Q3) | 3.0 | (1.0;3.0) | 3.0 | (2.0;5.0) | 3.0 |
| [Min–Max] | [1.0–12.0] | [1.0–12.0] | |||
| Intravenous iron, n (%) | 150 | (21.9) | 68 | (19.4) | 218 |
| Sucrose, n (%) | 32 | (23.0) | 12 | (19.0) | 44 |
| Ferric carboxymaltose, n (%) | 118 | (78.7) | 51 | (75.0) | 169 |
| Isomaltose, n (%) | 1 | (0.7) | 1 | (1.6) | 2 |
| Total dosage (mg), n(%) | |||||
| 100 | 5 | (3.4) | 1 | (1.5) | 6 |
| 200 | 14 | (9.4) | 3 | (4.6) | 17 |
| 300 | 9 | (6.0) | 5 | (7.7) | 14 |
| 400 | 1 | (0.7) | 0 | (0.0) | 1 |
| 500 | 21 | (14.1) | 8 | (12.3) | 29 |
| 600 | 3 | (2.0) | 2 | (3.1) | 5 |
| 700 | 0 | (0.0) | 1 | (1.5) | 1 |
| 750 | 1 | (0.7) | 0 | (0.0) | 1 |
| 800 | 1 | (0.7) | 0 | (0.0) | 1 |
| 900 | 4 | (2.7) | 2 | (3.1) | 6 |
| 1000 | 79 | (53.0) | 39 | (60.0) | 118 |
| 1500 | 4 | (2.7) | 1 | (1.5) | 5 |
| 2000 | 4 | (2.7) | 2 | (3.1) | 6 |
| 3000 | 1 | (0.7) | 0 | (0.0) | 1 |
| 4500 | 1 | (0.7) | 0 | (0.0) | 1 |
| 9000 | 1 | (0.7) | 1 | (1.5) | 2 |
UC ulcerative colitis; n number; SD standard deviation; Q quartile
Anemia and Iron Deficiency Evaluation
Anemia was found in 149/1013 patients (14.7%): mild in most patients (134/149, 89.9%) and moderate (14, 9.4%) and severe (1, 0.7%) in a limited number of people.
Most patients performed iron status monitoring during the hospitalization (823, 79.4%), while only a small proportion of subjects performed such tests in external laboratories (213, 20.6%).
The mean ferritin value was 136.6 ± 132.9 µg/L (Table 2). About half of the patients had a ferritin > 100 µg/L (502, 49.3%), while a third of the patients had a concentration between 30 and 100 µg/L (377, 37.0%) and a smaller percentage of patients had a ferritin < 30 µg/L (140, 13.7%).
The mean serum iron value was 80 ± 0.4 mg/L, while TSAT concentrations < 16% were reported only in a small group (185, 18.9%).
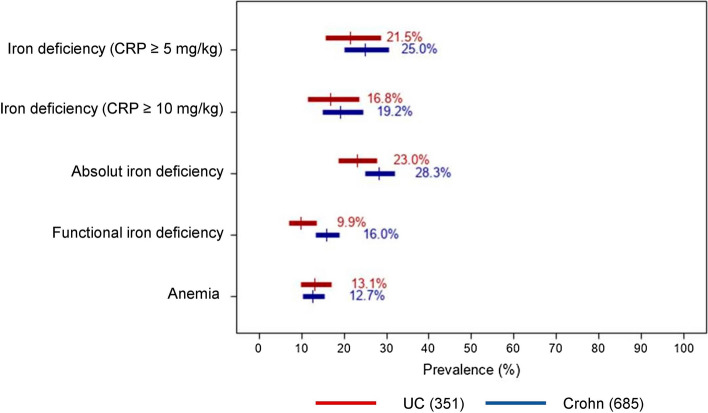
Iron deficiency occurred in a quarter of patients (97/409, 23.7% CI 95% 19.8–28.1) (Table 4). Of note, absolute iron deficiency was experienced by 261 patients (26.5%, CI 95% 23.9–29.4), while functional iron deficiency was in a smaller group (139, 13.9% CI 95% 11.9–16.2) (Fig. 1).
Table 4.
Prevalence of iron deficiency (absolute and functional iron deficiency) and anemia
| Crohn | UC | Study population | ||||
|---|---|---|---|---|---|---|
| (N = 685) | (N = 351) | (N = 1036) | ||||
| Iron deficiency | ||||||
| (Serum ferritin < 30 µg/L if CRP < 5 mg/L, or ≤ 100 µg/L, if CRP ≥ 5 mg/L), n (%) [CI] | 65a | (25.0) [20.1–30.6] | 32b | (21.5) [15.6–28.8] | 97c | (23.7) [19.8–28.1] |
| Iron deficiency | ||||||
| (Serum ferritin < 30 µg/L if CRP < 10 mg/L, or ≤ 100 µg/L, if CRP ≥ 10 mg/L), n (%) [CI] | 50 a | (19.2) [14.9–24.5] | 25b | (16.8) [11.6–23.7] | 75c | (18.3) [14.9–22.4] |
| Absolute iron deficiency | ||||||
| (Serum ferritin < 30 µg/L or TSAT < 16%), n (%) [CI] | 185 | (28.3) [25.0–31.9] | 76 | (23.0) [18.8–27.9] | 261 | (26.5) [23.9–29.4] |
| Functional iron deficiency | ||||||
| (Serum ferritin < 100 µg/L and TSAT < 16%), n (%) [CI] | 106 | (16.0) [13.4–19.0] | 33 | (9.9) [7.1–13.6] | 139 | (13.9) [11.9–16.2] |
| Anemia, n (%) [IC]* | 85 | (12.7) [10.4–15.4] | 45 | (13.1) [9.9–17.1] | 130 | (12.8) [10.9–15.0] |
UC ulcerative colitis; n number; CRP C reactive protein; TSAT transferrin saturation; IC confidence interval
aCRP levels were reported for 260 patients
bCRP levels were reported for 149 patients
cCRP levels were reported for 409 patients
Fig. 1.
Prevalence of iron deficiency and anemia by type of inflammatory bowel disease
Factors Associated with Iron Deficiency
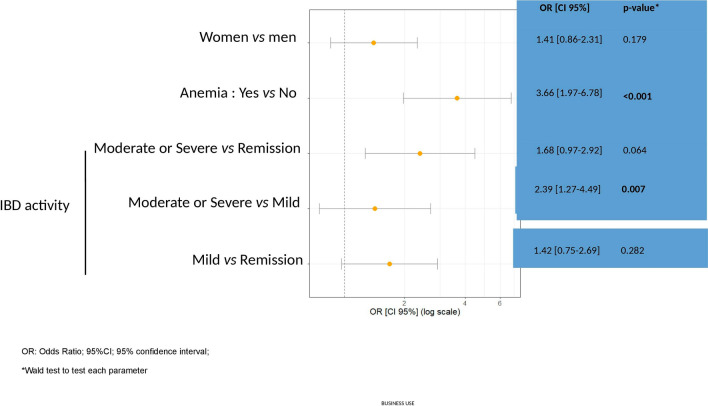
Several baseline patient characteristics were investigated to identify risk factors for iron deficiency. Of note, patients with moderate or severe IBD activity (OR: 3.66; CI 95% 24.4–61.2; p = 0.007) or concomitant anemia (OR: 3.66; CI 95% 1.97–6.78; p < 0.001) had an increased likelihood of having ID (Fig. 2).
Fig. 2.
ID diagnosis is increased in the presence of anemia and “moderate” or “severe” status of IBD
Discussion
In this prospective multicenter cross-sectional study, we evaluated the prevalence of iron deficiency in over 1000 patients with IBD. Iron deficiency was detected in about a quarter of patients, and its prevalence was higher in women and in subjects with severe IBD activity or concomitant anemia.
These data are in line with those of a Spanish observational cohort study which reported the presence of iron deficiency in about one-third of IBD outpatients with a higher risk for female patients and those with severe disease activity [18]. These data can probably be explained by menstrual losses in premenopausal women and by the increased risk of bloody diarrhea in patients with active disease, suggesting greater attention in this specific setting [19].
The iron deficiency diagnosis was based on serum ferritin levels although serum iron, TSAT, and Hb were also monitored. Ferritin levels can be increased in case of inflammation causing false-negative results [20]. For this reason, we have also investigated CRP levels to recognize patients with inflammation and to distinguish absolute versus functional iron deficiency. The serum iron concentration is reduced in patients with iron deficiency but it is also reduced in the case of inflammation, thus not being sufficient for the diagnosis of iron deficiency in patients with IBD [19].
Low Hb concentrations reflect the presence of concomitant anemia, while the measurement of the TSAT allows to distinguish the functional iron deficiencies from the absolute ones and should be routinely monitored [19]. No patient was subjected to the hepcidin dosage, a liver hormone, which regulates iron homeostasis and showed to be a valid marker of iron deficiency [21]. Interestingly, iron balance monitoring was performed within 3 months or no more than one year in most patients, with the exception of a limited group of subjects, as recommended by the ECCO, indicating acceptable adherence to international recommendations [9].
We also looked at the therapies used to supplement iron deficiency. About a quarter of the patients had been treated or had ongoing iron treatment. Importantly, most of the subjects had been treated with intravenous iron compared with oral iron (21.0% vs. 3.5%). The higher use of the intravenous formulation is probably due to the proportion of patients who were hospitalized following a disease flare or had concomitant anemia. It should also be noted that intravenous iron supplementation has been associated with greater efficacy and a lower rate of adverse events, resulting in better tolerance by patients [22]. The most frequent intravenous treatment used was ferric carboxymaltose. A recent Cochrane systematic review including 11 randomized clinical trials evaluated the efficacy and safety of the interventions for the treatment of iron deficiency anemia in patients with IBD [23]. Intravenous iron was associated with a greater response compared with oral iron, and intravenous ferric carboxymaltose probably led to a higher resolution rate of iron deficiency anemia than intravenous iron sucrose [23]. Another study by Stein et al. compared the long-term outcomes and costs of oral and intravenous iron supplementation in patients with IBD [12]. Interestingly, patients treated with intravenous iron formulations had a lower risk of hospitalization compared with the oral iron group (37% vs. 48%, p = 0.0019). Furthermore, although the absolute cost of intravenous treatment was higher than oral iron, inpatient costs were significantly lower for patients treated with intravenous iron. It should be emphasized that the iron administration route is a very hot topic and the development of new oral formulations is fueling the debate on the matter. Ferric maltol is a new oral formulation that allows greater iron bioavailability and less mucosal damage making it easier to tolerate [24] but is not available in France.
Iron supplementation therapy should be individualized based on patient characteristics. On the one hand, the intravenous formulation should be preferred in patients with severe anemia, active disease, or intolerant to oral iron. On the other hand, oral iron is a viable option in those with disease remission and who are not anemic. Of note, the European Society for Clinical Nutrition and Metabolism (ESPEN) guidelines recommend iron supplementation in all patients with IBD with iron deficiency anemia in order to normalize Hb levels and iron stores [25]. Based on these recommendations, oral iron should be the first choice for the treatment of patients with mild anemia, inactive disease, and those who are not intolerant to oral iron [25]. Intravenous iron should be considered as first-line treatment in patients with clinically active IBD, previous intolerance to oral iron, Hb < 100 g/L, and in subjects who need erythropoiesis-stimulating agents [25].
To the best of our knowledge, this is the largest study specifically designed to investigate the prevalence of iron deficiency in patients with IBD. There are other strengths of our study. Firstly, iron deficiency was defined according to the recommendations of ECCO. Secondly, several French centers participated in the enrollment of patients making our data reliable. However, there are also limitations that need to be addressed. There are no data on patient comorbidities (e.g., heart disease, diverticulosis, or gynecological disorders) that may have affected ferritin levels. There is no sufficient follow-up to establish long-term effects of iron deficiency and its treatment on disease outcomes. In the near future, it will be increasingly necessary to create international registries in order to collect as much data and define the optimal management of patients. To overcome this problem, a European online registry for clinical care and research in IBD, called United Registries for Clinical Assessment and Research (UR-CARE), has been created and will provide essential data to improve our knowledge about IBD.
In conclusion, about one quarter of patients with IBD are iron deficient. In particular, patients with concomitant anemia or moderate/severe disease activity are at increased risk of iron deficiency. Early detection and management of iron deficiency is recommended in patients with IBD to prevent the occurrence of poor disease outcomes.
Collaborators
MACAIGNE Gilles (CH De Marne La Vallée Site Jossigny, France); AMIL Morgane (CH La Roche S/Yon, France); AMIOT Aurélien (Hôpital Henri-Mondor, AP-HP, Créteil, France); LOCHER Christophe, (CH Meaux, Meaux, France): KAASSIS Mehdi (CH Cholet, Cholet, France); NAHON Stéphane (GHI Le Raincy Montfermeil, Montfermeil, France); BENAMOUZIG Robert (Hôpital Avicenne, APHP, France); ALTWEGG Romain (CHU Montpellier, Montpellier, France); HELUWAERT Frederic (CH Annecy, Annecy, France); CLAUDE Pierre (CH Mulhouse, Mulhouse, France); ANDRAU Pierre (CH Tarbes, Tarbes, France); VEYRARD Pauline (CHU Saint-Etienne, Saint-Etienne, France); DIB Nina (CHU Angers, Angers, France); ANDRIEU Valérie (Hôpital Bichat Claude Bernard, Paris, France); CHOUKROUN Gabriel (CHU Amiens, Site Sud, Amiens, France); COHEN-SOLAL Alain (Hôpital Lariboisière, Paris, France); LUPORSI Elisabeth (CHR-Metz- Thionville - Hôpital De Mercy, Metz, France); PEOC’H Katell (Hôpital Beaujon, Clichy, France)
Author’s Contribution
LPB, JNT, and PC conceived the study. LPB wrote the first draft of the paper and created tables and figure. GB, DL, GS, CG, CM, JNT, and PC critically reviewed the article and supervised the project. All authors contributed and approved the final version of the manuscript.
Declarations
Conflict of interest
L Peyrin-Biroulet reports personal fees from Galapagos, AbbVie, Janssen, Genentech, Ferring, Tillots, Celltrion, Takeda, Pfizer, Index Pharmaceuticals, Sandoz, Celgene, Biogen, Samsung Bioepis, Inotrem, Allergan, MSD, Roche, Arena, Gilead, Amgen, BMS, Vifor, Norgine, Mylan, Lilly, Fresenius Kabi, OSE Immunotherapeutics, Enthera, Theravance, Pandion Therapeutics, Gossamer Bio, Viatris, and Thermo Fisher; -Grants from Abbvie, MSD, Takeda, and Fresenius Kabi; and stock options from CTMA. G Bouguen received lecture fees from Abbvie, Ferring, MSD, Takeda, and Pfizer and consultant fees from Takeda and Janssen. D Laharie declares counseling, boards, transports or fees from Abbvie, Biogaran, Biogen, Ferring, HAC Pharma, Janssen, MSD, Novartis, Pfizer, Prometheus, Roche, Takeda, Theradiag and Tillots. G Savoye has received lecture fees from Vifor Pharma, Takeda, Pfizer, HAC Pharma, Abbvie, MSD, and Ferring France. This author has also received travel accommodations from Ferring, Abbvie, and MSD France as well as a research grant from Ferring. C Giletta has received lecture/consulting fees from Abbvie, Takeda, Pfizer, Celltrion, and Janssen. C Michiels received lecture fees from MSD and Abbvie and invitations to congresses form MSD, Abbvie and Ferring. M Fumery received lecture and consultant fees form Abbvie, MSD, Takeda, Janssen, Celgene, Boehringer, Biogen, Amgen, Celltrion, Pfizer, Galapagos and Lilly. A. Buisson disclosed consulting fees for Abbvie, Amgen, Arena, Biogen, Celltrion, CTMA, Ferring, Janssen, MSD, Nexbiome, Pfizer, Roche, Takeda, and Tillots, as well as lecture fees for Abbvie, Amgen, Biogen, Janssen, Mayoly-Spindler, MSD, Norgine Pfizer, Roche, Takeda, Tillots, and Vifor Pharma. P. Cacoub received consultancies and honoraria from Vifor Pharma.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Contributor Information
Laurent Peyrin-Biroulet, Email: peyrinbiroulet@gmail.com.
Patrice Cacoub, Email: patrice.cacoub@aphp.fr.
The CARENFER study group:
Gilles Macaigne, Morgane Amil, Aurélien Amiot, Christophe Locher, Mehdi Kaassis, Stéphane Nahon, Robert Benamouzig, Romain Altwegg, Frederic Heluwaert, Pierre Claude, Pierre Andrau, Pauline Veyrard, Nina Dib, Valérie Andrieu, Gabriel Choukroun, Alain Cohen-solal, Elisabeth Luporsi, and Katell Peoc’h
References
- 1.Lopez A, Cacoub P, Macdougall IC, Peyrin-Biroulet L. Iron deficiency anaemia. Lancet. 2016;387:907–916. doi: 10.1016/S0140-6736(15)60865-0. [DOI] [PubMed] [Google Scholar]
- 2.Parra RS, Feitosa MR, da Ferreira SC, Rocha JJRD, Troncon LEA, FÉres O. Anemia and iron deficiency in inflammatory bowel disease patients in a referral center in Brazil: prevalence and risk factors. Arq Gastroenterol. 2020;57:272–277. doi: 10.1590/s0004-2803.202000000-51. [DOI] [PubMed] [Google Scholar]
- 3.Stein J, Hartmann F, Dignass AU. Diagnosis and management of iron deficiency anemia in patients with IBD. Nat Rev Gastroenterol Hepatol. 2010;7:599–610. doi: 10.1038/nrgastro.2010.151. [DOI] [PubMed] [Google Scholar]
- 4.Gasche C, Lomer MCE, Cavill I, Weiss G. Iron, anaemia, and inflammatory bowel diseases. Gut. 2004;53:1190–1197. doi: 10.1136/gut.2003.035758. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Evstatiev R, Gasche C. Iron sensing and signalling. Gut. 2012;61:933–952. doi: 10.1136/gut.2010.214312. [DOI] [PubMed] [Google Scholar]
- 6.Cappellini MD, Musallam KM, Taher AT. Iron deficiency anaemia revisited. J Intern Med. 2020;287:153–170. doi: 10.1111/joim.13004. [DOI] [PubMed] [Google Scholar]
- 7.Cacoub P, Nicolas G, Peoc’h K. Iron deficiency markers in patients undergoing iron replacement therapy: a 9-year retrospective real-world evidence study using healthcare databases. Sci Rep. 2020;10:14983. doi: 10.1038/s41598-020-72057-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Peyrin-Biroulet L, Lopez A, Cummings JRF, Dignass A, Detlie TE, Danese S. Review article: treating-to-target for inflammatory bowel disease-associated anaemia. Aliment Pharmacol Ther. 2018;48:610–617. doi: 10.1111/apt.14922. [DOI] [PubMed] [Google Scholar]
- 9.Dignass AU, Gasche C, Bettenworth D, et al. European consensus on the diagnosis and management of iron deficiency and anaemia in inflammatory bowel diseases. J Crohns Colitis. 2015;9:211–222. doi: 10.1093/ecco-jcc/jju009. [DOI] [PubMed] [Google Scholar]
- 10.Dignass A, Farrag K, Stein J. Limitations of serum ferritin in diagnosing iron deficiency in inflammatory conditions. Int J Chronic Dis. 2018;2018:9394060. doi: 10.1155/2018/9394060. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Hou JK, Gasche C, Drazin NZ, et al. Assessment of gaps in care and the development of a care pathway for anemia in patients with inflammatory bowel diseases. Inflamm Bowel Dis. 2017;23:35–43. doi: 10.1097/MIB.0000000000000953. [DOI] [PubMed] [Google Scholar]
- 12.Stein J, Haas JS, Ong SH, et al. Oral versus intravenous iron therapy in patients with inflammatory bowel disease and iron deficiency with and without anemia in Germany - a real-world evidence analysis. Clinicoecon Outcomes Res. 2018;10:93–103. doi: 10.2147/CEOR.S150900. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Gasche C, Berstad A, Befrits R, et al. Guidelines on the diagnosis and management of iron deficiency and anemia in inflammatory bowel diseases. Inflamm Bowel Dis. 2007;13:1545–1553. doi: 10.1002/ibd.20285. [DOI] [PubMed] [Google Scholar]
- 14.https://www.has-sante.fr/jcms/c_1051506/fr/choix-des-examens-du-metabolisme-du-fer-en-cas-de-suspicion-de-carence-en-fer-rapport-d-evaluation; 2022 Accessed 15.02.22
- 15.https://www.who.int/vmnis/indicators/haemoglobin.pdf; 2022 Accessed 15.02.22
- 16.Silverberg MS, Satsangi J, Ahmad T, et al. Toward an integrated clinical, molecular and serological classification of inflammatory bowel disease: report of a Working Party of the 2005 Montreal World Congress of Gastroenterology. Can J Gastroenterol. 2005;19:5–36. doi: 10.1155/2005/269076. [DOI] [PubMed] [Google Scholar]
- 17.Turlot J-C, Petiot JF. Intervalles de confiance pour une proportion: lesquels doit-on enseigner ? arXiv:150101136 [math, stat] 2015. Available at: http://arxiv.org/abs/1501.01136 Accessed from 19 Jun 2021.
- 18.González Alayón C, Pedrajas Crespo C, Marín Pedrosa S, et al. Prevalence of iron deficiency without anaemia in inflammatory bowel disease and impact on health-related quality of life. Gastroenterol Hepatol. 2018;41:22–29. doi: 10.1016/j.gastrohep.2017.07.011. [DOI] [PubMed] [Google Scholar]
- 19.Pasricha S-R, Tye-Din J, Muckenthaler MU, Swinkels DW. Iron deficiency. Lancet. 2021;397:233–248. doi: 10.1016/S0140-6736(20)32594-0. [DOI] [PubMed] [Google Scholar]
- 20.Gomollón F, Gisbert JP. Current management of iron deficiency anemia in inflammatory bowel diseases: a practical guide. Drugs. 2013;73:1761–1770. doi: 10.1007/s40265-013-0131-2. [DOI] [PubMed] [Google Scholar]
- 21.Stojkovic Lalosevic M, Toncev L, Stankovic S, et al. Hepcidin is a reliable marker of iron deficiency anemia in newly diagnosed patients with inflammatory bowel disease. Dis Markers. 2020;2020:8523205. doi: 10.1155/2020/8523205. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Bonovas S, Fiorino G, Allocca M, et al. Intravenous versus oral iron for the treatment of anemia in inflammatory bowel disease: a systematic review and meta-analysis of randomized controlled trials. Medicine (Baltimore). 2016;95:e2308. doi: 10.1097/MD.0000000000002308. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Gordon M, Sinopoulou V, Iheozor-Ejiofor Z, et al. Interventions for treating iron deficiency anaemia in inflammatory bowel disease. Cochrane Database Syst Rev. 2021;1:CD013529. doi: 10.1002/14651858.CD013529.pub2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.D’Amico F, Peyrin-Biroulet L, Danese S. Oral iron for patients with IBD: lessons learned at time of COVID-19 pandemic. J Clin Med. 2020;9:E1536. doi: 10.3390/jcm9051536. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Forbes A, Escher J, Hébuterne X, et al. ESPEN guideline: clinical nutrition in inflammatory bowel disease. Clin Nutr. 2017;36:321–347. doi: 10.1016/j.clnu.2016.12.027. [DOI] [PubMed] [Google Scholar]