Abstract
The murine immune response to a pulmonary mycobacterial infection is slow to develop, allowing bacterial numbers to increase in the lung for several weeks after infection. We sought to enhance the protective immune response induced during Mycobacterium bovis BCG infection by administering an antibody that blocks the interaction of CTLA-4 with its ligands, CD80 and CD86. We found that injection of anti-CTLA-4 monoclonal antibody (MAb) greatly enhanced and accelerated the immune response, as measured by increased cellularity of the draining mediastinal lymph nodes, and enhanced antigen-inducible proliferation and gamma interferon production by mediastinal lymphocytes in vitro. However, despite the apparently enhanced immune response in the mediastinal lymph node following treatment with anti-CTLA-4 MAb, there was no improvement in clearance of mycobacteria in the lungs, liver, or spleen. Examination of the primary site of infection, the lung, revealed that CTLA-4 blockade had no effect on the number or function of lymphocytes infiltrating the infected lung tissue. Taken together, these data suggest that in vivo CTLA-4 blockade enhances mycobacterial-infection-induced lymphocyte expansion and effector cell cytokine production in the draining lymph node but does not alter the number or function of lymphocytes at the primary site of infection and therefore does not lead to enhanced clearance of the infection.
Interaction between the T-cell molecule CTLA-4 and its ligands, CD80 and CD86, on the surfaces of antigen-presenting cells has been shown to negatively regulate T-cell activation (16, 17, 28, 29). CTLA-4 is only expressed at high levels on the surfaces of activated T cells, with maximal expression occurring 48 to 72 h after T-cell receptor-mediated activation (20). By blocking the interaction between CTLA-4 and its ligands in vitro, with either whole anti-CTLA-4 monoclonal antibody (MAb) or Fab fragments, it is possible to enhance antigen-specific T-cell proliferative responses (28). Furthermore, recent in vivo studies have shown that CTLA-4 blockade can enhance antitumor responses (19), the Th2 immune response induced during nematode infection (21), and resistance to the intracellular pathogen Leishmania donovani (23). These findings suggest the exciting possibility of CTLA-4 blockade providing an immunotherapeutic strategy to enhance immunity against other infectious diseases.
We chose to study the effects of CTLA-4 blockade on the Th1 immune response induced during a lung infection with the intracellular pathogen Mycobacterium bovis bacille Calmette Guérin (BCG). Protective immunity against mycobacterial infections is considered to be mediated by CD4+ and CD8+ T cells (9, 18, 22, 24). CD4+ Th1 cells are considered to be the major source of gamma interferon (IFN-γ) (25), a cytokine that activates antibacterial effector functions in infected macrophages (7) and is essential in protection against mycobacteria (3, 8, 13). CD8+ T cells can also act as a source of IFN-γ (26) and may further contribute to protection by lysing infected alveolar macrophages (27). However, the murine protective immune response to a pulmonary mycobacterial infection is slow to develop, and bacterial numbers continue to increase in the lungs for several weeks after infection (4). We postulated that by blocking the interaction between CTLA-4 and its ligands during a mycobacterial infection, we could enhance the protective immune response which is induced and improve containment and clearance of the bacteria.
To investigate the effects of CTLA-4 blockade during a mycobacterial lung infection, mice were challenged intranasally with M. bovis BCG and treated with a MAb that blocked the interaction between CTLA-4 and CD80 and CD86. The effect of CTLA-4 blockade on antigen-specific T-cell responses was followed in vivo and in vitro, as was the ability of the immune response to contain and clear the invading microorganisms.
MATERIALS AND METHODS
BCG.
Aliquots of BCG Pasteur strain 1173P2 (11) were prepared from logarithmically growing cultures and frozen at −70°C in phosphate-buffered saline (PBS) plus 0.05% Tween 80. Immediately before use, the stocks were defrosted and briefly sonicated to dissociate clumped mycobacteria. To enumerate viable mycobacteria, samples were plated on Middlebrook 7H11 agar with 10% oleic acid-albumin-dextrose-catalase enrichment.
Intranasal infection.
Inbred 6- to 9-week-old male C57BL/6 mice were anesthetized by intraperitoneal injection of ketamine and xylazine to facilitate intranasal infection with 50 μl of BCG in PBS plus 0.05% Tween 80. The mice were sacrificed at 1, 2, 3, 4, 6, and 10 weeks postinfection, and the lungs, livers, and spleens were harvested for counts of viable bacteria. Blood was collected for analysis of circulating anti-CTLA-4 MAb levels, and draining mediastinal and nondraining inguinal lymph nodes were collected for analysis of antigen-specific T-cell-dependent proliferation and cytokine production. All experiments were approved by the Wellington School of Medicine Animal Ethics Committee and were in accordance with the University of Otago (New Zealand) guidelines.
Antibody treatment.
Anti-CTLA-4 MAb (4F10; hamster immunoglobulin G [IgG]) was purified from hybridoma supernatant with protein G affinity columns and was stored in PBS. Mice were injected intraperitoneally with one dose of 1 mg of anti-CTLA-4 MAb at weekly intervals to block the interaction between CTLA-4 and its ligands, CD80 and CD86. Antibody treatment started at day 0 of the BCG infection. The concentration of circulating anti-CTLA-4 MAb was directly measured by a sandwich enzyme-linked immunosorbent assay (ELISA) with plate-bound murine CTLA-4 (mCTLA-4)–Ig to capture, and anti-hamster IgG-biotin to detect, anti-CTLA-4 MAb in the serum. Anti-CTLA-4 MAb was found in the sera of all of the mice up to 2 weeks postinfection, in 88% of the mice at weeks 3 and 4 after infection, and in 56% of the mice at weeks 6 and 10 postinfection.
In vitro culture conditions for proliferation assays and cytokine production.
In vitro proliferative responses to mycobacterial antigens were measured by culturing total lymph node cells (4 × 105) from infected mice with macrophages (2 × 104) that had been pulsed overnight with BCG (2 × 105) in antibiotic-free medium. Macrophages were obtained from peritoneal lavages of uninfected male C57BL/6 mice. The lymphocytes and pulsed macrophages were incubated together in 96-well flat-bottom microplates with Iscove’s modified Dulbecco’s medium supplemented with 5% fetal bovine serum, penicillin and streptomycin, 2-mercaptoethanol, and l-glutamine for 72 h at 37°C and 5% CO2. [3H]thymidine (0.5 μCi/well) was added for the last 10 h of culture and then harvested, and thymidine incorporation was measured in a liquid scintillation counter (Wallac, Turku, Finland). To assay for mycobacterial-antigen-specific cytokine production, supernatants from the cultures of lymphocytes and infected macrophages were harvested after 72 h and kept frozen at −20°C until analysis. As a control for in vitro proliferative capacity, 4 × 105 lymphocytes were cultured in the presence of 100 U of recombinant human interleukin 2 (IL-2)/ml on 96-well flat-bottom microtiter plates coated with 10 μg of anti-CD3 MAb (2C11)/ml. The cells were pulsed with [3H]thymidine (0.5 μCi/well) for the last 10 h of the 48-h culture and then harvested, and thymidine incorporation was measured. For cytokine production assays, supernatants from lymphocytes restimulated in vitro with anti-CD3 MAb were harvested after 48 h and kept frozen at −20°C until analysis.
ELISA for the detection of cytokines.
A sandwich ELISA was used to measure cytokines, using R4 6A2 and XMG-D6-biotin conjugate (anti-IFN-γ), TRFK5 and TRFK4-biotin conjugate (anti-IL-5), 11B11 and BVD6-24G2-biotin conjugate (anti-IL-4), or JES5-2A5 and SXC-1-biotin conjugate (anti-IL-10) as capture and detecting reagents, respectively. Polyvinyl chloride 96-well plates were coated overnight with capture MAb at 4°C and then blocked for 1 h with 10% bovine serum albumin in PBS. Appropriate dilutions of test supernatants and mIFN-γ, mIL-5, mIL-4, or mIL-10 internal standards were added and incubated for 2 h at room temperature. Detecting antibody and then peroxidase-labelled streptavidin were added for 1 h at room temperature. Freshly prepared 1 mM ABTS [2,2′-azinobis(3-ethylbenzthiazoline-6-sulfonic acid)] in citrate phosphate buffer (pH 9.2) with 0.03% H2O2 was added to each well to develop the reaction. The reaction was stopped by adding 2 mM NaN3, and the plates were read at 415 nm with a Benchmark microplate reader (Bio-Rad Laboratories, Richmond, Calif.). Cytokine production is expressed in Genzyme units per milliliter or nanograms per milliliter; the limits of detection were as follows: IFN-γ, 30 U/ml; IL-5, 60 U/ml; IL-4, 0.2 U/ml; and IL-10, 0.5 ng/ml.
Flow cytometry.
Analysis of mediastinal lymphocytes was carried out by staining cells with anti-CD4-phycoerythrin (Pharmingen), anti-CD8a-fluorescein isothiocyanate (Pharmingen), and anti-B220 (6B2-biotin) with streptavidin-phycoerythrin (Pharmingen). 2.4G2 (10 μg/ml) was used to inhibit FcγRII-mediated uptake. Flow cytometric analysis was performed on a FACSort (Becton Dickinson) with CellQuest software.
RNA isolation and cDNA preparation.
RNA was isolated from lymph node and lung tissue with TRIzol (Gibco-BRL/Life Technologies). RNA was quantified with GeneQuant (Pharmacia, LBK Biochrom, England), and 1 μg of total RNA was used in the cDNA reaction. The cDNA reaction was carried out at 37°C for 1 h, using 200 U of Moloney murine leukemia virus reverse transcriptase (Gibco-BRL/Life Technologies) and 0.5 μg of oligo(dT)12–18 primer (Gibco-BRL/Life Technologies).
Quantitative PCR.
Primers and probes were designed with the Primer Express version 1.0 software (Perkin-Elmer [PE] Applied Biosystems, Foster City, Calif.). The primers were synthesized by Life Technologies, and the probes by PE Applied Biosystems. The probes were modified to incorporate a reporter dye at the 5′ end (6-carboxy-fluorescein [FAM] or tetrachloro-6-carboxy-fluorescein [TET]) and a quencher at the 3′ end (6-carboxy-tetramethyl-rhodamine [TAMRA]). The sequences of the oligonucleotides used are described in Table 1. Reactions were carried out in the Prism 7700 sequence detector (PE Applied Biosystems). The reactions were set up with the TaqMan core reagents (PE Applied Biosystems) according to the manufacturer’s instructions. Five millimolar MgCl2 was used in the IFN-γ and β2-microglobulin (β2m) reactions, and 7 mM MgCl2 was used in the tumor necrosis factor alpha (TNF-α) reactions. The PCR cycling conditions for IFN-γ and β2m reactions were 94°C for 2 min, followed by 35 cycles of 94°C for 15 s, 58°C for 30 s, and 72°C for 30 s. For TNF-α reactions, the conditions were 94°C for 2 min and then 35 cycles of 94°C for 15 and 60°C for 1 min.
TABLE 1.
Primer and probe sequences used for quantitative PCR
| mRNA | Primersa | Probe |
|---|---|---|
| β2m | F-5′ TGACCGGCTTGTATGCTATC 3′ | 5′ TET-TATACTCACGCCACCCACCGGAGAA-TAMRA 3′ |
| R-5′ CAGTGTGAGCCAGGATATAG 3′ | ||
| IFN-γ | F-5′ CCTCCTGCGGCCTAGCTC 3′ | 5′ 6FAM-CACACTGCATCTTGGCTTTGCAGCTC-TAMRA 3′ |
| R-5′ GTAACAGCCAGAAACAGCCATG 3′ | ||
| TNF-α | F-5′ AGGCGGTGCCTATGTCTCAG 3′ | 5′ 6FAM-CTCTTCTCATTCCTGCTTGTGGCAGGG-TAMRA 3′ |
| R-5′ GGGTCTGGGCCATAGAACTG 3′ |
F, forward; R, reverse.
DNA standards were made for each of the cytokines, and these were serially diluted and included with each PCR. Data analysis was carried out with Prism sequence detection systems version 1.6.3 software (PE Applied Biosystems). Amplifications were performed in duplicate, and the amount of DNA in the samples was calculated from the standard curve. All results were normalized to β2m to compensate for differences in the amount of cDNA in the samples. β2m was present at >1.5 × 108 copies/μl of cDNA in all samples. The TNF-α standard contained 1011 copies/μl of cDNA, and the IFN-γ standard contained 1010 copies/μl of cDNA.
Determination of viable bacterial counts.
Tissue homogenates were prepared in 1% Tween 80 in sterile distilled water, and samples were always kept on ice during preparation. Tenfold serial dilutions of homogenates were plated on Middlebrook 7H11 agar with 10% OADC supplement and incubated for 21 days at 37°C with 9% CO2. Colonies were counted and expressed as CFU per organ.
Histological analysis.
Tissues from infected mice were fixed in 10% phosphate-buffered formal saline for 24 h and embedded in paraffin wax. Sections (3 μm) were cut and stained with hematoxylin and eosin. The stained sections were examined by light microscopy.
RESULTS
CTLA-4 blockade increases total lymphocyte numbers in draining lymph nodes of BCG-infected mice.
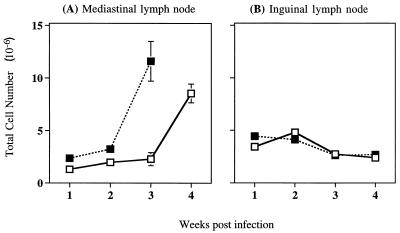
To investigate whether blockade of CTLA-4 signalling in vivo would alter T-cell proliferation induced by infection with BCG, we compared the total number of cells in the draining lymph nodes of infected mice that were treated with weekly injections of anti-CTLA-4 neutralizing MAb with those of mice that were similarly infected with BCG but were not treated with anti-CTLA-4 MAb. To establish the infection in the lung, the mice were intranasally infected with 5 × 105 BCG. The mediastinal lymph node, which drains the lung, normally enlarges following intranasal infection with BCG. Infected mice receiving anti-CTLA-4 MAb exhibited an enhanced increase in the number of lymphocytes obtained from the mediastinal lymph node as early as 1 week after infection (Fig. 1A). The greatest increase in response to anti-CTLA-4 MAb treatment occurred after 3 weeks of BCG infection and reflected a fivefold increase in mediastinal cell numbers over that observed in untreated, BCG-infected mice. At 4 weeks after infection, total mediastinal cell numbers in the control group and the anti-CTLA-4 MAb-treated group appeared to have reached the same level, although by visual inspection mediastinal lymph nodes from mice treated with anti-CTLA-4 MAb were always substantially larger. At this time the lymph nodes had become fibrous, and single-cell suspensions were difficult to obtain. Therefore, lymphocyte numbers obtained at later time points did not always reflect the size of the lymph node (data not shown).
FIG. 1.
Treatment with anti-CTLA-4 MAb increases total lymphocyte numbers in the draining mediastinal lymph nodes of BCG-infected mice. C57BL/6 mice were intranasally infected with 5 × 105 BCG and either treated with anti-CTLA-4 MAb (solid squares) or left untreated (open squares). The draining mediastinal lymph nodes (A) or nondraining inguinal lymph nodes (B) were collected at the times indicated. Single-cell suspensions were prepared from individual lymph nodes, and total lymphocytes were counted. The values represent mean cell numbers from four mice ± standard error, and the results are representative of three separate experiments.
To ensure that changes observed with anti-CTLA-4 MAb treatment were antigen induced, total lymphocyte numbers in the inguinal lymph nodes were also analyzed (Fig. 1B). This lymph node was not draining sites of BCG infection and made a useful internal control to observe what effect treatment with anti-CTLA-4 neutralizing MAb had in the absence of antigen. Anti-CTLA-4 MAb treatment had no effect on total lymphocyte numbers in the nondraining inguinal lymph nodes. When C57BL/6 mice were infected with a 10-fold-lower dose of BCG (5 × 104 CFU), the trend was the same as that shown for infection with 5 × 105 BCG. Anti-CTLA-4 MAb treatment led to a threefold increase in mediastinal lymph node cell numbers, with the greatest increase occurring 3 weeks postinfection (data not shown).
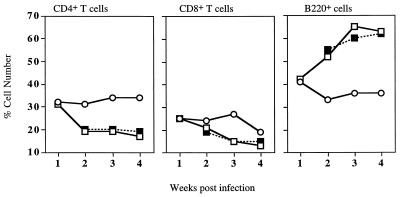
To determine whether the observed increase in mediastinal lymphocytes during CTLA-4 blockade was due to increased expansion or accumulation of one specific lymphocyte subpopulation, the proportion of B cells and CD8+ and CD4+ T cells present in the mediastinal lymph nodes was assessed by flow cytometry. BCG infection induced a significant increase in the proportion of B cells, and an identical profile was observed in the draining lymph nodes of anti-CTLA-4 MAb-treated mice (Fig. 2).
FIG. 2.
Treatment with anti-CTLA-4 MAb does not alter BCG-induced changes in mediastinal lymph node cell populations. C57BL/6 mice were left uninfected (open circles) or intranasally infected with BCG and either treated with anti-CTLA-4 MAb (solid squares) or left untreated (open squares). Mediastinal lymphocytes were stained for fluorescence-activated cell sorter analysis with anti-CD4, -CD8, or -B220 MAbs. The values represent the percentage of positive cells from lymphocytes pooled from four mice. The results shown are representative of two separate experiments.
CTLA-4 blockade in vivo enhances in vitro antigen-specific proliferative responses.
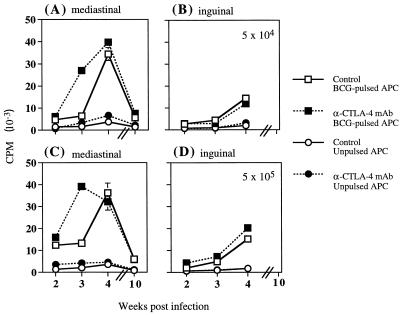
To establish whether the CTLA-4 blockade-induced increase in numbers of mediastinal lymphocytes corresponded to an increase in T-cell effector function, we analyzed antigen-inducible proliferation of mediastinal lymphocytes in vitro. BCG is actively taken up and presented to T cells by macrophages; therefore, BCG-pulsed macrophages were used to provide an antigen-specific stimulus. Resident peritoneal macrophages from uninfected C57BL/6 mice were incubated overnight with BCG. The BCG-pulsed macrophages were cultured with total mediastinal lymphocytes from BCG-infected mice that had been either treated weekly with anti-CTLA-4 neutralizing MAb or left untreated. Blockade of CTLA-4 ligation in vivo led to an enhanced in vitro antigen-specific proliferative response of lymphocytes taken from the draining mediastinal lymph node 3 weeks after BCG infection (Fig. 3A and C). Proliferation of mediastinal lymphocytes was antigen dependent, since macrophages that were not pulsed with BCG did not significantly stimulate proliferation (Fig. 3A and C) and this was not notably affected by in vivo treatment with anti-CTLA-4 MAb.
FIG. 3.
Treatment with anti-CTLA-4 MAb in vivo enhances in vitro antigen-specific proliferation of mediastinal lymphocytes. C57BL/6 mice were intranasally infected with 5 × 104 (A and B) or 5 × 105 (C and D) BCG and either treated with anti-CTLA-4 MAb or left untreated (controls). At the indicated times postinfection, total lymphocytes from the mediastinal lymph node (A and C) or inguinal lymph nodes (B and D) were stimulated in vitro with either BCG-pulsed or unpulsed peritoneal macrophages. Cultures were incubated for 72 h, and proliferation was measured by [3H]thymidine incorporation in the last 10 h of culture. The values at 2 weeks postinfection represent the mean counts per minute (CPM) of lymphocytes pooled from four mice ± standard error (SE). The values at other time points represent the mean CPM of separate samples from four mice ± SE. APC, antigen-presenting cells.
Although treatment with anti-CTLA-4 MAb had no effect on total inguinal lymph node cell numbers, antigen-specific proliferative responses of these cells were analyzed in vitro to determine whether there was any nonspecific activation induced by anti-CTLA-4 MAb treatment. As the BCG infection advanced, antigen-inducible proliferative responses of lymphocytes obtained from nondraining inguinal lymph nodes also increased marginally (Fig. 3B and D). This may reflect the spread of bacteria to organs other than the lung and increased peripheral circulation of antigen-specific effector cells. Proliferation of inguinal lymphocytes in the absence of antigen was very low and was not affected by in vivo treatment with anti-CTLA-4 MAb.
To ensure that the differences observed in the proliferative responses between anti-CTLA-4 MAb-treated and untreated animals did not merely reflect a difference in ability to respond in vitro, cells were also restimulated in vitro with immobilized anti-CD3 MAb. Stimulation of mediastinal lymphocytes with immobilized anti-CD3 MAb always induced the same degree of proliferation from anti-CTLA-4 MAb-treated and untreated mice (data not shown).
CTLA-4 blockade in vivo enhances in vitro antigen-specific IFN-γ production.
The development of protective immunity against BCG closely correlates with production of the Th1 inflammatory cytokine IFN-γ (25). The effect of in vivo CTLA-4 blockade on IFN-γ production during the course of BCG infection was analyzed by restimulating mediastinal lymphocytes in vitro with BCG-infected macrophages. After a 72-h incubation, culture supernatants were removed and the amount of IFN-γ produced was measured by ELISA. Cytokine secretion by mediastinal lymphocytes from infected, anti-CTLA-4 MAb-treated mice was compared with that of infected, untreated controls.
Antigen-specific, IFN-γ-producing lymphocytes could be detected earlier in anti-CTLA-4 MAb-treated, BCG-infected mice than in untreated, infected mice (Fig. 4A). The earlier response induced by anti-CTLA-4 MAb treatment resulted in an 8.5-fold increase in IFN-γ production at 3 weeks postinfection. The effect of anti-CTLA-4 MAb treatment on IFN-γ production was also observed when mice were infected with a 10-fold-higher concentration of BCG. Lymphocytes from anti-CTLA-4 MAb-treated mice produced fourfold more IFN-γ than lymphocytes from untreated controls. Production of IFN-γ was antigen dependent, as no IFN-γ production was detected from mediastinal lymphocytes that were cultured with unpulsed macrophages or in medium alone (data not shown).
FIG. 4.
In vivo treatment with anti-CTLA-4 MAb enhances in vitro cytokine production. C57BL/6 mice were intranasally infected with 5 × 104 BCG and treated with either anti-CTLA-4 MAb (solid squares) or left untreated (open squares). (A) At the indicated times postinfection, total lymph node cells from the draining mediastinal lymph nodes were stimulated in vitro with C57BL/6 peritoneal-derived macrophages pulsed with BCG. Supernatants were harvested after 72 h, and their IFN-γ content was measured by ELISA. The values at 2 weeks postinfection represent the mean IFN-γ production of lymphocytes pooled from four mice ± standard error (SE). The values at other time points represent the mean IFN-γ production of separate samples from four mice ± SE. (B) At the indicated times postinfection, total lymph node cells from the draining mediastinal lymph nodes were stimulated in vitro with immobilized anti-CD3 MAb. IL-5 levels in the supernatant after 48 h were measured by ELISA. The values at 1 and 2 weeks postinfection represent the mean IL-5 production of lymphocytes pooled from four mice ± SE. The values at other time points represent the mean IL-5 production of separate samples from four mice ± SE.
Effect of CTLA-4 blockade in vivo on Th2 cytokine production in vitro.
Although BCG infection typically induces a Th1 phenotype with characteristic IFN-γ production, blockade of CTLA-4 could potentially alter the cytokine pattern induced. In order to investigate whether treatment with anti-CTLA-4 MAb affected the Th1-Th2 balance during BCG infection, lymphocyte production of the Th2 cytokines IL-4, IL-5, and IL-10 was analyzed. Mediastinal lymphocytes from BCG-infected mice either treated with anti-CTLA-4 MAb or left untreated were restimulated in vitro with either immobilized anti-CD3 MAb for 48 h or BCG-infected macrophages for 72 h, and the concentration of cytokines in the culture supernatant was measured by ELISA.
Production of IL-5 by lymphocytes stimulated in vitro with BCG-pulsed macrophages was undetectable or very low, and no differences were detected between anti-CTLA-4 MAb-treated and untreated mice. Significant amounts of IL-5 were produced when lymphocytes were restimulated in vitro with anti-CD3 MAb (Fig. 4B). Interestingly, in vivo treatment with anti-CTLA-4 MAb during infection with 5 × 104 BCG led to a significant increase in the IL-5-producing capacity of lymphocytes in vitro compared to the amount of IL-5 produced by lymphocytes from infected, untreated mice (Fig. 4B). This increase in IL-5 production was dose dependent, since blockade of CTLA-4 ligation during infection with 5 × 105 BCG had a comparatively small effect on the amount of IL-5 produced following in vitro restimulation (data not shown). There was no detectable production of the Th2 cytokines IL-4 and IL-10 when lymphocytes were restimulated in vitro with either immobilized anti-CD3 MAb or macrophages pulsed with BCG, regardless of the infection dose and treatment.
Effect of CTLA-4 blockade on IFN-γ and TNF-α gene expression in vivo.
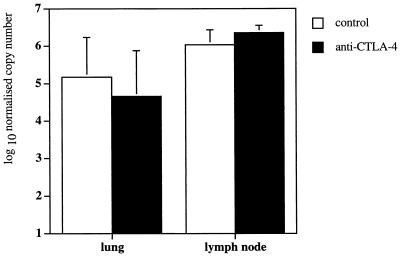
Despite evidence to suggest that CTLA-4 blockade enhanced antigen-driven proliferation and cytokine production in the draining mediastinal lymph node, it remained to be determined whether CTLA-4 blockade led to an accelerated immune response in the infected tissue in vivo. Therefore, we used a quantitative PCR method (12) to determine IFN-γ mRNA expression in infected tissue samples directly ex vivo. A threshold value was set in the exponential phase of amplification, and by comparison to a standard curve, the amount of cDNA in the samples was calculated and normalized to β2m. After 3 weeks of infection with 5 × 105 BCG, similar levels of IFN-γ mRNA expression were detected in the mediastinal lymph nodes of mice infected with 5 × 105 BCG and treated with anti-CTLA-4 MAb and those of control untreated mice (Fig. 5). Since levels of IFN-γ mRNA expression were normalized to β2m, and at this time point the cellularity of mediastinal lymph nodes was increased fivefold in anti-CTLA-4 MAb-treated mice (Fig. 1A), it can be extrapolated that there was approximately fivefold more total IFN-γ mRNA expression in mediastinal lymph nodes from anti-CTLA-4 MAb-treated mice than from untreated controls. Significantly, levels of IFN-γ mRNA expression at the primary site of infection, the lung, remained similar in mice treated with anti-CTLA-4 MAb and control untreated mice (Fig. 5).
FIG. 5.
Treatment with anti-CTLA-4 MAb has no detectable effect on IFN-γ gene expression in the mediastinal lymph nodes and lungs of BCG-infected mice. C57BL/6 mice were intranasally infected with 5 × 105 BCG and either treated with anti-CTLA-4 MAb (solid bars) or left untreated (open bars). The mice were sacrificed at 3 weeks after infection, and the mediastinal lymph nodes and lungs were collected and analyzed for IFN-γ mRNA levels by a real-time quantitative PCR method. The values were normalized to IFN-β2m, and represent the geometric mean of CFU from four mice ± standard error.
A further important point to consider was indirect effects of CTLA-4 blockade on non-T cells. We studied whether CTLA-4 blockade had a downstream effect on the production of TNF-α by macrophages, which do not express CTLA-4 (2). TNF-α is essential for protection against mycobacterial infection and acts synergistically with IFN-γ in activating the antimycobacterial action of macrophages (5, 6, 10, 13, 15).
However, TNF-α mRNA expression was present at extremely low or undetectable levels in BCG-infected lung samples from both anti-CTLA-4 MAb-treated and untreated mice (data not shown).
In vivo CTLA-4 blockade does not enhance clearance of BCG.
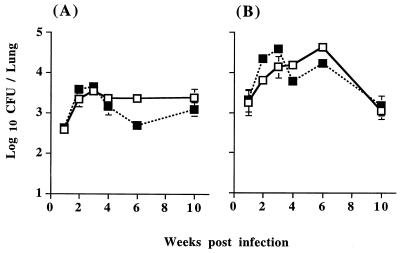
Since anti-CTLA-4 MAb treatment significantly enhanced antigen-induced lymphocyte expansion and cytokine production by T cells in the draining mediastinal lymph node, the effect of CTLA-4 blockade on mycobacterial growth in vivo was investigated. Tissue homogenates of lung, liver, and spleen from BCG-infected mice either treated with anti-CTLA-4 MAb or left untreated were cultured for growth of viable mycobacteria. Despite the accelerated and enhanced antigen-induced lymphocyte expansion in the draining mediastinal lymph node, the kinetics of BCG growth and clearance in the lung were not detectably affected by anti-CTLA-4 MAb treatment (Fig. 6). Most bacterial growth occurred within the lung, as expected with an intranasal infection; however, none of the animals were able to contain the infection within the lung. By 2 to 3 weeks postinfection, mycobacteria had spread to the liver and spleen in all animals regardless of treatment with anti-CTLA-4 MAb (data not shown).
FIG. 6.
Treatment with anti-CTLA-4 MAb has no detectable effect on the growth of BCG in the lung. C57BL/6 mice were intranasally infected with 5 × 104 (A) or 5 × 105 (B) BCG and either treated with anti-CTLA-4 MAb (solid squares) or left untreated (open squares). The mice were sacrificed at the indicated time points after infection, and the lungs were removed, homogenized, and plated on Middlebrook 7H11 agar for viable bacterial counts. The values represent the geometric mean of CFU from four mice ± standard error.
CTLA-4 blockade does not affect granuloma formation in tissues.
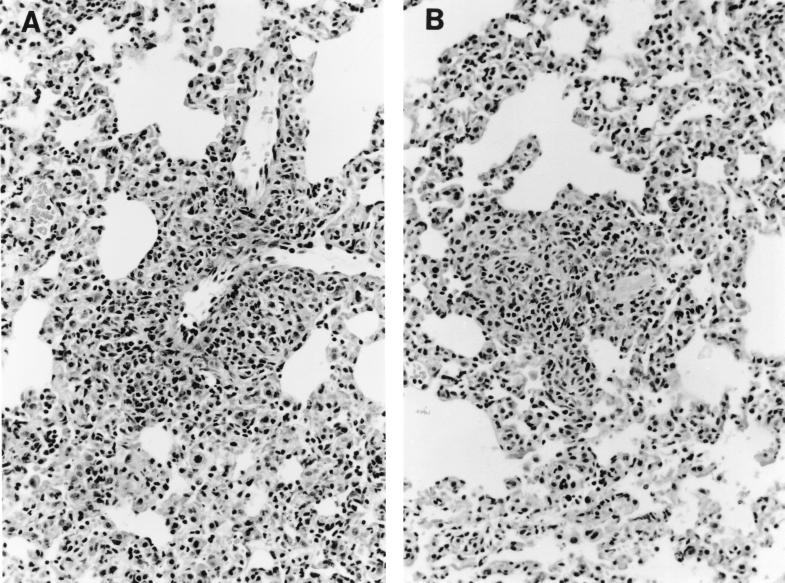
To establish whether CTLA-4 blockade had any effect on pathology, we analyzed histological sections from the lungs and livers of infected mice over the course of infection to look for differences in lymphoid infiltration and granuloma formation. All BCG-infected mice, independent of anti-CTLA-4 MAb treatment, developed mild-to-moderate granulatomous pneumonia by 4 weeks after infection. There was no difference in the sizes and densities of granulomas, and similar numbers of infiltrating lymphoid cells were seen in the lungs (Fig. 7A and B). Coinciding with the appearance of detectable levels of viable BCG in the liver, 2 weeks after intranasal BCG infection most mice developed focal lymphocytic hepatitis, with no differences between anti-CTLA-4 MAb-treated and untreated mice (data not shown).
FIG. 7.
Treatment with anti-CTLA-4 MAb does not affect granuloma formation in tissues. C57BL/6 mice were intranasally infected with 5 × 105 BCG and either treated with anti-CTLA-4 MAb (A) or left untreated (B). After 3 weeks of infection, the lungs were removed for histological analysis, fixed in formalin, and embedded in paraffin wax. Sections (3 μm) were stained with hematoxylin and eosin, and sections were analyzed by light microscopy (magnification, ×190). The results shown are representative of three separate experiments.
A similar level of lymphoid infiltration into the lung, taken together with data showing similar levels of IFN-γ mRNA expression in the lung tissue in infected anti-CTLA-4 MAb-treated mice and untreated controls (Fig. 5), strongly suggests that the immunity-enhancing effects of CTLA-4 blockade were limited to the draining mediastinal lymph node and that CTLA-4 blockade had no detectable effect on the number and potency of lymphocytes at the site of primary infection, the lung.
DISCUSSION
Several recent studies have shown that in vivo blockade of the interaction between CTLA-4 and its ligands, CD80 and CD86, can enhance immune responses generated against tumors and infectious agents and increase resistance (19, 21, 23). Here we report that administration of anti-CTLA-4 MAb during a BCG lung infection resulted in enhanced antigen-driven expansion of lymphocytes in the draining mediastinal lymph node but did not lead to a reduction in bacterial load.
CTLA-4 is thought to negatively regulate T-cell activation, and blockade of CTLA-4 ligation in vivo can enhance antigen-induced expansion of T cells (14). In keeping with the antigen-specific nature of the effect of CTLA-4 blockade, lymphocytes taken from the inguinal lymph nodes that were not draining sites of BCG infection did not demonstrate enhanced activity. In addition, treatment with anti-CTLA-4 MAb did not alter the cellular composition of the draining mediastinal lymph node, as the percentage of CD4+, CD8+, and B cells present remained the same regardless of CTLA-4 function.
Three weeks after intranasal BCG infection, cells from the draining mediastinal lymph node of anti-CTLA-4 MAb-treated mice showed enhanced proliferation when cultured with BCG-infected macrophages compared to cultures of lymphocytes from control, untreated mice. Parallel to the enhanced antigen-induced proliferative response, supernatants from these cultures also contained increased IFN-γ compared to the cultures of control, untreated mice. The enhanced cytokine production could be due either to an increase in the number of lymphocytes that had differentiated into effector cells or to an enhancement of the IFN-γ-producing abilities of individual effector cells. Analysis of IFN-γ mRNA levels in mediastinal lymphocytes directly ex vivo showed that IFN-γ gene expression remained similar in cells from infected anti-CTLA-4 MAb-treated mice and infected control mice. This suggests that the enhanced IFN-γ production detected in the cultures of lymphocytes from anti-CTLA-4 MAb-treated mice was likely due to the enhanced expansion of effector cells in vitro.
Despite CTLA-4 blockade in vivo leading to enhanced antigen-induced lymphocyte expansion and cytokine production in the draining mediastinal lymph nodes, this did not affect the course of the infection or the numbers of viable mycobacteria recovered from the lung, liver, or spleen. There are several possible explanations for the lack of enhanced BCG clearance in anti-CTLA-4 MAb-treated mice, despite the observed enhancement of the immune response. Since mice are considered to be very resistant to mycobacterial infections and as such represent an atypical host for these intracellular parasites (1), it could be argued that the primary murine immune response to an intranasal BCG infection is highly efficacious, with levels of IFN-γ that are already saturating and unable to be improved upon. However, recent studies from our laboratory show that priming the immune response by a primary BCG infection generates a greater degree of bacterial clearance and containment during a secondary infection (unpublished data). These results suggest that the primary immune response induced in mice infected with BCG can be improved upon.
A further explanation for the lack of enhanced protection observed with CTLA-4 blockade may be that anti-CTLA-4 MAb treatment amplified the Th2 response as well as the Th1 response, leading to inhibitory cross-regulation. We were unable to observe any production of the Th2 cytokines, IL-4, IL-5, and IL-10, secreted in response to in vitro antigen-specific stimulation regardless of CTLA-4 function, nor could we detect IL-4 or IL-10 production after polyclonal stimulation of lymphocytes during a 48-h culture with immobilized anti-CD3. However, in vivo blockade of CTLA-4 did increase mediastinal lymphocyte production of IL-5 in response to in vitro polyclonal stimulation by immobilized anti-CD3 MAb. Since CTLA-4 blockade in vivo could potentially increase lymphocyte production of any cytokine that is normally induced, and because low levels of IL-5 were induced with a low-level infection of BCG, treatment with anti-CTLA-4 MAb probably only served to enhance this production.
An alternative explanation for the lack of enhanced protection seen in BCG-infected mice treated with anti-CTLA-4 MAb is that another type of cell or cytokine, essential to the protective anti-mycobacterial response, was present in limiting amounts and remained unaffected by CTLA-4 blockade. One possibility we considered was TNF-α, which is produced by many types of cells, including macrophages infected with mycobacteria. As macrophages do not express CTLA-4 (2), production of TNF-α by macrophages may remain unaffected by CTLA-4 blockade. Levels of TNF-α gene expression were extremely low or undetectable in the lung tissue of infected mice and remained unaffected by in vivo CTLA-4 blockade. Therefore, lack of enhancement of another essential cytokine, such as TNF-α, may have limited the effectiveness of the CTLA-4 blockade-induced enhancement of the T-cell response.
However, the most likely explanation for the lack of enhanced mycobacterial clearance observed in anti-CTLA-4 MAb-treated mice is the apparent localization of the effects of anti-CTLA-4 MAb treatment to the draining mediastinal lymph node rather than the infected tissue. While enhanced lymphocyte expansion and cytokine production was detected in mice treated with anti-CTLA-4 MAb, these responses were detected in the lymph node that drains the lung and not at the actual site of infection. Although an increased number of potentially more reactive lymphocytes was present in the draining lymph node, these cells appeared not to have been trafficking through to the lung. The observation that the lymphoid infiltration and levels of IFN-γ gene expression in the lung were similar in anti-CTLA-4 MAb-treated mice and untreated mice supports this hypothesis.
Taken together, these results suggest that rather than skewing or amplifying the response in a nonspecific manner, CTLA-4 blockade increased the antigen-specific expansion and differentiation of lymphocytes in the draining lymph node that is typically induced in response to a BCG lung infection. However, treatment with anti-CTLA-4 MAb had no effect on IFN-γ mRNA expression and no effect on the level of lymphoid infiltration at the primary site of infection and therefore did not lead to enhanced clearance of the infection.
ACKNOWLEDGMENTS
The first two authors contributed equally to this work.
This work was supported by a Wellcome Trust Senior Research Fellowship, the Wellington Division of the Cancer Society of New Zealand, and the University of Otago.
We thank J. Bluestone for the 4F10 (anti-CTLA-4 MAb) hybridoma cell line, P. Cartwright for preparing histological sections, AgResearch Wallaceville for providing BCG (Pasteur), M. Camberis for technical assistance, F. Ronchese for helpful suggestions, and the Wellington School of Medicine Animal Facility.
REFERENCES
- 1.Brown I N. Animal models and immune mechanisms in mycobacterial infection. In: Ratledge C, Stanford J, editors. Biology of mycobacteria. Vol. 2. London, England: Academic Press; 1983. pp. 173–233. [Google Scholar]
- 2.Brunet J F, Denizot F, Luciani M F, Roux-Dosseto M, Suzan M, Mattei M G, Golstein P. A new member of the immunoglobulin superfamily—CTLA-4. Nature. 1987;328:267–270. doi: 10.1038/328267a0. [DOI] [PubMed] [Google Scholar]
- 3.Cooper A M, Dalton D K, Stewart T A, Griffin J P, Russell D G, Orme I M. Disseminated tuberculosis in IFNγ gene-disrupted mice. J Exp Med. 1993;178:2243–2249. doi: 10.1084/jem.178.6.2243. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Cooper A M, Callahan J E, Griffin J P, Roberts A D, Orme I M. Old mice are able to control low-dose aerogenic infections with Mycobacterium tuberculosis. Infect Immun. 1995;63:3259–3265. doi: 10.1128/iai.63.9.3259-3265.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Denis M. Involvement of cytokines in determining resistance and acquired immunity in murine tuberculosis. J Leukoc Biol. 1991;50:495–501. doi: 10.1002/jlb.50.5.495. [DOI] [PubMed] [Google Scholar]
- 6.Flesch I E A, Kaufmann S H E. Activation of tuberculostatic macrophage functions by interferon-γ, interleukin-4, and tumor necrosis factor. Infect Immun. 1990;58:2675–2677. doi: 10.1128/iai.58.8.2675-2677.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Flesch I E A, Kaufmann S H E. Mechanisms involved in mycobacterial growth inhibition by gamma-interferon-activated bone marrow macrophages: role of reactive nitrogen intermediates. Infect Immun. 1991;59:3213–3218. doi: 10.1128/iai.59.9.3213-3218.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Flynn J L, Chan J, Triebold K J, Dalton D K, Stewart T A, Bloom B R. An essential role for interferon γ in resistance to Mycobacterium tuberculosis infection. J Exp Med. 1993;178:2249–2252. doi: 10.1084/jem.178.6.2249. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Flynn J L, Goldstein M M, Triebold K J, Koller B, Bloom B R. Major histocompatibility complex class I-restricted T cells are required for resistance to Mycobacterium tuberculosis infection. Proc Natl Acad Sci USA. 1992;89:12013–12017. doi: 10.1073/pnas.89.24.12013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Flynn J L, Goldstein M M, Chan J, Triebold K J, Pfeffer K, Lowenstein C J, Schreiber R, Mak T W, Bloom B R. Tumor necrosis factor-α is required in the protective immune response against Mycobacterium tuberculosis in mice. Immunity. 1995;2:561–572. doi: 10.1016/1074-7613(95)90001-2. [DOI] [PubMed] [Google Scholar]
- 11.Gheorghiu M, Augier J, Lagrange P H. Maintenance and control of the French BCG strain 1173-P2 (primary and secondary seed-lots) Bull Inst Pasteur. 1983;81:281–288. [Google Scholar]
- 12.Heid C A, Stevens J, Livak K J, Williams P M. Real time quantitative PCR. Genome Res. 1996;6:986–994. doi: 10.1101/gr.6.10.986. [DOI] [PubMed] [Google Scholar]
- 13.Kamijo R, Shapiro D, Le J, Huang S, Aguet M, Vilcek J. Generation of nitric oxide and induction of major histocompatibility complex class II antigen in macrophages from mice lacking the interferon γ receptor. Proc Natl Acad Sci. 1993;90:6626–6630. doi: 10.1073/pnas.90.14.6626. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Kearney E R, Walunas T L, Karr R W, Morton P A, Loh D Y, Bluestone J A, Jenkins M K. Antigen-dependent clonal expansion of a trace population of antigen specific CD4+ T cells in vivo is dependent on CD28 costimulation and is inhibited by CTLA-4. J Immunol. 1995;155:1032–1036. [PubMed] [Google Scholar]
- 15.Kindler V, Sappino A-P, Grau G E, Piguet P-F, Vassalli P. The inducing role of tumor necrosis factor in the development of bactericidal granulomas during BCG infection. Cell. 1989;56:731–740. doi: 10.1016/0092-8674(89)90676-4. [DOI] [PubMed] [Google Scholar]
- 16.Krummel M F, Allison J P. CD28 and CTLA-4 have opposing effects on the response of T cells to stimulation. J Exp Med. 1995;182:459–465. doi: 10.1084/jem.182.2.459. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Krummel M F, Allison J P. CTLA-4 engagement inhibits IL-2 accumulation and cell cycle progression upon activation of resting T cells. J Exp Med. 1996;183:2533–2540. doi: 10.1084/jem.183.6.2533. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Ladel C H, Daugelat S, Kaufmann S H E. Immune response to Mycobacterium bovis bacille Calmette Guérin infection in major histocompatibility complex class I- and II-deficient knock-out mice: contribution of CD4 and CD8 T cells to acquired resistance. Eur J Immunol. 1995;25:377–384. doi: 10.1002/eji.1830250211. [DOI] [PubMed] [Google Scholar]
- 19.Leach D R, Krummel M F, Allison J P. Enhancement of antitumor immunity by CTLA-4 blockade. Science. 1996;271:1734–1736. doi: 10.1126/science.271.5256.1734. [DOI] [PubMed] [Google Scholar]
- 20.Linsley P S, Greene J L, Tan P, Bradshaw J, Ledbetter J A, Anasetti C, Damle N K. Coexpression and functional cooperation of CTLA-4 and CD28 on activated T lymphocytes. J Exp Med. 1992;176:1595–1604. doi: 10.1084/jem.176.6.1595. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.McCoy K, Camberis M, Le Gros G. Protective immunity to nematode infection is induced by CTLA-4 blockade. J Exp Med. 1997;186:183–187. doi: 10.1084/jem.186.2.183. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Müller I, Cobbold S P, Waldmann H, Kaufmann S H E. Impaired resistance to Mycobacterium tuberculosis infection after selective in vivo depletion of L3T4+ and Lyt-2+ T cells. Infect Immun. 1987;55:2037–2041. doi: 10.1128/iai.55.9.2037-2041.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Murphy M L, Cotterell S E J, Gorak P M A, Engwerda C R, Kaye P M. Blockade of CTLA-4 enhances host resistance to the intracellular pathogen, Leishmania donovani. J Immunol. 1998;161:4153–4160. [PubMed] [Google Scholar]
- 24.Orme I. Characteristics and specificity of acquired immunologic memory to Mycobacterium tuberculosis infection. J Immunol. 1988;140:3589–3593. [PubMed] [Google Scholar]
- 25.Orme I M, Roberts A D, Griffin J P, Abrams J S. Cytokine secretion by CD4 T lymphocytes acquired in response to Mycobacterium tuberculosis infection. J Immunol. 1993;151:518–525. [PubMed] [Google Scholar]
- 26.Orme I M, Miller E S, Roberts A D, Furney S K, Griffin J P, Dobos K M, Chi D, Rivoire B, Brennan P J. T lymphocytes mediating protection and cellular cytolysis during the course of Mycobacterium tuberculosis infection. J Immunol. 1992;148:189–196. [PubMed] [Google Scholar]
- 27.Stenger S, Mazzaccaro R J, Uyemura K, Cho S, Barnes P F, Rosat J-P, Sette A, Brenner M B, Porcelli S A, Bloom B R, Modlin R L. Differential effects of cytolytic T cell subsets on intracellular infection. Science. 1997;276:1684–1687. doi: 10.1126/science.276.5319.1684. [DOI] [PubMed] [Google Scholar]
- 28.Walunas T L, Lenschow D J, Bakker C Y, Linsley P S, Freeman G J, Green J M, Thompson C B, Bluestone J A. CTLA-4 can function as a negative regulator of T cell activation. Immunity. 1994;1:405–413. doi: 10.1016/1074-7613(94)90071-x. [DOI] [PubMed] [Google Scholar]
- 29.Walunas T L, Bakker C Y, Bluestone J A. CTLA-4 ligation blocks CD28-dependent T cell activation. J Exp Med. 1996;183:2541–2550. doi: 10.1084/jem.183.6.2541. [DOI] [PMC free article] [PubMed] [Google Scholar]