Abstract
Hypertension is a major public health concern due to its high prevalence and increased risk of cardiovascular disease and mortality. Complex traits resulting from both genetic and environmental factors affect the development of hypertension. Among environmental factors, a high salt diet is an important cause for hypertension. Humans show a heterogeneous blood pressure (BP) response to sodium intake. Although the precise mechanisms for the association between salt sensitivity and hypertension have not been fully elucidated, renal sodium handling has been considered to play a pivotal role. However, this conventional view has recently been challenged in that a third compartment, namely, skin may have a role in the regulation of sodium homeostasis. Skin is comprised of a significant portion of interstitium, which is a major extracellular fluid compartment, and its complex capillary network regulates body temperature and skin perfusion. Growing evidence indicates that local regulatory action of cutaneous blood flow as well as salt and water metabolism is associated with systemic BP control. Previous experimental studies have shown that dietary salt loading resulted in nonosmotic sodium accumulation via glycosaminoglycans and lymphatics embedded in the skin that were mediated by several endogenous factors and attenuated an increase in BP. Studies in humans have also suggested that the skin serves as a buffer system for sodium storage and that skin sodium contributes to salt sensitivity and hypertension. Thus, skin sodium storage provides the possibility of being an additional buffering system in response to salt loading and concomitant BP changes in humans.
Keywords: Skin, Sodium, Blood pressure, Salt sensitivity
INTRODUCTION
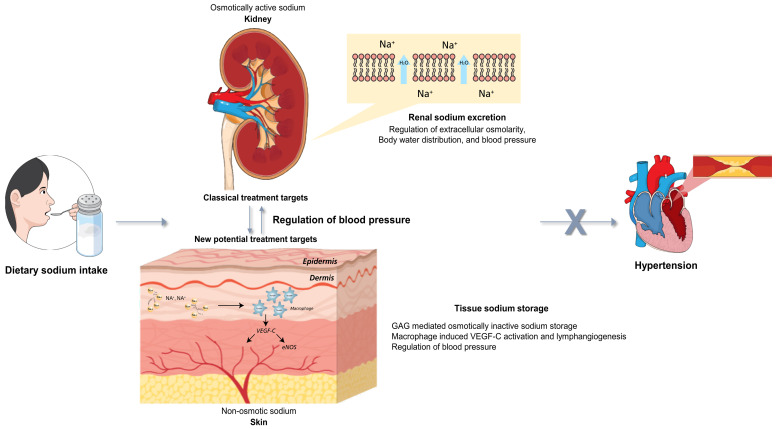
Hypertension is a major global health issue that leads to a high risk of cardiovascular events or death1). Currently, hypertension affects around one-third of the global population and the prevalence of hypertension is increasing worldwide2). Despite of prevailing prevalence, the precise mechanisms of hypertension remain unclear due to its complex origin resulting from both genetic and environmental factors3). Among the several factors that contribute to elevated blood pressure (BP), excessive dietary sodium intake is a crucial trigger for hypertension4,5,6). Sodium is the primary extracellular ion that mediates maintenance of effective circulatory volume and cell tonicity. Extracellular volume control and blood flow are closely linked to long-term BP control7). Increased sodium intake results in an accumulation of sodium in the extracellular body compartment with a parallel increase in extracellular volume, which is partially counterbalanced through pressure natriuresis. Thus, the kidney is known to play an important role in handling body sodium and maintaining BP in humans8). However, more recent studies have reported that large amounts of sodium can accumulate without corresponding water retention to maintain sodium balance9,10). In contrast to conventional understandings that sodium balance functions as a 2-compartment model, these recent findings indicate that the presence of nonosmotic sodium reservoirs act as a third compartment.
The skin accounts for approximately 6% of body weight and contributes a significant portion of the interstitium11,12). The skin is the body’s major extracellular fluid (ECF) compartment and a high volume of electrolytes, including sodium, chloride, and water, are embedded in the skin. Complex materials such as fibroblasts, blood vessels, lymphatics, and nerves are embedded in the dermal layer of the skin in the extracellular matrix of collagen, elastin, and glycosaminoglycans (GAGs)13). Among these, GAGs have a significant negative charge that enables the nonosmotic storage of sodium in the interstitial space14). Additionally, lymphatics in the skin are increased following dietary salt loads. Both processes are mediated by vascular endothelial growth factor-C (VEGF-C) and attenuate the elevation in BP15). Emerging evidence suggests that the skin acts as a buffer for sodium storage and that skin sodium may have a role in salt sensitivity and hypertension in humans.
These observations have led to suggestions that the skin may act as a ‘third compartment’ of body sodium distribution, with a dynamic capacity for sodium storage and buffering body volume and BP against dietary salt intake. In this brief review, recent findings on sodium distribution in skin and the role of skin sodium in the regulation of systemic BP are discussed.
Normal distribution of body sodium
Total body water (TBW) is distributed in intracellular fluid (ICF) or ECF compartments. ECF is compartmentalized with intravascular and interstitial spaces. Total body sodium (TBS) is also highly compartmentalized and almost 98% of TBS is distributed in ECF compartments16). Sodium and fluid located in intracellular space are tightly regulated to protect cells against detrimental volume changes. In contrast, intravascular and interstitial space sodium and fluid are generally in equilibrium to maintain homeostasis. According to the conventional view, TBS is divided into exchangeable and nonexchangeable parts on the basis of the extent of radioisotope sodium equilibration in the body contents17,18). Nonexchangeable sodium has been assumed to be localized primarily in bony structures and to be modified by bone restructuring only, independent of body fluid homeostasis19). However, this conventional view has been challenged in several studies, in which it is suggested that body sodium is not limited to conventional compartments. As dietary salt intake increases, sodium begins to accumulate in the extracellular space20). Thus, each 140 mmol of additional sodium should be theoretically joined with the accumulation of approximately 1 L of water in the extracellular fluid to maintain osmolality. However, several previous studies have evaluated changes in long-term sodium balance among healthy humans and have observed that large amounts of sodium are able to accumulate without corresponding water retention9,10,21,22). Recently, Olde et al. evaluated sodium and water excretion in healthy humans after infusion with hypertonic saline23). They observed that recovery of sodium in the urine was only half of the expected amount, suggesting that some of the infused sodium was retained in an osmotically inactive form. These findings support the presence of non-osmotic storage of excess sodium (sodium accumulation without concomitant water retention) in an additional third compartment, suggesting ‘extra-renal’ mechanisms are involved in sodium homeostasis.
Physiologic handling of sodium and salt sensitivity of blood pressure
Physiological BP responses to dietary sodium intake vary among individuals. Those individuals exhibiting BP changes parallel to changes in salt intake are defined as having a physiological “salt-sensitivity to BP” trait, whereas individuals who do not express this trait are considered to have “salt-resistance to BP”24). Salt-sensitive individuals show a sustained increase in BP with salt loading and a decrease with salt depletion, whereas salt-resistant individuals do not. In humans, these traits are normally distributed and determined by genetic as well as environmental factors. However, the inconsistency of this trait and underlying mechanisms by which sodium influences BP are not fully understood. According to Guyton’s model, the kidney has been considered to be the pivotal organ in the pathophysiology of salt-sensitivity and hypertension8,25). Excessive dietary sodium intake over the renal excretory capacity leads to an osmotically driven expansion of ECF volume. BP follows the equation: BP = pulse volume × peripheral resistance. Thus, salt loading increases the intravascular volume and the cardiac preload, which eventually increases pulse volume. If the kidney loses the ability to excrete excessive salt and water, the pulse volume will increase and, consequently, systemic BP rises. Thus, the inability to excrete sodium by kidney is the basis for salt sensitivity. In contrast, salt-resistant individuals are protected from salt-induced BP rises because they are able to rapidly excrete a salt load without retaining sodium26). Irrespective of possible causes due to abnormal sodium handling, the salt-sensitive trait is not usually characterized by alterations in sodium balance such as impaired natriuresis or volume expansion but rather by elevation of systemic BP to maintain homeostasis24).
However, this conventional paradigm has recently been challenged. Recent studies have reported that both salt-sensitive and salt-resistant individuals without hypertension showed similar degrees of body sodium retention after acute dietary salt loadings, showing that sodium retention occurred without an increase in BP in salt-resistant individuals27,28,29). These studies reported that salt-resistant individuals had an adaptive mechanism against a salt load through vasodilation concomitant to increased cardiac output, whereas this vasodilatory response is mitigated in salt-sensitive individuals. These findings are at variance with the conventional view that salt sensitivity is solely dependent on renal handling of the sodium mechanism and provides the possibility that another compartment is responsible for body sodium storage and BP regulation.
The skin: a third compartment of body sodium storage
The skin, accounting for approximately 6% of body weight and consisting of a significant portion of the interstitium11,12), comprises two layers, namely, the epidermis and the dermis. The dermis is an acellular layer mainly comprised of fibroblasts, blood vessels, lymphatics, and nerves in an extracellular matrix of collagen, elastin, and GAGs13). GAGs are long linear polysaccharides consisting of repeating disaccharide units30,31). Because carboxyl and sulphate functional groups are located on the disaccharide units, GAGs possess highly negative charges that can bind sodium and facilitate the nonosmotic storage of sodium in the interstitium14). Several electrolytes, including sodium, chloride, and water, are embedded in the skin, although their relevance was not fully understood in the early 1900s32). In 1978, Ivanova et al. experimentally showed that sodium stored in the skin of rats increased along with dietary salt loading, and observed that this was correlated with an increase in sulphated GAGs in the dermis33). In the early 2000s, Titze et al. first proposed the possibility of osmotically inactive sodium storage in the skin34). They observed that osmotically inactive sodium storage in salt-resistant rats was around 3-fold higher than in salt-sensitive rats, based on body sodium and body water measurements35). They also showed that male rats had a higher capacity for osmotically inactive skin sodium storage compared with female rats36). Finally, they showed that GAG polymerization induced sodium storage in the skin in an osmotically inactive form. Skin sodium concentrations have been reported to rise as high as 180 to 190 mmol/L without any commensurate increases in water content34). These findings concerning nonosmotic sodium storage in the skin suggest a potential mechanism for a buffering system to regulate body volume and BP after a dietary salt load. In 2013, Titze et al. expanded their scope to humans and performed an experimental study with space flight simulation settings22). They administered constant diets with 12, 9, and 6 g of sodium chloride daily for several months. Interestingly, the 24-hr urine collection results did not concur with conventional understanding that the amount of daily salt intake correlates to the amount of excreted urine sodium. Rather, excreted urine sodium followed a weekly rate dependent on aldosterone and cortisol levels. Furthermore, the total amount of sodium in the body did not correlate with BP levels or body weight. Thus, the authors concluded that sodium was being stored and released into the body without commensurate water changes. Further studies have confirmed the existence of skin sodium storage without an accompanied water content in humans using specialized magnetic resonance imaging (23Na-MRI). Titze et al. reported findings consistent with those in animal studies that showed sodium is stored in hypertensive individuals’ muscles and skin37,38). In another study with patients undergoing hemodialysis, skin sodium storage was elevated even though dialysis treatment eliminated some salt contents from the skin39). More recently, Kopp et al. also showed that patients with diabetes on hemodialysis had significantly higher skin sodium levels compared with those without diabetes using 23Na-MRI40). These findings provide evidence suggesting that the skin acts as a third compartment of body sodium distribution, with a dynamic capacity for sodium storage and buffering body volume and BP against dietary salt intake.
The role of skin sodium in blood pressure regulation: an extra-renal mechanism for buffering dietary salt
The skin is rich in nitric oxide (NO), which regulates vascular tone and dynamic blood flow. These skin properties also suggest a potential role in regulating systemic BP. More recently, immune cells from the innate and adaptive immune system have been shown to play a role in the pathogenesis of hypertension and cardiovascular disease41). Previous experimental studies have shown that macrophages in the dermal layer serve as mediators in modulating sodium homeostasis and BP levels during dietary salt loading15,42,43). Under normal conditions, sodium binds to negatively charged GAGs in the dermal interstitium without concomitant water retention. During high salt loading, the sodium-binding capacity of GAGs exceeds its normal capacity, and interstitial hypertonicity develops. Macrophages are then recruited to influx into the dermal layer and activate and release an osmosensitive transcription factor, namely, tonicity-responsive enhancer binding protein (TonEBP). In turn, TonEBP induces the expression of the VEGF-C gene in an autocrine signaling manner, leading to lymphangiogenesis. The increased lymphatic network enhances sodium transport from the skin into the systemic circulation, to eventually remove sodium by the kidney, preventing a BP rise against salt loading. VEGF-C also induces the expression of endothelial NO synthase (eNOS), causing vasodilation via NO production. In contrast, blocking or genetic depletion of VEGF-C and TonEBP or disruption of the above pathway has been reported to result in salt sensitivity in rats15,42,43). However, recent study by Rossito et al. reported that the skin sodium is isotonic, which could be mistakenly identified as hypertonic due to technical reasons during skin MRI44). This study showed the results against water-independent accumulation of skin sodium theory and suggested that systemic isotonic sodium excess may play an important role in the pathogenesis of hypertension and cardiovascular disease. Although these conflicting explanations need further investigation, it reinforces the importance of skin sodium.
In humans, 23Na-MRI spectroscopy and inductively coupled plasma optical emission spectrometry (ICP-OES) have enabled non-invasive assessment of sodium concentrations in human tissue, thus expanding research fields concerning skin sodium and its relevance to systemic BP. In 2014, Dahlmann et al. investigated the associations between skin sodium storage and VEGF-C in patients on hemodialysis39). They used 23Na-MRI to measure skin sodium and found that the amount of skin sodium was reduced by 19% following single dialysis. Their patients with higher serum VEGF-C levels showed greater amounts of removed sodium. Moreover, the concentrations of skin sodium were higher in patients on hemodialysis compared with healthy controls, and they observed an age-related increase in skin sodium corresponding with a decline in serum VEGF-C. Dahlmann et al. concluded that the sodium stored in the skin could be mobilized through hemodialysis, and that VEGF-C served as a mediator for sodium flow between the interstitium and the systemic circulation in humans. Schneider et al. also showed that sodium was stored in skin among individuals with hypertension and observed that skin sodium concentration correlated with left ventricular hypertrophy in patients with renal impairment45). Several recent studies have evaluated human skin content using ICP-OES, which is a highly sensitive analytical tool capable of simultaneously determining multiple elements down to the sub-billionth level. Fischereder et al. measured tissue sodium and GAGs in the skin in kidney transplant donors and in recipients46). They showed that skin and plasma sodium concentrations positively correlated with GAG contents. They also found that skin sodium correlated well with plasma sodium concentrations. These findings suggest that interstitial sodium storage is regulated by GAGs and through a possible association between the skin and systemic vasculature with regard to sodium homeostasis in humans. Selvarajah et al. undertook a randomized control study to assess skin electrolytes, BP, and plasma VEGF-C in 48 healthy individuals47) and administered placebo (70 mmol sodium/day) and slow sodium(200 mmol/day) for 7 days. Skin sodium(expressed as the Na+:K+ ratio) was higher in the slow sodium group compared with in the placebo group. This study also showed sex-specific differences that the male participants were found to have a significant increase in the skin Na+:K+ ratio (11.2%) after the administration of slow sodium whereas the female participants did not (4%). The female participants also showed a significant increase in 24-hr BP and body weight following salt loading, whereas the male participants did not. The gender difference may exist in the sodium content between skin and muscle. Elevated concentrations of sodium have also been documented in skeletal muscles. Previous study by Wang et al. showed that men have a higher sodium content in the skin than women while women have higher muscle sodium than skin sodium48). Hence, the skin Na+:K+ ratio positively correlated with BP, stroke volume, and peripheral vascular resistance in the male participants only. In summary, the female participants showed less capacity for skin sodium storage against salt loading and greater salt sensitivity of BP, which was in line with previous reports by Titze et al.34,35,36). However, Selvarajah et al.’s study showed no significant changes in plasma VEGF-C between the placebo and slow sodium groups, which indicated no clear involvement of Ton-EBP or VEGF-C activation. After all, the authors concluded that skin sodium increased with dietary salt loading and that skin sodium may have a buffering capacity in relation to dietary salt loading.
Despite major studies having been conducted, the pathophysiologic mechanism between skin sodium and systemic BP has not been fully clarified. The Ton-EBP and VEGF-C have been shown to mainly mediate skin sodium and systemic BP in dietary salt loading, but their relevance to changes in hemodynamics remains unclear. Sweat glands are suggested to have role in skin sodium excretion in response to a high salt diet49). After high sodium diet, sweat sodium concentration have been reported to significantly increase, suggesting that sweat may also play a role in regulating sodium balance in humans. In fact, the sweat gland has some similarity with the renal tubules as cells of the secretary coil of sweat glands contain ion channels and transporters, such as Na+-K+-Cl- cotransporter 1 (NKCC1), Na+-K+-ATPase, Na+/H+ exchanger 1 (NHE1) and aquaporin 5 (AQP5)50). Other etiological factors should be identified to help explain the link between skin sodium and its hemodynamic effects. In addition, the therapeutic effect of using diuretics to decrease skin sodium content in skin is unclear. As most diurectics have a natriuretic effect on the renal tubules, it is unclear whether diuretics have similar efficacy to excrete sodium from the skin and to lower blood pressure. Only few experimental studies showed that amilorides reduced skin sodium contents, but there is lack of evidence in human51). Recently, sodium-glucose cotransporter 2 (SGLT2) inhibitor, a anti-hyperglycemic agent which effectly reduces cardiovascular risks, have been demonstrated that it reduced skin sodium contents measured by 23Na MRI in type 2 diabetic patients11). The precise mechanism of decrease in skin sodium contents with SGLT2 inhibitor is still unknown. Whether it is a direct effect of SGLT-inhibition or a consequence of decrease in renal sodium excretion remains to be elucidated. Further studies with large number of patients are needed to fully uncover the treatment effect with diuretics or SGLT2 inhibitors on skin sodium and blood presure.
Extrarenal nonosmotic sodium handling acts as a “double-edged sword”
Current studies indicate that positively charged sodium binds to negatively charged GAGs in the skin and becomes osmotically inactive. High concentrations of sodium can stimulate GAG synthesis, expanding the storage capacity of sodium and contributing to buffering against systemic BP changes. However, when excessive sodium flows to the skin, it is unclear whether the skin acts as a preemptive reservoir for excessive sodium and prevents an overflow into the systemic circulation before contributing to adverse cardiovascular events, or whether the skin operates as an overflow reservoir once the excessive sodium has already caused vascular damage sufficient to leak into the adjacent tissue.
To reach the skin, dietary ingested sodium passes through the intestines and the blood stream, and is then secreted into the dermal interstitium. First, sodium absorption occurs across the apical membrane of enterocytes and colonocytes through several mechanisms, including sugar and phosphate co-transport via SGLT1, glucose transporter (GLUT) and NaPi2b; electroneutral proton exchange via NHE2, NHE3, and NHE8; and passive diffusion via the epithelial sodium channel (ENaC)52,53,54,55). Intracellular sodium is transported by active pump across the basolateral membrane and into the extracellular space by Na+-K+ ATPase. Once in the extracellular space, sodium diffuses into the intestinal capillaries for transport through the vasculature. What drives sodium out of vasculature and into the skin is not well elucidated, and the transit route from the vascular lumen to the dermis is not fully understood. However, sodium first encounters the endothelial glycocalyx as it is transited out from the vascular lumen. Excessive dietary sodium loads may negatively affect this process and damage the endothelial glycocalyx, thus promoting sodium leakage. The endothelial glycocalyx is comprised primarily of heparan sulphate proteoglycans (HSPGs) and its thickness ranges from 0.5 to 4.5 µm56). The anionic nature of the glycocalyx promotes the movement of red blood cells, provides protection from white blood cell adhesion, scavenges oxygen free radicals, and triggers a vasodilatory response57). Previous experimental studies have shown that chronic high sodium exposure to endothelial cells resulted in a decrease of glycocalyx HSPGs by 68% and caused endothelial stiffening58,59). This damaged glycocalyx enables excessive sodium movement into the interstitium through a paracellular diffusive mechanism or through increased vascular ENaC channels60). Furthermore, a damaged glycocalyx and consequent endothelial stiffening lead to an increase in leucocyte adhesion and infiltration, further injuring the vessel wall61). In summary, skin sodium storage, involving an extrarenal non-osmotic sodium handling, may have a double-edged sword effect. It may protect against the negative effects of excessive sodium buffering for sodium homeostasis. However, this beneficial buffering system may be reversed through sodium leakage and consequent damage to the endothelial glycocalyx barrier and infiltration of inflammatory cells, eventually increasing the risk of adverse cardiovascular outcomes.
CONCLUSIONS
The conventional paradigm understanding that renal sodium handling is the mainstream method of regulating sodium homeostasis and salt sensitivity of BP is shifting towards an understanding that the skin may act as a third compartment for sodium, and is capable of osmotically inactive sodium storage (Figure 1). Previous studies have shown that individuals predisposed to salt sensitive hypertension have defects in the skin sodium buffering system. These two understandings can intersect in that the skin may act as a buffer as well as a reservoir against dietary sodium loading, whereas the kidney handles sodium excretion and reabsorption through regulating serum osmolality and total body water. Nevertheless, little is known concerning the long-term effects of sodium loading on skin sodium storage. Skin sodium with short-term dietary salt loading protects against a rise in BP; however, long-term high skin sodium accumulation is associated with an increase in BP and salt sensitive hypertension. A potential explanation for these findings is that constantly high skin sodium damages the endothelial glycocalyx, with impaired VEGF-C activation and lymphangiogenesis, which then leads to a reduction in sodium flow from the skin to the systemic circulation and attenuates a vasodilatory response to salt loading. However, as noted, the mechanisms concerning the movement of dietary ingested sodium from the gut to the systemic vasculature and the skin have not yet been well elucidated. Further studies are needed to explore the mechanisms involved in this process. Moreover, longitudinal studies to evalute the causal association between skin sodium and hypertension are still lacking. Further longitudinal population based studies are needed whether increased amounts of skin sodium effects on BP levels or clinical outcomes.Nevertheless, findings indicating the potential of skin, which is the largest organ in the human body, to act as a third compartment responsible for sodium homeostasis and a buffer system for systemic BP, offer new insights into the pathogenesis of hypertension and related treatment challenges.
Fig. 1. Schematic summary of sodium homeostasis.
References
- 1.Kearney PM, Whelton M, Reynolds K, Muntner P, Whelton PK, He J. Global burden of hypertension: analysis of worldwide data. Lancet. 2005;365:217–223. doi: 10.1016/S0140-6736(05)17741-1. [DOI] [PubMed] [Google Scholar]
- 2.Mills KT, Bundy JD, Kelly TN, Reed JE, Kearney PM, Reynolds K, et al. Global Disparities of Hypertension Prevalence and Control: A Systematic Analysis of Population-Based Studies From 90 Countries. Circulation. 2016;134:441–450. doi: 10.1161/CIRCULATIONAHA.115.018912. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Choi HY, Park HC, Ha SK. Salt Sensitivity and Hypertension: A Paradigm Shift from Kidney Malfunction to Vascular Endothelial Dysfunction. Electrolyte Blood Press. 2015;13:7–16. doi: 10.5049/EBP.2015.13.1.7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Mente A, O'Donnell MJ, Rangarajan S, McQueen MJ, Poirier P, Wielgosz A, et al. Association of urinary sodium and potassium excretion with blood pressure. N Engl J Med. 2014;371:601–611. doi: 10.1056/NEJMoa1311989. [DOI] [PubMed] [Google Scholar]
- 5.Jackson SL, Cogswell ME, Zhao L, Terry AL, Wang CY, Wright J, et al. Association Between Urinary Sodium and Potassium Excretion and Blood Pressure Among Adults in the United States: National Health and Nutrition Examination Survey, 2014. Circulation. 2018;137:237–246. doi: 10.1161/CIRCULATIONAHA.117.029193. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Mente A, O'Donnell M, Rangarajan S, Dagenais G, Lear S, McQueen M, et al. Associations of urinary sodium excretion with cardiovascular events in individuals with and without hypertension: a pooled analysis of data from four studies. Lancet. 2016;388:465–475. doi: 10.1016/S0140-6736(16)30467-6. [DOI] [PubMed] [Google Scholar]
- 7.Johnson RS, Titze J, Weller R. Cutaneous control of blood pressure. Curr Opin Nephrol Hypertens. 2016;25:11–15. doi: 10.1097/MNH.0000000000000188. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Guyton AC. Blood pressure control--special role of the kidneys and body fluids. Science. 1991;252:1813–1816. doi: 10.1126/science.2063193. [DOI] [PubMed] [Google Scholar]
- 9.Heer M, Baisch F, Kropp J, Gerzer R, Drummer C. High dietary sodium chloride consumption may not induce body fluid retention in humans. Am J Physiol Renal Physiol. 2000;278:F585–F595. doi: 10.1152/ajprenal.2000.278.4.F585. [DOI] [PubMed] [Google Scholar]
- 10.Titze J, Maillet A, Lang R, Gunga HC, Johannes B, Gauquelin-Koch G, et al. Long-term sodium balance in humans in a terrestrial space station simulation study. Am J Kidney Dis. 2002;40:508–516. doi: 10.1053/ajkd.2002.34908. [DOI] [PubMed] [Google Scholar]
- 11.Aukland K, Reed RK. Interstitial-lymphatic mechanisms in the control of extracellular fluid volume. Physiol Rev. 1993;73:1–78. doi: 10.1152/physrev.1993.73.1.1. [DOI] [PubMed] [Google Scholar]
- 12.Tobin DJ. Biochemistry of human skin--our brain on the outside. Chem Soc Rev. 2006;35:52–67. doi: 10.1039/b505793k. [DOI] [PubMed] [Google Scholar]
- 13.LA G. Physiology, biochemistry, and molecular biology of the skin. USA: Oxford University Press; 1991. [Google Scholar]
- 14.Wiig H, Luft FC, Titze JM. The interstitium conducts extrarenal storage of sodium and represents a third compartment essential for extracellular volume and blood pressure homeostasis. Acta Physiol (Oxf) 2018;222 doi: 10.1111/apha.13006. [DOI] [PubMed] [Google Scholar]
- 15.Machnik A, Neuhofer W, Jantsch J, Dahlmann A, Tammela T, Machura K, et al. Macrophages regulate salt-dependent volume and blood pressure by a vascular endothelial growth factor-C-dependent buffering mechanism. Nat Med. 2009;15:545–552. doi: 10.1038/nm.1960. [DOI] [PubMed] [Google Scholar]
- 16.Bhave G, Neilson EG. Body fluid dynamics: back to the future. J Am Soc Nephrol. 2011;22:2166–2181. doi: 10.1681/ASN.2011080865. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Edelman IS, Leibman J. Anatomy of body water and electrolytes. Am J Med. 1959;27:256–277. doi: 10.1016/0002-9343(59)90346-8. [DOI] [PubMed] [Google Scholar]
- 18.Bie P. Mechanisms of sodium balance: total body sodium, surrogate variables, and renal sodium excretion. Am J Physiol Regul Integr Comp Physiol. 2018;315:R945–r962. doi: 10.1152/ajpregu.00363.2017. [DOI] [PubMed] [Google Scholar]
- 19.Davies RE, Kornberg HL, Wilson GM. Relation between total and exchangeable sodium in the body. Nature. 1952;170:979. doi: 10.1038/170979a0. [DOI] [PubMed] [Google Scholar]
- 20.Lee SH CY. Dietary Salt and Potassium Intake and Hypertension. Electrolyte Blood Press. 2005;3:36–43. [Google Scholar]
- 21.Palacios C, Wigertz K, Martin BR, Jackman L, Pratt JH, Peacock M, et al. Sodium retention in black and white female adolescents in response to salt intake. J Clin Endocrinol Metab. 2004;89:1858–1863. doi: 10.1210/jc.2003-031446. [DOI] [PubMed] [Google Scholar]
- 22.Rakova N, Jüttner K, Dahlmann A, Schröder A, Linz P, Kopp C, et al. Long-term space flight simulation reveals infradian rhythmicity in human Na(+) balance. Cell Metab. 2013;17:125–131. doi: 10.1016/j.cmet.2012.11.013. [DOI] [PubMed] [Google Scholar]
- 23.Olde Engberink RH, Rorije NM, van den Born BH, Vogt L. Quantification of nonosmotic sodium storage capacity following acute hypertonic saline infusion in healthy individuals. Kidney Int. 2017;91:738–745. doi: 10.1016/j.kint.2016.12.004. [DOI] [PubMed] [Google Scholar]
- 24.Elijovich F, Weinberger MH, Anderson CA, Appel LJ, Bursztyn M, Cook NR, et al. Salt Sensitivity of Blood Pressure: A Scientific Statement From the American Heart Association. Hypertension. 2016;68:e7–e46. doi: 10.1161/HYP.0000000000000047. [DOI] [PubMed] [Google Scholar]
- 25.YW K. Renal Sodium Handling and Hypertension. Electrolyte Blood Press. 2005;3:32–35. [Google Scholar]
- 26.Guyton AC. Long-term arterial pressure control: an analysis from animal experiments and computer and graphic models. Am J Physiol. 1990;259:R865–R877. doi: 10.1152/ajpregu.1990.259.5.R865. [DOI] [PubMed] [Google Scholar]
- 27.Schmidlin O, Sebastian AF, Morris RC., Jr What initiates the pressor effect of salt in salt-sensitive humans? Observations in normotensive blacks. Hypertension. 2007;49:1032–1039. doi: 10.1161/HYPERTENSIONAHA.106.084640. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Schmidlin O, Forman A, Leone A, Sebastian A, Morris RC., Jr Salt sensitivity in blacks: evidence that the initial pressor effect of NaCl involves inhibition of vasodilatation by asymmetrical dimethylarginine. Hypertension. 2011;58:380–385. doi: 10.1161/HYPERTENSIONAHA.111.170175. [DOI] [PubMed] [Google Scholar]
- 29.Laffer CL, Scott RC, 3rd, Titze JM, Luft FC, Elijovich F. Hemodynamics and Salt-and-Water Balance Link Sodium Storage and Vascular Dysfunction in Salt-Sensitive Subjects. Hypertension. 2016;68:195–203. doi: 10.1161/HYPERTENSIONAHA.116.07289. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Comper WD, Laurent TC. Physiological function of connective tissue polysaccharides. Physiol Rev. 1978;58:255–315. doi: 10.1152/physrev.1978.58.1.255. [DOI] [PubMed] [Google Scholar]
- 31.Schaefer L, Schaefer RM. Proteoglycans: from structural compounds to signaling molecules. Cell Tissue Res. 2010;339:237–246. doi: 10.1007/s00441-009-0821-y. [DOI] [PubMed] [Google Scholar]
- 32.Selvarajah V, Connolly K, McEniery C, Wilkinson I. Skin Sodium and Hypertension: a Paradigm Shift? Curr Hypertens Rep. 2018;20:94. doi: 10.1007/s11906-018-0892-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Ivanova LN, Archibasova VK, Shterental I. [Sodium-depositing function of the skin in white rats] Fiziol Zh SSSR Im I M Sechenova. 1978;64:358–363. [PubMed] [Google Scholar]
- 34.Titze J, Shakibaei M, Schafflhuber M, Schulze-Tanzil G, Porst M, Schwind KH, et al. Glycosaminoglycan polymerization may enable osmotically inactive Na+ storage in the skin. Am J Physiol Heart Circ Physiol. 2004;287:H203–H208. doi: 10.1152/ajpheart.01237.2003. [DOI] [PubMed] [Google Scholar]
- 35.Titze J, Krause H, Hecht H, Dietsch P, Rittweger J, Lang R, et al. Reduced osmotically inactive Na storage capacity and hypertension in the Dahl model. Am J Physiol Renal Physiol. 2002;283:F134–F141. doi: 10.1152/ajprenal.00323.2001. [DOI] [PubMed] [Google Scholar]
- 36.Titze J, Lang R, Ilies C, Schwind KH, Kirsch KA, Dietsch P, et al. Osmotically inactive skin Na+ storage in rats. Am J Physiol Renal Physiol. 2003;285:F1108–F1117. doi: 10.1152/ajprenal.00200.2003. [DOI] [PubMed] [Google Scholar]
- 37.Titze J, Dahlmann A, Lerchl K, Kopp C, Rakova N, Schröder A, et al. Spooky sodium balance. Kidney Int. 2014;85:759–767. doi: 10.1038/ki.2013.367. [DOI] [PubMed] [Google Scholar]
- 38.Kopp C, Linz P, Dahlmann A, Hammon M, Jantsch J, Müller DN, et al. 23Na magnetic resonance imaging-determined tissue sodium in healthy subjects and hypertensive patients. Hypertension. 2013;61:635–640. doi: 10.1161/HYPERTENSIONAHA.111.00566. [DOI] [PubMed] [Google Scholar]
- 39.Dahlmann A, Dörfelt K, Eicher F, Linz P, Kopp C, Mössinger I, et al. Magnetic resonance-determined sodium removal from tissue stores in hemodialysis patients. Kidney Int. 2015;87:434–441. doi: 10.1038/ki.2014.269. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Kopp C, Linz P, Maier C, Wabel P, Hammon M, Nagel AM, et al. Elevated tissue sodium deposition in patients with type 2 diabetes on hemodialysis detected by (23)Na magnetic resonance imaging. Kidney Int. 2018;93:1191–1197. doi: 10.1016/j.kint.2017.11.021. [DOI] [PubMed] [Google Scholar]
- 41.Harrison DG, Marvar PJ, Titze JM. Vascular inflammatory cells in hypertension. Front Physiol. 2012;3:128. doi: 10.3389/fphys.2012.00128. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Machnik A, Dahlmann A, Kopp C, Goss J, Wagner H, van Rooijen N, et al. Mononuclear phagocyte system depletion blocks interstitial tonicity-responsive enhancer binding protein/vascular endothelial growth factor C expression and induces salt-sensitive hypertension in rats. Hypertension. 2010;55:755–761. doi: 10.1161/HYPERTENSIONAHA.109.143339. [DOI] [PubMed] [Google Scholar]
- 43.Wiig H, Schröder A, Neuhofer W, Jantsch J, Kopp C, Karlsen TV, et al. Immune cells control skin lymphatic electrolyte homeostasis and blood pressure. J Clin Invest. 2013;123:2803–2815. doi: 10.1172/JCI60113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Rossitto G, Mary S, Chen JY, Boder P, Chew KS, Neves KB, et al. Tissue sodium excess is not hypertonic and reflects extracellular volume expansion. Nat Commun. 2020;11:4222. doi: 10.1038/s41467-020-17820-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Schneider MP, Raff U, Kopp C, Scheppach JB, Toncar S, Wanner C, et al. Skin Sodium Concentration Correlates with Left Ventricular Hypertrophy in CKD. J Am Soc Nephrol. 2017;28:1867–1876. doi: 10.1681/ASN.2016060662. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Yokota N, Burne-Taney M, Racusen L, Rabb H. Contrasting roles for STAT4 and STAT6 signal transduction pathways in murine renal ischemia-reperfusion injury. Am J Physiol Renal Physiol. 2003;285:F319–F325. doi: 10.1152/ajprenal.00432.2002. [DOI] [PubMed] [Google Scholar]
- 47.Selvarajah V, Mäki-Petäjä KM, Pedro L, Bruggraber SFA, Burling K, Goodhart AK, et al. Novel Mechanism for Buffering Dietary Salt in Humans: Effects of Salt Loading on Skin Sodium, Vascular Endothelial Growth Factor C, and Blood Pressure. Hypertension. 2017;70:930–937. doi: 10.1161/HYPERTENSIONAHA.117.10003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Wang P, Deger MS, Kang H, Ikizler TA, Titze J, Gore JC. Sex differences in sodium deposition in human muscle and skin. Magn Reson Imaging. 2017;36:93–97. doi: 10.1016/j.mri.2016.10.023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Polychronopoulou E, Braconnier P, Burnier M. New Insights on the Role of Sodium in the Physiological Regulation of Blood Pressure and Development of Hypertension. Frontiers in Cardiovascular Medicine. 2019;6 doi: 10.3389/fcvm.2019.00136. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Cui CY, Schlessinger D. Eccrine sweat gland development and sweat secretion. Exp Dermatol. 2015;24:644–650. doi: 10.1111/exd.12773. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Hammon M, Grossmann S, Linz P, Kopp C, Dahlmann A, Garlichs C, et al. 23Na Magnetic Resonance Imaging of the Lower Leg of Acute Heart Failure Patients during Diuretic Treatment. PLoS One. 2015;10:e0141336. doi: 10.1371/journal.pone.0141336. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Xu H, Chen R, Ghishan FK. Subcloning, localization, and expression of the rat intestinal sodium-hydrogen exchanger isoform 8. Am J Physiol Gastrointest Liver Physiol. 2005;289:G36–G41. doi: 10.1152/ajpgi.00552.2004. [DOI] [PubMed] [Google Scholar]
- 53.Collins JF, Honda T, Knobel S, Bulus NM, Conary J, DuBois R, et al. Molecular cloning, sequencing, tissue distribution, and functional expression of a Na+/H+ exchanger (NHE-2) Proc Natl Acad Sci U S A. 1993;90:3938–3942. doi: 10.1073/pnas.90.9.3938. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Collins JF, Xu H, Kiela PR, Zeng J, Ghishan FK. Functional and molecular characterization of NHE3 expression during ontogeny in rat jejunal epithelium. Am J Physiol. 1997;273:C1937–C1946. doi: 10.1152/ajpcell.1997.273.6.C1937. [DOI] [PubMed] [Google Scholar]
- 55.Kiela PR, Ghishan FK. Recent advances in the renal-skeletalgut axis that controls phosphate homeostasis. Lab Invest. 2009;89:7–14. doi: 10.1038/labinvest.2008.114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Reitsma S, Slaaf DW, Vink H, van Zandvoort MA, oude Egbrink MG. The endothelial glycocalyx: composition, functions, and visualization. Pflugers Arch. 2007;454:345–359. doi: 10.1007/s00424-007-0212-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Weinbaum S, Tarbell JM, Damiano ER. The structure and function of the endothelial glycocalyx layer. Annu Rev Biomed Eng. 2007;9:121–167. doi: 10.1146/annurev.bioeng.9.060906.151959. [DOI] [PubMed] [Google Scholar]
- 58.Oberleithner H, Peters W, Kusche-Vihrog K, Korte S, Schillers H, Kliche K, et al. Salt overload damages the glycocalyx sodium barrier of vascular endothelium. Pflugers Arch. 2011;462:519–528. doi: 10.1007/s00424-011-0999-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Oberleithner H, Riethmüller C, Schillers H, MacGregor GA, de Wardener HE, Hausberg M. Plasma sodium stiffens vascular endothelium and reduces nitric oxide release. Proc Natl Acad Sci U S A. 2007;104:16281–16286. doi: 10.1073/pnas.0707791104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Korte S, Wiesinger A, Straeter AS, Peters W, Oberleithner H, Kusche-Vihrog K. Firewall function of the endothelial glycocalyx in the regulation of sodium homeostasis. Pflugers Arch. 2012;463:269–278. doi: 10.1007/s00424-011-1038-y. [DOI] [PubMed] [Google Scholar]
- 61.McDonald KK, Cooper S, Danielzak L, Leask RL. Glycocalyx Degradation Induces a Proinflammatory Phenotype and Increased Leukocyte Adhesion in Cultured Endothelial Cells under Flow. PLoS One. 2016;11:e0167576. doi: 10.1371/journal.pone.0167576. [DOI] [PMC free article] [PubMed] [Google Scholar]