Abstract
Diabetes mellitus is one of the most prevalent metabolic diseases worldwide, causing an enormous burden on the economies of both developed and developing nations. Nonalcoholic fatty liver disease (NAFLD) is very closely associated with diabetes, and the two diseases are known to cause an increase in morbidity and mortality. Timely referral of a diabetic with NAFLD to a hepatologist can definitely delay disease progression and the related complications. Despite the magnitude, there are no guidelines that recommend a screening algorithm that must be followed for evaluating NAFLD in at-risk diabetics at the primary care level and their further referral to hepatologists. Nonalcoholic fatty liver disease management needs heightened awareness among primary care physicians/endocrinologists and hepatologists, and a collaborative care approach is paramount in these patients. Certain antidiabetic drugs are found to be beneficial in the treatment of NAFLD patients with diabetes, however, none of them are FDA approved.
How to cite this article
Khandelwal R, Dassanayake AS, Singh SP. Nonalcoholic Fatty Liver Disease in Diabetics: The Role of Hepatologist. Euroasian J Hepato-Gastroenterol 2022;12(Suppl 1):S37–S40.
Keywords: Antidiabetic drugs, Diabetes, Fibrosis, Hepatologist, Nonalcoholic fatty liver disease, Nonalcoholic steatohepatitis
Introduction
Nonalcoholic fatty liver disease and diabetes are well-known to be intricately connected. The relationship first came to the fore in 1884, when Pepper explained fatty infiltration in the liver of a patient with diabetes.1 Later, in 1938, Connor described fatty liver in diabetics and considered it as a possible cause of the development of cirrhosis in these patients.2 The overall prevalence of chronic metabolic diseases is rising worldwide, mainly due to an increase in the occurrence of Type 2 diabetes mellitus (T2DM) and obesity. Currently, the global prevalence of T2DM is about 8.8%, indicating that approximately 422 million people are affected by it throughout the world.3 There are various studies that show that among diabetics, 70−80% have NAFLD.4,5 Diabetes and obesity have also been found as important predictors of inflammatory progression in NAFLD patients, leading to nonalcoholic steatohepatitis (NASH) and severe fibrosis.6−8 Because diabetics with NAFLD are more likely to develop more severe and deadly forms of liver disease, there exists a need for early detection and timely institution of corrective measures. This can only be achieved if the treating physician or the treating endocrinologist screens diabetics for NAFLD similar to the way screening is done in these patients for other diabetes-related microvascular and macrovascular complications. This article is an attempt to highlight the close relationship that these two disease entities share, the current societal guidelines for their evaluation, and the available treatment options.
Relationship between NAFLD and Diabetes
Nonalcoholic fatty liver disease may be considered as metabolic syndrome's “hepatic manifestation”9 with lipid accumulation in the liver being the hallmark. The link between T2DM and NAFLD encompasses a spectrum of metabolic changes, which include insulin resistance (IR), defective triglyceride (TG) metabolism, and hepatic lipidic profile, which further contribute to fat accumulation, immune dysregulation, and/or hyperinsulinemia as determined by the β-cell dysfunction in T2DM.10 Insulin resistance, one of the key attributes of both NAFLD and T2DM, cannot be classified as either the cause or the consequence specific to any one of the two diseases.10 Genetic and environmental factors interfering with the insulin signaling cascade also add to the maintenance and progressive worsening of IR.11 Recently, the existence of a liver−pancreas axis, connecting the liver and pancreatic alpha cells, has also been suggested as a factor in pathogenesis.12 Epidemiological evidence supports the notion that NAFLD and T2DM have a bidirectional relationship, and that NAFLD may precede and/or accelerate the development of T2DM.13 Interestingly, it has been found that while NAFLD patients have a twofold increased risk of developing T2DM and metabolic syndrome,14 T2DM may also accelerate the course of NAFLD and has been proposed as a strong clinical predictor of progression of NAFLD to NASH and cirrhosis.15 Histological studies of the liver have shown that while up to 80% of patients with T2DM exhibited NASH, 30−40% may have advanced fibrosis.16 The estimated prevalence of NASH and advanced fibrosis in patients with coexisting T2DM and NAFLD is about 37.3% (95% CI 24.7–50.0) and 17.0% (95% CI 7.2–34.8), respectively, according to a recent systematic review and meta-analysis.17 Furthermore, the overall mortality in the study was found to be around 585 per 100,000 over a period of 5–10 years, which was significantly greater than mortality from other chronic liver illnesses related to different etiologies.17 Type 2 diabetes mellitus and hepatocellular carcinoma (HCC) have a strong pathophysiological relationship; T2DM alone can increase the probability of developing HCC by two to threefold.18 Diabetics have hyperinsulinemia and higher levels of inflammatory biomarkers, which have been linked to an increased risk of developing HCC.19 Concomitant NAFLD in diabetics makes management of diabetes difficult as there is inadequate control of blood glucose levels in these patients.20 Furthermore, NAFLD may enhance the risk of cardiovascular events in diabetics by up to 1.87-folds.21 Co-existent NAFLD also contributes to an increased risk of microvascular complications of diabetes such as retinopathy and chronic kidney disease.22 In addition, besides the effects on the liver, diabetics with NAFLD may also have sensory-motor and autonomic neuropathy.23,24 Therefore, T2DM and NAFLD can be considered as inextricably linked and this has been summarized in Figure 1.
Fig. 1.
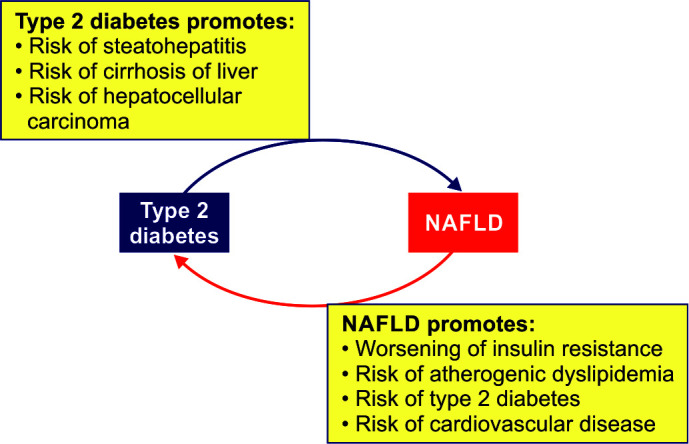
Type 2 diabetes and NAFLD: an intricate affair
Evaluation
As there is mounting evidence that diabetics are at a heightened risk of developing NASH and cirrhosis,25,26 the role of endocrinologists and primary care physicians gain center stage in the prevention and early detection of liver disease in such patients. It has also been noted that patients with NAFLD have a twofold augmented risk of developing diabetes, and thus, hepatologists can play a significant role in preventing the development of diabetes in these patients. Earlier, the requirement for screening of NAFLD in the community was often questioned, given the high testing costs involved, the low predictive value of noninvasive tests, and the risks associated with performing the cumbersome liver biopsy, the “gold standard” test for apt diagnosis of NAFLD.27 The earlier guidelines also did not mention any recommendations concerning screening of diabetics for liver fibrosis.
Noninvasive screening tests for steatosis, NASH, and fibrosis are now recommended in patients with obesity/diabetes or those with incidental high liver enzymes along with metabolic risk factors, as per the EASL–EASD–EASO guidelines of 2016.8 They recommend using ultrasonography in conjunction with steatosis biomarkers such as the Fatty Liver Index, SteatoTest, or NAFLD Fat Score for steatosis detection. Surrogate fibrosis markers (NFS, FIB-4, ELF, or FibroTest) need to be computed for ruling out severe fibrosis (PF2 by METAVIR), which, if discovered, would require expert referral for assessment.8
In 2019, the American Diabetes Association's (ADA) Standards of Medical Care in Diabetes External link also advised that individuals with prediabetes or diabetes and having increased liver enzymes or fatty liver on ultrasonography must undergo evaluation for NASH and fibrosis.28
However, debate continues to rage with regard to the threshold values of plasma alanine aminotransferase (ALT) that are to be used. Although a cut-off of 40 IU/L is commonly employed in clinical practice and studies, a few studies have demonstrated that using lower cut-offs for normal (i.e., 30 IU/L for males and 19 IU/L for females) may improve the identification of NAFLD prevalence.29 Noninvasive scoring systems, such as the FIB-4 or NAFLD fibrosis score (NFS), have recently been found as easy and sustainable techniques for identifying patients at high risk of developing advanced fibrosis.30 Higher liver fibrosis scores are known to be associated with greater liver disease and overall mortality.31 When the FIB-4 and NFS models are applied, their negative predictive values (NPV) are around 93.48% and 93.61%, respectively, in patients with liver fibrosis (stages 3 and 4), indicating that these models may help exclude severe liver disease.32 In one study,33 the use of vibration-controlled transient elastography (VCTE) in a high-risk population (hazardous alcohol and/or T2DM) in primary care, led to the diagnosis of cirrhosis in 3% of the population. Interestingly, 60% of these patients either presented with T2DM or were obese.33 In another study by Barbosa et al.,34 an algorithm was proposed involving an initial annual fibrosis-4 score (FIB-4) followed by VCTE for those with either indeterminate or high-risk scores (FIB-4 ≥1.3). As per their study, patients at low risk (FIB-4 <1.3 or VCTE <8 kPa) require follow-up by primary care providers, yearly calculation of FIB-4, and lifestyle changes, while patients at high risk (FIB-4 ≥1.3 and VCTE ≥8 kPa) need hepatology referral.34 These simple noninvasive tools need further validation as they can help in the easy and economical assessment of the risk of developing NAFLD in primary care clinics only. This will not only prevent the under-recognition of cases with significant cirrhosis but will also help in the avoidance of unnecessary referrals to specialists.35,36 Furthermore, primary care physicians also need to be aware of these algorithms, as it has been well demonstrated that many of them underestimate the prevalence of NAFLD and often fail to recognize its clinical spectrum.37
Management
Lifestyle interventions and weight reduction are found to be very advantageous38 and are often considered the cornerstone for both prevention and treatment of NAFLD in diabetics. Some of the antidiabetic drugs have been tried in these patients and have been found to be beneficial for both NAFLD and T2DM, such as pioglitazone, liraglutide, semaglutide, and SGLT-2 inhibitors– empagliflozin and dapagliflozin. However, none of them are FDA-approved. In both diabetic and nondiabetic patients, pioglitazone, a strong and specific agonist for peroxisome proliferator-activated receptor-gamma (PPAR gamma), is found to ameliorate NASH by effectively improving histological characteristics like ballooning or inflammation. However, the effects on liver fibrosis are modest.39 Fears regarding adverse effects of pioglitazone in these patients, such as weight gain, have been allayed by recent studies making it a reasonable treatment choice.40 GLP-1RAs have also been shown to decrease ALT and intrahepatic TG content when used in patients with diabetes in some studies, indicating their potential role in treatment.41,42 Table 1 shows the list of drugs that have been studied in diabetics with NAFLD and their effects.
Table 1.
List of drugs that have been studied in diabetics with NAFLD and their effects
| Drug class (Refs.) | Change in body weight | Change in body fat | Effect on ALT |
|---|---|---|---|
| Biguanides (Metformin)43,44 | Decreased | Decreased | Not known |
| Thiazolidinediones45,46 | Increased | Increased | Decreased |
| GLP-1r agonists47,48 | Decreased | Decreased | Decreased |
| DPP-4 inhibitors49,50 | Unchanged | Unchanged | Decreased/Unchanged |
| SGLT2 inhibitors51,52 | Decreased | Decreased | Decreased |
ALT, alanine transaminase; DPP-4, dipeptidyl peptidase-4 inhibitors; GLP-1r agonists, glucagon-like peptide 1 receptor agonists; SGLT2, sodium/glucose cotransporter-2 inhibitors
Conclusion
Nonalcoholic fatty liver disease is currently being considered the leading etiology of chronic liver disease in both developing and developed countries. The global burden of this disease is bound to increase further in the coming decades which would have obvious clinical, economic, and social implications. Furthermore, there is growing recognition of the fact that certain specific populations, such as patients with T2DM, are at a particularly increased risk of developing NAFLD/NASH. Thus, the formulation of more specific guidelines for such patients is the need of the hour. This would surely help endocrinologists/primary care physicians in the early identification of diabetics who are at risk of developing liver-related complications. A successful strategy would be to incorporate a simple, cost-effective algorithm into the present-day diabetes care system.
Orcid
Reshu Khandelwal https://orcid.org/0000-0003-3541-8119
Anuradha Supun Dassanayake https://orcid.org/0000-0001-5567-3542
Footnotes
Source of support: Nil
Conflict of interest: None
References
- 1.Pepper W. Saccharine diabetes. Med Rec. 1884;25:9–12. [Google Scholar]
- 2.Connor CL. Fatty infiltration of the liver and the development of cirrhosis in diabetes and chronic alcoholism. Am J Pathol. 1938;14(3):347–364. 19970395 [PMC free article] [PubMed] [Google Scholar]
- 3.Saklayen MG. The global epidemic of the metabolic syndrome. Curr Hypertens Rep. 2018;20(2):12. doi: 10.1007/s11906-018-0812-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Targher G, Bertolini L, Padovani R, et al. Prevalence of nonalcoholic fatty liver disease and its association with cardiovascular disease among type 2 diabetic patients. Diabetes Care. 2007;30(5):1212–1218. doi: 10.2337/dc06-2247. [DOI] [PubMed] [Google Scholar]
- 5.Williamson RM, Price JF, Glancy S, et al. Prevalence of and risk factors for hepatic steatosis and nonalcoholic fatty liver disease in people with type 2 diabetes: The Edinburgh Type 2 Diabetes Study. Diabetes Care. 2011;34(5):1139–1144. doi: 10.2337/dc10-2229. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Hamaguchi M, Kojima T, Takeda N, et al. The metabolic syndrome as a predictor of nonalcoholic fatty liver disease. Ann Intern Med. 2005;143(10):722–728. doi: 10.7326/0003-4819-143-10-200511150-00009. [DOI] [PubMed] [Google Scholar]
- 7.Ong JP, Elariny H, Collantes R, et al. Predictors of nonalcoholic steatohepatitis and advanced fibrosis in morbidly obese patients. Obes Surg. 2005;15(3):310–315. doi: 10.1381/0960892053576820. [DOI] [PubMed] [Google Scholar]
- 8.European Association for the Study of the Liver (EASL); European Association for the Study of Diabetes (EASD); European Association for the Study of Obesity (EASO). EASL-EASD-EASO Clinical Practice Guidelines for the management of non-alcoholic fatty liver disease. Diabetologia. 2016;59(6):1121–1140. doi: 10.1007/s00125-016-3902-y. [DOI] [PubMed] [Google Scholar]
- 9.Anstee QM, McPherson S, Day CP. How big a problem is non-alcoholic fatty liver disease? BMJ. 2011;343:d3897. doi: 10.1136/bmj.d3897. [DOI] [PubMed] [Google Scholar]
- 10.Tanase DM, Gosav EM, Costea CF, et al. The intricate relationship between Type 2 Diabetes Mellitus (T2DM), Insulin Resistance (IR), and Nonalcoholic Fatty Liver Disease (NAFLD). J Diabetes Res. 2020;2020:3920196. doi: 10.1155/2020/3920196. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Buzzetti E, Pinzani M, Tsochatzis EA. The multiple-hit pathogenesis of non-alcoholic fatty liver disease (NAFLD). Metabolism. 2016;65(8):1038–1048. doi: 10.1016/j.metabol.2015.12.012. [DOI] [PubMed] [Google Scholar]
- 12.Knop FK. EJE PRIZE 2018: A gut feeling about glucagon. Eur J Endocrinol. 2018;178(6):R267–R280. doi: 10.1530/EJE-18-0197. [DOI] [PubMed] [Google Scholar]
- 13.Targher G, Marchesini G, Byrne CD. Risk of type 2 diabetes in patients with non-alcoholic fatty liver disease: Causal association or epiphenomenon? Diabetes Metab. 2016;42(3):142–156. doi: 10.1016/j.diabet.2016.04.002. [DOI] [PubMed] [Google Scholar]
- 14.Ballestri S, Zona S, Targher G, et al. Nonalcoholic fatty liver disease is associated with an almost twofold increased risk of incident type 2 diabetes and metabolic syndrome. Evidence from a systematic review and meta-analysis. J Gastroenterol Hepatol. 2016;31(5):936–944. doi: 10.1111/jgh.13264. [DOI] [PubMed] [Google Scholar]
- 15.Adams LA, Lymp JF, St. Sauver J, et al. The natural history of nonalcoholic fatty liver disease: A population-based cohort study. Gastroenterology. 2005;129(1):113–121. doi: 10.1053/j.gastro.2005.04.014. [DOI] [PubMed] [Google Scholar]
- 16.Bazick J, Donithan M, Neuschwander-Tetri BA, et al. Clinical model for NASH and advanced fibrosis in adult patients with diabetes and NAFLD: Guidelines for referral in NAFLD. Diabetes Care. 2015;38(7):1347–1355. doi: 10.2337/dc14-1239. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Younossi ZM, Golabi P, de Avila L, et al. The global epidemiology of NAFLD and NASH in patients with type 2 diabetes: A systematic review and meta-analysis. J Hepatol. 2019;71(4):793–801. doi: 10.1016/j.jhep.2019.06.021. [DOI] [PubMed] [Google Scholar]
- 18.El-Serag HB, Tran T, Everhart JE. Diabetes increases the risk of chronic liver disease and hepatocellular carcinoma. Gastroenterology. 2004;126(2):460–468. doi: 10.1053/j.gastro.2003.10.065. [DOI] [PubMed] [Google Scholar]
- 19.Aleksandrova K, Boeing H, Nöthlings U, et al. Inflammatory and metabolic biomarkers and risk of liver and biliary tract cancer. Hepatology. 2014;60(3):858–871. doi: 10.1002/hep.27016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Afolabi BI, Ibitoye BO, Ikem RT, et al. The relationship between glycaemic control and non-alcoholic fatty liver disease in Nigerian type 2 diabetic patients. J Natl Med Assoc. 2018;110(3):256–264. doi: 10.1016/j.jnma.2017.06.001. [DOI] [PubMed] [Google Scholar]
- 21.Targher G, Bertolini L, Rodella S, et al. Nonalcoholic fatty liver disease is independently associated with an increased incidence of cardiovascular events in type 2 diabetic patients. Diabetes Care. 2007;30(8):2119–2121. doi: 10.2337/dc07-0349. [DOI] [PubMed] [Google Scholar]
- 22.Targher G, Bertolini L, Rodella S, et al. Non-alcoholic fatty liver disease is independently associated with an increased prevalence of chronic kidney disease and proliferative/laser-treated retinopathy in type 2 diabetic patients. Diabetologia. 2008;51(3):444–450. doi: 10.1007/s00125-007-0897-4. [DOI] [PubMed] [Google Scholar]
- 23.Williams KH, Burns K, Constantino M, et al. An association of large-fibre peripheral nerve dysfunction with non-invasive measures of liver fibrosis secondary to non-alcoholic fatty liver disease in diabetes. J Diabetes Complications. 2015;29(8):1240–1247. doi: 10.1016/j.jdiacomp.2015.06.015. [DOI] [PubMed] [Google Scholar]
- 24.Ziegler D, Strom A, Kupriyanova Y, et al. Association of lower cardiovagal tone and baroreflex sensitivity with higher liver fat content early in type 2 diabetes. J Clin Endocrinol Metab. 2018;103(3):1130–1138. doi: 10.1210/jc.2017-02294. [DOI] [PubMed] [Google Scholar]
- 25.Younossi ZM, Koenig AB, Abdelatif D, et al. Global epidemiology of nonalcoholic fatty liver disease-Meta-analytic assessment of prevalence, incidence, and outcomes. Hepatology. 2016;64(1):73–84. doi: 10.1002/hep.28431. [DOI] [PubMed] [Google Scholar]
- 26.Non-alcoholic Fatty Liver Disease Study Group, Lonardo A, Bellentani S, et al. Epidemiological modifiers of non-alcoholic fatty liver disease: Focus on high-risk groups. Dig Liver Dis. 2015;47(12):997–1006. doi: 10.1016/j.dld.2015.08.004. [DOI] [PubMed] [Google Scholar]
- 27.Chalasani N, Younossi Z, Lavine JE, et al. The diagnosis and management of non-alcoholic fatty liver disease: Practice Guideline by the American Association for the Study of Liver Diseases, American College of Gastroenterology, and the American Gastroenterological Association. Hepatology. 2012;55(6):2005–2023. doi: 10.1002/hep.25762. [DOI] [PubMed] [Google Scholar]
- 28.American Diabetes Association. 4. Comprehensive Medical Evaluation and Assessment of Comorbidities: Standards of Medical Care in Diabetes-2019. Diabetes Care. 2019;42(Suppl 1):S34–S45. doi: 10.2337/dc19-S004. [DOI] [PubMed] [Google Scholar]
- 29.Prati D, Taioli E, Zanella A, et al. Updated definitions of healthy ranges for serum alanine aminotransferase levels. Ann Intern Med. 2002;137(1):1–10. doi: 10.7326/0003-4819-137-1-200207020-00006. [DOI] [PubMed] [Google Scholar]
- 30.Armstrong MJ, Marchesini G. Referral pathways for NAFLD fibrosis in primary care–No longer a ‘needle in a haystack’. J Hepatol. 2019;71(2):246–248. doi: 10.1016/j.jhep.2019.05.010. [DOI] [PubMed] [Google Scholar]
- 31.Unalp-Arida A, Ruhl CE. Liver fibrosis scores predict liver disease mortality in the United States population. Hepatology. 2017;66(1):84–95. doi: 10.1002/hep.29113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Nones RB, Ivantes CP, Pedroso MLA. Can FIB4 and NAFLD fibrosis scores help endocrinologists refer patients with non-alcoholic fat liver disease to a hepatologist? Arch Endocrinol Metab. 2017;61(3):276–281. doi: 10.1590/2359-3997000000233. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Harman DJ, Ryder SD, James MW, et al. Obesity and type 2 diabetes are important risk factors underlying previously undiagnosed cirrhosis in general practice: A cross-sectional study using transient elastography. Aliment Pharmacol Ther. 2018;47(4):504–515. doi: 10.1111/apt.14463. [DOI] [PubMed] [Google Scholar]
- 34.Vieira Barbosa J, Lai M. Nonalcoholic fatty liver disease screening in type 2 diabetes mellitus patients in the primary care setting. Hepatol Commun. 2020;5(2):158–167. doi: 10.1002/hep4.1618. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Davyduke T, Tandon P, Al-Karaghouli M, et al. Impact of implementing a “FIB-4 First” strategy on a pathway for patients with NAFLD referred from primary care. Hepatol Commun. 2019;3(10):1322–1333. doi: 10.1002/hep4.1411. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Boursier J, Guillaume M, Leroy V, et al. New sequential combinations of non-invasive fibrosis tests provide an accurate diagnosis of advanced fibrosis in NAFLD. J Hepatol. 2019;71(2):389–396. doi: 10.1016/j.jhep.2019.04.020. [DOI] [PubMed] [Google Scholar]
- 37.Patel PJ, Banh X, Horsfall LU, et al. Underappreciation of non-alcoholic fatty liver disease by primary care clinicians: Limited awareness of surrogate markers of fibrosis. Intern Med J. 2018;48(2):144–151. doi: 10.1111/imj.13667. [DOI] [PubMed] [Google Scholar]
- 38.Sasso M, Beaugrand M, de Ledinghen V, et al. Controlled attenuation parameter (CAP): A novel VCTE™ guided ultrasonic attenuation measurement for the evaluation of hepatic steatosis: Preliminary study and validation in a cohort of patients with chronic liver disease from various causes. Ultrasound Med Biol. 2010;36(11):1825–1835. doi: 10.1016/j.ultrasmedbio.2010.07.005. [DOI] [PubMed] [Google Scholar]
- 39.Cusi K. A diabetologist's perspective of non-alcoholic steatohepatitis (NASH): Knowledge gaps and future directions. Liver Int. 2020;40(Suppl 1):82–88. doi: 10.1111/liv.14350. [DOI] [PubMed] [Google Scholar]
- 40.Sasso M, Tengher-Barna I, Ziol M, et al. Novel controlled attenuation parameter for noninvasive assessment of steatosis using Fibroscan(®): Validation in chronic hepatitis C. J Viral Hepat. 2012;19(4):244–253. doi: 10.1111/j.1365-2893.2011.01534.x. [DOI] [PubMed] [Google Scholar]
- 41.de Lédinghen V, Vergniol J, Capdepont M, et al. Controlled attenuation parameter (CAP) for the diagnosis of steatosis: A prospective study of 5323 examinations. J Hepatol. 2014;60(5):1026–1031. doi: 10.1016/j.jhep.2013.12.018. [DOI] [PubMed] [Google Scholar]
- 42.Lee JH, Kim D, Kim HJ, et al. Hepatic steatosis index: A simple screening tool reflecting nonalcoholic fatty liver disease. Dig Liver Dis. 2010;42(7):503–508. doi: 10.1016/j.dld.2009.08.002. [DOI] [PubMed] [Google Scholar]
- 43.Nair S, Diehl AM, Wiseman M, et al. Metformin in the treatment of non-alcoholic steatohepatitis: A pilot open label trial. Aliment Pharmacol Ther. 2004;20(1):23–28. doi: 10.1111/j.1365-2036.2004.02025.x. [DOI] [PubMed] [Google Scholar]
- 44.Haukeland JW, Konopski Z, Eggesbø HB, et al. Metformin in patients with non-alcoholic fatty liver disease: A randomized, controlled trial. Scand J Gastroenterol. 2009;44(7):853–860. doi: 10.1080/00365520902845268. [DOI] [PubMed] [Google Scholar]
- 45.Ratziu V, Giral P, Jacqueminet S, et al. Rosiglitazone for nonalcoholic steatohepatitis: One-year results of the randomized placebo-controlled Fatty Liver Improvement with Rosiglitazone Therapy (FLIRT) trial. Gastroenterology. 2008;135(1):100–110. doi: 10.1053/j.gastro.2008.03.078. [DOI] [PubMed] [Google Scholar]
- 46.Musso G, Cassader M, Paschetta E, et al. Thiazolidinediones and advanced liver fibrosis in nonalcoholic steatohepatitis: A meta-analysis. JAMA Intern Med. 2017;177(5):633–640. doi: 10.1001/jamainternmed.2016.9607. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.García Díaz E, Guagnozzi D, Gutiérrez V, et al. Effect of incretin therapies compared to pioglitazone and gliclazide in non-alcoholic fatty liver disease in diabetic patients not controlled on metformin alone: An observational, pilot study. Endocrinol Nutr. 2016;63(5):194–201. doi: 10.1016/j.endonu.2016.01.006. [DOI] [PubMed] [Google Scholar]
- 48.Shao N, Kuang HY, Hao M, et al. Benefits of exenatide on obesity and non-alcoholic fatty liver disease with elevated liver enzymes in patients with type 2 diabetes. Diabetes Metab Res Rev. 2014;30(6):521–529. doi: 10.1002/dmrr.2561. [DOI] [PubMed] [Google Scholar]
- 49.Cui J, Philo L, Nguyen P, et al. Sitagliptin vs. placebo for non-alcoholic fatty liver disease: A randomized controlled trial. J Hepatol. 2016;65(2):369–376. doi: 10.1016/j.jhep.2016.04.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Joy TR, McKenzie CA, Tirona RG, et al. Sitagliptin in patients with non-alcoholic steatohepatitis: A randomized, placebo-controlled trial. World J Gastroenterol. 2017;23(1):141–150. doi: 10.3748/wjg.v23.i1.141. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Ohki T, Isogawa A, Toda N, et al. Effectiveness of Ipragliflozin, a sodium-glucose co-transporter 2 inhibitor, as a second-line treatment for non-alcoholic fatty liver disease patients with type 2 diabetes mellitus who do not respond to incretin-based therapies including glucagon-like peptide-1 analogs and dipeptidyl peptidase-4 inhibitors. Clin Drug Investig. 2016;36(4):313–319. doi: 10.1007/s40261-016-0383-1. [DOI] [PubMed] [Google Scholar]
- 52.Takase T, Nakamura A, Miyoshi H, et al. Amelioration of fatty liver index in patients with type 2 diabetes on ipragliflozin: An association with glucose-lowering effects. Endocr J. 2017;64(3):363–367. doi: 10.1507/endocrj.EJ16-0295. [DOI] [PubMed] [Google Scholar]


