Abstract
Vocal activity and signal characteristics of mammals are driven by several factors that result in both stability and plasticity over multiple time scales. All three extant species of manatee communicate with several calls that are especially important for maintaining contact between cows and calves. Determining if calf calls differ across manatee species will provide insights into the evolution of species-specific acoustic communication traits. We investigated the interspecific differences in the vocalizations of calves of Amazonian manatees (Trichechus inunguis) and the two subspecies of the West Indian manatee (T. manatus). Vocalizations of individual calves were recorded in rehabilitation centers in Brazil, Puerto Rico, the United States, and Mexico. The acoustic structure of calls produced by manatee calves varied between species and with body size. Amazonian manatee calves produced shorter calls with multiple notes at higher frequency while West Indian calves produced modulated calls that were lower in frequency and longer in duration. Smaller West Indian calves produced frequency modulated, hill-shaped calls that flattened with an increase in body length. Our results provide evidence for divergence in the ontogeny of vocalizations across T. manatus and T. inunguis and suggest variation in body size contributed to the evolution of differences in the characteristics of their calls.
Subject terms: Ecology, Evolution
Introduction
Vocal behavior is a key modality in animal communication in which different evolutionary pressures and ecological factors influence changes in the acoustic structure of calls1,2. Vocal variation within related species can be the result of genetic differences, geographic separation, and effects of habitat type3,4. On a species-specific scale, the structure of vocalizations can vary with behavior (e.g., African elephants, Loxodonta africana)5, between individuals for identification (bottlenose dolphins, Tursiops truncatus)6, during ontogeny (e.g., harbor seal pups, Phoca vitulina7; meerkats, Suricata suricata)8, be genetic and/or learned9 and influenced by body size10,11.
The size and length of the vocal tract and vocal apparatus influences the acoustic features of a signal and may be a reliable indicator of body size10,11. The production of vocalizations in vertebrates can be described according to the two-stage process of the source filter theory12. The source-filter theory predicts the signal is generated by the vibration of the laryngeal folds and subsequently filtered in the supralaryngeal vocal tract12. The length of the vocal folds determines the minimum fundamental frequency of a vocalization and has an allometric relationship with body size13. Therefore, larger animals would have longer vocal folds and as a result have a lower fundamental frequency. Determining the factors that influence how communication systems evolve demands more study with taxa whose acoustic communication and the basic function of their acoustic calls are still poorly understood14. Such is the case with Sirenians, where we have limited knowledge about acoustic communication both within and amongst species.
Sirenians include three extant species of manatees: the West Indian manatee (Trichechus manatus)—divided into the Antillean manatee (T. m. manatus) and Florida manatee (T. m. latirostris) subspecies15, the Amazonian manatee (T. inunguis), the African manatee (T. senegalensis) and the dugong (Dugong dugon). All sirenians produce vocalizations16–19, but here we focus on comparisons within the West Indian and Amazonian manatee species due to their shared evolutionary paths and ecological contexts. Manatees in the Western Hemisphere (West Indian and Amazonian) inhabit tropical and subtropical coasts in the Americas. Amazonian manatees inhabit freshwaters throughout the Amazon River Basin of northern South America20. The Antillean manatee range includes Central America, northern South America, and the Caribbean21. Florida manatees are found throughout the southern United States with periodic sightings in Cuba22 and Mexico23. Both Florida and Antillean manatees inhabit salt, fresh and brackish environments24. The Antillean and Amazonian manatee species overlap in Brazil where both species live in sympatry but show low hybridization rates suggesting these species occupy different ecological niches, have evolved mechanisms of reproductive isolation, or hybrid offspring viability is low25.
In all manatee species, the only known stable association is the 1½—2 years of cow/calf dependency26,27. Calves are thought to be nutritionally independent by one year of age, but stay with their mother beyond this age, presumably to learn the location of warm water refuges (for Florida manatees) and food sources from cows28,29. Calves tend to be more vocal and vocalize about 2–6 times more frequently than their mothers17,19,26,30,31, although no significant difference in vocalization rate was found between calves and adults in Antillean manatees18. Calves produce vocalizations under a variety of conditions which include, traveling, during approach from conspecifics, when separated and prior to nursing19. Calf vocalizations have been described as ranging from tonal vocalizations with harmonics to atonal vocalizations17–19,30,32. Amazonian and Antillean calf calls contain signature information which may be useful in recognition between cow/calf pairs17,18.
Currently, there are no published studies investigating if there are differences in frequency and temporal parameters among calf species. A few studies compared vocalizations between West Indian33 and African manatee species34 and noted significant overlap in frequency and temporal parameters. Although these differences were observed, it is unclear if calls from the West Indian and African manatees were obtained from adults and/or calves. Amazonian manatees are the smallest in size of all manatee species and have a fundamental frequency range from 1 to 8 kHz17. This range is higher than the reported frequency parameters of other manatee species. Furthermore, Antillean and Amazonian manatees are smaller in body size than Florida manatees20 and it is unclear if there would be differences in frequency and temporal vocal parameters between populations based on body size. The source of manatee vocalizations is the larynx and sound vibrations are produced by forcing air through their thick vocal folds35. The smaller body size and subsequently the smaller length of the vocal folds of the Amazonian manatee could place physiological constraints on signal production throughout development and therefore affect signal transmission36.
In addition to a lack of information on differences in vocalizations across manatee populations, there are few studies investigating if and how calls change during development31. Manatees tend to grow rapidly from birth. In the first year of life, captive Antillean manatee calves gained an average of 106 kg and grew an average of 83 cm37. Research on Amazonian and Antillean manatees suggests that the fundamental frequency becomes more defined as the calf grows to a more adult-type call17,18. In Florida manatees, hill-shaped calls (high squeaks) are correlated with calf presence, but the study could not attribute calls to younger or older calves32. Evidence of the ontogeny of acoustic structure in calls is limited to the study of one individual Amazonian manatee. They found an Amazonian manatee calf produced calls with decreasing fundamental frequencies during the first year of life31. After the first year, recordings from this and five other individuals recorded during the transition from calf to juvenile showed vocalizations increased in frequency31.
Identifying the factors that shaped the acoustic structure of manatee calls across species and over the course of their ontogeny will facilitate a better understanding of their communication and vocal repertoire. Here, we investigated if the frequency and temporal variables of the vocalizations of manatee calves varies across species as a function of body size. Manatee vocalizations were compared using spectrographic analysis to identify if shifts in vocal parameters occur depending on species. Since body size can be correlated with fundamental frequency10,11, we would predict the smaller body size of the Amazonian manatee would lead to higher frequency parameters than the Florida and Antillean manatee.
Methods
Data collection
Data for this study were collected from captive manatee calves (n = 18) from two different species at eight different facilities in the USA (n = 1), Mexico (n = 1), and Brazil (n = 6) (Table 1). Except for the calf from Mexico and one calf in Brazil (Ere), all calves were born in the wild and were either orphaned or brought in with their mothers for rehabilitation. In all locations, acoustic recordings of manatee calf vocalizations were collected from healthy animals. The straight-line body length—a straight-line measure from the tip of the snout to the medial notch of the fluke—of each manatee calf was obtained using a measuring tape during routine health assessments by veterinary staff at each of the respective facilities38.
Table 1.
Information on the manatee calves recorded in this study. The body length of each calf was measured during routine veterinary procedures according to standard protocols at each facility38 (ZT ZooTampa, CMCC Caribbean Manatee Conservation Center, DD Dolphin Discovery, CMA Centro Mamíferos Aquáticos/IBAMA, INPA Laboratório de Mamíferos Aquáticos of the Instituto Nacional de Pesquisas da Amazônia, MPEG Parque Zoobotânico of the Museu Paraense Emílio Goeldi, CPPMA Centro de Preservação e Pesquisa de Mamíferos Aquáticos da Manaus Energia, BioMA Biologia e Conservação de Mamíferos Aquáticos da Amazônia).
| Country | State | Site name | Species | Subspecies | Individual | Body length (cm) | n calls analyzed |
|---|---|---|---|---|---|---|---|
| USA | Florida | ZT | West Indian | Florida | Agua | 132.0 | 11 |
| USA | Florida | ZT | West Indian | Florida | Emerald | 128.0 | 10 |
| USA | Florida | ZT | West Indian | Florida | Camlee | 204.0 | 10 |
| USA | Florida | ZT | West Indian | Florida | Furbie | 205.0 | 11 |
| USA | Florida | ZT | West Indian | Florida | Neuson | 192.0 | 10 |
| USA | Florida | ZT | West Indian | Florida | Muddy Barron | 175.0 | 11 |
| USA | Puerto Rico | CMCC | West Indian | Antillean | Aramaná | 106.0 | 10 |
| USA | Puerto Rico | CMCC | West Indian | Antillean | Guamá | 128.0 | 12 |
| Mexico | Quintana Roo | DD | West Indian | Antillean | BonBon | 115.0 | 4 |
| Brazil | Pernambuco | CMA | West Indian | Antillean | Guaju | 199.0 | 10 |
| Brazil | Pernambuco | CMA | West Indian | Antillean | Guape | 208.0 | 10 |
| Brazil | Pernambuco | CMA | West Indian | Antillean | Araqueto | 190.0 | 10 |
| Brazil | Amazonas | INPA | Amazonian | N/A | Anama | 112.5 | 10 |
| Brazil | Amazonas | INPA | Amazonian | N/A | Ere | 112.0 | 10 |
| Brazil | Amazonas | INPA | Amazonian | N/A | Aria | 113.0 | 10 |
| Brazil | Pará | MPEG | Amazonian | N/A | Uiara | 100.0 | 10 |
| Brazil | Amazonas | CPPMA | Amazonian | N/A | Acai | 103.0 | 10 |
| Brazil | Pará | BioMA | Amazonian | N/A | Neguinha | 98.0 | 12 |
West Indian manatees
The vocalizations of six Florida calves were recorded at ZooTampa in Florida, USA from 2005 to 2018. Recordings were made with a Cetacean Research Technology SQ26-08 hydrophone (sensitivity: − 169 dB re 1 V/µPa; sample rate: 48 kHz; resolution: 16 bit) (n = 5 calves) and an Aquarian H2A hydrophone (sensitivity: − 180 dB re: 1 µPa; sample rate: 48 kHz; resolution: 16 bit) (n = 1 calf) sampling to either a Sony PCM M-10, Sony DAT TCD-D8, or a M-Audio MicroTrack II Digital Multi Track Recorder.
Animals were routinely housed together at ZooTampa. To ensure calls were obtained from the same individual, calves were recorded under the following conditions: (1) when they were alone in a medical pool; (2) wrinkling of the nose was observed while a vocalization was being produced; and (3) the perceived level of loudness in relation to the hydrophone19. During sound production, manatees contract the muscles dorsal to the maxilla and caudal to the nostrils while maintaining them closed35. Such nasal cavity movements may have a role in sound modulation and filtering. Perceived loudness includes actively listening to calls as they are being produced and noting the location of animals in relation to the hydrophone. The closer a vocalizing animal is to a hydrophone, the louder the sound will be. When a call was produced, the call was annotated into a second, synchronized recorder. This happened when the calf vocalized within 1–2 m of the hydrophone.
Vocalizations from six isolated Antillean manatee calves were recorded from 1998 to 2018 at three locations: the Centro Mamíferos Aquáticos/IBAMA in Pernambuco, Brazil (n = 3): Dolphin Discovery, in the state of Quintana Roo, Mexico (n = 1); and the Caribbean Manatee Conservation Center at the Inter American University of Puerto Rico (n = 2). Recordings in Brazil were made with a Sony Walkman Pro WM-D6C Cassette Player (flat frequency response: 40–15,000 Hz ± 3 dB) and a Cetacean Research Technology 50Ca hydrophone (sensitivity: − 161 dB re: 1 µPa; sample rate: 44.1 kHz; resolution: 16 bits). Recordings of the two calves from Puerto Rico in 2011 were sampled with a SS03-10 Sea Phone directional hydrophone (sensitivity: − 169 dB re: 1 µPa; frequency range response: 20 Hz to 50 kHz) at a sample rate of 44.1 kHz in 16-bit resolution onto a Edirol R-44 digital recorder (frequency response 20 Hz to 40 kHz + 0/− 3 dB). The hydrophone was connected to two 30.5-cm suction cups, one placed on the throat and another on the nasal region caudal to the nares35. One calf recorded from Mexico was made with a Cetacean Research Technology 50Ca hydrophone (sensitivity: − 161 dB re: 1 µPa; sample rate: 44.1 kHz; resolution: 16 bits) sampling to an M-Audio MicroTrack II Digital Multi Track Recorder.
Amazonian manatees
Vocalizations from six isolated Amazonian manatee calves were recorded at four institutions in Brazil from 1998 to 2019: Instituto Nacional de Pesquisas da Amazônia in the state of Amazonas (n = 3 calves); Museu Paraense Emílio Goeldi in the state of Pará (n = 1 calf); and Centro de Preservação e Pesquisa de Mamíferos Aquáticos of Manaus Energia, in the state of Amazonas (n = 1 calf). More details on calf recordings can be found in Sousa-Lima et al.17,18. Neguinha’s vocalizations were recorded at Biologia e Conservação de Mamíferos Aquáticos da Amazônia in the state of Pará, Brazil using a SoundTrap HF300 acoustic recording system (Ocean Instruments, New Zealand) at a sample rate of 576 kHz in 16 bits. All other manatees (n = 5) were recorded with a Cetacean Research Technology 50Ca hydrophone (sensitivity: − 161 dB re: 1 µPa: sample rate: 44.1 kHz: resolution: 16 bits) on TDK SA60 cassette tapes using a Sony Walkman Pro WM-D6C Cassette Player and digitized at a sampling rate of 48 kHz.
The procedures during the recordings of Florida and Antillean manatees in Puerto Rico were approved by the US Fish and Wildlife Service (LOA #63658B and permit number M791721-4). Protocols on live animals in Puerto Rico were approved by the Inter American University’s Institutional Animal Care and Use Committee. Recordings of manatees in Brazil were approved by the Brazilian Government and the Animal Behavior Society’s Animal Care Committee. All methods were performed in accordance with relevant guidelines and regulations as suggested in the ARRIVE guidelines.
Acoustic analysis
To compare the acoustic parameters of the vocalizations of our three populations, we manually reviewed recordings by visually inspecting spectrograms to select high-quality calls for analysis. Spectrograms were calculated by fast Fourier transform (FFT) with a time resolution of 6.1 ms and a frequency resolution of 46.8 Hz (DFT: 1024 point; Hamming window; 50% overlap) in Raven Pro 1.539 Calls were selected if they had a signal-to-noise ratio (SNR) ≥ 6 dB and if they were clearly discernible with no overlapping calls or noise in the spectrogram. The fundamental frequency (the first harmonic in a series) of each call was boxed using the selection tool to draw a tight selection box (in the frequency and time domains) around the contour of the first harmonic. Amazonian manatees tend to vocalize in notes, i.e., short duration calls produced in a bout17; thus, all notes were included in the selection boundary when making measurements of the fundamental. Eight standard acoustic parameters were measured from the fundamental frequency: duration (ms); center frequency (Hz); duration (ms); bandwidth (Hz); maximum and minimum frequency (Hz); start and end frequency (Hz); and frequency modulation (Hz) (defined in Table 2 and illustrated in Fig. 1). Maximum and minimum frequency was derived from the average power spectrum across the entire measurement box using a 10 dB threshold40. Variables were chosen to be comparable with previous studies17,18.
Table 2.
Definitions of acoustic descriptors used in analysis of manatee calf vocalizations.
| Measurement | Description |
|---|---|
| Duration (ms) | Derived by subtracting the start and end time from the selection boundary |
| Center frequency (Hz) | The frequency that divides the selection into two frequency intervals of equal energy |
| Bandwidth (Hz) | Difference between the maximum and minimum frequency |
| Maximum frequency (Hz) | Maximum frequency of the call measured from the power spectrum |
| Minimum frequency (Hz) | Minimum frequency of the call measured from the power spectrum |
| Frequency modulation (Hz) | Calculated by subtracting the peak frequency contour min frequency from the peak frequency contour max frequency |
| Start and end peak frequency (Hz) | Start and end peak frequency obtained from pitch tracking algorithm within the selection boundaries |
Figure 1.
Schematics showing how the acoustic parameters of the vocalizations of manatee calves were extracted. The spectrogram on the left shows a single manatee call with the fundamental frequency of the call boxed in. The same box is expanded in the top right to show how duration (ms), frequency modulation (kHz) (peak frequency maximum minus peak frequency minimum), start frequency, and end frequency (kHz) were measured. The shaded area overlapping the call illustrates the power spectrum slice expanded in the bottom right showing the measurement of the power at the minimum and maximum frequencies (kHz) of the selected call. All measurements were conducted in Raven 1.639. Spectrogram parameters: DFT: 512 point; Hamming window; 50% overlap.
Statistical analysis
To determine if call variables differ across species, we performed a one-way multivariate analysis of covariance (MANCOVA) with species as a fixed factor and body length as a covariate. Calf call frequency and temporal variables were Box–Cox transformed to adjust their distribution to nearly normal41. For the purposes of this study, Florida and Antillean were treated as separate populations. Due to measuring several acoustic features on each call and performing multiple tests on the same data set, the chance of committing a Type 1 error increases. A sequential Bonferroni adjustment of 0.006 was used to reduce the possibility of an erroneous statistically significant result42. A test of significance between species was determined using Wilk's Lambda and the effect size was assessed with partial n2 statistics. A discriminant function analysis (DFA) was used to determine the call variables that best distinguished between species. We selected a regularized compromised discriminant method with lambda = 0 and gamma = 0.1 because the covariance matrices for the group compositions were significantly different (Box’s M = 448.32, df1 = 72, p < 0.001). The Kappa Index test was used to assess how well the discriminant function performed than expected by chance at a statistical significance level of 0.0543. The cross-validated method was used to calculate correct classification scores for the discriminant functions. Chi-square tests were used to evaluate the accuracy of the classification at a p-value of 0.0541. All calls from each species were treated as a group17,18. Prior probabilities were obtained from the number of calls used in analysis for each species. Although some variables were correlated, all variables were retained to minimize the loss of information44. All analyses were performed using SPSS version 28.045.
Results
Interspecies variation in call variables
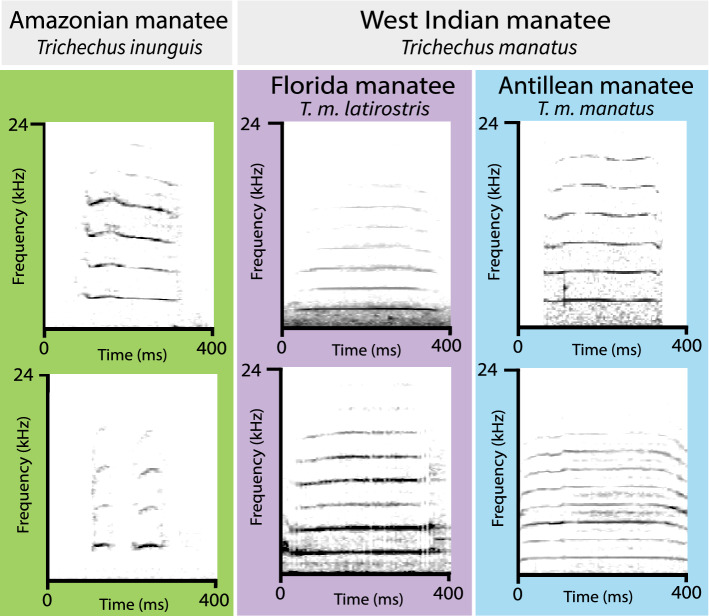
Florida and Antillean manatees (subspecies of the West Indian manatee) produced mostly tonal calls with hill-shaped contours (i.e., higher frequency modulation). Amazonian manatee calves also produced calls with frequency modulation, but it was not as accentuated as the hill-shaped calls observed in the Florida and Antillean manatees (Fig. 2). Amazonian calf calls contained one to three notes. The three smallest Amazonian calves produced calls that contained notes as well as the largest Amazonian manatee calf. Florida manatee calves produced calls that ranged in frequency from 2.25 to 3.42 kHz and call duration ranged from 0.089 to 0.371 ms. Antillean manatee calls had similar ranges to Florida manatee vocalizations for call duration (0.105–0.420 ms) and frequency range (1.87–3.96 kHz). The fundamental frequency and duration of Amazonian calf vocalizations ranged from 3.18 to 5.06 kHz and from 0.031 to 0.314 ms, respectively.
Figure 2.
Spectrograms of vocalizations recorded from individual calves in this study from the Amazonian manatee (Trichechus inunguis) and the two subspecies of the West Indian manatee (T. manatus)—the Antillean manatee (T. m. manatus) and the Florida manatee (T. m. longirostris).
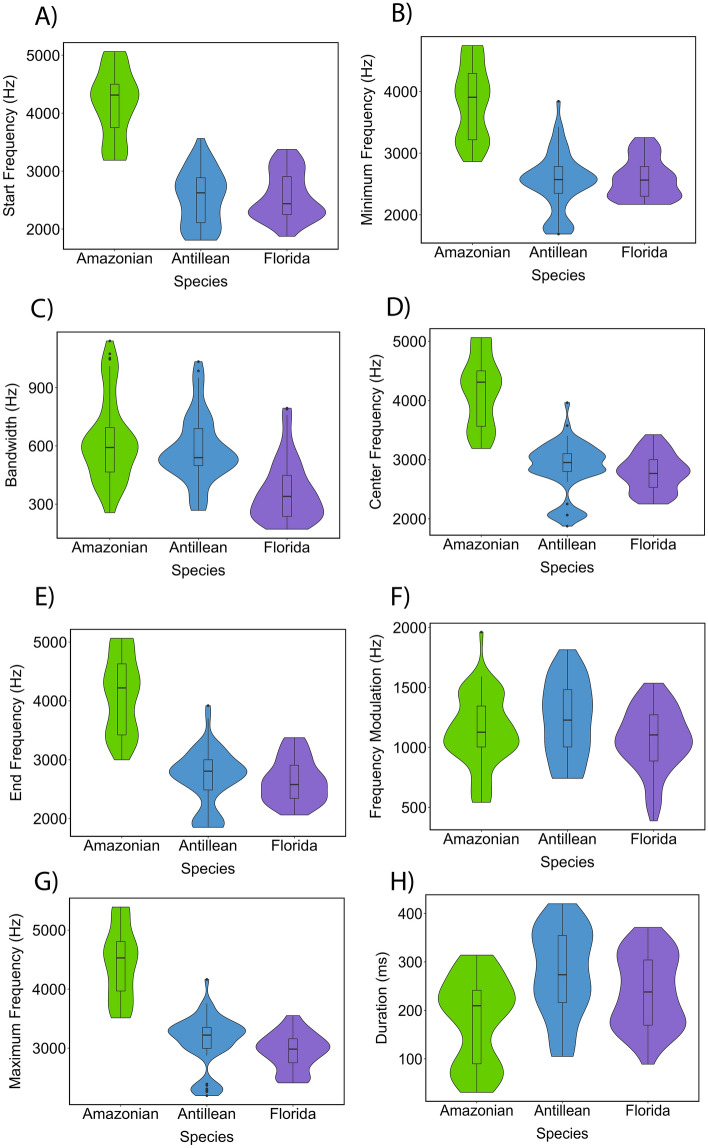
Amazonian manatees produced calls with shorter duration, higher start, center, minimum, end, and maximum frequencies compared to the Antillean and Florida manatees (Fig. 3A–H). After accounting for differences in body length, we found that Amazonian, Florida, and Antillean calves produce calls with acoustic features that are significantly different (Wilk’s λ = 0.21, F(16,340) = 24.53, p < 0.001). The multivariate n2 = 0.536, indicates that 53% of the multivariate variance of the call frequency and time variables is associated with the species.
Figure 3.
Manatees call frequency and temporal variation by populations. Box plots display medians, first and third quartiles, as well as standard error.
The discriminant analysis found that Functions 1 and 2 predict the differences in call acoustic variables between species (Function 1 through 2 Wilk’s λ = 0.198, χ2 = 282.97, p < 0.001 and Function 2 Wilk’s λ = 0.678, χ2 = 67.81, p < 0.001). Based on the standardized coefficients, function 1 was related primarily to minimum frequency and bandwidth, and function 2 was related to minimum, center, and start frequency, and bandwidth. Discriminant function 1 explained 70.8% of the differences in call acoustic structure among species (eigenvalue = 2.43, canonical correlation = 0.84) and function 2 explained 32.1% of the differences (eigenvalue = 0.475, canonical correlation = 0.567). The overall number of calls correctly classified to species was 82.9% (Kappa = 0.726, p < 0.001).
Differences between the West Indian (Florida and Antillean) manatee subspecies
We noted that the West Indian species had similar hill-shaped and linear call contours based on body size and differences in bandwidth as determined by the discriminant analysis. Frequency modulation and bandwidth can be correlated with body size17,18. Based on this, we performed an exploratory linear regression to predict if body length influenced frequency modulation and bandwidth across the two subspecies (Antillean and Florida) of the West Indian manatee. We used the mean values of each of the variables from each individual manatee calf across the two species17,41. Only frequency modulation was significantly correlated with body length (F (1,10) = 14.343, p < 0.004) with an R2 of 0.589. Frequency modulation decreased − 0.106 Hz with each increase in centimeter of body length, i.e., smaller animals vocalized with calls that tended to have a hill shape. As manatees grew in length, the hill shape appeared to flatten (Fig. 4).
Figure 4.

Ontogenetic changes from a hill-shape to a linear call contour of the vocalizations of the Florida manatee (T. m. latirostris) and the Antillean manatee (T. m. manatus). The Florida and Antillean manatee are subspecies of the West Indian manatee (Trichechus manatus),
Discussion
Identifying interspecific variation in the acoustic characteristics at different ages provides insights into the influence of different selective pressures on the ontogeny and function of acoustic calls. The goal of this study was to investigate how the vocalizations of the two subspecies of the West Indian manatee (Antillean and Florida) and the Amazonian manatee varied between populations. All three populations show similar morphology of the vocal tract35, which would suggest similarity in call contours and frequency and temporal characteristics. However, Amazonian calves tended to vocalize in multiple notes as opposed to one continuous call. In addition, Amazonian manatee calves produced calls that were higher in frequency and shorter in duration than Florida and Antillean manatees. This suggests that interspecific differences observed in Amazonian calf calls may have evolved due to factors unrelated to morphology of the vocal tract and may be related to body size. There were few differences in frequency and temporal values between Florida and Antillean manatee calves. Finally, we noted a decrease in frequency modulation in Florida and Antillean manatees that may be influenced by ontogeny.
The smaller body length of the Amazonian manatee may have caused the higher frequency and shorter duration vocalizations than the Antillean and Florida manatees. Amazonian manatees have a maximum total body length of 300 cm20 and are approximately 85–105 cm at birth46. Animals with a smaller body size tend to produce calls at higher frequencies than larger animals47. The body length of the Amazonian manatee remains relatively stable through the first year and increases slowly thereafter46,48. In contrast, the West Indian manatee species grows exponentially faster over the first year37,49. Body length may also partially explain the differences in duration of calls between species, i.e., Amazonian manatee calves produced shorter duration calls. In terrestrial animals, the duration of calls tends to lengthen with an increase in body size due to maturational changes in lung capacity50. The variability in growth patterns may influenced the frequency and temporal characteristics observed in Amazonian manatees.
Body size may have also influenced call contours in Florida and Antillean manatees. Call contours in younger, smaller animals were hill-shaped and tended to flatten in older, larger calves in the Florida and Antillean manatees. Observations from this and prior studies18,19,32 suggest that the hill-shaped call is a stereotypical call produced by smaller calves. Changes associated with lowering frequency modulation are likely due to the lengthening of the vocal tract with age17,18. The flattening of the hill shape over time as the animal becomes independent of its mother is likely associated with the transition during weaning and less dependency of the calf on the cow. Manatees may undergo periods of crystallization like passerine birds or as speculated for northern elephant seals in which after a certain age the structure of calls and variability of vocal repertoire are fixed51,52. These age-specific stereotypical hill-shaped features in Florida and Antillean manatees likely evolved to aid cows in discriminating calves from adult conspecifics. Furthermore, individualized acoustic parameters within calf calls can be used in recognition by lactating mothers17. This provides additional evidence of sirenian cognitive abilities to discriminate acoustic signals.
In addition to similarities in call contour, comparisons of measured variables of the fundamental frequency of Florida and Antillean manatee calves showed few statistically significant differences. Antillean and Florida manatees are genetically distinct from one another53 and diverged from a common ancestor approximately 1.34 Mya with some evidence for intermixing between populations54. Another plausible explanation for shared vocal characteristics is that West Indian manatees have similar habitats. Similar habitats can influence convergence in acoustic signals between genetically distinct populations55. Geographical variation in the call types of other species of marine mammals suggest the divergence of call types and structural components of calls are a common byproduct of local adaptation56,57.
Signal adaptation to different habitats could also have resulted in the differences observed in the Amazonian calf vocalizations. Many animals are reported to adjust the frequency and temporal content of vocalizations to optimize propagation of signals in their respective habitat58,59. The Amazonian manatee is completely freshwater dependent20 as opposed to Florida and Antillean manatees who are distributed throughout freshwater, brackish and marine habitats24. Freshwater and seawater ecosystems vary in sound propagation loss. For example, freshwater systems exhibit less sound absorption loss because they have less salt content than seawater systems. This could facilitate larger transmission distances of high-frequency signals60. However, a recent study found that frequency bands between 0.5–20 kHz have been shown to propagate efficiently in freshwater, marine and brackish habitats for the Antillean manatee and in seagrass beds for the Florida manatee59,61. Furthermore, behavioral audiograms identified hearing capabilities ranging from 0.4 to 90.5 kHz with peak sensitivity at 16 to 18 kHz in Florida manatees62,63. Evoked potential studies conducted with the Amazonian manatee show best hearing sensitivity between 5–20 kHz going up to a maximum of 50 kHz64. The overlap in hearing and frequency bands that propagate best in freshwater, marine and brackish environments suggest that habitat may not be the driver for the observed differences in vocalizations between Antillean, Amazonian and Florida manatee populations.
Similarly, the captive environment also may have influenced frequency and temporal parameters of vocalizations. Isolating manatees for recordings could have caused stress to the individual. Florida manatees under condition of stress (while being captured for health assessments) increased duration and had more frequency modulated calls32. Some of the Florida manatee calves were not isolated for this study, yet duration and frequency modulation of measured calls were not different from isolated Antillean manatees. Amazonian manatees were isolated, yet their calls were still shorter and showed less frequency modulation than Florida and Antillean manatees. Furthermore, fundamental frequencies recorded from wild Florida calves were reported ranging from 2.5 to 4 kHz19 and overlap with the fundamental frequency of vocalizations from captive Florida manatees in this study. Therefore, we believe the captive environment did not influence our results.
Future research should incorporate longitudinal comparisons of call structure of individuals to evaluate how the hill-shaped call structure changes in association with age, throughout development, and into adulthood. This would require localization of calls of individually recognizable animals and easiest done in captive facilities including aquariums and rehabilitation centers. Acoustic tags65 placed on young individuals could also reliably follow the ontogeny of vocalizations during development. Playback of hill-shaped calls of calves of different ages to female manatees (with dependent calves or recently independent calves) could be used to test whether the decreasing frequency modulation of call structure influences the behavior of a mother manatee. For example, if the calls of their own calf (known to elicit a strong response during lactation17) are recorded at various ages stimulate the same level of reactions when played back to the mother.
Conclusions
Our study provides evidence for interspecific variation in frequency and temporal parameters of manatee calf calls and in the ontogeny of call contours which is most likely related to body size. Amazonian manatee calves produce calls of higher frequency, shorter duration, and did not show the distinct hill-shaped contour used by both the Florida and Antillean manatee. Younger Florida and Antillean manatees tended to vocalize with a hill-shaped contour that appears to become more adult-like as it ages. Developmental changes in call structure seem to be absent in Amazonian manatees, corroborating previous evidence that strategies used by calves to call conspecific attention may be unique to Florida and Antillean manatees17,18. Our findings indicate Florida and Antillean calves retain call characteristics that make them distinguishable as calves from adults.
.
Acknowledgements
We would like to thank all the institutions mentioned in this manuscript for allowing us to record in their facilities. RSL thanks the Brazilian Council for Scientific and Technological Development (CNPq-Brazil) for her research fellowship (process number 312763/2019-0). This study was financed in part by the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior—Brasil (CAPES)—Finance Code 001, also by MacArthur Foundation, Fundação O Boticário de Proteção à Natureza, Conservation International, and U.S. Fish and Wildlife Service.
Author contributions
B.B., R.S.L. and E.R. wrote and edited the manuscript. E.R. prepared Figs. 1, 2, 3. L.M.C. assisted in statistical analysis. All authors reviewed the manuscript.
Data availability
All data is available upon reasonable request to the corresponding author.
Competing interests
The authors declare no competing interests.
Footnotes
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Podos J. Correlated evolution of morphology and vocal signal structure in Darwin's finches. Nature. 2001;409:185–188. doi: 10.1038/35051570. [DOI] [PubMed] [Google Scholar]
- 2.Bradbury JW, Vehrencamp S. Principles of Animal Communication. Sinauer Associated; 1998. [Google Scholar]
- 3.Podos J, Warren PS. The evolution of geographic variation in birdsong. Adv. Study Behav. 2007;37:403–458. [Google Scholar]
- 4.Charlton BD, Owen MA, Swaisgood RR. Coevolution of vocal signal characteristics and hearing sensitivity in forest mammals. Nat. Commun. 2019;10:1–7. doi: 10.1038/s41467-019-10768-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Soltis J. Vocal communication in African elephants (Loxodonta africana) Zoo Biol. 2010;29:192–209. doi: 10.1002/zoo.20251. [DOI] [PubMed] [Google Scholar]
- 6.King SL, Janik VM. Bottlenose dolphins can use learned vocal labels to address each other. Proc. Natl. Acad. Sci. 2013;110:13216–13221. doi: 10.1073/pnas.1304459110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Ravignani A, et al. Ontogeny of vocal rhythms in harbor seal pups: An exploratory study. Curr. Zool. 2019;65:107–120. doi: 10.1093/cz/zoy055. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Rauber R, Manser MB. Effect of group size and experience on the ontogeny of sentinel calling behaviour in meerkats. Anim. Behav. 2021;171:129–138. [Google Scholar]
- 9.Janik VM, Slater PJ. Vocal learning in mammals. Adv. Study Behav. 1997;26:59–100. [Google Scholar]
- 10.Fitch WT. Encyclopedia of Language and Linguistics. 2. Elsevier; 2006. Production of vocalizations in mammals; pp. 115–121. [Google Scholar]
- 11.Fletcher NH. Handbook of Behavioral Neuroscience. Elsevier; 2010. A frequency scaling rule in mammalian vocalization; pp. 51–56. [Google Scholar]
- 12.Fant G. Acoustic Theory of Speech Production. Mouton; 1960. [Google Scholar]
- 13.Taylor A, Reby D. The contribution of source–filter theory to mammal vocal communication research. J. Zool. 2010;280:221–236. [Google Scholar]
- 14.Fitch WT, Neubauer J, Herzel H. Calls out of chaos: The adaptive significance of nonlinear phenomena in mammalian vocal production. Anim. Behav. 2002;63:407–418. [Google Scholar]
- 15.Domning DP, Hayek LAC. Interspecific and intraspecific morphological variation in manatees (Sirenia: Trichechus) Mar. Mamm. Sci. 1986;2:87–144. [Google Scholar]
- 16.Anderson PK, Barclay RMR. Acoustic signals of solitary Dugongs: Physical characteristics and behavioral correlates. J. Mamm. 1995;76:1226–1237. [Google Scholar]
- 17.Sousa-Lima R, Paglia AP, Da Fonseca G. Signature information and individual recognition in the isolation calls of Amazonian manatees, Trichechus inunguis (Mammalia: Sirenia) Anim. Behav. 2002;63:301–310. [Google Scholar]
- 18.Sousa-Lima RS, Paglia AP, Fonseca GAB. Gender, age, and identity in the isolation calls of Antillean manatees (Trichechus manatus manatus) Aquat. Mamm. 2008;34:109–122. [Google Scholar]
- 19.O’Shea TJ, Poché LB. Aspects of underwater sound communication in Florida manatees (Trichechus manatus latirostris) J. Mamm. 2006;87:1061–1071. [Google Scholar]
- 20.Rosas FCW. Biology, conservation and status of the Amazonian manatee Trichechus inunguis. Mamm. Rev. 1994;24:49–59. [Google Scholar]
- 21.Meirelles, A. C. O. & Carvalho, V. L. Peixe-boi marinho: biologia e conservação no Brasil. Aquasis, Bambu Editora e Artes Gráficas, São Paulo (2016).
- 22.Alvarez-Alemán A, Beck CA, Powell JA. First report of a Florida manatee (Trichechus manatus latirostris) in Cuba. Aquat. Mamm. 2010;36:148. [Google Scholar]
- 23.Castelblanco-Martínez, D. N. et al. First documentation of long-distance travel by a Florida manatee to the Mexican Caribbean. Ethol. Ecol. Evol. 1–12 (2021).
- 24.Packard JM, Wetterqvist OF. Evaluation of manatee habitat systems on the northwestern Florida coast. Coast. Manag. 1986;14:279–310. [Google Scholar]
- 25.Luna FDO, et al. Genetic connectivity of the West Indian manatee in the southern range and limited evidence of hybridization with Amazonian manatees. Front. Mar. Sci. 2021;7:1089. [Google Scholar]
- 26.Hartman D. Ecology and behavior of the manatee (Trichechus manatus) in Florida. Spec. Publ. Am. Soc. Mammal. 1979;5:153. [Google Scholar]
- 27.D’AffonsecaNeto JA, Vergara-Parente JE. Sirenia (peixe-boi-da-Amazônia, Peixe-boi-marinho) In: Cubas ZS, Silva JCR, Catão-Dias JL, editors. Tratado de Animais Selvagens: medicina veterinária. Roca; 2006. pp. 701–714. [Google Scholar]
- 28.Deutsch, C. J., Reid, J. P., Bonde, R. K., Easton, D. E., Kochman, H. I. & O'Shea, T. J. Seasonal movements, migratory behavior, and site fidelity of West Indian manatees along the Atlantic coast of the United States. Wildl. Monogr. 1–77 (2003).
- 29.Laist DW, Reynolds JE., III Influence of power plants and other warm-water refuges on Florida manatees. Mar. Mamm. Sci. 2005;21:739–764. [Google Scholar]
- 30.Reynolds JE. Aspects of the social behaviour and herd structure of a semi-isolated colony of West Indian manatees, Trichechus manatus. Mammalia. 1981;45:431–452. [Google Scholar]
- 31.Dantas GA. Ontogenia do padrão vocal individual do peixe-boi da Amazônia Trichechus inunguis (Sirenia, trichechidae). Dissertação. Instituto Nacional de Pesquisas da Amazônia; 2009. [Google Scholar]
- 32.Brady, B., Moore, J. & Love, K. Behavior related vocalizations of the Florida manatee (Trichechus manatus latirostris). Mar. Mamm. Sci. 1–15 (2021).
- 33.Nowacek DP, Casper BM, Wells RS, Nowacek SM, Mann DA. Intraspecific and geographic variation of West Indian manatee (Trichechus manatus spp.) vocalizations. J. Acoust. Soc. Am. 2003;114:66–69. doi: 10.1121/1.1582862. [DOI] [PubMed] [Google Scholar]
- 34.Rycyk AM, et al. First characterization of vocalizations and passive acoustic monitoring of the vulnerable African manatee (Trichechus senegalensis) J. Acoust. Soc. Am. 2021;150:3028–3037. doi: 10.1121/10.0006734. [DOI] [PubMed] [Google Scholar]
- 35.Landrau-Giovannetti N, Mignucci-Giannoni AA, Reidenberg JS. Acoustical and anatomical determination of sound production and transmission in West Indian (Trichechus manatus) and Amazonian (T. inunguis) manatees. Anat. Rec. 2014;297:1896–1907. doi: 10.1002/ar.22993. [DOI] [PubMed] [Google Scholar]
- 36.Morton E. On the occurrence and significance of motivation-structural rules in some bird and mammal sounds. Am. Nat. 1977;111:855–869. [Google Scholar]
- 37.Borges JC, et al. Growth pattern differences of captive born Antillean manatee (Trichechus manatus) calves and those rescued in the Brazilian northeastern coast. J. Zoo Wildl. Med. 2012;43:494–500. doi: 10.1638/2011-0199R.1. [DOI] [PubMed] [Google Scholar]
- 38.Lima DS, Vergara-Parente JE, Young RJ, Paszkiewicz E. Training of Antillean manatee Trichechus manatus manatus Linnaeus, 1758 as a management technique for individual welfare. Lat. Am. J. Mar. Mamm. 2005;4:61–68. [Google Scholar]
- 39.K. Lisa Yang Center for Conservation Bioacoustics at the Cornell Lab of Ornithology. Raven Pro: Interactive Sound Analysis Software (Version 1.5) [Computer software]. https://ravensoundsoftware.com/ (The Cornell Lab of Ornithology, 2022).
- 40.Zollinger SA, Podos J, Nemeth E, Goller F, Brumm H. On the relationship between, and measurement of, amplitude and frequency in birdsong. Anim. Behav. 2012;84:e1–e9. [Google Scholar]
- 41.Sokal RR, Rohlf FJ. Biometry. W. H. Freeman and Co.; 1995. [Google Scholar]
- 42.Charrier I, Harcourt RG. Individual vocal identity in mother and pup Australian sea lions (Neophoca cinerea) J. Mamm. 2006;87:929–938. [Google Scholar]
- 43.Green S, Salkind NJ. Using SPSS for Windows and Macintosh: Analyzing and Understanding Data. 4. Prentice Hall; 2003. [Google Scholar]
- 44.Charlton BD, et al. Cues to body size in the formant spacing of male koala (Phascolarctos cinereus) bellows: Honesty in an exaggerated trait. J. Exp. Biol. 2011;214(20):3414–3422. doi: 10.1242/jeb.061358. [DOI] [PubMed] [Google Scholar]
- 45.IBM Corp. Released 2020. IBM SPSS Statistics for Windows, Version 27.0.
- 46.Best RC. The Amazon. Springer; 1984. The aquatic mammals and reptiles of the Amazon; pp. 371–412. [Google Scholar]
- 47.Gerhardt HC. The evolution of vocalization in frogs and toads. Annu. Rev. Ecol. Syst. 1994;25:293–324. [Google Scholar]
- 48.Mendoza, P. et al. Growth curve of Amazonian manatee (Trichechus inunguis) in captivity. Aquat. Mamm.45 (2019).
- 49.Schwarz LK. Methods and models to determine perinatal status of Florida manatee carcasses. Mar. Mamm. Sci. 2008;24:881–898. [Google Scholar]
- 50.Hauser MD, Chomsky N, Fitch WT. The faculty of language: What is it, who has it, and how did it evolve? Science. 2002;298:1569–1579. doi: 10.1126/science.298.5598.1569. [DOI] [PubMed] [Google Scholar]
- 51.Marler P, Peters S. Developmental overproduction and selective attrition: New processes in the epigenesis of birdsong. Dev. Psychol. J. Int. Soc. Dev. Psychol. 1982;15.4:369–378. doi: 10.1002/dev.420150409. [DOI] [PubMed] [Google Scholar]
- 52.Casey C, Reichmuth C, Costa DP, Le Boeuf B. The rise and fall of dialects in northern elephant seals. Proc. R. Soc. B. 2018;285:2018–2176. doi: 10.1098/rspb.2018.2176. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Hunter ME, et al. Puerto Rico and Florida manatees represent genetically distinct groups. Conserv. Genet. 2012;13:1623–1635. [Google Scholar]
- 54.Castelblanco-Martínez DN, et al. Analysis of body condition indices reveals different ecotypes of the Antillean manatee. Sci. Rep. 2021;11:1–14. doi: 10.1038/s41598-021-98890-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.McCracken KG, Sheldon FH. Avian vocalizations and phylogenetic signal. Proc. Natl. Acad. Sci. 1997;94:3833–3836. doi: 10.1073/pnas.94.8.3833. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Moron, J. R. et al. Whistle variability of Guiana dolphins in South America: Latitudinal variation or acoustic adaptation? Mar. Mamm. Sci. 1–32 (2018)
- 57.Luís AR, et al. Vocal universals and geographic variations in the acoustic repertoire of the common bottlenose dolphin. Sci. Rep. 2021;11:1–9. doi: 10.1038/s41598-021-90710-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Ey E, Fisher J. The ‘Acoustic adaptations hypothesis’ a review of the evidence from birds, anurans and mammals. Bioacoustics. 2009;19:21–48. [Google Scholar]
- 59.Miksis-Olds JL, Tyack PL. Manatee (Trichechus manatus) vocalization usage in relation to environmental noise levels. J. Acoust. Soc. Am. 2009;125:1806–1815. doi: 10.1121/1.3068455. [DOI] [PubMed] [Google Scholar]
- 60.Sun, W., Wang, Z., Jamalabdollahi, M. & Reza Zekavat, S. A. Experimental study on the difference between acoustic communication channels in freshwater rivers/lakes and in oceans. In 2014 48th Asilomar Conference on Signals, Systems and Computers, 333–337 (2004)
- 61.Rivera Chavarría M, Castro J, Camacho A. The relationship between acoustic habitat, hearing and tonal vocalizations in the Antillean manatee (Trichechus manatus manatus, Linnaeus, 1758) Biol. Open. 2015;4:1237–1242. doi: 10.1242/bio.013631. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Gaspard JC, III, et al. Audiogram and auditory critical ratios of two Florida manatees (Trichechus manatus latirostris) J. Exp. Biol. 2012;215:1442–1447. doi: 10.1242/jeb.065649. [DOI] [PubMed] [Google Scholar]
- 63.Gerstein ER, Gerstein L, Forsythe SE, Blue JE. The underwater audiogram of the West Indian manatee (Trichechus manatus) J. Acoust. Soc. Am. 1999;105:3575–3583. doi: 10.1121/1.424681. [DOI] [PubMed] [Google Scholar]
- 64.Klishin, V., Pezo, R., Popov, V., Ya, A. & Supin. Some characteristics of hearing of the Brazilian manatee, Trichechus inunguis. Aquat. Mamm. 16 (1990).
- 65.Johnson M, de Soto NA, Madsen PT. Studying the behaviour and sensory ecology of marine mammals using acoustic recording tags: A review. Mar. Ecol. Prog. Ser. 2009;395:55–73. [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
All data is available upon reasonable request to the corresponding author.