Abstract
Worldwide seagrass populations are in decline, calling for urgent measures in their conservation. Glyphosate is the most widely used herbicide globally, leading to increasing concern about its ecological impact, yet little is known about the prevalence or impact of glyphosate on seagrasses. In this study, we investigated the effect of sublethal glyphosate exposure on the endangered seagrass, Zostera capensis, to identify effects on growth, photosynthetic pigments and leaf morphology as measures of seagrass fitness. Seagrasses were exposed to a single dose of a commercial glyphosate formulation—ranging between 250 to 2,200 µg/L. After three weeks, the median leaf area decreased by up to 27%, with reductions of up to 31% in above ground biomass (p < 0.05). Photosynthetic pigment concentration showed no significant difference between groups. The observed effects on biomass and leaf area were seen at glyphosate levels below the regulatory limits set for surface water by several countries and may negatively affect the long-term resilience of this ecosystem engineer to additional stressors, such as those associated with climate change and anthropogenic pollution. As such, glyphosates and other herbicides that are washed into estuarine and marine ecosystems, pose a significant threat to the persistence of seagrasses and are important factors to consider in seagrass conservation, management and restoration efforts.
Keywords: Estuarine health, Herbicide, Seagrass conservation, Morphology, Zostera capensis
Introduction
Coastal vegetated ecosystems, including seagrass meadows, mangroves, and saltmarshes, play a vital role in sustaining marine biodiversity fisheries (Perkins-Visser, Wolcott & Wolcott, 1996; Verweij et al., 2008; Jänes et al., 2020). In addition, coastal vegetated systems are expected to play an increasingly important role in mitigating the impacts of climate change over the next decades, due their well-documented contributions to coastal protection (Ondiviela et al., 2014) and carbon sequestration (Fourqurean et al., 2012; Githaiga et al., 2017; Hilmi et al., 2021). Seagrass meadows, in particular, are known to be excellent carbon traps, especially in sediment, sequestering up to 30× more carbon than rainforest areas of similar size, and are estimated to contain between 4.2 and 8.4 PG carbon on a planetary scale (Fourqurean et al., 2012). In addition, seagrass meadows reduce the detrimental impact of pollutants and excess nutrients on estuaries and shallow marine environments (Levine et al., 1990; Olisah et al., 2021). However, seagrass meadows are globally in decline, as demonstrated by Waycott et al. (2009) who documented a decline in cover of approximately 7% per year since 1990, leading to urgent calls to increase seagrass conservation and management efforts.
Factors known to contribute to loss in seagrass cover include increased physical disturbance due to boating activity and bioturbation, as well as increased turbidity and eutrophication due to agricultural runoff (Burkholder, Tomasko & Touchette, 2007; Waycott et al., 2009; Mvungi & Pillay, 2019). Although eutrophication and decreased light availability (Ralph et al., 2007) due to turbidity are well-described drivers of seagrass decline, this is not the only impact agriculture has on seagrass meadows. Interest in the effects of herbicide and other pollutant runoff on this ecosystem has increased as ever-increasing environmental chemical loads become more evident (Persson et al., 2022). At this stage, the effects of pollutants on African seagrass species are poorly represented in the global literature, with research concentrated on Europe, the Americas and Australasia (Fraser & Kendrick, 2017; Gamain et al., 2018; Arcuri & Hendlin, 2019).
The Cape eelgrass, Zostera capensis, is the dominant seagrass species in South Africa, occurring in isolated shallow bays and estuaries along the coastline (Adams, 2016), with isolated populations also occurring in Mozambique and southern Kenya (Phair et al., 2019), but throughout its range it is poorly protected (van Niekerk et al., 2019). As of 2021, this species has been classified as endangered due to persistent population declines and the total loss of meadows in certain estuaries (Adams, 2016; van Niekerk et al., 2019), with associated losses in evolutionary resilience (Phair et al., 2020). Although some work has been done investigating regional declines, such as examining the impact of eutrophication on Zostera capensis (Mvungi & Pillay, 2019), very little is known about other contributing factors.
Even though glyphosate is now the most widely used herbicide globally by volume (Cuhra, Bøhn & Cuhra, 2016; Maggi et al., 2019), there is still relatively little research on its impact on aquatic and estuarine systems, compared to other pollutants and herbicides such as atrazine (Ralph, 2000; McMahon et al., 2005; Diepens et al., 2017) and heavy metals. Increasing agricultural application, together with glyphosate being licenced for the control of invasive aquatic weeds, has resulted in the detection of glyphosate in sediments and surface water across the globe, sometimes at concentrations above regulatory limits (Peruzzo, Porta & Ronco, 2008; Matozzo, Fabrello & Marin, 2020), although many uncertainties remain regarding its prevalence and persistence in aquatic systems. The exposure to and accumulation of glyphosate in estuarine and marine waters and sediments could as such significantly impact conservation efforts, including seagrass restoration if not measured and included in management actions (Lewis & Richard, 2009).
Comparatively little research has been done on the impact of this chemical on seagrasses, especially where long term exposure to sublethal levels is concerned. This is concerning, as even low levels of herbicides and pollutants have been shown to sometimes have significant, even synergistic effects on plant growth and fitness (Wilkinson et al., 2015; Diepens et al., 2017; Hughes et al., 2018), which is likely to be compounded by the increased stressors plants will be exposed to under climate change (Wilkinson et al., 2017). Further research into this area is thus needed, with a specific focus on the potential impact of pollutants and herbicides on coastal ecosystems in general and seagrass in particular.
In this study, we examine the impact on Z. capensis morphology, photosynthetic pigment concentration and growth, of prolonged exposure to sublethal levels of a commercial glyphosate formulation under a range of glyphosate concentrations. We used glyphosate levels that are based on levels detected by environmental sampling of surface waters in watersheds where glyphosate is extensively used for agriculture, as well as on published and proposed legislated regulatory limits in surface waters. We predicted that even at the lowest levels of glyphosate exposure (250 µg/L) which falls within legally permitted levels in many countries, seagrass morphology and biomass will be negatively impacted, with potential effects on long term survival and fitness. These results will add to the body of work demonstrating the need to revise and standardise regulatory glyphosate limits (Arcuri & Hendlin, 2019). Our work also highlights the importance of accounting for glyphosate for seagrass conservation, particularly for restoration efforts where ecologically intact environments increase restoration success. For example, exposure to environmental glyphosate is likely an important consideration when evaluating population declines (Diepens et al., 2017), prior to initiating restoration efforts and when prioritizing sites for restoration and protection (Devault, 2013).
Materials and Methods
Collection and acclimation
Zostera capensis ramets were collected in the Olifants river estuary (31.6982990S 18.2024690E; Fig. 1), from a 50 m transect along the intertidal zone. Shoots were collected at 5 m intervals, rinsed of sediment and stored in estuarine water in plastic resealable bags. Shoots were transported to the laboratory in an insulated box and planted within eight hours of collection. All collections were authorised under Department of Forestry, Fisheries and the Environment permit RES2021/68 and Cape Nature permit CN 35-87-15072.
Figure 1. Zostera capensis collection site, in the Western Cape province of South Africa, where the latter is shown as an inset.
Additional sediment was collected from an unvegetated area between seagrass beds and was well mixed by hand before being evenly distributed into plant containers. A total of 100 mL of beach sand was added to the top of the substrate to prevent disturbance and reduce turbidity when handling plants under experimental conditions. Containers were planted with one shoot from every transect point, resulting in 10 shoots per container. Shoots were selected to contain a set of roots and one leaf sheath with leaves. Shoot leaves were trimmed to 5 cm in length, and 1 cm of rhizome was planted, to approximate an equal starting biomass in every pot.
Polypropylene treatment tanks were prepared in advance, each containing 14 L artificial seawater (Red Sea salt™) at 31 ppt salinity, and two plant containers. The treatment tanks, each planted with 20 seagrass shoots, were placed in two, connected water baths to ensure consistent temperatures for all treatments. Polypropylene tanks were used rather than glass aquaria as glyphosate can be adsorbed onto glass surfaces, thus potentially decreasing concentrations of glyphosate (Goscinny et al., 2012).
Plants were acclimatised for four weeks at a water temperature of 21 °C (with daily measurements taken using a Thermochron iButton (iButtonLink, Whitewater, WI, USA)), while light was provided by a light array containing two T8 54 W actinic, and two T8 54 W 10,000 K daylight Odyssea fluorescent tubes, providing a surface irradiance of ~120 mmol/m2/s. A 16 h light/8 h dark cycle was maintained to provide >5 mol/m2/day PAR, in line with the minimum light requirement in a morphologically similar Zostera species occupying a similar environmental niche, Zostera muelleri (Bulmer, Kelly & Jeffs, 2016). A 20% water change was performed per week and aeration and filtration provided by one F-800 Dolphin aquarium filter per tank. Salinity was measured twice a week using a handheld Red Sea™ Seawater refractometer and maintained between 31 and 33 ppt by adding reverse osmosis water if needed. During acclimation, a marked increase in the number of shoots, roots and rhizomes occurred and plants grew well, which was important as glyphosate predominantly targets the actively growing parts of a plant (Kanissery et al., 2019). By the time glyphosate was added, all trimmed leaves had already senesced and the experiment was performed on new intact growth only.
After the acclimation period, treatment tanks were assigned to either control, 250, 750 or 2,200 µg/L treatment with Roundup™ using a randomised block design. The tested glyphosate concentrations were selected based on levels that have repeatedly been detected in environmental sampling (250 µg/L (Nielsen & Dahllöf, 2008; Battaglin et al., 2014)), legislated surface water limits (750 and 250 µg/L (Mensah, Palmer & Muller, 2013)) and levels experimentally shown to cause acute toxic effects in seagrasses (2,200 µg/L (de Castro et al., 2015)). Further, for studies comparing multiple treatments to a common control, it is recommended that the control group comprise at least 33% of the total samples (Bate & Karp, 2014), leading to a control group size of six tanks, compared to four tanks per treatment group.
Glyphosate based herbicides contain proprietary additives that may have significant additional toxic effects (Gandhi et al., 2021), although the exact nature of these additives is usually a trade secret. As these are the formulations used in practice, recent studies have aimed to use commercial formulations for toxicity testing, rather than pure glyphosate (Mesnage et al., 2015; Defarge et al., 2016; Parlapiano et al., 2021), to provide more representative effects such herbicides may be having in the environment. Roundup™ is the most commonly commercially available and applied (J. M. Dabrowski, 2019, personal communication) herbicide in South Africa and as such formed the basis of this study. A standard commercial agricultural product, Roundup Power 360 SL™ (glyphosate 360 g/L), was added to each individual tank at the beginning of the light cycle, at a dose calculated to achieve the desired concentration of the active ingredient, as glyphosate has been shown to have the greatest efficacy at this part of the circadian cycle in several plant species (Belbin et al., 2019).
A 20% water change was performed after seven days and again at 14 days to prevent fouling of seagrass by epiphytes. The experiment was conducted for 21 days, with chlorophyll, biomass and leaf morphology samples collected at the end of the experiment. Additionally, water samples were collected 24 h after treatment, to determine whether intended glyphosate concentrations had been achieved, and frozen at −20 °C until further analysis.
Morphometric analysis
In order to determine the impact of glyphosate on Z. capensis, the following measurements were taken; leaf length and width, above and below ground biomass and shoot density, as these have been found to adequately describe leaf morphology in seagrasses (Kuo & Hartog, 2006). Ten shoots were randomly selected from each of the two planted pots contained in every treatment tank (pot A and pot B) and the three longest leaves/shoot were measured for length and width, using Vernier callipers and leaf area calculated. Pot A samples were measured and harvested on day 21 and pot B samples on day 23, due to unforeseen circumstances. This was captured in the ‘Sample’ variable, which showed no statistically significant difference between groups on analysis. Plant material was washed and separated into above and below ground biomass, dried for eight hrs at 60 °C; dry matter weight was determined using an Explorer Ohaus electronic scale. Shoot density, above and below ground dry matter biomass, leaf length, width and area were compared using analysis of variance.
Glyphosate analysis
Water samples, collected 24 h after initiating treatment, were immediately frozen at −20 °C and maintained at that temperature until further analysis. Samples were analysed for glyphosate and its breakdown product, aminomethylphosphonic acid (AMPA), using the method described by Wang et al. (2016a) for seawater. Briefly, this entails derivatization of the sample with 9-fluorenylmethylchloroformate (FMOC-Cl), separation with high performance liquid chromatography (HPLC) and detection of glyphosate and AMPA by their fluorescence. At the Central Analytical Facility at Stellenbosch University, a Waters ACQUITY Ultra Performance Liquid Chromatography (UPLC) was coupled to a Xevo Fluorescence Detector (FLR) (Waters, Milford, MA, USA) and used for UPLC-FLR analysis. Glyphosate and AMPA were separated using a Waters ACQUITY BEH C18, 100 × 2.1 mm, 1.7 µm particle size column at 60 °C and a flow rate of 0.4 mL/min. An injection volume of 2 µl was used and the mobile phases consisted of 5 mmol/L ammonium acetate in water (Solvent A) and Methanol (Solvent B). The following gradient was used: 90% A, 0–0.54 min; 90–30% A, 0.5–6.0 min; 30–1% A, 6.0–7.0 min; 1–90% A, 7.0–8.0 min; 90% A, 8.0–10.0 min. The FLR was set at 265 nm (excitation) and 315 nm (emission).
Blank samples were run concurrently using distilled water adjusted to a salinity of 31 ppt. Calibration curves were created using an artificial seawater matrix, as Wang et al. (2016a) found no significant difference between natural seawater and artificial seawater calibration curves. The limit of detection (LOD) was determined to be 0.5 µg/L and limit of quantification (LOQ) 1 µg/L.
Chlorophyll and carotenoid content analysis
Leaf samples were collected at harvest and frozen at −20 °C until analysis. Chlorophyll and carotenoid content was extracted using a modified method, with 100% acetone used instead of 70% acetone (Pocock, Król & Huner, 2004). A total of 0.05 g of wet weight leaf sample (three samples from each tank) was ground in 1 mL of chilled acetone. The extract was centrifuged at 3,100 g for 5 min at a temperature of 10 °C. The supernatant was collected and chilled for 3 h at 4 °C, after which 50 µL of extract was added to 950 µL of 100% acetone. This was centrifuged at 2,000 g for seven minutes at a temperature of 4 °C. The absorbance of the supernatant was measured using a Biotec Power-wave HT. Total chlorophyll a and b, carotenoids and total chlorophyll concentration were calculated following Porra (2002), with details on equations provided in the Supplemental Table B.1.
Statistical analysis
Although Roundup™ was added to treatment tanks at levels calculated to achieve 250, 750 and 2,200 µg/L of the active ingredient glyphosate, there was a large variation in the concentration of glyphosate measurable in the tanks after 24 h, even within treatment groups, likely due to differences in adsorption to sediment and/or absorption into the plants themselves between different tanks. A total of 24 h post exposure, the 250 µg/L samples measured 106–181 µg/L (mean 148 µg/L), the 750 µg/L samples measured 370–750 µg/L (mean 533 µg/L) and the 2,200 µg/L samples measured 770–1,800 µg/L (mean 1,333 µg/L) (see Supplemental Material Table A.1).
In addition, the time weighted average exposure over the three-week treatment period is lower than initial exposure levels (see Supplementary Material Tables A.2 and A.3). Nonetheless, initial nominal values were used for all analyses, as glyphosate is known to need only 6 h of exposure for toxic effects to become apparent, with toxic effects taking up to three weeks to manifest according to the technical datasheet (Henderson et al., 2010). In addition to glyphosate, AMPA was detected during this experiment, with maximum levels of 25 µg/L at 24 h.
Analyses of variance (ANOVA) were performed on the different treatment groups, investigating effects on leaf area, leaf length, leaf width as morphological measurements, as well as above and below ground biomass as measures of growth. Groups were clustered according to the initial nominal dose of Roundup™, as well as on glyphosate concentration measured 24 h after administration. For leaf length, area and leaf width, 360 samples were measured for the control group and 240 for each of the three treatment groups.
While leaf width followed a normal distribution, leaf length, leaf area, chlorophyll and biomass data followed a gamma distribution. Leaf area and leaf length data were square root transformed to approximate a normal distribution of the residuals, for use in the ANOVA. Photosynthetic pigment data (control group n = 54, each treatment group n = 36) and biomass data (control group n = 12, each treatment group n = 8) could not be transformed and were analysed using a Kruskall-Wallis test, while residuals for leaf width did not need transformation.
All statistical analyses were carried out in R studio (RStudio Team, 2020), using the “car” and “ggplot2” libraries. The Aov() and Anova() functions were used, together with a Tukey Kramer post-hoc test for data following a normal distribution, and a Kruskall-Wallis and Dunn test performed on data that could not be transformed. Data were tested for normality using a Cullen and Frey graph from the R package “fitdistrplus”, and equal variance confirmed by the Levene test in “car” package. Tank 12 appeared as an outlier when inspecting the morphology data and two data sets were analysed; one including tank 12 and one without. Analysis on the reduced dataset is shown in the text (where n for the 750 µg/L treatment group was reduced to 180), while analysis of the full dataset is available in the Supplemental Materials. Biomass and chlorophyll data were analysed on the full dataset.
Data which had to be transformed for analysis are represented in their original form as boxplots in the Supplemental Material, while data with a normal distribution are represented with a plot of mean and confidence intervals in the text.
Results
Morphometric analysis
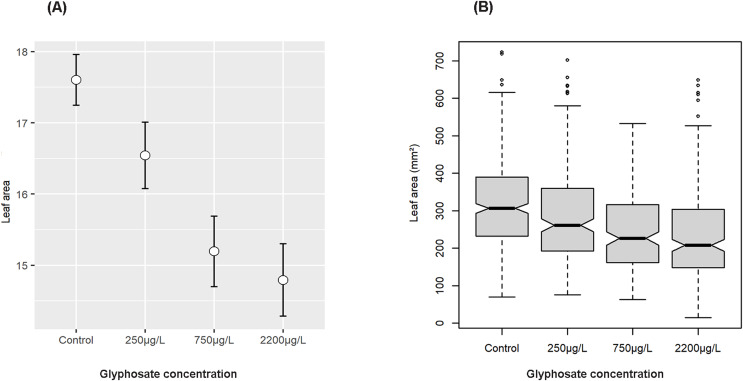
Analyses of leaf area, length and width performed on the reduced dataset, grouped according to nominal administered glyphosate levels, are presented. Glyphosate concentration had a strong, statistically significant negative effect on leaf area (F value 37.014, p = 2.2e−16, Figs. 2A and 2B), although an interaction between water baths and glyphosate treatment was also detected (F value 10.625, p = 7.002e−07; Type III ANOVA).
Figure 2. Effect of nominal glyphosate concentration on mean and confidence intervals of transformed leaf area (A) and notched boxplot showing median and interquartile range of leaf area (B) showing the decrease in leaf length with increasing glyphosate concentration.
Median leaf area decreased by 11% when treated with 250 µg/L of glyphosate and by 27% when treated with 2,200 µg/L.
The same was true for leaf length (F value 33.030, p = 2.2e−16, Figs. 3A and 3B), which also had an interaction with water bath (F value =12.104, p = 8.696e−08; Type III ANOVA). When the 24 h measured glyphosate concentrations instead of nominal administered treatment concentrations were used in analysis, this interaction fell away, indicating that the interaction may have been due to differing initial adsorption or absorption of glyphosate in the two baths, despite all efforts to standardise conditions.
Figure 3. Effect of nominal glyphosate concentration on mean and confidence intervals of transformed leaf length (A) and notched boxplot of median and interquartile range of leaf length (B) showing the decrease in leaf length with increasing glyphosate concentration.
Glyphosate concentration had a strong, statistically significant negative effect on leaf width (F value 31.225, p = 2.2e−16, Fig. 4), although an interaction between the two water baths and the glyphosate treatment was also detected (F value 8.135, p = 2.35e−05) (Type III ANOVA). Analyses on the full morphology dataset are provided in the Supplemental Material, showing the same trend as the curated dataset but with a weaker (although still statistically significant) effect on the 750 µg/L treatment group.
Figure 4. Mean and confidence intervals of leaf width (mm) as affected by nominal glyphosate concentration.
Shoot density between treatment and control groups was not statistically significant, and no trend was detected in the data (data not shown).
Above and below ground biomass data did not pass Q-Q plots for normality, therefore a Kruskal-Wallis rank sum test was performed. For above ground biomass, the effect was significant at the 90% confidence level (chi-squared 7.219, df = 3, p-value = 0.0653). A Dunn test was subsequently performed, only returning a significant p value for the difference between the control and 2,200 µg/L groups. However, a definite downward trend was observed in in the data (Fig. 5A), and the lack of statistical significance may be due to the smaller sample size for biomass data. The effect for 24 h glyphosate concentration was similar (data not shown).
Figure 5. Boxplot showing effect of nominal glyphosate concentration on (A) above and (B) below ground biomass.
Median above ground biomass declined with increasing glyphosate concentration, showing an 11% decrease when treated with 250 µg/L of glyphosate and 31% decrease when treated with 2,200 µg/L, while below ground biomass showed no trend.
Below ground biomass showed no pattern (Fig. 5B) and there was no statistically significant difference between different treatment groups (Kruskal-Wallis test, chi-squared 5.397, df = 3, p-value = 0.145). All data are available at https://github.com/vonderHeydenLab/van-Wyk-et-al_seagrass-and-glyhosate_raw-data.
Chlorophyll analysis
There were no statistically significant differences between groups for total chlorophyll content, chlorophyll a or chlorophyll b (Supplemental Material Table B.2). Carotenoid content was negligible.
Discussion
Brief overview of biodiversity impacts and global glyphosate legislation
Glyphosate is a post-emergent, non-selective herbicide that targets the shikimate pathway by inhibiting the enzyme 5-enolpyruvylshikimate-3-phosphate synthase (EPSPS) (Steinrücken & Amrhein, 1980). This enzyme is found in plants, algae, some bacteria, and fungi, but is absent in mammals. In plants, glyphosate disrupts folate, aromatic amino acid and lignin synthesis–leading to disrupted protein production, chlorophyll degradation and reduced growth—with maximum effect in areas of rapid metabolism such as root tips and shoot apices (Gomes et al., 2014). It is also a strong metal chelator, forming complexes with minerals such as iron and magnesium, leaving them unavailable to the plant (Cakmak et al., 2009). In seagrasses this is of particular concern, as iron availability reduces sulphide stress and promotes seagrass growth in anaerobic environments, such as that found under Zostera meadows (Holmer, Duarte & Marbá, 2005). In addition, there is evidence that glyphosate exposure can lead to increased susceptibility to disease, especially fungal root rot, in plants (Duke et al., 2012; van Bruggen et al., 2021).
Although glyphosate based herbicides were initially widely marketed as a safe alternative to older herbicides, there is increasing concern about their wider ecological impact (Mesnage et al., 2015; Sikorski et al., 2019; Matozzo, Fabrello & Marin, 2020; Corrales, Meerhoff & Antoniades, 2021) as well as their impact on human and animal health ( Peillex & Pelletier, 2020; van Bruggen et al., 2021; Vandenberg et al., 2017), with research revealing potential impacts on reproduction, renal health, carcinogenesis and even transgenerational effects due to epigenetic changes (Milesi et al., 2021). The potential long term effects on ecosystems may be severe, especially when considering the possibility of synergistic effects with other chemicals also present in the environment (Gamain et al., 2018).
This is of special relevance for organisms, such as aquatic plants, occupying lower trophic levels, where chronic sublethal effects on reproduction and fitness have the potential to disrupt entire ecosystems (Githaiga et al., 2019). In this regard, the limited research available on the impact of glyphosate and its formulations on marine and coastal systems is concerning, with indications that estuarine and marine organisms may be particularly sensitive to its effects. For example, it has recently been shown that glyphosate levels as low as 1 µg/L can lead to total loss of recruitment in the canopy forming marine macroalga, Carpodesmia crinita (de Caralt et al., 2020), with potential long term implications for population persistence.
In seawater, glyphosate has been shown to persist for between 47 and 315 days under low light conditions (Mercurio et al., 2014), while breakdown in sediment may take considerably longer than in water (Wang et al., 2016b) and sediment levels are often orders of magnitude higher than water levels in catchment areas where glyphosate is frequently applied, leading to organisms potentially being exposed to this herbicide for extended periods of time. Despite these concerns (Arcuri & Hendlin, 2019), regulatory limits vary immensely between countries and have been adjusted upwards in several jurisdictions as the demands of agriculture have increased. In the USA, where agriculture is dependent on massive herbicide use, the maximum residue level of glyphosate in drinking water is set at 700 µg/L (U.S. Environmental Protection Agency, 1995). Guidelines for the protection of the environment are set in a similar range. For example, the Australian water quality guideline for the protection of 99% of fresh water species sets a limit of 370 µg/L glyphosate in surface waters (Mercurio et al., 2014), while in South Africa, Mensah, Palmer & Muller (2013) suggested short and long term freshwater glyphosate limits of 250 µg/L and 2 µg/L respectively for the protection of indigenous aquatic biota. However, to date, no guidelines for glyphosate levels in South African waters have been legislated and it is rarely tested for.
Glyphosate impacts on an estuarine ecosystem engineer
There has been little research into the presence of agricultural pesticides (herbicides, insecticides and fungicides) in African waters (Porter et al., 2018; Horn, Pieters & Bøhn, 2019; Curchod et al., 2020), although there is a substantial body of research documenting the presence of herbicides and other agricultural chemicals in surface waters in other parts of the world (Wilkinson et al., 2015; Diepens et al., 2017) (Table 1). In this study, the glyphosate levels tested were based on environmentally realistic levels, as have been detected repeatedly in environmental sampling globally (Table 1), and also on the values proposed by regulatory authorities as allowable (U.S. Environmental Protection Agency, 1995; Mensah, Palmer & Muller, 2013). Ours is the first African study on the potential impacts of glyphosate on aquatic vegetation and suggests that even at levels within the suggested South African regulatory limit of 250 µg/L, and significantly below the USA regulatory limit of 700 µg/L, glyphosate was found to have a detrimental effect on seagrass leaf area, length, and width. The effect was strongly significant and increased with increasing levels of glyphosate, with declines in median leaf area of ~11% at the lowest level of glyphosate exposure and ~27% at the highest level, with median leaf length and width showing similar trajectories (Figs. 2–4). As expected, a decrease in above ground biomass was also noted. Further, the time weighted glyphosate exposure levels in this experiment were less than half the initial nominal dose of glyphosate administered (see Supplemental Material), which may indicate that Z. capensis is sensitive to even lower levels of glyphosate exposure than indicated by the initial doses. As our study only investigates the effect of glyphosate on a subsample of a single population of Zostera capensis, future studies should include specimens from multiple populations to determine if the effect holds true for genetically distinct populations (that have been demonstrated in Z. capensis; Phair et al., 2019), especially those from less pristine environments which may have acclimatised to herbicide exposure. In future, more studies should be carried out to identify clear chronic effect concentrations (EC10 values) for seagrass growth, based on measured concentrations.
Table 1. Glyphosate levels detected in recent environmental surface water samples.
| Country | Environment | Glyphosate concentration | Reference |
|---|---|---|---|
| USA | Groundwater | 2 µg/L | (Battaglin et al., 2014) |
| Precipitation | 2.5 µg/L | ||
| Large rivers | 3 µg/L | ||
| Streams | 73 µg/L | ||
| Wetlands | 302 µg/L | ||
| Ditches | 427 µg/L | ||
| Sediment | 476 µg/kg | ||
| Argentina | River | 125 µg/L | (Bonansea et al., 2017) |
| Argentina | River | Up to 0.7 µg/L | (Ronco et al., 2016) |
| Sediment | Up to 3,000 µg/kg | ||
| Argentina | River | Up to 1.80 µg/L | (Primost et al., 2017) |
| Sediment | Up to 3,294 µg/kg | ||
| Germany | Baltic Estuaries | 0.028 to 1.690 µg/l | (Skeff, Neumann & Schulz-Bull, 2015) |
| Sediment | 2 to 12 µg/kg | ||
| Denmark | Stream | 287 µg/L | (Nielsen & Dahllöf, 2008) |
| South Africa | Stream | 0.4 µg/L | (Horn, Pieters & Bøhn, 2019) |
In our study, Z. capensis leaves became consistently shorter and thinner as glyphosate concentration increased, with a concomitant decrease in above ground biomass. This is in contrast to research by Nielsen & Dahllöf (2008) which found an increase in above ground biomass and no effect on leaf morphology after three days of exposure to glyphosate levels of 169 and 1,600 µg/L. However, the lack of effect seen in Nielsen & Dahllöf (2008) may be due the short time frame used. Most research on glyphosate effects have been carried out over short time frames (<7 days), while glyphosate may take up to three weeks to have its full effect, even at commercial doses of 3.6 g/L (Kanissery et al., 2019). As such, the cumulative effect of long-term exposure to environmentally realistic levels are likely very different and future glyphosate impact studies will benefit from longer, more realistic exposure experiments.
Our findings of negative impacts on leaf morphology in aquatic plant species are not unique, as Sikorski et al. (2019) recorded similar levels of decline in response to similar concentrations of glyphosate-based formulations in the duckweed, Lemna minor. However, even with no significant differences in shoot density or chlorophyll concentration between treatment and control groups in Z. capensis, it is likely that the morphological changes observed may lead to decreased photosynthetic area available to the plant, in turn leading to decreased carbohydrate storage. This is supported by research showing that reducing above ground biomass during the growing season leads to lower carbohydrate storage in Zostera noltii, while exposure to low levels of glyphosate is also known to disrupt carbon metabolism in plants (Mateos-Naranjo & Perez-Martin, 2013). As lower carbohydrate stores are associated with decreased winter survival in seagrasses (Govers et al., 2015) and as decreased energy reserves may lead to lower resilience to stressors such as higher turbidity, freshwater run-off and disease, this may result in decreased fitness and survival over time. Reduced above ground biomass may also result in lower total carbon sequestration (Soissons et al., 2018) which may be of concern, as seagrass is estimated to store between 4.2 and 19.9 petagrams (Pg) of carbon globally (Fourqurean et al., 2012), and blue carbon is increasingly being investigated as a tool for climate mitigation (de Castro et al., 2015), including in South Africa (Adams et al., 2019). Although there was no statistically significant difference for below ground biomass in this study, it may that longer time frames would be needed to observe changes in this variable. The effect of chronic low grade glyphosate exposure on carbohydrate accumulation in rhizomes also invites further investigation, as chronic exposure to other herbicides like diuron has been shown to negatively affect below-ground starch reserves, even at concentrations lower than those that affected above ground leaf growth (Negri et al., 2015).
Future considerations for assessing herbicide impacts on the conservation of seagrasses
Our results support the assertion that in many countries, regulatory limits on the level of glyphosate-based herbicides allowed in surface waters may be set too high. In the view of increasing evidence of environmental harm at even relatively low levels, regulatory limits need to be reviewed and urgent attention given to alternative methods of weed control. Within the context of seagrass conservation, our work suggests that measuring glyphosate (and likely other herbicides) in for example, areas earmarked for restoration, is an important contributor to increasing restoration success and increasing both seagrass density and cover.
In addition, there is evidence in plants that reproductive life stages are more affected by glyphosate exposure than mature and seedling stages (Boutin et al., 2014), leading to reduced reproduction in plants exposed during the reproductive stage. As glyphosate is accumulating in sediments worldwide, associated with increasing application of the herbicide together with its slow breakdown in sediments, the effect of environmentally realistic levels in sediment on seed production (Boutin et al., 2014), germination and biomass of new shoots should be investigated. Although it has been shown that faba beans, oats and turnip rape had delayed germination and decreased shoot biomass after being grown in soil with a glyphosate concentration of 4.8 mg/kg (Helander et al., 2019), the effects in seagrasses and other marine macrophytes are still unknown. Given that many seagrass restoration projects rely on repopulating meadows by planting seeds (Unsworth et al., 2019; Govers et al., 2022), and that high levels of glyphosate in sediments may contribute to poor germination outcomes in certain plant species, determining sediment glyphosate concentrations may well be vital to restoration and conservation success.
In addition, synergistic effects of other environmental stressors and herbicide exposure should be further investigated. For example, research suggests that glyphosate may become less effective in some plant species at higher temperatures and CO2 concentrations (Matzrafi et al., 2019); however there remain limited insights and this has not been tested for seagrass species. Further, evidence suggests that sublethal combinations of chemicals, which on their own would have no effect, may have synergistic effects in combination (Nielsen & Dahllöf, 2008), potentially leading to reduced fitness, increased susceptibility to stressors such as increased water temperature and disease and even increased mortality (Gamain et al., 2018; Hughes et al., 2018). For example, Nielsen & Dahllöf (2008) showed that glyphosate mixed with other herbicides, at low levels such as those detected in the environment, act synergistically, having a far greater effect on growth and chlorophyll concentration of Zostera marina than would be suggested by individual toxicity testing. However, this is the only study we could find examining the effects of glyphosate in combination with other pollutants on seagrasses. Due to the increased chemical load in the environment, the impact of low-level glyphosate exposure when combined with environmentally realistic levels of other pollutants therefore warrant urgent further investigation in aquatic systems globally.
In conclusion, additional studies are urgently needed to investigate the effects of chronic long-term exposure to environmentally realistic levels of herbicides, including glyphosate, on seagrasses, particularly looking at effects on survival, carbohydrate storage and reproduction. Of special concern is the impact of cumulative stressors, such as increases in water temperatures and turbidity expected with climate change, as well as the potentially synergistic effects of multiple pollutants on seagrass resilience and survival in a rapidly changing world. This study improves our understanding of the interaction of anthropogenic pressures such as pollution on coastal aquatic ecosystems and the importance of accounting for these in conservation efforts of seagrasses worldwide.
Supplemental Information
Supplementary Materials for van Wyk et al._impacts of glyphosate on seagrass conservation
Acknowledgments
The authors wish to thank C. Hui for their advice on the statistical analyses and A. Ndhlovu for support during field lab work.
Funding Statement
This project was funded by the National Research Foundation South Africa through Marine and Coastal Research Grant Number 116048. The DSI/NRF Research Chair in Shallow Water Ecosystems (UID 84375) supported J. Adams. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Additional Information and Declarations
Competing Interests
The authors declare that they have no competing interests.
Author Contributions
Johanna W. van Wyk conceived and designed the experiments, performed the experiments, analyzed the data, prepared figures and/or tables, authored or reviewed drafts of the article, and approved the final draft.
Janine B. Adams conceived and designed the experiments, authored or reviewed drafts of the article, and approved the final draft.
Sophie von der Heyden conceived and designed the experiments, authored or reviewed drafts of the article, and approved the final draft.
Field Study Permissions
The following information was supplied relating to field study approvals (i.e., approving body and any reference numbers):
All collections were authorised under Department of Forestry, Fiserhies and the Environment permit RES2021/68 and Cape Nature permit CN 35-87-15072.
Data Availability
The following information was supplied regarding data availability:
The data is available at GitHub: www.github.com/vonderheydenlab/van-Wyk-et-al_seagrass-and-glyhosate_raw-data; Van Wyk, JW, Von der Heyden, S, & Adams, J. (2022). Conservation implications of herbicides on seagrasses: sublethal glyphosate exposure decreases fitness in the endangered Zostera capensis [Data set]. Zenodo. https://doi.org/10.5281/zenodo.7156564.
References
- Adams (2016).Adams JB. Distribution and status of Zostera capensis in South African estuaries—a review. South African Journal of Botany. 2016;107:63–73. doi: 10.1016/j.sajb.2016.07.007. [DOI] [Google Scholar]
- Adams et al. (2019).Adams J, Raw J, Banda-Mbense S, Bornman T, Rajkaran A, Van Niekerk L. Climate Change and South Africa’s blue carbon ecosystems. 2019. Water Research Commission Report No K5/2769.
- Arcuri & Hendlin (2019).Arcuri A, Hendlin YH. The chemical anthropocene: glyphosate as a case study of pesticide exposures. King’s Law Journal. 2019;30(2):234–253. doi: 10.1080/09615768.2019.1645436. [DOI] [Google Scholar]
- Bate & Karp (2014).Bate S, Karp NA. A common control group—optimising the experiment design to maximise sensitivity. PLOS ONE. 2014;9(12):1–12. doi: 10.1371/journal.pone.0114872. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Battaglin et al. (2014).Battaglin WA, Meyer MT, Kuivila KM, Dietze JE. Glyphosate and its degradation product AMPA occur frequently and widely in U.S. soils, surface water, groundwater, and precipitation. JAWRA Journal of the American Water Resources Association. 2014;50(2):275–290. doi: 10.1111/jawr.12159. [DOI] [Google Scholar]
- Belbin et al. (2019).Belbin FE, Hall GJ, Jackson AB, Schanschieff FE, Archibald G, Formstone C, Dodd AN. Plant circadian rhythms regulate the effectiveness of a glyphosate-based herbicide. Nature Communications. 2019;10:3704. doi: 10.1038/s41467-019-11709-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bonansea et al. (2017).Bonansea RI, Filippi I, Wunderlin DA, Marino DJG, Amé MV. The fate of glyphosate and AMPA in a freshwater endorheic basin: an ecotoxicological risk assessment. Toxics. 2017;6:1–13. doi: 10.3390/toxics6010003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Boutin et al. (2014).Boutin C, Strandberg B, Carpenter D, Mathiassen SK, Thomas PJ. Herbicide impact on non-target plant reproduction: what are the toxicological and ecological implications? Environmental Pollution. 2014;185(10):295–306. doi: 10.1016/j.envpol.2013.10.009. [DOI] [PubMed] [Google Scholar]
- Bulmer, Kelly & Jeffs (2016).Bulmer R, Kelly S, Jeffs A. Light requirements of the seagrass, Zostera muelleri, determined by observations at the maximum depth limit in a temperate estuary, New Zealand. New Zealand Journal of Marine and Freshwater Research. 2016;50(2):183–194. doi: 10.1080/00288330.2015.1120759. [DOI] [Google Scholar]
- Burkholder, Tomasko & Touchette (2007).Burkholder JAM, Tomasko DA, Touchette BW. Seagrasses and eutrophication. Journal of Experimental Marine Biology and Ecology. 2007;350(1–2):46–72. doi: 10.1016/j.jembe.2007.06.024. [DOI] [Google Scholar]
- Cakmak et al. (2009).Cakmak I, Yazici A, Tutus Y, Ozturk L. Glyphosate reduced seed and leaf concentrations of calcium, manganese, magnesium, and iron in non-glyphosate resistant soybean. European Journal of Agronomy. 2009;31(3):114–119. doi: 10.1016/j.eja.2009.07.001. [DOI] [Google Scholar]
- Corrales, Meerhoff & Antoniades (2021).Corrales N, Meerhoff M, Antoniades D. Glyphosate-based herbicide exposure affects diatom community development in natural biofilms. Environmental Pollution. 2021;284(3):117354. doi: 10.1016/j.envpol.2021.117354. [DOI] [PubMed] [Google Scholar]
- Cuhra, Bøhn & Cuhra (2016).Cuhra M, Bøhn T, Cuhra P. Glyphosate: too much of a good thing? Frontiers in Environmental Science. 2016;4:1–14. doi: 10.3389/fenvs.2016.00028. [DOI] [Google Scholar]
- Curchod et al. (2020).Curchod L, Oltramare C, Junghans M, Stamm C, Dalvie MA, Röösli M, Fuhrimann S. Temporal variation of pesticide mixtures in rivers of three agricultural watersheds during a major drought in the Western Cape, South Africa. Water Research X. 2020;6(27):100039. doi: 10.1016/j.wroa.2019.100039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- de Caralt et al. (2020).de Caralt S, Verdura J, Vergés A, Ballesteros E, Cebrian E. Differential effects of pollution on adult and recruits of a canopy-forming alga: implications for population viability under low pollutant levels. Scientific Reports. 2020;10(1):1–11. doi: 10.1038/s41598-020-73990-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- de Castro et al. (2015).de Castro AJV, Colares IG, dos Franco TCRS, Cutrim MVJ, Luvizotto-Santos R. Using a toxicity test with Ruppia maritima (Linnaeus) to assess the effects of Roundup. Marine Pollution Bulletin. 2015;91(2):506–510. doi: 10.1016/j.marpolbul.2014.10.006. [DOI] [PubMed] [Google Scholar]
- Defarge et al. (2016).Defarge N, Takács E, Lozano V, Mesnage R, Spiroux de Vendômois Jël, Séralini G-E, Székács A. Co-formulants in glyphosate-based herbicides disrupt aromatase activity in human cells below toxic levels. International Journal of Environmental Research and Public Health. 2016;13(3):264. doi: 10.3390/ijerph13030264. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Devault (2013).Devault D. Herbicide impact on seagrass communities. In: Price AJ, Kelton JA, editors. Herbicides - Current Research and Case Studies in Use. Houston: Intech; 2013. pp. 354–375. [Google Scholar]
- Diepens et al. (2017).Diepens NJ, Buffan-Dubau E, Budzinski H, Kallerhoff J, Merlina G, Silvestre J, Auby I, Nathalie T, Elger A. Toxicity effects of an environmental realistic herbicide mixture on the seagrass Zostera noltei. Environmental Pollution. 2017;222(8):393–403. doi: 10.1016/j.envpol.2016.12.021. [DOI] [PubMed] [Google Scholar]
- Duke et al. (2012).Duke SO, Lydon J, Koskinen WC, Moorman TB, Chaney RL, Hammerschmidt R. Glyphosate effects on plant mineral nutrition, crop rhizosphere microbiota, and plant disease in glyphosate-resistant crops. Journal of Agricultural and Food Chemistry. 2012;60(42):10375–10397. doi: 10.1021/jf302436u. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fourqurean et al. (2012).Fourqurean JW, Duarte CM, Kennedy H, Marbà N, Holmer M, Mateo MA, Apostolaki ET, Kendrick GA, Krause-Jensen D, McGlathery KJ, Serrano O. Seagrass ecosystems as a globally significant carbon stock. Nature Geoscience. 2012;5(7):505–509. doi: 10.1038/ngeo1477. [DOI] [Google Scholar]
- Fraser & Kendrick (2017).Fraser MW, Kendrick GA. Belowground stressors and long-term seagrass declines in a historically degraded seagrass ecosystem after improved water quality. Scientific Reports. 2017;7(1):14469. doi: 10.1038/s41598-017-14044-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gamain et al. (2018).Gamain P, Feurtet-Mazel A, Maury-Brachet R, Auby I, Pierron F, Belles A, Budzinski H, Daffe G, Gonzalez P. Can pesticides, copper and seasonal water temperature explain the seagrass Zostera noltei decline in the Arcachon bay? Marine Pollution Bulletin. 2018;134(4):66–74. doi: 10.1016/j.marpolbul.2017.10.024. [DOI] [PubMed] [Google Scholar]
- Gandhi et al. (2021).Gandhi K, Khan S, Patrikar M, Markad A, Kumar N, Choudhari A, Sagar P, Indurkar S. Exposure risk and environmental impacts of glyphosate: highlights on the toxicity of herbicide co-formulants. Environmental Challenges. 2021;4(8):100149. doi: 10.1016/j.envc.2021.100149. [DOI] [Google Scholar]
- Githaiga et al. (2019).Githaiga MN, Frouws AM, Kairo JG, Huxham M. Seagrass removal leads to rapid changes in Fauna and loss of carbon. Frontiers in Ecology and Evolution. 2019;7:1–12. doi: 10.3389/fevo.2019.00062. [DOI] [Google Scholar]
- Githaiga et al. (2017).Githaiga MN, Kairo JG, Gilpin L, Huxham M. Carbon storage in the seagrass meadows of Gazi Bay, Kenya. PLOS ONE. 2017;12(5):e0177001. doi: 10.1371/journal.pone.0177001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gomes et al. (2014).Gomes MP, Smedbol E, Chalifour A, Hénault-Ethier L, Labrecque M, Lepage L, Lucotte M, Juneau P. Alteration of plant physiology by glyphosate and its by-product aminomethylphosphonic acid: an overview. Journal of Experimental Botany. 2014;65(17):4691–4703. doi: 10.1093/jxb/eru269. [DOI] [PubMed] [Google Scholar]
- Goscinny et al. (2012).Goscinny S, Unterluggauer H, Aldrian J, Hanot V, Masselter S. Determination of glyphosate and its metabolite AMPA (Aminomethylphosphonic Acid) in cereals after derivatization by isotope dilution and UPLC-MS/MS. Food Analytical Methods. 2012;5(5):1177–1185. doi: 10.1007/s12161-011-9361-7. [DOI] [Google Scholar]
- Govers et al. (2015).Govers LL, Suykerbuyk W, Hoppenreijs JHT, Giesen K, Bouma TJ, van Katwijk MM. Rhizome starch as indicator for temperate seagrass winter survival. Ecological Indicators. 2015;49:53–60. doi: 10.1016/j.ecolind.2014.10.002. [DOI] [Google Scholar]
- Govers et al. (2022).Govers LL, Heusinkveld JHT, Gräfnings MLE, Smeele Q, van der Heide T. Adaptive intertidal seed-based seagrass restoration in the Dutch Wadden Sea. PLOS ONE. 2022;17:e0262845. doi: 10.1371/journal.pone.0262845. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Helander et al. (2019).Helander M, Pauna A, Saikkonen K, Saloniemi I. Glyphosate residues in soil affect crop plant germination and growth. Scientific Reports. 2019;9(1):19653. doi: 10.1038/s41598-019-56195-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Henderson et al. (2010).Henderson AM, Gervais JA, Luukinen B, Buhl K, Stone D, Strid A, Cross A, Jenkins J. Glyphosate technical fact sheet. 2010. http://npic.orst.edu/factsheets/archive/glyphotech.html http://npic.orst.edu/factsheets/archive/glyphotech.html National Pesticide Information Center, Oregon State University. ExtServices.
- Hilmi et al. (2021).Hilmi N, Chami R, Sutherland MD, Hall-Spencer JM, Lebleu L, Benitez MB, Levin LA. The role of blue carbon in climate change mitigation and carbon stock conservation. Frontiers in Climate. 2021;3:102. doi: 10.3389/fclim.2021.710546. [DOI] [Google Scholar]
- Holmer, Duarte & Marbá (2005).Holmer M, Duarte CM, Marbá N. Iron additions reduce sulfate reduction rates and improve seagrass growth on organic-enriched carbonate sediments. Ecosystems. 2005;8(6):721–730. doi: 10.1007/s10021-003-0180-6. [DOI] [Google Scholar]
- Horn, Pieters & Bøhn (2019).Horn S, Pieters R, Bøhn T. A first assessment of glyphosate, 2,4-D and Cry proteins in surface water of South Africa. South African Journal of Science. 2019;115(9/10):1–7. doi: 10.17159/sajs.2019/5988. [DOI] [Google Scholar]
- Hughes et al. (2018).Hughes RG, Potouroglou M, Ziauddin Z, Nicholls JC. Seagrass wasting disease: nitrate enrichment and exposure to a herbicide (Diuron) increases susceptibility of Zostera marina to infection. Marine Pollution Bulletin. 2018;134(170):94–98. doi: 10.1016/j.marpolbul.2017.08.032. [DOI] [PubMed] [Google Scholar]
- Jänes et al. (2020).Jänes H, Macreadie PI, Zu Ermgassen PSE, Gair JR, Treby S, Reeves S, Nicholson E, Ierodiaconou D, Carnell P. Quantifying fisheries enhancement from coastal vegetated ecosystems. Ecosystem Services. 2020;43(1):101105. doi: 10.1016/j.ecoser.2020.101105. [DOI] [Google Scholar]
- Kanissery et al. (2019).Kanissery R, Gairhe B, Kadyampakeni D, Batuman O, Alferez F. Glyphosate: its environmental persistence and impact on crop health and nutrition. Plants. 2019;8(11):499. doi: 10.3390/plants8110499. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kuo & Hartog (2006).Kuo J, Hartog CD. Seagrasses: Biology, Ecology and Conservation. Dordrecht: Springer Netherlands; 2006. Seagrass Morphology, Anatomy, and Ultrastructure; pp. 51–87. [Google Scholar]
- Levine et al. (1990).Levine SN, Rudnick DT, Kelly JR, Morton RD, Buttel LA, Carr KA. Pollutant dynamics as influenced by seagrass beds: experiments with tributyltin in thalassia microcosms. Marine Environmental Research. 1990;30(4):297–322. doi: 10.1016/0141-1136(90)90004-8. [DOI] [Google Scholar]
- Lewis & Richard (2009).Lewis MA, Richard D. Nonnutrient anthropogenic chemicals in seagrass ecosystems: fate and effects. Environmental Toxicology and Chemistry. 2009;28(3):644–661. doi: 10.1897/08-201.1. [DOI] [PubMed] [Google Scholar]
- Maggi et al. (2019).Maggi F, Tang FHM, La Cecilia D, Mcbratney A. PEST-CHEMGRIDS, global gridded maps of the top 20 crop-specific pesticide application rates from 2015 to 2025. Scientific Data. 2019;6(1):279. doi: 10.1038/s41597-019-0169-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mateos-Naranjo & Perez-Martin (2013).Mateos-Naranjo E, Perez-Martin A. Effects of sub-lethal glyphosate concentrations on growth and photosynthetic performance of non-target species Bolboschoenus maritimus. Chemosphere. 2013;93(10):2631–2638. doi: 10.1016/j.chemosphere.2013.09.094. [DOI] [PubMed] [Google Scholar]
- Matozzo, Fabrello & Marin (2020).Matozzo V, Fabrello J, Marin MG. The effects of glyphosate and its commercial formulations to marine invertebrates: a review. Journal of Marine Science and Engineering. 2020;8(6):399. doi: 10.3390/jmse8060399. [DOI] [Google Scholar]
- Matzrafi et al. (2019).Matzrafi M, Brunharo C, Tehranchian P, Hanson BD, Jasieniuk M. Increased temperatures and elevated CO2 levels reduce the sensitivity of Conyza canadensis and Chenopodium album to glyphosate. Scientific Reports. 2019;9(1):2228. doi: 10.1038/s41598-019-38729-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McMahon et al. (2005).McMahon K, Nash SB, Eaglesham G, Müller JF, Duke NC, Winderlich S. Herbicide contamination and the potential impact to seagrass meadows in Hervey Bay, Queensland, Australia. Marine Pollution Bulletin. 2005;51(1–4):325–334. doi: 10.1016/j.marpolbul.2004.10.045. [DOI] [PubMed] [Google Scholar]
- Mensah, Palmer & Muller (2013).Mensah PK, Palmer CG, Muller WJ. Derivation of South African water quality guidelines for Roundup® using species sensitivity distribution. Ecotoxicology and Environmental Safety. 2013;96(2):24–31. doi: 10.1016/j.ecoenv.2013.06.009. [DOI] [PubMed] [Google Scholar]
- Mercurio et al. (2014).Mercurio P, Flores F, Mueller JF, Carter S, Negri AP. Glyphosate persistence in seawater. Marine Pollution Bulletin. 2014;85(2):385–390. doi: 10.1016/j.marpolbul.2014.01.021. [DOI] [PubMed] [Google Scholar]
- Mesnage et al. (2015).Mesnage R, Defarge N, Spiroux de Vendômois J, Séralini GE. Potential toxic effects of glyphosate and its commercial formulations below regulatory limits. Food and Chemical Toxicology. 2015;84(1):133–153. doi: 10.1016/j.fct.2015.08.012. [DOI] [PubMed] [Google Scholar]
- Milesi et al. (2021).Milesi MM, Lorenz V, Durando M, Rossetti MF, Varayoud J. Glyphosate herbicide: reproductive outcomes and multigenerational effects. Frontiers in Endocrinology (Lausanne) 2021;12:672532. doi: 10.3389/FENDO.2021.672532/FULL. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mvungi & Pillay (2019).Mvungi EF, Pillay D. Eutrophication overrides warming as a stressor for a temperate African seagrass (Zostera capensis) PLOS ONE. 2019;14(4):1–17. doi: 10.1371/journal.pone.0215129. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Negri et al. (2015).Negri AP, Flores F, Mercurio P, Mueller JF, Collier CJ. Lethal and sub-lethal chronic effects of the herbicide diuron on seagrass. Aquatic Toxicology. 2015;165:73–83. doi: 10.1016/j.aquatox.2015.05.007. [DOI] [PubMed] [Google Scholar]
- Nielsen & Dahllöf (2008).Nielsen LW, Dahllöf I. Corrigendum to “Direct and indirect effects of the herbicides Glyphosate, Bentazone and MCPA on eelgrass (Zostera marina)” [Aquat. Toxicol. 82 (1) (2007) 47–54] Aquatic Toxicology. 2008;90(4):333. doi: 10.1016/j.aquatox.2008.09.001. [DOI] [PubMed] [Google Scholar]
- Olisah et al. (2021).Olisah C, Human LRD, Rubidge G, Adams JB. Organophosphate pesticides sequestered in tissues of a seagrass species—Zostera capensis from a polluted watershed. Journal of Environmental Management. 2021;300(6):113657. doi: 10.1016/j.jenvman.2021.113657. [DOI] [PubMed] [Google Scholar]
- Ondiviela et al. (2014).Ondiviela B, Losada IJ, Lara JL, Maza M, Galván C, Bouma TJ, van Belzen J. The role of seagrasses in coastal protection in a changing climate. Coastal Engineering. 2014;87(1):158–168. doi: 10.1016/j.coastaleng.2013.11.005. [DOI] [Google Scholar]
- Parlapiano et al. (2021).Parlapiano I, Biandolino F, Grattagliano A, Ruscito A, Libralato G, Prato E. Effects of commercial formulations of glyphosate on marine crustaceans and implications for risk assessment under temperature changes. Ecotoxicology and Environmental Safety. 2021;213(3):112068. doi: 10.1016/j.ecoenv.2021.112068. [DOI] [PubMed] [Google Scholar]
- Peillex & Pelletier (2020).Peillex C, Pelletier M. The impact and toxicity of glyphosate and glyphosate-based herbicides on health and immunity. Journal of Immunotoxicology. 2020;17(1):163–174. doi: 10.1080/1547691X.2020.1804492. [DOI] [PubMed] [Google Scholar]
- Perkins-Visser, Wolcott & Wolcott (1996).Perkins-Visser E, Wolcott TG, Wolcott DL. Nursery role of seagrass beds: enhanced growth of juvenile blue crabs (Callinectes sapidus Rathbun) Journal of Experimental Marine Biology and Ecology. 1996;198(2):155–173. doi: 10.1016/0022-0981(96)00014-7. [DOI] [Google Scholar]
- Persson et al. (2022).Persson L, Carney Almroth BM, Collins CD, Cornell S, de Wit CA, Diamond ML, Fantke P, Hassellöv M, MacLeod M, Ryberg MW, Søgaard Jørgensen P, Villarrubia-Gómez P, Wang Z, Hauschild MZ. Outside the safe operating space of the planetary boundary for novel entities. Environmental Science & Technology. 2022;56(3):1510–1521. doi: 10.1021/acs.est.1c04158. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Peruzzo, Porta & Ronco (2008).Peruzzo PJ, Porta AA, Ronco AE. Levels of glyphosate in surface waters, sediments and soils associated with direct sowing soybean cultivation in north pampasic region of Argentina. Environmental Pollution. 2008;156(1):61–66. doi: 10.1016/j.envpol.2008.01.015. [DOI] [PubMed] [Google Scholar]
- Phair et al. (2019).Phair NL, Toonen RJ, Knapp I, von der Heyden S. Shared genomic outliers across two divergent population clusters of a highly threatened seagrass. PeerJ. 2019;7:e6806. doi: 10.7717/peerj.6806. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Phair et al. (2020).Phair NL, Toonen RJ, Knapp IS, von der Heyden S. Anthropogenic pressures negatively impact genomic diversity of the vulnerable seagrass Zostera capensis. Journal of Environmental Management. 2020;255:109831. doi: 10.1016/j.jenvman.2019.109831. [DOI] [PubMed] [Google Scholar]
- Pocock, Król & Huner (2004).Pocock T, Król M, Huner NPA. The determination and quantification of photosynthetic pigments by reverse phase high-performance liquid chromatography, thin-layer chromatography, and spectrophotometry. Methods in Molecular Biology. 2004;274:137–148. doi: 10.1385/1592597998. [DOI] [PubMed] [Google Scholar]
- Porra (2002).Porra RJ. The chequered history of the development and use of simultaneous equations for the accurate determination of chlorophylls a and b. Photosynthesis Research. 2002;73(1/3):149–156. doi: 10.1023/A:1020470224740. [DOI] [PubMed] [Google Scholar]
- Porter et al. (2018).Porter SN, Humphries MS, Buah-Kwofie A, Schleyer MH. Accumulation of organochlorine pesticides in reef organisms from marginal coral reefs in South Africa and links with coastal groundwater. Marine Pollution Bulletin. 2018;137(11):295–305. doi: 10.1016/j.marpolbul.2018.10.028. [DOI] [PubMed] [Google Scholar]
- Primost et al. (2017).Primost JE, Marino DJG, Aparicio VC, Costa JL, Carriquiriborde P. Glyphosate and AMPA, “pseudo-persistent” pollutants under real-world agricultural management practices in the Mesopotamic Pampas agroecosystem, Argentina. Environmental Pollution. 2017;229:771–779. doi: 10.1016/j.envpol.2017.06.006. [DOI] [PubMed] [Google Scholar]
- Ralph (2000).Ralph PJ. Herbicide toxicity of Halophila ovalis assessed by chlorophyll a fluorescence. Aquatic Botany. 2000;66(2):141–152. doi: 10.1016/S0304-3770(99)00024-8. [DOI] [Google Scholar]
- Ralph et al. (2007).Ralph PJ, Durako MJ, Enríquez S, Collier CJ, Doblin MA. Impact of light limitation on seagrasses. Journal of Experimental Marine Biology and Ecology. 2007;350(1–2):176–193. doi: 10.1016/j.jembe.2007.06.017. [DOI] [Google Scholar]
- Ronco et al. (2016).Ronco AE, Marino DJG, Abelando M, Almada P, Apartin CD. Water quality of the main tributaries of the Paraná Basin: glyphosate and AMPA in surface water and bottom sediments. Environmental Monitoring and Assessment. 2016:188. doi: 10.1007/s10661-016-5467-0. [DOI] [PubMed] [Google Scholar]
- RStudio Team (2020).RStudio Team . RStudio: integrated development for R. RStudio, PBC, Boston, MA: 2020. [Google Scholar]
- Sikorski et al. (2019).Sikorski Ł, Baciak M, Bęś A, Adomas B. The effects of glyphosate-based herbicide formulations on Lemna minor, a non-target species. Aquatic Toxicology. 2019;209:70–80. doi: 10.1016/j.aquatox.2019.01.021. [DOI] [PubMed] [Google Scholar]
- Skeff, Neumann & Schulz-Bull (2015).Skeff W, Neumann C, Schulz-Bull DE. Glyphosate and AMPA in the estuaries of the Baltic Sea method optimization and field study. Marine Pollution Bulletin. 2015;100:577–585. doi: 10.1016/j.marpolbul.2015.08.015. [DOI] [PubMed] [Google Scholar]
- Soissons et al. (2018).Soissons LM, Haanstra EP, van Katwijk MM, Asmus R, Auby I, Barillé L, Brun FG, Cardoso PG, Desroy N, Fournier J, Ganthy F, Garmendia J-M, Godet L, Grilo TF, Kadel P, Ondiviela B, Peralta G, Puente A, Recio M, Rigouin L, Valle M, Herman PMJ, Bouma TJ. Latitudinal patterns in European seagrass carbon reserves: influence of seasonal fluctuations versus short-term stress and disturbance events. Frontiers in Plant Science. 2018;9:88. doi: 10.3389/fpls.2018.00088. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Steinrücken & Amrhein (1980).Steinrücken HC, Amrhein N. The herbicide glyphosate is a potent inhibitor of 5-enolpyruvylshikimic acid-3-phosphate synthase. Biochemical and Biophysical Research Communications. 1980;94(4):1207–1212. doi: 10.1016/0006-291X(80)90547-1. [DOI] [PubMed] [Google Scholar]
- U.S. Environmental Protection Agency (1995).U.S. Environmental Protection Agency National primary drinking water regulations: glyphosate {Technical Version} 1995. https://nepis.epa.gov/Exe/ZyPURL.cgi?Dockey=9100PO3S.txt https://nepis.epa.gov/Exe/ZyPURL.cgi?Dockey=9100PO3S.txt U.S. Environmental Protection Agency.
- Unsworth et al. (2019).Unsworth RKF, Bertelli CM, Cullen-Unsworth LC, Esteban N, Jones BL, Lilley R, Lowe C, Nuutila H, Rees SC. Sowing the seeds of seagrass recovery using Hessian Bags. Frontiers in Environmental Science. 2019;7:311. doi: 10.3389/fevo.2019.00311. [DOI] [Google Scholar]
- van Bruggen et al. (2021).van Bruggen AHC, Finckh MR, He M, Ritsema CJ, Harkes P, Knuth D, Geissen V. Indirect effects of the herbicide glyphosate on plant, animal and human health through its effects on microbial communities. Frontiers in Environmental Science. 2021;9:464. doi: 10.3389/fenvs.2021.763917. [DOI] [Google Scholar]
- van Niekerk et al. (2019).van Niekerk L, Adams J, Lamberth S, MacKay C, Taljaard S, Weerts S, Domitiila C. National biodiversity assessment 2018: technical report. 2019. http://hdl.handle.net/20.500.12143/6373 http://hdl.handle.net/20.500.12143/6373 Volume 3: Estuarine Realm. 3.
- Vandenberg et al. (2017).Vandenberg LN, Blumberg B, Antoniou MN, Benbrook CM, Carroll L, Colborn T, Everett LG, Hansen M, Landrigan PJ, Lanphear BP, Mesnage R, vom Saal FS, Welshons WV, Myers JP. Is it time to reassess current safety standards for glyphosate-based herbicides? Journal of Epidemiology and Community Health. 2017;71(6):613–618. doi: 10.1136/jech-2016-208463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Verweij et al. (2008).Verweij MC, Nagelkerken I, Hans I, Ruseler SM, Mason PRD. Seagrass nurseries contribute to coral reef fish populations. Limnology and Oceanography. 2008;53(4):1540–1547. doi: 10.4319/lo.2008.53.4.1540. [DOI] [Google Scholar]
- Wang et al. (2016a).Wang S, Liu B, Yuan D, Ma J. A simple method for the determination of glyphosate and aminomethylphosphonic acid in seawater matrix with high performance liquid chromatography and fluorescence detection. Talanta. 2016a;161:700–706. doi: 10.1016/j.talanta.2016.09.023. [DOI] [PubMed] [Google Scholar]
- Wang et al. (2016b).Wang S, Seiwert B, Kästner M, Miltner A, Schäffer A, Reemtsma T, Yang Q, Nowak KM. (Bio)degradation of glyphosate in water-sediment microcosms—a stable isotope co-labeling approach. Water Research. 2016b;99:91–100. doi: 10.1016/j.watres.2016.04.041. [DOI] [PubMed] [Google Scholar]
- Waycott et al. (2009).Waycott M, Duarte CM, Carruthers TJB, Orth RJ, Dennison WC, Olyarnik S, Calladine A, Fourqurean JW, Heck KL, Jr, Hughes AR, Kendrick GA, Kenworthy WJ, Short FT, Williams SL. Accelerating loss of seagrasses across the globe threatens coastal ecosystems. Proceedings of the National Academy of Sciences of the United States of America. 2009;106(30):12377–12381. doi: 10.1073/pnas.0905620106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wilkinson et al. (2017).Wilkinson AD, Collier CJ, Flores F, Langlois L, Ralph PJ, Negri AP. Combined effects of temperature and the herbicide diuron on Photosystem II activity of the tropical seagrass Halophila ovalis. Scientific Reports. 2017;7(1):1–11. doi: 10.1038/srep45404. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wilkinson et al. (2015).Wilkinson AD, Collier CJ, Flores F, Negri AP. Acute and additive toxicity of ten photosystem-II herbicides to seagrass. Scientific Reports. 2015;5(1):1–11. doi: 10.1038/srep17443. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Supplementary Materials for van Wyk et al._impacts of glyphosate on seagrass conservation
Data Availability Statement
The following information was supplied regarding data availability:
The data is available at GitHub: www.github.com/vonderheydenlab/van-Wyk-et-al_seagrass-and-glyhosate_raw-data; Van Wyk, JW, Von der Heyden, S, & Adams, J. (2022). Conservation implications of herbicides on seagrasses: sublethal glyphosate exposure decreases fitness in the endangered Zostera capensis [Data set]. Zenodo. https://doi.org/10.5281/zenodo.7156564.