Highlights
-
•
SDS-PAGE analysis of purified IgM of Asian seabass revealed three heavy chain bands of molecular weights 96, 82 and 76 kDa and a single light chain of 25 kDa.
-
•
Monoclonal antibodies raised against IgM of Asian seabass reacted with the 76 kDa heavy chain .
-
•
An Indirect ELISA was developed to detect and quantify specific immune response in Asian seabass.
-
•
The assay could be used to screen Asian seabass for VNN and to assess the immune response to VNN vaccine.
-
•
Immune response in Asian seabass immunized with inactivated RGNNV peaked at 4 weeks post-immunization.
Keywords: Viral nervous necrosis, Nervous necrosis virus, RGNNV, Non-lethal screening, Indirect ELISA
Abstract
Viral nervous necrosis (VNN) is a serious viral disease of several species of farmed and wild fishes. Adult fish are asymptomatic and become carriers of the virus when infected with nervous necrosis virus (NNV) and they transmit the virus to the offspring through eggs. ELISA is ideal for non-lethal screening of adult fish for VNN. Asian seabass (Lates calcarifer) IgM was purified using Protein A affinity column and hybridoma clones secreting monoclonal antibodies (MAb) specific to the heavy chain of IgM was developed. An Indirect ELISA using anti-seabass IgM MAb was developed by optimizing all the reagents. The assay was used to screen adult Asian seabass from grow-out farms in comparison to RT-PCR. The assay was also used to assess the immune response in Asian seabass immunized with inactivated Red-spotted grouper NNV (RGNNV). Seabass IgM on SDS-PAGE analysis revealed three heavy chain bands of size 96, 82 and 76 kDa and a single light chain of size 25 kDa. Out of 18 positive hybridoma clones, two selected clones reacted specifically with the 76 kDa heavy chain band. Out of 28 serum samples of Asian seabass from grow-out farms 26 were positive for NNV antibodies while 22 were positive by RT-PCR. Fish immunized with inactivated RGNNV showed immune response by one week post-immunization, and the peak immune response was observed four weeks post-immunization. The assay developed can be used for non-lethal screening of adult Asian seabass for VNN and to assess the immune response after vaccination.
1. Introduction
Viral nervous necrosis (VNN) also called as viral encephalopathy and retinopathy is a serious viral disease of fish affecting more than 120 species of farmed and wild fishes [1]. The disease is caused by nervous necrosis virus (NNV) belonging to the genus Betanodavirus under the family Nodaviridae. The virus is transmitted both horizontally and vertically. Disease outbreaks are more common in larval and early juveniles before they become fully immunocompetent [1]. In adult fish the infection is usually asymptomatic and the affected fish becomes a carrier of the virus transmitting it vertically through eggs and ovarian fluids to the offspring [2]. Hence, it is important to use virus free adults for breeding. VNN can be diagnosed by the detection of nucleic acid or antigen in the predilection organs such as brain, eyes and spinal cord. Since non-lethal sampling from these organs is not possible, brooders can be screened by detecting NNV from ovarian biopsy, blood, gills and caudal fin clippings by reverse transcription polymerase chain reaction (RT-PCR) [3,4]. However, detection of the virus was inconsistent due to low viral load in these tissues [3,4]. Immune response to betanodavirus can be detected as early as five days post infection [5] and antibodies can be detected up to one year following natural infection [6]. Hence, antibody detection coupled with antigen detection is the ideal way to screen adult fishes for VNN.
Serum neutralization test (SNT) and enzyme linked immunosorbent assay (ELISA) are ideal methods for antibody detection and quantification of serum antibodies in fish. SNT is laborious and is often used in the absence of anti-species antibodies. ELISA is the test of choice for species for which anti-fish antibodies are available. ELISA is also an important assay for serological survey to assess the prevalence of a disease in a particular geographical location. The assay can also be used to assess the efficacy of vaccines. Non-lethal sampling method, low cost and persistence of antibodies makes this an ideal test especially for valuable broodstock [7].
There are many formats of ELISA reported for antibody detection against NNV in several species [4,5,[8], [9], [10], [11], [12], [13], [14], [15], [16], [17]] (Table 1). In spite of this, antibody detection is not considered as a routine screening method for VNN due to insufficient research on antibody detection methods [18]. Concentration of each analyte such as antigen, test serum dilution, conjugate and substrate used in ELISA needs to be optimized for better reproducibility of the assay [19]. Most of the ELISA reported for antibody detection against NNV did not specify the concentration of antigen or the secondary antibody. ELISA for fish antibodies has the inherent problem of non-specific binding of the antibodies to blocking reagents and antigen [8,20]. Use of anti-species polyclonal antibodies adds to the non-specific binding. Use of monoclonal antibody (MAb) reduces non-specific bindings to a great extent. However, anti-species MAbs are commercially available only for a few species. The commercially available Asian seabass (Lates calcarifer) IgM MAb (Aquatic Diagnostic Ltd, Scotland, UK) at different dilutions did not give optical density (OD) higher than the background OD of the plate [17]. Hence, the study was aimed to develop hybridoma clones secreting MAbs against heavy chain of Asian seabass IgM and optimize all the reagents of ELISA to reduce the background OD. The ELISA developed can be used for non-lethal screening of VNN and to assess the immune response to VNN vaccines in Asian seabass.
Table 1.
ELISA formats reported for detection of anti-NNV antibodies in fish.
| S.No. | Capture antibody and antigen | Blocking agent | Test sample | Anti-species Antibody | Conjugate | Reference |
|---|---|---|---|---|---|---|
| 1* | Rabbit anti-NNV antibody (250 ng well−1) and Virus suspension (200 ng well−1) | skim milk (5%) |
Dicentrarchus labrax serum (1:400–1:800) |
Anti-seabass IgM biotinylated MAb (1:2000) | Avidin HRP conjugate (1:2500) |
[10] |
| 2 | Recombinant BFNNV coat protein (500 ng well−1) | skim milk (2%) | Barfin flounder serum (1:10–1:2560) |
Rabbit anti-barfin founder serum | Anti-rabbit HRP conjugate | [11] |
| 3 | Recombinant NNV (100 ng well−1) | Milk (5%) | Seabass (Lates calacrifer) plasma (1:25) | Rabbit anti-seabass serum (1:250) | Anti-rabbit HRP conjugate | [12] |
| 4 | NNV infected cell lysate (total protein content 7.6 µg ml–1) | Skim milk (5%) | Atlantic halibut plasma in PBS |
Anti-Atlantic halibut IgM antiserum (1:500) |
Anti-rabbit HRP conjugate (1:1000) | [13] |
| 5 | Recombinant NNV coat protein (10,000 ng well−1) |
Skim milk (3%) | Grouper (Epinephelus coioides) extract | Rabbit anti-grouper serum (1:1000) | Goat anti-rabbit ALP conjugate (1:1000) | [14] |
| 6 | Inactivated NNV diluted in coating buffer (1:2) | Bovine serum albumin (3%) |
D. labrax serum (1:100–1:6400) |
Anti-seabass IgM HRP conjugate | [15] | |
| 7 | NNV suspension diluted in distilled water or L15 medium (1:10–1:10,240) | skim milk (5%) | Seven banded grouper serum (1:10 or 1:320) |
Rabbit anti-sevenband grouper Ig (1:500) | HRP-conjugated anti-rabbit IgG (1:500) | [16] |
| 8* | Purified sheep anti-NNV antibodies (1:2000) and semi-purified NNV diluted in PBS (1:300) |
Skim milk (1%) | L. calcarifer or Macquaria novemaculeata sera (1:150) | Rabbit anti-Australian bass antiserum (1:1500) |
Donkey anti- rabbit Ig-HRP (1:1500) |
[17] |
| 9# | Purified NNV in deionized water (1:10) or Cultured NNV suspension diluted in L15 medium (1:300) | Skim milk (5%) | Sevenband grouper sera (1:40) | Anti-sevenband grouper IgM (1:500) | Swine anti-rabbit HRP conjugate (1:1000) | [8] |
| 10# | Purified NNV diluted in PBS (180 ng well−1) | Foetal bovine serum (50%) | European seabass (D. labrax) serum (1:200) | Anti-D. labrax IgM MAb (1:500) | Goat anti-mouse IgG HRP (1:1000) | [5] |
| 11 | NNV suspension (106 TCID50 well−1) | Skim milk (3%) | European seabass (D. labrax) serum (1:36–1:2048) | Anti-D. labrax IgM MAb (1:150) | Goat anti-mouse IgG HRP | [4] |
Purified anti-NNV antibodies were immobilized on wells to capture NNV particles which in turn capture anti-NNV IgM.
2. Materials and methods
2.1. Virus propagation, inactivation and production of recombinant capsid protein
Red-spotted grouper nervous necrosis virus (RGNNV) (RGNNVLCInd2016) isolated in this laboratory, was propagated in SSN-1 cells (RRID: CVCL_4306). The 50% tissue culture infective dose (TCID50 ml−1) was calculated using SSN-1 cells in 96 well tissue culture plate [21]. The virus was inactivated using 3 mM binary ethylenimine (BEI) by constant slow stirring at 25 °C for 48 h [22]. Complete inactivation of the virus was confirmed by inoculating the inactivated virus onto SSN-1 cells and observing no cytopathic effect even after 3 passages. The capsid protein gene of RGNNV was RT-PCR amplified, cloned into pET-28b(+) vector and the recombinant protein was expressed in Escherichia coli BL21 (DE3) cells as described earlier [23]. The purified recombinant capsid protein was used to coat ELISA plates.
2.2. Purification and characterization of Asian seabass IgM
Three adult Asian seabass (weighing 3.4 ± 0.45 Kg) maintained at fish hatchery of Muttukadu experimental station of ICAR-Central Institute of Brackishwater Aquaculture, Chennai, India was used for blood collection. All invasive procedures involving fish were carried out with the approval of the institute animal ethics committee of Central Institute of Brackishwater Aquaculture, Chennai (Approval number: CIBA/IAEC/2019-02). The fish were anaesthetized by adding 2-phenoxyethanol at a dose of 200 µl l−1 of seawater to the holding tank and blood was collected from the gill arches. The blood was allowed to clot and the serum was separated. The serum was clarified at 5000 × g for 10 min and the supernatant was used to purify IgM using HiTrapTM protein A HP column (GE Healthcare, Sweden) following the manufacturer's protocol. The purified IgM was quantified using Pierce™ BCA Protein Assay Kit (Thermo Fisher Scientific, USA). The molecular weight of the heavy chain and light chain of IgM was estimated by SDS-PAGE analysis [24]. Briefly, the IgM was boiled in 4X sample loading buffer (VWR Life sciences, India) for 5 min and electrophoresed on a polyacrylamide gel consisting of 5% stacking gel and 12% separating gel. The gel was stained with Coomassie brilliant blue R-250 and destained. The gel was observed under Molecular imager® Gel Doc™ XR+ system (Bio-Rad, India) and the molecular weight of the heavy chain and light chain was estimated using Image Lab Software 5.1 (Bio-Rad, India).
2.3. Development and characterization of MAbs against Asian seabass IgM
All animal procedures for hybridoma development in this study were approved by Institutional Animal Ethics Committee (IAEC) of Vellore Institute of Technology, Vellore, India (VIT/IAEC/14/Nov5/29). Four weeks old BALB/c mice (n=2) were immunized subcutaneously with approximately 50 µg of purified Asian seabass IgM per mouse, emulsified in Freund's complete adjuvant (Sigma-Aldrich, USA). Two booster doses emulsified in Freund's incomplete adjuvant (Sigma-Aldrich, USA) were administered similarly at three weeks interval. Blood collected from orbital plexus seven days after the second booster dose was used to determine anti-Asian seabass IgM antibody titer by indirect ELISA [25]. The mouse with highest serum antibody titer was given a final booster dose (100 µg) intraperitoneally. Four days later, the spleen cells were isolated and fused with Sp2/0-Ag14 myeloma cells (ATCC CRL-1581) and plated on 96 well tissue culture plate containing feeder cells in DMEM supplemented with 1X HAT (Thermo Fisher Scientific, USA). Hybridoma clones were screened for secretion of anti-Asian seabass IgM antibodies by indirect ELISA [25] and positive hybridoma cells showing high OD405 values were cloned twice by limiting dilution and then cryopreserved. MAbs were purified from hybridoma culture supernatants using MAbtrap kit (GE Healthcare, Sweden) following the manufacturer's protocol. The isotype of MAb was determined by IsoStrip Mouse Monoclonal Antibody Isotyping Kit (Sigma-Aldrich, USA). The specificity of the MAbs was tested by Western blot against purified Asian seabass IgM. MAbs were also tested against serum from milkfish (Chanos chanos), tilapia (Oreochromis mossambicus), pearlspot (Etroplus suratensis) and Mangrove red snapper (Lutjanus argentimaculatus) by Western blot for possible cross-reaction. Sera of these fish were diluted 1:150 in PBS and electrophoresed on 12% SDS-PAGE gel, transferred to PVDF membrane and blotted against two selected MAbs. MAb (1E12) showing high specific reactivity with Asian seabass IgM heavy chain was selected for optimizing ELISA for anti-NNV antibody detection.
2.4. Collection of samples
Seventy eight serum samples collected from the caudal vein of Asian seabass fingerlings (36±3.6 g) raised in fish hatchery (28±1 °C), free of VNN as tested by RT-PCR, were used as negative serum samples. Positive serum samples were obtained from ten adult Asian seabass (3.4 ± 0.45 kg) immunized intraperitoneally each with one ml of inactivated RGNNV (108 TCID50 ml−1) emulsified in Montanide™ ISA 763 A VG (Seppic, France). Blood samples were collected from the gill arches one month after the immunization. Test serum samples (n=28) from adult Asian seabass (weighing 4.5 ± 0.85 kg) from two grow out farms located at coastal areas near Chennai, were collected from the gill arches for screening for antibodies against NNV. Ovarian biopsy samples were also collected from the corresponding fish using a cut down cannula in RNAlater (Sigma-Aldrich, USA) for RT-PCR for NNV. Test serum samples were also collected from Asian seabass fingerlings immunized with inactivated RGNNV. Asian seabass fingerlings (36±3.6 g; n=30) raised in fish hatchery (28±1 °C) free of VNN as tested by RT-PCR, were immunized intraperitoneally each with 0.1 ml of inactivated RGNNV (108 TCID50 ml−1), emulsified with Montanide™ ISA 763 A VG (Seppic, France). Control fish (n=30) were immunized with adjuvant only. Blood samples (0.1 ml) (n=8) were collected from the immunized and control groups before and every week post immunization up to 8 weeks.
2.5. Indirect ELISA
The ideal concentration of antigen (recombinant NNV capsid protein), secondary antibody (anti-Asian seabass IgM mouse MAb) and conjugate (anti-mouse HRP conjugate) (Genei laboratories Pvt Ltd, India) required for indirect ELISA were determined by checker-board titration. Recombinant capsid protein of RGNNV (5 µg ml−1) suspended in 100 mM carbonate buffer (pH 9.6) was used to coat 96-well polystyrene plates (Nunc MaxiSorp™) at 100 µl well−1 and incubated for 2 h at room temperature (RT). The plates were washed three times with 300 µl well−1 of wash buffer (Phosphate buffered saline containing 0.05% v/v Tween 20, PBST). The wells were blocked with 3% (w/v) skim milk powder in PBS (SM-PBS) overnight at 4 °C. After washing three times with wash buffer, serum samples diluted in PBS were added in two-fold serial dilutions starting from 1:20 in each well in duplicate and incubated at RT for 3 h. The plates were washed three times and 100 µl of purified anti-Asian seabass IgM MAb (5 µg ml−1) in 0.3% SM-PBS were added to each well and incubated at RT for 1 h. After the subsequent wash step, 100 µl of anti-mouse HRP conjugate diluted 1:2000 in 0.3% SM-PBS was added to each well and incubated at RT for 1 h. After the final wash, 100 µl of the chromogenic solution containing ABTS (15 mg ml−1) and 10 µl of 30% (w/w) Hydrogen peroxide in citrate buffer, pH 4.2 was added to each well and incubated at RT for 20 min. The reaction was stopped with 50 µl of 1% SDS solution well−1 and OD was measured at 405 nm in a microplate reader (Tecan, Switzerland). Blank, positive control and negative control were included in all the assay plates. The cut-off value of the indirect ELISA was determined as the average OD405 at 1:40 dilution + 3 standard deviations (SD) of the negative samples. Test samples showing OD405 values above cut-off at 1:40 dilution were considered positive for NNV antibodies.
2.6. RT-PCR
Total RNA was extracted from the ovarian biopsy samples collected in RNAlater using TRIzol reagent (ThermoFisher Scientific, USA) following the manufacturer's protocol. One microgram of RNA was reverse transcribed into cDNA using iScriptTM cDNA synthesis Kit (Bio-Rad, India). Nested RT-PCR for NNV was carried out using gene-specific primers [3] as described earlier [23]. The PCR products were electrophoresed on 1.5% (w/v) agarose gel and stained with ethidium bromide. The gel was analyzed in a gel documentation system (Bio-Rad, India).
2.7. Statistical analysis
ELISA OD405 values were given as the average of the duplicate samples. The OD405 values between the control and immunized groups was analyzed by one way ANOVA and p < 0.05 was considered statistically significant.
3. Results
3.1. Purification and characterization of Asian seabass IgM
Purification of immunoglobulin from adult Asian seabass serum using protein A column yielded 0.9 mg of IgM ml−1 of serum. On SDS-PAGE analysis the immunoglobulin yielded three heavy chain bands of molecular weight of approximately 96, 82 and 76 kDa and a single light chain of molecular weight 25 kDa respectively (Fig. 1).
Fig. 1.
SDS-PAGE of purified Asian seabass IgM. Lane 1: Protein marker; Lane 2: purified Asian seabass IgM.
3.2. Development and characterization of MAbs against Asian seabass IgM
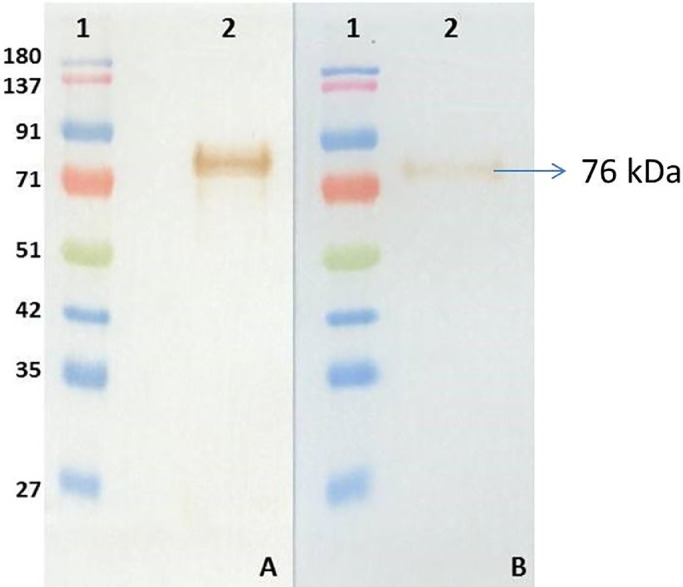
The average indirect ELISA OD405 values of immunized mice were 2.023 and 1.916 at 1:100 dilutions while the control mouse serum OD405 value was 0.069. A total of 18 positive hybridoma clones were obtained upon screening the cell culture supernatant of hybridoma clones by ELISA. Two clones (2B6 and 1E12) which produced high OD405 in indirect ELISA were selected and subcloned twice to ensure monoclonality. Both the clones reacted with the 76 kDa heavy chain of Asian seabass IgM (Fig. 2). The heavy chain isotype of the MAbs were 2B6: IgG1 and 1E12: IgG2b and the light chain isotype of both the MAbs were kappa. The MAbs did not react with IgM of any of the other fish species tested.
Fig. 2.
Western blot of purified Asian seabass IgM with anti-seabass IgM monoclonal antibodies. A- Western blot with MAb 1E12 and B- Western blot with MAb 2B6; Lane 1: Protein marker; Lane 2: MAb blotted against purified Asian seabass IgM.
3.3. Indirect ELISA and RT-PCR
The negative samples gave an average OD405 of 0.132 ± 0.02819 at 1:40 dilution. The cut-off value of the ELISA was determined as average OD405 + 3 SD= 0.217. The positive serum samples obtained from immunized adult produced an OD405 of 1.191 ± 0.143 at 1:40 dilution by indirect ELISA. Twenty six out of 28 test serum samples collected from grow-out farms tested positive for NNV antibodies by indirect ELISA. Out of the 28 samples six samples tested negative and the rest of the samples tested positive for NNV by nested RT-PCR (Fig. 3; Table 2). Test serum samples from Asian seabass fingerlings immunized with inactivated RGNNV showed immune response from one week post-immunization although significant immune response (p < 0.05) could be observed only by two weeks post-immunization. The immune response peaked at four weeks and decreased thereafter. The control serum showed OD405 below the cut-off value throughout the experimental period (Fig. 4).
Fig. 3.
Nested RT-PCR for NNV for Asian seabass ovarian biopsy samples. Lane M: 100 bp DNA marker; Lanes 1–28: Ovarian biopsy samples; Lane 29: Nested RT-PCR positive control (255 bp); Lane 30: Nested RT-PCR Negative control; Lane 31: First step RT-PCR positive control (605 bp).
Table 2.
Comparison of anti-NNV antibody detection by indirect ELISA and NNV detection by RT-PCR.
| Sample | Indirect ELISA (OD) | NNV-specific RT-PCR | NNV-specific nested RT-PCR |
|---|---|---|---|
| 1 | + (0.557 ± 0.021) | − | − |
| 2 | + (0.613 ± 0.018) | − | + |
| 3 | + (0.504 ± 0.028) | − | − |
| 4 | + (0.502 ± 0.012) | − | + |
| 5 | + (0.370 ± 0.025) | + | + |
| 6 | + (0.683 ± 0.011) | + | + |
| 7 | + (0.304 ± 0.013) | + | + |
| 8 | + (0.699 ± 0.016) | − | + |
| 9 | + (0.602 ± 0.019) | − | + |
| 10 | + (0.469 ± 0.011) | + | + |
| 11 | + (0.552 ± 0.014) | − | − |
| 12 | + (0.403 ± 0.010) | − | − |
| 13 | + (0.634 ± 0.013) | + | + |
| 14 | + (0.741 ± 0.018) | − | − |
| 15 | + (0.679 ± 0.015) | + | + |
| 16 | + (0.905 ± 0.021) | − | + |
| 17 | + (0.655 ± 0.019) | − | + |
| 18 | + (0.699 ± 0.018) | − | + |
| 19 | + (0.901 ± 0.028) | − | + |
| 20 | + (0.656 ± 0.034) | − | + |
| 21 | + (0.828 ± 0.020) | + | + |
| 22 | + (0.660 ± 0.025) | + | + |
| 23 | (0.144 ± 0.018) | − | + |
| 24 | + (0.439 ± 0.028) | + | + |
| 25 | + (0.342 ± 0.030) | + | + |
| 26 | + (0.556 ± 0.025) | − | + |
| 27 | + (0.307 ± 0.027) | − | + |
| 28 | (0.142 ± 0.012) | − | − |
+ = Positive; − = Negative; Average indirect ELISA OD405 ± SD values of Asian seabass serum at 1:40 dilution are given in bracket. OD values above 0.217 (Average negative sample OD + 3 SD) were considered as ELISA positive.
Fig. 4.
Anti-NNV serum antibody levels (OD405 at 1:40 dilution of serum) in control and immunized Asian seabass fingerlings (n=8) at different time points. The bar represents mean OD values of 8 samples in duplicate. Error bar represents standard deviation for the mean of 8 samples.
4. Discussion
IgM is the predominant immunoglobulin isotype present in serum of teleosts and is expressed as a tetramer [26]. Each monomer consists of two heavy chains and two light chains and the size of the heavy and light chains vary with species. The size of heavy chain and light chain of Asian seabass was previously reported as 86 and 24 kDa by Crosbie and Nowak [27] and as 83 and 27 kDa by Choudhury and Pani Prasad [28] respectively. However, in the present study, three types of heavy chain measuring approximately 96, 82 and 76 kDa were observed in the IgM purified using protein A affinity column. The SDS-PAGE image of the purified IgM obtained was similar to that reported earlier for Asian seabass by Crosbie and Nowak [27]. The MAbs developed against purified IgM reacted with the 76 kDa band only. The band was confirmed as IgM heavy chain by LC-MS/MS analysis . Although reports of more than one IgM isotype in teleosts are rare, two types of IgM differing in net charge which can be separated by anion exchange chromatography was reported in Atlantic salmon (Salmo salar) [29], [30], [31], [32]. The other two bands (96 and 82 kDa) obtained in the present study needs to be characterized further to ascertain whether more than one isotype of IgM exists in Asian seabass. IgM concentration in serum of teleost fish is reported to be between 800 and 9000 µg ml−1 [33]. The serum IgM concentration of salmon, halibut, haddock and cod were ≤ 1, 4, 7 and 11.5 mg ml−1 of serum of adult fish [34]. The serum IgM concentration of adult Asian seabass measured was within this range (0.9 mg ml−1). The serum IgM concentration depends on the age, size, sex, season, and environment in addition to the infection and vaccination status [31]. The serum collected for IgM purification was from naive fish with no history of vaccination.
Development of MAbs against IgM of several fish species has been reported and MAbs against a few species are available commercially. Although the MAb against Asian seabass is available commercially, it did not work in ELISA format for quantifying the serum antibody levels in Asian seabass [17]. We had a similar experience when using the commercially available Asian seabass IgM MAb. In the present work hybridoma clones secreting MAbs against heavy chain of IgM of Asian seabass were developed which could be used to quantify specific antibodies in serum.
Coating of ELISA plate with recombinant capsid protein of NNV is a better option since the use of NNV infected cell culture supernatant will inhibit binding of NNV to the substrate [16] and purification of NNV from cell culture supernatants is laborious [11]. Use of carbonate buffer (pH 9.6) to coat NNV antigen to plates was reported to lower the OD values significantly due to the possible denaturation of the surface projection of NNV [35]. However, in our study, there was no reduction in the OD405 values when carbonate buffer (pH 9.6) was used instead of PBS to coat recombinant coat protein of NNV. Although different antigen concentration and dilutions were reported for coating ELISA plate, checker board titrations revealed that 500 ng well−1 to be optimum in terms of sensitivity and reagent economy. Blocking the plate with 3% skim milk overnight resulted in low background OD405 compared to 1 h blocking at RT. Further, 1:40 dilution of test serum was found optimum as it provided a good balance between sensitivity and low background OD. The duration and temperature of each incubation step was also optimized by incubating the plates at different temperature and duration. Earlier reports on ELISA for anti-NNV antibody detection used different dilutions of secondary antibody. Unless the secondary antibody is commercially available, it is always better to quantify the concentration of the antibody used for better reproducibility and to avoid batch to batch variations. In our study, a concentration of 500 ng well−1 of anti-Asian seabass IgM MAb was found to be optimum. Use of MAbs as secondary antibodies reduces non-specific binding of secondary antibodies with other analytes. Further, dilution of secondary antibodies and conjugate in PBS containing 0.3% skim milk reduced non-specific binding and reduced background OD. The study describes an indirect ELISA with all the reagent concentrations and steps optimized for low background OD and better sensitivity.
The assay can be used to screen adult Asian seabass for the presence of antibodies against NNV. The assay correlated with virus detection by nested RT-PCR. A good correlation between ELISA and PCR was reported earlier [17]. All the fish except two tested positive for NNV antibodies while six samples tested negative by nested RT-PCR indicating that PCR alone would be insufficient to screen fish for NNV probably due to low viral load in the sample. Brain, retina and spinal cord being the predilection organs [36,37], samples from these organs are ideal for screening. Since non-lethal sample collection from these organs is not possible, samples like ovarian biopsy, blood, gills and caudal fin clips are usually used for screening [4]. However, these samples are not ideal for screening since the viral load in these organs are very low [3,4] and hence it can be coupled with antibody screening to assess the status of exposure of fish to NNV.
The assay can also be used to quantify the immune response of Asian seabass to vaccination against NNV. The antibody response can be detected as early as one week post immunization and reached a peak by four weeks post immunization. Similar results were also reported in European seabass (Dicentrarchus labrax) [5] where the immune response to betanodavirus was detected as early as five days post-infection and a peak immune response was reported in Australian bass (Macquaria novemaculeata) [17] at four weeks post-immunization.
Declaration of Competing Interest
None.
Acknowledgment
This work was carried out under the consortium research platform on vaccines and diagnostics funded by the Indian Council of Agricultural Research.
Footnotes
Supplementary material associated with this article can be found, in the online version, at doi:10.1016/j.fsirep.2021.100011.
Appendix. Supplementary materials
References
- 1.Costa J.Z., Thompson K.D. Understanding the interaction between Betanodavirus and its host for the development of prophylactic measures for viral encephalopathy and retinopathy. Fish Shellfish Immunol. 2016;53:35–49. doi: 10.1016/j.fsi.2016.03.033. [DOI] [PubMed] [Google Scholar]
- 2.Breuil G., Pépin J.F.P., Boscher S., Thiéry R. Experimental vertical transmission of nodavirus from broodfish to eggs and larvae of the sea bass, Dicentrarchus labrax (L.) J. Fish Dis. 2002;25 doi: 10.1046/j.1365-2761.2002.00406.x. [DOI] [Google Scholar]
- 3.Dalla Valle L., Zanella L., Patarnello P., Paolucci L., Belvedere P., Colombo L. Development of a sensitive diagnostic assay for fish nervous necrosis virus based on RT-PCR plus nested PCR. J. Fish Dis. 2000;23:321–327. doi: 10.1046/j.1365-2761.2000.00255.x. [DOI] [Google Scholar]
- 4.Ferreira I.A., Costa J.Z., Macchia V., Thompson K.Dawn, Baptista T. Detection of Betanodavirus in experimentally infected European seabass (Dicentrarchus labrax, Linnaeus 1758) using non-lethal sampling methods. J. Fish Dis. 2019;42:1097–1105. doi: 10.1111/jfd.13015. [DOI] [PubMed] [Google Scholar]
- 5.Tarrab K., Ravid-Peretz S., Ucko M. Immunoserology of European seabass (Dicentrarchus labrax) and white grouper (Epinephelus aeneus) as a non-lethal diagnostic tool for viral nervous necrosis. Aquac. Int. 2019;27:63–77. doi: 10.1007/s10499-018-0307-6. [DOI] [Google Scholar]
- 6.Johansen R., Grove S., Svendsen A.K., Modahl I., Dannevig B. A sequential study of pathological findings in Atlantic halibut, Hippoglossus hippoglossus (L.), throughout one year after an acute outbreak of viral encephalopathy and retinopathy. J. Fish Dis. 2004;27 doi: 10.1111/j.1365-2761.2004.00548.x. [DOI] [PubMed] [Google Scholar]
- 7.Jaramillo D., Peeler E.J., Laurin E., Gardner I.A., Whittington R.J. Serology in finfish for diagnosis, surveillance, and research: a systematic review. J. Aquat. Anim. Health. 2017;29 doi: 10.1080/08997659.2016.1244577. [DOI] [PubMed] [Google Scholar]
- 8.Gye H.J., Nishizawa T. Reducing background optical density in enzyme-linked immunosorbent assay for detecting nervous necrosis virus (NNV)-specific IgM by immobilizing fish sera. Aquaculture. 2018;485:93–100. doi: 10.1016/j.aquaculture.2017.11.039. https://doi.org/https://doi.org/ [DOI] [Google Scholar]
- 9.S. Vimal, N. Madan, M.A. Farook, K.S.N. Nambi, S.A. Majeed, T. Rajkumar, S. Venu, A.R. Thirunavukkarasu, A.S.S. Hameed, Production of recombinant vaccine using capsid gene of nodavirus to protect Asian sea bass, Lates calcarifer (Bloch, 1790), Aquaculture. 418–419 (2014) 148–154. https://doi.org/https://doi.org/ 10.1016/j.aquaculture.2013.10.017.
- 10.Breuil G., Romestand B. A rapid ELISA method for detecting specific antibody level against nodavirus in the serum of the sea bass, Dicentrarchus labrax (L.): application to the screening of spawners in a sea bass hatchery. J. Fish Dis. 1999;22:45–52. doi: 10.1046/j.1365-2761.1999.00136.x. [DOI] [Google Scholar]
- 11.Watanabe K.I., Nishizawa T., Yoshimizu M. Selection of brood stock candidates of barfin flounder using an ELISA system with recombinant protein of barfin flounder nervous necrosis virus. Dis. Aquat. Organ. 2000;41 doi: 10.3354/dao041219. [DOI] [PubMed] [Google Scholar]
- 12.Huang Tan, Chang Munday, Mathew Ngoh, Kwang Detection of nodavirus in barramundi, Lates calcarifer (Bloch), using recombinant coat protein-based ELISA and RT-PCR. J. Fish Dis. 2001;24:135–141. doi: 10.1046/j.1365-2761.2001.00270.x. [DOI] [Google Scholar]
- 13.Grove S., Johansen R., Dannevig B.H., Reitan L.J., Ranheim T. Experimental infection of Atlantic halibut Hippoglossus hippoglossus with nodavirus: tissue distribution and immune response. Dis. Aquat. Organ. 2003;53 doi: 10.3354/dao053211. [DOI] [PubMed] [Google Scholar]
- 14.Lin C.C., Lin J.H.Y., Chen M.S., Yang H.L. An oral nervous necrosis virus vaccine that induces protective immunity in larvae of grouper (Epinephelus coioides) Aquaculture. 2007;268 doi: 10.1016/j.aquaculture.2007.04.066. [DOI] [Google Scholar]
- 15.Scapigliati G., Buonocore F., Randelli E., Casani D., Meloni S., Zarletti G., Tiberi M., Pietretti D., Boschi I., Manchado M., Martin-Antonio B., Jimenez-Cantizano R., Bovo G., Borghesan F., Lorenzen N., Einer-Jensen K., Adams S., Thompson K., Alonso C., Bejar J., Cano I., Borrego J.J., Alvarez M.C. Cellular and molecular immune responses of the sea bass (Dicentrarchus labrax) experimentally infected with betanodavirus. Fish Shellfish Immunol. 2010;28:303–311. doi: 10.1016/j.fsi.2009.11.008. [DOI] [PubMed] [Google Scholar]
- 16.Choi B., Gye H.J., Oh M.J., Nishizawa T. Cell culture medium inhibits antigen binding used in an ELISA for detection of antibodies against nervous necrosis virus. J. Aquat. Anim. Health. 2014;26 doi: 10.1080/08997659.2014.922516. [DOI] [PubMed] [Google Scholar]
- 17.Jaramillo D., Hick P., Deece K., Tweedie A., Kirkland P., Arzey E., Whittington R.J. Comparison of ELISA formats for detection of antibodies specific for nervous necrosis virus (Betanodavirus) in the serum of immunized barramundi Lates calcarifer and Australian bass Macquaria novemaculeata. Aquaculture. 2016;451 doi: 10.1016/j.aquaculture.2015.08.015. [DOI] [Google Scholar]
- 18.World Organisation for Animal Health [OIE] Chapter 2.3.12. Viral encephalopathy and retinopathy. Proceedings of the Manual on Diagnostic Tests for Aquatic Animals; OIE Paris, France; 2019. https://doi.org/ [Google Scholar]
- 19.World Organisation for Animal Health [OIE] Chapter 2.2.1. Development and optimisation of antibody detection assays. Proceedings of the OIE Terrestrial Manual 2018. OIE; Paris, France; 2018. pp. 172–184. https://doi.org/ [Google Scholar]
- 20.Kim W.S., Nishizawa T., Yoshimizu M. Non-specific adsorption of fish immunoglobulin M (IgM) to blocking reagents on ELISA plate wells. Dis. Aquat. Org. 2007;78:55–59. doi: 10.3354/dao01843. [DOI] [PubMed] [Google Scholar]
- 21.Reed L.J., Muench H. A simple method of estimating fifty per cent endpoints. Am. J. Epidemiol. 1938;27:493–497. doi: 10.1093/oxfordjournals.aje.a118408. [DOI] [Google Scholar]
- 22.Kai Y.-H., Chi S.-C. Efficacies of inactivated vaccines against betanodavirus in grouper larvae (Epinephelus coioides) by bath immunization. Vaccine. 2008;26:1450–1457. doi: 10.1016/j.vaccine.2007.12.043. [DOI] [PubMed] [Google Scholar]
- 23.Marappan M., Rengarajan A., Nallala V.S., Sukumaran K., Bera A., Sivaramakrishnan T., Thiagarajan G., Kailasam M., Kizhakedath V.Koyadan. Resistance of pearlspot larvae, Etroplus suratensis, to redspotted grouper nervous necrosis virus by immersion challenge. J. Fish Dis. 2019;42:249–256. doi: 10.1111/jfd.12930. [DOI] [PubMed] [Google Scholar]
- 24.Laemmli U.K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970;227 doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- 25.Qiu S., Zheng L., Qin C., Yin X., Ma J., Liu J., Yang Z., Li C., Wang Y., Wang M., Qi Y., Huo Y. Production and characterization of monoclonal antibodies against GII.6 norovirus virus-like particles. Microb. Pathog. 2020;142 doi: 10.1016/j.micpath.2020.104100. [DOI] [PubMed] [Google Scholar]
- 26.Bengtén E., Clem L.W., Miller N.W., Warr G.W., Wilson M. Channel catfish immunoglobulins: repertoire and expression. Dev. Comp. Immunol. 2006;30 doi: 10.1016/j.dci.2005.06.016. [DOI] [PubMed] [Google Scholar]
- 27.Crosbie P.B.B., Nowak B.F. Production of polyclonal antisera against barramundi (Lates calcarifer Bloch) serum immunoglobulin derived from affinity columns containing mannan-binding protein or staphylococcal protein A. Aquaculture. 2002;211:49–63. [Google Scholar]
- 28.Choudhury M., Pani Prasad K. Isolation and characterization of immunoglobulin M of Asian sea bass, Lates calcarifer and its level in serum. Open Life Sci. 2011;6:1064. doi: 10.2478/s11535-011-0092-y. https://doi.org/doi. [DOI] [Google Scholar]
- 29.Håvarstein L.S., Aasjord P.M., Ness S., Endresen C. Purification and partial characterization of an IgM-like serum immunoglobulin from atlantic salmon (Salmo salar) Dev. Comp. Immunol. 1988;12 doi: 10.1016/0145-305X(88)90052-3. [DOI] [PubMed] [Google Scholar]
- 30.Kamil A., Raae A., Fjelldal P.G., Koppang E.O., Fladmark K.E., Hordvik I. Comparative analysis of IgM sub-variants in salmonid fish and identification of a residue in μ3 which is essential for MAb4C10 reactivity. Fish Shellfish Immunol. 2013;34 doi: 10.1016/j.fsi.2012.12.006. [DOI] [PubMed] [Google Scholar]
- 31.Hordvik I. Immunoglobulin isotypes in atlantic salmon, salmo salar. Biomolecules. 2015;5 doi: 10.3390/biom5010166. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Kamil A., Falk K., Sharma A., Raae A., Berven F., Koppang E.O., Hordvik I. A monoclonal antibody distinguishes between two IgM heavy chain isotypes in Atlantic salmon and brown trout: protein characterization, 3D modeling and epitope mapping. Mol. Immunol. 2011;48:1859–1867. doi: 10.1016/j.molimm.2011.05.005. [DOI] [PubMed] [Google Scholar]
- 33.Ye J., Kaattari I.M., Ma C., Kaattari S. The teleost humoral immune response. Fish Shellfish Immunol. 2013;35 doi: 10.1016/j.fsi.2013.10.015. [DOI] [PubMed] [Google Scholar]
- 34.Magnadottir B. Comparison of immunoglobulin (IgM) from four fish species. Icelandic Agric. Sci. 1998:49–54. [Google Scholar]
- 35.Gye H.J., Nishizawa T. Reproducibility of antigen-immobilized enzyme-linked immunosorbent assay (ELISA) and sandwich ELISA for quantitative detection of NNV particles. J. Virol. Methods. 2020;275 doi: 10.1016/j.jviromet.2019.113754. [DOI] [PubMed] [Google Scholar]
- 36.Skliris G.P., Richards R.H. Virus Res. 1999. Induction of nodavirus disease in seabass, Dicentrarchus labrax, using different infection models. [DOI] [PubMed] [Google Scholar]
- 37.Tanaka S., Takagi M., Miyazaki T. Histopathological studies on viral nervous necrosis of sevenband grouper, Epinephelus septemfasciatus Thunberg, at the grow-out stage. J. Fish Dis. 2004;27 doi: 10.1111/j.1365-2761.2004.00559.x. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.