Abstract
AtDjC5 belongs to the J-protein family in Arabidopsis thaliana. Its biological functions remain unclear. In this study, we examined the roles of AtDjC5 in resisting heat stress using reverse genetic analysis. After the seedlings were exposed directly to 44 °C for 90 min, AtDjC5 knockout seedlings displayed decreases in the survival rate, membrane system stability, and cell vitality compared to WT seedlings, indicating that AtDjC5 is involved in plant basal thermotolerance. The AtDjC5 knockout seedlings pre-exposed to 37 °C for 30 min exhibited decreases in the survival rate and total chlorophyll contents and increased cell death when they were subsequently exposed to 45 °C compared to the WT seedlings, indicating that AtDjC5 plays an important role in plant acquired thermotolerance. AtDjC5 was found to localize to the endoplasmic reticulum. The expression of the AtDjC5 gene was induced by heat and TM (an ER stress inducer) treatment. Furthermore, we found that the knockout of AtDjC5 inhibited ER stress-induced autophagy and the expression of ER stress-related genes. Taken together, these results suggest that AtDjC5 facilitates thermotolerance, likely by aiding in the ER stress response.
Keywords: AtDjC5, thermotolerance, ER stress response
1. Introduction
A higher growing season temperature has great impacts on agricultural production, farm income, and food security [1,2,3]. High-temperature stress disturbs cellular protein homeostasis, leading to protein denaturation or aggregation [4,5,6]. Thermotolerance is an important component of organisms to adapt to high-temperature stress and is divided into basal and acquired thermotolerance [7,8]. Plants pre-exposed to a nonlethal high-temperature condition for a certain period of time exhibit an evidently increased tolerance to lethal high temperature. This is called acquired thermotolerance. The best-characterized aspect of acquired thermotolerance is the production of heat-shock proteins (HSPs), which are regulated by heat shock transcription factors [9,10,11,12]. HSPs function as molecular chaperones to allow cellular proteins to avoid and/or recover from stress-induced protein aggregation [13,14,15,16].
The accumulation of unfolded and/or misfolded proteins in the endoplasmic reticulum (ER) generates a condition called ER stress, which may activate the unfolded protein response (UPR), also called the ER stress response. The UPR is a stress mitigation mechanism that monitors protein folding in the cellular secretory pathway [17,18,19]. To alleviate ER stress, the UPR is activated to produce molecular chaperones or HSPs to aid in protein folding [20,21]. The molecular mechanism of the UPR in plant cells is much less well-understood than those in yeast and animal cells. In Arabidopsis thaliana, bZIP28 and bZIP60 of the basic domain/leucine zipper (bZIP) family have been identified as ER stress-activated membrane-related transcription factors [22,23]. bZIP60 mRNA is spliced by inositol-requiring protein 1 (IRE1) in response to ER stress, and the resulting mRNA encodes a nuclear targeted bZIP60, which activates the expression of UPR genes in the nucleus (IRE1-bZIP60 pathway) [24]. The activation of bZIP28 by unfolded proteins requires ER-localized molecular chaperones, BiPs (ER-localized HSP70s) [25]. Under nonstress conditions, BiPs bind to the lumen-facing domain of bZIP28. In response to ER stress, bZIP28 is released because BiPs bind to the unfolded protein accumulated in the ER, moves from the ER to the Golgi, and is then subjected to proteolysis by Golgi-resident proteases, which release its cytoplasmic domain, which relocates to the nucleus to activate the expression of UPR genes encoding chaperones, protein folding, and protein degradation factors such as BiPs (BiP-bZIP28 pathway) [23,24,26]. There are three BiP coding genes in A. thaliana (BiP1, BiP2, and BiP3), and BiP3, in particular, is the most highly upregulated by ER stress agents such as tunicamycin (TM) and dithiothreitol (DTT), two potent inducers of ER stress [27,28]. In addition, autophagy eliminates misfolded proteins by disposing of large protein aggregates and whole organelles through the formation of an autophagosome [29,30,31]. Plant autophagy has been shown to be involved in senescence, nutrient deprivation, oxidative stress, salt and drought stresses, and pathogen infection [32,33,34,35,36,37,38].
J-proteins (also known as Hsp40s) are defined by a J-domain consisting of approximately 75 conserved amino acid residues. They are located in various subcellular compartments and act as molecular chaperones, either alone or in association with HSP70s. Hsp70–J-protein chaperone machines play active roles in protein homeostasis by transiently binding to many different polypeptide substrates, restoring the native state of misfolded polypeptides trapped in aggregates [39,40]. One hundred and twenty J-proteins have been identified in the Arabidopsis genome [41,42]. J-proteins have been reported to regulate plant growth and development and to participate in the adaptation to various environmental stresses [43,44,45,46,47,48,49,50,51,52,53,54]. AtDjC5 is a member of the Arabidopsis J-protein family. The open reading frame of AtDjC5 encodes a protein consisting of 113 amino acid residues. The roles of AtDjC5 in regulating the growth and development of plants and resistance to environmental stresses have not been reported. Based on our previous large-scale phenotype screening for the J-protein family with heat stress treatment, AtDjC5 mutants possessed the heat sensitive phenotype. Therefore, in this work, we used reverse genetic analysis to define the roles of AtDjC5 in resisting heat stress in A. thaliana.
2. Results
2.1. Knockout of the AtDjC5 Gene Decreased Basal Thermotolerance
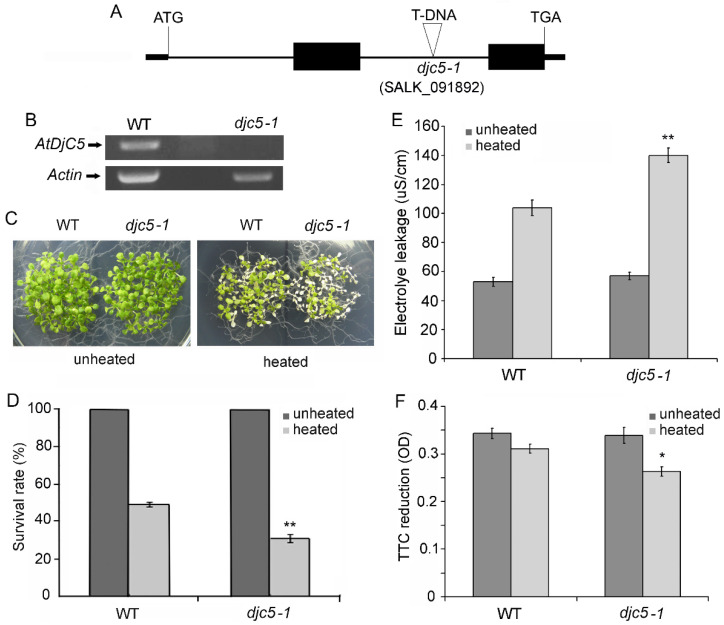
To understand the role of AtDjC5 in adapting to heat stress, we identified a homozygous T-DNA insertion mutant for the AtDjC5 gene, djc5-1. Sequence analysis of the T-DNA flanking regions revealed that djc5-1 contained a T-DNA insertion in intron 2 of AtDjC5 (Figure 1A). RT–PCR showed no detectable full AtDjC5 transcript in the djc5-1 plants (Figure 1B). To assess the effect of AtDjC5 knockout on plant basal thermotolerance, the survival rates were compared between the WT and djc5-1 plants after the seedlings were exposed directly to 44 °C for 90 min. The survival rate of the djc5-1 seedlings was not different from that of the WT seedlings under normal growth conditions. However, the djc5-1 seedlings exhibited hypersensitivity after heat shock (HS). While 49% of the WT seedlings were alive after HS, the survival rate after HS was 31% for the djc5-1 seedlings (Figure 1C,D).
Figure 1.
AtDjC5 knockout decreased the basal thermotolerance. WT, wild-type plants; djc5-1, a T-DNA insertion mutant line for the AtDjC5 gene; unheated, under normal conditions; heated, treated at 44 °C for 90 min. (A) Intron/exon organization of the AtDjC5 coding region and T-DNA insertion location. Solid boxes, exons; lines, introns; triangles, T-DNA insertion position. (B) RT–PCR analysis of the AtDjC5 full transcript in WT and djc5-1 plants. (C) Comparison of WT and djc5-1 seedling viability. (D) Survival rates of WT and atdj5c-1 seedlings. Each value is the mean ± SD of ten biological replicates. (E) Electrolyte leakage assay for the WT and djc5-1 leaves. The data are the means ± SDs of three independent experiments. (F) 2,3,5-triphenyltetrazolium chloride (TTC) reduction activity of roots for WT and djc5-1. The data are the means ± SDs of four biological replicates, with a total of 30 roots per sample. Asterisks indicate significant differences from the WT of the same treatment (t test, * p < 0.05, ** p < 0.01).
As an additional test of heat sensitivity, the electrolyte leakage level was compared between the WT and djc5-1 leaves. Under non-HS conditions, there were no significant differences in the ion conductivity of the incubation medium between the WT and djc5-1 leaves (Figure 1E). After HS at 45 °C for 120 min, the ion conductivity of the incubation medium was markedly increased in WT and djc5-1, and the ion conductivity in djc5-1 was distinctly higher than that in WT (Figure 1E). The results indicated that AtDjC5 is important for the stability of the membrane system during HS.
To further confirm the role of AtDjC5 in plant thermotolerance, we compared the difference in cell vitality between the WT and djc5-1 roots. Under non-HS conditions, the TTC reduction activity in the djc5-1 roots was not obviously different from that in the WT roots; however, after HS at 45 °C for 1 h, the TTC reduction activity in the WT and djc5-1 roots was markedly decreased, and the TTC reduction activity in djc5-1 was markedly lower than that in WT (Figure 1F). HS prevented the reduction in TTC much more in atdjc5-1 than in WT. Taken together, our data suggest that AtDjC5 knockout accelerated HS-induced cell death and improved the basal thermotolerance of plants.
2.2. AtDjC5 Is Involved in the Regulation of Acquired Thermotolerance
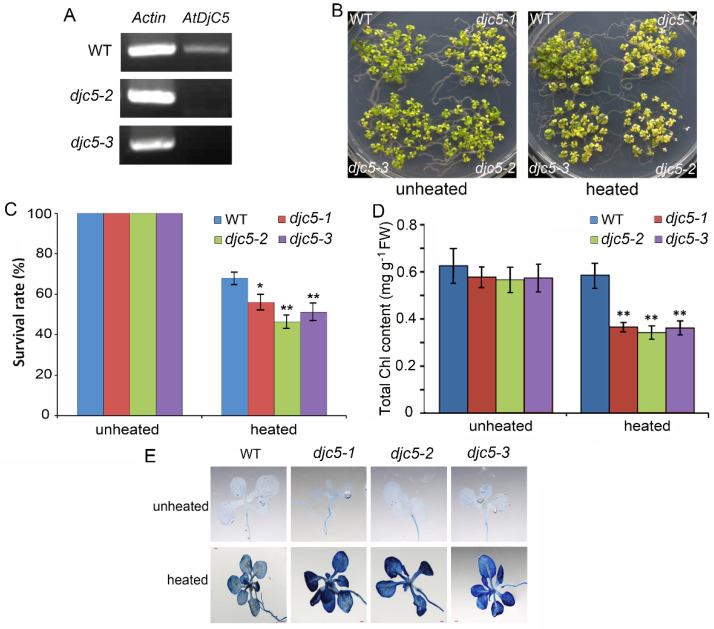
To determine the role of AtDjC5 in plant acquired thermotolerance, we generated two other AtDjC5 mutant lines (djc5-2 and djc5-3) through the artificial microRNA method. RT–PCR showed no detectable full AtDjC5 transcript in the djc5-2 and djc5-3 seedlings (Figure 2A). The 11-day-old seedlings grown at 22 °C were acclimated at 37 °C for 30 min and then returned to 22 °C for 120 min before challenge at 45 °C for 120 min. The survival rate of seedlings after HS was compared among WT, djc5-1, djc5-2, and djc5-3. The results showed that these three AtDjC5 mutant seedlings were hypersensitive to HS. While 68% of WT seedlings were alive after HS, the survival rates after HS were 56% for djc5-1, 46% for djc5-2, and 51% for djc5-3 (Figure 2B,C). The decrease in acquired thermotolerance in the AtDjC5 mutants was confirmed by testing the total chlorophyll contents of seedlings. Before HS, the total chlorophyll contents in the three AtDjC5 mutant lines were not obviously different from those in WT; however, the total chlorophyll contents in the three AtDjC5 mutant lines were markedly lower than those in WT after HS (Figure 2D). As an additional test of heat sensitivity, heat-induced cell death was assayed in WT, djc5-1, djc5-2, and djc5-3 by trypan blue staining. The three AtDjC5 mutant lines exhibited increased cell death after HS compared to WT (Figure 2E).
Figure 2.
AtDjC5 knockout decreased the acquired thermotolerance. WT, wild-type plants; djc5-1, a T-DNA insertion mutant line for AtDjC5 gene; djc5-2 and djc5-3, two AtDjC5 mutant lines by artificial microRNA; unheated, under normal conditions; heated, treated at 37 °C for 30 min, then 22 °C for 120 min, followed by 45 °C for 120 min. (A) RT–PCR analysis of the AtDjC5 full transcript in WT, djc5-2, and djc5-3 plants. (B) Comparison of the WT and AtDjC5 mutant seedling viability. (C) Survival rates of WT and AtDjC5 mutant seedlings. Each value is the mean ± SD of ten biological replicates. (D) Total chlorophyll contents in WT, djc5-1, djc5-2, and djc5-3 leaves. The data are the means ± SDs of four biological replicates. (E) Analysis of cell death in the WT and three AtDjC5 mutant lines before and after heat stress by trypan blue staining. Asterisks indicate significant differences from the WT of the same treatment (t test, * p < 0.05, ** p < 0.01).
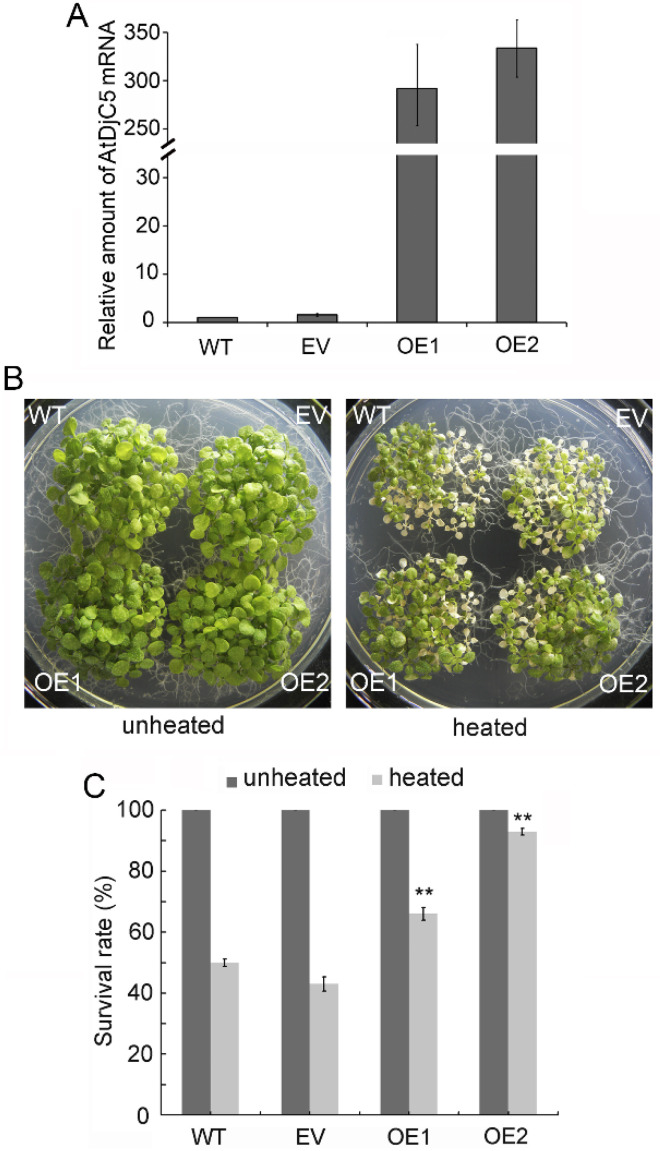
Furthermore, we examined the effect of AtDjC5 overexpression on plant acquired thermotolerance. A binary vector containing the AtDjC5 coding region (pCAMBIA1300-35S::AtDjC5) or an empty vector (pCAMBIA1300-35S) was transformed into Col plants. PCR assays confirmed that the AtDjC5 gene was introduced into all AtDjC5 transgenic lines obtained. Q-PCR assays showed that the levels of AtDjC5 mRNA were strongly increased in the OE1 and OE2 overexpression lines compared to the WT and transgenic lines containing an empty vector (EV) (Figure 3A). The WT, EV, and AtDjC5 overexpression seedlings grown at 22 °C were acclimated at 37 °C for 30 min and then returned to 22 °C for 120 min before challenge at 45 °C for 120 min. While 50% of the WT seedlings were alive after HS, the survival rates after HS were 44% for EV, 66% for OE1, and 93% for OE2 (Figure 3B,C), indicating that AtDjC5 overexpression increased the acquired thermotolerance. Taken together, our data suggest a role for AtDjC5 in improving the acquired thermotolerance of A. thaliana seedlings.
Figure 3.
AtDjC5 overexpression increased the acquired thermotolerance. WT, wild-type plants; EV, a transgenic line containing empty vector pCAMBIA1300-35S; OE1 and OE2, two AtDjC5 overexpression lines. (A) Analysis of AtDjC5 transcript levels through real-time quantitative RT–PCR (Q-PCR). The AtDjC5 transcript level in the WT sample was used as the calibrator and was set to 1. Data are the means ± SDs of three biological replicates. (B) Comparison of WT and AtDjC5-overexpressing seedling viability. (C) Survival rates of WT and AtDjC5 overexpression seedlings. Each value is the mean ± SD of ten biological replicates. Asterisks indicate significant differences from WT of the same treatment (t test, ** p < 0.01).
2.3. Expression Pattern and Subcellular Localization of AtDjC5
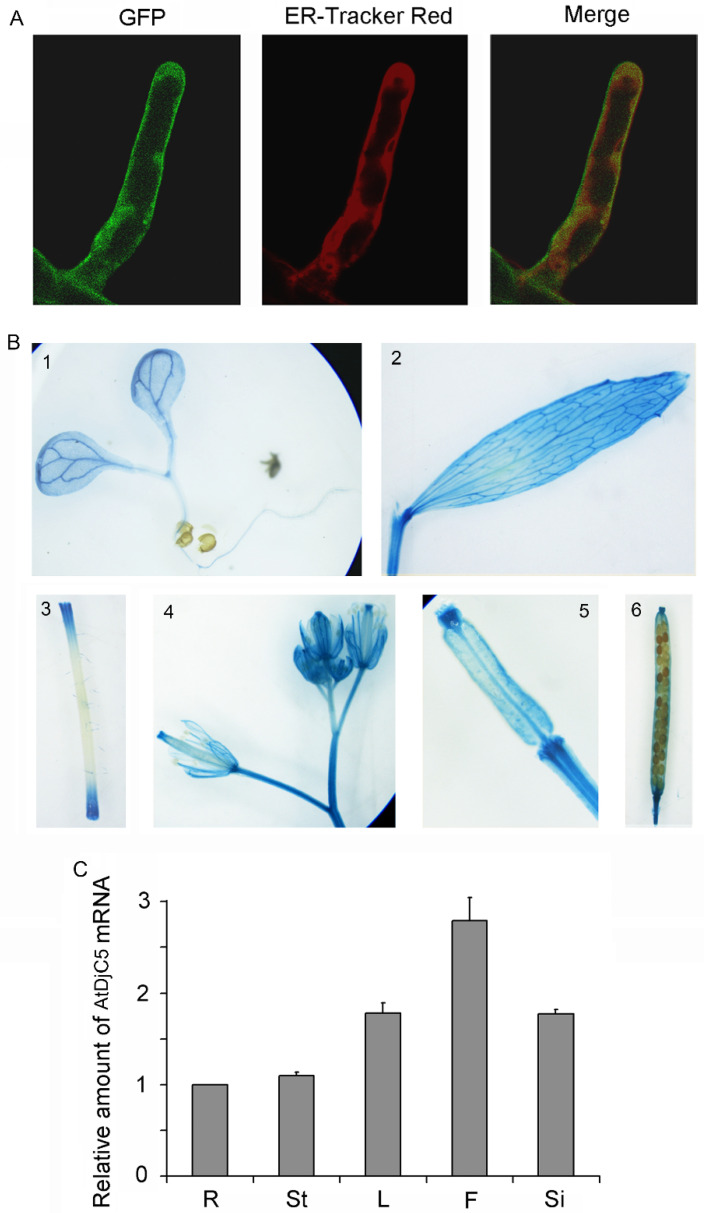
Bioinformatics analysis showed that AtDjC5 may exist in the cytosol, endoplasmic reticulum (ER), Golgi apparatus, and other cellular compartments (http://utoronto.ca/cell_efp/cgi-bin/cell_efp.cgi). We examined the subcellular localization of AtDjC5 using transgenic plants stably expressing 35S::AtDjC5-sGFP. ER-Tracker Red, an ER-specific fluorescent dye, was used to highlight the ER in the cells of root hairs. Merged images of GFP fluorescence (green) and ER-Tracker Red fluorescence (red) showed bright yellow fluorescence, indicating that the AtDjC5-sGFP fusion protein was partially present in the ER (Figure 4A).
Figure 4.
Spatiotemporal expression pattern and subcellular localization of AtDjC5 in Arabidopsis thaliana. (A) Analysis of AtDjC5 subcellular localization in the root hair cells. Transgenic Arabidopsis seedlings harboring P35S::AtDjC5-sGFP were stained with ER-Tracker Red. GFP (green) and ER-Tracker Red (red) fluorescence in the root hair cells was observed using a confocal laser microscope. In the photomicrographs on the right, a merge of the two images is presented, and yellow fluorescence was visible in the cells. (B) Transgenic plants harboring PAtDjC5::GUS were histochemically stained. GUS staining in 1, 6-day-old seedling; 2, cauline leaf; 3, stem; 4, anthotaxy; 5, young silique; 6, mature silique. (C) Tissue-specific expression of the AtDjC5 gene by Q-PCR. The expression level in roots was set to 1 and used for normalization. Data are the means ± SDs of three biological replicates. R, roots; St, stems; L, leaves; F, flowers; Si, siliques.
The spatiotemporal expression pattern of the AtDjC5 gene was examined using two approaches. First, the GUS reporter assay was used to monitor the activity of the promoter of the AtDjC5 gene. The results obtained from transgenic plants harboring PAtDjC5::GUS showed that GUS under the AtDjC5 promoter was expressed in cotyledons, roots, stems, cauline leaves, flowers, and young siliques (Figure 4B). Then, Q-PCR was used to compare the expression levels of AtDjC5 in different tissues. AtDjC5 was expressed to different degrees in all of the tissues analyzed, consistent with the results obtained by the GUS reporter assay (Figure 4C).
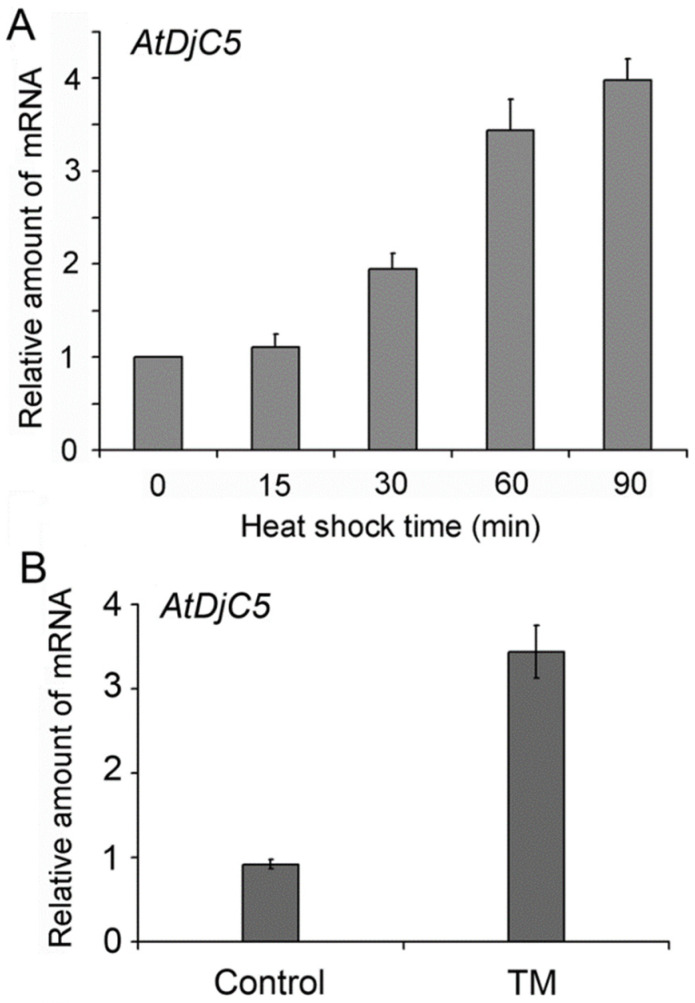
Furthermore, we investigated the effect of heat shock treatment on the expression of the AtDjC5 gene using Q-PCR. Ten-day-old seedlings grown at 22 °C were exposed to 37 °C for 0, 15, 30, 60 or 90 min, which induced an increase in the expression level of the AtDjC5 gene. The level of AtDjC5 mRNA began to increase 30 min after HS and continued to rise until 90 min (Figure 5A). Due to the ER localization of AtDjC5, we evaluated the effect of tunicamycin (TM) treatment, an ER stress inducer, on AtDjC5 gene expression. After the 10-day-old seedlings grown at 22 °C were treated with 5 μg/mL TM for 2 h, the expression of the AtDjC5 gene was markedly increased, reaching 3.5 times that detected before treatment (Figure 5B). The data above suggest that the expression of the AtDjC5 gene was induced by heat stress and ER stress.
Figure 5.
The effects of heat or TM treatment on the expression of the AtDjC5 gene by Q-PCR. (A) Time course of heat-induced AtDjC5 expression. Ten-day-old seedlings grown at 22 °C were heated at 37 °C for the length of time indicated. The level of AtDjC5 in the unheated seedlings was normalized to 1. (B) The effect of TM treatment on AtDjC5 expression. Ten-day-old seedlings grown at 22 °C were treated with 5 μg/mL TM for 2 h. The level of AtDjC5 in the nontreated seedlings was normalized to 1. Q-PCR was performed with AtDjC5-specific primers (Table S1). Actin was used as an internal control. Data are the mean ± SD from three independent experiments.
2.4. AtDjC5 Is Required for ER Stress–Induced Autophagy and the ER Stress Response
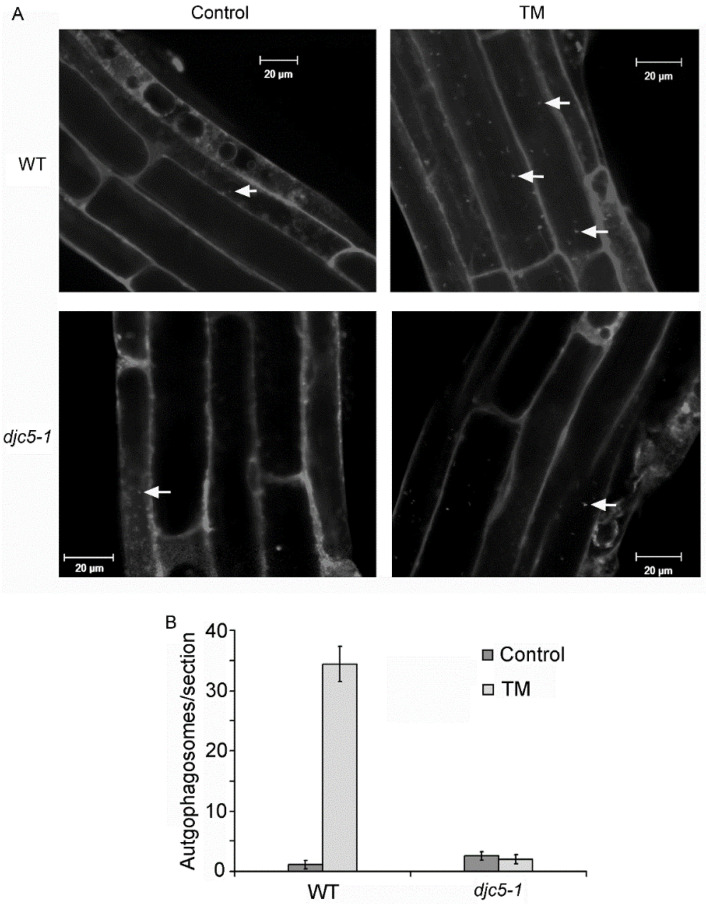
To clarify whether AtDjC5 is related to ER stress-induced autophagy, the induction of autophagy was examined in WT and djc5-1 mutant plants. Seven-day-old WT and djc5-1 seedlings were treated with 5 μg/mL TM for 8 h, followed by MDC staining; nontreated seedlings were used as a control. Both WT and djc5-1 seedlings showed very few autophagosomes in the control group (Figure 6A). The WT seedlings showed markedly elevated autophagy after TM treatment; however, autophagy induction was not observed in response to TM treatment in djc5-1 seedlings (Figure 6A). To quantify these results, autophagosome numbers were analyzed per root section for WT and djc5-1. Autophagosome numbers in the WT roots strongly increased after TM treatment; however, the djc5-1 roots did not show obvious differences from WT roots in autophagosome numbers after TM treatment (Figure 6B), indicating that AtDjC5 is likely to be involved in the regulation of ER stress-induced autophagy.
Figure 6.
AtDjC5 knockout inhibited TM-induced autophagy. WT, wild-type plants; djc5-1, a T-DNA insertion mutant line for the AtDjC5 gene; Control, nontreated roots; TM, roots treated with 5 μg/mL TM for 8 h. (A) MDC-stained roots before and after TM treatment were observed using a confocal microscope. Arrows indicate autophagosomes. Bar = 20 μm. (B) The numbers of MDC-stained autophagosomes per root section before and after TM treatment were counted, and the average numbers were determined for 20 seedlings per treatment. Error bars represent SD.
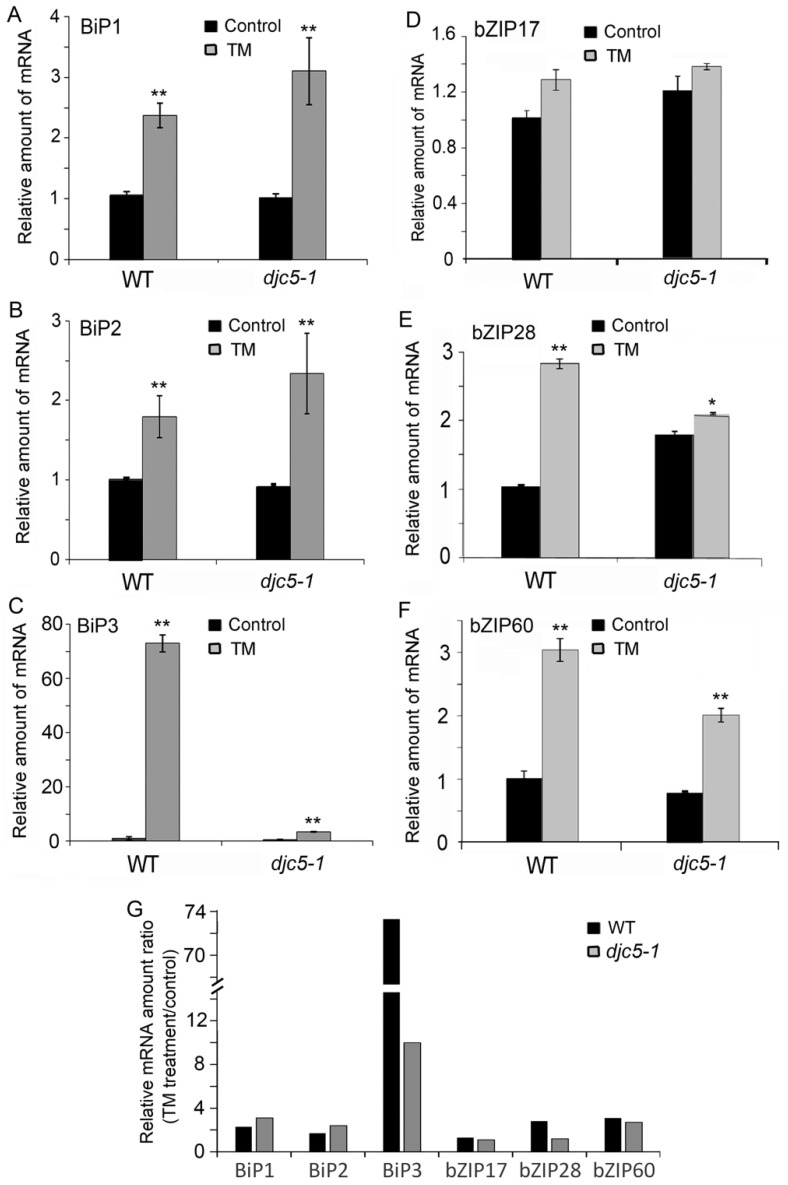
Furthermore, we explored the possibility of AtDjC5 participating in UPR regulation. We examined the effects of TM treatment on the expression levels of the BiP1, BiP2, BiP3, bZIP17, bZIP28, and bZIP60 genes. Except for bZIP17, the expression levels of the other five genes were upregulated to different degrees in the WT and djc5-1 seedlings after treatment with 5 μg/mL TM for 30 min (Figure 7A–F). Meanwhile, we found that AtDjC5 knockout significantly suppressed the TM-induced upregulation of BiP3 and bZIP28 expression (Figure 7G). The above results indicate that AtDjC5 is required for the ER stress response.
Figure 7.
The effects of AtDjC5 knockout on the expression levels of the three BiP and three bZIP genes before and after TM treatment. WT, wild-type plants; djc5-1, a T-DNA insertion mutant line for the AtDjC5 gene; Control, nontreated seedlings; TM, seedlings treated with 5 μg/mL TM for 30 min. (A–F) Analysis of the expression levels of the BiP1, BiP2, BiP3, bZIP17, bZIP28, and bZIP60 genes in WT and djc5-1. Q-PCR was performed with gene-specific primers (Table S1). Actin was used as an internal control. The expression levels of these genes in the nontreated WT seedlings were set to 1 and used for normalization. Data are the mean ± SD from three independent experiments. Asterisks indicate significant differences from the control group. (G) Comparison of the relative mRNA amount ratios (TM treatment/control) of BiP1, BiP2, BiP3, bZIP17, bZIP28, or bZIP60 between WT and the djc5-1 seedlings. (* p < 0.05, ** p < 0.01).
3. Discussion
In plants, J-domain proteins have been reported to localize in different subcellular compartments and participate in various biological processes. A previous study showed that AtDjC5 (also named AtPam18-3) mainly exists in mitochondria [55]. However, in this study, we found that AtDjC5 showed a partial overlap with the ER maker (Figure 4A), indicating that AtDjC5 possibly has multiple subcellular localization.
Eukaryotic have two evolutionarily highly conserved systems to adapt to environmental stress conditions: the heat shock response (HSR) and the UPR. Various environmental stresses may lead to the accumulation of unfolded proteins or denatured proteins in plant cells. HSPs/chaperones respond to environmental stress, while cells respond to the accumulation of unfolded proteins in the ER by activating the UPR to generate HSPs/chaperones. It is likely that there is crosstalk between the HSR and the UPR. Heat stress has been shown to transiently induce X-box binding protein 1 (XBP1) splicing and to lead to increases in the mRNA expression levels of HSPA5 and DNAJB9 (ERdj4) [56,57,58,59], two typical UPR genes. The IRE1-bZIP60 pathway in plants is activated by heat stress [56]. Gao reported that bZIP28 is an important component of the plant response to heat stress and is involved in the regulation of chaperone expression [60]. Further work has confirmed the role of bZIP28 in the plant response to heat stress and connected it to the maintenance of fertility under heat stress [61]. In this study, we showed that AtDjC5 was induced by heat and TM treatment, and the knockout of AtDjC5 led to decreased thermotolerance and inhibited the ER stress-induced upregulation of BiP3 and bZIP28 expression (Figure 5 and Figure 7), suggesting that AtDjC5 promotes plant thermotolerance, likely by assisting the BiP3-bZIP28 pathway. The dissociation of BiP3 from bZIP28 is a major switch that activates the UPR signaling pathway in plants. In response to heat stress, BiP3 competes with unfolded proteins in the ER to release bZIP28, allowing it to be mobilized from the ER to the Golgi apparatus and then enter the nucleus to activate the UPR. AtDjC5, as a chaperone, is likely to play an important role in assisting the dissociation of BiP3 from the C-terminal tail of bZIP28. Loss of AtDjC5 function might block bZIP28 exit from the ER and inhibit the UPR, which downregulates the expression of chaperone proteins, leading to decreased thermotolerance. Whether AtDjC5 plays a role as a partner of BiP3 and how they regulate UPR still need answers.
4. Materials and Methods
4.1. Plant Materials and Growth Conditions
Seeds of Arabidopsis thaliana (ecotype Columbia-0) were surface-sterilized, plated on Murashige and Skoog (MS) medium containing 1.0% (w/v) sucrose and 0.8% (w/v) agar, and kept at 4 °C for 3 d. Then, plants were cultured in a growth chamber under long-day conditions (16/8 h photoperiod) at approximately 100 μmol photons m−2 s−1 and 22 °C. Two-week-old seedlings were transplanted to soil and cultured under the original growth conditions.
4.2. Identification and Isolation of AtDjC5 T-DNA Insertional Mutants
Seeds of a putative T-DNA insertional mutant for AtDjC5 (At5g03030) and SALK_091892 (djc5-1) were obtained from the Arabidopsis Biological Resource Center (Columbus, OH, USA). The homozygous AtDjC5 mutant was identified by PCR as described by the Salk Institute Genomic Analysis Laboratory (http://signal.salk.edu/tdna_FAQs.html). PCR was conducted with genomic DNA from T2 generation seedlings using AtDjC5-specific primers (LP, 5′- GCGTCTGTAATCGACGGTAAG-3′; RP, 5′-CTGGCTATACACAGGCTACGC-3′) and a T-DNA left border primer LBb1.3 (5′-ATTTTGCCGATTTCGGAAC-3′). The position of the T-DNA insertion was determined by PCR product sequencing.
The transcript levels of AtDjC5 in WT and djc5-1 were detected by RT–PCR using a RT–PCR Kit (TaKaRa, Otsu, Japan). Total RNA was isolated from 10-day-old seedlings with TRIzol reagent (Life Technologies, Carlsbad, CA, USA). The AtDjC5 coding region was amplified using a forward primer (FP) 5′- ATGGCTACGCCAATGATTGC-3′ and a reverse primer (RP) 5′-TCAAAAGGCAGAACCGCTGT-3′. Actin was used as a loading control and amplified using FP 5′-AGGCACCTCTTAACCCTAAAGC-3′ and RP 5′-GGACAACGGAATCTCTCAGC-3′. PCR products were analyzed by agarose gel electrophoresis.
4.3. Heat Stress Treatment and Chlorophyll Measurement
Seeds of different genotypes were planted on separate regions of the same MS plate, with 30 seeds per genotype and experiment. For the basal thermotolerance assay, 11-day-old seedlings grown at 22 °C were exposed to 44 °C for 90 min and recovered at 22 °C for 3 to 7 d prior to calculating the survival rate. For the acquired thermotolerance assay, 11-d-old seedlings grown at 22 °C were acclimated at 37 °C for 30 min and returned to 22 °C for 120 min before challenge at 45 °C for 120 min. Then, the seedlings recovered at 22 °C for 3 to 7 d prior to calculating the survival rate or analyzing the total chlorophyll content. Plants that were still green and produced new leaves were scored as surviving. The total chlorophyll content was determined as described by Porra [62].
4.4. Electrolyte Leakage Measurement
Membrane system stability was evaluated by an electrolyte leakage assay before and after heat treatment. Green leaves from 11-day-old seedlings grown at 22 °C were washed three times with ion-free water and then incubated in 5 mL of ion-free water at 45 °C for 120 min. The conductivity of the incubation medium was measured using a Leici conductivity meter (DDS-IIA, Shanghai, China).
4.5. Assay of Root Vitality
The vitality of the root tissue was detected with 2,3,5-triphenyltetrazolium chloride (TTC) according to Gong et al. [63] with modifications. Eleven-day-old seedlings grown at 22 °C were exposed to 45 °C for 1 h. Heat-treated seedlings were transferred to 0.6% (w/v) TTC solution and cultured at 22 °C for 20 h. After washing three times, 1.0-cm tips of primary roots (total 30 per sample) were homogenized in 95% (v/v) ethanol and centrifuged at 4 000× g for 5 min. The absorbance of the supernatant at 530 nm was measured using a spectrophotometer (TU-1900, PERSEE, Beijing, China).
4.6. Plant Transformation
To generate the P35S::AtDjC5 construct, the AtDjC5 coding region was PCR-amplified with cDNA from A. thaliana (Columbia-0) seedlings using FP 5′-TCTAGAATGGCTACGCCAATGATT-3′ and RP′-GAGCTCTCAAAAGGCAGAACCGCT-3′. The PCR product was cloned into the binary vector pCAMBIA1300-35S digested with Xba I/Sac I.
To generate the PAtDjC5::β-glucuronidase (GUS) construct, a 1239-bp DNA fragment upstream of the AtDjC5 translational start codon was PCR-amplified with genomic DNA from A. thaliana seedlings using FP 5′-CTGCAGAACTCCCTCAAGGCTAAACC-3′ and RP 5′-TCTAGATATTCAGCTAAGTAGTTGTTCGG-3′. The PCR fragment was ligated into the binary vector pCAMBIA1300-GUS digested with Pst I/Xba I.
To generate the P35S::AtDjC5-sGFP construct, the AtDjC5 coding region was obtained from a plasmid containing the AtDjC5 coding region by digestion with Xba I/Sac I. The AtDjC5 coding region was fused to the N terminus of sGFP in the binary vector pCAMBIA1300-35S::sGFP digested with XbaI/SacI.
The resulting plasmids were introduced into Agrobacterium tumefaciens (GV3101). Transformation of A. thaliana was performed by the floral-dipping method [64]. Transgenic lines were selected on MS medium containing 25 μg mL−1 hygromycin.
4.7. Real-Time Quantitative RT–PCR
Total RNA was isolated from different tissues or different genotypic seedlings using TRIzol reagent (Life Technologies, Carlsbad, CA, USA). Real-time quantitative RT–PCR (Q-PCR) was performed as described by Zhang et al. [65]. Primer pairs were designed using Primer Express software (Applied Biosystems) (Table S1). ACTIN was used as the internal control.
4.8. Obtaining AtDjC5 amiRNA Mutants
Using WMD3-Web MicroRNA Designer (http://wmd3.weigelworld.org/cgi-bin/webapp.cgi, accessed on 10 February 2016), we obtained two AtDjC5-specific artificial microRNAs (amiRNAs) (“TAAGCAAGATAGTTACTGCCT” and “TTCTGGGTGATTTACAACCAT”) and four oligonucleotide sequences (I to IV) (Table S2), which were used to engineer amiRNA into the endogenous miR319a precursor by site-directed mutagenesis. The detailed protocol for amiRNA cloning is as described by Schwab [66]. The resulting two amiRNAs were cloned into the binary vector pCAMBIA1300 via the BamH I/EcoR I site. The resulting plasmids were introduced into Agrobacterium tumefaciens (GV3101). Transformation of A. thaliana was performed by the floral-dipping method [64]. Transgenic lines were selected on MS medium containing 25 μg mL−1 hygromycin. The transgenic plants introduced into the above two amiRNAs were defined as djc5-2 and djc5-3, respectively.
4.9. Subcellular Localization of AtDjC5
Roots of 5-day-old transgenic seedlings harboring P35S::AtDjC5-sGFP were incubated in 1 μM ER-Tracker Red (Molecular Probes, Carlsbad, CA) solution for 30 min in the dark. Green fluorescent protein (GFP) fluorescence and ER-Tracker Red fluorescence were observed using a laser scanning confocal microscope (FV3000; OLYMPUS, Tokyo Prefecture, Japan) with excitation/emission wavelengths of 488/510 nm for GFP and 587/615 nm for ER-Tracker Red.
4.10. Histochemical Staining
Trypan blue staining was conducted as described by Choi et al. [67]. Histochemical staining for GUS expression was performed according to Jefferson et al. [68].
4.11. MDC Staining and Microscopy
Seven-day-old WT or djc5-1 seedlings were incubated in MS liquid medium with or without 5 μg/mL TM for 8 h, subjected to MDC staining with 0.05 mM monodansylcadaverine (MDC) for 10 min, and washed three times with PBS. MDC fluorescence was visualized using a laser scanning confocal microscope (LSM 710; Zeiss, Oberkochen, Germany) with an excitation/emission of 335/508 nm.
4.12. Statistical Analysis
Statistical analyses were carried out with STATISTICA 6.0 (StatSoft, Inc., Tulsa, OK, USA). The significance of differences was determined at p < 0.05 using ANOVA with Tukey’s HSD test.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/ijms232113134/s1, Additional supporting information may be found in the online version of this article. Table S1 Primers for real-time PCR amplification. Table S2 Primers for cloning of AtDjC5-specific amiRNAs.
Author Contributions
Methodology, T.-T.S., L.W., C.-H.S., Y.-C.Z. and Y.-L.F.; validation, T.-T.S., L.W. and C.-H.S.; investigation, T.-T.S., L.-L.W., C.-H.S., Y.-C.Z., T.Z. and Y.-L.F.; writing—original draft preparation, B.L. and J.-T.B.; writing—review and editing, B.L. and J.-T.B.; supervision, B.L. and J.-T.B.; project administration, B.L. and J.-T.B.; funding acquisition, B.L. and J.-T.B. All authors have read and agreed to the published version of the manuscript.
Data Availability Statement
Data is contained within the article and Supplementary Materials.
Conflicts of Interest
The authors declare no conflict of interests.
Funding Statement
This research was funded by “the National Natural Science Foundation of China, grant number 31870222” and the “Science and Technology Project of Hebei Education Department, China, grant number QN2021084”.
Footnotes
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Battisti D.S., Naylor R.L. Historical warnings of future food insecurity with unprecedented seasonal heat. Science. 2009;323:240–244. doi: 10.1126/science.1164363. [DOI] [PubMed] [Google Scholar]
- 2.Li B.J., Gao K., Ren H.M., Tang W.Q. Molecular mechanisms governing plant responses to high temperatures. J. Integr. Plant Biol. 2018;60:757–779. doi: 10.1111/jipb.12701. [DOI] [PubMed] [Google Scholar]
- 3.Lohani N., Singh M.B., Bhalla P.L. High temperature susceptibility of sexual reproduction in crop plants. J. Exp. Bot. 2020;71:555–568. doi: 10.1093/jxb/erz426. [DOI] [PubMed] [Google Scholar]
- 4.Timperio A.M., Egidi M.G., Zolla L. Proteomics applied on plant abiotic stresses: Role of heat shock proteins (HSP) J. Proteom. 2008;71:391. doi: 10.1016/j.jprot.2008.07.005. [DOI] [PubMed] [Google Scholar]
- 5.Hassan M.U., Chattha M.U., Khan I., Chattha M.B., Aslam M.T. Heat stress in cultivated plants: Nature, impact, mechanisms, and mitigation strategies-A review. Plant Biosyst. 2020;7:1–56. doi: 10.1080/11263504.2020.1727987. [DOI] [Google Scholar]
- 6.Janni M., Gullì M., Maestri E., Marmiroli M., Valliyodan B., Nguyen H.T., Marmiroli N. Molecular and genetic bases of heat stress responses in crop plants and breeding for increased resilience and productivity. J. Exp. Bot. 2020;71:3780–3802. doi: 10.1093/jxb/eraa034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Suzuki N., Bajad S., Shuman J., Shulaev V., Mittler R. The transcriptional co-activator MBF1c is a key regulator of thermotolerance in Arabidopsis thaliana. J. Biol. Chem. 2008;283:9269–9275. doi: 10.1074/jbc.M709187200. [DOI] [PubMed] [Google Scholar]
- 8.Frank G., Pressman E., Ophir R., Althan L., Shaked R., Freedman M., Shen S., Firon N. Transcriptional profiling of maturing tomato microspores reveals the involvement of heat shock proteins, ROS scavengers, hormones, and sugars in the heat stress response. J. Exp. Bot. 2009;60:3891–3908. doi: 10.1093/jxb/erp234. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Vierling E. The roles of heat shock proteins in plants. Annu. Rev. Plant Physiol. Plant Mol. Biol. 1991;42:579–620. doi: 10.1146/annurev.pp.42.060191.003051. [DOI] [Google Scholar]
- 10.Larkindale J., Hall J.D., Knight M.R., Vierling E. Heat stress phenotypes of Arabidopsis mutants implicate multiple signaling pathways in the acquisition of thermotolerance. Plant Physiol. 2005;138:882–897. doi: 10.1104/pp.105.062257. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Charng Y.Y., Liu H.C., Liu N.Y., Chi W.T., Wang C.N., Chang S.H., Wang T.T. A heat-inducible transcription factor, HsfA2, is required for extension of acquired thermotolerance in Arabidopsis. Plant Physiol. 2007;143:251–262. doi: 10.1104/pp.106.091322. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Wang R., Mao C.J., Jiang C.H., Zhang L., Peng S.Y., Zhang Y., Feng S.C., Ming F. One heat shock transcription factor confers high thermal tolerance in Clematis plants. Int. J. Mol. Sci. 2021;22:2900. doi: 10.3390/ijms22062900. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Miernyk J.A. Protein folding in the plant cell. Plant Physiol. 1999;121:695–703. doi: 10.1104/pp.121.3.695. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Nollen E.A., Morimoto R.I. Chaperoning signaling pathways: Molecular chaperones as stress-sensing ‘heat shock’ proteins. J. Cell Sci. 2002;115:2809–2816. doi: 10.1242/jcs.115.14.2809. [DOI] [PubMed] [Google Scholar]
- 15.Mayer M.P., Bukau B. Hsp70 chaperones: Cellular functions and molecular mechanism. Cell. Mol. Life Sci. 2005;62:670–684. doi: 10.1007/s00018-004-4464-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Shan Q., Ma F.T., Wei J.Y., Li H.Y., Ma H., Sun P. Physiological Functions of Heat Shock Proteins. Curr. Protein Pept. Sci. 2020;21:751–760. doi: 10.2174/1389203720666191111113726. [DOI] [PubMed] [Google Scholar]
- 17.Schroder M., Kaufman R.J. The mammalian unfolded protein response. Annu. Rev. Biochem. 2005;74:739–789. doi: 10.1146/annurev.biochem.73.011303.074134. [DOI] [PubMed] [Google Scholar]
- 18.Schroder M., Kaufman R.J. ER stress and the unfolded protein response. Mutat. Res. 2005;569:29–63. doi: 10.1016/j.mrfmmm.2004.06.056. [DOI] [PubMed] [Google Scholar]
- 19.Bernales S., McDonald K.L., Walter P. Autophagy counterbalances endoplasmic reticulum expansion during the unfolded protein response. PLoS Biol. 2006;4:e423. doi: 10.1371/journal.pbio.0040423. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Howell S.H. Endoplasmic reticulum stress responses in plants. Annu. Rev. Plant Biol. 2013;64:477–499. doi: 10.1146/annurev-arplant-050312-120053. [DOI] [PubMed] [Google Scholar]
- 21.Reyes-Impellizzeri S., Moreno A.A. The Endoplasmic Reticulum Role in the Plant Response to Abiotic Stress. Front. Plant Sci. 2013;12:755447. doi: 10.3389/fpls.2021.755447. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Iwata Y., Koizumi N. An Arabidopsis transcription factor, AtbZIP60, regulates the endoplasmic reticulum stress response in a manner unique to plants. Proc. Natl. Acad. Sci. USA. 2005;102:5280–5285. doi: 10.1073/pnas.0408941102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Liu J.X., Srivastava R., Che P., Howell S.H. An endoplasmic reticulum stress response in Arabidopsis is mediated by proteolytic processing and nuclear relocation of a membraneassociated transcription factor, bZIP28. Plant Cell. 2007;19:4111–4119. doi: 10.1105/tpc.106.050021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Liu J.X., Howell S.H. Managing the protein folding demands in the endoplasmic reticulum of plants. New Phytol. 2016;38:82–97. doi: 10.1111/nph.13915. [DOI] [PubMed] [Google Scholar]
- 25.Srivastava R., Deng Y., Shah S., Rao A.G., Howell S.H. Binding protein is a master regulator of the endoplasmic reticulum stress sensor/transducer bZIP28 in Arabidopsis. Plant Cell. 2013;25:1416–1429. doi: 10.1105/tpc.113.110684. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Che P., Bussell J.D., Zhou W., Estavillo G.M., Pogson B.J., Smith S.M. Signaling from the endoplasmic reticulum activates brassinosteroid signaling and promotes acclimation to stress in Arabidopsis. Sci. Signal. 2010;3:ra69. doi: 10.1126/scisignal.2001140. [DOI] [PubMed] [Google Scholar]
- 27.Iwata Y., Fedoroff N.V., Koizumi N. Arabidopsis bZIP60 is a proteolysis-activated transcription factor involved in the endoplasmic reticulum stress response. Plant Cell. 2008;20:3107–3121. doi: 10.1105/tpc.108.061002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Tajima H., Iwata Y., Iwano M., Takayama S., Koizumi N. Identification of an Arabidopsis transmembrane bZIP transcription factor involved in the endoplasmic reticulum stress response. Biochem. Biophys. Res. Commun. 2008;374:242–247. doi: 10.1016/j.bbrc.2008.07.021. [DOI] [PubMed] [Google Scholar]
- 29.Houck S.A., Ren H.Y., Madden V.J., Bonner J.N., Conlin M.P., Janovick J.A., Conn P.M., Cyr D.M. Quality control autophagy degrades soluble ERAD-resistant conformers of the misfolded membrane protein GnRHR. Mol. Cell. 2014;54:166–179. doi: 10.1016/j.molcel.2014.02.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Zhuang X., Jiang L. Autophagosome biogenesis in plants. Autophagy. 2014;10:704–705. doi: 10.4161/auto.28060. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Zeng Y.L., Li B.Y., Zhang W.X., Jiang L.W. ER-phagy and ER stress response (ERSR) in plants. Front. Plant Sci. 2019;10:1192. doi: 10.3389/fpls.2019.01192. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Doelling J.H., Walker J.M., Friedman E.M., Thompson A.R., Vierstra R.D. The APG8/12-activating enzyme APG7 is required for proper nutrient recycling and senescence in Arabidopsis thaliana. J. Biol. Chem. 2002;277:33105–33114. doi: 10.1074/jbc.M204630200. [DOI] [PubMed] [Google Scholar]
- 33.Hanaoka H., Noda T., Shirano Y., Kato T., Hayashi H., Shibata D., Tabata S., Ohsumi Y. Leaf senescence and starvationinduced chlorosis are accelerated by the disruption of an Arabidopsis autophagy gene. Plant Physiol. 2002;129:1181–1193. doi: 10.1104/pp.011024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Liu Y., Schiff M., Czymmek K., Tallóczy Z., Levine B., Dinesh-Kumar S.P. Autophagy regulates programmed cell death during the plant innate immune response. Cell. 2005;121:567–577. doi: 10.1016/j.cell.2005.03.007. [DOI] [PubMed] [Google Scholar]
- 35.Liu Y., Xiong Y., Bassham D.C. Autophagy is required for tolerance of drought and salt stress in plants. Autophagy. 2009;5:954–963. doi: 10.4161/auto.5.7.9290. [DOI] [PubMed] [Google Scholar]
- 36.Xiong Y., Contento A.L., Bassham D.C. AtATG18a is required for the formation of autophagosomes during nutrient stress and senescence in Arabidopsis thaliana. Plant J. 2005;42:535–546. doi: 10.1111/j.1365-313X.2005.02397.x. [DOI] [PubMed] [Google Scholar]
- 37.Xiong Y., Contento A.L., Bassham D.C. Disruption of autophagy results in constitutive oxidative stress in Arabidopsis. Autophagy. 2007;3:257–258. doi: 10.4161/auto.3847. [DOI] [PubMed] [Google Scholar]
- 38.Liu Y., Burgos J.S., Deng Y., Srivastava R., Howell S.H., Bassham D.C. Degradation of the endoplasmic reticulum by autophagy during endoplasmic reticulum stress in Arabidopsis. Plant Cell. 2012;24:4635–4651. doi: 10.1105/tpc.112.101535. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Kampinga H.H., Andreasson C., Barducci A., Cheetham M.E., Cyr D., Emanuelsson C., Genevaux P., Gestwicki J.E., Goloubinoff P., Huerta-Cepas J., et al. Function, evolution, and structure of J-domain proteins. Cell Stress Chaperones. 2019;24:7–15. doi: 10.1007/s12192-018-0948-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Wyszkowski H., Janta A., Sztangierska W., Obuchowski I., Chamera T., Kłosowska A., Liberek K. Class-specific interactions between Sis1 J-domain protein and Hsp70 chaperone potentiate disaggregation of misfolded proteins. Proc. Natl. Acad. Sci. USA. 2021;118:e2108163118. doi: 10.1073/pnas.2108163118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Miernyk J.A. The J-domain proteins of Arabidopsis thaliana: An unexpectedly large and diverse family of chaperones. Cell Stress Chaperones. 2001;6:209–218. doi: 10.1379/1466-1268(2001)006<0209:TJDPOA>2.0.CO;2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Rajan V.B., D’Silva P. Arabidopsis thaliana J-class heat shock proteins: Cellular stress sensors. Funct. Integr. Genom. 2009;9:433–446. doi: 10.1007/s10142-009-0132-0. [DOI] [PubMed] [Google Scholar]
- 43.Christensen C.A., Gorsich S.W., Brown R.H., Jones L.G., Brown J., Shaw J.M., Drews G.N. Mitochondrial GFA2 is required for synergid cell death in Arabidopsis. Plant Cell. 2002;14:2215–2232. doi: 10.1105/tpc.002170. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Li G.L., Chang H., Li B., Zhou W., Sun D.Y., Zhou R.G. The roles of the atDjA2 and atDjA3 molecular chaperone proteins in improving thermotolerance of Arabidopsis thaliana seedlings. Plant Sci. 2007;173:408–416. doi: 10.1016/j.plantsci.2007.06.013. [DOI] [Google Scholar]
- 45.Tamura K., Takahashi H., Kunieda T., Fuji K., Shimada T., Hara-Nishimura I. Arabidopsis KAM2⁄GRV2 is required for proper endosome formation and functions in vacuolar sorting and determination of the embryo growth axis. Plant Cell. 2007;19:320–332. doi: 10.1105/tpc.106.046631. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Glynn J.M., Froehlich J.E., Osteryoungc K.W. Arabidopsis ARC6 coordinates the division machineries of the inner and outer chloroplast membranes through interaction with PDV2 in the intermembrane space. Plant Cell. 2008;20:2460–2470. doi: 10.1105/tpc.108.061440. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Yang K.Z., Xia C., Liu X.L., Dou X.Y., Wang W., Chen L.Q., Zhang X.Q., Xie L.F., He L.Y., Ma X., et al. A mutation in Thermosensitive Male Sterile 1, encoding a heat shock protein with DnaJ and PDI domains, leads to thermosensitive gametophytic male sterility in Arabidopsis. Plant J. 2009;57:870–882. doi: 10.1111/j.1365-313X.2008.03732.x. [DOI] [PubMed] [Google Scholar]
- 48.Yang Y.Q., Qin Y.X., Xie C.G., Zhao F.Y., Zhao J.F., Liu D.F., Chen S.Y., Fuglsang A.T., Palmgren M.G., Schumaker K.S., et al. The Arabidopsis chaperone J3 regulates the plasma membrane H+-ATPase through interaction with the PKS5 kinase. Plant Cell. 2010;22:1313–1332. doi: 10.1105/tpc.109.069609. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Chen K.M., Piippo M., Holmstr¨om M., Nurmi M., Pakula E., Suorsa M., Aro E.M. A chloroplast-targeted DnaJ protein AtJ8 is negatively regulated by light and has rapid turnover in darkness. J. Plant Physiol. 2011;168:1780–1783. doi: 10.1016/j.jplph.2011.04.001. [DOI] [PubMed] [Google Scholar]
- 50.Shen L., Kang Y.G., Liu L., Yu H. The J-domain protein J3 mediates the integration of flowering signals in Arabidopsis. Plant Cell. 2011;23:499–514. doi: 10.1105/tpc.111.083048. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Zhou W., Zhou T., Li M.X., Zhao C.L., Jia N., Wang X.X., Sun Y.Z., Li G.L., Xu M., Zhou R.G. The Arabidopsis J-protein AtDjB1 facilitates thermotolerance by protecting cells against heat-induced oxidative damage. New Phytol. 2012;194:364–378. doi: 10.1111/j.1469-8137.2012.04070.x. [DOI] [PubMed] [Google Scholar]
- 52.Pulido P., Toledo-Ortiz G., Phillips M.A., Wright L.P., Rodríguez-Concepción M. Arabidopsis j-protein j20 delivers the first enzyme of the plastidial isoprenoid pathway to protein quality control. Plant Cell. 2013;25:4183–4194. doi: 10.1105/tpc.113.113001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Wang X., Jia N., Zhao C., Fang Y., Lv T., Zhou W., Sun Y., Li B. Knockout of AtDjB1, a J-domain protein from Arabidopsis thaliana, alters plant responses to osmotic stress and abscisic acid. Physiol. Plant. 2014;45:59–63. doi: 10.1111/ppl.12169. [DOI] [PubMed] [Google Scholar]
- 54.Jia N., Lv T.T., Li M.X., Wei S.S., Li Y.Y., Zhao C.L., Li B. The J-protein AtDjB1 is required for mitochondrial complex I activity and regulates growth and development through ROS-mediated auxin signalling. J. Exp. Bot. 2020;71:3780–3802. doi: 10.1093/jxb/erw171. [DOI] [PubMed] [Google Scholar]
- 55.Tamadaddi C., Sagar V., Verma A.K., Afsal F., Sahi C. Expansion of the evolutionarily conserved network of J-domain proteins in the Arabidopsis mitochondrial import complex. Plant Mol. Biol. 2021;105:385–403. doi: 10.1007/s11103-020-01095-8. [DOI] [PubMed] [Google Scholar]
- 56.Deng Y., Humbert S., Liu J.X., Srivastava R., Rothstein S.J., Howell S.H. Heat induces the splicing by IRE1 of a mRNA encoding a transcription factor involved in the unfolded protein response in Arabidopsis. Proc. Natl. Acad. Sci. USA. 2011;108:7247–7252. doi: 10.1073/pnas.1102117108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Adachi M., Liu Y., Fujii K., Calderwood S.K., Nakai A., Imai K., Shinomura Y. Oxidative stress impairs the heat stress response and delays unfolded protein recovery. PLoS ONE. 2009;4:e7719. doi: 10.1371/journal.pone.0007719. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Finka A., Mattoo R.U., Goloubinoff P. Meta-analysis of heat- and chemically upregulated chaperone genes in plant and human cells. Cell Stress Chaperones. 2011;16:15–31. doi: 10.1007/s12192-010-0216-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Heldens L., Hensen S.M.M., Onnekink C., van Genesen S.T., Dirks R.P., Lubsen N.H. An atypical unfolded protein response in heat shocked cells. PLoS ONE. 2011;6:e23512. doi: 10.1371/journal.pone.0023512. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Gao H., Brandizzi F., Benning C., Larkin R.M. A membrane-tethered transcription factor defines a branch of the heat stress response in Arabidopsis thaliana. Proc. Natl. Acad. Sci. USA. 2008;105:16398–16403. doi: 10.1073/pnas.0808463105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Zhang S.S., Yang H.X., Ding L., Song Z.T., Ma H., Chang F., Liu J.X. Tissue-Specific Transcriptomics Reveals an important role of the unfolded protein response in maintaining fertility upon heat stress in Arabidopsis. Plant Cell. 2017;29:1007–1023. doi: 10.1105/tpc.16.00916. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Porra R.J., Thompson W.A., Kriedemann P.E. Determination of accurate extinction coefficients and simultaneous equations for assaying chlorophyll a and b extracted with four different solvents: Verification of the concentration of chlorophyll standards by atomic absorption spectroscopy. Biochem. Biophys. Acta. 1989;975:384–394. doi: 10.1016/S0005-2728(89)80347-0. [DOI] [Google Scholar]
- 63.Gong M., Chen S.N., Song Y.Q., Li Z.G. Effect of calcium and calmodulin on intrinsic heat tolerance in relation to antioxidant systems in maize seedlings. J. Plant Physiol. 1997;24:371–379. doi: 10.1071/PP96118. [DOI] [Google Scholar]
- 64.Clough S.J., Bent A.F. Floral dip: A simplified method for Agrobacteriummediated transformation of Arabidopsis thaliana. Plant J. 1998;16:735–743. doi: 10.1046/j.1365-313x.1998.00343.x. [DOI] [PubMed] [Google Scholar]
- 65.Zhang W., Zhou R.G., Gao Y.J., Zheng S.Z., Xu P., Zhang S.Q., Sun D.Y. Molecular and genetic evidence for the key role of AtCaM3 in heat-shock signal transduction in Arabidopsis. Plant Physiol. 2009;149:1773–1784. doi: 10.1104/pp.108.133744. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Schwab R., Ossowski S., Riester M., Warthmann N., Weigel D. Highly Specific Gene Silencing by Artificial MicroRNAs in Arabidopsis. Plant Cell. 2006;18:1121–1133. doi: 10.1105/tpc.105.039834. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Choi H.W., Kim Y.J., Lee S.C., Hong J.K., Hwang B.K. Hydrogen peroxide generation by the pepper extracellular peroxidase CaPO2 activates local and systemic cell death and defense response to bacterial pathogens. Plant Physiol. 2007;145:890–904. doi: 10.1104/pp.107.103325. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Jefferson R.A., Kavanagh T.A., Bevan M.W. GUS fusions: β-glucuronidase as a sensitive and versatile gene marker in higher plants. Eur. Mol. Biol. Organ. J. 1987;6:3901–3907. doi: 10.1002/j.1460-2075.1987.tb02730.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
Data is contained within the article and Supplementary Materials.