Abstract
Since 1980, the number of cases of meningococcal disease caused by serogroup B isolates with the P1.4 serosubtype has greatly increased in The Netherlands. Screening for this serosubtype in the strain collection of The Netherlands Reference Laboratory for Bacterial Meningitis revealed that a low number of P1.4 strains had been present in the Dutch meningococcal population since 1965. Genotyping of P1.4 strains showed that one cluster of strains, the hyperendemic lineage III (D. A. Caugant et al., J. Infect. Dis. 162:867–874, 1990), is responsible for the increase since 1980. The diversity of the porA genes, which encode the P1 protein on which serosubtyping is based, was studied for genotypically different P1.4 strains and for lineage III strains expressing antigenically different P1 proteins. Sequence analysis showed that porA genes of genotypically distinct strains that express antigenically indistinguishable P1 proteins are identical only in the epitope-encoding region, suggesting that this region has spread through the meningococcal population via horizontal gene transfer. Analysis of porA genes of lineage III strains showed that both horizontal gene transfer and partial deletion of the epitope-encoding region may contribute to the different antigenic properties for P1 of these strains. Phase variation of expression of the porA gene seems to account for most nonreacting strains. These results show that serosubtyping may underestimate the rise of a hyperendemic clone.
Neisseria meningitidis, an encapsulated gram-negative bacterium, causes worldwide meningococcal disease. Acute meningococcal disease manifests clinically as meningitis, sepsis, or both.
Characterization of the surface structures of N. meningitidis has led to a classification system for this bacterium. Capsular polysaccharide antigenic differences determine the 12 serogroups, of which A, B, C, W-135, and Y are predominant. The serotypes are based on antigenic differences of the class 2/3 outer membrane protein (OMP). The antigenic heterogeneity of class 1 OMP, or P1, determines the serosubtypes (8).
Serogroup A meningococcal disease is associated with epidemics in underdeveloped countries. In Europe and North America, endemic meningococcal disease is due predominantly to serogroups B and C. In The Netherlands, P1.4 is the predominant serogroup B serosubtype among isolates since 1983, whereas only 7 isolates with this subtype were encountered among 444 isolates isolated in the period 1985 to 1980 (21). In 1990, the 158 P1.4 isolates represented 41% of all serogroup B isolates from clinical meningococcal disease cases in 1990 (21). Multilocus enzyme electrophoresis (MLEE) revealed that the serogroup B P1.4 serosubtype strains isolated since 1980 were genetically closely related, forming the so-called lineage III cluster, whereas five P1.4 strains recovered before 1983 belonged to lineages IV and II (5). More recently, strains belonging to lineage III were isolated from cases in other European countries, Chile, and New Zealand (6). Since it has been noted that lineage III strains are associated particularly with an increase of endemic meningococcal cases, it was termed a hypervirulent lineage (14).
For serogroup B meningococci, the class 1 OMP is not only important for typing purposes but also a major component of various vaccines that are currently under development (10). The porA genes, encoding P1 (2), of different N. meningitidis subtypes have been sequenced (4, 7, 9, 13, 17, 23). Two highly variable regions of porA, VR1 and VR2, generate two separate serosubtype-specific antigenic determinants (16). These regions correspond to the first and fourth predicted surface-exposed loops of the protein (26). The P1.4 epitope is located on the fourth loop of P1. For serogroup A strains, it has been reported that antigenic variation of porA is due mainly to horizontal gene transfer (23). However, for serogroup B strains, which differ in epidemiological behavior and population genetics from serogroup A strains, it is thought that point mutations and small duplications and deletions may be more important factors. Indeed, Suker et al. (24) showed that antigenic variation of the P1.10 epitope was caused mainly by point mutations. However, such phenomena have not been studied within one particular genotype and not for epitope families other than P1.10.
In this work, we studied the antigenic variation of P1.4 and the P1 variation in lineage III.
The porA genes of genotypically different P1.4 strains were sequenced in order to investigate the variation in porA genes that encode the P1.4 epitope. The porA sequences of lineage III strains that could not be serosubtyped as P1.4 were compared to determine the mechanisms by which expression of the P1.4 epitope was lost.
MATERIALS AND METHODS
Bacterial strains and serosubtype determination.
All strains are clinical isolates from patients with meningitis or meningococcemia from The Netherlands, sent to The Netherlands Reference Laboratory for Bacterial Meningitis by clinical microbiological laboratories throughout the country. Isolates were grown on heated blood (chocolate) agar at 37°C in a humidified atmosphere of 5% CO2 in air. Selection of the isolates used in this study was based on previous serosubtyping and MLEE data (5, 21, 22). Previously determined serosubtypes were confirmed by using reference antibodies for the determination of serosubtype specificity (1). In addition, previously nonsubtypeable serogroup B meningococci isolated in the Netherlands in 1978 (n = 74), 1979 (n = 49), 1981 (n = 117), and 1982 (n = 98) were screened for the P1.4 subtype with this method.
The strains for which porA sequences were determined (Table 1) comprise the known P1.4 strains that are not part of lineage III (n = 6), the known lineage III strains that do not express the P1.4 subtype (n = 6), and five P1.4 lineage III strains, isolated in 1980, 1982, or 1988.
TABLE 1.
Strains of which the porA sequence was determined
| Strain | Yr of isolation | Serogroup:serotype: serosubtype | ETa | Lineage III | porA sequence typeb |
|---|---|---|---|---|---|
| 520 | 1965 | B:15:P1.4 | 38.2 | − | a |
| 575 | 1966 | B:15:P1.4 | 48.1 | − | b |
| 1730 | 1970 | B:15:P1.4 | 38.2 | − | a |
| 3532 | 1975 | B:15:P1.4 | 30.0 | − | a |
| 800580 | 1980 | B:ntc:P1.4 | 22.2 | − | c |
| 800615 | 1980 | B:nt:P1.4 | 24.0 | + | c |
| 820220 | 1982 | B:4:P1.4 | NDd | + | c |
| 820287 | 1982 | B:nt:P1.4 | ND | + | c |
| 820555 | 1982 | B:4:P1.4 | ND | + | c |
| 830248 | 1983 | B:16:P1.4,12 | 46.0 | − | d |
| 850008 | 1985 | B:4:nt | 24.0 | + | c |
| 882066 | 1988 | B:4:P1.4 | 24.0 | + | c |
| 900278 | 1990 | B:4:P1.6 | 24.0 | + | e |
| 900974 | 1990 | B:4:P1.12 | 24.0 | + | f |
| 901005 | 1990 | B:4:P1.7 | 24.0 | + | g |
| 901063 | 1990 | B:4:P1.12 | 24.0 | + | f |
| 902641 | 1990 | B:4:nt | 24.0 | + | c |
Randomly amplified polymorphic DNA analysis.
To genotype newly identified P1.4 strains and to confirm the previously inferred phylogenetic relations between the strains by MLEE (5, 21, 22), randomly amplified polymorphic DNA (RAPD) analysis was performed. Four separate PCRs using four different primers were used, as described earlier (3). The resulting patterns were analyzed with the GelCompar software (Applied Maths BVBA, Kortrijk, Belgium).
PCR amplification of porA genes.
Template chromosomal DNA was prepared as described by Suker et al. (23), with minor variations. Primers used for amplification of the porA gene were AB01 (5′-TGT AAA ACG ACG GCC AGT GTT TGC CCG ATG TTT TTA GGT T-3′), AB02 (5′-CAG GAA ACA GCT ATG ACC CGG CGT ATA GGC GGA CTT GCT G-3′), AB03 (5′-TGT AAA ACG ACG GCC AGT CAG CGG CAG CGT CCA ATT CGT T-3′), and AB04 (5′-CAG GAA ACA GCT ATG ACC CGT ATC CGC TTC ACC GCC CCG A-3′), synthesized by Perkin-Elmer Nederland B.V., Gouda, The Netherlands. Sequences identical or complementary to the -21M13 and M13Reverse primers, necessary for dyed primer sequencing, are underlined. First, primers AB01 and AB04 were used to amplify the porA gene, using chromosomal DNA as a template. Subsequently, this product was used for reactions with primer set AB01-AB02 or AB03-AB04.
The reaction mixture contained 50 ng of template DNA, 1 μM each primer, 200 μM each nucleotide (dATP, dCTP, dGTP, and dTTP), 0.01% gelatin, 2.5 mM MgCl2, 50 mM KCl, 10 mM Tris-Cl (pH 8.3), and 2 U of Taq polymerase (Perkin-Elmer Cetus) in a final volume of 100 μl. Following initial denaturation at 95°C for 5 min, 30 amplification cycles were performed in a Trio Thermoblock (Biometra), each cycle comprising 2 min at 95°C, 2 min at 60°C, and 2 min at 72°C, with a final incubation at 72°C for 10 min. PCR products of three separate reactions were pooled and agarose gel purified by using a kit from Qiagen Inc. (Chatsworth, Calif.).
Fluorescence-based sequencing.
Sequences of the porA genes were obtained by PCR-based sequencing (according to the instructions supplied by Applied Biosystems Inc., Foster City, Calif.) with fluorescence dye-labeled universal primers -21M13 and M13Reverse of the products obtained with primer pairs AB01-AB02 and AB03-AB04, respectively. The products were analyzed on an automatic sequenator (model 370A or 373; Applied Biosystems).
Analysis of porA sequences.
porA sequences were analyzed by using computer programs included in the 1991 PC/GENE (IntelliGenetics, Inc.) program package. Alignments were also performed with the Clustal program in the Genetics Computer Group package, according to the method developed by Higgins and Sharp (11); alignments were also analyzed with the programs included in the 1993 MEGA program (12). Distribution of polymorphic positions was performed with the computer program Maximum Chi-Squared for Macintosh (version 1.0, 1995; developed by Nick Ross, Molecular Microbiology Group, School of Biological Sciences, University of Sussex, Brighton, England) from the original implementation of the maximum chi-squared method by Maynard Smith (15).
Nucleotide sequence accession numbers.
The nucleotide sequence data will appear in the EMBL/Genbank/DDBJ nucleotide sequence databases under accession no. AF112448, AF112449, AF112450, AF112451, AF112452, AF112453, and AF112454.
RESULTS
Identification of P1.4 strains among clinical isolates in The Netherlands during the period 1959 to 1994.
Previous studies (5, 21, 22) showed that the increase in the number of P1.4 isolates in The Netherlands started in the early 1980s. To assess the onset of this rise more precisely, all nonsubtypeable serogroup B patient isolates recovered in 1978, 1979, 1981, and 1982 were screened for the P1.4 subtype. We identified no P1.4 strains among the isolates recovered in 1978 and 1979, one P1.4 strain among the 1981 isolates, and seven P1.4 strains among the 1982 isolates.
Genotyping of P1.4 and non-P1.4 isolates.
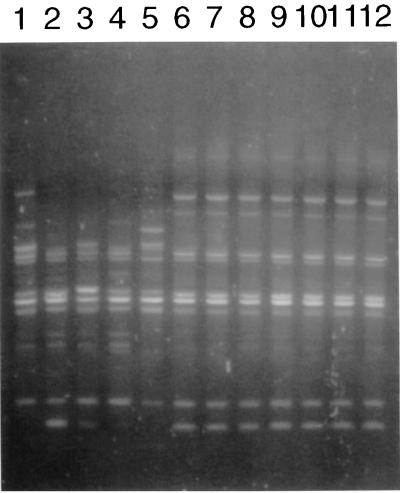
Strains that were previously genotyped by MLEE (listed in Table 1) were reanalyzed by RAPD. The patterns of the 11 lineage III strains were identical but differed from the patterns of the 6 non-lineage III strains, confirming the previous MLEE data (5). Also, the 1981 P1.4 isolate and the seven 1982 P1.4 isolates were genotyped by RAPD. Their RAPD patterns were identical to those of known lineage III strains, showing that they belong to lineage III. These data are illustrated in Fig. 1, which shows RAPD patterns obtained with one of the four primers used for five non-lineage III isolates, two lineage III isolates, and five previously not genotyped isolates.
FIG. 1.
RAPD patterns obtained with primer 1254 (2) of non-lineage III, lineage III, and previously not genotyped strains. Lane 1, strain 520 (non-lineage III); lane 2, strain 575 (non-lineage III); lane 3, 1730 (non-lineage III); lane 4, 3532 (non-lineage III); lane 5, 800580 (non-lineage III); lane 6, 800615 (lineage III); lane 7, 800616 (lineage III); lanes 8 to 12, previously not genotyped P1.4 isolates from cases in the period 1981 to 1982.
Sequence analysis.
The porA genes of the 17 strains listed in Table 1 were sequenced to analyze the variation in porA genes among the 11 P1.4 strains with different genotypes and among the 6 lineage III strains with different serosubtypes. Seven different sequences were found, indicated as porA sequence types a to g in Table 1. Four different porA genes (a to d in Table 1) were identified in the P1.4 strains. Strikingly, the porA sequence of strain 800580 (sequence type c) was identical to that of all lineage III strains, whereas by both RAPD and MLEE these strains were genotypically different. The sequence type c genes of two not subtypeable strains of lineage III and of the P1.4 strains of lineage III were identical.
An alignment of the polymorphic sites of the different porA genes is given in Fig. 2. Sequence types a to d, the genes of the P1.4 strains, shared an identical 366-bp region (positions 538 to 904) containing the P1.4 epitope. The sites that were polymorphic between sequence types d and c and between sequence type a or b and sequence type c were highly nonrandomly distributed (even if the P1.4-encoding VR2, for which the selection was made, is excluded from the analysis), as determined by the maximum chi-squared procedure (15). The number of polymorphic sites flanking VR2 was significantly lower than in the 5′ part of the gene. The six sites that were polymorphic between sequence type a and sequence type b were not significantly nonrandomly distributed.
FIG. 2.
Alignment of the polymorphic positions of the eight different porA types. Genes are indicated on the left by the sequence types indicated in Table 1; nucleotide position in the alignment is indicated vertically. Identical nucleotides are indicated with dots; gaps created by the alignment are indicated by dashes. Variable regions VR1 and VR2 are indicated above the nucleotide positions.
No nonrandom distribution of polymorphic sites in the sequences of sequence types e and f was observed compared to the sequence types c of strains with the same genotype. However, the percentage of polymorphic sites throughout the porA gene was comparable to the percentage in the 5′ part of the P1.4 porA genes (sequence types a or b, c, and d).
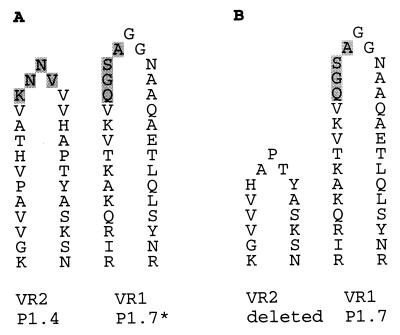
Sequence type g was identical to sequence type c of the P1.4 lineage III strains except for a deletion of part of VR2, between positions 587 and 627. Therefore, the P1.4 epitope was not present in the P1 of strain 901005.
The designations and amino acid sequences of the VR1 and VR2 regions of the different sequence types are given in Table 2. The deduced VR1 and VR2 sequences agreed well with the serosubtyping data for sequence types a, b, c, d, and f. Sequence type e of P1.6 strain 900278 corresponds to an Ea-type VR1 and a P1.3a-type VR1. The deduced amino acid sequence of the seventh loop of the PorA protein of this strain is common to strains that express the P1.6 serosubtype (20). Since the P1.6 monoclonal antibody is supposed to recognize a three-dimensional epitope of VR1, VR2, and the seventh loop of the PorA protein, serosubtype and the porA sequence were in agreement. VR1 of sequence types c and d was of the P1.7b type, which contains a P1.7 epitope that in the serosubtyping method used (1) does not react with the P1.7 monoclonal antibody, denoted P1.7* (27). However, serosubtyping of strain 901005 with sequence type d showed that this strain was weakly positive for P1.7. This may be due to the deletion of the VR2 region in this sequence type. In Fig. 3, the implications that a shorter VR2-encoded loop may have for the exposure of the P1.7* epitope are shown schematically.
TABLE 2.
Deduced amino acid sequences and designations for VR1 and VR2 regions
| porA typea | VR1 sequence | VR1 typeb | VR2 sequence | VR2 typeb |
|---|---|---|---|---|
| a | QPSKGQVGNKVTKG | Eb | HVVVNNKVATHVP | 4 |
| b | QPSKGQVGNKVTKG | Eb | HVVVNNKVATHVP | 4 |
| c | AQAANGGASGQVKVTKA | 7b | HVVVNNKVATHVP | 4 |
| g | AQAANGGASGQVKVTKA | 7b | HVV | |
| d | KPSSTNAKTGNKVEVTKA | 12a | HVVVNNKVATHVP | 4 |
| e | PPSQGQTGNKVTKG | Ea | TVANGANNTIIRVP | 3ac |
| f | KPSSTNAKTGNKVEVTKA | 12a | YWTTVNTGSATTTFVP | 13d |
FIG. 3.
Schematic representation of the implications of the deletion of the VR2 loop in sequence type g. (A) Positions of the P1.7 epitope on loop 1 and the P1.4 epitope on loop 4 relative to each other, as for sequence type c. The P1.4 epitope and nonreactive P1.7 epitope are shaded. (B) Position of the P1.7 epitope relative to the fourth loop of the predicted P1 protein of strain 901005 (sequence type g). The silent P1.7 epitope is no longer shielded by the fourth loop.
DISCUSSION
Since 1980, an increase in the number of serosubtype P1.4 isolates from patients with meningococcal disease has occurred in The Netherlands. Our data prove that the VR2 that encodes P1.4 had been present in the meningococcal population circulating in The Netherlands since 1965. As was shown by Caugant et al. (5) and Scholten et al. (21, 22), the rise in P1.4 isolates in The Netherlands since 1980 is due to the rise of one specific clone, constituting lineage III. Serosubtyping proved to underestimate the rise of this clone, as 6 of 61 lineage III isolates were not of the P1.4 serosubtype (22). Therefore, this clone is also partly responsible for the increase of non-P1.4 serosubtypes since 1980. Apparently, lineage III isolates have certain characteristics, besides the P1.4 serosubtype, that render them successful pathogens.
The porA genes of all P1.4 strains contain an exactly conserved region of more than 350 bp, including and flanking the VR2 region. This, together with the highly nonrandom distribution of the polymorphic sites of these sequences, indicates that this part of the gene was derived from a common ancestor gene. Parts of the porA gene outside this conserved region, which include the VR1 region, show large interstrain variability: 35 sites are polymorphic in the region from nucleotide positions 1 to 318 of sequence type c compared to sequence types a and b. Similarly, 41 sites are polymorphic in the region from nucleotide positions 1 to 537 between sequence types c and d. The resulting mosaic structure of the genes indicates that horizontal gene transfer had taken place. The fact that the VR2 that encodes P1.4 was found in genotypically different strains also indicates that horizontal gene transfer has occurred. Natural transformation in N. meningitidis is mediated by a DNA uptake sequence, of which the copy nearest VR2 is present just downstream of porA (2). Therefore, it is expected that an incoming stretch of VR2-containing DNA also contains the porA sequence downstream of VR2. The low number and random distribution of the polymorphic sites found in the 250-bp 3′ parts of sequence types a to d is consistent with this hypothesis, which posits that these six polymorphic sites would reflect point mutations. The finding that sequence type c is shared by strains from the lineage III cluster and the electrophoretic type 22.2 strain suggests that the entire porA has been transferred between different strains. The two nontypeable lineage III strains contained the P1.4-encoding gene, although the gene apparently was not expressed. It has been reported that phase variation of PorA expression occurs through variation in the spacing between the −35 and −10 parts of the porA promoter (25). Lineage III strains that expressed a subtype other than P1.4 seem to have lost the P1.4 epitope through horizontal gene transfer of the whole porA gene. This is indicated by the finding that the polymorphic sites between these strains and the P1.4 lineage III strains were located throughout the porA sequence and that the percentage of polymorphic sites in the whole gene was similar to the percentage of polymorphic sites in the nonconserved part of the genes of genotypically different P1.4 strains.
One isolate, strain 901005, lost the P1.4 epitope due to a deletion of 39 bp of VR2. This deletion apparently occurred via recombination of the flanking TGTTGT direct repeats (positions 582 to 587 and 621 to 626) in the parent sequence type c porA. A similar deletion was described by Suker et al. (23). In that case, 87 bp were deleted between two TCGTTCCGGC direct repeats. The VR1 of porA of strain 901005 contains a masked P1.7 epitope (P1.7b), which usually reacts only with the P1.7 monoclonal antibody after treatment with sodium dodecyl sulfate (17, 27). Serosubtyping of strain 901005 was negative for P1.4 but weakly positive for P1.7. Possibly, the fourth loop of the P1 of this strain is too short to mask the P1.7 epitope on loop 1 of either the same monomer or a neighboring P1 monomer in a P1 trimer, thereby exposing it to the P1.7 monoclonal antibody. Since a recent report suggests that the PorA monomer forms heterotrimers with PorB rather than homotrimers (18), this demasking might occur within one monomer. Masking by other molecules, e.g., lipopolysaccharide, may be the reason for the fact that no strong reaction for P1.7 was observed.
No sequences with point mutations in the P1.4 epitope were found, although recently one variant was described (20). In contrast, Suker et al. (24) found eight variants for the P1.10 epitope. It is tempting to speculate that the human immune system played a role in the selection of the large number of P1.10 variants found by Suker et al. (24), since P1.10 may be a better immunogen than P1.4. Therefore, there will be less selection for changes in the top of the epitope-encoding loop, and thus fewer variants will be found. Interestingly, in a vaccine trial with an hexavalent outer membrane vesicle vaccine containing both P1.10 and P1.4, stronger responses to P1.10 than to P1.4 were found (19). It may be that a reduced antibody response against the P1.4 epitope plays a role in the rise of meningococcal disease due to lineage III strains.
In conclusion, most variation in porA genes in the strains analyzed seems due to horizontal gene transfer instead of other sorts of mutations. This implies that serology may underestimate the rise of a specific clone, since the serosubtype can be replaced by novel subtype combinations.
REFERENCES
- 1.Abdillahi H, Poolman J T. Neisseria meningitidis group B serosubtyping using monoclonal antibodies in whole-cell ELISA. Microb Pathog. 1988;4:27–32. doi: 10.1016/0882-4010(88)90045-9. [DOI] [PubMed] [Google Scholar]
- 2.Barlow A K, Heckels J E, Clarke I N. The class 1 outer membrane protein of Neisseria meningitidis: gene sequence and structural and immunological similarities to gonococcal porins. Mol Microbiol. 1989;3:131–139. doi: 10.1111/j.1365-2958.1989.tb01802.x. [DOI] [PubMed] [Google Scholar]
- 3.Bart A, Schuurman I G, Achtman M, Caugant D A, Dankert J, van der Ende A. Randomly amplified polymorphic DNA genotyping of serogroup A meningococci yields results similar to those obtained by multilocus enzyme electrophoresis and reveals new genotypes. J Clin Microbiol. 1998;36:1746–1749. doi: 10.1128/jcm.36.6.1746-1749.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Brooks J L, Fallon R J, Heckels J E. Sequence variation in class 1 outer membrane protein in Neisseria meningitidis isolated from patients with meningococcal infection and close household contacts. FEMS Microbiol Lett. 1995;128:145–150. doi: 10.1111/j.1574-6968.1995.tb07514.x. [DOI] [PubMed] [Google Scholar]
- 5.Caugant D A, Bol P, Høiby E A, Zanen H C, Frøholm L O. Clones of serogroup B Neisseria meningitidis causing systemic disease in the Netherlands, 1958–1986. J Infect Dis. 1990;162:867–874. doi: 10.1093/infdis/162.4.867. [DOI] [PubMed] [Google Scholar]
- 6.Caugant D A. Population genetics and molecular epidemiology of Neisseria meningitidis. APMIS. 1998;106:505–525. [PubMed] [Google Scholar]
- 7.Feavers I M, Heath A B, Bygraves J A, Maiden M C J. Role of horizontal genetic exchange in the antigenic variation of the class I outer membrane protein of Neisseria meningitidis. Mol Microbiol. 1992;6:489–495. doi: 10.1111/j.1365-2958.1992.tb01493.x. [DOI] [PubMed] [Google Scholar]
- 8.Frasch C E, Zollinger W D, Poolman J T. Serotype antigens of Neisseria meningitidis and a proposed scheme for designation of serotypes. Rev Infect Dis. 1985;7:504–510. doi: 10.1093/clinids/7.4.504. [DOI] [PubMed] [Google Scholar]
- 9.Guillén G, Alvarez A, Lemos G, Paredes T, Silva R, Martín A. Comparison of the DNA sequence of nine different genes for the class 1 outer membrane protein from Neisseria meningitidis. Biotecnol Apl. 1993;10:108–113. [Google Scholar]
- 10.Herbert M A, Heath P T, Mayon-White R T. Meningococcal vaccines for the United Kingdom. Communicable Disease Report CDR Rev. 1995;5:R130–R135. [PubMed] [Google Scholar]
- 11.Higgins D G, Sharp P M. CLUSTAL: a package for performing multiple sequences alignment on microcomputer. Gene. 1988;73:237–244. doi: 10.1016/0378-1119(88)90330-7. [DOI] [PubMed] [Google Scholar]
- 12.Kumar S, Tamura K, Nei M. MEGA, Molecular Evolutionary Genetics Analysis, version 1.01. University Park, Pa: The Pennsylvania State University; 1993. . (Distributed by the authors.) [Google Scholar]
- 13.Maiden M C J, Suker J, McKenna A J, Bygraves J A, Feavers I M. Comparison of the class 1 outer membrane proteins of eight serological reference strains of Neisseria meningitidis. Mol Microbiol. 1991;5:727–736. doi: 10.1111/j.1365-2958.1991.tb00743.x. [DOI] [PubMed] [Google Scholar]
- 14.Maiden M C J, Bygraves J A, Feil E, Morelli G, Russell J E, Urwin R, Zhang Q, Zhou J, Zurth K, Caugant D A, Feavers I M, Achtman M, Spratt B G. Multilocus sequence typing: a portable approach to the identification of clones within populations of pathogenic microorganisms. Proc Natl Acad Sci USA. 1998;95:3140–3145. doi: 10.1073/pnas.95.6.3140. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Maynard Smith J. Analyzing the mosaic structure of genes. J Mol Evol. 1992;34:126–129. doi: 10.1007/BF00182389. [DOI] [PubMed] [Google Scholar]
- 16.McGuiness B T, Barlow A K, Clarke I N, Farley J E, Anilionis A, Poolman J T, Jones D M, Heckels J E. Deduced amino acid sequences of class 1 protein (porA) from 3 strains of Neisseria meningitidis: synthetic peptides define the epitopes responsible for serosubtype specificity. J Exp Med. 1990;171:1871–1882. doi: 10.1084/jem.171.6.1871. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.McGuiness B T, Lambden P R, Heckels J E. Class 1 outer membrane protein of Neisseria meningitidis: epitope analysis of the antigenic diversity between strains, implications for serosubtype definition and molecular epidemiology. Mol Microbiol. 1993;7:505–514. doi: 10.1111/j.1365-2958.1993.tb01141.x. [DOI] [PubMed] [Google Scholar]
- 18.Minetti C, Song J, Colombini M, Blake M S. Abstracts of the 98th General Meeting of the American Society for Microbiology 1998. Washington, D.C: American Society for Microbiology; 1998. Structural, functional, and antigenic relationship of meningococcal PorA and PorB, abstr. D-119; p. 232. [Google Scholar]
- 19.Rouppe van der Voort E, Van Dijken H, Kuipers B, Van der Biezen J, Van der Ley P, Meylis J, Claassen I, Poolman J. Human B- and T-cell responses after immunization with a hexavalent PorA meningococcal outer membrane vesicle vaccine. Infect Immun. 1997;65:5184–5190. doi: 10.1128/iai.65.12.5184-5190.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Sacchi C T, Lemos A P S, Brandt M E, Whitney A M, Melles C E, Solari C A, Frasch C E, Mayer L W. Proposed standardization of Neisseria meningitidis PorA variable-region typing nomenclature. Clin Diagn Lab Immunol. 1998;5:845–855. doi: 10.1128/cdli.5.6.845-855.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Scholten R J P M, Bijlmer H A, Poolman J T, Kuipers B, Caugant D A, Van Alphen L, Dankert J, Valkenburg H A. Meningococcal disease in the Netherlands, 1958–1990: a steady increase in the incidence since 1982 partially caused by new serotypes and subtypes of Neisseria meningitidis. Clin Infect Dis. 1993;16:237–246. doi: 10.1093/clind/16.2.237. [DOI] [PubMed] [Google Scholar]
- 22.Scholten R J P M, Poolman J T, Valkenburg H A, Bijlmer H A, Dankert J, Caugant D A. Phenotypic and genotypic changes in a new clone complex of Neisseria meningitidis causing disease in the Netherlands, 1958–1990. J Infect Dis. 1994;169:673–676. doi: 10.1093/infdis/169.3.673. [DOI] [PubMed] [Google Scholar]
- 23.Suker J, Feavers I M, Achtman M, Morelli G, Wang J-F, Maiden M C J. The porA gene in serogroup A meningococci: evolutionary stability and mechanism of genetic variation. Mol Microbiol. 1994;12:253–265. doi: 10.1111/j.1365-2958.1994.tb01014.x. [DOI] [PubMed] [Google Scholar]
- 24.Suker J, Feavers I M, Maiden M C J. Monoclonal antibody recognition of members of the meningococcal P1.10 variable region family: implications for serological typing and vaccine design. Microbiology. 1996;142:63–69. doi: 10.1099/13500872-142-1-63. [DOI] [PubMed] [Google Scholar]
- 25.Van der Ende A, Hopman C T P, Zaat S, Oude Essink B B, Berkhout B, Dankert J. Phase variation of class 1 outer membrane protein in Neisseria meningitidis by transcription from a variable promoter region. J Bacteriol. 1995;177:2475–2480. doi: 10.1128/jb.177.9.2475-2480.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Van der Ley P, Heckels J E, Virji M, Hoogerhout P, Poolman J T. Topology of outer membrane porins in pathogenic Neisseria spp. Infect Immun. 1991;59:2963–2971. doi: 10.1128/iai.59.9.2963-2971.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Wedege E, Dalseg R, Caugant D A, Poolman J T, Froholm L O. Expression of an inaccessible P1.7 subtype epitope on meningococcal class 1 proteins. J Med Microbiol. 1993;38:23–28. doi: 10.1099/00222615-38-1-23. [DOI] [PubMed] [Google Scholar]