Abstract
Salmonella typhimurium strains harboring independent, defined mutations in aroA, invA, ssrA, or msbB were assessed for their ability to induce fluid accumulation, tissue damage, and local inflammation in rabbit ileal loops. Three wild-type strains of S. typhimurium, TML, HWSH, and SL1344, and two mutant strains, S. typhimurium SL1344 ssrA and S. typhimurium SL1344 msbB, consistently induced fluid accumulation in the lumen of loops and inflammation of loop-associated tissues. In contrast, three different S. typhimurium aroA strains and an invA mutant of SL1344 did not induce significant fluid accumulation in the rabbit ileal loops. However, the S. typhimurium aroA strains did induce an inflammatory infiltrate and some local villus-associated damage, but the invA mutant did not. Histologically, wild-type S. typhimurium, S. typhimurium SL1344 ssrA, and S. typhimurium SL1344 msbB demonstrated more severe effects on villus architecture than S. typhimurium aroA strains, whereas S. typhimurium invA-infected loops showed no detectable damage. This suggests that villus damage most likely contributes to fluid accumulation within the loop.
Salmonella-associated infections can be grouped into two general types according to their clinical features and underlying pathogenicities (18). Isolates of most Salmonella enterica serovars cause localized enteric infections or gastroenteritis associated with diarrhea and abdominal pain during which the infecting salmonella bacteria invade the gut wall but remain predominantly associated with the gut tissue and local lymphatics. These Salmonella serotypes can sometimes spread systemically in very old, very young, or immunocompromised individuals. Other organisms in the genus Salmonella, including S. enterica serovar Typhi (S. typhi), cause systemic diseases, including typhoid, in which bacteria spread to organs of the reticuloendothelial system, including the liver and spleen (20, 21).
Salmonella is an attractive target organism for scientific investigators interested in the genetic basis of virulence. In particular, the availability of excellent genetic manipulation techniques and the murine model of infection has allowed a huge amount of work to be performed. However, the ready accessibility of mice has also resulted in most in vivo studies of salmonella virulence being carried out in this model. Mouse-virulent Salmonella strains cause a systemic disease in mice that superficially resembles human typhoid (5, 6). In this model, mice do not develop diarrhea but do succumb to overwhelming bacterial growth in deep tissues. With the murine model, many Salmonella genes have been shown to contribute to virulence (7). The identification of attenuating mutations has encouraged the development of novel live salmonella vaccine strains (2, 5–7, 16, 17, 19, 24, 28, 32–35). Most work toward developing this vaccine for humans has involved attempts to develop live oral typhoid vaccines based on genetically defined S. typhi (17).
Identification of candidate attenuating mutations for use in experimental typhoid vaccines has been, in large part, dependent on studies of Salmonella typhimurium in the mouse, as S. typhi isolates exhibit low virulence rates in nonhumans. Few attenuated mutants identified in the mouse have been examined for their role in the diarrhea associated with Salmonella infections, as the mouse model is inappropriate for this purpose. Many of the virulence-associated genes identified by using the murine model fall into distinct classes. Some encode enzymes for critical biosynthetic pathways essential to sustain in vivo growth, such as the aro genes, which encode enzymes of the chorismate pathway (16, 17). Some encode type III secretion systems and are known as pathogenicity islands. Salmonella spp. have at least two such loci. One, named Salmonella pathogenicity island 1 (SPI-1), contributes to eucaryotic cell invasion (8, 22, 25, 26). A second, SPI-2, contributes to survival in deep tissue (29). Another group of genes are involved in the biosynthesis of lipopolysaccharide. For example, msbB is involved in the biosynthesis of the highly toxic lipid A component of lipopolysaccharide, and S. typhimurium msbB mutants have an altered pathogenicity in the mouse and a reduced ability to induce production of tumor necrosis factor alpha, interleukin 1, and nitric oxide (23).
In contrast to studies of systemic disease, investigations of the contributions of different Salmonella genes to gastroenteritis and diarrhea are more difficult to undertake because of the lack of readily available experimental systems. The rabbit loop model has been utilized as a method for identifying Salmonella strains that are able to induce fluid accumulation and potentially diarrhea (3, 12, 13, 15, 30, 36, 37, 40). For example, S. typhimurium TML has been shown to be a strong inducer of fluid accumulation and associated pathology in this model (12, 37). We employed the rabbit ileal loop assay to investigate the contributions of different classes of virulence-associated S. typhimurium genes, previously characterized in the murine model, to the pathogenesis of gastroenteritis and diarrhea.
MATERIALS AND METHODS
Bacterial strains and growth conditions.
S. typhimurium TML was isolated from a clinical case of human gastroenteritis, causes diarrhea in monkeys, and has been shown in previous studies to invade rabbit ileal loop mucosa and cause an intense inflammatory response and fluid accumulation within 18 h (10–14, 30, 31, 36, 37). S. typhimurium TML aroA was provided by G. Douce (Imperial College, London, United Kingdom). S. typhimurium HWSH was initially isolated from a calf dying of systemic salmonellosis (27). S. typhimurium HWSH aroA harbors a deletion mutant in the aroA gene and has been described previously (27). S. typhimurium SL1344 and SL1344 aroA (strain 3261) have been described previously (15). S. typhimurium SL1344 msbB was constructed by inserting a DNA sequence encoding kanamycin resistance into a cloned S. typhimurium msbB gene and replacing the wild-type msbB gene with the inactivated gene by allelic exchange (23). S. typhimurium SL1344 ssrA is defective in expression of the type III secretion system encoded within SPI-2. S. typhimurium SL1344 invA was provided by Tahir Ali (Imperial College). S. typhimurium SL1344 invA is defective in the expression of SPI-1 (9) (Table 1).
TABLE 1.
Strains used in this study
| S. typhimurium | Mutation |
|---|---|
| TML | S. typhimurium isolated from clinical case of gastroenteritis (10) |
| TML aroA | aroA mutant of TML |
| HWSH | Wild-type S. typhimurium |
| HWSH aroA | aroA mutant of HWSH |
| SL1344 | Wild-type S. typhimurium |
| SL1344 aroA | aroA mutant of SL1344 |
| SL1344 msbB | msbB mutant of SL1344 (lipid A mutant) |
| SL1344 ssrA | ssrA mutant of SL1344 (SPI-2 mutant) |
| SL1344 invA | invA mutant of SL1344 (SPI-1 mutant) |
Bacteria were grown statically and aerobically in Luria broth overnight at 37°C. For intestinal infection, bacteria were harvested by centrifugation and resuspended at a concentration of 108 bacteria/ml in phosphate-buffered saline (PBS) for injection into rabbit ileal loops.
Ileal loop procedure.
Specific-pathogen-free New Zealand White rabbits weighing less than 2 kg were anesthetized. The peritoneal cavity was opened by a sterile surgical technique, and the bowel was carefully washed with prewarmed PBS and clamped at least 20 cm distal to the ligament of Trietz. The required number of loops (loop length, 5 cm; interloop length, 5 cm) was measured, and the bowel was clamped proximal to the ileo-cecal junction. The length of bowel making the loops was cut at both ends, and all cut surfaces were clamped. The remaining intestine was reanastomosed and placed back into the peritoneal cavity. The resected ends of the isolated intestine were closed with sutures, and, to avoid compromising the integrity of blood supply to the tissue, the required number of loops was constructed by tying with ligatures. While closing the loop tie, we injected the bacterial suspension containing approximately 108 viable bacteria in a 0.5-ml volume of PBS into the proximal end of the loop. Positive-control loops were inoculated with 1 μg of cholera toxin (CT) (a gift from M.-G. Pizza, Instituto Richerche Immunologique Siena, Siena, Italy) in 0.5 ml of PBS; negative-control loops received 0.5 ml of PBS alone. The resected intestine was replaced into the peritoneal cavity, the cavity was closed, and the animal was allowed to recover. After 18 h, the animal was anesthetized, and the loops were removed and weighed, and loop fluids were placed in sterile containers.
Analysis of fluids from loops.
Fluid from infected loops was measured by volume, and the consistency, in terms of color and viscosity, was noted. Bacterial and cellular contents (erythrocytes and leukocytes) of the fluid were determined by wet preparation and Gram and Giemsa staining. The predominant cell types in the fluid exudate were determined by differential counts after Giemsa staining. Loop fluids were centrifuged to remove cellular components and assayed for bicarbonate, pH, and hemoglobin levels with a laboratory biochemical analyzer (Radiometer, Copenhagen, Denmark) and for total protein by the method of Bradford (1).
Histopathology of intestinal tissue.
After postmortem removal, the loop tissue was weighed and cut longitudinally to release any fluid contained within. Any gross changes were noted, and the mucosal side was inspected for macroscopic tissue damage. Loop tissue was washed in PBS, and small samples of loop tissue were placed into 10% formaldehyde in PBS at pH 7.2. Tissue was wax embedded, and thin-cut sections were stained with hematoxylin and eosin and examined under bright-field illumination with a Nikon Axiophot microscope.
Quantitation of bacteria in loops and fluids.
Loop fluid was assessed for number of viable bacteria by dilution and surface-viable counting by using a modification of the technique of Miles et al. (25). Loop tissue samples taken post mortem were cultured for S. typhimurium to demonstrate the presence of bacteria on or within loop tissue. Tissue samples (0.5 g [wet weight]) were washed with PBS before homogenization. Tissue was homogenized in 5 ml of Luria broth in a stomacher, and the resulting suspension was serially diluted for surface viable counting. All samples were plated on Luria agar.
RESULTS
Fluid secretion in rabbit ileal loops induced by different wild-type S. typhimurium strains and their mutant derivatives.
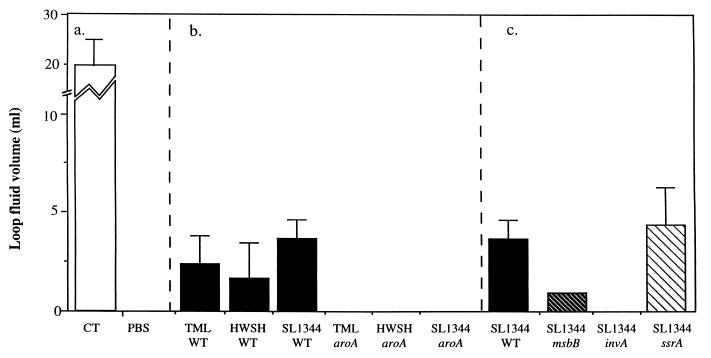
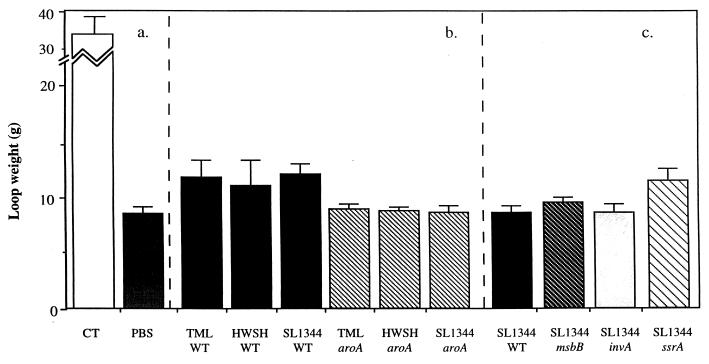
Different S. typhimurium mutant derivatives and the appropriate S. typhimurium wild-type controls were inoculated into ligated rabbit ileal loops, and the level of fluid accumulation over the next several hours was noted. Wild-type S. typhimurium TML, HWSH, and SL1344 consistently caused significant levels of fluid to accumulate within the gut lumen compared to uninfected control loops. The loop-derived fluid was blood stained and, when examined microscopically, found to contain many erythrocytes and polymorphonuclear leukocytes (PMNs). Fluid taken from loops infected with S. typhimurium HWSH contained blood clots which were not detectable in similar loops infected with other Salmonella strains. The volumes of fluids varied between 3 and 6 ml for the inoculations of different wild-type strains (Fig. 1). Loops infected with SL1344 ssrA or SL1344 msbB were macroscopically indistinguishable from loops infected with wild-type S. typhimurium controls. In contrast, the S. typhimurium aroA mutants and S. typhimurium SL1344 invA failed to induce any significant fluid accumulation. However, on the apical, lumen-exposed surface of loops infected with S. typhimurium aroA derivatives, a thin layer of pus was consistently associated with the mucosal surface, which, upon microscopic examination, was shown to be composed mainly of sheets of PMNs. Interestingly, this thin layer of pus was not observed when the invA mutant was tested. The increase in tissue weights for the wild-type S. typhimurium-infected loops reflected the fluid contained within them (Fig. 2).
FIG. 1.
Volumes of fluid recovered from infected rabbit ileal loops 18 h after infection. (a) Fluid recovery in control loops inoculated with either CT as a positive control or PBS as a negative control. (b) Fluid accumulation in loops immunized with wild type S. typhimurium strain TML, HWSH, or SL1344 or the equivalent aroA mutant. (c) Fluid accumulation in loops inoculated with S. typhimurium in which deletions have been engineered in the msbB, invA, or ssrA gene. These mutants were all created in wild-type S. typhimurium SL1344, which is included for comparison. Error bars represent standard errors of the means. WT, wild type.
FIG. 2.
Weight of infected loops recovered from animals 18 h after infection. (a) Weight of control loops inoculated with either CT as a positive control or PBS as a negative control. (b) Weight of loops immunized with wild-type S. typhimurium strain TML, HWSH, or SL1344 or the equivalent aroA mutant. (c) Weight of loops inoculated with S. typhimurium msbB, invA, or ssrA. These mutants were all created in wild-type S. typhimurium SL1344, which is included for comparison. Error bars represent standard deviations. WT, wild type.
Control loops injected with CT contained an average of 20 ml of non-blood-stained fluid, and leukocytes were absent from these fluids. Loops injected with PBS contained no free fluid.
Histopathology of rabbit ileal loops infected with different S. typhimurium wild-type strains, S. typhimurium SL1344 msbB, and S. typhimurium SL1344 ssrA.
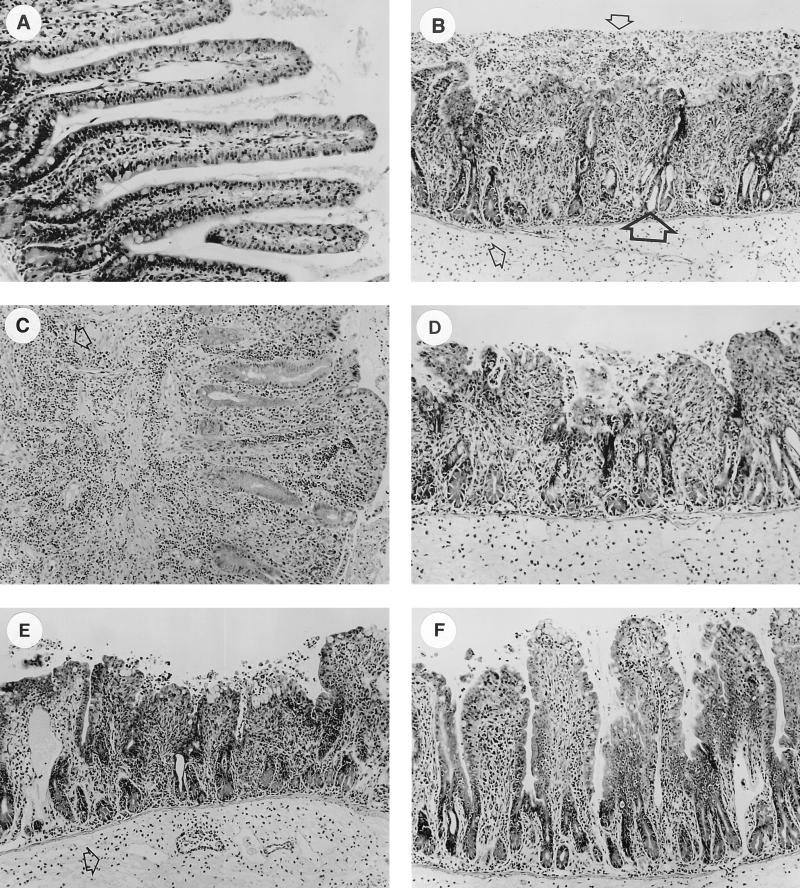
Tissue was taken from all loops and studied for histological changes. Normal tissue taken from a loop inoculated with PBS is shown in Fig. 3A. Contrasting tissue taken from a loop infected with wild-type HWSH is shown in Fig. 3B. The changes shown are typical of the histological changes observed in all loops infected with the wild-type strains. In all cases, villi were shortened, and in some cases villi were completely absent in tissue samples taken from loops infected with any of the S. typhimurium wild-type strains (Fig. 3C and E). There were large infiltrates of PMNs present in the damaged villus structure of the lamina propria and on the luminal surface of the mucosa. The lamina propria and submucosal blood vessels also contained large numbers of infiltrating PMNs. The villus enterocyte layer was severely damaged. There was bleeding into the submucosa and free blood in the lumen. Tissue edema was evident, unlike in PBS-injected controls (Fig. 3A). Loops infected with wild-type S. typhimurium exhibited dilatation of the crypts, suggesting an active secretory response to infection. Tissue taken from loops infected with either SL1344 ssrA or SL1344 msbB (Fig. 3D) showed morphological changes identical to those observed in loops infected with the appropriate wild-type control.
FIG. 3.
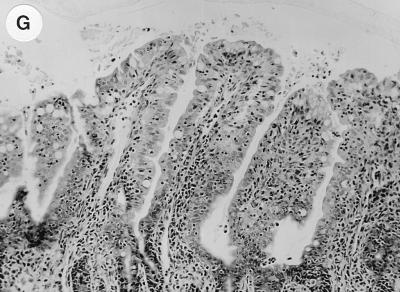
Histological sections showing cross sections (magnification, ×40) of loop-associated tissues stained with hematoxylin and eosin. (A) Uninfected loop showing normal villus height. (B) Loop infected with wild-type S. typhimurium HWSH. Large numbers of inflammatory cells are highlighted in the lamina propria (small arrow) and on top of damaged villi in the lumen (small arrow). Dilation of the crypts suggests an active secretory response (large arrow). Identical histology was also observed with wild-type strains TML and SL1344. (C) Loop infected with S. typhimurium SL1344 showing large numbers of inflammatory cells (arrow), predominantly neutrophils (architecture as described above). (D) Loop infected with S. typhimurium SL1344 msbB. Histological changes were consistent with those described above for tissues infected with wild-type strains. Similar effects were observed with S. typhimurium SL1344 ssrA. (E) Loop infected with S. typhimurium TML. There is extensive villus damage and a large number of inflammatory cells in the lamina propria (arrow). (F) Loop infected with S. typhimurium TML aroA. Villus shortening and architectural damage are less severe than in panel E, and significantly less crypt dilation is apparent. Tissue taken from loops infected with either SL1344 aroA or HWSH aroA displayed identical histology. (G) Submucosal loop infected with S. typhimurium SL1344 invA.
Histopathology of rabbit ileal loops infected with S. typhimurium invA and the different S. typhimurium aroA mutants.
Intestinal tissue infected with S. typhimurium aroA strains exhibited some histopathological similarities to intestinal tissue infected with wild-type S. typhimurium (Fig. 3E and F). However, there was a discernible difference in the degree of tissue damage to the villi. The villus structures were shortened but retained reasonable tissue architecture, unlike the flattened mucosa exhibited after wild-type S. typhimurium infection. Large numbers of PMNs were present within the lamina propria of S. typhimurium aroA-infected tissue, demonstrating an ongoing inflammatory response. Loops infected with S. typhimurium aroA strains showed significantly less crypt dilatation than wild-type-infected tissue. No loops infected with S. typhimurium invA showed significant and reproducible differences from uninfected control loops (Fig. 3G).
Analysis of fluid from rabbit ileal loops.
The fluids collected from loops infected with different S. typhimurium derivatives were subjected to biochemical analysis (Table 2). The total protein content of the fluid was high, reflecting the increased cellular content and, presumably, leakage of plasma proteins from damaged capillaries, in contrast to the secretory nature of the fluid from CT-treated loops. There was also a high hemoglobin content in the fluid, reflecting the visible presence of blood. Bicarbonate concentrations in the fluids from S. typhimurium-infected tissue were within the normal range for rabbit blood (16.2 to 31.8 mM). Fluid pH ranged from 4.5 to 5.2, in contrast to the alkaline pH of 8 in CT-induced fluid, reflecting the high bicarbonate levels in the CT-induced fluids.
TABLE 2.
Analysis of fluid production in Salmonella-infected rabbit ileal loops
| Strain or agent inoculated | Positive loops (n = 4)a | Mean fluid volb | Mean bicarbonate concnc | pHd | Avg protein concne
|
|
|---|---|---|---|---|---|---|
| Total | Hemoglobinf | |||||
| CT | 4 | 20 (15–25) | 90.1 (70–118) | 8 | 0.3 | 0.1 |
| TML (WT)g | 4 | 2.5 (1.5–3.5) | 30.6 (12–46) | 5 | 1.8 | 0.7 |
| HWSH (WT) | 4 | 1.75 (0.5–3) | 26.3 (18–39) | 5 | 3.0 | 0.5 |
| SL1344 (WT) | 4 | 3.75 (3–4.5) | 30 (18–42) | 5.2 | 2.4 | 0.35 |
| TML aroA | 0 | No fluid to test | ||||
| HWSH aroA | 0 | No fluid to test | ||||
| SL1344 aroA | 0 | No fluid to test | ||||
| SL1344 msbB | 4 | 1 (0.5–1.5) | 21 (7.4–35) | 4.5 | 3.5 | 0.8 |
| SL1344 ssrA | 4 | 4.5 (3–6) | 25 (11–39) | 5 | 3.5 | 0.9 |
| SL1344 invA | 0 | No fluid to test | ||||
| PBS control | 0 | NDh | ||||
Loops showing fluid present in the lumen when opened postmortem were considered positive.
Total volume in milliliters collected from each loop divided by the number of positive loops (numbers in parentheses are ranges).
Total bicarbonate content (in millimolar units) of the fluids present in the loops divided by the number of positive loops (numbers in parentheses are ranges).
pH of fluids in the loop. These were found to be consistent for each strain tested.
Total protein content in the fluids (in milligrams per milliliter) divided by the number of positive loops.
Total hemoglobin content in the fluids (in milligrams per milliliter) divided by the number of positive loops.
WT, wild type.
ND, not done.
Microbiological analysis of infected rabbit ileal loops.
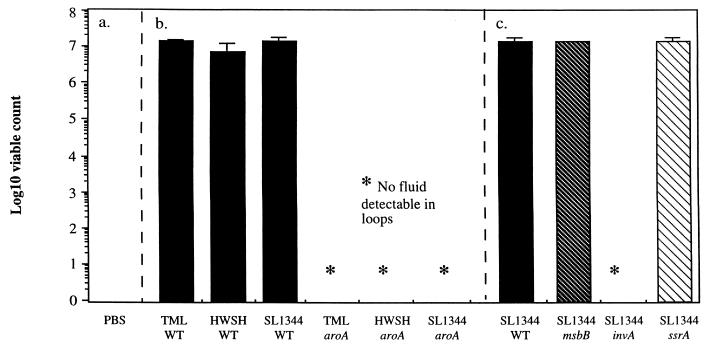
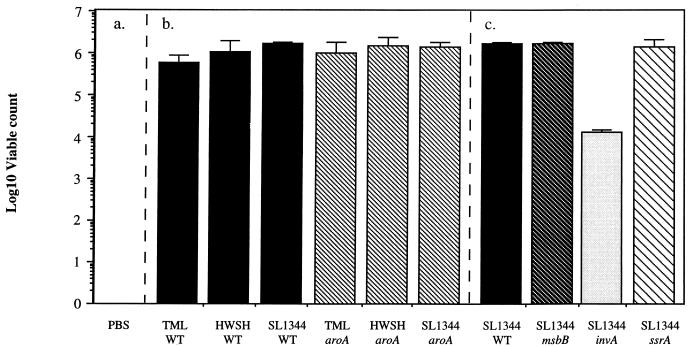
There were large numbers of wild-type and mutant S. typhimurium strains within all loops where fluid accumulation occurred (Fig. 4). Further, viable S. typhimurium strains could be recovered from homogenized intestinal mucosa from all infected loops (Fig. 5). The numbers of S. typhimurium mutants and wild-type controls present in the tissues were similar in all cases, with the exception of the S. typhimurium invA mutant. Counts of these bacteria from infected loops were significantly below those of the corresponding wild-type S. typhimurium control.
FIG. 4.
S. typhimurium viable counts in fluid recovered from ileal loops. (a) PBS control. (b) Viable count of bacteria in the fluid recovered from loops infected with one of three wild-type S. typhimurium strains: TML, HWSH, or SL1344. (c) Viable count of bacteria in the fluid recovered from loops infected with S. typhimurium msbB, invA, or ssrA. These mutants were all created in wild-type S. typhimurium SL1344, which is included for comparison. Error bars represent standard deviations. WT, wild type.
FIG. 5.
S. typhimurium viable counts from infected ileal loop tissue homogenates. (a) PBS control loop was not cross-infected by S. typhimurium during the course of the experiment. (b) Viable count of bacteria in loops infected with wild-type S. typhimurium strain TML, HWSH, or SL1344 or the equivalent aroA mutant. (c) Viable count of bacteria in loops infected with S. typhimurium msbB, invA, or ssrA. These mutants were all created in wild-type S. typhimurium SL1344, which is included for comparison. Error bars represent standard deviations. WT, wild type.
DISCUSSION
Here we demonstrate that genes associated with S. typhimurium virulence in the murine model of typhoid fever show significant differences in their influences on S. typhimurium virulence in the rabbit ileal loop model of gastroenteritis. Wild-type strains of S. typhimurium SL1344, TML, and HWSH and mutants SL1344 ssrA and SL1344 msbB exhibited similar patterns of pathogenicity in the rabbit ileum with respect to levels of fluid secretion, histological damage, inflammation intensity, and PMN infiltration. These data suggest that SPI-2 and a fully toxic lipid A component of endotoxin are not required for the induction of fluid accumulation in this model. Further, these data indicate that these genes are not essential for the induction of diarrhea in gastroenteritis. In contrast, S. typhimurium TML aroA, HWSH aroA, and SL1344 aroA each induced significant tissue inflammation but no significant fluid accumulation. In addition, S. typhimurium SL1344 invA was found to be essentially avirulent in this assay, inducing no significant fluid accumulation or tissue damage, which is in agreement with published studies showing that SPI-1 function is required for fluid secretion and tissue inflammation in calf ileal loops (38, 39).
Giannella et al. and Gots et al. (11–15) observed a close correlation between fluid secretion in rabbit ileal loops and the virulence for humans of gastroenteritis-causing S. typhimurium. Stephen et al. (4, 30, 31, 36, 37) showed that strains of S. typhimurium in the rabbit ileal loop model could be separated into three different groups: invasive and diarrheagenic, invasive and nondiarrheagenic, and noninvasive and nondiarrheagenic. Strains SL1027 and LT-7 were invasive but nondiarrheagenic and failed to cause a net secretion of chloride in Ussing chambers. This is in contrast to strains isolated from cases of human gastroenteritis, including S. typhimurium TML (30), which were found to be invasive in the rabbit ileal loop, to cause fluid secretion, and to induce chloride secretion and depressed sodium absorption in Ussing chambers. S. typhimurium TML was also shown to cause cell damage at villus tips and to induce a large influx of PMNs into the mucosa. Structural damage to villus tips led to shortened villi, which were suspected to contribute to diarrhea by altering absorption/secretion ratios (30, 31). Fluid secretion in rabbit loops infected with wild-type S. typhimurium was not observed in the absence of leukocytes, but this leukocyte influx, by itself, did not induce fluid secretion (36, 37).
Fluid accumulation induced by wild-type S. typhimurium was associated with damaged and shortened villi. Although S. typhimurium infection had reduced the villi to almost flattened mucosa by 18 h, most villi retained their structural integrity during S. typhimurium aroA infection, although some shortened villi were detectable. Both S. typhimurium wild type and aroA derivatives induced large influxes of PMNs into the submucosal tissue and lamina propria and onto the surface of the mucosa. Thus, S. typhimurium aroA derivatives behave like the invasive, non-fluid-accumulating strains described by Stephen et al. (30). Loops infected with S. typhimurium invA, which is poorly invasive in tissue culture cells, had normal-looking villi and no inflammatory infiltrate, whereas S. typhimurium SL1344 ssrA, required for virulence beyond the mucosa in mice, was not impaired in its ability to induce infiltration and damage.
Thus, these data indicate that the main determinant of fluid accumulation within the rabbit ileal loop could be invasion-induced damage to the villus structure. Wallis et al. (37), after performing ileal loop experiments in cattle, proposed that villus height reduction resulted in the loss of the upper absorptive region of the villus, which in turn led to physiological secretion. They also proposed that the regeneration of damaged villi could give rise to hypersecretion. This is not observed in the S. typhimurium aroA-infected loops, as villus damage is apparently not severe enough to reduce the absorptive capability of the ileum or elicit hypersecretion due to villus regeneration. The other main histological difference observed in our study was that in ileal tissue infected with wild-type S. typhimurium, the crypts were markedly dilated compared to the crypts in S. typhimurium aroA-infected tissue, where no crypt dilation was observed. In view of the fact that fluid accumulation was observed only in wild-type S. typhimurium-infected loops, crypt secretion may be another contributor to fluid accumulation in this model. The fact that S. typhimurium aroA derivatives failed to induce fluid accumulation may be due to the large PMN infiltrate within the tissues controlling infection before more extensive villus damage can occur.
One aim of this study was to determine which mutations might be incorporated into candidate live oral S. typhimurium vaccine strains suitable for use in humans. It is essential that any candidate live vaccine be incapable of causing diarrhea. By using the rabbit ileal loop, we have demonstrated that the aroA and invA (SPI-1) mutations are appropriate candidate genes for this purpose, whereas in this assay ssrA (SPI-2) and msbB gave no evidence of producing an attenuating effect. The inability of S. typhimurium invA mutants to induce local inflammation may suggest that they are poorly immunogenic, but this can only be evaluated properly in volunteer studies.
ACKNOWLEDGMENTS
We acknowledge the technical support and advice of the Biomedical Services staff at Leicester University.
G.D. and P.E. were supported by a program grant from the Wellcome Trust. J.K. was a Royal Society University Research Fellow at the University of Leicester. S.H. acknowledges support from the Wellcome Trust (047407/Z/96).
REFERENCES
- 1.Bradford M M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem. 1976;72:248–254. doi: 10.1016/0003-2697(76)90527-3. [DOI] [PubMed] [Google Scholar]
- 2.Chatfield S N, Strahan K, Pickard D, Charles I G, Hormaeche C E, Dougan G. Evaluation of Salmonella typhimurium strains harbouring defined mutations in htrA and aroA in the murine salmonellosis model. Microb Pathog. 1992;12:145–151. doi: 10.1016/0882-4010(92)90117-7. [DOI] [PubMed] [Google Scholar]
- 3.Chopra A K, Houston C W, Peterson J W, Prasad R, Mekalanos J J. Cloning and expression of the Salmonella enterotoxin gene. J Bacteriol. 1987;169:5095–5100. doi: 10.1128/jb.169.11.5095-5100.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Clarke G J, Qui G M, Wallis T S, Starkey W G, Collins J, Spencer A J, Haddon S J, Osborne M P, Worton K J, Stephen J. Expression of an antigen in strains of Salmonella typhimurium which reacts with antibodies to cholera toxin. J Med Microbiol. 1988;25:139–146. doi: 10.1099/00222615-25-2-139. [DOI] [PubMed] [Google Scholar]
- 5.Collins F M. Vaccines and cell-mediated immunity. Bacteriol Rev. 1974;38:371–402. doi: 10.1128/br.38.4.371-402.1974. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Collins F M, Mackaness G B, Blanden R V. Infection and immunity in experimental salmonellosis. J Exp Med. 1966;124:601–619. doi: 10.1084/jem.124.4.601. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Dougan G. Genetics as a route toward mucosal vaccine development. In: Miller V L, Kaper J B, Portnoy D A, Isberg R R, editors. Molecular genetics of bacterial pathogenesis. Washington, D.C: ASM Press; 1994. pp. 491–506. [Google Scholar]
- 8.Finlay B B, Falkow S. Salmonella interaction with polarised human intestinal Caco-2 epithelial cells. J Infect Dis. 1990;162:1096–1106. doi: 10.1093/infdis/162.5.1096. [DOI] [PubMed] [Google Scholar]
- 9.Galan J E, Curtiss R. Cloning and molecular characterization of genes whose products allow Salmonella typhimurium to penetrate tissue culture cells. Proc Natl Acad Sci USA. 1989;86:6383–6387. doi: 10.1073/pnas.86.16.6383. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Galyov E E, Wood M W, Rosqvist R, Mullan P B, Watson P R, Hedges S, Wallis T S. A secreted effector protein of Salmonella dublin is translocated into eukaryotic cells and mediates inflammation and fluid secretion in infected ileal mucosa. Mol Microbiol. 1997;25:903–912. doi: 10.1111/j.1365-2958.1997.mmi525.x. [DOI] [PubMed] [Google Scholar]
- 11.Giannella R A. Importance of the intestinal inflammatory reaction in salmonella-mediated intestinal secretion. Infect Immun. 1979;23:140–145. doi: 10.1128/iai.23.1.140-145.1979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Giannella R A, Formal S B, Dammin G J, Collins J. Pathogenesis of salmonellosis. Studies of fluid secretion, mucosal invasion and morphological reaction in the rabbit ileum. J Clin Investig. 1973;52:441–453. doi: 10.1172/JCI107201. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Giannella R A, Gots R E, Charney A N, Greenough W B, Formal S B. Pathogenesis of Salmonella mediated intestinal fluid secretion. Gastroenterology. 1975;69:1238–1245. [PubMed] [Google Scholar]
- 14.Giannella R A, Rout W R, Formal S B. Effect of indomethacin on intestinal water transport in salmonella-infected rhesus monkeys. Infect Immun. 1977;17:136–139. doi: 10.1128/iai.17.1.136-139.1977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Gots R E, Formal S B, Giannella R A. Indomethacin inhibition of Salmonella typhimurium, Shigella flexneri and cholera-mediated rabbit ileal secretion. J Infect Dis. 1974;130:280–284. doi: 10.1093/infdis/130.3.280. [DOI] [PubMed] [Google Scholar]
- 16.Hoiseth S K, Stocker B A. Aromatic-dependent Salmonella typhimurium are non-virulent and effective as live vaccines. Nature. 1981;291:238–239. doi: 10.1038/291238a0. [DOI] [PubMed] [Google Scholar]
- 17.Hone D M, Harris A M, Chatfield S, Dougan G, Levine M M. Construction of genetically defined double aro mutants of Salmonella typhi. Vaccine. 1991;9:810–816. doi: 10.1016/0264-410x(91)90218-u. [DOI] [PubMed] [Google Scholar]
- 18.Hook E W. Salmonella species (including typhoid fever) In: Mandell G L, Douglas R G, Bennett J E, editors. Principles and practice of infectious diseases. 2nd ed. London, England: Churchill Livingstone; 1988. p. 1256. [Google Scholar]
- 19.Hormaeche C E, Khan A C M, Mastroeni P, Villareal B, Dougan G, Roberts M, Chatfield S N. Salmonella vaccines: mechanisms of immunity and their use as carriers of recombinant antigens. In: Ala’Aldeen D, Hormaeche C E, editors. Molecular and clinical aspects of bacterial vaccine development. Chichester, England: John Wiley; 1994. pp. 119–153. [Google Scholar]
- 20.Hornick R B, Greisman S E, Woodward T E, DuPont H L, Dawkins A T, Snyder M J. Typhoid fever: pathogenesis and immunologic control. N Engl J Med. 1970;283:686–691. doi: 10.1056/NEJM197009242831306. [DOI] [PubMed] [Google Scholar]
- 21.Hornick R B, Greisman S E, Woodward T E, DuPont H L, Dawkins A T, Snyder M J. Typhoid fever: pathogenesis and immunological control. 2. N Engl J Med. 1970;283:739–746. doi: 10.1056/NEJM197010012831406. [DOI] [PubMed] [Google Scholar]
- 22.Jepson M A, Collares-Buzato C B, Clark M A, Hirst B H, Simmons N L. Rapid disruption of epithelial barrier function by Salmonella typhimurium is associated with structural modification of intercellular junctions. Infect Immun. 1995;63:356–359. doi: 10.1128/iai.63.1.356-359.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Khan S A, Everest P, Servos S, Foxwell N, Zahringer U, Brade H, Rietschel E T, Dougan G, Charles I G, Maskell D J. A lethal role for lipid A in Salmonella infections. Mol Microbiol. 1998;29:571–579. doi: 10.1046/j.1365-2958.1998.00952.x. [DOI] [PubMed] [Google Scholar]
- 24.Mastroeni P, Villareal B, Hormaeche C E. Role of T-cells, TNF and IFN in recall of immunity to oral challenge with virulent salmonellae in mice vaccinated with live attenuated aro Salmonella vaccines. Microb Pathog. 1992;13:477–491. doi: 10.1016/0882-4010(92)90014-f. [DOI] [PubMed] [Google Scholar]
- 25.Miles A A, Misra S S, Irwin J O. The estimation of the bactericidal action of the blood. J Hyg Camb. 1938;38:732–749. doi: 10.1017/s002217240001158x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Mills D M, Bajaj V, Lee C A. A 40 kb chromosomal fragment encoding Salmonella typhimurium invasion genes is absent from the corresponding region of the E. coli K12 chromosome. Mol Microbiol. 1995;15:749–759. doi: 10.1111/j.1365-2958.1995.tb02382.x. [DOI] [PubMed] [Google Scholar]
- 27.O’Callaghan D, Maskell D, Liew F Y, Easmon C S F, Dougan G. Characterization of aromatic- and purine-dependent Salmonella typhimurium: attenuation, persistence, and ability to induce protective immunity in BALB/c mice. Infect Immun. 1988;56:419–423. doi: 10.1128/iai.56.2.419-423.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Roberts M, Chatfield S N, Dougan G. Salmonella as carriers of heterologous antigens. In: O’Hagan D T, editor. Novel delivery systems for oral vaccines. Boca Raton, Fla: CRC Press; 1994. pp. 27–58. [Google Scholar]
- 29.Shea J E, Hensel M, Gleeson C, Holden D W. Identification of of a virulence locus encoding a second type III secretion system in Salmonella typhimurium. Proc Natl Acad Sci USA. 1996;93:2593–2597. doi: 10.1073/pnas.93.6.2593. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Stephen J, Wallis T S, Starkey W G, Candy D C A, Osborne M P, Haddon S. Salmonellosis: in retrospect and prospect. Ciba Found Symp. 1985;112:175–192. [PubMed] [Google Scholar]
- 31.Stephen J, Amin I, Douce G R. Experimental Salmonella typhimurium-induced gastroenteritis. In: Cabello F, Hormaeche C, Mastreoni P, Bonina L, editors. Biology of Salmonella. New York, N.Y: Plenum Press; 1993. pp. 199–209. [Google Scholar]
- 32.Stocker B A D. Auxotrophic Salmonella typhi as a live vaccine. Vaccine. 1988;6:141–145. doi: 10.1016/s0264-410x(88)80017-3. [DOI] [PubMed] [Google Scholar]
- 33.Tacket C, Levine M M. Typhoid vaccines—old and new. In: Ala’Aldeen D, Hormaeche C E, editors. Molecular and clinical aspects of bacterial vaccine development. Chichester, England: John Wiley; 1994. pp. 155–178. [Google Scholar]
- 34.Takeuchi A. Electron microscope studies of experimental Salmonella infection. 1. Penetration into the intestinal epithelium by Salmonella typhimurium. Am J Pathol. 1967;50:109–136. [PMC free article] [PubMed] [Google Scholar]
- 35.Vancott J L, Staats H F, Pascual D W, Roberts M, Chatfield S N, Yamamoto M, Coste M, Carter P B, Kiyono H, McGhee J R. Regulation of mucosal and systemic antibody responses by T helper subsets, macrophages, and derived cytokines following oral immunisation with live recombinant Salmonella. J Immunol. 1996;156:1504–1514. [PubMed] [Google Scholar]
- 36.Wallis T S, Hawker R J H, Candy D C A, Qui G-M, Clarke G J, Worton K J, Osborne M P, Stephen J. Quantification of the leucocyte influx into rabbit ileal loops induced by strains of Salmonella typhimurium of different virulence. J Med Microbiol. 1989;30:149–156. doi: 10.1099/00222615-30-2-149. [DOI] [PubMed] [Google Scholar]
- 37.Wallis T S, Starkey W G, Stephen J, Haddon S J, Osbourne M P, Candy D C A. The nature and role of mucosal damage in relation to Salmonella typhimurium-induced fluid secretion in the rabbit ileum. J Med Microbiol. 1986;22:39–49. doi: 10.1099/00222615-22-1-39. [DOI] [PubMed] [Google Scholar]
- 38.Watson P R, Paulin S M, Bland A P, Jones P W, Wallis T S. Characterization of intestinal invasion by Salmonella typhimurium and Salmonella dublin and effect of a mutation in the invH gene. Infect Immun. 1995;63:2743–2754. doi: 10.1128/iai.63.7.2743-2754.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Watson P R, Galyov E E, Paulin S M, Jones P W, Wallis T S. Mutation of invH, but not stn, reduces Salmonella-induced enteritis in cattle. Infect Immun. 1998;66:1432–1438. doi: 10.1128/iai.66.4.1432-1438.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Worton K J, Candy D C A, Wallis T S, Clarke G J, Osbourne M P, Haddon S J, Stephen J. Studies on early association of Salmonella typhimurium with intestinal mucosa in vivo and in vitro: relationship to virulence. J Med Microbiol. 1989;29:283–294. doi: 10.1099/00222615-29-4-283. [DOI] [PubMed] [Google Scholar]